Glucosinolate Bioactivation by Apis mellifera Workers and Its Impact on Nosema ceranae Infection at the Colony Level
Abstract
:1. Introduction
2. Materials and Methods
2.1. Honey Bee Colony Selection
2.2. Brassicaceae DSMs
2.3. Feed Formulation
2.4. Treatments and Colony Handling
2.5. N. ceranae Infection Quantification: DNA Extraction and qPCR
2.6. Bee Gut MYR Enzyme Extraction and Activity Assay
2.6.1. MYR Activity Assay
2.7. Bee Gut Total ITC Extraction and Quantification
2.8. Honey GSL and ITC Detection
2.9. Calculations and Statistics
3. Results
3.1. DSM Characterization and Patty Formulation
3.2. Bee Treatment Feeding Trials
3.2.1. Palatability of DSM Patties
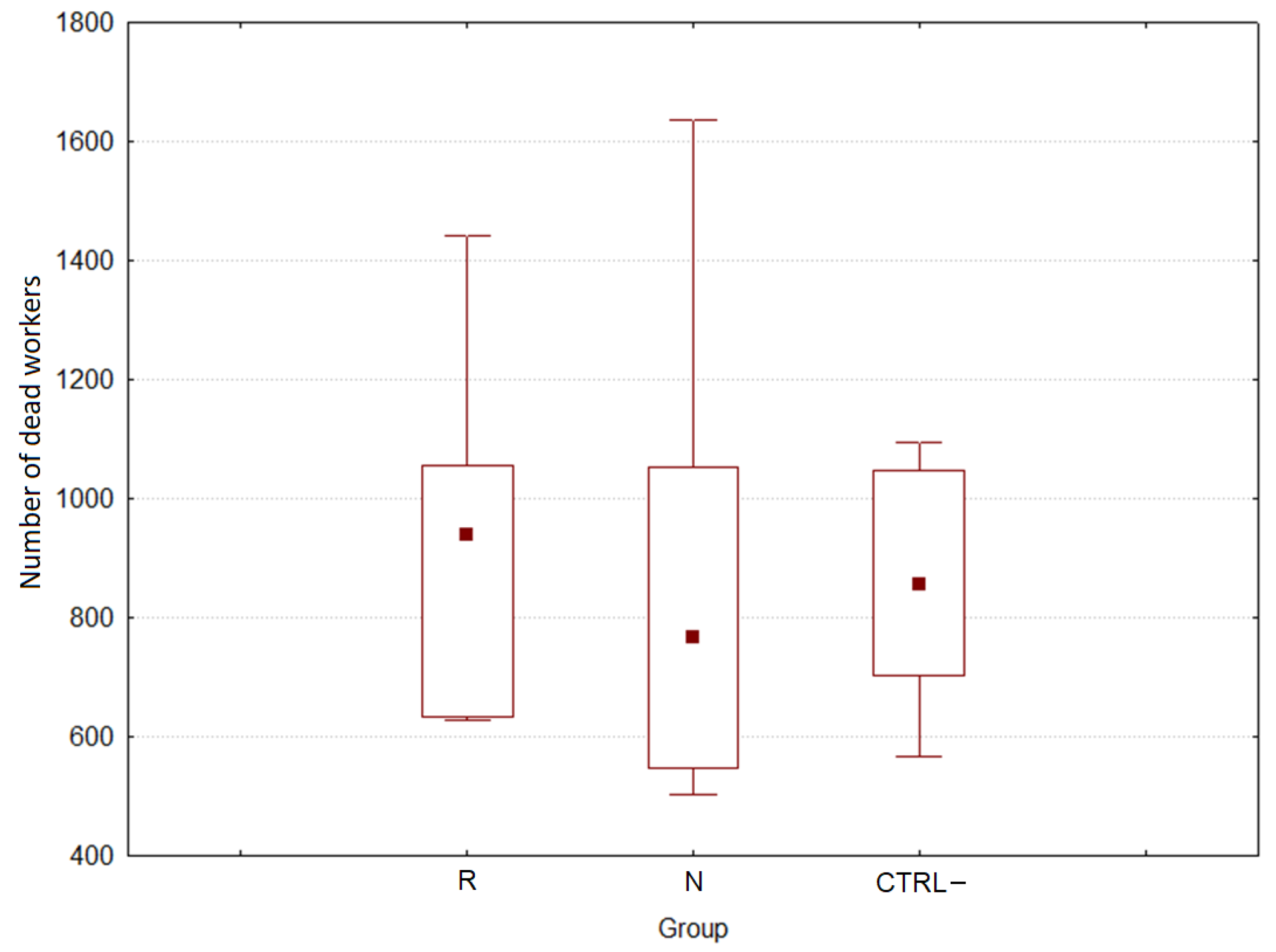
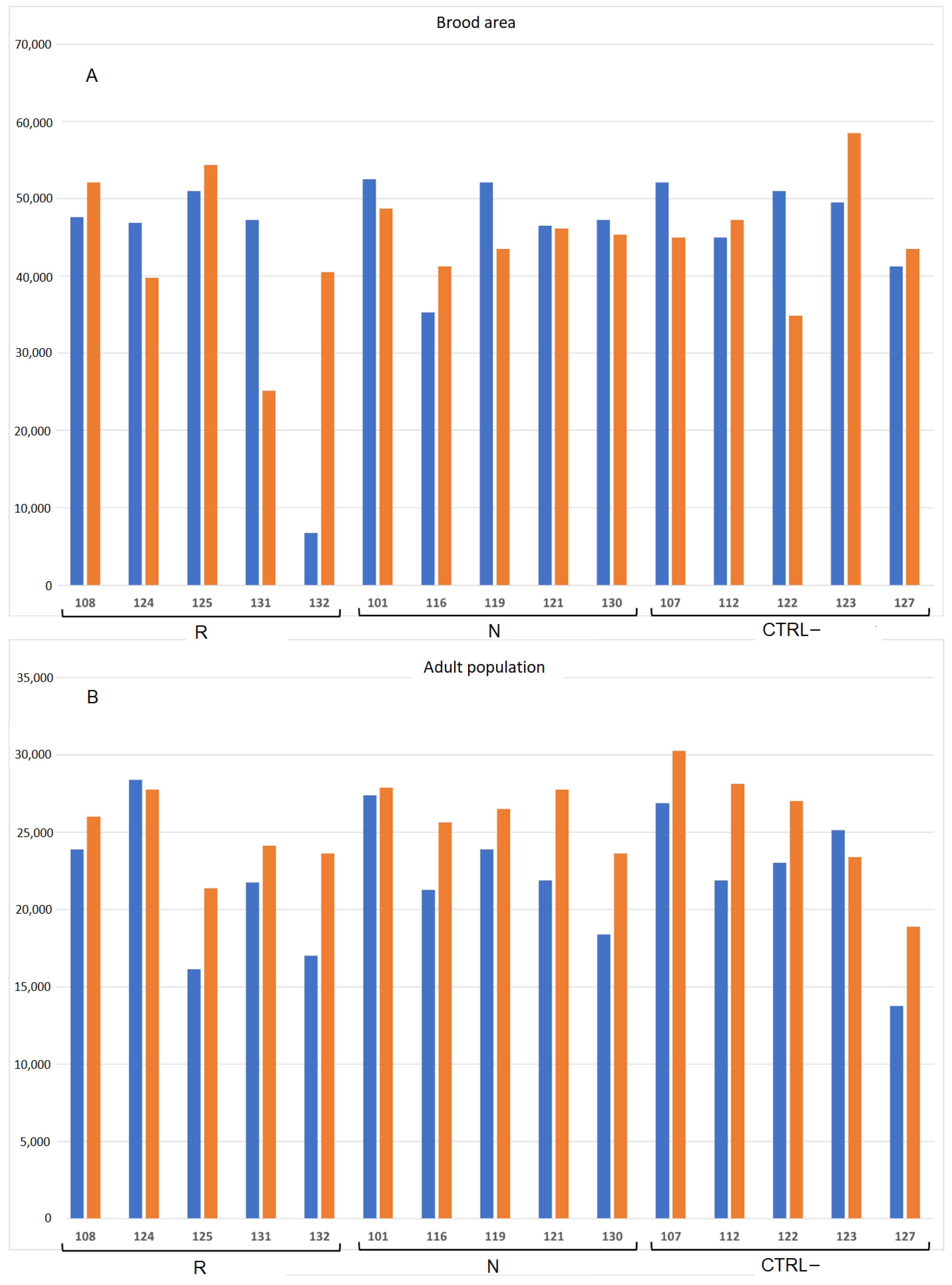
3.2.2. Effect of DSM Patties on Bee Survival
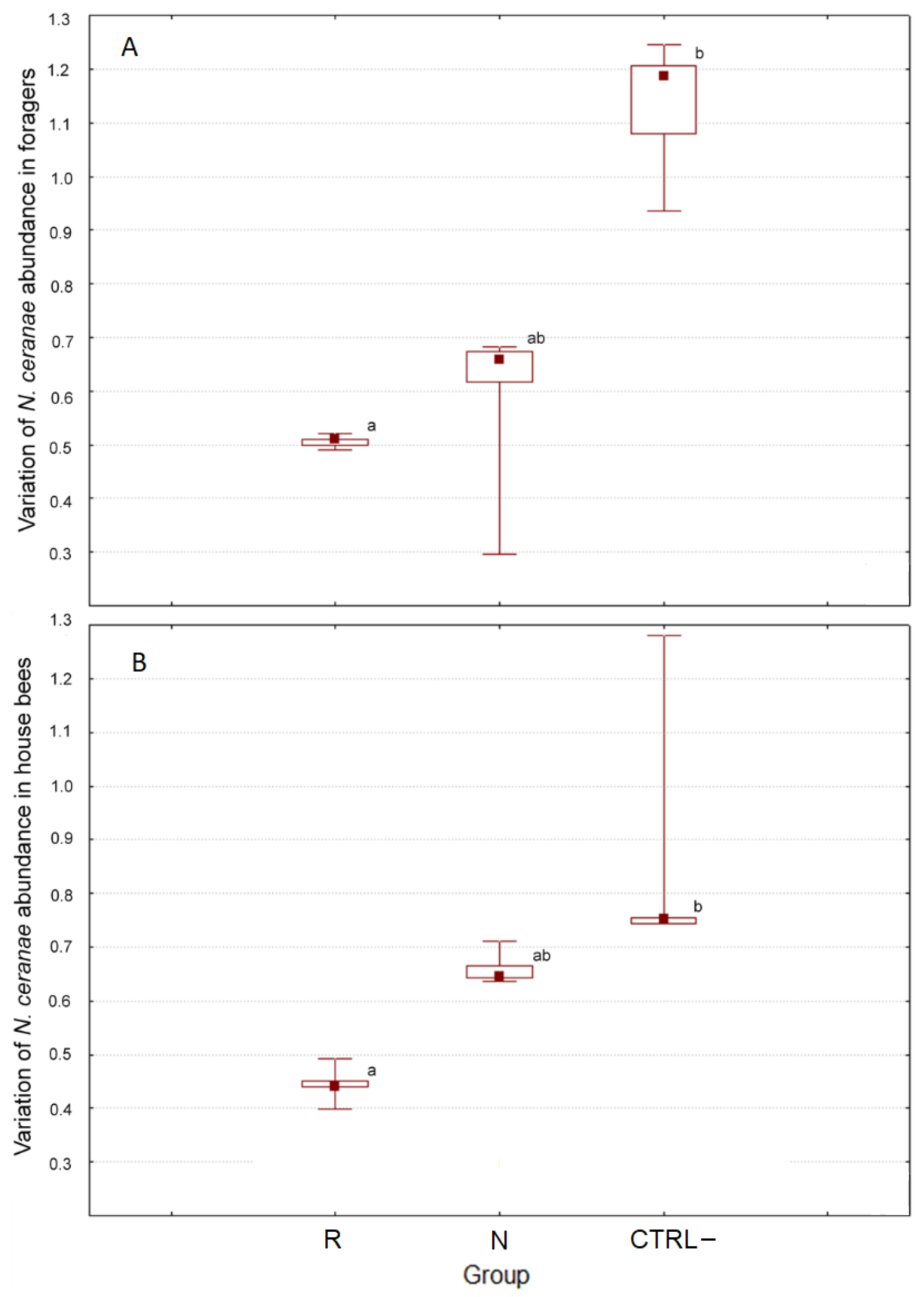
3.2.3. Effect of DSM Patties on N. ceranae
3.3. MYR-Like Enzymatic Activity in the Bee Gut
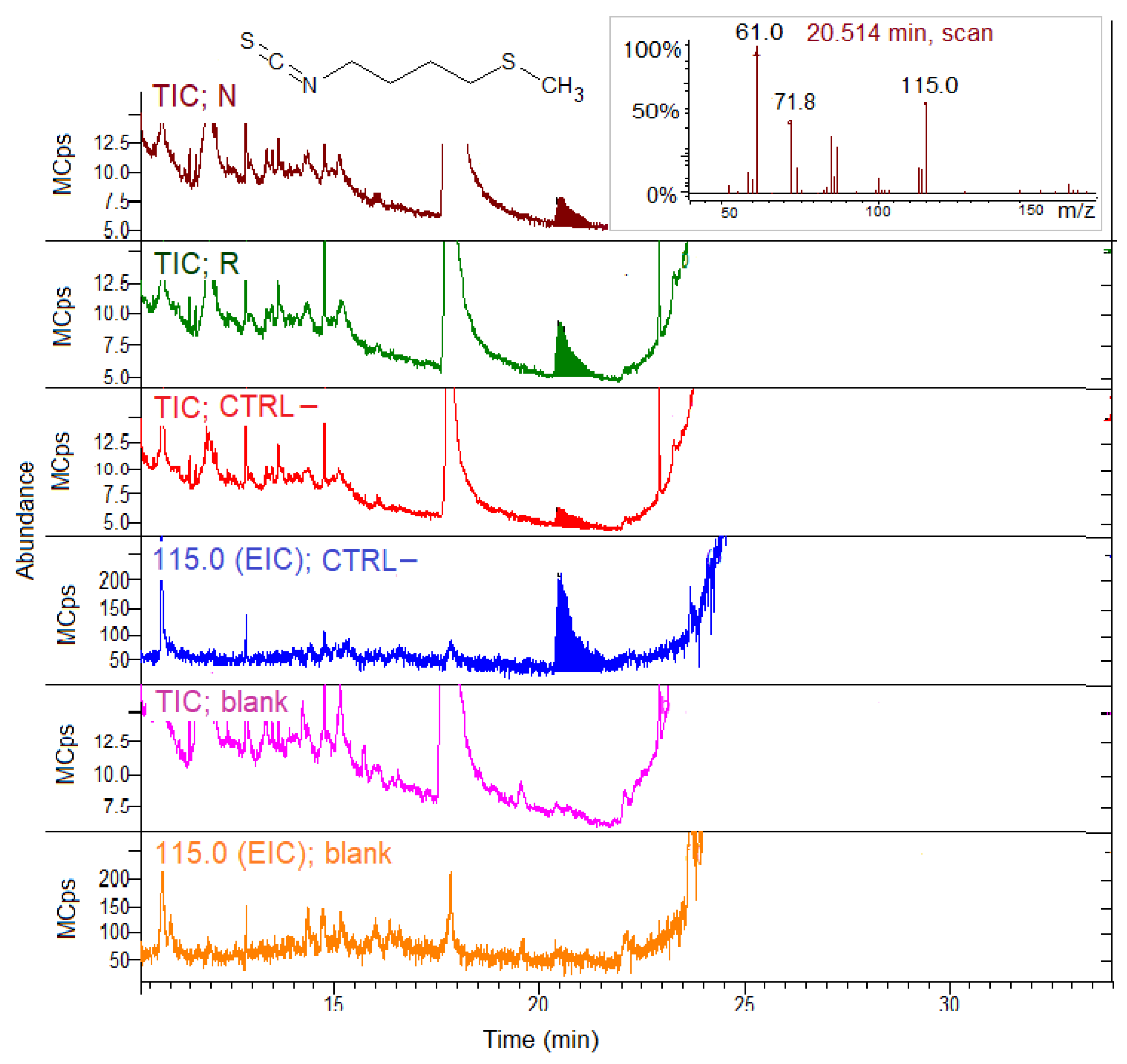
3.4. Total ITC Gut Analysis
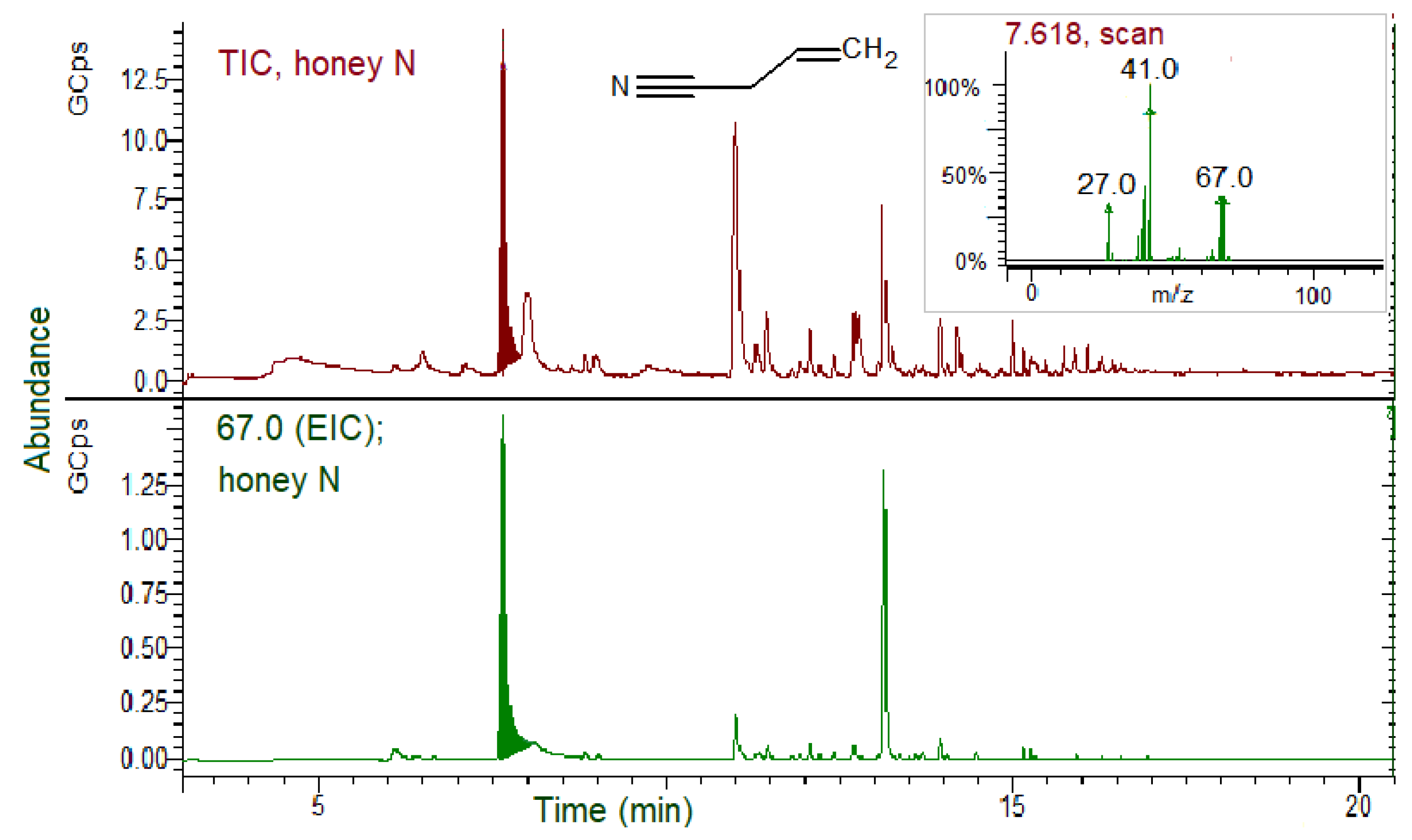
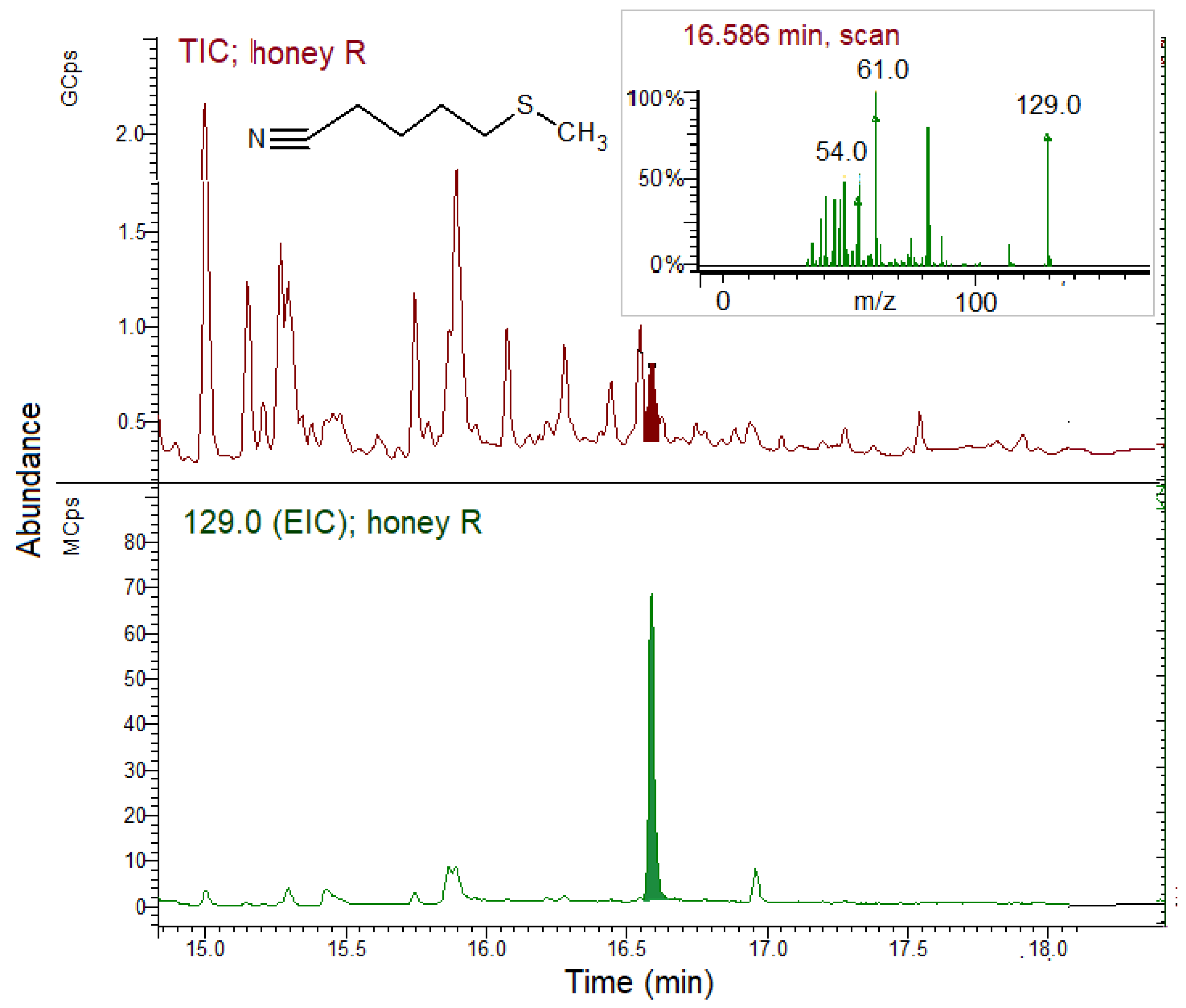
3.5. GSLs and GSL Hydrolysis Products in Honey
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Higes, M.; Meana, A.; Bartolomé, C.; Botías, C.; Martín-Hernández, R. Nosema ceranae (Microsporidia), a controversial 21st century honey bee pathogen. Environ. Microbiol. Rep. 2013, 5, 17–29. [Google Scholar] [CrossRef]
- Fries, I.; Martín, R.; Meana, A.; García-Palencia, P.; Higes, M. Natural infections of Nosema ceranae in European honey bees. J. Apic. Res. 2006, 45, 230–233. [Google Scholar] [CrossRef]
- Fries, I.; Feng, F.; da Silva, A.; Slemenda, S.B.; Pieniazek, N.J. Nosema ceranae n. sp. (Microspora, Nosematidae), morphological and molecular characterization of a microsporidian parasite of the Asian honey bee Apis cerana (Hymenoptera, Apidae). Eur. J. Protistol. 1996, 32, 356–365. [Google Scholar] [CrossRef]
- Higes, M.; García-Palencia, P.; Urbieta, A.; Nanetti, A.; Martín-Hernández, R. Nosema apis and Nosema ceranae Tissue Tropism in Worker Honey Bees (Apis mellifera). Vet. Pathol. 2020, 57, 132–138. [Google Scholar] [CrossRef]
- Li, W.; Chen, Y.; Cook, S.C. Chronic Nosema ceranae infection inflicts comprehensive and persistent immunosuppression and accelerated lipid loss in host Apis mellifera honey bees. Int. J. Parasitol. 2018, 48, 433–444. [Google Scholar] [CrossRef]
- Goblirsch, M.; Huang, Z.Y.; Spivak, M. Physiological and Behavioral Changes in Honey Bees (Apis mellifera) Induced by Nosema ceranae Infection. PLoS ONE 2013, 8, e58165. [Google Scholar] [CrossRef]
- Higes, M.; García-Palencia, P.; Martín-Hernández, R.; Meana, A. Experimental infection of Apis mellifera honeybees with Nosema ceranae (Microsporidia). J. Invertebr. Pathol. 2007, 94, 211–217. [Google Scholar] [CrossRef]
- Huang, W.-F.; Solter, L.F.; Yau, P.M.; Imai, B.S. Nosema ceranae Escapes Fumagillin Control in Honey Bees. PLoS Pathog. 2013, 9, e1003185. [Google Scholar] [CrossRef] [Green Version]
- Klassen, S.S.; VanBlyderveen, W.; Eccles, L.; Kelly, P.G.; Borges, D.; Goodwin, P.H.; Petukhova, T.; Wang, Q.; Guzman-Novoa, E. Nosema ceranae Infections in Honey Bees (Apis mellifera) Treated with Pre/Probiotics and Impacts on Colonies in the Field. Vet. Sci. 2021, 8, 107. [Google Scholar] [CrossRef]
