Circadian Blood Pressure Rhythm in Cardiovascular and Renal Health and Disease
Abstract
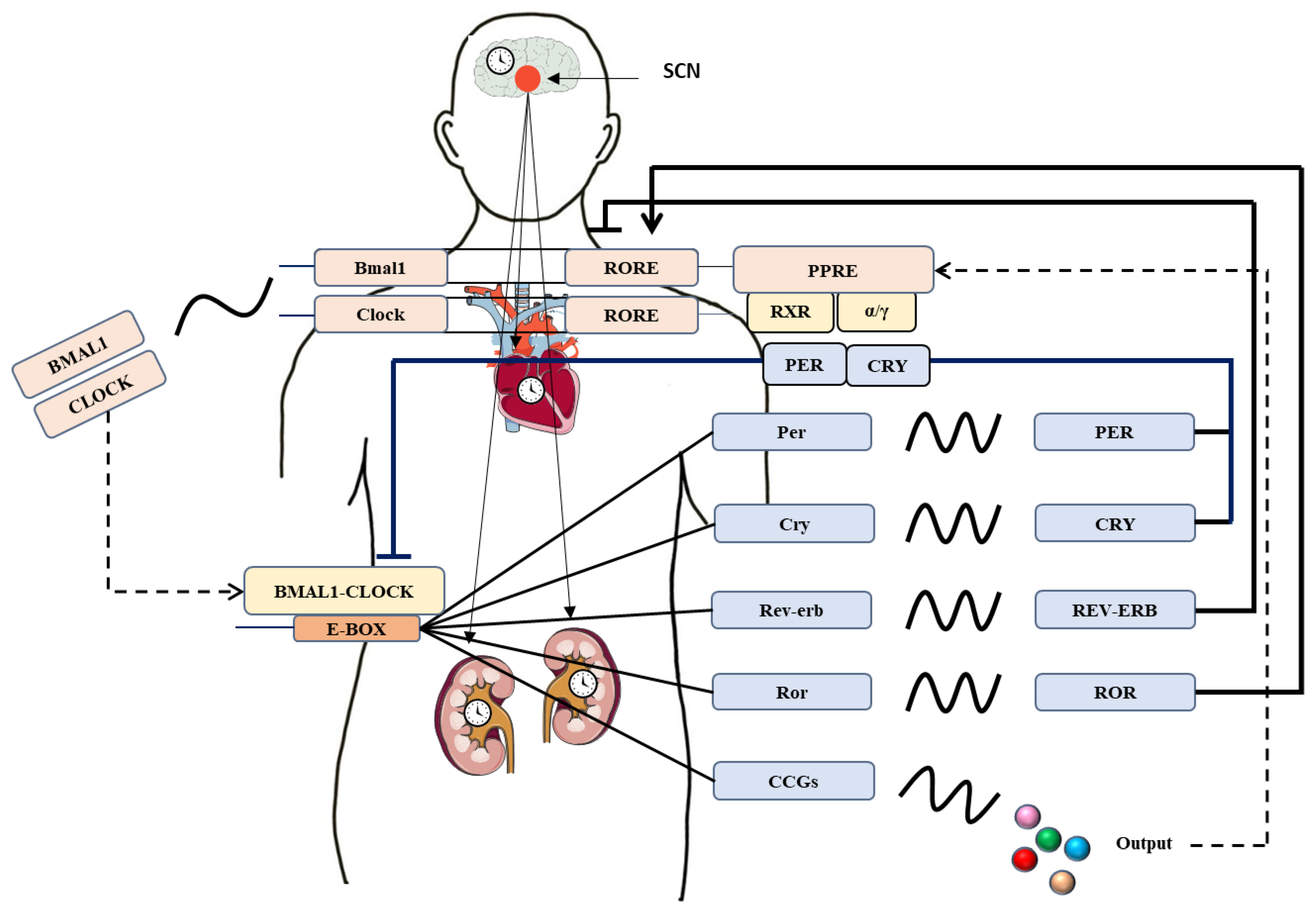
1. Introduction of Circadian BP Rhythm
2. Canonical Clock Genes and BP Regulation
2.1. Bmal1
2.2. Per
2.3. Clock and Cry
3. Disruption of Circadian BP Rhythm and Cardiovascular Disease
4. Disruption of Circadian BP Rhythm and Renal Damage
5. Chronotherapy of Anti-Hypertensive Medications
5.1. ACEI
5.2. ARB
5.3. Other Anti-Hypertension Medications
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Roenneberg, T.; Merrow, M. Circadian clocks—The fall and rise of physiology. Nat. Rev. Mol. Cell Biol. 2005, 6, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Harmer, S.L.; Panda, S.; Kay, S.A. Molecular bases of circadian rhythms. Annu. Rev. Cell Dev. Biol. 2001, 17, 215–253. [Google Scholar] [CrossRef]
- Lowrey, P.L.; Takahashi, J.S. Genetics of circadian rhythms in Mammalian model organisms. Adv. Genet. 2011, 74, 175–230. [Google Scholar] [CrossRef]
- Dibner, C.; Schibler, U.; Albrecht, U. The mammalian circadian timing system: Organization and coordination of central and peripheral clocks. Annu. Rev. Physiol. 2010, 72, 517–549. [Google Scholar] [CrossRef]
- Zhang, D.; Pollock, D.M. Diurnal regulation of renal electrolyte excretion: The role of paracrine factors. Annu. Rev. Physiol. 2019. [Google Scholar] [CrossRef]
- Lutshumba, J.; Liu, S.; Zhong, Y.; Hou, T.; Daugherty, A.; Lu, H.; Guo, Z.; Gong, M.C. Deletion of BMAL1 in smooth muscle cells protects mice from abdominal aortic aneurysms. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 1063–1075. [Google Scholar] [CrossRef]
- Duong, H.A.; Robles, M.S.; Knutti, D.; Weitz, C.J. A molecular mechanism for circadian clock negative feedback. Science 2011, 332, 1436–1439. [Google Scholar] [CrossRef]
- Guillaumond, F.; Dardente, H.; Giguere, V.; Cermakian, N. Differential control of Bmal1 circadian transcription by REV-ERB and ROR nuclear receptors. J. Biol. Rhythm. 2005, 20, 391–403. [Google Scholar] [CrossRef]
- Wang, N.; Yang, G.; Jia, Z.; Zhang, H.; Aoyagi, T.; Soodvilai, S.; Symons, J.D.; Schnermann, J.B.; Gonzalez, F.J.; Litwin, S.E.; et al. Vascular PPARgamma controls circadian variation in blood pressure and heart rate through Bmal1. Cell Metab. 2008, 8, 482–491. [Google Scholar] [CrossRef]
- Yang, X.; Downes, M.; Yu, R.T.; Bookout, A.L.; He, W.; Straume, M.; Mangelsdorf, D.J.; Evans, R.M. Nuclear receptor expression links the circadian clock to metabolism. Cell 2006, 126, 801–810. [Google Scholar] [CrossRef]
- Zhang, R.; Lahens, N.F.; Ballance, H.I.; Hughes, M.E.; Hogenesch, J.B. A circadian gene expression atlas in mammals: Implications for biology and medicine. Proc. Natl. Acad. Sci. USA 2014, 111, 16219–16224. [Google Scholar] [CrossRef] [PubMed]
- Smolensky, M.H.; Hermida, R.C.; Portaluppi, F. Circadian mechanisms of 24-hour blood pressure regulation and patterning. Sleep Med. Rev. 2017, 33, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Scheer, F.A.; Van Montfrans, G.A.; van Someren, E.J.; Mairuhu, G.; Buijs, R.M. Daily nighttime melatonin reduces blood pressure in male patients with essential hypertension. Hypertension 2004, 43, 192–197. [Google Scholar] [CrossRef]
- Portaluppi, F.; Montanari, L.; Bagni, B.; degli Uberti, E.; Trasforini, G.; Margutti, A. Circadian rhythms of atrial natriuretic peptide, blood pressure and heart rate in normal subjects. Cardiology 1989, 76, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Gordon, R.D.; Wolfe, L.K.; Island, D.P.; Liddle, G.W. A diurnal rhythm in plasma renin activity in man. J. Clin. Investig. 1966, 45, 1587–1592. [Google Scholar] [CrossRef] [PubMed]
- Sica, D.A. What are the influences of salt, potassium, the sympathetic nervous system, and the renin-angiotensin system on the circadian variation in blood pressure? Blood Press. Monit. 1999, 4 (Suppl. 2), S9–S16. [Google Scholar]
- Veglio, F.; Pietrandrea, R.; Ossola, M.; Vignani, A.; Angeli, A. Circadian rhythm of the angiotensin converting enzyme (ACE) activity in serum of healthy adult subjects. Chronobiologia 1987, 14, 21–25. [Google Scholar] [PubMed]
- Li, H.; Sun, N.L.; Wang, J.; Liu, A.J.; Su, D.F. Circadian expression of clock genes and angiotensin II type 1 receptors in suprachiasmatic nuclei of sinoaortic-denervated rats. Acta Pharmacol. Sin. 2007, 28, 484–492. [Google Scholar] [CrossRef]
