Anticancer Plants: A Review of the Active Phytochemicals, Applications in Animal Models, and Regulatory Aspects
Abstract
1. Introduction
2. Sources and Methodology
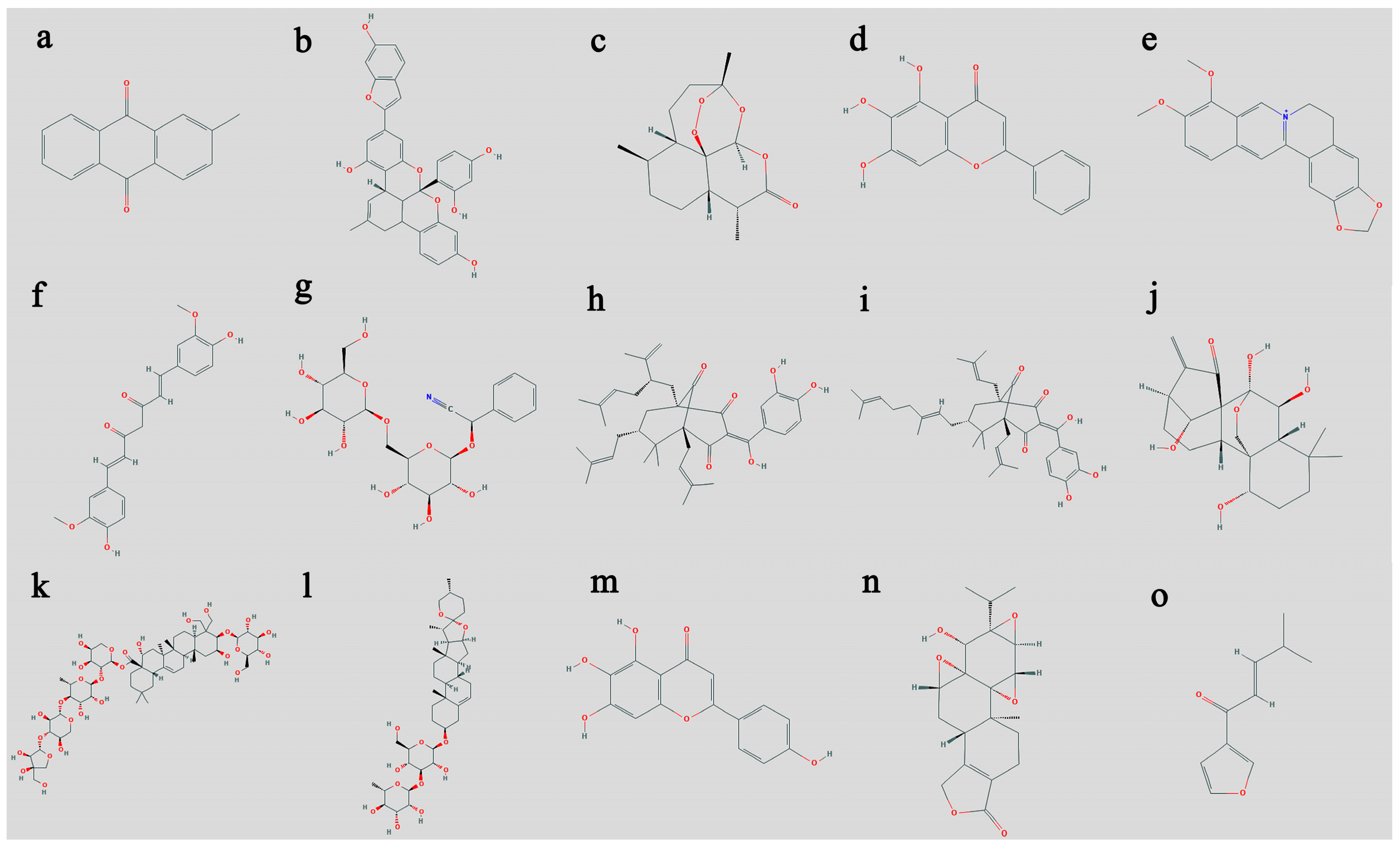
3. Selected Plants and Their Anticancer Activity
3.1. Artemisia annua
3.2. Coptis chinensis
3.3. Curcuma longa
3.4. Fagonia indica
3.5. Garcinia oblongifolia
3.6. Garcinia indica
3.7. Hedyotis diffusa
3.8. Loranthus parasiticus and Scurrulus parasitica
3.9. Morus alba
3.10. Paris polyphylla
3.11. Perilla frutescens
3.12. Platycodon grandiflorus
3.13. Prunus armeniaca
3.14. Rabdosiae rubescens
3.15. Scutellaria baicalensis
3.16. Scutellaria barbata
3.17. Tripterygium wilfordii
3.18. Tussilago farfara
3.19. Wedelia chinensis
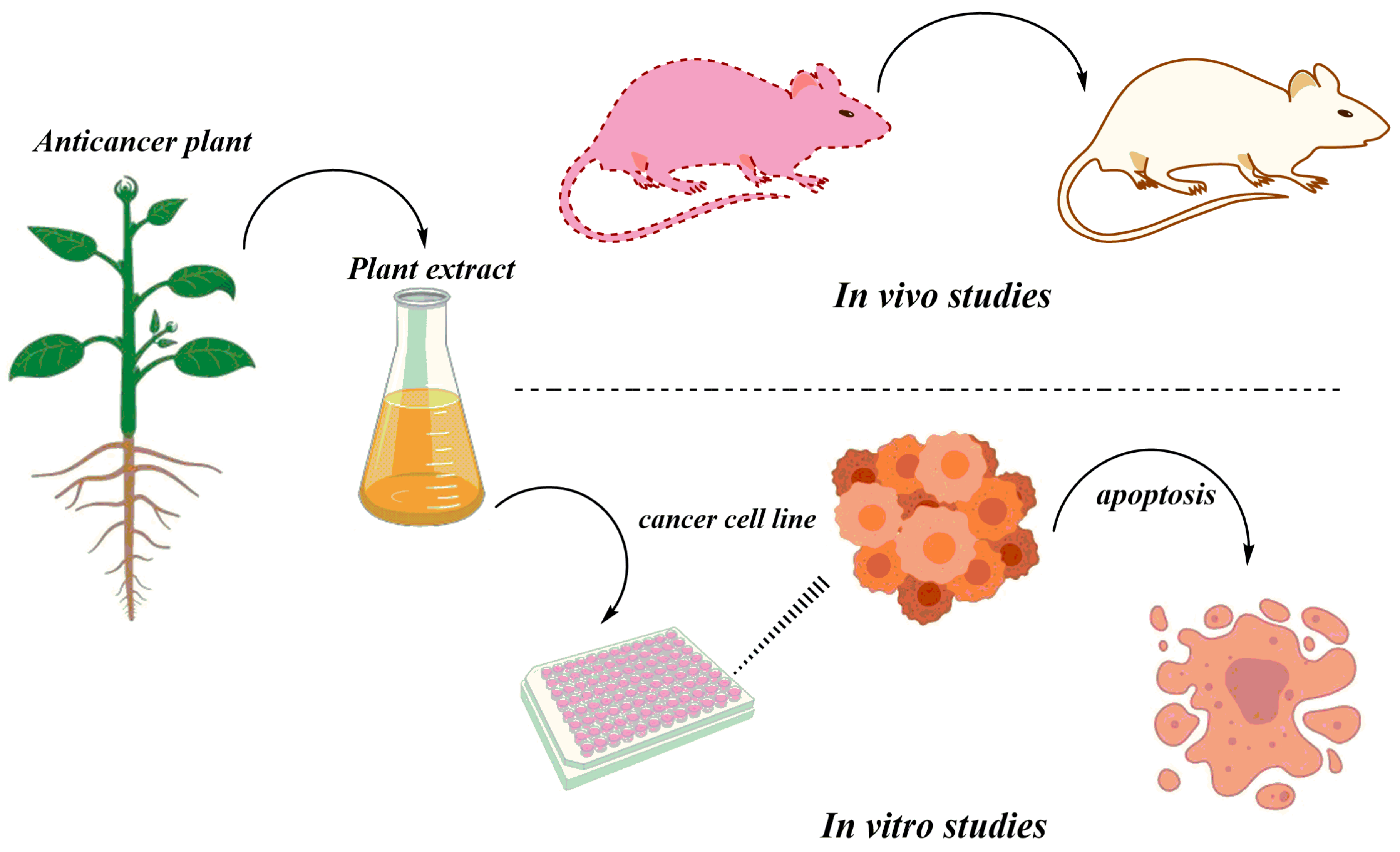
4. In Vivo Studies of Anticancer Herbal Medicine: An Overview
5. Regulatory Aspects of Herbal Anticancer Drugs
6. Modern Trends in Traditional Medicine Informatics and Opportunities for Anticancer Plant Products
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Karpuz, M.; Silindir-Gunay, M.; Ozer, A.Y. Current and Future Approaches for Effective Cancer Imaging and Treatment. Cancer Biother. Radiopharm. 2018, 33, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Nobili, S.; Lippi, D.; Witort, E.; Donnini, M.; Bausi, L.; Mini, E.; Capaccioli, S. Natural compounds for cancer treatment and prevention. Pharmacol. Res. 2009, 59, 365–378. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H. Advanced Textbook on Traditional Chinese Medicine and Pharmacology; New World Press: Beijing, China, 1995. [Google Scholar]
- Shehab, N.G.; Mahdy, A.; Khan, S.A.; Noureddin, S.M. Chemical constituents and biological activities of Fagonia indica Burm F. Res. J. Med. Plant. 2011, 5, 531–546. [Google Scholar] [CrossRef][Green Version]
- Faried, A.; Kurnia, D.; Faried, L.; Usman, N.; Miyazaki, T.; Kato, H.; Kuwano, H. Anticancer effects of gallic acid isolated from Indonesian herbal medicine, Phaleria macrocarpa (Scheff.) Boerl, on human cancer cell lines. Int. J. Oncol. 2007, 30, 605–613. [Google Scholar] [CrossRef]
- Sohi, K.K.; Mittal, N.; Hundal, M.K.; Khanduja, K.L. Gallic acid, an antioxidant, exhibits antiapoptotic potential in normal human lymphocytes: A Bcl-2 independent mechanism. J. Nutr. Sci. Vitaminol. (Tokyo) 2003, 49, 221–227. [Google Scholar] [CrossRef]
- Inoue, M.; Suzuki, R.; Koide, T.; Sakaguchi, N.; Ogihara, Y.; Yabu, Y. Antioxidant, gallic acid, induces apoptosis in HL-60RG cells. Biochem. Biophys. Res. Commun. 1994, 204, 898–904. [Google Scholar] [CrossRef]
- Sun, J.; Chu, Y.-F.; Wu, X.; Liu, R.H. Antioxidant and antiproliferative activities of common fruits. J. Agric. Food Chem. 2002, 50, 7449–7454. [Google Scholar] [CrossRef]
- Taraphdar, A.K.; Roy, M.; Bhattacharya, R. Natural products as inducers of apoptosis: Implication for cancer therapy and prevention. Curr. Sci. 2001, 1387–1396. [Google Scholar]
- Pal, S.K.; Shukla, Y. Herbal medicine: Current status and the future. Asian Pac. J. Cancer Prev. 2003, 4, 281–288. [Google Scholar]
- Koehn, F.E.; Carter, G.T. The evolving role of natural products in drug discovery. Nat. Rev. Drug Discov. 2005, 4, 206–220. [Google Scholar] [CrossRef] [PubMed]
- Saklani, A.; Kutty, S.K. Plant-derived compounds in clinical trials. Drug Discov. Today 2008, 13, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-Z.; Calway, T.; Yuan, C.-S. Herbal medicines as adjuvants for cancer therapeutics. Am. J. Chin. Med. 2012, 40, 657–669. [Google Scholar] [CrossRef] [PubMed]
- Fattovich, G.; Stroffolini, T.; Zagni, I.; Donato, F. Hepatocellular carcinoma in cirrhosis: Incidence and risk factors. Gastroenterology 2004, 127, S35–S50. [Google Scholar] [CrossRef]
- Llovet, J.M. Updated treatment approach to hepatocellular carcinoma. J. Gastroenterol. 2005, 40, 225–235. [Google Scholar] [CrossRef]
- Ruan, W.-J.; Lai, M.-D.; Zhou, J.-G. Anticancer effects of Chinese herbal medicine, science or myth? J. Zhejiang Univ. Sci. B 2006, 7, 1006–1014. [Google Scholar] [CrossRef]
- Trease, G.; Evans, W. Textbook of Pharmacognosy; Balliere: London, UK, 1983; pp. 57–59. [Google Scholar]
- Ghorani-Azam, A.; Sepahi, S.; Riahi-Zanjani, B.; Ghamsari, A.A.; Mohajeri, S.A.; Balali-Mood, M. Plant toxins and acute medicinal plant poisoning in children: A systematic literature review. J. Res. Med. Sci. 2018, 23, 26. [Google Scholar]
- Abad, M.J.; Bedoya, L.M.; Bermejo, P. Essential Oils from the Asteraceae Family Active against Multidrug-Resistant Bacteria. In Fighting Multidrug Resistance with Herbal Extracts, Essential Oils and Their Components; Rai, M.K., Kon, K.V., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 205–221. [Google Scholar]
- Tan, R.X.; Zheng, W.; Tang, H. Biologically active substances from the genus Artemisia. Planta Med. 1998, 64, 295–302. [Google Scholar] [CrossRef]
- Lu, H.; Zou, W.X.; Meng, J.C.; Hu, J.; Tan, R.X. New bioactive metabolites produced by Colletotrichum sp., an endophytic fungus in Artemisia annua. Plant. Sci. 2000, 151, 67–73. [Google Scholar] [CrossRef]
- Efferth, T. Mechanistic perspectives for 1, 2, 4-trioxanes in anti-cancer therapy. Drug Resist. Updat. 2005, 8, 85–97. [Google Scholar] [CrossRef]
- Ryu, J.-H.; Lee, S.-J.; Kim, M.-J.; Shin, J.-H.; Kang, S.-K.; Cho, K.-M.; Sung, N.-J. Antioxidant and anticancer activities of Artemisia annua L. and determination of functional compounds. J. Korean Soc. Food Sci. Nutr. 2011, 40, 509–516. [Google Scholar] [CrossRef]
- Xie, W.; Gu, D.; Li, J.; Cui, K.; Zhang, Y. Effects and action mechanisms of berberine and Rhizoma coptidis on gut microbes and obesity in high-fat diet-fed C57BL/6J mice. PLoS ONE 2011, 6, e24520. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Zhang, H.; Ye, J. Traditional Chinese medicine in treatment of metabolic syndrome. Endocr. Metab. Immune Disord. Drug Targets 2008, 8, 99–111. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Davies, G.E. Berberine inhibits adipogenesis in high-fat diet-induced obesity mice. Fitoterapia 2010, 81, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Feng, Y.; Tsao, S.; Wang, N.; Curtain, R.; Wang, Y. Berberine and Coptidis rhizoma as novel antineoplastic agents: A review of traditional use and biomedical investigations. J. Ethnopharmacol. 2009, 126, 5–17. [Google Scholar] [CrossRef]
- Ammon, H.P.; Wahl, M.A. Pharmacology of Curcuma longa. Planta Med. 1991, 57, 1–7. [Google Scholar] [CrossRef]
- Liu, F.; Ng, T. Antioxidative and free radical scavenging activities of selected medicinal herbs. Life Sci. 2000, 66, 725–735. [Google Scholar] [CrossRef]
- Schinella, G.; Tournier, H.; Prieto, J.; De Buschiazzo, P.M.; Rıos, J. Antioxidant activity of anti-inflammatory plant extracts. Life Sci. 2002, 70, 1023–1033. [Google Scholar] [CrossRef]
- Goel, A.; Boland, C.R.; Chauhan, D.P. Specific inhibition of cyclooxygenase-2 (COX-2) expression by dietary curcumin in HT-29 human colon cancer cells. Cancer Lett. 2001, 172, 111–118. [Google Scholar] [CrossRef]
- Wang, X.; Hang, Y.; Liu, J.; Hou, Y.; Wang, N.; Wang, M. Anticancer effect of curcumin inhibits cell growth through miR-21/PTEN/Akt pathway in breast cancer cell. Oncol. Lett. 2017, 13, 4825–4831. [Google Scholar] [CrossRef]
- Beier, B.A. A revision of the desert shrub Fagonia (Zygophyllaceae). Syst. Biodivers. 2005, 3, 221–263. [Google Scholar] [CrossRef]
- Akhtar, N.; Begum, S. Ethnopharmacological important plants of Jalala, district Mardan, Pakistan. Pak. J. Pl. Sci. 2009, 15, 95–100. [Google Scholar]
- Sharrma, S.; Gupta, V.; Sharma, G. Phytopharmacology of Fagonia indica (L): A review. J. Nat. Cons. 2010, 1, 143–147. [Google Scholar]