- van den Heever, J.P.; Thompson, T.S.; Curtis, J.M.; Ibrahim, A.; Pernal, S.F. Fumagillin: An Overview of Recent Scientific Advances and Their Significance for Apiculture. J. Agric. Food Chem. 2014, 62, 2728–2737. [Google Scholar] [CrossRef]
- Burnham, A.J. Scientific Advances in Controlling Nosema ceranae (Microsporidia) Infections in Honey Bees (Apis mellifera). Front. Vet. Sci. 2019, 6, 79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaggìa, F.; Baffoni, L.; Alberoni, D. Probiotics for Honeybees’ Health. In Probiotics and Prebiotics in Animal Health and Food Safety; Springer International Publishing: Cham, Switzerland, 2018. [Google Scholar] [CrossRef]
- Tlak Gajger, I.; Vlainić, J.; Šoštarić, P.; Prešern, J.; Bubnič, J.; Smodiš Škerl, M.I. Effects on Some Therapeutical, Biochemical, and Immunological Parameters of Honey Bee (Apis mellifera) Exposed to Probiotic Treatments, in Field and Laboratory Conditions. Insects 2020, 11, 638. [Google Scholar] [CrossRef]
- Baffoni, L.; Gaggìa, F.; Alberoni, D.; Cabbri, R.; Nanetti, A.; Biavati, B.; Di Gioia, D. Effect of dietary supplementation of Bifidobacterium and Lactobacillus strains in Apis mellifera L. against Nosema ceranae. Benef. Microbes 2016, 7, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Borges, D.; Guzman-Novoa, E.; Goodwin, P.H. Effects of Prebiotics and Probiotics on Honey Bees (Apis mellifera) Infected with the Microsporidian Parasite Nosema ceranae. Microorganisms 2021, 9, 481. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Su, M.; Wang, L.; Huang, S.; Su, S.; Huang, W.-F. Vairimorpha (Nosema) ceranae Infection Alters Honey Bee Microbiota Composition and Sustains the Survival of Adult Honey Bees. Biology 2021, 10, 905. [Google Scholar] [CrossRef]
- Costa, C.; Lodesani, M.; Maistrello, L. Effect of thymol and resveratrol administered with candy or syrup on the development of Nosema ceranae and on the longevity of honeybees (Apis mellifera L.) in laboratory conditions. Apidologie 2010, 41, 141–150. [Google Scholar] [CrossRef] [Green Version]
- Maistrello, L.; Lodesani, M.; Costa, C.; Leonardi, F.; Marani, G.; Caldon, M.; Mutinelli, F.; Granato, A. Screening of natural compounds for the control of nosema disease in honeybees (Apis mellifera). Apidologie 2008, 39, 436–445. [Google Scholar] [CrossRef] [Green Version]
- Glavinic, U.; Stankovic, B.; Draskovic, V.; Stevanovic, J.; Petrovic, T.; Lakic, N.; Stanimirovic, Z. Dietary amino acid and vitamin complex protects honey bee from immunosuppression caused by Nosema ceranae. PLoS ONE 2017, 12, e0187726. [Google Scholar] [CrossRef]
- Michalczyk, M.; Sokół, R.; Koziatek, S. Evaluation of the effectiveness of selected treatments of Nosema spp. infection by the hemocytometric method and duplex PCR. Acta Vet. 2016, 66, 115–124. [Google Scholar] [CrossRef] [Green Version]
- Cilia, G.; Fratini, F.; Tafi, E.; Turchi, B.; Mancini, S.; Sagona, S.; Nanetti, A.; Cerri, D.; Felicioli, A. Microbial Profile of the Ventriculum of Honey Bee (Apis mellifera ligustica Spinola, 1806) Fed with Veterinary Drugs, Dietary Supplements and Non-Protein Amino Acids. Vet. Sci. 2020, 7, 76. [Google Scholar] [CrossRef]
