The Link between Homocysteine and Omega-3 Polyunsaturated Fatty Acid: Critical Appraisal and Future Directions
Abstract
1. Introduction
2. Methods
3. Involvement of Homocysteine in Human Diseases
4. The Role of Omega-3 Polyunsaturated Fatty Acids in Human Diseases
5. Evidence about the Interaction between Homocysteine and Omega-3 Polyunsaturated Fatty Acids from Systematic Reviews and Meta-Analyses
6. Latest Insight and Implications for Cognitive Performance and Neurobiology
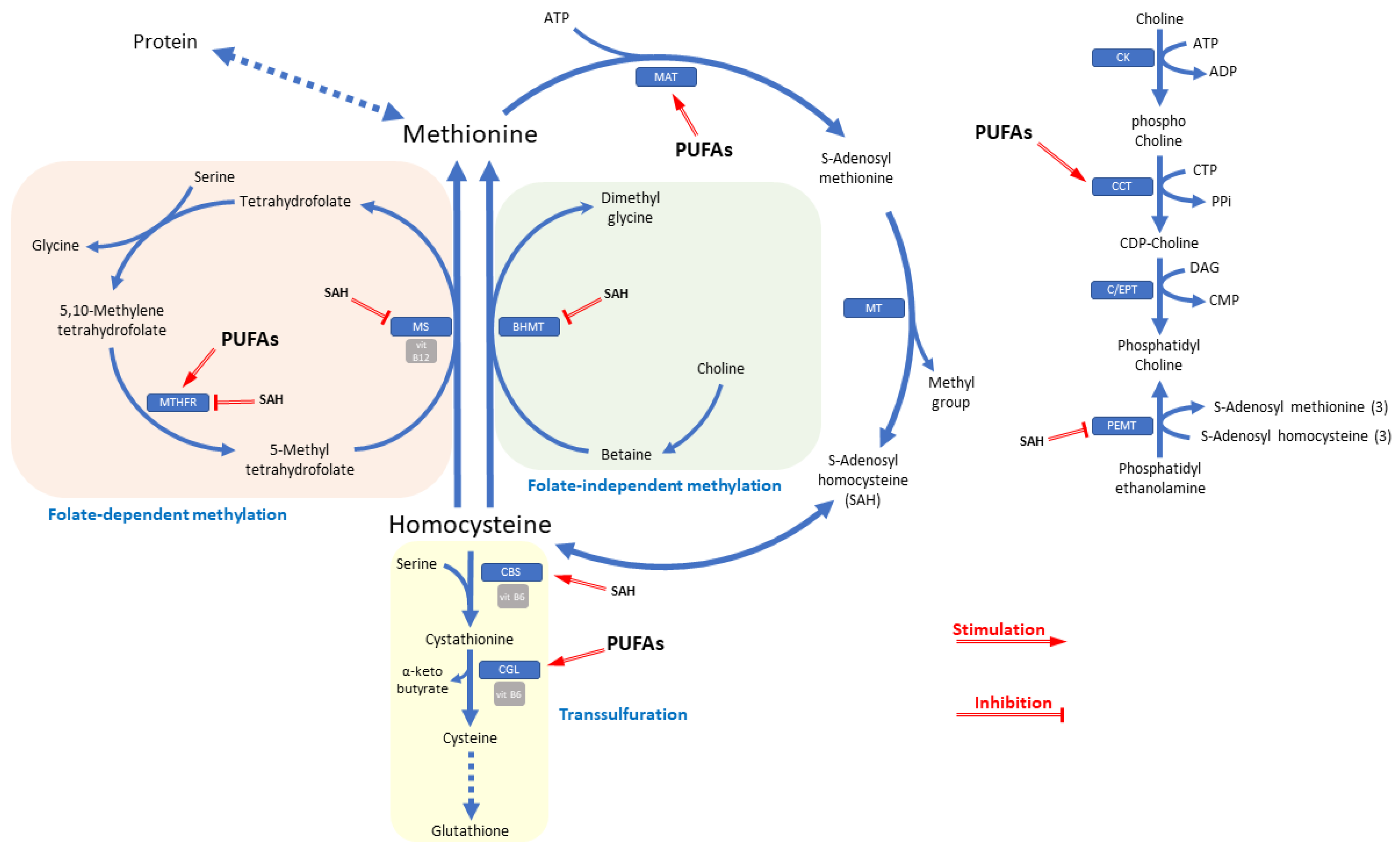
7. Possible Mechanisms of Interaction between Homocysteine and Omega-3 Polyunsaturated Fatty Acids
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Finkelstein, J.D. Methionine metabolism in mammals. J. Nutr. Biochem. 1990, 1, 228–237. [Google Scholar] [CrossRef]
- Finkelstein, J.D. Pathways and regulation of homocysteine metabolism in mammals. Semin. Thromb. Hemost. 2000, 26, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, J.D. The metabolism of homocysteine: Pathways and regulation. Eur. J. Pediatr. 1998, 157 (Suppl. 2), S40–S44. [Google Scholar] [CrossRef]
- Huang, T.; Yuan, G.; Zhang, Z.; Zou, Z.; Li, D. Cardiovascular pathogenesis in hyperhomocysteinemia. Asia Pac. J. Clin. Nutr. 2008, 17, 8–16. [Google Scholar] [PubMed]
- Green, R.; Allen, L.H.; Bjørke-Monsen, A.-L.; Brito, A.; Guéant, J.-L.; Miller, J.W.; Molloy, A.M.; Nexo, E.; Stabler, S.; Toh, B.-H.; et al. Vitamin B12 deficiency. Nat. Rev. Dis. Primers 2017, 3, 17040. [Google Scholar] [CrossRef]
- Miller, E.R.; Juraschek, S.; Pastor-Barriuso, R.; Bazzano, L.A.; Appel, L.J.; Guallar, E. Meta-analysis of folic acid supplementation trials on risk of cardiovascular disease and risk interaction with baseline homocysteine levels. Am. J. Cardiol. 2010, 106, 517–527. [Google Scholar] [CrossRef]
- Jacques, P.F.; Selhub, J.; Bostom, A.G.; Wilson, P.W.; Rosenberg, I.H. The effect of folic acid fortification on plasma folate and total homocysteine concentrations. N. Engl. J. Med. 1999, 340, 1449–1454. [Google Scholar] [CrossRef]
- Green, R.; Miller, J.W. Vitamin B12 deficiency is the dominant nutritional cause of hyperhomocysteinemia in a folic acid-fortified population. Clin. Chem. Lab. Med. 2005, 43, 1048–1051. [Google Scholar] [CrossRef]
- de Benoist, B. Conclusions of a WHO Technical Consultation on folate and vitamin B12 deficiencies. Food Nutr. Bull. 2008, 29, S238–S244. [Google Scholar] [CrossRef]
- Rizzo, G.; Laganà, A.S.; Rapisarda, A.M.C.; La Ferrera, G.M.G.; Buscema, M.; Rossetti, P.; Nigro, A.; Muscia, V.; Valenti, G.; Sapia, F.; et al. Vitamin B12 among Vegetarians: Status, Assessment and Supplementation. Nutrients 2016, 8, 767. [Google Scholar] [CrossRef]
- Gjesdal, C.G.; Vollset, S.E.; Ueland, P.M.; Refsum, H.; Drevon, C.A.; Gjessing, H.K.; Tell, G.S. Plasma total homocysteine level and bone mineral density: The Hordaland Homocysteine Study. Arch. Intern. Med. 2006, 166, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Boushey, C.J.; Beresford, S.A.; Omenn, G.S.; Motulsky, A.G. A quantitative assessment of plasma homocysteine as a risk factor for vascular disease. Probable benefits of increasing folic acid intakes. JAMA 1995, 274, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Kivipelto, M.; Annerbo, S.; Hultdin, J.; Bäckman, L.; Viitanen, M.; Fratiglioni, L.; Lökk, J. Homocysteine and holo-transcobalamin and the risk of dementia and Alzheimers disease: A prospective study. Eur. J. Neurol. 2009, 16, 808–813. [Google Scholar] [CrossRef] [PubMed]
