1. Introduction
Human islet amyloid polypeptide (hIAPP) is a hormone that slows down gastric emptying and participates in the regulation of plasmatic glucose associated with functions such as glucagon-release inhibition and leptin sensitization [
1,
2]. hIAPP is a monomeric peptide processed in the Golgi complex and secreted in insulin granules in response to β-cell secretagogues [
3]. However, amyloid fibril formation in hIAPP could trigger an amplified toxicity response that leads to failure of pancreatic β-cells, a hallmark of type 2 diabetes mellitus (T2DM). Several variants of hIAPP modify their stability accelerating amyloid formation; for instance, in Asian populations, the S
20G mutation has been associated with early-stage cases of DM2 [
4]. Likewise, variants have been described in the Maori populations of New Zealand [
5]. By contrast, rat IAPP (rIAPP) containing structural differences with hIAPP in six residues situated in region 18–29 (H
18R, F
23L, A
25P, I
26V, S
28P, and S
29P) show a diminished trend to produce amyloid fibrils [
6]. Although these changes allowed the development of pramlintide, an amylinomimetic peptide with three variants (A
25P, S
28P, and S
29P) used in DM2 therapy; the propensity to aggregation of this sequence is not completely avoided.
Several reports suggest that the C-native segment of IAPP (residues 23–37) is a critical domain in the structural transitions that trigger amyloid formation [
6], hence, it could be a target for the development of strategies to reduce aggregation. Based on multiple sequence alignment among N- and C-domains on 240 sequences of different species, the N-domain (residues 1–20) has been described as a conserved sequence among a wide variety of organisms, while the C-domain (residues 22–37) has been restricted to phylogenetically close groups [
7]. Therefore, we proposed a panel of optimized IAPP sequences, which could reduce aggregation with only one residue substitution [
7].
Localized changes in the secondary structure of proteins and peptides are believed to work as a molecular switch regulating function or, in some cases, as a trigger for misfolding. In this context, we have described these conditions by studying the cholesteryl-ester transfer protein (CETP) and a series of apolipoproteins [
8,
9,
10,
11,
12,
13]. In addition, other studies have reported that phosphatidylserine (PS) vesicles could increase the peptide/aggregation ratio, suggesting an electrostatic factor as a triggering condition for the induction of conformational changes [
14,
15]. In this sense, within insulin secretory granules derived from the endoplasmic reticulum (ER) [
16], the concentration of PS and phosphatidylinositol has been described to be fivefold higher compared to that of the cell membrane [
17]. Therefore, lipids could be a factor that promotes structural changes in hIAPP, inducing misfolding phenomena.
However, highly sophisticated mechanisms that modulate protein structure and function have evolved to maintain cellular homeostasis and counteract misfolding [
11]. Perturbations in these mechanisms can lead to protein dysfunction as well as deleterious cell processes. Specifically, imbalances in secretory protein synthesis pathways lead to a condition known as ER stress, which elicits the adaptive unfolded protein response (UPR) [
11], a phenomenon that could be critical during insulin maturation. Importantly, proinsulin is folded in the ER by chaperones such as protein disulfide isomerases (PDI) and binding immunoglobulin protein (BiP) or GRP-78 [
18]. Therefore, the transducers of the UPR pathway, IRE1, ATF6α, and PERK, promote the translation of target chaperones through XBPIs, ATF6α, and CHOP transcription factors, respectively, when unfolded proteins accumulate in the lumen [
19]. The three branches of UPR are essential in cell homeostasis to reduce ER stress and to ensure adequate synthesis of peptides such as insulin [
20]. Indeed, in several reports, we have described the effect of metabolic overload on the dysregulation of UPR arms [
21,
22].
Given this situation, the initial events of misfolding and amyloid aggregation promote a cascade of pathological processes considered the hallmark in the progression of several chronic degenerative diseases [
8,
9,
10,
11,
12,
13] that might be associated with conditions related to metabolic overload [
21,
22]. Having this in mind, we herein characterized the role of lipid systems on the conformational transitions of the most aggregative C-terminal domain of hIAPP, and through a biomimetic approach, evaluated this condition on variants F
23R and I
26A that potentially could reduce aggregation. In addition, we also characterized the association with cell responses involved in protein homeostasis such as UPR.
2. Materials and Methods
2.1. De Novo IAPP Sequences
Based on network analysis, different residues from 240 species reported in the NCBI database were replaced on the hIAPP sequence. The effect of the substitutions was characterized through physicochemical assays, as well as by the computational identification of regions with a high intrinsic propensity for aggregation [
7]. Aggregation propensity range was obtained considering the aggregation value of hIAPP as a reference, based on the AGGRESCAN algorithm (Na4vSS) [
23].
2.2. Materials
Cell culture reagents were purchased from Thermo Fisher (Carlsbad, CA, USA), while tissue culture plates and other plastic materials were obtained from Corning Inc. (Corning, NY, USA). Salts and buffers were obtained from Sigma-Aldrich (St. Luis, MI, USA), as well as Thioflavin T (ThT), black Sudan B, Congo red, sodium dodecyl sulfate (SDS), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT). l-α-phosphatidylcholine (PC), l-α-phosphatidylserine (PS), l-α-phosphatidyl-ethanolamine (PE), 1-oleoyl-2-hydroxy-sn-glycero-3-phosphate (LPA), 1-palmitoyl-2-oleoyl-sn-glycerol (POPG), and cholesterol were obtained from Avanti Polar Lipids, Inc. (Alabaster, AL, USA). Antibodies anti-XBP1s and anti-BiP/GRP78 were purchased from Abcam (Cambridge, UK) and anti-β-actin was obtained from Santa Cruz Biotechnology (Dallas, TX, USA). Anti-PDI was donated by Dr. Marco A. Ramos Ibarra.