- Braglia, C.; Alberoni, D.; Porrini, M.P.; Garrido, P.M.; Baffoni, L.; Di Gioia, D. Screening of Dietary Ingredients against the Honey Bee Parasite Nosema ceranae. Pathogens 2021, 10, 1117. [Google Scholar] [CrossRef]
- Tlak Gajger, I.; Smodiš Škerl, M.I.; Šoštarić, P.; Šuran, J.; Sikirić, P.; Vlainić, J. Physiological and Immunological Status of Adult Honeybees (Apis mellifera) Fed Sugar Syrup Supplemented with Pentadecapeptide BPC 157. Biology 2021, 10, 891. [Google Scholar] [CrossRef]
- Bones, A.M.; Rossiter, J.T. The enzymic and chemically induced decomposition of glucosinolates. Phytochemistry 2006, 67, 1053–1067. [Google Scholar] [CrossRef]
- Fenwick, G.R.; Heaney, R.K.; Mullin, W.J.; VanEtten, C.H. Glucosinolates and their breakdown products in food and food plants. CRC Crit. Rev. Food Sci. Nutr. 1983, 18, 123–201. [Google Scholar] [CrossRef] [PubMed]
- Wittstock, U.; Burow, M. Tipping the Scales—Specifier Proteins in Glucosinolate Hydrolysis. IUBMB Life 2007, 59, 744–751. [Google Scholar] [CrossRef] [PubMed]
- Agerbirk, N.; Olsen, C.E. Glucosinolate structures in evolution. Phytochemistry 2012, 77, 16–45. [Google Scholar] [CrossRef]
- Blažević, I.; Montaut, S.; Burčul, F.; Olsen, C.E.; Burow, M.; Rollin, P.; Agerbirk, N. Glucosinolate structural diversity, identification, chemical synthesis and metabolism in plants. Phytochemistry 2020, 169, 112100. [Google Scholar] [CrossRef] [PubMed]
- Plaszkó, T.; Szűcs, Z.; Vasas, G.; Gonda, S. Effects of Glucosinolate-Derived Isothiocyanates on Fungi: A Comprehensive Review on Direct Effects, Mechanisms, Structure-Activity Relationship Data and Possible Agricultural Applications. J. Fungi 2021, 7, 539. [Google Scholar] [CrossRef]
- Pessina, A.; Thomas, R.M.; Palmieri, S.; Luisi, P.L. An improved method for the purification of myrosinase and its physicochemical characterization. Arch. Biochem. Biophys. 1990, 280, 383–389. [Google Scholar] [CrossRef]
- Burmeister, W.P.; Cottaz, S.; Driguez, H.; Iori, R.; Palmieri, S.; Henrissat, B. The crystal structures of Sinapis alba myrosinase and a covalent glycosyl–enzyme intermediate provide insights into the substrate recognition and active-site machinery of an S-glycosidase. Structure 1997, 5, 663–676. [Google Scholar] [CrossRef] [Green Version]
- Bridges, M.; Jones, A.M.E.; Bones, A.M.; Hodgson, C.; Cole, R.; Bartlet, E.; Wallsgrove, R.; Karapapa, V.K.; Watts, N.; Rossiter, J.T. Spatial organization of the glucosinolate–myrosinase system in brassica specialist aphids is similar to that of the host plant. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2002, 269, 187–191. [Google Scholar] [CrossRef] [Green Version]
- Beran, F.; Pauchet, Y.; Kunert, G.; Reichelt, M.; Wielsch, N.; Vogel, H.; Reinecke, A.; Svato, A.; Mewis, I.; Schmid, D.; et al. Phyllotreta striolata flea beetles use host plant defense compounds to create their own glucosinolate-myrosinase system. Proc. Natl. Acad. Sci. USA 2014, 111, 7349–7354. [Google Scholar] [CrossRef] [Green Version]
