Activin A Modulates Betaglycan Shedding via the ALK4-SMAD3-Dependent Pathway in Endometriotic Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells
2.2. Cell Culture
2.3. Recombinant Proteins, Inhibitors, and ELISAs
2.4. Treatment of Cells with Various Agents
2.5. Betaglycan, MMP2 and MMP3 ELISAs
2.6. Western Blot
2.7. SiRNA Transfection, RNA Isolation and Real Time-qPCR
2.8. CCK-8 Cell Viability and BrdU Proliferation Assays
2.9. Statistical Analyses
3. Results
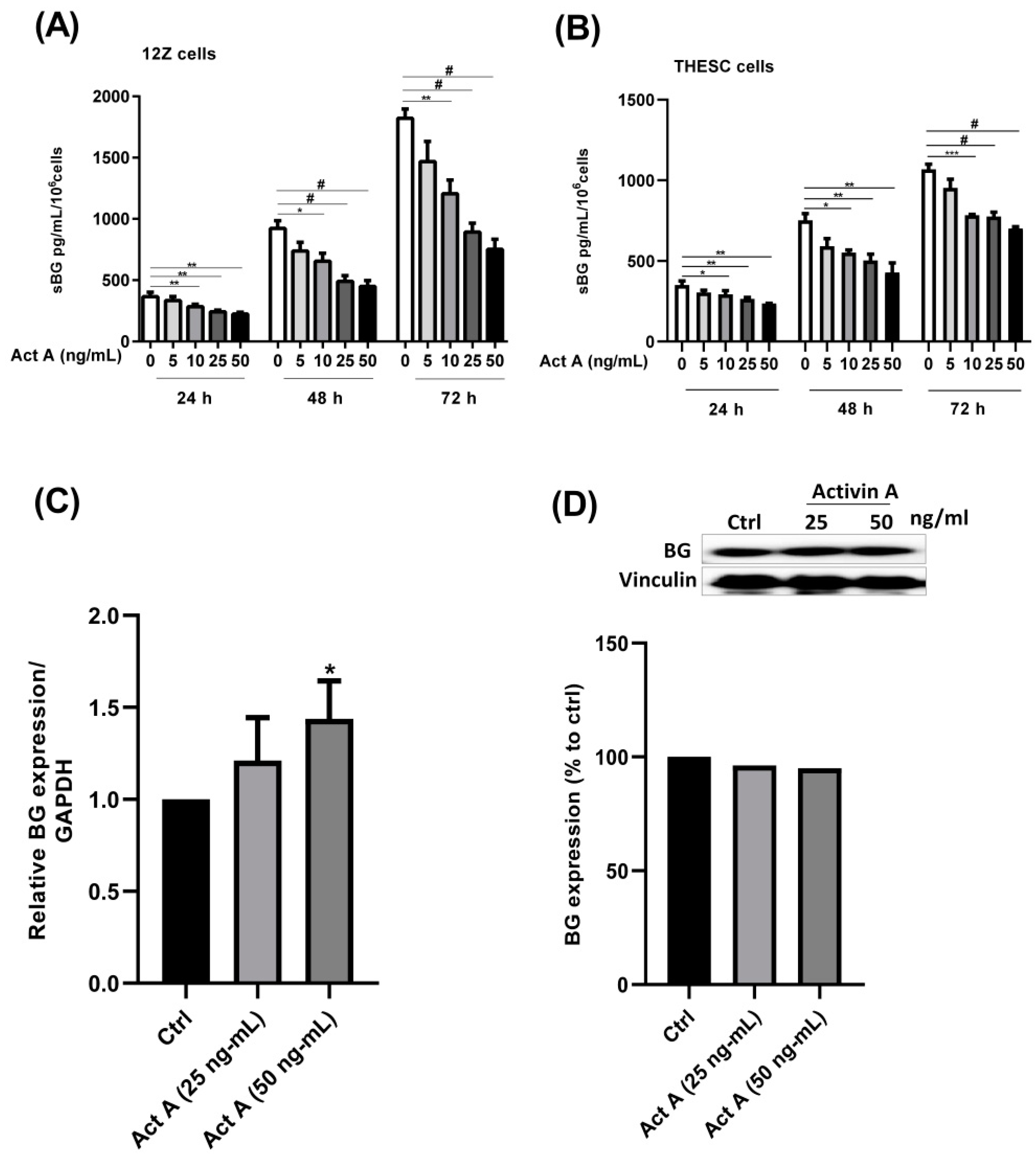
3.1. Modulation of BG Shedding and Expression by Activin A in Endometrial Cells
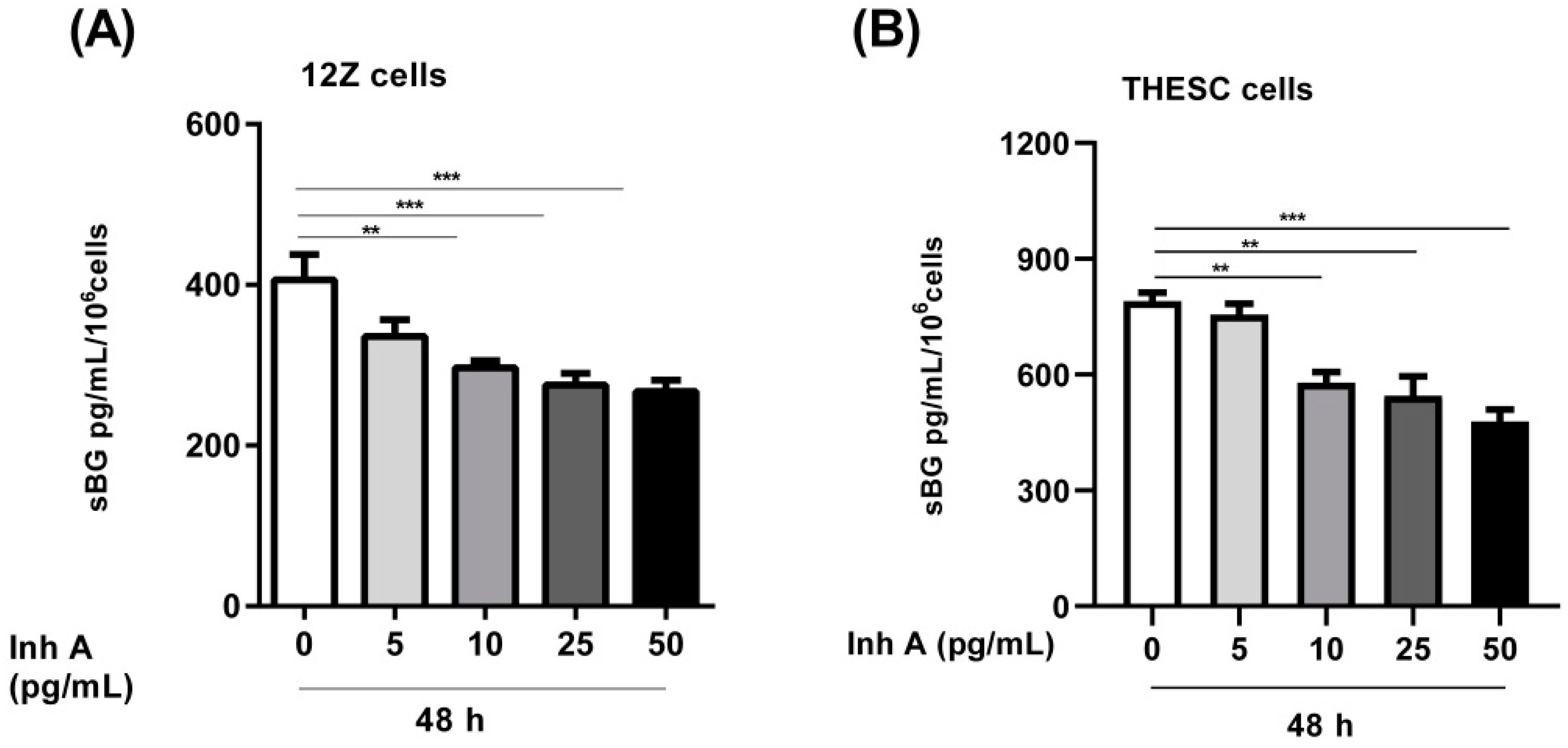
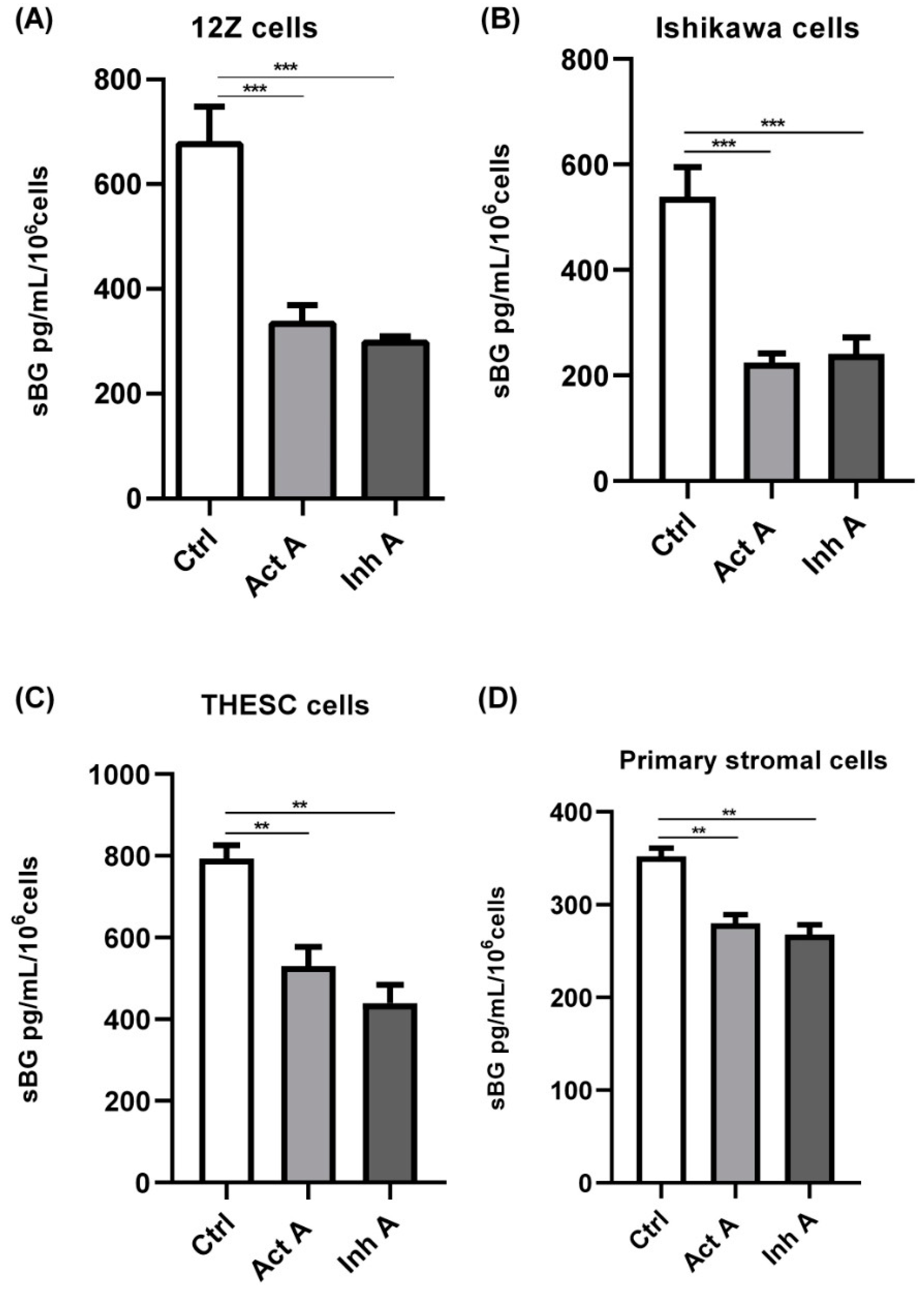
3.2. Modulation of BG Shedding by Activin A and Inhibin A in Different Cell Types
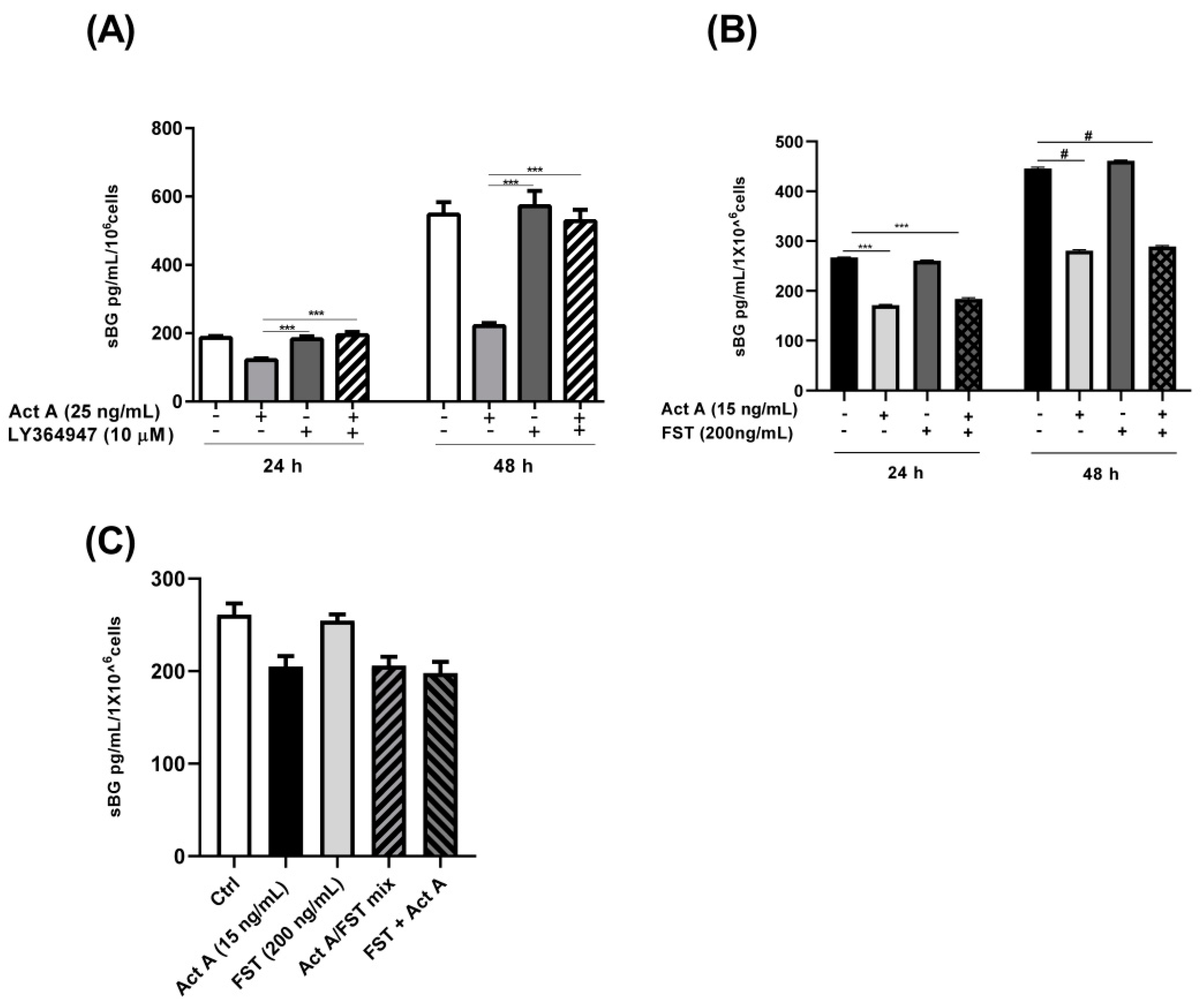
3.3. Involvement of Activin Receptor Type-1B (ALK-4) in BG Shedding
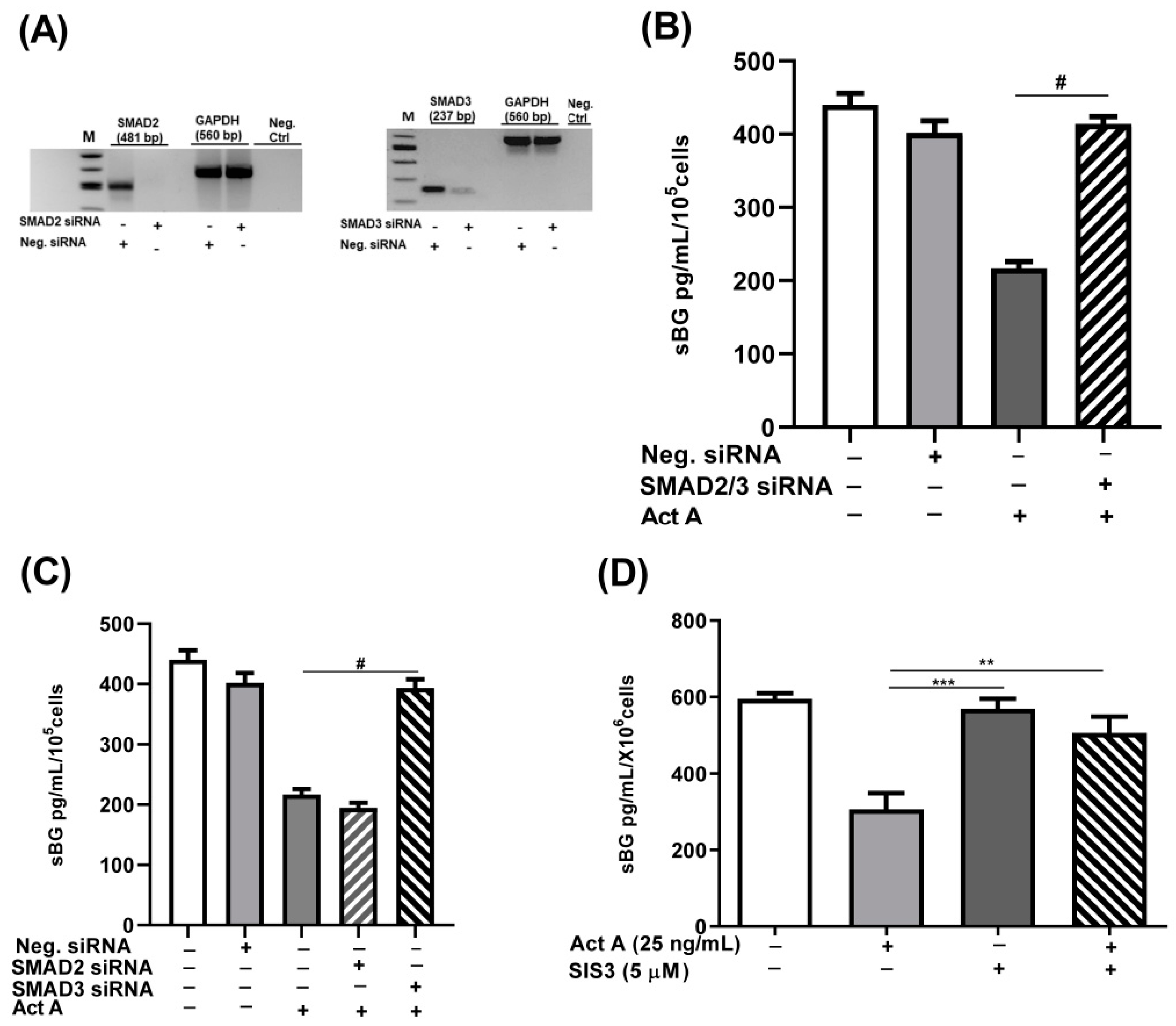
3.4. Involvement of SMAD2/3 in Activin A-Mediated Reduction in BG Shedding
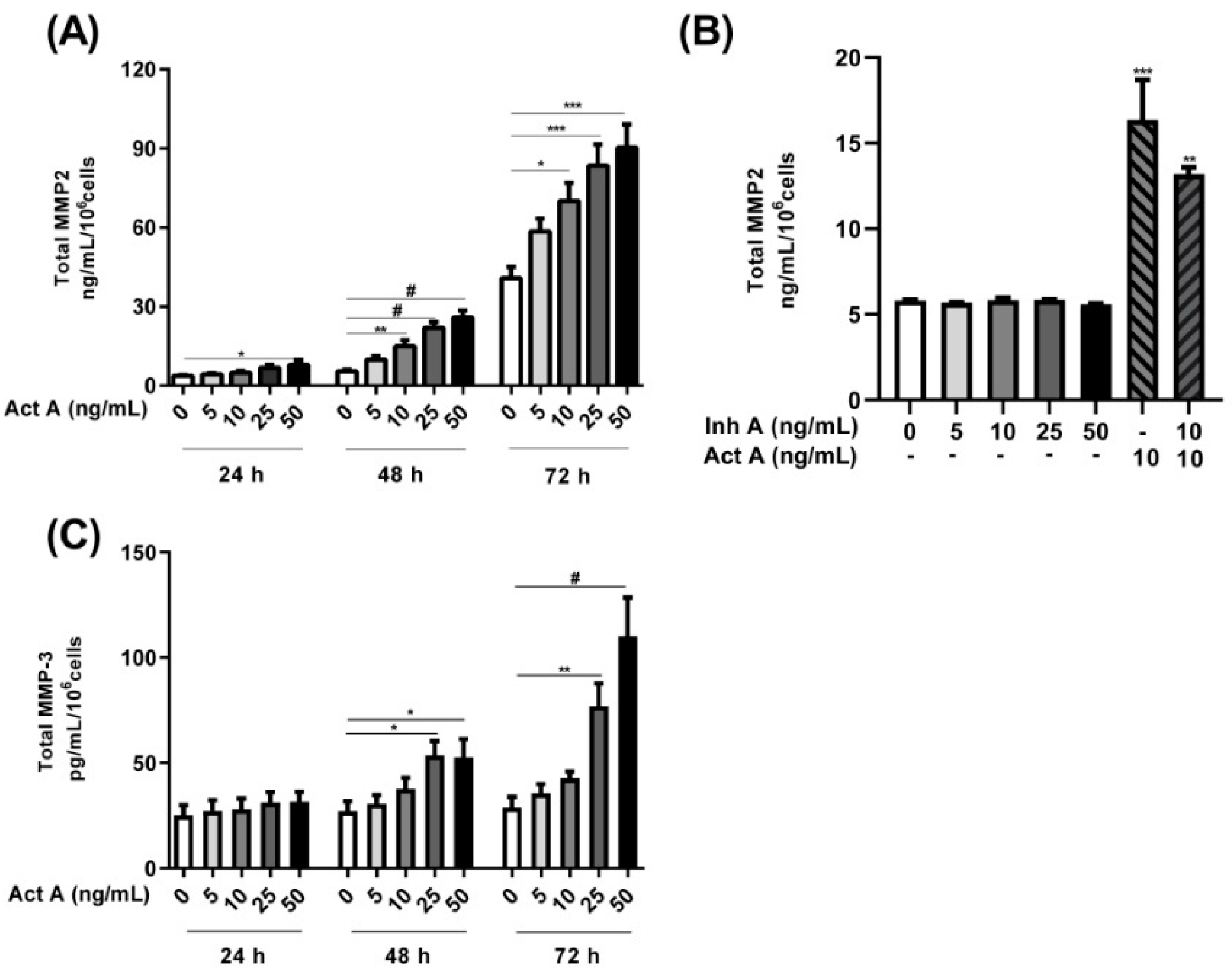
3.5. Influence of Activin A and Inhibin A in MMP2 and MMP3 Secretion
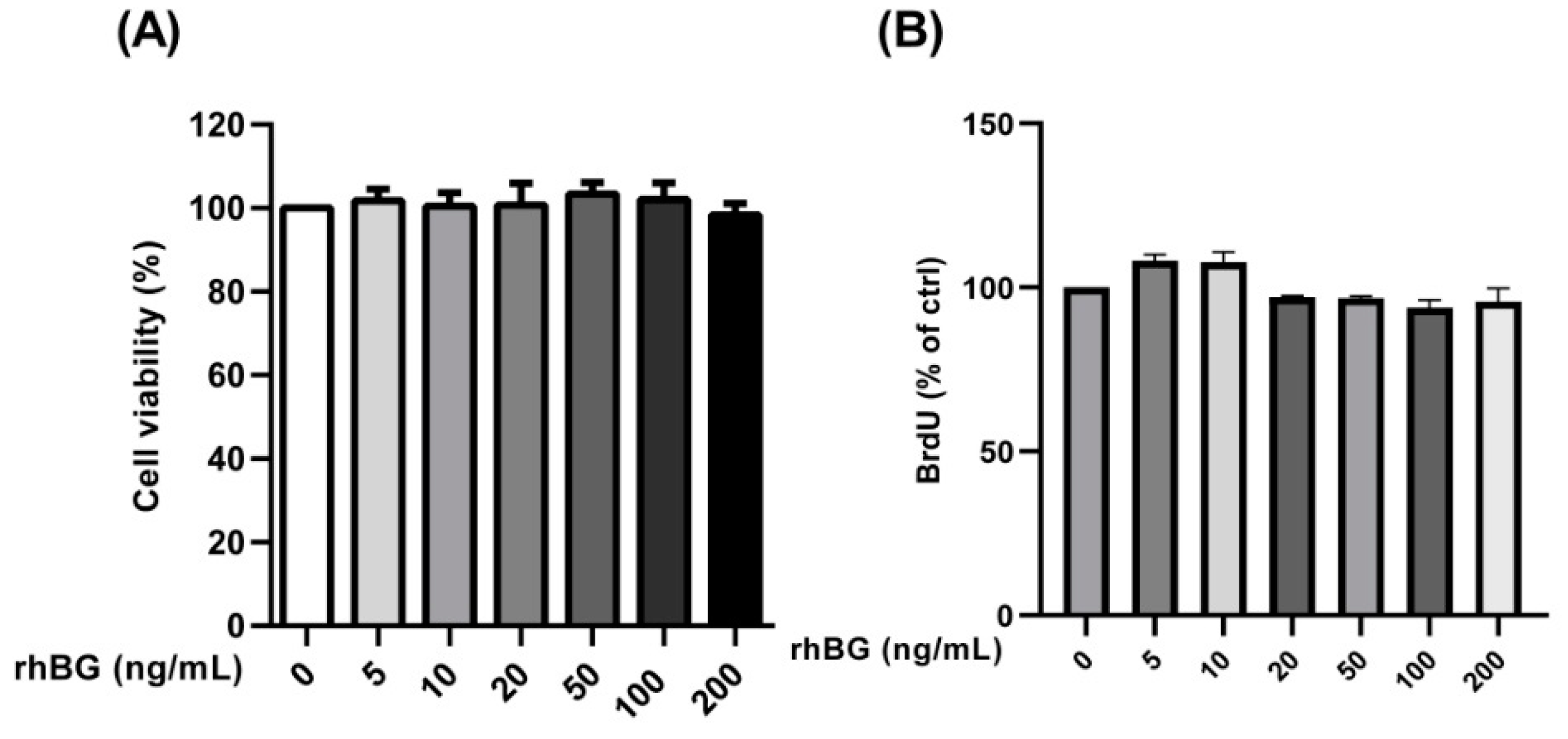
3.6. Influence of Recombinant Betaglycan on Cell Viability and Proliferation
4. Discussion
4.1. Action of Activin A and Inhibin A on BG Shedding in Endometrial Cells
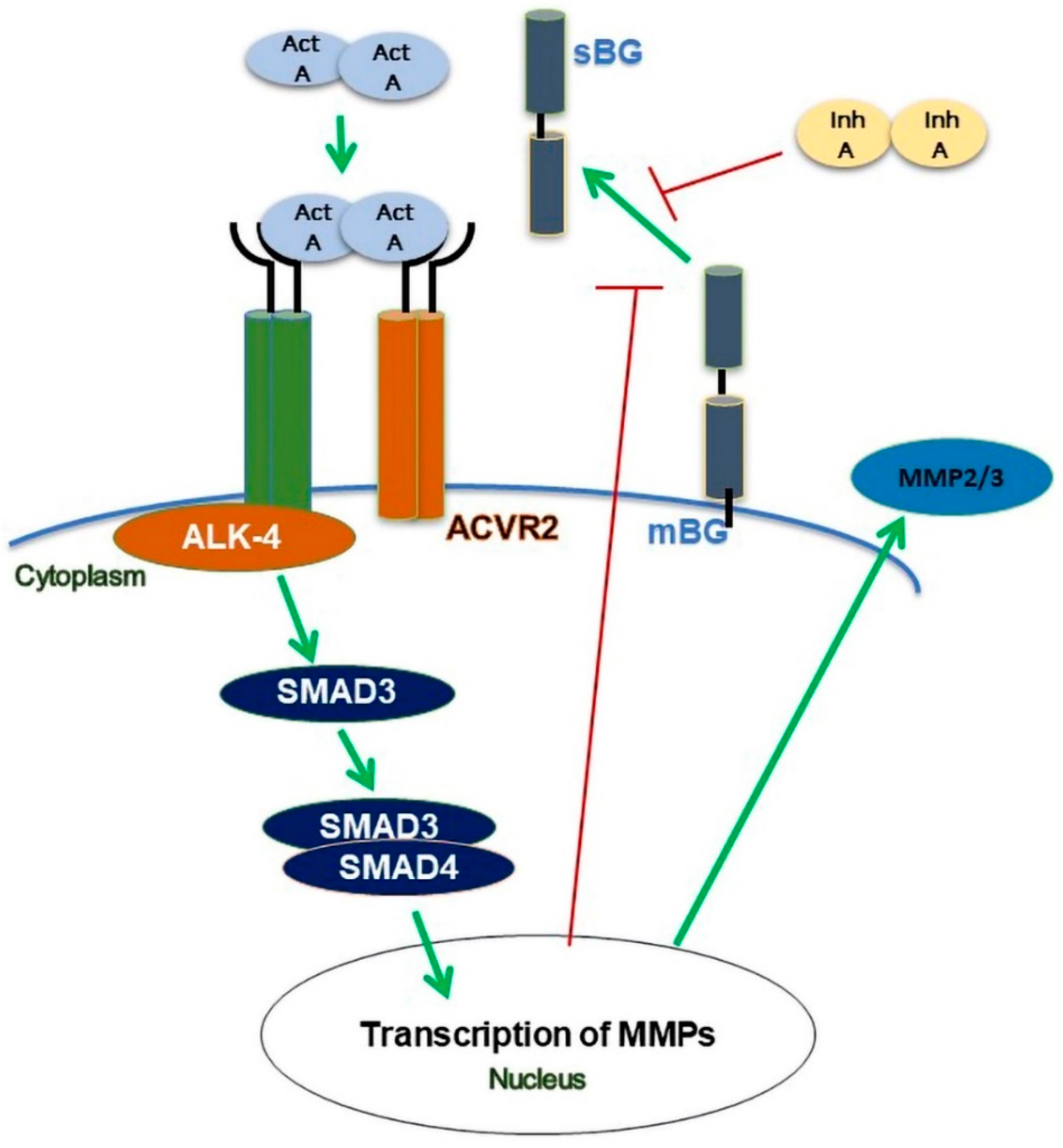
4.2. Mechanisms Involved in Activin A-Mediated Modulation of BG Shedding
4.3. Role of Activin A and Inhibin A in MMP2 and MMP3 Secretion
4.4. Role of Betaglycan in Cell Viability
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zondervan, K.T.; Becker, C.M.; Koga, K.; Missmer, S.A.; Taylor, R.N.; Viganò, P. Endometriosis. Nat. Rev. Dis. Prim. 2018, 4, 95. [Google Scholar] [CrossRef]
- Ballard, K.D.; Seaman, H.E.; De Vries, C.S.; Wright, J.T. Can symptomatology help in the diagnosis of endometriosis? Findings from a national case-control study—Part 1. BJOG Int. J. Obstet. Gynaecol. 2008, 115, 1382–1391. [Google Scholar] [CrossRef]
- Vercellini, P.; Viganò, P.; Somigliana, E.; Fedele, L. Endometriosis: Pathogenesis and treatment. Nat. Rev. Endocrinol. 2014, 10, 261–275. [Google Scholar] [CrossRef]
