Panax ginseng Pharmacopuncture: Current Status of the Research and Future Challenges
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Study Selection, Data Extraction, and Data Analysis
3. Results
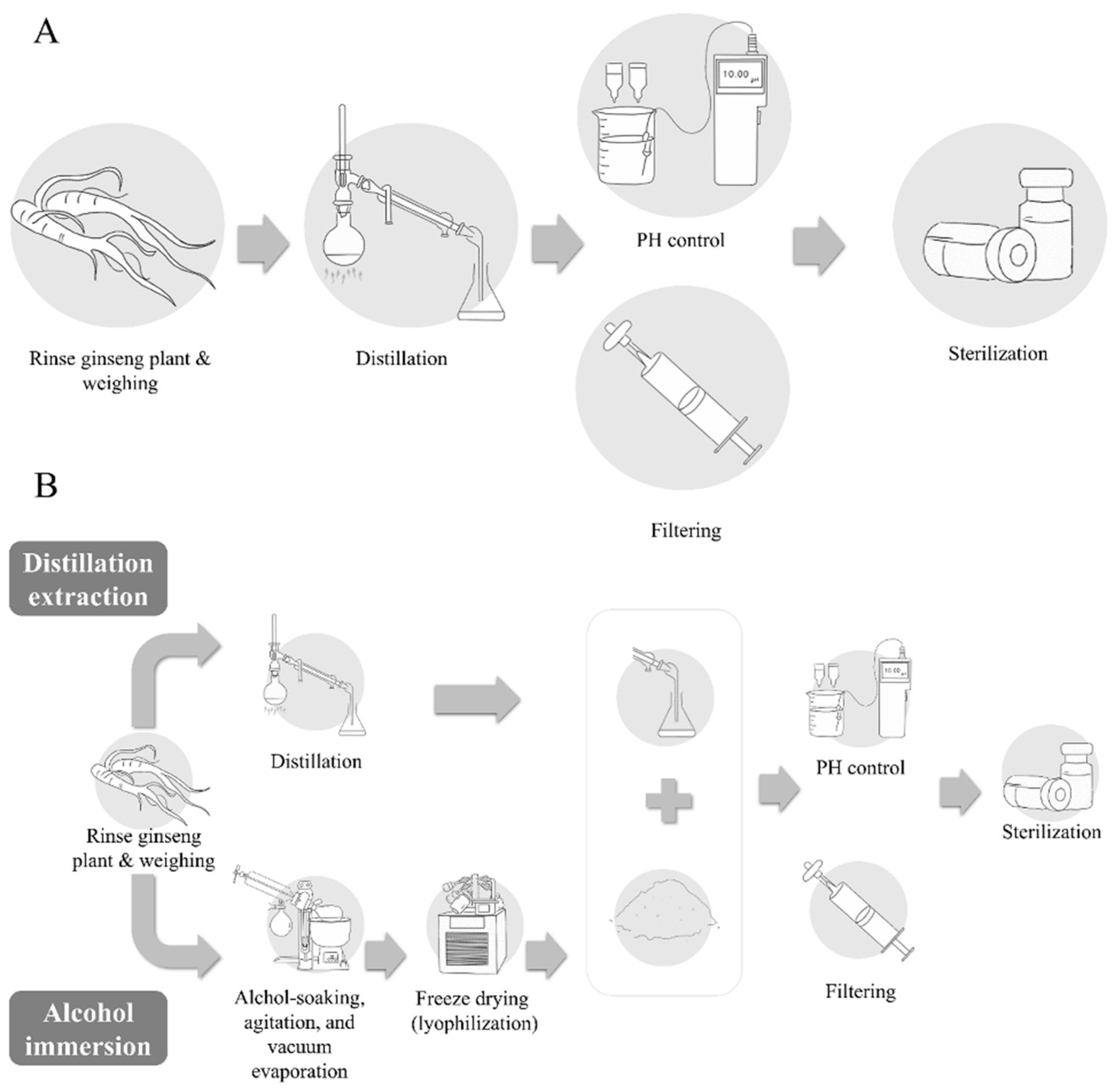
3.1. The Manufacturing Process of the Ginseng Pharmacopuncture Solution
3.2. Physiological Response to Ginseng Pharmacopuncture in Human Subjects
3.3. Safety Tests of Ginseng Pharmacopuncture
3.4. Systematic Review of Clinical Studies
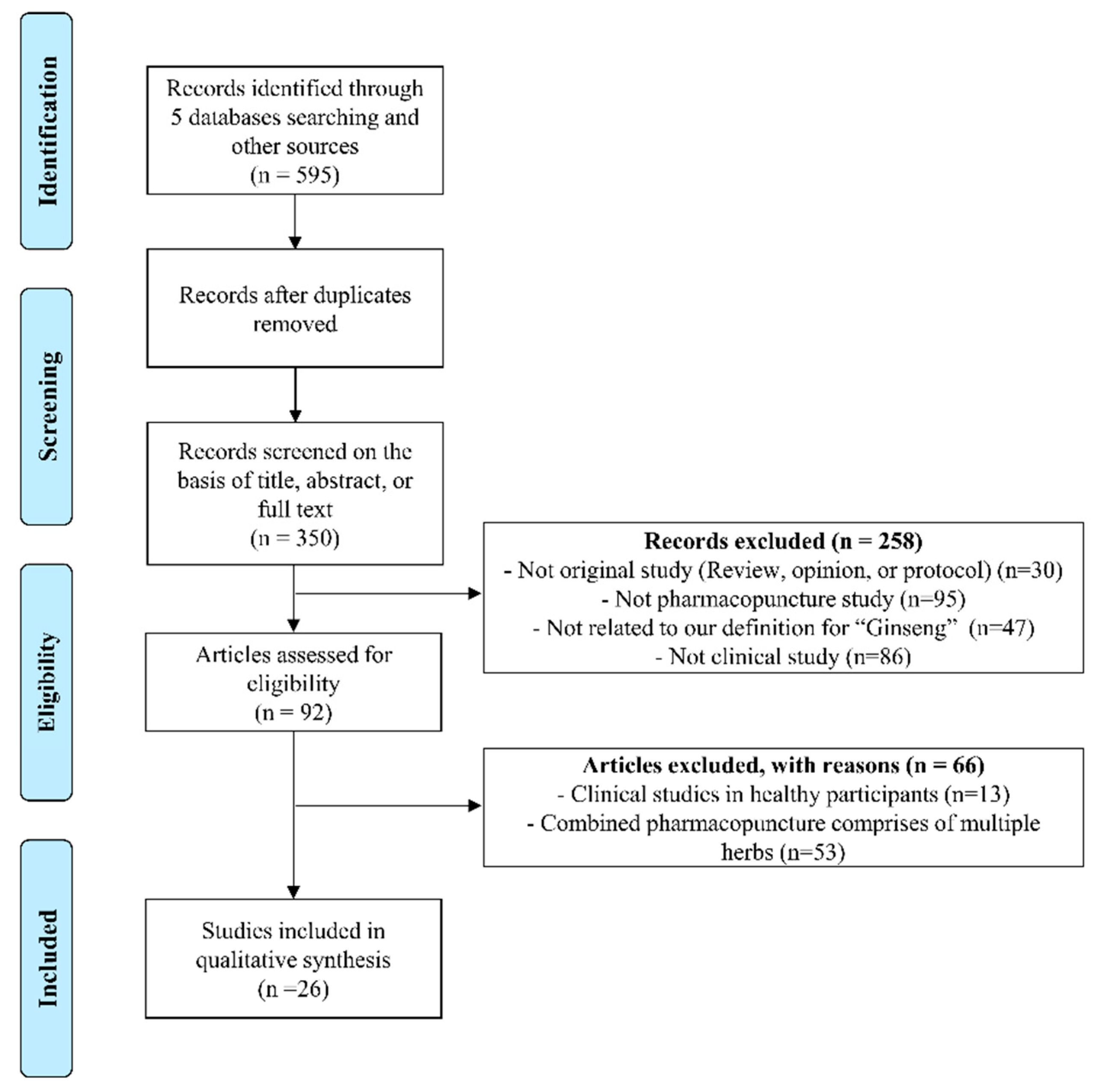
3.4.1. Search Results
3.4.2. Participants and Settings
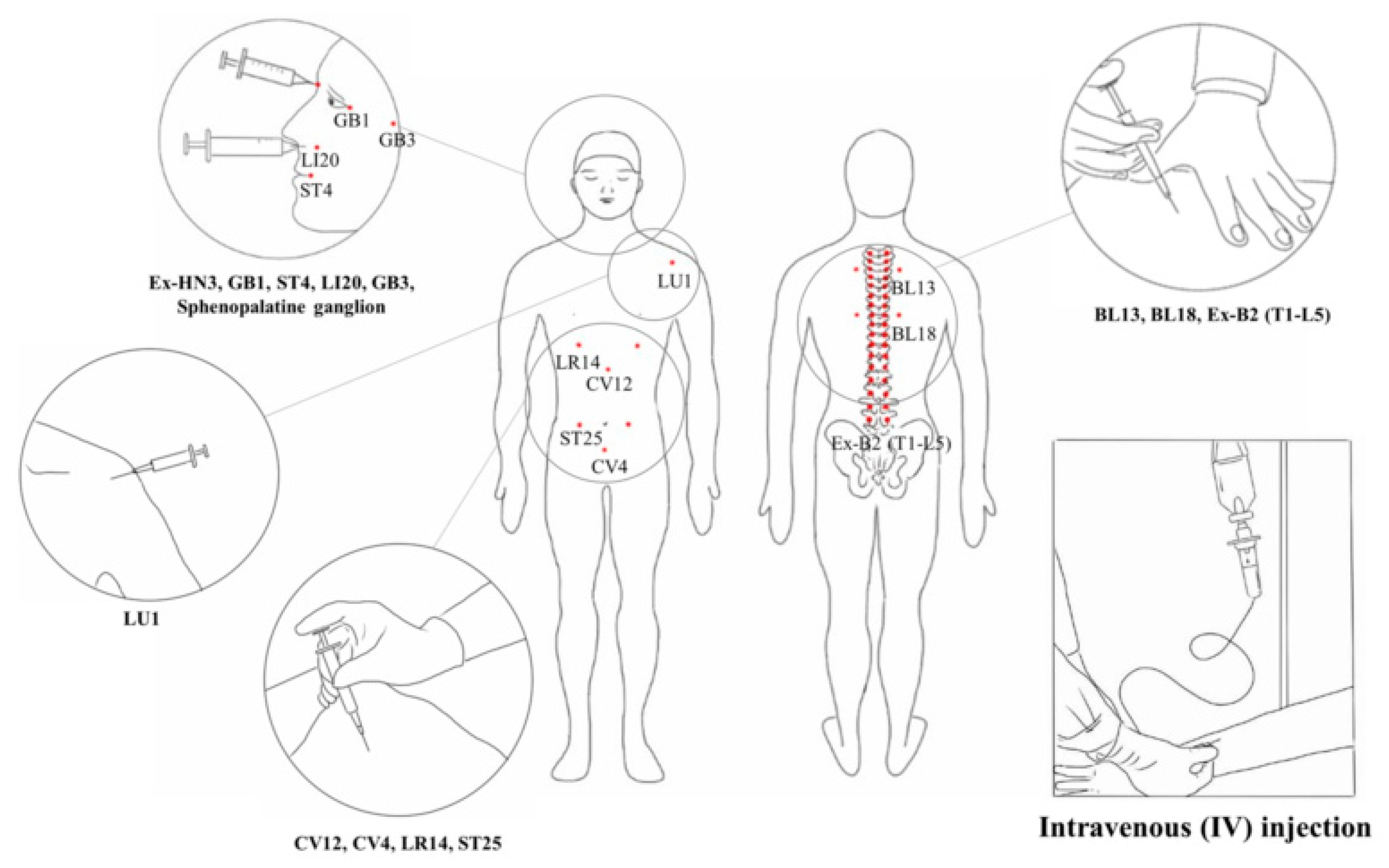
3.4.3. Ginseng Pharmacopuncture Interventions
3.4.4. Outcome Measures
3.4.5. Changes in Clinical Outcomes: Examples from Case Reports
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Park, S.Y.; Park, J.H.; Kim, H.S.; Lee, C.Y.; Lee, H.J.; Kang, K.S.; Kim, C.E. Systems-level mechanisms of action of Panax ginseng: A network pharmacological approach. J. Ginseng Res. 2018, 42, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.C. Immune Pharmacopuncturology; Meridian Medicine Publishing Company: Daejeon, Korea, 2009; pp. 1–475. [Google Scholar]
- Jeong, H.S.; Lim, C.S.; Cha, B.C.; Choi, S.H.; Kwon, K.R. Component analysis of cultivated ginseng, cultivated wild ginseng, and wild ginseng and the change of ginsenoside components in the process of red ginseng. J. Pharmacopunct. 2010, 13, 63–77. [Google Scholar] [CrossRef][Green Version]
- Sun, H.; Liu, F.; Sun, L.; Liu, J.; Wang, M.; Chen, X.; Xu, X.; Ma, R.; Feng, K.; Jiang, R. Proteomic analysis of amino acid metabolism differences between wild and cultivated Panax ginseng. J. Ginseng Res. 2016, 40, 113–120. [Google Scholar] [CrossRef]
- Sung, I.J.; Ghimeray, A.K.; Chang, K.J.; Park, C.H. Changes in Contents of Ginsenoside Due to Boiling Process of Panax ginseng C.A. Mayer. Korea J. Plant Res. 2013, 26, 726–730. [Google Scholar] [CrossRef][Green Version]
- Lee, S.M.; Bae, B.S.; Park, H.W.; Ahn, N.G.; Cho, B.G.; Cho, Y.L.; Kwak, Y.S. Characterization of Korean Red Ginseng (Panax ginseng Meyer): History, preparation method, and chemical composition. J. Ginseng Res. 2015, 39, 384–391. [Google Scholar] [CrossRef]
- Park, J.Y.; Lee, D.S.; Kim, C.E.; Shin, M.S.; Seo, C.S.; Shin, H.K.; Hwang, G.S.; An, J.M.; Kim, S.N.; Kang, K.S. Effects of fermented black ginseng on wound healing mediated by angiogenesis through the mitogen-activated protein kinase pathway in human umbilical vein endothelial cells. J. Ginseng Res. 2018, 42, 524–531. [Google Scholar] [CrossRef]
- Jin, X.; Che, D.B.; Zhang, Z.H.; Yan, H.M.; Jia, Z.Y.; Jia, X.B. Ginseng consumption and risk of cancer: A meta-analysis. J. Ginseng Res. 2016, 40, 269–277. [Google Scholar] [CrossRef]
- Hernandez-Garcia, D.; Granado-Serrano, A.B.; Martin-Gari, M.; Naudi, A.; Serrano, J.C. Efficacy of Panax ginseng supplementation on blood lipid profile. A meta-analysis and systematic review of clinical randomized trials. J. Ethnopharmacol. 2019, 243, 112090. [Google Scholar] [CrossRef]
- Lee, K.H.; Cho, Y.Y.; Kim, S.; Sun, S.H. History of research on pharmacopuncture in Korea. J. Pharmacopunct. 2016, 19, 101. [Google Scholar]
- Korean Pharmacopuncture Institute. Pharmacopuncturology-Principles and Clinical Applications; Elsevier Korea: Seoul, Korea, 2011; pp. 76–89. [Google Scholar]
- Lee, D.Y.; Choi, B.S.; Lee, I.H.; Kim, J.H.; Gwon, P.S. Comparison of index compounds content and antioxidative activity of Wild Ginseng Pharmacopuncture by extraction method. Korean J. Intern. Med. 2018, 39, 313–322. [Google Scholar] [CrossRef][Green Version]
- Kang, S.K.; Lee, H.J.; Park, Y.B. Experimental studies on the effect of ginseng radix aqua-acupuncture. Int. Symp. East-West Med. 1989, 1989, 61–83. [Google Scholar]
- Lim, C.; Kwon, K.; Lee, K. Plexiform neurofibroma treated with pharmacopuncture. J. Pharmacopunct. 2014, 17, 74. [Google Scholar] [CrossRef]
- Lee, K.; Yu, J.; Sun, S.; Kwon, K.; Lim, C. A 4-week, repeated, intravenous dose, toxicity test of mountain ginseng pharmacopuncture in sprague-dawley rats. J. Pharmacopunct. 2014, 17, 27–35. [Google Scholar] [CrossRef]
