Identification of miRNAs Enriched in Extracellular Vesicles Derived from Serum Samples of Breast Cancer Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Breast Cancer Patients and Controls
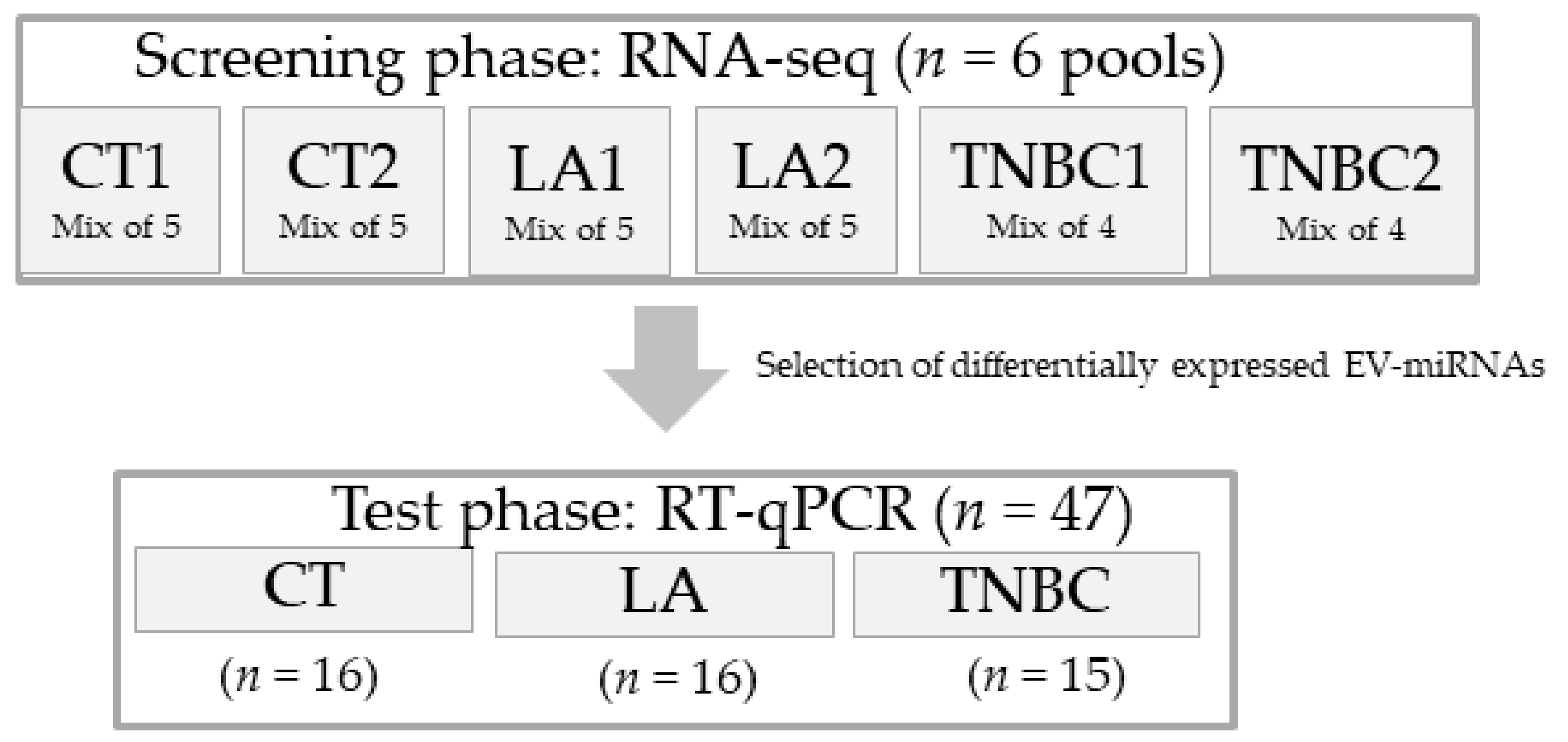
2.2. Study Design
2.3. EV Isolation
2.4. EV Characterization
2.5. RNA Extraction
2.6. Profiling of Small RNA Cargo of EVs
2.7. RT-qPCR
2.8. Statistical Analysis
3. Results
3.1. EVs’ Isolation and Characterization
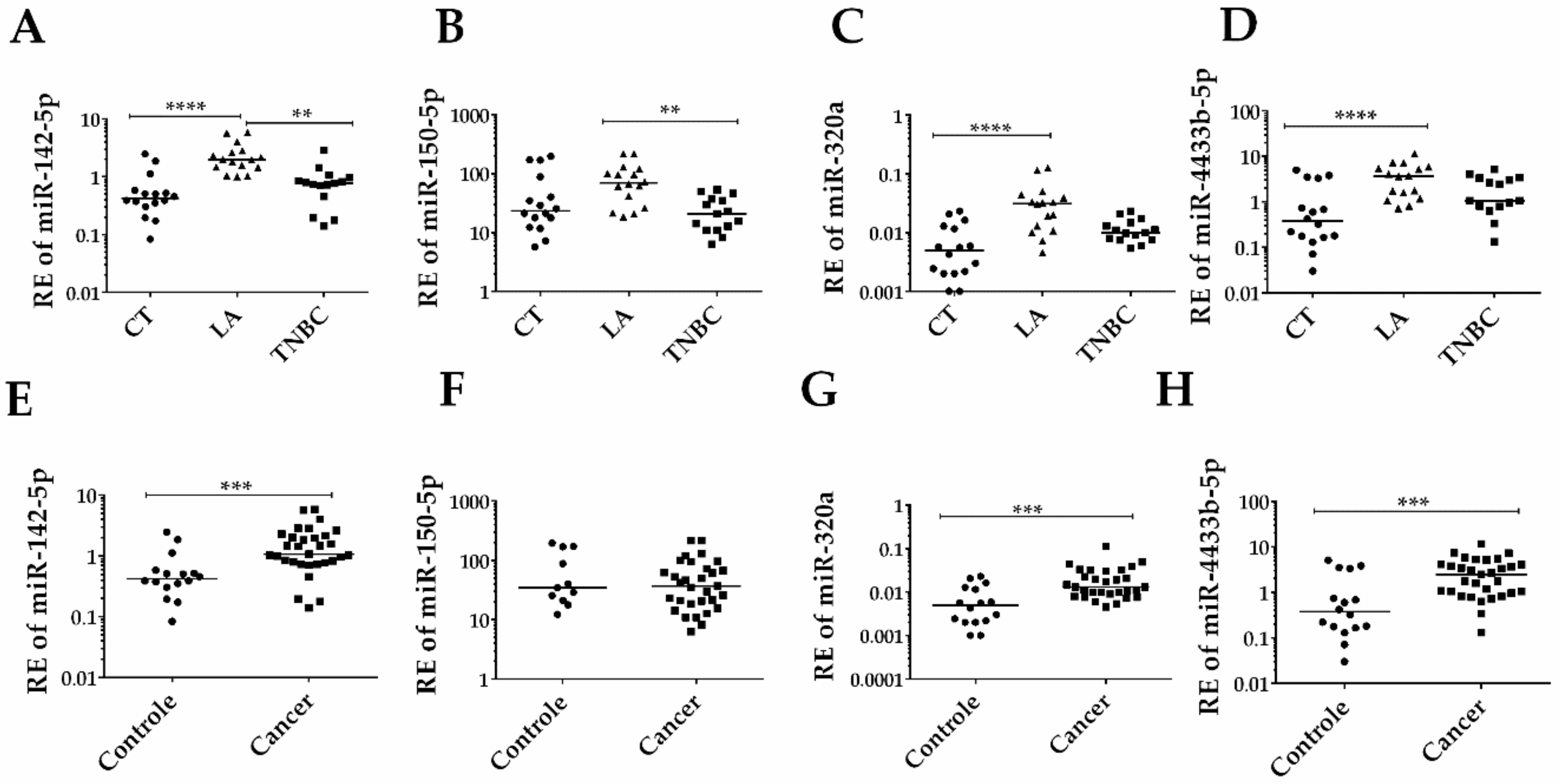
3.2. Differentially Expressed EV-miRNAs Identified by RNA-seq are Suitable Biomarkers for Diagnosis of BC and Its Subtypes
3.3. EV-miRNAs Expression in Individual Serum Samples
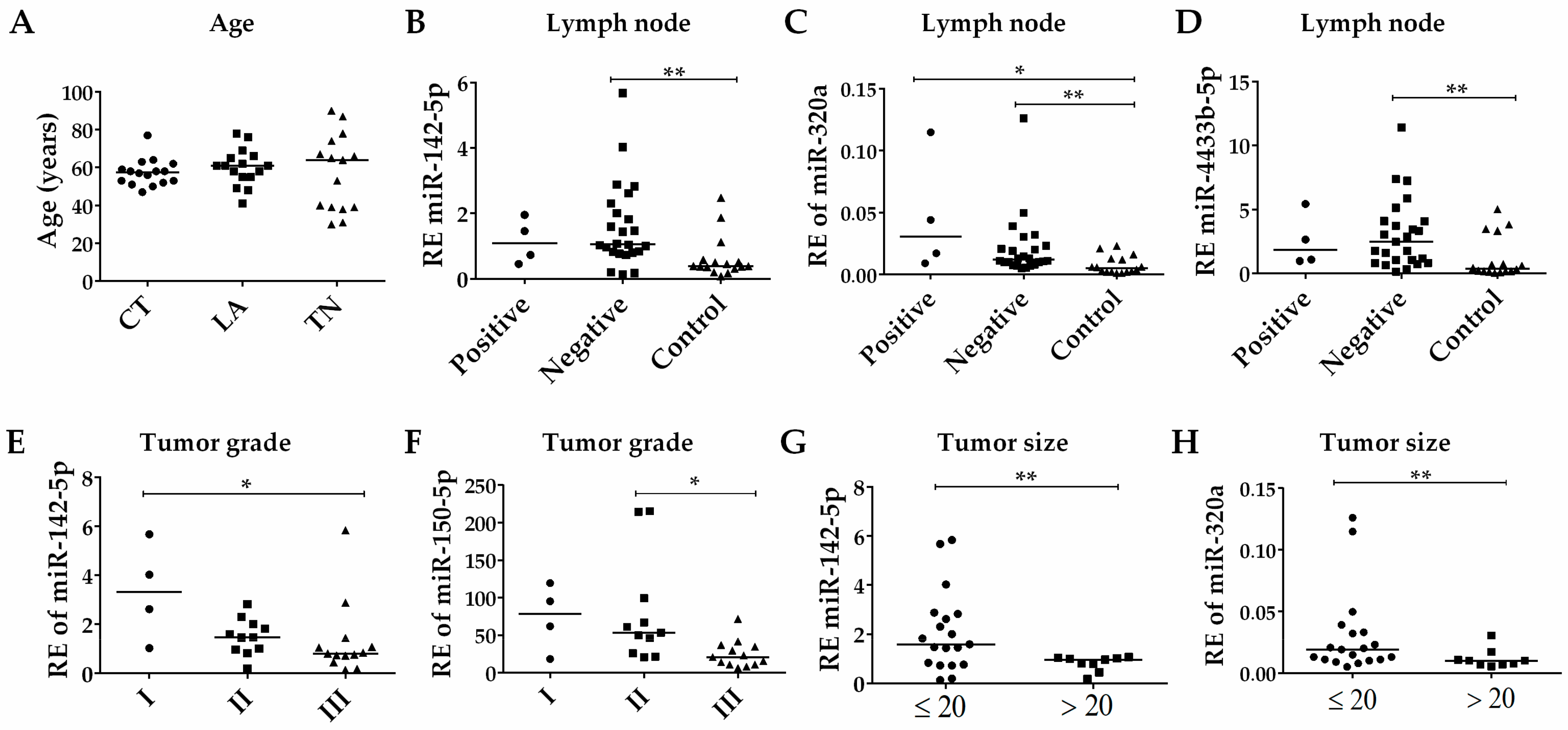
3.4. Association of EV-miRNAs’ Expression with the Clinicopathological Parameters of BC Patients
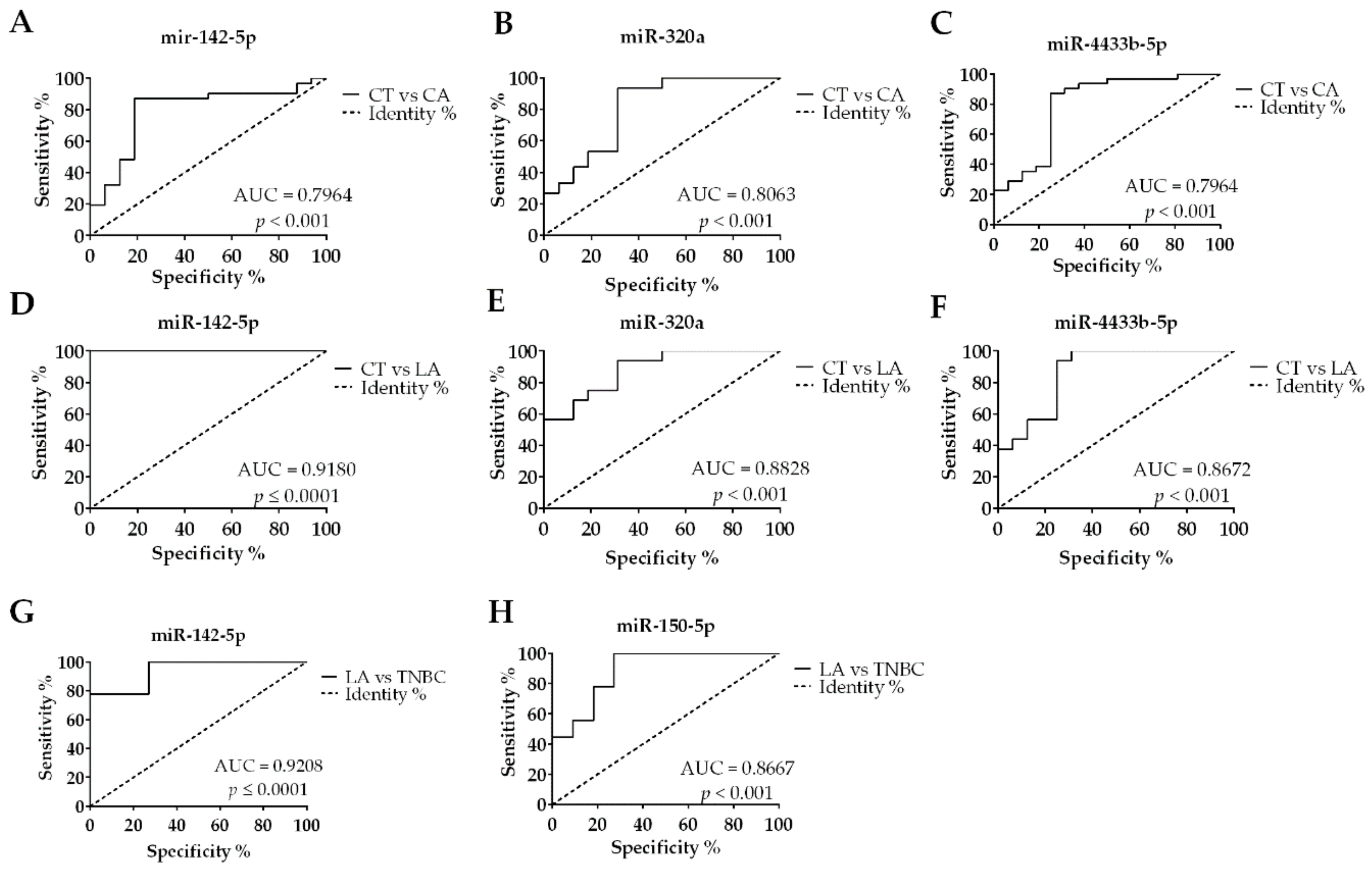
3.5. Diagnostic Potential of EV-miRNAs in Serum Samples
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Caby, M.P.; Lankar, D.; Vincendeau-Scherrer, C.; Raposo, G.; Bonnerot, C. Exosomal-like vesicles are present in human blood plasma. Int. Immunol. 2005, 17, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Mathivanan, S.; Ji, H.; Simpson, R.J. Exosomes: extracellular organelles important in intercellular communication. J. Proteom. 2010, 73, 1907–1920. [Google Scholar] [CrossRef] [PubMed]
- Skog, J.; Wurdinger, T.; van Rijn, S.; Meijer, D.H.; Gainche, L.; Sena-Esteves, M.; Curry, W.T., Jr.; Carter, B.S.; Krichevsky, A.M.; Breakefield, X.O. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat. Cell Biol. 2008, 10, 1470–1476. [Google Scholar] [CrossRef] [PubMed]
- Ambros, V. The functions of animal microRNAs. Nature 2004, 431, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Qian, F.; Yang, X.; Jiang, H.; Chen, Y.; Liu, S. Circulating miR-222 in plasma and its potential diagnostic and prognostic value in gastric cancer. Med. Oncol. 2014, 31, 164. [Google Scholar] [CrossRef]
- Wang, J.; Huang, S.K.; Zhao, M.; Yang, M.; Zhong, J.L.; Gu, Y.Y.; Peng, H.; Che, Y.Q.; Huang, C.Z. Identification of a circulating microRNA signature for colorectal cancer detection. PloS ONE 2014, 9, e87451. [Google Scholar] [CrossRef]
- Hannafon, B.N.; Trigoso, Y.D.; Calloway, C.L.; Zhao, Y.D.; Lum, D.H.; Welm, A.L.; Zhao, Z.J.; Blick, K.E.; Dooley, W.C.; Ding, W.Q. Plasma exosome microRNAs are indicative of breast cancer. Breast Cancer Res. BCR 2016, 18, 90. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018. [Google Scholar] [CrossRef]