- Ibrahim, L.F.; Kawashty, S.A.; El-Hagrassy, A.M.; Nassar, M.I.; Mabry, T.J. A new kaempferol triglycoside from Fagonia taeckholmiana: Cytotoxic activity of its extracts. Carbohydr. Res. 2008, 343, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Sharawy, S.M.; Alshammari, A.M. Checklist of poisonous plants and animals in Aja Mountain, Ha’il Region, Saudi Arabia. Aust. J. Basic Appl. Sci. 2009, 3, 2217–2225. [Google Scholar]
- Shaker, K.H.; Bernhardt, M.; Elgamal, M.H.A.; Seifert, K. Triterpenoid saponins from Fagonia indica. Phytochemistry 1999, 51, 1049–1053. [Google Scholar] [CrossRef]
- Perrone, A.; Masullo, M.; Bassarello, C.; Hamed, A.I.; Belisario, M.A.; Pizza, C.; Piacente, S. Sulfated triterpene derivatives from Fagonia arabica. J. Nat. Prod. 2007, 70, 584–588. [Google Scholar] [CrossRef]
- Bagban, I.; Roy, S.; Chaudhary, A.; Das, S.; Gohil, K.; Bhandari, K. Hepatoprotective activity of the methanolic extract of Fagonia indica Burm in carbon tetra chloride induced hepatotoxicity in albino rats. Asian Pac. J. Trop. Biomed. 2012, 2, S1457–S1460. [Google Scholar] [CrossRef]
- Eman, A.A. Morphological, phytochemical and biological screening on three Egyptian species of Fagonia. Acad Arena 2011, 3, 18–27. [Google Scholar]
- Waheed, A.; Barker, J.; Barton, S.J.; Owen, C.P.; Ahmed, S.; Carew, M.A. A novel steroidal saponin glycoside from Fagonia indica induces cell-selective apoptosis or necrosis in cancer cells. Eur. J. Pharm. Sci. 2012, 47, 464–473. [Google Scholar] [CrossRef]
- Lam, M.; Carmichael, A.R.; Griffiths, H.R. An aqueous extract of Fagonia cretica induces DNA damage, cell cycle arrest and apoptosis in breast cancer cells via FOXO3a and p53 expression. PLoS ONE 2012, 7, e40152. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-B.; Long, C.; Kennelly, E.J. Structural diversity and bioactivities of natural benzophenones. Nat. Prod. Rep. 2014, 31, 1158–1174. [Google Scholar] [CrossRef] [PubMed]
- Hemshekhar, M.; Sunitha, K.; Santhosh, M.S.; Devaraja, S.; Kemparaju, K.; Vishwanath, B.; Niranjana, S.; Girish, K. An overview on genus Garcinia: Phytochemical and therapeutical aspects. Phytochem. Rev. 2011, 10, 325–351. [Google Scholar] [CrossRef]
- Huang, S.-X.; Feng, C.; Zhou, Y.; Xu, G.; Han, Q.-B.; Qiao, C.-F.; Chang, D.C.; Luo, K.Q.; Xu, H.-X. Bioassay-guided isolation of xanthones and polycyclic prenylated acylphloroglucinols from Garcinia oblongifolia. J. Nat. Prod. 2008, 72, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.; Huang, S.-X.; Gao, X.-M.; Xu, H.-X.; Luo, K.Q. Characterization of Proapoptotic Compounds from the Bark of Garcinia oblongifolia. J. Nat. Prod. 2014, 77, 1111–1116. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; AnandhiSenthilkumar, H.; Wu, S.-B.; Liu, B.; Guo, Z.-Y.; Fata, J.E.; Kennelly, E.J.; Long, C.-L. Comparative UPLC-QTOF-MS-based metabolomics and bioactivities analyses of Garcinia oblongifolia. J. Chromatogr. B 2016, 1011, 179–195. [Google Scholar] [CrossRef]
- Hong, J.; Kwon, S.J.; Sang, S.; Ju, J.; Zhou, J.-N.; Ho, C.-T.; Huang, M.-T.; Yang, C.S. Effects of garcinol and its derivatives on intestinal cell growth: Inhibitory effects and autoxidation-dependent growth-stimulatory effects. Free Radic. Biol. Med. 2007, 42, 1211–1221. [Google Scholar] [CrossRef]
- Liao, C.H.; Sang, S.; Ho, C.T.; Lin, J.K. Garcinol modulates tyrosine phosphorylation of FAK and subsequently induces apoptosis through down-regulation of Src, ERK, and Akt survival signaling in human colon cancer cells. J. Cell. Biochem. 2005, 96, 155–169. [Google Scholar] [CrossRef]
- Pan, M.-H.; Chang, W.-L.; Lin-Shiau, S.-Y.; Ho, C.-T.; Lin, J.-K. Induction of apoptosis by garcinol and curcumin through cytochrome c release and activation of caspases in human leukemia HL-60 cells. J. Agric. Food Chem. 2001, 49, 1464–1474. [Google Scholar] [CrossRef]
- Lin, C.-C.; Ng, L.-T.; Yang, J.-J.; Hsu, Y.-F. Anti-inflammatory and hepatoprotective activity of peh-hue-juwa-chi-cao in male rats. Am. J. Chin. Med. 2002, 30, 225–234. [Google Scholar] [CrossRef]
- Ahmad, R.; Ali, A.M.; Israf, D.A.; Ismail, N.H.; Shaari, K.; Lajis, N.H. Antioxidant, radical-scavenging, anti-inflammatory, cytotoxic and antibacterial activities of methanolic extracts of some Hedyotis species. Life Sci. 2005, 76, 1953–1964. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Zhang, Y.; Chen, M.; Zheng, H.; Zhang, K. The active component of Hedyotis diffusa Willd. Chin. Tradit. Plant. Med. 2004, 26, 577–579. [Google Scholar]
- Ahmad, R.; Shaari, K.; Lajis, N.H.; Hamzah, A.S.; Ismail, N.H.; Kitajima, M. Anthraquinones from Hedyotis capitellata. Phytochemistry 2005, 66, 1141–1147. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Xue, X.; Zhou, D.; Zhang, F.; Xu, Q.; Ren, L.; Liang, X. Analysis of iridoid glucosides in Hedyotis diffusa by high-performance liquid chromatography/electrospray ionization tandem mass spectrometry. J. Pharm. Biomed. Anal. 2008, 48, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Liu, M.; Liu, M.; Li, J. Methylanthraquinone from Hedyotis diffusa WILLD induces Ca2+-mediated apoptosis in human breast cancer cells. Toxicol. Vitr. 2010, 24, 142–147. [Google Scholar] [CrossRef]
- Wu, Z.; Jin, C.; Li, J.; Chen, X.; Yao, Q.; Zhu, Q. Inhibition of Colon Cancer Cells by Ethanol Extract of Oldenlandia diffusa. J. Kunming Med. Univ. 2013, 10, 31–34. [Google Scholar]
- Vidal-Russell, R.; Nickrent, D.L. Evolutionary relationships in the showy mistletoe family (Loranthaceae). Am. J. Bot. 2008, 95, 1015–1029. [Google Scholar] [CrossRef]
- Fukunaga, T.; Kajikawa, I.; Nishiya, K.; Takeya, K.; Itokawa, H. Studies on the constituents of the Japanese mistletoe, Viscum album L. var. coloratum Ohwi grown on different host trees. Chem. Pharm. Bull. 1989, 37, 1300–1303. [Google Scholar] [CrossRef]
- Osadebe, P.; Okide, G.; Akabogu, I. Study on anti-diabetic activities of crude methanolic extracts of Loranthus micranthus (Linn.) sourced from five different host trees. J. Ethnopharmacol. 2004, 95, 133–138. [Google Scholar] [CrossRef]
- Qin, Y.; Mo, T.; Liu, X.; Pan, Z.; Ye, M. Effects of Loranthus parasiticus on Nutrition Metabolism of Osmanthus fragrans and Cinnamomum burmannii. Acta Agric. Boreali-Occident. Sin. 2010, 4, 34. [Google Scholar]
- Powell, C.B.; Fung, P.; Jackson, J.; Dall’Era, J.; Lewkowicz, D.; Cohen, I.; Smith-McCune, K. Aqueous extract of herba Scutellaria barbatae, a Chinese herb used for ovarian cancer, induces apoptosis of ovarian cancer cell lines. Gynecol. Oncol. 2003, 91, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.-J.; Chen, Y.-Z.; Chen, B.-H.; Chen, J.-H.; Lin, Z.-X.; Fan, Y.-L. Study on cytotoxic activities on human leukemia cell line HL-60 by flavonoids extracts of Scurrula parasitica from four different host trees. China J. Chin. Mater. Med. 2008, 33, 427–432. [Google Scholar]
- Xiao, Y.; Fan, Y.; Chen, B.; Zhang, Q.; Zeng, H. Polysaccharides from Scurrula parasitica L. inhibit sarcoma S180 growth in mice. China J. Chin. Mater. Med. 2010, 35, 381–384. [Google Scholar]
- Devi, B.; Sharma, N.; Kumar, D.; Jeet, K. Morus alba Linn: A phytopharmacological review. Int. J. Pharm. Pharm. Sci. 2013, 5, 14–18. [Google Scholar]
- Chon, S.-U.; Kim, Y.-M.; Park, Y.-J.; Heo, B.-G.; Park, Y.-S.; Gorinstein, S. Antioxidant and antiproliferative effects of methanol extracts from raw and fermented parts of mulberry plant (Morus alba L.). Eur. Food Res. Technol. 2009, 230, 231–237. [Google Scholar] [CrossRef]
- Kikuchi, T.; Nihei, M.; Nagai, H.; Fukushi, H.; Tabata, K.; Suzuki, T.; Akihisa, T. Albanol A from the root bark of Morus alba L. induces apoptotic cell death in HL60 human leukemia cell line. Chem. Pharm. Bull. 2010, 58, 568–571. [Google Scholar] [CrossRef]