- Liu, P. A clinical controlled trial of ginseng polysaccharide pharmacopuncture in the treatment of depressive disorder. Neimong J. Tradit. Chin. Med. 2017, 36, 103. [Google Scholar]
- Sung, S.H.; Shin, B.C.; Park, M.J.; Kim, K.H.; Kim, J.W.; Ryu, J.Y.; Park, J.K. Current Status of Management on Pharmacopuncture in Korea through Introduction of an Accreditation System. J. Pharmacopunct. 2019, 22, 75–82. [Google Scholar]
- Kwon, K.A. Clinical Study on the Effects of Intravenous Wild Ginseng Herbal Acupuncture on the Human Body. J. Pharmacopunct. 2004, 7, 15–26. [Google Scholar]
- Lee, H.Y.; You, J.S.; Yook, T.H.; Hong, K.E. The Effects of Distilled Astragali Radix Herbal Acupuncture, Wild Ginseng Herbal Acupuncture and Rehmannia Glutinosa Herbal Acupuncture on Vital Sign—A Randomized, Placebo-controlled, Double-blind Clinical Trial. J. Acupunct. Res. 2007, 24, 207–217. [Google Scholar]
- Lee, E.S.; Oh, J.Y.; Kim, Y.J.; Yu, A.M.; Jang, S.H.; Cho, H.S.; Kim, K.H.; Lee, S.D.; Kim, K.S.; Kim, E.J. The Clinical Study about Qualitative and Quantitative Characteristics of Acupuncture Sensation According to the Type of Pharmacopuncture: Study about BUM Pharmacopuncture, Mountain Ginseng Pharmacopuncture and Sciatica No. 5 Pharmacopuncture. J. Korean Acupunct. Moxib. Soc. 2013, 30, 25–39. [Google Scholar] [CrossRef]
- Choi, Y.N.; Oh, J.Y.; Cho, H.S.; Kim, K.H.; Kim, K.S.; Lee, S.D.; Kim, E.J. Research on the Amount of Stimulus Differences According to Pharmacopuncture Injected dose and Characters Method. Acupuncture 2015, 32, 89–95. [Google Scholar] [CrossRef]
- Roh, J.D.; Kim, L.H.; Song, B.Y.; Yook, T.H. The Effects of distilled Wild Ginseng Herbal Acupuncture on the Heart Rate Variability(HRV). J. Pharmacopunct. 2008, 11, 55–69. [Google Scholar] [CrossRef]
- Yook, T.; Yu, J.; Lee, H.; Song, B.; Kim, L.; Roh, J.; Shin, J.; Lim, S. Comparing the effects of distilled Rehmannia glutinosa, Wild Ginseng and Astragali Radix pharmacopuncture with heart rate variability (HRV): A randomized, sham-controlled and double-blind clinical trial. J. Acupunct. Meridian Stud. 2009, 2, 239–247. [Google Scholar] [PubMed]
- Lee, J.B. The Effects of Distilled Rehmannia Glutinosa and Wild Ginseng Pharmacopuncture CV17, CV4 on Heart Rate Variability—A Randomized and Double-Blind Clinical Trial. Ph.D. Thesis, Department of Korean Medicine Graduate School of Woosuk University, Wanju, Korea, 2012. [Google Scholar]
- Seol, H.; Song, B.Y.; Yook, T.H. The Effects of Panax Ginseng Radix Pharmacopuncture and Zizyphi Spinosi Semen Pharmacopuncture on the Heart Rate Variability. J. Acupunct. Res. 2009, 26, 19–28. [Google Scholar]
- Kim, G.C.; Park, S.W.; Kim, Y.S. Effect of heart rate variability, pulse wave velocity in women of breast cancer patients care by mountain cultivated ginseng pharmacopuncture. J. Korea Inst. Orient. Med. Diagn. 2011, 15, 245–260. [Google Scholar]
- Park, S.W.; Kim, Y.S.; Hwang, W.D.; Kim, G.C. Effect of mountain cultivated ginseng pharmacopuncture on Heart Rate Variability (HRV), Pulse Wave Velocity (PWV) in middle aged women. J. Acupunct. Res. 2011, 28, 97–105. [Google Scholar]
- Park, S.W.; Kim, Y.S.; Hwang, W.D.; Kim, G.C. Effect of pulse-wave factors in middle aged women by mountain cultivated ginseng pharmacopuncture original articles. J. Pharmacopunct. 2011, 14, 35–49. [Google Scholar] [CrossRef]
- Lee, D.H.; Kwon, K.R. Analysis of Serum Proteom after Intravenous Injection of cultivated wild ginseng pharmacopuncture. J. Pharmacopunct. 2006, 9, 17–37. [Google Scholar]
- Kang, T.S.; Lee, S.G.; Kwon, K.R. Analysis of Serum proteom before and after Intravenous Injection of wild ginseng herbal acupuncture. J. Pharmacopunct. 2004, 7, 5–25. [Google Scholar]
- Yu, J.; Sun, S.; Lee, K.; Kwon, K. Single-dose Toxicity of Water-soluble Ginseng Pharmacopuncture Injected Intramuscularly in Rats. J. Pharmacopunct. 2015, 18, 76–85. [Google Scholar]
- Yu, J.S.; Sun, S.H.; Lee, K.H.; Kwon, K.R. Intravenous Toxicity Study of Water-soluble Ginseng Pharmacopuncture in SD Rats. J. Pharmacopunct. 2015, 18, 38–44. [Google Scholar]
- Jo, H.G.; Jung, P.S.; Kim, H.Y.; Bae, S.Y.; Jo, M.J.; Shin, J.H.; Han, S.H.; Na, J.I.; Sul, J.U.; Lee, S.Y. Case of Suspected Drug-Induced Liver Injury after Intravenous Wild Ginseng Pharmacopuncture. J. Physiol. Pathol. Korean Med. 2014, 28, 102–106. [Google Scholar] [CrossRef]