- INCA. Estimate/2018 – Cancer Incidence in Brazil; Department, H., Ed.; INCA: Rio de Janeiro, Brazil, 2017; p. 128. [Google Scholar]
- Goldhirsch, A.; Wood, W.C.; Coates, A.S.; Gelber, R.D.; Thurlimann, B.; Senn, H.J. Strategies for subtypes—dealing with the diversity of breast cancer: highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2011, 22, 1736–1747. [Google Scholar] [CrossRef]
- Nishimura, R.; Arima, N. Is triple negative a prognostic factor in breast cancer? Breast Cancer 2008, 15, 303–308. [Google Scholar] [CrossRef]
- Heneghan, H.M.; Miller, N.; Lowery, A.J.; Sweeney, K.J.; Newell, J.; Kerin, M.J. Circulating microRNAs as novel minimally invasive biomarkers for breast cancer. Ann. Surg. 2010, 251, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Kodahl, A.R.; Lyng, M.B.; Binder, H.; Cold, S.; Gravgaard, K.; Knoop, A.S.; Ditzel, H.J. Novel circulating microRNA signature as a potential non-invasive multi-marker test in ER-positive early-stage breast cancer: A case control study. Mol. Oncol. 2014, 8, 874–883. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhou, Y.; Xia, T.; Zhou, X.; Huang, Z.; Zhang, H.; Zhu, W.; Ding, Q.; Wang, S. Circulating microRNAs from the miR-106a-363 cluster on chromosome X as novel diagnostic biomarkers for breast cancer. Breast Cancer Res. Treat. 2018, 170, 257–270. [Google Scholar] [CrossRef] [PubMed]
- Mar-Aguilar, F.; Mendoza-Ramirez, J.A.; Malagon-Santiago, I.; Espino-Silva, P.K.; Santuario-Facio, S.K.; Ruiz-Flores, P.; Rodriguez-Padilla, C.; Resendez-Perez, D. Serum circulating microRNA profiling for identification of potential breast cancer biomarkers. Dis. Mark. 2013, 34, 163–169. [Google Scholar] [CrossRef]
- Eichelser, C.; Stuckrath, I.; Muller, V.; Milde-Langosch, K.; Wikman, H.; Pantel, K.; Schwarzenbach, H. Increased serum levels of circulating exosomal microRNA-373 in receptor-negative breast cancer patients. Oncotarget 2014, 5, 9650–9663. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.K.; Park, S.R.; Jung, B.K.; Jeon, Y.K.; Lee, Y.S.; Kim, M.K.; Kim, Y.G.; Jang, J.Y.; Kim, C.W. Exosomes derived from mesenchymal stem cells suppress angiogenesis by down-regulating VEGF expression in breast cancer cells. PloS ONE 2013, 8, e84256. [Google Scholar] [CrossRef]
