Oxidative Stress and Phototherapy in Atopic Dermatitis: Mechanisms, Role, and Future Perspectives
Abstract
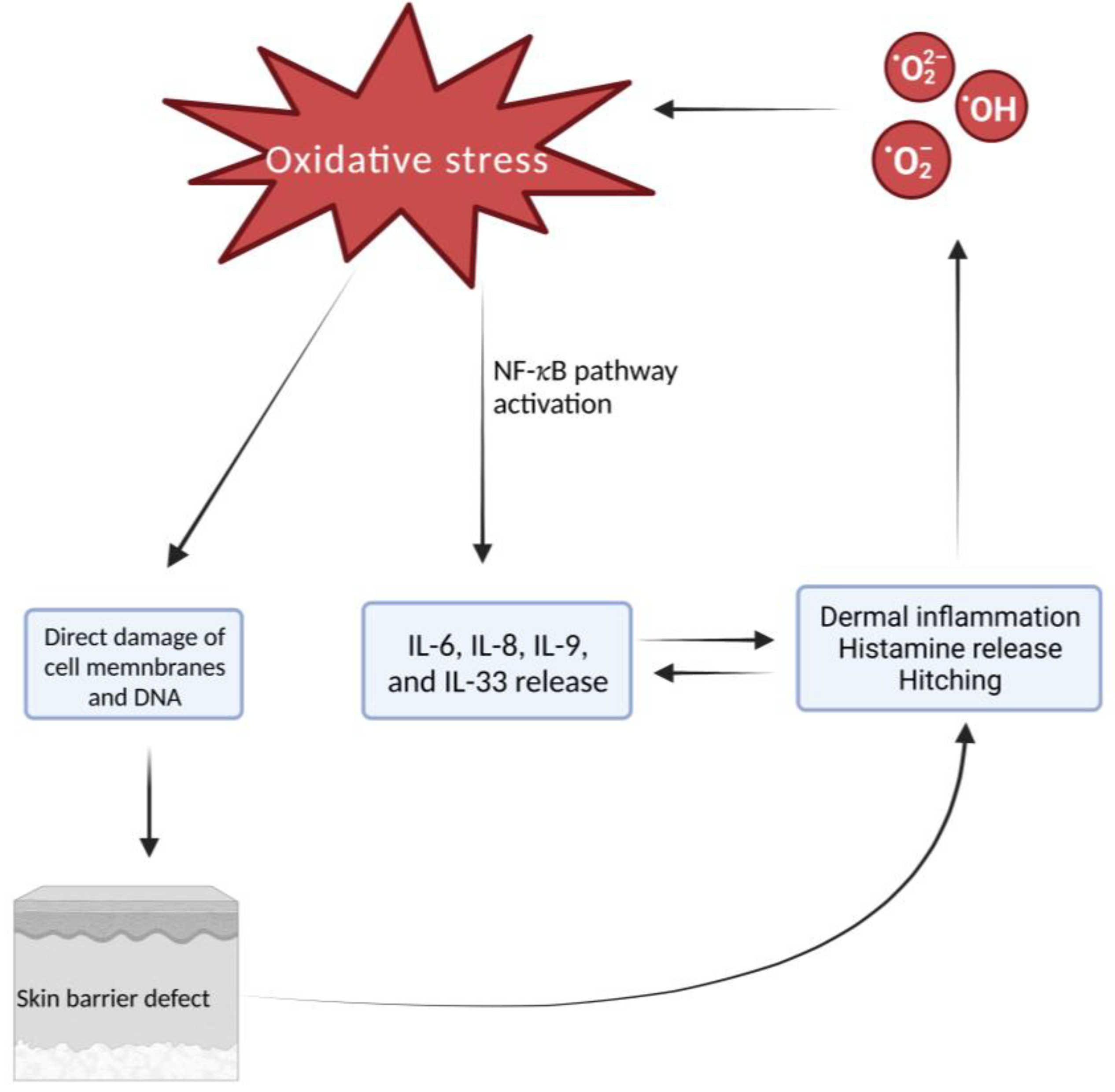
1. Introduction
1.1. Atopic Dermatitis Pathogenesis: Role of Oxidative Stress
1.2. Marker of Oxidative Stress in Atopic Dermatitis
1.2.1. AHR/ARNT-NRF2 Crosstalk
1.2.2. PONs
2. Atopic Dermatitis Treatment
2.1. Topical, Systemic, and Biological Treatment
2.2. Phototherapy
2.2.1. Ultraviolet Sources
2.2.2. Phototherapy Immunomodulatory Effects
2.2.3. Role of Phototherapy during COVID-19 Pandemic
3. Atopic Dermatitis and Other Chronic Inflammatory Cutaneous Diseases
4. Potential Targets of Oxidative Stress during Phototherapy in Atopic Dermatitis
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Campanati, A.; Bianchelli, T.; Gesuita, R.; Foti, C.; Malara, G.; Micali, G.; Amerio, P.; Rongioletti, F.; Corazza, M.; Patrizi, A.; et al. Comorbidities and treatment patterns in adult patients with atopic dermatitis: Results from a nationwide multicenter study. Arch. Dermatol. Res. 2021, 314, 593–603. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Gu, X.; Huang, Y.; Zhu, W.; Shen, M. Phototherapy for atopic dermatitis: Systematic review and network meta-analysis of randomized controlled trials. Photodermatol. Photoimmunol. Photomed. 2022, 38, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Mastorino, L.; Rosset, F.; Gelato, F.; Ortoncelli, M.; Cavaliere, G.; Quaglino, P.; Ribero, S. Chronic pruritus in atopic patients treated with dupilumab: Real life response and related parameters in 354 patients. Pharmaceuticals 2022, 15, 883. [Google Scholar] [CrossRef] [PubMed]
- Miniotti, M.; Lazzarin, G.; Ortoncelli, M.; Mastorino, L.; Ribero, S.; Leombruni, P. Impact on health-related quality of life and symptoms of anxiety and depression after 32 weeks of dupilumab treatment for moderate-to-severe atopic dermatitis. Dermatol. Ther. 2022, 35. [Google Scholar] [CrossRef] [PubMed]
- Tintle, S.; Shemer, A.; Suárez-Fariñas, M.; Fujita, H.; Gilleaudeau, P.; Sullivan-Whalen, M.; Johnson-Huang, L.; Chiricozzi, A.; Cardinale, I.; Duan, S.; et al. Reversal of atopic dermatitis with narrow-band uvb phototherapy and biomarkers for therapeutic response. J. Allergy Clin. Immunol. 2011, 128, 583–593. [Google Scholar] [CrossRef]
- Kamiya, K.; Saeki, H.; Tokura, Y.; Yoshihara, S.; Sugai, J.; Ohtsuki, M. Clinical practice guidelines for the diagnosis and management of atopic dermatitis. Asian. Pac. J. Allergy Immunol. 2021, 11, 6477. [Google Scholar] [CrossRef]
- Galiniak, S.; Mołoń, M.; Biesiadecki, M.; Bożek, A.; Rachel, M. The role of oxidative stress in atopic dermatitis and chronic urticaria. Antioxidants 2022, 11, 1590. [Google Scholar] [CrossRef]
- Bertino, L.; Guarneri, F.; Cannavò, S.P.; Casciaro, M.; Pioggia, G.; Gangemi, S. Oxidative stress and atopic dermatitis. Antioxidants 2020, 9, 196. [Google Scholar] [CrossRef]