- Harker, L.A.; Slichter, S.J.; Scott, C.R.; Ross, R. Homocystinemia. Vascular injury and arterial thrombosis. N. Engl. J. Med. 1974, 291, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Clarke, R.; Daly, L.; Robinson, K.; Naughten, E.; Cahalane, S.; Fowler, B.; Graham, I. Hyperhomocysteinemia: An independent risk factor for vascular disease. N. Engl. J. Med. 1991, 324, 1149–1155. [Google Scholar] [CrossRef]
- Devalia, V.; Hamilton, M.S.; Molloy, A.M. British Committee for Standards in Haematology Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br. J. Haematol. 2014, 166, 496–513. [Google Scholar] [CrossRef]
- Wald, D.S.; Law, M.; Morris, J.K. Homocysteine and cardiovascular disease: Evidence on causality from a meta-analysis. BMJ 2002, 325, 1202. [Google Scholar] [CrossRef]
- van Meurs, J.B.J.; Pare, G.; Schwartz, S.M.; Hazra, A.; Tanaka, T.; Vermeulen, S.H.; Cotlarciuc, I.; Yuan, X.; Mälarstig, A.; Bandinelli, S.; et al. Common genetic loci influencing plasma homocysteine concentrations and their effect on risk of coronary artery disease. Am. J. Clin. Nutr. 2013, 98, 668–676. [Google Scholar] [CrossRef]
- Rizzo, G.; Laganà, A.S. Chapter 6—A review of vitamin B12. In Molecular Nutrition; Patel, V.B., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 105–129. ISBN 978-0-12-811907-5. [Google Scholar]
- Himmelfarb, J.; Phinney, S.; Ikizler, T.A.; Kane, J.; McMonagle, E.; Miller, G. Gamma-tocopherol and docosahexaenoic acid decrease inflammation in dialysis patients. J. Ren. Nutr. 2007, 17, 296–304. [Google Scholar] [CrossRef]
- Vernaglione, L.; Cristofano, C.; Chimienti, S. Omega-3 polyunsaturated fatty acids and proxies of cardiovascular disease in hemodialysis: A prospective cohort study. J. Nephrol. 2008, 21, 99–105. [Google Scholar]
- Martí-Carvajal, A.J.; Solà, I.; Lathyris, D.; Dayer, M. Homocysteine-lowering interventions for preventing cardiovascular events. Cochrane Database Syst. Rev. 2017, 8, CD006612. [Google Scholar] [CrossRef]
- Kuratko, C.N.; Nolan, C.C.; Salem, N. Long-chain omega-3 fatty acids and cardiovascular health. Nutrafoods 2014, 13, 49–60. [Google Scholar] [CrossRef]
- Bang, H.O.; Dyerberg, J.; Sinclair, H.M. The composition of the Eskimo food in north western Greenland. Am. J. Clin. Nutr. 1980, 33, 2657–2661. [Google Scholar] [CrossRef] [PubMed]
- Dyerberg, J.; Bang, H.O. A hypothesis on the development of acute myocardial infarction in Greenlanders. Scand. J. Clin. Lab. Invest. Suppl. 1982, 161, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.; Kirsch, H.; Domergue, F.; Abbadi, A.; Sperling, P.; Bauer, J.; Cirpus, P.; Zank, T.K.; Moreau, H.; Roscoe, T.J.; et al. Novel fatty acid elongases and their use for the reconstitution of docosahexaenoic acid biosynthesis. J. Lipid Res. 2004, 45, 1899–1909. [Google Scholar] [CrossRef]
- Bernstein, A.M.; Ding, E.L.; Willett, W.C.; Rimm, E.B. A meta-analysis shows that docosahexaenoic acid from algal oil reduces serum triglycerides and increases HDL-cholesterol and LDL-cholesterol in persons without coronary heart disease. J. Nutr. 2012, 142, 99–104. [Google Scholar] [CrossRef]
- Rizzo, G.; Baroni, L. Health and ecological implications of fish consumption: A deeper insight. Mediterr. J. Nutr. Metab. 2016, 9, 7–22. [Google Scholar] [CrossRef]
- Schmitz, G.; Ecker, J. The opposing effects of n-3 and n-6 fatty acids. Prog. Lipid Res. 2008, 47, 147–155. [Google Scholar] [CrossRef]
- Omega-3 Fatty Acids in Brain and Neurological Health; Elsevier: Amsterdam, The Netherlands, 2014; ISBN 978-0-12-410527-0.
- Harris, W.S. n-3 fatty acids and serum lipoproteins: Human studies. Am. J. Clin. Nutr. 1997, 65, 1645S–1654S. [Google Scholar] [CrossRef]
