A Critical Regulation of Th17 Cell Responses and Autoimmune Neuro-Inflammation by Ginsenoside Rg3
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Mice and EAE Model
2.3. Bone-Marrow-Derived Dendritic Cells (BMDCs) Generation
2.4. In Vitro Th17 Cell Differentiation
2.5. ELISA
2.6. Real-Time RT-PCR
2.7. Flow Cytometry Analysis
2.8. Statistical Analysis
3. Results
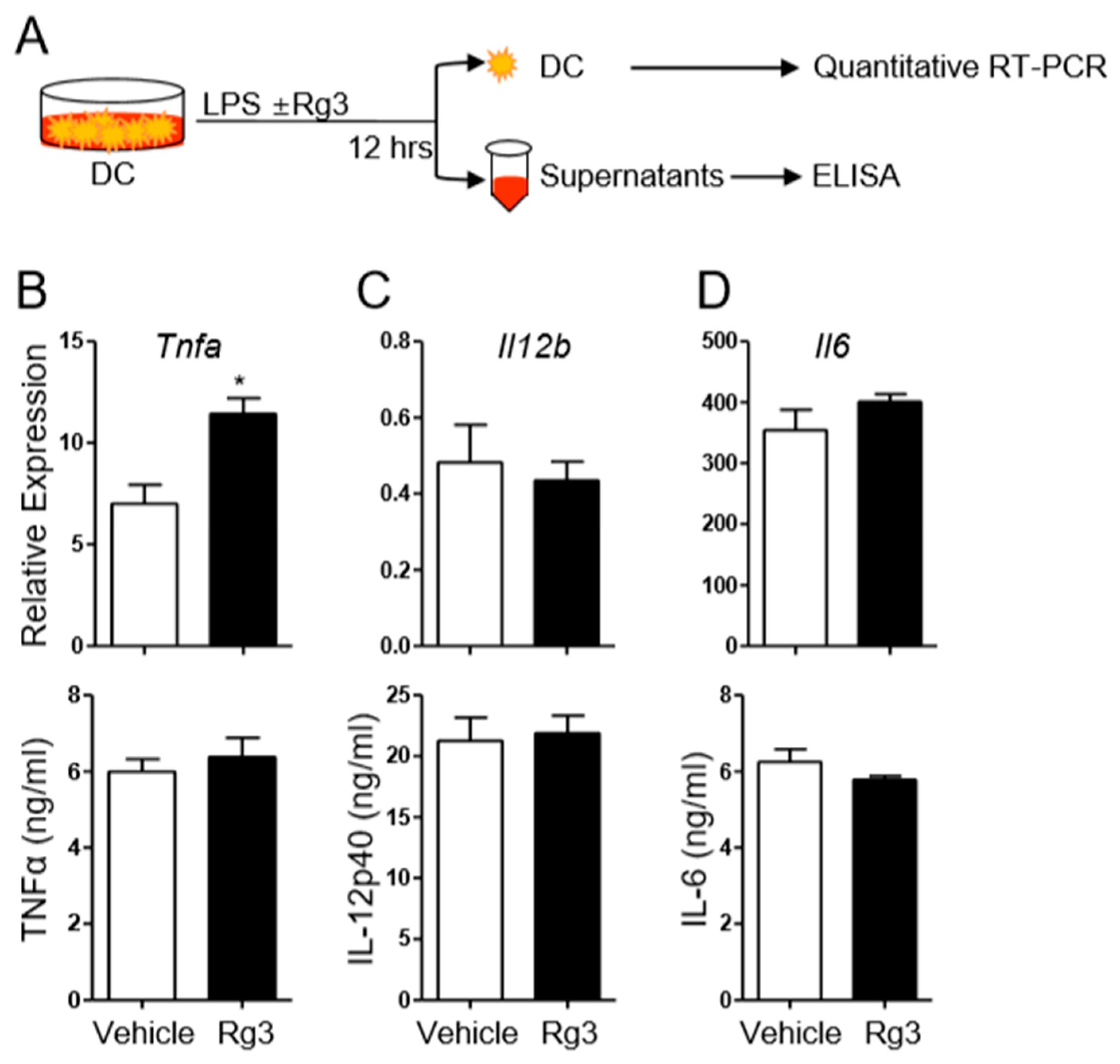
3.1. Rg3 Minimally Affects the Production of Th17-Promoting Cytokines from DCs
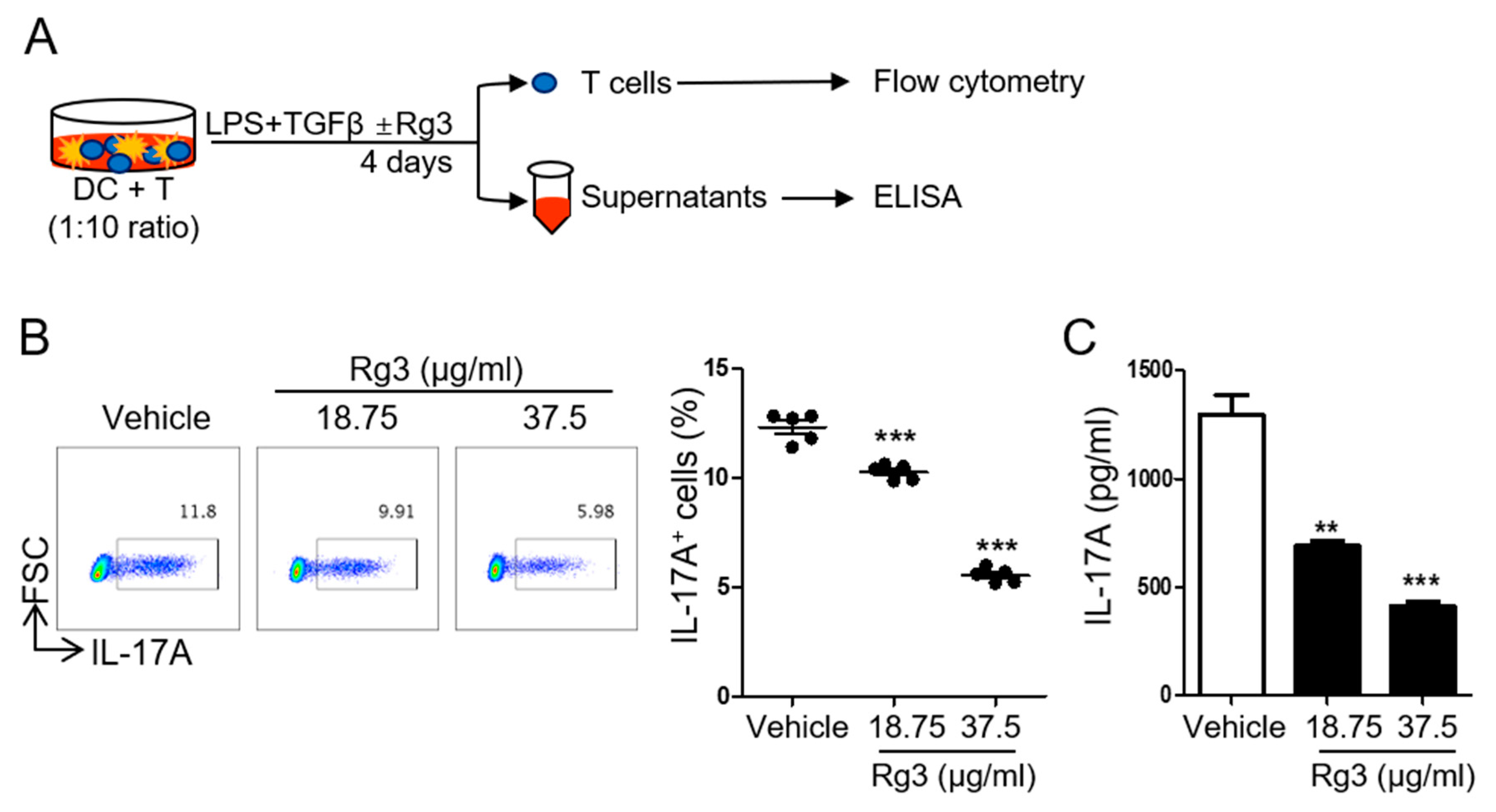
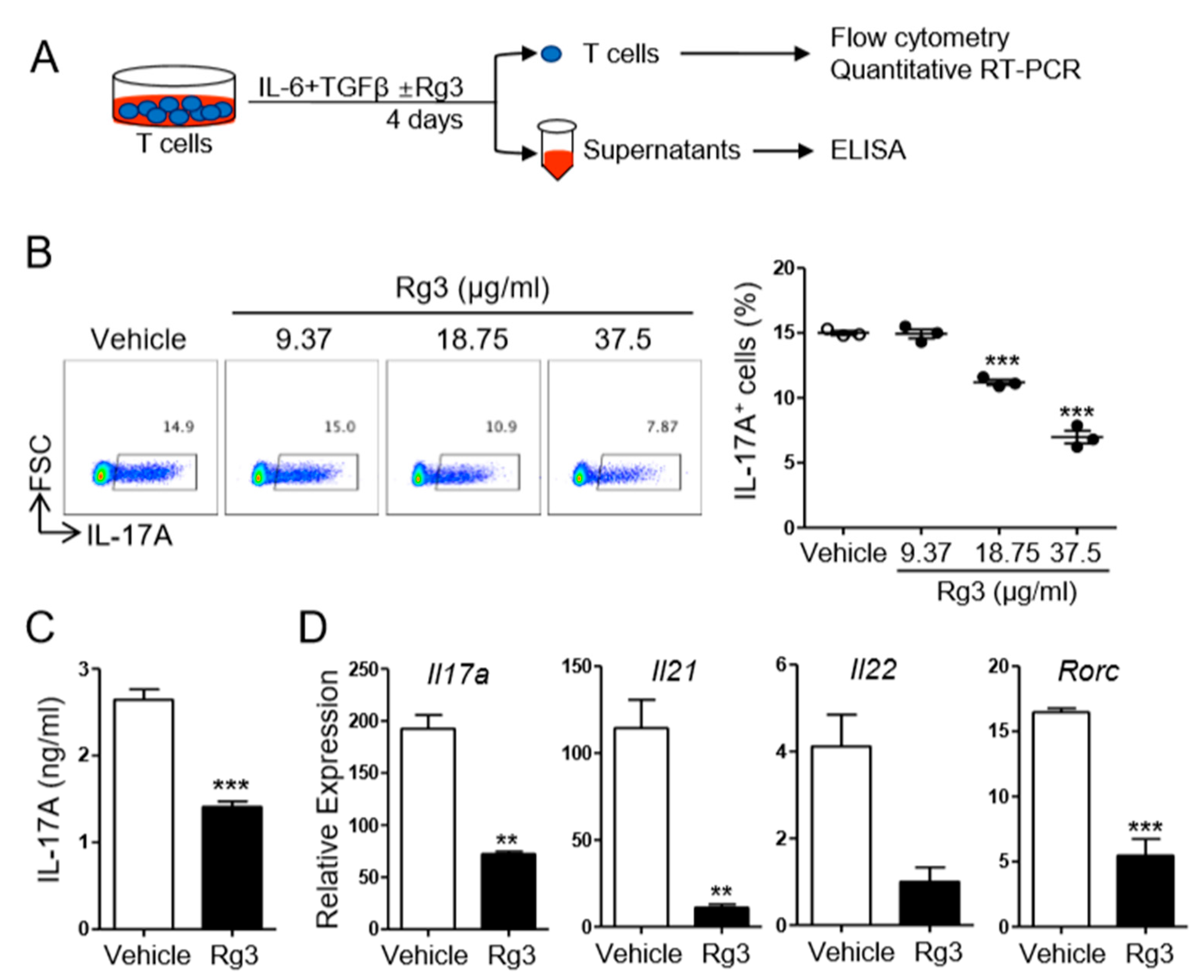
3.2. Rg3 Inhibits Th17 Cell Differentiation in a T-Cell-Intrinsic Manner
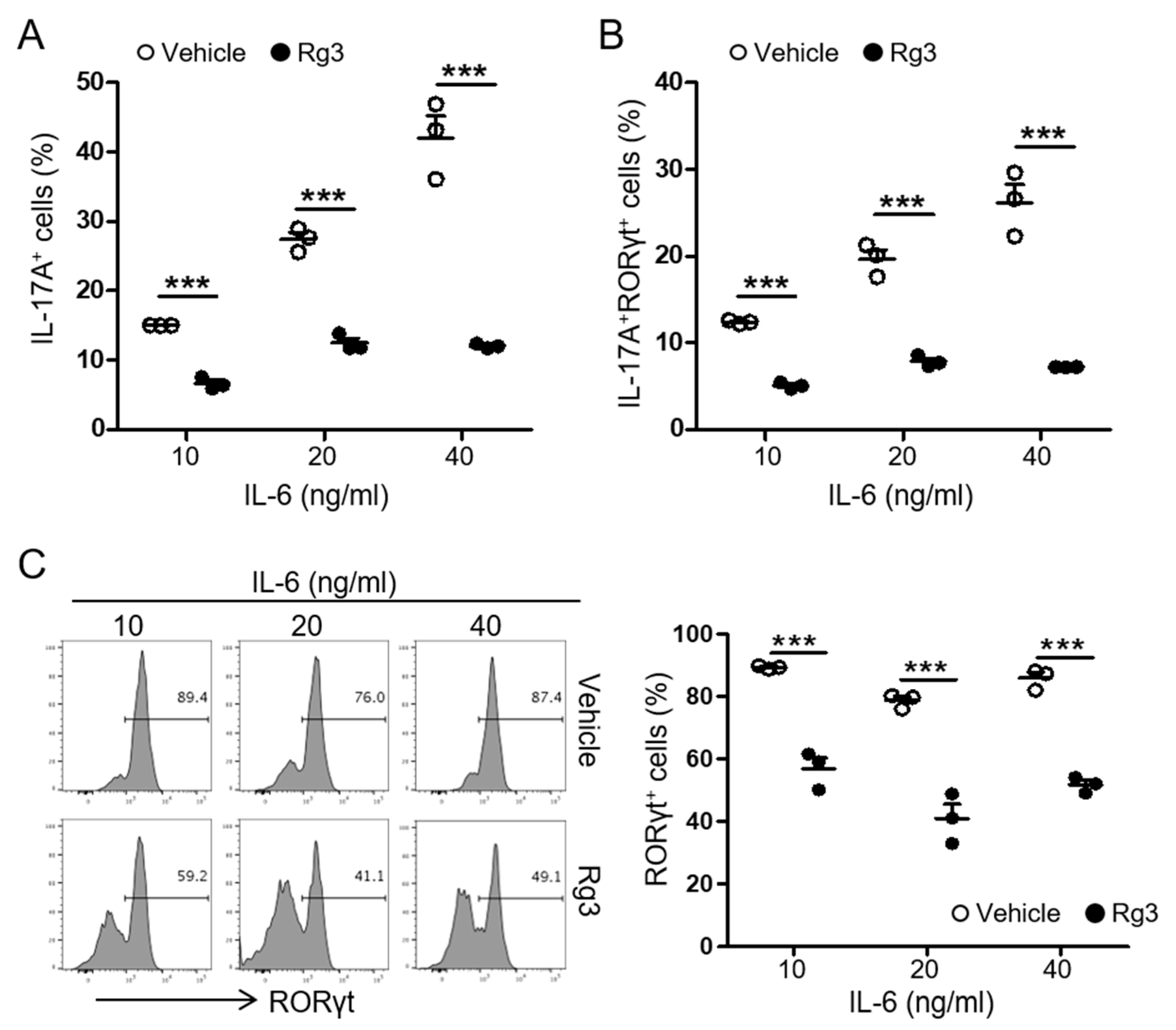
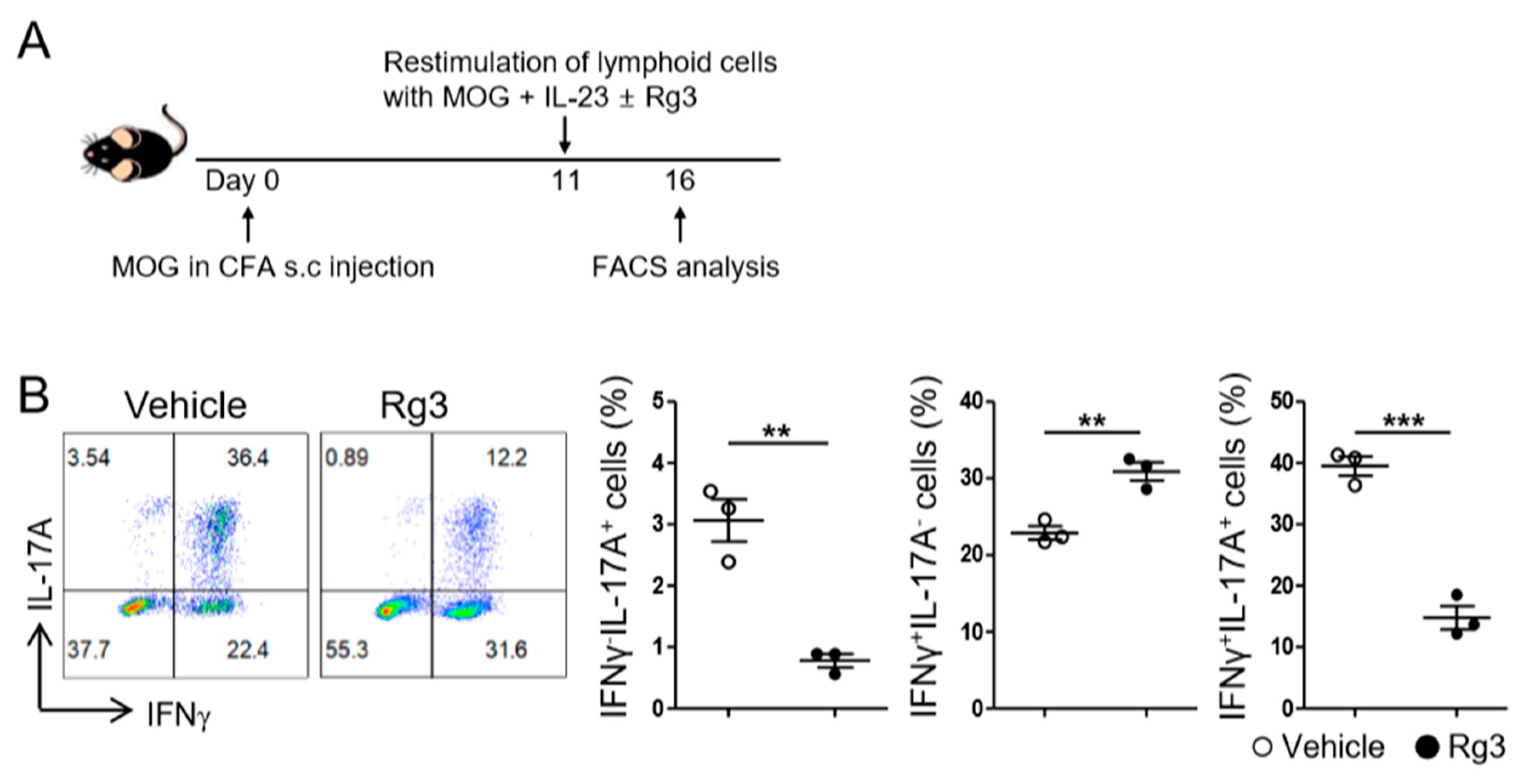
3.3. Rg3 Inhibits RORγt Expression in CD4+ T Cells during Th17 Cell Differentiation
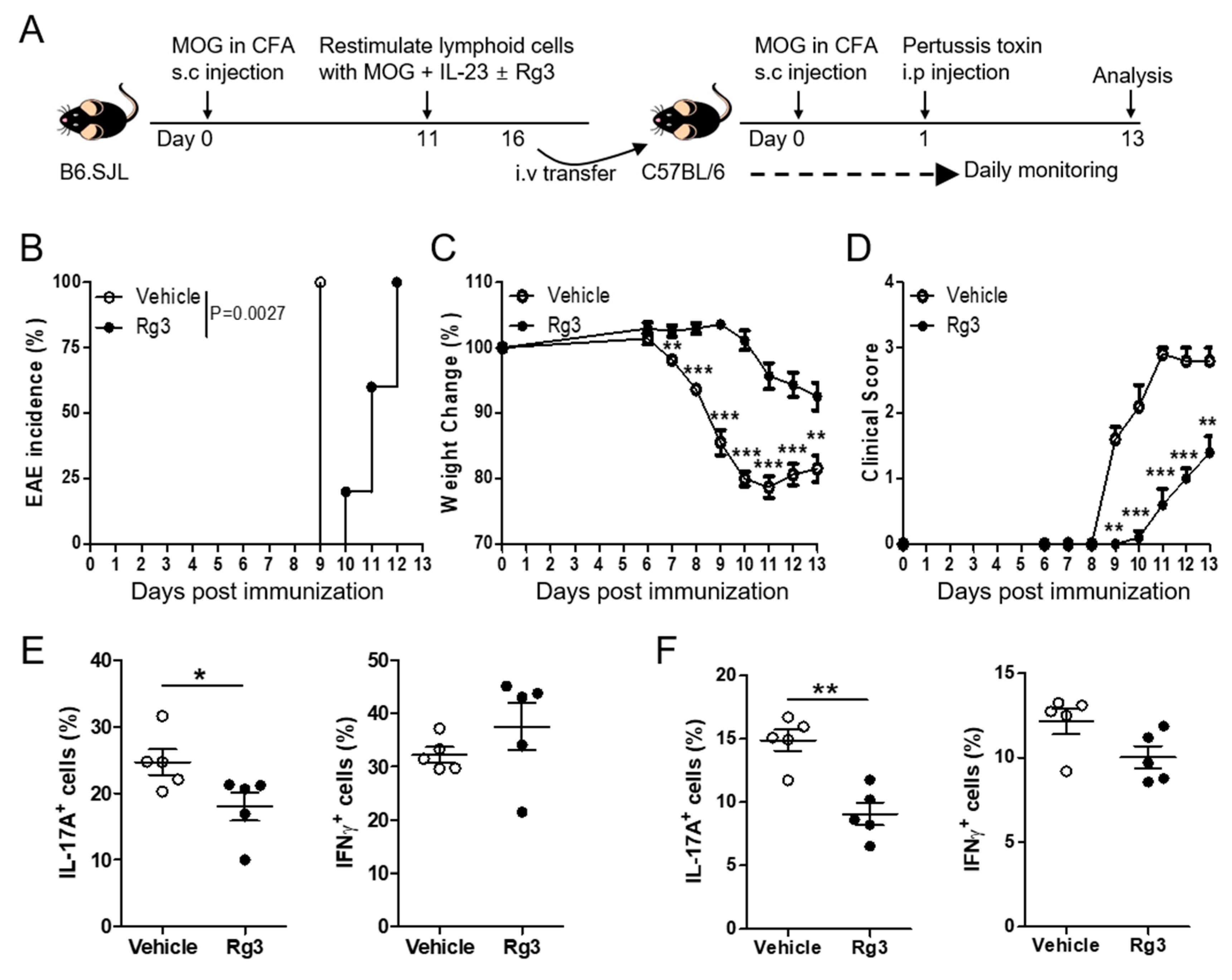
3.4. Rg3 Attenuates the Reactivation of Autoreactive Th17 Cells and Experimental Autoimmune Encephalomyelitis Induced by MOG-Reactive CD4+ T-Cell Transfer
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lee, J.-I.; Park, K.S.; Cho, I.-H. Panax ginseng: A candidate herbal medicine for autoimmune disease. J Ginseng Res. 2019, 43, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Yi, Y.