Focusing on the Role of Natural Products in Overcoming Cancer Drug Resistance: An Autophagy-Based Perspective
Abstract
1. Introduction
2. Methods
3. Mechanisms of Autophagy
4. Autophagy and Tumor
4.1. The Dual Role of Autophagy in Tumors
4.2. Autophagy and Tumor Drug Resistance
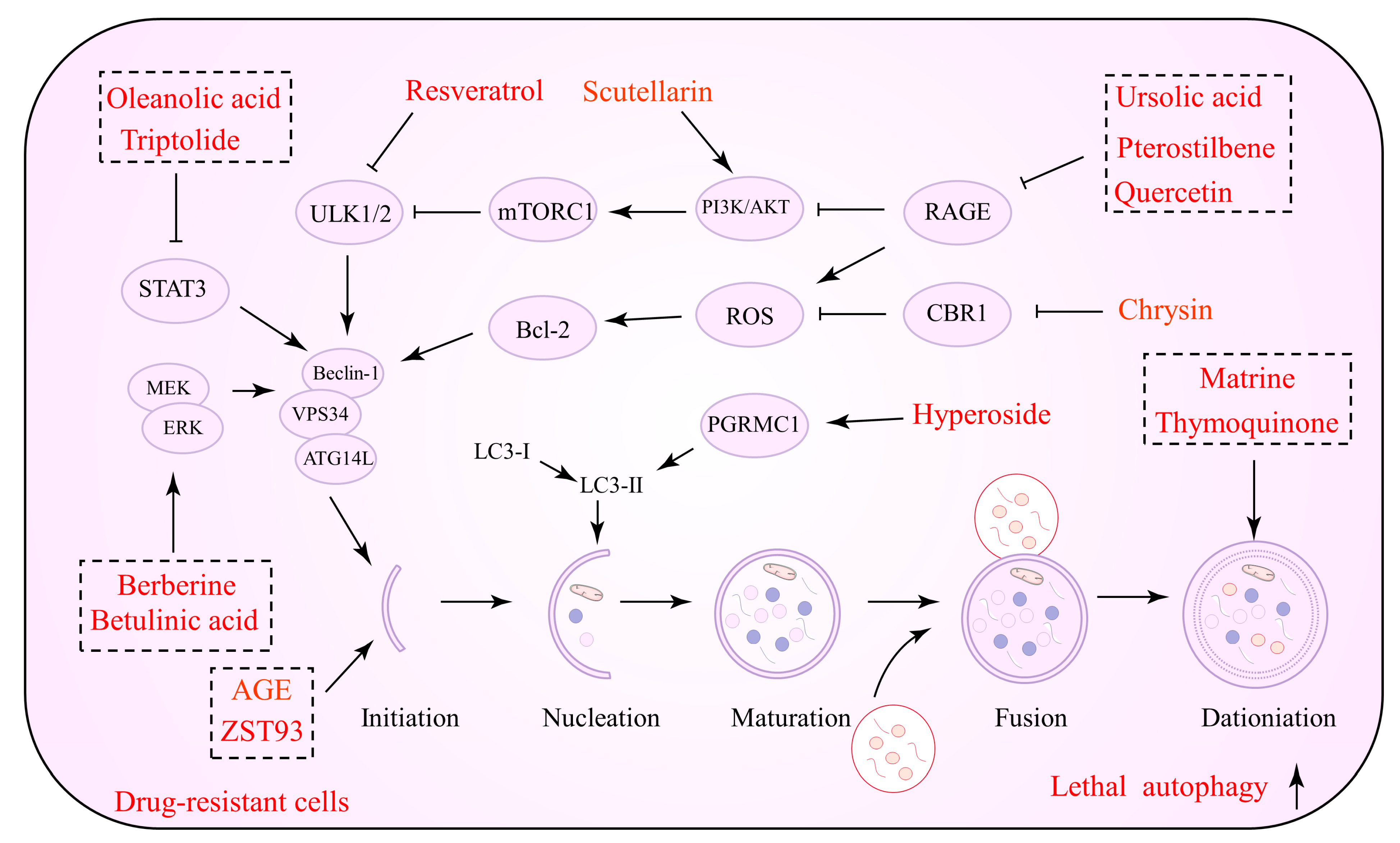
5. Natural Products Overcome Autophagy-Mediated Tumor Drug Resistance
5.1. Natural Products as An Inhibitor for Protective Autophagy
| Compounds | Plant Origin | Classification | Cancer Types | Models and Dosage | Mechanism | Main Effects | References |
|---|---|---|---|---|---|---|---|
| Andrographolide | Andrographis paniculata | Terpenoid | Non-small-cell lung cancer (NSCLC) | In vitro: A549/DDP cell line (30 µM) In vivo: mice (5 mg/kg) | Inhibits autophagy and promotes cell apoptosis | Facilitates cisplatin sensitivity | [59] |
| α-Hederin | Hedera helix | Terpenoid | NSCLC | NCI-H1299 and NCI-H1650 cell lines (12.5 µM, 24 h) | Inhibits autophagy and promotes ROS accumulation | Facilitates paclitaxel sensitivity | [62] |
| Jolkinolide B | Euphorbia fischeriana Steud | Terpenoid | Bladder cancer | In vitro: UM-UC-3 and T24 cell lines (2.5 or 5 µM, 48 h) In vivo: mice (intraperitoneally injected, 10 mg/kg/day) | Inhibits autophagy and promotes cell apoptosis | Facilitates temsirolimus, rapamycin, and everolimus sensitivity | [63] |
| PC3-15 | Schisandra propinqua (Wall.) Baill. var. propinqua | Terpenoid | Breast cancer | In vitro: MDA-MB-468 and HEK293T cell lines (20 µM, 6 h) In vivo: mice (oral gavage, 50 mg/kg/day) | Facilitates lapatinib sensitivity | [64] | |
| Pristimerin | Celastraceae/ Hippocrateaceae | Terpenoid | Lung cancer (LC) | In vitro: A549 and NCI-H446 cell lines (0.25 µM, 24 h) In vivo: mice (0.8 mg/kg/day) | Inhibits autophagy and promotes cell apoptosis | Facilitates cisplatin sensitivity | [65] |
| Celastrol | Tripterygium wilfordii Hook F | Terpenoid | LC | A549, HCC-15, and Calu-3 cell lines (1–4 µM, 12 h) | Inhibits autophagy and promotes cell apoptosis | Facilitates TRAIL sensitivity | [66] |
| Icariin | Epimedium brevicornum Maxim. | Polyphenol | Breast cancer | MCF-7 and T47D cell lines (10–75 µM, 24 h) | Inhibits autophagy and promotes cell apoptosis | Facilitates tamoxifen sensitivity | [67] |
| Ovarian cancer | SKVCR cell line (10–75 µM, 24 h) | Inhibits autophagy and promotes cell apoptosis | Facilitates cisplatin sensitivity | [68] | |||
| Apigenin | Apiaceae | Polyphenol | Hepatocellular carcinoma (HCC) | In vitro: BEL-7402/ADM cell line In vivo: mice (intratumorally injected, 50 mg/kg/day) | Inhibits autophagy | Facilitates doxorubicin sensitivity | [69] |
| Tea polyphenol | Camellia sinensis | Polyphenol | Bladder cancer | T24 and BIU87 cell lines (100 µM, 24 h) | Inhibits autophagy and promotes cell apoptosis | Facilitates epirubicin sensitivity | [70] |
| Genistein | Soybeans and soy products | Polyphenol | LC | A549 cell line (40 µM, 12 h) | Inhibits autophagy and promotes cell apoptosis | Facilitates TRAIL sensitivity | [71] |
| Phloretin | Apples | Polyphenol | Breast cancer | In vitro: MCF7 and MDA-MB-231 cell lines (100–300 µM, 24 h) In vivo: mice (oral gavage, 100 mg/kg/day) | Inhibits autophagy | Facilitates TMX and DOX sensitivity | [72] |
| Formononetin | Astragalus membranaceus | Polyphenol | Breast cancer | In vitro: MDA-MB-231 cell line (15 µM) In vivo: mice (given orally, 30 mg/kg/3 day) | Inhibits autophagy and promotes cell apoptosis | Facilitates taxol sensitivity | [73] |
| Rutin | Potentilla discolor Bunge | Polyphenol | HCC | In vitro: HepG2 and HCCLM3 cell lines (75 µM, 24 h) In vivo: mice (intratumorally injected, 3 mg/kg/2 day) | Inhibits autophagy | Facilitates sorafenib sensitivity | [74] |
5.2. Natural Products as Promoters of Lethal Autophagy
| Compounds | Plant Origin | Classification | Cancer Types | Models and Dosage | Mechanism | Main Effects | References |
|---|---|---|---|---|---|---|---|
| Berberine | Coptis chinensis | Alkaloids | Glioblastoma | In vitro: U87 and U251 cell lines (10 µM, 24 h) In vivo: mice (intraperitoneally injected, 50 mg/kg/day) | Induces autophagy and promotes cell apoptosis | Facilitates temozolomide sensitivity | [86] |
| Non-small-cell lung cancer (NSCLC) | In vitro: H460 and H1299 cell lines (10, 25, 50 µM, 72 h) In vivo: mice (oral gavage, 80 mg/kg/day) | Induces autophagy and promotes cell apoptosis | Facilitates icotinib sensitivity | [87] | |||
| Matrine | Sophora flavescens | Alkaloids | Leukemia | K562/ADM cell line (0.5, 1, 2 mg/mL, 48 h) | Induces autophagy and promotes cell apoptosis | Facilitates vincristine and adriamycin sensitivity | [88] |
| Ursolic acid | Lamiaceae | Terpenoids | Pancreatic cancer (PC) | MIA Paca-2 cell line (50 μM, 24 h) | Induces autophagy and promotes cell apoptosis | Facilitates gemcitabine (GEM) sensitivity | [89] |
| Betulinic acid | Mirabilis jalapa | Terpenoids | Lung cancer (LC) | HCC827 and H1975 cell lines (20 µM, 48 h) | Induces autophagy and promotes cell apoptosis | Facilitates Iressa and Tarceva sensitivity | [90] |
| Triptolide | Tripterygium wilfordii | Terpenoids | PC | In vitro: MIA PaCa-2 and PANC-1 cell lines (50 nM, 48 h) | Induces autophagy and promotes cell apoptosis | Facilitates TNF-related apoptosis-inducing ligand (TRAIL) sensitivity | [91] |
| Ovarian cancer | In vitro: SKOV3/DDP cell line (100 nM, 12 h) In vivo: mice (intraperitoneally injected, 0.15 mg/kg/day) | Induces autophagy and ROS accumulation | Facilitates cisplatin sensitivity | [92] | |||
| Oleanolic acid | Oleaceae | Terpenoids | Cervical cancer | In vitro: HeLa cell line (30 µM, 24 h) In vivo: mice (oral gavage, 10 and 40 mg/kg/day) | Induces autophagy | Facilitates cisplatin sensitivity | [93] |
| AGE | Sanguisorba officinalis L. | Terpenoids | Colorectal cancer | RKO-R, HCT15-R, RKO-P, and HCT15-P cell lines (10 and 20 µM, 24 and 48 h) | Induces autophagy and promotes cell apoptosis | Facilitates 5-FU sensitivity | [94] |
| Demethylzeylasteral | Tripterygium wilfordii Hook F | Terpenoids | PC | In vitro: MIA PaCa-2 and PANC-1 cell lines (0.1–1 pg/mL, 24–72 h) In vivo: mice (oral gavage, 80, 160, and 200 mg/kg/day) | Induces autophagy and promotes cell apoptosis | Facilitates GEM sensitivity | [95] |
| Resveratrol | Veratrum gandiflorum | Polyphenols | Oral cancer | CAR cell line (50 µM, 48 h) | Induces autophagy and promotes cell apoptosis | Facilitates cisplatin sensitivity | [96] |
| Ovarian Cancer | SKOV3 and OVCAR3 cell lines (100 µM, 72 h) | Induces autophagy and promotes cell apoptosis | Facilitates platinum sensitivity | [97] | |||