- Borgia, F.; Custurone, P.; Peterle, L.; Pioggia, G.; Gangemi, S. Role of epithelium-derived cytokines in atopic dermatitis and psoriasis: Evidence and therapeutic perspectives. Biomolecules 2021, 11, 1843. [Google Scholar] [CrossRef]
- Albano, G.D.; Gagliardo, R.P.; Montalbano, A.M.; Profita, M. Overview of the Mechanisms of Oxidative Stress: Impact in Inflammation of the Airway Diseases. Antioxidants 2022, 11, 2237. [Google Scholar] [CrossRef]
- Khan, A.Q.; Agha, M.V.; Sheikhan, K.S.A.M.; Younis, S.M.; Tamimi, M.A.; Alam, M.; Ahmad, A.; Uddin, S.; Buddenkotte, J.; Steinhoff, M. Targeting Deregulated Oxidative Stress in Skin Inflammatory Diseases: An Update on Clinical Importance. Biomed. Pharmacother. 2022, 154, 113601. [Google Scholar] [CrossRef] [PubMed]
- Moliteo, E.; Sciacca, M.; Palmeri, A.; Papale, M.; Manti, S.; Parisi, G.F.; Leonardi, S. Cystic Fibrosis and Oxidative Stress: The Role of CFTR. Molecules 2022, 27, 5324. [Google Scholar] [CrossRef] [PubMed]
- Dizdaroglu, M. Oxidatively Induced DNA Damage and Its Repair in Cancer. Mutat. Res. Rev. Mutat. Res. 2015, 763, 212–245. [Google Scholar] [CrossRef] [PubMed]
- Hebert, A.A. Oxidative Stress as a Treatment Target in Atopic Dermatitis: The Role of Furfuryl Palmitate in Mild-to-Moderate Atopic Dermatitis. Int. J. Womens. Dermatol. 2020, 6, 331–333. [Google Scholar] [CrossRef]
- Almela, R.M.; Rubio, C.P.; Cerón, J.J.; Ansón, A.; Tichy, A.; Mayer, U. Selected Serum Oxidative Stress Biomarkers in Dogs with Non-Food-Induced and Food-Induced Atopic Dermatitis. Vet. Dermatol. 2018, 29, 229-e82. [Google Scholar] [CrossRef]
- Furue, M.; Hashimoto-Hachiya, A.; Tsuji, G. Aryl Hydrocarbon Receptor in Atopic Dermatitis and Psoriasis. Int. J. Mol. Sci. 2019, 20, 5424. [Google Scholar] [CrossRef]
- Furue, M. Regulation of Filaggrin, Loricrin, and Involucrin by IL-4, IL-13, IL-17A, IL-22, AHR, and NRF2: Pathogenic Implications in Atopic Dermatitis. Int. J. Mol. Sci. 2020, 21, 5382. [Google Scholar] [CrossRef]
- Napolitano, M.; Fabbrocini, G.; Martora, F.; Picone, V.; Morelli, P.; Patruno, C. Role of Aryl Hydrocarbon Receptor Activation in Inflammatory Chronic Skin Diseases. Cells 2021, 10, 3559. [Google Scholar] [CrossRef]
- Szelest, M.; Walczak, K.; Plech, T. A New Insight into the Potential Role of Tryptophan-Derived AhR Ligands in Skin Physiological and Pathological Processes. Int. J. Mol. Sci. 2021, 22, 1104. [Google Scholar] [CrossRef]
- Morris, G.; Gevezova, M.; Sarafian, V.; Maes, M. Redox Regulation of the Immune Response. Cell. Mol. Immunol. 2022, 19, 1079–1101. [Google Scholar] [CrossRef]
- Edamitsu, T.; Taguchi, K.; Okuyama, R.; Yamamoto, M. AHR and NRF2 in Skin Homeostasis and Atopic Dermatitis. Antioxidants 2022, 11, 227. [Google Scholar] [CrossRef] [PubMed]
- Wysocka, A.; Zwolak, A. The Relevance of Noncoding DNA Variations of Paraoxonase Gene Cluster in Atherosclerosis-Related Diseases. Int. J. Mol. Sci. 2021, 22, 2137. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, C.J.; Lamichhane, S.; Connolly, J.A.; Soehnlen, S.M.; Khalaf, F.K.; Malhotra, D.; Haller, S.T.; Isailovic, D.; Kennedy, D.J. A PON for All Seasons: Comparing Paraoxonase Enzyme Substrates, Activity and Action Including the Role of PON3 in Health and Disease. Antioxidants 2022, 11, 590. [Google Scholar] [CrossRef] [PubMed]
