On the Role of Paraoxonase-1 and Chemokine Ligand 2 (C-C motif) in Metabolic Alterations Linked to Inflammation and Disease. A 2021 Update
Abstract
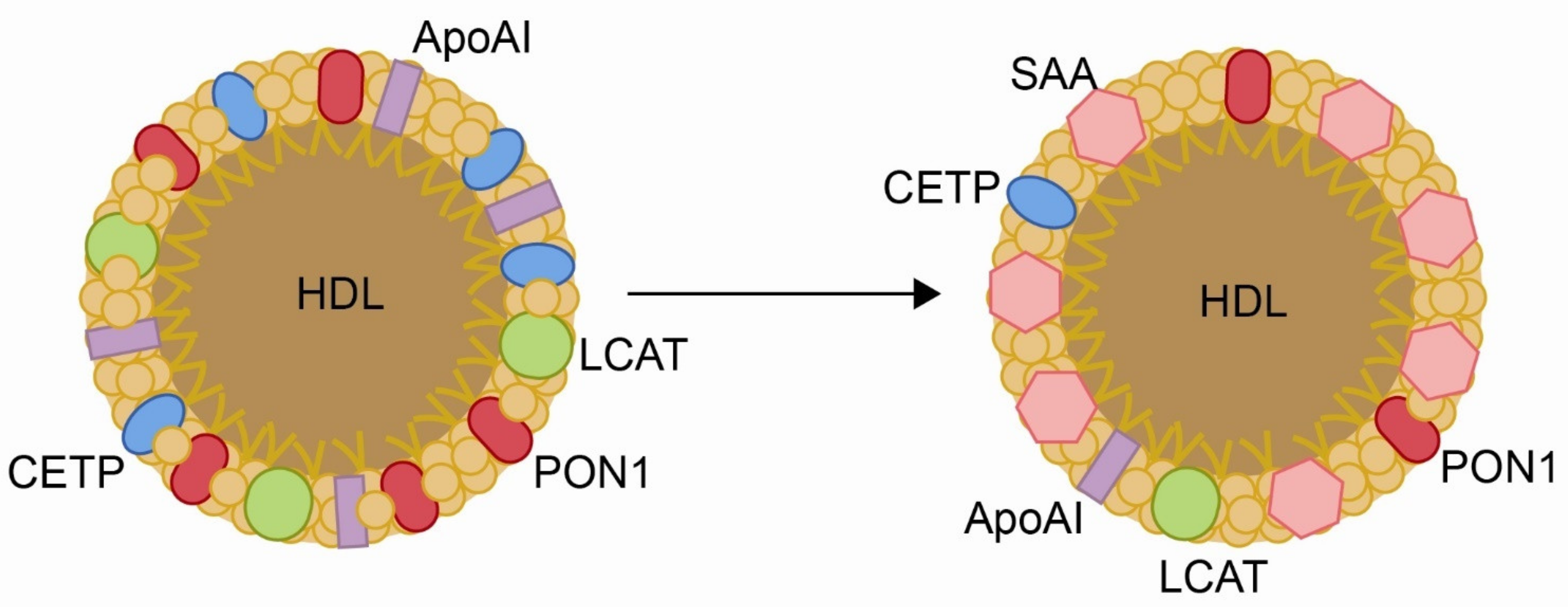
1. Oxidation, Inflammation and Disease
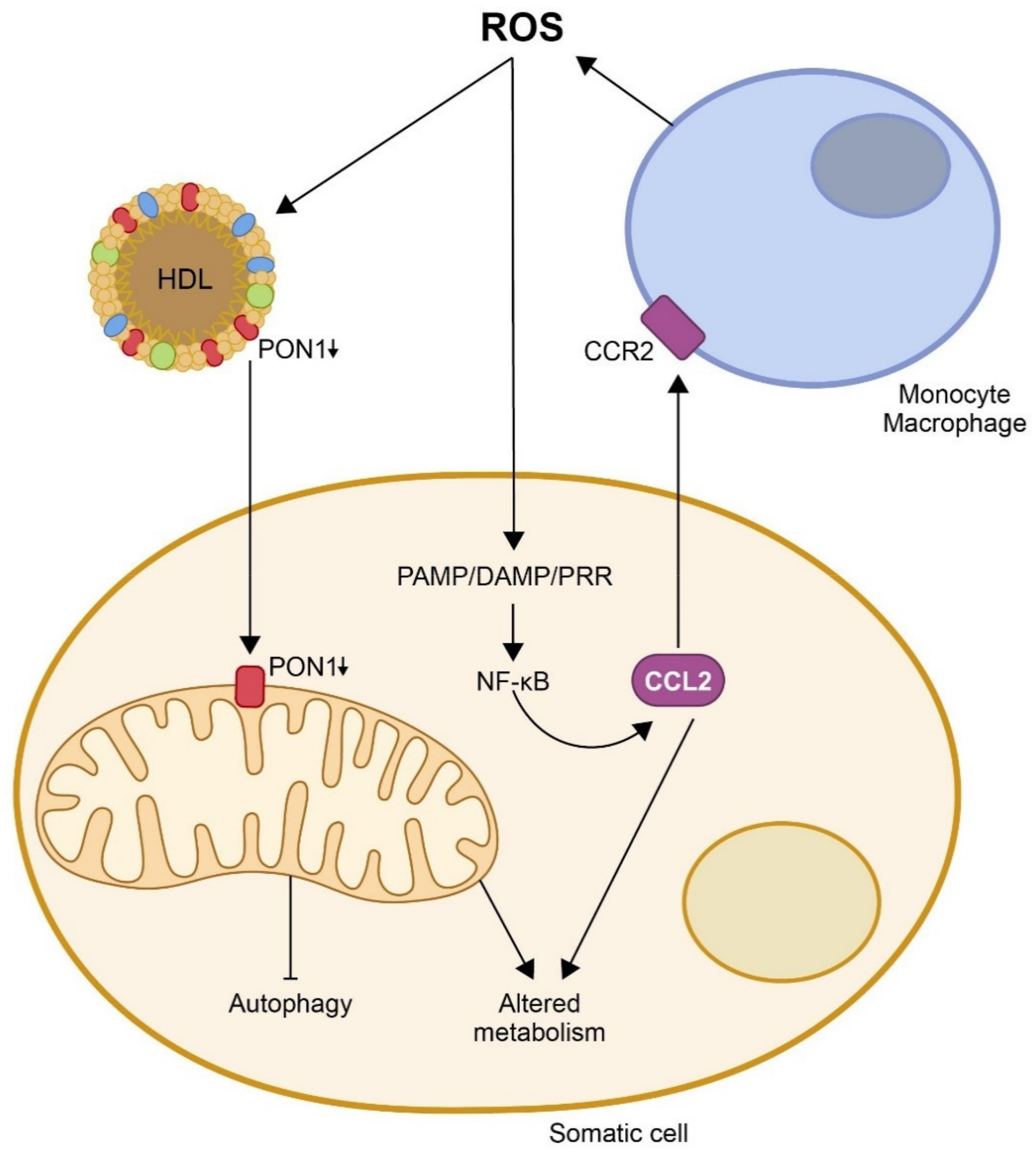
2. Paraoxonase-1 Is an Antioxidant Enzyme That Participates in the Innate Immune System
3. The Protective Role of Paraoxonases on CCL2 Expression, Mitochondrial Function, and Metabolism
4. Mechanism of Action of CCL2 in the Immune Response and Inflammation and Its Relationship with Multiple Metabolic Alterations
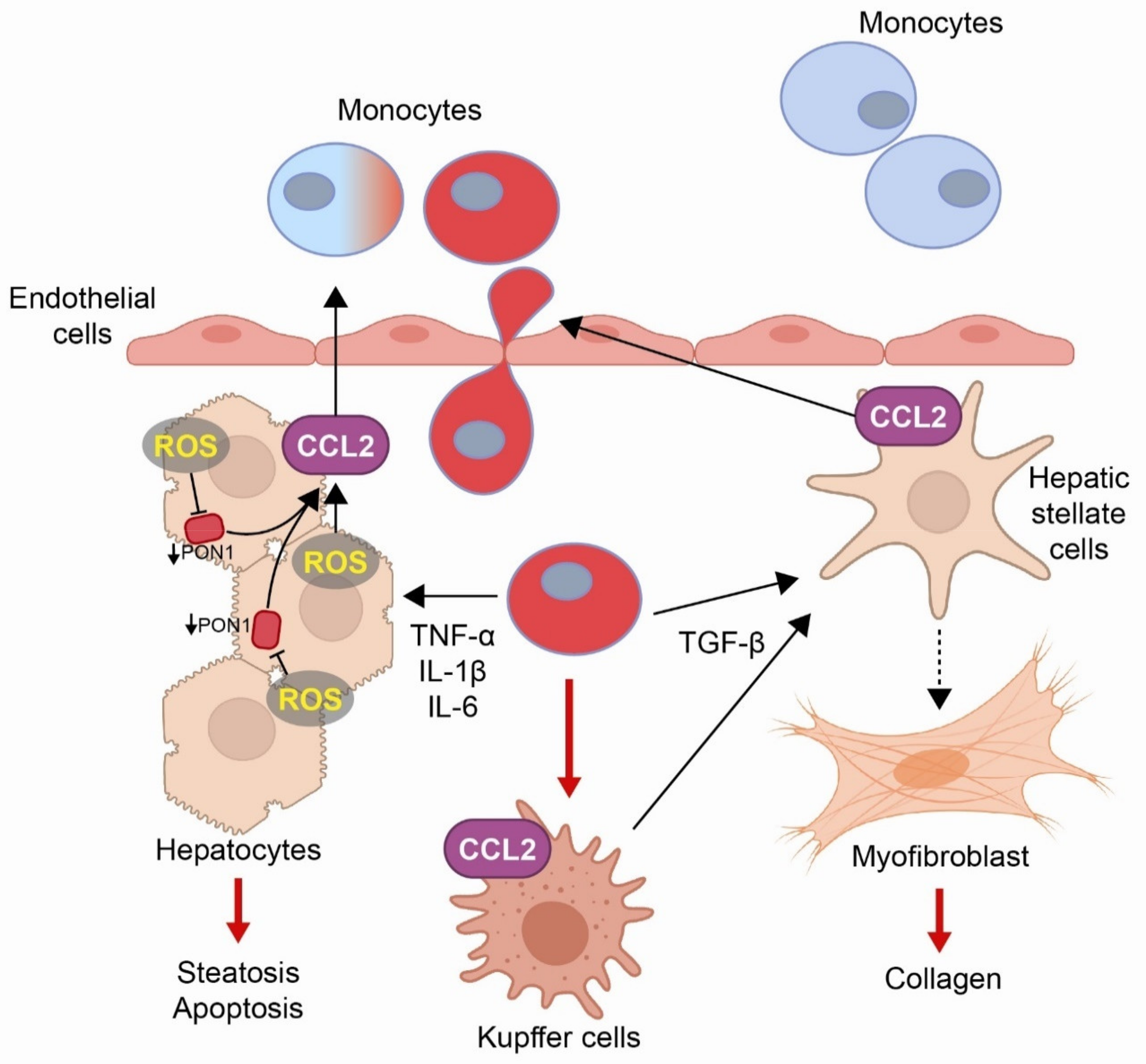
5. Obesity and the Associated Liver Disease
6. Cardiovascular Diseases
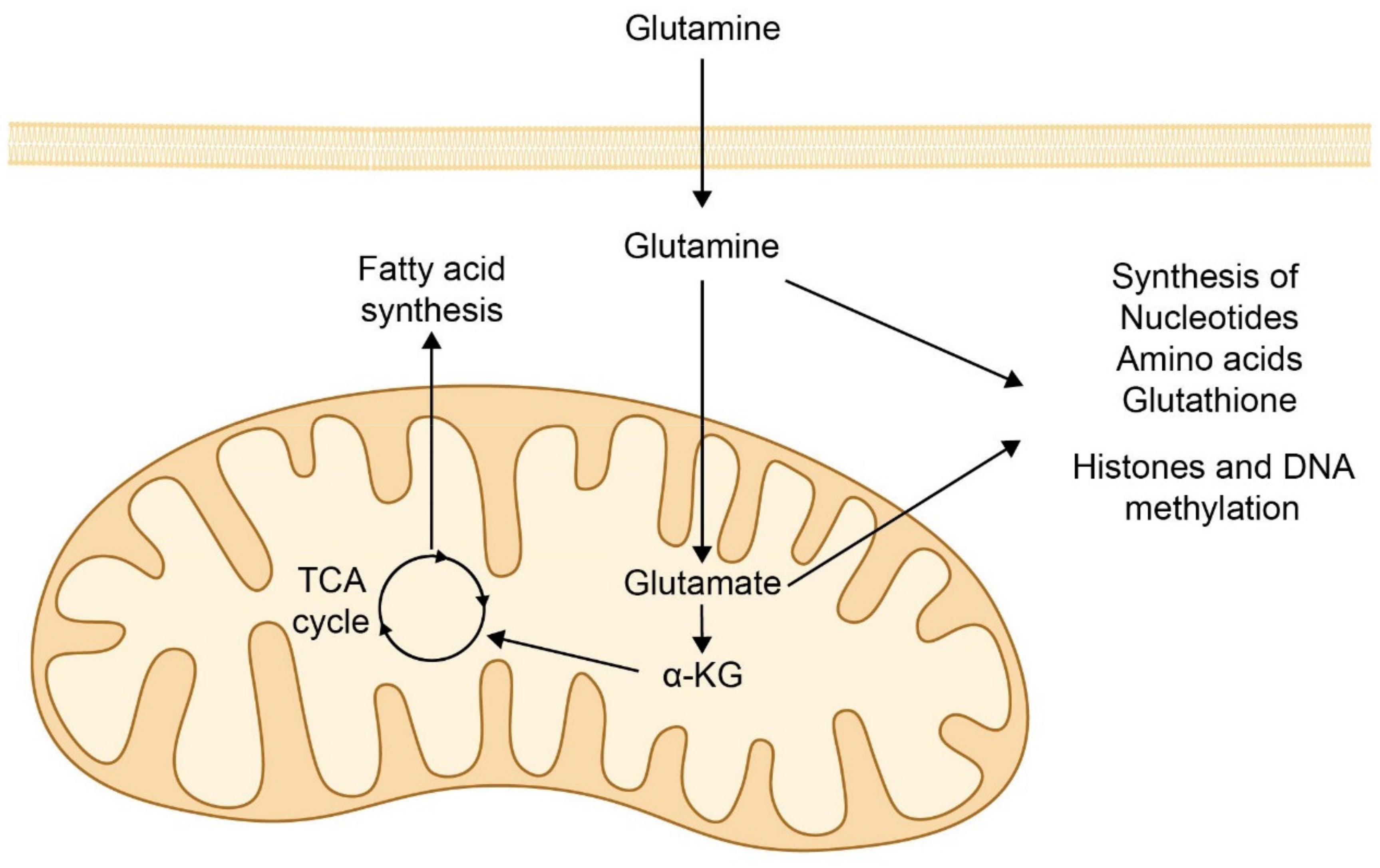
7. Cancer
8. Infectious Diseases
9. Final Remarks
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative stress: Harms and benefits for human health. Oxid. Med. Cell. Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef] [PubMed]
- Masud, S.; Torraca, V.; Meijer, A.H. Modeling infectious diseases in the context of a developing immune system. Curr. Top. Dev. Biol. 2017, 124, 277–329. [Google Scholar] [CrossRef]
- He, J.; Liu, J.; Huang, Y.; Tang, X.; Xiao, H.; Hu, Z. Oxidative Stress, inflammation, and autophagy: Potential targets of mesenchymal stem cells-based therapies in ischemic stroke. Front. Neurosci. 2021, 15, 641157. [Google Scholar] [CrossRef] [PubMed]
- Kibel, A.; Lukinac, A.M.; Dambic, V.; Juric, I.; Relatic, K.S. Oxidative stress in ischemic heart disease. Oxid. Med. Cell. Longev. 2020, 2020, 6627144. [Google Scholar] [CrossRef]
- Atri, C.; Guerfali, F.Z.; Laouini, D. Role of human macrophage polarization in inflammation during infectious diseases. Int. J. Mol. Sci. 2018, 19, 1801. [Google Scholar] [CrossRef]
- Parisi, L.; Gini, E.; Baci, D.; Tremolati, M.; Fanuli, M.; Bassani, B.; Farronato, G.; Bruno, A.; Mortara, L. Macrophage polarization in chronic inflammatory diseases: Killers or builders? J. Immunol. Res. 2018, 2018, 8917804. [Google Scholar] [CrossRef]
- Poltavets, A.S.; Vishnyakova, P.A.; Elchaninov, A.V.; Sukhikh, G.T.; Fatkhudinov, T.K. Macrophage modification strategies for efficient cell therapy. Cells 2020, 9, 1535. [Google Scholar] [CrossRef] [PubMed]
- Viola, A.; Munari, F.; Sánchez-Rodríguez, R.; Scolaro, T.; Castegna, A. The metabolic signature of macrophage responses. Front. Immunol. 2019, 10, 1462. [Google Scholar] [CrossRef]
- Turner, M.D.; Nedjai, B.; Hurst, T.; Pennington, D.J. Cytokines and chemokines: At the crossroads of cell signalling and inflammatory disease. Biochim. Biophys. Acta 2014, 1843, 2563–2582. [Google Scholar] [CrossRef]
- Gschwandtner, M.; Derler, R.; Midwood, K.S. More than just attractive: How CCL2 influences myeloid cell behavior beyond chemotaxis. Front. Immunol. 2019, 10, 2759. [Google Scholar] [CrossRef]
- Huang, R.; Guo, L.; Gao, M.; Li, J.; Xiang, S. Research trends and regulation of CCL5 in prostate cancer. Onco. Targets Ther. 2021, 14, 1417–1427. [Google Scholar] [CrossRef]
- Agresti, N.; Lalezari, J.P.; Amodeo, P.P.; Mody, K.; Mosher, S.F.; Seethamraju, H.; Kelly, S.A.; Pourhassan, N.Z.; Sudduth, C.D.; Bovinet, C.; et al. Disruption of CCR5 signaling to treat COVID-19-associated cytokine storm: Case series of four critically ill patients treated with leronlimab. J. Transl. Autoimmun. 2021, 4, 100083. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Matosevic, S. Chemokine networks modulating natural killer cell trafficking to solid tumors. Cytokine Growth Factor Rev. 2021, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Necula, D.; Riviere-Cazaux, C.; Shen, Y.; Zhou, M. Insight into the roles of CCR5 in learning and memory in normal and disordered states. Brain Behav. Immun. 2021, 92, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Taler-Verčič, A.; Goličnik, M.; Bavec, A. The structure and function of paraoxonase-1 and its comparison to paraoxonase-2 and -3. Molecules 2020, 25, 5980. [Google Scholar] [CrossRef]
- Levy, D.; Reichert, C.O.; Bydlowski, S.P. Paraoxonases activities and polymorphisms in elderly and old-age diseases: An overview. Antioxidants 2019, 8, 118. [Google Scholar] [CrossRef] [PubMed]
- Camps, J.; Marsillach, J.; Joven, J. The paraoxonases: Role in human diseases and methodological difficulties in measurement. Crit. Rev. Clin. Lab. Sci. 2009, 46, 83–106. [Google Scholar] [CrossRef]
- Costa, L.G.; Cole, T.B.; Vitalone, A.; Furlong, C.E. Measurement of paraoxonase (PON1) status as a potential biomarker of susceptibility to organophosphate toxicity. Clin. Chim. Acta 2005, 352, 37–47. [Google Scholar] [CrossRef]
- Draganov, D.I.; Teiber, J.F.; Speelman, A.; Osawa, Y.; Sunahara, R.; La Du, B.N. Human paraoxonases (PON1, PON2, and PON3) are lactonases with overlapping and distinct substrate specificities. J. Lipid Res. 2005, 46, 1239–1247. [Google Scholar] [CrossRef]
- Jaouad, L.; de Guise, C.; Berrougui, H.; Cloutier, M.; Isabelle, M.; Fulop, T.; Payette, H.; Khalil, A. Age-related decrease in high-density lipoproteins antioxidant activity is due to an alteration in the PON1′s free sulfhydryl groups. Atherosclerosis 2006, 185, 191–200. [Google Scholar] [CrossRef]
- Leviev, I.; Negro, F.; James, R.W. Two alleles of the human paraoxonase gene produce different amounts of mRNA. An explanation for differences in serum concentrations of paraoxonase associated with the (Leu-Met54) polymorphism. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 2935–2939. [Google Scholar] [CrossRef] [PubMed]
- Sierksma, A.; van der Gaag, M.S.; van Tol, A.; James, R.W.; Hendriks, H.F. Kinetics of HDL cholesterol and paraoxonase activity in moderate alcohol consumers. Alcohol. Clin. Exp. Res. 2002, 26, 1430–1435. [Google Scholar] [CrossRef]
- Deakin, S.P.; Bioletto, S.; Bochaton-Piallat, M.L.; James, R.W. HDL-associated paraoxonase-1 can redistribute to cell membranes and influence sensitivity to oxidative stress. Free Radic. Biol. Med. 2011, 50, 102–109. [Google Scholar] [CrossRef]
- Marsillach, J.; Camps, J.; Beltran-Debón, R.; Rull, A.; Aragones, G.; Maestre-Martínez, C.; Sabench, F.; Hernández, M.; Castillo, D.D.; Joven, J.; et al. Immunohistochemical analysis of paraoxonases-1 and 3 in human atheromatous plaques. Eur. J. Clin. Investig. 2011, 41, 308–314. [Google Scholar] [CrossRef]
- Marsillach, J.; Mackness, B.; Mackness, M.; Riu, F.; Beltrán, R.; Joven, J.; Camps, J. Immunohistochemical analysis of paraoxonases-1, 2, and 3 expression in normal mouse tissues. Free Radic. Biol. Med. 2008, 45, 146–157. [Google Scholar] [CrossRef]
- Rodríguez-Sanabria, F.; Rull, A.; Beltrán-Debón, R.; Aragonès, G.; Camps, J.; Mackness, B.; Mackness, M.; Joven, J. Tissue distribution and expression of paraoxonases and chemokines in mouse: The ubiquitous and joint localisation suggest a systemic and coordinated role. J. Mol. Histol. 2010, 41, 379–386. [Google Scholar] [CrossRef]
- Manco, G.; Porzio, E.; Carusone, T.M. Human paraoxonase-2 (PON2): Protein functions and modulation. Antioxidants 2021, 10, 256. [Google Scholar] [CrossRef] [PubMed]
- Mackness, B.; Hine, D.; Liu, Y.; Mastorikou, M.; Mackness, M. Paraoxonase-1 inhibits oxidised LDL-induced MCP-1 production by endothelial cells. Biochem. Biophys. Res. Commun. 2004, 318, 680–683. [Google Scholar] [CrossRef] [PubMed]
- Camps, J.; Rodríguez-Gallego, E.; García-Heredia, A.; Triguero, I.; Riera-Borrull, M.; Hernández-Aguilera, A.; Luciano-Mateo, F.; Fernández-Arroyo, S.; Joven, J. Paraoxonases and chemokine (C-C motif) ligand-2 in noncommunicable diseases. Adv. Clin. Chem. 2014, 63, 247–308. [Google Scholar] [CrossRef]
- Watson, A.D.; Berliner, J.A.; Hama, S.Y.; La Du, B.N.; Faull, K.F.; Fogelman, A.M.; Navab, M. Protective effect of high density lipoprotein associated paraoxonase. Inhibition of the biological activity of minimally oxidized low density lipoprotein. J. Clin. Investig. 1995, 96, 2882–2891. [Google Scholar] [CrossRef] [PubMed]
- Shih, D.M.; Gu, L.; Xia, Y.R.; Navab, M.; Li, W.F.; Hama, S.; Castellani, L.W.; Furlong, C.E.; Costa, L.G.; Fogelman, A.M.; et al. Mice lacking serum paraoxonase are susceptible to organophosphate toxicity and atherosclerosis. Nature 1998, 394, 284–287. [Google Scholar] [CrossRef]
- Rozenberg, O.; Rosenblat, M.; Coleman, R.; Shih, D.M.; Aviram, M. Paraoxonase (PON1) deficiency is associated with increased macrophage oxidative stress: Studies in PON1-knockout mice. Free Radic. Biol. Med. 2003, 34, 774–784. [Google Scholar] [CrossRef]
- Mackness, M.; Mackness, B. Human paraoxonase-1 (PON1): Gene structure and expression, promiscuous activities and multiple physiological roles. Gene 2015, 567, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Singh, S.K.; Chowdhury, I.; Singh, R. Understanding the mechanism of bacterial biofilms resistance to antimicrobial agents. Open Microbiol. J. 2017, 11, 53–62. [Google Scholar] [CrossRef]
- Gebreyohannes, G.; Nyerere, A.; Bii, C.; Sbhatu, D.B. Challenges of intervention, treatment, and antibiotic resistance of biofilm-forming microorganisms. Heliyon 2019, 5, e02192. [Google Scholar] [CrossRef]
