Evolution and Adaptation of Legionella pneumophila to Manipulate the Ubiquitination Machinery of Its Amoebae and Mammalian Hosts
Abstract
:1. Introduction
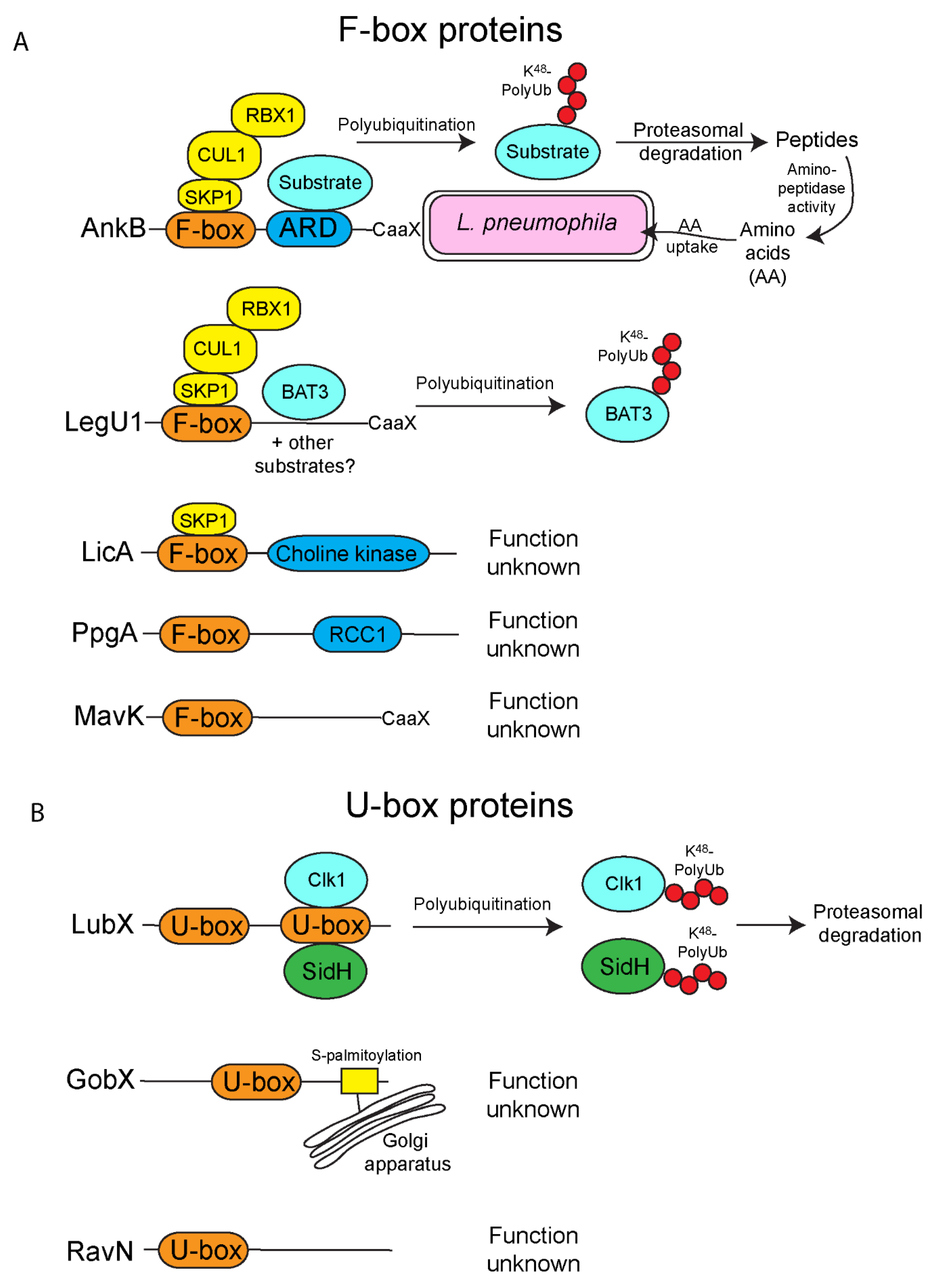
2. F-Box Effectors
3. U-Box Effectors
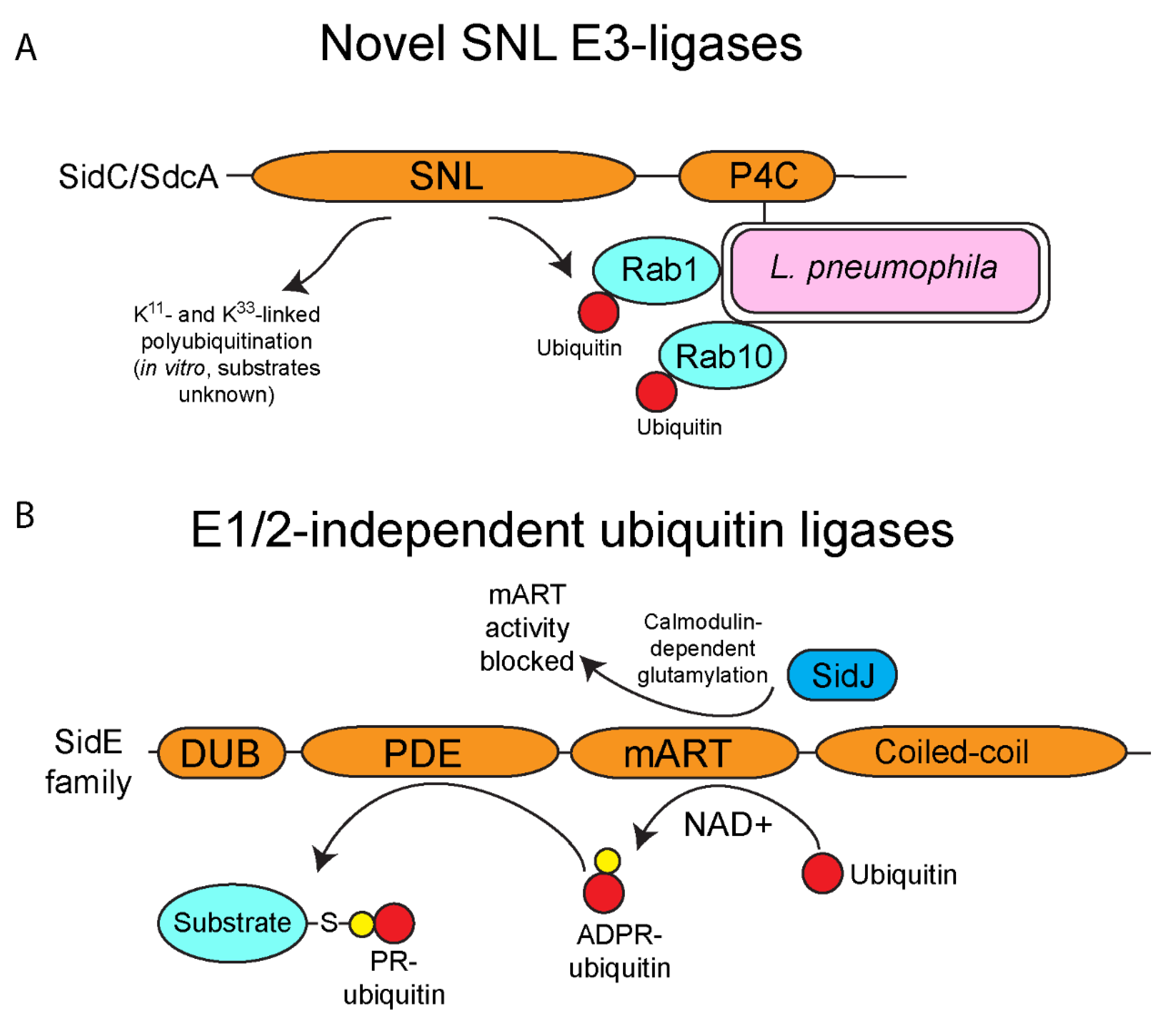
4. E3-Ubiquitin Ligase Activity of SidC/SdcA
5. One Enzyme-Mediated Phosphoribosyl-Ubiquitination by the SidE Family Effectors
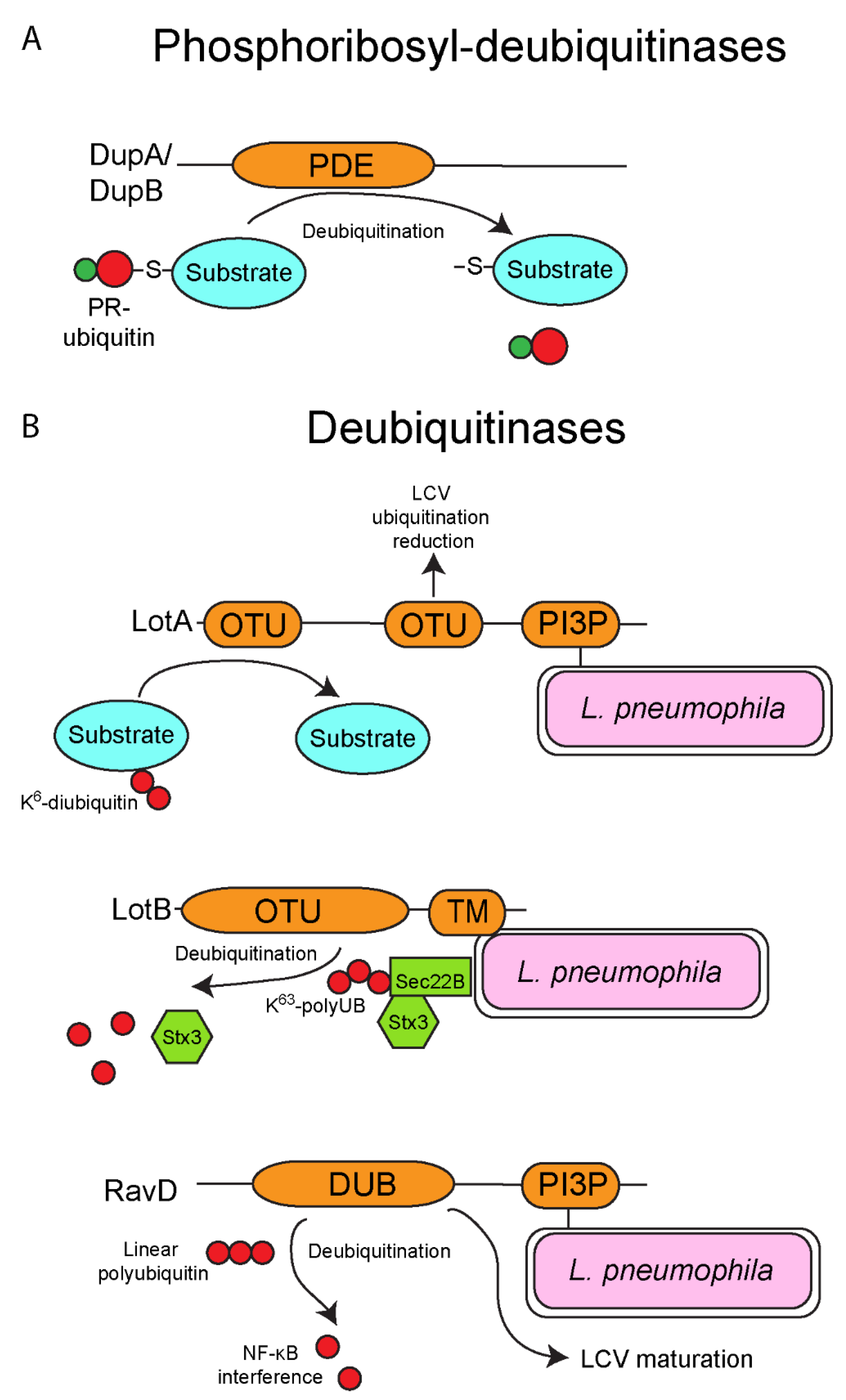
6. Deubiquitinases
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vere, G.; Kealy, R.; Kessler, B.M.; Pinto-Fernandez, A. Ubiquitomics: An Overview and Future. Biomolecules 2020, 10, 1453. [Google Scholar] [CrossRef] [PubMed]
- Kliza, K.; Husnjak, K. Resolving the Complexity of Ubiquitin Networks. Front. Mol. Biosci. 2020, 7, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, L.; Luo, Z.-Q. Post-translational regulation of ubiquitin signaling. J. Cell Biol. 2019, 218, 1776–1786. [Google Scholar] [CrossRef] [PubMed]
- Grice, G.L.; Nathan, J.A. The recognition of ubiquitinated proteins by the proteasome. Cell. Mol. Life Sci. 2016, 73, 3497–3506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kudriaeva, A.A.; Belogurov, A.A. Proteasome: A Nanomachinery of Creative Destruction. Biochemistry 2019, 84, 159–192. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, A.; Nethe, M.; Hordijk, P.L. Ubiquitin links to cytoskeletal dynamics, cell adhesion and migration. Biochem. J. 2012, 442, 13–25. [Google Scholar] [CrossRef] [Green Version]
- Aquila, L.; Atanassov, B.S. Regulation of Histone Ubiquitination in Response to DNA Double Strand Breaks. Cells 2020, 9, 1699. [Google Scholar] [CrossRef]
- Wang, Y.; Argiles-Castillo, D.; Kane, E.I.; Zhou, A.; Spratt, D.E. HECT E3 ubiquitin ligases—emerging insights into their biological roles and disease relevance. J. Cell Sci. 2020, 133, jcs228072. [Google Scholar] [CrossRef]
- Leseigneur, C.; Lê-Bury, P.; Pizarro-Cerdá, J.; Dussurget, O. Emerging Evasion Mechanisms of Macrophage Defenses by Pathogenic Bacteria. Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef]
- Sachdeva, K.; Sundaramurthy, V. The Interplay of Host Lysosomes and Intracellular Pathogens. Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef]
- Kubelkova, K.; Macela, A. Innate Immune Recognition: An Issue More Complex Than Expected. Front. Cell. Infect. Microbiol. 2019, 9, 241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Curto, P.; Riley, S.P.; Simões, I.; Martinez, J.J. Macrophages Infected by a Pathogen and a Non-pathogen Spotted Fever Group Rickettsia Reveal Differential Reprogramming Signatures Early in Infection. Front. Cell. Infect. Microbiol. 2019, 9, 97. [Google Scholar] [CrossRef] [PubMed]
- Faris, R.; Andersen, S.E.; McCullough, A.; Gourronc, F.; Klingelhutz, A.J.; Weber, M.M. Chlamydia trachomatis Serovars Drive Differential Production of Proinflammatory Cytokines and Chemokines Depending on the Type of Cell Infected. Front. Cell. Infect. Microbiol. 2019, 9, 399. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.R.; Newton, P.; Lau, N.; Newton, H.J. Interfering with Autophagy: The Opposing Strategies Deployed by Legionella pneumophila and Coxiella burnetii Effector Proteins. Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Snäkä, T.; Fasel, N. Behind the Scenes: Nod-Like Receptor X1 Controls Inflammation and Metabolism. Front. Cell. Infect. Microbiol. 2020, 10, 609812. [Google Scholar] [CrossRef] [PubMed]
- Mambu, J.; Barilleau, E.; Fragnet-Trapp, L.; Le Vern, Y.; Olivier, M.; Sadrin, G.; Grépinet, O.; Taieb, F.; Velge, P.; Wiedemann, A. Rck of Salmonella typhimurium Delays the Host Cell Cycle to Facilitate Bacterial Invasion. Front. Cell. Infect. Microbiol. 2020, 10, 586934. [Google Scholar] [CrossRef] [PubMed]