- Liebau, H.; Manitius, J. Diurnal and daily variations of PRA, plasma catecholamines and blood pressure in normotensive and hypertensive man. Contrib. Nephrol. 1982, 30, 57–63. [Google Scholar] [CrossRef]
- Bode-Boger, S.M.; Boger, R.H.; Kielstein, J.T.; Loffler, M.; Schaffer, J.; Frolich, J.C. Role of endogenous nitric oxide in circadian blood pressure regulation in healthy humans and in patients with hypertension or atherosclerosis. J. Investig. Med. 2000, 48, 125–132. [Google Scholar]
- Anea, C.B.; Cheng, B.; Sharma, S.; Kumar, S.; Caldwell, R.W.; Yao, L.; Ali, M.I.; Merloiu, A.M.; Stepp, D.W.; Black, S.M.; et al. Increased superoxide and endothelial NO synthase uncoupling in blood vessels of Bmal1-knockout mice. Circ. Res. 2012, 111, 1157–1165. [Google Scholar] [CrossRef]
- Katinas, G.S.; Halberg, F.; Cornelissen, G.; Hawkins, D.; Bueva, M.V.; Korzhevsky, D.E.; Sapozhnikova, L.R.; Rhodus, N.; Schaffer, E. About 8- and approximately 84-h rhythms in endotheliocytes as in endothelin-1 and effect of trauma. Peptides 2001, 22, 647–659. [Google Scholar] [CrossRef]
- Thosar, S.S.; Berman, A.M.; Herzig, M.X.; McHill, A.W.; Bowles, N.P.; Swanson, C.M.; Clemons, N.A.; Butler, M.P.; Clemons, A.A.; Emens, J.S.; et al. Circadian rhythm of vascular function in midlife adults. Arterioscler. Thromb. Vasc. Biol. 2019, 39, 1203–1211. [Google Scholar] [CrossRef]
- Hwang, Y.S.; Hsieh, T.J.; Lee, Y.J.; Tsai, J.H. Circadian rhythm of urinary endothelin-1 excretion in mild hypertensive patients. Am. J. Hypertens. 1998, 11, 1344–1351. [Google Scholar] [CrossRef][Green Version]
- Soliman, R.H.; Johnston, J.G.; Gohar, E.Y.; Taylor, C.M.; Pollock, D.M. Greater natriuretic response to ENaC inhibition in male versus female Sprague-Dawley rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2020, 318, R418–R427. [Google Scholar] [CrossRef] [PubMed]
- Rautureau, Y.; Schiffrin, E.L. Endothelin in hypertension: An update. Curr. Opin. Nephrol. Hypertens. 2012, 21, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Cuspidi, C.; Tadic, M.; Sala, C.; Gherbesi, E.; Grassi, G.; Mancia, G. Blood pressure non-dipping and obstructive sleep apnea syndrome: A meta-analysis. J. Clin. Med. 2019, 8, 1367. [Google Scholar] [CrossRef]
- Kario, K.; Schwartz, J.E.; Pickering, T.G. Ambulatory physical activity as a determinant of diurnal blood pressure variation. Hypertension 1999, 34, 685–691. [Google Scholar] [CrossRef][Green Version]
- Riegel, B.; Daus, M.; Lozano, A.J.; Malone, S.K.; Patterson, F.; Hanlon, A.L. Shift workers have higher blood pressure medicine use, but only when they are short sleepers: A longitudinal UK biobank study. J. Am. Heart Assoc. 2019, 8, e013269. [Google Scholar] [CrossRef] [PubMed]
- Motohashi, Y.; Higuchi, S.; Maeda, A.; Liu, Y.; Yuasa, T.; Motohashi, K.; Nakamura, K. Alteration of circadian time structure of blood pressure caused by night shift schedule. Occup. Med. 1998, 48, 523–528. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gumenyuk, V.; Roth, T.; Drake, C.L. Circadian phase, sleepiness, and light exposure assessment in night workers with and without shift work disorder. Chronobiol. Int. 2012, 29, 928–936. [Google Scholar] [CrossRef]
- Morris, C.J.; Purvis, T.E.; Mistretta, J.; Hu, K.; Scheer, F. Circadian misalignment increases c-reactive protein and blood pressure in chronic shift workers. J. Biol. Rhythm. 2017, 32, 154–164. [Google Scholar] [CrossRef]
- Yang, G.; Chen, L.; Grant, G.R.; Paschos, G.; Song, W.L.; Musiek, E.S.; Lee, V.; McLoughlin, S.C.; Grosser, T.; Cotsarelis, G.; et al. Timing of expression of the core clock gene Bmal1 influences its effects on aging and survival. Sci. Transl. Med. 2016, 8, 324ra316. [Google Scholar] [CrossRef]
- Yang, G.; Jia, Z.; Aoyagi, T.; McClain, D.; Mortensen, R.M.; Yang, T. Systemic PPARgamma deletion impairs circadian rhythms of behavior and metabolism. PLoS ONE 2012, 7, e38117. [Google Scholar] [CrossRef]
- Zhang, D.; Colson, J.C.; Jin, C.; Becker, B.K.; Rhoads, M.K.; Pati, P.; Neder, T.H.; King, M.A.; Valcin, J.A.; Tao, B.; et al. Timing of food intake drives the circadian rhythm of blood pressure. Function 2021, 2, zqaa034. [Google Scholar] [CrossRef]