- Kwon, K.R.; Park, C.W.; Ra, M.S.; Cho, C.K. Clinical observation of multiple metastatic cancer patient with hepatocellular carcinoma treated with cultivated wild ginseng herbal acupuncture therapy. J. Korean Acupunct. Moxib. Soc. 2005, 22, 211–217. [Google Scholar]
- Lee, D.H.; Kim, S.S.; Seong, S.; Kim, N.; Han, J.B. Korean medicine therapy as a substitute for chemotherapy for metastatic breast cancer: A case report. Case Rep. Oncol. 2015, 8, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Kim, C.W.; Lee, K.H. A case report of monitoring PSA level changes in two prostate cancer patients treated with mountain ginseng pharmacopuncture and sweet bee venom along with western anticancer therapy. J. Pharmacopunct. 2011, 14, 81–88. [Google Scholar] [CrossRef]
- Park, B.K.; Cho, C.K.; Kwon, K.R.; Yoo, H.S. A Case Report for Stage IIIB Squamous Cell Lung Carcinoma Patient Treated with Cultured Wild Ginseng Pharmacopuncture Therapy. J. Pharmacopunct. 2007, 10, 143–147. [Google Scholar] [CrossRef]
- Bang, S.H.; Kwon, K.R.; Yoo, H.S. Two Cases of Non-Small Cell Lung Cancer Treated with Intravenous Cultivated Wild Ginseng Pharmacopuncture. J. Pharmacopunct. 2008, 11, 13–19. [Google Scholar] [CrossRef]
- Lee, J.H.; Kwon, K.R.; Cho, C.K.; Han, S.S.R.; Yoo, H.S. Advanced cancer cases treated with cultivated wild ginseng phamacopuncture. J. Acupunct. Meridian Stud. 2010, 3, 119–124. [Google Scholar] [CrossRef][Green Version]
- Kwon, K.R.; Kim, H.; Kim, J.S.; Yoo, H.S.; Cho, C.K. Case series of non-small cell lung cancer treated with mountain Ginseng pharmacopuncture. J. Acupunct. Meridian Stud. 2011, 4, 61–68. [Google Scholar] [CrossRef][Green Version]
- Kim, K.; Choi, Y.S.; Joo, J.C.; Moon, G. A case report for lung cancer patient showing remission treated with cultivated Wild Ginseng pharmacopuncture. J. Pharmacopunct. 2011, 14, 33–37. [Google Scholar] [CrossRef]
- Im, C.R.; Kwon, K.; Sur, Y.C.; Bang, S.H.; Kim, S.S.; Seong, S. A case of hepatic and pulmonary metastatic colorectal cancer patient treated by traditional korean therapy and XELOX chemotherapy. J. Korean Tradit. Oncol. 2012, 17, 17–25. [Google Scholar]
- Ha, T.H.; Seong, S.; Lee, D.H.; Kim, S.S. Improved case of recurred and metastatic ascending colon cancer by combination of Oriental medical therapy and FOLFIRI chemotherapy. J. Physiol. Pathol. Korean Med. 2013, 27, 148–151. [Google Scholar]
- Han, J.B.; Ha, T.H.; Kim, S.S.; Seong, S. Case of complete remission of breast cancer metastasized to lung treated by traditional Korean therapy. J. Physiol. Pathol. Korean Med. 2013, 27, 818–822. [Google Scholar]
- Kang, H.J.; Yoon, J.W.; Park, J.H.; Cho, C.K.; Yoo, H.S. Cancer pain control for advanced cancer patients by using autonomic nerve pharmacopuncture. J. Pharmacopunct. 2014, 17, 62. [Google Scholar] [CrossRef] [PubMed]
- Yun, H.; Cho, C.; Yoo, H.; Bang, S. A Case Report of Unresectable Pancreatic Carcinoma Patient for Relieving Cancer Related Pain and Improving Quality of Life by Korean Medical Treatment. J. Korean Tradit. Oncol. 2013, 18, 9–15. [Google Scholar]
- Lee, D.H.; Seong, S.; Kim, S.S.; Han, J.B. A case of stage IV non-small cell lung cancer treated with Korean medicine therapy alone. Case Rep. Oncol. 2013, 6, 574–578. [Google Scholar] [CrossRef]
- Park, J.H.; Jeon, H.J.; Kang, H.J.; Jeong, I.S.; Cho, C.K.; Yoo, H.S. Cancer-related fatigue in patients with advanced cancer treated with autonomic nerve pharmacopuncture. J. Acupunct. Meridian Stud. 2015, 8, 142–146. [Google Scholar] [CrossRef][Green Version]
- Kim, J.S.; Lee, H.J.; Lee, S.H.; Lee, B.H. Recurrent Oligodendroglioma Treated with Acupuncture and Pharmacopuncture. J. Acupunct. Meridian Stud. 2015, 8, 147–151. [Google Scholar] [CrossRef]
- Kim, H.R.; Jeong, H.R.; Jang, C.Y.; Woo, C.; Ha, Y.J.; Moon, G.; Baek, D.G. Cirrhosis after liver lobectomy managed with traditional Korean medical treatment: A case report. J. Int. Korean Med. 2015, 36, 410–418. [Google Scholar]
- Lee, D.H.; Kim, S.S.; Seong, S. A Case Report of Metastatic Breast Cancer Treated with Korean Medicine Therapy as a Substitute for Chemotherapy. Case Rep. Oncol. 2017, 10, 27–36. [Google Scholar] [CrossRef]