- Simonetti, O.; Bacchetti, T.; Ferretti, G.; Molinelli, E.; Rizzetto, G.; Bellachioma, L.; Offidani, A. Oxidative Stress and Alterations of Paraoxonases in Atopic Dermatitis. Antioxidants 2021, 10, 697. [Google Scholar] [CrossRef]
- Ji, H.; Li, X. Oxidative Stress in Atopic Dermatitis. Oxid. Med. Cell. Longev. 2016, 23, 1–8. [Google Scholar] [CrossRef]
- Kleinman, E.; Laborada, J.; Metterle, L.; Eichenfield, L.F. What’s New in Topicals for Atopic Dermatitis? Am. J. Clin. Dermatol. 2022, 23, 595–603. [Google Scholar] [CrossRef]
- Eichenfield, L.F.; Tom, W.L.; Berger, T.G.; Krol, A.; Paller, A.S.; Schwarzenberger, K.; Bergman, J.N.; Chamlin, S.L.; Cohen, D.E.; Cooper, K.D.; et al. Guidelines of Care for the Management of Atopic Dermatitis. J. Am. Acad. Dermatol. 2014, 71, 116–132. [Google Scholar] [CrossRef]
- Asgari, M.M.; Tsai, A.-L.; Avalos, L.; Sokil, M.; Quesenberry, C.P. Association Between Topical Calcineurin Inhibitor Use and Keratinocyte Carcinoma Risk Among Adults With Atopic Dermatitis. JAMA. Dermatol. 2020, 156, 1066. [Google Scholar] [CrossRef]
- Paller, A.S.; Tom, W.L.; Lebwohl, M.G.; Blumenthal, R.L.; Boguniewicz, M.; Call, R.S.; Eichenfield, L.F.; Forsha, D.W.; Rees, W.C.; Simpson, E.L.; et al. Efficacy and Safety of Crisaborole Ointment, a Novel, Nonsteroidal Phosphodiesterase 4 (PDE4) Inhibitor for the Topical Treatment of Atopic Dermatitis (AD) in Children and Adults. J. Am. Acad. Dermatol. 2016, 75, 494–503e6. [Google Scholar] [CrossRef]
- Pao-Ling Lin, C.; Gordon, S.; Her, M.J.; Rosmarin, D. A Retrospective Study: Application Site Pain with the Use of Crisaborole, a Topical Phosphodiesterase 4 Inhibitor. J. Am. Acad. Dermatol. 2019, 80, 1451–1453. [Google Scholar] [CrossRef]
- Anonymous. Revolutionizing Atopic Dermatitis, 13–14 December 2020. Br. J. Dermatol. 2021, 184. [Google Scholar] [CrossRef]
- Saeki, H.; Imamura, T.; Yokota, D.; Tsubouchi, H. Difamilast Ointment in Japanese Adult and Pediatric Patients with Atopic Dermatitis: A Phase III, Long-Term, Open-Label Study. Dermatol. Ther. 2022, 12, 1589–1601. [Google Scholar] [CrossRef]
- Owji, S.; Caldas, S.A.; Ungar, B. Management of Atopic Dermatitis: Clinical Utility of Ruxolitinib. J. Asthma Allergy 2022, 15, 1527–1537. [Google Scholar] [CrossRef] [PubMed]
- Blauvelt, A.; Szepietowski, J.C.; Papp, K.; Simpson, E.L.; Silverberg, J.I.; Kim, B.S.; Kwatra, S.G.; Kuligowski, M.E.; Venturanza, M.E.; Sun, K.; et al. 26884 Ruxolitinib Cream Rapidly Decreases Pruritus in Atopic Dermatitis: Pooled Results from Two Phase 3 Studies. J. Am. Acad. Dermatol. 2021, 85, AB121. [Google Scholar] [CrossRef]
- Chovatiya, R.; Paller, A.S. JAK Inhibitors in the Treatment of Atopic Dermatitis. J. Allergy. Clin. Immunol. 2021, 148, 927–940. [Google Scholar] [CrossRef] [PubMed]
