Ingestion of Diazotrophs Makes Corals More Resistant to Heat Stress
Abstract
:1. Introduction
2. Materials and Methods
2.1. Diazotrophic Strains Acclimation
2.2. Coral Preparation and Acclimation
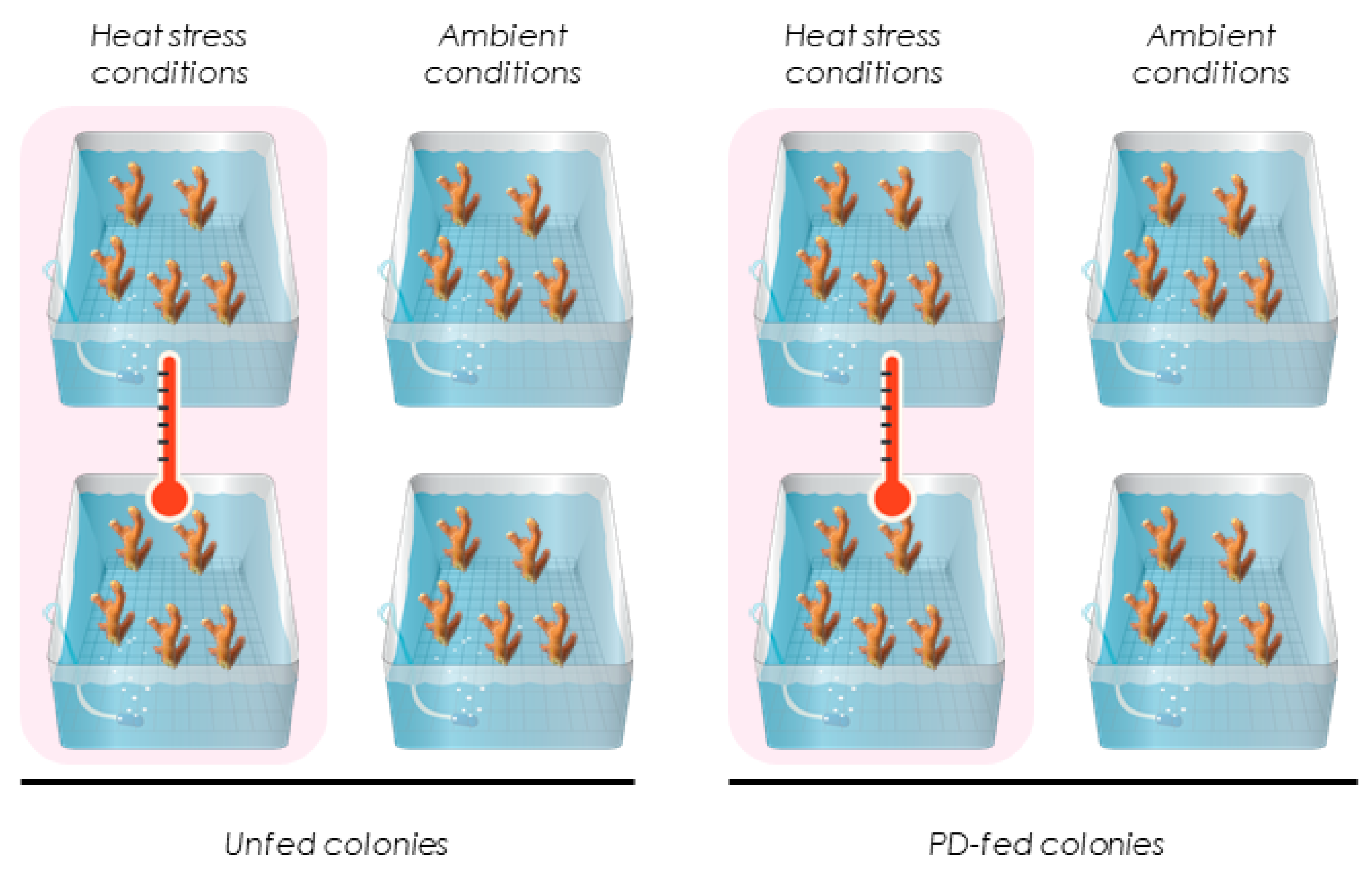
2.3. Experimental Setup and Conditions
2.4. Symbiodiniaceae Density and Total Chlorophyll, and Protein Content
2.5. Growth Rates
2.6. Photosynthetic Efficiency Measurements
2.7. Statistical Analyses
3. Results
3.1. Symbiodiniaceae Density, Chlorophyll, and Protein Concentrations
3.2. Growth Rates
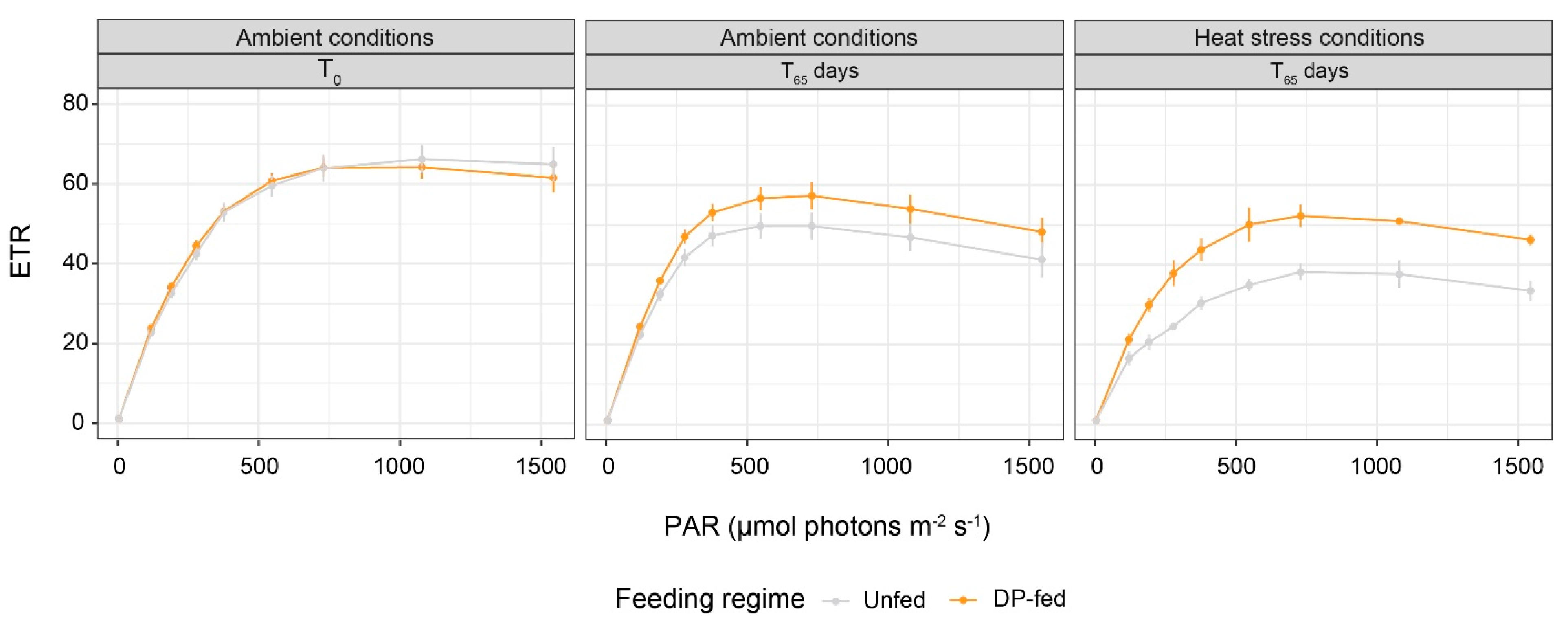
3.3. Fv/Fm and Electron Transport Rates of Symbiodiniaceae
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- LaJeunesse, C.T.; Parkinson, J.; Barielson, P.; Jeong, H.; Reimer, J.; Voolstra, C.R.; Santos, S. Systematic revision of Symbiodiniaceae highlights the antiquity and diversity of coral endosymbionts. Curr. Biol. 2018, 28, 2570–2580. [Google Scholar] [CrossRef] [Green Version]
- Muscatine, L.; Porter, J.W. Reef Corals: Mutualistic Symbioses Adapted to Nutrient-Poor Environments. Bioscience 1977, 27, 454–460. [Google Scholar] [CrossRef]
- Hughes, T.; Kerry, J.; Álvarez-Noriega, M.; Álvarez-Romero, J.; Anderson, K.; Baird, A.; Babcock, R.; Beger, M.; Bellwood, D.; Berkelmans, R.; et al. Global warming and recurrent mass bleaching of corals. Nature 2017, 543, 373–377. [Google Scholar] [CrossRef]
- Muscatine, L. The role of symbiotic algae in carbon and energy flux in reef corals. In Ecosystems of the World Coral Reefs; Dubinsky, Z., Ed.; Elsevier: Amsterdam, The Netherlands, 1990; pp. 75–87. [Google Scholar]
- Lesser, M.P.; Mazel, C.H.; Gorbunov, M.Y.; Falkowski, P.G. Discovery of symbiotic nitrogen-fixing cyanobacteria in corals. Science 2004, 305, 997–1000. [Google Scholar] [CrossRef] [PubMed]
- Fisk, D.D.A.; Done, T.T.J. Taxonomic and bathymetric patterns of bleaching in corals, Myrmidon reef. In Proceedings of the 5th International Coral Reef Congress, Tahiti, France, 27 May–1 June 1985. [Google Scholar]
- Grottoli, G.; Rodrigues, L.J.; Juarez, C. Lipids and stable carbon isotopes in two species of Hawaiian corals, Porites compressa and Montipora verrucosa, following a bleaching event. Mar. Biol. 2004, 145, 621–631. [Google Scholar] [CrossRef]
- Marshall, P.A.; Baird, A.H. Bleaching of corals on the Great Barrier Reef: Differential susceptibilities among taxa. Coral Reefs 2000, 19, 155–163. [Google Scholar] [CrossRef]
- Stimson, J.; Sakai, K.; Sembali, H. Interspecific comparison of the symbiotic relationship in corals with high and low rates of bleaching-induced mortality. Coral Reefs 2002, 21, 409–421. [Google Scholar] [CrossRef]
- Wilkinson, C.; Hodgson, G. Coral reefs and the 1997-1998 mass bleaching and mortality. Nat. Resour. 1999, 35, 16–25. [Google Scholar]
- Loya, Y.; Sakai, K.; Yamazato, K.; Nakano, Y.; Sambali, H.; van Woesik, R. Coral bleaching: The winners and the losers. Ecol. Lett. 2001, 4, 122–131. [Google Scholar] [CrossRef]
- Fitt, W.K.; Gates, R.D.; Hoegh-Guldberg, O.; Bythell, J.C.; Jatkar, A.; Grottoli, A.G.; Gomez, M.; Fisher, P.; Lajuenesse, T.C.; Pantos, O.; et al. Response of two species of Indo-Pacific corals, Porites cylindrica and Stylophora pistillata, to short-term thermal stress: The host does matter in determining the tolerance of corals to bleaching. J. Exp. Mar. Biol. Ecol. 2009, 373, 102–110. [Google Scholar] [CrossRef]
