Fitness Shifts the Balance of BDNF and IL-6 from Inflammation to Repair among People with Progressive Multiple Sclerosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Performance Outcomes
2.3. Blood Samples
2.4. Data Analysis
3. Results
3.1. Participant Characteristics
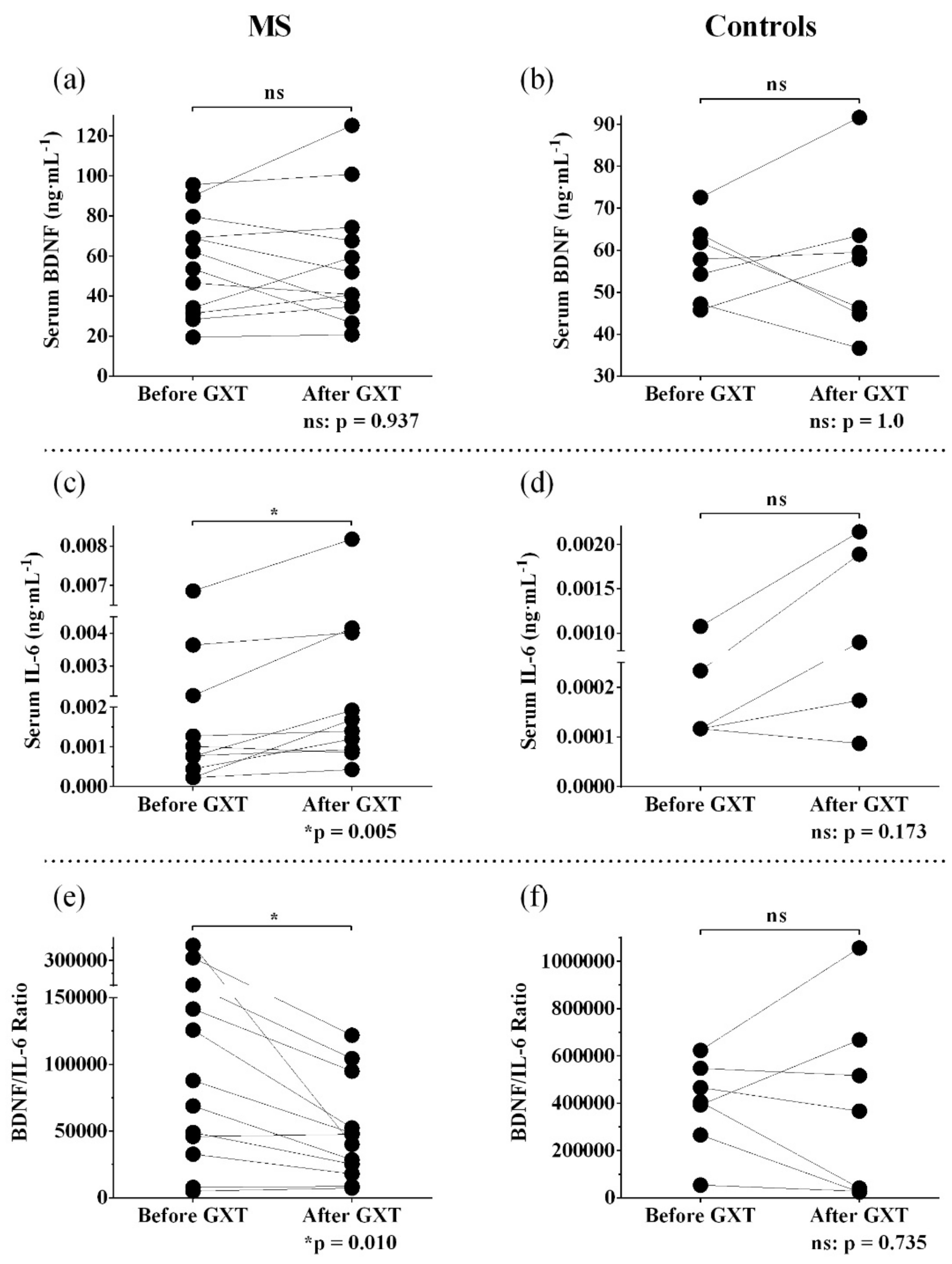
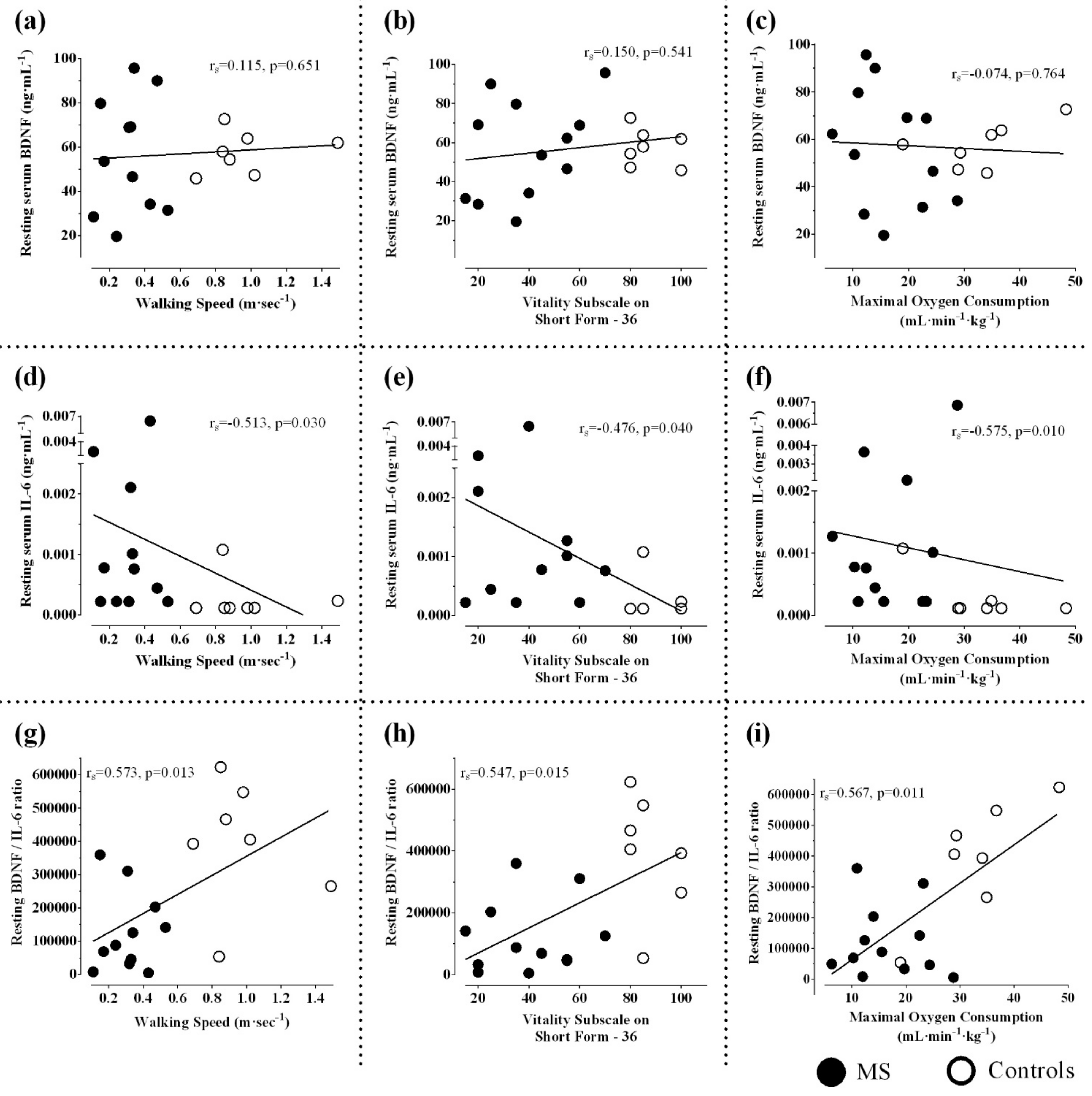
3.2. Resting and Post-Exercise Serum BDNF
3.3. Resting and Post-Exercise Serum IL-6
3.4. Resting and Post-Exercise Serum BDNF/IL-6 ratio
4. Discussion
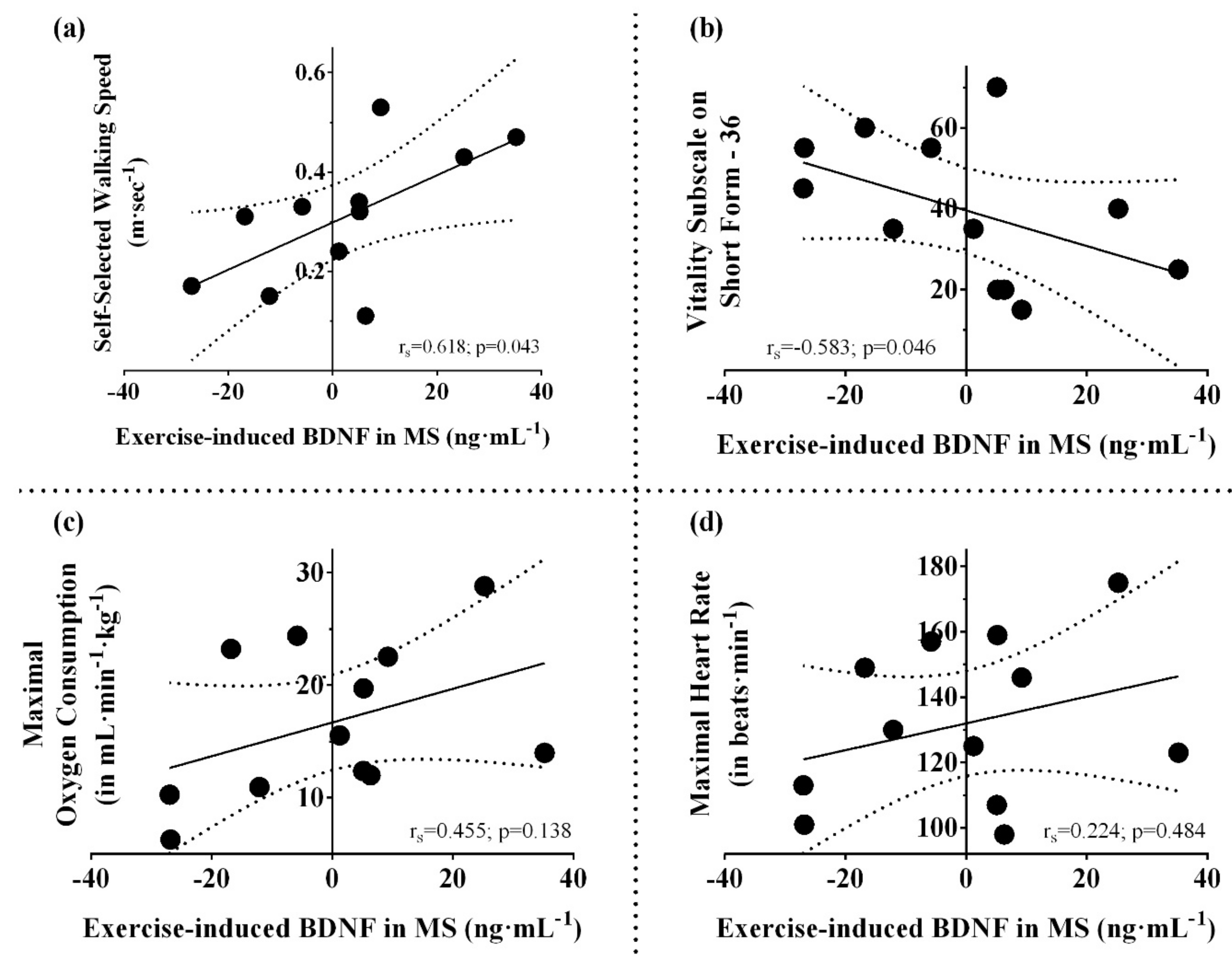
4.1. Aerobic Fitness, Disability, and Expression of Neurotrophins
4.2. Skeletal Muscle and Serum BDNF Induction
4.3. Factors Influencing Cytokine Responses in MS
4.4. Pattern of Serum BDNF/IL-6 Ratio
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Browne, P.; Chandraratna, D.; Angood, C.; Tremlett, H.; Baker, C.; Taylor, B.V.; Thompson, A.J. Atlas of Multiple Sclerosis 2013: A growing global problem with widespread inequity. Neurology 2014, 83, 1022–1024. [Google Scholar] [CrossRef]
- Zaratin, P.; Comi, G.; Coetzee, T.; Ramsey, K.; Smith, K.; Thompson, A.; Panzara, M. Progressive MS Alliance Industry Forum: Maximizing Collective Impact To Enable Drug Development. Trends Pharmacol. Sci. 2016, 37, 808–810. [Google Scholar] [CrossRef]
- Miller, D.H.; Thompson, A.J. Advancing trial design in progressive multiple sclerosis. Mult. Scler. J. 2017, 23, 1571–1572. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.J. A much-needed focus on progression in multiple sclerosis. Lancet Neurol. 2015, 14, 133–135. [Google Scholar] [CrossRef]
- Ploughman, M.; Kelly, L.P. Four birds with one stone? Reparative, neuroplastic, cardiorespiratory, and metabolic benefits of aerobic exercise poststroke. Curr. Opin. Neurol. 2016, 29, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, M.; Bishop, N.C.; Stensel, D.J.; Lindley, M.R.; Mastana, S.S.; Nimmo, M.A. The anti-inflammatory effects of exercise: Mechanisms and implications for the prevention and treatment of disease. Nat. Rev. Immunol. 2011, 11, 607–615. [Google Scholar] [CrossRef]
- Stampanoni Bassi, M.; Iezzi, E.; Mori, F.; Simonelli, I.; Gilio, L.; Buttari, F.; Sica, F.; De Paolis, N.; Mandolesi, G.; Musella, A.; et al. Interleukin-6 Disrupts Synaptic Plasticity and Impairs Tissue Damage Compensation in Multiple Sclerosis. Neurorehabil. Neural Repair 2019, 1545968319868713. [Google Scholar] [CrossRef] [PubMed]