- Balaure, P.C.; Grumezescu, A.M. Recent advances in surface nanoengineering for biofilm prevention and control. Part I: Molecular basis of biofilm recalcitrance. Passive anti-biofouling nanocoatings. Nanomaterials 2020, 10, 1230. [Google Scholar] [CrossRef]
- Camps, J.; Iftimie, S.; García-Heredia, A.; Castro, A.; Joven, J. Paraoxonases and infectious diseases. Clin. Biochem. 2017, 50, 804–811. [Google Scholar] [CrossRef]
- Coquant, G.; Grill, J.P.; Seksik, P. Impact of N-acyl-homoserine lactones, quorum sensing molecules, on gut immunity. Front. Immunol. 2020, 11, 1827. [Google Scholar] [CrossRef] [PubMed]
- Billot, R.; Plener, L.; Jacquet, P.; Elias, M.; Chabrière, E.; Daudé, D. Engineering acyl-homoserine lactone-interfering enzymes toward bacterial control. J. Biol. Chem. 2020, 295, 12993–13007. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Singh, P.; Sarmah, B.K.; Nandi, S.P. Quorum sensing: Its role in microbial social networking. Res. Microbiol. 2020, 171, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Chun, C.K.; Ozer, E.A.; Welsh, M.J.; Zabner, J.; Greenberg, E.P. Inactivation of a Pseudomonas aeruginosa quorum-sensing signal by human airway epithelia. Proc. Natl. Acad. Sci. USA 2004, 101, 3587–3590. [Google Scholar] [CrossRef] [PubMed]
- Stoltz, D.A.; Ozer, E.A.; Zabner, J. Paraoxonases, quorum sensing, and Pseudomonas aeruginosa. In The Paraoxonases: Their Role in Disease Development and Xenobiotic Metabolism; Mackness, B., Mackness, M., Aviram, M., Paragh, G., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 307–319. [Google Scholar]
- Chen, F.; Gao, Y.; Chen, X.; Yu, Z.; Li, X. Quorum quenching enzymes and their application in degrading signal molecules to block quorum sensing-dependent infection. Int. J. Mol. Sci. 2013, 14, 17477–17500. [Google Scholar] [CrossRef]
- Ozer, E.A.; Pezzulo, A.; Shih, D.M.; Chun, C.; Furlong, C.; Lusis, A.J.; Greenberg, E.P.; Zabner, J. Human and murine paraoxonase 1 are host modulators of Pseudomonas aeruginosa quorum-sensing. FEMS Microbiol. Lett. 2005, 253, 29–37. [Google Scholar] [CrossRef]
- Richter, R.J.; Jarvik, G.P.; Furlong, C.E. Paraoxonase 1 status as a risk factor for disease or exposure. Adv. Exp. Med. Biol. 2010, 660, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Horke, S.; Witte, I.; Wilgenbus, P.; Krüger, M.; Strand, D.; Förstermann, U. Paraoxonase-2 reduces oxidative stress in vascular cells and decreases endoplasmic reticulum stress-induced caspase activation. Circulation 2007, 115, 2055–2064. [Google Scholar] [CrossRef] [PubMed]
- Horke, S.; Witte, I.; Wilgenbus, P.; Altenhöfer, S.; Krüger, M.; Li, H.; Förstermann, U. Protective effect of paraoxonase-2 against endoplasmic reticulum stress-induced apoptosis is lost upon disturbance of calcium homoeostasis. Biochem. J. 2008, 416, 395–405. [Google Scholar] [CrossRef]
- Devarajan, A.; Grijalva, V.R.; Bourquard, N.; Meriwether, D.; Imaizumi, S.; Shin, B.C.; Devaskar, S.U.; Reddy, S.T. Macrophage paraoxonase 2 regulates calcium homeostasis and cell survival under endoplasmic reticulum stress conditions and is sufficient to prevent the development of aggravated atherosclerosis in paraoxonase 2 deficiency/apoE-/- mice on a Western diet. Mol. Genet. Metab. 2012, 107, 416–427. [Google Scholar] [CrossRef]
- Sulaiman, D.; Li, J.; Devarajan, A.; Cunningham, C.M.; Li, M.; Fishbein, G.A.; Fogelman, A.M.; Eghbali, M.; Reddy, S.T. Paraoxonase 2 protects against acute myocardial ischemia-reperfusion injury by modulating mitochondrial function and oxidative stress via the PI3K/Akt/GSK-3β RISK pathway. J. Mol. Cell. Cardiol. 2019, 129, 154–164. [Google Scholar] [CrossRef]
- Tao, S.; Niu, L.; Cai, L.; Geng, Y.; Hua, C.; Ni, Y.; Zhao, R. N-(3-oxododecanoyl)-l-homoserine lactone modulates mitochondrial function and suppresses proliferation in intestinal goblet cells. Life Sci. 2018, 201, 81–88. [Google Scholar] [CrossRef]
- Tao, S.; Luo, Y.; Bin, H.; Liu, J.; Qian, X.; Ni, Y.; Zhao, R. Paraoxonase 2 modulates a proapoptotic function in LS174T cells in response to quorum sensing molecule N-(3-oxododecanoyl)-L-homoserine lactone. Sci. Rep. 2016, 6, 28778. [Google Scholar] [CrossRef]
- García-Heredia, A.; Kensicki, E.; Mohney, R.P.; Rull, A.; Triguero, I.; Marsillach, J.; Tormos, C.; Mackness, B.; Mackness, M.; Shih, D.M.; et al. Paraoxonase-1 deficiency is associated with severe liver steatosis in mice fed a high-fat high-cholesterol diet: A metabolomic approach. J. Proteome Res. 2013, 12, 1946–1955. [Google Scholar] [CrossRef] [PubMed]
- Luciano-Mateo, F.; Cabré, N.; Fernández-Arroyo, S.; Baiges-Gaya, G.; Hernández- Aguilera, A.; Rodríguez-Tomàs, E.; Mercado-Gómez, M.; Menendez, J.A.; Camps, J.; Joven, J. Chemokine (C-C motif) ligand 2 gene ablation protects low-density lipoprotein and paraoxonase-1 double deficient mice from liver injury, oxidative stress and inflammation. Biochim. Biophys. Acta Mold Basis Dis. 2019, 1865, 1555–1566. [Google Scholar] [CrossRef] [PubMed]
- Meneses, M.J.; Silvestre, R.; Sousa-Lima, I.; Macedo, M.P. Paraoxonase-1 as a regulator of glucose and lipid homeostasis: Impact on the onset and progression of metabolic disorders. Int. J. Mol. Sci. 2019, 20, 4049. [Google Scholar] [CrossRef] [PubMed]
- Calvo, N.; Beltrán-Debón, R.; Rodríguez-Gallego, E.; Hernández-Aguilera, A.; Guirro, M.; Mariné-Casadó, R.; Millá, L.; Alegret, J.M.; Sabench, F.; del Castillo, D.; et al. Liver fat deposition and mitochondrial dysfunction in morbid obesity: An approach combining metabolomics with liver imaging and histology. World J. Gastroenterol. 2015, 21, 7529–7544. [Google Scholar] [CrossRef]
- Cabré, N.; Luciano-Mateo, F.; Fernández-Arroyo, S.; Baiges-Gayà, G.; Hernández-Aguilera, A.; Fibla, M.; Fernández-Julià, R.; París, M.; Sabench, F.; Castillo, D.D.; et al. Laparoscopic sleeve gastrectomy reverses non-alcoholic fatty liver disease modulating oxidative stress and inflammation. Metabolism 2019, 99, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Cabré, N.; Luciano-Mateo, F.; Baiges-Gayà, G.; Fernández-Arroyo, S.; Rodríguez-Tomàs, E.; Hernández-Aguilera, A.; París, M.; Sabench, F.; Del Castillo, D.; López-Miranda, J.; et al. Plasma metabolic alterations in patients with severe obesity and non-alcoholic steatohepatitis. Aliment. Pharmacol. Ther. 2020, 51, 374–387. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, C.; Lecuona, E.; Wu, Q.; Yang, W.; Núñez-Santana, F.L.; Akbarpour, M.; Liu, X.; Ren, Z.; Li, W.; Querrey, M.; et al. Crosstalk between nonclassical monocytes and alveolar macrophages mediates transplant ischemia-reperfusion injury through classical monocyte recruitment. JCI Insight 2021, 6, 147282. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, S.; Nicklas, E.H.; Thadathil, N.; Selvarani, R.; Royce, G.H.; Kinter, M.; Richardson, A.; Deepa, S.S. Role of necroptosis in chronic hepatic inflammation and fibrosis in a mouse model of increased oxidative stress. Free Radic. Biol. Med. 2021, 164, 315–328. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Willette-Brown, J.; Zhang, J.; Ferre, E.M.N.; Sun, Z.; Wu, X.; Lionakis, M.S.; Hu, Y. NLRP3 inhibition ameliorates severe cutaneous autoimmune manifestations in a mouse model of autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy-like disease. J. Investig. Dermatol. 2020, 141, 1404–1415. [Google Scholar] [CrossRef]
- McKnight, A.H.; Katzenberger, D.R.; Britnell, S.R. Colchicine in acute coronary syndrome: A systematic review. Ann. Pharmacother. 2021, 55, 187–197. [Google Scholar] [CrossRef]
- Kolattukudy, P.E.; Niu, J. Inflammation, endoplasmic reticulum stress, autophagy, and the monocyte chemoattractant protein-1/CCR2 pathway. Circ. Res. 2012, 110, 174–189. [Google Scholar] [CrossRef]
- Pandey, E.; Nour, A.S.; Harris, E.N. Prominent receptors of liver sinusoidal endothelial cells in liver homeostasis and disease. Front. Physiol. 2020, 11, 873. [Google Scholar] [CrossRef]
- Andersson, U.; Ottestad, W.; Tracey, K.J. Extracellular HMGB1: A therapeutic target in severe pulmonary inflammation including COVID-19? Mol. Med. 2020, 26, 42. [Google Scholar] [CrossRef] [PubMed]
- Shamilov, R.; Ackley, T.W.; Aneskievich, B.J. Enhanced wound healing- and inflammasome-associated gene expression in TNFAIP3-interacting protein 1-(TNIP1-) deficient HaCaT keratinocytes parallels reduced reepithelialization. Mediat. Inflamm. 2020, 2020, 5919150. [Google Scholar] [CrossRef] [PubMed]
- Relja, B.; Land, W.G. Damage-associated molecular patterns in trauma. Eur. J. Trauma. Emerg. Surg. 2020, 46, 751–775. [Google Scholar] [CrossRef] [PubMed]
- Afrose, S.S.; Junaid, M.; Akter, Y.; Tania, M.; Zheng, M.; Khan, M.A. Targeting kinases with thymoquinone: A molecular approach to cancer therapeutics. Drug Discov. Today. 2020, 25, 2294–2306. [Google Scholar] [CrossRef] [PubMed]
- Dantonio, P.M.; Klein, M.O.; Freire, M.R.V.B.; Araujo, C.N.; Chiacetti, A.C.; Correa, R.G. Exploring major signaling cascades in melanomagenesis: A rationale route for targetted skin cancer therapy. Biosci. Rep. 2018, 38, BSR20180511. [Google Scholar] [CrossRef] [PubMed]
- Stawski, L.; Trojanowska, M. Oncostatin M and its role in fibrosis. Connect. Tissue Res. 2019, 60, 40–49. [Google Scholar] [CrossRef]