- Dunn, S.R.; Pernice, M.; Green, K.; Hoegh-Guldberg, O.; Dove, S.G. Thermal stress promotes host mitochondrial degradation in symbiotic cnidarians: Are the batteries of the reef going to run out? PLoS ONE 2012, 7, e39024. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Glynn, P.W.; Maté, J.L.; Baker, A.C.; Calderón, M.O. Coral bleaching and mortality in Panama and Ecuador during the 1997–1998 El Niño-Southern Oscillation event: Spatial/temporal patterns and comparisons with the 1982-1983 event. Bull. Mar. Sci. 2001, 69, 79–109. [Google Scholar]
- Howells, E.J.; Beltran, V.H.; Larsen, N.W.; Bay, L.K.; Willis, B.L.; van Oppen, M.J.H. Coral thermal tolerance shaped by local adaptation of photosymbionts. Nat. Clim. Chang. 2012, 2, 116–120. [Google Scholar] [CrossRef]
- Warner, M.E.; Fitt, W.K.; Schmidt, G.W. Damage to photosystem II in symbiotic dinoflagellates: A determinant of coral bleaching. Proc. Natl. Acad. Sci. USA 1999, 96, 8007–8012. [Google Scholar] [CrossRef] [Green Version]
- Houlbrèque, F.; Tambutté, E.; Richard, C.; Ferrier-pagès, C. Importance of a micro-diet for scleractinian corals. Mar. Ecol. Prog. Ser. 2004, 282, 151–160. [Google Scholar] [CrossRef] [Green Version]
- Houlbrèque, F.; Ferrier-Pagès, C. Heterotrophy in Tropical Scleractinian Corals. Biol. Rev. 2009, 84, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, W.M. Coral Food, Feeding, Nutrition, and Secretion: A Review. Mar. Org. Model Syst. Biol. Med. 2018, 65, 377–421. [Google Scholar]
- Houlbrèque, F.; Tambutté, E.; Ferrier-Pagès, C. Effect of zooplankton availability on the rates of photosynthesis, and tissue and skeletal growth in the scleractinian coral Stylophora pistillata. J. Exp. Mar. Biol. Ecol. 2003, 296, 145–166. [Google Scholar] [CrossRef]
- Borell, E.M.; Yuliantri, A.R.; Bischof, K.; Richter, C. The effect of heterotrophy on photosynthesis and tissue composition of two scleractinian corals under elevated temperature. J. Exp. Mar. Biol. Ecol. 2008, 364, 116–123. [Google Scholar] [CrossRef]
- Ferrier-Pagès, C.; Rottier, C.; Beraud, E.; Levy, O. Experimental assessment of the feeding effort of three scleractinian coral species during a thermal stress: Effect on the rates of photosynthesis. J. Exp. Mar. Biol. Ecol. 2010, 390, 118–124. [Google Scholar] [CrossRef]
- Ferrier-Pagès, C.; Peiranoet, A.; Abbate, M.; Cocito, S.; Negri, A.; Rottier, C.; Riera, P.; Rodolfo-Metalpaal, R.; Reynaud, S. Summer autotrophy and winter heterotrophy in the temperate symbiotic coral Cladocora caespitosa. Limnol. Oceanogr. 2011, 56, 1429–1438. [Google Scholar] [CrossRef]
- Treignier, C.; Grover, R.; Ferrier-Pagès, C.; Tolosa, I. Effect of light and feeding on the fatty acid and sterol composition of zooxanthellae and host tissue isolated from the scleractinian coral Turbinaria reniformis. Limnol. Oceanogr. 2008, 53, 2702–2710. [Google Scholar] [CrossRef] [Green Version]
- Anthony, K.R.N.; Hoogenboom, M.O.; Maynard, J.A.; Grottoli, A.G.; Middlebrook, R. Energetics approach to predicting mortality risk from environmental stress: A case study of coral bleaching. Funct. Ecol. 2009, 23, 539–550. [Google Scholar] [CrossRef]
- Cox, E.F. Continuation of sexual reproduction in Montipora capitata following bleaching. Coral Reefs 2007, 26, 721–724. [Google Scholar] [CrossRef]
