Probing Structure and Function of Alkali Sensor IRR with Monoclonal Antibodies
Abstract
1. Introduction
2. Materials and Methods
2.1. Monoclonal Antibodies (mAbs) Production and Purification
2.2. Immunoprecipitation
2.3. SDS-PAGE and Western Blotting
2.4. Immunocytochemistry
2.5. Effect of Monoclonal Antibodies on IRR Activation
3. Results
3.1. Purification of IRR Ectodomain and Preparation of Monoclonal Antibodies
3.2. Western Blotting
3.3. Immunoprecipitation
3.4. Immunocytochemistry
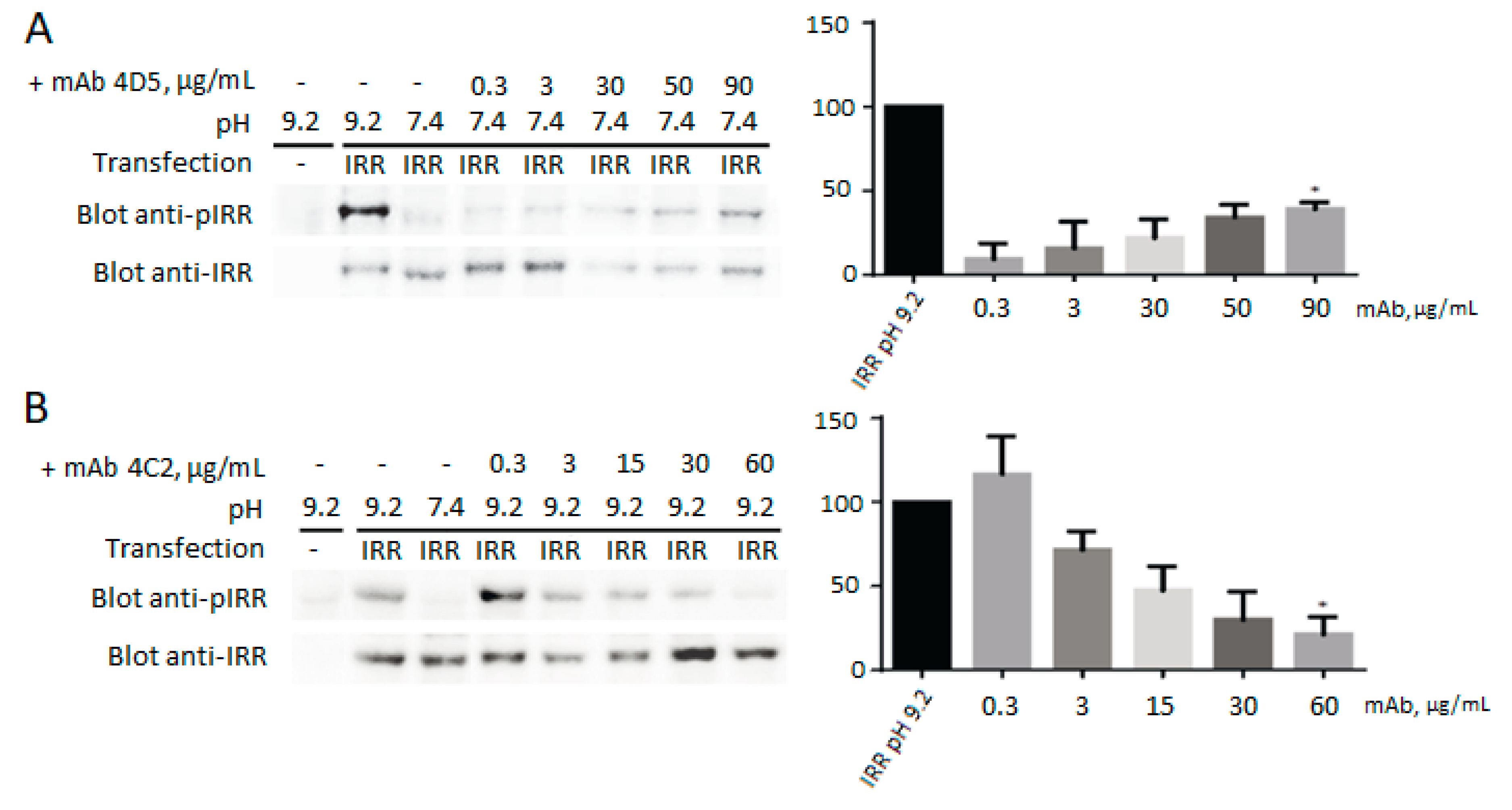
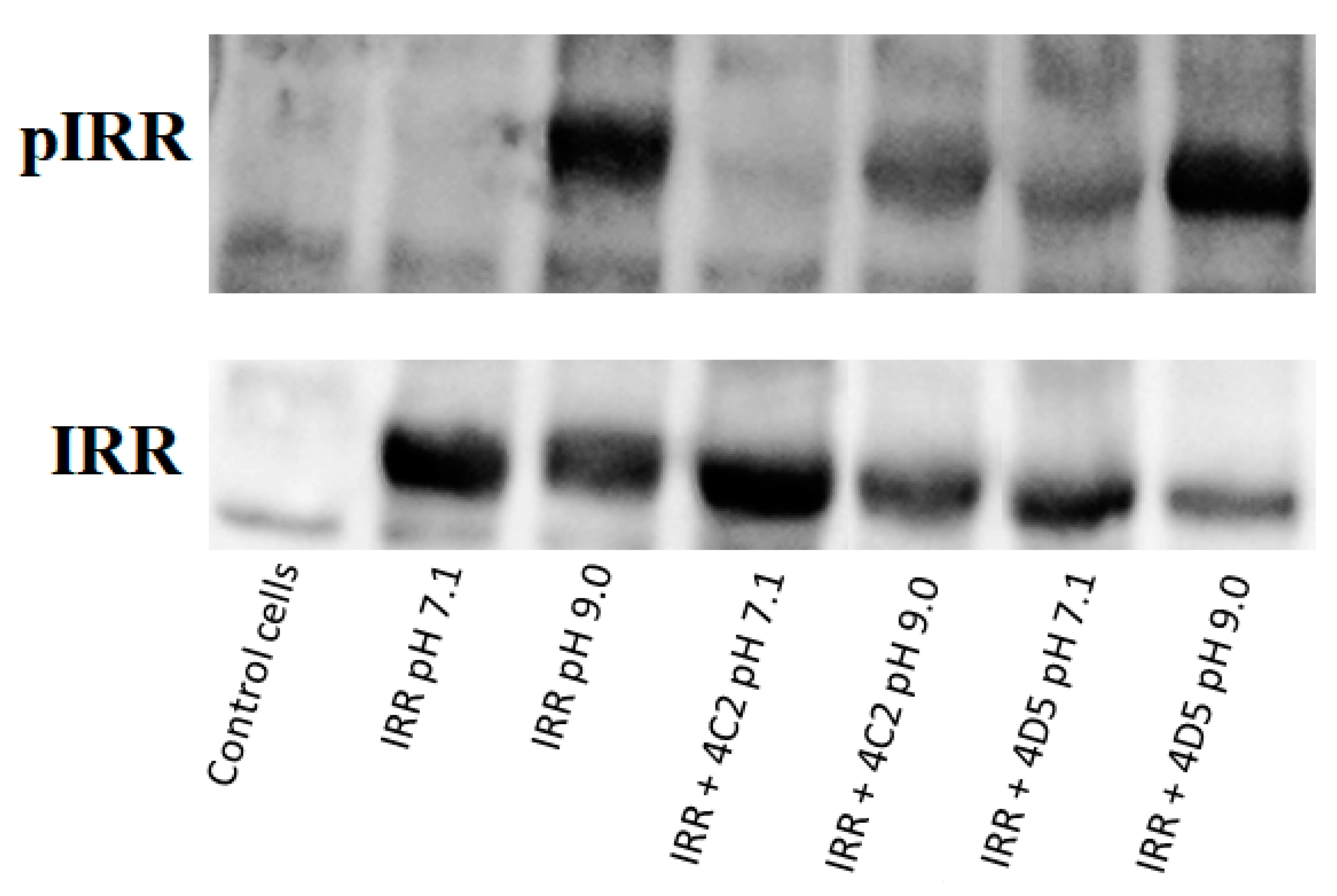
3.5. Influence of the Monoclonal Antibodies on the IRR Activation by Mildly Alkaline pH
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Blume-Jensen, P.; Hunter, T. Oncogenic kinase signalling. Nature 2001, 411, 355–365. [Google Scholar] [CrossRef] [PubMed]
- Ségaliny, A.I.; Tellez-Gabriel, M.; Heymann, M.F.; Heymann, D. Receptor tyrosine kinases: Characterisation, mechanism of action and therapeutic interests for bone cancers. J. Bone Oncol. 2015, 4, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, S.R. Juxtamembrane autoinhibition in receptor tyrosine kinases. Nat. Rev. Mol. Cell Biol. 2004, 5, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Pawson, T.; Gish, G.D.; Nash, P. SH2 domains, interaction modules and cellular wiring. Trends Cell Biol. 2001, 11, 504–511. [Google Scholar] [CrossRef]
- Czech, M.P. The Nature and Regulation of the Insulin Receptor: Structure and Function. Annu. Rev. Physiol. 1985, 47, 357–381. [Google Scholar] [CrossRef]
- Tatulian, S.A. Structural Dynamics of Insulin Receptor and Transmembrane Signaling. Biochemistry 2015, 54, 5523–5532. [Google Scholar] [CrossRef]
- Shier, P.; Watt, V.M. Primary structure of a putative receptor for a ligand of the insulin family. J. Biol. Chem. 1989, 264, 14605–14608. [Google Scholar]
- Deyev, I.E.; Sohet, F.; Vassilenko, K.P.; Serova, O.V.; Popova, N.V.; Zozulya, S.A.; Burova, E.B.; Houillier, P.; Rzhevsky, D.I.; Berchatova, A.A.; et al. Insulin receptor-related receptor as an extracellular alkali sensor. Cell Metab. 2011, 13, 679–689. [Google Scholar] [CrossRef]