- Sueta, A.; Yamamoto, Y.; Tomiguchi, M.; Takeshita, T.; Yamamoto-Ibusuki, M.; Iwase, H. Differential expression of exosomal miRNAs between breast cancer patients with and without recurrence. Oncotarget 2017, 8, 69934–69944. [Google Scholar] [CrossRef]
- Zhang, G.; Zhang, W.; Li, B.; Stringer-Reasor, E.; Chu, C.; Sun, L.; Bae, S.; Chen, D.; Wei, S.; Jiao, K.; et al. MicroRNA-200c and microRNA- 141 are regulated by a FOXP3-KAT2B axis and associated with tumor metastasis in breast cancer. Breast Cancer Res. Bcr 2017, 19, 73. [Google Scholar] [CrossRef]
- Tamkovich, S.; Tutanov, O.; Efimenko, A.; Grigor’eva, A.; Ryabchikova, E.; Kirushina, N.; Vlassov, V.; Tkachuk, V.; Laktionov, P. Blood Circulating Exosomes Contain Distinguishable Fractions of Free and Cell-Surface-Associated Vesicles. Curr. Mol. Med. 2019, 19, 273–285. [Google Scholar] [CrossRef]
- Thery, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Fehlmann, T.; Backes, C.; Kahraman, M.; Haas, J.; Ludwig, N.; Posch, A.E.; Wurstle, M.L.; Hubenthal, M.; Franke, A.; Meder, B.; et al. Web-based NGS data analysis using miRMaster: a large-scale meta-analysis of human miRNAs. Nucleic Acids Res. 2017, 45, 8731–8744. [Google Scholar] [CrossRef] [PubMed]
- Vlachos, I.S.; Zagganas, K.; Paraskevopoulou, M.D.; Georgakilas, G.; Karagkouni, D.; Vergoulis, T.; Dalamagas, T.; Hatzigeorgiou, A.G. DIANA-miRPath v3.0: deciphering microRNA function with experimental support. Nucleic Acids Res. 2015, 43, W460–W466. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Team, R. RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA, 2016. [Google Scholar]
- Team, R.C. R: A language and environment for statistical computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Anders, S.; Huber, W. Differential expression analysis for sequence count data. Genome Biol. 2010, 11, R106. [Google Scholar] [CrossRef]
- Sugita, B.; Gill, M.; Mahajan, A.; Duttargi, A.; Kirolikar, S.; Almeida, R.; Regis, K.; Oluwasanmi, O.L.; Marchi, F.; Marian, C.; et al. Differentially expressed miRNAs in triple negative breast cancer between African-American and non-Hispanic white women. Oncotarget 2016, 7, 79274–79291. [Google Scholar] [CrossRef]
- Hamam, R.; Hamam, D.; Alsaleh, K.A.; Kassem, M.; Zaher, W.; Alfayez, M.; Aldahmash, A.; Alajez, N.M. Circulating microRNAs in breast cancer: novel diagnostic and prognostic biomarkers. Cell Death Dis. 2017, 8, e3045. [Google Scholar] [CrossRef]