- Slaine, P.D.; Kleer, M.; Duguay, B.A.; Pringle, E.S.; Kadijk, E.; Ying, S.; Balgi, A.; Roberge, M.; McCormick, C.; Khaperskyy, D.A. Thiopurines activate an antiviral unfolded protein response that blocks influenza A virus glycoprotein accumulation. J. Virol. 2021, 95, e00453-21. [Google Scholar] [CrossRef] [PubMed]
- Féral, K.; Jaud, M.; Philippe, C.; Di Bella, D.; Pyronnet, S.; Rouault-Pierre, K.; Mazzolini, L.; Touriol, C. ER Stress and unfolded protein response in leukemia: Friend, foe, or both? Biomolecules 2021, 11, 199. [Google Scholar] [CrossRef]
- Huang, J.; Pan, H.; Wang, J.; Wang, T.; Huo, X.; Ma, Y.; Lu, Z.; Sun, B.; Jiang, H. Unfolded protein response in colorectal cancer. Cell. Biosci. 2021, 11, 26. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.M.; Talty, A.; Logue, S.E.; Mnich, K.; Gorman, A.M.; Samali, A. An emerging role for the unfolded protein response in pancreatic cancer. Cancers 2021, 13, 261. [Google Scholar] [CrossRef]
- Morris, G.; Puri, B.K.; Walder, K.; Berk, M.; Stubbs, B.; Maes, M.; Carvalho, A.F. The endoplasmic reticulum stress response in neuroprogressive diseases: Emerging pathophysiological role and translational implications. Mol. Neurobiol. 2018, 55, 8765–8787. [Google Scholar] [CrossRef] [PubMed]
- Rashid, H.O.; Yadav, R.K.; Kim, H.R.; Chae, H.J. ER stress: Autophagy induction, inhibition and selection. Autophagy 2015, 11, 1956–1977. [Google Scholar] [CrossRef] [PubMed]
- Dymkowska, D. The involvement of autophagy in the maintenance of endothelial homeostasis: The role of mitochondria. Mitochondrion 2021, 57, 131–147. [Google Scholar] [CrossRef]
- Picca, A.; Calvani, R.; Coelho-Junior, H.J.; Marzetti, E. Cell death and inflammation: The role of mitochondria in health and disease. Cells 2021, 10, 537. [Google Scholar] [CrossRef]
- Su, Y.J.; Wang, P.W.; Weng, S.W. The role of mitochondria in immune-cell-mediated tissue regeneration and ageing. Int. J. Mol Sci. 2021, 22, 2668. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.Y.; Blum, A.; Liu, J.; Finkel, T. The role of mitochondria in aging. J. Clin. Investig. 2018, 128, 3662–3670. [Google Scholar] [CrossRef] [PubMed]
- Meyers, A.K.; Zhu, X. The NLRP3 Inflammasome: Metabolic regulation and contribution to inflammaging. Cells 2020, 9, 1808. [Google Scholar] [CrossRef] [PubMed]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [PubMed]
- Fulop, T.; Witkowski, J.M.; Olivieri, F.; Larbi, A. The integration of inflammaging in age-related diseases. Semin. Immunol. 2018, 40, 17–35. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell 2010, 140, 900–917. [Google Scholar] [CrossRef] [PubMed]
- Berent-Maoz, B.; Montecino-Rodriguez, E.; Signer, R.A.; Dorshkind, K. Fibroblast growth factor-7 partially reverses murine thymocyte progenitor aging by repression of Ink4a. Blood 2012, 119, 5715–5721. [Google Scholar] [CrossRef]
- Sierra-Filardi, E.; Nieto, C.; Domínguez-Soto, A.; Barroso, R.; Sánchez-Mateos, P.; Puig-Kroger, A.; López-Bravo, M.; Joven, J.; Ardavín, C.; Rodríguez-Fernández, J.L.; et al. CCL2 shapes macrophage polarization by GM-CSF and M-CSF: Identification of CCL2/CCR2-dependent gene expression profile. J. Immunol. 2014, 192, 3858–3867. [Google Scholar] [CrossRef]
- Yousefzadeh, M.J.; Schafer, M.J.; Noren Hooten, N.; Atkinson, E.J.; Evans, M.K.; Baker, D.J.; Quarles, E.K.; Robbins, P.D.; Ladiges, W.C.; LeBrasseur, N.K.; et al. Circulating levels of monocyte chemoattractant protein-1 as a potential measure of biological age in mice and frailty in humans. Aging Cell 2018, 17, e12706. [Google Scholar] [CrossRef] [PubMed]
- Acosta, J.C.; Banito, A.; Wuestefeld, T.; Georgilis, A.; Janich, P.; Morton, J.P.; Athineos, D.; Kang, T.W.; Lasitschka, F.; Andrulis, M.; et al. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat. Cell. Biol. 2013, 15, 978–990. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Yu, K.R.; Lee, B.C.; Kang, I.; Kim, J.J.; Jung, E.J.; Kim, H.S.; Seo, Y.; Choi, S.W.; Kang, K.S. GATA4-dependent regulation of the secretory phenotype via MCP-1 underlies lamin a-mediated human mesenchymal stem cell aging. Exp. Mol. Med. 2018, 50, 1–12. [Google Scholar] [CrossRef]
- Luciano-Mateo, F.; Cabré, N.; Baiges-Gaya, G.; Fernández-Arroyo, S.; Hernández-Aguilera, A.; Rodríguez-Tomàs, E.; Arenas, M.; Camps, J.; Menéndez, J.A.; Joven, J. Systemic overexpression of C-C motif chemokine ligand 2 promotes metabolic dysregulation and premature death in mice with accelerated aging. Aging 2020, 12, 20001–20023. [Google Scholar] [CrossRef] [PubMed]
- Hoyt, L.R.; Randall, M.J.; Ather, J.L.; DePuccio, D.P.; Landry, C.C.; Qian, X.; Janssen-Heininger, Y.M.; van der Vliet, A.; Dixon, A.E.; Amiel, E.; et al. Mitochondrial ROS induced by chronic ethanol exposure promote hyper-activation of the NLRP3 inflammasome. Redox. Biol. 2017, 12, 883–896. [Google Scholar] [CrossRef]
- Lavallard, V.; Cottet-Dumoulin, D.; Wassmer, C.H.; Rouget, C.; Parnaud, G.; Brioudes, E.; Lebreton, F.; Bellofatto, K.; Berishvili, E.; Berney, T.; et al. NLRP3 inflammasome is activated in rat pancreatic islets by transplantation and hypoxia. Sci. Rep. 2020, 10, 7011. [Google Scholar] [CrossRef]
- Fusco, R.; Gugliandolo, E.; Siracusa, R.; Scuto, M.; Cordaro, M.; D’Amico, R.; Evangelista, M.; Peli, A.; Peritore, A.F.; Impellizzeri, D.; et al. Formyl peptide receptor 1 signaling in acute inflammation and neural differentiation induced by traumatic brain injury. Biology 2020, 9, 238. [Google Scholar] [CrossRef]
- González-Muniesa, P.; Mártinez-González, M.A.; Hu, F.B.; Després, J.P.; Matsuzawa, Y.; Loos, R.J.F.; Moreno, L.A.; Bray, G.A.; Martinez, J.A. Obesity. Nat. Rev. Dis. Primers. 2017, 15, 17034. [Google Scholar] [CrossRef]
- Friedman, S.L.; Neuschwander-Tetri, B.A.; Rinella, M.; Sanyal, A.J. Mechanisms of NAFLD development and therapeutic strategies. Nat. Med. 2018, 24, 908–922. [Google Scholar] [CrossRef]
- You, K.; Wang, L.; Chou, C.H.; Liu, K.; Nakata, T.; Jaiswal, A.; Yao, J.; Lefkovith, A.; Omar, A.; Perrigoue, J.G.; et al. QRICH1 dictates the outcome of ER stress through transcriptional control of proteostasis. Science 2021, 371, eabb6896. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Park, S.; Lee, D.; Ginting, R.P.; Lee, M.R.; Lee, M.W.; Han, J. Reduction in endoplasmic reticulum stress activates beige adipocytes differentiation and alleviates high fat diet-induced metabolic phenotypes. Biochim. Biophys. Acta Mol. Basis Dis. 2021, 1867, 166099. [Google Scholar] [CrossRef] [PubMed]
- Scully, T.; Ettela, A.; LeRoith, D.; Gallagher, E.J. Obesity, type 2 diabetes, and cancer risk. Front. Oncol. 2021, 10, 615375. [Google Scholar] [CrossRef] [PubMed]
- Srinivas, A.N.; Suresh, D.; Santhekadur, P.K.; Suvarna, D.; Kumar, D.P. Extracellular vesicles as inflammatory drivers in NAFLD. Front. Immunol. 2021, 11, 627424. [Google Scholar] [CrossRef] [PubMed]
- Dommel, S.; Blüher, M. Does C-C motif chemokine ligand 2 (CCL2) link obesity to a pro-inflammatory State? Int. J. Mol. Sci. 2021, 22, 1500. [Google Scholar] [CrossRef]
- Vieira, E.; Mirizio, G.G.; Barin, G.R.; de Andrade, R.V.; Nimer, N.F.S.; La Sala, L. Clock genes, inflammation and the immune system-implications for diabetes, obesity and neurodegenerative diseases. Int. J. Mol. Sci. 2020, 21, 9743. [Google Scholar] [CrossRef]
- Kawai, T.; Autieri, M.V.; Scalia, R. Adipose tissue inflammation and metabolic dysfunction in obesity. Am. J. Physiol. Cell. Physiol. 2021, 320, C375–C391. [Google Scholar] [CrossRef]
- Michailidou, Z.; Gomez-Salazar, M.; Alexaki, V.I. Innate immune cells in the adipose tissue in health and metabolic disease. J. Innate Immun. 2021, 13, 1–27. [Google Scholar] [CrossRef]
- Ruggiero, A.D.; Key, C.C.; Kavanagh, K. Adipose tissue macrophage polarization in healthy and unhealthy obesity. Front. Nutr. 2021, 8, 625331. [Google Scholar] [CrossRef]
- Kwaifa, I.K.; Bahari, H.; Yong, Y.K.; Noor, S.M. Endothelial dsfunction in obesity-induced inflammation: Molecular mechanisms and clinical implications. Biomolecules 2020, 10, 291. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.; Ni, L.; Zhuge, F.; Xu, L.; Fu, Z.; Ota, T. Adipose tissue macrophage phenotypes and characteristics: The key to insulin resistance in obesity and metabolic disorders. Obesity 2020, 28, 225–234. [Google Scholar] [CrossRef]
- Baiges-Gaya, G.; Fernández-Arroyo, S.; Luciano-Mateo, F.; Cabré, N.; Rodríguez-Tomàs, E.; Hernández-Aguilera, A.; Castañé, H.; Romeu, M.; Nogués, M.R.; Camps, J.; et al. Hepatic metabolic adaptation and adipose tissue expansion are altered in mice with steatohepatitis induced by high-fat high sucrose diet. J. Nutr. Biochem. 2021, 89, 108559. [Google Scholar] [CrossRef]
- Ferré, N.; Feliu, A.; García-Heredia, A.; Marsillach, J.; París, N.; Zaragoza-Jordana, M.; Mackness, B.; Mackness, M.; Escribano, J.; Closa-Monasterolo, R.; et al. Impaired paraoxonase-1 status in obese children. Relationships with insulin resistance and metabolic syndrome. Clin. Biochem. 2013, 46, 1830–1836. [Google Scholar] [CrossRef]