| Pterostilbene | Resveratrol | Polyphenols | Pancreatic ductal adenocarcinoma | MIA PaCa-2 cell line (25, 50 µM, 72 h) | Induces autophagy and promotes cell apoptosis | Facilitates gemcitabine (GEM) sensitivity | [98] |
| Quercetin | Vegetables, fruits, and herbs | Polyphenols | PC | MIA Paca-2, BxPC-3, AsPC-1, HPAC, and PANC-1 cell lines (25–50 µM, 24 h) | Induces autophagy and promotes cell apoptosis | Facilitates GEM sensitivity | [99] |
| Hyperoside | Hypericum and Crataegus | Polyphenols | Ovarian cancer | SKOV-3 and HO-8910 cell lines (100 µM, 48 h) | Induces autophagy and promotes cell apoptosis | Facilitates cisplatin sensitivity | [100] |
| Scutellarin | Erigeron breviscapus Hand-Mazz | Polyphenols | NSCLC | In vitro: PC-9, H1975, and A549/DDP cell lines (120 µM, 24–48 h) In vivo: mice (oral gavage, 60 mg/kg/day) | Induces autophagy and promotes cell apoptosis | Facilitates cisplatin sensitivity | [101] |
| Chrysin | Passiflora caerulea | Polyphenols | PC | In vitro: ANC-1, Capan-2, BxPC-3 and AsPC-1 cell lines (50 µM, 24 h) In vivo: mice (oral gavage, 30 mg/kg/day) | Induces autophagy and promotes cell ferroptosis | Facilitates GEM sensitivity | [102] |
| Thymoquinone | Nigella sativa | Anthraquinones | Breast cancer | In vitro: MCF-7 and T47D cell lines (10–100 µM, 24–48 h) | Induces autophagy and promotes cell ferroptosis | Facilitates GEM sensitivity | [103] |
5.3. Natural Products with a Dual Role in Autophagy Regulation
| Compounds | Plant Origin | Classification | Cancer Types | Models and Dosage | Mechanism | Main Effects | References |
|---|---|---|---|---|---|---|---|
| Tetrandrine | Stephania tetrandra S. Moore | Alkaloids | Non-small-cell lung cancer (NSCLC) | A549/DDP cell line (0.25 µg/mL, 12 h) | Induces autophagy and promotes cell apoptosis | Facilitates cisplatin sensitivity | [130] |
| Pancreatic cancer | PANC-1 cell line (40 µg/mL, 24 h) | Induces autophagy and promotes cell apoptosis | Facilitates gemcitabine sensitivity | [131] | |||
| Breast cancer | TAM-R and MCF-7 cell lines (1.8 µg/mL, 24 h) | Inhibits autophagy and promotes cell apoptosis | Facilitates tamoxifen sensitivity | [132] | |||
| Lung cancer | PC14 cell line (3 µM, 72 h) | Inhibits autophagy | Facilitates gefitinib sensitivity | [133] | |||
| Lycorine | Amaryllidaceae | Alkaloids | Multiple myeloma | ANBL6, ARP-1, ARH-77, H929, and MM.1S cell lines (10 µM, 24 h) | Inhibits autophagy and promotes cell apoptosis | Facilitates bortezomib sensitivity | [134] |
| Hepatocellular carcinoma (HCC) | In vitro: PLC/PRF/5 and MHCC-97H cell lines (1, 2, 4 µmol/L) In vivo: mice (oral gavage, 10 mg/kg/day) | Induces autophagy and promotes cell apoptosis | Facilitates sorafenib sensitivity | [135] | |||
| Carnosic acid | Rosmarinus officinalis/Salvia officinalis | Terpenoids | Glioma | U251 and LN229 cell lines (10 µM, 24 h) | Induces autophagy and promotes cell apoptosis | Facilitates temozolomide sensitivity | [136] |
| Hepatocellular carcinoma | Huh7 and HCO2 cell lines (10 µM, 48 h) | Induces autophagy and promotes cell apoptosis | Facilitates sorafenib sensitivity | [137] | |||
| Breast cancer | SKBR-3, BT474, MCF7, and MDA-MB-231 cell lines (27.5, 37.5 µM, 48 h) | Inhibits autophagy and promotes cell apoptosis | Facilitates trastuzumab sensitivity | [138] | |||
| β-Elemene | Curcuma longa L. | Terpenoids | Colorectal cancer | In vitro: HCT116p53 +/+ and HCT116p53 –/– cell line (40 µg/mL, 24 h) In vivo: mice (intraperitoneally injected, 100 mg/kg/day) | Induces autophagy | Facilitates 5-FU sensitivity | [139] |
| NSCLC | In vitro: PC9GR and HCC827GR cell lines (120 µg/mL, 24 h) In vivo: mice (intraperitoneally injected, 100 mg/kg/day) | Inhibits autophagy and promotes cell apoptosis | Facilitates gefitinib sensitivity | [140] | |||
| Curcumin | Rosmarinus officinalis/Salvia officinalis | Polyphenols | NSCLC | A549, H460, H1299, and H1066 cell lines (10–30 µM, 48 h) | Inhibits autophagy and promotes cell apoptosis | Facilitates grizotinib sensitivity | [141] |
| NSCLC | In vitro: H157, H1299, and PC9 cell lines (5 µM, 48 h) In vivo: mice (oral gavage, 1 g/kg/day) | Induces autophagy and promotes cell apoptosis | Facilitates gefitinib sensitivity | [142] | |||
| Luteolin | Polyphenols | HCC | Huh7 and Hep3B cell lines (20 µM, 18 h) | Induces autophagy and promotes cell apoptosis | Facilitates TNF-related apoptosis-inducing ligand (TRAIL) sensitivity | [143] | |
| Ovarian cancer | Inhibits autophagy and promotes cell apoptosis | Facilitates cisplatin sensitivity | [138] | ||||
| Epigallocatechin gallate | Camellia sinensis | Polyphenols | Oral cancer | SKVCR cell line (10–75 µM, 24 h) | Induces autophagy and promotes cell apoptosis | Facilitates cisplatin sensitivity | [144] |
| NSCLC | In vitro: A549 cell line (34 µM, 48 h) In vivo: mice (oral gavage, 200 mg/kg/day) | Inhibits autophagy and promotes cell apoptosis | Facilitates gefitinib sensitivity | [145] |
6. Limitations
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lee, J.; Xie, C. Autophagy Regulation on Cancer Stem Cell Maintenance, Metastasis, and Therapy Resistance. Cancers 2022, 14, 381. [Google Scholar] [CrossRef]
- Klionsky, D.J.; Petroni, G.; Amaravadi, R.K.; Baehrecke, E.H.; Ballabio, A.; Boya, P.; Bravo-San Pedro, J.M.; Cadwell, K.; Cecconi, F.; Choi, A.; et al. Autophagy in major human diseases. EMBO J. 2021, 40, e108863. [Google Scholar] [CrossRef] [PubMed]
- Raudenska, M.; Balvan, J.; Masarik, M. Crosstalk between autophagy inhibitors and endosome-related secretory pathways: A challenge for autophagy-based treatment of solid cancers. Mol. Cancer 2021, 20, 140. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Ahmed, K.R.; Rahman, M.H.; Park, M.N.; Kim, B. Potential Therapeutic Action of Autophagy in Gastric Cancer Managements: Novel Treatment Strategies and Pharmacological Interventions. Front. Pharmacol. 2021, 12, 813703. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Castellanos, G.; Abdel Hadi, N.; Carrier, A. Autophagy Contributes to Metabolic Reprogramming and Therapeutic Resistance in Pancreatic Tumors. Cells 2022, 11, 426. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Gewirtz, D.A. Is Autophagy Always a Barrier to Cisplatin Therapy. Biomolecules 2022, 12, 463. [Google Scholar] [CrossRef]
- Mohsen, S.; Sobash, P.T.; Algwaiz, G.F.; Nasef, N.; Al-Zeidaneen, S.A.; Karim, N.A. Autophagy Agents in Clinical Trials for Cancer Therapy: A Brief Review. Curr. Oncol. 2022, 29, 1695–1708. [Google Scholar] [CrossRef]