- Keam, S.J. Tapinarof Cream 1%: First Approval. Drugs 2022, 82, 1221–1228. [Google Scholar] [CrossRef]
- Paller, A.S.; Stein Gold, L.; Soung, J.; Tallman, A.M.; Rubenstein, D.S.; Gooderham, M. Efficacy and Patient-Reported Outcomes from a Phase 2b, Randomized Clinical Trial of Tapinarof Cream for the Treatment of Adolescents and Adults with Atopic Dermatitis. J. Am. Acad. Dermatol. 2021, 84, 632–638. [Google Scholar] [CrossRef]
- Smith, S.H.; Jayawickreme, C.; Rickard, D.J.; Nicodeme, E.; Bui, T.; Simmons, C.; Coquery, C.M.; Neil, J.; Pryor, W.M.; Mayhew, D.; et al. Tapinarof Is a Natural AhR Agonist That Resolves Skin Inflammation in Mice and Humans. J. Investig. Dermatol. 2017, 137, 2110–2119. [Google Scholar] [CrossRef]
- Simpson, E.L.; Bieber, T.; Guttman-Yassky, E.; Beck, L.A.; Blauvelt, A.; Cork, M.J.; Silverberg, J.I.; Deleuran, M.; Kataoka, Y.; Lacour, J.-P.; et al. Two Phase 3 Trials of Dupilumab versus Placebo in Atopic Dermatitis. N. Engl. J. Med. 2016, 375, 2335–2348. [Google Scholar] [CrossRef]
- Tazawa, T.; Sugiura, H.; Sugiura, Y.; Uehara, M. Relative Importance of IL-4 and IL-13 in Lesional Skin of Atopic Dermatitis. Arch. Dermatol. Res. 2004, 295, 459–464. [Google Scholar] [CrossRef]
- Zhang, Y.; Jing, D.; Cheng, J.; Chen, X.; Shen, M.; Liu, H. The Efficacy and Safety of IL-13 Inhibitors in Atopic Dermatitis: A Systematic Review and Meta-Analysis. Front. Immunol. 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Puar, N.; Chovatiya, R.; Paller, A.S. New Treatments in Atopic Dermatitis. Ann. Allergy Asthma Immunol. 2021, 126, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Borgia, F.; Custurone, P.; Li Pomi, F.; Cordiano, R.; Alessandrello, C.; Gangemi, S. IL-31: State of the Art for an Inflammation-Oriented Interleukin. Int. J. Mol. Sci. 2022, 23, 6507. [Google Scholar] [CrossRef] [PubMed]
- Kabashima, K.; Matsumura, T.; Komazaki, H.; Kawashima, M. Nemolizumab plus Topical Agents in Patients with Atopic Dermatitis (AD) and Moderate-to-severe Pruritus Provide Improvement in Pruritus and Signs of AD for up to 68 Weeks: Results from Two Phase III, Long-term Studies*. Br. J. Dermatol. 2022, 186, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Myers, E.; Kheradmand, S.; Miller, R. An Update on Narrowband Ultraviolet B Therapy for the Treatment of Skin Diseases. Cureus 2021, 13, e19182. [Google Scholar] [CrossRef] [PubMed]
- Barros, N.D.M.; Sbroglio, L.L.; Buffara, M.D.O.; Baka, J.L.C.E.S.; Pessoa, A.D.S.; Azulay-Abulafia, L. Phototherapy. An. Bras. Dermatol. 2021, 96, 397–407. [Google Scholar] [CrossRef]
- Gittler, J.K.; Krueger, J.G.; Guttman-Yassky, E. Atopic Dermatitis Results in Intrinsic Barrier and Immune Abnormalities: Implications for Contact Dermatitis. J. Allergy Clin. Immunol. 2013, 131, 300–313. [Google Scholar] [CrossRef]
- Vieyra-Garcia, P.A.; Wolf, P. A Deep Dive into UV-Based Phototherapy: Mechanisms of Action and Emerging Molecular Targets in Inflammation and Cancer. Pharmacol. Ther. 2021, 222, 107784. [Google Scholar] [CrossRef]