- Edmunds, P.J. Zooplanktivory ameliorates the effects of ocean acidification on the reef coral Porites spp. Limnol. Oceanogr. 2011, 56, 2402–2410. [Google Scholar] [CrossRef]
- Houlbrèque, F.; Reynaud, S.; Godinot, C.; Oberhänsli, F.; Rodolfo-Metalpa, R.; Ferrier-Pagés, C. Ocean acidification reduces feeding rates in the scleractinian coral Stylophora pistillata. Limnol. Oceanogr. 2015, 60, 89–99. [Google Scholar] [CrossRef]
- Drenkard, E.J.; Cohen, A.L.; McCorkle, D.C.; de Putron, S.J.; Starczak, V.R.; Zicht, A.E. Calcification by juvenile corals under heterotrophy and elevated CO2. Coral Reefs 2013, 32, 727–735. [Google Scholar] [CrossRef]
- Grottoli, G.; Rodrigues, L.J.; Palardy, J.E. Heterotrophic plasticity and resilience in bleached corals. Nature 2006, 440, 1186–1189. [Google Scholar] [CrossRef]
- Bessell-Browne, P.; Stat, M.; Thomson, D.; Clode, P.L. Coscinaraea marshae corals that have survived prolonged bleaching exhibit signs of increased heterotrophic feeding. Coral Reefs 2014, 33, 795–804. [Google Scholar] [CrossRef]
- Palardy, J.E.; Rodrigues, L.J.; Grottoli, A.G. The importance of zooplankton to the daily metabolic carbon requirements of healthy and bleached corals at two depths. J. Exp. Mar. Biol. Ecol. 2008, 367, 180–188. [Google Scholar] [CrossRef]
- Sangmanee, K.; Casareto, B.; Nguyen, T.D.; Sangsawang, L.; Toyoda, K.; Suzuki, T.; Suzuki, Y. Influence of thermal stress and bleaching on heterotrophic feeding of two scleractinian corals on pico-nanoplankton. Mar. Pollut. Bull. 2020, 158, 111405. [Google Scholar] [CrossRef] [PubMed]
- Conti-Jerpe, E.; Thompson, P.; Wong, C.W.M.; Oliveira, N.; Duprey, N.; Moynihan, M.; Baker, D. Trophic strategy and bleaching resistance in reef-building corals. Sci. Adv. 2020, 6, eaaz5443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tremblay, P.; Naumann, M.S.; Sikorski, S.; Grover, R.; Ferrier-Pagès, C. Experimental assessment of organic carbon fluxes in the scleractinian coral Stylophora pistillata during a thermal and photo stress event. Mar. Ecol. Prog. Ser. 2012, 453, 63–77. [Google Scholar] [CrossRef]
- Meunier, V.; Bonnet, S.; Pernice, M.; Benavides, M.; Lorrain, A.; Grosso, O.; Lambert, C.; Houlbrèque, F. Bleaching forces coral’s heterotrophy on diazotrophs and Synechococcus. ISME J. 2019, 13, 2882–2886. [Google Scholar] [CrossRef]
- Turk-Kubo, K.A.; Frank, I.E.; Hogan, M.E.; Desnues, A.; Bonnet, S.; Zehr, J.P. Diazotroph community succession during the VAHINE mesocosms experiment (New Caledonia Lagoon). Biogeosciences Discuss. 2015, 12, 9043–9079. [Google Scholar] [CrossRef] [Green Version]
- Messer, F.; Mahaffey, C.; Robinson, C.M.; Jeffries, T.C.; Baker, K.G.; Isaksson, J.B.; Ostrowski, M.; Doblin, M.A.; Brown, M.V.; Seymour, J.R. High levels of heterogeneity in diazotroph diversity and activity within a putative hotspot for marine nitrogen fixation. ISME J. 2016, 10, 1499–1513. [Google Scholar] [CrossRef]
- Messer, F.; Brown, M.V.; Furnas, M.J.; Carney, R.L.; McKinnon, A.D.; Seymour, J.R. Diversity and activity of diazotrophs in great barrier reef surface waters. Front. Microbiol. 2017, 8, 967. [Google Scholar] [CrossRef]
- Bonnet, S.; Caffin, M.; Berthelot, H.; Moutin, T. Hot spot of N2 fixation in the western tropical South Pacific pleads for a spatial decoupling between N2 fixation and denitrification. Proc. Natl. Acad. Sci. USA 2017, 114, E2800–E2801. [Google Scholar] [CrossRef] [Green Version]