- Ndlovu, T.; van Jaarsveld, F.; Caleb, O.J. French and Mediterranean-style diets: Contradictions, misconceptions and scientific facts-A review. Food Res. Int. 2019, 116, 840–858. [Google Scholar] [CrossRef] [PubMed]
- Augimeri, G.; Bonofiglio, D. The Mediterranean Diet as a Source of Natural Compounds: Does It Represent a Protective Choice against Cancer? Pharmaceuticals 2021, 14, 920. [Google Scholar] [CrossRef]
- Maruyama, K.; Khairunnisa, S.; Saito, I.; Tanigawa, T.; Tomooka, K.; Minato-Inokawa, S.; Sano, M.; Takakado, M.; Kawamura, R.; Takata, Y.; et al. Association of Fish and Omega-3 Fatty Acid Intake with Carotid Intima-Media Thickness in Middle-Aged to Elderly Japanese Men and Women: The Toon Health Study. Nutrients 2022, 14, 3644. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Chen, Y.; Tan, H.; Liu, B.; Zheng, L.L.; Mu, Y. Targeting Autophagy with Natural Compounds in Cancer: A Renewed Perspective from Molecular Mechanisms to Targeted Therapy. Front Pharmacol. 2021, 12, 748149. [Google Scholar] [CrossRef]
- Zhai, K.; Mazurakova, A.; Koklesova, L.; Kubatka, P.; Büsselberg, D. Flavonoids Synergistically Enhance the Anti-Glioblastoma Effects of Chemotherapeutic Drugs. Biomolecules 2021, 11, 1841. [Google Scholar] [CrossRef] [PubMed]
- Maleki Dana, P.; Sadoughi, F.; Asemi, Z.; Yousefi, B. The role of polyphenols in overcoming cancer drug resistance: A comprehensive review. Cell. Mol. Biol. Lett. 2022, 27, 1. [Google Scholar] [CrossRef] [PubMed]
- Denton, D.; Nicolson, S.; Kumar, S. Cell death by autophagy: Facts and apparent artefacts. Cell Death Differ. 2012, 19, 87–95. [Google Scholar] [CrossRef]
- Chang, H.; Zou, Z. Targeting autophagy to overcome drug resistance: Further developments. J. Hematol. Oncol. 2020, 13, 159. [Google Scholar] [CrossRef] [PubMed]
- Poillet-Perez, L.; Sarry, J.E.; Joffre, C. Autophagy is a major metabolic regulator involved in cancer therapy resistance. Cell Rep. 2021, 36, 109528. [Google Scholar] [CrossRef]
- Jiang, T.; Chen, X.; Ren, X.; Yang, J.M.; Cheng, Y. Emerging role of autophagy in anti-tumor immunity: Implications for the modulation of immunotherapy resistance. Drug Resist. Updat. 2021, 56, 100752. [Google Scholar] [CrossRef]
- Zhang, X.W.; Lv, X.X.; Zhou, J.C.; Jin, C.C.; Qiao, L.Y.; Hu, Z.W. Autophagic Flux Detection: Significance and Methods Involved. Adv. Exp. Med. Biol. 2021, 1208, 131–173. [Google Scholar]
- Peng, Y.; Wang, Y.; Zhou, C.; Mei, W.; Zeng, C. PI3K/Akt/mTOR Pathway and Its Role in Cancer Therapeutics: Are We Making Headway. Front Oncol. 2022, 12, 819128. [Google Scholar] [CrossRef]
- Rakesh, R.; PriyaDharshini, L.C.; Sakthivel, K.M.; Rasmi, R.R. Role and regulation of autophagy in cancer. Biochim. Biophys. Acta Mol. Basis Dis. 2022, 1868, 166400. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Park, M.N.; Rahman, M.H.; Rashid, M.M.; Islam, R.; Uddin, M.J.; Hannan, M.A.; Kim, B. p53 Modulation of Autophagy Signaling in Cancer Therapies: Perspectives Mechanism and Therapeutic Targets. Front. Cell Dev. Biol. 2022, 10, 761080. [Google Scholar] [CrossRef] [PubMed]
- Passaro, E.; Papulino, C.; Chianese, U.; Toraldo, A.; Congi, R.; Del Gaudio, N.; Nicoletti, M.M.; Benedetti, R.; Altucci, L. HDAC6 Inhibition Extinguishes Autophagy in Cancer: Recent Insights. Cancers 2021, 13, 6280. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Ge, P.; Wang, M.; Chen, H.; Liu, J.; Wei, T.; Jiang, Y.; Qu, J.; Chen, H. Research progress of DLX6-AS1 in human cancers. Hum. Cell 2021, 34, 1642–1652. [Google Scholar] [CrossRef]
- De la Cruz-Ojeda, P.; Flores-Campos, R.; Navarro-Villarán, E.; Muntané, J. The Role of Non-Coding RNAs in Autophagy During Carcinogenesis. Front. Cell Dev. Biol. 2022, 10, 799392. [Google Scholar] [CrossRef]
- Gao, W.; Wang, X.; Zhou, Y.; Wang, X.; Yu, Y. Autophagy, ferroptosis, pyroptosis, and necroptosis in tumor immunotherapy. Signal Transduct. Target Ther. 2022, 7, 196. [Google Scholar] [CrossRef] [PubMed]
- Patergnani, S.; Missiroli, S.; Morciano, G.; Perrone, M.; Mantovani, C.M.; Anania, G.; Fiorica, F.; Pinton, P.; Giorgi, C. Understanding the Role of Autophagy in Cancer Formation and Progression Is a Real Opportunity to Treat and Cure Human Cancers. Cancers 2021, 13, 5622. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Du, J.; Wu, X.; Abdelrehem, A.; Ren, Y.; Liu, C.; Zhou, X.; Wang, S. Crosstalk between autophagy and microbiota in cancer progression. Mol. Cancer 2021, 20, 163. [Google Scholar] [CrossRef]
- Ziegler, D.V.; Huber, K.; Fajas, L. The Intricate Interplay between Cell Cycle Regulators and Autophagy in Cancer. Cancers 2021, 14, 153. [Google Scholar] [CrossRef] [PubMed]
- Marcucci, F.; Rumio, C. Tumor Cell Glycolysis-At the Crossroad of Epithelial-Mesenchymal Transition and Autophagy. Cells 2022, 11, 1041. [Google Scholar] [CrossRef]
- Fan, Y.; Wang, Y.; Zhang, J.; Dong, X.; Gao, P.; Liu, K.; Ma, C.; Zhao, G. Breaking Bad: Autophagy Tweaks the Interplay Between Glioma and the Tumor Immune Microenvironment. Front. Immunol. 2021, 12, 746621. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gibson, S.B. Three dimensions of autophagy in regulating tumor growth: Cell survival/death, cell proliferation, and tumor dormancy. Biochim. Biophys. Acta Mol. Basis Dis. 2021, 1867, 166265. [Google Scholar] [CrossRef] [PubMed]
- Vitto, V.; Bianchin, S.; Zolondick, A.A.; Pellielo, G.; Rimessi, A.; Chianese, D.; Yang, H.; Carbone, M.; Pinton, P.; Giorgi, C.; et al. Molecular Mechanisms of Autophagy in Cancer Development, Progression, and Therapy. Biomedicines 2022, 10, 1596. [Google Scholar] [CrossRef]
- D’Orazi, G.; Cirone, M. Interconnected Adaptive Responses: A Way Out for Cancer Cells to Avoid Cellular Demise. Cancers 2022, 14, 2780. [Google Scholar] [CrossRef] [PubMed]
- Emran, T.B.; Shahriar, A.; Mahmud, A.R.; Rahman, T.; Abir, M.H.; Siddiquee, M.F.; Ahmed, H.; Rahman, N.; Nainu, F.; Wahyudin, E.; et al. Multidrug Resistance in Cancer: Understanding Molecular Mechanisms, Immunoprevention and Therapeutic Approaches. Front. Oncol. 2022, 12, 891652. [Google Scholar] [CrossRef] [PubMed]
- Hasan, A.; Rizvi, S.F.; Parveen, S.; Pathak, N.; Nazir, A.; Mir, S.S. Crosstalk Between ROS and Autophagy in Tumorigenesis: Understanding the Multifaceted Paradox. Front. Oncol. 2022, 12, 852424. [Google Scholar] [CrossRef] [PubMed]
- Cocco, S.; Leone, A.; Roca, M.S.; Lombardi, R.; Piezzo, M.; Caputo, R.; Ciardiello, C.; Costantini, S.; Bruzzese, F.; Sisalli, M.J.; et al. Inhibition of autophagy by chloroquine prevents resistance to PI3K/AKT inhibitors and potentiates their antitumor effect in combination with paclitaxel in triple negative breast cancer models. J. Transl. Med. 2022, 20, 290. [Google Scholar] [CrossRef]