- Müschen, A.; Mirmohammadsadegh, A.; Jarzebska-Deussen, B.; Abts, H.F.; Ruzicka, T.; Michel, G. Differential IL-10 Receptor Gene Expression in Acute versus Chronic Atopic Eczema. Modulation by Immunosuppressive Drugs and Cytokines in Normal Cultured Keratinocytes. Inflamm. Res. 1999, 48, 539–543. [Google Scholar] [CrossRef]
- Howell, M.D.; Kim, B.E.; Gao, P.; Grant, A.V.; Boguniewicz, M.; DeBenedetto, A.; Schneider, L.; Beck, L.A.; Barnes, K.C.; Leung, D.Y.M. Cytokine Modulation of Atopic Dermatitis Filaggrin Skin Expression. J. Allergy Clin. Immunol. 2009, 124, R7–R12. [Google Scholar] [CrossRef]
- Choi, J.Y.; Owusu-Ayim, M.; Dawe, R.; Ibbotson, S.; Fleming, C.; Foerster, J. Narrowband Ultraviolet B Phototherapy Is Associated with a Reduction in Topical Corticosteroid and Clinical Improvement in Atopic Dermatitis: A Historical Inception Cohort Study. Clin. Exp. Dermatol. 2021, 46, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Malinowska, K.; Woźniacka, A.; Bogaczewicz, J. The Impact of Medium Dose UVA1 Phototherapy on Pruritus, DLQI and SCORAD Index in Patients with Atopic Dermatitis. Postepy Dermatol. Alergol. 2020, 37, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T.; Chiang, K.-C.; Chen, C.-C.; Chen, P.-J.; Lai, C.-Y.; Hsu, L.-W.; Ohmori, N.; Goto, T.; Chen, C.-L.; Goto, S. Sunlight Exposure and Phototherapy: Perspectives for Healthy Aging in an Era of COVID-19. Int. J. Environ. Res. Public Health 2021, 18, 10950. [Google Scholar] [CrossRef] [PubMed]
- Granahan, A.; McCavana, J.; Lally, A.; Morgan, I.; Fitzgerald, S.; Moriarty, B. A Review of the Selection Process and Decontamination Methods with the Use of Face Shields in UV Phototherapy during the SARS-CoV-2 Pandemic. Skin Health Dis. 2022, 2. [Google Scholar] [CrossRef] [PubMed]
- Hanna, R.; Dalvi, S.; Sălăgean, T.; Bordea, I.R.; Benedicenti, S. Phototherapy as a Rational Antioxidant Treatment Modality in COVID-19 Management; New Concept and Strategic Approach: Critical Review. Antioxidants 2020, 9, 875. [Google Scholar] [CrossRef]
- Hanna, R.; Dalvi, S.; Sălăgean, T.; Pop, I.D.; Bordea, I.R.; Benedicenti, S. Understanding COVID-19 Pandemic: Molecular Mechanisms and Potential Therapeutic Strategies. An Evidence-Based Review. J. Inflamm. Res. 2021, 14, 13–56. [Google Scholar] [CrossRef]
- Conrado, P.C.V.; Sakita, K.M.; Arita, G.S.; Galinari, C.B.; Gonçalves, R.S.; Lopes, L.D.G.; Lonardoni, M.V.C.; Teixeira, J.J.V.; Bonfim-Mendonça, P.S.; Kioshima, E.S. A Systematic Review of Photodynamic Therapy as an Antiviral Treatment: Potential Guidance for Dealing with SARS-CoV-2. Photodiagnosis Photodyn. Ther. 2021, 34, 102221. [Google Scholar] [CrossRef]
- Speeckaert, R.; Lambert, J.; Puig, L.; Speeckaert, M.; Lapeere, H.; de Schepper, S.; van Geel, N. Vaccinations in Patients Receiving Systemic Drugs for Skin Disorders: What Can We Learn for SARS-Cov-2 Vaccination Strategies? Drugs R&D 2021, 21, 341–350. [Google Scholar] [CrossRef]