-S.; Kim, M.-Y.; Cho, J.Y. Role of ginsenosides, the main active components of Panax ginseng, in inflammatory responses and diseases. J. Ginseng Res. 2017, 41, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Helms, S. Cancer prevention and therapeutics: Panax ginseng. Altern. Med. Rev. 2004, 9, 259–274. [Google Scholar] [PubMed]
- Hong, B.N.; Ji, M.G.; Kang, T.H. The efficacy of red ginseng in type 1 and type 2 diabetes in animals. Evid. Based Complement. Alternat. Med. 2013, 2013, 593181. [Google Scholar] [CrossRef] [PubMed]
- Jeong, C.-S. Effect of butanol fraction of Panax ginseng head on gastric lesion and ulcer. Arch. Pharm. Res. 2002, 25, 61–66. [Google Scholar] [CrossRef]
- Attele, A.S.; Wu, J.A.; Yuan, C.-S. Ginseng pharmacology: Multiple constituents and multiple actions. Biochem. Pharmacol. 1999, 58, 1685–1693. [Google Scholar] [CrossRef]
- Yi, Y.-S. Ameliorative effects of ginseng and ginsenosides on rheumatic diseases. J. Ginseng Res. 2019, 43, 335–341. [Google Scholar] [CrossRef]
- Liu, C.-X.; Xiao, P.-G. Recent advances on ginseng research in China. J. Ethnopharmacol. 1992, 36, 27–38. [Google Scholar]