- De Sanctis, J.B.; Charris, J.; Blanco, Z.; Ramírez, H.; Martínez, G.P.; Mijares, M.R. Molecular Mechanisms of Chloroquine and Hydroxychloroquine use in Cancer Therapy. Anticancer Agents Med. Chem. 2022, 20. [Google Scholar] [CrossRef]
- Aga, T.; Endo, K.; Tsuji, A.; Aga, M.; Moriyama-Kita, M.; Ueno, T.; Nakanishi, Y.; Hatano, M.; Kondo, S.; Sugimoto, H.; et al. Inhibition of autophagy by chloroquine makes chemotherapy in nasopharyngeal carcinoma more efficient. Auris Nasus Larynx 2019, 46, 443–450. [Google Scholar] [CrossRef]
- Wang, F.T.; Wang, H.; Wang, Q.W.; Pan, M.S.; Li, X.P.; Sun, W.; Fan, Y.Z. Inhibition of autophagy by chloroquine enhances the antitumor activity of gemcitabine for gallbladder cancer. Cancer Chemother. Pharmacol. 2020, 86, 221–232. [Google Scholar] [CrossRef]
- Zheng, S.; Shu, Y.; Lu, Y.; Sun, Y. Chloroquine Combined with Imatinib Overcomes Imatinib Resistance in Gastrointestinal Stromal Tumors by Inhibiting Autophagy via the MAPK/ERK Pathway. Onco. Targets Ther. 2020, 13, 6433–6441. [Google Scholar] [CrossRef]
- Lamprou, I.; Tsolou, A.; Kakouratos, C.; Mitrakas, A.G.; Xanthopoulou, E.T.; Kassela, K.; Karakasiliotis, I.; Zois, C.E.; Giatromanolaki, A.; Koukourakis, M.I. Suppressed PLIN3 frequently occurs in prostate cancer, promoting docetaxel resistance via intensified autophagy, an event reversed by chloroquine. Med. Oncol. 2021, 38, 116. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.H.; Ye, C.; Bai, E.H.; Zhang, L.L.; Huo, S.J.; Yu, H.H.; Xiang, S.Y.; Yu, S.Q. Co-delivery nanoparticles of doxorubicin and chloroquine for improving the anti-cancer effect in vitro. Nanotechnology 2019, 30, 085101. [Google Scholar] [CrossRef] [PubMed]
- Truong, A.; Yoo, J.H.; Scherzer, M.T.; Sanchez, J.; Dale, K.J.; Kinsey, C.G.; Richards, J.R.; Shin, D.; Ghazi, P.C.; Onken, M.D.; et al. Chloroquine Sensitizes GNAQ/11-mutated Melanoma to MEK1/2 Inhibition. Clin. Cancer Res. 2020, 26, 6374–6386. [Google Scholar] [CrossRef]
- Ovejero-Sánchez, M.; González-Sarmiento, R.; Herrero, A.B. Synergistic effect of Chloroquine and Panobinostat in ovarian cancer through induction of DNA damage and inhibition of DNA repair. Neoplasia 2021, 23, 515–528. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, M.R.; Ye, X.; Supko, J.G.; Desideri, S.; Grossman, S.A.; Brem, S.; Mikkelson, T.; Wang, D.; Chang, Y.C.; Hu, J.; et al. A phase I/II trial of hydroxychloroquine in conjunction with radiation therapy and concurrent and adjuvant temozolomide in patients with newly diagnosed glioblastoma multiforme. Autophagy 2014, 10, 1359–1368. [Google Scholar] [CrossRef]
- Anand, K.; Niravath, P.; Patel, T.; Ensor, J.; Rodriguez, A.; Boone, T.; Wong, S.T.; Chang, J.C. A Phase II Study of the Efficacy and Safety of Chloroquine in Combination with Taxanes in the Treatment of Patients With Advanced or Metastatic Anthracycline-refractory Breast Cancer. Clin. Breast Cancer 2021, 21, 199–204. [Google Scholar] [CrossRef]
- Samaras, P.; Tusup, M.; Nguyen-Kim, T.; Seifert, B.; Bachmann, H.; von Moos, R.; Knuth, A.; Pascolo, S. Phase I study of a chloroquine-gemcitabine combination in patients with metastatic or unresectable pancreatic cancer. Cancer Chemother. Pharmacol. 2017, 80, 1005–1012. [Google Scholar] [CrossRef]
- Arnaout, A.; Robertson, S.J.; Pond, G.R.; Lee, H.; Jeong, A.; Ianni, L.; Kroeger, L.; Hilton, J.; Coupland, S.; Gottlieb, C.; et al. A randomized, double-blind, window of opportunity trial evaluating the effects of chloroquine in breast cancer patients. Breast Cancer Res. Treat. 2019, 178, 327–335. [Google Scholar] [CrossRef]
- Mele, L.; Del Vecchio, V.; Liccardo, D.; Prisco, C.; Schwerdtfeger, M.; Robinson, N.; Desiderio, V.; Tirino, V.; Papaccio, G.; La Noce, M. The role of autophagy in resistance to targeted therapies. Cancer Treat. Rev. 2020, 88, 102043. [Google Scholar] [CrossRef]
- Shao, B.Z.; Chai, N.L.; Yao, Y.; Li, J.P.; Law, H.; Linghu, E.Q. Autophagy in gastrointestinal cancers. Front. Oncol. 2022, 12, 975758. [Google Scholar] [CrossRef]
- Finnegan, R.M.; Elshazly, A.M.; Schoenlein, P.V.; Gewirtz, D.A. Therapeutic Potential for Targeting Autophagy in ER+ Breast Cancer. Cancers 2022, 14, 4289. [Google Scholar] [CrossRef]
- Yang, M.; Lu, Y.; Piao, W.; Jin, H. The Translational Regulation in mTOR Pathway. Biomolecules 2022, 12, 802. [Google Scholar] [CrossRef] [PubMed]
- Hashem, S.; Ali, T.A.; Akhtar, S.; Nisar, S.; Sageena, G.; Ali, S.; Al-Mannai, S.; Therachiyil, L.; Mir, R.; Elfaki, I.; et al. Targeting cancer signaling pathways by natural products: Exploring promising anti-cancer agents. Biomed. Pharmacother. 2022, 150, 113054. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.; Bouyahya, A.; Hachlafi, N.; Menyiy, N.E.; Akram, M.; Sultana, S.; Zengin, G.; Ponomareva, L.; Shariati, M.A.; Ojo, O.A.; et al. Anticancer properties of medicinal plants and their bioactive compounds against breast cancer: A review on recent investigations. Environ. Sci. Pollut. Res. Int. 2022, 29, 24411–24444. [Google Scholar] [CrossRef]
- Kamran, S.; Sinniah, A.; Abdulghani, M.; Alshawsh, M.A. Therapeutic Potential of Certain Terpenoids as Anticancer Agents: A Scoping Review. Cancers 2022, 14, 1100. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Zhang, L.; Hou, X. Potential roles and molecular mechanisms of phytochemicals against cancer. Food Funct. 2022, 13, 9208–9225. [Google Scholar] [CrossRef] [PubMed]
- Malik, Z.; Parveen, R.; Parveen, B.; Zahiruddin, S.; Aasif Khan, M.; Khan, A.; Massey, S.; Ahmad, S.; Husain, S.A. Anticancer potential of andrographolide from Andrographis paniculata (Burm.f.) Nees and its mechanisms of action. J. Ethnopharmacol. 2021, 272, 113936. [Google Scholar] [CrossRef]
- Mi, S.; Xiang, G.; Yuwen, D.; Gao, J.; Guo, W.; Wu, X.; Wu, X.; Sun, Y.; Su, Y.; Shen, Y.; et al. Inhibition of autophagy by andrographolide resensitizes cisplatin-resistant non-small cell lung carcinoma cells via activation of the Akt/mTOR pathway. Toxicol. Appl. Pharmacol. 2016, 310, 78–86. [Google Scholar] [CrossRef]
- Yuwen, D.; Mi, S.; Ma, Y.; Guo, W.; Xu, Q.; Shen, Y.; Shu, Y. Andrographolide enhances cisplatin-mediated anticancer effects in lung cancer cells through blockade of autophagy. Anticancer Drugs 2017, 28, 967–976. [Google Scholar] [CrossRef]
- Zhang, M.; Xue, E.; Shao, W. Andrographolide promotes vincristine-induced SK-NEP-1 tumor cell death via PI3K-AKT-p53 signaling pathway. Drug Des. Devel. Ther. 2016, 10, 3143–3152. [Google Scholar] [PubMed]