- Kim, Y.J.; Park, D.I.; Kwon, K.R. Case report on the improvement of liver functions by mountain cultivated wild ginseng pharmacopuncture. J. Pharmacopunct. 2009, 12, 107–112. [Google Scholar] [CrossRef]
- Ryu, Y.J.; Lee, K.H.; Kwon, K.R.; Lee, Y.H.; An, J.C.; Sun, S.H.; Lee, S.J. Mountain ginseng pharmacopuncture treatment on three amyotrophic lateral sclerosis patients. J. Pharmacopunct. 2010, 13, 119–128. [Google Scholar] [CrossRef]
- Han, J.H.; Kim, S.S.; Seong, S.; Bang, S.H. A Case of Intractable Cervical Dysplasia Patient Treated with Korean Medicine for Three Months. Korean J. Obstet. Gynecol. 2012, 25, 154–160. [Google Scholar]
- Park, J.H.; Oh, D.J.; Jang, S.H.; Hur, H.S. A Clinical Case Report on a Patient of Acute Disseminated Encephalomyelitis Using Korean Medicine. J. Korean Med. Rehabil. 2014, 24, 141–153. [Google Scholar]
- Li, Y. Preliminary study on 100 cases of allergic rhinitis treated by ginseng pharmacopuncture on sphenopalatine ganglion. Xinjiang Tradit. Chin. Med. 1994, 2, 25–26. [Google Scholar]
- Kwon, K.R. Anticancer effect of mountain ginseng Pharmacopuncture to the nude mouse oflung carcinoma induced by NCI-H460 human non-small cell lung cancer cells. J. Pharmacopunct. 2010, 13, 5–14. [Google Scholar] [CrossRef]
- Cho, B.J.; Kwon, K.R. Effects of Cultivated Wild Ginseng Herbal Acupuncture to the serum cytokine on Hepatic Metastatic Model using Colon26-L5 Carcinoma Cells. J. Pharmacopunct. 2006, 9, 127–137. [Google Scholar]
- Baek, S.H.; Lee, I.H.; Kim, M.J.; Kim, E.J.; Ha, I.H.; Lee, J.H.; Lee, J.W. Component analysis and toxicity study of combined cultivated wild ginseng pharmacopuncture. J. Int. Korean Med. 2015, 36, 189–199. [Google Scholar]

| Groups | Contents | Ingredients |
|---|---|---|
| Saponin | Saponin (3–6%) |
|
| Non-saponin | N-containing substances (12–15%) |
|
| Fat-soluble components (1–2%) |
| |
| Carbohydrates (50–60%) |
| |
| Others | Ash (4–6%) |
|
| Vitamin (0.05%) |
|
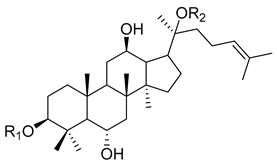
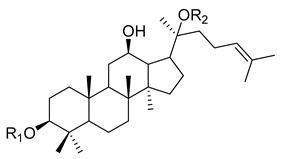 | ||
|---|---|---|
| Ginsenoside | R1 | R2 |
| ginsenoside-Ra1 | - glc(2→1)glc | - glc(6→1)arap(4→1)xyl |
| ginsenoside-Ra2 | - glc(2→1)glc | - glc(6→1)araf(2→1)xyl |
| ginsenoside-Ra3 | - glc(2→1)glc | - glc(6→1)glc(3→1)xyl |
| ginsenoside-Rb1 | - glc(2→1)glc | - glc(6→1)glc |
| ginsenoside-Rb2 | - glc(2→1)glc | - glc(6→1)arap |
| ginsenoside-Rb3 | - glc(2→1)glc | - glc(6→1)xyl |
| ginsenoside-Rc | - glc(2→1)glc | - glc(6→1)araf |
| ginsenoside-Rd | - glc(2→1)glc | - glc |
| ginsenoside-Rg3 | - glc(2→1)glc | - H |
| ginsenoside-F2 | - glc | - glc |
| ginsenoside-Rh2 | - glc | - H |
| ginsenoside-R1 | - glc(2→1)glc(6)Ac | - glc(6→1)glc |
| ginsenoside-Rs1 | - glc(2→1)glc(6)Ac | - glc(6→1)arap |
| ginsenoside-Rs2 | - glc(2→1)glc(6)Ac | - glc(6→1)araf |
| ginsenoside-Rs3 | - glc(2→1)glc(6)Ac | - H |
| ginsenoside-Rb1 | - glc(2→1)glc(6)Ma | - glc(6→1)glc |
| ginsenoside-Rb2 | - glc(2→1)glc(6)Ma | - glc(6→1)arap |
| ginsenoside-Rc | - glc(2→1)glc(6)Ma | - glc(6→1)araf |
| ginsenoside-Rd | - glc(2→1)glc(6)Ma | - glc |
| ginsenoside-R4 | - glc(2→1)glc | - glc(6→1)glc(6→1)xyl |
| ginsenoside-Fa | - glc(2→1)glc(2→1)xyl | - glc(6→1)glc |
| ginsenoside-X VII | - glc | - glc(6→1)glc |
 | ||
|---|---|---|
| Ginsenoside | R1 | R2 |
| ginsenoside-Re | - glc(2→1)rha | - glc |
| ginsenoside-Rf | - glc(2→1)glc | - H |
| 20-gluco-ginsenoside-Rf | - glc(2→1)glc | - glc |
| ginsenoside-Rg1 | - glc | - glc |
| ginsenoside-Rg2 | - glc(2→1)rha | - H |
| ginsenoside-Rh1 | - glc | - H |
| ginsenoside-F1 | - H | - glc |
| ginsenoside-F3 | - H | - glc(6→1)arap |
| ginsenoside-F5 | - H | - glc(6→1)araf |
| ginsenoside-R1 | - glc(2→1)xyl | - glc |
| ginsenoside-R2 | - glc(2→1)xyl | - H |
| ginsenoside-R3 | - glc –glc(6→1) | - glc |
| ginsenoside-R6 | - glc –glc(6→1) | - glc* |
| No. Author (Year), Article Type | Disorder (Symptoms), n *, Gender, Age | Treatment (1, 2, 3…: Serial Treatment or Group; a, b, c…: Combined Treatment) | Significant Results | |||
|---|---|---|---|---|---|---|
| Pharmacopuncture | Co-Interventions | |||||
