Essential Oils of Lamiaceae Family Plants as Antifungals
Abstract
1. Introduction
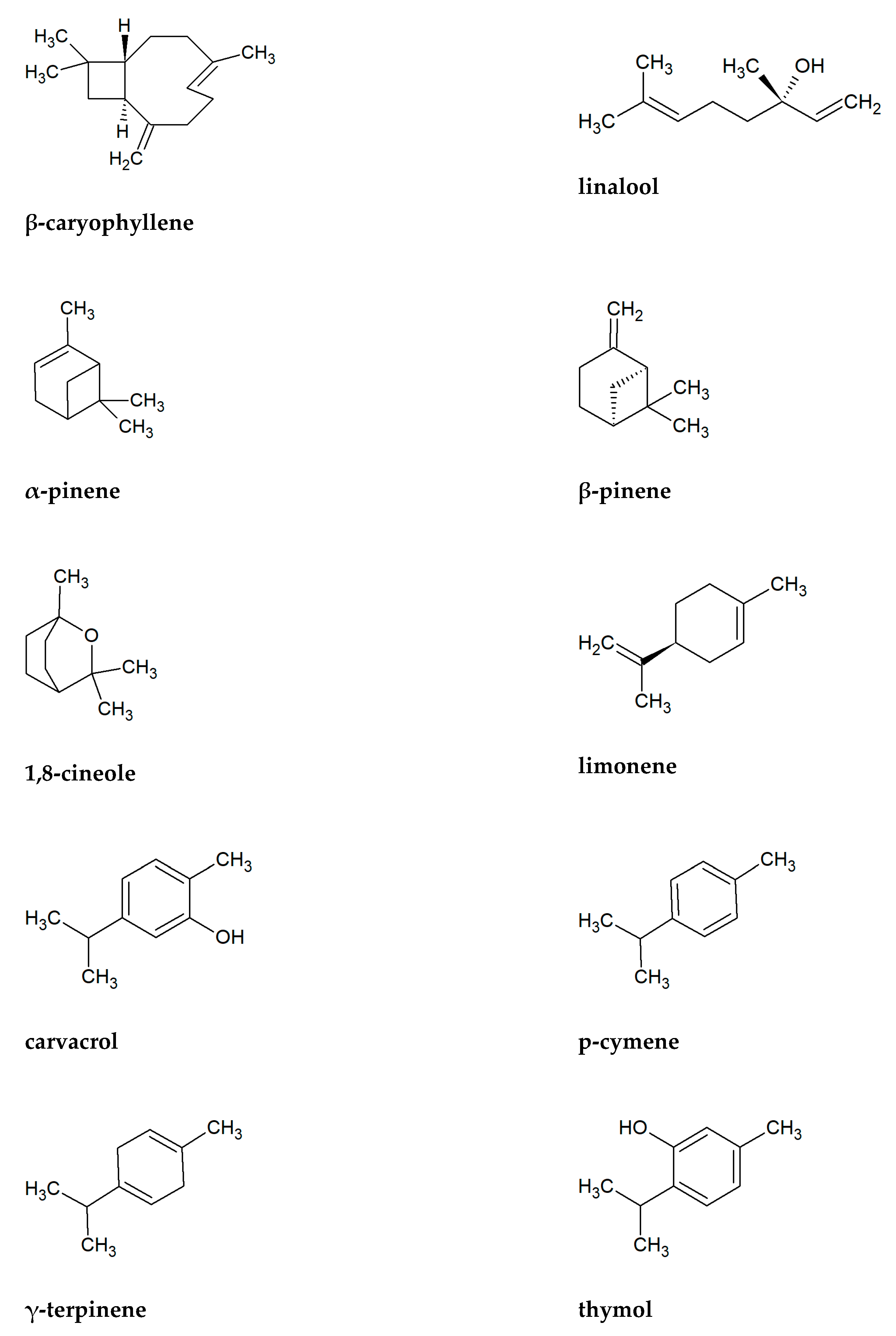
2. Components of Essential Oils of Lamiaceae Family
3. Antifungal Activity of Essential Oils of Lamiaceae Family
4. Essential Oils of Lamiaceae Plants in Cosmetics and Medicines
5. Conclusions
Funding
Conflicts of Interest
References
- Brown, G.D.; Denning, D.W.; Gow, N.A.; Levitz, S.M.; Netea, M.G.; White, T.C. Hidden killers: Human fungal infections. Sci. Transl. Med. 2012, 4. [Google Scholar] [CrossRef] [PubMed]
- White, T.C.; Findley, K.; Dawson, T.L., Jr.; Scheynius, A.; Boekhout, T.; Cuomo, C.A.; Xu, J.; Saunders, C.W. Fungi on the skin: Dermatophytes and Malassezia. Cold Spring Harb. Perspect. Med. 2014, 4. [Google Scholar] [CrossRef]
- Bongomin, F.; Gago, S.; Oladele, R.O.; Denning, D.W. Global and multi-national prevalence of fungal diseases—estimate precision. J. Fungi 2017, 3, 57. [Google Scholar] [CrossRef] [PubMed]
- Park, B.J.; Wannemuehler, K.A.; Marston, B.J.; Govender, N.; Pappas, P.G.; Chiller, T.M. Estimation of the current global burden of cryptococcal meningitis among persons living with HIV/AIDS. AIDS 2009, 23, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Drgona, L.; Khachatryan, A.; Stephens, J.; Charbonneau, C.; Kantecki, M.; Haider, S.; Barnes, R. Clinical and economic burden of invasive fungal diseases in Europe: Focus on pre-emptive and empirical treatment of Aspergillus and Candida species. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 7–21. [Google Scholar] [CrossRef] [PubMed]
- Murray, P.R.; Rosenthal, K.S.; Pfaller, M.A. Section 6. Mycology. In Medical Microbiology, 7th ed.; Saunders: Philadelphia, PA, USA, 2013; pp. 605–711. [Google Scholar]
- Reddy, K.R. Fungal infections (Mycoses): Dermatophytoses (Tinea, Ringworm). J. Gandaki Med. Coll. Nepal 2017, 10. [Google Scholar] [CrossRef]
- Lortholary, O.; Desnos-Ollivier, M.; Sitbon, K.; Fontanet, A.; Bretagne, S.; Dromer, F. Recent exposure to caspofungin or fluconazole influences the epidemiology of candidemia: A prospective multicenter study involving 2,441 patients. Antimicrob. Agents Chemother. 2011, 55, 532–538. [Google Scholar] [CrossRef]
- Alexander, B.D.; Johnson, M.D.; Pfeiffer, C.D.; Jiménez-Ortigosa, C.; Catania, J.; Booker, R.; Castanheira, M.; Messer, S.A.; Perlin, D.S.; Pfaller, M.A. Increasing echinocandin resistance in Candida glabrata: Clinical failure correlates with presence of FKS mutations and elevated minimum inhibitory concentrations. Clin. Infect. Dis. 2013, 56, 1724–1732. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Perlin, D.S. Echinocandin resistance: An emerging clinical problem? Curr. Opin. Infect. Dis. 2014, 27, 484–492. [Google Scholar] [CrossRef]
- Van der Linden, J.W.; Arendrup, M.C.; Warris, A.; Lagrou, K.; Pelloux, H.; Hauser, P.M.; Chryssanthou, E.; Mellado, E.; Kidd, S.E.; Tortorano, A.M.; et al. Prospective multicenter international surveillance of azole resistance in Aspergillus fumigatus. Emerg. Infect. Dis. 2015, 21, 1041–1044. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Diekema, D.J. Rare and emerging opportunistic fungal pathogens: Concern for resistance beyond Candida albicans and Aspergillus fumigatus. J. Clin. Microbiol. 2004, 42, 4419–4431. [Google Scholar] [CrossRef] [PubMed]
- Perlin, D.S.; Rautemaa-Richardson, R.; Alastruey-Izquierdo, A. The global problem of antifungal resistance: Prevalence, mechanisms, and management. Lancet Infect. Dis. 2017, 17, e383–e392. [Google Scholar] [CrossRef]
- Krcmery, V., Jr.; Spanik, S.; Kunova, A.; Trupl, J. Breakthrough fungemia appearing during empiric therapy with amphotericin B. Chemotherapy 1997, 43, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Hull, C.M.; Bader, O.; Parker, J.E.; Weig, M.; Gross, U.; Warrilow, A.G.; Kelly, D.E.; Kelly, S.L. Two clinical isolates of Candida glabrata exhibiting reduced sensitivity to amphotericin B both harbor mutations in ERG2. Antimicrob. Agents Chemother. 2012, 56, 6417–6421. [Google Scholar] [CrossRef]
- Woods, R.A.; Bard, M.; Jackson, I.E.; Drutz, D.J. Resistance to polyene antibiotics and correlated sterol changes in two isolates of Candida tropicalis from a patient with an amphotericin B-resistant funguria. J. Infect. Dis. 1974, 129, 53–58. [Google Scholar] [CrossRef]
- Satoh, K.; Makimura, K.; Hasumi, Y.; Nishiyama, Y.; Uchida, K.; Yamaguchi, H. Candida auris sp. nov, a novel ascomycetous yeast isolated from the external ear canal of an inpatient in a Japanese hospital. Microbiol. Immunol. 2009, 53, 41–44. [Google Scholar] [CrossRef]
- Tracking Candida Auris. Case Count Updated as of July 31; 2019; CDC. Available online: https://www.cdc.gov/fungal/candida-auris/tracking-c-auris.html (accessed on 9 September 2019).
- Lockhart, S.R.; Etienne, K.A.; Vallabhaneni, S.; Farooqi, J.; Chowdhary, A.; Govender, N.P.; Colombo, A.L.; Calvo, B.; Cuomo, C.A.; Desjardins, C.A.; et al. Simultaneous emergence of multidrug-resistant Candida auris on 3 continents confirmed by whole-genome sequencing and epidemiological analyses. Clin. Infect. Dis. 2017, 64, 134–140. [Google Scholar] [CrossRef]
- Friedman, D.Z.P.; Schwartz, I.S. Emerging fungal infections: New patients, new patterns, and new pathogens. J. Fungi 2019, 5, 67. [Google Scholar] [CrossRef]
- Hata, D.J.; Humphries, R.; Lockhart, S.R.; College of American Pathologists Microbiology Committee. Candida auris: An emerging yeast pathogen posing distinct challenges for laboratory diagnostics, treatment, and infection prevention. Arch. Pathol. Lab. Med. 2019. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeast, 3rd ed.; Approved Standard, CLSI document M27-A3, Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008. [Google Scholar]
- Clinical and Laboratory Standards Institute. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi, 2nd ed.; Approved Standard, CLSI document M38-A2, Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008. [Google Scholar]
- Przybyłek, I.; Karpiński, T.M. Antibacterial properties of propolis. Molecule 2019, 24, 47. [Google Scholar] [CrossRef]
- Ramasubramania Raja, R. Medicinally potential plants of Labiatae (Lamiaceae) family: An overview. Res. J. Med. Plant. 2012, 6, 203–213. [Google Scholar]
- Carović-Stanko, K.; Petek, M.; Grdiša, M.; Pintar, J.; Bedeković, D.; Herak Ćustić, M.; Satovic, Z. Medicinal plants of the family Lamiaceae as functional foods—A review. Czech J. Food Sci. 2016, 34, 377–390. [Google Scholar] [CrossRef]
- Radulović, N.S.; Blagojević, P.D.; Stojanović-Radić, Z.Z.; Stojanowić, N.M. Antimicrobial plant metabolites: Structural diversity and mechanism of action. Curr. Med. Chem. 2013, 20, 932–952. [Google Scholar] [PubMed]
- Vainstein, A.; Lewinsohn, E.; Pichersky, E.; Weiss, D. Floral fragrance. New inroads into an old commodity. Plant Physiol. 2001, 127, 1383–1389. [Google Scholar] [CrossRef]
- De Groot, A.C.; Schmidt, E. Essential oils, Part III: Chemical composition. Dermatitis 2016, 27, 161–619. [Google Scholar] [CrossRef]
- Piątkowska, E.; Rusiecka-Ziółkowska, J. Influence of essential oils on infectious agents. Adv. Clin. Exp. Med. 2016, 25, 989–995. [Google Scholar] [CrossRef]
- Dhifi, W.; Bellili, S.; Jazi, S.; Bahloul, N.; Mnif, W. Essential oils’ chemical characterization and investigation of some biological activities: A critical review. Medicines 2016, 3, 25. [Google Scholar] [CrossRef]
- The Plant List. Available online: http://www.theplantlist.org (accessed on 12 September 2019).
- PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov (accessed on 23 September 2019).