- Luciano-Mateo, F.; Cabré, N.; Nadal, M.; García-Heredia, A.; Baiges-Gaya, G.; Hernández-Aguilera, A.; Camps, J.; Joven, J.; Domingo, J.L. Serum concentrations of trace elements and their relationships with paraoxonase-1 in morbidly obese women. J. Trace Elem. Med. Biol. 2018, 48, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Sureda, A.; Mestre-Alfaro, A.; Banquells, M.; Riera, J.; Drobnic, F.; Camps, J.; Joven, J.; Tur., J.A.; Pons, A. Exercise in a hot environment influences plasma anti-inflammatory and antioxidant status in well-trained athletes. J. Therm. Biol. 2015, 47, 91–98. [Google Scholar] [CrossRef]
- Ferré, N.; Camps, J.; Fernández-Ballart, J.; Arija, V.; Murphy, M.M.; Ceruelo, S.; Biarnés, E.; Vilella, E.; Tous, M.; Joven, J. Regulation of serum paraoxonase activity by genetic, nutritional, and lifestyle factors in the general population. Clin. Chem. 2003, 49, 1491–1497. [Google Scholar] [CrossRef] [PubMed]
- Kupczyk, D.; Bilski, R.; Sokołowski, K.; Pawłowska, M.; Woźniak, A.; Szewczyk-Golec, K. Paraoxonase 1: The lectin-like oxidized LDL receptor type I and oxidative stress in the blood of men with type II obesity. Dis. Mark. 2019, 2019, 6178017. [Google Scholar] [CrossRef] [PubMed]
- Camps, J.; Joven, J. Chemokine ligand 2 and paraoxonase-1 in non-alcoholic fatty liver disease: The search for alternative causative factors. World J. Gastroenterol. 2015, 21, 2875–2882. [Google Scholar] [CrossRef] [PubMed]
- Camps, J.; Hernandez-Aguilera, A.; Garcia-Heredia, A.; Cabre, N.; Luciano-Mateo, F.; Arenas, M.; Joven, J. Relationships between metformin, paraoxonase-1 and the chemokine (C-C Motif) ligand 2. Curr. Clin. Pharmacol. 2016, 11, 250–258. [Google Scholar] [CrossRef]
- Marsillach, J.; Bertran, N.; Camps, J.; Ferré, N.; Riu, F.; Tous, M.; Coll, B.; Alonso- Villaverde, C.; Joven, J. The role of circulating monocyte chemoattractant protein-1 as a marker of hepatic inflammation in patients with chronic liver disease. Clin. Biochem. 2005, 38, 1138–1140. [Google Scholar] [CrossRef]
- Diaz Soto, M.P.; Lim, J.K. Evaluating the therapeutic potential of cenicriviroc in the treatment of nonalcoholic steatohepatitis with fibrosis: A brief report on emerging data. Hepat. Med. 2020, 12, 115–123. [Google Scholar] [CrossRef]
- Samaan, M.C.; Obeid, J.; Nguyen, T.; Thabane, L.; Timmons, B.W. Chemokine (C-C motif) Ligand 2 is a potential biomarker of inflammation & physical fitness in obese children: A cross-sectional study. BMC Pediatr. 2013, 13, 47. [Google Scholar] [CrossRef]
- Bodo, M.J.; Jimenez, E.Y.; Conn, C.; Dye, A.; Pomo, P.; Kolkmeyer, D.; Orlando, R.; Kong, A.S. Association between circulating CCL2 levels and modifiable behaviors in overweight and obese adolescents: A cross-sectional pilot study. J. Pediatr. Endocrinol. Metab. 2016, 29, 441–449. [Google Scholar] [CrossRef]
- Rull, A.; Camps, J.; Alonso-Villaverde, C.; Joven, J. Insulin resistance, inflammation, and obesity: Role of monocyte chemoattractant protein-1 (or CCL2) in the regulation of metabolism. Mediat. Inflamm. 2010, 2010, 326580. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Fan, M.; Zhao, L.; Yun, H.; Yang, Y.; Wang, C.; Qin, D. Fibroblast growth factor 1 ameliorates adipose tissue inflammation and systemic insulin resistance via enhancing adipocyte mTORC2/Rictor signal. J. Cell. Mol. Med. 2020, 24, 12813–12825. [Google Scholar] [CrossRef] [PubMed]
- Luciano-Mateo, F.; Cabré, N.; Fernández-Arroyo, S.; Baiges-Gaya, G.; Hernández- Aguilera, A.; Rodríguez-Tomàs, E.; Muñoz-Pinedo, C.; Menéndez, J.A.; Camps, J.; Joven, J. Chemokine C-C motif ligand 2 overexpression drives tissue-specific metabolic responses in the liver and muscle of mice. Sci. Rep. 2020, 10, 11954. [Google Scholar] [CrossRef] [PubMed]
- Mato, J.M.; Lu, S.C. Role of S-adenosyl-L-methionine in liver health and injury. Hepatology 2007, 45, 1306–1312. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Alvarez, M.I.; Sebastián, D.; Vives, S.; Ivanova, S.; Bartoccioni, P.; Kakimoto, P.; Plana, N.; Veiga, S.R.; Hernández, V.; Vasconcelos, N.; et al. Deficient endoplasmic reticulum-mitochondrial phosphatidylserine transfer causes liver disease. Cell 2019, 177, 881–895.e17. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Gallego, E.; Guirro, M.; Riera-Borrull, M.; Hernández-Aguilera, A.; Mariné-Casadó, R.; Fernández-Arroyo, S.; Beltrán-Debón, R.; Sabench, F.; Hernández, M.; del Castillo, D.; et al. Mapping of the circulating metabolome reveals α-ketoglutarate as a predictor of morbid obesity-associated non-alcoholic fatty liver disease. Int. J. Obes. 2015, 39, 279–287. [Google Scholar] [CrossRef]
- Hernández-Aguilera, A.; Casacuberta, N.; Castañé, H.; Fibla, M.; Fernández-Arroyo, S.; Fort-Gallifa, I.; París, M.; Sabench, F.; Del Castillo, D.; Baiges-Gaya, G.; et al. Nonalcoholic steatohepatitis modifies serum iron-related variables in patients with morbid obesity. Biol. Trace Elem. Res. 2021. [Google Scholar] [CrossRef] [PubMed]
- Auguet, T.; Bertran, L.; Binetti, J.; Aguilar, C.; Martínez, S.; Sabench, F.; Lopez-Dupla, J.M.; Porras, J.A.; Riesco, D.; Del Castillo, D.; et al. Relationship between IL-8 circulating levels and TLR2 hepatic expression in women with morbid obesity and nonalcoholic steatohepatitis. Int. J. Mol. Sci. 2020, 21, 4189. [Google Scholar] [CrossRef]
- Cabré, N.; Gil, M.; Amigó, N.; Luciano-Mateo, F.; Baiges-Gaya, G.; Fernández-Arroyo, S.; Rodríguez-Tomàs, E.; Hernández-Aguilera, A.; Castañé, H.; París, M.; et al. Laparoscopic sleeve gastrectomy alters 1H-NMR-measured lipoprotein and glycoprotein profile in patients with severe obesity and nonalcoholic fatty liver disease. Sci. Rep. 2021, 11, 1343. [Google Scholar] [CrossRef]
- Cabré, N.; Luciano-Mateo, F.; Chapski, D.J.; Baiges-Gaya, G.; Fernández-Arroyo, S.; Hernández-Aguilera, A.; Castañé, H.; Rodríguez-Tomàs, E.; París, M.; Sabench, F.; et al. Glutaminolysis-induced mTORC1 activation drives non-alcoholic steatohepatitis progression. J. Hepatol. 2021. Epub ahead of print. [Google Scholar] [CrossRef]
- Roth, C.L.; Molica, F.; Kwak, B.R. Browning of white adipose tissue as a therapeutic tool in the fight against atherosclerosis. Metabolites 2021, 11, 319. [Google Scholar] [CrossRef]
- Skiba, D.S.; Nosalski, R.; Mikolajczyk, T.P.; Siedlinski, M.; Rios, F.J.; Montezano, A.C.; Jawien, J.; Olszanecki, R.; Korbut, R.; Czesnikiewicz-Guzik, M.; et al. Anti-atherosclerotic effect of the angiotensin 1-7 mimetic AVE0991 is mediated by inhibition of perivascular and plaque inflammation in early atherosclerosis. Br. J. Pharmacol. 2017, 174, 4055–4069. [Google Scholar] [CrossRef] [PubMed]
- Nosalski, R.; Guzik, T.J. Perivascular adipose tissue inflammation in vascular disease. Br. J. Pharmacol. 2017, 174, 3496–3513. [Google Scholar] [CrossRef]
- Ravindran, D.; Ridiandries, A.; Vanags, L.Z.; Henriquez, R.; Cartland, S.; Tan, J.T.; Bursill, C.A. Chemokine binding protein ‘M3’ limits atherosclerosis in apolipoprotein E-/- mice. PLoS ONE 2017, 12, e0173224. [Google Scholar] [CrossRef] [PubMed]
- Braunersreuther, V.; Pellieux, C.; Pelli, G.; Burger, F.; Steffens, S.; Montessuit, C.; Weber, C.; Proudfoot, A.; Mach, F.; Arnaud, C. Chemokine CCL5/RANTES inhibition reduces myocardial reperfusion injury in atherosclerotic mice. J. Mol. Cell. Cardiol. 2010, 48, 789–798. [Google Scholar] [CrossRef]
- França, C.N.; Izar, M.C.O.; Hortêncio, M.N.S.; do Amaral, J.B.; Ferreira, C.E.S.; Tuleta, I.D.; Fonseca, F.A.H. Monocyte subtypes and the CCR2 chemokine receptor in cardiovascular disease. Clin. Sci. 2017, 131, 1215–1224. [Google Scholar] [CrossRef]
- Bianconi, V.; Sahebkar, A.; Atkin, S.L.; Pirro, M. The regulation and importance of monocyte chemoattractant protein-1. Curr. Opin. Hematol. 2018, 25, 44–51. [Google Scholar] [CrossRef]
- Hartmann, P.; Schober, A.; Weber, C. Chemokines and microRNAs in atherosclerosis. Cell. Mol. Life Sci. 2015, 72, 3253–3266. [Google Scholar] [CrossRef] [PubMed]
- Takeya, M.; Yoshimura, T.; Leonard, E.J.; Takahashi, K. Detection of monocyte chemoattractant protein-1 in human atherosclerotic lesions by an anti-monocyte chemoattractant protein-1 monoclonal antibody. Hum. Pathol. 1993, 24, 534–539. [Google Scholar] [CrossRef]
- Nelken, N.A.; Coughlin, S.R.; Gordon, D.; Wilcox, J.N. Monocyte chemoattractant protein-1 in human atheromatous plaques. J. Clin. Investig. 1991, 88, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Aguilera, A.; Fibla, M.; Cabré, N.; Luciano-Mateo, F.; Camps, J.; Fernández-Arroyo, S.; Martín-Paredero, V.; Menendez, J.A.; Sirvent, J.J.; Joven, J. Chemokine (C-C motif) ligand 2 and coronary artery disease: Tissue expression of functional and atypical receptors. Cytokine 2020, 126, 154923. [Google Scholar] [CrossRef]
- Georgakis, M.K.; Malik, R.; Björkbacka, H.; Pana, T.A.; Demissie, S.; Ayers, C.; Elhadad, M.A.; Fornage, M.; Beiser, A.S.; Benjamin, E.J.; et al. Circulating monocyte chemoattractant protein-1 and risk of stroke: Meta-analysis of population-based studies involving 17180 Individuals. Circ. Res. 2019, 125, 773–782. [Google Scholar] [CrossRef]