- Hennessy, E.; Gormley, S.; Lopez-Rodriguez, A.B.; Murray, C.; Murray, C.; Cunningham, C. Systemic TNF-alpha produces acute cognitive dysfunction and exaggerated sickness behavior when superimposed upon progressive neurodegeneration. Brain Behav. Immun. 2017, 59, 233–244. [Google Scholar] [CrossRef]
- Mee-Inta, O.; Zhao, Z.W.; Kuo, Y.M. Physical Exercise Inhibits Inflammation and Microglial Activation. Cells 2019, 8, 691. [Google Scholar] [CrossRef]
- Ploughman, M.; Austin, M.W.; Glynn, L.; Corbett, D. The effects of poststroke aerobic exercise on neuroplasticity: A systematic review of animal and clinical studies. Transl. Stroke Res. 2015, 6, 13–28. [Google Scholar] [CrossRef]
- Briken, S.; Rosenkranz, S.C.; Keminer, O.; Patra, S.; Ketels, G.; Heesen, C.; Hellweg, R.; Pless, O.; Schulz, K.-H.; Gold, S.M. Effects of exercise on Irisin, BDNF and IL-6 serum levels in patients with progressive multiple sclerosis. J. Neuroimmunol. 2016, 299, 53–58. [Google Scholar] [CrossRef]
- Castellano, V.; White, L.J. Serum brain-derived neurotrophic factor response to aerobic exercise in multiple sclerosis. J. Neurol. Sci. 2008, 269, 85–91. [Google Scholar] [CrossRef]
- Gold, S.M.; Schulz, K.H.; Hartmann, S.; Mladek, M.; Lang, U.E.; Hellweg, R.; Reer, R.; Braumann, K.M.; Heesen, C. Basal serum levels and reactivity of nerve growth factor and brain-derived neurotrophic factor to standardized acute exercise in multiple sclerosis and controls. J. Neuroimmunol. 2003, 138, 99–105. [Google Scholar] [CrossRef]
- Döring, A.; Pfueller, C.F.; Paul, F.; Dörr, J. Exercise in multiple sclerosis—An integral component of disease management. EPMA J. 2011, 3, 2. [Google Scholar] [CrossRef] [PubMed]
- Barry, A.; Cronin, O.; Ryan, A.M.; Sweeney, B.; Yap, S.M.; O’Toole, O.; Allen, A.P.; Clarke, G.; O’Halloran, K.D.; Downer, E.J. Impact of Exercise on Innate Immunity in Multiple Sclerosis Progression and Symptomatology. Front. Physiol. 2016, 7, 194. [Google Scholar] [CrossRef] [PubMed]
- Waschbisch, A.; Tallner, A.; Pfeifer, K.; Maurer, M. Multiple sclerosis and exercise: Effects of physical activity on the immune system. Nervenarzt 2009, 80, 688–692. [Google Scholar] [CrossRef] [PubMed]
- Castellano, V.; Patel, D.I.; White, L.J. Cytokine responses to acute and chronic exercise in multiple sclerosis. J. Appl. Physiol. 2008, 104, 1697–1702. [Google Scholar] [CrossRef]