- Ngo Mback, M.N.; Agnaniet, H.; Nguimatsia, F.; Jazet Dongmo, P.M.; Hzounda Fokou, J.B.; Bakarnga-Via, I.; Fekam Boyom, F.; Menut, C. Optimization of antifungal activity of Aeollanthus heliotropioides oliv essential oil and Time Kill Kinetic Assay. J. Mycol. Med. 2016, 26, 233–243. [Google Scholar] [CrossRef]
- Ivanov, I.G.; Vrancheva, R.Z.; Petkova, N.T.; Tumbarski, Y.; Dincheva, I.N.; Badjakov, I.K. Phytochemical compounds of anise hyssop (Agastache foeniculum) and antibacterial, antioxidant, and acetylcholinesterase inhibitory properties of its essential oil. J. Appl. Pharmac. Sci. 2019, 9, 72–78. [Google Scholar]
- Li, H.Q.; Liu, Q.Z.; Liu, Z.L.; Du, S.S.; Deng, Z.W. Chemical composition and nematicidal activity of essential oil of Agastache rugosa against Meloidogyne incognita. Molecules 2013, 18, 4170–4180. [Google Scholar] [CrossRef]
- Fraternale, D.; Ricci, D. Essential oil composition and antifungal activity of aerial parts of Ballota nigra ssp foetida collected at flowering and fruiting times. Nat. Prod. Commun. 2014, 9, 1015–1018. [Google Scholar] [CrossRef] [PubMed]
- Marinković, B.; Marin, P.D.; Knezević-Vukcević, J.; Soković, M.D.; Brkić, D. Activity of essential oils of three Micromeria species (Lamiaceae) against micromycetes and bacteria. Phytother. Res. 2002, 16, 336–339. [Google Scholar] [CrossRef] [PubMed]
- Monforte, M.T.; Tzakou, O.; Nostro, A.; Zimbalatti, V.; Galati, E.M. Chemical composition and biological activities of Calamintha officinalis Moench essential oil. J. Med. Food 2011, 14, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Ćavar, S.; Vidic, D.; Maksimović, M. Volatile constituents, phenolic compounds, and antioxidant activity of Calamintha glandulosa (Req.) Bentham. J. Sci. Food Agric. 2013, 93, 1758–1764. [Google Scholar] [CrossRef]
- Marongiu, B.; Piras, A.; Porcedda, S.; Falconieri, D.; Maxia, A.; Gonçalves, M.J.; Cavaleiro, C.; Salgueiro, L. Chemical composition and biological assays of essential oils of Calamintha nepeta (L.) Savi subsp. nepeta (Lamiaceae). Nat. Prod. Res. 2010, 24, 1734–1742. [Google Scholar] [CrossRef] [PubMed]
- Božović, M.; Garzoli, S.; Sabatino, M.; Pepi, F.; Baldisserotto, A.; Andreotti, E.; Romagnoli, C.; Mai, A.; Manfredini, S.; Ragno, R. Essential oil extraction, chemical analysis and anti-Candida activity of Calamintha nepeta (L.) Savi subsp. landulosa (Req.) Ball—New approaches. Molecules 2017, 22, 203. [Google Scholar] [CrossRef]
- Kumar, V.; Mathela, C.S.; Tewari, A.K.; Bisht, K.S. In vitro inhibition activity of essential oils from some Lamiaceae species against phytopathogenic fungi. Pestic. Biochem. Physiol. 2014, 114, 67–71. [Google Scholar] [CrossRef]
- Stappen, I.; Wanner, J.; Tabanca, N.; Wedge, D.E.; Ali, A.; Kaul, V.K.; Lal, B.; Jaitak, V.; Gochev, V.K.; Schmidt, E.; et al. Chemical composition and biological activity of essential oils of Dracocephalum heterophyllum and Hyssopus officinalis from Western Himalaya. Nat. Prod. Commun. 2015, 10, 133–138. [Google Scholar] [CrossRef]
- Ahmadi, F.; Sadeghi, S.; Modarresi, M.; Abiri, R.; Mikaeli, A. Chemical composition, in vitro anti-microbial, antifungal an d antioxidant activities of the essential oil and methanolic extract of Hymenocrater longiflorus Benth., of Iran. Food Chem. Toxicol. 2010, 48, 1137–1144. [Google Scholar] [CrossRef]
- De Oliveira, C.M.A.; Silva, M.R.R.; Kato, L.; da Silva, C.C.; Ferreira, H.D.; Souza, L.K.H. Chemical composition and antifungal activity of the essential oil of Hyptis ovalifolia Benth. (Lamiaceae). J. Braz. Chem. Soc. 2004, 15, 756–759. [Google Scholar] [CrossRef]
- Džamić, A.M.; Soković, M.D.; Novaković, M.; Jadranin, M.; Ristić, M.S.; Tešević, V.; Marin, P.D. Composition, antifungal and antioxidant properties of Hyssopus officinalis L. subsp. pilifer (Pant.) Murb. essential oil and deodorized extracts. Ind. Crops Prod. 2013, 51, 401–407. [Google Scholar]
- Hristova, Y.; Wanner, J.; Jirovetz, L.; Stappen, I.; Iliev, I.; Gochev, V. Chemical composition and antifungal activity of essential oil of Hyssopus officinalis L. from Bulgaria against clinical isolates of Candida species. Biotechnol. Biotechnol. Equip. 2015, 29, 592–601. [Google Scholar] [CrossRef]
- Adam, K.; Sivropoulou, A.; Kokkini, S.; Lanaras, T.; Arsenakis, M. Antifungal activities of Origanum vulgare subsp. hirtum, Mentha spicata, Lavandula angustifolia, and Salvia fruticosa essential oils against human pathogenic fungi. J. Agric. Food Chem. 1998, 46, 1739–1745. [Google Scholar]
- D’Auria, F.D.; Tecca, M.; Strippoli, V.; Salvatore, G.; Battinelli, L.; Mazzanti, G. Antifungal activity of Lavandula angustifolia essential oil against Candida albicans yeast and mycelial form. Med. Mycol. 2005, 43, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Khoury, M.; Stien, D.; Eparvier, V.; Ouaini, N.; El Beyrouthy, M. Report on the medicinal use of eleven Lamiaceae species in Lebanon and rationalization of their antimicrobial potential by examination of the chemical composition and antimicrobial activity of their essential oils. Evid. Based Compl. Altern. Med. 2016, 2016. [Google Scholar] [CrossRef]
- Zuzarte, M.; Vale-Silva, L.; Gonçalves, M.J.; Cavaleiro, C.; Vaz, S.; Canhoto, J.; Pinto, E.; Salgueiro, L. Antifungal activity of phenolic-rich Lavandula multifida L. essential oil. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1359–1366. [Google Scholar] [CrossRef]
- Zuzarte, M.; Gonçalves, M.J.; Cavaleiro, C.; Dinis, A.M.; Canhoto, J.M.; Salgueiro, L.R. Chemical composition and antifungal activity of the essential oils of Lavandula pedunculata (Miller) Cav. Chem. Biodivers. 2009, 6, 1283–1292. [Google Scholar] [CrossRef]
- Zuzarte, M.; Gonçalves, M.J.; Cruz, M.T.; Cavaleiro, C.; Canhoto, J.; Vaz, S.; Pinto, E.; Salgueiro, L. Lavandula luisieri essential oil as a source of antifungal drugs. Food Chem. 2012, 135, 1505–1510. [Google Scholar] [CrossRef]
- Zuzarte, M.; Gonçalves, M.J.; Cavaleiro, C.; Canhoto, J.; Vale-Silva, L.; Silva, M.J.; Pinto, E.; Salgueiro, L. Chemical composition and antifungal activity of the essential oils of Lavandula viridis L’Her. J. Med. Microbiol. 2011, 60, 612–618. [Google Scholar] [CrossRef]
- Ramírez, J.; Gilardoni, G.; Jácome, M.; Montesinos, J.; Rodolfi, M.; Guglielminetti, M.L.; Cagliero, C.; Bicchi, C.; Vidari, G. Chemical composition, enantiomeric analysis, AEDA sensorial evaluation and antifungal activity of the essential oil from the Ecuadorian plant Lepechinia mutica Benth (Lamiaceae). Chem. Biodivers. 2017, 14, e1700292. [Google Scholar] [CrossRef]
- Ramírez, J.; Gilardoni, G.; Ramón, E.; Tosi, S.; Picco, A.M.; Bicchi, C.; Vidari, G. Phytochemical study of the Ecuadorian species Lepechinia mutica (Benth.) Epling and high antifungal activity of carnosol against Pyricularia oryzae. Pharmaceuticals 2018, 11, 33. [Google Scholar] [CrossRef] [PubMed]
- Zarai, Z.; Kadri, A.; Ben Chobba, I.; Ben Mansour, R.; Bekir, A.; Mejdoub, H.; Gharsallah, N. The in-vitro evaluation of antibacterial, antifungal and cytotoxic properties of Marrubium vulgare L. essential oil grown in Tunisia. Lipids Health Dis. 2011, 10, 161. [Google Scholar] [CrossRef] [PubMed]
- Mimica-Dukic, N.; Bozin, B.; Sokovic, M.; Simin, N. Antimicrobial and antioxidant activities of Melissa officinalis L. (Lamiaceae) essential oil. J. Agric. Food Chem. 2004, 52, 2485–2489. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, M.J.; Vicente, A.M.; Cavaleiro, C.; Salgueiro, L. Composition and antifungal activity of the essential oil of Mentha cervina from Portugal. Nat. Prod. Res. 2007, 21, 867–871. [Google Scholar] [CrossRef] [PubMed]
- Soković, M.D.; Vukojević, J.; Marin, P.D.; Brkić, D.D.; Vajs, V.; van Griensven, L.J. Chemical composition of essential oils of Thymus and Mentha species and their antifungal activities. Molecules 2009, 14, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Camiletti, B.X.; Asensio, C.M.; Pecci Mde, L.; Lucini, E.I. Natural control of corn postharvest fungi Aspergillus flavus and Penicillium sp. using essential oils from plants grown in Argentina. J. Food Sci. 2014, 79, M2499–M2506. [Google Scholar] [CrossRef]
- Samber, N.; Khan, A.; Varma, A.; Manzoor, N. Synergistic anti-candidal activity and mode of action of Mentha piperita essential oil and its major components. Pharm. Biol. 2015, 53, 1496–1504. [Google Scholar] [CrossRef]
- Hossain, F.; Follett, P.; Dang Vu, K.; Harich, M.; Salmieri, S.; Lacroix, M. Evidence for synergistic activity of plant-derived essential oils against fungal pathogens of food. Food Microbiol. 2016, 53, 24–30. [Google Scholar] [CrossRef]
- Mahboubi, M.; Haghi, G. Antimicrobial activity and chemical composition of Mentha pulegium L. essential oil. J. Ethnopharmacol. 2008, 119, 325–327. [Google Scholar] [CrossRef]
- Abdelli, M.; Moghrani, H.; Aboun, A.; Maachi, R. Algerian Mentha pulegium L. leaves essential oil: Chemical composition, antimicrobial, insecticidal and antioxidant activities. Ind. Crops Prod. 2016, 94, 197–205. [Google Scholar] [CrossRef]
- Chessa, M.; Sias, A.; Piana, A.; Mangano, G.S.; Petretto, G.L.; Masia, M.D.; Tirillini, B.; Pintore, G. Chemical composition and antibacterial activity of the essential oil from Mentha requienii Bentham. Nat. Prod. Res. 2013, 27, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Nardoni, S.; Giovanelli, S.; Pistelli, L.; Mugnaini, L.; Profili, G.; Pisseri, F.; Mancianti, F. In vitro activity of twenty commercially available, plant-derived essential oils against selected dermatophyte species. Nat. Prod. Commun. 2015, 10, 1473–1478. [Google Scholar] [CrossRef] [PubMed]
- Oumzil, H.; Ghoulami, S.; Rhajaoui, M.; Ilidrissi, A.; Fkih-Tetouani, S.; Faid, M.; Benjouad, A. Antibacterial and antifungal activity of essential oils of Mentha suaveolens. Phytother. Res. 2002, 16, 727–731. [Google Scholar] [CrossRef] [PubMed]
- El-Kashoury, S.A.; El-Askary, H.I.; Kandil, Z.A.; Salem, M.A. Chemical composition of the essential oil and botanical study of the flowers of Mentha suaveolens. Pharm. Biol. 2014, 52, 688–697. [Google Scholar] [CrossRef] [PubMed]
- Garzoli, S.; Pirolli, A.; Vavala, E.; Di Sotto, A.; Sartorelli, G.; Božović, M.; Angiolella, L.; Mazzanti, G.; Pepi, F.; Ragno, R. Multidisciplinary approach to determine the optimal time and period for extracting the essential oil from Mentha suaveolens Ehrh. Molecules 2015, 20, 9640–9655. [Google Scholar] [CrossRef] [PubMed]
- Casiglia, S.; Jemia, M.B.; Riccobono, L.; Bruno, M.; Scandolera, E.; Senatore, F. Chemical composition of the essential oil of Moluccella spinosa L. (Lamiaceae) collected wild in Sicily and its activity on microorganisms affecting historical textiles. Nat. Prod. Res. 2015, 29, 1201–1206. [Google Scholar] [CrossRef]
- Sacchetti, G.; Medici, A.; Maietti, S.; Radice, M.; Muzzoli, M.; Manfredini, S.; Braccioli, E.; Bruni, R. Composition and functional properties of the essential oil of amazonian basil, Ocimum micranthum Willd., Labiatae in comparison with commercial essential oils. J. Agric. Food Chem. 2004, 52, 3486–3491. [Google Scholar] [CrossRef]
- Bozin, B.; Mimica-Dukic, N.; Simin, N.; Anackov, G. Characterization of the volatile composition of essential oils of some Lamiaceae spices and the antimicrobial and antioxidant activities of the entire oils. J. Agric. Food Chem. 2006, 54, 1822–1828. [Google Scholar] [CrossRef]
- Al-Maskri, A.Y.; Hanif, M.A.; Al-Maskari, M.Y.; Abraham, A.S.; Al-sabahi, J.N.; Al-Mantheri, O. Essential oil from Ocimum basilicum (Omani Basil): A desert crop. Nat. Prod. Commun. 2011, 6, 1487–1490. [Google Scholar]
- Avetisyan, A.; Markosian, A.; Petrosyan, M.; Sahakyan, N.; Babayan, A.; Aloyan, S.; Trchounian, A. Chemical composition and some biological activities of the essential oils from basil Ocimum different cultivars. BMC Complement Altern. Med. 2017, 17, 60. [Google Scholar] [CrossRef]
- Ali, N.A.A.; Chhetri, B.K.; Dosoky, N.S.; Shari, K.; Al-Fahad, A.J.A.; Wessjohann, L.; Setzer, W.N. Antimicrobial, antioxidant, and cytotoxic activities of Ocimum forskolei and Teucrium yemense (Lamiaceae) essential oils. Medicines 2017, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Dubey, N.K.; Tiwari, T.N.; Mandin, D.; Andriamboavonjy, H.; Chaumont, J.P. Antifungal properties of Ocimum gratissimum essential oil (ethyl cinnamate chemotype). Fitoterapia 2000, 71, 567–569. [Google Scholar] [CrossRef]
- Lemos Jde, A.; Passos, X.S.; Fernandes Ode, F.; Paula, J.R.; Ferri, P.H.; Souza, L.K.; Lemos Ade, A.; Silva Mdo, R. Antifungal activity from Ocimum gratissimum L. towards Cryptococcus neoformans. Mem. Inst. Oswaldo Cruz 2005, 100, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Mohr, F.B.; Lermen, C.; Gazim, Z.C.; Gonçalves, J.E.; Alberton, O. Antifungal activity, yield, and composition of Ocimum gratissimum essential oil. Genet. Mol. Res. 2017, 16. [Google Scholar] [CrossRef]
- Amber, K.; Aijaz, A.; Immaculata, X.; Luqman, K.A.; Nikhat, M. Anticandidal effect of Ocimum sanctum essential oil and its synergy with fluconazole and ketoconazole. Phytomedicine 2010, 17, 921–925. [Google Scholar] [CrossRef]
- Khan, A.; Ahmad, A.; Akhtar, F.; Yousuf, S.; Xess, I.; Khan, L.A.; Manzoor, N. Ocimum sanctum essential oil and its active principles exert their antifungal activity by disrupting ergosterol biosynthesis and membrane integrity. Res. Microbiol. 2010, 161, 816–823. [Google Scholar] [CrossRef]
- Kumar, A.; Shukla, R.; Singh, P.; Dubey, N.K. Chemical composition, antifungal and antiaflatoxigenic activities of Ocimum sanctum L. essential oil and its safety assessment as plant based antimicrobial. Food Chem. Toxicol. 2010, 48, 539–543. [Google Scholar] [CrossRef]
- Santamarina, M.P.; Roselló, J.; Sempere, F.; Giménez, S.; Blázquez, M.A. Commercial Origanum compactum Benth. and Cinnamomum zeylanicum Blume essential oils against natural mycoflora in Valencia rice. Nat. Prod. Res. 2015, 29, 2215–2258. [Google Scholar] [CrossRef]
- Homa, M.; Fekete, I.P.; Böszörményi, A.; Singh, Y.R.; Selvam, K.P.; Shobana, C.S.; Manikandan, P.; Kredics, L.; Vágvölgyi, C.; Galgóczy, L. Antifungal effect of essential oils against Fusarium keratitis isolates. Planta Med. 2015, 81, 1277–1284. [Google Scholar] [CrossRef]
