Mechanism and Role of Endoplasmic Reticulum Stress in Osteosarcoma
Abstract
1. Introduction
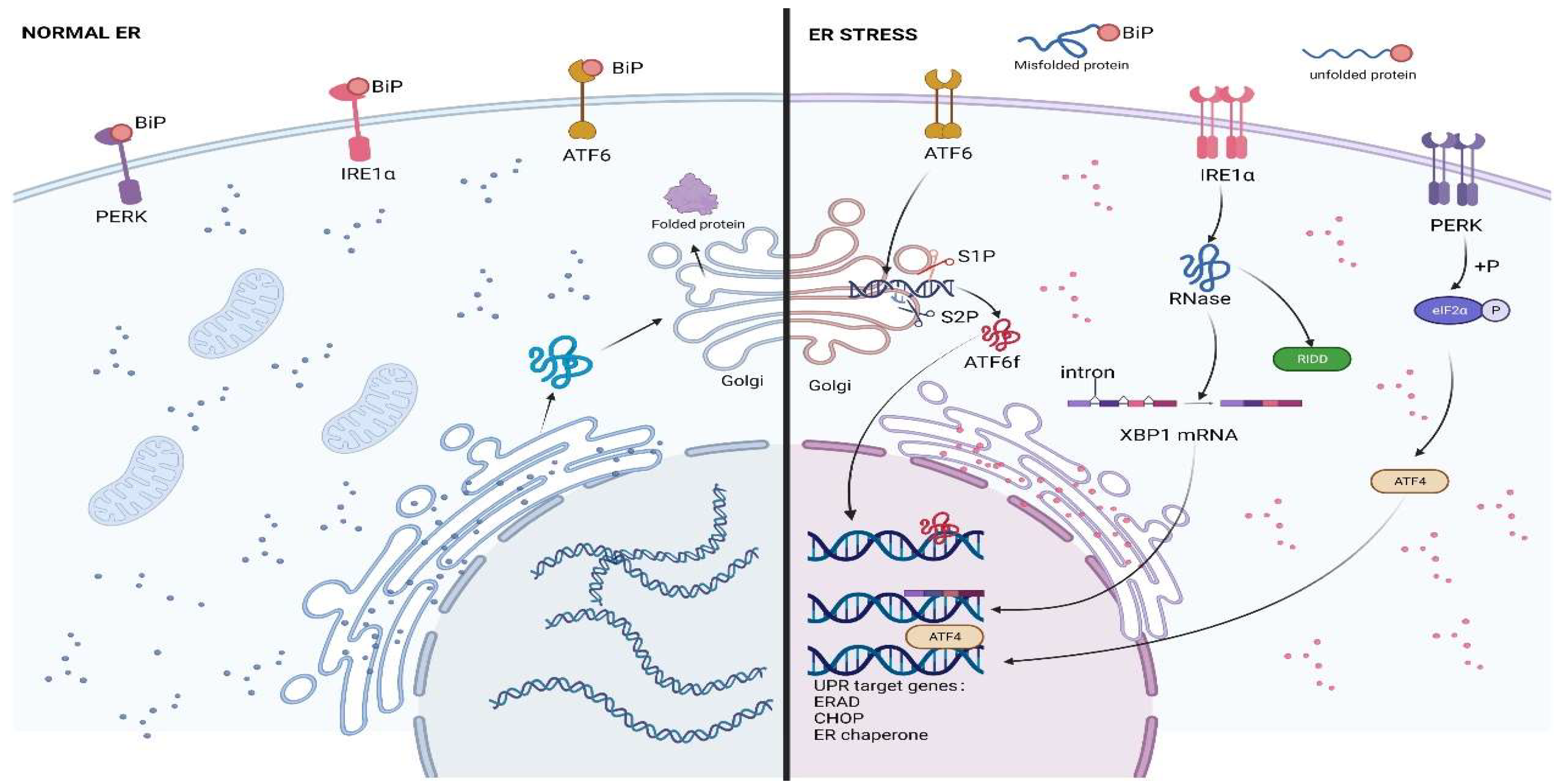
2. ERS and UPR
2.1. The Three Branches of UPR
2.1.1. IRE1
2.1.2. PERK
2.1.3. ATF6
3. ERS Signaling in Osteosarcoma
3.1. IRE1α-XBP1s Pathway
3.2. ATF6
3.3. GPR78 and GPR94
3.4. Crosstalk of ERS with Other Pathways
3.4.1. Integrated Stress Response (ISR)
3.4.2. Autophagy
3.4.3. Oxidative Stress
3.4.4. PI3K/Akt Pathway
3.4.5. Wnt/β-Catenin Pathway
3.4.6. MicroRNAs
4. Treatment of Osteosarcoma
4.1. Current Treatment Methods of Osteosarcoma and Their Limitations
4.2. Potential Osteosarcoma Treatments That Target ERS
4.2.1. IRE1
4.2.2. PERK
4.2.3. PI3K/Akt
4.2.4. Other Therapeutic Targets
5. Future Perspectives
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ritter, J.; Bielack, S.S. Osteosarcoma. Ann. Oncol. 2010, 21 (Suppl. 7), vii320–vii325. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Zhu, J.; Wang, Y.; Wang, J.; Fang, W.; Xia, K.; Shao, J.; Wu, M.; Liu, B.; Liang, C.; et al. A review and outlook in the treatment of osteosarcoma and other deep tumors with photodynamic therapy: From basic to deep. Oncotarget 2017, 8, 39833–39848. [Google Scholar] [CrossRef] [PubMed]
- Corre, I.; Verrecchia, F.; Crenn, V.; Redini, F.; Trichet, V. The Osteosarcoma Microenvironment: A Complex but Targetable Ecosystem. Cells 2020, 9, 976. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Yi, X.; Huang, D.; Liu, P.; Chen, L.; Du, Y.; Hao, L. ROCK2 mediates osteosarcoma progression and TRAIL resistance by modulating O-GlcNAc transferase degradation. Am. J. Cancer Res. 2020, 10, 781–798. [Google Scholar]
- Chen, C.; Xie, L.; Ren, T.; Huang, Y.; Xu, J.; Guo, W. Immunotherapy for osteosarcoma: Fundamental mechanism, rationale, and recent breakthroughs. Cancer Lett. 2021, 500, 1–10. [Google Scholar] [CrossRef]
- Deng, X.; Deng, J.; Yi, X.; Zou, Y.; Liu, H.; Li, C.; Deng, B.; Fan, H.; Hao, L. Ubiquitin-like protein FAT10 promotes osteosarcoma glycolysis and growth by upregulating PFKFB3 via stabilization of EGFR. Am. J. Cancer Res. 2020, 10, 2066–2082. [Google Scholar]
- Sergi, C.M. Targeting the ‘garbage-bin’ to fight cancer: HDAC6 inhibitor WT161 has an anti-tumor effect on osteosarcoma and synergistically interacts with 5-FU. Biosci. Rep. 2021, 41, BSR20210952. [Google Scholar] [CrossRef]
- Eaton, B.R.; Schwarz, R.; Vatner, R.; Yeh, B.; Claude, L.; Indelicato, D.J.; Laack, N. Osteosarcoma. Pediatr. Blood Cancer 2021, 68 (Suppl. 2), e28352. [Google Scholar] [CrossRef]
- Dejeans, N.; Manié, S.; Hetz, C.; Bard, F.; Hupp, T.; Agostinis, P.; Samali, A.; Chevet, E. Addicted to secrete—Novel concepts and targets in cancer therapy. Trends Mol. Med. 2014, 20, 242–250. [Google Scholar] [CrossRef]
- Urra, H.; Dufey, E.; Avril, T.; Chevet, E.; Hetz, C. Endoplasmic Reticulum Stress and the Hallmarks of Cancer. Trends Cancer 2016, 2, 252–262. [Google Scholar] [CrossRef]
- Sisinni, L.; Pietrafesa, M.; Lepore, S.; Maddalena, F.; Condelli, V.; Esposito, F.; Landriscina, M. Endoplasmic Reticulum Stress and Unfolded Protein Response in Breast Cancer: The Balance between Apoptosis and Autophagy and Its Role in Drug Resistance. Int. J. Mol. Sci. 2019, 20, 857. [Google Scholar] [CrossRef]
- Hong, S.Y.; Hagen, T. Multiple myeloma Leu167Ile (c.499C>A) mutation prevents XBP1 mRNA splicing. Br. J. Haematol. 2013, 161, 898–901. [Google Scholar] [CrossRef]
- Ajiro, M.; Zheng, Z.-M. E6^E7, a novel splice isoform protein of human papillomavirus 16, stabilizes viral E6 and E7 oncoproteins via HSP90 and GRP78. mBio 2015, 6, e02014–e02068. [Google Scholar] [CrossRef]
- Oakes, S.A. Endoplasmic Reticulum Stress Signaling in Cancer Cells. Am. J. Pathol. 2020, 190, 934–946. [Google Scholar] [CrossRef]
- Chen, X.; Cubillos-Ruiz, J.R. Endoplasmic reticulum stress signals in the tumour and its microenvironment. Nat. Rev. Cancer 2021, 21, 71–88. [Google Scholar] [CrossRef]
- Zhao, A.; Zhang, Z.; Zhou, Y.; Li, X.; Li, X.; Ma, B.; Zhang, Q. β-Elemonic acid inhibits the growth of human Osteosarcoma through endoplasmic reticulum (ER) stress-mediated PERK/eIF2α/ATF4/CHOP activation and Wnt/β-catenin signal suppression. Phytomedicine 2020, 69, 153183. [Google Scholar] [CrossRef]
- Cubillos-Ruiz, J.R.; Bettigole, S.E.; Glimcher, L.H. Tumorigenic and Immunosuppressive Effects of Endoplasmic Reticulum Stress in Cancer. Cell 2017, 168, 692–706. [Google Scholar] [CrossRef]
- Oakes, S.A.; Papa, F.R. The role of endoplasmic reticulum stress in human pathology. Annu. Rev. Pathol. 2015, 10, 173–194. [Google Scholar] [CrossRef]
- Braakman, I.; Hebert, D.N. Protein folding in the endoplasmic reticulum. Cold Spring Harb. Perspect. Biol. 2013, 5, a013201. [Google Scholar] [CrossRef]
- So, J.-S. Roles of Endoplasmic Reticulum Stress in Immune Responses. Mol. Cells 2018, 41, 705–716. [Google Scholar] [CrossRef]
- Rahmati, M.; Moosavi, M.A.; McDermott, M.F. ER Stress: A Therapeutic Target in Rheumatoid Arthritis? Trends Pharmacol. Sci. 2018, 39, 610–623. [Google Scholar] [CrossRef]
- Hetz, C. The unfolded protein response: Controlling cell fate decisions under ER stress and beyond. Nat. Rev. Mol. Cell. Biol. 2012, 13, 89–102. [Google Scholar] [CrossRef]
- Qi, L.; Tsai, B.; Arvan, P. New Insights into the Physiological Role of Endoplasmic Reticulum-Associated Degradation. Trends Cell. Biol. 2017, 27, 430–440. [Google Scholar] [CrossRef]
- Zhang, T.; Xu, Y.; Liu, Y.; Ye, Y. gp78 functions downstream of Hrd1 to promote degradation of misfolded proteins of the endoplasmic reticulum. Mol. Biol. Cell 2015, 26, 4438–4450. [Google Scholar] [CrossRef]
- Baldridge, R.D.; Rapoport, T.A. Autoubiquitination of the Hrd1 Ligase Triggers Protein Retrotranslocation in ERAD. Cell 2016, 166, 394–407. [Google Scholar] [CrossRef]