- Zhan, Y.; Wang, K.; Li, Q.; Zou, Y.; Chen, B.; Gong, Q.; Ho, H.I.; Yin, T.; Zhang, F.; Lu, Y.; et al. The Novel Autophagy Inhibitor Alpha-Hederin Promoted Paclitaxel Cytotoxicity by Increasing Reactive Oxygen Species Accumulation in Non-Small Cell Lung Cancer Cells. Int. J. Mol. Sci. 2018, 19, 3221. [Google Scholar] [CrossRef] [PubMed]
- Sang, J.; Gan, L.; Zou, M.F.; Lin, Z.J.; Fan, R.Z.; Huang, J.L.; Li, W.; Tang, G.H.; Yin, S. Jolkinolide B sensitizes bladder cancer to mTOR inhibitors via dual inhibition of Akt signaling and autophagy. Cancer Lett. 2022, 526, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Zhou, Y.; Duan, D.; Yang, C.; Zhou, Z.; Li, F.; Kong, Y.; Hsieh, Y.C.; Zhang, R.; Ding, W.; et al. Targeting ubiquitin conjugating enzyme UbcH5b by a triterpenoid PC3-15 from Schisandra plants sensitizes triple-negative breast cancer cells to lapatinib. Cancer Lett. 2021, 504, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, J.; Hui, B.; Sun, W.; Li, B.; Shi, F.; Che, S.; Chai, L.; Song, L. Pristimerin enhances the effect of cisplatin by inhibiting the miR-23a/Akt/GSK3β signaling pathway and suppressing autophagy in lung cancer cells. Int. J. Mol. Med. 2019, 43, 1382–1394. [Google Scholar] [CrossRef] [PubMed]
- Nazim, U.M.; Yin, H.; Park, S.Y. Autophagy flux inhibition mediated by celastrol sensitized lung cancer cells to TRAIL-induced apoptosis via regulation of mitochondrial transmembrane potential and reactive oxygen species. Mol. Med. Rep. 2019, 19, 984–993. [Google Scholar] [CrossRef]
- Cheng, X.; Tan, S.; Duan, F.; Yuan, Q.; Li, Q.; Deng, G. Icariin induces apoptosis by suppressing autophagy in tamoxifen-resistant breast cancer cell line MCF-7/TAM. Breast Cancer 2019, 26, 766–775. [Google Scholar] [CrossRef]
- Jiang, S.; Chang, H.; Deng, S.; Fan, D. Icariin enhances the chemosensitivity of cisplatin-resistant ovarian cancer cells by suppressing autophagy via activation of the AKT/mTOR/ATG5 pathway. Int. J. Oncol. 2019, 54, 1933–1942. [Google Scholar] [CrossRef]
- Gao, A.M.; Zhang, X.Y.; Hu, J.N.; Ke, Z.P. Apigenin sensitizes hepatocellular carcinoma cells to doxorubic through regulating miR-520b/ATG7 axis. Chem. Biol. Interact. 2018, 280, 45–50. [Google Scholar] [CrossRef]
- Gu, W.; Lin, Y.; Gou, X.; He, W. Tea Polyphenol inhibits autophagy to sensitize Epirubicin-induced apoptosis in human bladder cancer cells. Neoplasma 2017, 64, 674–680. [Google Scholar] [CrossRef]
- Nazim, U.M.; Park, S.Y. Genistein enhances TRAIL-induced cancer cell death via inactivation of autophagic flux. Oncol. Rep. 2015, 34, 2692–2698. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Gowd, V.; Wang, M.; Chen, F.; Cheng, K.W. The apple dihydrochalcone phloretin suppresses growth and improves chemosensitivity of breast cancer cells via inhibition of cytoprotective autophagy. Food Funct. 2021, 12, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Zhang, S.; Chen, F.; Hu, J.; Yuan, S.; Li, C.; Wang, X.; Zhang, W.; Tang, R. Formononetin ameliorates the drug resistance of Taxol resistant triple negative breast cancer by inhibiting autophagy. Am. J. Transl. Res. 2021, 13, 497–514. [Google Scholar] [PubMed]
- Zhou, M.; Zhang, G.; Hu, J.; Zhu, Y.; Lan, H.; Shen, X.; Lv, Y.; Huang, L. Rutin attenuates Sorafenib-induced Chemoresistance and Autophagy in Hepatocellular Carcinoma by regulating BANCR/miRNA-590-5P/OLR1 Axis. Int. J. Biol. Sci. 2021, 17, 3595–3607. [Google Scholar] [CrossRef] [PubMed]
- Chu, A.J. Quarter-Century Explorations of Bioactive Polyphenols: Diverse Health Benefits. Front. Biosci. (Landmark Ed.) 2022, 27, 134. [Google Scholar] [CrossRef]
- Szabó, R.; Rácz, C.P.; Dulf, F.V. Bioavailability Improvement Strategies for Icariin and Its Derivates: A Review. Int. J. Mol. Sci. 2022, 23, 7519. [Google Scholar] [CrossRef]
- Bai, R.; Li, Y.; Jian, L.; Yang, Y.; Zhao, L.; Wei, M. The hypoxia-driven crosstalk between tumor and tumor-associated macrophages: Mechanisms and clinical treatment strategies. Mol. Cancer 2022, 21, 177. [Google Scholar] [CrossRef]
- Bao, M.H.; Wong, C.C. Hypoxia, Metabolic Reprogramming, and Drug Resistance in Liver Cancer. Cells 2021, 10, 1715. [Google Scholar] [CrossRef]
- Sharma, A.; Sinha, S.; Shrivastava, N. Therapeutic Targeting Hypoxia-Inducible Factor (HIF-1) in Cancer: Cutting Gordian Knot of Cancer Cell Metabolism. Front Genet. 2022, 13, 849040. [Google Scholar] [CrossRef]
- Zhou, Y.; Yu, Y.; Lv, H.; Zhang, H.; Liang, T.; Zhou, G.; Huang, L.; Tian, Y.; Liang, W. Apigenin in cancer therapy: From mechanism of action to nano-therapeutic agent. Food Chem. Toxicol. 2022, 168, 113385. [Google Scholar] [CrossRef]
- Kim, T.W.; Lee, H.G. Apigenin Induces Autophagy and Cell Death by Targeting EZH2 under Hypoxia Conditions in Gastric Cancer Cells. Int. J. Mol. Sci. 2021, 22, 13455. [Google Scholar] [CrossRef] [PubMed]
- Islam, F.; Mitra, S.; Emran, T.B.; Khan, Z.; Nath, N.; Das, R.; Sharma, R.; Awadh, A.; Park, M.N.; Kim, B. Natural Small Molecules in Gastrointestinal Tract and Associated Cancers: Molecular Insights and Targeted Therapies. Molecules 2022, 27, 5686. [Google Scholar] [CrossRef] [PubMed]
- Talib, W.H.; Awajan, D.; Hamed, R.A.; Azzam, A.O.; Mahmod, A.I.; Al-Yasari, I.H. Combination Anticancer Therapies Using Selected Phytochemicals. Molecules 2022, 27, 5452. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Qiao, H.; Chen, S.; Chen, X.; Dou, K.; Wei, L.; Zhang, J. Berberine reverses lapatinib resistance of HER2-positive breast cancer cells by increasing the level of ROS. Cancer Biol. Ther. 2016, 17, 925–934. [Google Scholar] [CrossRef]
- Mohammadinejad, R.; Ahmadi, Z.; Tavakol, S.; Ashrafizadeh, M. Berberine as a potential autophagy modulator. J. Cell. Physiol. 2019, 9, 14914–14926. [Google Scholar] [CrossRef]
- Qu, H.; Song, X.; Song, Z.; Jiang, X.; Gao, X.; Bai, L.; Wu, J.; Na, L.; Yao, Z. Berberine reduces temozolomide resistance by inducing autophagy via the ERK1/2 signaling pathway in glioblastoma. Cancer Cell Int. 2020, 20, 592. [Google Scholar] [CrossRef]
- Chen, P.; Dai, C.H.; Shi, Z.H.; Wang, Y.; Wu, J.N.; Chen, K.; Su, J.Y.; Li, J. Synergistic inhibitory effect of berberine and icotinib on non-small cell lung cancer cells via inducing autophagic cell death and apoptosis. Apoptosis 2021, 26, 639–656. [Google Scholar] [CrossRef]
- Li, Z.; Wang, N.; Yue, T.; Liu, L. Matrine reverses the drug resistance of K562/ADM cells to ADM and VCR via promoting autophagy. Transl. Cancer Res. 2020, 9, 786–794. [Google Scholar] [CrossRef]
- Lin, J.H.; Chen, S.Y.; Lu, C.C.; Lin, J.A.; Yen, G.C. Ursolic acid promotes apoptosis, autophagy, and chemosensitivity in gemcitabine-resistant human pancreatic cancer cells. Phytother Res. 2020, 34, 2053–2066. [Google Scholar] [CrossRef]