- Waller, S.B.; Madrid, I.M.; Ferraz, V.; Picoli, T.; Cleff, M.B.; de Faria, R.O.; Meireles, M.C.; de Mello, J.R. Cytotoxicity and anti-Sporothrix brasiliensis activity of the Origanum majorana Linn. oil. Braz. J. Microbiol. 2016, 47, 896–901. [Google Scholar] [CrossRef]
- Nikkhah, M.; Hashemi, M.; Habibi Najafi, M.B.; Farhoosh, R. Synergistic effects of some essential oils against fungal spoilage on pear fruit. Int. J. Food Microbiol. 2017, 257, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Rosato, A.; Vitali, C.; Piarulli, M.; Mazzotta, M.; Argentieri, M.P.; Mallamaci, R. In vitro synergic efficacy of the combination of Nystatin with the essential oils of Origanum vulgare and Pelargonium graveolens against some Candida species. Phytomedicine 2009, 16, 972–975. [Google Scholar] [CrossRef] [PubMed]
- Vale-Silva, L.; Silva, M.J.; Oliveira, D.; Gonçalves, M.J.; Cavaleiro, C.; Salgueiro, L.; Pinto, E. Correlation of the chemical composition of essential oils from Origanum vulgare subsp. virens with their in vitro activity against pathogenic yeasts and filamentous fungi. J. Med. Microbiol. 2012, 61, 252–260. [Google Scholar] [PubMed]
- Jeršek, B.; Poklar Ulrih, N.; Skrt, M.; Gavarić, N.; Božin, B.; Smole Možina, S. Effects of selected essential oils on the growth and production of ochratoxin A by Penicillium verrucosum. Arhiv Higijenu i Toksikologiju 2014, 65, 199–208. [Google Scholar] [CrossRef]
- Elansary, H.O.; Abdelgaleil, S.A.M.; Mahmoud, E.A.; Yessoufou, K.; Elhindi, K.; El-Hendawy, S. Effective antioxidant, antimicrobial and anticancer activities of essential oils of horticultural aromatic crops in northern Egypt. BMC Complement Altern. Med. 2018, 18, 214. [Google Scholar] [CrossRef]
- Kocevski, D.; Du, M.; Kan, J.; Jing, C.; Lačanin, I.; Pavlović, H. Antifungal effect of Allium tuberosum, Cinnamomum cassia, and Pogostemon cablin essential oils and their components against population of Aspergillus species. J. Food Sci. 2013, 78, M731–M737. [Google Scholar] [CrossRef]
- Murugan, R.; Mallavarapu, G.R.; Padmashree, K.V.; Rao, R.R.; Livingstone, C. Volatile oil composition of Pogostemon heyneanus and comparison of its composition with patchouli oil. Nat. Prod. Commun. 2010, 5, 1961–1964. [Google Scholar] [CrossRef]
- Zhang, H.Y.; Gao, Y.; Lai, P.X. Chemical composition, antioxidant, antimicrobial and cytotoxic activities of essential oil from Premna microphylla Turczaninow. Molecules 2017, 22, 381. [Google Scholar] [CrossRef]
- Angioni, A.; Barra, A.; Cereti, E.; Barile, D.; Coïsson, J.D.; Arlorio, M.; Dessi, S.; Coroneo, V.; Cabras, P. Chemical composition, plant genetic differences, antimicrobial and antifungal activity investigation of the essential oil of Rosmarinus officinalis L. J. Agric. Food Chem. 2004, 52, 3530–3535. [Google Scholar] [CrossRef]
- Bozin, B.; Mimica-Dukic, N.; Samojlik, I.; Jovin, E. Antimicrobial and antioxidant properties of rosemary and sage (Rosmarinus officinalis L. and Salvia officinalis L., Lamiaceae) essential oils. J. Agric. Food Chem. 2007, 55, 7879–7885. [Google Scholar] [CrossRef]
- Ozcan, M.M.; Chalchat, J.C. Chemical composition and antifungal activity of rosemary (Rosmarinus officinalis L.) oil from Turkey. Int. J. Food Sci. Nutr. 2008, 59, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Wu, N.; Fu, Y.J.; Wang, W.; Luo, M.; Zhao, C.J.; Zu, Y.G.; Liu, X.L. Chemical composition and antimicrobial activity of the essential oil of Rosemary. Environ. Toxicol. Pharmacol. 2011, 32, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Pitarokili, D.; Tzakou, O.; Loukis, A.; Harvala, C. Volatile metabolites from Salvia fruticosa as antifungal agents in soilborne pathogens. J. Agric. Food Chem. 2003, 51, 3294–3301. [Google Scholar] [CrossRef] [PubMed]
- Zomorodian, K.; Moein, M.; Pakshir, K.; Karami, F.; Sabahi, Z. Chemical composition and antimicrobial activities of the essential oil from Salvia mirzayanii leaves. J. Evid. Based Complementary Altern. Med. 2017, 22, 770–776. [Google Scholar] [CrossRef]
- Abu-Darwish, M.S.; Cabral, C.; Ferreira, I.V.; Gonçalves, M.J.; Cavaleiro, C.; Cruz, M.T.; Al-bdour, T.H.; Salgueiro, L. Essential oil of common sage (Salvia officinalis L.) from Jordan: Assessment of safety in mammalian cells and its antifungal and anti-inflammatory potential. BioMed Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Yuce, E.; Yildirim, N.; Yildirim, N.C.; Paksoy, M.Y.; Bagci, E. Essential oil composition, antioxidant and antifungal activities of Salvia sclarea L. from Munzur Valley in Tunceli, Turkey. Cell. Mol. Biol. 2014, 60, 1–5. [Google Scholar]
- Güllüce, M.; Sökmen, M.; Daferera, D.; Ağar, G.; Ozkan, H.; Kartal, N.; Polissiou, M.; Sökmen, A.; Sahin, F. In vitro antibacterial, antifungal, and antioxidant activities of the essential oil and methanol extracts of herbal parts and callus cultures of Satureja hortensis L. J. Agric. Food Chem. 2003, 51, 3958–3965. [Google Scholar] [CrossRef]
- Omidbeygi, M.; Barzegar, M.; Hamidi, Z.; Naghdibadi, H. Antifungal activity of thyme, summer savory and clove essential oils against Aspergillus flavus in liquid medium and tomato paste. Food Control 2007, 18, 1518–1523. [Google Scholar] [CrossRef]
- Giweli, A.; Džamić, A.M.; Soković, M.; Ristić, M.S.; Marin, P.D. Antimicrobial and antioxidant activities of essential oils of Satureja thymbra growing wild in Libya. Molecules 2012, 17, 4836–4850. [Google Scholar] [CrossRef]
- Serbetçi, T.; Demirci, B.; Güzel, C.B.; Kültür, S.; Ergüven, M.; Başer, K.H. Essential oil composition, antimicrobial and cytotoxic activities of two endemic Stachys cretica subspecies (Lamiaceae) from Turkey. Nat. Prod. Commun. 2010, 5, 1369–1374. [Google Scholar] [CrossRef]
- Lazarević, J.S.; Đorđević, A.S.; Kitić, D.V.; Zlatković, B.K.; Stojanović, G.S. Chemical composition and antimicrobial activity of the essential oil of Stachys officinalis (L.) Trevis. (Lamiaceae). Chem. Biodivers. 2013, 10, 1335–1349. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, A.; Nazari, H.; Imani, S.; Amrollahi, H. Antifungal activities and chemical composition of some medicinal plants. J. Mycol. Med. 2014, 24, e1–e8. [Google Scholar] [CrossRef] [PubMed]
- Salah, K.B.; Mahjoub, M.A.; Chaumont, J.P.; Michel, L.; Millet-Clerc, J.; Chraeif, I.; Ammar, S.; Mighri, Z.; Aouni, M. Chemical composition and in vitro antifungal and antioxidant activity of the essential oil and methanolic extract of Teucrium sauvagei Le Houerou. Nat. Prod. Res. 2006, 20, 1089–1097. [Google Scholar] [CrossRef] [PubMed]
- Goren, A.C.; Bilsel, G.; Bilsel, M.; Demir, H.; Kocabaş, E.E. Analysis of essential oil of Coridothymus capitatus (L.) and its antibacterial and antifungal activity. Zeitschrift für Naturforschung C 2003, 58, 687–690. [Google Scholar] [CrossRef]
- Salgueiro, L.R.; Pinto, E.; Gonçalves, M.J.; Pina-Vaz, C.; Cavaleiro, C.; Rodrigues, A.G.; Palmeira, A.; Tavares, C.; Costa-de-Oliveira, S.; Martinez-de-Oliveira, J. Chemical composition and antifungal activity of the essential oil of Thymbra capitata. Planta Med. 2004, 70, 572–575. [Google Scholar] [CrossRef]
- Palmeira-de-Oliveira, A.; Gaspar, C.; Palmeira-de-Oliveira, R.; Silva-Dias, A.; Salgueiro, L.; Cavaleiro, C.; Pina-Vaz, C.; Martinez-de-Oliveira, J.; Queiroz, J.A.; Rodrigues, A.G. The anti-Candida activity of Thymbra capitata essential oil: Effect upon pre-formed biofilm. J. Ethnopharmacol. 2012, 140, 379–383. [Google Scholar] [CrossRef]
- Kiliç, T. Analysis of essential oil composition of Thymbra spicata var. spicata: Antifungal, antibacterial and antimycobacterial activities. Z. Naturforsch. C 2006, 61, 324–328. [Google Scholar]
- Unlü, M.; Vardar-Unlü, G.; Vural, N.; Dönmez, E.; Ozbaş, Z.Y. Chemical composition, antibacterial and antifungal activity of the essential oil of Thymbra spicata L. from Turkey. Nat. Prod. Res. 2009, 23, 572–579. [Google Scholar] [CrossRef]
- Jaradat, N.; Adwan, L.; Kaibni, S.; Shraim, N.; Zaid, A.N. Chemical composition, anthelmintic, antibacterial and antioxidant effects of Thymus bovei essential oil. BMC Complement Altern. Med. 2016, 16, 418. [Google Scholar] [CrossRef]
- Pina-Vaz, C.; Gonçalves Rodrigues, A.; Pinto, E.; Costa-de-Oliveira, S.; Tavares, C.; Salgueiro, L.; Cavaleiro, C.; Gonçalves, M.J.; Martinez-de-Oliveira, J. Antifungal activity of Thymus oils and their major compounds. J. Eur. Acad Dermatol. Venereol. 2004, 18, 73–78. [Google Scholar] [CrossRef]
- Alizadeh, A.; Zamani, E.; Sharaifi, R.; Javan-Nikkhah, M.; Nazari, S. Antifungal activity of some essential oils against toxigenic Aspergillus species. Commun. Agric. Appl. Biol. Sci. 2010, 75, 761–767. [Google Scholar] [PubMed]
- Alizadeh, A.; Sharaifi, R.; Javan-Nikkhah, M.; Sedaghat, N. Survey of Thymus migricus essential oil on aflatoxin inhibition in Aspergillus flavus. Commun. Agric. Appl. Biol. Sci. 2010, 75, 769–776. [Google Scholar] [PubMed]
- Pinto, E.; Pina-Vaz, C.; Salgueiro, L.; Gonçalves, M.J.; Costa-de-Oliveira, S.; Cavaleiro, C.; Palmeira, A.; Rodrigues, A.; Martinez-de-Oliveira, J. Antifungal activity of the essential oil of Thymus pulegioides on Candida, Aspergillus and dermatophyte species. J. Med. Microbiol. 2006, 55, 1367–1373. [Google Scholar] [CrossRef] [PubMed]
- Pagiotti, R.; Angelini, P.; Rubini, A.; Tirillini, B.; Granetti, B.; Venanzoni, R. Identification and characterisation of human pathogenic filamentous fungi and susceptibility to Thymus schimperi essential oil. Mycoses 2011, 54, e364–e376. [Google Scholar] [CrossRef]
- Couladis, M.; Tzakou, O.; Kujundzic, S.; Sokovic, M.; Mimica-Dukic, N. Chemical analysis and antifungal activity of Thymus striatus. Phytother. Res. 2004, 18, 40–42. [Google Scholar] [CrossRef]
- Segvić Klarić, M.; Kosalec, I.; Mastelić, J.; Piecková, E.; Pepeljnak, S. Antifungal activity of thyme (Thymus vulgaris L.) essential oil and thymol against moulds from damp dwellings. Lett. Appl. Microbiol. 2007, 44, 36–42. [Google Scholar] [CrossRef]
- De Lira Mota, K.S.; de Oliveira Pereira, F.; de Oliveira, W.A.; Lima, I.O.; de Oliveira Lima, E. Antifungal activity of Thymus vulgaris L. essential oil and its constituent phytochemicals against Rhizopus oryzae: Interaction with ergosterol. Molecules 2012, 17, 14418–14433. [Google Scholar] [CrossRef]
- Khan, M.S.; Ahmad, I.; Cameotra, S.S. Carum copticum and Thymus vulgaris oils inhibit virulence in Trichophyton rubrum and Aspergillus spp. Braz. J. Microbiol. 2014, 45, 523–531. [Google Scholar] [CrossRef]
- Kohiyama, C.Y.; Yamamoto Ribeiro, M.M.; Mossini, S.A.; Bando, E.; Bomfim Nda, S.; Nerilo, S.B.; Rocha, G.H.; Grespan, R.; Mikcha, J.M.; Machinski, M., Jr. Antifungal properties and inhibitory effects upon aflatoxin production of Thymus vulgaris L. by Aspergillus flavus Link. Food Chem. 2015, 173, 1006–1010. [Google Scholar] [CrossRef]
- Divband, K.; Shokri, H.; Khosravi, A.R. Down-regulatory effect of Thymus vulgaris L. on growth and Tri4 gene expression in Fusarium oxysporum strains. Microb. Pathog. 2017, 104, 1–5. [Google Scholar] [CrossRef]
- Lagha, R.; Ben Abdallah, F.; Al-Sarhan, B.O.; Al-Sodany, Y. Antibacterial and biofilm inhibitory activity of medicinal plant essential oils against Escherichia coli isolated from UTI patients. Molecules 2019, 24, 1161. [Google Scholar] [CrossRef] [PubMed]
- Marongiu, B.; Piras, A.; Porcedda, S.; Falconieri, D.; Goncalves, M.J.; Salgueiro, L.; Maxia, A.; Lai, R. Extraction, separation and isolation of volatiles from Vitex agnus-castus L. (Verbenaceae) wild species of Sardinia, Italy, by supercritical CO2. Nat. Prod. Res. 2010, 24, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Asdadi, A.; Hamdouch, A.; Oukacha, A.; Moutaj, R.; Gharby, S.; Harhar, H.; El Hadek, M.; Chebli, B.; Idrissi Hassani, L.M. Study on chemical analysis, antioxidant and in vitro antifungal activities of essential oil from wild Vitex agnus-castus L. seeds growing in area of Argan Tree of Morocco against clinical strains of Candida responsible for nosocomial infections. J. Mycol. Med. 2015, 25, e118–e127. [Google Scholar] [CrossRef] [PubMed]
- Mahboubi, M.; Heidary Tabar, R.; Mahdizadeh, E. The anti-dermatophyte activity of Zataria multiflora essential oils. J. Mycol. Med. 2017, 27, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Shahbazi, Y. Chemical compositions, antioxidant and antimicrobial properties of Ziziphora clinopodioides Lam. essential oils collected from different parts of Iran. J. Food Sci. Technol. 2017, 54, 3491–3503. [Google Scholar] [CrossRef] [PubMed]
- Mohammadifard, F.; Alimohammadi, S. Chemical composition and role of opioidergic system in antinociceptive effect of Ziziphora clinopodioides essential oil. Basic Clin. Neurosci. 2018, 9, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Abu-Darwish, M.S.; Cabral, C.; Gonçalves, M.J.; Cavaleiro, C.; Cruz, M.T.; Paoli, M.; Tomi, F.; Efferth, T.; Salgueiro, L. Ziziphora tenuior L. essential oil from Dana Biosphere Reserve (Southern Jordan); Chemical characterization and assessment of biological activities. J. Ethnopharmacol. 2016, 194, 963–970. [Google Scholar] [CrossRef]
- Yang, D.; Michel, L.; Chaumont, J.P.; Millet-Clerc, J. Use of caryophyllene oxide as an antifungal agent in an in vitro experimental model of onychomycosis. Mycopathologia 1999, 148, 79–82. [Google Scholar] [CrossRef]
- Dahham, S.S.; Tabana, Y.M.; Iqbal, M.A.; Ahamed, M.B.; Ezzat, M.O.; Majid, A.S.; Majid, A.M. The anticancer, antioxidant and antimicrobial properties of the sesquiterpene β-caryophyllene from the essential oil of Aquilaria crassna. Molecules 2015, 20, 11808–11829. [Google Scholar] [CrossRef]
- Selestino Neta, M.C.; Vittorazzi, C.; Guimarães, A.C.; Martins, J.D.; Fronza, M.; Endringer, D.C.; Scherer, R. Effects of β-caryophyllene and Murraya paniculata essential oil in the murine hepatoma cells and in the bacteria and fungi 24-h time-kill curve studies. Pharm. Biol. 2017, 55, 190–197. [Google Scholar] [CrossRef]
- Bona, E.; Cantamessa, S.; Pavan, M.; Novello, G.; Massa, N.; Rocchetti, A.; Berta, G.; Gamalero, E. Sensitivity of Candida albicans to essential oils: Are they an alternative to antifungal agents? J. Appl. Microbiol. 2016, 121, 1530–1545. [Google Scholar] [CrossRef]