- Lee, S.-Y.; Jeong, J.-J.; Eun, S.-H.; Kim, D.-H. Anti-inflammatory effects of ginsenoside Rg1 and its metabolites ginsenoside Rh1 and 20(S)-protopanaxatriol in mice with TNBS-induced colitis. Eur. J. Pharmacol. 2015, 762, 333–343. [Google Scholar] [CrossRef]
- Cho, M.; Choi, G.; Shim, I.; Chung, Y. Enhanced Rg3 negatively regulates Th1 cell responses. J. Ginseng Res. 2019, 43, 49–57. [Google Scholar] [CrossRef]
- Kang, S.; Park, S.-J.; Lee, A.-Y.; Huang, J.; Chung, H.-Y.; Im, D.-S. Ginsenoside Rg3 promotes inflammation resolution through M2 macrophage polarization. J. Ginseng Res. 2018, 42, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Stockinger, B.; Omenetti, S. The dichotomous nature of T helper 17 cells. Nat. Rev. Immunol. 2017, 17, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Muranski, P.; Restifo, N.P. Essentials of Th17 cell commitment and plasticity. Blood 2013, 121, 2402–2414. [Google Scholar] [CrossRef] [PubMed]
- Mohanan, P.; Subramaniyam, S.; Mathiyalagan, R.; Yang, D.-C. Molecular signaling of ginsenosides Rb1, Rg1, and Rg3 and their mode of actions. J. Ginseng Res. 2018, 42, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Mangan, P.R.; Harrington, L.E.; O’Quinn, D.B.; Helms, W.S.; Bullard, D.C.; Elson, C.O.; Hatton, R.D.; Wahl, S.M.; Schloeb, T.R.; Weaver, C.T. Transforming growth factor-beta induces development of the T(H)17 lineage. Nature 2006, 441, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, I.I.; McKenzie, B.S.; Zhou, L.; Tadokoro, C.E.; Lepelley, A.; Lafaille, J.J.; Cua, D.J.; Littman, D.R. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 2006, 126, 1121–1133. [Google Scholar] [CrossRef]
- Yang, X.O.; Panopoulos, A.D.; Nurieva, R.; Chang, S.H.; Wang, D.; Watowich, S.S.; Dong, C. STAT3 regulates cytokine-mediated generation of inflammatory helper T cells. J. Biol. Chem. 2007, 282, 9358–9363. [Google Scholar] [CrossRef]
- Heo, S.B.; Lim, S.W.; Jhun, J.Y.; Cho, M.L.; Chung, B.H.; Yang, C.W. Immunological benefits by ginseng through reciprocal regulation of Th17 and Treg cells during cyclosporine-induced immunosuppression. J. Ginseng Res. 2016, 40, 18–27. [Google Scholar] [CrossRef]
- Jhun, J.; Lee, J.; Byun, J.-K.; Kim, E.-K.; Woo, J.-W.; Lee, J.-H.; Kwok, S.-K.; Ju, J.-H.; Park, K.-S.; Kim, H.-Y.; et al. Red ginseng extract ameliorates autoimmune arthritis via regulation of STAT3 pathway, Th17/Treg balance, and osteoclastogenesis in mice and human. Mediat. Inflamm. 2014, 2014, 351856. [Google Scholar] [CrossRef]