2.3. Peptide Synthesis and Preparation
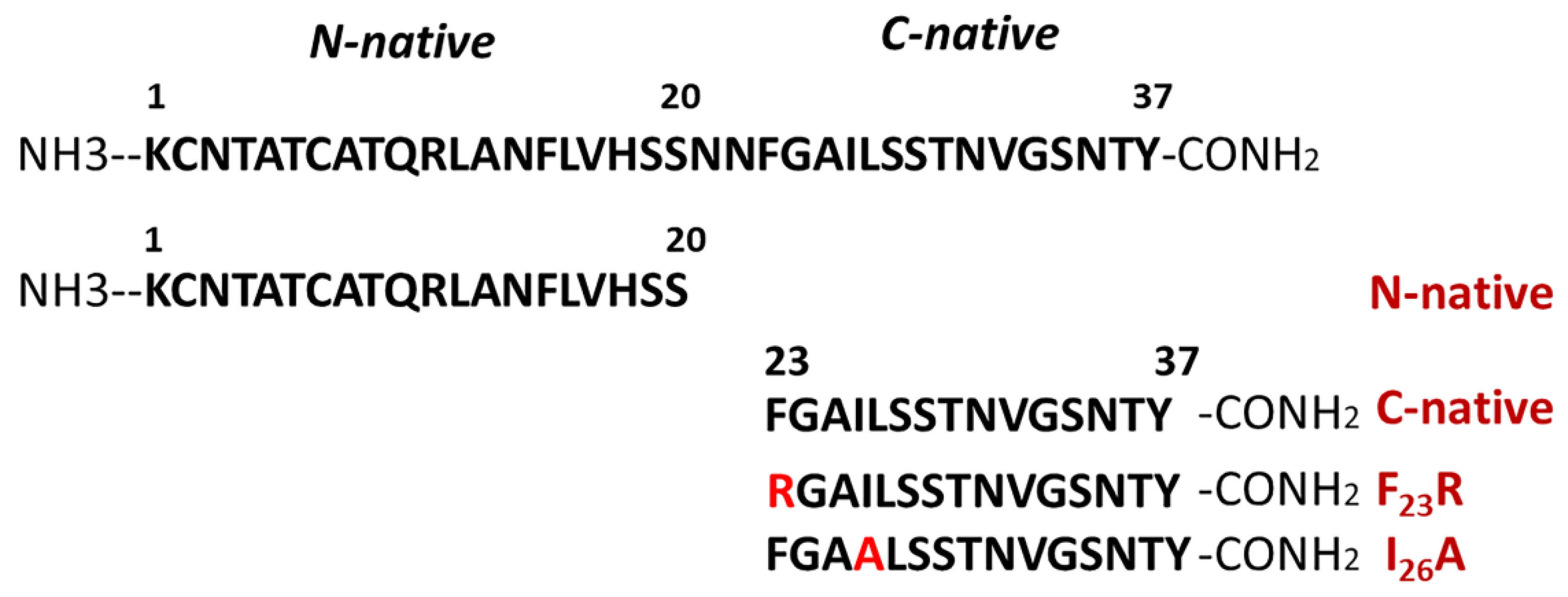
Several peptides were synthesized considering the physicochemical properties of hIAPP such as: N-native segment (
1KCNTATCATQRLANFLVHSS
20); C-native segment (
23FGAILSSTNVGSNTY
37); F
23R variant (
23RGAILSSTNVGSNTY
37); and I26A variant (
23FGAALSSTNVGSNTY
37) (
Figure 1). Likewise, the aggregative core of amyloid beta (Aβ) peptide was used as a control
25GSNKGAIIGLM
35. All solutions were filtered through 0.22 μm membrane filters (Millipore, Burlington, MA, USA) before the experiments. Peptide purity greater than 98% was confirmed by mass spectrometry and HPLC (GenScript, Piscataway, NY, USA). The best condition for peptide solubilization was the use of ultrapure H
2O (600 µg/mL), subsequently diluted in phosphate buffer pH 7.4 (60 µg/mL).
2.4. Preparation of Small Unilamellar Vesicles (SUVs)
SUVs were prepared from PC, PS, PE, cholesterol, and POPG (600 µg/mL) upon their mixing and conditioning at varying concentrations. PC, PS, PE, cholesterol, and POPG were dissolved in chloroform and dried for 90 min under a gentle stream of N
2 with an additional incubation of 5 h at 30 °C in an Eppendorf Vacufuge concentrator (Eppendorf, Hamburg, Germany), according to protocols established by our working group [
9]. After drying, samples were hydrated in phosphate buffer to pH 7.4 and processed through 4 cycles of freezing in liquid N
2 and thawing at 37 °C, and finally subjected to a sonication process (for 10 min under 15 s on/30 s off cycles at 9.5–10 W) in a Cole-Parmer Ultrasonic Homogenizer (Vernon Hills, IL, USA). Samples were stabilized for 1 h at 25 °C and centrifuged at 13,000 rpm for 10 min.
LPA samples in chloroform were placed under a gentle flow of N2 for 90 min and additional 12 h in a vacuum equipment. The samples were hydrated in phosphate buffer and afterwards processed through 4 cycles of freezing in liquid N2 and thawing at 37 °C. Solutions were left to equilibrate for 2 h and centrifuged at 13,000 rpm for 10 min.
2.5. Preparation of Large Unilamellar Vesicles (LUVs)
LUVs were prepared from PC and PS by the reverse-phase evaporation methodology [
24], with some adaptations. Specifically, PC LUVs were prepared by dissolving the lipid (600 µg/mL) in a 1:3 mixture of diethylether:phosphate buffer (pH 7.4). Then, the solution was sonicated for 5 min in an ultrasonic homogenizer. Finally, the solution was added to a rotary evaporator working at an initial pressure of 400 mmHg for 5 min, followed by a final pressure of 650 mmHg until complete removal of diethylether. Likewise, PS LUVs were prepared by dissolving the lipid (600 µg/mL) in chloroform and drying the resulting solution for 90 min under a gentle stream of N
2 with an additional incubation of 2 h at 30 °C in an Eppendorf Vacufuge concentrator, according to protocols established by our working group [
9]. After drying, samples were hydrated in phosphate buffer, pH 7.4, and processed through 4 cycles of freezing in liquid N
2 and thawing at 37 °C. Finally, samples were subjected to a sonication process (for 5 min under 15 s on/30 s off cycles at 9.5–10 W) in an ultrasonic homogenizer.
2.6. Dynamic Light Scattering Experimentation and Optical Density Characterization
In the characterization of vesicle size, PC (300 µM) and PS (300 µM) vesicles were evaluated. The hydrodynamic diameter (Dh) and Z-Potential of the vesicles were assessed by dynamic light scattering (DLS), employing a Zetasizer Nanoseries spectrophotometer (Malvern Instruments, Malvern, UK). All measurements were carried out at 25 °C using polystyrene disposable cells and folded capillary cells for the size and ζ potential measurements, respectively. Before their characterization, the samples were vortexed for 10 s and left to rest for 30 min at 25 °C. The results are the average of five measurements. In another batch of vesicles (PS), we completed the characterization by using a Microtrac equipment. In a complementary way, we performed the optical density characterization of vesicles employing a BioRad Smart spectrophotometer with diode array (Hercules, CA, USA).