- Friedrichs, J.; Schweiger, R.; Geisler, S.; Mix, A.; Wittstock, U.; Müller, C. Novel glucosinolate metabolism in larvae of the leaf beetle Phaedon cochleariae. Insect Biochem. Mol. Biol. 2020, 124, 103431. [Google Scholar] [CrossRef] [PubMed]
- Narbad, A.; Rossiter, J.T. Gut Glucosinolate Metabolism and Isothiocyanate Production. Mol. Nutr. Food Res. 2018, 62, e1700991. [Google Scholar] [CrossRef] [PubMed]
- Albaser, A.; Kazana, E.; Bennett, M.H.; Cebeci, F.; Luang-In, V.; Spanu, P.D.; Rossiter, J.T. Discovery of a Bacterial Glycoside Hydrolase Family 3 (GH3) β-Glucosidase with Myrosinase Activity from a Citrobacter Strain Isolated from Soil. J. Agric. Food Chem. 2016, 64, 1520–1527. [Google Scholar] [CrossRef] [Green Version]
- Hammer, T.J.; Bowers, M.D. Gut microbes may facilitate insect herbivory of chemically defended plants. Oecologia 2015, 179, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Romeo, L.; Iori, R.; Rollin, P.; Bramanti, P.; Mazzon, E. Isothiocyanates: An Overview of Their Antimicrobial Activity against Human Infections. Molecules 2018, 23, 624. [Google Scholar] [CrossRef] [Green Version]
- Dufour, V.; Stahl, M.; Baysse, C. The antibacterial properties of isothiocyanates. Microbiology 2015, 161, 229–243. [Google Scholar] [CrossRef] [Green Version]
- Ugolini, L.; Martini, C.; Lazzeri, L.; D’Avino, L.; Mari, M. Control of postharvest grey mould (Botrytis cinerea Per.: Fr.) on strawberries by glucosinolate-derived allyl-isothiocyanate treatments. Postharvest Biol. Technol. 2014, 90, 34–39. [Google Scholar] [CrossRef]
- Matthiessen, J.N.; Kirkegaard, J.A. Biofumigation and Enhanced Biodegradation: Opportunity and Challenge in Soilborne Pest and Disease Management. CRC. Crit. Rev. Plant Sci. 2006, 25, 235–265. [Google Scholar] [CrossRef]
- Kawakishi, S.; Kaneko, T. Interaction of proteins with allyl isothiocyanate. J. Agric. Food Chem. 1987, 35, 85–88. [Google Scholar] [CrossRef]
- Bertóti, R.; Vasas, G.; Gonda, S.; Nguyen, N.M.; Szőke, É.; Jakab, Á.; Pócsi, I.; Emri, T. Glutathione protects Candida albicans against horseradish volatile oil. J. Basic Microbiol. 2016, 56, 1071–1079. [Google Scholar] [CrossRef]
- Melchini, A.; Traka, M. Biological Profile of Erucin: A New Promising Anticancer Agent from Cruciferous Vegetables. Toxins 2010, 2, 593–612. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y. Allyl isothiocyanate as a cancer chemopreventive phytochemical. Mol. Nutr. Food Res. 2010, 54, 127–135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, P.; Kim, B.; Kim, S.-H.; Srivastava, S.K. Molecular targets of isothiocyanates in cancer: Recent advances. Mol. Nutr. Food Res. 2014, 58, 1685–1707. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramirez, D.; Abellán-Victorio, A.; Beretta, V.; Camargo, A.; Moreno, D.A. Functional Ingredients from Brassicaceae Species: Overview and Perspectives. Int. J. Mol. Sci. 2020, 21, 1998. [Google Scholar] [CrossRef] [Green Version]
- Matteo, R.; Back, M.A.; Reade, J.P.H.; Ugolini, L.; Pagnotta, E.; Lazzeri, L. Effectiveness of defatted seed meals from Brassicaceae with or without crude glycerin against black grass (Alopecurus myosuroides Huds.). Ind. Crop. Prod. 2018, 111, 506–512. [Google Scholar] [CrossRef]