- Bansi, J.; Bloch, W.; Gamper, U.; Kesselring, J. Training in MS: Influence of two different endurance training protocols (aquatic versus overland) on cytokine and neurotrophin concentrations during three week randomized controlled trial. Mult. Scler. J. 2013, 19, 613–621. [Google Scholar] [CrossRef]
- Ozkul, C.; Guclu-Gunduz, A.; Irkec, C.; Fidan, I.; Aydin, Y.; Ozkan, T.; Yazici, G. Effect of combined exercise training on serum brain-derived neurotrophic factor, suppressors of cytokine signaling 1 and 3 in patients with multiple sclerosis. J. Neuroimmunol. 2018, 316, 121–129. [Google Scholar] [CrossRef]
- Florindo, M. Inflammatory cytokines and physical activity in multiple sclerosis. Int. Sch. Res. Not. 2014, 2014, 151572. [Google Scholar] [CrossRef]
- Devasahayam, A.J.; Downer, M.B.; Ploughman, M. The effects of aerobic exercise on the recovery of walking ability and neuroplasticity in people with multiple sclerosis: A systematic review of animal and clinical studies. Mult. Scler. Int. 2017, 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Murphy, P.G.; Borthwick, L.A.; Altares, M.; Gauldie, J.; Kaplan, D.; Richardson, P.M. Reciprocal actions of interleukin-6 and brain-derived neurotrophic factor on rat and mouse primary sensory neurons. Eur. J. Neurosci. 2000, 12, 1891–1899. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.P.; Liu, H.B.; Qin, L.Y.; Chen, B.; Zhang, X.X.; Hu, X.P.; Xiao, L.P.; Qin, S.H. Oxidative stress-mediated influence of plasma DPP4 activity to BDNF ratio on mild cognitive impairment in elderly type 2 diabetic patients: Results from the GDMD study in China. Metabolism 2018, 87, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Motl, R.W.; Dlugonski, D.; Suh, Y.; Weikert, M.; Agiovlasitis, S.; Fernhall, B.; Goldman, M. Multiple Sclerosis Walking Scale-12 and oxygen cost of walking. Gait Posture 2010, 31, 506–510. [Google Scholar] [CrossRef]
- Graham, J.E.; Ostir, G.V.; Fisher, S.R.; Ottenbacher, K.J. Assessing walking speed in clinical research: A systematic review. J. Eval. Clin. Pract. 2008, 14, 552–562. [Google Scholar] [CrossRef] [PubMed]
- Kieseier, B.C.; Pozzilli, C. Assessing walking disability in multiple sclerosis. Mult. Scler. J. 2012, 18, 914–924. [Google Scholar] [CrossRef]
- Bisht, B.; Darling, W.G.; White, E.C.; White, K.A.; Shivapour, E.T.; Zimmerman, M.B.; Wahls, T.L. Effects of a multimodal intervention on gait and balance of subjects with progressive multiple sclerosis: A prospective longitudinal pilot study. Degener. Neurol. Neuromuscul. Dis. 2017, 7, 79–93. [Google Scholar] [CrossRef]