| Herb, Location, Dose, Injection Methods | Duration, Number | Interventions | Duration, Number | |||
| Case report/series in cancer patients | ||||||
| 1. Kwon (2005) [34] | Hepatocellular carcinoma, liver cirrhosis, hepatitis B+, lung metastasis (abdominal discomfort), m, 41 | a. Cultivated wild ginseng, 0.5–1 cc, IV (at the points of BL13, BL18, LR14, CV12) total 4–40 cc | 5 months, 5 times/week | b. Moxibustion, CV4, CV6 c. Cupping | - | CT: near elimination of the cancer cells metastasized into lungs |
| 2. Park (2007) [37] | Squamous cell carcinoma Stage 3B (severe cough, dyspnea, shoulder discomfort), m, 58 | 2. Cultivated wild ginseng, 10 mL, IV 3. Cultivated wild ginseng, 10 mL, PI (LU1) 4. Cultivated wild ginseng, 10 mL, PI (LU1) | 2. 54 days, 1/day 3. 6 days, 1/day 4. 28 days, - | 1. Herbal medicine | - | 1. CT: aggravation of cancer 2. CT: mass size increased and invaded the mediastinum after the first trial, stabilized after 54 treatments 3. Discontinue the treatment due to a slow of speech and hemiplegia (cerebral infarction) 4. CT: mass invaded the heart Deceased |
| 3. Bang (2008) [38] | Case 1. Lung cancer (adenocarcinoma, Stage T2N3 3B, cough, phlegm), f, 68 | a. Cultivated wild ginseng, 20 mL, IV | a. 8 months, 1/day | b. Herbal medication (globule, Hangamdan) c. Herbal medication (granule, decoction) | b. 3/day, 8 months | Overall decrease/maintain of cough and phlegm CT, PET-CT: stabilized mass size |
| Case 2. NSCLC stage T1N2 3A, m, 64 | Cultivated wild ginseng, IV | a. 5 months | - | - | Overall decrease/maintain of cough, weakness, phlegm CT, PET-CT: stabilized mass size | |
| 4. Lee (2010) [39] | Lung cancer (n = 3), colorectal cancer (n = 2), stomach cancer (n = 1), malignant mesothelioma (n = 1) (f, 1), median 56 | Cultivated wild ginseng, 20 mL, IV | 14 days, 1/day (1 cycle) | - | - | median survival days: 544; 1 year survival rate: 57.1%; drop out (n = 2), progressive (n = 2), stable disease (n = 3) |
| Case 1: Colon cancer stage 3B, ECOG 3 | 2 cycles | - | - | Median survival days: 26 days; drop out | ||
| Case 2: Mesothelioma stage 4, ECOG 3 | 2 cycles | - | - | Median survival days: 56 days; progressive disease; increased size and number of mass | ||
| Case 3: NSCLC stage 4, ECOG 3 | 1 cycle | - | - | Median survival days: 140 days; drop out | ||
| Case 4: Gastric carcinoma stage 4, ECOG 2, m, 55 | 13 cycles | - | - | Median survival days: 544 days; stable disease; no changes of stomach mass, increased liver metastasis | ||
| Case 5: Colon cancer stage 4, ECOG 2 | 3 cycles | - | - | Median survival days: 596 days; progressive disease; increased rectal mass, liver and lung metastasis | ||
| Cace 6: NSCLC stage 3A, ECOG 1, m, 63 | 13 cycles | - | - | Median survival days: 718 days; stable disease; no changes (early)/increased mass (later) | ||
| Case 7: NSCLC stage 3, ECOG 1, f, 67 | 21 cycles | - | - | Median survival days: 898 days; stable disease; slight increased mass in left lower lobe (early)/no changes (later), right adrenal gland metastasis (later) | ||
| 5. Lee (2011) [36] | Prostate cancer, n = 2 (f, 0), mean 52 | a. Cultivated wild ginseng, 20 mL, IV | - | b. AKDH pharmacopuncture c. Sweet BV pharmacopuncture | - | |
| Case 1. Prostate adenocarcinoma (T3bN0M0, fatigue, pain, nocturia, impotence), m, 51 | a. | 2/week | b. AKDH pharmacopuncture c. Acupuncture | - | Decreased PSA and prostate volume; prostate cancer disappeared, remain cancer in a seminal vesicle and left apex, T3bN0M0 (25 weeks) PSA maintained, no discomfort symptoms (30 months) | |
| Case 2. Prostate cancer T3bN0M1c (metastasis to bone/lung, pelvic pain, knee pain, short breath, weakness, abnormal urination), m, 53 | a. | 5/week | c. Sweet BV pharmacopuncture d. Acupuncture e. Medication | 2–3/week | Ups and downs of PSA and symptoms, stable health condition (19 months) | |
| 6. Kwon (2011) [40] | NSCLC, n = 6 (f, 5), median 67 | a. Wild ginseng, 20 mL, IV b. Wild ginseng, 10 mL, PI (LU1) | a, b. 4 weeks/cycle | |||
| Case 1. Adenocarcinoma stage 4, m, 64 | 2. b 3. a | 2. 2 cycles 3. 4 cycles | 1. Chemotherapy, operation | - | 2. CT: tumor progressed (progressed disease) 3. normal range of blood cancer markers, tumor growth has stopped for 9 weeks, increase of mass (stable disease) | |