- Wack, S.; Patton, T.; Ferris, L.K. COVID-19 Vaccine Safety and Efficacy in Patients with Immune-Mediated Inflammatory Disease: Review of Available Evidence. J. Am. Acad. Dermatol. 2021, 85, 1274–1284. [Google Scholar] [CrossRef]
- Liew, S.-Y.; Tree, T.; Smith, C.H.; Mahil, S.K. The Impact of Immune-Modifying Treatments for Skin Diseases on the Immune Response to COVID-19 Vaccines: A Narrative Review. Curr. Dermatol. Rep. 2022, 11, 263–288. [Google Scholar] [CrossRef]
- Haddad, I.; Kozman, K.; Kibbi, A.-G. Navigating Patients with Atopic Dermatitis or Chronic Spontaneous Urticaria during the COVID-19 Pandemic. Front. Allergy 2022, 3, 809646. [Google Scholar] [CrossRef] [PubMed]
- Grabbe, S.; Beissert, S.; Enk, A. Systemic Immunosuppression in Times of COVID-19: Do We Need to Rethink Our Standards? J. Dtsch. Dermatol. Ges. 2020, 18, 810–813. [Google Scholar] [CrossRef] [PubMed]
- Sadeghinia, A.; Daneshpazhooh, M. Immunosuppressive Drugs for Patients with Psoriasis during the COVID-19 Pandemic Era. A Review. Dermatol. Ther. 2021, 34. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, C.M.; Hsiao, J.L.; Lio, P.A.; Shi, V.Y. Transitioning From Immunosuppressants to Dupilumab in Pediatric Atopic Dermatitis. Dermatitis 2021, 32, S4–S7. [Google Scholar] [CrossRef]
- Patruno, C.; Stingeni, L.; Fabbrocini, G.; Hansel, K.; Napolitano, M. Dupilumab and COVID-19: What Should We Expect? Dermatol. Ther. 2020, 33, e13502. [Google Scholar] [CrossRef]
- Jones, M.E.; Kohn, A.H.; Pourali, S.P.; Rajkumar, J.R.; Gutierrez, Y.; Yim, R.M.; Armstrong, A.W. The Use of Biologics During the COVID-19 Pandemic. Dermatol. Clin. 2021, 39, 545–553. [Google Scholar] [CrossRef]
- El-Qushayri, A.E.; Mahmoud, M.A.; Salman, S.; Sarsik, S.; Nardone, B. Dupilumab Therapy in Atopic Dermatitis Is Safe during COVID-19 Infection Era: A Systematic Review and Meta-analysis of 1611 Patients. Dermatol. Ther. 2022, 35, e15476. [Google Scholar] [CrossRef]
- Shah, A.A.; Sinha, A.A. Oxidative Stress and Autoimmune Skin Disease. Eur. J. Dermatol. 2013, 23, 5–13. [Google Scholar] [CrossRef]
- Li Pomi, F.; Borgia, F.; Custurone, P.; Vaccaro, M.; Pioggia, G.; Gangemi, S. Role of HMGB1 in Cutaneous Melanoma: State of the Art. Int. J. Mol. Sci. 2022, 23, 9327. [Google Scholar] [CrossRef]
- Georgescu, S.R.; Mitran, C.I.; Mitran, M.I.; Nicolae, I.; Matei, C.; Ene, C.D.; Popa, G.L.; Tampa, M. Oxidative Stress in Cutaneous Lichen Planus—A Narrative Review. J. Clin. Med. 2021, 10, 2692. [Google Scholar] [CrossRef]
- Prie, B.E.; Voiculescu, V.M.; Ionescu-Bozdog, O.B.; Petrutescu, B.; Iosif, L.; Gaman, L.E.; Clatici, V.G.; Stoian, I.; Giurcaneanu, C. Oxidative Stress and Alopecia Areata. J. Med. Life 2015, 8, 43–46. [Google Scholar] [PubMed]
- Vaccaro, M.; Irrera, N.; Cutroneo, G.; Rizzo, G.; Vaccaro, F.; Anastasi, G.; Borgia, F.; Cannavò, S.; Altavilla, D.; Squadrito, F. Differential Expression of Nitric Oxide Synthase Isoforms NNOS and INOS in Patients with Non-Segmental Generalized Vitiligo. Int. J. Mol. Sci. 2017, 18, 2533. [Google Scholar] [CrossRef] [PubMed]