- Rossini, P.M.; Pasqualetti, P.; Pozzilli, C.; Grasso, M.G.; Millefiorini, E.; Graceffa, A.; Carlesimo, G.A.; Zibellini, G.; Caltagirone, C. Fatigue in progressive multiple sclerosis: Results of a randomized, double-blind, placebo-controlled, crossover trial of oral 4-aminopyridine. Mult. Scler. J. 2001, 7, 354–358. [Google Scholar] [CrossRef]
- Hojsgaard Chow, H.; Schreiber, K.; Magyari, M.; Ammitzboll, C.; Bornsen, L.; Romme Christensen, J.; Ratzer, R.; Soelberg Sorensen, P.; Sellebjerg, F. Progressive multiple sclerosis, cognitive function, and quality of life. Brain Behav. 2018, 8, e00875. [Google Scholar] [CrossRef]
- Sawyer, D.B.; Vasan, R.S. Encyclopedia of Cardiovascular Research and Medicine; Elsevier Science: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Day, J.R.; Rossiter, H.B.; Coats, E.M.; Skasick, A.; Whipp, B.J. The maximally attainable during exercise in humans: The peak vs. maximum issue. J. Appl. Physiol. 2003, 95, 1901–1907. [Google Scholar] [CrossRef]
- Langeskov-Christensen, M.; Heine, M.; Kwakkel, G.; Dalgas, U. Aerobic Capacity in Persons with Multiple Sclerosis: A Systematic Review and Meta-Analysis. Sports Med. 2015, 45, 905–923. [Google Scholar] [CrossRef]
- Group, B.D.W.; Atkinson, A.J., Jr.; Colburn, W.A.; DeGruttola, V.G.; DeMets, D.L.; Downing, G.J.; Hoth, D.F.; Oates, J.A.; Peck, C.C.; Schooley, R.T. Biomarkers and surrogate endpoints: Preferred definitions and conceptual framework. Clin. Pharmacol. Ther. 2001, 69, 89–95. [Google Scholar]
- Katsavos, S.; Anagnostouli, M. Biomarkers in multiple sclerosis: An up-to-date overview. Mult. Scler. Int. 2013, 2013, 20. [Google Scholar] [CrossRef]
- Strimbu, K.; Tavel, J.A. What are biomarkers? Curr. Opin. HIV Aids 2010, 5, 463–466. [Google Scholar] [CrossRef] [PubMed]
- Harris, V.K.; Sadiq, S.A. Disease biomarkers in multiple sclerosis. Mol. Diagn. Ther. 2009, 13, 225–244. [Google Scholar] [CrossRef]
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann. Neurol. 2011, 69, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.; Reading, J.; Shephard, R.J. Revision of the physical activity readiness questionnaire (PAR-Q). Can. J. Sport Sci. 1992, 17, 338–345. [Google Scholar] [PubMed]