- Georgakis, M.K.; Gill, D.; Rannikmäe, K.; Traylor, M.; Anderson, C.D.; Lee, J.M.; Kamatani, Y.; Hopewell, J.C.; Worrall, B.B.; Bernhagen, J.; et al. Genetically determined levels of circulating cytokines and risk of stroke. Circulation 2019, 139, 256–268. [Google Scholar] [CrossRef] [PubMed]
- Savelieva, I.; Camm, A.J. A new biomarker in atrial fibrillation: Monocyte chemoattractant protein-1-induced protein. Cardiology 2019, 144, 122–124. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lou, W.; Li, H.; Zhu, Y.; Huang, X. Upregulated C-C motif chemokine ligand 2 promotes ischemic stroke via chemokine signaling pathway. Ann. Vasc. Surg. 2020, 68, 476–486. [Google Scholar] [CrossRef]
- Feingold, K.R.; Grunfeld, C. Effect of inflammation on HDL structure and function. Curr. Opin. Lipidol. 2016, 27, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Cervellati, C.; Vigna, G.B.; Trentini, A.; Sanz, J.M.; Zimetti, F.; Dalla Nora, E.; Morieri, M.L.; Zuliani, G.; Passaro, A. Paraoxonase-1 activities in individuals with different HDL circulating levels: Implication in reverse cholesterol transport and early vascular damage. Atherosclerosis 2019, 285, 64–70. [Google Scholar] [CrossRef]
- Vaisar, T.; Kanter, J.E.; Wimberger, J.; Irwin, A.D.; Gauthier, J.; Wolfson, E.; Bahnam, V.; Wu, I.H.; Shah, H.; Keenan, H.A.; et al. High concentration of medium-sized HDL particles and enrichment in HDL paraoxonase 1 associate with protection from vascular complications in people with long-standing type 1 diabetes. Diabetes Care 2020, 43, 178–186. [Google Scholar] [CrossRef] [PubMed]
- White, C.R.; Anantharamaiah, G.M. Cholesterol reduction and macrophage function: Role of paraoxonases. Curr. Opin. Lipidol. 2017, 28, 397–402. [Google Scholar] [CrossRef]
- Sikora, M.; Bretes, E.; Perła-Kaján, J.; Lewandowska, I.; Marczak, Ł.; Jakubowski, H. Genetic attenuation of paraoxonase 1 activity induces proatherogenic changes in plasma proteomes of mice and humans. Antioxidants 2020, 9, 1198. [Google Scholar] [CrossRef]
- Çetin, M.; Tunçdemir, P.; Karaman, K.; Yel, S.; Karaman, E.; Özgökçe, M.; Kömüroğlu, A.U. Cardiovascular evaluation and serum paraoxonase-1 levels in adolescents with polycystic ovary syndrome. J. Obstet. Gynaecol. 2020, 40, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Shokri, Y.; Variji, A.; Nosrati, M.; Khonakdar-Tarsi, A.; Kianmehr, A.; Kashi, Z.; Bahar, A.; Bagheri, A.; Mahrooz, A. Importance of paraoxonase 1 (PON1) as an antioxidant and antiatherogenic enzyme in the cardiovascular complications of type 2 diabetes: Genotypic and phenotypic evaluation. Diabetes Res. Clin. Pract. 2020, 161, 108067. [Google Scholar] [CrossRef]
- Khalil, A.; Fulop, T.; Berrougui, H. Role of paraoxonase1 in the regulation of high-density lipoprotein functionality and in cardiovascular protection. Antioxid. Redox Signal. 2021, 34, 191–200. [Google Scholar] [CrossRef]
- Khosravi, M.; Poursaleh, A.; Ghasempour, G.; Farhad, S.; Najafi, M. The effects of oxidative stress on the development of atherosclerosis. Biol. Chem. 2019, 400, 711–732. [Google Scholar] [CrossRef]
- Ponce-Ruiz, N.; Murillo-González, F.E.; Rojas-García, A.E.; Barrón-Vivanco, B.S.; Bernal-Hernández, Y.Y.; González-Arias, C.A.; Ortega-Cervantes, L.; Ponce-Gallegos, J.; López-Guarnido, O.; Medina-Díaz, I.M. PON1 status and homocysteine levels as potential biomarkers for cardiovascular disease. Exp. Gerontol. 2020, 140, 111062. [Google Scholar] [CrossRef]
- Corsetti, J.P.; Sparks, C.E.; James, R.W.; Bakker, S.J.L.; Dullaart, R.P.F. Low serum paraoxonase-1 activity associates with incident cardiovascular disease risk in subjects with concurrently high levels of high-density lipoprotein cholesterol and C-reactive protein. J. Clin. Med. 2019, 8, 1357. [Google Scholar] [CrossRef]
- Kunutsor, S.K.; Bakker, S.J.; James, R.W.; Dullaart, R.P. Serum paraoxonase-1 activity and risk of incident cardiovascular disease: The PREVEND study and meta-analysis of prospective population studies. Atherosclerosis 2016, 245, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Lent-Schochet, D.; McLaughlin, M.; Ramakrishnan, N.; Jialal, I. Exploratory metabolomics of metabolic syndrome: A status report. World J. Diabetes 2019, 10, 23–36. [Google Scholar] [CrossRef]
- Reddy, P.; Lent-Schochet, D.; Ramakrishnan, N.; McLaughlin, M.; Jialal, I. Metabolic syndrome is an inflammatory disorder: A conspiracy between adipose tissue and phagocytes. Clin. Chim. Acta 2019, 496, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Pouralijan Amiri, M.; Khoshkam, M.; Salek, R.M.; Madadi, R.; Faghanzadeh Ganji, G.; Ramazani, A. Metabolomics in early detection and prognosis of acute coronary syndrome. Clin. Chim. Acta 2019, 495, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Iida, M.; Harada, S.; Takebayashi, T. Application of metabolomics to epidemiological studies of atherosclerosis and cardiovascular disease. J. Atheroscler. Thromb. 2019, 26, 747–757. [Google Scholar] [CrossRef] [PubMed]
- Ganna, A.; Salihovic, S.; Sundström, J.; Broeckling, C.D.; Hedman, A.K.; Magnusson, P.K.; Pedersen, N.L.; Larsson, A.; Siegbahn, A.; Zilmer, M.; et al. Large-scale metabolomic profiling identifies novel biomarkers for incident coronary heart disease. PLoS Genet. 2014, 10, e1004801. [Google Scholar] [CrossRef]
- Stegemann, C.; Pechlaner, R.; Willeit, P.; Langley, S.R.; Mangino, M.; Mayr, U.; Menni, C.; Moayyeri, A.; Santer, P.; Rungger, G.; et al. Lipidomics profiling and risk of cardiovascular disease in the prospective population-based Bruneck study. Circulation 2014, 129, 1821–1831. [Google Scholar] [CrossRef]
- Shah, S.H.; Sun, J.L.; Stevens, R.D.; Bain, J.R.; Muehlbauer, M.J.; Pieper, K.S.; Haynes, C.; Hauser, E.R.; Kraus, W.E.; Granger, C.B.; et al. Baseline metabolomic profiles predict cardiovascular events in patients at risk for coronary artery disease. Am. Heart J. 2012, 163, 844–850.e1. [Google Scholar] [CrossRef]
- Haas, J.; Frese, K.S.; Sedaghat-Hamedani, F.; Kayvanpour, E.; Tappu, R.; Nietsch, R.; Tugrul, O.F.; Wisdom, M.; Dietrich, C.; Amr, A.; et al. Energy Metabolites as Biomarkers in Ischemic and Dilated Cardiomyopathy. Int. J. Mol. Sci. 2021, 22, 1999. [Google Scholar] [CrossRef]
- Van Driel, B.O.; Schuldt, M.; Algül, S.; Levin, E.; Güclü, A.; Germans, T.; Rossum, A.C.V.; Pei, J.; Harakalova, M.; Baas, A.; et al. Metabolomics in Severe Aortic Stenosis Reveals Intermediates of Nitric Oxide Synthesis as Most Distinctive Markers. Int. J. Mol. Sci. 2021, 22, 3569. [Google Scholar] [CrossRef]
- Rull, A.; Hernandez-Aguilera, A.; Fibla, M.; Sepulveda, J.; Rodríguez-Gallego, E.; Riera-Borrull, M.; Sirvent, J.J.; Martín-Paredero, V.; Menendez, J.A.; Camps, J.; et al. Understanding the role of circulating chemokine (C-C motif) ligand 2 in patients with chronic ischemia threatening the lower extremities. Vasc. Med. 2014, 19, 442–451. [Google Scholar] [CrossRef] [PubMed]
- Fort-Gallifa, I.; García-Heredia, A.; Hernández-Aguilera, A.; Simó, J.M.; Sepúlveda, J.; Martín-Paredero, V.; Camps, J.; Joven, J. Biochemical indices of oxidative stress and inflammation in the evaluation of peripheral artery disease. Free Radic. Biol. Med. 2016, 97, 568–576. [Google Scholar] [CrossRef]
- Fort-Gallifa, I.; Hernández-Aguilera, A.; García-Heredia, A.; Cabré, N.; Luciano-Mateo, F.; Simó, J.M.; Martín-Paredero, V.; Camps, J.; Joven, J. Galectin-3 in peripheral artery disease. Relationships with markers of oxidative stress and inflammation. Int. J. Mol Sci. 2017, 18, 973. [Google Scholar] [CrossRef]
- Cabré, N.; Luciano-Mateo, F.; Arenas, M.; Nadal, M.; Baiges-Gaya, G.; Hernández-Aguilera, A.; Fort-Gallifa, I.; Rodríguez, E.; Riu, F.; Camps, J.; et al. Trace element concentrations in breast cancer patients. Breast 2018, 42, 142–149. [Google Scholar] [CrossRef]
- Hernández-Aguilera, A.; Fernández-Arroyo, S.; Cabre, N.; Luciano-Mateo, F.; Baiges-Gaya, G.; Fibla, M.; Martín-Paredero, V.; Menendez, J.A.; Camps, J.; Joven, J. Plasma energy-balance metabolites discriminate asymptomatic patients with peripheral artery disease. Mediat. Inflamm. 2018, 2018, 2760272. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.A. Metabolic pathways in obesity-related breast cancer. Nat. Rev. Endocrinol. 2021, 17, 350–363. [Google Scholar] [CrossRef]
- Michalak, S.; Szubert, S.; Moszynski, R.; Sajdak, S.; Szpurek, D. Serum arylesterase and paraoxonase activities in patients with ovarian tumors. Taiwan J. Obstet. Gynecol. 2014, 53, 490–493. [Google Scholar] [CrossRef] [PubMed]
- Arenas, M.; Rodríguez, E.; Sahebkar, A.; Sabater, S.; Rizo, D.; Pallisé, O.; Hernández, M.; Riu, F.; Camps, J.; Joven, J. Paraoxonase-1 activity in patients with cancer: A systematic review and meta-analysis. Crit. Rev. Oncol. Hematol. 2018, 127, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Arenas, M.; García-Heredia, A.; Cabré, N.; Luciano-Mateo, F.; Hernández-Aguilera, A.; Sabater, S.; Bonet, M.; Gascón, M.; Fernández-Arroyo, S.; Fort-Gallifa, I.; et al. Effect of radiotherapy on activity and concentration of serum paraoxonase-1 in breast cancer patients. PLoS ONE 2017, 12, e0188633. [Google Scholar] [CrossRef]