- Motolese, A.; Ceccarelli, M.; Macca, L.; Li Pomi, F.; Ingrasciotta, Y.; Nunnari, G.; Guarneri, C. Novel Therapeutic Approaches to Psoriasis and Risk of Infectious Disease. Biomedicines 2022, 10, 228. [Google Scholar] [CrossRef]
- Dobrică, E.-C.; Cozma, M.-A.; Găman, M.-A.; Voiculescu, V.-M.; Găman, A.M. The Involvement of Oxidative Stress in Psoriasis: A Systematic Review. Antioxidants 2022, 11, 282. [Google Scholar] [CrossRef] [PubMed]
- Koca, R.; Armutcu, F.; Altinyazar, C.; Gürel, A. Evaluation of Lipid Peroxidation, Oxidant/Antioxidant Status, and Serum Nitric Oxide Levels in Alopecia Areata. Med. Sci. Monit. 2005, 11, CR296-299. [Google Scholar] [PubMed]
- Naziroglu, M.; Kokcam, I. Antioxidants and Lipid Peroxidation Status in the Blood of Patients with Alopecia. Cell. Biochem. Funct. 2000, 18, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Vaccaro, M.; Bagnato, G.; Cristani, M.; Borgia, F.; Spatari, G.; Tigano, V.; Saja, A.; Guarneri, F.; Cannavò, S.P.; Gangemi, S. Oxidation Products Are Increased in Patients Affected by Non-Segmental Generalized Vitiligo. Arch. Dermatol. Res. 2017, 309, 485–490. [Google Scholar] [CrossRef]
- Faraj, S.; Kemp, E.H.; Gawkrodger, D.J. Patho-Immunological Mechanisms of Vitiligo: The Role of the Innate and Adaptive Immunities and Environmental Stress Factors. Clin. Exp. Immunol. 2022, 207, 27–43. [Google Scholar] [CrossRef]
- Chen, S.; Wang, X.; Nisar, M.F.; Lin, M.; Zhong, J.L. Heme Oxygenases: Cellular Multifunctional and Protective Molecules against UV-Induced Oxidative Stress. Oxid. Med. Cell. Longev. 2019, 1–17. [Google Scholar] [CrossRef]
- Sorour, N.E.; Abd El-Kareem, H.M.; Ibrahim, A.E.; Salem, R.M. Nuclear Factor Erythroid-2-Related Factor 2 Gene Polymorphisms in Vitiligo. J. Clin. Aesthet. Dermatol. 2021, 14, 14–17. [Google Scholar]
- Sharma, C.K.; Sharma, M.; Prasad, K. Involvement of Different Genes Expressions during Immunological and Inflammatory Responses in Vitiligo. Crit. Rev. Eukaryot. Gene Expr. 2017, 27, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Romano-Lozano, V.; Cruz-Avelar, A.; Peralta Pedrero, M.L. Factor Nuclear Eritroide Similar al Factor 2 En El Vitíligo. Actas. Dermosifiliogr. 2022, 113, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Xuan, Y.; Yang, Y.; Xiang, L.; Zhang, C. The Role of Oxidative Stress in the Pathogenesis of Vitiligo: A Culprit for Melanocyte Death. Oxid. Med. Cell Longev. 2022, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Meng, X.; Song, Z.; Lin, J. Nuclear Factor Erythroid 2-Related Factor 2 (Nrf2) as a Potential Therapeutic Target for Vitiligo. Arch. Biochem. Biophys. 2020, 696, 108670. [Google Scholar] [CrossRef] [PubMed]
- Panieri, E.; Telkoparan-Akillilar, P.; Saso, L. NRF2, a Crucial Modulator of Skin Cells Protection against Vitiligo, Psoriasis, and Cancer. BioFactors 2022. [Google Scholar] [CrossRef]