- Maurya, R.K.; Bharti, S.; Krishnan, M.Y. Triacylglycerols: Fuelling the Hibernating Mycobacterium tuberculosis. Front. Cell. Infect. Microbiol. 2019, 8, 450. [Google Scholar] [CrossRef]
- Del Portillo, P.; García-Morales, L.; Menéndez, M.C.; Anzola, J.M.; Rodríguez, J.G.; Helguera-Repetto, A.C.; Ares, M.A.; Prados-Rosales, R.; Gonzalez-Y-Merchand, J.A.; García, M.J. Hypoxia Is Not a Main Stress When Mycobacterium tuberculosis Is in a Dormancy-Like Long-Chain Fatty Acid Environment. Front. Cell. Infect. Microbiol. 2019, 8, 449. [Google Scholar] [CrossRef] [Green Version]
- Augenstreich, J.; Briken, V. Host Cell Targets of Released Lipid and Secreted Protein Effectors of Mycobacterium tuberculosis. Front. Cell. Infect. Microbiol. 2020, 10, 595029. [Google Scholar] [CrossRef]
- Arora, S.K.; Naqvi, N.; Alam, A.; Ahmad, J.; Alsati, B.S.; Sheikh, J.A.; Kumar, P.; Mitra, D.K.; Rahman, S.A.; Hasnain, S.E.; et al. Mycobacterium smegmatis Bacteria Expressing Mycobacterium tuberculosis-Specific Rv1954A Induce Macrophage Activation and Modulate the Immune Response. Front. Cell. Infect. Microbiol. 2020, 10, 564565. [Google Scholar] [CrossRef]
- Garg, R.; Borbora, S.M.; Bansia, H.; Rao, S.; Singh, P.; Verma, R.; Balaji, K.N.; Nagaraja, V. Mycobacterium tuberculosis Calcium Pump CtpF Modulates the Autophagosome in an mTOR-Dependent Manner. Front. Cell. Infect. Microbiol. 2020, 10, 461. [Google Scholar] [CrossRef] [PubMed]
- Çakır, T.; Panagiotou, G.; Uddin, R.; Durmuş, S. Novel Approaches for Systems Biology of Metabolism-Oriented Pathogen-Human Interactions: A Mini-Review. Front. Cell. Infect. Microbiol. 2020, 10, 52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Price, C.T.D.; Al-Quadan, T.; Santic, M.; Rosenshine, I.; Abu Kwaik, Y. Host Proteasomal Degradation Generates Amino Acids Essential for Intracellular Bacterial Growth. Science 2011, 334, 1553–1557. [Google Scholar] [CrossRef] [PubMed]
- Mohareer, K.; Medikonda, J.; Vadankula, G.R.; Banerjee, S. Mycobacterial Control of Host Mitochondria: Bioenergetic and Metabolic Changes Shaping Cell Fate and Infection Outcome. Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, R.; Chowdhury, A.R.; Mukherjee, D.; Chakravortty, D. Lipid larceny: Channelizing host lipids for establishing successful pathogenesis by bacteria. Virulence 2021, 12, 195–216. [Google Scholar] [CrossRef] [PubMed]
- Kamanova, J. Bordetella Type III Secretion Injectosome and Effector Proteins. Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sun, J.; Wan, L.; Yang, X.; Lin, H.; Zhang, Y.; He, X.; Zhong, H.; Guan, K.; Min, M.; et al. The Shigella Type III Secretion Effector IpaH4.5 Targets NLRP3 to Activate Inflammasome Signaling. Front. Cell. Infect. Microbiol. 2020, 10, 511798. [Google Scholar] [CrossRef]
- Gan, J.; Scott, N.E.; Newson, J.P.M.; Wibawa, R.R.; Lung, T.W.F.; Pollock, G.L.; Ng, G.Z.; Van Driel, I.; Pearson, J.S.; Hartland, E.L.; et al. The Salmonella Effector SseK3 Targets Small Rab GTPases. Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef]
- Feria, J.M.; Valvano, M.A. An Overview of Anti-Eukaryotic T6SS Effectors. Front. Cell. Infect. Microbiol. 2020, 10, 584751. [Google Scholar] [CrossRef]
- Lou, L.; Zhang, P.; Piao, R.; Wang, Y. Salmonella Pathogenicity Island 1 (SPI-1) and Its Complex Regulatory Network. Front. Cell. Infect. Microbiol. 2019, 9, 270. [Google Scholar] [CrossRef] [Green Version]
- Green, R.S.; Izac, J.R.; Naimi, W.A.; O’Bier, N.; Breitschwerdt, E.B.; Marconi, R.T.; Carlyon, J.A. Ehrlichia chaffeensis EplA Interaction with Host Cell Protein Disulfide Isomerase Promotes Infection. Front. Cell. Infect. Microbiol. 2020, 10, 500. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Luo, J.; Li, S. Bacteria-Catalyzed Arginine Glycosylation in Pathogens and Host. Front. Cell. Infect. Microbiol. 2020, 10, 185. [Google Scholar] [CrossRef] [PubMed]