- Luo, Y.-W.; Doney, S.C.; Anderson, L.A.; Benavides, M.; Berman-Frank, I.; Bode, A.; Bonnet, S.; Boström, K.H.; Böttjer, D.; Capone, D.G.; et al. Database of diazotrophs in global ocean: Abundance, biomass and nitrogen fixation rates. Earth Syst. Sci. Data 2012, 4, 47–73. [Google Scholar] [CrossRef] [Green Version]
- Foster, R.A.; Paytan, A.; Zehr, J.P. Seasonality of N2 fixation and nifH gene diversity in the Gulf of Aqaba (Red Sea). Limnol. Oceanogr. 2009, 54, 219–233. [Google Scholar] [CrossRef]
- Rahav, E.; Bar-Zeev, E.; Ohayon, S.; Elifantz, H.; Belkin, N.; Herut, B.; Mulholland, M.; Berman-Frank, I. Dinitrogen fixation in aphotic oxygenated marine environments. Front. Microbiol. 2013, 4, 227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saulia, E.; Benavides, M.; Henke, B.; Turk-Kubo, K.; Cooperguard, H.; Grosso, O.; Desnues, A.; Rodier, M.; Dupouy, C.; Riemann, L.; et al. Seasonal Shifts in Diazotrophs Players: Patterns Observed Over a Two-Year Time Series in the New Caledonian Lagoon (Western Tropical South Pacific Ocean). Front. Mar. Sci. 2020, 7, 915. [Google Scholar] [CrossRef]
- Berthelot, H.; Moutin, T.; L’Helguen, S.; Leblanc, K.; Hélias, S.; Grosso, O.; Leblond, N.; Charrière, B.; Bonnet, S. Dinitrogen fixation and dissolved organic nitrogen fueled primary production and particulate export during the VAHINE mesocosm experiment (New Caledonia lagoon). Biogeosciences 2015, 12, 4099–4112. [Google Scholar] [CrossRef] [Green Version]
- Rodier, M.; Le Borgne, R. Population and trophic dynamics of Trichodesmium thiebautii in the SE lagoon of New Caledonia. Comparison with T. erythraeum in the SW lagoon. Mar. Pollut. Bull. 2010, 61, 349–359. [Google Scholar] [CrossRef]
- Bonnet, S.; Berthelot, H.; Turk-Kubo, K.; Fawcett, S.; Rahav, E.; L’Helguen, S.; Berman-Frank, I. Dynamics of N2 fixation and fate of diazotroph-derived nitrogen in a low-nutrient, low-chlorophyll ecosystem: Results from the VAHINE mesocosm experiment (New Caledonia). Biogeosciences 2016, 13, 2653–2673. [Google Scholar] [CrossRef] [Green Version]
- 48. White, E.; Spitz, Y.H.; Karl, D.M.; Letelier, R.M. Flexible elemental stoichiometry in Trichodesmium spp. and its ecological implications. Limnol. Oceanogr. 2006, 51, 1777–1790. [Google Scholar] [CrossRef]
- Berthelot, H.; Bonnet, S.; Camps, M.; Grosso, O.; Moutin, T. Assessment of the dinitrogen released as ammonium and dissolved organic nitrogen by unicellular and filamentous marine diazotrophic cyanobacteria grown in culture. Front. Mar. Sci. 2015, 2, 80. [Google Scholar] [CrossRef] [Green Version]
- Benavides, M.; Houlbreque, F.; Camps, M.; Lorrain, A.; Grosso, O.; Bonnet, S. Diazotrophs: A non-negligible source of nitrogen for the tropical coral Stylophora pistillata. J. Exp. Biol. 2016, 219, 2608–2612. [Google Scholar] [CrossRef] [Green Version]
- Guillard, R.R.; Ryther, J.H. Studies on marine planktonic diatoms. I. Cyclotella nana Hustedt and Detonula confervacaea (Cleve) Gran. Can. J. Microbiol. 1962, 8, 229–239. [Google Scholar] [CrossRef]
- Biegala, I.; Raimbault, P. High abundance of diazotrophic picocyanobacteria (<3 μm) in a Southwest Pacific coral lagoon. Aquat. Microb. Ecol. 2008, 51, 45–53. [Google Scholar] [CrossRef]