2.7. Peptide Bond Conformational Changes
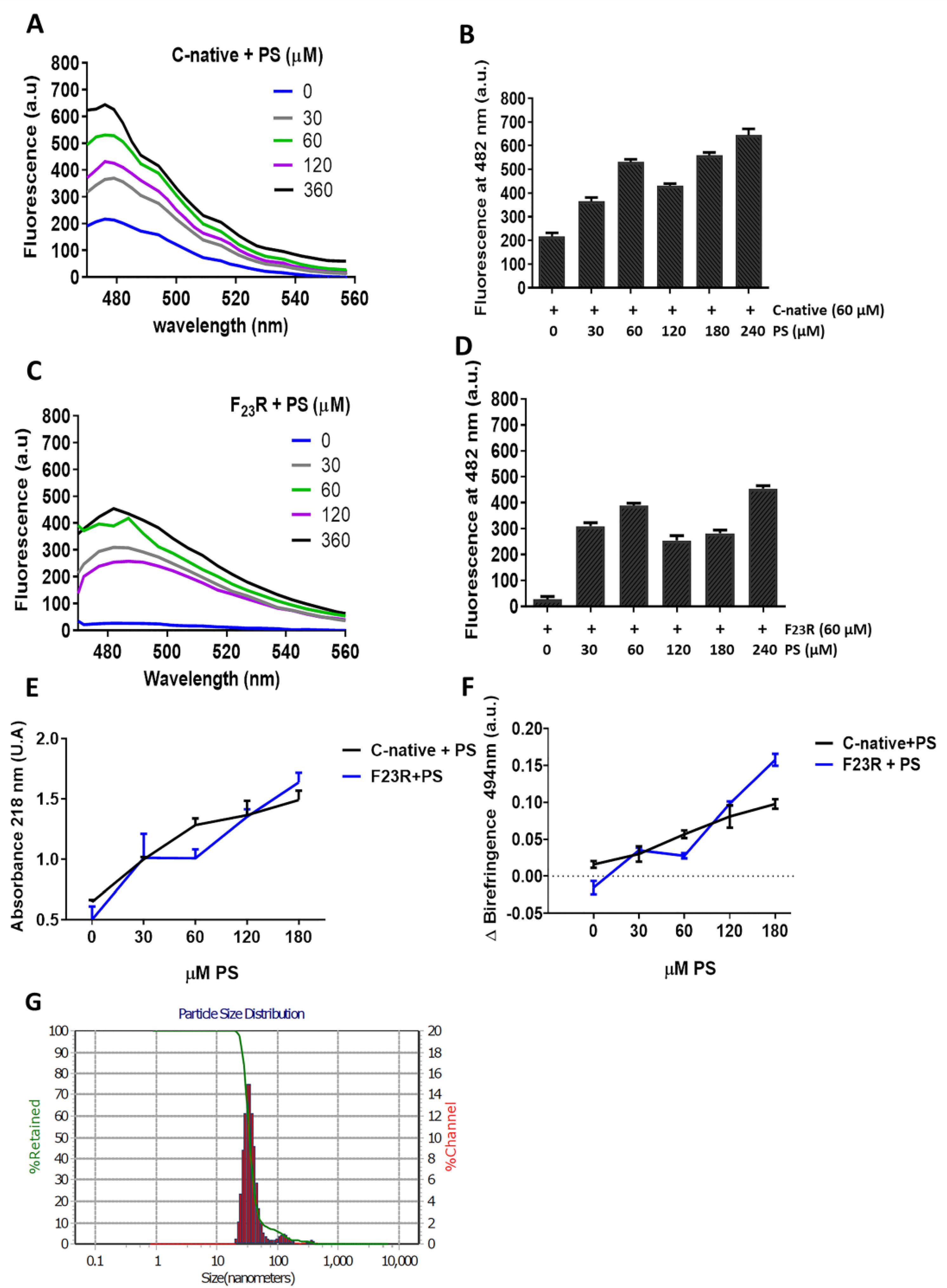
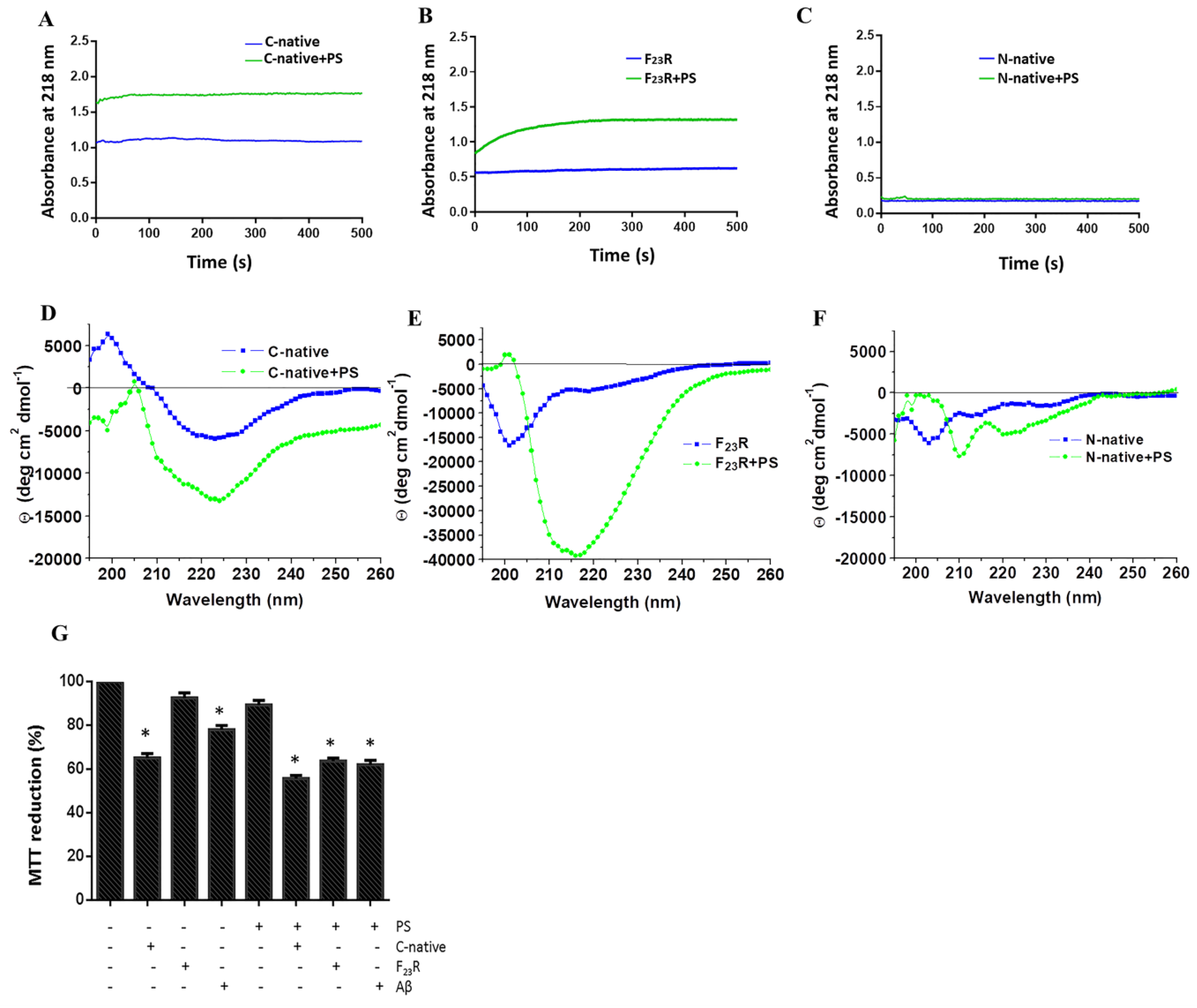
Experiments were performed through the characterization of optical density at 218 nm, which is associated with conformational changes along the formation of β-sheet structures [
8,
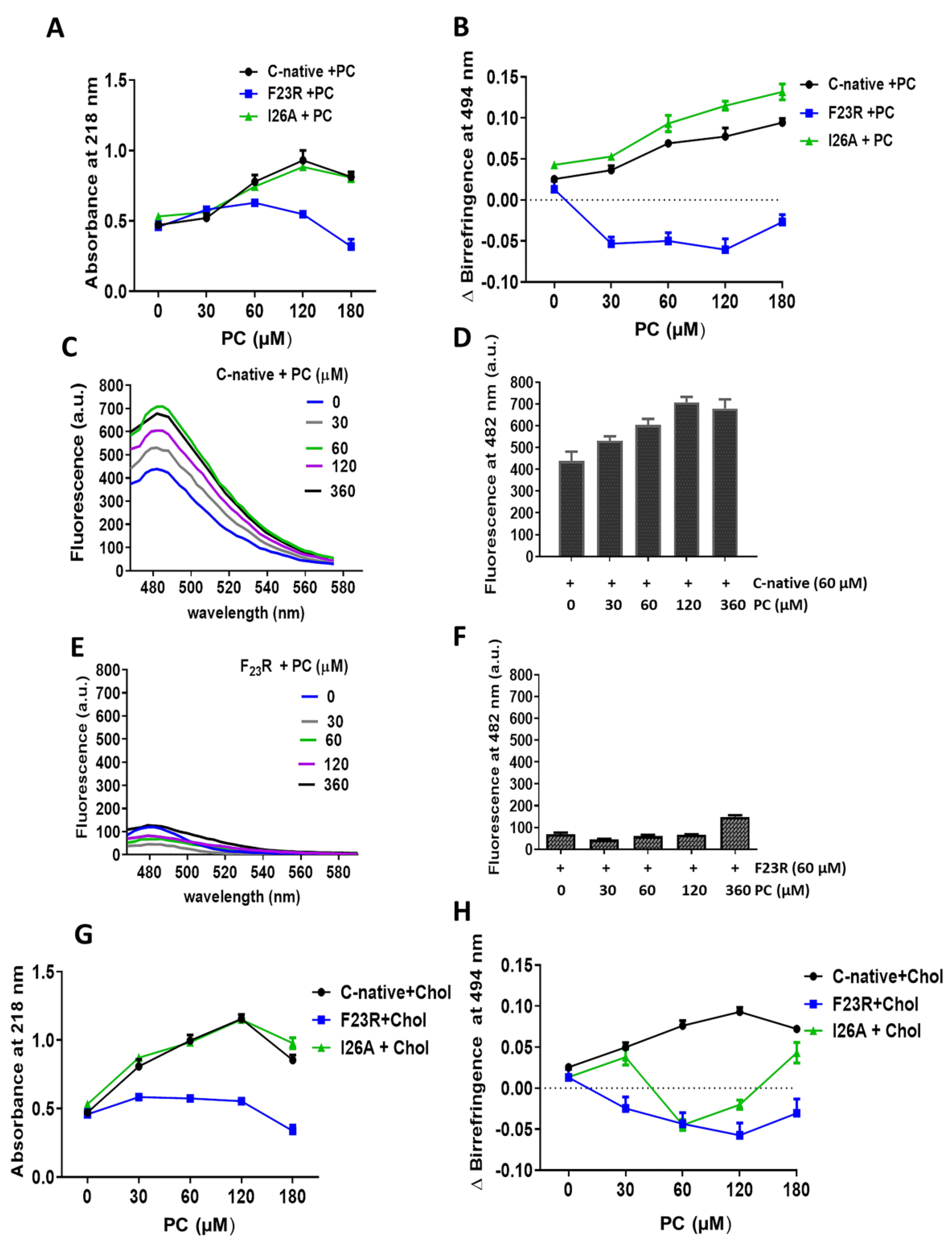
10]. The effect of lipid vesicles composed of several lipids on conformational changes of hIAPP-derived peptides was evaluated. Measurements were obtained using the above-described BioRad Smart spectrophotometer, employing a peptide concentration of 60 µg/mL and then evaluating the effect of the lipid vesicles.
2.8. Congo Red Birefringence Spectroscopy
Assays were performed based on a previous protocol [
8], employing 10.6 μM Congo red and 60 μg/mL peptides solutions. The optical density was measured at 494 nm, employing the above-described BioRad Smart spectrophotometer, under varying solution conditions.
2.9. ThT-Fluorescence Assay
β-sheet structures of peptides were characterized through the ThT-fluorescence assay. Samples were incubated for 12 h at 37 °C and monitored with the ThT (20 μM) treatment. Fluorescence emission spectra were registered at 25 °C from 460 to 610 nm with an excitation wavelength of 450 nm in a Cary Eclipse Fluorescence spectrophotometer (Agilent Technologies, Inc., Santa Clara, CA, USA).
2.10. Circular Dichroism (CD)