- Ghiasi, M.; Kulkarni, M.T.; Missmer, S.A. Is endometriosis more common and more severe than it was 30 years ago? J. Minim. Invasive Gynecol. 2020, 27, 452–461. [Google Scholar] [CrossRef] [Green Version]
- Busacca, M.; Vignali, M. Ovarian endometriosis: From pathogenesis to surgical treatment. Curr. Opin. Obstet. Gynecol. 2003, 15, 321–326. [Google Scholar] [CrossRef]
- Sampson, J.A. Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. Am. J. Obstet. Gynecol. 1927, 14, 422–469. [Google Scholar] [CrossRef]
- Derynck, R.; Budi, E.H. Specificity, versatility, and control of TGF-β family signaling. Sci. Signal. 2019, 12, eaav5183. [Google Scholar] [CrossRef] [Green Version]
- Omwandho, C.O.A.; Konrad, L.; Halis, G.; Oehmke, F.; Tinneberg, H. Role of TGF-βs in normal human endometrium and endometriosis. Hum. Reprod. 2010, 25, 101–109. [Google Scholar] [CrossRef] [Green Version]
- Finnson, K.W.; McLean, S.; Di Guglielmo, G.M.; Philip, A. Dynamics of transforming growth factor beta signaling in wound healing and scarring. Adv. Wound Care 2013, 2, 195–214. [Google Scholar] [CrossRef] [Green Version]
- Cruz, C.D.; Reis, F.M. The role of TGF β superfamily members in the pathophysiology of endometriosis. Gynecol. Endocrinol. 2015, 31, 511–515. [Google Scholar] [CrossRef]
- Young, V.J.; Ahmad, S.F.; Duncan, W.C.; Horne, A.W. The role of TGF-β in the pathophysiology of peritoneal endometriosis. Hum. Reprod. Update 2017, 23, 548–559. [Google Scholar] [CrossRef] [Green Version]
- Balkowiec, M.; Maksym, R.B.; Wlodarski, P.K. The bimodal role of matrix metalloproteinases and their inhibitors in etiology and pathogenesis of endometriosis (Review). Mol. Med. Rep. 2018, 18, 3123–3136. [Google Scholar] [CrossRef] [Green Version]
- Namwanje, M.; Brown, C.W. Activins and inhibins: Roles in development, physiology, and disease. Cold Spring Harb. Perspect. Biol. 2016, 8, a021881. [Google Scholar] [CrossRef] [Green Version]