| Case 2. Squamous cell carcinoma stage 4, m, 60 | 2. a | 4 cycles | 1. Chemotherapy, operation | - | 1. tumor progressed 2. tumor growth has shown stable condition (4 weeks; stable disease); CT: slight progress | |
| Case 3. Squamous cell carcinoma stage 4, f, 62 | b. | 2 cycles | CT: Tumor markedly increased (progressed disease) | |||
| Case 4. Breathing difficulties with weight loss, m, 70 | b. | 1 cycle | - | - | CT: Tumor size slightly increased (stable disease) | |
| Case 5. Squamous cell carcinoma stage 3A, m, 65 | 1. a 2. b | 1. 5 cycles 2. 2 cycles | - | - | 1. CT: tumor growth showed stable condition for 20 weeks (stable disease) 2. CT: tumor size slightly increased (progressed disease) | |
| Case 6. Squamous cell carcinoma stage 3B (dyspnea, hemoptysis, fever and weight loss), m, 78 | a. | 16 cycles | - | - | a. all cancer related symptoms and the tumor growth showed stable condition (16 months, stable disease) | |
| 7. Kim (2011) [41] | Squamous cell lung carcinoma T2bN1, stage 2B (dyspnea, phlegm, hemoptysis, weight lose), m, 75 | a. Cultivated wild ginseng, 0.5 mL, PI (CV12, CV4, BL13) | 1/day, 12 days or 2/week, 3 weeks | b. Acupuncture c. Cupping d. Moxibustion | Dyspnea maintained, phlegm and hemoptysis disappeared CT: decrease of mass | |
| 8. Im (2012) [42] | Colorectal cancer (metastasis in liver, lung, ovary, chest pain, insomnia), f, 47 | a. Cultivated wild ginseng, 10 mL, IV | 3–5/week | b. Soram pharmacopuncture ** c. Herbal medicine d. Moxibustion e. Acupuncture | b. 1, 2, 5/day | Decreased pain (2 weeks) Decreased size of metastasized mass in lung (13 weeks) CEA, CA19-9 decreased |
| 9. Ha (2013) [43] | Colorectal adenocarcinoma stage 4B (recurrence, metastasis in liver, spleen, lung), m, 42 | a. Cultivated wild ginseng, 30 mL, IV | a. 3/week, 34 weeks | b. FOLFIRI chemotherapy | b. 12/2 weeks, 31 weeks | Recurrence of colorectal adenocarcinoma and metastasis disappeared, no adverse events reported to FOLFIRA chemotherapy |
| 10. Han (2013) [44] | Breast cancer (recurrence, metastasis in lung, chest pain, cough, short breath, shoulder pain, excessive sweating), f, 53 | a. Cultivated wild ginseng, 10 mL, IV | 3/week | b. Cordyceps militaris pharmacopuncture c. Acupuncture d. Moxibustion e. Herbal medicine | 3/week | Chest pain and cough disappeared (1 month) Tubercles in lung disappeared (6 weeks) PET-CT: Recurrence disappeared (50 days) |
| 11. Yun (2013) [46] | Pancreatic cancer (abdominal pain, indigestion, post-prandial pain, abdominal inflation), f, 68 | a. Cultivated wild ginseng, 20 mL, NA | 3/week, 5 months | b. Medication c. Herbal medication (globule, Hangamdan; globule, Ginseno-pil) d. Acupuncture | b. 4/week c. 3/day | Decreased pain and intake of analgesics (5 months) Improvement of all symptom; CT: decreased hydrothorax and mass size (stable disease) |
| 12. Lee (2013) [47] | NSCLC squamous cell carcinoma stage 4 (cough, dyspnoea, weakness), m, 79 | a. Wild ginseng, 10 mL, IV | a. 1/week, 7 months | b. Berbal medication (globule, Soramdan) c. Cordyceps sinensis pharmacopuncture, 10 mL, IV d. Trichosanthes kirilowii pharmacopuncture, 10mL, IV | b. 2/week, 7 months c, d. 1/week, 7 months | ECOG scale maintained (4 weeks) and decreased (5 week) Tumor size decreased |
| 13. Kang (2014) [45] | Case 1. Signet ring cell carcinoma stage 4 (abdominal pain, diarrhea, nausea, fatigue, bloating, heartburn), f, 41 | a. Cultivated wild ginseng, 1 mL, subcutaneous injection (EX-B2) | a. 20 times | b. Acupuncture c. Analgesics | b. 46 days, 1/day | Reduced analgesics (10 days) Improvement of symptoms and adverse events of analgesics (sedation, nausea) |
| Case 2. bronchioalveolar carcinoma, adenocarcinoma stage 4 (dizziness, nausea, vomiting, insomnia, fatigue, diarrhea, dyspnea), f, 51 | a. | a. 5 times | b. Chemotherapy | - | Pain disappeared, able to perform daily activities without analgesics | |
| Case 3. tubulovillous adenoma, neuroendocrine carcinoma stage 4A (diarrhea, abdominal discomfort, rash, dyspnea), m, 81 | a. | a. 13 times | b. Analgesics | - | Decreased pain, improved health condition, reduced analgesics Worsening of liver and kidney functions | |