CD experiments were performed at a peptide concentration of 120 μg/mL in a 1-mm path length quartz cuvette, using the CD neural network (CDNN) based software. Spectra were recorded with a 1-mm bandwidth, using 1 nm increments and 2.5 s accumulation time. CD spectra were recorded with an AVIV 62DS spectropolarimeter (AVIV Instruments, Lakewood, NJ, USA) at 25 °C employing far UV wavelengths (190–260 nm). CD results were reported as mean molar ellipticity (deg cm2 dmol−1).
2.11. Lipid–Peptide Interactions
Lipid/peptide samples were analyzed with a nondenaturing electrophoresis technique adapted by our group for lipid–peptide characterization. We established a new methodology through the use of 0.8–15% native gradient gel electrophoresis [
8]. Later, gels were stained following the Sudan black and silver nitrate protocols [
8].
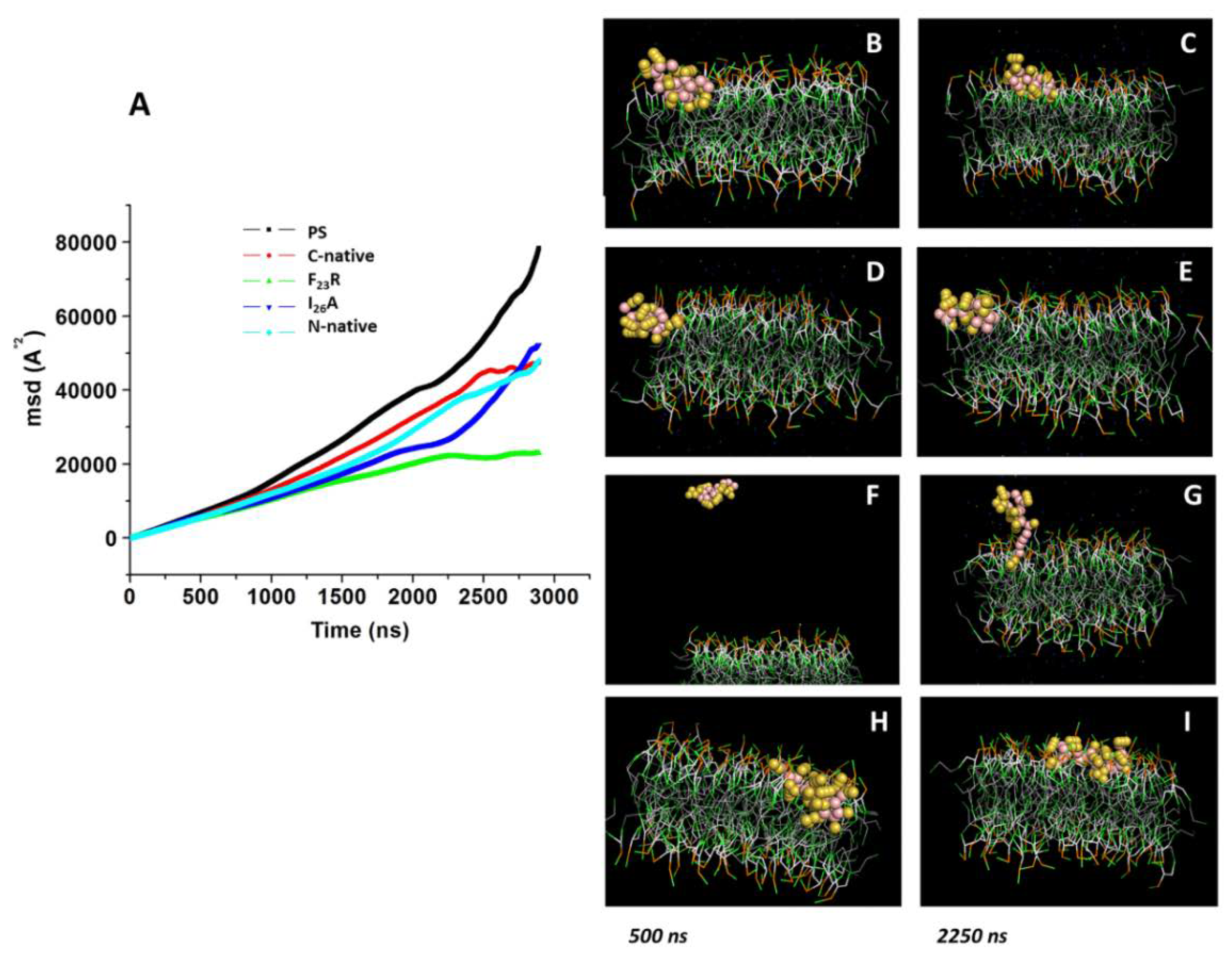
2.12. Molecular Dynamics
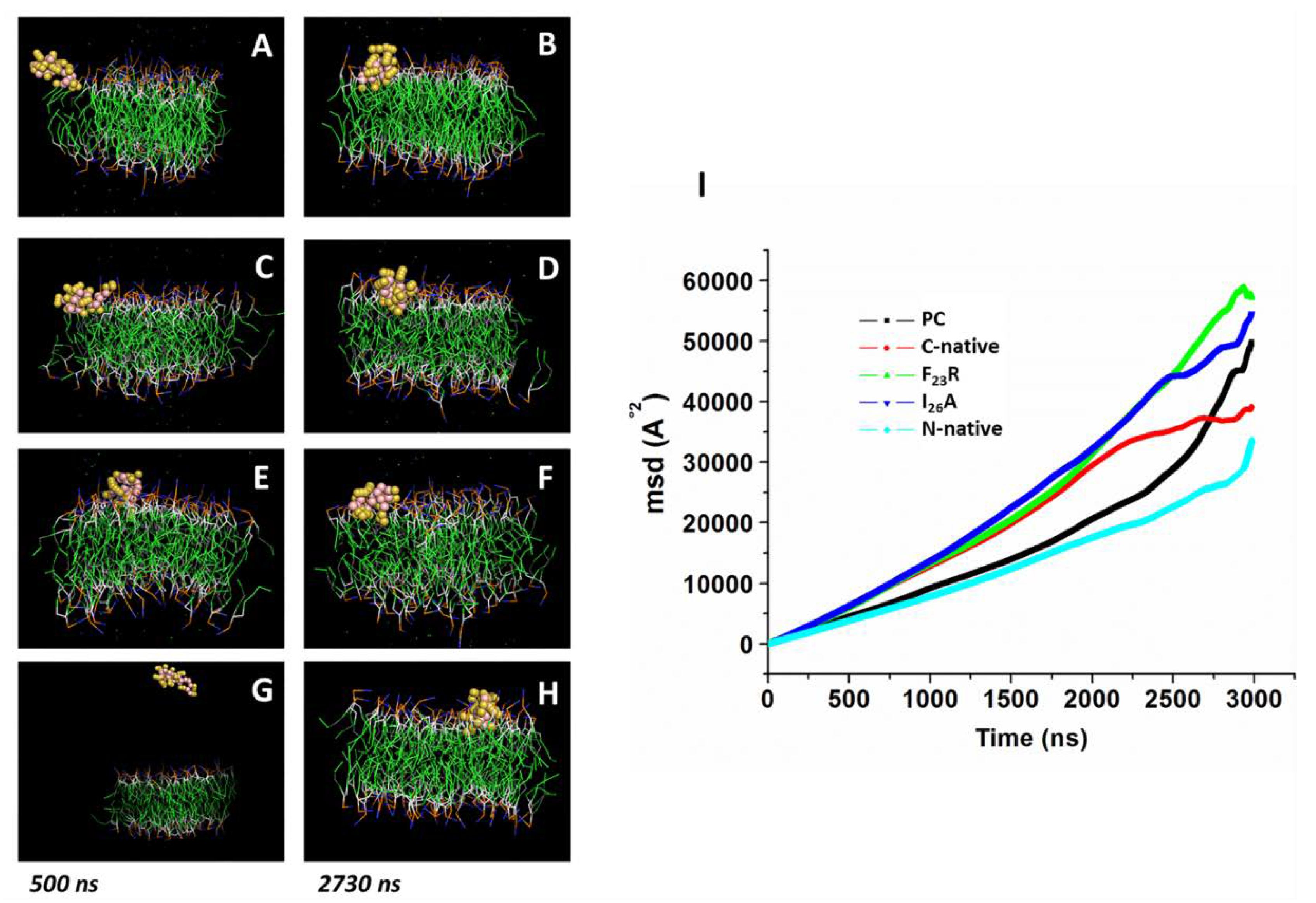
Peptide-membrane systems were generated with the CHARMM-GUI input generator. For all systems, a MARTINI force field for polarizable amino acids and water was used. In our assays, we used the peptides in a simple lipid bilayer system, each lipid bilayer consisting of a homogenous array of 8 × 8 lipid molecules of PS (DIPS 18:2–18:2) or PC (3:1, DPOC 16:1–18:1 and POPC 16:0–18:0). Peptide models of N-native, C-native, and variants F
23R and I
26A were generated using the ITASSER online server; for all peptides, we used the predicted model with highest TM value. The systems were minimized using steepest descent and conjugate gradient methods. Then, five equilibration steps were performed for each system. Simulations were conducted in a 1:1 peptide-membrane system during 3000 ns at 303.15 K and 1 atm pressure. For each system, we evaluated the lipid bilayer lateral displacement from lipids as an indicator of bilayer fluidity. Our analysis was based on the GROMACS built-in function for MSD analysis. To this end, we used the built-in functions of GROMACS, which are calculated using the following equation:
where
ri (
t) indicates molecule position a t time,
ri (
0) indicates the position at molecules at time zero, and is calculated for molecules included in
A set of molecules. Additionally,
6DAt represents diffusion coefficient over time (
t) for
A set of molecules, using Einstein correlation adjusted for long simulations.
In this sense, the analysis along the membrane was performed in one single plane. Before analysis of MSD, periodic boundary conditions were converted used built-in conversion functions to ensure the continuous trajectory of molecules.
In a complementary way, we performed molecular dynamics reruns, started from previously equilibrated systems that were used in original reported simulations. The systems were evaluated under the same conditions of temperature, pressure, and number of molecules. The simulation time was reduced to 30,000 ps; 100 consecutive simulations were performed for each system where the previous simulation was used as a starting point for the subsequent one. All trajectories obtained from short simulations were joined using built-in functions of GROMACS to reach the same simulation time reported previously; the resulting trajectory file was converted into a continuous trajectory file. MSD analysis for this system is reported in
Figures S3 and S6. This strategy was based on previous reports [
25,
26].