- Lazzeri, L.; Malaguti, L.; Cinti, S.; Ugolini, L.; De Nicola, G.R.; Bagatta, M.; Casadei, N.; D’Avino, L.; Matteo, R.; Patalano, G. The Brassicaceae Biofumigation System for Plant Cultivation and Defence. An Italian Twenty-Year Esperience of Study and Application. Acta Hortic. 2013, 1005, 375–382. [Google Scholar] [CrossRef]
- Ugolini, L.; Pagnotta, E.; Matteo, R.; Malaguti, L.; Di Francesco, A.; Lazzeri, L. Brassica meal-derived allyl-isothiocyanate postharvest application: Influence on strawberry nutraceutical and biochemical parameters. J. Sci. Food Agric. 2019, 99, 4235–4241. [Google Scholar] [CrossRef]
- Canistro, D.; Vivarelli, F.; Ugolini, L.; Pinna, C.; Grandi, M.; Antonazzo, I.C.; Cirillo, S.; Sapone, A.; Cinti, S.; Lazzeri, L.; et al. Digestibility, toxicity and metabolic effects of rapeseed and sunflower protein hydrolysates in mice. Ital. J. Anim. Sci. 2017, 16, 462–473. [Google Scholar] [CrossRef] [Green Version]
- Lucarini, E.; Pagnotta, E.; Micheli, L.; Parisio, C.; Testai, L.; Martelli, A.; Calderone, V.; Matteo, R.; Lazzeri, L.; Mannelli, C. Eruca sativa Meal against Diabetic Neuropathic Pain: An H2S-Mediated Effect of Glucoerucin. Molecules 2019, 24, 3006. [Google Scholar] [CrossRef] [Green Version]
- Franco, P.; Spinozzi, S.; Pagnotta, E.; Lazzeri, L.; Ugolini, L.; Camborata, C.; Roda, A. Development of a liquid chromatography–electrospray ionization–tandem mass spectrometry method for the simultaneous analysis of intact glucosinolates and isothiocyanates in Brassicaceae seeds and functional foods. J. Chromatogr. A 2016, 1428, 154–161. [Google Scholar] [CrossRef]
- Borges, D.; Guzman-Novoa, E.; Goodwin, P.H. Control of the microsporidian parasite Nosema ceranae in honey bees (Apis mellifera) using nutraceutical and immuno-stimulatory compounds. PLoS ONE 2020, 15, e0227484. [Google Scholar] [CrossRef]
- Nanetti, A.; Ugolini, L.; Cilia, G.; Pagnotta, E.; Malaguti, L.; Cardaio, I.; Matteo, R.; Lazzeri, L. Seed Meals from Brassica nigra and Eruca sativa Control Artificial Nosema ceranae Infections in Apis mellifera. Microorganisms 2021, 9, 949. [Google Scholar] [CrossRef] [PubMed]
- Accorti, M. Valutazione numerica degli adulti di Apis mellifica L.: Variazioni e modifiche al metodo dei sesti. Apicoltura 1985, 1, 63–73. [Google Scholar]
- Marchetti, S. Il “Metodo dei sesti” per la valutazione numerica degli adulti in famiglie di Apis mellifera L. Apicoltura 1985, 1, 41–61. [Google Scholar]
- Accorti, M.; Luti, F.; Tarducci, F. Methods for collecting data on natural mortality in bee. Ethol. Ecol. Evol. 1991, 3, 123–126. [Google Scholar] [CrossRef]
- Pérez, J.L.; Higes, M.; Suárez, M.; Llorente, J.; Meana, A. Easy ways to determine honey bee mortality using dead-bee traps. J. Apic. Res. 2015, 40, 25–28. [Google Scholar] [CrossRef]
- Cilia, G.; Garrido, C.; Bonetto, M.; Tesoriero, D.; Nanetti, A. Effect of Api-Bioxal ® and ApiHerb ® Treatments against Nosema ceranae Infection in Apis mellifera Investigated by Two qPCR Methods. Vet. Sci. 2020, 7, 125. [Google Scholar] [CrossRef]