- Bucher, H.C.; Hengstler, P.; Schindler, C.; Meier, G. N-3 polyunsaturated fatty acids in coronary heart disease: A meta-analysis of randomized controlled trials. Am. J. Med. 2002, 112, 298–304. [Google Scholar] [CrossRef]
- Ubeda, N.; Achón, M.; Varela-Moreiras, G. Omega 3 fatty acids in the elderly. Br. J. Nutr. 2012, 107 (Suppl. 2), S137–S151. [Google Scholar] [CrossRef] [PubMed]
- Caleyachetty, R.; Echouffo-Tcheugui, J.B.; Tait, C.A.; Schilsky, S.; Forrester, T.; Kengne, A.P. Prevalence of behavioural risk factors for cardiovascular disease in adolescents in low-income and middle-income countries: An individual participant data meta-analysis. Lancet Diabetes Endocrinol. 2015, 3, 535–544. [Google Scholar] [CrossRef]
- Brambilla, P.; Lissau, I.; Flodmark, C.-E.; Moreno, L.A.; Widhalm, K.; Wabitsch, M.; Pietrobelli, A. Metabolic risk-factor clustering estimation in children: To draw a line across pediatric metabolic syndrome. Int. J. Obes. (Lond) 2007, 31, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.J.; Shiri-Feshki, M. Rate of progression of mild cognitive impairment to dementia-meta-analysis of 41 robust inception cohort studies. Acta Psychiatr. Scand. 2009, 119, 252–265. [Google Scholar] [CrossRef]
- Peng, H.; Man, C.; Xu, J.; Fan, Y. Elevated homocysteine levels and risk of cardiovascular and all-cause mortality: A meta-analysis of prospective studies. J. Zhejiang Univ. Sci. B 2015, 16, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Lonn, E.; Yusuf, S.; Arnold, M.J.; Sheridan, P.; Pogue, J.; Micks, M.; McQueen, M.J.; Probstfield, J.; Fodor, G.; Held, C.; et al. Homocysteine lowering with folic acid and B vitamins in vascular disease. N. Engl. J. Med. 2006, 354, 1567–1577. [Google Scholar]
- Veeranna, V.; Zalawadiya, S.K.; Niraj, A.; Pradhan, J.; Ference, B.; Burack, R.C.; Jacob, S.; Afonso, L. Homocysteine and reclassification of cardiovascular disease risk. J. Am. Coll. Cardiol. 2011, 58, 1025–1033. [Google Scholar] [CrossRef]
- Ganguly, P.; Alam, S.F. Role of homocysteine in the development of cardiovascular disease. Nutr. J. 2015, 14, 6. [Google Scholar] [CrossRef]
- Miao, L.; Deng, G.-X.; Yin, R.-X.; Nie, R.-J.; Yang, S.; Wang, Y.; Li, H. No causal effects of plasma homocysteine levels on the risk of coronary heart disease or acute myocardial infarction: A Mendelian randomization study. Eur. J. Prev. Cardiol. 2019. [Google Scholar] [CrossRef]
- Lewis, S.J.; Ebrahim, S.; Davey Smith, G. Meta-analysis of MTHFR 677C->T polymorphism and coronary heart disease: Does totality of evidence support causal role for homocysteine and preventive potential of folate? BMJ 2005, 331, 1053. [Google Scholar] [CrossRef]
- Fezeu, L.K.; Ducros, V.; Guéant, J.-L.; Guilland, J.-C.; Andreeva, V.A.; Hercberg, S.; Galan, P. MTHFR 677C → T genotype modulates the effect of a 5-year supplementation with B-vitamins on homocysteine concentration: The SU.FOL.OM3 randomized controlled trial. PLoS ONE 2018, 13, e0193352. [Google Scholar] [CrossRef] [PubMed]
- Cosentino, F.; Rubattu, S.; Savoia, C.; Venturelli, V.; Pagannonne, E.; Volpe, M. Endothelial dysfunction and stroke. J. Cardiovasc. Pharmacol. 2001, 38 (Suppl. 2), S75–S78. [Google Scholar] [CrossRef] [PubMed]
- Stamler, J.S.; Osborne, J.A.; Jaraki, O.; Rabbani, L.E.; Mullins, M.; Singel, D.; Loscalzo, J. Adverse vascular effects of homocysteine are modulated by endothelium-derived relaxing factor and related oxides of nitrogen. J. Clin. Investig. 1993, 91, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Starkebaum, G.; Harlan, J.M. Endothelial cell injury due to copper-catalyzed hydrogen peroxide generation from homocysteine. J. Clin. Investig. 1986, 77, 1370–1376. [Google Scholar] [CrossRef] [PubMed]