- Wanke, F.; Moos, S.; Croxford, A.L.; Heinen, A.P.; Gräf, S.; Kalt, B.; Tischner, D.; Zhang, J.; Christen, I.; Bruttger, J.; et al. EBI2 Is Highly Expressed in Multiple Sclerosis Lesions and Promotes Early CNS Migration of Encephalitogenic CD4 T Cells. Cell Rep. 2017, 18, 1270–1284. [Google Scholar] [CrossRef]
- Veldhoen, M.; Hocking, R.J.; Atkins, C.J.; Locksley, R.M.; Stockinger, B. TGFbeta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 2006, 24, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Kim, Y.U.; Sun, H.; Lee, J.H.; Reynolds, J.M.; Hanabuchi, S.; Wu, H.; Teng, B.-B.; Chung, Y. Proatherogenic conditions promote autoimmune T helper 17 cell responses in vivo. Immunity 2014, 40, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Quintana, F.J. Old dog, new tricks: IL-6 cluster signaling promotes pathogenic TH17 cell differentiation. Nat. Immunol. 2016, 18, 8–10. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.; Chang, S.H.; Martinez, G.J.; Yang, X.O.; Nurieva, R.; Kang, H.S.; Ma, L.; Watowich, S.S.; Jetten, A.M.; Tian, Q.; et al. Critical regulation of early Th17 cell differentiation by interleukin-1 signaling. Immunity 2009, 30, 576–587. [Google Scholar] [CrossRef]