- D’Elia, L.; La Fata, E.; Giaquinto, A.; Strazzullo, P.; Galletti, F. Effect of dietary salt restriction on central blood pressure: A systematic review and meta-analysis of the intervention studies. J. Clin. Hypertens. 2020, 22, 814–825. [Google Scholar] [CrossRef]
- Wang, Q.; Li, C.; Guo, Y.; Barnett, A.G.; Tong, S.; Phung, D.; Chu, C.; Dear, K.; Wang, X.; Huang, C. Environmental ambient temperature and blood pressure in adults: A systematic review and meta-analysis. Sci. Total Environ. 2017, 575, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.Y.; Lai, Y.A.; Hsieh, H.H.; Lai, J.S.; Liu, C.S. Effects of environmental noise exposure on ambulatory blood pressure in young adults. Environ. Res. 2009, 109, 900–905. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.M.; Cheng, Y.; Kapoor, S.; Reilly, D.; Price, T.S.; Fitzgerald, G.A. Circadian variation of blood pressure and the vascular response to asynchronous stress. Proc. Natl. Acad. Sci. USA 2007, 104, 3450–3455. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.G.; Speed, J.S.; Becker, B.K.; Kasztan, M.; Soliman, R.H.; Rhoads, M.K.; Tao, B.; Jin, C.; Geurts, A.M.; Hyndman, K.A.; et al. Diurnal control of blood pressure is uncoupled from sodium excretion. Hypertension 2020, 75, 1624–1634. [Google Scholar] [CrossRef] [PubMed]
- Zuber, A.M.; Centeno, G.; Pradervand, S.; Nikolaeva, S.; Maquelin, L.; Cardinaux, L.; Bonny, O.; Firsov, D. Molecular clock is involved in predictive circadian adjustment of renal function. Proc. Natl. Acad. Sci. USA 2009, 106, 16523–16528. [Google Scholar] [CrossRef]
- Sei, H.; Oishi, K.; Chikahisa, S.; Kitaoka, K.; Takeda, E.; Ishida, N. Diurnal amplitudes of arterial pressure and heart rate are dampened in clock mutant mice and adrenalectomized mice. Endocrinology 2008, 149, 3576–3580. [Google Scholar] [CrossRef][Green Version]
- Stow, L.R.; Richards, J.; Cheng, K.Y.; Lynch, I.J.; Jeffers, L.A.; Greenlee, M.M.; Cain, B.D.; Wingo, C.S.; Gumz, M.L. The circadian protein period 1 contributes to blood pressure control and coordinately regulates renal sodium transport genes. Hypertension 2012, 59, 1151–1156. [Google Scholar] [CrossRef] [PubMed]
- Solocinski, K.; Holzworth, M.; Wen, X.; Cheng, K.Y.; Lynch, I.J.; Cain, B.D.; Wingo, C.S.; Gumz, M.L. Desoxycorticosterone pivalate-salt treatment leads to non-dipping hypertension in Per1 knockout mice. Acta Physiol. 2017, 220, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Pati, P.; Fulton, D.J.; Bagi, Z.; Chen, F.; Wang, Y.; Kitchens, J.; Cassis, L.A.; Stepp, D.W.; Rudic, R.D. Low-salt diet and circadian dysfunction synergize to induce angiotensin II-dependent hypertension in mice. Hypertension 2016, 67, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Vukolic, A.; Antic, V.; Van Vliet, B.N.; Yang, Z.; Albrecht, U.; Montani, J.P. Role of mutation of the circadian clock gene Per2 in cardiovascular circadian rhythms. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R627–R634. [Google Scholar] [CrossRef]
- Doi, M.; Takahashi, Y.; Komatsu, R.; Yamazaki, F.; Yamada, H.; Haraguchi, S.; Emoto, N.; Okuno, Y.; Tsujimoto, G.; Kanematsu, A.; et al. Salt-sensitive hypertension in circadian clock-deficient Cry-null mice involves dysregulated adrenal Hsd3b6. Nat. Med. 2010, 16, 67–74. [Google Scholar] [CrossRef]
- Xie, Z.; Su, W.; Liu, S.; Zhao, G.; Esser, K.; Schroder, E.A.; Lefta, M.; Stauss, H.M.; Guo, Z.; Gong, M.C. Smooth-muscle BMAL1 participates in blood pressure circadian rhythm regulation. J. Clin. Investig. 2015, 125, 324–336. [Google Scholar] [CrossRef]
- Chang, L.; Xiong, W.; Zhao, X.; Fan, Y.; Guo, Y.; Garcia-Barrio, M.; Zhang, J.; Jiang, Z.; Lin, J.D.; Chen, Y.E. Bmal1 in perivascular adipose tissue regulates resting-phase blood pressure through transcriptional regulation of angiotensinogen. Circulation 2018, 138, 67–79. [Google Scholar] [CrossRef]
- Westgate, E.J.; Cheng, Y.; Reilly, D.F.; Price, T.S.; Walisser, J.A.; Bradfield, C.A.; FitzGerald, G.A. Genetic components of the circadian clock regulate thrombogenesis in vivo. Circulation 2008, 117, 2087–2095. [Google Scholar] [CrossRef]
- Nikolaeva, S.; Ansermet, C.; Centeno, G.; Pradervand, S.; Bize, V.; Mordasini, D.; Henry, H.; Koesters, R.; Maillard, M.; Bonny, O.; et al. Nephron-specific deletion of circadian clock gene bmal1 alters the plasma and renal metabolome and impairs drug disposition. J. Am. Soc. Nephrol. 2016, 27, 2997–3004. [Google Scholar] [CrossRef] [PubMed]