- Kaitu’u-Lino, T.J.; Phillips, D.J.; Morison, N.B.; Salamonsen, L.A. A new role for activin in endometrial repair after menses. Endocrinology 2009, 150, 1904–1911. [Google Scholar] [CrossRef] [Green Version]
- Appiah Adu-Gyamfi, E.; Tanam Djankpa, F.; Nelson, W.; Czika, A.; Kumar Sah, S.; Lamptey, J.; Ding, Y.B.; Wang, Y.X. Activin and inhibin signaling: From regulation of physiology to involvement in the pathology of the female reproductive system. Cytokine 2020, 133, 155105. [Google Scholar] [CrossRef]
- Reis, F.M.; Blasio, A.M.D.; Florio, P.; Ambrosini, G.; Loreto, D.C.; Petraglia, F. Evidence for local production of inhibin A and activin A in patients with ovarian endometriosis. Fertil. Steril. 2001, 75, 367–373. [Google Scholar] [CrossRef]
- Rombauts, L.; Donoghue, J.; Cann, L.; Jones, R.L.; Healy, D.L. Activin-A secretion is increased in the eutopic endometrium from women with endometriosis. Aust. N. Z. J. Obstet. Gynaecol. 2006, 46, 148–153. [Google Scholar] [CrossRef]
- Mabuchi, Y.; Yamoto, M.; Minami, S.; Umesaki, N. Immunohistochemical localization of inhibin and activin subunits, activin receptors and Smads in ovarian endometriosis. Int. J. Mol. Med. 2010, 25, 17–23. [Google Scholar] [CrossRef]
- Onichtchouk, D.; Chen, Y.G.; Dosch, R.; Gawantka, V.; Delius, H.; Massagué, J.; Niehrs, C. Silencing of TGF-β signalling by the pseudoreceptor BAMBI. Nature 1999, 401, 480–485. [Google Scholar] [CrossRef]
- Wang, X.F.; Lin, H.Y.; Ng-Eaton, E.; Downward, J.; Lodish, H.F.; Weinberg, R.A. Expression, cloning and characterization of the TGF-β type III receptor. Cell 1991, 67, 797–805. [Google Scholar] [CrossRef]
- López-Casillas, F.; Payne, H.M.; Andres, J.L.; Massagué, J. Betaglycan can act as a dual modulator of TGF-β access to signaling receptors: Mapping of ligand binding and GAG attachment sites. J. Cell. Biol. 1994, 124, 557–568. [Google Scholar] [CrossRef] [Green Version]