- Henke, B.A.; Turk-Kubo, K.A.; Bonnet, S.; Zehr, J.P. Distributions and abundances of sublineages of the N2-fixing cyanobacterium Candidatus Atelocyanobacterium thalassa (UCYN-A) in the New Caledonian coral lagoon. Front. Microbiol. 2018, 9, 554. [Google Scholar] [CrossRef] [Green Version]
- Rodolfo-Metalpa, R.; Martin, S.; Ferrier-Pagès, C.; Gattuso, J.-P. Response of the temperate coral Cladocora caespitosa to mid- and long-term exposure to pCO2 and temperature levels projected for the year 2100 AD. Biogeosciences 2010, 7, 289–300. [Google Scholar] [CrossRef] [Green Version]
- Godinot, C.; Houlbrèque, F.; Grover, R.; Ferrier-Pagès, C. Coral uptake of inorganic phosphorus and nitrogen negatively affected by simultaneous changes in temperature and pH. PLoS ONE 2011, 6, e25024. [Google Scholar] [CrossRef] [Green Version]
- Jeffrey, S.W.; Humphrey, G.F. New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem. Und Physiol. Der Pflanz. 1975, 167, 191–194. [Google Scholar] [CrossRef]
- Hoogenboom, M.; Beraud, E.; Ferrier-Pagès, C. Relationship between symbiont density and photosynthetic carbon acquisition in the temperate coral Cladocora caespitosa. Coral Reefs 2010, 29, 21–29. [Google Scholar] [CrossRef]
- Stimson, J.; Kinzie, R.A., III. The temporal pattern and rate of release of zooxanthellae from the reef coral Pocillopora damicornis (Linnaeus) under nitrogen-enrichment and control conditions. J. Exp. Mar. Biol. Ecol. 1991, 153, 63–74. [Google Scholar] [CrossRef]
- Naumann, M.S.; Richter, C.; El-Zibdah, M.; Wild, C. Coral mucus as an efficient trap for picoplanktonic cyanobacteria: Implications for pelagic-benthic coupling in the reef ecosystem. Mar. Ecol. Prog. Ser. 2009, 385, 65–76. [Google Scholar] [CrossRef]
- Smith, K.; Krohn, R.I.; Hermanson, G.T.; Mallia, A.K.; Gartner, F.H.; Provenzano, M.D.; Fujimoto, E.K.; Goeke, N.M.; Olson, B.J.; Klenk, B.C. Measurement of protein using bicinchoninic acid. Anal. Biochem. 1985, 150, 76–85. [Google Scholar] [CrossRef]
- Spencer Davies, P. Short-term growth measurements of corals using an accurate buoyant weighing technique. Mar. Biol. 1989, 101, 389–395. [Google Scholar] [CrossRef]
- Schreiber, U.; Schliwa, U.; Bilger, W. Continuous recording of photochemical and non-photochemical chlorophyll fluorescence quenching with a new type of modulation fluorometer. Photosynth. Res. 1986, 10, 51–62. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Jones, R.J. Photoinhibition and photoprotection in symbiotic dinoflagellates from reef-building corals. Mar. Ecol. Prog. Ser. 1999, 183, 73–86. [Google Scholar] [CrossRef] [Green Version]
- Biscéré, T.; Rodolfo-Metalpa, R.; Lorrain, A.; Chauvaud, L.; Thébault, J.; Clavier, J.; Houlbrèque, F. Responses of two scleractinian corals to cobalt pollution and ocean acidification. PLoS ONE 2015, 10, e0122898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fitt, W.; Brown, B.; Warner, M.; Dunne, R. Coral bleaching: Interpretation of thermal tolerance limits and thermal thresholds in tropical corals. Coral Reefs 2001, 20, 51–65. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Grottoli, G.; Rodrigues, L.J. Bleached Porites compressa and Montipora capitata corals catabolize δ13C-enriched lipids. Coral Reefs 2011, 30, 687. [Google Scholar] [CrossRef]