- Wu, W.; Peng, G.; Yang, F.; Zhang, Y.; Mu, Z.; Han, X. Sulforaphane Has a Therapeutic Effect in an Atopic Dermatitis Murine Model and Activates the Nrf2/HO-1 Axis. Mol. Med. Rep. 2019, Aug 20, 1761–1771. [Google Scholar] [CrossRef]
- York, N.R.; Jacobe, H.T. UVA1 Phototherapy: A Review of Mechanism and Therapeutic Application. Int. J. Dermatol. 2010, 49, 623–630. [Google Scholar] [CrossRef]
- Bernerd, F.; Passeron, T.; Castiel, I.; Marionnet, C. The Damaging Effects of Long UVA (UVA1) Rays: A Major Challenge to Preserve Skin Health and Integrity. Int. J. Mol. Sci. 2022, 23, 8243. [Google Scholar] [CrossRef]
- Broekgaarden, M.; Weijer, R.; van Gulik, T.M.; Hamblin, M.R.; Heger, M. Tumor Cell Survival Pathways Activated by Photodynamic Therapy: A Molecular Basis for Pharmacological Inhibition Strategies. Cancer Metast. Rev. 2015, 34, 643–690. [Google Scholar] [CrossRef]
- Ryšavá, A.; Vostálová, J.; Rajnochová Svobodová, A. Effect of Ultraviolet Radiation on the Nrf2 Signaling Pathway in Skin Cells. Int. J. Radiat. Biol. 2021, 97, 1383–1403. [Google Scholar] [CrossRef]
- Vieyra-Garcia, P.A.; Wolf, P. From Early Immunomodulatory Triggers to Immunosuppressive Outcome: Therapeutic Implications of the Complex Interplay Between the Wavebands of Sunlight and the Skin. Front. Med. 2018, 5, 232. [Google Scholar] [CrossRef] [PubMed]

| Topic | Author, Reference | Os Marker | Cytoprotective Mechanism | Cellular Damage Mechanism |
|---|---|---|---|---|
| UVA1 | Bernerd et al. [31] | NRF2 | Upregulation of NRF2 target genes: HMOX1, TXNRD1, NQO1, FTL, GCLM, AKR1C2, AKR1C3 | // |
| PDT | Broekgaarden et al. [32] | NRF2 | Activation of JNK and NRF2-KEAP1 dissociation. Activation of ARE sequences by NRF2-AP1 and transcription of NRF2 target genes. Upregulation of HMOX1 HIF-1-mediated genes. | // |
| UVB | Ryšavá et al. [33] | NRF2 | // | Direct DNA damage and downregulation of NRF2 target genes |
| UVB | Vieyra-Garcia et al. [34] | // | // | NLRP3 inflammasome activation |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borgia, F.; Li Pomi, F.; Vaccaro, M.; Alessandrello, C.; Papa, V.; Gangemi, S. Oxidative Stress and Phototherapy in Atopic Dermatitis: Mechanisms, Role, and Future Perspectives. Biomolecules 2022, 12, 1904. https://doi.org/10.3390/biom12121904
Borgia F, Li Pomi F, Vaccaro M, Alessandrello C, Papa V, Gangemi S. Oxidative Stress and Phototherapy in Atopic Dermatitis: Mechanisms, Role, and Future Perspectives. Biomolecules. 2022; 12(12):1904. https://doi.org/10.3390/biom12121904
Chicago/Turabian StyleBorgia, Francesco, Federica Li Pomi, Mario Vaccaro, Clara Alessandrello, Vincenzo Papa, and Sebastiano Gangemi. 2022. "Oxidative Stress and Phototherapy in Atopic Dermatitis: Mechanisms, Role, and Future Perspectives" Biomolecules 12, no. 12: 1904. https://doi.org/10.3390/biom12121904
APA StyleBorgia, F., Li Pomi, F., Vaccaro, M., Alessandrello, C., Papa, V., & Gangemi, S. (2022). Oxidative Stress and Phototherapy in Atopic Dermatitis: Mechanisms, Role, and Future Perspectives. Biomolecules, 12(12), 1904. https://doi.org/10.3390/biom12121904