- Shephard, R.J. PAR-Q, Canadian Home Fitness Test and exercise screening alternatives. Sports Med. 1988, 5, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Warburton, D.E.; Gledhill, N.; Jamnik, V.K.; Bredin, S.S.; McKenzie, D.C.; Stone, J.; Charlesworth, S.; Shephard, R.J. Evidence-based risk assessment and recommendations for physical activity clearance: Consensus Document 2011. Appl. Physiol. Nutr. Metab. 2011, 36, S266–S298. [Google Scholar] [CrossRef] [PubMed]
- Ware, J.E. Scoring the SF-36. In SF-36 Health Survey. Manual and Interpretation Guide; The Health Institute, New England Medical Center: Boston, MA, USA, 1993. [Google Scholar]
- Ware, J.E., Jr. SF-36 health survey update. Spine 2000, 25, 3130–3139. [Google Scholar] [CrossRef]
- Deng, N.; Guyer, R.; Ware, J.E. Energy, fatigue, or both? A bifactor modeling approach to the conceptualization and measurement of vitality. Qual. Life Res. 2015, 24, 81–93. [Google Scholar] [CrossRef]
- Kelly, L.P.; Devasahayam, A.J.; Chaves, A.R.; Wallack, E.M.; McCarthy, J.; Basset, F.A.; Ploughman, M. Intensifying functional task practice to meet aerobic training guidelines in stroke survivors. Front. Physiol. 2017, 8, 809. [Google Scholar] [CrossRef]
- American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013. [Google Scholar]
- Price, R.D.; Milne, S.A.; Sharkey, J.; Matsuoka, N. Advances in small molecules promoting neurotrophic function. Pharmacol. Ther. 2007, 115, 292–306. [Google Scholar] [CrossRef] [PubMed]
- Öztuna, D.; Elhan, A.H.; Tüccar, E. Investigation of four different normality tests in terms of type 1 error rate and power under different distributions. Turk. J. Med. Sci. 2006, 36, 171–176. [Google Scholar]
- Ghasemi, A.; Zahediasl, S. Normality tests for statistical analysis: A guide for non-statisticians. Int. J. Endocrinol. Metab. 2012, 10, 486. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioural Sciences; Academic Press: New York, NY, USA, 1969. [Google Scholar]
- Hornung, R.W.; Reed, L.D. Estimation of Average Concentration in the Presence of Nondetectable Values. Appl. Occup. Environ. Hyg. 1990, 5, 46–51. [Google Scholar] [CrossRef]
- Marques-Vidal, P.; Bochud, M.; Bastardot, F.; Lüscher, T.; Ferrero, F.; Gaspoz, J.-M.; Paccaud, F.; Urwyler, A.; von Känel, R.; Hock, C.; et al. Levels and Determinants of Inflammatory Biomarkers in a Swiss Population-Based Sample (CoLaus Study). PLoS ONE 2011, 6, e21002. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Guidance for Data Quality Assessment: Practical Methods for Data Analysis EPA QA/G-9, (QA97 Version); U.S. Environmental Protection Agency: Washington, DC, USA, 1998.