- Cilia, G.; Sagona, S.; Giusti, M.; Jarmela dos Santos, P.E.; Nanetti, A.; Felicioli, A. Nosema ceranae infection in honeybee samples from Tuscanian Archipelago (Central Italy) investigated by two qPCR methods. Saudi J. Biol. Sci. 2019, 26, 1553–1556. [Google Scholar] [CrossRef]
- Cilia, G.; Cardaio, I.; dos Santos, P.E.J.; Ellis, J.D.; Nanetti, A. The first detection of Nosema ceranae (Microsporidia) in the small hive beetle, Aethina tumida Murray (Coleoptera: Nitidulidae). Apidologie 2018, 49, 619–624. [Google Scholar] [CrossRef] [Green Version]
- Cilia, G.; Cabbri, R.; Maiorana, G.; Cardaio, I.; Dall’Olio, R.; Nanetti, A. A novel TaqMan ® assay for Nosema ceranae quantification in honey bee, based on the protein coding gene Hsp70. Eur. J. Protistol. 2018, 63, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Citi, V.; Piragine, E.; Pagnotta, E.; Ugolini, L.; Di Cesare Mannelli, L.; Testai, L.; Ghelardini, C.; Lazzeri, L.; Calderone, V.; Martelli, A. Anticancer properties of erucin, an H2S-releasing isothiocyanate, on human pancreatic adenocarcinoma cells (AsPC-1). Phyther. Res. 2019, 33, 845–855. [Google Scholar] [CrossRef] [Green Version]
- Pasini, F.; Gardini, S.; Marcazzan, G.L.; Caboni, M.F. Buckwheat honeys: Screening of composition and properties. Food Chem. 2013, 141, 2802–2811. [Google Scholar] [CrossRef] [PubMed]
- Galletti, S.; Bernardi, R.; Leoni, O.; Rollin, P.; Palmieri, S. Preparation and Biological Activity of Four Epiprogoitrin Myrosinase-Derived Products. J. Agric. Food Chem. 2001, 49, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Pagnotta, E.; Ugolini, L.; Matteo, R.; Lazzeri, L.; Foschi, L.; Angelini, L.G.; Tavarini, S. Exploring the Camelina sativa value chain: A new opportunity for bio-based products and overall crop sustainability. Riv. Ital. Delle Sostanze Grasse 2019, 96, 259–268. [Google Scholar]
- Zhang, Y. The 1,2-Benzenedithiole-Based Cyclocondensation Assay: A Valuable Tool for the Measurement of Chemopreventive Isothiocyanates. Crit. Rev. Food Sci. Nutr. 2012, 52, 525–532. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clarke, J.D.; Hsu, A.; Riedl, K.; Bella, D.; Schwartz, S.J.; Stevens, J.F.; Ho, E. Bioavailability and inter-conversion of sulforaphane and erucin in human subjects consuming broccoli sprouts or broccoli supplement in a cross-over study design. Pharmacol. Res. 2011, 64, 456–463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costa, C.; Lodesani, M.; Bienefeld, K. Differences in colony phenotypes across different origins and locations: Evidence for genotype by environment interactions in the Italian honeybee (Apis mellifera ligustica)? Apidologie 2012, 43, 634–642. [Google Scholar] [CrossRef] [Green Version]
- DeGrandi-Hoffman, G.; Gage, S.L.; Corby-Harris, V.; Carroll, M.; Chambers, M.; Graham, H.; Watkins deJong, E.; Hidalgo, G.; Calle, S.; Azzouz-Olden, F.; et al. Connecting the nutrient composition of seasonal pollens with changing nutritional needs of honey bee (Apis mellifera L.) colonies. J. Insect Physiol. 2018, 109, 114–124. [Google Scholar] [CrossRef]
- Laomettachit, T.; Liangruksa, M.; Termsaithong, T.; Tangthanawatsakul, A.; Duangphakdee, O. A model of infection in honeybee colonies with social immunity. PLoS ONE 2021, 16, e0247294. [Google Scholar] [CrossRef]