| 14. Park (2015) [48] | Case 1. Thymic cancer (fatigue, fever, anorexia, itching), f, 40 | a. Wild ginseng, 1 mL, total 20 mL, subcutaneous injection (EX-B2) | a. 1/2–3 days, 4 times | - | - | Increased white blood cells Decreased erythrocyte sedimentation rate, thyroglobulin Ag level, Korean version of the Revised Piper Fatigue Scale total score |
| Case 2. cervical cancer (fatigue, lower limb edema and pain, lower back pain, gait disturbance), f, 61 | a. Wild ginseng, 1 mL, total 20 mL, subcutaneous injection (EX-B2) | a. 12 times | - | - | Increased total protein level, lower limb pain Decreased g-glutamyl transferase level, C-reactive protein, fatigue | |
| 15. Lee (2015) [35] | Breast cancer stage 3, f, 46 | 2, 3, 4-a. Wild ginseng, 10 mL, IV | all 3 times/week except 4-e. daily | 1. Neoadjuvant chemotherapy, breast conservation surgery, adjuvant radiotherapy 2-b. Cordyceps sinensis 2-c. Trichosanthes kirilowii 2-d. Euonymus alatus 2-e. Astragalus membranaceus pharmacopuncture 3-b. C. sinensis 3-c. T. kirilowii 3-d. E. alatus 3-e. P. vulgaris pharmacopuncture 4-b. E. alatus 4-c. Soram nebulizer solution 4-d. Nebulizer solution 4-e. Herbal medication (globule, Hangamdan) | 1. nodule in right upper lung increased 2. 15 mm lymphadenopathies in right paratracheal, interlobar hilar, and subcarinal area 3. lymph node decreased to less than 10 mm 4. no recurrence was observed | |
| 16. Kim (2015a) [49] | recurrent oligodendroglioma (hemiparesis, dysarthria, severe daily seizures, headache, drowsiness, constipation, dysuria), m, 54 | a. Wild ginseng, 0.5 mL, PI (BL13) | a. 1/week | b. BV pharmacopuncture, GV20, EX-HN1, 0.2 mL c. Acupuncture d. Herbal medicine (fermented red ginseng solution) | c, d. 1/day | brain MRI: decreased tumor size (18 months) free from seizure, left-side hemiparesis was improved, symptoms disappeared (21 months) maintain the treated status without any deterioration (5 years) |
| 17. Kim (2015b) [50] | Hepatocellular carcinoma (stage 2, cirrhosis, abdominal pain, lack of appetite, sleep disorder, fatigue, weakness), m, 57 | 3-a. Cultivated wild ginseng, 0.25-0.5cc, PI (BL18, CV12, ST25) | 3-a. 1/day | 1. Ascites puncture, albumin injection, diuretic 2. Ascites puncture 3-b. Medication 3-c. Herbal medication (globule, Hangamdan) 3-d. Acupuncture | various | 1. unmanageable liver index and ascites 2. Ascites and symptoms maintained 3. symptoms relieved, abdominal circumference decreased (22 days) normal ALP (50 days) Ascites reduced (64 days) Symptoms disappeared except slight and seldom abdominal pain, increase of ALP/AST (71 days) Symptoms disappeared, normal blood test index (340 days) Ascites almost disappeared (351 days) |
| 18.Lee (2017) [51] | Right breast invasive ductal carcinoma T1N0M0 (metastases in the liver, retroperitoneum, mesentery, pelvic bones, cranium, whole-body bone, pleural effusion, back pain, jaundice, ascites), f, 45 | 4-a. Wild ginseng, IV or PI (acupoint) | 1/day or 2 days | 1.Chemotherapy (trastuzumab, paclitaxel) 2. Medication (trastuzumab, vancomycin, tazoperan) 3. Chemotherapy (trastuzumab, paclitaxel) 4-b. Herbal medication (Soramdan) 4-c. Herbal medication (Jeobgoldan) | 4-b. 1/day or 3/week 4-c. 2/day | 1. - 2. CT, bone scan: reduction of liver metastases, pleural effusion increased, new lesions on the sternum, ribs, acetabulum 3. find vancomycin-resistant enterococci 4-a, b, c. CT: reduction of the liver metastasis lesion, scar, contraction of the liver parenchyma PET-CT: reduction of the tumor size in the right breast, the primary site of the tumor, right axillary lymph node, liver, bone metastases |
| Case report/series in patients with other diseases | ||||||
| 19. Li (1994) [56] | Allergic rhinitis, n = 100 (f, 44), range 16-51 | Panax ginseng, 4 mL, Sphenopalatine ganglion | 1/week, 4–12 week | - | - | Complete symptom relief without recurrence (68%), significant symptom relief (29%), not significant results (3%) |
| 20. Kim (2009) [52] | Case 1. Behçet’s disease (canker sore, edema, chronic fatigue, drug-induced hepatitis), m, 47 | a. Cultivated wild ginseng, 10 mL, IV | 1–2/week | b. Acupuncture c. AKDH pharmacopuncture | - | Improvement of all symptoms |