- Alvarenga-Filho, H.; Sacramento, P.M.; Ferreira, T.B.; Hygino, J.; Abreu, J.E.C.; Carvalho, S.R.; Wing, A.C.; Alvarenga, R.M.P.; Bento, C.A.M. Combined exercise training reduces fatigue and modulates the cytokine profile of T-cells from multiple sclerosis patients in response to neuromediators. J. Neuroimmunol. 2016, 293, 91–99. [Google Scholar] [CrossRef]
- Langeskov-Christensen, M.; Bisson, E.J.; Finlayson, M.L.; Dalgas, U. Potential pathophysiological pathways that can explain the positive effects of exercise on fatigue in multiple sclerosis: A scoping review. J. Neurol. Sci. 2017, 373, 307–320. [Google Scholar] [CrossRef]
- Motl, R.W.; Sandroff, B.M. Exercise as a Countermeasure to Declining Central Nervous System Function in Multiple Sclerosis. Clin. Ther. 2018, 40, 16–25. [Google Scholar] [CrossRef]
- Devasahayam, A.J.; Kelly, L.P.; Wallack, E.M.; Ploughman, M. Oxygen cost during mobility tasks and its relationship to fatigue in progressive Multiple Sclerosis. Arch. Phys. Med. Rehabil. 2019. [Google Scholar] [CrossRef]
- Coote, S.; O’Dwyer, C. Energy expenditure during everyday activities—A study comparing people with varying mobility limitations due to multiple sclerosis and healthy controls. Disabil. Rehabil. 2014, 36, 2059–2064. [Google Scholar] [CrossRef]
- Rasmussen, P.; Brassard, P.; Adser, H.; Pedersen, M.V.; Leick, L.; Hart, E.; Secher, N.H.; Pedersen, B.K.; Pilegaard, H. Evidence for a release of brain-derived neurotrophic factor from the brain during exercise. Exp. Physiol. 2009, 94, 1062–1069. [Google Scholar] [CrossRef]
- Cabral-Santos, C.; Castrillón, C.I.M.; Miranda, R.A.T.; Monteiro, P.A.; Inoue, D.S.; Campos, E.Z.; Hofmann, P.; Lira, F.S. Inflammatory Cytokines and BDNF Response to High-Intensity Intermittent Exercise: Effect the Exercise Volume. Front. Physiol. 2016, 7, 509. [Google Scholar] [CrossRef]
- Ogborn, D.I.; Gardiner, P.F. Effects of exercise and muscle type on BDNF, NT-4/5, and TrKB expression in skeletal muscle. Muscle Nerve 2010, 41, 385–391. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscles, exercise and obesity: Skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 2012, 8, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Banks, W.A.; Fasold, M.B.; Bluth, J.; Kastin, A.J. Transport of brain-derived neurotrophic factor across the blood-brain barrier. Neuropharmacology 1998, 37, 1553–1561. [Google Scholar] [CrossRef]
- Bohannon, R.W. Comfortable and maximum walking speed of adults aged 20–79 years: Reference values and determinants. Age Ageing 1997, 26, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Chiu, M.-C.; Wang, M.-J. The effect of gait speed and gender on perceived exertion, muscle activity, joint motion of lower extremity, ground reaction force and heart rate during normal walking. Gait Posture 2007, 25, 385–392. [Google Scholar] [CrossRef] [PubMed]
- den Otter, A.R.; Geurts, A.C.H.; Mulder, T.; Duysens, J. Speed related changes in muscle activity from normal to very slow walking speeds. Gait Posture 2004, 19, 270–278. [Google Scholar] [CrossRef]
- Hesse, S.; Werner, C.; Paul, T.; Bardeleben, A.; Chaler, J. Influence of walking speed on lower limb muscle activity and energy consumption during treadmill walking of hemiparetic patients. Arch. Phys. Med. Rehabil. 2001, 82, 1547–1550. [Google Scholar] [CrossRef]
- Murray, M.P.; Mollinger, L.A.; Gardner, G.M.; Sepic, S.B. Kinematic and EMG patterns during slow, free, and fast walking. J. Orthop. Res. 1984, 2, 272–280. [Google Scholar] [CrossRef] [PubMed]
- van Hedel, H.J.A.; Tomatis, L.; Müller, R. Modulation of leg muscle activity and gait kinematics by walking speed and bodyweight unloading. Gait Posture 2006, 24, 35–45. [Google Scholar] [CrossRef]
- Warren, G.L.; Maher, R.M.; Higbie, E.J. Temporal patterns of plantar pressures and lower-leg muscle activity during walking: Effect of speed. Gait Posture 2004, 19, 91–100. [Google Scholar] [CrossRef]