- Naowaratwattana, W.; De-Eknamkul, W.; De Mejia, E.G. Phenolic-containing organic extracts of mulberry (Morus alba L.) leaves inhibit HepG2 hepatoma cells through G2/M phase arrest, induction of apoptosis, and inhibition of topoisomerase IIα activity. J. Med. Food 2010, 13, 1045–1056. [Google Scholar] [CrossRef]
- Deepa, M.; Priya, S. Purification and characterization of a novel anti-proliferative lectin from Morus alba L. leaves. Protein Pept. Lett. 2012, 19, 839–845. [Google Scholar] [CrossRef]
- Fathy, S.A.; Singab, A.N.B.; Agwa, S.A.; El Hamid, D.M.A.; Zahra, F.A.; El Moneim, S.M.A. The antiproliferative effect of mulberry (Morus alba L.) plant on hepatocarcinoma cell line HepG2. Egypt. J. Med. Hum. Genet. 2013, 14, 375–382. [Google Scholar] [CrossRef]
- Qin, J.; Fan, M.; He, J.; Wu, X.-D.; Peng, L.-Y.; Su, J.; Cheng, X.; Li, Y.; Kong, L.-M.; Li, R.-T. New cytotoxic and anti-inflammatory compounds isolated from Morus alba L. Nat. Prod. Res. 2015, 29, 1711–1718. [Google Scholar] [CrossRef]
- Huang, X.; Gao, W.; Man, S.; Zhao, Z. Advances in studies on saponins in plants of Paris L. and their biosynthetic approach. Chin. Tradit. Herb. Drugs 1994. [Google Scholar]
- Negi, J.S.; Bisht, V.K.; Bhandari, A.K.; Bhatt, V.P.; Singh, P.; Singh, N. Paris polyphylla: Chemical and biological prospectives. Anti-Cancer Agents Med. Chem. 2014, 14, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-F.; Cui, Y.; Huang, J.-J.; Zhang, Y.-Z.; Nie, Z.; Wang, L.-F.; Yan, B.-Z.; Tang, Y.-L.; Liu, Y. Immuno-stimulating properties of diosgenyl saponins isolated from Paris polyphylla. Bioorg. Med. Chem. Lett. 2007, 17, 2408–2413. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Liu, H.; Liu, X.-T.; Chen, X.-Q.; Wang, Q. Steroidal saponins from the rhizomes of Paris delavayi. Steroids 2009, 74, 809–813. [Google Scholar] [CrossRef] [PubMed]
- Deng, D.; Lauren, D.R.; Cooney, J.M.; Jensen, D.J.; Wurms, K.V.; Upritchard, J.E.; Cannon, R.D.; Wang, M.Z.; Li, M.Z. Antifungal saponins from Paris polyphylla Smith. Planta Med. 2008, 74, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Kang, L.-P.; Liu, Y.-X.; Liang, Y.-G.; Tan, D.-W.; Yu, Z.-Y.; Cong, Y.-W.; Ma, B.-P. Steroidal saponins from the rhizome of Paris polyphylla and their cytotoxic activities. Planta Med. 2009, 75, 356. [Google Scholar] [CrossRef]
- Fu, Y.L.; Yu, Z.Y.; Tang, X.M.; Zhao, Y.; Yuan, X.L.; Wang, S.; Ma, B.P.; Cong, Y.W. Pennogenin glycosides with a spirostanol structure are strong platelet agonists: Structural requirement for activity and mode of platelet agonist synergism. J. Thromb. Haemost. 2008, 6, 524–533. [Google Scholar] [CrossRef]
- Guo, L.; Su, J.; Deng, B.; Yu, Z.; Kang, L.; Zhao, Z.; Shan, Y.; Chen, J.; Ma, B.; Cong, Y. Active pharmaceutical ingredients and mechanisms underlying phasic myometrial contractions stimulated with the saponin extract from Paris polyphylla Sm. var. yunnanensis used for abnormal uterine bleeding. Hum. Reprod. 2008, 23, 964–971. [Google Scholar] [CrossRef]
- Sun, J.; Liu, B.R.; Hu, W.J.; Yu, L.X.; Qian, X.P. In vitro anticancer activity of aqueous extracts and ethanol extracts of fifteen taditional Chinese medicines on human digestive tumor cell lines. Phytother. Res. 2007, 21, 1102–1104. [Google Scholar] [CrossRef]
- Li, F.-R.; Jiao, P.; Yao, S.-T.; Sang, H.; Qin, S.-C.; Zhang, W.; Zhang, Y.-B.; Gao, L.-L. Paris polyphylla Smith extract induces apoptosis and activates cancer suppressor gene connexin26 expression. Asian Pac. J. Cancer Prev. 2012, 13, 205–209. [Google Scholar] [CrossRef]
- Heci, Y. Valuable ingredients from herb perilla: A mini review. Innov. Food Technol. 2001, 29, 32–33. [Google Scholar]
- Asif, M.; Kumar, A. Nutritional and functional characterization of Perilla frutescens seed oil and evaluation of its effect on gastrointestinal motility. Malay. J. Pharm. Sci. 2010, 8, 1–12. [Google Scholar]
- Lee, J.K.; Ohnishi, O. Geographic differentiation of morphological characters among Perilla crops and their weedy types in East Asia. Breed. Sci. 2001, 51, 247–255. [Google Scholar] [CrossRef]
- Lee, J.K.; Ohnishi, O. Genetic relationships among cultivated types of Perilla frutescens and their weedy types in East Asia revealed by AFLP markers. Genet. Resour. Crop. Evol. 2003, 50, 65–74. [Google Scholar] [CrossRef]
- Mao, Q.-Q.; Huang, Z.; Zhong, X.-M.; Feng, C.-R.; Pan, A.-J.; Li, Z.-Y.; Ip, S.-P.; Che, C.-T. Effects of SYJN, a Chinese herbal formula, on chronic unpredictable stress-induced changes in behavior and brain BDNF in rats. J. Ethnopharmacol. 2010, 128, 336–341. [Google Scholar] [CrossRef]
- Lin, C.-S.; Kuo, C.-L.; Wang, J.-P.; Cheng, J.-S.; Huang, Z.-W.; Chen, C.-F. Growth inhibitory and apoptosis inducing effect of Perilla frutescens extract on human hepatoma HepG2 cells. J. Ethnopharmacol. 2007, 112, 557–567. [Google Scholar] [CrossRef]
- Osakabe, N.; Yasuda, A.; Natsume, M.; Yoshikawa, T. Rosmarinic acid inhibits epidermal inflammatory responses: Anticarcinogenic effect of Perilla frutescens extract in the murine two-stage skin model. Carcinogenesis 2004, 25, 549–557. [Google Scholar] [CrossRef]
- Kwak, C.S.; Yeo, E.J.; Moon, S.C.; Kim, Y.W.; Ahn, H.J.; Park, S.C. Perilla leaf, Perilla frutescens, induces apoptosis and G1 phase arrest in human leukemia HL-60 cells through the combinations of death receptor-mediated, mitochondrial, and endoplasmic reticulum stress-induced pathways. J. Med. Food 2009, 12, 508–517. [Google Scholar] [CrossRef]
- Cho, B.O.; Jin, C.H.; Park, Y.D.; Ryu, H.W.; Byun, M.W.; Seo, K.I.; Jeong, I.Y. Isoegomaketone induces apoptosis through caspase-dependent and caspase-independent pathways in human DLD1 cells. Biosci. Biotechnol. Biochem. 2011, 75, 1306–1311. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Y.; Yang, D.; Zhang, C.; Zhang, N.; Li, M.; Liu, Y. Platycodon grandiflorus—An ethnopharmacological, phytochemical and pharmacological review. J. Ethnopharmacol. 2015, 164, 147–161. [Google Scholar] [CrossRef]
- Han, S.B.; Park, S.H.; Lee, K.H.; Lee, C.W.; Lee, S.H.; Kim, H.C.; Kim, Y.S.; Lee, H.S.; Kim, H.M. Polysaccharide isolated from the radix of Platycodon grandiflorum selectively activates B cells and macrophages but not T cells. Int. Immunopharmacol. 2001, 1, 1969–1978. [Google Scholar] [CrossRef]
- Xie, Y.; Pan, H.; Sun, H.; Li, D. A promising balanced Th1 and Th2 directing immunological adjuvant, saponins from the root of Platycodon grandiflorum. Vaccine 2008, 26, 3937–3945. [Google Scholar] [CrossRef] [PubMed]
- Khanal, T.; Choi, J.H.; Hwang, Y.P.; Chung, Y.C.; Jeong, H.G. Saponins isolated from the root of Platycodon grandiflorum protect against acute ethanol-induced hepatotoxicity in mice. Food Chem. Toxicol. 2009, 47, 530–535. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.J.; Hwang, S.J.; Choi, J.H.; Jeong, H.G. Saponins derived from the roots of Platycodon grandiflorum inhibit HT-1080 cell invasion and MMPs activities: Regulation of NF-κB activation via ROS signal pathway. Cancer Lett. 2008, 268, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.S.; Kim, A.K. Platycodin D induces apoptosis in MCF-7 human breast cancer cells. J. Med. Food 2010, 13, 298–305. [Google Scholar] [CrossRef]
- Kim, M.-O.; Moon, D.-O.; Choi, Y.H.; Shin, D.Y.; Kang, H.S.; Choi, B.T.; Lee, J.-D.; Li, W.; Kim, G.-Y. Platycodin D induces apoptosis and decreases telomerase activity in human leukemia cells. Cancer Lett. 2008, 261, 98–107. [Google Scholar] [CrossRef]
- Shin, D.Y.; Kim, G.Y.; Li, W.; Choi, B.T.; Kim, N.D.; Kang, H.S.; Choi, Y.H. Implication of intracellular ROS formation, caspase-3 activation and Egr-1 induction in platycodon D-induced apoptosis of U937 human leukemia cells. Biomed. Pharmacother. 2009, 63, 86–94. [Google Scholar] [CrossRef]