- Kordali, S.; Cakir, A.; Ozer, H.; Cakmakci, R.; Kesdek, M.; Mete, E. Antifungal, phytotoxic and insecticidal properties of essential oil isolated from Turkish Origanum acutidens and its three components, carvacrol, thymol and p-cymene. Bioresour. Technol. 2008, 99, 8788–8795. [Google Scholar] [CrossRef]
- Marei, G.I.K.; Abdel Rasoul, M.A.; Abdelgaleil, S.A.M. Comparative antifungal activities and biochemical effects of monoterpenes on plant pathogenic fungi. Pesticide Biochem. Physiol. 2012, 103, 56–61. [Google Scholar] [CrossRef]
- Abbaszadeh, S.; Sharifzadeh, A.; Shokri, H.; Khosravi, A.R.; Abbaszadeh, A. Antifungal efficacy of thymol, carvacrol, eugenol and menthol as alternative agents to control the growth of food-relevant fungi. J. Mycol. Med. 2014, 24, e51–e56. [Google Scholar] [CrossRef]
- Rivera-Yañez, C.R.; Terrazas, L.I.; Jimenez-Estrada, M.; Campos, J.E.; Flores-Ortiz, C.M.; Hernandez, L.B.; Cruz-Sanchez, T.; Garrido-Fariña, G.I.; Rodriguez-Monroy, M.A.; Canales-Martinez, M.M. Anti-Candida activity of Bursera morelensis Ramirez essential oil and two compounds, α-pinene and γ-terpinene—an in vitro study. Molecules 2017, 22, 95. [Google Scholar] [CrossRef]
- de Oliveira Lima, M.I.; Araújo de Medeiros, A.C.; Souza Silva, K.V.; Cardoso, G.N.; de Oliveira Lima, E.; de Oliveira Pereira, F. Investigation of the antifungal potential of linalool against clinical isolates of fluconazole resistant Trichophyton rubrum. J. Mycol. Med. 2017, 27, 195–202. [Google Scholar] [CrossRef] [PubMed]
- de Macêdo Andrade, A.C.; Rosalen, P.L.; Freires, I.A.; Scotti, L.; Scotti, M.T.; Aquino, S.G.; de Castro, R.D. Antifungal activity, mode of action, docking prediction and anti-biofilm effects of (+)-β-pinene enantiomers against Candida spp. Curr. Top. Med. Chem. 2018, 18, 2481–2490. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Jiang, S.; Pu, T.; Fan, L.; Su, F.; Ye, M. Antifungal activity of phenolic monoterpenes and structure-related compounds against plant pathogenic fungi. Nat. Prod. Res. 2019, 33, 1423–1430. [Google Scholar] [CrossRef]
- Shi, Y.; Si, H.; Wang, P.; Chen, S.; Shang, S.; Song, Z.; Wang, Z.; Liao, S. Derivatization of natural compound β-pinene enhances its in vitro antifungal activity against plant pathogens. Molecules 2019, 24, 3144. [Google Scholar] [CrossRef]
- Wojtunik-Kulesza, K.A.; Kasprzak, K.; Oniszczuk, T.; Oniszczuk, A. Natural monoterpenes: Much more than only a scent. Chem. Biodiv. 2019, 16, e19004. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Helal, G.A.; Sarhan, M.M.; Abu Shahla, A.N.K.; Abou El-Khair, E.K. Effects of Cymbopogon citratus L. essential oil on the growth, lipid content and morphogenesis of Aspergillus niger ML2-strain. J. Basic Microbiol. 2006, 46, 456–469. [Google Scholar] [CrossRef] [PubMed]
- Rammanee, K.; Hongpattarakere, T. Effects of tropical citrus essential oils on growth, aflatoxin production, and ultrastructure alterations of Aspergillus flavus and Aspergillus parasiticus. Food Bioprocess Technol. 2011, 4, 1050–1059. [Google Scholar] [CrossRef]
- Hyldgaard, M.; Mygind, T.; Meyer, R.L. Essential oils in food preservation: Mode of action, synergies, and interactions with food matrix components. Front. Microbiol. 2012, 3, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Basak, S.; Guha, P. A review on antifungal activity and mode of action of essential oils and their delivery as nano-sized oil droplets in food system. J. Food Sci. Technol. 2018, 55, 4701–4710. [Google Scholar] [CrossRef]
- Tariq, S.; Wani, S.; Rasool, W.; Shafi, K.; Bhat, M.A.; Prabhakar, A.; Shalla, A.H.; Rather, M.A. A comprehensive review of the antibacterial, antifungal and antiviral potential of essential oils and their chemical constituents against drug-resistant microbial pathogens. Microb. Pathog. 2019, 134. [Google Scholar] [CrossRef]
- Shin, S.; Kang, C.A. Antifungal activity of the essential oil of Agastache rugosa Kuntze and its synergism with ketoconazole. Lett. Appl. Microbiol. 2003, 36, 111–115. [Google Scholar] [CrossRef]
- Gong, H.; Li, S.; He, L.; Kasimu, R. Microscopic identification and in vitro activity of Agastache rugosa (Fisch. et Mey) from Xinjiang, China. BMC Complement Altern. Med. 2017, 17, 95. [Google Scholar] [CrossRef]
- Zhang, C.; Li, H.; Yun, T.; Fu, Y.; Liu, C.; Gong, B.; Neng, B. Chemical composition, antimicrobial and antioxidant activities of the essential oil of Tibetan herbal medicine Dracocephalum heterophyllum Benth. Nat. Prod. Res. 2008, 22, 1–11. [Google Scholar] [CrossRef]
- Souza, L.K.; de Oliveira, C.M.; Ferri, P.H.; de Oliveira Júnior, J.G.; de Souza Júnior, A.H.; Fernandes Ode, F.; Silva Mdo, R. Antimicrobial activity of Hyptis ovalifolia towards dermatophytes. Memórias do Instituto Oswaldo Cruz 2003, 98, 963–965. [Google Scholar] [CrossRef]
- Dolatabadi, S.; Salari, Z.; Mahboubi, M. Antifungal effects of Ziziphora tenuior, Lavandula angustifolia, Cuminum cyminum essential oils against clinical isolates of Candida albicans from women suffering from vulvovaginal candidiasis. Infect 2019, 23, 222–226. [Google Scholar] [CrossRef]
- Powers, C.N.; Osier, J.L.; McFeeters, R.L.; Brazell, C.B.; Olsen, E.L.; Moriarity, D.M.; Satyal, P.; Setzer, W.N. Antifungal and cytotoxic activities of sixty commercially-available essential oils. Molecules 2018, 23, 1549. [Google Scholar] [CrossRef] [PubMed]
- Ozcakmak, S.; Dervisoglu, M.; Yilmaz, A. Antifungal activity of lemon balm and sage essential oils on the growth of ochratoxigenic Penicillium verrucosum. Afr. J. Microbiol. Res. 2012, 6, 3079–3084. [Google Scholar] [CrossRef]
- Tyagi, A.K.; Malik, A. Liquid and vapour-phase antifungal activities of selected essential oils against Candida albicans: Microscopic observations and chemical characterization of Cymbopogon citratus. BMC Complement Altern. Med. 2010, 10, 65. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Rajendran, S.; Srivastava, A.; Sharma, S.; Kundu, B. Antifungal activities of selected essential oils against Fusarium oxysporum f. sp. lycopersici 1322, with emphasis on Syzygium aromaticum essential oil. J. Biosci. Bioeng. 2017, 123, 308–313. [Google Scholar]
- Piras, A.; Porcedda, S.; Falconieri, D.; Maxia, A.; Gonçalves, M.; Cavaleiro, C.; Salgueiro, L. Antifungal activity of essential oil from Mentha spicata L. and Mentha pulegium L. growing wild in Sardinia island (Italy). Nat. Prod. Res. 2019. [Google Scholar] [CrossRef]
- Fancello, F.; Zara, S.; Petretto, G.L.; Chessa, M.; Addis, R.; Rourke, J.P.; Pintore, G. Essential oils from three species of Mentha harvested in Sardinia: Chemical characterization and evaluation of their biological activity. Int. J. Food Prop. 2017, 20, 1751–1761. [Google Scholar] [CrossRef]
- Houicher, A.; Hechachna, H.; Teldji, H.; Ozogul, F. In vitro study of the antifungal activity of essential oils obtained from Mentha spicata, Thymus vulgaris, and Laurus nobilis. Recent Pat. Food Nutr. Agric. 2016, 8, 99–106. [Google Scholar] [CrossRef]
- Ji, H.; Kim, H.; Beuchat, L.R.; Ryu, J.H. Synergistic antimicrobial activities of essential oil vapours against Penicillium corylophilum on a laboratory medium and beef jerky. Int. J. Food Microbiol. 2019, 291, 104–110. [Google Scholar] [CrossRef]
- Pietrella, D.; Angiolella, L.; Vavala, E.; Rachini, A.; Mondello, F.; Ragno, R.; Bistoni, F.; Vecchiarelli, A. Beneficial effect of Mentha suaveolens essential oil in the treatment of vaginal candidiasis assessed by real-time monitoring of infection. BMC Complement Altern. Med. 2011, 11, 18. [Google Scholar] [CrossRef]
- Angiolella, L.; Vavala, E.; Sivric, S.; D’Auria, F.D.; Ragno, R. In vitro activity of Mentha suaveolens essential oil against Cryptococcus neoformans and dermatophytes. Int. J. Essent. Oil Ther. 2010, 4, 35–36. [Google Scholar]
- Bisht, D.S.; Padalia, R.C.; Singh, L.; Pande, V.; Lal, P.; Mathela, C.S. Constituents and antimicrobial activity of the essential oils of six Himalayan Nepeta species. J. Serb. Chem. Soc. 2010, 75, 739–747. [Google Scholar] [CrossRef]
- Cardoso, N.N.; Alviano, C.S.; Blank, A.F.; Arrigoni-Blank, M.F.; Romanos, M.T.; Cunha, M.M.; da Silva, A.J.; Alviano, D.S. Anti-cryptococcal activity of ethanol crude extract and hexane fraction from Ocimum basilicum var. Maria bonita: Mechanisms of action and synergism with amphotericin B and Ocimum basilicum essential oil. Pharm. Biol. 2017, 55, 1380–1388. [Google Scholar] [CrossRef] [PubMed]
- Al-Hajj, N.Q.M.; Wang, H.X.; Ma, C.; Lou, Z.; Bashari, M.; Thabit, R. Antimicrobial and antioxidant activities of the essential oils of some aromatic medicinal plants (Pulicaria inuloides-Asteraceae and Ocimum forskolei-Lamiaceae). Trop. J. Pharmaceut. Res. 2014, 13, 1287–1293. [Google Scholar] [CrossRef]
- Nakamura, C.V.; Ishida, K.; Faccin, L.C.; Filho, B.P.; Cortez, D.A.; Rozental, S.; de Souza, W.; Ueda-Nakamura, T. In vitro activity of essential oil from Ocimum gratissimum L. against four Candida species. Res. Microbiol. 2004, 155, 579–586. [Google Scholar] [CrossRef]
- Silva, M.R.; Oliveira, J.G., Jr.; Fernandes, O.F.; Passos, X.S.; Costa, C.R.; Souza, L.K.; Lemos, J.A.; Paula, J.R. Antifungal activity of Ocimum gratissimum towards dermatophytes. Mycoses 2005, 48, 172–175. [Google Scholar] [CrossRef]
- Bedoya-Serna, C.M.; Dacanal, G.C.; Fernandes, A.M.; Pinho, S.C. Antifungal activity of nanoemulsions encapsulating oregano (Origanum vulgare) essential oil: In vitro study and application in Minas Padrão cheese. Braz. J. Microbiol. 2018, 49, 929–935. [Google Scholar] [CrossRef]
- Vinciguerra, V.; Rojas, F.; Tedesco, V.; Giusiano, G.; Angiolella, L. Chemical characterization and antifungal activity of Origanum vulgare, Thymus vulgaris essential oils and carvacrol against Malassezia furfur. Nat. Prod. Res. 2018, 33, 3273–3277. [Google Scholar] [CrossRef]
- Wang, G.S.; Deng, J.H.; Ma, Y.H.; Shi, M.; Li, B. Mechanisms, clinically curative effects, and antifungal activities of cinnamon oil and pogostemon oil complex against three species of Candida. J. Tradit. Chin. Med. 2012, 32, 19–24. [Google Scholar] [CrossRef]
- Farisa Banu, S.; Rubini, D.; Shanmugavelan, P.; Murugan, R.; Gowrishankar, S.; Karutha Pandian, S.; Nithyanand, P. Effects of patchouli and cinnamon essential oils on biofilm and hyphae formation by Candida species. J. Mycol. Med. 2018, 28, 332–339. [Google Scholar] [CrossRef]
- Sookto, T.; Srithavaj, T.; Thaweboon, S.; Thaweboon, B.; Shrestha, B. In vitro effects of Salvia officinalis L. essential oil on Candida albicans. Asian Pac. J. Trop. Biomed. 2013, 3, 376–380. [Google Scholar] [CrossRef]
- Sharifzadeh, A.; Khosravi, A.R.; Ahmadian, S. Chemical composition and antifungal activity of Satureja hortensis L. essentiall oil against planktonic and biofilm growth of Candida albicans isolates from buccal lesions of HIV(+) individuals. Microb. Pathog. 2016, 96, 1–9. [Google Scholar] [CrossRef]
- Marković, T.; Chatzopoulou, P.; Šiljegović, J.; Nikolić, M.; Glamočlija, J.; Ćirić, A.; Soković, M. Chemical analysis and antimicrobial activities of the essential oils of Satureja thymbra L. and Thymbra spicata L. and their main components. Arch. Biol. Sci. Belgrade 2011, 63, 457–464. [Google Scholar] [CrossRef]
- Tabti, L.; Dib Mel, A.; Gaouar, N.; Samira, B.; Tabti, B. Antioxidant and antifungal activity of extracts of the aerial parts of Thymus capitatus (L.) Hoffmanns against four phytopathogenic fungi of Citrus sinensis. Jundishapur J. Nat. Pharm. Prod. 2014, 9, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Nasir, M.; Tafess, K.; Abate, D. Antimicrobial potential of the Ethiopian Thymus schimperi essential oil in comparison with others against certain fungal and bacterial species. BMC Complement Altern. Med. 2015, 15, 260. [Google Scholar] [CrossRef] [PubMed]
- Sokolić-Mihalak, D.; Frece, J.; Slavica, A.; Delaš, F.; Pavlović, H.; Markov, K. The effects of wild thyme (Thymus serpyllum L.) essential oil components against ochratoxin-producing Aspergilli. Arhiv za Higijenu i Toksikologiju 2012, 63, 457–462. [Google Scholar] [CrossRef]
- Perina, F.J.; Amaral, D.C.; Fernandes, R.S.; Labory, C.R.; Teixeira, G.A.; Alves, E. Thymus vulgaris essential oil and thymol against Alternaria alternata (Fr.) Keissler: Effects on growth, viability, early infection and cellular mode of action. Pest Manag. Sci. 2015, 71, 1371–1378. [Google Scholar] [CrossRef]
- Sharifzadeh, A.; Javan, A.J.; Shokri, H.; Abbaszadeh, S.; Keykhosravy, K. Evaluation of antioxidant and antifungal properties of the traditional plants against foodborne fungal pathogens. J. Mycol. Med. 2016, 26, e11–e17. [Google Scholar] [CrossRef]
- Liu, J.; Sui, G.; He, Y.; Liu, D.; Yan, J.; Liu, S.; Qin, W. Prolonging storage time of baby ginger by using a sand-based storage medium and essential oil treatment. J. Food Sci. 2014, 79, M593–M599. [Google Scholar] [CrossRef]
- Moghadam, H.D.; Sani, A.M.; Sangatash, M.M. Antifungal activity of essential oil of Ziziphora clinopodioides and the inhibition of aflatoxin B1 production in maize grain. Toxicol. Ind. Health 2016, 32, 493–499. [Google Scholar] [CrossRef]
- Dreger, M.; Wielgus, K. Application of essential oils as natural cosmetic preservatives. Herba Pol. 2013, 59, 142–156. [Google Scholar] [CrossRef]
- Sarkic, A.; Stappen, I. Essential oils and their single compounds in cosmetics—A critical review. Cosmetics 2018, 5, 11. [Google Scholar] [CrossRef]
- Ali, B.; Al-Wabel, N.A.; Shams, S.; Ahamad, A.; Khan, S.A.; Anwar, F. Essential oils used in aromatherapy: A systemic review. Asian Pac. J. Trop. Biomed. 2015, 5, 601–611. [Google Scholar] [CrossRef]
- Helms, S.; Miller, A.L. Natural treatment of chronic rhinosinusitis. Altern. Med. Rev. 2006, 11, 196–207. [Google Scholar]
- Schilcher, H. Efficient phytotherapy. Herbal medicines in the upper respiratory tract for catarrh. Herba Pol. 2000, 46, 52–57. [Google Scholar]
- Vranic, E.; Lacević, A.; Mehmedagić, A.; Uzunovic, A. Formulation ingredients for toothpastes and mouthwashes. Bosnian J. Basic Med. Sci. 2004, 4, 51–58. [Google Scholar] [CrossRef]
- Guven, Y.; Ustun, N.; Tuna, E.B.; Aktoren, O. Antimicrobial effect of newly formulated toothpastes and a mouthrinse on specific microorganisms: An in vitro study. Eur. J. Dent. 2019, 13, 172–177. [Google Scholar] [CrossRef]
- DOZ Pharmacy. Available online: https://www.doz.pl/ (accessed on 17 December 2019).