- Almanza, A.; Carlesso, A.; Chintha, C.; Creedican, S.; Doultsinos, D.; Leuzzi, B.; Luís, A.; McCarthy, N.; Montibeller, L.; More, S.; et al. Endoplasmic reticulum stress signalling—From basic mechanisms to clinical applications. FEBS J. 2019, 286, 241–278. [Google Scholar] [CrossRef]
- Walter, P.; Ron, D. The unfolded protein response: From stress pathway to homeostatic regulation. Science 2011, 334, 1081–1086. [Google Scholar] [CrossRef]
- McGrath, E.P.; Logue, S.E.; Mnich, K.; Deegan, S.; Jäger, R.; Gorman, A.M.; Samali, A. The Unfolded Protein Response in Breast Cancer. Cancers 2018, 10, 344. [Google Scholar] [CrossRef]
- Acosta-Alvear, D.; Karagöz, G.E.; Fröhlich, F.; Li, H.; Walther, T.C.; Walter, P. The unfolded protein response and endoplasmic reticulum protein targeting machineries converge on the stress sensor IRE1. eLife 2018, 7, e43036. [Google Scholar] [CrossRef]
- Adams, C.J.; Kopp, M.C.; Larburu, N.; Nowak, P.R.; Ali, M.M.U. Structure and Molecular Mechanism of ER Stress Signaling by the Unfolded Protein Response Signal Activator IRE1. Front. Mol. Biosci. 2019, 6, 11. [Google Scholar] [CrossRef]
- Siwecka, N.; Rozpędek-Kamińska, W.; Wawrzynkiewicz, A.; Pytel, D.; Diehl, J.A.; Majsterek, I. The Structure, Activation and Signaling of IRE1 and Its Role in Determining Cell Fate. Biomedicines 2021, 9, 156. [Google Scholar] [CrossRef] [PubMed]
- Hetz, C.; Papa, F.R. The Unfolded Protein Response and Cell Fate Control. Mol. Cell 2018, 69, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Korennykh, A.; Walter, P. Structural basis of the unfolded protein response. Annu. Rev. Cell Dev. Biol. 2012, 28, 251–277. [Google Scholar] [CrossRef] [PubMed]
- Tabas, I.; Ron, D. Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat. Cell Biol. 2011, 13, 184–190. [Google Scholar] [CrossRef]
- Imagawa, Y.; Hosoda, A.; Sasaka, S.-I.; Tsuru, A.; Kohno, K. RNase domains determine the functional difference between IRE1alpha and IRE1beta. FEBS Lett. 2008, 582, 656–660. [Google Scholar] [CrossRef]
- Grandjean, J.M.D.; Wiseman, R.L. Small molecule strategies to harness the unfolded protein response: Where do we go from here? J. Biol. Chem. 2020, 295, 15692–15711. [Google Scholar] [CrossRef]
- Yoshida, H.; Matsui, T.; Yamamoto, A.; Okada, T.; Mori, K. XBP1 mRNA is induced by ATF6 and spliced by IRE1 in response to ER stress to produce a highly active transcription factor. Cell 2001, 107, 881–891. [Google Scholar] [CrossRef]
- Travers, K.J.; Patil, C.K.; Wodicka, L.; Lockhart, D.J.; Weissman, J.S.; Walter, P. Functional and genomic analyses reveal an essential coordination between the unfolded protein response and ER-associated degradation. Cell 2000, 101, 249–258. [Google Scholar] [CrossRef]
- Hollien, J.; Lin, J.H.; Li, H.; Stevens, N.; Walter, P.; Weissman, J.S. Regulated Ire1-dependent decay of messenger RNAs in mammalian cells. J. Cell Biol. 2009, 186, 323–331. [Google Scholar] [CrossRef]
- Hollien, J.; Weissman, J.S. Decay of endoplasmic reticulum-localized mRNAs during the unfolded protein response. Science 2006, 313, 104–107. [Google Scholar] [CrossRef]
- Huang, J.; Pan, H.; Wang, J.; Wang, T.; Huo, X.; Ma, Y.; Lu, Z.; Sun, B.; Jiang, H. Unfolded protein response in colorectal cancer. Cell Biosci. 2021, 11, 26. [Google Scholar] [CrossRef] [PubMed]
- Moncan, M.; Mnich, K.; Blomme, A.; Almanza, A.; Samali, A.; Gorman, A.M. Regulation of lipid metabolism by the unfolded protein response. J. Cell. Mol. Med. 2021, 25, 1359–1370. [Google Scholar] [CrossRef] [PubMed]
- Fusakio, M.E.; Willy, J.A.; Wang, Y.; Mirek, E.T.; Al Baghdadi, R.J.T.; Adams, C.M.; Anthony, T.G.; Wek, R.C. Transcription factor ATF4 directs basal and stress-induced gene expression in the unfolded protein response and cholesterol metabolism in the liver. Mol. Biol. Cell 2016, 27, 1536–1551. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Chen, X.; Hendershot, L.; Prywes, R. ER stress regulation of ATF6 localization by dissociation of BiP/GRP78 binding and unmasking of Golgi localization signals. Dev. Cell 2002, 3, 99–111. [Google Scholar] [CrossRef]
- Yamamoto, K.; Sato, T.; Matsui, T.; Sato, M.; Okada, T.; Yoshida, H.; Harada, A.; Mori, K. Transcriptional induction of mammalian ER quality control proteins is mediated by single or combined action of ATF6alpha and XBP1. Dev. Cell 2007, 13, 365–376. [Google Scholar] [CrossRef]
- Jin, J.-K.; Blackwood, E.A.; Azizi, K.; Thuerauf, D.J.; Fahem, A.G.; Hofmann, C.; Kaufman, R.J.; Doroudgar, S.; Glembotski, C.C. ATF6 Decreases Myocardial Ischemia/Reperfusion Damage and Links ER Stress and Oxidative Stress Signaling Pathways in the Heart. Circ. Res. 2017, 120, 862–875. [Google Scholar] [CrossRef]
- Chen, S.; Chen, J.; Hua, X.; Sun, Y.; Cui, R.; Sha, J.; Zhu, X. The emerging role of XBP1 in cancer. Biomed. Pharmacother. 2020, 127, 110069. [Google Scholar] [CrossRef]
- Shajahan, A.N.; Riggins, R.B.; Clarke, R. The role of X-box binding protein-1 in tumorigenicity. Drug. News Perspect. 2009, 22, 241–246. [Google Scholar] [CrossRef]
- Yang, J.; Cheng, D.; Zhou, S.; Zhu, B.; Hu, T.; Yang, Q. Overexpression of X-Box Binding Protein 1 (XBP1) Correlates to Poor Prognosis and Up-Regulation of PI3K/mTOR in Human Osteosarcoma. Int. J. Mol. Sci. 2015, 16, 28635–28646. [Google Scholar] [CrossRef]
- Erzberger, J.P.; Berger, J.M. Evolutionary relationships and structural mechanisms of AAA+ proteins. Annu. Rev. Biophys. Biomol. Struct. 2006, 35, 93–114. [Google Scholar] [CrossRef]
- Meyer, H.; Bug, M.; Bremer, S. Emerging functions of the VCP/p97 AAA-ATPase in the ubiquitin system. Nat. Cell Biol. 2012, 14, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Vembar, S.S.; Brodsky, J.L. One step at a time: Endoplasmic reticulum-associated degradation. Nat. Rev. Mol. Cell Biol. 2008, 9, 944–957. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Wu, M.; Zhang, X.; Jin, Q.; Wang, Y.; Zou, C.; Huang, G.; Yin, J.; Xie, X.; Shen, J. CB-5083, an inhibitor of P97, suppresses osteosarcoma growth and stem cell properties by altering protein homeostasis. Am. J. Transl. Res. 2020, 12, 2956–2967. [Google Scholar] [PubMed]