- Ko, J.L.; Lin, C.H.; Chen, H.C.; Hung, W.H.; Chien, P.J.; Chang, H.Y.; Wang, B.Y. Effects and mechanisms of betulinic acid on improving EGFR TKI-resistance of lung cancer cells. Environ. Toxicol. 2018, 33, 1153–1159. [Google Scholar] [CrossRef]
- Dai, H.; Jiang, Y.; Luo, Y.; Bie, P.; Chen, Z. Triptolide enhances TRAIL sensitivity of pancreatic cancer cells by activating autophagy via downregulation of PUM1. Phytomedicine 2019, 62, 152953. [Google Scholar] [CrossRef]
- Zhong, Y.; Le, F.; Cheng, J.; Luo, C.; Zhang, X.; Wu, X.; Xu, F.; Zuo, Q.; Tan, B. Triptolide inhibits JAK2/STAT3 signaling and induces lethal autophagy through ROS generation in cisplatin-resistant SKOV3/DDP ovarian cancer cells. Oncol. Rep. 2021, 45, 69. [Google Scholar] [CrossRef] [PubMed]
- Potočnjak, I.; Šimić, L.; Vukelić, I.; Domitrović, R. Oleanolic acid attenuates cisplatin-induced nephrotoxicity in mice and chemosensitizes human cervical cancer cells to cisplatin cytotoxicity. Food Chem. Toxicol. 2019, 132, 110676. [Google Scholar]
- Zhang, W.; Peng, C.; Shen, X.; Yuan, Y.; Zhang, W.; Yang, C.; Yao, M. A Bioactive Compound from Sanguisorba officinalis L. Inhibits Cell Proliferation and Induces Cell Death in 5-Fluorouracil-Sensitive/Resistant Colorectal Cancer Cells. Molecules 2021, 26, 3843. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Tian, X.; Zhang, Z.; Ma, Y.; Xie, X.; Liang, J.; Yang, C.; Yang, Y. Demethylzeylasteral (ZST93) inhibits cell growth and enhances cell chemosensitivity to gemcitabine in human pancreatic cancer cells via apoptotic and autophagic pathways. Int. J. Cancer 2018, 142, 1938–1951. [Google Scholar] [CrossRef]
- Chang, C.H.; Lee, C.Y.; Lu, C.C.; Tsai, F.J.; Hsu, Y.M.; Tsao, J.W.; Juan, Y.N.; Chiu, H.Y.; Yang, J.S.; Wang, C.C. Resveratrol-induced autophagy and apoptosis in cisplatin-resistant human oral cancer CAR cells: A key role of AMPK and Akt/mTOR signaling. Int. J. Oncol. 2017, 50, 873–882. [Google Scholar] [CrossRef]
- Ferraresi, A.; Esposito, A.; Girone, C.; Vallino, L.; Salwa, A.; Ghezzi, I.; Thongchot, S.; Vidoni, C.; Dhanasekaran, D.N.; Isidoro, C. Resveratrol Contrasts LPA-Induced Ovarian Cancer Cell Migration and Platinum Resistance by Rescuing Hedgehog-Mediated Autophagy. Cells 2021, 10, 3213. [Google Scholar] [CrossRef]
- Hsu, Y.H.; Chen, S.Y.; Wang, S.Y.; Lin, J.A.; Yen, G.C. Pterostilbene Enhances Cytotoxicity and Chemosensitivity in Human Pancreatic Cancer Cells. Biomolecules 2020, 10, 709. [Google Scholar] [CrossRef]
- Lan, C.Y.; Chen, S.Y.; Kuo, C.W.; Lu, C.C.; Yen, G.C. Quercetin facilitates cell death and chemosensitivity through RAGE/PI3K/AKT/mTOR axis in human pancreatic cancer cells. J. Food Drug. Anal. 2019, 27, 887–896. [Google Scholar] [CrossRef]
- Zhu, X.; Ji, M.; Han, Y.; Guo, Y.; Zhu, W.; Gao, F.; Yang, X.; Zhang, C. PGRMC1-dependent autophagy by hyperoside induces apoptosis and sensitizes ovarian cancer cells to cisplatin treatment. Int. J. Oncol. 2017, 50, 835–846. [Google Scholar] [CrossRef]
- Sun, C.Y.; Zhu, Y.; Li, X.F.; Wang, X.Q.; Tang, L.P.; Su, Z.Q.; Li, C.Y.; Zheng, G.J.; Feng, B. Scutellarin Increases Cisplatin-Induced Apoptosis and Autophagy to Overcome Cisplatin Resistance in Non-small Cell Lung Cancer via ERK/p53 and c-met/AKT Signaling Pathways. Front Pharmacol. 2018, 9, 92. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Yang, C.; Zhong, W.; Wang, Q.; Zhang, D.; Zhang, J.; Xie, S.; Xu, M. Chrysin induces autophagy-dependent ferroptosis to increase chemosensitivity to gemcitabine by targeting CBR1 in pancreatic cancer cells. Biochem. Pharmacol. 2021, 193, 114813. [Google Scholar] [CrossRef] [PubMed]
- Bashmail, H.A.; Alamoudi, A.A.; Noorwali, A.; Hegazy, G.A.; AJabnoor, G.; Choudhry, H.; Al-Abd, A.M. Thymoquinone synergizes gemcitabine anti-breast cancer activity via modulating its apoptotic and autophagic activities. Sci. Rep. 2018, 8, 11674. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Dong, H.; Li, T.; Wang, N.; Wei, X.; Wu, H.; Liu, Y.; Wang, W.; Guo, Z.; Xiao, X. The Synergistic Reducing Drug Resistance Effect of Cisplatin and Ursolic Acid on Osteosarcoma through a Multistep Mechanism Involving Ferritinophagy. Oxid. Med. Cell Longev. 2021, 2021, 5192271. [Google Scholar] [CrossRef]
- Wang, H.; Wang, H.; Ge, L.; Zhao, Y.; Zhu, K.; Chen, Z.; Wu, Q.; Xin, Y.; Guo, J. Betulinic acid targets drug-resistant human gastric cancer cells by inducing autophagic cell death, suppresses cell migration and invasion, and modulates the ERK/MEK signaling pathway. Acta Biochim. Pol. 2021, 69, 25–30. [Google Scholar] [CrossRef]
- Sun, C.Y.; Cao, D.; Ren, Q.N.; Zhang, S.S.; Zhou, N.N.; Mai, S.J.; Feng, B.; Wang, H.Y. Combination Treatment With Inhibitors of ERK and Autophagy Enhances Antitumor Activity of Betulinic Acid in Non-small-Cell Lung Cancer In Vivo and In Vitro. Front Pharmacol. 2021, 12, 684243. [Google Scholar] [CrossRef]
- Salehi, B.; Albayrak, S.; Antolak, H.; Kręgiel, D.; Pawlikowska, E.; Sharifi-Rad, M.; Uprety, Y.; Tsouh Fokou, P.V.; Yousef, Z.; Amiruddin Zakaria, Z.; et al. Aloe Genus Plants: From Farm to Food Applications and Phytopharmacotherapy. Int. J. Mol. Sci. 2018, 19, 2843. [Google Scholar] [CrossRef]
- Şeker Karatoprak, G.; Küpeli Akkol, E.; Yücel, Ç.; Bahadır, A.Ö.; Sobarzo-Sánchez, E. Advances in Understanding the Role of Aloe Emodin and Targeted Drug Delivery Systems in Cancer. Oxid. Med. Cell Longev. 2022, 2022, 7928200. [Google Scholar] [CrossRef]
- Cheng, G.; Pi, Z.; Zhuang, X.; Zheng, Z.; Liu, S.; Liu, Z.; Song, F. The effects and mechanisms of aloe-emodin on reversing adriamycin-induced resistance of MCF-7/ADR cells. Phytother Res. 2021, 35, 3886–3897. [Google Scholar] [CrossRef]
- Zhang, C.; He, X.J.; Li, L.; Lu, C.; Lu, A.P. Effect of the Natural Product Triptolide on Pancreatic Cancer: A Systematic Review of Preclinical Studies. Front Pharmacol. 2017, 8, 490. [Google Scholar] [CrossRef]
- Gao, J.; Zhang, Y.; Liu, X.; Wu, X.; Huang, L.; Gao, W. Triptolide: Pharmacological spectrum, biosynthesis, chemical synthesis and derivatives. Theranostics 2021, 11, 7199–7221. [Google Scholar] [CrossRef] [PubMed]
- Quiroz-Reyes, A.G.; Delgado-Gonzalez, P.; Islas, J.F.; Gallegos, J.; Martínez Garza, J.H.; Garza-Treviño, E.N. Behind the Adaptive and Resistance Mechanisms of Cancer Stem Cells to TRAIL. Pharmaceutics 2021, 13, 1062. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Li, F.; Zhang, L.; Liu, W.; Wang, X.; Zhu, R.; Qiao, Z.A.; Yu, B.; Yu, X. TRAIL-modified, doxorubicin-embedded periodic mesoporous organosilica nanoparticles for targeted drug delivery and efficient antitumor immunotherapy. Acta Biomater. 2022, 143, 392–405. [Google Scholar] [CrossRef] [PubMed]