- Rodríguez-Tomàs, E.; Murcia, M.; Arenas, M.; Arguís, M.; Gil, M.; Amigó, N.; Correig, X.; Torres, L.; Sabater, S.; Baiges-Gayà, G.; et al. Serum paraoxonase-1-related variables and lipoprotein profile in patients with lung or head and neck cancer: Effect of radiotherapy. Antioxidants 2019, 8, 213. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Tomàs, E.; Arenas, M.; Gómez, J.; Acosta, J.; Trilla, J.; López, Y.; Árquez, M.; Torres, L.; Araguas, P.; Hernández-Aguilera, A.; et al. Identification of potential metabolic biomarkers of rectal cancer and of the effect of neoadjuvant radiochemotherapy. PLoS ONE 2021, 16, e0250453. [Google Scholar] [CrossRef]
- Camuzcuoglu, H.; Arioz, D.T.; Toy, H.; Kurt, S.; Celik, H.; Erel, O. Serum paraoxonase and arylesterase activities in patients with epithelial ovarian cancer. Gynecol. Oncol. 2009, 112, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Krzystek-Korpacka, M.; Boehm, D.; Matusiewicz, M.; Diakowska, D.; Grabowski, K.; Gamian, A. Paraoxonase 1 (PON1) status in gastroesophageal malignancies and associated paraneoplastic syndromes-connection with inflammation. Clin. Biochem. 2008, 41, 804–811. [Google Scholar] [CrossRef]
- Afsar, C.U.; Gunaldı, M.; Okuturlar, Y.; Gedikbası, A.; Tiken, E.E.; Kahraman, S.; Karaca, F.; Ercolak, V.; Karabulut, M. Paraoxonase-1 and arylesterase activities in patients with colorectal cancer. Int. J. Clin. Exp. Med. 2015, 8, 21599–21604. [Google Scholar] [PubMed]
- Balci, H.; Genc, H.; Papila, C.; Can, G.; Papila, B.; Yanardag, H.; Uzun, H. Serum lipid hydroperoxide levels and paraoxonase activity in patients with lung, breast, and colorectal cancer. J. Clin. Lab. Anal. 2012, 26, 155–160. [Google Scholar] [CrossRef]
- Sehitogulları, A.; Aslan, M.; Sayır, F.; Kahraman, A.; Demir, H. Serum paraoxonase-1 enzyme activities and oxidative stress levels in patients with esophageal squamous cell carcinoma. Redox Rep. 2014, 19, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Bulbuller, N.; Eren, E.; Ellidag, H.Y.; Oner, O.Z.; Sezer, C.; Aydin, O.; Yılmaz, N. Diagnostic value of thiols, paraoxonase 1, arylesterase and oxidative balance in colorectal cancer in human. Neoplasma 2013, 60, 419–424. [Google Scholar] [CrossRef]
- Malik, U.U.; Siddiqui, I.A.; Hashim, Z.; Zarina, S. Measurement of serum paraoxonase activity and MDA concentrations in patients suffering with oral squamous cell carcinoma. Clin. Chim. Acta. 2014, 430, 38–42. [Google Scholar] [CrossRef]
- Iftimie, S.; García-Heredia, A.; Pujol-Bosch, F.; Pont-Salvadó, A.; López-Azcona, A.F.; Hernández-Aguilera, A.; Cabré, N.; Luciano-Mateo, F.; Fort-Gallifa, I.; Castro, A.; et al. Serum paraoxonase-1 concentration as a potential predictor of urinary bladder cancer recurrence. A five year follow-up study. Arch. Med. Res. 2018, 49, 119–122. [Google Scholar] [CrossRef]
- Li, R.; Wen, A.; Lin, J. Pro-inflammatory cytokines in the formation of the pre-metastatic niche. Cancers 2020, 12, 3752. [Google Scholar] [CrossRef]
- Tao, L.; Liu, S.; Xiong, J.; Yang, H.; Wu, Y.; Xu, A.; Gong, Y. IL-1β promotes cervical cancer through activating NF-κB/CCL-2. Int. J. Clin. Exp. Pathol. 2021, 14, 426–433. [Google Scholar] [PubMed]
- Adekoya, T.O.; Richardson, R.M. Cytokines and chemokines as mediators of prostate cancer metastasis. Int. J. Mol. Sci. 2020, 21, 4449. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.B.; Sofia Acevedo, D.; Smart, C.; Zinda, B.; Alissa, N.; Warren, K.; Fraga, G.; Huang, L.C.; Shyr, Y.; Li, W.; et al. Expression of CCL2/CCR2 signaling proteins in breast carcinoma cells is associated with invasive progression. Sci. Rep. 2021, 11, 8708. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Hong, J.H.; Kim, J.S.; Yoon, J.S.; Chun, S.H.; Hong, S.A.; Kim, E.J.; Kang, K.; Lee Kang, J.; Ko, Y.H.; et al. Cancer-associated fibroblasts activated by miR-196a promote the migration and invasion of lung cancer cells. Cancer Lett. 2021, 508, 92–103. [Google Scholar] [CrossRef]
- Liu, X.; Zheng, S.; Peng, Y.; Zhuang, J.; Yang, Y.; Xu, Y.; Guan, G. Construction of the prediction model for locally advanced rectal cancer following neoadjuvant chemoradiotherapy based on pretreatment tumor-infiltrating macrophage-associated biomarkers. Onco. Targets Ther. 2021, 14, 2599–2610. [Google Scholar] [CrossRef]
- Sun, X.; Ye, D.; Du, L.; Qian, Y.; Jiang, X.; Mao, Y. Genetically predicted levels of circulating cytokines and prostate cancer risk: A Mendelian randomization study. Int. J. Cancer 2020, 147, 2469–2478. [Google Scholar] [CrossRef] [PubMed]
- Vander Heiden, M.G.; Cantley, L.C.; Thompson, C.B. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science 2009, 324, 1029–1033. [Google Scholar] [CrossRef]
- Warburg, O. On the origin of cancer cells. Science 1956, 123, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Bott, A.J.; Maimouni, S.; Zong, W.X. The pleiotropic effects of glutamine metabolism in cancer. Cancers 2019, 11, 770. [Google Scholar] [CrossRef] [PubMed]
- Trilla-Fuertes, L.; Gámez-Pozo, A.; López-Camacho, E.; Prado-Vázquez, G.; Zapater- Moros, A.; López-Vacas, R.; Arevalillo, J.M.; Díaz-Almirón, M.; Navarro, H.; Maín, P.; et al. Computational models applied to metabolomics data hints at the relevance of glutamine metabolism in breast cancer. BMC Cancer 2020, 20, 307. [Google Scholar] [CrossRef]
- Nguyen, T.; Kirsch, B.J.; Asaka, R.; Nabi, K.; Quinones, A.; Tan, J.; Antonio, M.J.; Camelo, F.; Li, T.; Nguyen, S.; et al. Uncovering the role of N-acetyl-aspartyl-glutamate as a glutamate reservoir in cancer. Cell Rep. 2019, 27, 491–501.e6. [Google Scholar] [CrossRef]
- Udupa, S.; Nguyen, S.; Hoang, G.; Nguyen, T.; Quinones, A.; Pham, K.; Asaka, R.; Nguyen, K.; Zhang, C.; Elgogary, A.; et al. Upregulation of the glutaminase II pathway contributes to glutamate production upon glutaminase 1 inhibition in pancreatic cancer. Proteomics 2019, 19, e1800451. [Google Scholar] [CrossRef] [PubMed]
- Karsli-Uzunbas, G.; Guo, J.Y.; Price, S.; Teng, X.; Laddha, S.V.; Khor, S.; Kalaany, N.Y.; Jacks, T.; Chan, C.S.; Rabinowitz, J.D.; et al. Autophagy is required for glucose homeostasis and lung tumor maintenance. Cancer Discov. 2014, 4, 914–927. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.Y.; Teng, X.; Laddha, S.V.; Ma, S.; Van Nostrand, S.C.; Yang, Y.; Khor, S.; Chan, C.S.; Rabinowitz, J.D.; White, E. Autophagy provides metabolic substrates to maintain energy charge and nucleotide pools in Ras-driven lung cancer cells. Genes Dev. 2016, 30, 1704–1717. [Google Scholar] [CrossRef]
- Rabinowitz, J.D.; White, E. Autophagy and metabolism. Science 2010, 330, 1344–1348. [Google Scholar] [CrossRef]
- Sappington, D.R.; Siegel, E.R.; Hiatt, G.; Desai, A.; Penney, R.B.; Jamshidi-Parsian, A.; Griffin, R.J.; Boysen, G. Glutamine drives glutathione synthesis and contributes to radiation sensitivity of A549 and H460 lung cancer cell lines. Biochim. Biophys. Acta 2016, 1860, 836–843. [Google Scholar] [CrossRef]
- Zeng, C.; Wu, Q.; Wang, J.; Yao, B.; Ma, L.; Yang, Z.; Li, J.; Liu, B. NOX4 supports glycolysis and promotes glutamine metabolism in non-small cell lung cancer cells. Free Radic. Biol. Med. 2016, 101, 236–248. [Google Scholar] [CrossRef]
- Rodríguez-Tomàs, E.; Arguís, M.; Arenas, M.; Fernández-Arroyo, S.; Murcia, M.; Sabater, S.; Torres, L.; Baiges-Gayà, G.; Hernández-Aguilera, A.; Camps, J.; et al. Alterations in plasma concentrations of energy-balance-related metabolites in patients with lung, or head & neck, cancers: Effects of radiotherapy. J. Proteom. 2020, 213, 103605. [Google Scholar] [CrossRef]
- Vanhove, K.; Giesen, P.; Owokotomo, O.E.; Mesotten, L.; Louis, E.; Shkedy, Z.; Thomeer, M.; Adriaensens, P. The plasma glutamate concentration as a complementary tool to differentiate benign PET-positive lung lesions from lung cancer. BMC Cancer 2018, 18, 868. [Google Scholar] [CrossRef]
- Berker, Y.; Vandergrift, L.A.; Wagner, I.; Su, L.; Kurth, J.; Schuler, A.; Dinges, S.S.; Habbel, P.; Nowak, J.; Mark, E.; et al. Magnetic resonance spectroscopy-based metabolomic biomarkers for typing, staging, and survival estimation of early-stage human lung cancer. Sci. Rep. 2019, 9, 10319. [Google Scholar] [CrossRef]
- Michalak, S.; Rybacka-Mossakowska, J.; Ambrosius, W.; Gazdulska, J.; Gołda-Gocka, I.; Kozubski, W.; Ramlau, R. The markers of glutamate metabolism in peripheral blood mononuclear cells and neurological complications in lung cancer patients. Dis. Mark. 2016, 2016, 2895972. [Google Scholar] [CrossRef]
- Ye, G.; Liu, Y.; Yin, P.; Zeng, Z.; Huang, Q.; Kong, H.; Lu, X.; Zhong, L.; Zhang, Z.; Xu, G. Study of induction chemotherapy efficacy in oral squamous cell carcinoma using pseudotargeted metabolomics. J. Proteome Res. 2014, 13, 1994–2004. [Google Scholar] [CrossRef] [PubMed]
- Long, N.P.; Yoon, S.J.; Anh, N.H.; Nghi, T.D.; Lim, D.K.; Hong, Y.J.; Hong, S.S.; Kwon, S.W. A systematic review on metabolomics-based diagnostic biomarker discovery and validation in pancreatic cancer. Metabolomics 2018, 14, 109. [Google Scholar] [CrossRef]
- Arenas, M.; Rodríguez, E.; García-Heredia, A.; Fernández-Arroyo, S.; Sabater, S.; Robaina, R.; Gascón, M.; Rodríguez-Pla, M.; Cabré, N.; Luciano-Mateo, F.; et al. Metabolite normalization with local radiotherapy following breast tumor resection. PLoS ONE 2018, 13, e0207474. [Google Scholar] [CrossRef]