- Upchurch, G.R.; Welch, G.N.; Fabian, A.J.; Freedman, J.E.; Johnson, J.L.; Keaney, J.F.; Loscalzo, J. Homocyst(e)ine decreases bioavailable nitric oxide by a mechanism involving glutathione peroxidase. J. Biol. Chem. 1997, 272, 17012–17017. [Google Scholar] [CrossRef]
- Brown, A.A.; Hu, F.B. Dietary modulation of endothelial function: Implications for cardiovascular disease. Am. J. Clin. Nutr. 2001, 73, 673–686. [Google Scholar] [CrossRef]
- McCully, K.S. Homocysteine and the pathogenesis of atherosclerosis. Expert Rev. Clin. Pharmacol. 2015, 8, 211–219. [Google Scholar] [CrossRef]
- Wang, J.; Dudman, N.P.; Wilcken, D.E.; Lynch, J.F. Homocysteine catabolism: Levels of 3 enzymes in cultured human vascular endothelium and their relevance to vascular disease. Atherosclerosis 1992, 97, 97–106. [Google Scholar] [CrossRef]
- Taoka, S.; Ohja, S.; Shan, X.; Kruger, W.D.; Banerjee, R. Evidence for heme-mediated redox regulation of human cystathionine beta-synthase activity. J. Biol. Chem. 1998, 273, 25179–25184. [Google Scholar] [CrossRef]
- Bostom, A.G.; Lathrop, L. Hyperhomocysteinemia in end-stage renal disease: Prevalence, etiology, and potential relationship to arteriosclerotic outcomes. Kidney Int. 1997, 52, 10–20. [Google Scholar] [CrossRef]
- Wijekoon, E.P.; Brosnan, M.E.; Brosnan, J.T. Homocysteine metabolism in diabetes. Biochem. Soc. Trans. 2007, 35, 1175–1179. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Nam, J.H.; Oh, D.H.; Park, Y. Erythrocyte α-linolenic acid is associated with the risk for mild dementia in Korean elderly. Nutr. Res. 2010, 30, 756–761. [Google Scholar] [CrossRef] [PubMed]
- von Schacky, C. n-3 fatty acids and the prevention of coronary atherosclerosis. Am. J. Clin. Nutr. 2000, 71, 224S–227S. [Google Scholar] [CrossRef] [PubMed]
- Lenihan-Geels, G.; Bishop, K.S.; Ferguson, L.R. Alternative Sources of Omega-3 Fats: Can We Find a Sustainable Substitute for Fish? Nutrients 2013, 5, 1301–1315. [Google Scholar] [CrossRef]
- Leaf, A.; Weber, P.C. Cardiovascular effects of n-3 fatty acids. N. Engl. J. Med. 1988, 318, 549–557. [Google Scholar] [CrossRef]
- Bowden, R.G.; Wilson, R.L.; Deike, E.; Gentile, M. Fish oil supplementation lowers C-reactive protein levels independent of triglyceride reduction in patients with end-stage renal disease. Nutr. Clin. Pract. 2009, 24, 508–512. [Google Scholar] [CrossRef]
- Abeywardena, M.Y.; Belobrajdic, D.P. Long-Chain Omega-3 Polyunsaturated Fatty Acids and Obesity. In Obesity: A Practical Guide; Ahmad, S.I., Imam, S.K., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 29–44. ISBN 978-3-319-19821-7. [Google Scholar]
- Robinson, L.E.; Buchholz, A.C.; Mazurak, V.C. Inflammation, obesity, and fatty acid metabolism: Influence of n-3 polyunsaturated fatty acids on factors contributing to metabolic syndrome. Appl. Physiol. Nutr. Metab. 2007, 32, 1008–1024. [Google Scholar] [CrossRef]
- Jenkins, C.M.; Cedars, A.; Gross, R.W. Eicosanoid signalling pathways in the heart. Cardiovasc. Res. 2009, 82, 240–249. [Google Scholar] [CrossRef]
- Akbar, M.; Calderon, F.; Wen, Z.; Kim, H.-Y. Docosahexaenoic acid: A positive modulator of Akt signaling in neuronal survival. Proc. Natl. Acad. Sci. USA 2005, 102, 10858–10863. [Google Scholar] [CrossRef]
- Yang, R.; Chiang, N.; Oh, S.F.; Serhan, C.N. Metabolomics-lipidomics of eicosanoids and docosanoids generated by phagocytes. Curr. Protoc. Immunol. 2011, Chapter 14, Unit 14.26. [Google Scholar] [CrossRef]