- Ehlers, V.; Helm, S.; Kasaj, A.; Willershausen, B. The effect of Parodontax® on the MMP-8 concentration in gingivitis patients. Schweiz Monatsschr. Zahnmed. 2011, 121, 1041–1051. [Google Scholar]
- Adwan, G.; Salameh, Y.; Adwan, K.; Barakat, A. Assessment of antifungal activity of herbal and conventional toothpastes against clinical isolates of Candida albicans. Asian Pac. J. Trop. Biomed. 2012, 2, 375–379. [Google Scholar] [CrossRef]
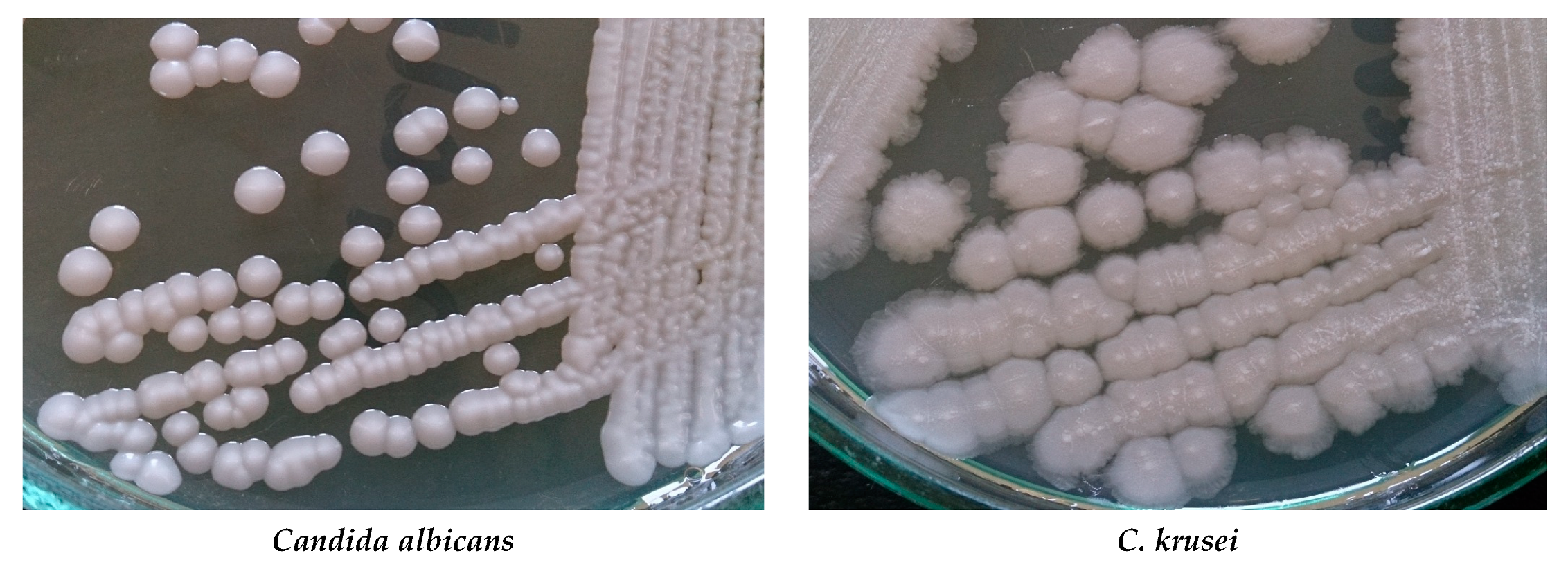
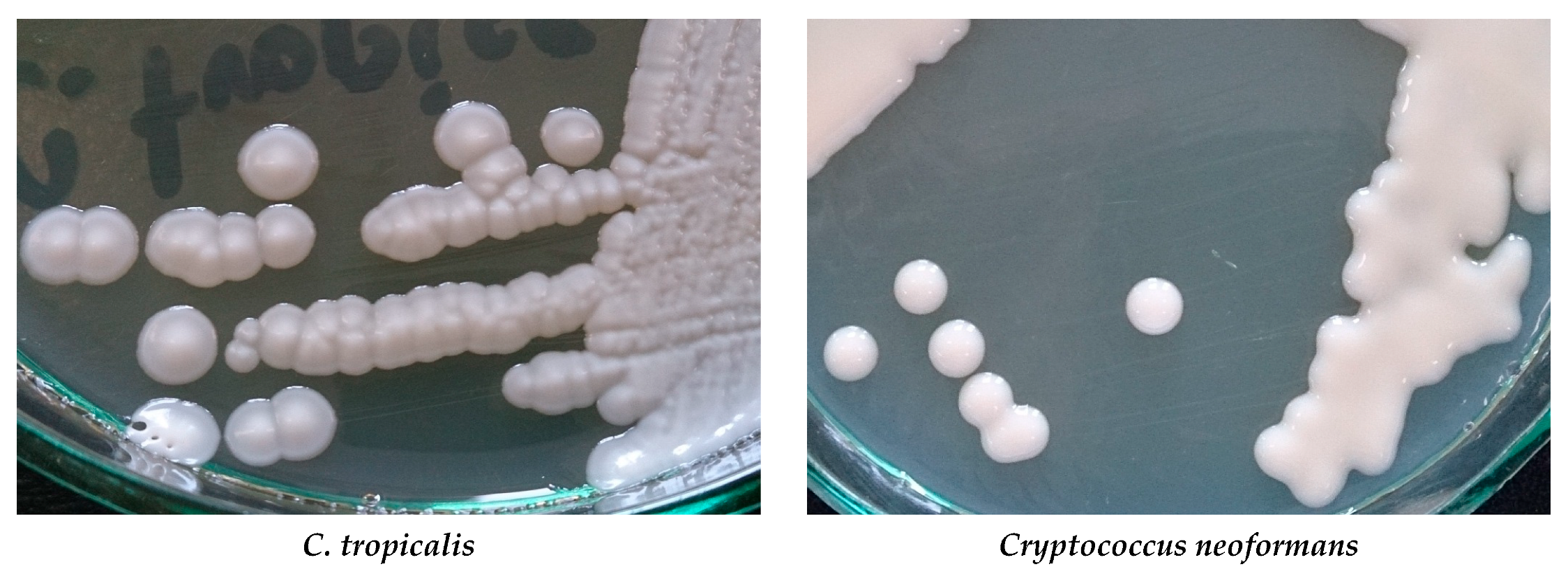
| Superficial mycoses |
|
| Cutaneous and subcutaneous mycoses |
|
| Endemic mycoses |
|
| Opportunistic mycoses |
|
| Essential Oil | Main Chemical Components | References |
|---|---|---|
| Aeollanthus suaveolens Mart. ex Spreng. = A. heliotropioides Oliv. | Linalool (38.5%), α-Farnesene (25.1%), Massoialactone (4.5%), β-Caryophyllene (3.6%), Germacrene D (2.0%) | [34] |
| Agastache rugosa (Fisch. and C.A.Mey.) Kuntze | Methyl chavicol (93.45%), Methyl eugenol (2.48–50.51%), Estragole (8.55%), Eugenol (0.15–7.54%), Thymol (3.62%), Pulegone (2.56%), Limonene (2.49%), β-Caryophyllene (1.19–2.38%), | [35,36] |
| Ballota nigra subsp. foetida (Vis.) Hayek | β-Caryophyllene (21.8–22.6%), Caryophyllene oxide (18.0–20.5%), Germacrene D (13.1–16.5%), 2-Hexenal (6.5–11.2%), 1-Octen-3-ol (3.5–5.5%), β-Pinene (1.6–4.4%), Limonene (2.2–4.1%), Linalool (1.2–3.5%), β-Bourbonene (1.5–2.7%), α-Humulene (2.2–2.6%), α-Copaene (1.5–2.2%) | [37] |
| Clinopodium dalmaticum (Benth.) Bräuchler and Heubl = Micromeria dalmatica Benth. | Piperitenone oxide (41.77%), Pulegone (15.94%), Piperitenone (10.19%), Limonene (5.77%), Piperitone (3.39%), α-Pinene (2.9%), β-Pinene (2.16%), | [38] |
| Clinopodium nepeta subsp. glandulosum (Req.) Govaerts = Calamintha glandulosa (Req.) Bentham = Calamintha officinalis Moench | Piperitenone (trace–42.6%), Piperitone (0.0–40.3%), Carvone (1–38.7%), Pulegone (0.6–9.7%), Shisofuran (0.1–9.7%), Menthone (trace–8.3%), Dihydrocarveol acetate (0.1–7.6%), Dihydrocarveol (0–6.9%),1,8-Cineole (0.0–6.4%), cis-Carvyl acetate (0.0–6.1%), | [39,40] |
| Clinopodium nepeta (L.) Kuntze = Calamintha nepeta (L.) Savi | Pulegone (2.4–84.7%), Isomenthone (1.9–51.3%), Menthone (0.0–35.4%), Crysanthenone (1.3–33.9%), 1,8-Cineole (0.3–21.4%), Piperitenone oxide (0.0–19.1%), Limonene (0.0–13.6%), Isopulegone (0.0–9.4%), Piperitenone (0.0–7.7%), Cinerolone (0.0–5.8%), Isopulegol (0.0–4.1%), Isomenthol (0.0–3.9%), β-Caryophyllene (0.0–3.8%), 3-Octanol (0.0–3.0%), β-Pinene (0.0–2.3%), cis-Piperitone oxide (0.0–2.2%) | [41,42] |
| Clinopodium thymifolium (Scop.) Kuntze = Micromeria thymifolia (Scop.) Fritsch | Pulegone (32.81%), Piperitenone (25.7%), Piperitone (11.71%), Isomenthone (4.98%), Limonene (2.4%), β-Caryophyllene (2.39%) | [38] |
| Clinopodium umbrosum (M.Bieb.) Kuntze = Calamintha umbrosa Benth. | β-Caryophyllene (13.9%), Germacrene D (11.6%), Spathulenol (10.6%) | [43] |
| Dracocephalum heterophyllum Benth. | Citronellol (74.2%), Geraniol (2.8%), cis-Rose oxide (2.2%), Citronellyl acetate (1.7%) | [44] |
| Hymenocrater longiflorus Benth. | δ-Cadinol (18.49%), α-Pinene (10.16%), p-Menth-1-en-8-ol (9.82%), Hedycaryol (6.42%), β-Eudesmol (4.56%), Spathulenol (4.14%), δ-Cadenene (3.02%), Linalool (2.98%), Caryophyllene oxide (2.81%), β-Bourbonene (2.72%), β-Caryophyllene (2.29%) | [45] |
| Hyptis ovalifolia Benth. | (R)-6-[(Z)-1-Heptenyl]-5,6-dihydro-2H-pyran-2-one (60.0%), γ-Cadinene (6.6%), Viridiflorol (6.08%), Caryophyllene oxide (4.98%), γ-Elemene (4.38%) | [46] |
| Hyssopus officinalis L. | Pinocamphone (5.78–50.77%), 1,8-Cineole (0.47–36.43%), Pinocarvone (0.44–23.4%), β-Pinene (13.38–19.55%), Isopinocamphone (15.32%), α-Phellandrene (trace–3.74%), Sabinene (1.7–2.9%), Myrtenol (1.39–2.7%), α-Pinene (1.01–2.57%), cis-Sabinene hydrate (0.0–2.5%), Myrtenyl methyl ether (1.64–2.1%) | [44,47,48] |
| Lavandula angustifolia Mill. | Linalool (20.18–45.8%), Linalyl acetate (4.6–43.13%), Lavandulyl acetate (0–16.01%), 1,8-Cineole (0.6–13.1%), Camphor (0.52–11.2%), Borneol (0.76–7.5%), Terpinen-4-ol (1.05–5.8%), β-Caryophyllene (0.6–4.95%), Lavandulol (0–3.09%), β-Ocimene (1.5–2.84%), Myrcene (0.4–2.41%) | [49,50,51] |
| Lavandula multifida L. | Carvacrol (41.5–42.8%), β-Ocimene (27.0–27.4%), Myrcene (5.5–5.7%), β-Bisabolene (5.0–5.6%), Terpinolene (2.1–3.1%), α-Farnesene (2.6–2.8%) | [52] |
| Lavandula pedunculata (Mill.) Cav. | Fenchone (6.2–44.5%), 1,8-Cineole (5.1–34.3%), Camphor (8.7–34.0%), β-Pinene (1.4–9.0%), α-Pinene (2.5–8.0%), Camphene (0.8–6.1%), Linalool (0.5–3.8%), Bornyl acetate (0.9–3.5%), Borneol (0.6–3.4%), α-Cadinol (0.2–3.1%), cis-Verbenol (0.2–2.8%), Myrtenal (0.8–2.4%), trans-Verbenol (1.1–2.0%) | [53] |
| Lavandula stoechas L. | Fenchone (0.0–36.2%), 1,8-Cineole (0–33.9%), Camphor (2.2–18%), α-trans-Necrodyl acetate (0.0–17.4%), Lavandulyl acetate (0.0–7.6%), α-trans-Necrodol (0.0–7.1%), Linalool (0.0–6.2%), α-Copaene-8-ol (0.7–4.7%), Viridiflorol (1.4–3.6%), α-Pinene (1.1–3.2%), 2,3,4,4-Tetramethyl-5-methylene-cyclopenten-1-one (0.0–2.8%), Lyratyl acetate (0–2.4%), Myrtenyl acetate (1.0–2.0%), 1,1,2,3-Tetramethyl-4-hidroximethyl-2-cyclopentene (0.0–2.0%) | [51,54] |
| Lavandula viridis L’Her. | 1,8-Cineole (34.5–42.2%), Camphor (13.4%), α-Pinene (9.0%), Linalool (6.7–7.9%) | [55] |
| Lepechinia mutica (Benth.) Epling | Δ3-Carene (8.69–24.23%), Thujopsan-2-α-ol (0.0–11.9%), Shyobunol (0.0–10.8%), β-Pinene (3.78–7.96%), δ-Cadinene (0.0–6.96%), Globulol (0.0–5.91%), Valerianol (0.0–5.19%), epi-Cubebol (0.0–4.62%), β-Caryophyllene (0.0–4.55%), Limonene (3.79–4.47%), α-Eudesmol (0.0–4.47%), α-Phellandrene (0.34–3.8%), β-Phellandrene (3.79%), γ-Cadinene (0.0–2.86%), α-Pinene (1.23–2.68%), o-Cymene (0.0–2.04%), Isobornyl acetate (0.0–2.2%) | [56,57] |
| Marrubium vulgare L. | γ-Eudesmol (11.93%), β-Citronellol (9.9%), Citronellyl formate (9.5%), Germacrene-D (9.37%), Geranyl formate (6.25%), Geranyl tiglate (5.53%), Ledene (5.35%), 1,8-Cineole (3.72%), Neryl acetate (3.41%), δ-Cadinene (3.3%), Cyclononasiloxane octadecamethyl (3.08%), Geraniol (2.74%), N-trimethylsilyl trifluoroacetamide (2.35%), Eicosamethylcyclodecasiloxane (2.29%), α-Thujone (2.29%), trans-Caryophyllene (2.15%) | [58] |
| Melissa officinalis L. | Geranial (23.4%), Neral (16.5%), Citronellal (13.7%), β-Caryophyllene (4.6%), Geraniol (3.4%), Isomenthone (3.0%), Menthol (2.9%), Methyl citronellate (2.7%), Germacrene D (2.4%), Limonene (2.2%) | [59] |
| Mentha cervina L. | Isomenthone (8.7–77%), Pulegone (12.9–75.1%), Menthone (0.8–4.4%), Limonene (0.8–4.3%) | [60] |
| Mentha × piperita L. | Menthol (34.82–43.85%), Menthone (9.1–31.68%), Carvone (0.0–19.54%), Menthyl acetate (1.64–17.4%), Anethole (0.0–9.54%), Isomenthone (4.71–8.08%), Limonene (0.86–6.9%), Menthofuran (6.8%), Eucalyptol (4.36–6.21%), 1,8-Cineole (5.6%), Pulegone (0.47–5.15%), Isomenthol acetate (4.56–4.91%), Isomenthol (0.68–3.58%), Sabinene (0.0–2.5%) | [61,62,63,64] |
| Mentha pulegium L. | Pulegone (2.3–70.66%), Piperitone (0.24–38.0%), Piperitenone (1.58–33.0%), Neomenthol (11.21%), α-Terpineol (0.0–4.7%), 1,8-Cineole (0.11–4.0%), Piperitenone oxide (0.0–3.4%), Menthone (2.63–3.0%), Borneol (0.0–2.9%), Isopulegone (2.33%) | [65,66] |
| Mentha requienii Benth. | Pulegone (77.6%), Isomenthone (18.2%), Limonene (1.76%) | [67] |
| Mentha spicata L. | Pulegone (0.0–78.7%), Carvone (0.0–59.12%), Menthol (0.0–39%), Menthone (5.1–21.9%), Neomenthol (11.2%), Menthyl acetate (0.0–6.9%), Dihydrocarveol (0.0–6.27%), Limonene (1.0–5.8%), 1,8-Cineole (3.0–5.42%), cis-Dihydrocarvone (0.0–4.9%), cis-Carveol (0.0–3.9%), β-Caryophyllene (0.7–2.8%), β-Myrcene (0.3–2.3%) | [49,51,61,68] |
| Mentha suaveolens Ehrh. | Piperitenone oxide (0.0–87.25%), Carvone (0.0–50.59%), Pulegone (0.0–50.0%), Demelverine (0.0–43.46%), Cinerolone (0.0–38.79%), p-Cymenene (0.0–35.22%), Limonene (0.0–31.25%), Piperitone oxide (0.0–26.0%), p-Cymenol-8 (0.0–20.6%), Spathulenol (0.0–18.35%), β-Caryophyllene oxide (0.3–17.25%), α-Pharnesene (0.0–16.54%), α-Cadinol (0.09–10.69%), Calamenene (0.44–10.63%), α-Cubenene (0.0–10.08%), α-Caryophyllene (2.0–9.8%), Veridiflorol (0.0–7.59%), Cubenol (0.0–7.46%), Verbenone (0.0–6.56%), δ-Fenchol (0.3–5.9%), Menthone (0.0–5.7%), Borneol (0.12–5.6%), Citronellyl acetate (0.0–5.45%), δ-Cadinene (0.0–4.89), Eucalyptol (0.0–4.21%), cis-8-Menthene (0.3–4.2%), Fenchone (0.1–3.6%), Geraniol (1.0–3.4%), τ-Muurolol (0.0–3.29%), α-Pinene (0.1–2.7%), β-Caryophyllene (2.56%), cis-Carveol (2.31%), Germacrene D (0.0–2.04%) | [69,70,71] |
| Micromeria albanica (K. Maly) Silic | Piperitenone oxide (38.73%), Pulegone (13.43%), Piperitenone (9.72%), Piperitone (5.62%), Limonene (3.2%), α-Copaene (2.12%) | [38] |
| Moluccella spinosa L. | α-Pinene (26.6%), Caryophyllene oxide (16.8%), β-Caryophyllene (8.6%), α-Thujene (5.9%), Nonacosane (5.5%), Heptacosane (5.3%), Ethylbenzaldehyde (3.4%), Pentacosane (2.5%), Tetracosane (2.3%), Sabinene (2.2%) | [72] |
| Nepeta ciliaris Benth. = Nepeta leucophylla Benth. | Caryophyllene oxide (14.8–26.3%), β-Caryophyllene (18.0%), β-Sesquiphellandrene (15.0%), Iridodial b-monoenol acetate (9.8%) | [43] |
| Nepeta clarkei Hook. f. | β-Sesquiphellandrene (22.0%), Actinidine (10.0%), Germacrene D (8.0%) | [43] |
| Ocimum basilicum L. | Linalool (18.0–68.0%), Methyl chavicol (0.0–57.3%), Geraniol (0.0–16.5%), 1,8-Cineole (1.4–15.1%), p-Allylanisole (0.2–13.8%), Eugenol (0.0–12.32%), Limonene (0.2–10.4%), β-Farnesene (0.0–6.3%), τ-Cadinol (trace–5.8%), β-Caryophyllene (0.0–4.5%), α-Bergamotene (0.0–4.34%), α-Cadinol (0.0–4.05%), β-Elemene (0.0–3.62%), δ-Cadinene (0.0–3.6%), Germacrene D (0.0–3.5%), γ-Cadinene (0.0–2.8%), Camphor (0.0–2.4%), β-Myrcene (0.2–2.3%), Terpinen-4-ol (0.0–2.2%), Guaiene (0.0–2.1%), Estragole (0.0–2.03%), Isolimonene (0.0–2.0%), α-Bulnesene (0.0–2.0%), γ-Terpinene (0.0–2.0%) | [64,68,73,74,75,76] |
| Ocimum × africanum Lour. = Ocimum × citriodorum | Nerol (23.0%), Geranial (15.77%), Methyl chavicol (9.45%), Linalool (9.42%), β-Bisabolenene (8.31%), β-Caryophyllene (7.8%), Geraniol (5.2%), Neral (4.93%), α-Bergamotene (3.52%), α-Bisabolene (2.29%), β-Cubebene (2.26%) | [76] |