- Bilandzic, M.; Stenvers, K.L. Betaglycan: A multifunctional accessory. Mol. Cell. Endocrinol. 2011, 339, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Gatza, C.E.; Oh, S.Y.; Blobe, G.C. Roles for the type III TGF-β receptor in human cancer. Cell Signal. 2010, 22, 1163–1174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.D.; Hempel, N.; Lee, N.Y.; Blobe, G.C. The type III TGF-β receptor suppresses breast cancer progression through GIPC-mediated inhibition of TGF-β signaling. Carcinogenesis 2010, 31, 175–183. [Google Scholar] [CrossRef]
- Nishida, J.; Miyazono, K.; Ehata, S. Decreased TGFBR3/betaglycan expression enhances the metastatic abilities of renal cell carcinoma cells through TGF-β-dependent and -independent mechanisms. Oncogene 2018, 37, 2197–2212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grgurevic, L.; Novak, R.; Trkulja, V.; Hrkac, S.; Salai, G.; Bilandzic, J.; Hamzic, L.F.; Milas, I.; Vucemilo, T.; Balja, M.P.; et al. Plasma levels and tissue expression of soluble TGFβrIII receptor in women with early-stage breast cancer and in healthy women: A prospective observational study. J. Transl. Med. 2020, 18, 478. [Google Scholar] [CrossRef]
- Andres, J.L.; Stanley, K.; Cheifetz, S.; Massague, J. Membrane-anchored and soluble forms of betaglycan, a polymorphic proteoglycan that binds transforming growth factor-β. J. Cell. Biol. 1989, 109, 3137–3145. [Google Scholar] [CrossRef]
- Zhang, M.F.; Zola, H.; Read, L.C.; Penttila, I.A. Identification of soluble transforming growth factor-β receptor III (sTβIII) in rat milk. Immunol. Cell Biol. 2001, 79, 291–297. [Google Scholar] [CrossRef]
- Velasco-Loyden, G.; Arribas, J.; López-Casillas, F. The shedding of betaglycan is regulated by pervanadate and mediated by membrane type matrix metalloprotease-1. J. Biol. Chem. 2004, 279, 7721–7733. [Google Scholar] [CrossRef] [Green Version]
- Mwaura, A.N.; Riaz, M.A.; Maoga, J.B.; Mecha, E.; Omwandho, C.O.A.; Scheiner-Bobis, G.; Meinhold-Heerlein, I.; Konrad, L. Role of betaglycan in TGF-β signaling and wound healing in human endometriotic epithelial cells and in endometriosis. Biology 2022, 11, 513. [Google Scholar] [CrossRef]
