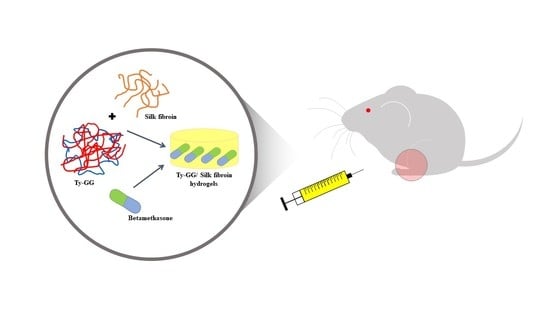
Anti-Inflammatory Properties of Injectable Betamethasone-Loaded Tyramine-Modified Gellan Gum/Silk Fibroin Hydrogels
Abstract
:1. Introduction
2. Materials and Methods
2.1. Physicochemical Characterization
2.1.1. Preparation of Silk Fibroin Solution
2.1.2. Horseradish Peroxidase to Induce Ty–GG/SF Hydrogel Formation
2.1.3. Analysis of the Secondary Protein Structure of Silk Fibroin
2.1.4. Water Uptake and Enzymatic Degradation of Ty–GG/SF Hydrogels
2.1.5. Gelation Time, Injectability and Rheological Properties of the Ty–GG/SF Hydrogels
2.1.6. Betamethasone Release Studies of the Ty–GG/SF Hydrogels
2.2. In Vitro Studies
2.2.1. Chondrogenic Cell Isolation
2.2.2. Cell Culture
2.2.3. Metabolic Activity
2.2.4. DNA Quantification
2.2.5. Fluorescence Microscopy
2.2.6. The Therapeutic Efficacy of Ty–GG/SF with Betamethasone Encapsulated
2.3. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Characterization
3.2. In Vitro Studies
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chancay, M.G.; Guendsechadze, S.N.; Blanco, I. Types of pain and their psychosocial impact in women with rheumatoid arthritis. Women’s Midlife Health 2019, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Sardar, S.; Andersson, Å. Old and new therapeutics for rheumatoid arthritis: In vivo models and drug development. Immunopharmacol. Immunotoxicol. 2016, 38, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Kesharwani, D.; Paliwal, R.; Satapathy, T.; Paul, S.D. Rheumatiod arthritis: An updated overview of latest therapy and drug delivery. J. Pharmacopuncture 2019, 22, 210. [Google Scholar]
- Yasir, M.; Sonthalia, S. Corticosteroid Adverse Effects; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Oliveira, I.M.; Gonçalves, C.; Reis, R.L.; Oliveira, J.M. Engineering nanoparticles for targeting rheumatoid arthritis: Past, present, and future trends. Nano Res. 2018, 11, 4489–4506. [Google Scholar] [CrossRef] [Green Version]
- Guo, Q.; Wang, Y.; Xu, D.; Nossent, J.; Pavlos, N.J.; Xu, J. Rheumatoid arthritis: Pathological mechanisms and modern pharmacologic therapies. Bone Res. 2018, 6, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H. Injectable hydrogels delivering therapeutic agents for disease treatment and tissue engineering. Biomater. Res. 2018, 22, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.-S.; Yang, J.; Kim, K.; Shin, U.S. Biodegradable and injectable hydrogels as an immunosuppressive drug delivery system. Mater. Sci. Eng. C 2019, 98, 472–481. [Google Scholar] [CrossRef]
- Liang, K.; Bae, K.H.; Kurisawa, M. Recent advances in the design of injectable hydrogels for stem cell-based therapy. J. Mater. Chem. B 2019, 7, 3775–3791. [Google Scholar] [CrossRef]
- Onaciu, A.; Munteanu, R.A.; Moldovan, A.I.; Moldovan, C.S.; Berindan-Neagoe, I. Hydrogels Based Drug Delivery Synthesis, Characterization and Administration. Pharmaceutics 2019, 11, 432. [Google Scholar] [CrossRef] [Green Version]
- Spicer, C.D. Hydrogel scaffolds for tissue engineering: The importance of polymer choice. Polym. Chem. 2020, 11, 184–219. [Google Scholar] [CrossRef]