- Wang, F.; Zheng, Z.; Guo, J.; Ding, X. Correlation and quantitation of microRNA aberrant expression in tissues and sera from patients with breast tumor. Gynecol. Oncol. 2010, 119, 586–593. [Google Scholar] [CrossRef]
- Arroyo, J.D.; Chevillet, J.R.; Kroh, E.M.; Ruf, I.K.; Pritchard, C.C.; Gibson, D.F.; Mitchell, P.S.; Bennett, C.F.; Pogosova-Agadjanyan, E.L.; Stirewalt, D.L.; et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl. Acad. Sci. USA 2011, 108, 5003–5008. [Google Scholar] [CrossRef]
- Gao, J.; Zhang, Q.; Xu, J.; Guo, L.; Li, X. Clinical significance of serum miR-21 in breast cancer compared with CA153 and CEA. Chin. J. Cancer Res. = Chung-Kuo Yen Cheng Yen Chiu 2013, 25, 743–748. [Google Scholar] [CrossRef]
- Heneghan, H.M.; Miller, N.; Kelly, R.; Newell, J.; Kerin, M.J. Systemic miRNA-195 differentiates breast cancer from other malignancies and is a potential biomarker for detecting noninvasive and early stage disease. Oncologist 2010, 15, 673–682. [Google Scholar] [CrossRef] [PubMed]
- Schrauder, M.G.; Strick, R.; Schulz-Wendtland, R.; Strissel, P.L.; Kahmann, L.; Loehberg, C.R.; Lux, M.P.; Jud, S.M.; Hartmann, A.; Hein, A.; et al. Circulating micro-RNAs as potential blood-based markers for early stage breast cancer detection. PloS ONE 2012, 7, e29770. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wang, M.; Lin, G.; Sun, S.; Li, X.; Qi, J.; Li, J. Serum microRNA-155 as a potential biomarker to track disease in breast cancer. PloS ONE 2012, 7, e47003. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Chen, X.; Zhu, D.; Luo, Z.; Yang, M. Low Expression of Circulating MicroRNA-34c is Associated with Poor Prognosis in Triple-Negative Breast Cancer. Yonsei Med. J. 2017, 58, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Ng, E.K.; Li, R.; Shin, V.Y.; Jin, H.C.; Leung, C.P.; Ma, E.S.; Pang, R.; Chua, D.; Chu, K.M.; Law, W.L.; et al. Circulating microRNAs as specific biomarkers for breast cancer detection. PloS ONE 2013, 8, e53141. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Hu, Q.; Schrauder, M.; Yan, L.; Wang, D.; Medico, L.; Guo, Y.; Yao, S.; Zhu, Q.; Liu, B.; et al. Circulating miR-148b and miR-133a as biomarkers for breast cancer detection. Oncotarget 2014, 5, 5284–5294. [Google Scholar] [CrossRef]
- Shimomura, A.; Shiino, S.; Kawauchi, J.; Takizawa, S.; Sakamoto, H.; Matsuzaki, J.; Ono, M.; Takeshita, F.; Niida, S.; Shimizu, C.; et al. Novel combination of serum microRNA for detecting breast cancer in the early stage. Cancer Sci. 2016, 107, 326–334. [Google Scholar] [CrossRef]