- Soria, A.C.; Martínez-Castro, I.; Sanz, J. Analysis of volatile composition of honey by solid phase microextraction and gas chromatography-mass spectrometry. J. Sep. Sci. USA 2003, 26, 793–801. [Google Scholar] [CrossRef]
- Abdalsamee, M.K.; Giampà, M.; Niehaus, K.; Müller, C. Rapid incorporation of glucosinolates as a strategy used by a herbivore to prevent activation by myrosinases. Insect Biochem. Mol. Biol. 2014, 52, 115–123. [Google Scholar] [CrossRef]
- Zheng, H.; Powell, J.E.; Steele, M.I.; Dietrich, C.; Moran, N.A. Honeybee gut microbiota promotes host weight gain via bacterial metabolism and hormonal signaling. Proc. Natl. Acad. Sci. USA 2017, 114, 4775–4780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Angelino, D.; Dosz, E.B.; Sun, J.; Hoeflinger, J.L.; Van Tassell, M.L.; Chen, P.; Harnly, J.M.; Miller, M.J.; Jeffery, E.H. Myrosinase-dependent and -independent formation and control of isothiocyanate products of glucosinolate hydrolysis. Front. Plant Sci. 2015, 6, 831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Charron, C.S.; Vinyard, B.T.; Ross, S.A.; Seifried, H.E.; Jeffery, E.H.; Novotny, J.A. Absorption and metabolism of isothiocyanates formed from broccoli glucosinolates: Effects of BMI and daily consumption in a randomised clinical trial. Br. J. Nutr. 2018, 120, 1370–1379. [Google Scholar] [CrossRef] [Green Version]
- Charron, C.S.; Vinyard, B.T.; Jeffery, E.H.; Ross, S.A.; Seifried, H.E.; Novotny, J.A. BMI Is Associated with Increased Plasma and Urine Appearance of Glucosinolate Metabolites after Consumption of Cooked Broccoli. Front. Nutr. 2020, 7, 575092. [Google Scholar] [CrossRef]
- Pontoh, J.; Low, N. Purification and characterization of β-glucosidase from honey bees (Apis mellifera). Insect Biochem. Mol. Biol. 2002, 32, 679–690. [Google Scholar] [CrossRef]
- Ricigliano, V.A.; Fitz, W.; Copeland, D.C.; Mott, B.M.; Maes, P.; Floyd, A.S.; Dockstader, A.; Anderson, K.E. The impact of pollen consumption on honey bee (Apis mellifera) digestive physiology and carbohydrate metabolism. Arch. Insect Biochem. Physiol. 2017, 96, e21406. [Google Scholar] [CrossRef]
- Truchado, P.; Tourn, E.; Gallez, L.M.; Moreno, D.A.; Ferreres, F.; Tomás-Barberán, F.A. Identification of Botanical Biomarkers in Argentinean Diplotaxis Honeys: Flavonoids and Glucosinolates. J. Agric. Food Chem. 2010, 58, 12678–12685. [Google Scholar] [CrossRef]
- Zhang, H.; Dong, J.; Zhang, X.; Gao, Y. Identification of Glucosinolate and Its Degradation Product in Honey of Brassica napus L. Food Sci. 2009, 30, 363–366. [Google Scholar]
- Ares, A.M.; Valverde, S.; Nozal, M.J.; Bernal, J.L.; Bernal, J. Development and validation of a specific method to quantify intact glucosinolates in honey by LC–MS/MS. J. Food Compos. Anal. 2016, 46, 114–122. [Google Scholar] [CrossRef]
- Houghton, C.A. Sulforaphane: Its “Coming of Age” as a Clinically Relevant Nutraceutical in the Prevention and Treatment of Chronic Disease. Oxid. Med. Cell. Longev. 2019, 2019, 1–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soria, A.C.; Martínez-Castro, I.; de Lorenzo, C.; Sanz, J. Occurrence of nitriles in Taraxacum labelled honeys. Food Chem. 2008, 107, 439–443. [Google Scholar] [CrossRef]