- Kim, B.-S.; Park, Y.-J.; Chung, Y. Targeting IL-17 in autoimmunity and inflammation. Arch. Pharm. Res. 2016, 39, 1537–1547. [Google Scholar] [CrossRef]
- Ciofani, M.; Madar, A.; Galan, C.; Sellars, M.; Mace, K.; Pauli, F.; Agarwal, A.; Huang, W.; Parkurst, C.N.; Muratet, M.; et al. A validated regulatory network for Th17 cell specification. Cell 2012, 151, 289–303. [Google Scholar] [CrossRef]
- Li, J.; Liu, T.; Zhao, L.; Chen, W.; Hou, H.; Ye, Z.; Li, X. Ginsenoside 20(S)Rg3 inhibits the Warburg effect through STAT3 pathways in ovarian cancer cells. Int. J. Oncol. 2015, 46, 775–781. [Google Scholar] [CrossRef]
- Nurieva, R.; Yang, X.O.; Martinez, G.; Zhang, Y.; Panopoulos, A.D.; Ma, L.; Schluns, K.; Tian, Q.; Watowich, S.S.; Jetten, A.M.; et al. Essential autocrine regulation by IL-21 in the generation of inflammatory T cells. Nature 2007, 448, 480–483. [Google Scholar] [CrossRef]
- Santori, F.R.; Huang, P.; van de Pavert, S.A.; Douglass, E.F., Jr.; Leaver, D.J.; Haubrich, B.A.; Keber, R.; Lorbek, G.; Konijn, T.; Rosales, B.N.; et al. Identification of natural RORgamma ligands that regulate the development of lymphoid cells. Cell Metab. 2015, 21, 286–298. [Google Scholar] [CrossRef]