- Seib, F.P. Reverse-engineered silk hydrogels for cell and drug delivery. Ther. Deliv. 2018, 9, 469–487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribeiro, V.P.; da Silva Morais, A.; Maia, F.R.; Canadas, R.F.; Costa, J.B.; Oliveira, A.L.; Oliveira, J.M.; Reis, R.L. Combinatory approach for developing silk fibroin scaffolds for cartilage regeneration. Acta Biomater. 2018, 72, 167–181. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, V.P.; Silva-Correia, J.; Gonçalves, C.; Pina, S.; Radhouani, H.; Montonen, T.; Hyttinen, J.; Roy, A.; Oliveira, A.L.; Reis, R.L. Rapidly responsive silk fibroin hydrogels as an artificial matrix for the programmed tumor cells death. PLoS ONE 2018, 13, e0194441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, Z.; Wu, F.; Xing, T.; Yadavalli, V.K.; Kundu, S.C.; Lu, S. A silk fibroin hydrogel with reversible sol–gel transition. RSC Adv. 2017, 7, 24085–24096. [Google Scholar] [CrossRef] [Green Version]
- Khanmohammadi, M.; Dastjerdi, M.B.; Ai, A.; Ahmadi, A.; Godarzi, A.; Rahimi, A.; Ai, J. Horseradish peroxidase-catalyzed hydrogelation for biomedical applications. Biomater. Sci. 2018, 6, 1286–1298. [Google Scholar] [CrossRef]
- Lee, F.; Bae, K.H.; Kurisawa, M. Injectable hydrogel systems crosslinked by horseradish peroxidase. Biomed. Mater. 2015, 11, 014101. [Google Scholar] [CrossRef]
- Liu, H.; Yuan, M.; Sonamuthu, J.; Yan, S.; Huang, W.; Cai, Y.; Yao, J. A dopamine-functionalized aqueous-based silk protein hydrogel bioadhesive for biomedical wound closure. New J. Chem. 2020, 44, 884–891. [Google Scholar] [CrossRef]
- Tan, H.; Wu, J.; Lao, L.; Gao, C. Gelatin/chitosan/hyaluronan scaffold integrated with PLGA microspheres for cartilage tissue engineering. Acta Biomater. 2009, 5, 328–337. [Google Scholar] [CrossRef]
- Zhu, L.; Luo, D.; Liu, Y. Effect of the nano/microscale structure of biomaterial scaffolds on bone regeneration. Int. J. Oral Sci. 2020, 12, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Milivojevic, M.; Pajic-Lijakovic, I.; Bugarski, B.; Nayak, A.K.; Hasnain, M.S. Chapter 6—Gellan gum in drug delivery applications. In Natural Polysaccharides in Drug Delivery and Biomedical Applications; Hasnain, M.S., Nayak, A.K., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 145–186. [Google Scholar] [CrossRef]
- Muthukumar, T.; Song, J.E.; Khang, G. Biological Role of Gellan Gum in Improving Scaffold Drug Delivery, Cell Adhesion Properties for Tissue Engineering Applications. Molecules 2019, 24, 4514. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, I.M.; Gonçalves, C.; Shin, M.E.; Lee, S.; Reis, R.L.; Khang, G.; Oliveira, J.M. Enzymatically crosslinked tyramine-gellan gum hydrogels as drug delivery system for rheumatoid arthritis treatment. Drug Deliv. Transl. Res. 2020, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Rockwood, D.N.; Preda, R.C.; Yücel, T.; Wang, X.; Lovett, M.L.; Kaplan, D.L. Materials fabrication from Bombyx mori silk fibroin. Nat. Protoc. 2011, 6, 1612. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Tripathy, N.; Shin, J.H.; Song, J.E.; Cha, J.G.; Min, K.D.; Park, C.H.; Khang, G. Enhanced osteogenesis of β-tricalcium phosphate reinforced silk fibroin scaffold for bone tissue biofabrication. Int. J. Biol. Macromol. 2017, 95, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Litvinov, R.I.; Faizullin, D.A.; Zuev, Y.F.; Weisel, J.W. The α-helix to β-sheet transition in stretched and compressed hydrated fibrin clots. Biophys. J. 2012, 103, 1020–1027. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wulandari, L.; Indrayanto, G. HPTLC determination of betamethasone in tablets and its validation. J. Liq. Chromatogr. Relat. Technol. 2003, 26, 2709–2717. [Google Scholar] [CrossRef]
- Manning, W.K.; Bonner, W.M., Jr. Isolation and culture of chondrocytes from human adult articular cartilage. Arthritis Rheum. 1967, 10, 235–239. [Google Scholar] [CrossRef]