- Herhaus, L.; Dikic, I. Regulation of Salmonella-host cell interactions via the ubiquitin system. Int. J. Med Microbiol. 2018, 308, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Chai, Q.; Wang, L.; Liu, C.H.; Ge, B. New insights into the evasion of host innate immunity by Mycobacterium tuberculosis. Cell. Mol. Immunol. 2020, 17, 901–913. [Google Scholar] [CrossRef] [PubMed]
- Tanner, K.; Brzovic, P.; Rohde, J.R. The bacterial pathogen-ubiquitin interface: Lessons learned from Shigella. Cell. Microbiol. 2014, 17, 35–44. [Google Scholar] [CrossRef]
- Qiu, J.; Luo, Z.-Q. Legionella and Coxiella effectors: Strength in diversity and activity. Nat. Rev. Genet. 2017, 15, 591–605. [Google Scholar] [CrossRef]
- Huang, B.; Ojogun, N.; Ragland, S.A.; Carlyon, J.A. Monoubiquitinated proteins decorate the Anaplasma phagocytophilum-occupied vacuolar membrane. FEMS Immunol. Med. Microbiol. 2012, 64, 32–41. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Zhu, Y. Diversity of bacterial manipulation of the host ubiquitin pathways. Cell. Microbiol. 2015, 17, 26–34. [Google Scholar] [CrossRef]
- Hayward, R.J.; Marsh, J.W.; Humphrys, M.S.; Huston, W.M.; Myers, G.S.A. Early Transcriptional Landscapes of Chlamydia trachomatis-Infected Epithelial Cells at Single Cell Resolution. Front. Cell. Infect. Microbiol. 2019, 9, 392. [Google Scholar] [CrossRef] [Green Version]
- Kunz, T.C.; Götz, R.; Sauer, M.; Rudel, T. Detection of Chlamydia Developmental Forms and Secreted Effectors by Expansion Microscopy. Front. Cell. Infect. Microbiol. 2019, 9, 276. [Google Scholar] [CrossRef] [Green Version]
- Bartra, S.S.; Lorica, C.; Qian, L.; Gong, X.; Bahnan, W.; Barreras, H.B., Jr.; Hernandez, R.; Li, Z.; Plano, G.V.; Schesser, K. Chromosomally-Encoded Yersinia pestis Type III Secretion Effector Proteins Promote Infection in Cells and in Mice. Front. Cell. Infect. Microbiol. 2019, 9, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kitao, T.; Nagai, H.; Kubori, T. Divergence of Legionella Effectors Reversing Conventional and Unconventional Ubiquitination. Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Best, A.; Abu Kwaik, Y. Evolution of the Arsenal of Legionella pneumophila Effectors to Modulate Protist Hosts. mBio 2018, 9, e01313-18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomez-Valero, L.; Buchrieser, C. Intracellular parasitism, the driving force of evolution of Legionella pneumophila and the genus Legionella. Genes Immun. 2019, 20, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Mondino, S.; Schmidt, S.; Buchrieser, C. Molecular Mimicry: A Paradigm of Host-Microbe Coevolution Illustrated by Legionella. mBio 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Vassiliadis, D.; Ong, S.Y.; Bennett-Wood, V.; Sugimoto, C.; Yamagishi, J.; Hartland, E.L.; Pasricha, S. Legionella pneumophila Infection Rewires the Acanthamoeba castellanii Transcriptome, Highlighting a Class of Sirtuin Genes. Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Fields, B.S. The molecular ecology of legionellae. Trends Microbiol. 1996, 4, 286–290. [Google Scholar] [CrossRef]
- Harb, O.S.; Gao, L.-Y.; Abu Kwaik, Y. From protozoa to mammalian cells: A new paradigm in the life cycle of intracellular bacterial pathogens. Minireview. Environ. Microbiol. 2000, 2, 251–265. [Google Scholar] [CrossRef]
- Molmeret, M.; Horn, M.; Wagner, M.; Santic, M.; Abu Kwaik, Y. Amoebae as Training Grounds for Intracellular Bacterial Pathogens. Appl. Environ. Microbiol. 2005, 71, 20–28. [Google Scholar] [CrossRef] [Green Version]