- Gor, R.; Sampath, S.S.; Lazer, L.M.; Ramalingam, S. RNA binding protein PUM1 promotes colon cancer cell proliferation and migration. Int. J. Biol. Macromol. 2021, 174, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.Y.; Lim, S.C.; Lee, T.B.; Han, S.I. Molecular Basis of Resveratrol-Induced Resensitization of Acquired Drug-Resistant Cancer Cells. Nutrients 2022, 14, 699. [Google Scholar] [CrossRef]
- Siddamurthi, S.; Gutti, G.; Jana, S.; Kumar, A.; Singh, S.K. Anthraquinone: A promising scaffold for the discovery and development of therapeutic agents in cancer therapy. Future Med. Chem. 2020, 12, 1037–1069. [Google Scholar] [CrossRef]
- Yang, X.; Zhai, D.; Song, J.; Qing, R.; Wang, B.; Ji, J.; Chen, X.; Hao, S. Rhein-PEG-nHA conjugate as a bone targeted drug delivery vehicle for enhanced cancer chemoradiotherapy. Nanomedicine (Lond.) 2020, 27, 102196. [Google Scholar] [CrossRef]
- Sun, J.; Xu, Z.; Hou, Y.; Yao, W.; Fan, X.; Zheng, H.; Piao, J.; Li, F.; Wei, Y. Hierarchically structured microcapsules for oral delivery of emodin and tanshinone IIA to treat renal fibrosis. Int. J. Pharm. 2022, 616, 121490. [Google Scholar] [CrossRef]
- Cheng, W.; Xiang, W.; Wang, S.; Xu, K. Tanshinone IIA ameliorates oxaliplatin-induced neurotoxicity via mitochondrial protection and autophagy promotion. Am. J. Transl. Res. 2019, 11, 3140–3149. [Google Scholar]
- Wang, X.; Li, C.; Wang, Q.; Li, W.; Guo, D.; Zhang, X.; Shao, M.; Chen, X.; Ma, L.; Zhang, Q.; et al. Tanshinone IIA Restores Dynamic Balance of Autophagosome/Autolysosome in Doxorubicin-Induced Cardiotoxicity via Targeting Beclin1/LAMP1. Cancers 2019, 11, 910. [Google Scholar] [CrossRef]
- Xu, Z.; Chen, L.; Xiao, Z.; Zhu, Y.; Jiang, H.; Jin, Y.; Gu, C.; Wu, Y.; Wang, L.; Zhang, W.; et al. Potentiation of the anticancer effect of doxorubicinin drug-resistant gastric cancer cells by tanshinone IIA. Phytomedicine 2018, 51, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Jehan, S.; Huang, J.; Farooq, U.; Basheer, I.; Zhou, W. Combinatorial effect of thymoquinone with chemo agents for tumor therapy. Phytomedicine 2022, 98, 153936. [Google Scholar] [CrossRef] [PubMed]
- Llaguno-Munive, M.; Vazquez-Lopez, M.I.; Jurado, R.; Garcia-Lopez, P. Mifepristone Repurposing in Treatment of High-Grade Gliomas. Front Oncol. 2021, 11, 606907. [Google Scholar] [CrossRef] [PubMed]
- Saha, P.; Kumar, S.; Datta, K.; Tyagi, R.K. Upsurge in autophagy, associated with mifepristone-treated polycystic ovarian condition, is reversed upon thymoquinone treatment. J. Steroid Biochem. Mol. Biol. 2021, 208, 105823. [Google Scholar]
- Bhagya, N.; Chandrashekar, K.R. Autophagy and cancer: Can tetrandrine be a potent anticancer drug in the near future. Biomed. Pharmacother. 2022, 148, 112727. [Google Scholar] [CrossRef]
- Gong, K.; Chen, C.; Zhan, Y.; Chen, Y.; Huang, Z.; Li, W. Autophagy-related gene 7 (ATG7) and reactive oxygen species/extracellular signal-regulated kinase regulate tetrandrine-induced autophagy in human hepatocellular carcinoma. J. Biol. Chem. 2012, 287, 35576–35588. [Google Scholar] [CrossRef]
- Wang, H.; Liu, T.; Li, L.; Wang, Q.; Yu, C.; Liu, X.; Li, W. Tetrandrine is a potent cell autophagy agonist via activated intracellular reactive oxygen species. Cell Biosci. 2015, 5, 4. [Google Scholar] [CrossRef]
- Mei, L.; Chen, Y.; Wang, Z.; Wang, J.; Wan, J.; Yu, C.; Liu, X.; Li, W. Synergistic anti-tumour effects of tetrandrine and chloroquine combination therapy in human cancer: A potential antagonistic role for p21. Br. J. Pharmacol. 2015, 172, 2232–2245. [Google Scholar] [CrossRef]
- Wong, V.; Zeng, W.; Chen, J.; Yao, X.J.; Leung, E.; Wang, Q.Q.; Chiu, P.; Ko, B.; Law, B. Tetrandrine, an Activator of Autophagy, Induces Autophagic Cell Death via PKC-α Inhibition and mTOR-Dependent Mechanisms. Front Pharmacol. 2017, 8, 351. [Google Scholar] [CrossRef]
- Ye, L.Y.; Hu, S.; Xu, H.E.; Xu, R.R.; Kong, H.; Zeng, X.N.; Xie, W.P.; Wang, H. The effect of tetrandrine combined with cisplatin on proliferation and apoptosis of A549/DDP cells and A549 cells. Cancer Cell Int. 2017, 17, 40. [Google Scholar] [CrossRef]
- Song, J.; Xu, J.; Guo, J.; Shang, Y.; Wang, J.; Wang, T. The enhancement of Tetrandrine to gemcitabine-resistant PANC-1 cytochemical sensitivity involves the promotion of PI3K/Akt/mTOR-mediated apoptosis and AMPK-regulated autophagy. Acta. Histochem. 2021, 123, 151769. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yue, W.; Lang, H.; Ding, X.; Chen, X.; Chen, H. Resuming Sensitivity of Tamoxifen-Resistant Breast Cancer Cells to Tamoxifen by Tetrandrine. Integr. Cancer Ther. 2021, 20, 1534735421996822. [Google Scholar] [CrossRef] [PubMed]
- Sato, E.; Ohta, S.; Kawakami, K.; Ikeda, M.; Takahashi, T.; Kobayashi, S.; Nomura, M. Tetrandrine Increases the Sensitivity of Human Lung Adenocarcinoma PC14 Cells to Gefitinib by Lysosomal Inhibition. Anticancer Res. 2019, 39, 6585–6593. [Google Scholar] [CrossRef] [PubMed]
- Roy, M.; Liang, L.; Xiao, X.; Peng, Y.; Luo, Y.; Zhou, W.; Zhang, J.; Qiu, L.; Zhang, S.; Liu, F.; et al. Lycorine Downregulates HMGB1 to Inhibit Autophagy and Enhances Bortezomib Activity in Multiple Myeloma. Theranostics 2016, 6, 2209–2224. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Yao, Y.; Rao, Y.; Huang, X.; Wei, L.; You, Z.; Zheng, G.; Hou, X.; Su, Y.; Varghese, Z.; et al. Cholesterol sensor SCAP contributes to sorafenib resistance by regulating autophagy in hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 2022, 41, 116. [Google Scholar] [CrossRef]
- Shao, N.; Mao, J.; Xue, L.; Wang, R.; Zhi, F.; Lan, Q. Carnosic acid potentiates the anticancer effect of temozolomide by inducing apoptosis and autophagy in glioma. J. Neurooncol. 2019, 141, 277–288. [Google Scholar] [CrossRef]
- Wu, Q.; Wang, X.; Pham, K.; Luna, A.; Studzinski, G.P.; Liu, C. Enhancement of sorafenib-mediated death of Hepatocellular carcinoma cells by Carnosic acid and Vitamin D2 analog combination. J. Steroid Biochem. Mol. Biol. 2020, 197, 105524. [Google Scholar]
- D’Alesio, C.; Bellese, G.; Gagliani, M.C.; Aiello, C.; Grasselli, E.; Marcocci, G.; Bisio, A.; Tavella, S.; Daniele, T.; Cortese, K.; et al. Cooperative antitumor activities of carnosic acid and Trastuzumab in ERBB2+ breast cancer cells. J. Exp. Clin. Cancer Res. 2017, 36, 154. [Google Scholar]