- Baumann, J.; Grottoli, A.G.; Hughes, A.D.; Matsui, Y. Photoautotrophic and heterotrophic carbon in bleached and non-bleached coral lipid acquisition and storage. J. Exp. Mar. Biol. Ecol. 2014, 461, 469–478. [Google Scholar] [CrossRef]
- Meunier, V.; Bonnet, S.; Benavides, M.; Ravache, A.; Grosso, O.; Lambert, C.; Houlbrèque, F. Diazotroph-Derived Nitrogen Assimilation Strategies Differ by Scleractinian Coral Species. Front. Mar. Sci. 2021, 8, 1018. [Google Scholar] [CrossRef]
- Connolly, S.R.; Lopez-Yglesias, M.A.; Anthony, K.R.N. Food availability promotes rapid recovery from thermal stress in a scleractinian coral. Coral Reefs 2012, 31, 951–960. [Google Scholar] [CrossRef]
- Tremblay, P.; Gori, A.; Maguer, J.F.; Hoogenboom, M.; Ferrier-Pagès, C. Heterotrophy promotes the re-establishment of photosynthate translocation in a symbiotic coral after heat stress. Sci. Rep. 2016, 6, 38112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benavides, M.; Agawin, N.; Arístegui, J.; Peene, J.; Stal, L. Dissolved organic nitrogen and carbon release by a marine unicellular diazotrophic cyanobacterium. Aquat. Microb. Ecol. 2013, 69, 69–80. [Google Scholar] [CrossRef] [Green Version]
- Glibert, P.M.; Bronk, D.A. Release of dissolved organic nitrogen by marine diazotrophic cyanobacteria, Trichodesmium spp. Appl. Environ. Microbiol. 1994, 60, 3996–4000. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muscatine, L.; Falkowski, P.G.; Dubinsky, Z.; Cook, P.A.; McCloskey, L.R. The effect of external nutrient resources on the population dynamics of zooxanthellae in a reef coral. Proc. R. Soc. Lond. 1989, 236, 311–324. [Google Scholar] [CrossRef]
- Houlbrèque, F.; Tambutté, E.; Allemand, D.; Ferrier-Pagès, C. Interactions between zooplankton feeding, photosynthesis and skeletal growth in the scleractinian coral Stylophora pistillata. J. Exp. Biol. 2004, 207, 1461–1469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lyndby, N.H.; Holm, J.B.; Wangpraseurt, D.; Grover, R.; Rottier, C.; Kühl, M.; Ferrier-Pagès, C. Effect of temperature and feeding on carbon budgets and O2 dynamics in Pocillopora damicornis. Mar. Ecol. Prog. Ser. 2020, 652, 49–62. [Google Scholar] [CrossRef]
- Grover, R.; Maguer, J.F.; Reynaud-Vaganay, S.; Ferrier-Pagès, C. Uptake of ammonium by the scleractinian coral Stylophora pistillata: Effect of feeding, light, and ammonium concentrations. Limnol. Oceanogr. 2002, 47, 782–790. [Google Scholar] [CrossRef] [Green Version]
- Pernice, M.; Meibom, A.; Heuvel, A.V.D.; Kopp, C.; Domart-Coulon, I.; Hoegh-Guldberg, O.; Dove, S. A single-cell view of ammonium assimilation in coral-dinoflagellate symbiosis. ISME J. 2012, 6, 1314–1324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanaka, Y.; Grottoli, A.G.; Matsui, Y.; Suzuki, A.; Sakai, K. Partitioning of nitrogen sources to algal endosymbionts of corals with long-term 15N-labelling and a mixing model. Ecol. Modell. 2015, 309–310, 163–169. [Google Scholar] [CrossRef]
- Bonnet, S.; Webb, E.A.; Panzeca, C.; Karl, D.M.; Capone, D.G.; Wilhelmy, S.A.S. Vitamin B 12 excretion by cultures of the marine cyanobacteria Crocosphaera and Synechococcus. Limnol. Oceanogr. 2010, 55, 1959–1964. [Google Scholar] [CrossRef] [Green Version]