- Haenssler, E.; Ramabhadran, V.; Murphy, C.S.; Heidtman, M.I.; Isberg, R.R. Endoplasmic Reticulum Tubule Protein Reticulon 4 Associates with the Legionella pneumophila Vacuole and with Translocated Substrate Ceg9. Infect. Immun. 2015, 83, 3479–3489. [Google Scholar] [CrossRef] [Green Version]
- Isberg, R.R.; O’Connor, T.J.; Heidtman, M. The Legionella pneumophila replication vacuole: Making a cosy niche inside host cells. Nat. Rev. Genet. 2008, 7, 13–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kagan, J.C.; Roy, C.R. Legionella phagosomes intercept vesicular traffic from endoplasmic reticulum exit sites. Nat. Cell Biol. 2002, 4, 945–954. [Google Scholar] [CrossRef] [PubMed]
- Younes, S.; Al-Sulaiti, A.; Nasser, E.A.A.; Najjar, H.; Kamareddine, L. Drosophila as a Model Organism in Host–Pathogen Interaction Studies. Front. Cell. Infect. Microbiol. 2020, 10, 214. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, M.A. Phagocytosis of the legionnaires’ disease bacterium (legionella pneumophila) occurs by a novel mechanism: Engulfment within a Pseudopod coil. Cell 1984, 36, 27–33. [Google Scholar] [CrossRef]
- Burstein, D.; Amaro, F.; Zusman, T.; Lifshitz, Z.; Cohen, O.; Gilbert, J.A.; Pupko, T.; Shuman, H.A.; Segal, G. Genomic analysis of 38 Legionella species identifies large and diverse effector repertoires. Nat. Genet. 2016, 48, 167–175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, W.; Banga, S.; Tan, Y.; Zheng, C.; Stephenson, R.; Gately, J.; Luo, Z.-Q. Comprehensive Identification of Protein Substrates of the Dot/Icm Type IV Transporter of Legionella pneumophila. PLoS ONE 2011, 6, e17638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Price, C.T.; Al-Khodor, S.; Al-Quadan, T.; Santic, M.; Habyarimana, F.; Kalia, A.; Abu Kwaik, Y. Molecular Mimicry by an F-Box Effector of Legionella pneumophila Hijacks a Conserved Polyubiquitination Machinery within Macrophages and Protozoa. PLOS Pathog. 2009, 5, e1000704. [Google Scholar] [CrossRef]
- Horwitz, M.A. The Legionnaires’ disease bacterium (Legionella pneumophila) inhibits phagosome-lysosome fusion in human monocytes. J. Exp. Med. 1983, 158, 2108–2126. [Google Scholar] [CrossRef] [Green Version]
- Horwitz, M.A. Formation of a novel phagosome by the Legionnaires’ disease bacterium (Legionella pneumophila) in human monocytes. J. Exp. Med. 1983, 158, 1319–1331. [Google Scholar] [CrossRef] [Green Version]
- Horwitz, M.A.; Silverstein, S.C. Legionnaires’ Disease Bacterium (Legionella pneumophila) Multiplies Intracellularly in Human Monocytes. J. Clin. Investig. 1980, 66, 441–450. [Google Scholar] [CrossRef] [Green Version]
- Al-Khodor, S.; Price, C.T.; Habyarimana, F.; Kalia, A.; Abu Kwaik, Y. A Dot/Icm-translocated ankyrin protein of Legionella pneumophila is required for intracellular proliferation within human macrophages and protozoa. Mol. Microbiol. 2008, 70, 908–923. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ensminger, A.W.; Isberg, R.R. E3 Ubiquitin Ligase Activity and Targeting of BAT3 by Multiple Legionella pneumophila Translocated Substrates. Infect. Immun. 2010, 78, 3905–3919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, K.M.; Busino, L. The Biology of F-box Proteins: The SCF Family of E3 Ubiquitin Ligases. Adv. Exp. Med. Biol. 2020, 1217, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Tekcham, D.S.; Chen, D.; Liu, Y.; Ling, T.; Zhang, Y.; Chen, H.; Wang, W.; Otkur, W.; Qi, H.; Xia, T.; et al. F-box proteins and cancer: An update from functional and regulatory mechanism to therapeutic clinical prospects. Theranostics 2020, 10, 4150–4167. [Google Scholar] [CrossRef] [PubMed]