- Gilder, M.; Ramsbottom, R.; Currie, J.; Sheridan, B.; Nevill, A.M. Effect of fat free mass on serum and plasma BDNF concentrations during exercise and recovery in healthy young men. Neurosci. Lett. 2014, 560, 137–141. [Google Scholar] [CrossRef]
- Thoumie, P.; Lamotte, D.; Cantalloube, S.; Faucher, M.; Amarenco, G. Motor determinants of gait in 100 ambulatory patients with multiple sclerosis. Mult. Scler. J. 2005, 11, 485–491. [Google Scholar] [CrossRef]
- Ireland, S.J.; Monson, N.L.; Davis, L.S. Seeking balance: Potentiation and inhibition of multiple sclerosis autoimmune responses by IL-6 and IL-10. Cytokine 2015, 73, 236–244. [Google Scholar] [CrossRef]
- Starkie, R.; Ostrowski, S.R.; Jauffred, S.; Febbraio, M.; Pedersen, B.K. Exercise and IL-6 infusion inhibit endotoxin-induced TNF-α production in humans. FASEB J. 2003, 17, 884–886. [Google Scholar] [CrossRef]
- Knudsen, J.G.; Gudiksen, A.; Bertholdt, L.; Overby, P.; Villesen, I.; Schwartz, C.L.; Pilegaard, H. Skeletal muscle IL-6 regulates muscle substrate utilization and adipose tissue metabolism during recovery from an acute bout of exercise. PLoS ONE 2017, 12, e0189301. [Google Scholar] [CrossRef]
- Pedersen, B. IL-6 Signalling in Exercise and Disease; Portland Press Limited: London, UK, 2007. [Google Scholar]
- Brown, W.M.; Davison, G.W.; McClean, C.M.; Murphy, M.H. A systematic review of the acute effects of exercise on immune and inflammatory indices in untrained adults. Sports Med. Open 2015, 1, 35. [Google Scholar] [CrossRef]
- MacPherson, R.E.; Huber, J.S.; Frendo-Cumbo, S.; Simpson, J.A.; Wright, D.C. Adipose tissue insulin action and IL-6 signaling after exercise in obese mice. Med. Sci. Sports Exerc. 2015, 47, 2034–2042. [Google Scholar] [CrossRef]
- Audet, M.-C.; McQuaid, R.J.; Merali, Z.; Anisman, H. Cytokine variations and mood disorders: Influence of social stressors and social support. Front. Neurosci. 2014, 8, 416. [Google Scholar] [CrossRef]
- Cryan, J.F.; Dinan, T.G. Mind-altering microorganisms: The impact of the gut microbiota on brain and behaviour. Nat. Rev. Neurosci. 2012, 13, 701. [Google Scholar] [CrossRef] [PubMed]
- Grimble, R.F.; Tappia, P.S. Modulation of pro-inflammatory cytokine biology by unsaturated fatty acids. Z. Ernahr. 1998, 37 (Suppl. 1), 57–65. [Google Scholar]
- Grimble, R.F. Nutritional modulation of cytokine biology. Nutrition 1998, 14, 634–640. [Google Scholar] [CrossRef]
- Vgontzas, A.N.; Zoumakis, E.; Bixler, E.O.; Lin, H.M.; Follett, H.; Kales, A.; Chrousos, G.P. Adverse effects of modest sleep restriction on sleepiness, performance, and inflammatory cytokines. J. Clin. Endocrinol. Metab. 2004, 89, 2119–2126. [Google Scholar] [CrossRef] [PubMed]
- Pejovic, S.; Basta, M.; Vgontzas, A.N.; Kritikou, I.; Shaffer, M.L.; Tsaoussoglou, M.; Stiffler, D.; Stefanakis, Z.; Bixler, E.O.; Chrousos, G.P. Effects of recovery sleep after one work week of mild sleep restriction on interleukin-6 and cortisol secretion and daytime sleepiness and performance. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E890–E896. [Google Scholar] [CrossRef] [PubMed]
- Altara, R.; Manca, M.; Hermans, K.C.; Daskalopoulos, E.P.; Brunner-La Rocca, H.P.; Hermans, R.J.; Struijker-Boudier, H.A.; Blankesteijn, M.W. Diurnal rhythms of serum and plasma cytokine profiles in healthy elderly individuals assessed using membrane based multiplexed immunoassay. J. Transl. Med. 2015, 13, 129. [Google Scholar] [CrossRef] [PubMed]
- Haack, M.; Pollmacher, T.; Mullington, J.M. Diurnal and sleep-wake dependent variations of soluble TNF- and IL-2 receptors in healthy volunteers. Brain Behav. Immun. 2004, 18, 361–367. [Google Scholar] [CrossRef]
- Scheff, J.D.; Calvano, S.E.; Lowry, S.F.; Androulakis, I.P. Modeling the influence of circadian rhythms on the acute inflammatory response. J. Theor. Biol. 2010, 264, 1068–1076. [Google Scholar] [CrossRef]