- Zhang, R.; Pan, T.; Xiang, Y.; Zhang, M.; Feng, J.; Liu, S.; Duan, T.; Chen, P.; Zhai, B.; Chen, X.; et al. β-Elemene Reverses the Resistance of p53-Deficient Colorectal Cancer Cells to 5-Fluorouracil by Inducing Pro-death Autophagy and Cyclin D3-Dependent Cycle Arrest. Front. Bioeng. Biotechnol. 2020, 8, 378. [Google Scholar] [CrossRef]
- Liu, S.; Li, Q.; Li, G.; Zhang, Q.; Zhuo, L.; Han, X.; Zhang, M.; Chen, X.; Pan, T.; Yan, L.; et al. The mechanism of m6A methyltransferase METTL3-mediated autophagy in reversing gefitinib resistance in NSCLC cells by β-elemene. Cell Death Dis. 2020, 11, 969. [Google Scholar] [CrossRef]
- He, Y.Z.; Yu, S.L.; Li, X.N.; Bai, X.H.; Li, H.T.; Liu, Y.C.; Lv, B.L.; Zhao, X.M.; Wei, D.; Zhang, H.L.; et al. Curcumin increases crizotinib sensitivity through the inactivation of autophagy via epigenetic modulation of the miR-142-5p/Ulk1 axis in non-small cell lung cancer. Cancer Biomark. 2021, 34, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Huang, H.P.; Wang, Y.; Jin, J.; Long, W.G.; Chen, K.; Zhao, X.H.; Chen, C.G.; Li, J. Curcumin overcome primary gefitinib resistance in non-small-cell lung cancer cells through inducing autophagy-related cell death. J. Exp. Clin. Cancer Res. 2019, 38, 254. [Google Scholar] [CrossRef] [PubMed]
- Nazim, U.M.; Park, S.Y. Luteolin sensitizes human liver cancer cells to TRAIL-induced apoptosis via autophagy and JNK-mediated death receptor 5 upregulation. Int. J. Oncol. 2019, 54, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Yuan, C.H.; Horng, C.T.; Lee, C.F.; Chiang, N.N.; Tsai, F.J.; Lu, C.C.; Chiang, J.H.; Hsu, Y.M.; Yang, J.S.; Chen, F.A. Epigallocatechin gallate sensitizes cisplatin-resistant oral cancer CAR cell apoptosis and autophagy through stimulating AKT/STAT3 pathway and suppressing multidrug resistance 1 signaling. Environ. Toxicol. 2017, 32, 845–855. [Google Scholar] [CrossRef]
- Meng, J.; Chang, C.; Chen, Y.; Bi, F.; Ji, C.; Liu, W. EGCG overcomes gefitinib resistance by inhibiting autophagy and augmenting cell death through targeting ERK phosphorylation in NSCLC. Onco. Targets Ther. 2019, 12, 6033–6043. [Google Scholar] [CrossRef]
- Yin, S.; Yang, S.; Luo, Y.; Lu, J.; Hu, G.; Wang, K.; Shao, Y.; Zhou, S.; Koo, S.; Qiu, Y.; et al. Cyclin-dependent kinase 1 as a potential target for lycorine against hepatocellular carcinoma. Biochem. Pharmacol. 2021, 193, 114806. [Google Scholar] [CrossRef]
- Hu, H.; Yang, W.; Liang, Z.; Zhou, Z.; Song, Q.; Liu, W.; Deng, X.; Zhu, J.; Xing, X.; Zhong, B.; et al. Amplification of oxidative stress with lycorine and gold-based nanocomposites for synergistic cascade cancer therapy. J. Nanobiotechnology 2021, 19, 221. [Google Scholar] [CrossRef]
- Jia, X.; Zhang, Z.; Luo, K.; Zheng, G.; Lu, M.; Song, Y.; Liu, H.; Qiu, H.; He, Z. TCRP1 transcriptionally regulated by c-Myc confers cancer chemoresistance in tongue and lung cancer. Sci. Rep. 2017, 7, 3744. [Google Scholar] [CrossRef]
- Zhao, S.; Li, X.; Yin, L.; Hou, L.; Lan, J.; Zhu, X. TCRP1 induces tamoxifen resistance by promoting the activation of SGK1 in MCF-7 cells. Oncol. Rep. 2020, 43, 2017–2027. [Google Scholar] [CrossRef]
- Gu, Y.; Fan, S.; Liu, B.; Zheng, G.; Yu, Y.; Ouyang, Y.; He, Z. TCRP1 promotes radioresistance of oral squamous cell carcinoma cells via Akt signal pathway. Mol. Cell. Biochem. 2011, 357, 107–113. [Google Scholar] [CrossRef]
- Yu, H.; Qiu, Y.; Pang, X.; Li, J.; Wu, S.; Yin, S.; Han, L.; Zhang, Y.; Jin, C.; Gao, X.; et al. Lycorine Promotes Autophagy and Apoptosis via TCRP1/Akt/mTOR Axis Inactivation in Human Hepatocellular Carcinoma. Mol. Cancer Ther. 2017, 16, 2711–2723. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.H.; Zhang, P.; Yu, T.T.; Huang, H.K.; Zhang, L.L.; Yang, C.M.; Tan, T.; Yang, S.D.; Luo, X.J.; Luo, J.Y. Lycorine inhibits tumor growth of human osteosarcoma cells by blocking Wnt/β-catenin, ERK1/2/MAPK and PI3K/AKT signaling pathway. Am. J. Transl. Res. 2020, 12, 5381–5398. [Google Scholar] [PubMed]
- Shakeri, A.; Cicero, A.; Panahi, Y.; Mohajeri, M.; Sahebkar, A. Curcumin: A naturally occurring autophagy modulator. J. Cell. Physiol. 2019, 234, 5643–5654. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.L.; Zhan, T.C.; Hu, J.P.; Zhang, C.L.; Zhu, K.P. Doxorubicin-induced novel circRNA_0004674 facilitates osteosarcoma progression and chemoresistance by upregulating MCL1 through miR-142-5p. Cell Death Discov. 2021, 7, 309. [Google Scholar] [CrossRef] [PubMed]
- Monti, E.; Marras, E.; Prini, P.; Gariboldi, M.B. Luteolin impairs hypoxia adaptation and progression in human breast and colon cancer cells. Eur. J. Pharmacol. 2020, 881, 173210. [Google Scholar] [CrossRef]
- Liu, Q.; Zhu, D.; Hao, B.; Zhang, Z.; Tian, Y. Luteolin promotes the sensitivity of cisplatin in ovarian cancer by decreasing PRPA1-medicated autophagy. Cell. Mol. Biol. (Noisy-le-Grand) 2018, 64, 17–22. [Google Scholar] [CrossRef]
- Song, L.; Xiong, P.; Zhang, W.; Hu, H.; Tang, S.; Jia, B.; Huang, W. Mechanism of Citri Reticulatae Pericarpium as an Anticancer Agent from the Perspective of Flavonoids: A Review. Molecules 2022, 27, 5622. [Google Scholar] [CrossRef]
- Wu, J.; Zhou, T.; Wang, Y.; Jiang, Y.; Wang, Y. Mechanisms and Advances in Anti-Ovarian Cancer with Natural Plants Component. Molecules 2021, 26, 5949. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, L. Autophagy is a double-edged sword in the therapy of colorectal cancer. Oncol. Lett. 2021, 21, 378. [Google Scholar] [CrossRef]
- Xu, Z.; Han, X.; Ou, D.; Liu, T.; Li, Z.; Jiang, G.; Liu, J.; Zhang, J. Targeting PI3K/AKT/mTOR-mediated autophagy for tumor therapy. Appl. Microbiol. Biotechnol. 2020, 104, 575–587. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, J.; Ma, C.; Feng, K.; Tan, G.; Wen, Q. Focusing on the Role of Natural Products in Overcoming Cancer Drug Resistance: An Autophagy-Based Perspective. Biomolecules 2022, 12, 1565. https://doi.org/10.3390/biom12111565
Yao J, Ma C, Feng K, Tan G, Wen Q. Focusing on the Role of Natural Products in Overcoming Cancer Drug Resistance: An Autophagy-Based Perspective. Biomolecules. 2022; 12(11):1565. https://doi.org/10.3390/biom12111565
Chicago/Turabian StyleYao, Jiaqi, Chi Ma, Kaixuan Feng, Guang Tan, and Qingping Wen. 2022. "Focusing on the Role of Natural Products in Overcoming Cancer Drug Resistance: An Autophagy-Based Perspective" Biomolecules 12, no. 11: 1565. https://doi.org/10.3390/biom12111565
APA StyleYao, J., Ma, C., Feng, K., Tan, G., & Wen, Q. (2022). Focusing on the Role of Natural Products in Overcoming Cancer Drug Resistance: An Autophagy-Based Perspective. Biomolecules, 12(11), 1565. https://doi.org/10.3390/biom12111565