- Serova, O.V.; Gantsova, E.A.; Deyev, I.E.; Petrenko, A.G. The Value of pH Sensors in Maintaining Homeostasis of the Nervous System. Russ. J. Bioorganic Chem. 2020, 46, 506–519. [Google Scholar]
- Petrenko, A.G.; Zozulya, S.A.; Deyev, I.E.; Eladari, D. Insulin receptor-related receptor as an extracellular pH sensor involved in the regulation of acid-base balance. Biochim. Biophys. Acta Proteins Proteomics 2013, 1834, 2170–2175. [Google Scholar] [CrossRef]
- Deyev, I.E.; Popova, N.V.; Serova, O.V.; Zhenilo, S.V.; Regoli, M.; Bertelli, E.; Petrenko, A.G. Alkaline pH induces IRR-mediated phosphorylation of IRS-1 and actin cytoskeleton remodeling in a pancreatic beta cell line. Biochimie 2017, 138, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Deyev, I.E.; Rzhevsky, D.I.; Berchatova, A.A.; Serova, O.V.; Popova, N.V. Deficient Response to Experimentally Induced Alkalosis in Mice with the Inactivated insrr Gene. Acta Naturae 2011, 3, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Zubkov, E.A.; Morozova, A.Y.; Chachina, N.A.; Shayakhmetova, D.M.; Mozhaev, A.A.; Deyev, I.E.; Chekhonin, V.P.; Petrenko, A.G. Behavioral Characteristics of Mice with Knockout of the IRR Alkali Sensor Gene. Neurosci. Behav. Physiol. 2018, 48, 483–487. [Google Scholar] [CrossRef]
- Shayahmetova, D.M.; Zhevlenev, E.S.; Mozhaev, A.A.; Deyev, I.E.; Petrenko, A.G. Genetic link between IRR-receptor and Ly6/PLAUR protein. Russ. J. Bioorganic Chem. 2016, 42, 449–452. [Google Scholar] [CrossRef]
- Deyev, I.E.; Chachina, N.A.; Zhevlenev, E.S.; Petrenko, A.G. Site-directed mutagenesis of the fibronectin domains in insulin receptor-related receptor. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef]
- Deyev, I.E.; Chachina, N.A.; Shayahmetova, D.M.; Serova, O.V.; Petrenko, A.G. Mapping of alkali-sensing sites of the insulin receptor-related receptor. the role of L2 and fibronectin domains. Biochimie 2015, 1–9. [Google Scholar] [CrossRef]
- Mozhaev, A.A.; Erokhina, T.N.; Serova, O.V.; Deyev, I.E.; Petrenko, A.G. Production and immunochemical characterization of monoclonal antibody to IRR ectodomain. Russ. J. Bioorg. Chem. 2017, 43, 653–657. [Google Scholar] [CrossRef]