- Yarapureddy, S.; Abril, J.; Foote, J.; Kumar, S.; Asad, O.; Sharath, V.; Faraj, J.; Daniel, D.; Dickman, P.; White-Collins, A.; et al. ATF6α Activation Enhances Survival against Chemotherapy and Serves as a Prognostic Indicator in Osteosarcoma. Neoplasia 2019, 21, 516–532. [Google Scholar] [CrossRef]
- Ledoux, A.C.; Sellier, H.; Gillies, K.; Iannetti, A.; James, J.; Perkins, N.D. NFκB regulates expression of Polo-like kinase 4. Cell Cycle 2013, 12, 3052–3062. [Google Scholar] [CrossRef]
- Shen, T.; Li, Y.; Chen, Z.; Liang, S.; Qiu, Y.; Zhu, L.; Ba, G.; Lu, G.; Qiu, L. Activating transcription factor 6 (ATF6) negatively regulates Polo-like kinase 4 expression via recruiting C/EBPβ to the upstream-promoter during ER stress. Biochim. Biophys. Acta Gene Regul. Mech. 2020, 1863, 194488. [Google Scholar] [CrossRef]
- Pagliarini, V.; Giglio, P.; Bernardoni, P.; De Zio, D.; Fimia, G.M.; Piacentini, M.; Corazzari, M. Downregulation of E2F1 during ER stress is required to induce apoptosis. J. Cell Sci. 2015, 128, 1166–1179. [Google Scholar] [CrossRef]
- Du, L.; He, F.; Kuang, L.; Tang, W.; Li, Y.; Chen, D. eNOS/iNOS and endoplasmic reticulum stress-induced apoptosis in the placentas of patients with preeclampsia. J. Hum. Hypertens. 2017, 31, 49–55. [Google Scholar] [CrossRef]
- Huang, Q.; Lan, T.; Lu, J.; Zhang, H.; Zhang, D.; Lou, T.; Xu, P.; Ren, J.; Zhao, D.; Sun, L.; et al. DiDang Tang Inhibits Endoplasmic Reticulum Stress-Mediated Apoptosis Induced by Oxygen Glucose Deprivation and Intracerebral Hemorrhage Through Blockade of the GRP78-IRE1/PERK Pathways. Front. Pharmacol. 2018, 9, 1423. [Google Scholar] [CrossRef]
- Chaiyawat, P.; Sungngam, P.; Teeyakasem, P.; Sirikaew, N.; Klangjorhor, J.; Settakorn, J.; Diskul-Na-Ayudthaya, P.; Chokchaichamnankit, D.; Srisomsap, C.; Svasti, J.; et al. Protein profiling of osteosarcoma tissue and soft callus unveils activation of the unfolded protein response pathway. Int. J. Oncol. 2019, 54, 1704–1718. [Google Scholar] [CrossRef]
- Luo, J.; Xia, Y.; Yin, Y.; Luo, J.; Liu, M.; Zhang, H.; Zhang, C.; Zhao, Y.; Yang, L.; Kong, L. ATF4 destabilizes RET through nonclassical GRP78 inhibition to enhance chemosensitivity to bortezomib in human osteosarcoma. Theranostics 2019, 9, 6334–6353. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-Y.; Wei, P.-L.; Wang, J.-W.; Makondi, P.T.; Huang, M.-T.; Chen, H.-A.; Chang, Y.-J. Glucose-Regulated Protein 94 Modulates the Response of Osteosarcoma to Chemotherapy. Dis. Mark. 2019, 2019, 4569718. [Google Scholar] [CrossRef] [PubMed]
- Ron, D. Translational control in the endoplasmic reticulum stress response. J. Clin. Investig. 2002, 110, 1383–1388. [Google Scholar] [CrossRef] [PubMed]
- Harding, H.P.; Zhang, Y.; Zeng, H.; Novoa, I.; Lu, P.D.; Calfon, M.; Sadri, N.; Yun, C.; Popko, B.; Paules, R.; et al. An integrated stress response regulates amino acid metabolism and resistance to oxidative stress. Mol. Cell 2003, 11, 619–633. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, Z.; Li, Y.; Li, Y. The Regulation of Integrated Stress Response Signaling Pathway on Viral Infection and Viral Antagonism. Front. Microbiol. 2021, 12, 814635. [Google Scholar] [CrossRef]
- Harding, H.P.; Zhang, Y.; Ron, D. Protein translation and folding are coupled by an endoplasmic-reticulum-resident kinase. Nature 1999, 397, 271–274. [Google Scholar] [CrossRef]
- Costa-Mattioli, M.; Walter, P. The integrated stress response: From mechanism to disease. Science 2020, 368, eaat5314. [Google Scholar] [CrossRef]
- Pakos-Zebrucka, K.; Koryga, I.; Mnich, K.; Ljujic, M.; Samali, A.; Gorman, A.M. The integrated stress response. EMBO Rep. 2016, 17, 1374–1395. [Google Scholar] [CrossRef]
- White, E. Deconvoluting the context-dependent role for autophagy in cancer. Nat. Rev. Cancer 2012, 12, 401–410. [Google Scholar] [CrossRef]
- Niu, J.; Yan, T.; Guo, W.; Wang, W.; Zhao, Z. Insight into the Role of Autophagy in Osteosarcoma and Its Therapeutic Implication. Front. Oncol. 2019, 9, 1232. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Yan, T.; Guo, W.; Sun, K.; Wang, S.; Bao, X.; Liu, K.; Zheng, B.; Zhang, H.; Ren, T. Novel oncogene COPS3 interacts with Beclin1 and Raf-1 to regulate metastasis of osteosarcoma through autophagy. J. Exp. Clin. Cancer Res. 2018, 37, 135. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yin, F.; Xu, J.; Zhang, T.; Wang, G.; Mao, M.; Wang, Z.; Sun, W.; Han, J.; Yang, M.; et al. CYT997(Lexibulin) induces apoptosis and autophagy through the activation of mutually reinforced ER stress and ROS in osteosarcoma. J. Exp. Clin. Cancer Res. 2019, 38, 44. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Wei, X.; Li, T.; Wang, W.; Wu, H.; Dong, H.; Liu, Y.; Wei, F.; Shi, L.; Li, X.; et al. Sestrin2-Mediated Autophagy Contributes to Drug Resistance Endoplasmic Reticulum Stress in Human Osteosarcoma. Front. Cell Dev. Biol. 2021, 9, 722960. [Google Scholar] [CrossRef]
- Ji, G.-R.; Yu, N.-C.; Xue, X.; Li, Z.-G. PERK-mediated Autophagy in Osteosarcoma Cells Resists ER Stress-induced Cell Apoptosis. Int. J. Biol. Sci. 2015, 11, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.H.; Ro, S.-H.; Cao, J.; Otto, N.M.; Kim, D.-H. mTOR regulation of autophagy. FEBS Lett. 2010, 584, 1287–1295. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Huang, Q.; Niu, K.; Wang, B.; Li, Y.; Dai, C.; Chen, Z.; Tao, K.; Dai, J. Sestrin 2 confers primary resistance to sorafenib by simultaneously activating AKT and AMPK in hepatocellular carcinoma. Cancer Med. 2018, 7, 5691–5703. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.Y.; Lee, Y.-J.; Sai, S.; Ohno, T.; Kong, C.-B.; Lim, S.H.; Kim, E.H. The Unfolded Protein Response: Neutron-Induced Therapy Autophagy as a Promising Treatment Option for Osteosarcoma. Int. J. Mol. Sci. 2020, 21, 3766. [Google Scholar] [CrossRef] [PubMed]