- de Bree, A.; Mennen, L.I.; Hercberg, S.; Galan, P. Evidence for a protective (synergistic?) effect of B-vitamins and omega-3 fatty acids on cardiovascular diseases. Eur. J. Clin. Nutr. 2004, 58, 732–744. [Google Scholar] [CrossRef] [PubMed]
- Olszewski, A.J.; McCully, K.S. Fish oil decreases serum homocysteine in hyperlipemic men. Coron. Artery Dis. 1993, 4, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Haglund, O.; Wallin, R.; Wretling, S.; Hultberg, B.; Saldeen, T. Effects of fish oil alone and combined with long chain (n-6) fatty acids on some coronary risk factors in male subjects. J. Nutr. Biochem. 1998, 9, 629–635. [Google Scholar] [CrossRef]
- Piolot, A.; Blache, D.; Boulet, L.; Fortin, L.J.; Dubreuil, D.; Marcoux, C.; Davignon, J.; Lussier-Cacan, S. Effect of fish oil on LDL oxidation and plasma homocysteine concentrations in health. J. Lab. Clin. Med. 2003, 141, 41–49. [Google Scholar] [CrossRef]
- Huang, T.; Zheng, J.; Chen, Y.; Yang, B.; Wahlqvist, M.L.; Li, D. High consumption of Ω-3 polyunsaturated fatty acids decrease plasma homocysteine: A meta-analysis of randomized, placebo-controlled trials. Nutrition 2011, 27, 863–867. [Google Scholar] [CrossRef]
- Beavers, K.M.; Beavers, D.P.; Bowden, R.G.; Wilson, R.L.; Gentile, M. Omega-3 fatty acid supplementation and total homocysteine levels in end-stage renal disease patients. Nephrology (Carlton) 2008, 13, 284–288. [Google Scholar] [CrossRef]
- Zeman, M.; Zák, A.; Vecka, M.; Tvrzická, E.; Písaríková, A.; Stanková, B. N-3 fatty acid supplementation decreases plasma homocysteine in diabetic dyslipidemia treated with statin-fibrate combination. J. Nutr. Biochem. 2006, 17, 379–384. [Google Scholar] [CrossRef]
- Dawson, S.L.; Bowe, S.J.; Crowe, T.C. A combination of omega-3 fatty acids, folic acid and B-group vitamins is superior at lowering homocysteine than omega-3 alone: A meta-analysis. Nutr. Res. 2016, 36, 499–508. [Google Scholar] [CrossRef]
- Galan, P.; Kesse-Guyot, E.; Czernichow, S.; Briancon, S.; Blacher, J.; Hercberg, S. SU.FOL.OM3 Collaborative Group Effects of B vitamins and omega 3 fatty acids on cardiovascular diseases: A randomised placebo controlled trial. BMJ 2010, 341, c6273. [Google Scholar] [CrossRef]
- Xu, T.; Sun, Y.; Sun, W.; Yao, L.; Sun, L.; Liu, L.; Ma, J.; Wang, L. Effect of omega-3 fatty acid supplementation on serum lipids and vascular inflammation in patients with end-stage renal disease: A meta-analysis. Sci. Rep. 2016, 6, 39346. [Google Scholar] [CrossRef]
- Beydoun, M.A.; Beydoun, H.A.; Gamaldo, A.A.; Teel, A.; Zonderman, A.B.; Wang, Y. Epidemiologic studies of modifiable factors associated with cognition and dementia: Systematic review and meta-analysis. BMC Public Health 2014, 14, 643. [Google Scholar] [CrossRef] [PubMed]
- Wald, D.S.; Kasturiratne, A.; Simmonds, M. Serum homocysteine and dementia: Meta-analysis of eight cohort studies including 8669 participants. Alzheimers Dement. 2011, 7, 412–417. [Google Scholar] [CrossRef] [PubMed]
- Baroni, L.; Bonetto, C.; Rizzo, G.; Bertola, C.; Caberlotto, L.; Bazzerla, G. Association Between Cognitive Impairment and Vitamin B12, Folate, and Homocysteine Status in Elderly Adults: A Retrospective Study. J. Alzheimers Dis. 2019, 70, 443–453. [Google Scholar] [CrossRef]
- Velho, S.; Marques-Vidal, P.; Baptista, F.; Camilo, M.E. Dietary intake adequacy and cognitive function in free-living active elderly: A cross-sectional and short-term prospective study. Clin. Nutr. 2008, 27, 77–86. [Google Scholar] [CrossRef]
- Yurko-Mauro, K.; McCarthy, D.; Rom, D.; Nelson, E.B.; Ryan, A.S.; Blackwell, A.; Salem, N.; Stedman, M. MIDAS Investigators Beneficial effects of docosahexaenoic acid on cognition in age-related cognitive decline. Alzheimers Dement. 2010, 6, 456–464. [Google Scholar] [CrossRef]