- Agostini, S.; Suzuki, Y.; Higuchi, T.; Casareto, B.E.; Yoshinaga, K.; Nakano, Y.; Fujimura, H. Biological and chemical characteristics of the coral gastric cavity. Coral Reefs 2012, 31, 147–156. [Google Scholar] [CrossRef]
- Dubinsky, Z.; Stambler, N.; Ben-Zion, M.; McCloskey, L.R.; Muscatine, L.; Falkowski, P.G. The effect of external nutrient resources on the optical properties and photosynthetic efficiency of Stylophora pistillata. Proc. R. Soc. B Biol. Sci. 1990, 239, 231–246. [Google Scholar] [CrossRef]
- Davy, S.; Cook, C. The relationship between nutritional status and carbon flux in the zooxanthellate sea anemone Aiptasia pallida. Mar. Biol. 2001, 139, 999–1005. [Google Scholar] [CrossRef]
- Ferrier-Pagès, C.; Witting, J.; Tambutté, E.; Sebens, K.P. Effect of natural zooplankton feeding on the tissue and skeletal growth of the scleractinian coral Stylophora pistillata. Coral Reefs 2003, 22, 229–240. [Google Scholar] [CrossRef]
- Jacques, G.; Pilson, M.E.Q. Experimental ecology of the temperate scleractinian coral Astrangia danae I. Partition of respiration, photosynthesis and calcification between host and symbionts. Mar. Biol. 1980, 60, 167–178. [Google Scholar] [CrossRef]
- Witting, J.H. Zooplankton capture and coral growth: The role of heterotrophy in Carribean reef corals. Ph.D. Thesis, Northeastern University, Boston, MA, USA, 1999. [Google Scholar]
- Simkiss, K.; Wilbur, K.M. Biomineralization: Cell Biology and Mineral Deposition; Academic Press: San Diego, CA, USA, 1989. [Google Scholar]
- Puverel, S.; Tambutté, E.; Pereira-Mouriès, L.; Zoccola, D.; Allemand, D.; Tambutté, S. Soluble organic matrix of two Scleractinian corals: Partial and comparative analysis. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2005, 141, 480–487. [Google Scholar] [CrossRef]
- Puverel, S.; Houlbrèque, F.; Tambutté, E.; Zoccola, D.; Payan, P.; Caminiti, N.; Tambutté, S.; Allemand, D. Evidence of low molecular weight components in the organic matrix of the reef building coral, Stylophora pistillata. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2007, 147, 850–856. [Google Scholar] [CrossRef]
- Schoepf, V.; Grottoli, A.G.; Warner, M.E.; Cai, W.-J.; Melman, T.F.; Hoadley, K.D.; Pettay, D.T.; Hu, X.P.; Li, Q.; Xu, H.; et al. Coral Energy Reserves and Calcification in a High-CO2 World at Two Temperatures. PLoS ONE 2013, 8, e75049. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Béraud, E.; Gevaert, F.; Rottier, C.; Ferrier-Pagès, C. The response of the scleractinian coral Turbinaria reniformis to thermal stress depends on the nitrogen status of the coral holobiont. J. Exp. Biol. 2013, 216, 2665–2674. [Google Scholar] [CrossRef] [Green Version]
- Borell, E.M.; Bischof, K. Feeding sustains photosynthetic quantum yield of a scleractinian coral during thermal stress. Oecologia 2008, 157, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.J.; Suggett, D.J.; Baker, N.R. Is photoinhibition of zooxanthellae photosynthesis the primary cause of thermal bleaching in corals? Glob. Chang. Biol. 2005, 11, 1–11. [Google Scholar] [CrossRef]
- Yakovleva, I.; Bhagooli, R.; Takemura, A.; Hidaka, M. Differential susceptibility to oxidative stress of two scleractinian corals: Antioxidant functioning of mycosporine-glycine. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2004, 139, 721–730. [Google Scholar] [CrossRef]