- Tokonami, N.; Mordasini, D.; Pradervand, S.; Centeno, G.; Jouffe, C.; Maillard, M.; Bonny, O.; Gachon, F.; Gomez, R.A.; Sequeira-Lopez, M.L.; et al. Local renal circadian clocks control fluid-electrolyte homeostasis and BP. J. Am. Soc. Nephrol. 2014, 25, 1430–1439. [Google Scholar] [CrossRef]
- Zhang, D.; Jin, C.; Obi, I.E.; Rhoads, M.K.; Soliman, R.H.; Sedaka, R.S.; Allan, J.M.; Tao, B.; Speed, J.S.; Pollock, J.S.; et al. Loss of circadian gene Bmal1 in the collecting duct lowers blood pressure in male, but not female, mice. Am. J. Physiol. Ren. Physiol. 2020, 318, F710–F719. [Google Scholar] [CrossRef]
- Crislip, G.R.; Douma, L.G.; Masten, S.H.; Cheng, K.Y.; Lynch, I.J.; Johnston, J.G.; Barral, D.; Glasford, K.B.; Holzworth, M.R.; Verlander, J.W.; et al. Differences in renal BMAL1 contribution to sodium homeostasis and blood pressure control in male and female mice. Am. J. Physiol. Ren. Physiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Ingle, K.A.; Kain, V.; Goel, M.; Prabhu, S.D.; Young, M.E.; Halade, G.V. Cardiomyocyte-specific Bmal1 deletion in mice triggers diastolic dysfunction, extracellular matrix response, and impaired resolution of inflammation. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H1827–H1836. [Google Scholar] [CrossRef]
- Lefta, M.; Campbell, K.S.; Feng, H.Z.; Jin, J.P.; Esser, K.A. Development of dilated cardiomyopathy in Bmal1-deficient mice. Am. J. Physiol. Heart Circ. Physiol. 2012, 303, H475–H485. [Google Scholar] [CrossRef]
- Douma, L.G.; Solocinski, K.; Holzworth, M.R.; Crislip, G.R.; Masten, S.H.; Miller, A.H.; Cheng, K.Y.; Lynch, I.J.; Cain, B.D.; Wingo, C.S.; et al. Female C57BL/6J mice lacking the circadian clock protein PER1 are protected from nondipping hypertension. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2019, 316, R50–R58. [Google Scholar] [CrossRef]
- Douma, L.G.; Holzworth, M.R.; Solocinski, K.; Masten, S.H.; Miller, A.H.; Cheng, K.Y.; Lynch, I.J.; Cain, B.D.; Wingo, C.S.; Gumz, M.L. Renal Na-handling defect associated with PER1-dependent nondipping hypertension in male mice. Am. J. Physiol. Ren. Physiol. 2018, 314, F1138–F1144. [Google Scholar] [CrossRef]
- Alli, A.; Yu, L.; Holzworth, M.; Richards, J.; Cheng, K.Y.; Lynch, I.J.; Wingo, C.S.; Gumz, M.L. Direct and indirect inhibition of the circadian clock protein Per1: Effects on ENaC and blood pressure. Am. J. Physiol. Ren. Physiol. 2019, 316, F807–F813. [Google Scholar] [CrossRef]
- Gumz, M.L.; Stow, L.R.; Lynch, I.J.; Greenlee, M.M.; Rudin, A.; Cain, B.D.; Weaver, D.R.; Wingo, C.S. The circadian clock protein Period 1 regulates expression of the renal epithelial sodium channel in mice. J. Clin. Investig. 2009, 119, 2423–2434. [Google Scholar] [CrossRef] [PubMed]
- Solocinski, K.; Richards, J.; All, S.; Cheng, K.Y.; Khundmiri, S.J.; Gumz, M.L. Transcriptional regulation of NHE3 and SGLT1 by the circadian clock protein Per1 in proximal tubule cells. Am. J. Physiol. Ren. Physiol. 2015, 309, F933–F942. [Google Scholar] [CrossRef]
- Lemmer, B.; Mattes, A.; Bohm, M.; Ganten, D. Circadian blood pressure variation in transgenic hypertensive rats. Hypertension 1993, 22, 97–101. [Google Scholar] [CrossRef]
- Lemmer, B.; Witte, K.; Enzminger, H.; Schiffer, S.; Hauptfleisch, S. Transgenic TGR(mREN2)27 rats as a model for disturbed circadian organization at the level of the brain, the heart, and the kidneys. Chronobiol. Int. 2003, 20, 711–738. [Google Scholar] [CrossRef]
- Zeman, M.; Szantoova, K.; Stebelova, K.; Mravec, B.; Herichova, I. Effect of rhythmic melatonin administration on clock gene expression in the suprachiasmatic nucleus and the heart of hypertensive TGR(mRen2)27 rats. J. Hypertens. Suppl. 2009, 27, S21–S26. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yang, G. Recent advances in circadian rhythms in cardiovascular system. Front. Pharmacol. 2015, 6, 71. [Google Scholar] [CrossRef]
- Ohkubo, T.; Hozawa, A.; Yamaguchi, J.; Kikuya, M.; Ohmori, K.; Michimata, M.; Matsubara, M.; Hashimoto, J.; Hoshi, H.; Araki, T.; et al. Prognostic significance of the nocturnal decline in blood pressure in individuals with and without high 24-h blood pressure: The Ohasama study. J. Hypertens. 2002, 20, 2183–2189. [Google Scholar] [CrossRef]
- Virag, J.A.; Lust, R.M. Circadian influences on myocardial infarction. Front. Physiol. 2014, 5, 422. [Google Scholar] [CrossRef] [PubMed]