- Hu, Q.; Pan, R.; Wang, L.; Peng, B.; Tang, J.; Liu, X. Platycodon grandiflorum induces apoptosis in SKOV3 human ovarian cancer cells through mitochondrial-dependent pathway. Am. J. Chin. Med. 2010, 38, 373–386. [Google Scholar] [CrossRef]
- Yiğit, D.; Yiğit, N.; Mavi, A. Antioxidant and antimicrobial activities of bitter and sweet apricot (Prunus armeniaca L.) kernels. Braz. J. Med. Biol. Res. 2009, 42, 346–352. [Google Scholar] [CrossRef]
- Hacıseferoğulları, H.; Gezer, I.; Özcan, M.M.; MuratAsma, B. Post-harvest chemical and physical–mechanical properties of some apricot varieties cultivated in Turkey. J. Food Eng. 2007, 79, 364–373. [Google Scholar] [CrossRef]
- Yan, J.; Tong, S.; Li, J.; Lou, J. Preparative isolation and purification of amygdalin from Prunus armeniaca L. with high recovery by high-speed countercurrent chromatography. J. Liq. Chromatogr. Relat. Technol. 2006, 29, 1271–1279. [Google Scholar] [CrossRef]
- Akcicek, E.; Otles, S.; Esiyok, D. Cancer and its prevention by some horticultural and field crops in Turkey. Asian Pac. J. Cancer Prev 2005, 6, 224–230. [Google Scholar] [PubMed]
- Gomaa, E.Z. In vitro antioxidant, antimicrobial, and antitumor activities of bitter almond and sweet apricot (Prunus armeniaca L.) kernels. Food Sci. Biotechnol. 2013, 22, 455–463. [Google Scholar] [CrossRef]
- Madrau, M.A.; Piscopo, A.; Sanguinetti, A.M.; Del Caro, A.; Poiana, M.; Romeo, F.V.; Piga, A. Effect of drying temperature on polyphenolic content and antioxidant activity of apricots. Eur. Food Res. Technol. 2009, 228, 441. [Google Scholar] [CrossRef]
- Liu, H.; Chen, F.; Yang, H.; Yao, Y.; Gong, X.; Xin, Y.; Ding, C. Effect of calcium treatment on nanostructure of chelate-soluble pectin and physicochemical and textural properties of apricot fruits. Food Res. Int. 2009, 42, 1131–1140. [Google Scholar] [CrossRef]
- Vardi, N.; Parlakpinar, H.; Ozturk, F.; Ates, B.; Gul, M.; Cetin, A.; Erdogan, A.; Otlu, A. Potent protective effect of apricot and β-carotene on methotrexate-induced intestinal oxidative damage in rats. Food Chem. Toxicol. 2008, 46, 3015–3022. [Google Scholar] [CrossRef]
- Sun, H.-D.; Huang, S.-X.; Han, Q.-B. Diterpenoids from Isodon species and their biological activities. Nat. Prod. Rep. 2006, 23, 673–698. [Google Scholar] [CrossRef]
- Liu, H.-M.; Yan, X.; Kiuchi, F.; Liu, Z. A new diterpene glycoside from Rabdosia rubescens. Chem. Pharm. Bull. 2000, 48, 148–149. [Google Scholar] [CrossRef]
- Zhang, Y.; Liang, Y.; He, C. Anticancer activities and mechanisms of heat-clearing and detoxicating traditional Chinese herbal medicine. Chin. Med. 2017, 12, 20. [Google Scholar] [CrossRef]
- Bao, R.; Shu, Y.; Wu, X.; Weng, H.; Ding, Q.; Cao, Y.; Li, M.; Mu, J.; Wu, W.; Ding, Q. Oridonin induces apoptosis and cell cycle arrest of gallbladder cancer cells via the mitochondrial pathway. BMC Cancer 2014, 14, 217. [Google Scholar] [CrossRef]
- Wang, H.; Ye, Y.; Pan, S.-Y.; Zhu, G.-Y.; Li, Y.-W.; Fong, D.W.; Yu, Z.-L. Proteomic identification of proteins involved in the anticancer activities of oridonin in HepG2 cells. Phytomedicine 2011, 18, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhong, Z.; Wan, J.; Tan, W.; Wu, G.; Chen, M.; Wang, Y. Oridonin induces apoptosis, inhibits migration and invasion on highly-metastatic human breast cancer cells. Am. J. Chin. Med. 2013, 41, 177–196. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.; He, X.; He, X.; Li, M.; Zhang, R.; Fan, P.; Zhang, Q.; Jia, Z. The genus Scutellaria an ethnopharmacological and phytochemical review. J. Ethnopharmacol. 2010, 128, 279–313. [Google Scholar] [CrossRef] [PubMed]
- Gasiorowski, K.; Lamer-Zarawska, E.; Leszek, J.; Parvathaneni, K.; Yendluri, B.B.; Błach-Olszewska, Z.; Aliev, G. Flavones from root of Scutellaria baicalensis Georgi: Drugs of the future in neurodegeneration? CNS Neurol. Disord. Drug Targets 2011, 10, 184–191. [Google Scholar] [CrossRef]
- Matkowski, A.; Jamiolkowska-Kozlowska, W.; Nawrot, I. Chinese Medicinal Herbs as Source of Antioxidant Compounds—Where Tradition Meets the Future. Curr. Med. Chem. 2013, 20, 984–1004. [Google Scholar] [PubMed]
- Wozniak, D.; Lamer-Zarawska, E.; Matkowski, A. Antimutagenic and antiradical properties of flavones from the roots of Scutellaria baicalensis Georgi. Food/Nahrung 2004, 48, 9–12. [Google Scholar] [CrossRef]
- Woźniak, D.; Dryś, A.; Matkowski, A. Antiradical and antioxidant activity of flavones from Scutellariae baicalensis radix. Nat. Prod. Res. 2015, 29, 1567–1570. [Google Scholar] [CrossRef]
- Kumagai, T.; Müller, C.I.; Desmond, J.C.; Imai, Y.; Heber, D.; Koeffler, H.P. Scutellaria baicalensis, a herbal medicine: Anti-proliferative and apoptotic activity against acute lymphocytic leukemia, lymphoma and myeloma cell lines. Leuk. Res. 2007, 31, 523–530. [Google Scholar] [CrossRef]
- Scheck, A.C.; Perry, K.; Hank, N.C.; Clark, W.D. Anticancer activity of extracts derived from the mature roots of Scutellaria baicalensis on human malignant brain tumor cells. BMC Complement. Altern. Med. 2006, 6, 27. [Google Scholar] [CrossRef]
- Ye, F.; Jiang, S.; Volshonok, H.; Wu, J.; Zhang, D.Y. Molecular mechanism of anti-prostate cancer activity of Scutellaria baicalensis extract. Nutr. Cancer 2007, 57, 100–110. [Google Scholar] [CrossRef]
- Zhang, D.Y.; Wu, J.; Ye, F.; Xue, L.; Jiang, S.; Yi, J.; Zhang, W.; Wei, H.; Sung, M.; Wang, W. Inhibition of cancer cell proliferation and prostaglandin E2 synthesis by Scutellaria baicalensis. Cancer Res. 2003, 63, 4037–4043. [Google Scholar] [PubMed]
- Wang, H.; Li, H.; Luo, J.; Gu, J.; Wang, H.; Wu, H.; Xu, Y. Baicalin extracted from Huang qin (Radix Scutellariae Baicalensis) induces apoptosis in gastric cancer cells by regulating B cell lymphoma (Bcl-2)/Bcl-2-associated X protein and activating caspase-3 and caspase-9. J. Tradit. Chin. Med. 2017, 37, 229–235. [Google Scholar] [PubMed]
- Zhu, N.; Wang, S.; Lawless, J.; He, J.; Zheng, Z. Dose Dependent Dual Effect of Baicalin and Herb Huang Qin Extract on Angiogenesis. PLoS ONE 2016, 11, e0167125. [Google Scholar] [CrossRef] [PubMed]
- Sato, D.; Kondo, S.; Yazawa, K.; Mukudai, Y.; Li, C.; Kamatani, T.; Katsuta, H.; Yoshihama, Y.; Shirota, T.; Shintani, S. The potential anticancer activity of extracts derived from the roots of Scutellaria baicalensis on human oral squamous cell carcinoma cells. Mol. Clin. Oncol. 2013, 1, 105–111. [Google Scholar] [CrossRef]
- Suh, S.J.; Yoon, J.W.; Lee, T.K.; Jin, U.H.; Kim, S.L.; Kim, M.S.; Kwon, D.Y.; Lee, Y.C.; Kim, C.H. Chemoprevention of Scutellaria bardata on human cancer cells and tumorigenesis in skin cancer. Phytother. Res. 2007, 21, 135–141. [Google Scholar] [CrossRef]
- Qu, G.-W.; Yue, X.-D.; Li, G.-S.; Yu, Q.-Y.; Dai, S.-J. Two new cytotoxic ent-clerodane diterpenoids from Scutellaria barbata. J. Asian Nat. Prod. Res. 2010, 12, 859–864. [Google Scholar] [CrossRef]
- Dai, Z.-J.; Gao, J.; Li, Z.-F.; Ji, Z.-Z.; Kang, H.-F.; Guan, H.-T.; Diao, Y.; Wang, B.-F.; Wang, X.-J. In Vitro and In Vivo Antitumor Activity of Scutellaria barbate Extract on Murine Liver Cancer. Molecules 2011, 16, 4389–4400. [Google Scholar] [CrossRef]
- Chen, V.; Staub, R.E.; Baggett, S.; Chimmani, R.; Tagliaferri, M.; Cohen, I.; Shtivelman, E. Identification and Analysis of the Active Phytochemicals from the Anti-Cancer Botanical Extract Bezielle. PLoS ONE 2012, 7, e30107. [Google Scholar] [CrossRef]
- Androutsopoulos, V.P.; Ruparelia, K.; Arroo, R.R.; Tsatsakis, A.M.; Spandidos, D.A. CYP1-mediated antiproliferative activity of dietary flavonoids in MDA-MB-468 breast cancer cells. Toxicology 2009, 264, 162–170. [Google Scholar] [CrossRef]
- Qiu, D.; Kao, P.N. Immunosuppressive and anti-inflammatory mechanisms of triptolide, the principal active diterpenoid from the Chinese medicinal herb Tripterygium wilfordii Hook. f. Drugs R D 2003, 4, 1–18. [Google Scholar] [CrossRef]