- Kume, A.; Kurotani, K.; Sato, M.; Ejima, Y.; Pham, N.M.; Nanri, A.; Kuwahara, K.; Mizoue, T. Polyunsaturated fatty acids in serum and homocysteine concentrations in Japanese men and women: A cross-sectional study. Nutr. Metab. (Lond) 2013, 10, 41. [Google Scholar] [CrossRef]
- Laurin, D.; Verreault, R.; Lindsay, J.; Dewailly, E.; Holub, B.J. Omega-3 fatty acids and risk of cognitive impairment and dementia. J. Alzheimers Dis. 2003, 5, 315–322. [Google Scholar] [CrossRef]
- Lauritzen, L.; Hansen, H.S.; Jørgensen, M.H.; Michaelsen, K.F. The essentiality of long chain n-3 fatty acids in relation to development and function of the brain and retina. Prog. Lipid Res. 2001, 40, 1–94. [Google Scholar] [CrossRef]
- Selley, M.L. A metabolic link between S-adenosylhomocysteine and polyunsaturated fatty acid metabolism in Alzheimer’s disease. Neurobiol. Aging 2007, 28, 1834–1839. [Google Scholar] [CrossRef]
- Baierle, M.; Vencato, P.H.; Oldenburg, L.; Bordignon, S.; Zibetti, M.; Trentini, C.M.; Duarte, M.M.M.F.; Veit, J.C.; Somacal, S.; Emanuelli, T.; et al. Fatty acid status and its relationship to cognitive decline and homocysteine levels in the elderly. Nutrients 2014, 6, 3624–3640. [Google Scholar] [CrossRef]
- Jernerén, F.; Elshorbagy, A.K.; Oulhaj, A.; Smith, S.M.; Refsum, H.; Smith, A.D. Brain atrophy in cognitively impaired elderly: The importance of long-chain ω-3 fatty acids and B vitamin status in a randomized controlled trial. Am. J. Clin. Nutr. 2015, 102, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Oulhaj, A.; Jernerén, F.; Refsum, H.; Smith, A.D.; de Jager, C.A. Omega-3 Fatty Acid Status Enhances the Prevention of Cognitive Decline by B Vitamins in Mild Cognitive Impairment. J. Alzheimers Dis. 2016, 50, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Hooper, C.; De Souto Barreto, P.; Coley, N.; Caussé, E.; Payoux, P.; Salabert, A.S.; Cesari, M.; Andrieu, S.; Bowman, G.-L.; Weiner, M.; et al. Cross-Sectional Associations of Total Plasma Homocysteine with Cortical β-Amyloid Independently and as a Function of Omega 3 Polyunsaturated Fatty Acid Status in Older Adults at Risk of Dementia. J. Nutr. Health Aging 2017, 21, 1075–1080. [Google Scholar] [CrossRef] [PubMed]
- Hooper, C.; De Souto Barreto, P.; Payoux, P.; Salabert, A.S.; Guyonnet, S.; Andrieu, S.; Vellas, B. MAPT/DSA study group Cross-sectional associations of cortical β-amyloid with erythrocyte membrane long-chain polyunsaturated fatty acids in older adults with subjective memory complaints. J. Neurochem. 2017, 142, 589–596. [Google Scholar] [CrossRef]
- Uchida, K. 4-Hydroxy-2-nonenal: A product and mediator of oxidative stress. Prog. Lipid Res. 2003, 42, 318–343. [Google Scholar] [CrossRef]
- Schebb, N.H.; Ostermann, A.I.; Yang, J.; Hammock, B.D.; Hahn, A.; Schuchardt, J.P. Comparison of the effects of long-chain omega-3 fatty acid supplementation on plasma levels of free and esterified oxylipins. Prostaglandins Other Lipid Mediat. 2014, 113–115, 21–29. [Google Scholar] [CrossRef]
- Cipollina, C. Endogenous Generation and Signaling Actions of Omega-3 Fatty Acid Electrophilic Derivatives. Biomed. Res. Int. 2015, 2015, 501792. [Google Scholar] [CrossRef]
- Obeid, R.; Herrmann, W. Homocysteine and lipids: S-adenosyl methionine as a key intermediate. FEBS Lett. 2009, 583, 1215–1225. [Google Scholar] [CrossRef]
- Mallampalli, R.K.; Salome, R.G.; Spector, A.A. Regulation of CTP:choline-phosphate cytidylyltransferase by polyunsaturated n-3 fatty acids. Am. J. Physiol. 1994, 267, L641–L648. [Google Scholar] [CrossRef]