- Thune, J.J.; Signorovitch, J.; Kober, L.; Velazquez, E.J.; McMurray, J.J.; Califf, R.M.; Maggioni, A.P.; Rouleau, J.L.; Howlett, J.; Zelenkofske, S.; et al. Effect of antecedent hypertension and follow-up blood pressure on outcomes after high-risk myocardial infarction. Hypertension 2008, 51, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Laukkanen, J.A.; Kurl, S.; Salonen, R.; Lakka, T.A.; Rauramaa, R.; Salonen, J.T. Systolic blood pressure during recovery from exercise and the risk of acute myocardial infarction in middle-aged men. Hypertension 2004, 44, 820–825. [Google Scholar] [CrossRef]
- Safar, M. Relation of pulse pressure and blood pressure reduction to the incidence of myocardial infarction. Hypertension 1994, 24, 368. [Google Scholar] [CrossRef]
- Skrlec, I.; Milic, J.; Steiner, R. The impact of the circadian genes CLOCK and ARNTL on myocardial infarction. J. Clin. Med. 2020, 9, 484. [Google Scholar] [CrossRef]
- Skrlec, I.; Milic, J.; Heffer, M.; Wagner, J.; Peterlin, B. Circadian clock genes and circadian phenotypes in patients with myocardial infarction. Adv. Med. Sci. 2019, 64, 224–229. [Google Scholar] [CrossRef]
- Wei, F.F.; Zhou, Y.; Thijs, L.; Xue, R.; Dong, B.; He, X.; Liang, W.; Wu, Y.; Jiang, J.; Tan, W.; et al. Visit-to-visit blood pressure variability and clinical outcomes in patients with heart failure with preserved ejection fraction. Hypertension 2021. [Google Scholar] [CrossRef] [PubMed]
- Wei, F.F.; Xue, R.; Thijs, L.; Liang, W.; Owusu-Agyeman, M.; He, X.; Staessen, J.A.; Dong, Y.; Liu, C. Associations of left ventricular structure and function with blood pressure in heart failure with preserved ejection fraction: Analysis of the TOPCAT trial. J. Am. Heart Assoc. 2020, 9, e016009. [Google Scholar] [CrossRef] [PubMed]
- Bohm, M.; Young, R.; Jhund, P.S.; Solomon, S.D.; Gong, J.; Lefkowitz, M.P.; Rizkala, A.R.; Rouleau, J.L.; Shi, V.C.; Swedberg, K.; et al. Systolic blood pressure, cardiovascular outcomes and efficacy and safety of sacubitril/valsartan (LCZ696) in patients with chronic heart failure and reduced ejection fraction: Results from PARADIGM-HF. Eur. Heart J. 2017, 38, 1132–1143. [Google Scholar] [CrossRef]
- Tsujimoto, T.; Kajio, H. Low diastolic blood pressure and adverse outcomes in heart failure with preserved ejection fraction. Int. J. Cardiol. 2018, 263, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Gotte, J.; Zittermann, A.; Deutsch, M.A.; Schramm, R.; Bleiziffer, S.; Hata, M.; Gummert, J.F. Daytime variation in aortic valve surgery and clinical outcome: A propensity score-matched analysis. Ann. Thorac. Surg. 2020, 110, 558–566. [Google Scholar] [CrossRef]
- Kenney, P.S.; Nielsen, P.H.; Modrau, I.S. Daytime-dependent cardioprotection in cardiac surgery: A large propensity-matched cohort study. Ann. Thorac. Surg. 2020, 110, 1629–1636. [Google Scholar] [CrossRef]
- Michaud, M.; Beland, V.; Noiseux, N.; Forcillo, J.; Stevens, L.M. Daytime variation of clinical outcome in cardiac surgery: A propensity-matched cohort study. J. Cardiothorac. Vasc. Anesth. 2021. [Google Scholar] [CrossRef]
- Montaigne, D.; Marechal, X.; Modine, T.; Coisne, A.; Mouton, S.; Fayad, G.; Ninni, S.; Klein, C.; Ortmans, S.; Seunes, C.; et al. Daytime variation of perioperative myocardial injury in cardiac surgery and its prevention by Rev-Erbalpha antagonism: A single-centre propensity-matched cohort study and a randomised study. Lancet 2018, 391, 59–69. [Google Scholar] [CrossRef]
- du Fay de Lavallaz, J.; Puelacher, C.; Buse, G.L.; Bolliger, D.; Germanier, D.; Hidvegi, R.; Walter, J.E.; Twerenbold, R.; Strebel, I.; Badertscher, P.; et al. Daytime variation of perioperative myocardial injury in non-cardiac surgery and effect on outcome. Heart 2019, 105, 826–833. [Google Scholar] [CrossRef]
- Voulgaris, A.; Marrone, O.; Bonsignore, M.R.; Steiropoulos, P. Chronic kidney disease in patients with obstructive sleep apnea. A narrative review. Sleep Med. Rev. 2019, 47, 74–89. [Google Scholar] [CrossRef] [PubMed]
- Rucker, D.; Tonelli, M. Cardiovascular risk and management in chronic kidney disease. Nat. Rev. Nephrol. 2009, 5, 287–296. [Google Scholar] [CrossRef]
- Agarwal, R.; Andersen, M.J. Prognostic importance of ambulatory blood pressure recordings in patients with chronic kidney disease. Kidney Int. 2006, 69, 1175–1180. [Google Scholar] [CrossRef]