- Wang, H.; Peng, R.; Wang, J.; Qin, Z.; Xue, L. Circulating microRNAs as potential cancer biomarkers: the advantage and disadvantage. Clin. Epigenet. 2018, 10, 59. [Google Scholar] [CrossRef]
- Palma, J.; Yaddanapudi, S.C.; Pigati, L.; Havens, M.A.; Jeong, S.; Weiner, G.A.; Weimer, K.M.; Stern, B.; Hastings, M.L.; Duelli, D.M. MicroRNAs are exported from malignant cells in customized particles. Nucleic Acids Res. 2012, 40, 9125–9138. [Google Scholar] [CrossRef]
- Pigati, L.; Yaddanapudi, S.C.; Iyengar, R.; Kim, D.J.; Hearn, S.A.; Danforth, D.; Hastings, M.L.; Duelli, D.M. Selective release of microRNA species from normal and malignant mammary epithelial cells. PloS ONE 2010, 5, e13515. [Google Scholar] [CrossRef]
- Hrustincova, A.; Votavova, H.; Dostalova Merkerova, M. Circulating MicroRNAs: Methodological Aspects in Detection of These Biomarkers. Folia Biol. 2015, 61, 203–218. [Google Scholar]
- Chang, J.T.; Wang, F.; Chapin, W.; Huang, R.S. Identification of MicroRNAs as Breast Cancer Prognosis Markers through the Cancer Genome Atlas. PloS ONE 2016, 11, e0168284. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Y.; Jia, S.; Zhang, W.H.; Zhang, Y.; Kang, Y.; Li, P.S. Differential distribution of microRNAs in breast cancer grouped by clinicopathological subtypes. Asian Pac. J. Cancer Prev. Apjcp 2013, 14, 3197–3203. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Li, J.; Sun, M.; Sun, L.; Zhang, X. miRNA expression in breast cancer varies with lymph node metastasis and other clinicopathologic features. IUBMB Life 2014, 66, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Wang, W. MicroRNA1425p modulates breast cancer cell proliferation and apoptosis by targeting phosphatase and tensin homolog. Mol. Med. Rep. 2018, 17, 7529–7536. [Google Scholar] [CrossRef]
- Buffa, F.M.; Camps, C.; Winchester, L.; Snell, C.E.; Gee, H.E.; Sheldon, H.; Taylor, M.; Harris, A.L.; Ragoussis, J. microRNA-associated progression pathways and potential therapeutic targets identified by integrated mRNA and microRNA expression profiling in breast cancer. Cancer Res. 2011, 71, 5635–5645. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.P.; Huang, S.F.; Li, C.F.; Chien, H.T.; Chen, S.C. Differential microRNA expression in breast cancer with different onset age. PloS ONE 2018, 13, e0191195. [Google Scholar] [CrossRef]