2.13. Cell Culture
β-cell line RIN-m5F (American Type Culture Collection) was grown using RPMI-1640 culture medium supplemented with 10% fetal bovine serum, 10 U/mL penicillin, 10 µg/mL streptomycin, and 25 µg/mL amphotericin B. Cultures were maintained at 37 °C in a humidified atmosphere of 95% air and 5% CO2.
2.14. Cell Viability Assay
Peptide cytotoxicity was assessed through MTT assays RIN-m5F cells, under different peptide and lipid–peptide treatments. Cells were seeded into 96-well plates at a density of 20,000 cells/well and allowed to grow to 90% of confluence. Next, the culture medium was replaced with Opti-MEM medium. After 1 h under this condition, cells were incubated under the different treatments and subsequently processed according to previous protocols [
8].
2.15. Western Blotting Analysis
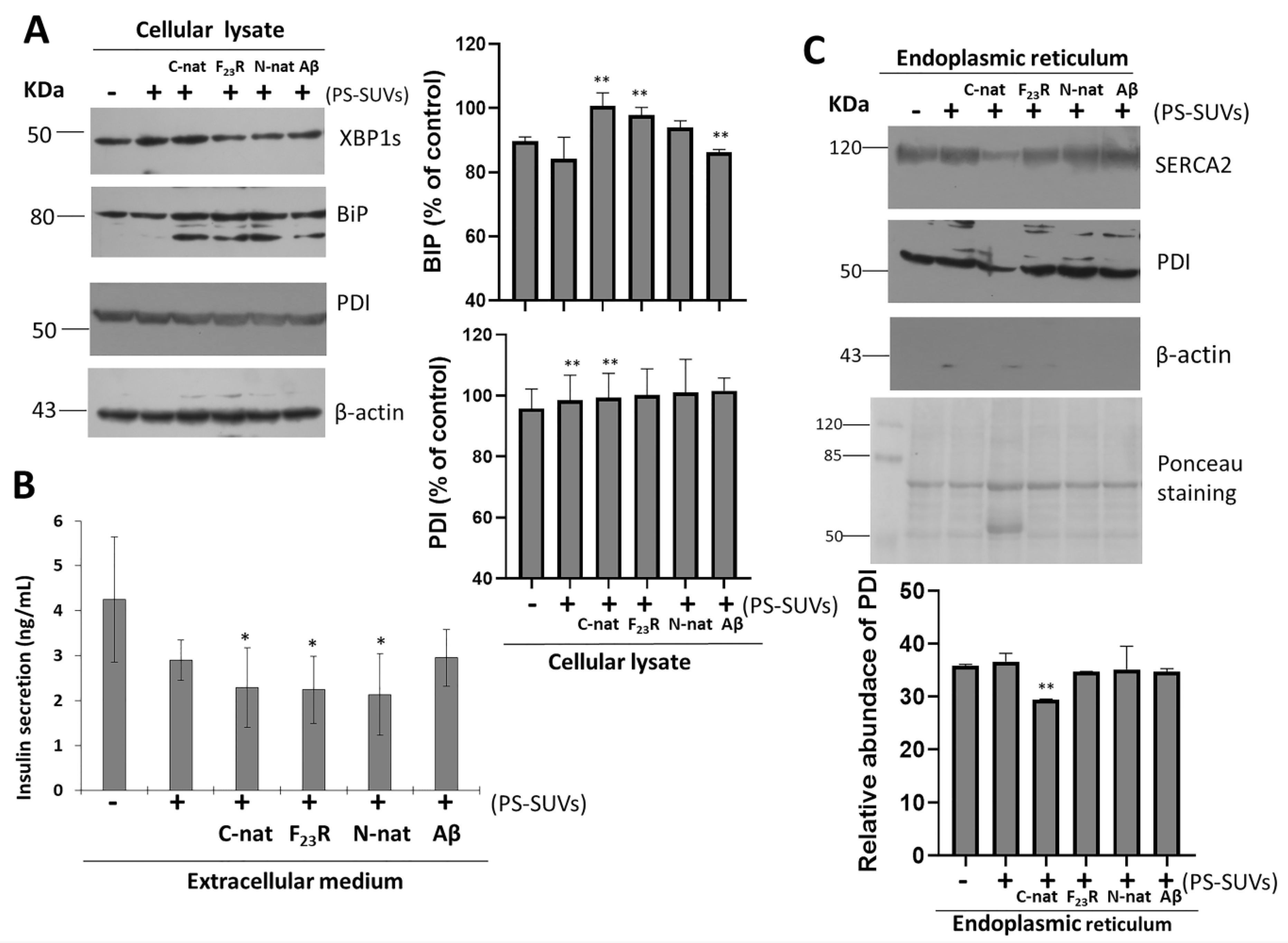
Under different peptide and PS-SUVs treatments, the expression of proteins associated with the UPR pathway and insulin folding were evaluated. After experimentation on RIN-m5F cells, proteins were extracted from cell cultures using ice-cold protein lysis buffer (150 mM NaCl, 10 mM Tris, pH 7.4, 1% Triton X-100, 0.5% NP40, 1 mM EDTA, 1 mM EGTA, 0.2 mM sodium orthovanadate, 10 mM benzamidine, 10 µg/mL leupeptin, 10 µg/mL aprotinin, and 250 µM PMSF). An average of 25 µg of protein lysates were separated on 8% SDS-PAGE electrophoresis and transferred to polyvinylidene difluoride (PVDF) membranes. The membranes were blocked with 5% nonfat milk in Tris-buffered saline 0.1% Tween-20 (TBS-T) for 1 h at 37 °C and incubated at 4 °C overnight with primary antibody (anti-XBP1s, anti-BiP/GRP78, anti-PDI, anti-SERCA2, and anti-β-actin). Following washing with TBS-T, the membranes were further incubated for 1.5 h at 37 °C with the corresponding horseradish peroxidase-conjugated secondary antibodies. Proteins were detected with the enhanced chemiluminescence reagent (Immobilon Western from Millipore, Burlington, MA, USA).
2.16. Endoplasmic Reticulum Isolation
ER fractions were obtained from RIN-m5F cells under several PS/peptides treatments. The methodology was based on the report of Prajapati et al. [
27]. Cells were proliferated in 100 mm cell culture plates at a density of 2.3 × 10
5 cells/mL. Cells were maintained in proliferation for 72 h to reach 95% of confluence, and later, different treatments were performed on a volume of 5 mL. Later, culture cells were washed with PBS 1x, recovered, and treated with 1 mL of homogenizer buffer (30 mM Tris–HCl pH 7.4, 225 mM mannitol, 75 mM sucrose, 0.5 mM EGTA, protease inhibitor, and 0.5% BSA). Homogenates were briefly sonicated (two cycles of 15 s on/30 s off at 9.5–10 W). Then, the homogenate was centrifuged at 630×
g for 5 min at 4 °C. Supernatant was collected and conserved, and the pellet was newly processed by sonication and centrifuged under the same conditions. The combined supernatant was centrifuged again at 630×
g for 5 min at 4 °C (nuclei-free lysate), and 150 µL was conserved. Then, the nuclei-free lysate fraction was centrifuged at 6300×
g for 10 min. The supernatant was transferred into a new tube, and then, it was centrifuged at 20,000×
g for 30 min at 4 °C. The supernatant was recovered and centrifuged at 100,000×
g for 60 min at 4 °C, using a S140-AT 2555 rotor. The supernatant corresponds to the cytoplasm fraction, whereas the pellet corresponds to the ER fraction. Protein markers (anti-BiP, anti-PDI, anti-SERCA2, and anti-β-actin) were used to evaluate the quality of the isolations (
Figure S9).