- Rädecker, N.; Pogoreutz, C.; Gegner, H.M.; Cardenas, A.; Perna, G.; Geissler, L.; Roth, F.; Bougoure, J.; Guagliardo, P.; Struck, U.; et al. Heat stress reduces the contribution of diazotrophs to coral holobiont nitrogen cycling. ISME J. 2021. [Google Scholar] [CrossRef]
- Benavides, M.; Bednarz, V.N.; Ferrier-Pagès, C. Diazotrophs: Overlooked key players within the coral symbiosis and tropical reef ecosystems? Front. Mar. Sci. 2017, 4. [Google Scholar] [CrossRef] [Green Version]
- Meunier, V.; Geissler, L.; Bonnet, S.; Rädecker, N.; Perna, G.; Grosso, O.; Lambert, C.; Rodolfo-Metalpa, R.; Voolstra, C.R.; Houlbrèque, F. Microbes support enhanced nitrogen requirements of coral holobionts in a high CO2 environment. Mol. Ecol. 2021, 30, 5888–5899. [Google Scholar] [CrossRef] [PubMed]
- Bindoff, L.N.; Cheung, W.W.; Kairo, G.J.; Aristegui, J.; Guinder, A.V.; Hallberg, R. Changing Ocean, marine ecosystems, and dependent communities. In IPCC Special Report on the Ocean and Cryosphere in a Changing Climate; Pörtner, H.O., Roberts, D.C., Masson-Delmotte, V., Zhai, P., Tignor, M., Poloczanska, E., Mintenbeck, K., Alegria, A., Nicolai, M., Okem, A., et al., Eds.; IPCC: Geneva, Switzerland, 2019. [Google Scholar]
- Hughes, T.P.; Anderson, K.D.; Connolly, S.R.; Heron, S.F.; Kerry, J.T.; Baird, A.H.; Baum, J.K.; Lough, J.M.; Berumen, M.L.; Bridge, T.C.; et al. Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science 2018, 359, 80–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoey, A.; Howells, E.; Johansen, J.; Hobbs, J.; Messmer, V.; McCowan, D.; Wilson, S.; Pratchett, M. Recent advances in understanding the effects of climate change on coral reefs. Diversity 2016, 8, 12. [Google Scholar] [CrossRef] [Green Version]
- Jin, K.; Lundgren, P.; Lutz, A.; Raina, J.-B.; Howells, E.J.; Paley, A.S.; Willis, B.L.; van Oppen, M.J.H. Genetic markers for antioxidant capacity in a reef-building coral. Sci. Adv. 2016, 2, e1500842. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Genus | Morphological Type | Size | Location |
|---|---|---|---|
| Xenococcus spp. | Unicellular | ∼6 µm | Noumea lagoon |
| Cyanothece spp. | Unicellular | ∼5.5 µm | Noumea lagoon |
| Calothrix spp. | Filamentous heterocystous | ∼50 µm | WTSP Ocean |
| Rivularia spp. | Filamentous heterocystous | ∼30 µm | WTSP Ocean |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meunier, V.; Bonnet, S.; Camps, M.; Benavides, M.; Dubosc, J.; Rodolfo-Metalpa, R.; Houlbrèque, F. Ingestion of Diazotrophs Makes Corals More Resistant to Heat Stress. Biomolecules 2022, 12, 537. https://doi.org/10.3390/biom12040537
Meunier V, Bonnet S, Camps M, Benavides M, Dubosc J, Rodolfo-Metalpa R, Houlbrèque F. Ingestion of Diazotrophs Makes Corals More Resistant to Heat Stress. Biomolecules. 2022; 12(4):537. https://doi.org/10.3390/biom12040537
Chicago/Turabian StyleMeunier, Valentine, Sophie Bonnet, Mercedes Camps, Mar Benavides, Jeff Dubosc, Riccardo Rodolfo-Metalpa, and Fanny Houlbrèque. 2022. "Ingestion of Diazotrophs Makes Corals More Resistant to Heat Stress" Biomolecules 12, no. 4: 537. https://doi.org/10.3390/biom12040537
APA StyleMeunier, V., Bonnet, S., Camps, M., Benavides, M., Dubosc, J., Rodolfo-Metalpa, R., & Houlbrèque, F. (2022). Ingestion of Diazotrophs Makes Corals More Resistant to Heat Stress. Biomolecules, 12(4), 537. https://doi.org/10.3390/biom12040537