- Shtykova, E.V.; Petoukhov, M.V.; Mozhaev, A.A.; Deyev, I.E.; Dadinova, L.A.; Loshkarev, N.A.; Goryashchenko, A.S.; Bocharov, E.V.; Jeffries, C.M.; Svergun, D.I.; et al. The dimeric ectodomain of the alkali-sensing insulin receptor-related receptor (ectoIRR) has a droplike shape. J. Biol. Chem. 2019, 294, 17790–17798. [Google Scholar] [CrossRef]
- Erokhina, T.N.; Zinovkin, R.A.; Vitushkina, M.V.; Jelkmann, W.; Agranovsky, A.A. Detection of beet yellows closterovirus methyltransferase-like and helicase-like proteins in vivo using monoclonal antibodies. J. Gen. Virol. 2000, 81, 597–603. [Google Scholar] [CrossRef]
- Mozhaev, A.A.; Orsa, A.N.; Deyev, I.E.; Shvets, V.I.; Petrenko, A.G. Optimization of heterological expression of insulin receptor-related receptor ectodomain (Article in Russian). Dokl. Akad. Nauk 2019, 485, 110–123. [Google Scholar] [CrossRef]
- Fauvel, B.; Yasri, A. Antibodies directed against receptor tyrosine kinases: Current and future strategies to fight cancer. mAbs 2014, 6, 838–851. [Google Scholar] [CrossRef] [PubMed]
- Soos, M.A.; Siddle, K.; Baron, M.D.; Heward, J.M.; Luzio, J.P.; Bellatin, J.; Lennox, E.S. Monoclonal antibodies reacting with multiple epitopes on the human insulin receptor. Biochem. J. 1986, 235, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Soos, M.A.; Field, C.E.; Lammers, R.; Ullrich, A.; Zhang, B.; Roth, R.A.; Andersen, A.S.; Kjeldsen, T.; Siddle, K. A Panel of Monoclonal Antibodies for the Type I Insulin-like Growth Factor Receptor. Biochemistry 1992, 267, 12955–12963. [Google Scholar]
- Taylor, R.; Soos, M.A.; Wells, A.; Argyraki, M.; Siddle, K. Insulin-like and insulin-inhibitory effects of monoclonal antibodies for different epitopes on the human insulin receptor. Biochem. J. 1987, 242, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Prigent, S.A.; Stanley, K.K.; Siddle, K. Identification of epitopes on the human insulin receptor reacting with rabbit polyclonal antisera nad mouse monoclonal antibodies. J. Biol. Chem. 1990, 265, 9970–9977. [Google Scholar]