- Milioli, H.H.; Tishchenko, I.; Riveros, C.; Berretta, R.; Moscato, P. Basal-like breast cancer: molecular profiles, clinical features and survival outcomes. BMC Med. Genom. 2017, 10, 19. [Google Scholar] [CrossRef]
- Du, F.; Yuan, P.; Zhao, Z.T.; Yang, Z.; Wang, T.; Zhao, J.D.; Luo, Y.; Ma, F.; Wang, J.Y.; Fan, Y.; et al. A miRNA-based signature predicts development of disease recurrence in HER2 positive breast cancer after adjuvant trastuzumab-based treatment. Sci. Rep. 2016, 6, 33825. [Google Scholar] [CrossRef]
- Van Schooneveld, E.; Wildiers, H.; Vergote, I.; Vermeulen, P.B.; Dirix, L.Y.; Van Laere, S.J. Dysregulation of microRNAs in breast cancer and their potential role as prognostic and predictive biomarkers in patient management. Breast Cancer Res. BCR 2015, 17, 21. [Google Scholar] [CrossRef]
- Wang, B.; Yang, Z.; Wang, H.; Cao, Z.; Zhao, Y.; Gong, C.; Ma, L.; Wang, X.; Hu, X.; Chen, S. MicroRNA-320a inhibits proliferation and invasion of breast cancer cells by targeting RAB11A. Am. J. Cancer Res. 2015, 5, 2719–2729. [Google Scholar] [PubMed]
- Yu, J.; Wang, J.G.; Zhang, L.; Yang, H.P.; Wang, L.; Ding, D.; Chen, Q.; Yang, W.L.; Ren, K.H.; Zhou, D.M.; et al. MicroRNA-320a inhibits breast cancer metastasis by targeting metadherin. Oncotarget 2016, 7, 38612–38625. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Wang, L.; Yang, H.; Ding, D.; Zhang, L.; Wang, J.; Chen, Q.; Zou, Q.; Jin, Y.; Liu, X. Rab14 Suppression Mediated by MiR-320a Inhibits Cell Proliferation, Migration and Invasion in Breast Cancer. J. Cancer 2016, 7, 2317–2326. [Google Scholar] [CrossRef] [PubMed]
- Cuk, K.; Zucknick, M.; Heil, J.; Madhavan, D.; Schott, S.; Turchinovich, A.; Arlt, D.; Rath, M.; Sohn, C.; Benner, A.; et al. Circulating microRNAs in plasma as early detection markers for breast cancer. Int. J. Cancer 2013, 132, 1602–1612. [Google Scholar] [CrossRef] [PubMed]
- Freres, P.; Wenric, S.; Boukerroucha, M.; Fasquelle, C.; Thiry, J.; Bovy, N.; Struman, I.; Geurts, P.; Collignon, J.; Schroeder, H.; et al. Circulating microRNA-based screening tool for breast cancer. Oncotarget 2015, 7, 5416–5428. [Google Scholar] [CrossRef]

| Parameters | BC Patients |
|---|---|
| Sample size (n) | 31 |
| Age (mean ± SD) | 58.8 ± 15.5 |
| Range in years | 30–90 |
| Tumor Size (mm) | |
| ≤ 20 | 19 |
| > 20 | 9 |