- Kudipudi, P.K.; Galuska, S.P.; Dietze, R.; Scheiner-bobis, G.; Loveland, K.L.; Konrad, L. Betaglycan (TβRIII) is a key factor in TGF-β2 signaling in prepubertal rat Sertoli cells. Int. J. Mol. Sci. 2019, 20, 6214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Escalona, R.M.; Stenvers, K.L.; Farnworth, P.G.; Findlay, J.K.; Ooi, G.T. Reducing betaglycan expression by RNA interference (RNAi) attenuates inhibin bioactivity in LβT2 gonadotropes. Mol. Cell. Endocrinol. 2009, 307, 149–156. [Google Scholar] [CrossRef]
- Lewis, K.A.; Gray, P.C.; Blount, A.L.; MacConell, L.A.; Wiater, E.; Bitezikjian, L.M.; Vale, W. Betaglycan binds inhibin and can mediate functional antagonism of activin signalling. Nature 2000, 404, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Zeitvogel, A.; Baumann, R.; Starzinski-Powitz, A. Identification of an invasive, N-cadherin-expressing epithelial cell type in endometriosis using a new cell culture model. Am. J. Pathol. 2001, 159, 1839–1852. [Google Scholar] [CrossRef] [Green Version]
- Banu, S.K.; Lee, J.H.; Starzinski-Powitz, A.; Arosh, J.A. Gene expression profiles and functional characterization of human immortalized endometriotic epithelial and stromal cells. Fertil. Steril. 2008, 90, 972–987. [Google Scholar] [CrossRef] [PubMed]
- Krikun, G.; Mor, G.; Alvero, A.; Guller, S.; Schatz, F.; Sapi, E.; Rahman, M.; Caze, R.; Qumsiyeh, M.; Lockwood, C.J. A novel immortalized human endometrial stromal cell line with normal progestational response. Endocrinology 2004, 145, 2291–2296. [Google Scholar] [CrossRef]
- Swangchan-Uthai, T.; Lavender, C.R.M.; Cheng, Z.; Fouladi-Nashta, A.A.; Wathes, D.C. Time course of defense mechanisms in bovine endometrium in response to lipopolysaccharide. Biol. Reprod. 2012, 87, 135. [Google Scholar] [CrossRef]
- Raheem, K.; Fouladi-Nashta, A. Isolation and characterization of endometrial luminal epithelial and stromal cells in vitro. Sokoto J. Vet. Sci. 2014, 12, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Fortier, M.A.; Guilbault, L.A.; Grasso, F. Specific properties of epithelial and stromal cells from the endometrium of cows. Reproduction 1988, 83, 239–248. [Google Scholar] [CrossRef] [Green Version]