- Jeon, H.Y.; Shin, E.Y.; Choi, J.H.; Song, J.E.; Reis, R.L.; Khang, G. Evaluation of saponin loaded gellan gum hydrogel scaffold for cartilage regeneration. Macromol. Res. 2018, 26, 724–729. [Google Scholar] [CrossRef] [Green Version]
- Yan, C.; Yang, B.; Yu, Z. Retracted Article: Determination of silk fibroin secondary structure by terahertz time domain spectroscopy. Anal. Methods 2013, 6, 248–252. [Google Scholar] [CrossRef]
- Johari, N.; Moroni, L.; Samadikuchaksaraei, A. Tuning the conformation and mechanical properties of silk fibroin hydrogels. Eur. Polym. J. 2020, 109842. [Google Scholar] [CrossRef]
- Wei, W.; Liu, J.; Peng, Z.; Liang, M.; Wang, Y.; Wang, X. Gellable silk fibroin-polyethylene sponge for hemostasis. Artif. Cells Nanomed. Biotechnol. 2020, 48, 28–36. [Google Scholar] [CrossRef]
- Taketani, I.; Nakayama, S.; Nagare, S.; Senna, M. The secondary structure control of silk fibroin thin films by post treatment. Appl. Surf. Sci. 2005, 244, 623–626. [Google Scholar] [CrossRef]
- Vedadghavami, A.; Minooei, F.; Mohammadi, M.H.; Khetani, S.; Kolahchi, A.R.; Mashayekhan, S.; Sanati-Nezhad, A. Manufacturing of hydrogel biomaterials with controlled mechanical properties for tissue engineering applications. Acta Biomater. 2017, 62, 42–63. [Google Scholar] [CrossRef]
- Huang, Y.; Yu, H.; Xiao, C. pH-sensitive cationic guar gum/poly (acrylic acid) polyelectrolyte hydrogels: Swelling and in vitro drug release. Carbohydr. Polym. 2007, 69, 774–783. [Google Scholar] [CrossRef]
- Xu, Y.; Li, Y.; Chen, Q.; Fu, L.; Tao, L.; Wei, Y. Injectable and self-healing chitosan hydrogel based on imine bonds: Design and therapeutic applications. Int. J. Mol. Sci. 2018, 19, 2198. [Google Scholar] [CrossRef] [Green Version]
- Mihaila, S.M.; Gaharwar, A.K.; Reis, R.L.; Marques, A.P.; Gomes, M.E.; Khademhosseini, A. Photocrosslinkable kappa-carrageenan hydrogels for tissue engineering applications. Adv. Heal. Mater. 2013, 2, 895–907. [Google Scholar] [CrossRef]
- da Silva, L.P.; Cerqueira, M.T.; Sousa, R.A.; Reis, R.L.; Correlo, V.M.; Marques, A.P. Engineering cell-adhesive gellan gum spongy-like hydrogels for regenerative medicine purposes. Acta Biomater. 2014, 10, 4787–4797. [Google Scholar] [CrossRef]
- Coutinho, D.F.; Sant, S.V.; Shin, H.; Oliveira, J.T.; Gomes, M.E.; Neves, N.M.; Khademhosseini, A.; Reis, R.L. Modified Gellan Gum hydrogels with tunable physical and mechanical properties. Biomaterials 2010, 31, 7494–7502. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, V.P.; Pina, S.; Costa, J.o.B.; Cengiz, I.F.; García-Fernández, L.; Fernández-Gutiérrez, M.d.M.; Paiva, O.C.; Oliveira, A.L.; San-Román, J.; Oliveira, J.M. Enzymatically cross-linked silk fibroin-based hierarchical scaffolds for osteochondral regeneration. ACS Appl. Mater. Interfaces 2019, 11, 3781–3799. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.; Hiemstra, C.; Zhong, Z.; Feijen, J. Enzyme-mediated fast in situ formation of hydrogels from dextran–tyramine conjugates. Biomaterials 2007, 28, 2791–2800. [Google Scholar] [CrossRef]
- Park, K.M.; Shin, Y.M.; Joung, Y.K.; Shin, H.; Park, K.D. In situ forming hydrogels based on tyramine conjugated 4-Arm-PPO-PEO via enzymatic oxidative reaction. Biomacromolecules 2010, 11, 706–712. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.W.; Choi, J.H.; Lee, Y.; Park, K.D. Horseradish peroxidase-catalysed in situ-forming hydrogels for tissue-engineering applications. J. Tissue Eng. Regen. Med. 2015, 9, 1225–1232. [Google Scholar] [CrossRef] [PubMed]
- Byju, A.G.; Kulkarni, A.; Gundiah, N. Mechanics of Gelatin and Elastin Based Hydrogels as Tissue Engineered Constructs. In Proceedings of the 13th International Conference on Fracture 2013, ICF 2013, Beijing, China, 16–21 June 2013; Volume 6, pp. 4406–4415. [Google Scholar]