| Ocimum campechianum Mill. = Ocimum micranthum Willd. | Eugenol (46.55%), β-Caryophyllene (11.94%), β-Elemene (9.06%), 1,8-Cineole (5.35%), δ-Elemene (4.17%), Bicyclogermacrene (2.9%), cis-Ocimene (2.69%), allo-Ocimene (2.42%), α-Humulene (2.4%) | [73] |
| Ocimum forskolei Benth. | endo-Fenchol (31.1%), τ-Cadinol (12.2%), Fenchone (12.2%), Camphor (6.2%), Linalool (5.7%), Methyl(E)-cinnamate (5.1%), α-Bergamotene (3.1%), γ-Cadinene (2.9%), endo-Fenchyl acetate (2.8%), Limonene (2.5%) | [77] |
| Ocimum gratissimum L. | Eugenol (7.42–57.82%), Ethyl cinnamate (0.0–34.0%), Linalool (30.0–32.95%), 1,8-Cineole (6.5–21.91%), α-Bisabolene (0.0–17.19%), Camphor (3.8–11.97%), Thymol (0.0–9.8%), α-Cadinol (5.18%), Germacrene D (0.79–4.76%), α-Terpineol (3.36%), γ-Terpinene (0.0–3.06%), β-Caryophyllene (1.68–3.03%), p-Cymene (0.0–2.11%) | [78,79,80] |
| Ocimum tenuiflorum L. = Ocimum sanctum L. | Eugenol (0.0–61.3%), Methyl chavicol (0.0–44.63%), Linalool (0.26–21.84%), α-Caryophyllene (3.3–11.89%), Germacrene D (0.37–9.14%), Carvone (0.0–6.31%), Limonene (0.71–4.39%), β-Caryophyllene (1.4–3.3%), α-Cubebene (0.0–2.54%), Carvacrol (0.0–2.04%) | [81,82,83] |
| Origanum compactum Benth. | Carvacrol (43.26%), Thymol (21.64%), p-Cymene (13.95%), γ-Terpinene (11.28%) | [84] |
| Origanum majorana L. | Terpinen-4-ol (6.66–33.84%), Sabinene hydrate (2.31–28.33%), 1,8-Cineole (0.0–20.9%), Carvacrol (0.0–20.8%), γ-Terpinene (7.59–19.5%), Thymol (0.0–12.18%), α-Terpinene (3.03–10.08%), β-Phellandrene (1.96–8.0%), p-Cymene (2.45–7.84%), Sabinene (3.2–6.7%), Limonene (0.0–5.3%), α-Terpineol (2.7–4.7%), Linalool (0.0–4.4%), Terpinolene (0.98–3.76%), Linalool acetate (1.82–3.2%), Geraniol (2.7%), β-Caryophyllene (1.7–2.38%), α-Pinene (0.0–2.0%) | [62,68,85,86,87] |
| Origanum vulgare L. | Pulegone (0.0–77.45%), Carvacrol (0.21–65.9%), Cymenol (0.0–58.6%), Thymol (3.7–45.22%), o-Cymene (0.0–14.33%), Terpinen-4-ol (0.03–12.55%), β-Terpineol (0.0–10.46%), p-Cymene (0.5–9.3%), γ-Terpinene (3.1–9.12%), Borneol (0.0–6.1%), α-Pinene (0.0–5.1%), Menthone (0.0–4.86%), Linalool (0.0–4.8%), β-Bisabolene (0.0–4.5%), Caryophyllene oxide (0.0–4.5%), Sabinene (0.0–3.91%), β-Phellandrene (0.0–3.74%), β-Caryophyllene (00–3.7%), α-Terpineol (0.0–3.35%), Sabinene hydrate (0.0–3.31%), α-Cadinol (0.0–3.3%), α-Terpinene (1.63–3.1%), Eucalyptol (0.0–2.8%), β-Ocimene (0.0–2.77%), cis-Isopulegone (2.22%), β-Myrcene (0.0–2.2%), Anisole (0.0–2.13%), Piperitenone (0.0–2.13%), Germacrene D (0.0–1.23%) | [49,62,64,68,74,88,89,90,91] |
| Pogostemon cablin (Blanco) Benth. | Patchouli alcohol (38.3–44.52%), α-Bulnesene (0.0–13.3%), δ-Guaiene (12.64%), α-Guaiene (8.89–9.6%), Pogostol (0.0–6.2%), Seychellene (5.8%), α-Bergamotene (5.76%), Eremophilene (4.34%), β-Guaiene (3.54%), β-Caryophyllene (1.93–3.0%), β-Patchoulene (1.8–2.77%) | [92,93] |
| Pogostemon heyneanus Benth. | Acetophenone (51.0%), Patchouli alcohol (14.0%), Nerolidol (5.4%), β-Pinene (5.3%), Limonene (4.0%), Benzoyl acetone (3.1%), α-Pinene (2.4%), β-Caryophyllene (2.0%) | [93] |
| Premna microphylla Turcz. | Blumenol C (49.7%), β-Cedrene (6.1%), Limonene (3.8%), α-Guaiene (3.3%), Cryptone (3.1%), α-Cyperone (2.7%), cis-14-nor-Muurol-5-en-4-one (2.4%) | [94] |
| Rosmarinus officinalis L. | α-Pinene (5.4–37.9%), 1,8-Cineole (0.88–26.54%), Eucalyptol (0.0–24.34%), Limonene (0.0–21.7%), Camphor (2.45–21.6%), Myrcene (0.9–20.18%), Borneol (0.0–18.08%), Bornyl acetete (0.92–14.9%), Verbenone (1.36–12.0%)Camphene (1.7–11.38%), Linalool oxide (0.0–10.8%), β-Pinene (0.0–6.95%), β-Caryophyllene (0.0–6.3%), Linalool (00–5.32%), o-Cymene (0.0–4.43%), p-Cymene (0.0–4.34%), β-Phellandrene (0.0–3.9%), Sabinene (0.0–3.72%), α-Terpineol (1.19–3.36%), Isobornyl acetate (0.0–3.3%), Carvacrol (0.0–3.15%), Verbenol (0.7–3.03%), α-Humulene (0.0–2.6%), α-Terpinene (0.21–2.4%), Terpinen-4-ol (0.34–2.15%) | [51,62,68,87,91,95,96,97,98] |
| Salvia fruticosa Miller | 1,8-Cineole (16.9–54.4%), Camphor (0.6–18.34%), Manool (0–11.2%), β-Thujone (0.6–9.0%), β-Pinene (0.0–9.0%), Sabinene (0.0–8.6%), Viridiflorol (0.0–8.4%)β-Caryophyllene (1.53–8.3%), α-Thujone (trace–8.1%), Borneol (0.0–8.0%), Camphene (0.0–7.0%), α-Pinene (1.5–6.85%), Bornyl acetate (0.0–6.8%), α-Terpineol (trace–6.7%), Myrcene (1.3–5.2%), Caryophyllene oxide (0.0–3.9%), α-Terpinyl acetate (0.0–2.2%), α-Humulene (0.16–1.5%) | [49,51,99] |
| Salvia mirzayanii Rech. f. and Esfand | 1,8-Cineole (41.2%), Linalool acetate (10.7%), α-Terpinyl acetate (5.7%), Myrcene (4.7%), Geranyl acetate (3.7%), γ-Cadinene (3.3%), Linalool (2.5%), Neryl acetate (2.3%) | [100] |
| Salvia officinalis L. | 1,8-Cineole (4.2–50.3%), Camphor (8.8–25.0%), α-Thujone (1.2–19.9%), Viridiflorol (0.5–17.5%), β-Thujone (0.1–9.9%), β-Pinene (0.8–7.3%), β-Caryophyllene (1.4–5.5%), Borneol (1.5–5.4%), α-Pinene (0.5–4.8%), Camphene (0.2–3.9%), Bornyl acetate (0.2–3.3%), α-Terpineol (0.0–3.1%), α-Terpenyl acetate (1.4–2.9%), α-Humulene (0.4–2.6%),α-Farnesene (0.0–2.5%), Eicosane (0.0–2.0%) | [96,101] |
| Salvia sclarea L. | Linalyl acetate (84%), Caryophyllene oxide (24.1%), Linalool (13.6%), 1H-Naphtho(2,1,6)pyran (8.6%), Sclareol (11.5%), Spathulenol (11.4%), β-Caryophyllene (5.1%) | [85,102] |
| Satureja hortensis L. | Thymol (23.12–29.0%), Carvacrol (24.5–26.5%), γ-Terpinene (20.72–22.6%), p-Cymene (6.3–9.3%), α-Terpinene (2.2–2.93%), α-Pinene (2.6–2.91%), β-Pinene (0.92–2.7%), Limonene (0.0–2.55%), β-Bisabolene (0.2–2.2%) | [103,104] |
| Satureja montana L. | Carvacrol (47.1%), p-Cymene (9.0%), γ-Terpinene (6.1%), β-Caryophyllene (3.6%), Linalool (3.1%), Thymol (2.6%), Borneol (2.1%) | [68] |
| Satureja thymbra L. | Thymol (25.16–44.5%), γ-Terpinene (11.1–39.23%), p-Cymene (7.17–21.7%), Carvacrol (4.18–5.3%), Carvacrol methyl ether (0.1–3.33%), α-Terpinene (1.0–3.26%), β-Caryophyllene (1.2–2.76%), Caryophyllene oxide (0.32–2.0%) | [51,105] |
| Stachys cretica L. | Germacrene D (12.9–20.3%), β-Caryophyllene (0.9–9.5%), α-Pinene (0.7–8.6%), Octacosane (0.0–7.2%), β-Pinene (1.5–6.2%), Linalyl acetate (0.0–5.2%), Nonacosane (0.4–4.9%), 9-Geranyl-p-cymene (0.0–4.9%), Heptacosane (0.3–4.8%), cis-Chrysanthenyl acetate (0.0–4.8%), β-Farnesene (3.1–4.0%), Hexadecanoic acid (1.3–3.5%), Caryophyllene oxide (0.5–2.9%), β-Bisabolene (1.6–2.8%), Linalool (0.0–2.6%), Pentacosane (0.0–2.5%), Sesquisabinene (2.1%), Geranyl acetate (0.0–2.1%) | [106] |
| Stachys officinalis (L.) Trevis | Germacrene D (19.9%), β-Caryophyllene (14.1%), α-Humulene (7.5%), δ-Cadinene (4.0%), β-Bourbonene (3.8%), α-Selinene (3.4%), γ-Muurolene (3.2%), Oct-1-en-3-ol (2.9%), Caryophyllene oxide (2.5%), Hexadecanoic acid (2.4%), β-Selinene (2.1%), γ-Cadinene (2.0%), τ-Muurolol (2.0%) | [107] |
| Stachys pubescens Ten. | Germacrene (22.4%), δ-Cadinene (19.7%), 2,6-Octadien (11.5%), Linalool (9.7%), Limonene (6.3%), δ-Elemene (5.4%), β-Ocimene (2.8%), α-Terpinene (2.7%), 2,6-Octadienal (2.1%) | [108] |
| Teucrium sauvagei Le Houerou | β-Eudesmol (28.8%), τ-Cadinol (17.5%), α-Thujene (8.7%), γ-Cadinene (5.6%), Sabinene (4.8%), β-Selinene (4.2%), Limonene (2.8%), γ-Selinene (2.8%), α-Selinene (2.8%), δ-Cadinene (2.2%), Terpinen-4-ol (2.2%), p-Cymene (2.0%), | [109] |
| Teucrium yemense Deflers. | Caryophyllene oxide (4.3–20.1%), 7-epi-α-Selinene (1.3–20.1%), β-Caryophyllene (11.2–19.1%), α-Cadinol (2.0–9.5%), α-Pinene (2.3–6.6%), δ-Cadinene (0.4–6.5%), α-Humulene (4.0–6.4%), τ-Cadinol (2.0–5.7%), γ-Selinene (0.4–5.5%), τ-Muurolol (0.6–4.9%), Shyobunol (0.0–4.6%), Valencene (0.0–3.7%), Ledol (0.5–3.6%), cis-Sesquisabinene hydrate (0.9–3.4%), β-Pinene (1.1–3.1%), Germacrene D-4-ol (0.0–3.1%), γ-Cadinene (0.0–2.7%), β-Selinene (0.3–2.5%), Alloaromadendrene (trace–2.2%) | [77] |
| Thymbra capitata (L.) Cav. = Thymus capitatus (L.) Hoffmanns. and Link = Coridothymus capitatus (L.) Rchb.f. Solms | Carvacrol (35.6–75.0%), Thymol (0.1–29.3%), p-Cymene (5.0–21.0%), γ-Terpinene (4.0–12.3%), α-Terpinene (1.0–3.0%), β-Myrcene (0.8–3.0%), Linalool (0.5–2.9%), β-Caryophyllene (0.2–2.5%) | [51,110,111,112] |
| Thymbra spicata L. | Carvacrol (20.1–64.0%), γ-Terpinene (11.6–31.2%), p-Cymene (9.6–26.0%), α-Terpinene (1.2–10.1%), β-Myrcene (0.9–7.7%), Thujene (trace–5.2%), β-Caryophyllene (0.5–5.1%) | [51,113,114] |
| Thymus bovei Benth. | Geraniol (35.38%), α-Citral (20.37%), β-Citral (14.76%), Nerol (7.38%), 3-Octanol (4.38%) | [115] |
| Thymus daenensis Celak. | Carvacrol (31.46%), α-Terpineol (22.95%), Thymol (20.2%), Camphene (6.27%), 2,6-Octadien (2.22%), Borneol (2.17%), Cyclohexanone (2.1%) | [108] |
| Thymus kotschyanus Boiss. and Hohen. | Thymol (46.72%), Benzene (6.88%), Carvacrol (3.73%), γ-Terpinene (3.58%), β-Caryophyllene (3.39%), Linalool (2.88%), Phenol (2.61%), Borneol (2.51%), Isopropyl (2.07%) | [108] |
| Thymus mastichina (L.) L. | 1,8-Cineole (67.4%), Linalool (4.3%), β-Pinene (4.0%), α-Terpineol (3.5%), α-Pinene (3.0%), Sabinene (2.4%) | [116] |
| Thymus migricus Klokov et Des.-Shost. | Thymol (44.9%), Geraniol (10.8%), γ-Terpinene (10.3%), Citronellol (8.5%), p-Cymene (7.2%) | [117,118] |
| Thymus pulegioides L. | Thymol (26.0%), Carvacrol (21.0%), γ-Terpinene (8.8%), p-Cymene (7.8%), Octan-3-one (3.9%), Camphor (3.9%), β-Bisabolene (3.0%), Borneol (2.9%), Oct-1-en-3-ol (2.0%) | [119] |
| Thymus schimperi Ronniger | Carvacrol (13.91–39.07%), Thymol (11.53–34.66%), o-Cymene (18.72–27.06%), γ-Terpinene (4.13–13.73%), Linalool (3.34–3.59%), 3-Octanone (1.05–2.67%), α-Terpinene (1.67–2.37%) | [120] |
| Thymus serpyllum L. | Thymol (52.6%), p-Cymene (15.3%), β-Caryophyllene (6.8%), Sabinene hydrate (3.8%), γ-Terpinene (2.9%), Terpinen-4-ol (2.4%) | [68] |
| Thymus striatus Vahl. | Thymol (59.5%), γ-Terpinene (11.6%), p-Cymene (6.4%), Carvacrol methyl ether (5.9%), Carvacrol (4.9%), α-Terpinene (3.3%), β-Caryophyllene (2.3%) | [121] |
| Thymus vulgaris L. | Carvacrol (3.5–70.3%), Thymol (0.6–51.8%), Borneol (0.0–40.6%), p-Cymene (2.9–38.9%), o-Cymene (0.0–31.7%), α-Terpineol (0.0–19.9%), Linalool (0.0–16.0%), γ-Terpinene (0.3–12.65%), Camphene (0.0–12.3%), 1,8-Cineole (0.0–11.3%), α-Pinene (0.2–6.1%), β-Caryophyllene (0.0–3.5%), Neomenthol (0.0–2.8%), β-Cubebene (0.0–2.4%), Geraniol (0.0–2.32%), Menthone (0.0–2.2%) | [61,64,74,85,87,104,116,122,123,124,125,126] |