- Davidson, M.B.; Hix, J.K.; Vidt, D.G.; Brotman, D.J. Association of impaired diurnal blood pressure variation with a subsequent decline in glomerular filtration rate. Arch. Intern. Med. 2006, 166, 846–852. [Google Scholar] [CrossRef]
- Elung-Jensen, T.; Strandgaard, S.; Kamper, A.L. Longitudinal observations on circadian blood pressure variation in chronic kidney disease stages 3-5. Nephrol. Dial. Transplant. 2008, 23, 2873–2878. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Agarwal, R.; Kariyanna, S.S.; Light, R.P. Circadian blood pressure classification scheme and the health of patients with chronic kidney disease. Am. J. Nephrol. 2009, 30, 536–546. [Google Scholar] [CrossRef]
- Agarwal, R.; Kariyanna, S.S.; Light, R.P. Prognostic value of circadian blood pressure variation in chronic kidney disease. Am. J. Nephrol. 2009, 30, 547–553. [Google Scholar] [CrossRef]
- Fukuda, M.; Munemura, M.; Usami, T.; Nakao, N.; Takeuchi, O.; Kamiya, Y.; Yoshida, A.; Kimura, G. Nocturnal blood pressure is elevated with natriuresis and proteinuria as renal function deteriorates in nephropathy. Kidney Int. 2004, 65, 621–625. [Google Scholar] [CrossRef]
- Hussein, A.M.; Malek, H.A.; Saad, M.A. Renoprotective effects of aliskiren on adenine-induced tubulointerstitial nephropathy: Possible underlying mechanisms. Can. J. Physiol. Pharmacol. 2016, 94, 829–837. [Google Scholar] [CrossRef]
- Motohashi, H.; Tahara, Y.; Whittaker, D.S.; Wang, H.B.; Yamaji, T.; Wakui, H.; Haraguchi, A.; Yamazaki, M.; Miyakawa, H.; Hama, K.; et al. The circadian clock is disrupted in mice with adenine-induced tubulointerstitial nephropathy. Kidney Int. 2020, 97, 728–740. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, C.; Liang, Q.; Zheng, F.; Guan, Y.; Yang, G.; Chen, L. Postnatal deletion of Bmal1 in mice protects against obstructive renal fibrosis via suppressing Gli2 transcription. FASEB J. 2021, 35, e21530. [Google Scholar] [CrossRef]
- Rahman, A.; Hasan, A.U.; Nishiyama, A.; Kobori, H. Altered circadian timing system-mediated non-dipping pattern of blood pressure and associated cardiovascular disorders in metabolic and kidney diseases. Int. J. Mol. Sci. 2018, 19, 400. [Google Scholar] [CrossRef]
- Otsuka, K.; Cornelissen, G.; Halberg, F.; Oehlerts, G. Excessive circadian amplitude of blood pressure increases risk of ischaemic stroke and nephropathy. J. Med. Eng. Technol. 1997, 21, 23–30. [Google Scholar] [CrossRef]
- Velasquez, M.T.; Beddhu, S.; Nobakht, E.; Rahman, M.; Raj, D.S. Ambulatory blood pressure in chronic kidney disease: Ready for prime time? Kidney Int. Rep. 2016, 1, 94–104. [Google Scholar] [CrossRef]
- Kuroda, T.; Kario, K.; Hoshide, S.; Hashimoto, T.; Nomura, Y.; Saito, Y.; Mito, H.; Shimada, K. Effects of bedtime vs. morning administration of the long-acting lipophilic angiotensin-converting enzyme inhibitor trandolapril on morning blood pressure in hypertensive patients. Hypertens. Res. 2004, 27, 15–20. [Google Scholar] [CrossRef]
- Middeke, M.; Kluglich, M.; Holzgreve, H. Chronopharmacology of captopril plus hydrochlorothiazide in hypertension: Morning versus evening dosing. Chronobiol. Int. 1991, 8, 506–510. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Fontao, M.J.; Mojon, A.; Alonso, I.; Fernandez, J.R. Administration-time-dependent effects of spirapril on ambulatory blood pressure in uncomplicated essential hypertension. Chronobiol. Int. 2010, 27, 560–574. [Google Scholar] [CrossRef]
- Witte, K.; Weisser, K.; Neubeck, M.; Mutschler, E.; Lehmann, K.; Hopf, R.; Lemmer, B. Cardiovascular effects, pharmacokinetics, and converting enzyme inhibition of enalapril after morning versus evening administration. Clin. Pharmacol. Ther. 1993, 54, 177–186. [Google Scholar] [CrossRef]
- Macchiarulo, C.; Pieri, R.; Mitolo, D.C.; Pirrelli, A. Management of antihypertensive treatment with Lisinopril: A chronotherapeutic approach. Eur. Rev. Med. Pharmacol. Sci. 1999, 3, 269–275. [Google Scholar]
- Palatini, P.; Racioppa, A.; Raule, G.; Zaninotto, M.; Penzo, M.; Pessina, A.C. Effect of timing of administration on the plasma ACE inhibitory activity and the antihypertensive effect of quinapril. Clin. Pharmacol. Ther. 1992, 52, 378–383. [Google Scholar] [CrossRef]