- Matés, J.M.; Segura, J.A.; Alonso, F.J.; Márquez, J. Oxidative stress in apoptosis and cancer: An update. Arch. Toxicol. 2012, 86, 1649–1665. [Google Scholar] [CrossRef]
- Malhotra, J.D.; Kaufman, R.J. Endoplasmic reticulum stress and oxidative stress: A vicious cycle or a double-edged sword? Antioxid. Redox Signal. 2007, 9, 2277–2293. [Google Scholar] [CrossRef]
- Verfaillie, T.; Rubio, N.; Garg, A.D.; Bultynck, G.; Rizzuto, R.; Decuypere, J.P.; Piette, J.; Linehan, C.; Gupta, S.; Samali, A.; et al. PERK is required at the ER-mitochondrial contact sites to convey apoptosis after ROS-based ER stress. Cell Death Differ. 2012, 19, 1880–1891. [Google Scholar] [CrossRef]
- Xian, M.; Cao, H.; Cao, J.; Shao, X.; Zhu, D.; Zhang, N.; Huang, P.; Li, W.; Yang, B.; Ying, M.; et al. Bortezomib sensitizes human osteosarcoma cells to adriamycin-induced apoptosis through ROS-dependent activation of p-eIF2α/ATF4/CHOP axis. Int. J. Cancer 2017, 141, 1029–1041. [Google Scholar] [CrossRef]
- Huang, X.; Chen, J.; Wu, W.; Yang, W.; Zhong, B.; Qing, X.; Shao, Z. Delivery of MutT homolog 1 inhibitor by functionalized graphene oxide nanoparticles for enhanced chemo-photodynamic therapy triggers cell death in osteosarcoma. Acta Biomater. 2020, 109, 229–243. [Google Scholar] [CrossRef]
- Tu, B.P.; Weissman, J.S. Oxidative protein folding in eukaryotes: Mechanisms and consequences. J. Cell Biol. 2004, 164, 341–346. [Google Scholar] [CrossRef]
- Zito, E. ERO1: A protein disulfide oxidase and H2O2 producer. Free Radic. Biol. Med. 2015, 83, 299–304. [Google Scholar] [CrossRef]
- Rao, J.; Zhang, C.; Wang, P.; Lu, L.; Qian, X.; Qin, J.; Pan, X.; Li, G.; Wang, X.; Zhang, F. C/EBP homologous protein (CHOP) contributes to hepatocyte death via the promotion of ERO1α signalling in acute liver failure. Biochem. J. 2015, 466, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Hemmings, B.A.; Restuccia, D.F. PI3K-PKB/Akt pathway. Cold Spring Harb. Perspect. Biol. 2012, 4, a011189. [Google Scholar] [CrossRef]
- Aoki, M.; Fujishita, T. Oncogenic Roles of the PI3K/AKT/mTOR Axis. Curr. Top. Microbiol. Immunol. 2017, 407, 153–189. [Google Scholar] [CrossRef] [PubMed]
- Murugan, A.K. mTOR: Role in cancer, metastasis and drug resistance. Semin. Cancer Biol. 2019, 59, 92–111. [Google Scholar] [CrossRef] [PubMed]
- Porta, C.; Paglino, C.; Mosca, A. Targeting PI3K/Akt/mTOR Signaling in Cancer. Front. Oncol. 2014, 4, 64. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Han, X.; Ou, D.; Liu, T.; Li, Z.; Jiang, G.; Liu, J.; Zhang, J. Targeting PI3K/AKT/mTOR-mediated autophagy for tumor therapy. Appl. Microbiol. Biotechnol. 2020, 104, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Wang, S.; Wang, Z.; Wang, Z.; Sun, C.; Zhang, Y. Inhibition of PTEN Attenuates Endoplasmic Reticulum Stress and Apoptosis via Activation of PI3K/AKT Pathway in Alzheimer’s Disease. Neurochem. Res. 2017, 42, 3052–3060. [Google Scholar] [CrossRef]
- Koga, T.; Suico, M.A.; Shimasaki, S.; Watanabe, E.; Kai, Y.; Koyama, K.; Omachi, K.; Morino-Koga, S.; Sato, T.; Shuto, T.; et al. Endoplasmic Reticulum (ER) Stress Induces Sirtuin 1 (SIRT1) Expression via the PI3K-Akt-GSK3β Signaling Pathway and Promotes Hepatocellular Injury. J. Biol. Chem. 2015, 290, 30366–30374. [Google Scholar] [CrossRef] [PubMed]
- Oeckinghaus, A.; Hayden, M.S.; Ghosh, S. Crosstalk in NF-κB signaling pathways. Nat. Immunol. 2011, 12, 695–708. [Google Scholar] [CrossRef] [PubMed]
- Castro-Gamero, A.M.; Borges, K.S.; da Silva Silveira, V.; Lira, R.C.P.; de Paula Gomes Queiroz, R.; Valera, F.C.P.; Scrideli, C.A.; Umezawa, K.; Tone, L.G. Inhibition of nuclear factor-κB by dehydroxymethylepoxyquinomicin induces schedule-dependent chemosensitivity to anticancer drugs and enhances chemoinduced apoptosis in osteosarcoma cells. Anticancer Drugs 2012, 23, 638–650. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Wang, Q.; Zhou, X.; Fu, C.; Cheng, M.; Guo, R.; Liu, H.; Zhang, B.; Dai, M. Celastrol negatively regulates cell invasion and migration ability of human osteosarcoma via downregulation of the PI3K/Akt/NF-κB signaling pathway. Oncol. Lett. 2016, 12, 3423–3428. [Google Scholar] [CrossRef]
- Yan, M.; Ni, J.; Song, D.; Ding, M.; Huang, J. Activation of unfolded protein response protects osteosarcoma cells from cisplatin-induced apoptosis through NF-κB pathway. Int. J. Clin. Exp. Pathol. 2015, 8, 10204–10215. [Google Scholar] [PubMed]
- Jiang, C.; Ma, S.; Hu, R.; Wang, X.; Li, M.; Tian, F.; Jiang, W.; Zhu, L.; Bian, Z. Effect of CXCR4 on Apoptosis in Osteosarcoma Cells via the PI3K/Akt/NF-κβ Signaling Pathway. Cell. Physiol. Biochem. 2018, 46, 2250–2260. [Google Scholar] [CrossRef]
- Jiang, J.; Wang, W.; Xiang, W.; Jiang, L.; Zhou, Q. The phosphoinositide 3-kinase inhibitor ZSTK474 increases the susceptibility of osteosarcoma cells to oncolytic vesicular stomatitis virus VSVΔ51 via aggravating endoplasmic reticulum stress. Bioengineered 2021, 12, 11847–11857. [Google Scholar] [CrossRef]
- Xu, X.; Rajamanicham, V.; Xu, S.; Liu, Z.; Yan, T.; Liang, G.; Guo, G.; Zhou, H.; Wang, Y. Schisandrin A inhibits triple negative breast cancer cells by regulating Wnt/ER stress signaling pathway. Biomed. Pharmacother. 2019, 115, 108922. [Google Scholar] [CrossRef]
- Grigoryan, T.; Wend, P.; Klaus, A.; Birchmeier, W. Deciphering the function of canonical Wnt signals in development and disease: Conditional loss- and gain-of-function mutations of beta-catenin in mice. Genes Dev. 2008, 22, 2308–2341. [Google Scholar] [CrossRef]
- Malanchi, I.; Peinado, H.; Kassen, D.; Hussenet, T.; Metzger, D.; Chambon, P.; Huber, M.; Hohl, D.; Cano, A.; Birchmeier, W.; et al. Cutaneous cancer stem cell maintenance is dependent on beta-catenin signalling. Nature 2008, 452, 650–653. [Google Scholar] [CrossRef]
- Horndasch, M.; Lienkamp, S.; Springer, E.; Schmitt, A.; Pavenstädt, H.; Walz, G.; Gloy, J. The C/EBP homologous protein CHOP (GADD153) is an inhibitor of Wnt/TCF signals. Oncogene 2006, 25, 3397–3407. [Google Scholar] [CrossRef]