- Price, C.T.D.; Abu Kwaik, Y. Exploitation of Host Polyubiquitination Machinery through Molecular Mimicry by Eukaryotic-Like Bacterial F-Box Effectors. Front. Microbiol. 2010, 1, 122. [Google Scholar] [CrossRef] [Green Version]
- Lomma, M.; Dervins-Ravault, D.; Rolando, M.; Nora, T.; Newton, H.J.; Sansom, F.M.; Sahr, T.; Gomez-Valero, L.; Jules, M.; Hartland, E.L.; et al. The Legionella pneumophila F-box protein Lpp2082 (AnkB) modulates ubiquitination of the host protein parvin B and promotes intracellular replication. Cell. Microbiol. 2010, 12, 1272–1291. [Google Scholar] [CrossRef]
- Price, C.T.; Al-Quadan, T.; Santic, M.; Jones, S.C.; Abu Kwaik, Y. Exploitation of conserved eukaryotic host cell farnesylation machinery by an F-box effector of Legionella pneumophila. J. Exp. Med. 2010, 207, 1713–1726. [Google Scholar] [CrossRef] [Green Version]
- Wong, K.; Perpich, J.D.; Kozlov, G.; Cygler, M.; Abu Kwaik, Y.; Gehring, K. Structural Mimicry by a Bacterial F Box Effector Hijacks the Host Ubiquitin-Proteasome System. Structure 2017, 25, 376–383. [Google Scholar] [CrossRef] [Green Version]
- Price, C.T.D.; Richards, A.M.; Von Dwingelo, J.E.; Samara, H.A.; Abu Kwaik, Y. Amoeba host-Legionella synchronization of amino acid auxotrophy and its role in bacterial adaptation and pathogenic evolution. Environ. Microbiol. 2013, 16, 350–358. [Google Scholar] [CrossRef] [Green Version]
- Jiang, H.; Zhang, X.; Chen, X.; Aramsangtienchai, P.; Tong, Z.; Lin, H. Protein Lipidation: Occurrence, Mechanisms, Biological Functions, and Enabling Technologies. Chem. Rev. 2018, 118, 919–988. [Google Scholar] [CrossRef]
- Perpich, J.D.; Kalia, A.; Price, C.T.D.; Jones, S.C.; Wong, K.; Gehring, K.; Abu Kwaik, Y. Divergent evolution of Di-lysine ER retention vs. farnesylation motif-mediated anchoring of the AnkB virulence effector to the Legionella-containing vacuolar membrane. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Price, C.T.D.; Merchant, M.; Jones, S.; Best, A.; Von Dwingelo, J.; Lawrenz, M.B.; Alam, N.; Schueler-Furman, O.; Abu Kwaik, Y. Host FIH-Mediated Asparaginyl Hydroxylation of Translocated Legionella pneumophila Effectors. Front. Cell. Infect. Microbiol. 2017, 7, 54. [Google Scholar] [CrossRef] [PubMed]
- Strowitzki, M.J.; Cummins, E.P.; Taylor, C.T. Protein Hydroxylation by Hypoxia-Inducible Factor (HIF) Hydroxylases: Unique or Ubiquitous? Cells 2019, 8, 384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bruckert, W.M.; Abu Kwaik, Y. Lysine11-Linked Polyubiquitination of the AnkB F-Box Effector of Legionella pneumophila. Infect. Immun. 2015, 84, 99–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rothmeier, E.; Pfaffinger, G.; Hoffmann, C.; Harrison, C.F.; Grabmayr, H.; Repnik, U.; Hannemann, M.; Wölke, S.; Bausch, A.; Griffiths, G.; et al. Activation of Ran GTPase by a Legionella Effector Promotes Microtubule Polymerization, Pathogen Vacuole Motility and Infection. PLOS Pathog. 2013, 9, e1003598. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.-G.; Ye, Y. Bag6/Bat3/Scythe: A novel chaperone activity with diverse regulatory functions in protein biogenesis and degradation. BioEssays 2013, 35, 377–385. [Google Scholar] [CrossRef]
- Ivanov, S.S.; Charron, G.; Hang, H.C.; Roy, C.R. Lipidation by the Host Prenyltransferase Machinery Facilitates Membrane Localization of Legionella pneumophila Effector Proteins. J. Biol. Chem. 2010, 285, 34686–34698. [Google Scholar] [CrossRef] [Green Version]
- Kubori, T.; Hyakutake, A.; Nagai, H. Legionella translocates an E3 ubiquitin ligase that has multiple U-boxes with distinct functions. Mol. Microbiol. 2008, 67, 1307–1319. [Google Scholar] [CrossRef]
- Kubori, T.; Shinzawa, N.; Kanuka, H.; Nagai, H. Legionella Metaeffector Exploits Host Proteasome to Temporally Regulate Cognate Effector. PLOS Pathog. 2010, 6, e1001216. [Google Scholar] [CrossRef] [Green Version]