| Case 2. Drug-induced hepatitis (weakness, fatigue, low back pain, indigestion), m, 51 | a. Cultivated wild ginseng, 10 mL, IV | 1/week | b. Acupuncture c. AKDH pharmacopuncture | - | Improvement of symptoms, satisfied by the results | |
| Case 3. Hepatocirrhosis (bleeding, weakness, indigestion), m, 72 | Cultivated wild ginseng, 20 mL, IV | 3/week | - | - | Improvement of all symptoms, normalization of AST/ALT | |
| 21. Ryu (2010) [53] | ALS | a. Cultivated wild ginseng, 20 mL, IV | a. 1–2/2 weeks or 1/2 days | b. AKDH pharmacopuncture c. Sweet BV pharmacopuncture | b. 1–2/2 weeks or 1/2 days | |
| Case 1. ALS (myo-atrophy, tetraparesis), f, 51 | Improvement of general health condition without muscle weakness (2–3 months) Slow movement, became pessimistic and depressed, slight muscle atrophy (4–5.5 months) Decrease of thigh thickness, ALS functional rating scale, ALS severity score (motor ability) (26.5 weeks) | |||||
| Case 2. ALS (myo-atrophy, tetraparesis), m, 47 | Increased strength of limbs and ALS functional rating scale (2–6 weeks) Increase of muscular strength, decrease of thickness of thigh/upper limbs and ALS functional rating scale (6–9 weeks) | |||||
| Case 3. ALS (myo-atrophy, tetraparesis), f, 70 | Increased pain (2–3 months) Decreased low back pain, discomfort (4–5 months) Decreased weight, thickness of body, muscular strength, ALS functional rating scale, ALS severity scale | |||||
| 22. Han (2012) [54] | Cervical dysplasia (genital itching, HPV 52 positive), f, 49 | a. Cultivated wild ginseng, 30 mL, IV | 6/week, 3 months | b. Herbal medicine c. Moxibustion | Improvement of symptoms, negative HPV 52 test | |
| 23. Lim (2014) [14] | Plexiform neurofibroma (general weakness, coldness of the hands/feet, skin rashes), f, 16 | 2-a. Wild ginseng, 20 mL, IV | 1/2 weeks | 1. Surgeries 2-b. Sweet BV, 5 mL, intracutaneous | 2-b. 1/2 weeks | 2. Tumor stopped growing, range of motion improved, no bones or organs affected |
| 24. Park (2014) [55] | Acute demyelinating encephalomyelitis (paraplegia), m, 16 | a. Cultivated wild ginseng, 20 mL, IV | 1/week, 8 weeks | b. Acupuncture c. Moxibustion d. Herbal medicine e. Rehabilitation f. Medication | b. 2/day c. 1/day | Increased muscular strength, Improved Modified Bathel Index, Normalized muscular motor ability, Decreased pain |
| 25. Lee (2015) [21] | Skin wrinkles, n = 23 (f, 20), mean 34 | Cultivated wild ginseng, 0.5cc, PI (Ex-HN3, GB1, GB3, LI20, ST4) | 2/week, 5 times | - | - | Decreased width and depth of skinfold |
| Randomized controlled trial in patients with other diseases | ||||||
| 26. Liu (2017) [16] | Depression, Intervention group: n = 51 (f, 28), range 22-54 Control group: n = 51 (f, 29), range 21-56 | Intervention group: 1.a. ginseng polysaccharide, 1 mL, PI (BL15, BL20, ST36), 5/week 1.b. Bupleurum pharmacopuncture, 1 mL, PI (BL18, LR8) 1.c. paroxetine | 30 days (10 days/session, 3 sessions) | Control group: 2. paroxetine (40–60 mg/day, 1–2 times/day) | - | Intervention group was significantly improved symptoms more than control group at 2, 4, 6 weeks after treatment (The Hamilton Depression Scale (HAMD), p < 0.05). Intervention group showed significantly higher total effective rate (98%) than control group (82%). Intervention group reported significantly lower adverse events (7 cases) than control group (21cases). |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, I.-S.; Kang, K.S.; Kim, S.-Y. Panax ginseng Pharmacopuncture: Current Status of the Research and Future Challenges. Biomolecules 2020, 10, 33. https://doi.org/10.3390/biom10010033
Lee I-S, Kang KS, Kim S-Y. Panax ginseng Pharmacopuncture: Current Status of the Research and Future Challenges. Biomolecules. 2020; 10(1):33. https://doi.org/10.3390/biom10010033
Chicago/Turabian StyleLee, In-Seon, Ki Sung Kang, and Song-Yi Kim. 2020. "Panax ginseng Pharmacopuncture: Current Status of the Research and Future Challenges" Biomolecules 10, no. 1: 33. https://doi.org/10.3390/biom10010033
APA StyleLee, I.-S., Kang, K. S., & Kim, S.-Y. (2020). Panax ginseng Pharmacopuncture: Current Status of the Research and Future Challenges. Biomolecules, 10(1), 33. https://doi.org/10.3390/biom10010033