- Arenas, M.; Fernández-Arroyo, S.; Rodríguez-Tomàs, E.; Sabater, S.; Murria, Y.; Gascón, M.; Amillano, K.; Melé, M.; Camps, J.; Joven, J. Effects of radiotherapy on plasma energy metabolites in patients with breast cancer who received neoadjuvant chemotherapy. Clin. Transl. Oncol. 2020, 22, 1078–1085. [Google Scholar] [CrossRef] [PubMed]
- Holecek, M. Relation between glutamine, branched-chain amino acids, and protein metabolism. Nutrition 2002, 18, 130–133. [Google Scholar] [CrossRef]
- Hensley, C.T.; Wasti, A.T.; DeBerardinis, R.J. Glutamine and cancer: Cell biology, physiology, and clinical opportunities. J. Clin. Investig. 2013, 123, 3678–3684. [Google Scholar] [CrossRef]
- Miller, V.J.; Villamena, F.A.; Volek, J.S. Nutritional ketosis and mitohormesis: Potential implications for mitochondrial function and human health. J. Nutr. Metab. 2018, 8, 5157645. [Google Scholar] [CrossRef]
- Shimazu, T.; Hirschey, M.D.; Newman, J.; He, W.; Shirakawa, K.; Le Moan, N.; Grueter, C.A.; Lim, H.; Saunders, L.R.; Stevens, R.D.; et al. Suppression of oxidative stress by β-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science 2013, 339, 211–214. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Hu, M.; Zhang, X.; Hu, J.Z. NMR-based metabolomics analysis of liver from C57BL/6 mouse exposed to ionizing radiation. Radiat. Res. 2017, 188, 44–55. [Google Scholar] [CrossRef]
- Mörén, L.; Wibom, C.; Bergström, P.; Johansson, M.; Antti, H.; Bergenheim, A.T. Characterization of the serum metabolome following radiation treatment in patients with high-grade gliomas. Radiat. Oncol. 2016, 11, 51. [Google Scholar] [CrossRef]
- Chai, Y.; Wang, J.; Wang, T.; Yang, Y.; Su, J.; Shi, F.; Wang, J.; Zhou, X.; He, B.; Ma, H.; et al. Application of 1H NMR spectroscopy-based metabonomics to feces of cervical cancer patients with radiation-induced acute intestinal symptoms. Radiother. Oncol. 2015, 117, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Webb, N.R. High-density lipoproteins and serum amyloid A (SAA). Curr. Atheroscler. Rep. 2021, 23, 7. [Google Scholar] [CrossRef]
- Namiri-Kalantari, R.; Gao, F.; Chattopadhyay, A.; Wheeler, A.A.; Navab, K.D.; Farias-Eisner, R.; Reddy, S.T. The dual nature of HDL: Anti-inflammatory and pro-inflammatory. Biofactors 2015, 41, 153–159. [Google Scholar] [CrossRef]
- Birner-Gruenberger, R.; Schittmayer, M.; Holzer, M.; Marsche, G. Understanding high-density lipoprotein function in disease: Recent advances in proteomics unravel the complexity of its composition and biology. Prog. Lipid Res. 2014, 56, 36–46. [Google Scholar] [CrossRef]
- Sharma, N.K.; Ferreira, B.L.; Tashima, A.K.; Brunialti, M.K.C.; Torquato, R.J.S.; Bafi, A.; Assuncao, M.; Azevedo, L.C.P.; Salomao, R. Lipid metabolism impairment in patients withsepsis secondary to hospital acquired pneumonia, a proteomic analysis. Clin. Proteom. 2019, 16, 29. [Google Scholar] [CrossRef] [PubMed]
- Iftimie, S.; García-Heredia, A.; Pujol, I.; Ballester, F.; Fort-Gallifa, I.; Simó, J.M.; Joven, J.; Castro, A.; Camps, J. A preliminary study of paraoxonase-1 in infected patients with an indwelling central venous catheter. Clin. Biochem. 2016, 49, 449–457. [Google Scholar] [CrossRef]
- Iftimie, S.; García-Heredia, A.; Pujol, I.; Ballester, F.; Fort-Gallifa, I.; Simó, J.M.; Joven, J.; Camps, J.; Castro, A. Preliminary study on serum paraoxonase-1 status and chemokine (C-C motif) ligand 2 in hospitalized elderly patients with catheter-associated asymptomatic bacteriuria. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 1417–1424. [Google Scholar] [CrossRef]
- Sans, T.; Rull, A.; Luna, J.; Mackness, B.; Mackness, M.; Joven, J.; Ibañez, M.; Pariente, R.; Rodriguez, M.; Ortin, X.; et al. Monocyte chemoattractant protein-1 and paraoxonase-1 and 3 levels in patients with sepsis treated in an intensive care unit: A preliminary report. Clin. Chem. Lab. Med. 2012, 50, 1409–1415. [Google Scholar] [CrossRef] [PubMed]
- Garrido, P.; Rovira, C.; Cueto, P.; Fort-Gallifa, I.; Hernández-Aguilera, A.; Cabré, N.; Luciano-Mateo, F.; García-Heredia, A.; Camps, J.; Joven, J.; et al. Effect of continuous renal-replacement therapy on paraoxonase-1-related variables in patients with acute renal failure caused by septic shock. Clin. Biochem. 2018, 61, 1–6. [Google Scholar] [CrossRef]
- Kucukazman, M.; Yeniova, O.; Dal, K.; Yavuz, B. Helicobacter pylori and cardiovascular disease. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 3731–3741. [Google Scholar] [PubMed]
- Vijayvergiya, R.; Vadivelu, R. Role of Helicobacter pylori infection in pathogenesis of atherosclerosis. World J. Cardiol. 2015, 7, 134–143. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Yang, Z.; Lu, N.H. Helicobacter pylori: An infectious risk factor for atherosclerosis? J. Atheroscler. Thromb. 2014, 21, 1229–1242. [Google Scholar] [CrossRef][Green Version]
- Aslan, M.; Nazligul, Y.; Horoz, M.; Bolukbas, C.; Bolukbas, F.F.; Gur, M.; Celik, H.; Erel, O. Serum paraoxonase-1 activity in Helicobacter pylori infected subjects. Atherosclerosis 2008, 196, 270–274. [Google Scholar] [CrossRef]
- Akbas, H.S.; Basyigit, S.; Suleymanlar, I.; Kemaloglu, D.; Koc, S.; Davran, F.; Demir, I.; Suleymanlar, G. The assessment of carotid intima media thickness and serum paraoxonase-1 activity in Helicobacter pylori positive subjects. Lipids Health Dis. 2010, 9, 92. [Google Scholar] [CrossRef]
- Demirpençe, O.; Sevim, B.; Yıldırım, M.; Ayan Nurlu, N.; Mert, D.; Evliyaoğlu, O. Serum paraoxonase, TAS, TOS and ceruloplasmin in brucellosis. Int. J. Clin. Exp. Med. 2014, 7, 1592–1597. [Google Scholar]
- Selek, S.; Cosar, N.; Kocyigit, A.; Erel, O.; Aksoy, N.; Gencer, M.; Gunak, F.; Aslan, M. PON1 activity and total oxidant status in patients with active pulmonary tuberculosis. Clin. Biochem. 2008, 41, 140–144. [Google Scholar] [CrossRef]
- Coll, B.; van Wijk, J.P.; Parra, S.; Castro Cabezas, M.; Hoepelman, I.M.; Alonso-Villaverde, C.; de Koning, E.J.; Camps, J.; Ferre, N.; Rabelink, T.J.; et al. Effects of rosiglitazone and metformin on postprandial paraoxonase-1 and monocyte chemoattractant protein-1 in human immunodeficiency virus-infected patients with lipodystrophy. Eur. J. Pharmacol. 2006, 544, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Parra, S.; Marsillach, J.; Aragonès, G.; Rull, A.; Beltrán-Debón, R.; Alonso-Villaverde, C.; Joven, J.; Camps, J. Methodological constraints in interpreting serum paraoxonase-1 activity measurements: An example from a study in HIV-infected patients. Lipids Health Dis. 2010, 9, 32. [Google Scholar] [CrossRef]
- Parra, S.; Alonso-Villaverde, C.; Coll, B.; Ferré, N.; Marsillach, J.; Aragonès, G.; Mackness, M.; Mackness, B.; Masana, L.; Joven, J.; et al. Serum paraoxonase-1 activity and concentration are influenced by human immunodeficiency virus infection. Atherosclerosis 2007, 194, 175–181. [Google Scholar] [CrossRef]
- Parra, S.; Marsillach, J.; Aragonés, G.; Beltrán, R.; Montero, M.; Coll, B.; Mackness, B.; Mackness, M.; Alonso-Villaverde, C.; Joven, J.; et al. Paraoxonase-1 gene haplotypes are associated with metabolic disturbances, atherosclerosis, and immunologic outcome in HIV-infected patients. J. Infect. Dis. 2010, 201, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Viladés, C.; Broch, M.; Plana, M.; Domingo, P.; Alonso-Villaverde, C.; Pedrol, E.; Knobel, H.; Dalmau, D.; Peraire, J.; Gutiérrez, C.; et al. Effect of genetic variants of CCR2 and CCL2 on the natural history of HIV-1 infection: CCL2-2518GG is overrepresented in a cohort of Spanish HIV-1-infected subjects. J. Acquir. Immune Defic. Syndr. 2007, 44, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Villaverde, C.; Aragonès, G.; Beltrán-Debón, R.; Fernández-Sender, L.; Rull, A.; Rodríguez-Sanabria, F.; Marsillach, J.; Pardo-Reche, P.; Camps, J.; Joven, J. Host—Pathogen interactions in the development of metabolic disturbances and atherosclerosis in HIV infection: The role of CCL2 genetic variants. Cytokine 2010, 51, 251–258. [Google Scholar] [CrossRef]
- Aragonès, G.; Beltrán-Debón, R.; Rull, A.; Rodríguez-Sanabria, F.; Fernández-Sender, L.; Camps, J.; Joven, J.; Alonso-Villaverde, C. Human immunodeficiency virus-infection induces major changes in high-density lipoprotein particle size distribution and composition: The effect of antiretroviral treatment and disease severity. Clin. Chem. Lab. Med. 2010, 48, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- Coll, B.; Parra, S.; Alonso-Villaverde, C.; Aragonés, G.; Montero, M.; Camps, J.; Joven, J.; Masana, L. The role of immunity and inflammation in the progression of atherosclerosis in patients with HIV infection. Stroke 2007, 38, 2477–2484. [Google Scholar] [CrossRef][Green Version]
- Joven, J.; Coll, B.; Tous, M.; Ferré, N.; Alonso-Villaverde, C.; Parra, S.; Camps, J. The influence of HIV infection on the correlation between plasma concentrations of monocyte chemoattractant protein-1 and carotid atherosclerosis. Clin. Chim. Acta. 2006, 368, 114–119. [Google Scholar] [CrossRef]