- Mueller, J.F.; Iacono, J.M. Effect of Desoxypyridoxine-Induced Vitamin B6 Deficiency on Polyunsaturated Fatty Acid Metabolism in Human Beings. Am. J. Clin. Nutr. 1963, 12, 358–367. [Google Scholar] [CrossRef]
- Akesson, B.; Fehling, C.; Jägerstad, M.; Stenram, U. Effect of experimental folate deficiency on lipid metabolism in liver and brain. Br. J. Nutr. 1982, 47, 505–520. [Google Scholar] [CrossRef] [PubMed]
- da Silva, R.P.; Kelly, K.B.; Al Rajabi, A.; Jacobs, R.L. Novel insights on interactions between folate and lipid metabolism. Biofactors 2014, 40, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Lamers, Y.; Ralat, M.A.; Coats, B.S.; Chi, Y.-Y.; Muller, K.E.; Bain, J.R.; Shankar, M.N.; Newgard, C.B.; Stacpoole, P.W.; et al. Marginal vitamin B-6 deficiency decreases plasma (n-3) and (n-6) PUFA concentrations in healthy men and women. J. Nutr. 2012, 142, 1791–1797. [Google Scholar] [CrossRef] [PubMed][Green Version]
- da Costa, K.-A.; Sanders, L.M.; Fischer, L.M.; Zeisel, S.H. Docosahexaenoic acid in plasma phosphatidylcholine may be a potential marker for in vivo phosphatidylethanolamine N-methyltransferase activity in humans. Am. J. Clin. Nutr. 2011, 93, 968–974. [Google Scholar] [CrossRef]
- Serafim, V.; Chirita-Emandi, A.; Andreescu, N.; Tiugan, D.-A.; Tutac, P.; Paul, C.; Velea, I.; Mihailescu, A.; Șerban, C.L.; Zimbru, C.G.; et al. Single Nucleotide Polymorphisms in PEMT and MTHFR Genes are Associated with Omega 3 and 6 Fatty Acid Levels in the Red Blood Cells of Children with Obesity. Nutrients 2019, 11, 2600. [Google Scholar] [CrossRef] [PubMed]
- DeLong, C.J.; Shen, Y.J.; Thomas, M.J.; Cui, Z. Molecular distinction of phosphatidylcholine synthesis between the CDP-choline pathway and phosphatidylethanolamine methylation pathway. J. Biol. Chem. 1999, 274, 29683–29688. [Google Scholar] [CrossRef]
- Svingen, G.F.T.; Schartum-Hansen, H.; Ueland, P.M.; Pedersen, E.R.; Seifert, R.; Ebbing, M.; Bønaa, K.H.; Mellgren, G.; Nilsen, D.W.T.; Nordrehaug, J.E.; et al. Elevated plasma dimethylglycine is a risk marker of mortality in patients with coronary heart disease. Eur. J. Prev. Cardiol. 2015, 22, 743–752. [Google Scholar] [CrossRef]
- Svingen, G.F.T.; Ueland, P.M.; Pedersen, E.K.R.; Schartum-Hansen, H.; Seifert, R.; Ebbing, M.; Løland, K.H.; Tell, G.S.; Nygård, O. Plasma dimethylglycine and risk of incident acute myocardial infarction in patients with stable angina pectoris. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 2041–2048. [Google Scholar] [CrossRef]
- Li, D.; Yu, X.M.; Xie, H.B.; Zhang, Y.H.; Wang, Q.; Zhou, X.Q.; Yu, P.; Wang, L.J. Platelet phospholipid n-3 PUFA negatively associated with plasma homocysteine in middle-aged and geriatric hyperlipaemia patients. Prostaglandins Leukot. Essent. Fatty Acids 2007, 76, 293–297. [Google Scholar] [CrossRef]
- Huang, T.; Wahlqvist, M.L.; Li, D. Effect of n-3 polyunsaturated fatty acid on gene expression of the critical enzymes involved in homocysteine metabolism. Nutr. J. 2012, 11, 6. [Google Scholar] [CrossRef]
- Green, P.S.; Mendez, A.J.; Jacob, J.S.; Crowley, J.R.; Growdon, W.; Hyman, B.T.; Heinecke, J.W. Neuronal expression of myeloperoxidase is increased in Alzheimer’s disease. J. Neurochem. 2004, 90, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Wahlqvist, M.L.; Li, D. Docosahexaenoic acid decreases plasma homocysteine via regulating enzyme activity and mRNA expression involved in methionine metabolism. Nutrition 2010, 26, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Żebrowska, A.; Mizia-Stec, K.; Mizia, M.; Gąsior, Z.; Poprzęcki, S. Omega-3 fatty acids supplementation improves endothelial function and maximal oxygen uptake in endurance-trained athletes. Eur. J. Sport Sci. 2015, 15, 305–314. [Google Scholar] [CrossRef]