- Yan, C.; Altunbas, A.; Yucel, T.; Nagarkar, R.P.; Schneider, J.P.; Pochan, D.J. Injectable solid hydrogel: Mechanism of shear-thinning and immediate recovery of injectable β-hairpin peptide hydrogels. Soft Matter 2010, 6, 5143–5156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Franck, A.; Germany, T.I. Viscoelasticity and Dynamic Mechanical Testing; TA Instruments: New Castle, DE, USA, 1993. [Google Scholar]
- Lawless, B.M.; Sadeghi, H.; Temple, D.K.; Dhaliwal, H.; Espino, D.M.; Hukins, D.W. Viscoelasticity of articular cartilage: Analysing the effect of induced stress and the restraint of bone in a dynamic environment. J. Mech. Behav. Biomed. Mater. 2017, 75, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, S.J.; Beckworth, W.J. Commonly Used Medications in Procedures. In Pain Procedures in Clinical Practice E-Book; Elsevier BV: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Firestein, G.S.; Budd, R.; Gabriel, S.E.; McInnes, I.B.; O’Dell, J.R. Kelley and Firestein’s Textbook of Rheumatology E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Narayanaswamy, R.; Torchilin, V.P. Hydrogels and their applications in targeted drug delivery. Molecules 2019, 24, 603. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Mooney, D.J. Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 2016, 1, 1–17. [Google Scholar] [CrossRef]
- Khang, G.; Lee, S.; Kim, H.; Silva-Correia, J.; Gomes, M.E.; Viegas, C.; Dias, I.R.; Oliveira, J.M.; Reis, R. Biological evaluation of intervertebral disc cells in different formulations of gellan gum-based hydrogels. J. Tissue Eng. Regen. Med. 2015, 9, 265–275. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.H.; Park, W.H. Chemically cross-linked silk fibroin hydrogel with enhanced elastic properties, biodegradability, and biocompatibility. Int. J. Nanomed. 2016, 11, 2967–2978. [Google Scholar] [CrossRef] [Green Version]
- Wenk, E.; Merkle, H.P.; Meinel, L. Silk fibroin as a vehicle for drug delivery applications. J. Control. Release 2011, 150, 128–141. [Google Scholar] [CrossRef]
- D’Arrigo, G.; Navarro, G.; Di Meo, C.; Matricardi, P.; Torchilin, V. Gellan gum nanohydrogel containing anti-inflammatory and anti-cancer drugs: A multi-drug delivery system for a combination therapy in cancer treatment. Eur. J. Pharm. Biopharm. 2014, 87, 208–216. [Google Scholar] [CrossRef]








| Ty–GG | SF | HRP | H2O2 | |
|---|---|---|---|---|
| C1 | 83.5 µL | 83.5 µL | 16.6 µL | 10.83 µL |
| C2 | 83.5 µL | 83.5 µL | 18.3 µL | 15 µL |
| C3 | 83.5 µL | 83.5 µL | 20 µL | 13.3 µL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, I.M.; Gonçalves, C.; Shin, M.E.; Lee, S.; Reis, R.L.; Khang, G.; Oliveira, J.M. Anti-Inflammatory Properties of Injectable Betamethasone-Loaded Tyramine-Modified Gellan Gum/Silk Fibroin Hydrogels. Biomolecules 2020, 10, 1456. https://doi.org/10.3390/biom10101456
Oliveira IM, Gonçalves C, Shin ME, Lee S, Reis RL, Khang G, Oliveira JM. Anti-Inflammatory Properties of Injectable Betamethasone-Loaded Tyramine-Modified Gellan Gum/Silk Fibroin Hydrogels. Biomolecules. 2020; 10(10):1456. https://doi.org/10.3390/biom10101456
Chicago/Turabian StyleOliveira, Isabel Matos, Cristiana Gonçalves, Myeong Eun Shin, Sumi Lee, Rui Luis Reis, Gilson Khang, and Joaquim Miguel Oliveira. 2020. "Anti-Inflammatory Properties of Injectable Betamethasone-Loaded Tyramine-Modified Gellan Gum/Silk Fibroin Hydrogels" Biomolecules 10, no. 10: 1456. https://doi.org/10.3390/biom10101456