- Williams, M.E.; Joska, J.A.; Amod, A.R.; Paul, R.H.; Stein, D.J.; Ipser, J.C.; Naudé, P.J.W. The association of peripheral immune markers with brain cortical thickness and surface area in South African people living with HIV. J. Neurovirol. 2020, 26, 908–919. [Google Scholar] [CrossRef] [PubMed]
- Force, G.; Ghout, I.; Ropers, J.; Carcelain, G.; Marigot-Outtandy, D.; Hahn, V.; Darchy, N.; Defferriere, H.; Bouaziz-Amar, E.; Carlier, R.; et al. Improvement of HIV-associated neurocognitive disorders after antiretroviral therapy intensification: The Neuro+3 study. J. Antimicrob. Chemother. 2021, 76, 743–752. [Google Scholar] [CrossRef]
- Zhang, J.; Gong, X.; Xiong, H. Significant higher-level C-C motif chemokine ligand 2/3 and chemotactic power in cerebral white matter than grey matter in rat and human. Eur. J. Neurosci. 2021. [Google Scholar] [CrossRef]
- Hermes, D.J.; Yadav-Samudrala, B.J.; Xu, C.; Paniccia, J.E.; Meeker, R.B.; Armstrong, M.L.; Reisdorph, N.; Cravatt, B.F.; Mackie, K.; Lichtman, A.H.; et al. GPR18 drives FAAH inhibition-induced neuroprotection against HIV-1 Tat-induced neurodegeneration. Exp. Neurol. 2021, 341, 113699. [Google Scholar] [CrossRef]
- Karsen, H.; Binici, I.; Sunnetcioglu, M.; Baran, A.I.; Ceylan, M.R.; Selek, S.; Celik, H. Association of paraoxonase activity and atherosclerosis in patients with chronic hepatitis B. Afr. Health Sci. 2012, 12, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Duygu, F.; Tekin Koruk, S.; Aksoy, N. Serum paraoxonase and arylesterase activities in various forms of hepatitis B virus infection. J. Clin. Lab. Anal. 2011, 25, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Schulpis, K.H.; Barzeliotou, A.; Papadakis, M.; Rodolakis, A.; Antsaklis, A.; Papassotiriou, I.; Vlachos, G.D. Maternal chronic hepatitis B virus is implicated with low neonatal paraoxonase/arylesterase activities. Clin. Biochem. 2008, 41, 282–287. [Google Scholar] [CrossRef]
- Ferré, N.; Marsillach, J.; Camps, J.; Rull, A.; Coll, B.; Tous, M.; Joven, J. Genetic association of paraoxonase-1 polymorphisms and chronic hepatitis C virus infection. Clin. Chim. Acta. 2005, 361, 206–210. [Google Scholar] [CrossRef]
- Ali, E.M.; Shehata, H.H.; Ali-Labib, R.; Esmail Zahra, L.M. Oxidant and antioxidant of arylesterase and paraoxonase as biomarkers in patients with hepatitis C virus. Clin. Biochem. 2009, 42, 1394–1400. [Google Scholar] [CrossRef]
- Chandrasena, L.G.; Peiris, H.; Kamani, J.; Wanigasuriya, P.; Jayaratne, S.D.; Wijayasiri, W.A.; Wijesekara, G.U. Antioxidants in patients with dengue viral infection. Southeast Asian J. Trop. Med. Public Health 2014, 45, 1015–1022. [Google Scholar]
- Peltenburg, N.C.; Schoeman, J.C.; Hou, J.; Mora, F.; Harms, A.C.; Lowe, S.H.; Bierau, J.; Bakker, J.A.; Verbon, A.; Hankemeier, T.; et al. Persistent metabolic changes in HIV-infected patients during the first year of combination antiretroviral therapy. Sci. Rep. 2018, 8, 16947. [Google Scholar] [CrossRef]
- Rodríguez-Gallego, E.; Gómez, J.; Domingo, P.; Ferrando-Martínez, S.; Peraire, J.; Viladés, C.; Veloso, S.; López-Dupla, M.; Beltrán-Debón, R.; Alba, V.; et al. Circulating metabolomic profile can predict dyslipidemia in HIV patients undergoing antiretroviral therapy. Atherosclerosis 2018, 273, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Okeke, N.L.; Craig, D.M.; Muehlbauer, M.J.; Ilkayeva, O.; Clement, M.E.; Naggie, S.; Shah, S.H. Metabolites predict cardiovascular disease events in persons living with HIV: A pilot case-control study. Metabolomics 2018, 14, 23. [Google Scholar] [CrossRef]
- Begue, F.; Tanaka, S.; Mouktadi, Z.; Rondeau, P.; Veeren, B.; Diotel, N.; Tran-Dinh, A.; Robert, T.; Vélia, E.; Mavingui, P.; et al. Altered high-density lipoprotein composition and functions during severe COVID-19. Sci. Rep. 2021, 11, 2291. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.H.; Kim, J.R.; Lee, I.C.; Kwon, H.J. Native high-density lipoproteins (HDL) with higher paraoxonase exerts a potent antiviral effect against SARS-CoV-2 (COVID-19), while glycated HDL lost the antiviral activity. Antioxidants 2021, 10, 209. [Google Scholar] [CrossRef]
- Cava, C.; Bertoli, G.; Castiglioni, I. In silico discovery of candidate drugs against Covid-19. Viruses 2020, 12, 404. [Google Scholar] [CrossRef] [PubMed]
- Keller, B.T.; Borchardt, R.T. Adenosine dialdehyde: A potent inhibitor of vaccinia virus multiplication in mouse L929 cells. Mol. Pharmacol. 1987, 31, 485–492. [Google Scholar] [PubMed]
- Haljasmägi, L.; Salumets, A.; Rumm, A.P.; Jürgenson, M.; Krassohhina, E.; Remm, A.; Sein, H.; Kareinen, L.; Vapalahti, O.; Sironen, T.; et al. Longitudinal proteomic profiling reveals increased early inflammation and sustained apoptosis proteins in severe COVID-19. Sci. Rep. 2020, 10, 20533. [Google Scholar] [CrossRef] [PubMed]
- Jøntvedt Jørgensen, M.; Holter, J.C.; Christensen, E.E.; Schjalm, C.; Tonby, K.; Pischke, S.E.; Jenum, S.; Skeie, L.G.; Nur, S.; Lind, A.; et al. Increased interleukin-6 and macrophage chemoattractant protein-1 are associated with respiratory failure in COVID-19. Sci. Rep. 2020, 10, 21697. [Google Scholar] [CrossRef]
- Li, S.; Jiang, L.; Li, X.; Lin, F.; Wang, Y.; Li, B.; Jiang, T.; An, W.; Liu, S.; Liu, H.; et al. Clinical and pathological investigation of patients with severe COVID-19. JCI Insight 2020, 5, e138070. [Google Scholar] [CrossRef]
- Xi, X.; Guo, Y.; Zhu, M.; Wei, Y.; Li, G.; Du, B.; Wang, Y. Higher expression of monocyte chemotactic protein 1 in mild COVID-19 patients might be correlated with inhibition of Type I IFN signaling. Virol. J. 2021, 18, 12. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Mears, J.R.; Shakib, L.; Beynor, J.I.; Shanaj, S.; Korsunsky, I.; Nathan, A.; Donlin, L.T.; Raychaudhuri, S.; Accelerating Medicines Partnership Rheumatoid Arthritis and Systemic Lupus Erythematosus (AMP RA/SLE) Consortium. IFN-γ and TNF-α drive a CXCL10+ CCL2+ macrophage phenotype expanded in severe COVID-19 lungs and inflammatory diseases with tissue inflammation. Genome Med. 2021, 13, 64. [Google Scholar] [CrossRef]
- Chi, Y.; Ge, Y.; Wu, B.; Zhang, W.; Wu, T.; Wen, T.; Liu, J.; Guo, X.; Huang, C.; Jiao, Y.; et al. Serum cytokine and chemokine profile in relation to the severity of coronavirus disease 2019 in China. J. Infect. Dis. 2020, 222, 746–754. [Google Scholar] [CrossRef]
- Xiong, Y.; Liu, Y.; Cao, L.; Wang, D.; Guo, M.; Jiang, A.; Guo, D.; Hu, W.; Yang, J.; Tang, Z.; et al. Transcriptomic characteristics of bronchoalveolar lavage fluid and peripheral blood mononuclear cells in COVID-19 patients. Emerg. Microbes Infect. 2020, 9, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.; Chan, J.F.; Wang, Y.; Yuen, T.T.; Chai, Y.; Hou, Y.; Shuai, H.; Yang, D.; Hu, B.; Huang, X.; et al. Comparative replication and immune activation profiles of SARS-CoV-2 and SARS-CoV in human lungs: An ex vivo study with implications for the pathogenesis of COVID-19. Clin. Infect. Dis. 2020, 71, 1400–1409. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Lu, L.; Cao, W.; Li, T. Hypothesis for potential pathogenesis of SARS-CoV-2 infection-a review of immune changes in patients with viral pneumonia. Emerg. Microbes Infect. 2020, 9, 727–732. [Google Scholar] [CrossRef]
- Ferraro, E.; Germanò, M.; Mollace, R.; Mollace, V.; Malara, N. HIF-1, the Warburg effect, and macrophage/microglia polarization potential role in COVID-19 pathogenesis. Oxid. Med. Cell. Longev. 2021, 2021, 8841911. [Google Scholar] [CrossRef] [PubMed]
- Burtscher, J.; Burtscher, M.; Millet, G.P. The central role of mitochondrial fitness on antiviral defenses: An advocacy for physical activity during the COVID-19 pandemic. Redox Biol. 2021, 43, 101976. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camps, J.; Castañé, H.; Rodríguez-Tomàs, E.; Baiges-Gaya, G.; Hernández-Aguilera, A.; Arenas, M.; Iftimie, S.; Joven, J. On the Role of Paraoxonase-1 and Chemokine Ligand 2 (C-C motif) in Metabolic Alterations Linked to Inflammation and Disease. A 2021 Update. Biomolecules 2021, 11, 971. https://doi.org/10.3390/biom11070971
Camps J, Castañé H, Rodríguez-Tomàs E, Baiges-Gaya G, Hernández-Aguilera A, Arenas M, Iftimie S, Joven J. On the Role of Paraoxonase-1 and Chemokine Ligand 2 (C-C motif) in Metabolic Alterations Linked to Inflammation and Disease. A 2021 Update. Biomolecules. 2021; 11(7):971. https://doi.org/10.3390/biom11070971
Chicago/Turabian StyleCamps, Jordi, Helena Castañé, Elisabet Rodríguez-Tomàs, Gerard Baiges-Gaya, Anna Hernández-Aguilera, Meritxell Arenas, Simona Iftimie, and Jorge Joven. 2021. "On the Role of Paraoxonase-1 and Chemokine Ligand 2 (C-C motif) in Metabolic Alterations Linked to Inflammation and Disease. A 2021 Update" Biomolecules 11, no. 7: 971. https://doi.org/10.3390/biom11070971
APA StyleCamps, J., Castañé, H., Rodríguez-Tomàs, E., Baiges-Gaya, G., Hernández-Aguilera, A., Arenas, M., Iftimie, S., & Joven, J. (2021). On the Role of Paraoxonase-1 and Chemokine Ligand 2 (C-C motif) in Metabolic Alterations Linked to Inflammation and Disease. A 2021 Update. Biomolecules, 11(7), 971. https://doi.org/10.3390/biom11070971