- Brinker, A.M.; Ma, J.; Lipsky, P.E.; Raskin, I.J.P. Medicinal chemistry and pharmacology of genus Tripterygium (Celastraceae). Phytochemistry 2007, 68, 732–766. [Google Scholar] [CrossRef] [PubMed]
- Ziaei, S.; Halaby, R. Immunosuppressive, anti-inflammatory and anti-cancer properties of triptolide: A mini review. Avicenna J. Phytomed. 2016, 6, 149. [Google Scholar] [PubMed]
- Sarkar, S.; Paul, S. Triptolide Mediated Amelioration of Breast Cancer via Modulation of Molecular Pathways. Pharmacogn. J. 2017, 9, 838–845. [Google Scholar] [CrossRef]
- Kiviharju, T.M.; Lecane, P.S.; Sellers, R.G.; Peehl, D.M. Antiproliferative and proapoptotic activities of triptolide (PG490), a natural product entering clinical trials, on primary cultures of human prostatic epithelial cells. Clin. Cancer Res. 2002, 8, 2666–2674. [Google Scholar]
- Chang, W.-T.; Kang, J.J.; Lee, K.-Y.; Wei, K.; Anderson, E.; Gotmare, S.; Ross, J.A.; Rosen, G.D. Triptolide and chemotherapy cooperate in tumor cell apoptosis A role for the p53 pathway. J. Biol. Chem. 2001, 276, 2221–2227. [Google Scholar] [CrossRef]
- Lou, Y.J.; Jin, J. Triptolide down-regulates bcr-abl expression and induces apoptosis in chronic myelogenous leukemia cells. Leuk. Lymphoma 2004, 45, 373–376. [Google Scholar] [CrossRef]
- He, M.-F.; Huang, Y.-H.; Wu, L.-W.; Ge, W.; Shaw, P.-C.; But, P.P.-H. Triptolide functions as a potent angiogenesis inhibitor. Int. J. Cancer 2010, 126, 266–278. [Google Scholar] [CrossRef]
- Yao, J.; Jiang, Z.; Duan, W.; Huang, J.; Zhang, L.; Hu, L.; He, L.; Li, F.; Xiao, Y.; Shu, B.; et al. Involvement of mitochondrial pathway in triptolide-induced cytotoxicity in human normal liver L-02 cells. Boil. Pharm. Bull. 2008, 31, 592–597. [Google Scholar] [CrossRef]
- Yang, H.; Chen, D.; Cui, Q.C.; Yuan, X.; Dou, Q.P. Celastrol, a triterpene extracted from the Chinese “Thunder of God Vine,” is a potent proteasome inhibitor and suppresses human prostate cancer growth in nude mice. Cancer Res. 2006, 66, 4758–4765. [Google Scholar] [CrossRef]
- Sethi, G.; Ahn, K.S.; Pandey, M.K.; Aggarwal, B.B. Celastrol, a novel triterpene, potentiates TNF-induced apoptosis and suppresses invasion of tumor cells by inhibiting NF-κB–regulated gene products and TAK1-mediated NF-κB activation. Blood 2007, 109, 2727–2735. [Google Scholar] [CrossRef]
- Lee, J.-H.; Koo, T.H.; Yoon, H.; Jung, H.S.; Jin, H.Z.; Lee, K.; Hong, Y.-S.; Lee, J.J. Inhibition of NF-κB activation through targeting IκB kinase by celastrol, a quinone methide triterpenoid. Biochem. Pharmacol. 2006, 72, 1311–1321. [Google Scholar] [CrossRef] [PubMed]
- Antonoff, M.B.; Chugh, R.; Borja-Cacho, D.; Dudeja, V.; Clawson, K.A.; Skube, S.J.; Sorenson, B.S.; Saltzman, D.A.; Vickers, S.M.; Saluja, A.K. Triptolide therapy for neuroblastoma decreases cell viability in vitro and inhibits tumor growth in vivo. Surgery 2009, 146, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Chen, J.; Guo, Z.; Xu, X.-M.; Wang, L.; Pei, X.-F.; Yang, J.; Underhill, C.B.; Zhang, L. Triptolide inhibits the growth and metastasis of solid tumors1. Mol. Cancer Ther. 2003, 2, 65–72. [Google Scholar] [PubMed]
- Ravipati, A.S.; Zhang, L.; Koyyalamudi, S.R.; Jeong, S.C.; Reddy, N.; Bartlett, J.; Smith, P.T.; Shanmugam, K.; Münch, G.; Wu, M.J.; et al. Antioxidant and anti-inflammatory activities of selected Chinese medicinal plants and their relation with antioxidant content. BMC Complement. Altern. Med. 2012, 12, 173. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-R.; Lee, J.Y.; Lee, H.-H.; Aryal, D.K.; Kim, Y.G.; Kim, S.K.; Woo, E.-R.; Kang, K.W. Antioxidative effects of quercetin-glycosides isolated from the flower buds of Tussilago farfara L. Food Chem. Toxicol. 2006, 44, 1299–1307. [Google Scholar] [CrossRef]
- Lee, M.-R.; Cha, M.-R.; Jo, K.-J.; Yoon, M.-Y.; Park, H.-R. Cytotoxic and apoptotic activities of Tussilago farfara extract in HT-29 human colon cancer cells. Food Sci. Biotechnol. 2008, 17, 308–312. [Google Scholar]
- Fatykhova, D.G.; Karamova, N.S.; Abdrahimova, Y.R.; Ilinskaya, O.N. Evaluation of antigenotoxic effects of juices of plants Chelidonium majus L., Plantago major L. и Tussilago farfara L. Ecol. Genet. 2010, 8, 56–65. [Google Scholar] [CrossRef]
- Lee, H.-J.; Cho, H.-S.; Jun, S.; Lee, J.-J.; Yoon, J.; Lee, J.-H.; Song, H.; Choi, S.; Kim, S.; Saloura, V.; et al. Tussilago farfara L. augments TRAIL-induced apoptosis through MKK7/JNK activation by inhibition of MKK7 TIPRL in human hepatocellular carcinoma cells. Oncol. Rep. 2014, 32, 1117–1123. [Google Scholar] [CrossRef]
- Nair, N.C.; Sheela, D. Quantification of secondary metabolites and anti-oxidant potential of selected members of the tribe Heliantheae. J. Pharmacogn. Phytochem. 2016, 5, 163–166. [Google Scholar]
- Manjamalai, A.; Grace, V. Antioxidant activity of essential oils from Wedelia chinensis (Osbeck) in vitro and in vivo lung cancer bearing C57BL/6 mice. Asian Pac. J. Cancer Prev. 2012, 13, 3065–3071. [Google Scholar] [CrossRef]
- Noori, S.; Hassan, Z.M. Dihydroartemisinin shift the immune response towards Th1, inhibit the tumor growth in vitro and in vivo. Cell. Immunol. 2011, 271, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Dell’Eva, R.; Pfeffer, U.; Vené, R.; Anfosso, L.; Forlani, A.; Albini, A.; Efferth, T. Inhibition of angiogenesis in vivo and growth of Kaposi’s sarcoma xenograft tumors by the anti-malarial artesunate. Biochem. Pharmacol. 2004, 68, 2359–2366. [Google Scholar] [CrossRef] [PubMed]
- Katiyar, S.K.; Meeran, S.M.; Katiyar, N.; Akhtar, S. p53 cooperates berberine-induced growth inhibition and apoptosis of non-small cell human lung cancer cells in vitro and tumor xenograft growth in vivo. Mol. Carcinog. 2009, 48, 24–37. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.S.; Oh, J.H.; Kim, S.M.; Jung, H.Y.; Yoo, H.S.; Lee, Y.M.; Moon, D.C.; Han, S.B.; Hong, J.T. Berberine inhibits p53-dependent cell growth through induction of apoptosis of prostate cancer cells. Int. J. Oncol. 2009, 34, 1221–1230. [Google Scholar]
- Harikumar, K.B.; Kuttan, G.; Kuttan, R. Inhibition of progression of erythroleukemia induced by Friend virus in BALB/c mice by natural products—Berberine, Curcumin and Picroliv. J. Exp. Ther. Oncol. 2008, 7, 275–284. [Google Scholar]
- Peng, P.-L.; Kuo, W.-H.; Tseng, H.-C.; Chou, F.-P. Synergistic Tumor-Killing Effect of Radiation and Berberine Combined Treatment in Lung Cancer: The Contribution of Autophagic Cell Death. Int. J. Radiat. Oncol. 2008, 70, 529–542. [Google Scholar] [CrossRef]
- Huang, T.; Xiao, Y.; Yi, L.; Li, L.; Wang, M.; Tian, C.; Ma, H.; He, K.; Wang, Y.; Han, B.; et al. Coptisine from Rhizoma coptidis Suppresses HCT-116 Cells-related Tumor Growth in vitro and in vivo. Sci. Rep. 2017, 7, 38524. [Google Scholar] [CrossRef]
- Chen, X.-Z.; Cao, Z.-Y.; Chen, T.-S.; Zhang, Y.-Q.; Liu, Z.-Z.; Su, Y.-T.; Liao, L.-M.; Du, J. Water extract of Hedyotis diffusa Willd suppresses proliferation of human HepG2 cells and potentiates the anticancer efficacy of low-dose 5-fluorouracil by inhibiting the CDK2-E2F1 pathway. Oncol. Rep. 2012, 28, 742–748. [Google Scholar] [CrossRef]
- Yang, X.; Yang, Y.; Tang, S.; Tang, H.; Yang, G.; Xu, Q.; Wu, J. Anti-tumor effect of polysaccharides from Scutellaria barbata D. Don on the 95-D xenograft model via inhibition of the C-met pathway. J. Pharmacol. Sci. 2014, 125, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Gu, J.-F.; Zou, X.; Wu, J.; Zhang, M.-H.; Jiang, J.; Qin, D.; Zhou, J.-Y.; Liu, B.-X.-Z.; Zhu, Y.-T. The anti-lung cancer activities of steroidal saponins of P. polyphylla Smith var. chinensis (Franch.) Hara through enhanced immunostimulation in experimental Lewis tumor-bearing C57BL/6 mice and induction of apoptosis in the A549 cell line. Molecules 2013, 18, 12916–12936. [Google Scholar] [CrossRef] [PubMed]