- Song, L.; De Sarno, P.; Jope, R.S. Central role of glycogen synthase kinase-3beta in endoplasmic reticulum stress-induced caspase-3 activation. J. Biol. Chem. 2002, 277, 44701–44708. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Zhang, Y.; Niu, J.; Nie, Z.; Liu, Q.; Lv, C. Zinc promotes cell apoptosis via activating the Wnt-3a/β-catenin signaling pathway in osteosarcoma. J. Orthop. Surg. Res. 2020, 15, 57. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Sun, S.; Xu, J.; Luo, Y.; Xin, Y.; Wang, Y. MicroRNA-152 inhibits cell proliferation of osteosarcoma by directly targeting Wnt/β-catenin signaling pathway in a DKK1-dependent manner. Oncol. Rep. 2018, 40, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhou, F.; Dong, Y.; Ren, F. α-Mangostin Induces Apoptosis in Human Osteosarcoma Cells Through ROS-Mediated Endoplasmic Reticulum Stress via the WNT Pathway. Cell Transplant. 2021, 30, 9636897211035080. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; He, C.; Wu, Z.; Li, F.; Xiao, J. Histone methyltransferase SETD2 regulates osteosarcoma cell growth and chemosensitivity by suppressing Wnt/β-catenin signaling. Biochem. Biophys. Res. Commun. 2018, 502, 382–388. [Google Scholar] [CrossRef]
- Guo, J.; Yang, Z.; Yang, X.; Li, T.; Liu, M.; Tang, H. miR-346 functions as a pro-survival factor under ER stress by activating mitophagy. Cancer Lett. 2018, 413, 69–81. [Google Scholar] [CrossRef]
- Jin, L.; Hu, W.L.; Jiang, C.C.; Wang, J.X.; Han, C.C.; Chu, P.; Zhang, L.J.; Thorne, R.F.; Wilmott, J.; Scolyer, R.A.; et al. MicroRNA-149*, a p53-responsive microRNA, functions as an oncogenic regulator in human melanoma. Proc. Natl. Acad. Sci. USA 2011, 108, 15840–15845. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Zhang, D.; Fan, Y.; Chao, Y.; Chang, J.; Li, N.; Han, L.; Han, C. Regulation of Cancer Stem Cell Self-Renewal by HOXB9 Antagonizes Endoplasmic Reticulum Stress-Induced Melanoma Cell Apoptosis via the miR-765-FOXA2 Axis. J. Investig. Dermatol. 2018, 138, 1609–1619. [Google Scholar] [CrossRef]
- Jiang, J.; Ma, B.; Li, X.; Jin, W.; Han, C.; Wang, L.; Wang, H. MiR-1281, a p53-responsive microRNA, impairs the survival of human osteosarcoma cells upon ER stress via targeting USP39. Am. J. Cancer Res. 2018, 8, 1764–1774. [Google Scholar] [PubMed]
- Zhang, L.; Wang, Y.; Zhang, L.; Xia, X.; Chao, Y.; He, R.; Han, C.; Zhao, W. ZBTB7A, a miR-663a target gene, protects osteosarcoma from endoplasmic reticulum stress-induced apoptosis by suppressing LncRNA GAS5 expression. Cancer Lett. 2019, 448, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Maeda, T.; Hobbs, R.M.; Merghoub, T.; Guernah, I.; Zelent, A.; Cordon-Cardo, C.; Teruya-Feldstein, J.; Pandolfi, P.P. Role of the proto-oncogene Pokemon in cellular transformation and ARF repression. Nature 2005, 433, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.-B.; Wang, Y.-W.; Xing, A.-Y.; Gao, J.-W.; Zhang, H.; Guo, X.-Y.; Gao, P. C/EBPα-induced miR-100 expression suppresses tumor metastasis and growth by targeting ZBTB7A in gastric cancer. Cancer Lett. 2015, 369, 376–385. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Y.; Li, X.; Xia, X.; Li, N.; He, R.; He, H.; Han, C.; Zhao, W. ZBTB7A Enhances Osteosarcoma Chemoresistance by Transcriptionally Repressing lncRNALINC00473-IL24 Activity. Neoplasia 2017, 19, 908–918. [Google Scholar] [CrossRef]
- Harrison, D.J.; Geller, D.S.; Gill, J.D.; Lewis, V.O.; Gorlick, R. Current and future therapeutic approaches for osteosarcoma. Expert Rev. Anticancer Ther. 2018, 18, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Belayneh, R.; Fourman, M.S.; Bhogal, S.; Weiss, K.R. Update on Osteosarcoma. Curr. Oncol. Rep. 2021, 23, 71. [Google Scholar] [CrossRef] [PubMed]
- Grimer, R.J. Surgical options for children with osteosarcoma. Lancet Oncol. 2005, 6, 85–92. [Google Scholar] [CrossRef]
- Fan, X.-L.; Cai, G.-P.; Zhu, L.-L.; Ding, G.-M. Efficacy and safety of ifosfamide-based chemotherapy for osteosarcoma: A meta-analysis. Drug Des. Dev. Ther. 2015, 9, 5925–5932. [Google Scholar] [CrossRef] [PubMed]
- Smrke, A.; Anderson, P.M.; Gulia, A.; Gennatas, S.; Huang, P.H.; Jones, R.L. Future Directions in the Treatment of Osteosarcoma. Cells 2021, 10, 172. [Google Scholar] [CrossRef]
- Yamamoto, N.; Tsuchiya, H. Chemotherapy for osteosarcoma—Where does it come from? What is it? Where is it going? Expert Opin. Pharmacother. 2013, 14, 2183–2193. [Google Scholar] [CrossRef]
- Kopp, L.M.; Malempati, S.; Krailo, M.; Gao, Y.; Buxton, A.; Weigel, B.J.; Hawthorne, T.; Crowley, E.; Moscow, J.A.; Reid, J.M.; et al. Phase II trial of the glycoprotein non-metastatic B-targeted antibody-drug conjugate, glembatumumab vedotin (CDX-011), in recurrent osteosarcoma AOST1521: A report from the Children’s Oncology Group. Eur. J. Cancer 2019, 121, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Goorin, A.M.; Schwartzentruber, D.J.; Devidas, M.; Gebhardt, M.C.; Ayala, A.G.; Harris, M.B.; Helman, L.J.; Grier, H.E.; Link, M.P. Presurgical chemotherapy compared with immediate surgery and adjuvant chemotherapy for nonmetastatic osteosarcoma: Pediatric Oncology Group Study POG-8651. J. Clin. Oncol. 2003, 21, 1574–1580. [Google Scholar] [CrossRef]
- Boyer, K.M.; Gotoff, S.P. Prevention of early-onset neonatal group B streptococcal disease with selective intrapartum chemoprophylaxis. N. Engl. J. Med. 1986, 314, 1665–1669. [Google Scholar] [CrossRef] [PubMed]
- Livshits, Z.; Rao, R.B.; Smith, S.W. An approach to chemotherapy-associated toxicity. Emerg. Med. Clin. N. Am. 2014, 32, 167–203. [Google Scholar] [CrossRef] [PubMed]
- Gordon, N.; Felix, K.; Daw, N.C. Aerosolized Chemotherapy for Osteosarcoma. Adv. Exp. Med. Biol. 2020, 1257, 67–73. [Google Scholar] [CrossRef]
- Lilienthal, I.; Herold, N. Targeting Molecular Mechanisms Underlying Treatment Efficacy and Resistance in Osteosarcoma: A Review of Current and Future Strategies. Int. J. Mol. Sci. 2020, 21, 6885. [Google Scholar] [CrossRef]