- Issafras, H.; Bedinger, D.H.; Corbin, J.A.; Goldfine, I.D.; Bhaskar, V.; White, M.L.; Rubin, P.; Scannon, P.J. Selective allosteric antibodies to the insulin receptor for the treatment of hyperglycemic and hypoglycemic disorders. J. Diabetes Sci. Technol. 2014, 8, 865–873. [Google Scholar] [CrossRef]
- Rowinsky, E.K.; Youssoufian, H.; Tonra, J.R.; Solomon, P.; Burtrum, D.; Ludwig, D.L. IMC-A12, a human IgG1 monoclonal antibody to the insulin-like growth factor I receptor. Clin. Cancer Res. 2007, 13, 5549–5555. [Google Scholar] [CrossRef]
- Gualberto, A.; Karp, D.D. Development of the monoclonal antibody figitumumab, targeting the insulin-like growth factor-1 receptor, for the treatment of patients with non-small-cell lung cancer. Clin. Lung Cancer 2009, 10, 273–280. [Google Scholar] [CrossRef]
- Scartozzi, M.; Bianconi, M.; MacCaroni, E.; Giampieri, R.; Berardi, R.; Cascinu, S. Dalotuzumab, a recombinant humanized mab targeted against IGFR1 for the treatment of cancer. Curr. Opin. Mol. Ther. 2010, 12, 361–371. [Google Scholar]
- Beltran, P.J.; Calzone, F.J.; Mitchell, P.; Chung, Y.A.; Cajulis, E.; Moody, G.; Belmontes, B.; Li, C.M.; Vonderfecht, S.; Velculescu, V.E.; et al. Ganitumab (AMG 479) inhibits IGF-II-dependent ovarian cancer growth and potentiates platinum-based chemotherapy. Clin. Cancer Res. 2014, 20, 2947–2958. [Google Scholar] [CrossRef]
- Pappo, A.S.; Patel, S.R.; Crowley, J.; Reinke, D.K.; Kuenkele, K.P.; Chawla, S.P.; Toner, G.C.; Maki, R.G.; Meyers, P.A.; Chugh, R.; et al. R1507, a monoclonal antibody to the insulin-like growth factor 1 receptor, in patients with recurrent or refractory Ewing sarcoma family of tumors: Results of a phase II Sarcoma Alliance for research through Collaboration study. J. Clin. Oncol. 2011, 29, 4541–4547. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.H.; Lenz, H.J.; Saleh, M.N.; Mackenzie, M.J.; Knost, J.A.; Pathiraja, K.; Langdon, R.B.; Yao, S.L.; Lu, B.D. A randomized, phase II study of the anti-insulin-like growth factor receptor type 1 (IGF-1R) monoclonal antibody robatumumab (SCH 717454) in patients with advanced colorectal cancer. Cancer Med. 2014, 3, 988–997. [Google Scholar] [CrossRef] [PubMed]