- Palatini, P.; Mos, L.; Motolese, M.; Mormino, P.; Del Torre, M.; Varotto, L.; Pavan, E.; Pessina, A.C. Effect of evening versus morning benazepril on 24-hour blood pressure: A comparative study with continuous intraarterial monitoring. Int. J. Clin. Pharmacol. Ther. Toxicol. 1993, 31, 295–300. [Google Scholar]
- Zaslavskaia, R.M.; Narmanova, O.Z.; Teiblium, M.M.; Kalimurzina, B.S. Time-dependent effects of ramipril in patients with hypertension of 2 stage. Klin. Meditsina 1999, 77, 41–44. [Google Scholar]
- Kohno, I.; Ijiri, H.; Takusagawa, M.; Yin, D.F.; Sano, S.; Ishihara, T.; Sawanobori, T.; Komori, S.; Tamura, K. Effect of imidapril in dipper and nondipper hypertensive patients: Comparison between morning and evening administration. Chronobiol. Int. 2000, 17, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Hermida, R.C.; Calvo, C.; Ayala, D.E.; Dominguez, M.J.; Covelo, M.; Fernandez, J.R.; Mojon, A.; Lopez, J.E. Administration time-dependent effects of valsartan on ambulatory blood pressure in hypertensive subjects. Hypertension 2003, 42, 283–290. [Google Scholar] [CrossRef]
- Hermida, R.C.; Calvo, C.; Ayala, D.E.; Fernandez, J.R.; Covelo, M.; Mojon, A.; Lopez, J.E. Treatment of non-dipper hypertension with bedtime administration of valsartan. J. Hypertens. 2005, 23, 1913–1922. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Fontao, M.J.; Mojon, A.; Fernandez, J.R. Chronotherapy with valsartan/amlodipine fixed combination: Improved blood pressure control of essential hypertension with bedtime dosing. Chronobiol. Int. 2010, 27, 1287–1303. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Fernandez, J.R.; Calvo, C. Comparison of the efficacy of morning versus evening administration of telmisartan in essential hypertension. Hypertension 2007, 50, 715–722. [Google Scholar] [CrossRef]
- Fukuda, M.; Yamanaka, T.; Mizuno, M.; Motokawa, M.; Shirasawa, Y.; Miyagi, S.; Nishio, T.; Yoshida, A.; Kimura, G. Angiotensin II type 1 receptor blocker, olmesartan, restores nocturnal blood pressure decline by enhancing daytime natriuresis. J. Hypertens. 2008, 26, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Bankir, L.; Bochud, M.; Maillard, M.; Bovet, P.; Gabriel, A.; Burnier, M. Nighttime blood pressure and nocturnal dipping are associated with daytime urinary sodium excretion in African subjects. Hypertension 2008, 51, 891–898. [Google Scholar] [CrossRef]
- Del Giorno, R.; Troiani, C.; Gabutti, S.; Stefanelli, K.; Puggelli, S.; Gabutti, L. Impaired daytime urinary sodium excretion impacts nighttime blood pressure and nocturnal dipping at older ages in the general population. Nutrients 2020, 12, 2013. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Chayan, L.; Mojon, A.; Fernandez, J.R. Administration-time-dependent effects of olmesartan on the ambulatory blood pressure of essential hypertension patients. Chronobiol. Int. 2009, 26, 61–79. [Google Scholar] [CrossRef]
- Smolensky, M.H.; Hermida, R.C.; Portaluppi, F. Comparison of the efficacy of morning versus evening administration of olmesartan in uncomplicated essential hypertension. Chronobiol. Int. 2007, 24, 171–181. [Google Scholar] [CrossRef]
- Hermida, R.C.; Fernandez, J.R.; Ayala, D.E.; Mojon, A.; Iglesias, M. Influence of aspirin usage on blood pressure: Dose and administration-time dependencies. Chronobiol. Int. 1997, 14, 619–637. [Google Scholar] [CrossRef] [PubMed]
- Hermida, R.C.; Ayala, D.E.; Iglesias, M.; Mojon, A.; Silva, I.; Ucieda, R.; Fernandez, J.R. Time-dependent effects of low-dose aspirin administration on blood pressure in pregnant women. Hypertension 1997, 30, 589–595. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Mojon, A.; Fernandez, J.R. Ambulatory blood pressure control with bedtime aspirin administration in subjects with prehypertension. Am. J. Hypertens. 2009, 22, 896–903. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Calvo, C.; Lopez, J.E.; Mojon, A.; Rodriguez, M.; Fernandez, J.R. Differing administration time-dependent effects of aspirin on blood pressure in dipper and non-dipper hypertensives. Hypertension 2005, 46, 1060–1068. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Calvo, C.; Lopez, J.E. Aspirin administered at bedtime, but not on awakening, has an effect on ambulatory blood pressure in hypertensive patients. J. Am. Coll. Cardiol. 2005, 46, 975–983. [Google Scholar] [CrossRef]