- Lin, Y.-H.; Lucas, M.; Evans, T.R.; Abascal-Palacios, G.; Doms, A.G.; Beauchene, N.A.; Rojas, A.L.; Hierro, A.; Machner, M.P. RavN is a member of a previously unrecognized group of Legionella pneumophila E3 ubiquitin ligases. PLOS Pathog. 2018, 14, e1006897. [Google Scholar] [CrossRef] [Green Version]
- Lin, Y.-H.; Doms, A.G.; Cheng, E.; Kim, B.; Evans, T.R.; Machner, M.P. Host Cell-catalyzed S-Palmitoylation Mediates Golgi Targeting of the Legionella Ubiquitin Ligase GobX. J. Biol. Chem. 2015, 290, 25766–25781. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hatakeyama, S.; Nakayama, K.-I. U-box proteins as a new family of ubiquitin ligases. Biochem. Biophys. Res. Commun. 2003, 302, 635–645. [Google Scholar] [CrossRef]
- Quaile, A.T.; Urbanus, M.L.; Stogios, P.J.; Nocek, B.; Skarina, T.; Ensminger, A.W.; Savchenko, A. Molecular Characterization of LubX: Functional Divergence of the U-Box Fold by Legionella pneumophila. Structure 2015, 23, 1459–1469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Urbanus, M.L.; Quaile, A.T.; Stogios, P.J.; Morar, M.; Rao, C.; Di Leo, R.; Evdokimova, E.; Lam, M.; Oatway, C.; Cuff, M.E.; et al. Diverse mechanisms of metaeffector activity in an intracellular bacterial pathogen, Legionella pneumophila. Mol. Syst. Biol. 2016, 12, 893. [Google Scholar] [CrossRef] [PubMed]
- Ragaz, C.; Pietsch, H.; Urwyler, S.; Tiaden, A.; Weber, S.S.; Hilbi, H. The Legionella pneumophila phosphatidylinositol-4 phosphate-binding type IV substrate SidC recruits endoplasmic reticulum vesicles to a replication-permissive vacuole. Cell. Microbiol. 2008, 10, 2416–2433. [Google Scholar] [CrossRef]
- Brombacher, E.; Urwyler, S.; Ragaz, C.; Weber, S.S.; Kami, K.; Overduin, M.; Hilbi, H. Rab1 Guanine Nucleotide Exchange Factor SidM Is a Major Phosphatidylinositol 4-Phosphate-binding Effector Protein of Legionella pneumophila. J. Biol. Chem. 2009, 284, 4846–4856. [Google Scholar] [CrossRef] [Green Version]
- Horenkamp, F.A.; Mukherjee, S.; Alix, E.; Schauder, C.M.; Hubber, A.M.; Roy, C.R.; Reinisch, K.M. Legionella pneumophila Subversion of Host Vesicular Transport by SidC Effector Proteins. Traffic 2014, 15, 488–499. [Google Scholar] [CrossRef] [Green Version]
- Dolinsky, S.; Haneburger, I.; Cichy, A.; Hannemann, M.; Itzen, A.; Hilbi, H. The Legionella longbeachae Icm/Dot Substrate SidC Selectively Binds Phosphatidylinositol 4-Phosphate with Nanomolar Affinity and Promotes Pathogen Vacuole-Endoplasmic Reticulum Interactions. Infect. Immun. 2014, 82, 4021–4033. [Google Scholar] [CrossRef] [Green Version]
- Luo, X.; Wasilko, D.J.; Liu, Y.; Sun, J.; Wu, X.; Luo, Z.-Q.; Mao, Y. Structure of the Legionella Virulence Factor, SidC Reveals a Unique PI(4)P-Specific Binding Domain Essential for Its Targeting to the Bacterial Phagosome. PLOS Pathog. 2015, 11, e1004965. [Google Scholar] [CrossRef]
- Hsu, F.; Luo, X.; Qiu, J.; Teng, Y.-B.; Jin, J.; Smolka, M.B.; Luo, Z.-Q.; Mao, Y. The Legionella effector SidC defines a unique family of ubiquitin ligases important for bacterial phagosomal remodeling. Proc. Natl. Acad. Sci. USA 2014, 111, 10538–10543. [Google Scholar] [CrossRef] [Green Version]
- Wasilko, D.J.; Huang, Q.; Mao, Y. Insights into the ubiquitin transfer cascade catalyzed by the Legionella effector SidC. eLife 2018, 7, e36154. [Google Scholar] [CrossRef] [PubMed]