- Madretsma, G.S.; Donze, G.J.; van Dijk, A.P.; Tak, C.J.; Wilson, J.H.; Zijlstra, F.J. Nicotine inhibits the in vitro production of interleukin 2 and tumour necrosis factor-alpha by human mononuclear cells. Immunopharmacology 1996, 35, 47–51. [Google Scholar] [CrossRef]
- Achur, R.N.; Freeman, W.M.; Vrana, K.E. Circulating cytokines as biomarkers of alcohol abuse and alcoholism. J. Neuroimmune Pharmacol. Off. J. Soc. Neuroimmune Pharmacol. 2010, 5, 83–91. [Google Scholar] [CrossRef]
- You, Z.; Luo, C.; Zhang, W.; Chen, Y.; He, J.; Zhao, Q.; Zuo, R.; Wu, Y. Pro- and anti-inflammatory cytokines expression in rat’s brain and spleen exposed to chronic mild stress: Involvement in depression. Behav. Brain Res. 2011, 225, 135–141. [Google Scholar] [CrossRef]
- Tavakoli-Ardakani, M.; Mehrpooya, M.; Mehdizadeh, M.; Hajifathali, A.; Abdolahi, A. Association between Interlukin-6 (IL-6), Interlukin-10 (IL-10) and depression in patients undergoing Hematopoietic stem cell transplantation. Int. J. Hematol. Oncol. Stem Cell Res. 2015, 9, 80–87. [Google Scholar]
- Kang, S.; Tanaka, T.; Narazaki, M.; Kishimoto, T. Targeting Interleukin-6 Signaling in Clinic. Immunity 2019, 50, 1007–1023. [Google Scholar] [CrossRef] [PubMed]
| Parameters | Characteristic | MS | Controls | p Values |
|---|---|---|---|---|
| Mean (SD) | Mean (SD) | |||
| Demographics | ||||
| Age (in years) | 54.07 (8.46) | 50.71 (12.08) | 0.585 | |
| Sex | Females/Males | 10/4 | 4/3 | 0.513 |
| Body mass index (in kg−1·m−1) | 27.74 (7.56) | 27.44 (3.76) | 1.0 | |
| Years since MS diagnosis | 16.57 (9.69) | NA | NA | |
| Ambulatory assistive device used | Cane | 6 | 0 | NA |
| 2 Canes or Walker | 8 | 0 | NA | |
| Type of MS (n) | SPMS | 11 | NA | NA |
| PPMS | 3 | NA | NA | |
| Biomarkers † | ||||
| BDNF (ng·mL−1) | At rest | 56.56 (25.12) | 57.63 (9.48) | 0.967 |
| Post-Pre | −0.09 (18.99) | −0.43 (14.79) | 0.837 | |
| IL-6 (ng·mL−1) | At rest | 0.0015 (0.002) | 0.0003 (0.0004) | 0.010 |
| Post-Pre | 0.0007 (0.0007) | 0.0005 (0.0007) | 0.384 | |
| BDNF/IL-6 ratio | At rest | 119808.50 (116307.59) | 393501.90 (188730.57) | 0.004 |
| Post-Pre | −70038.97 (95439.75) | −7787.75 (278552.43) | 1.0 | |
| Functional measures | ||||
| Comfortable walking speed (m·s−1) ‡ | 0.32 (0.13) | 0.96 (0.26) | <0.001 | |
| SF-36 (Vitality/Energy/Fatigue) | 37.14 (18.16) | 87.14 (9.06) | <0.001 | |
| Maximal (mL·min−1·kg−1) | 16.35 (6.39) | 33.04 (8.95) | <0.001 | |
| Duration of GXT (s) | 793.29 (259.84) | 1087.71 (207.95) | 0.046 | |
| Maximal workload (Watts) | 99.69 (33.84) | 271.43 (127.46) | <0.001 | |
| Maximal heart rate (beats·min−1) | 131.57 (23.16) | 168.26 (16.83) | 0.002 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Devasahayam, A.J.; Kelly, L.P.; Williams, J.B.; Moore, C.S.; Ploughman, M. Fitness Shifts the Balance of BDNF and IL-6 from Inflammation to Repair among People with Progressive Multiple Sclerosis. Biomolecules 2021, 11, 504. https://doi.org/10.3390/biom11040504
Devasahayam AJ, Kelly LP, Williams JB, Moore CS, Ploughman M. Fitness Shifts the Balance of BDNF and IL-6 from Inflammation to Repair among People with Progressive Multiple Sclerosis. Biomolecules. 2021; 11(4):504. https://doi.org/10.3390/biom11040504
Chicago/Turabian StyleDevasahayam, Augustine Joshua, Liam Patrick Kelly, John Bradley Williams, Craig Stephen Moore, and Michelle Ploughman. 2021. "Fitness Shifts the Balance of BDNF and IL-6 from Inflammation to Repair among People with Progressive Multiple Sclerosis" Biomolecules 11, no. 4: 504. https://doi.org/10.3390/biom11040504
APA StyleDevasahayam, A. J., Kelly, L. P., Williams, J. B., Moore, C. S., & Ploughman, M. (2021). Fitness Shifts the Balance of BDNF and IL-6 from Inflammation to Repair among People with Progressive Multiple Sclerosis. Biomolecules, 11(4), 504. https://doi.org/10.3390/biom11040504