2.17. Fabrication of Vesicles Composed of PS (30 µM) and the Fluorescent Probe BODIPY-Leu (6 µM)
BODIPY-Leu probe was synthesized according to protocols developed by our group [
22]. Then, the required quantities of
l-α-phosphatidylserine and BODIPY-Leu were dissolved in chloroform and mixed vigorously to obtain a clear solution. Then, the solution was dried for 90 min under a gentle stream of N
2 with an additional incubation of 5 h at 30 °C in an Eppendorf Vacufuge concentrator, according to protocols established by our working group [
9]. After drying, samples were hydrated in phosphate buffer pH 7.4 and processed through 4 cycles of freezing in liquid N
2 and thawing at 37 °C, and finally subjected to a sonication process (for 10 min under 15 s on/30 s off cycles at 9.5–10 W) in an ultrasonic homogenizer. Samples were stabilized for 1 h at 25 °C and centrifuged at 13,000 rpm for 10 min.
2.18. Confocal Microscopy
A LEICA TCS-SP8 confocal scanning biological microscope (LEICA Microsystems Heidelberg GmbH, Nussloch, Germany) was employed in the characterization of the subcellular localization of PS/BODIPY-Leu vesicles (PS 30 µM/BODIPY-Leu 6 µM). RIN-m5F cells were proliferated to 90% of confluence and treated with PS/BODIPY-Leu vesicles and hIAPP-derived peptides for 20 h. Later, culture cells were washed with HBSS buffer, and then, the ER-tracker probe (1 µM) was added and incubated for 25 min at 37 °C. Cells were washed once with HBSS buffer and fixed with 4% formaldehyde for 2 min at 37 °C and mounted for observation. Macroscopically different zones were recorded, preferentially at the center of the specimens, to depict representative images. Images were recorded at excitation/emission wavelengths of 488/495-545 and 552/562-700 nm for detection of PS 30 µM/BODIPY-Leu (green) and ER-tracker (red), respectively.
2.19. Insulin ELISA Assays
Cells were proliferated in 100 mm cell culture plates at a density of 2.3 × 105 cells/mL. Cells were maintained in proliferation for 72 h, and later, different treatments were performed on a volume of 5 mL. Cell culture medium was recovered and centrifuged for 5 min at 5000 rpm. The supernatant medium was recovered and diluted (1/3) in PBS. Insulin concentrations were quantified with the Rat Ultrasensitive Insulin ELISA kit (80-INSRTU-E01, E10; ALPCO Diagnostics, Salem, NH, USA) through several adaptations according to manufacturer’s recommendations. Absorbance readings were performed at 450 nm, and results were reported as ng/mL.
2.20. Statistical Analysis
Data were expressed as mean ± SD. The statistical analyses were conducted with one-way ANOVA. In MTT assays data were expressed as mean ± SD.
4. Discussion
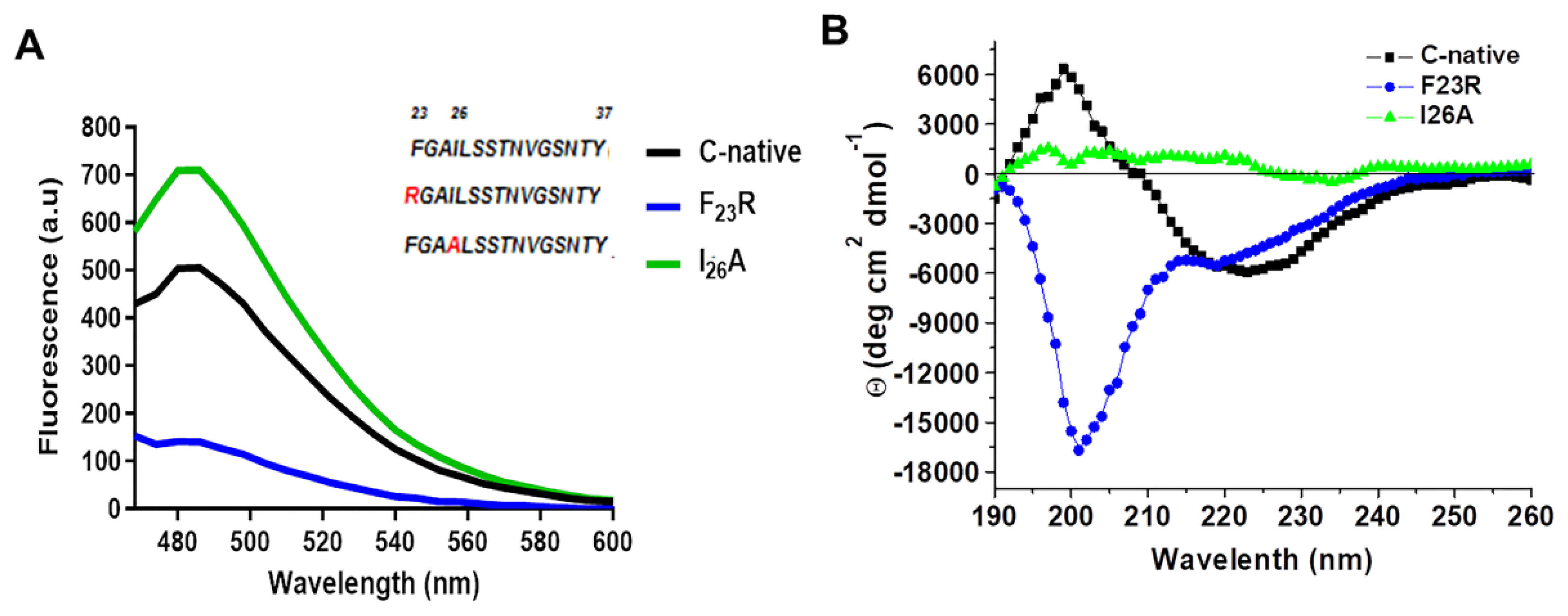
During the course of the present investigation, employing a biomimetic approach, we have been able to develop and evaluate new variants of IAPP with the property to show fewer propensities to form β-sheet structures. New sequences such as F
23R have shown to increase the stability of the C-native segment of IAPP, with an important involvement of the anionic nature lipids. This behavior can be associated at pH 7.4 with charged lipids that contributed to structural changes towards the β-sheet formation, whereas the I
26A variant shows a neutral charge and an isoelectric point similar to the C-native segment, promoting a strong interaction with anionic phospholipids. Molecular dynamics showing an absorption phenomenon of F
23R at the surface of lipid bilayer indicate the possibility for the formation of peptide-PS aggregates that, by means of lipotoxicity, could have contributed to the observed cytotoxicity (
Figure 6G). Further experiments are nowadays in progress in our laboratories to confirm this assumption (data to be published).