- Jeng, E.E.; Bhadkamkar, V.; Ibe, N.U.; Gause, H.; Jiang, L.; Chan, J.; Jian, R.; Jimenez-Morales, D.; Stevenson, E.; Krogan, N.J.; et al. Systematic Identification of Host Cell Regulators of Legionella pneumophila Pathogenesis Using a Genome-wide CRISPR Screen. Cell Host Microbe 2019, 26, 551–563.e6. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Sheedlo, M.J.; Yu, K.; Tan, Y.; Nakayasu, E.S.; Das, C.; Liu, X.; Luo, Z.Q. Ubiquitination independent of E1 and E2 enzymes by bacterial effectors. Nature 2016, 533, 120–124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhogaraju, S.; Kalayil, S.; Liu, Y.; Bonn, F.; Colby, T.; Matic, I.; Dikic, I. Phosphoribosylation of Ubiquitin Promotes Serine Ubiquitination and Impairs Conventional Ubiquitination. Cell 2016, 167, 1636–1649.e13. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shi, M.; Feng, H.; Zhu, Y.; Liu, S.; Gao, A.; Gao, P. Structural Insights into Non-canonical Ubiquitination Catalyzed by SidE. Cell 2018, 173, 1231–1243.e16. [Google Scholar] [CrossRef] [Green Version]
- Dong, Y.; Mu, Y.; Xie, Y.; Zhang, Y.; Han, Y.; Zhou, Y.; Wang, W.; Liu, Z.; Wu, M.; Wang, H.; et al. Structural basis of ubiquitin modification by the Legionella effector SdeA. Nature 2018, 557, 674–678. [Google Scholar] [CrossRef]
- Akturk, A.; Wasilko, D.J.; Wu, X.; Liu, Y.; Zhang, Y.; Qiu, J.; Luo, Z.-Q.; Reiter, K.H.; Brzovic, P.S.; Klevit, R.E.; et al. Mechanism of phosphoribosyl-ubiquitination mediated by a single Legionella effector. Nature 2018, 557, 729–733. [Google Scholar] [CrossRef]
- Kalayil, S.; Bhogaraju, S.; Bonn, F.; Shin, D.; Liu, Y.; Gan, N.; Basquin, J.; Grumati, P.; Luo, Z.-Q.; Dikic, I. Insights into catalysis and function of phosphoribosyl-linked serine ubiquitination. Nature 2018, 557, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Kim, L.; Kwon, D.H.; Kim, B.H.; Kim, J.; Park, M.R.; Park, Z.-Y.; Song, H.K. Structural and Biochemical Study of the Mono-ADP-Ribosyltransferase Domain of SdeA, a Ubiquitylating/Deubiquitylating Enzyme from Legionella pneumophila. J. Mol. Biol. 2018, 430, 2843–2856. [Google Scholar] [CrossRef] [PubMed]
- Kotewicz, K.M.; Ramabhadran, V.; Sjoblom, N.; Vogel, J.P.; Haenssler, E.; Zhang, M.; Behringer, J.; Scheck, R.A.; Isberg, R.R. A Single Legionella Effector Catalyzes a Multistep Ubiquitination Pathway to Rearrange Tubular Endoplasmic Reticulum for Replication. Cell Host Microbe 2017, 21, 169–181. [Google Scholar] [CrossRef] [Green Version]
- Havey, J.C.; Roy, C.R. Toxicity and SidJ-Mediated Suppression of Toxicity Require Distinct Regions in the SidE Family of Legionella pneumophila Effectors. Infect. Immun. 2015, 83, 3506–3514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeong, K.C.; Sexton, J.A.; Vogel, J.P. Spatiotemporal Regulation of a Legionella pneumophila T4SS Substrate by the Metaeffector SidJ. PLoS Pathog. 2015, 11, e1004695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, J.; Yu, K.; Fei, X.; Liu, Y.; Nakayasu, E.S.; Piehowski, P.D.; Shaw, J.B.; Puvar, K.; Das, C.; Liu, X.; et al. A unique deubiquitinase that deconjugates phosphoribosyl-linked protein ubiquitination. Cell Res. 2017, 27, 865–881. [Google Scholar] [CrossRef] [PubMed]
- Black, M.H.; Osinski, A.; Gradowski, M.; Servage, K.; Pawłowski, K.; Tomchick, D.R.; Tagliabracci, V. Bacterial pseudokinase catalyzes protein polyglutamylation to inhibit the SidE-family ubiquitin ligases. Science 2019, 364, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Gan, N.; Zhen, X.; Liu, Y.; Xu, X.; He, C.; Qiu, J.; Liu, Y.; Fujimoto, G.M.; Nakayasu, E.S.; Zhou, B.; et al. Regulation of phosphoribosyl ubiquitination by a calmodulin-dependent glutamylase. Nature 2019, 572, 387–391. [Google Scholar] [CrossRef]
- Bhogaraju, S.; Bonn, F.; Mukherjee, R.; Adams, M.; Pfleiderer, M.M.; Galej, W.P.; Matkovic, V.; Lopez-Mosqueda, J.; Kalayil, S.; Shin, D.; et al. Inhibition of bacterial ubiquitin ligases by SidJ–calmodulin catalysed glutamylation. Nature 2019, 572, 382–386. [Google Scholar] [CrossRef]