- Macaulay, V.M.; Middleton, M.R.; Protheroe, A.S.; Tolcher, A.; Dieras, V.; Sessa, C.; Bahleda, R.; Blay, J.Y.; LoRusso, P.; Mery-Mignard, D.; et al. Phase I study of humanized monoclonal antibody AVE1642 directed against the type 1 insulin-like growth factor receptor (IGF-1R), administered in combination with anticancer therapies to patients with advanced solid tumors. Ann. Oncol. 2013, 24, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Von Mehren, M.; Britten, C.D.; Pieslor, P.; Saville, W.; Vassos, A.; Harris, S.; Galluppi, G.R.; Darif, M.; Wainberg, Z.A.; Cohen, R.B.; et al. A phase 1, open-label, dose-escalation study of BIIB022 (anti-IGF-1R monoclonal antibody) in subjects with relapsed or refractory solid tumors. Invest. New Drugs 2014, 32, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhang, Y.; Lv, M.; Feng, J.; Peng, H.; Geng, J.; Lin, Z.; Zhou, T.; Li, X.; Shen, B.; et al. Anti-IGF-1R monoclonal antibody inhibits the carcinogenicity activity of acquired trastuzumab-resistant SKOV3. J. Ovarian Res. 2014, 7. [Google Scholar] [CrossRef] [PubMed]
- Burtrum, D.; Zhu, Z.; Lu, D.; Anderson, D.M.; Prewett, M.; Pereira, D.S.; Bassi, R.; Abdullah, R.; Hooper, A.T.; Koo, H.; et al. A Fully Human Monoclonal Antibody to the Insulin-Like Growth Factor I Receptor Blocks Ligand-Dependent Signaling and Inhibits Human Tumor Growth in Vivo. Cancer Res. 2003, 63, 8912–8921. [Google Scholar] [PubMed]
- Kiselyov, V.V.; Versteyhe, S.; Gauguin, L.; De Meyts, P. Harmonic oscillator model of the insulin and IGF1 receptors’ allosteric binding and activation. Mol. Syst. Biol. 2009, 5, 243–255. [Google Scholar] [CrossRef]
- Uchikawa, E.; Choi, E.; Shang, G.; Yu, H.; Xiao-Chen, B. Activation mechanism of the insulin receptor revealed by cryo-EM structure of the fully liganded receptor-ligand complex. Elife 2019, 8, 18–23. [Google Scholar] [CrossRef]
- De Meyts, P. The Insulin Receptor and Its Signal Transduction Network; Endotext: South Dartmouth, MA, USA, 2000. [Google Scholar]


| Antibody | With Mercaptoethanol | Without Mercaptoethanol | ||||
|---|---|---|---|---|---|---|
| hIRR | mIRR | ectoIRR | hIRR | mIRR | ectoIRR | |