- Chen, L.; Yang, G.; Zhang, J.; Ren, B.; Tang, S.; Li, X.; FitzGerald, G.A. Time-Dependent Hypotensive Effect of Aspirin in Mice. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 2819–2826. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Mojon, A.; Fernandez, J.R. Chronotherapy with nifedipine GITS in hypertensive patients: Improved efficacy and safety with bedtime dosing. Am. J. Hypertens. 2008, 21, 948–954. [Google Scholar] [CrossRef]
- Hermida, R.C.; Calvo, C.; Ayala, D.E.; Lopez, J.E.; Rodriguez, M.; Chayan, L.; Mojon, A.; Fontao, M.J.; Fernandez, J.R. Dose- and administration time-dependent effects of nifedipine gits on ambulatory blood pressure in hypertensive subjects. Chronobiol. Int. 2007, 24, 471–493. [Google Scholar] [CrossRef] [PubMed]
- Hermida, R.C.; Ayala, D.E.; Mojon, A.; Alonso, I.; Fernandez, J.R. Reduction of morning blood pressure surge after treatment with nifedipine GITS at bedtime, but not upon awakening, in essential hypertension. Blood Press. Monit. 2009, 14, 152–159. [Google Scholar] [CrossRef]
- Hermida, R.C.; Crespo, J.J.; Dominguez-Sardina, M.; Otero, A.; Moya, A.; Rios, M.T.; Sineiro, E.; Castineira, M.C.; Callejas, P.A.; Pousa, L.; et al. Bedtime hypertension treatment improves cardiovascular risk reduction: The Hygia Chronotherapy Trial. Eur. Heart J. 2020, 41, 4565–4576. [Google Scholar] [CrossRef] [PubMed]
- Kreutz, R.; Kjeldsen, S.E.; Burnier, M.; Narkiewicz, K.; Oparil, S.; Mancia, G. Disregard the reported data from the HYGIA project: Blood pressure medication not to be routinely dosed at bedtime. J. Hypertens. 2020, 38, 2144–2145. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Greene, T.; Phillips, R.A.; Agodoa, L.Y.; Bakris, G.L.; Charleston, J.; Contreras, G.; Gabbai, F.; Hiremath, L.; Jamerson, K.; et al. A trial of 2 strategies to reduce nocturnal blood pressure in blacks with chronic kidney disease. Hypertension 2013, 61, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Kohno, I.; Yuzo, S.; Wakasugi, K.; Achiwa, S.; Imanishi, Y.; Cugini, P.; Halberg, F. Antihypertensive individualized therapeutic strategy. Dif. Soc. 1991, 6, 109–124. [Google Scholar]
- Watanabe, Y.; Halberg, F.; Otsuka, K.; Cornelissen, G. Toward a personalized chronotherapy of high blood pressure and a circadian overswing. Clin. Exp. Hypertens. 2013, 35, 257–266. [Google Scholar] [CrossRef]
| Gene Editing | Species | Position | Phenotype of BP | Circadian Rhythmof BP | Reference |
| Bmal1 KO | mouse | global | ↓ | No | [33,39] |
| Bmal1 KO | rat | global | ↓ | Yes | [40] |
| Clock KO | mouse | global | ↓ | Yes | [41] |
| Clock mutant | mouse | global | Day-night variation↓ | Yes | [42] |
| Per1 KO (129/sv) | mouse | global | ↓ | Yes | [43] |
| Per1 KO (C57BL/6) | mouse | global | ↑ | Yes | [44] |
| Per2 KO | mouse | global | = | Yes | [45] |
| Per2 mutant | mouse | global | DBP↓ | Yes | [46] |
| Cry1/2 KO | mouse | global | Salt-sensitive hypertension | No | [47] |
| Bmal1 KO | mouse | smooth muscle | ↓ | Yes | [48] |
| Bmal1 KO | mouse | brown adipocytes | ↓ | No | [49] |
| Bmal1 KO | mouse | endothelial cells | ↓ | Yes | [50] |
| Bmal1 KO | mouse | tubular cells | SBP↓ | Yes | [51] |
| Bmal1 KO | mouse | renin-secreting cells | ↓ | Yes | [52] |
| Bmal1 KO | mouse | collecting duct | ↓ | Yes | [53] |
| Bmal1 KO | mouse | thick ascending limb, distal convoluted tubule, and collecting duct | ↓ | Yes | [54] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Sun, R.; Jiang, T.; Yang, G.; Chen, L. Circadian Blood Pressure Rhythm in Cardiovascular and Renal Health and Disease. Biomolecules 2021, 11, 868. https://doi.org/10.3390/biom11060868
Zhang J, Sun R, Jiang T, Yang G, Chen L. Circadian Blood Pressure Rhythm in Cardiovascular and Renal Health and Disease. Biomolecules. 2021; 11(6):868. https://doi.org/10.3390/biom11060868
Chicago/Turabian StyleZhang, Jiayang, Ruoyu Sun, Tingting Jiang, Guangrui Yang, and Lihong Chen. 2021. "Circadian Blood Pressure Rhythm in Cardiovascular and Renal Health and Disease" Biomolecules 11, no. 6: 868. https://doi.org/10.3390/biom11060868
APA StyleZhang, J., Sun, R., Jiang, T., Yang, G., & Chen, L. (2021). Circadian Blood Pressure Rhythm in Cardiovascular and Renal Health and Disease. Biomolecules, 11(6), 868. https://doi.org/10.3390/biom11060868