| Tumor Grade | |
| I | 4 |
| II | 11 |
| III | 13 |
| Lymph Node Metastasis | |
| Positive | 4 |
| Negative | 24 |
| Estrogen Receptor | |
| Positive | 16 |
| Negative | 15 |
| Progesterone Receptor | |
| Positive | 16 |
| Negative | 15 |
| HER2 Overexpression | |
| Positive | 0 |
| Negative | 31 |
| Proliferation Index (Ki-67) | |
| ≤ 14% | 16 |
| > 14% | 13 |
| EV-miRNAs | log2 FC | padj | |
|---|---|---|---|
| CT versus CA | hsa-miR-320a | −2.03 | 6.94 × 10–5 |
| hsa-miR-126-5p | 2.7 | 1.23 × 10−4 | |
| hsa-miR-423-5p | –1.73 | 1.28 × 10−3 | |
| hsa-miR-378a-3p | −2.29 | 1.64 × 10−3 | |
| hsa-miR-185-5p | –1.81 | 1.64 × 10−3 | |
| hsa-miR-150-5p | 2.47 | 1.64 × 10−3 | |
| hsa-miR-4454 | 2.93 | 2.14 × 10−3 | |
| CT versus LA | hsa-miR-320a | −2.23 | 1.33 × 10−6 |
| hsa-miR-423-5p | −1.91 | 2.06 × 10−4 | |
| hsa-miR-744-5p | −1.75 | 4.67 × 10−3 | |
| hsa-miR-103a-3p | −1.52 | 5.55 × 10−3 | |
| hsa-miR-183-5p | 1.4 | 6.55 × 10−3 | |
| hsa-miR-126-5p | 2.31 | 6.55 × 10−3 | |
| hsa-let-7f-5p | 1.23 | 1.20 × 10−2 | |
| CT versus TNBC | hsa-miR-185-5p | −2.2 | 5.26 × 10−6 |
| hsa-miR-195-5p | −2.74 | 5.94 × 10−5 | |
| hsa-miR-150-5p | 3.4 | 5.94 × 10−5 | |
| hsa-miR-126-5p | 3.35 | 6.82 × 10−5 | |
| hsa-miR-320a | −1.79 | 6.98 × 10−5 | |
| hsa-miR-26a-5p | 2.18 | 6.98 × 10−5 | |
| hsa-miR-4454 | 3.61 | 2.07 × 10−3 | |
| LA versus TNBC | hsa-miR-4433b-5p | 2.233 | 2.52 × 10−5 |
| hsa-miR-26a-5p | 1.768 | 8.62 × 10−3 | |
| hsa-miR-142-5p | 1.853 | 1.83 × 10−2 | |
| hsa-let-7f-5p | −1.114 | 3.77 × 10−2 | |
| hsa-miR-484 | 1.275 | 3.77 × 10−2 | |
| hsa-miR-486-5p | 1.252 | 4.17 × 10−2 | |
| hsa-miR-15b-5p | 2.39 | 4.77 × 10−2 |
| KEGG Pathway | p-value | #genes | #miRNAs |
|---|---|---|---|
| Pathways in cancer | 9.95 × 10−5 | 62 | 3 |
| Viral carcinogenesis | 3.10 × 10−5 | 42 | 3 |
| Human T-cell lymphotropic virus type 1 (HTLV-I) infection | 0.044937 | 40 | 3 |
| MicroRNAs in cancer | 0.038582 | 37 | 3 |
| Mitogen-activated protein kinase (MAPK) signaling pathway | 0.03917 | 37 | 3 |
| Transcriptional misregulation in cancer | 0.001051 | 33 | 3 |
| Protein processing in endoplasmic reticulum | 0.028384 | 31 | 3 |
| Thyroid hormone signaling pathway | 0.001618 | 29 | 3 |
| Proteoglycans in cancer | 0.004657 | 29 | 3 |
| RNA transport | 0.029074 | 28 | 3 |
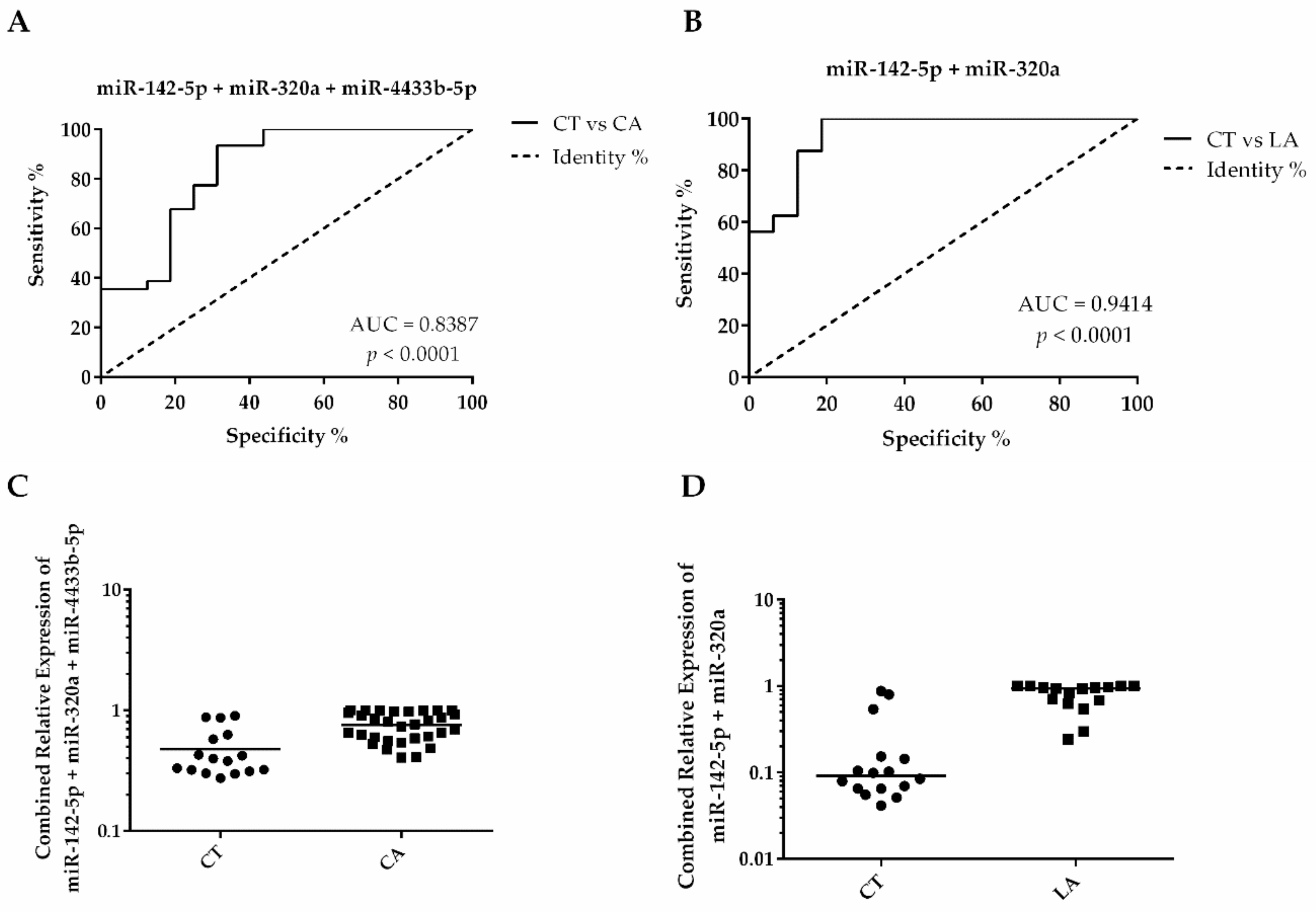
| EV-miRNA | Comparison | AUC | p-Value | Cutoff | Sensitivity | Specificity |