| 1D2 | − | − | − | − | − | + |
| 4D5 | + | − | + | − | − | − |
| 4C2 | − | − | − | − | − | + |
| 4A2 | − | − | − | − | − | − |
| 3C5 | − | − | − | − | − | − |
| 3B4 | − | − | − | − | − | + |
| Anti-IRR ectodomain antibody | + | + | + | + | + | + |
| Protein/Mutant | Relative Activity, % | Аntibody | |||||
|---|---|---|---|---|---|---|---|
| 1D2 | 3C5 | 4C2 | 4D5 | 4A2 | 3B4 | ||
| IRR | 100 | ++ | ++ | ++ | ++ | ++ | −− |
| L1C | 35 [16] | −− | ++ | ++ | ++ | ++ | −− |
| L2 | 64 [16] | ++ | ++ | ++ | ++ | +− | −− |
| FnIII-1 | 36 [16] | ++ | −− | +− | −− | +− | −− |
| FnIII-2/3 | 0 [16] | ++ | −− | −− | ++ | −− | −− |
| IRR-5A | 96 [16] | −− | −− | ++ | ++ | ++ | −− |
| C-pept-TK(N) | 100 [16] | ++ | ++ | ++ | ++ | ++ | −− |
| MVD | 28 [16] | ++ | −− | ++ | ++ | +− | −− |
| PV | 154 [16] | ++ | ++ | ++ | ++ | +− | −− |
| T(A) | 64 [15] | ++ | ++ | ++ | ++ | ++ | −− |
| RL-Fn | 100 [15] | ++ | ++ | ++ | ++ | +− | −− |
| Result | L1C | Fn(III)-1 | Fn(III)-2/3 | Fn(III)-1 | Fn(III)-2/3 | − | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goryashchenko, A.S.; Mozhaev, A.A.; Serova, O.V.; Erokhina, T.N.; Orsa, A.N.; Deyev, I.E.; Petrenko, A.G. Probing Structure and Function of Alkali Sensor IRR with Monoclonal Antibodies. Biomolecules 2020, 10, 1060. https://doi.org/10.3390/biom10071060
Goryashchenko AS, Mozhaev AA, Serova OV, Erokhina TN, Orsa AN, Deyev IE, Petrenko AG. Probing Structure and Function of Alkali Sensor IRR with Monoclonal Antibodies. Biomolecules. 2020; 10(7):1060. https://doi.org/10.3390/biom10071060
Chicago/Turabian StyleGoryashchenko, Alexander S., Andrey A. Mozhaev, Oxana V. Serova, Tatiana N. Erokhina, Alexander N. Orsa, Igor E. Deyev, and Alexander G. Petrenko. 2020. "Probing Structure and Function of Alkali Sensor IRR with Monoclonal Antibodies" Biomolecules 10, no. 7: 1060. https://doi.org/10.3390/biom10071060
APA StyleGoryashchenko, A. S., Mozhaev, A. A., Serova, O. V., Erokhina, T. N., Orsa, A. N., Deyev, I. E., & Petrenko, A. G. (2020). Probing Structure and Function of Alkali Sensor IRR with Monoclonal Antibodies. Biomolecules, 10(7), 1060. https://doi.org/10.3390/biom10071060