The content of anionic phospholipids in β-cell membranes is reported to range from 2.5% to 13.2%, a proportion mainly situated in the inner leaflet of the membrane [
38], proportion much similar as found in insulin secretory granules where a higher content of PS and phosphatidylinositol has been described [
17,
42], which are derived from ER membranes during insulin maturation. This contributes to the possibility that hIAPP aggregation could be increased at the intracellular space, especially during the process of maturation of insulin secretory granules. The phenomenon of membrane asymmetry that could enhance hIAPP amyloid formation and membrane damage in vivo [
43] could modify the PS-biodisponibility and, therefore, triggers the β-sheets formation.
Therefore, considering the critical role of PS in the promotion of β-sheet conformational transitions on hIAPP-derived peptides, the impact of phospholipid negative-electrostatic charge on UPR regulation was characterized. In this sense, PS-SUVs with the hIAPP-derived peptides under a 1 to 2 peptide/PS ratio were incubated on β-cell cultures for 20 h. In the first case, critical UPR-targets were evaluated under the peptide/PS treatments in complete cellular lysates (
Figure 8A) results suggest the slight activation of XBPIs, a transcription factor of the activation of IRE1-arm of UPR. Likewise, an increase in the expression of chaperone BiP under peptides/PS treatments was registered, and this phenomenon was more evident under the C-native treatment. Possibly, these modifications could be partly related to a cellular compensatory response aimed to maintain protein homeostasis in ER (
Figure 8A), as well as critical functions in the physiology of β-cells. Importantly, when insulin concentrations were evaluated in extracellular media, we found a diminution of the insulin levels upon treatment with the peptide/PS mixtures (
Figure 8B).
PDI is a chaperone that regulates folding of proinsulin, participates in disulfide bond formation, and maintains ER redox homeostasis [
44]. In our conditions, when total β-cell lysates were evaluated under peptide/PS treatments, we did not find changes in PDI expression (
Figure 8A). Constituting a chaperone-protein with critical functions, high expression levels of PDI have been found in the ER lumen, to a lower extend in the cytosol, and also in different cellular membranes. [
45]. In addition, we have also detected PDI in the extracellular medium of β-cells treated with peptide/PS mixtures (data not shown). However, more experimental evidence is required to establish a mechanistic proposal.
To dissect the role of PDI, we performed the isolation of ER of β-cells cultures, evaluating sarco/endoplasmic reticulum Ca
2+-ATPase-2 (SERCA2) and β-actin as controls of ER isolations. Results indicated the isolation of pure ER-fractions (
Figure S9). Then, under the same treatments of hIAPP-peptides/PS-SUVs, we characterized the expression of PDI. We found that the PDI levels diminished upon treatment with C-native/PS, as well as the levels of SERCA2 resident of ER (
Figure 8C). Results suggest that the affectation of SERCA2 and PDI under C-native/PS treatment might be related to both their ER localization and the activation of UPR, affecting insulin secretion.
ER-lumen and the function of chaperones BiP and PDI are critical during proinsulin folding. Considering our results, and in an attempt to characterize the effect of the PS vesicles and possibly trace their cellular localization, confocal microscopy experiments were carried out. To this end, we prepared PS and peptide/PS vesicles (30 µM) tagged with the green fluorescent probe BODIPY-Leu [
46] (6 µM) (referred to as BODIPY-Leu/PS and BODIPY-Leu/peptides/PS vesicles, respectively), with which RIN-m5F cells were treated. In the first instance, our results demonstrated that BODIPY-Leu/PS vesicles are internalized in RIN-m5F cells (
Figure 9A–C, stained in green). Then, we used ER-tracker (red staining) for characterize the colocalizing in ER sites (
Figure 9D–I). Results suggest that the system is located in the ER. Importantly, when we evaluated the localization of the BODIPY-Leu/PS vesicles incubated with hIAPP-derived peptides (C-native and F
23R variant) under a molar relationship (1/2; peptide/vesicle), the BODIPY-Leu/PS signal was localized under C-native treatment in ER sites (
Figure 9J–L), however, the signal diminished slightly under F
23R treatment (
Figure 9M–O). In an important way, C-native/PS treatment promoted the higher levels of cytotoxicity in the studied cells (
Figure 6G), affecting insulin secretion (
Figure 8B). Moreover, these phenomena coincide with UPR activation and affectation of localization of PDI and SERCA2 in the ER (
Figure 8C). Therefore, it appears that the localization of peptide/PS is a critical condition to induce the alterations in homeostasis of the ER. This phenomenon was evidenced after treatment with the C-native/PS system, whereupon the higher fluorescence signal of the BODIPY-Leu/PS vesicles along the ER very likely corresponds with the alterations in localization of PDI, SERCA2, and the insulin secretion. This phenomenon was not evident upon treatment with the F
23R variant.
Therefore, our results suggest that the lipid-anionic electrostatic charge is a critical condition that could modulate the UPR pathway and the conformational transition of IAPP-derived peptides. Thus, a negative electrostatic-charge environment could be critical in insulin secretion as well. Interestingly, the content of anionic lipids of β-cells and insulin secretory granules has been related to an altered glucose-stimulated insulin exocytosis [
17,
43]. Therefore, a condition of metabolic overload also has been associated with the biodisponibility of fatty acids [
22], very likely contributing to the deleterious phenomenon correlated with the concentration of anionic phospholipids, promoting misfolded transitions on hIAPP. Having in mind this phenomenon, our group has generated new materials of polymeric films of polyvinyl dimethylazlactone (PVDMA) and polyethylene imine (PEI) to evaluate the effect of fatty acids on β-cell membranes [
47] and diverse critical physiological functions.
In a complementary way, current results of our laboratory suggest that the induction of oligomers at the C-native domain of IAPP accelerates β-sheet formation when treated with oleic acid/PC vesicles. By contrast, when palmitic acid/PC vesicles are used, this result is not found (data not shown). Then, considering that unsaturation and shorter fatty acids of phospholipids facilitate the curvature and fluidity of membranes favoring their fusion [
17], although increasing the risk of aggregation, there is a subtle regulation in the conservation of the structure of the hIAPP. Moreover, a report reveals synergic implications of free fatty acids and hIAPP in ER stress and apoptosis of islet β-cells [
48]. In this context, we have documented the role of metabolic overload by saturated fatty acids on proteostasis and its impact on insulin secretion, specifically the dysregulation of targets that control intracellular calcium homeostasis [
22], as also documented in this report for SERCA2.
Recently, following peptidomimetic design strategies, research has been developed to find a way to inhibit the formation of β-sheet structures in segments of an important series of polypeptides and proteins as a therapeutic way to fight amyloid disease. There is still a field of action in amyloidogenesis design. Although prolines residues in rIAPP promote disordered structures, results of coincubation of rIAPP and hIAPP suggest that rat amylin does not block β-sheet and also forms its own β-sheet, most probably on the outside of the human fibrils [
49], revealing the complex behavior in the development of an amyloid fibril inhibitor. We and other authors have documented that it is critical to consider the impact of lipid environment. In the light of our results, the F
23R variant of IAPP showed a low propensity to form β-sheet structures even under the effect of zwitterionic lipids. However, anionic charge of lipid vesicles and degree of solvation were factors for the modulation of β-sheet formation of the F
23R and I
26A variants, as well as in the C-native segment of IAPP, all associated to the cytotoxicity phenomena of β-cells. In conclusion, our results show the potential implications of modulating the structure and stability of IAPP for the design of analog therapeutics based on peptides and proteins.