| Thymus zygis L. | Linalool (5.5–39.7%), Thymol (0.52–39.6%), p-Cymene (2.2–21.2%), Terpinen-4-ol (1.0–11.7%), β-Myrcene (3.0–8.6%), γ-Terpinene (7.6–7.9%), α-Terpinene (1.2–4.2%), β-Caryophyllene (1.6–3.6%), α-Pinene (0.9–3.6%), Limonene (1.7–2.6%), Carvacrol (0.08–2.4%), Terpinolene (0.2–2.0%) | [116,127] |
| Vitex agnus-castus L. | Eucalyptol (20.5%), 1,8-Cineole (1.5–19.61%), Bicyclogermacrene (0.0–16.2%), β-Farnesene (0.0–16.1%), Sabinene (0.0–14.57%), Sclarene (0.0–10.9%), α-Pinene (0.9–9.76%), Manool (0.0–8.2%), β-Caryophyllene (3.0–6.6%), β-Caryophyllene oxide (0.0–5.83%), Limonene (0.0–4.89%), Vulgarol B (0.0–4.7%), β-Pinene (0.4–4.4%), α-Terpinyl acetate (1.2–4.21%), β-Sitosterol (3.13%), p-Cymene (0.0–3.11%), Geranyl linalool (0.0–3.1%), β-Phellandrene (0.0–3.0%), Cembrene A (0.7–2.8%), Beyrene (0.0–2.6%), β-Myrcene (trace–2.12%), γ-Elemene (2.11%), s-Cadinol (2.01%) | [51,128,129] |
| Zataria multiflora Boiss. | Thymol (25.8–48.4%), Carvacrol (1.5–34.36%), Carvacrol methyl ether (0.0–28.32%), p-Cymene (2.27–13.2%), γ-Terpinene (0.92–10.6%), Linalool (0.9–6.52%), α-Terpinenyl acetate (5.4%), α-Terpineol (0.5–3.69%), α-Pinene (0.02–3.13%), β-Caryophyllene (2.24–3.12%), Carvacrol acetate (0.0–2.26%), Terpinen-4-ol (0.0–2.21%) | [117,130] |
| Ziziphora clinopodioides L. | Carvacrol (0.63–74.29%), Thymol (7.28–55.6%), γ-Terpinene (1.54–24.56%), p-Cymene (2.21–10.25%), α-Terpinene (0.39–2.77%) | [131,132] |
| Ziziphora tenuior L. | Pulegone (46.8%), p-Menth-3-en-8-ol (12.5%), Isomenthone (6.6%), 8-Hydroxymenthone (6.2%), Isomenthol (4.7%), Limonene (3.2%) | [133] |
| Source of the Essential Oil | Targeted Fungus | MICs (µg/mL; µl/mL) | Reference(s) |
|---|---|---|---|
| Aeollanthus suaveolens Mart. ex Spreng. = A. heliotropioides Oliv. | Candida albicans | 1200–5000 | [34] |
| Candida glabrata | 5000 | [34] | |
| Candida krusei | 2500 | [34] | |
| Candida parapsilosis | 2500 | [34] | |
| Candida tropicalis | 1200 | [34] | |
| Cryptococcus neoformans | 600–5000 | [34] | |
| Agastache rugosa (Fisch. and C.A.Mey.) Kuntze | Aspergillus flavus | 10,000 | [153] |
| Aspergillus niger | 5000 | [153] | |
| Blastoschizomyces capitatus | 5000 | [153] | |
| Candida albicans | 28–5000 | [153,154] | |
| Candida utilis | 5000 | [153] | |
| Candida tropicalis | 5000 | [153] | |
| Cryptococcus neoformans | 10,000 | [153] | |
| Trichoderma viride | 5000 | [153] | |
| Trichophyton erinacei | 780 | [153] | |
| Trichophyton mentagrophytes | 3120 | [153] | |
| Trichophyton rubrum | 1560 | [153] | |
| Trichophyton schoenleinii | 1560 | [153] | |
| Trichophyton soudanense | 1560 | [153] | |
| Trichophyton tonsurans | 10,000 | [153] | |
| Trichosporon mucoides | 5000 | [153] | |
| Ballota nigra subsp. foetida (Vis.) Hayek | Alternaria solani | 750 | [37] |
| Botrytis cinerea | 600 | [37] | |
| Fusarium coeruleum | 350 | [37] | |
| Fusarium culmorum | 300 | [37] | |
| Fusarium oxysporum | 300 | [37] | |
| Fusarium solani | 350 | [37] | |
| Fusarium sporotrichioides | 350 | [37] | |
| Fusarium tabacinum | 350 | [37] | |
| Fusarium verticillioides | 300 | [37] | |
| Clinopodium dalmaticum (Benth.) Bräuchler and Heubl = Micromeria dalmatica Benth. | Aspergillus niger | 0.4 | [38] |
| Aspergillus ochraceus | 0.4 | [38] | |
| Cladosporium cladosporioides | 0.4 | [38] | |
| Fusarium tricinctum | 0.4 | [38] | |
| Penicilium ochrochloron | 0.4 | [38] | |
| Phomopsis helianthi | 0.2 | [38] | |
| Trichoderma viride | 0.4 | [38] | |
| Clinopodium nepeta subsp. glandulosum (Req.) Govaerts = Calamintha glandulosa (Req.) Bentham = Calamintha officinalis Moench | Aspergillus niger | 1250 | [39] |
| Candida albicans | 2500 | [39] | |
| Clinopodium nepeta (L.) Kuntze = Calamintha nepeta (L.) Savi | Aspergillus flavus | 1.25–10 | [41] |
| Aspergillus fumigatus | 0.64–5 | [41] | |
| Aspergillus niger | 0.32–10 | [41] | |
| Candida albicans | 1.25–12,480 | [41,42] | |
| Candida guillermondii | 1.25–2.5 | [41] | |
| Candida krusei | 1.25–2.5 | [41] | |
| Candida parapsilosis | 1.25–2.5 | [41] | |
| Candida tropicalis | 1.25–2.5 | [41] | |
| Cryptococcus neoformans | 0.32–1.25 | [41] | |
| Epidermophyton floccosum | 0.64–2.5 | [41] | |
| Microsporum canis | 0.64–2.5 | [41] | |
| Microsporum gypseum | 1.25–5 | [41] | |
| Trichophyton mentagrophytes | 0.64–5 | [41] | |
| Trichophyton rubrum | 0.64–5 | [41] | |
| Clinopodium thymifolium (Scop.) Kuntze = Micromeria thymifolia (Scop.) Fritsch | Aspergillus niger | 2 | [38] |
| Aspergillus ochraceus | 2 | [38] | |
| Cladosporium cladosporioides | 2 | [38] | |
| Fusarium tricinctum | 2 | [38] | |
| Penicillium ochrochloron | 2 | [38] | |
| Phomopsis helianthi | 0.4 | [38] | |
| Trichoderma viride | 2 | [38] | |
| Clinopodium umbrosum (M.Bieb.) Kuntze = Calamintha umbrosa Benth. | Alternaria solani | 3000 | [43] |
| Fusarium oxysporum | 2000 | [43] | |
| Helminthosporium maydis | 1500 | [43] | |
| Dracocephalum heterophyllum Benth. | Alternaria solani | 625 | [155] |
| Candida albicans | 625–1000 | [44,155] | |
| Epidermophyton floccosum | 2500 | [155] | |
| Fusarium semitectum | 313 | [155] | |
| Hymenocrater longiflorus Benth. | Aspergillus niger | 480 | [45] |
| Candida albicans | 240 | [45] | |
| Hyptis ovalifolia Benth. | Microsporum canis | 15.6–1000 | [46,156] |
| Microsporum gypseum | 7.8–1000 | [46,156] | |
| Trichophyton mentagrophytes | 15.6–1000 | [46,156] | |
| Trichophyton rubrum | 7.8–1000 | [46,156] | |
| Hyssopus officinalis L. | Aspergillus niger | 52,200 | [47] |
| Aspergillus ochraceus | 26,100 | [47] | |
| Aspergillus versicolor | 10,440 | [47] | |
| Candida albicans | 128–1000 | [44,48] | |
| Candida glabrata | 512–1024 | [48] | |
| Candida krusei | 128–256 | [48] | |
| Candida parapsilosis | 256–512 | [48] | |
| Candida tropicalis | 512–1024 | [48] | |
| Cladosporium cladosporioides | 10,440 | [47] | |
| Cladosporium fulvum | 26,100 | [47] | |
| Penicillium funiculosum | 52,200 | [47] | |
| Penicillium ochrochloron | 26,100 | [47] | |
| Trichoderma viride | 10,440 | [47] | |
| Lavandula angustifolia Mill. | Candida albicans | 0.125–512 | [50,51,157] |
| Malassezia furfur | >4 | [49] | |
| Trichophyton rubrum | 1–512 | [49,51] | |
| Trichosporon beigelii | 2 | [49] | |
| Lavandula multifida L. | Aspergillus flavus | 0.64 | [52] |
| Aspergillus fumigatus | 0.32 | [52] | |
| Aspergillus niger | 0.32 | [52] | |
| Candida albicans | 0.32 | [52] | |
| Candida guilliermondii | 0.32 | [52] | |
| Candida krusei | 0.64 | [52] | |
| Candida parapsilosis | 0.32 | [52] | |
| Candida tropicalis | 0.32 | [52] | |
| Cryptococcus neoformans | 0.16 | [52] | |
| Epidermophyton floccosum | 0.16 | [52] | |
| Microsporum canis | 0.16 | [52] | |
| Microsporum gypseum | 0.16 | [52] | |
| Trichophyton mentagrophytes | 0.16 | [52] | |
| Trichophyton mentagrophytes var. interdigitale | 0.16 | [52] | |
| Trichophyton rubrum | 0.16 | [52] | |
| Trichophyton verrucosum | 0.16 | [52] | |
| Lavandula pedunculata (Miller) Cav. | Aspergillus flavus | 5–10 | [53] |
| Aspergillus fumigatus | 2.5–5 | [53] | |
| Aspergillus niger | 5 | [53] | |
| Candida albicans | 2.5 | [53] | |
| Candida guillermondii | 1.25 | [53] | |
| Candida krusei | 1.25–2.5 | [53] | |
| Candida parapsilosis | 2.5–5 | [53] | |
| Candida tropicalis | 1.25–2.5 | [53] | |
| Cryptococcus neoformans | 0.32–1.25 | [53] | |
| Epidermophyton floccosum | 0.32–0.64 | [53] | |
| Microsporum canis | 0.32–1.25 | [53] | |
| Microsporum gypseum | 0.64–2.5 | [53] | |
| Trichophyton mentagrophytes | 0.64–1.25 | [53] | |
| Trichophyton rubrum | 0.32–1.25 | [53] | |
| Lavandula stoechas L. | Aspergillus flavus | 1.25–10 | [54] |
| Aspergillus fumigatus | 0.64–1.25 | [54] | |
| Aspergillus niger | 0.32–1.25 | [54] | |
| Candida albicans | 0.64–512 | [51,54] | |
| Candida guillermondii | 1.25 | [54] | |
| Candida krusei | 2.5 | [54] | |
| Candida parapsilosis | 2.5 | [54] | |
| Candida tropicalis | 2.5 | [54] | |
| Cryptococcus neoformans | 0.64 | [54] | |
| Epidermophyton floccosum | 0.16–0.32 | [54] | |
| Microsporum canis | 0.16–0.64 | [54] | |
| Microsporum gypseum | 0.32–0.64 | [54] | |
| Trichophyton mentagrophytes | 0.32–0.64 | [54] | |
| Trichophyton mentagrophytes var. interdigitale | 0.16–0.64 | [54] | |
| Trichophyton rubrum | 0.16–256 | [51,54] | |
| Trichophyton verrucosum | 0.32 | [54] | |
| Lavandula viridis L’Her. | Aspergillus flavus | 5 | [55] |
| Aspergillus fumigatus | 2.5 | [55] | |
| Aspergillus niger | 2.5 | [55] | |
| Candida albicans | 1.25–2.5 | [55] | |
| Candida guillermondii | 0.64–1.25 | [55] | |
| Candida krusei | 1.25–2.5 | [55] | |
| Candida parapsilosis | 1.25 | [55] | |
| Candida tropicalis | 1.25–2.5 | [55] | |
| Cryptococcus neoformans | 0.64 | [55] | |
| Epidermophyton floccosum | 0.32 | [55] | |
| Microsporum canis | 0.32 | [55] | |
| Microsporum gypseum | 0.64 | [55] | |
| Trichophyton mentagrophytes | 0.32–0.64 | [55] | |
| Trichophyton mentagrophytes var. interdigitale | 0.32–0.64 | [55] | |
| Trichophyton rubrum | 0.32 | [55] | |
| Trichophyton verrucosum | 0.32 | [55] | |
| Lepechinia mutica (Benth.) Epling | Candida albicans | >9000 | [56] |
| Fusarium graminearum | >9000 | [56] | |
| Microsporum canis | 2200–4500 | [56] | |
| Pyricularia oryzae | >9000 | [56] | |
| Trichophyton rubrum | 2200–4500 | [56] | |
| Marrubium vulgare L. | Aspergillus niger | >1180 | [58] |
| Botrytis cinerea | >1100 | [58] | |
| Fusarium solani | >1190 | [58] | |
| Penicillium digitatum | >1120 | [58] | |
| Melissa officinalis L. | Aspergillus niger | 313 | [158] |
| Candida albicans | 30–313 | [59,158] | |
| Cryptococcus neoformans | 78 | [158] | |
| Epidermophyton floccosum | 30 | [59] | |
| Microsporum canis | 30 | [59] | |
| Penicillium verrucosum | 125 | [159] | |
| Trichophyton mentagrophytes var. mentagrophytes | 15 | [59] | |
| Trichophyton rubrum | 15 | [59] | |
| Trichophyton tonsurans | 15 | [59] | |
| Mentha cervina L. | Aspergillus flavus | 2.5–5 | [60] |
| Aspergillus fumigatus | 1.25–2.5 | [60] | |
| Aspergillus niger | 1.25–2.5 | [60] | |
| Candida albicans | 1.25–2.5 | [60] | |
| Candida guillermondii | 1.25–2.5 | [60] | |
| Candida krusei | 1.25–2.5 | [60] | |
| Candida parapsilosis | 1.25–2.5 | [60] | |
| Candida tropicalis | 1.25–2.5 | [60] | |
| Cryptococcus neoformans | 1.25 | [60] | |
| Epidermophyton floccosum | 0.64–1.25 | [60] | |
| Microsporum canis | 1.25 | [60] | |
| Microsporum gypseum | 1.25–2.5 | [60] | |