- Masuda, A.; Katoh, N.; Nakabayashi, K.; Kato, K.; Sonoda, K.; Kitade, M.; Takeda, S.; Hata, K.; Tomikawa, J. An improved method for isolation of epithelial and stromal cells from the human endometrium. J. Reprod. Dev. 2016, 62, 2015–2137. [Google Scholar] [CrossRef]
- Cui, X.; Shang, S.; Lv, X.; Zhao, J.; Qi, Y.; Liu, Z. Perspectives of small molecule inhibitors of activin receptor-like kinase in anti-tumor treatment and stem cell differentiation (Review). Mol. Med. Rep. 2019, 19, 5053–5062. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, R.L.; Findlay, J.K.; Farnworth, P.G.; Robertson, D.M.; Wallace, E.; Salamonsen, L.A. Activin A and inhibin a differentially regulate human uterine matrix metalloproteinases: Potential interactions during decidualization and trophoblast invasion. Endocrinology 2006, 147, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Criswell, T.L.; Dumont, N.; Barnett, J.V.; Arteaga, C.L. Knockdown of the transforming growth factor-β type III receptor impairs motility and invasion of metastatic cancer cells. Cancer Res. 2008, 68, 7304–7312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-Casillas, F.; Cheifetz, S.; Doody, J.; Andres, J.L.; Lane, W.S.; Massague, J. Structure and expression of the membrane proteoglycan betaglycan, a component of the TGF-β receptor system. Cell 1991, 67, 785–795. [Google Scholar] [CrossRef] [PubMed]
- López-Casillas, F.; Wrana, J.L.; Massagué, J. Betaglycan presents ligand to the TGFβ signaling receptor. Cell 1993, 73, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Toufaily, C.; Bernard, D.J. Mechanisms of inhibin action. In Reference Module in Neuroscience and Biobehavioral Psychology; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 297–303. [Google Scholar]
- Farnworth, P.G.; Stanton, P.G.; Wang, Y.; Escalona, R.; Findlay, J.K.; Ooi, G.T. Inhibins differentially antagonize activin and bone morphogenetic protein action in a mouse adrenocortical cell line. Endocrinology 2006, 147, 3462–3471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Looyenga, B.D.; Wiater, E.; Vale, W.; Hammer, G.D. Inhibin-A antagonizes TGFβ2 signaling by down-regulating cell surface expression of the TGFβ coreceptor betaglycan. Mol. Endocrinol. 