- Zeisel, S. Choline, Other Methyl-Donors and Epigenetics. Nutrients 2017, 9, 445. [Google Scholar] [CrossRef]
- Pogribny, I.P.; James, S.J.; Beland, F.A. Molecular alterations in hepatocarcinogenesis induced by dietary methyl deficiency. Mol. Nutr. Food Res. 2012, 56, 116–125. [Google Scholar] [CrossRef]
- Loscalzo, J.; Handy, D.E. Epigenetic modifications: Basic mechanisms and role in cardiovascular disease (2013 Grover Conference series). Pulm Circ. 2014, 4, 169–174. [Google Scholar] [CrossRef]
- Perła-Kaján, J.; Jakubowski, H. Dysregulation of Epigenetic Mechanisms of Gene Expression in the Pathologies of Hyperhomocysteinemia. Int. J. Mol. Sci. 2019, 20. [Google Scholar]
- Codreanu, S.G.; Zhang, B.; Sobecki, S.M.; Billheimer, D.D.; Liebler, D.C. Global Analysis of Protein Damage by the Lipid Electrophile 4-Hydroxy-2-nonenal. Mol. Cell. Proteom. 2009, 8, 670–680. [Google Scholar] [CrossRef]
- Doyle, K.; Fitzpatrick, F.A. Redox signaling, alkylation (carbonylation) of conserved cysteines inactivates class I histone deacetylases 1, 2, and 3 and antagonizes their transcriptional repressor function. J. Biol. Chem. 2010, 285, 17417–17424. [Google Scholar] [CrossRef]
- Lee, H.-S.; Barraza-Villarreal, A.; Biessy, C.; Duarte-Salles, T.; Sly, P.D.; Ramakrishnan, U.; Rivera, J.; Herceg, Z.; Romieu, I. Dietary supplementation with polyunsaturated fatty acid during pregnancy modulates DNA methylation at IGF2/H19 imprinted genes and growth of infants. Physiol. Genom. 2014, 46, 851–857. [Google Scholar] [CrossRef]
- Resseguie, M.E.; da Costa, K.-A.; Galanko, J.A.; Patel, M.; Davis, I.J.; Zeisel, S.H. Aberrant Estrogen Regulation of PEMT Results in Choline Deficiency-associated Liver Dysfunction. J. Biol. Chem. 2011, 286, 1649–1658. [Google Scholar] [CrossRef]
| References | Participants | Trials | Patients | Daily Doses | Duration | Results | 95% CI | I2 |
|---|---|---|---|---|---|---|---|---|
| Huang et al. 2011 | T: 352; C: 355 | 11 | CHD; ESRD; HL; HT; MI; MS; PVD; T2D | 0.2–6 g | 6 Wk–12 Mo | MD: −1.59 µmol/L; p < 0.0001 | −2.34, −0,83 | 52%; p = 0.02 |
| Dawson et al. 2016 | T: 1134; C: 1121 T: 787; C: 786 | 13 8 | CVD; ESRD; HL; HT; MI; T2D CHD; CVD; HT; MS; PVD | 0.5–6 g 0.2–2 g | 2 Wk–12 Mo 3–12 Mo | MD: −1.09 µmol/L; p = 0.026 (B) MD: −1.37 µmol/L; p = 0.008 (C) | −2.04, −0.13 −2.38, −0.36 | 57.08%; p = 0.006 43.77%; p = 0,087 |
| Xu et al. 2016 | T: 182; C: 181 | 3 | ESRD | 1.7–6 g | 2–6 Mo | SMD: −0.46 µmol/L; p = 0.001 (Fixed) SMD: −1.63 µmol/L; p = 0.219 (Random) | −0.72, −0.20 −4.24, 0.97 | 98.3%; p < 0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rizzo, G.; Laganà, A.S. The Link between Homocysteine and Omega-3 Polyunsaturated Fatty Acid: Critical Appraisal and Future Directions. Biomolecules 2020, 10, 219. https://doi.org/10.3390/biom10020219
Rizzo G, Laganà AS. The Link between Homocysteine and Omega-3 Polyunsaturated Fatty Acid: Critical Appraisal and Future Directions. Biomolecules. 2020; 10(2):219. https://doi.org/10.3390/biom10020219
Chicago/Turabian StyleRizzo, Gianluca, and Antonio Simone Laganà. 2020. "The Link between Homocysteine and Omega-3 Polyunsaturated Fatty Acid: Critical Appraisal and Future Directions" Biomolecules 10, no. 2: 219. https://doi.org/10.3390/biom10020219
APA StyleRizzo, G., & Laganà, A. S. (2020). The Link between Homocysteine and Omega-3 Polyunsaturated Fatty Acid: Critical Appraisal and Future Directions. Biomolecules, 10(2), 219. https://doi.org/10.3390/biom10020219