- Wanga, Y.; Huangb, X.; Hanc, J.; Zhenga, W.; Maa, W. Extract of Perilla frutescens inhibits tumor proliferation of HCC via PI3K/AKT signal pathway. Afr. J. Tradit. Complementary Altern. Med. 2013, 10, 251–257. [Google Scholar] [CrossRef]
- Fukutake, M.; Yokota, S.; Kawamura, H.; Iizuka, A.; Amagaya, S.; Fukuda, K.; Komatsu, Y. Inhibitory Effect of Coptidis rhizoma and Scutellariae Radix on Azoxymethane-Induced Aberrant Crypt Foci Formation in Rat Colon. Biol. Pharm. Bull. 1998, 21, 814–817. [Google Scholar] [CrossRef] [PubMed]
- Manjamalai, A.; Grace, B. Chemotherapeutic effect of essential oil of Wedelia chinensis (Osbeck) on inducing apoptosis, suppressing angiogenesis and lung metastasis in C57BL/6 mice model. J. Cancer Sci. Ther. 2013, 5, 271–281. [Google Scholar] [CrossRef]
- Singh, N.P.; Lai, H.C.; Park, J.S.; Gerhardt, T.E.; Kim, B.J.; Wang, S.; Sasaki, T. Effects of artemisinin dimers on rat breast cancer cells in vitro and in vivo. Anticancer Res. 2011, 31, 4111–4114. [Google Scholar] [PubMed]
- Lai, H.; Singh, N.P. Oral artemisinin prevents and delays the development of 7, 12-dimethylbenz [a] anthracene (DMBA)-induced breast cancer in the rat. Cancer Lett. 2006, 231, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Joe, B.; Rao, U.J.; Lokesh, B.R. Presence of an acidic glycoprotein in the serum of arthritic rats: Modulation by capsaicin and curcumin. Mol. Cell. Biochem. 1997, 169, 125–134. [Google Scholar] [CrossRef]
- Tanaka, T.; Kohno, H.; Shimada, R.; Kagami, S.; Yamaguchi, F.; Kataoka, S.; Ariga, T.; Murakami, A.; Koshimizu, K.; Ohigashi, H. Prevention of colonic aberrant crypt foci by dietary feeding of garcinol in male F344 rats. Carcinogenesis 2000, 21, 1183–1189. [Google Scholar] [CrossRef]
- Liu, S.; Leach, S.D. Zebrafish models for cancer. Annu. Rev. Pathol. 2011, 6, 71–93. [Google Scholar] [CrossRef]
- Zhu, X.-Y.; Guo, D.-W.; Lao, Q.-C.; Xu, Y.-Q.; Meng, Z.-K.; Xia, B.; Yang, H.; Li, C.-Q.; Li, P. Sensitization and synergistic anti-cancer effects of Furanodiene identified in zebrafish models. Sci. Rep. 2019, 9, 4541. [Google Scholar] [CrossRef]
- Efferth, T.; Dunstan, H.; Sauerbrey, A.; Miyachi, H.; Chitambar, C.R. The anti-malarial artesunate is also active against cancer. Int. J. Oncol. 2001, 18, 767–773. [Google Scholar] [CrossRef]
- Efferth, T. Molecular pharmacology and pharmacogenomics of artemisinin and its derivatives in cancer cells. Curr. Drug Targets 2006, 7, 407–421. [Google Scholar] [CrossRef] [PubMed]
- Breuer, E.; Efferth, T. Treatment of Iron-Loaded Veterinary Sarcoma by Artemisia annua. Nat. Prod. Bioprospecting 2014, 4, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Apolone, G.; Joppi, R.; Garattini, S. Ten years of marketing approvals of anticancer drugs in Europe: Regulatory policy and guidance documents need to find a balance between different pressures. Br. J. Cancer 2005, 93, 504. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Farrell, A.; Papadouli, I.; Hori, A.; Harczy, M.; Harrison, B.; Asakura, W.; Marty, M.; Dagher, R.; Pazdur, R. The advisory process for anticancer drug regulation: A global perspective. Ann. Oncol. 2005, 17, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Mezher, M. FDA Adopts ICH Guideline on Nonclinical Evaluation for Anticancer Drugs; Regulatory Affairs Professional Society: Rockville, MD, USA, 2018. [Google Scholar]
- Calixto, J. Efficacy, safety, quality control, marketing and regulatory guidelines for herbal medicines (phytotherapeutic agents). Braz. J. Med. Biol. Res. 2000, 33, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.C.D.; Silva, J.M.D.; Ramos, M.A. What Factors Guide the Selection of Medicinal Plants in a Local Pharmacopoeia? A Case Study in a Rural Community from a Historically Transformed Atlantic Forest Landscape. Evid.-Based Complement. Altern. Med. 2018, 2018, 2519212. [Google Scholar] [CrossRef]
- Khan, T.; Abbasi, B.H.; Khan, M.A.; Shinwari, Z.K. Differential Effects of Thidiazuron on Production of Anticancer Phenolic Compounds in Callus Cultures of Fagonia indica. Appl. Biochem. Biotechnol. 2016, 179, 46–58. [Google Scholar] [CrossRef]
- Khan, T.; Abbasi, B.H.; Khan, M.A.; Azeem, M. Production of biomass and useful compounds through elicitation in adventitious root cultures of Fagonia indica. Ind Crop. Prod. 2017, 108, 451–457. [Google Scholar] [CrossRef]
- Huang, L.; Xie, D.; Yu, Y.; Liu, H.; Shi, Y.; Shi, T.; Wen, C. TCMID 2.0: A comprehensive resource for TCM. Nucleic Acids Res. 2018, 46, D1117–D1120. [Google Scholar] [CrossRef]
- Zeng, X.; Zhang, P.; Wang, Y.; Qin, C.; Chen, S.; He, W.; Tao, L.; Tan, Y.; Gao, D.; Wang, B.; et al. CMAUP: A database of collective molecular activities of useful plants. Nucleic Acids Res. 2018, 47, D1118–D1127. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, F.; Yang, K.; Fang, S.; Bu, D.; Li, H.; Sun, L.; Hu, H.; Gao, K.; Wang, W.; et al. SymMap: An integrative database of traditional Chinese medicine enhanced by symptom mapping. Nucleic Acids Res. 2018, 47, D1110–D1117. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.-Y.; Zhang, Y.-Q.; Liu, Z.-M.; Chen, T.; Lv, C.-Y.; Tang, S.-H.; Zhang, X.-B.; Zhang, W.; Li, Z.-Y.; Zhou, R.-R.; et al. ETCM: An encyclopaedia of traditional Chinese medicine. Nucleic Acids Res. 2018, 47, D976–D982. [Google Scholar] [CrossRef] [PubMed]
- Jia, C.-Y.; Li, J.-Y.; Hao, G.-F.; Yang, G.-F. A drug-likeness toolbox facilitates ADMET study in drug discovery. Drug Discov. Today 2019. [Google Scholar] [CrossRef] [PubMed]
- Keefe, L.J.; Stoll, V.S. Accelerating pharmaceutical structure-guided drug design: A successful model. Drug Discov. Today 2019, 24, 377–381. [Google Scholar] [CrossRef]
- Ferreira, L.L.G.; Andricopulo, A.D. ADMET modeling approaches in drug discovery. Drug Discov. Today 2019, 24, 1157–1165. [Google Scholar] [CrossRef]
- Bergström, F.; Lindmark, B. Accelerated drug discovery by rapid candidate drug identification. Drug Discov. Today 2019, 24, 1237–1241. [Google Scholar] [CrossRef]
- Fang, J.; Liu, C.; Wang, Q.; Lin, P.; Cheng, F. In silico polypharmacology of natural products. Brief. Bioinform. 2018, 19, 1153–1171. [Google Scholar] [CrossRef]
- Yang, B.; Mao, J.; Gao, B.; Lu, X. Computer-Assisted Drug Virtual Screening Based on the Natural Product Databases. Curr. Pharm. Biotechnol. 2019, 20, 293–301. [Google Scholar] [CrossRef]
- Qu, Y.; Zhang, Z.; Lu, Y.; Zheng, D.; Wei, Y. Network Pharmacology Reveals the Molecular Mechanism of Cuyuxunxi Prescription in Promoting Wound Healing in Patients with Anal Fistula. Evidence-Based Complement. Altern. Med. 2019, 2019, 3865121-9. [Google Scholar] [CrossRef]
- Lv, Y.; Hou, X.; Zhang, Q.; Li, R.; Xu, L.; Chen, Y.; Tian, Y.; Sun, R.; Zhang, Z.; Xu, F. Untargeted Metabolomics Study of the In vitro Anti-Hepatoma Effect of Saikosaponin d in Combination with NRP-1 Knockdown. Molecules 2019, 24, 1423. [Google Scholar] [CrossRef]
- Wang, Y.; Jafari, M.; Tang, Y.; Tang, J. Predicting Meridian in Chinese traditional medicine using machine learning approaches. PLoS Comput. Boil. 2019, 15, e1007249. [Google Scholar] [CrossRef] [PubMed]