2010, 24, 608–620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ethier, J.-F.; Farnworth, P.G.; Findlay, J.K.; Ooi, G.T. Transforming growth factor-β modulates inhibin A bioactivity in the LβT2 gonadotrope cell line by competing for binding to betaglycan. Mol. Endocrinol. 2002, 16, 2754–2763. [Google Scholar] [CrossRef] [PubMed]
- Harrington, A.E.; Morris-Triggs, S.A.; Ruotolo, B.T.; Robinson, C.V.; Ohnuma, S.I.; Hyvönen, M. Structural basis for the inhibition of activin signalling by follistatin. EMBO J. 2006, 25, 1035–1045. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mather, P.; Roberts, P.E.; Krummen, L.A. Follistatin modulates activin activity in a cell- and tissue specific-manner. Endocrinology 1993, 132, 2732–2734. [Google Scholar] [CrossRef] [PubMed]
- Gilabert-Estellés, J.; Ramón, L.A.; España, F.; Gilabert, J.; Vila, V.; Réganon, E.; Castelló, R.; Chirivella, M.; Estellés, A. Expression of angiogenic factors in endometriosis: Relationship to fibrinolytic and metalloproteinase systems. Hum. Reprod. 2007, 22, 2120–2127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lv, X.; Chen, P.; Liu, W. Down regulation of MiR-93 contributes to endometriosis through targeting MMP3 and VEGFA. Am. J. Cancer Res. 2015, 5, 1706–1717. [Google Scholar] [PubMed]
- Mecha, E.O.; Sui, C.; Omwandho, C.O.A.; Tinneberg, H.-R.; Konrad, L. Transforming growth factor betas induce MMP-2 and MMP-9 secretion via smad-dependent signaling in human endometrial and endometriotic Cells. Int. J. Sci. Eng. Technol. 2015, 4, 567–572. [Google Scholar]
- Ogawa, K.; Funaba, M.; Mathews, L.S.; Mizutani, T. Activin A stimulates type IV collagenase (matrix metalloproteinase-2) production in mouse peritoneal macrophages. J. Immunol. 2000, 165, 2997–3003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Klausen, C.; Zhu, H.; Leung, P.C.K. Activin A increases human trophoblast invasion by inducing SNAIL-mediated MMP2 up-regulation through ALK4. J. Clin. Endocrinol. Metab. 2015, 100, E1415–E1427. [Google Scholar] [CrossRef] [PubMed]