|---|---|---|---|---|---|---|
| miR-142-5p, miR-320a and miR-4433b-5p panel | CT versus CA | 0.8387 | <0.0001 | 0.4504 | 93.55% | 68.75 |
| miR-320a | CT versus CA | 0.8063 | <0.001 | 0.0060 | 93.33% | 68.75% |
| miR-142-5p | CT versus CA | 0.7964 | <0.001 | 0.6435 | 87.10% | 81.25% |
| miR-4433b-5p | CT versus CA | 0.7964 | <0.002 | 0.7578 | 87.10% | 75% |
| miR-142-5p and miR-320a panel | CT versus LA | 0.9410 | <0.0001 | 0.0871 | 100% | 93.80% |
| miR-142-5p | CT versus LA | 0.9180 | <0.0001 | 0.7926 | 100% | 81.25% |
| miR-320a | CT versus LA | 0.8828 | <0.001 | 0.0065 | 93.75% | 68.75% |
| miR-4433b-5p | CT versus LA | 0.8672 | <0.001 | 0.7743 | 93.75% | 75% |
| miR-142-5p | TNBC versus LA | 0.9208 | <0.0001 | 0.6435 | 87.10% | 81.25% |
| miR-150-5p | TNBC versus LA | 0.8667 | <0.001 | 39.3800 | 80% | 75% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ozawa, P.M.M.; Vieira, E.; Lemos, D.S.; Souza, I.L.M.; Zanata, S.M.; Pankievicz, V.C.; Tuleski, T.R.; Souza, E.M.; Wowk, P.F.; Urban, C.d.A.; et al. Identification of miRNAs Enriched in Extracellular Vesicles Derived from Serum Samples of Breast Cancer Patients. Biomolecules 2020, 10, 150. https://doi.org/10.3390/biom10010150
Ozawa PMM, Vieira E, Lemos DS, Souza ILM, Zanata SM, Pankievicz VC, Tuleski TR, Souza EM, Wowk PF, Urban CdA, et al. Identification of miRNAs Enriched in Extracellular Vesicles Derived from Serum Samples of Breast Cancer Patients. Biomolecules. 2020; 10(1):150. https://doi.org/10.3390/biom10010150
Chicago/Turabian StyleOzawa, Patricia M. M., Evelyn Vieira, Débora S. Lemos, Ingrid L. Melo Souza, Silvio M. Zanata, Vânia C. Pankievicz, Thalita R. Tuleski, Emanuel M. Souza, Pryscilla F. Wowk, Cícero de Andrade Urban, and et al. 2020. "Identification of miRNAs Enriched in Extracellular Vesicles Derived from Serum Samples of Breast Cancer Patients" Biomolecules 10, no. 1: 150. https://doi.org/10.3390/biom10010150
APA StyleOzawa, P. M. M., Vieira, E., Lemos, D. S., Souza, I. L. M., Zanata, S. M., Pankievicz, V. C., Tuleski, T. R., Souza, E. M., Wowk, P. F., Urban, C. d. A., Kuroda, F., Lima, R. S., Almeida, R. C., Gradia, D. F., Cavalli, I. J., Cavalli, L. R., Malheiros, D., & Ribeiro, E. M. S. F. (2020). Identification of miRNAs Enriched in Extracellular Vesicles Derived from Serum Samples of Breast Cancer Patients. Biomolecules, 10(1), 150. https://doi.org/10.3390/biom10010150