- Gaur, R.; Yadav, D.K.; Kumar, S.; Darokar, M.P.; Khan, F.; Bhakuni, R.S. Molecular modeling based synthesis and evaluation of in vitro anticancer activity of indolyl chalcones. Curr. Top. Med. Chem. 2015, 15, 1003–1012. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tabana, Y.M.; Hassan, L.E.; Ahamed, M.B.; Dahham, S.S.; Iqbal, M.A.; Saeed, M.A.; Khan, M.S.; Sandai, D.; Majid, A.S.; Oon, C.E.; et al. Scopoletin, an active principle of tree tobacco (Nicotiana glauca) inhibits human tumor vascularization in xenograft models and modulates ERK1, VEGF-A, and FGF-2 in computer model. Microvasc Res. 2016, 107, 17–33. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Prakash, O.; Shukla, A.; Rajpurohit, C.S.; Vasudev, P.G.; Luqman, S.; Srivastava, S.K.; Pant, A.B.; Khan, F. Structure-Activity Relationship Studies on Holy Basil (Ocimum sanctum L.) Based Flavonoid Orientin and its Analogue for Cytotoxic Activity in Liver Cancer Cell Line HepG2. Comb. Chem. High Throughput Screen. 2016, 19, 656–666. [Google Scholar] [CrossRef] [PubMed]
- Ntie-Kang, F.; Simoben, C.V.; Karaman, B.; Ngwa, V.F.; Judson, P.N.; Sippl, W.; Mbaze, L.M. Pharmacophore modeling and in silico toxicity assessment of potential anticancer agents from African medicinal plants. Drug Des. Dev. Ther. 2016, 10, 2137–2154. [Google Scholar] [CrossRef]
- Li, Y.; Wang, J.; Lin, F.; Yang, Y.; Chen, S.S. A Methodology for Cancer Therapeutics by Systems Pharmacology-Based Analysis: A Case Study on Breast Cancer-Related Traditional Chinese Medicines. PLoS ONE 2017, 12, e0169363. [Google Scholar] [CrossRef]
- Sharma, P.; Shukla, A.; Kalani, K.; Dubey, V.; Luqman, S.; Srivastava, S.K.; Khan, F. In-silico & In-vitro Identification of Structure-Activity Relationship Pattern of Serpentine & Gallic Acid Targeting PI3Kgamma as Potential Anticancer Target. Curr. Cancer Drug Targets 2017, 17, 722–734. [Google Scholar] [CrossRef]
- Shirzad, H.; Taji, F.; Rafieian-Kopaei, M. Correlation between antioxidant activity of garlic extracts and WEHI-164 fibrosarcoma tumor growth in BALB/c mice. J. Med. Food 2011, 14, 969–974. [Google Scholar] [CrossRef]
- Lakshmi, S.; Suresh, S.; Rahul, B.; Saikant, R.; Maya, V.; Gopi, M.; Padmaja, G.; Remani, P. In vitro and in vivo studies of 5, 7-dihydroxy flavones isolated from Alpinia galanga (L.) against human lung cancer and ascetic lymphoma. Med. Chem. Res. 2019, 28, 39–51. [Google Scholar] [CrossRef]
- Jagetia, G.C.; Baliga, M.S. Evaluation of anticancer activity of the alkaloid fraction of Alstonia scholaris (Sapthaparna) in vitro and in vivo. Phytother. Res. Int. J. Devoted Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 2006, 20, 103–109. [Google Scholar]
- Kumar, R.A.; Sridevi, K.; Kumar, N.V.; Nanduri, S.; Rajagopal, S. Anticancer and immunostimulatory compounds from Andrographis paniculata. J. Ethnopharmacol. 2004, 92, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.R.; Spindola, D.G.; Garcia, D.M.; Erustes, A.; Bechara, A.; Palmeira-dos-Santos, C.; Smaili, S.S.; Pereira, G.J.; Hinsberger, A.; Viriato, E.P. Medicinal properties of Angelica archangelica root extract: Cytotoxicity in breast cancer cells and its protective effects against in vivo tumor development. J. Integr. Med. 2019, 17, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Wang, W.; Xiao, H. The evaluation of anti-breast cancer activity and safety pharmacology of the ethanol extract of Aralia elata Seem. leaves. Drug Chem. Toxicol. 2019, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Baskar, A.A.; Ignacimuthu, S.; Paulraj, G.M.; Al Numair, K.S. Chemopreventive potential of β-sitosterol in experimental colon cancer model-an in vitro and in vivo study. BMC Complement. Altern. Med. 2010, 10, 24. [Google Scholar] [CrossRef]
- Yang, B.; Xiao, B.; Sun, T. Antitumor and immunomodulatory activity of Astragalus membranaceus polysaccharides in H22 tumor-bearing mice. Int. J. Biol. Macromol. 2013, 62, 287–290. [Google Scholar] [CrossRef]
- Lima, S.R.; Junior, V.F.V.; Christo, H.B.; Pinto, A.C.; Fernandes, P.D. In vivo and in vitro studies on the anticancer activity of Copaifera multijuga Hayne and its fractions. Phytother. Res. 2003, 17, 1048–1053. [Google Scholar] [CrossRef]
- Wang, N.; Feng, Y.; Zhu, M.; Tsang, C.M.; Man, K.; Tong, Y.; Tsao, S.W. Berberine induces autophagic cell death and mitochondrial apoptosis in liver cancer cells: The cellular mechanism. J. Cell. Biochem. 2010, 111, 1426–1436. [Google Scholar] [CrossRef]
- Kabeer, F.A.; Rajalekshmi, D.S.; Nair, M.S.; Prathapan, R. In vitro and in vivo antitumor activity of deoxyelephantopin from a potential medicinal plant Elephantopus scaber against Ehrlich ascites carcinoma. Biocatal. Agric. Biotechnol. 2019, 19, 101106. [Google Scholar] [CrossRef]
- Pareek, A.; Godavarthi, A.; Issarani, R.; Nagori, B.P. Antioxidant and hepatoprotective activity of Fagonia schweinfurthii (Hadidi) Hadidi extract in carbon tetrachloride induced hepatotoxicity in HepG2 cell line and rats. J. Ethnopharmacol. 2013, 150, 973–981. [Google Scholar] [CrossRef]
- Biloa Messi, B.; Ho, R.; Meli Lannang, A.; Cressend, D.; Perron, K.; Nkengfack, A.E.; Carrupt, P.-A.; Hostettmann, K.; Cuendet, M. Isolation and biological activity of compounds from Garcinia preussii. Pharm. Biol. 2014, 52, 706–711. [Google Scholar] [CrossRef]
- Li, J.; Sun, J.; Song, J. Experimental research on effect of Hedyotis diffusa Willd on blood metastasis in H22 mice. Lishizhen Med. Mater. Med. Res. 2012, 23, 2434–2435. [Google Scholar]
- Paramee, S.; Sookkhee, S.; Sakonwasun, C.; Takuathung, M.N.; Mungkornasawakul, P.; Nimlamool, W.; Potikanond, S. Anti-cancer effects of Kaempferia parviflora on ovarian cancer SKOV3 cells. BMC Complement. Altern. Med. 2018, 18, 178. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yuan, S.; Wang, J.; Lin, P.; Liu, G.; Lu, Y.; Zhang, J.; Wang, W.; Wei, Y. Anticancer activity of litchi fruit pericarp extract against human breast cancer in vitro and in vivo. Toxicol. Appl. Pharmacol. 2006, 215, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk, T.; Sitarek, P.; Skała, E.; Toma, M.; Wielanek, M.; Pytel, D.; Wieczfińska, J.; Szemraj, J.; Śliwiński, T. Induction of apoptosis by in vitro and in vivo plant extracts derived from Menyanthes trifoliata L. in human cancer cells. Cytotechnology 2019, 71, 165–180. [Google Scholar] [CrossRef] [PubMed]
- Turan, I.; Demir, S.; Kilinc, K.; Burnaz, N.A.; Yaman, S.O.; Akbulut, K.; Mentese, A.; Aliyazicioglu, Y.; Deger, O. Antiproliferative and apoptotic effect of Morus nigra extract on human prostate cancer cells. Saudi Pharm. J. 2017, 25, 241–248. [Google Scholar] [CrossRef]
- Boubaker, J.; Ben Toumia, I.; Sassi, A.; Bzouich-Mokded, I.; Ghoul Mazgar, S.; Sioud, F.; Bedoui, A.; Safta Skhiri, S.; Ghedira, K.; Chekir-Ghedira, L. Antitumoral potency by immunomodulation of chloroform extract from leaves of Nitraria retusa, Tunisian medicinal plant, via its major compounds β-sitosterol and palmitic acid in BALB/c mice bearing induced tumor. Nutr. Cancer 2018, 70, 650–662. [Google Scholar] [CrossRef]
- Lee, S.M.Y.; Li, M.L.Y.; Tse, Y.C.; Leung, S.C.L.; Lee, M.M.S.; Tsui, S.K.W.; Fung, K.P.; Lee, C.Y.; Waye, M.M.Y. Paeoniae Radix, a Chinese herbal extract, inhibit hepatoma cells growth by inducing apoptosis in a p53 independent pathway. Life Sci. 2002, 71, 2267–2277. [Google Scholar] [CrossRef]
- Xu, W.W.; Li, B.; Lai, E.T.C.; Chen, L.; Huang, J.J.H.; Cheung, A.L.M.; Cheung, P.C.K. Water extract from Pleurotus pulmonarius with antioxidant activity exerts in vivo chemoprophylaxis and chemosensitization for liver cancer. Nutr. Cancer 2014, 66, 989–998. [Google Scholar] [CrossRef]
- Wang, X.-N.; Xu, L.-N.; Peng, J.-Y.; Liu, K.-X.; Zhang, L.-H.; Zhang, Y.-K. In vivo inhibition of S180 tumors by the synergistic effect of the Chinese medicinal herbs Coptis chinensis and Evodia rutaecarpa. Planta Med. 2009, 75, 1215–1220. [Google Scholar] [CrossRef]
| S.No. | Plant Name | Common Name | Parts Used | Extract Used (Aqueous/Methanolic etc.) | Active Components Used | Dose Concentration | Cancer Cell Line Applied To | Animal Models Applied To | References |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Allium sativum | Garlic | Leaves | Aqueous extracts | Allicin, flavonoids, and phenolic components | 20 mg/kg/0.2 mL | Wehi-164 tumor cells | Balb/c mice | [202] |