- Anderson, P.M. Radiopharmaceuticals for Treatment of Osteosarcoma. Adv. Exp. Med. Biol. 2020, 1257, 45–53. [Google Scholar] [CrossRef]
- Chen, Y.; Ou, Y.; Tao, Y.; Liu, H.; Yin, H.; Zhong, S.; Yu, H.; Zhao, Z.; He, B. Effect and mechanisms of celastrol on the apoptosis of HOS osteosarcoma cells. Oncol. Rep. 2018, 40, 2260–2268. [Google Scholar] [CrossRef]
- Wang, G.-S.; Chen, J.-Y.; Chen, W.-C.; Wei, I.C.; Lin, S.-W.; Liao, K.-W.; Yang, T.-S.; Liu, J.-F. Surfactin induces ER stress-mediated apoptosis via IRE1-ASK1-JNK signaling in human osteosarcoma. Environ. Toxicol. 2022, 37, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.; Hu, Y.; Pang, H.; Sun, J.; Wang, Z.; Li, J. Melittin protein inhibits the proliferation of MG63 cells by activating inositol-requiring protein-1α and X-box binding protein 1-mediated apoptosis. Mol. Med. Rep. 2014, 9, 1365–1370. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, C.-W.; Chi, M.-C.; Chang, T.-M.; Liu, J.-F. Artocarpin induces cell apoptosis in human osteosarcoma cells through endoplasmic reticulum stress and reactive oxygen species. J. Cell. Physiol. 2019, 234, 13157–13168. [Google Scholar] [CrossRef]
- Machihara, K.; Namba, T. Kuanoniamine C stimulates bortezomib-induced cell death via suppression of glucose-regulated protein 78 in osteosarcoma. Biochem. Biophys. Res. Commun. 2020, 527, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Buondonno, I.; Gazzano, E.; Tavanti, E.; Chegaev, K.; Kopecka, J.; Fanelli, M.; Rolando, B.; Fruttero, R.; Gasco, A.; Hattinger, C.; et al. Endoplasmic reticulum-targeting doxorubicin: A new tool effective against doxorubicin-resistant osteosarcoma. Cell. Mol. Life Sci. 2019, 76, 609–625. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Kamel, W.A.; Yamaguchi-Iwai, S.; Fukuchi, Y.; Muto, A.; Saya, H. Calcitriol exerts an anti-tumor effect in osteosarcoma by inducing the endoplasmic reticulum stress response. Cancer Sci. 2017, 108, 1793–1802. [Google Scholar] [CrossRef]
- Chao, C.-C.; Hou, S.-M.; Huang, C.C.; Hou, C.-H.; Chen, P.-C.; Liu, J.-F. Plumbagin induces apoptosis in human osteosarcoma through ROS generation, endoplasmic reticulum stress and mitochondrial apoptosis pathway. Mol. Med. Rep. 2017, 16, 5480–5488. [Google Scholar] [CrossRef]
- Sun, X.; Wei, Q.; Cheng, J.; Bian, Y.; Tian, C.; Hu, Y.; Li, H. Enhanced Stim1 expression is associated with acquired chemo-resistance of cisplatin in osteosarcoma cells. Hum. Cell 2017, 30, 216–225. [Google Scholar] [CrossRef]
- Gazzano, E.; Buondonno, I.; Marengo, A.; Rolando, B.; Chegaev, K.; Kopecka, J.; Saponara, S.; Sorge, M.; Hattinger, C.M.; Gasco, A.; et al. Hyaluronated liposomes containing H2S-releasing doxorubicin are effective against P-glycoprotein-positive/doxorubicin-resistant osteosarcoma cells and xenografts. Cancer Lett. 2019, 456, 29–39. [Google Scholar] [CrossRef]
- Wirries, A.; Jabari, S.; Jansen, E.P.; Roth, S.; Figueroa-Juárez, E.; Wissniowski, T.T.; Neureiter, D.; Klieser, E.; Lechler, P.; Ruchholtz, S.; et al. Panobinostat mediated cell death: A novel therapeutic approach for osteosarcoma. Oncotarget 2018, 9, 32997–33010. [Google Scholar] [CrossRef]
- Song, M.-Z.; Zhang, F.-L.; Lin, L.-J. (3)-5,6,7-trihydroxy-3-isopropyl-3-methylisochroman-1-one inhibited osteosarcoma growth by inducing apoptosis. Exp. Ther. Med. 2019, 18, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-C.; Kuo, C.-L.; Lee, M.-H.; Lai, K.-C.; Lin, J.-P.; Yang, J.-S.; Yu, C.-S.; Lu, C.-C.; Chiang, J.-H.; Chueh, F.-S.; et al. Wogonin triggers apoptosis in human osteosarcoma U-2 OS cells through the endoplasmic reticulum stress, mitochondrial dysfunction and caspase-3-dependent signaling pathways. Int. J. Oncol. 2011, 39, 217–224. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhu, W.; Mao, X.; Wang, W.; Chen, Y.; Li, D.; Li, H.; Dou, P. Anti-ganglioside GD2 monoclonal antibody synergizes with cisplatin to induce endoplasmic reticulum-associated apoptosis in osteosarcoma cells. Pharmazie 2018, 73, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.-F.; Huang, C.-W.; Chiang, J.-H.; Tsai, F.-J.; Hsu, Y.-M.; Lu, C.-C.; Hsiao, C.-Y.; Yang, J.-S. Gadolinium chloride elicits apoptosis in human osteosarcoma U-2 OS cells through extrinsic signaling, intrinsic pathway and endoplasmic reticulum stress. Oncol. Rep. 2016, 36, 3421–3426. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tu, H. Psoralen inhibits the proliferation and promotes apoptosis through endoplasmic reticulum stress in human osteosarcoma cells. Folia Histochem. Cytobiol. 2022, 60, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Leonard, M.A.; Milich, R.; Lorch, E.P. The role of pragmatic language use in mediating the relation between hyperactivity and inattention and social skills problems. J. Speech Lang. Hear. Res. 2011, 54, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Duong, H.-Q.; Kim, H.J.; Kang, H.J.; Seong, Y.-S.; Bae, I. ZSTK474, a PI3K inhibitor, suppresses proliferation and sensitizes human pancreatic adenocarcinoma cells to gemcitabine. Oncol. Rep. 2012, 27, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Namatame, N.; Tamaki, N.; Yoshizawa, Y.; Okamura, M.; Nishimura, Y.; Yamazaki, K.; Tanaka, M.; Nakamura, T.; Semba, K.; Yamori, T.; et al. Antitumor profile of the PI3K inhibitor ZSTK474 in human sarcoma cell lines. Oncotarget 2018, 9, 35141–35161. [Google Scholar] [CrossRef]
- Yu, L.; Chen, X.; Wang, L.; Chen, S. The sweet trap in tumors: Aerobic glycolysis and potential targets for therapy. Oncotarget 2016, 7, 38908–38926. [Google Scholar] [CrossRef]