- Xu, T.; Wang, X.; Zhong, B.; Nurieva, R.I.; Ding, S.; Dong, C. Ursolic acid suppresses interleukin-17 (IL-17) production by selectively antagonizing the function of RORgamma t protein. J. Biol. Chem. 2011, 286, 22707–22710. [Google Scholar] [CrossRef]
- Na, H.; Lim, H.; Choi, G.; Kim, B.-K.; Kim, S.-H.; Chang, Y.-S.; Nurieva, R.; Dong, C.; Chang, S.H.; Chung, Y. Concomitant suppression of TH2 and TH17 cell responses in allergic asthma by targeting retinoic acid receptor-related orphan receptor gammat. J. Allergy Clin. Immunol. 2018, 141, 2061–2073. [Google Scholar] [CrossRef] [PubMed]
- Sonnenberg, G.F.; Artis, D. Innate lymphoid cells in the initiation, regulation and resolution of inflammation. Nat. Med. 2015, 21, 698–708. [Google Scholar] [CrossRef] [PubMed]
- Cherrier, M.; Eberl, G. The development of LTi cells. Curr. Opin. Immunol. 2012, 24, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Dyring-Andersen, B.; Skov, L.; Zachariae, C. Targeting IL-17 with ixekizumab in patients with psoriasis. Immunotherapy 2015, 7, 957–966. [Google Scholar] [CrossRef]