| 2 | Alpinia galangal | Lengkuas, greater galangal, and blue ginger | Rhizomes | Ethyl acetate extract | Chrysin | 1.3 mg/kg | Murine daltons lymphoma ascite (dla) and human lung cancer (a549) cells | Balb/c mice | [203] |
| 3 | Alstonia scholaris | Blackboard or devil’s tree | Stem bark | Ethyl Alcohol extract | -- | 210 mg/kg | Hela cells lines | Swiss Albino mice | [204] |
| 4 | Andrographis paniculata | Creat or green chireta | Aerial parts | Methanolic extract | Diterpenes | 10 µg/mL | Cancer cell lines sw620 and a498 | Swiss Albino mice | [205] |
| 5 | Angelica archangelica | Garden angelica, wild celery, and Norwegian angelica | Root and rhizome | Ethanolic extract | Angelicin | 500mg/kg | Mcf7 and 4t1 cell lines | Female balb/c mice | [206] |
| 6 | Aralia elata | Chinese angelica-tree, Japanese angelica-tree, and Korean angelica-tree | Leaves | Ethanol extract | -- | 300 mg/kg | Mcf-7 cells | Tumor bearing-nude mice | [207] |
| 7 | Artemisia annua | Sweet wormwood, sweet annie, and sweet sagewort | -- | -- | Artemisinin | 0.02% | Breast cancer | Rats | [168] |
| 8 | Asclepia scurassavica | Tropical milkweed | Shade dried leaves | Ethyl acetate and methanolextract | Β-sitosterol | 10–20 mg/kg b.w. | Human colo 320 dm and monkey vero cell lines | Male wistar rats | [208] |
| 9 | Astragalus membranaceus | Mongolian milkvetch | -- | -- | Polysaccharide | 400 mg/kg | Liver cancer | H22 hepatocarcinoma transplanted balb/c mice | [209] |
| 10 | Copaifera multijuga | Hayne oil, Copaiba | Trunk of the tree | Oil resin | Clerodane Diterpenes | 2 g/Kg | B16f10 melanoma cells | Male C57/black mice | [210] |
| 11 | Coptidis rhizoma | Huanglian, Copaiba, and Copaibera | -- | -- | Berberine | 200 µM and 400 µM | Human hepatic carcinoma cell lines HepG2 and mhcc97-l. | -- | [211] |
| 12 | Curcuma longa | Turmeric | -- | -- | Curcumin | 75 µM | Ht-29 colon cancer cells of human | -- | [32] |
| 13 | Elephantopus scaber | Elephant′s Foot | -- | Dimethyl sulfoxide extract | Deoxyelephantopin (doe) | 25mg/kg | Murine ehrlich ascites carcinoma (eac) | Male swiss albino mice | [212] |
| 14 | Fagonia schweinfurthii | bush candle | Whole plant | Ethanolic extract | Carbon tetrachloride (ccl4) | 200 µg/mL | HepG2 cell line | Male albino rats | [213] |
| 15 | Garcinia indica | Kokum | Fruits | Ethanol extract | Garcinol | <1 μM | Ht-29 and hct-116 colon cancer cells | -- | [50] |
| 16 | Garcinia oblongifolia | Lingnan garcinia | Branch | Methanol extract | Xanthone | 1000 μg/mL | Mcf-7 breast cancer cell line | -- | [49] |
| 17 | Garcinia preussii | -- | Fruits and leaves | Meohextract | Benzophenones | Du145, hela, ht-29, and a431 cell lines | -- | [214] | |
| 18 | Hedyotis diffusa | Snake-needle grass | -- | -- | Hela cells | Nude mice xenograft | [215] | ||
| 19 | Hedyotis spp. | -- | Aerial parts, stem and leaves | Methanol extract | -- | 20 μM | Cem-ss cell line | -- | [54] |
| 20 | Kaempferia parviflora | Black ginger | Rhizomes | Ethanolic extract | -- | 1 mg/mL | Ovarian cancer cell line, skov3 | -- | [216] |
| 21 | Litchi chinensis | litchi or lychee | Fruit pericarp | Ethanolic extract | Polyphenolic compounds | 0.3 mg/mL | Human smmc-7721 hepatocellular carcinoma cell Line | Murine hepatoma bearing-mice | [217] |
| 22 | Menyanthes trifoliata | Bogbean, Buckbean, and Marsh Trefoil | Aerial part and root | Aqueous methanol extract | Polyphenolic compounds | 1.5 mg/mL | Grade iv glioma cells | -- | [218] |
| 23 | Morus alba | white mulberry | Root | N-hexane and methanolextracts. | Albanol a | 30 µM | HL-60 (human leukemia) and Crl1579 (human melanoma) cell lines | -- | [69] |
| 24 | Morus nigra | Black mulberry or blackberry | Aerial parts | dimethyl sulfoxide extract | Phenolic compounds especially Ascorbic acid and chlorogenic acid | 1000 μg/mL | human prostate adenocarcinoma (PC-3) | -- | [219] |
| 25 | Nitraria retusa | Salt tree or Nitre bush | Leaves | Chloroform extract | Β-sitosterol and palmitic acid | 50 mg/Kg b.w | B16-f10 cells lines | Balb/c mice | [220] |
| 26 | Paeonia lactiflora | Chinese Peony | Root | Aqueous extract | -- | 15 mg/mL | Human hepatoma cell lines (HepG2 and hep3b) | -- | [221] |
| 27 | Paris polyphylla | Herb Paris | Rhizomes | Methanol extract | Steroidal saponins | 7.5 mg/kg | A549 cell line | Tumor-bearing c57bl/6 mice | [163] |
| 28 | Perilla frutescens | Beafsteak plant | Leaves | Meoh extract | Isoegomaketone | 10nmol/l | Huh-7 hepatoma cell carcinoma | Tumor-xenograft nude mice | [164] |
| 29 | Perilla frutescens | Beafsteak plant | Leaf | -- | Rosmarinic acid | 105 µg/mL | Human hepatoma (HepG2) cells | -- | [89] |
| 30 | Platycodon grandiflorus | balloon-flower | Root | Platycodin d was dissolved in Phosphate-buffered saline | Platycodin D | 8 µg/mL | Human breast cancer cell line, mcf-7 | -- | [98] |
| 31 | Pleurotus pulmonarius | Indian Oyster, Italian Oyster, Phoenix Mushroom, or the Lung Oyster | Edible part | Aqueous extract | -- | 20 mg/kg | Huh7 liver cancer cells | Nude mice | [222] |
| 32 | Rabdosia rubescens | Bing Ling Cao, BlushredRabdosia, and Isodonrubescens | -- | -- | Oridonin | 30 μmol/L | Human gallbladder cancer cell lines sgc996 and noz | Athymic nude mice | [113] |
| 33 | Rhodamnia rubescens | Scrub stringybark, brush turpentine, or brown malletwood | -- | -- | Tetracycline diterpenoidoridonin | 50 μM | Human breast (mcf-7 and mda-mb-231) cancer cells | -- | [115] |
| 34 | Scutellaria barbata | Barbed Skullcap | -- | -- | Polysaccharides | 40 µg/mL | 95-d cell line | Xenograft model | [162] |
| 35 | Scutellaria baicalensis | Baikal skullcap | Root | Aqueous extract | Baicalin | 100 µg/mL | Human oral squamous cell carcinoma (oscc) cell line | -- | [127] |
| 36 | Tripterygium wilfordii | Thunder god vine | -- | -- | Triptolide | 250 nmol/L | Neuroblastoma cell lines (n2a and sknsh) | Neuroblastoma (nude mice model) | [145] |
| 37 | Tussilago farfara | Coltsfoot | Flower buds | Methanol extract | Quercetin-glycosides | Ht-29 human colon cancer cells | -- | [149] | |
| 38 | Wedelia chinensis | Chinese Wedelia | Leaves | Essential oils | Carvocrol and trans-caryophyllene | B16f-10 melanoma metastatic cell line | C57bl/6 mice | [153] | |
| 39 | Zuojin wan | -- | Aqueous extract | Palmatine, berberine, epiberberine, and coptisine | 10 mg/mL | S180 tumor cells | Chinese kunming (km) mice | [223] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, T.; Ali, M.; Khan, A.; Nisar, P.; Jan, S.A.; Afridi, S.; Shinwari, Z.K. Anticancer Plants: A Review of the Active Phytochemicals, Applications in Animal Models, and Regulatory Aspects. Biomolecules 2020, 10, 47. https://doi.org/10.3390/biom10010047
Khan T, Ali M, Khan A, Nisar P, Jan SA, Afridi S, Shinwari ZK. Anticancer Plants: A Review of the Active Phytochemicals, Applications in Animal Models, and Regulatory Aspects. Biomolecules. 2020; 10(1):47. https://doi.org/10.3390/biom10010047
Chicago/Turabian StyleKhan, Tariq, Muhammad Ali, Ajmal Khan, Parveen Nisar, Sohail Ahmad Jan, Shakeeb Afridi, and Zabta Khan Shinwari. 2020. "Anticancer Plants: A Review of the Active Phytochemicals, Applications in Animal Models, and Regulatory Aspects" Biomolecules 10, no. 1: 47. https://doi.org/10.3390/biom10010047
APA StyleKhan, T., Ali, M., Khan, A., Nisar, P., Jan, S. A., Afridi, S., & Shinwari, Z. K. (2020). Anticancer Plants: A Review of the Active Phytochemicals, Applications in Animal Models, and Regulatory Aspects. Biomolecules, 10(1), 47. https://doi.org/10.3390/biom10010047