- Burns, C.J.; Fantino, E.; Powell, A.K.; Shnyder, S.D.; Cooper, P.A.; Nelson, S.; Christophi, C.; Malcontenti-Wilson, C.; Dubljevic, V.; Harte, M.F.; et al. The microtubule depolymerizing agent CYT997 causes extensive ablation of tumor vasculature in vivo. J. Pharmacol. Exp. Ther. 2011, 339, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Lang, L.; He, L.; Gao, L.; Chyan, D.; Xiong, Y.; Li, H.; Peng, H.; Teng, Y. Intracellular reduction in ATP levels contributes to CYT997-induced suppression of metastasis of head and neck squamous carcinoma. J. Cell. Mol. Med. 2019, 23, 1174–1182. [Google Scholar] [CrossRef]
- Monaghan, K.; Khong, T.; Smith, G.; Spencer, A. CYT997 causes apoptosis in human multiple myeloma. Investig. New Drugs 2011, 29, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wan, C.; Cheng, J.; Huang, H.; Lovell, J.F.; Jin, H. Delivery Strategies for Melittin-Based Cancer Therapy. ACS Appl. Mater. Interfaces 2021, 13, 17158–17173. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhao, Y.; Luo, W.; Chen, S.; Lin, F.; Zhang, X.; Fan, S.; Shen, X.; Wang, Y.; Liang, G. Celastrol induces ROS-mediated apoptosis via directly targeting peroxiredoxin-2 in gastric cancer cells. Theranostics 2020, 10, 10290–10308. [Google Scholar] [CrossRef] [PubMed]
- Tzeng, C.-W.; Tzeng, W.-S.; Lin, L.-T.; Lee, C.-W.; Yen, F.-L.; Lin, C.-C. Enhanced autophagic activity of artocarpin in human hepatocellular carcinoma cells through improving its solubility by a nanoparticle system. Phytomedicine 2016, 23, 528–540. [Google Scholar] [CrossRef] [PubMed]
- Berdeja, J.G.; Laubach, J.P.; Richter, J.; Stricker, S.; Spencer, A.; Richardson, P.G.; Chari, A. Panobinostat from Bench to Bedside: Rethinking the Treatment Paradigm for Multiple Myeloma. Clin. Lymphoma Myeloma Leuk. 2021, 21, 752–765. [Google Scholar] [CrossRef] [PubMed]
- Ron, D.; Hubbard, S.R. How IRE1 reacts to ER stress. Cell 2008, 132, 24–26. [Google Scholar] [CrossRef] [PubMed]
- Sano, R.; Reed, J.C. ER stress-induced cell death mechanisms. Biochim. Biophys. Acta 2013, 1833, 3460–3470. [Google Scholar] [CrossRef]
- Miao, J.-H.; Wang, S.-Q.; Zhang, M.-H.; Yu, F.-B.; Zhang, L.; Yu, Z.-X.; Kuang, Y. Knockdown of galectin-1 suppresses the growth and invasion of osteosarcoma cells through inhibition of the MAPK/ERK pathway. Oncol. Rep. 2014, 32, 1497–1504. [Google Scholar] [CrossRef]
- Rozpedek, W.; Pytel, D.; Mucha, B.; Leszczynska, H.; Diehl, J.A.; Majsterek, I. The Role of the PERK/eIF2α/ATF4/CHOP Signaling Pathway in Tumor Progression During Endoplasmic Reticulum Stress. Curr. Mol. Med. 2016, 16, 533–544. [Google Scholar] [CrossRef]
- Nishitoh, H. CHOP is a multifunctional transcription factor in the ER stress response. J. Biochem. 2012, 151, 217–219. [Google Scholar] [CrossRef] [PubMed]
- Laplante, M.; Sabatini, D.M. mTOR signaling in growth control and disease. Cell 2012, 149, 274–293. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Almasan, A. Autophagy as a mechanism of Apo2L/TRAIL resistance. Cancer Biol. Ther. 2018, 19, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Hattori, T.; Takahashi, Y.; Chen, L.; Tang, Z.; Wills, C.A.; Liang, X.; Wang, H.-G. Targeting the ESCRT-III component CHMP2A for noncanonical Caspase-8 activation on autophagosomal membranes. Cell Death Differ. 2021, 28, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Martini, M.; Ciraolo, E.; Gulluni, F.; Hirsch, E. Targeting PI3K in Cancer: Any Good News? Front. Oncol. 2013, 3, 108. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.K.; Reckamp, K.; Yu, H.; Figlin, R.A. Akt inhibitors in clinical development for the treatment of cancer. Expert Opin. Investig. Drugs 2010, 19, 1355–1366. [Google Scholar] [CrossRef] [PubMed]
- Baselga, J.; Campone, M.; Piccart, M.; Burris, H.A.; Rugo, H.S.; Sahmoud, T.; Noguchi, S.; Gnant, M.; Pritchard, K.I.; Lebrun, F.; et al. Everolimus in postmenopausal hormone-receptor-positive advanced breast cancer. N. Engl. J. Med. 2012, 366, 520–529. [Google Scholar] [CrossRef]
- Martelli, A.M.; Chiarini, F.; Evangelisti, C.; Cappellini, A.; Buontempo, F.; Bressanin, D.; Fini, M.; McCubrey, J.A. Two hits are better than one: Targeting both phosphatidylinositol 3-kinase and mammalian target of rapamycin as a therapeutic strategy for acute leukemia treatment. Oncotarget 2012, 3, 371–394. [Google Scholar] [CrossRef]
- Yu, F.; Yu, C.; Li, F.; Zuo, Y.; Wang, Y.; Yao, L.; Wu, C.; Wang, C.; Ye, L. Wnt/β-catenin signaling in cancers and targeted therapies. Signal Transduct. Target. Ther. 2021, 6, 307. [Google Scholar] [CrossRef]
- Nusse, R.; Clevers, H. Wnt/β-Catenin Signaling, Disease, and Emerging Therapeutic Modalities. Cell 2017, 169, 985–999. [Google Scholar] [CrossRef]
- Zuo, Q.; Ou, Y.; Zhong, S.; Yu, H.; Zhan, F.; Zhang, M. Targeting GRP78 enhances the sensitivity of HOS osteosarcoma cells to pyropheophorbide-α methyl ester-mediated photodynamic therapy via the Wnt/β-catenin signaling pathway. Acta Biochim. Biophys. Sin. 2021, 53, 1387–1397. [Google Scholar] [CrossRef]
- Chow, W.A. Chondrosarcoma: Biology, genetics, and epigenetics. F1000Research 2018, 7, 1826. [Google Scholar] [CrossRef] [PubMed]
- Marks, K.E.; Bauer, T.W. Fibrous tumors of bone. Orthop. Clin. N. Am. 1989, 20, 377–393. [Google Scholar]
- Fernandez, P.M.; Tabbara, S.O.; Jacobs, L.K.; Manning, F.C.; Tsangaris, T.N.; Schwartz, A.M.; Kennedy, K.A.; Patierno, S.R. Overexpression of the glucose-regulated stress gene GRP78 in malignant but not benign human breast lesions. Breast Cancer Res. Treat. 2000, 59, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, Y.; Hattori, Y.; Kawasaki, T.; Kanoh, K.; Adachi, K.; Shizuri, Y.; Shin-Ya, K. Efrapeptin J, a new down-regulator of the molecular chaperone GRP78 from a marine Tolypocladium sp. J. Antibiot. 2008, 61, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, Y.; Saito, J.; Izawa, M.; Shin-Ya, K. Actinopyrone D, a new downregulator of the molecular chaperone GRP78 from Streptomyces sp. J. Antibiot. 2014, 67, 831–834. [Google Scholar] [CrossRef]