- Farahnik, B.; Beroukhim, K.; Abrouk, M.; Nakamura, M.; Zhu, T.H.; Singh, R.; Lee, K.; Bhutani, T.; Koo, J. Brodalumab for the Treatment of Psoriasis: A Review of Phase III Trials. Dermatol. Ther. 2016, 6, 111–124. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, Y.-J.; Cho, M.; Choi, G.; Na, H.; Chung, Y. A Critical Regulation of Th17 Cell Responses and Autoimmune Neuro-Inflammation by Ginsenoside Rg3. Biomolecules 2020, 10, 122. https://doi.org/10.3390/biom10010122
Park Y-J, Cho M, Choi G, Na H, Chung Y. A Critical Regulation of Th17 Cell Responses and Autoimmune Neuro-Inflammation by Ginsenoside Rg3. Biomolecules. 2020; 10(1):122. https://doi.org/10.3390/biom10010122
Chicago/Turabian StylePark, Young-Jun, Minkyoung Cho, Garam Choi, Hyeongjin Na, and Yeonseok Chung. 2020. "A Critical Regulation of Th17 Cell Responses and Autoimmune Neuro-Inflammation by Ginsenoside Rg3" Biomolecules 10, no. 1: 122. https://doi.org/10.3390/biom10010122
APA StylePark, Y.-J., Cho, M., Choi, G., Na, H., & Chung, Y. (2020). A Critical Regulation of Th17 Cell Responses and Autoimmune Neuro-Inflammation by Ginsenoside Rg3. Biomolecules, 10(1), 122. https://doi.org/10.3390/biom10010122