- Sulpizio, A.; Minelli, M.E.; Wan, M.; Burrowes, P.D.; Wu, X.; Sanford, E.J.; Shin, J.-H.; Williams, B.C.; Goldberg, M.L.; Smolka, M.B.; et al. Protein polyglutamylation catalyzed by the bacterial calmodulin-dependent pseudokinase SidJ. eLife 2019, 8, 8. [Google Scholar] [CrossRef]
- Wan, M.; Sulpizio, A.G.; Akturk, A.; Beck, W.H.J.; Lanz, M.; Faça, V.M.; Smolka, M.B.; Vogel, J.P.; Mao, Y. Deubiquitination of phosphoribosyl-ubiquitin conjugates by phosphodiesterase-domain–containing Legionella effectors. Proc. Natl. Acad. Sci. USA 2019, 116, 23518–23526. [Google Scholar] [CrossRef]
- Shin, D.; Mukherjee, R.; Liu, Y.; Gonzalez, A.; Bonn, F.; Liu, Y.; Rogov, V.V.; Heinz, M.; Stolz, A.; Hummer, G.; et al. Regulation of Phosphoribosyl-Linked Serine Ubiquitination by Deubiquitinases DupA and DupB. Mol. Cell 2020, 77, 164–179.e6. [Google Scholar] [CrossRef] [Green Version]
- Kubori, T.; Kitao, T.; Ando, H.; Nagai, H. LotA, a Legionella deubiquitinase, has dual catalytic activity and contributes to intracellular growth. Cell. Microbiol. 2018, 20, e12840. [Google Scholar] [CrossRef]
- Ma, K.; Zhen, X.; Zhou, B.; Gan, N.; Cao, Y.; Fan, C.; Ouyang, S.; Luo, Z.-Q.; Qiu, J. The bacterial deubiquitinase Ceg23 regulates the association of Lys-63–linked polyubiquitin molecules on the Legionella phagosome. J. Biol. Chem. 2020, 295, 1646–1657. [Google Scholar] [CrossRef] [PubMed]
- Kitao, T.; Taguchi, K.; Seto, S.; Arasaki, K.; Ando, H.; Nagai, H.; Kubori, T. Legionella Manipulates Non-canonical SNARE Pairing Using a Bacterial Deubiquitinase. Cell Rep. 2020, 32, 108107. [Google Scholar] [CrossRef] [PubMed]
- Risselada, H.; Mayer, A. SNAREs, tethers and SM proteins: How to overcome the final barriers to membrane fusion? Biochem. J. 2020, 477, 243–258. [Google Scholar] [CrossRef] [PubMed]
- Derré, I.; Isberg, R.R. Legionella pneumophila Replication Vacuole Formation Involves Rapid Recruitment of Proteins of the Early Secretory System. Infect. Immun. 2004, 72, 3048–3053. [Google Scholar] [CrossRef] [Green Version]
- Kagan, J.C.; Stein, M.-P.; Pypaert, M.; Roy, C.R. Legionella Subvert the Functions of Rab1 and Sec22b to Create a Replicative Organelle. J. Exp. Med. 2004, 199, 1201–1211. [Google Scholar] [CrossRef] [Green Version]
- Arasaki, K.; Roy, C.R. Legionella pneumophila Promotes Functional Interactions between Plasma Membrane Syntaxins and Sec22b. Traffic 2010, 11, 587–600. [Google Scholar] [CrossRef] [Green Version]
- Wan, M.; Wang, X.; Huang, C.; Xu, D.; Wang, Z.; Zhou, Y.; Zhu, Y. A bacterial effector deubiquitinase specifically hydrolyses linear ubiquitin chains to inhibit host inflammatory signalling. Nat. Microbiol. 2019, 4, 1282–1293. [Google Scholar] [CrossRef]
- Pike, C.M.; Boyer-Andersen, R.; Kinch, L.N.; Caplan, J.L.; Neunuebel, M.R. The Legionella effector RavD binds phosphatidylinositol-3-phosphate and helps suppress endolysosomal maturation of the Legionella-containing vacuole. J. Biol. Chem. 2019, 294, 6405–6415. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Price, C.T.D.; Abu Kwaik, Y. Evolution and Adaptation of Legionella pneumophila to Manipulate the Ubiquitination Machinery of Its Amoebae and Mammalian Hosts. Biomolecules 2021, 11, 112. https://doi.org/10.3390/biom11010112
Price CTD, Abu Kwaik Y. Evolution and Adaptation of Legionella pneumophila to Manipulate the Ubiquitination Machinery of Its Amoebae and Mammalian Hosts. Biomolecules. 2021; 11(1):112. https://doi.org/10.3390/biom11010112
Chicago/Turabian StylePrice, Christopher T.D., and Yousef Abu Kwaik. 2021. "Evolution and Adaptation of Legionella pneumophila to Manipulate the Ubiquitination Machinery of Its Amoebae and Mammalian Hosts" Biomolecules 11, no. 1: 112. https://doi.org/10.3390/biom11010112