- Su, C.-M.; Chen, C.-Y.; Lu, T.; Sun, Y.; Li, W.; Huang, Y.-L.; Tsai, C.-H.; Chang, C.-S.; Tang, C.-H. A novel benzofuran derivative, ACDB, induces apoptosis of human chondrosarcoma cells through mitochondrial dysfunction and endoplasmic reticulum stress. Oncotarget 2016, 7, 83530–83543. [Google Scholar] [CrossRef]
- Su, C.-M.; Wang, S.-W.; Lee, T.-H.; Tzeng, W.-P.; Hsiao, C.-J.; Liu, S.-C.; Tang, C.-H. Trichodermin induces cell apoptosis through mitochondrial dysfunction and endoplasmic reticulum stress in human chondrosarcoma cells. Toxicol. Appl. Pharmacol. 2013, 272, 335–344. [Google Scholar] [CrossRef]
- Chen, Y.-J.; Wu, C.-L.; Liu, J.-F.; Fong, Y.-C.; Hsu, S.-F.; Li, T.-M.; Su, Y.-C.; Liu, S.-H.; Tang, C.-H. Honokiol induces cell apoptosis in human chondrosarcoma cells through mitochondrial dysfunction and endoplasmic reticulum stress. Cancer Lett. 2010, 291, 20–30. [Google Scholar] [CrossRef]
- Gazit, G.; Kane, S.E.; Nichols, P.; Lee, A.S. Use of the stress-inducible grp78/BiP promoter in targeting high level gene expression in fibrosarcoma in vivo. Cancer Res. 1995, 55, 1660–1663. [Google Scholar] [PubMed]
- Jamora, C.; Dennert, G.; Lee, A.S. Inhibition of tumor progression by suppression of stress protein GRP78/BiP induction in fibrosarcoma B/C10ME. Proc. Natl. Acad. Sci. USA 1996, 93, 7690–7694. [Google Scholar] [CrossRef]

| Medicine | Drug Category | Natural or Chemical Synthesis | Drug Action Target | Mode of Action | Stage of Drug | Reference |
|---|---|---|---|---|---|---|
| ZSTK474 | Phosphoinositi-de 3 kinase inhibitor | Chemical synthesis | PI3K/AKT/mTOR | ZSTK474-VSVΔ51 combination therapy | Experimental stage | [98] |
| CXCR4 | Receptor protein | Natural | PI3K/AKT/NF-κβ | Down-regulated expression and inhibition pathway | Experimental stage | [97] |
| GPR94 | Glucose related protein | Natural | GPR94 | Upregulated expression and improving chemotherapy sensitivity | Experimental stage | [62] |
| Sestrin2 | Gene target | Natural | Sestrin2/PERK/eIF2α/CHOP | Knocking out and improving chemotherapy sensitivity | Experimental stage | [73] |
| CYT997 | Microtubule targeting agent | Chemical synthesis | PERK/eIF2α/CHOP/ERO1 | Activating ROS, inducing autophagy and apoptosis | Experimental stage | [72] |
| β-Elemonic acid | Active ingredient | Natural | PERK/eIF2α/ATF4/CHOP, Wnt/β-catenin | Direct treatment | Experimental stage | [16] |
| celastrol | Quinone methylamine triterpenoids | Natural | PERK/eIF2α | Inducing autophagy and apoptosis | Applied treatment | [129] |
| Surfactant | Cyclic lipopeptide | Natural | IRE1/ASK1/JNK | Abnormal Ca2+ Release and strengthening routine treatment | Experimental stage | [130] |
| E2F1 | Transcription factor | Natural | IRE1/Xbp-1 | Down, combined with ATF6, inhibition GPR78 | Experimental stage | [57] |
| Melittin | Protein | Natural | IRE1/Xbp-1 | Inhibition MG63 cell proliferation | Applied treatment | [131] |
| Artocarpin | Flavonoid derivatives | Natural | GPR78 | Activation of ROS, ER stress and other pathways, inducing apoptosis | Experimental stage | [132] |
| Kuanoniami-ne C | Nitramine | Natural | GPR78 | Degradation GPR78 mRNA, stimulating bortezomib to induce apoptosis | Experimental stage | [133] |
| α-Mangostin | Enzyme | Natural | Wnt/β-catenin | Inducing apoptosis | Experimental stage | [106] |
| ZBTB7A | miR663 target gene | Natural | miR-663a/ZBTB7A/LncRNAGAS5 | Down regulation, promoting apoptosis | Experimental stage | [112] |
| Endoplasmic reticulum targeted adriamycin | Adriamycin | Chemical synthesis | C/EBP-β LIP/CHOP/PUMA/caspases 12-7-3 | Increasing sensitivity | Experimental stage | [134] |
| Calcitriol | Alcohol | Natural | Cell cycle | Inhibition of AXT cell proliferation | Approved by FDA | [135] |
| Plumbagin | Plant isolate | Natural | Apoptotic pathway | Induction of ROS and mitochondrial dysfunction | Experimental stage | [136] |
| Stim1 | Medium factor | Natural | GPR78, CHOP, ATF4 | Knocking down, increasing cisplation sensitivity | Experimental stage | [137] |
| Grphene oxide nanoparticles | MTH1 inhibitor | Chemical synthesis | JNK/p53/p21 | Combined photodynamic therapy | Experimental stage | [82] |
| HA-Lsdox | Conjugated liposome | Chemical synthesis | Pgp, CHOP | Improving the sensitivity of adrianycin treatment | Experimental stage | [138] |
| Panobinostat | Anticancer drugs | Chemical synthesis | P21, TP53, Bip, CHOP | Inhibiting OS cell survival | Experimental stage | [139] |
| TIM | Plant isolate | Natural | IRE1, ATF6 | Antitumor | Experimental stage | [140] |
| Wogonin | Flavone | Natural | GPR78 | Cleaving GPR78 and promoting apoptosis | Experimental stage | [141] |
| An-GD2-mAb | Antibody | Chemical synthesis | elF2α, CHOP | Cisplatin and An-GD2 mAb combination therapy | Experimental stage | [142] |
| GdCl3 | Chemical anticancer agent | Chemical synthesis | DNA | Inducing ERS and promoting apoptosis | Experimental stage | [143] |
| Psoralen | Pseudomonas active ingredient | Natural | ATF6, CHOP | Promoting apoptosis | Experimental stage | [144] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, P.; Li, T.; Li, Q.; Gu, Y.; Shu, Y.; Hu, K.; Chen, L.; Peng, X.; Peng, J.; Hao, L. Mechanism and Role of Endoplasmic Reticulum Stress in Osteosarcoma. Biomolecules 2022, 12, 1882. https://doi.org/10.3390/biom12121882
Zhu P, Li T, Li Q, Gu Y, Shu Y, Hu K, Chen L, Peng X, Peng J, Hao L. Mechanism and Role of Endoplasmic Reticulum Stress in Osteosarcoma. Biomolecules. 2022; 12(12):1882. https://doi.org/10.3390/biom12121882
Chicago/Turabian StyleZhu, Peijun, Ting Li, Qingqing Li, Yawen Gu, Yuan Shu, Kaibo Hu, Leifeng Chen, Xiaogang Peng, Jie Peng, and Liang Hao. 2022. "Mechanism and Role of Endoplasmic Reticulum Stress in Osteosarcoma" Biomolecules 12, no. 12: 1882. https://doi.org/10.3390/biom12121882
APA StyleZhu, P., Li, T., Li, Q., Gu, Y., Shu, Y., Hu, K., Chen, L., Peng, X., Peng, J., & Hao, L. (2022). Mechanism and Role of Endoplasmic Reticulum Stress in Osteosarcoma. Biomolecules, 12(12), 1882. https://doi.org/10.3390/biom12121882