- Turley, R.S.; Finger, E.C.; Hempel, N.; How, T.; Fields, T.A.; Blobe, G.C. The type III transforming growth factor-β receptor as a novel tumor suppressor gene in prostate cancer. Cancer Res. 2007, 67, 1090–1098. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bilandzic, M.; Chu, S.; Farnworth, P.G.; Harrison, C.; Nicholls, P.; Wang, Y.; Escalona, R.M.; Fuller, P.J.; Findlay, J.K.; Stenvers, K.L. Loss of betaglycan contributes to the malignant properties of human granulosa tumor cells. Mol. Endocrinol. 2009, 23, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Finger, E.C.; Turley, R.S.; Dong, M.; How, T.; Fields, T.A.; Blobe, G.C. TβRIII suppresses non-small cell lung cancer invasiveness and tumorigenicity. Carcinogenesis 2008, 29, 528–535. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mwaura, A.N.; Riaz, M.A.; Maoga, J.B.; Mecha, E.; Omwandho, C.O.A.; Scheiner-Bobis, G.; Meinhold-Heerlein, I.; Konrad, L. Activin A Modulates Betaglycan Shedding via the ALK4-SMAD3-Dependent Pathway in Endometriotic Cells. Biomolecules 2022, 12, 1749. https://doi.org/10.3390/biom12121749
Mwaura AN, Riaz MA, Maoga JB, Mecha E, Omwandho COA, Scheiner-Bobis G, Meinhold-Heerlein I, Konrad L. Activin A Modulates Betaglycan Shedding via the ALK4-SMAD3-Dependent Pathway in Endometriotic Cells. Biomolecules. 2022; 12(12):1749. https://doi.org/10.3390/biom12121749
Chicago/Turabian StyleMwaura, Agnes N., Muhammad A. Riaz, Jane B. Maoga, Ezekiel Mecha, Charles O. A. Omwandho, Georgios Scheiner-Bobis, Ivo Meinhold-Heerlein, and Lutz Konrad. 2022. "Activin A Modulates Betaglycan Shedding via the ALK4-SMAD3-Dependent Pathway in Endometriotic Cells" Biomolecules 12, no. 12: 1749. https://doi.org/10.3390/biom12121749
APA StyleMwaura, A. N., Riaz, M. A., Maoga, J. B., Mecha, E., Omwandho, C. O. A., Scheiner-Bobis, G., Meinhold-Heerlein, I., & Konrad, L. (2022). Activin A Modulates Betaglycan Shedding via the ALK4-SMAD3-Dependent Pathway in Endometriotic Cells. Biomolecules, 12(12), 1749. https://doi.org/10.3390/biom12121749