- Winde, I.; Wittstock, U. Insect herbivore counteradaptations to the plant glucosinolate–myrosinase system. Phytochemistry 2011, 72, 1566–1575. [Google Scholar] [CrossRef]
- Hanschen, F.S.; Schreiner, M. Isothiocyanates, Nitriles, and Epithionitriles from Glucosinolates Are Affected by Genotype and Developmental Stage in Brassica oleracea Varieties. Front. Plant Sci. 2017, 8, 1095. [Google Scholar] [CrossRef] [Green Version]
- Matusheski, N.V.; Jeffery, E.H. Comparison of the Bioactivity of Two Glucoraphanin Hydrolysis Products Found in Broccoli, Sulforaphane and Sulforaphane Nitrile. J. Agric. Food Chem. 2001, 49, 5743–5749. [Google Scholar] [CrossRef]
- Tanii, H. Allyl nitrile: Toxicity and health effects. J. Occup. Health 2017, 59, 104–111. [Google Scholar] [CrossRef] [Green Version]

| E. sativa DSM | B. nigra DSM | |
|---|---|---|
| Moisture (%) | 5.9 ± 0.1 | 6.0 ± 0.1 |
| Protein (%) | 35 | 43.8 |
| Oil (%) | 17.9 ± 0.2 | 8.5 ± 0.2 |
| GSL1 (μmol/g) side chain (R1) | GER 90.5 ± 1.9 | SIN 130.7 ± 4.3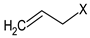 |
| GSL2 (μmol/g) side chain (R2) | GRA 9.3 ± 0.4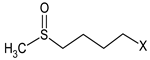 | |
| Phenols (mg GAE/g) | 7.8 ± 0.7 b | 9.5 ± 0.9 a |
| R | N | CTRL– | All Groups | ||
|---|---|---|---|---|---|
| Foragers | PRE | 213.00 ± 3.31 | 199.65 ± 37.01 | 1284.35 ± 297.34 | 565.67 ± 164.32 |
| 7.40 | 82.75 | 664.88 | 636.40 | ||
| POST | 107.79 ± 1.92 | 106.17 ±1.59 | 1448.35 ± 723.73 | 555.10 ± 196.32 | |
| 4.30 | 3.56 | 323.88 | 760.35 | ||
| House bees | PRE | 202.36 ± 8.33 | 264.30 ± 4.67 | 1105.27 ± 245.39 | 523.98 ± 133.63 |
| 18.63 | 10.45 | 548.71 | 517.55 | ||
| POST | 89.62 ± 3.08 | 174.51 ± 4.83 | 840.86 ±170.56 | 368.33 ± 104.09 | |
| 6.88 | 10.79 | 381.38 | 403.12 |
| DSM Patty | ITC Content (pmol/mg) | |
|---|---|---|
| Midgut | Hindgut | |
| R | 38.5 bc | 102.9 a |
| N | 8.3 c | 80.4 ab |
| Honey | GSL Content (nmol/g) | |
|---|---|---|
| SIN | GRA | |
| R | – | 12 ± 3 |
| N | 40 ± 20 | – |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ugolini, L.; Cilia, G.; Pagnotta, E.; Malaguti, L.; Capano, V.; Guerra, I.; Zavatta, L.; Albertazzi, S.; Matteo, R.; Lazzeri, L.; et al. Glucosinolate Bioactivation by Apis mellifera Workers and Its Impact on Nosema ceranae Infection at the Colony Level. Biomolecules 2021, 11, 1657. https://doi.org/10.3390/biom11111657
Ugolini L, Cilia G, Pagnotta E, Malaguti L, Capano V, Guerra I, Zavatta L, Albertazzi S, Matteo R, Lazzeri L, et al. Glucosinolate Bioactivation by Apis mellifera Workers and Its Impact on Nosema ceranae Infection at the Colony Level. Biomolecules. 2021; 11(11):1657. https://doi.org/10.3390/biom11111657
Chicago/Turabian StyleUgolini, Luisa, Giovanni Cilia, Eleonora Pagnotta, Lorena Malaguti, Vittorio Capano, Irene Guerra, Laura Zavatta, Sergio Albertazzi, Roberto Matteo, Luca Lazzeri, and et al. 2021. "Glucosinolate Bioactivation by Apis mellifera Workers and Its Impact on Nosema ceranae Infection at the Colony Level" Biomolecules 11, no. 11: 1657. https://doi.org/10.3390/biom11111657