| Trichophyton mentagrophytes | 1.25–2.5 | [60] | |
| Trichophyton rubrum | 1.25 | [60] | |
| Mentha × piperita L. | Aspergillus flavus | 1450–5000 | [62,64] |
| Aspergillus niger | 625–10,000 | [64,158] | |
| Aspergillus parasiticus | 2500 | [64] | |
| Candida albicans | 225–1125 | [63,158,160] | |
| Candida glabrata | 225 | [62] | |
| Candida tropicalis | 225–230 | [62] | |
| Cryptococcus neoformans | 313 | [158] | |
| Fusarium oxysporum | 125 | [161] | |
| Penicillium chrysogenum | 1250 | [64] | |
| Penicillium minioluteum | 2050–2200 | [62] | |
| Penicillium oxalicum | 1300–2050 | [62] | |
| Penicillium verrucosum | 2500 | [90] | |
| Mentha pulegium L. | Aspergillus niger | 0.25–1.25 | [65,162] |
| Aspergillus flavus | 1.25 | [162] | |
| Aspergillus fumigatus | 1.25 | [162] | |
| Candida albicans | 0.94–3.75 | [65,66,162,163] | |
| Candida bracarensis | 3.75 | [163] | |
| Candida guillermondii | 1.25 | [162] | |
| Candida krusei | 0.94–1.25 | [162,163] | |
| Candida parapsilosis | 1.25 | [162] | |
| Candida tropicalis | 1.25 | [162] | |
| Cryptococcus neoformans | 0.64 | [162] | |
| Epidermophyton floccosum | 1.25 | [162] | |
| Microsporum canis | 1.25 | [162] | |
| Microsporum gypseum | 1.25–2.5 | [162] | |
| Saccharomyces cervisiae | <0.3–0.94 | [66,163] | |
| Trichophyton mentagrophytes | 1.25–2.5 | [162] | |
| Trichophyton mentagrophytes var. interdigitale | 2.5 | [162] | |
| Trichophyton rubrum | 1.25 | [162] | |
| Trichophyton verrucosum | 1.25 | [162] | |
| Mentha requienii Bentham | Alternaria spp. | >40 | [67] |
| Aspergillus fumigatus | >60 | [67] | |
| Candida albicans | 0.94–40 | [67,163] | |
| Candida bracarensis | 3.75 | [163] | |
| Candida krusei | 0.94 | [163] | |
| Fusarium spp. | >40 | [67] | |
| Penicillum spp. | >60 | [67] | |
| Rhodotorula spp. | 45 | [67] | |
| Saccharomyces cerevisiae | 0.94 | [163] | |
| Mentha spicata L. | Aspergillus flavus | 1.25 | [162] |
| Aspergillus fumigatus | 0.64 | [162] | |
| Aspergillus niger | 0.64–313 | [158,162] | |
| Candida albicans | 1.25–625 | [51,158,162] | |
| Candida guillermondii | 1.25 | [162] | |
| Candida krusei | 1.25 | [162] | |
| Candida parapsilosis | 1.25 | [162] | |
| Candida tropicalis | 1.25 | [162] | |
| Cryptococcus neoformans | 0.32–313 | [158,162] | |
| Epidermophyton floccosum | 0.64 | [162] | |
| Fusarium graminearum | 2.5 | [164] | |
| Fusarium moniliforme | 2.5 | [164] | |
| Malassezia furfur | >4 | [49] | |
| Microsporum canis | 0.64–2 | [68,162] | |
| Microsporum gypseum | 0.64–3 | [162] | |
| Penicillium corylophilum | 0.625 | [165] | |
| Penicillium expansum | 2.5 | [164] | |
| Trichophyton erinacei | 3 | [68] | |
| Trichophyton mentagrophytes | 0.64–3 | [68,162] | |
| Trichophyton mentagrophytes var. interdigitale | 0.64 | [162] | |
| Trichophyton rubrum | 0.25–512 | [49,51,162] | |
| Trichophyton terrestre | 3 | [68] | |
| Trichophyton verrucosum | 0.32 | [162] | |
| Trichosporon beigelii | 0.25 | [49] | |
| Mentha suaveolens Ehrh. | Candida albicans | 0.34–1250 | [69,71,166] |
| Candida glabrata | 0.69–2.77 | [69] | |
| Cryptococcus neoformans | 300 | [167] | |
| Microsporum canis | 1250 | [167] | |
| Microsporum gypseum | 1250 | [167] | |
| Trichophyton mentagrophytes | 600–1250 | [167] | |
| Trichophyton rubrum | 5000 | [167] | |
| Trichophyton violaceum | 600 | [167] | |
| Micromeria albanica (Griseb. ex K. Maly) Silic | Aspergillus niger | 0.2 | [38] |
| Aspergillus ochraceus | 0.2 | [38] | |
| Cladosporium cladosporioides | 0.2 | [38] | |
| Fusarium tricinctum | 0.4 | [38] | |
| Penicilium ochrochloron | 0.2 | [38] | |
| Phomopsis helianthi | 0.2 | [38] | |
| Trichoderma viride | 0.4 | [38] | |
| Moluccella spinosa L. | Aspergillus niger | 50 | [72] |
| Candida albicans | 100 | [72] | |
| Fusarium oxysporum | 100 | [72] | |
| Nepeta ciliaris Benth. = Nepeta leucophylla Benth. | Alternaria solani | 3000 | [43] |
| Candida albicans | 0.78 | [168] | |
| Fusarium oxysporum | 1000 | [43] | |
| Trichophyton rubrum | 0.19 | [168] | |
| Helminthosporium maydis | 1500 | [43] | |
| Nepeta clarkei Hook. f. | Alternaria solani | 3000 | [43] |
| Fusarium oxysporum | 2000 | [43] | |
| Helminthosporium maydis | 2000 | [43] | |
| Ocimum basilicum L. | Aspergillus flavus | 10,000 | [64] |
| Aspergillus fumigatus | >50 | [75] | |
| Aspergillus niger | >50–10,000 | [64,75,158] | |
| Aspergillus parasiticus | 5000 | [64] | |
| Candida albicans | 30–625 | [73,74,158] | |
| Candida guilliermondii | 3.125–6.25 | [76] | |
| Cryptococcus neoformans | 313–1250 | [158,169] | |
| Debaryomyces hansenii | 6.25 | [76] | |
| Epidermophyton floccosum | 15 | [74] | |
| Microsporum canis | 1–15.2 | [68,74] | |
| Microsporum gypseum | 3 | [68] | |
| Penicillium chrysogenum | 10,000 | [64] | |
| Penicillium italicum | >50 | [75] | |
| Rhizopus stolonifer | >50 | [75] | |
| Rhodotorula glutinis | 86 | [73] | |
| Trichophyton erinacei | 2.5 | [68] | |
| Trichophyton mentagrophytes | 2.5–8.3 | [68,74] | |
| Trichophyton terrestre | 3 | [68] | |
| Saccharomyces cerevisiae | 28 | [73] | |
| Schizosaccharomyces pombe | 86 | [73] | |
| Trichophyton rubrum | 8.3 | [74] | |
| Trichophyton tonsurans | 8 | [74] | |
| Yarrowia lypolytica | 57 | [73] | |
| Ocimum × africanum Lour. = Ocimum × citriodorum | Candida guilliermondii | 3.125 | [76] |
| Debaryomyces hansenii | 1.56 | [76] | |
| Ocimum campechianum Mill. = Ocimum micranthum Willd. | Candida albicans | 69 | [73] |
| Rhodotorula glutinis | 139 | [73] | |
| Saccharomyces cerevisiae | 69 | [73] | |
| Schizosaccharomyces pombe | 104 | [73] | |
| Yarrowia lypolytica | 69 | [73] | |
| Ocimum forskolei Benth. | Candida albicans | 35.3–8600 | [77,170] |
| Ocimum gratissimum L. | Aspergillus fumigatus | >1000 | [78] |
| Candida albicans | 350–1500 | [78,171] | |
| Candida krusei | 750 | [171] | |
| Candida parapsilosis | 380 | [171] | |
| Candida tropicalis | 1500 | [171] | |
| Cryptococcus neoformans | 250–300 | [78,79] | |
| Fusarium oxysporum f. sp. cubense | 62.5 | [80] | |
| Fusarium oxysporum f. sp. lycopersici | 31.25 | [80] | |
| Fusarium oxysporum f. sp. tracheiphilum | 62.5 | [80] | |
| Fusarium solani | 62.5 | [80] | |
| Macrophomina phaseolina | 62.5–125 | [80] | |
| Malassezia pachydermatis | 300 | [78] | |
| Microsporum canis | 200–500 | [78,172] | |
| Microsporum gypseum | 150–250 | [78,172] | |
| Rhizoctonia solani | 31.25 | [80] | |
| Scopulariopsis brevicaulis | 400 | [78] | |
| Trichophyton interdigitale | 250 | [78] | |
| Trichophyton mentagrophytes | 200–250 | [78,172] | |
| Trichophyton rubrum | 150–250 | [78,172] | |
| Ocimum tenuiflorum L. = Ocimum sanctum L. | Aspergillus flavus | 300 | [83] |
| Candida albicans | 0.1–300 | [81,82] | |
| Candida glabrata | 0.15–300 | [81,82] | |
| Candida krusei | 0.35–450 | [81,82] | |
| Candida parapsilosis | 0.25–500 | [81,82] | |
| Candida tropicalis | 0.1–300 | [81,82] | |
| Origanum compactum Benth. | Alternaria alternata | 300 | [84] |
| Bipolaris oryzae | 300 | [84] | |
| Fusarium equiseti | 300 | [84] | |
| Fusarium graminearum | 300 | [84] | |
| Fusarium verticillioides | 300 | [84] | |
| Origanum majorana L. | Aspergillus flavus | 450–650 | [62] |
| Aspergillus niger | 625 | [158] | |
| Botrytis cinerea | 5000 | [87] | |
| Candida albicans | 625 | [158] | |
| Cryptococcus neoformans | 313 | [158] | |
| Fusarium delphinoides | 1800–14,400 | [85] | |
| Fusarium incarnatum-equiseti | 450–3600 | [85] | |
| Fusarium napiforme | 3600–14,400 | [85] | |
| Fusarium oxysporum | 900–3600 | [85] | |
| Fusarium solani | 900–3600 | [85] | |
| Fusarium verticillioides | 14,400 | [85] | |
| Microsporum canis | 0.5 | [68] | |
| Microsporum gypseum | 2 | [68] | |
| Penicillium expansum | 10,000 | [87] | |
| Penicillium minioluteum | 400–500 | [62] | |
| Penicillium oxalicum | 350–400 | [62] | |
| Sporothrix brasiliensis | ≤2250–9000 | [86] | |
| Sporothrix schenckii | ≤2250–9000 | [86] | |
| Trichophyton erinacei | 1 | [68] | |
| Trichophyton mentagrophytes | 1.5 | [68] | |
| Trichophyton terrestre | 2 | [68] | |
| Origanum vulgare L. | Aspergillus flavus | 0.64–2500 | [64,89,91] |
| Aspergillus fumigatus | 0.32–0.64 | [89] | |
| Aspergillus niger | 0.32–623 | [62,89,91,158] | |
| Aspergillus ochraceus | 470 | [91] | |
| Aspergillus parasiticus | 2500 | [64] | |
| Candida albicans | 0.32–700 | [74,88,89,91,158] | |
| Candida glabrata | 350 | [88] | |
| Candida guillermondii | 0.64–1.25 | [89] | |
| Candida krusei | 0.64–700 | [88,89] | |
| Candida parapsilosis | 0.64–170 | [88,89] | |
| Candida tropicalis | 0.32–700 | [88,89] | |
| Cladosporium sp. | 0.05–0.3 | [173] | |
| Cryptococcus neoformans | 0.16–78 | [89,158] | |
| Epidermophyton floccosum | 0.32–2 | [74,89] | |
| Fusarium sp. | 0.1–0.5 | [173] | |
| Malassezia furfur | 1–780 | [49,174] | |
| Microsporum canis | 0.025–2 | [68,74,89] | |
| Microsporum gypseum | 0.025–1.25 | [68,89] | |
| Penicillium sp. | 0.1–0.5 | [173] | |
| Penicillium chrysogenum | 625 | [64] | |
| Penicillium corylophilum | 0.625 | [165] | |
| Penicillium funiculosum | 610 | [91] | |
| Penicillium ochrochloron | 710 | [91] | |
| Penicillium verrucosum | 1.1719 | [90,91] | |
| Trichophyton mentagrophytes | 0.32–1.25 | [74,89] | |
| Trichophyton rubrum | 0.16–1.25 | [49,74,89] | |
| Trichophyton tonsurans | 1 | [74] | |
| Trichosporon beigelii | 0.25 | [49] | |
| Trichophyton erinacei | 0.5 | [68] | |
| Trichophyton mentagrophytes | 0.5 | [68] | |
| Trichophyton terrestre | 0.25 | [68] | |
| Pogostemon cablin (Blanco) Benth. | Aspergillus flavus | >1500 | [92] |
| Aspergillus niger | 156 | [158] | |
| Aspergillus oryzae | >1500 | [92] | |
| Candida albicans | 32–625 | [158,175] | |
| Candida krusei | 64–257 | [175] | |
| Candida tropicalis | 32–257 | [175] | |
| Cryptococcus neoformans | 20 | [158] | |
| Pogostemon heyneanus Benth. | Candida albicans | 6000 | [176] |
| Candida glabrata | 6000 | [176] | |
| Candida tropicalis | 10,000 | [176] | |
| Premna microphylla Turcz. | Aspergillus niger | >500 | [94] |
| Candida albicans | >500 | [94] | |
| Fusarium oxysporum | >500 | [94] | |
| Rosmarinus officinalis L. | Aspergillus flavus | 330 | [91] |
| Aspergillus ochraceus | 590 | [91] | |
| Aspergillus niger | 380–10,000 | [91,98,158] | |
| Botrytis cinerea | 2500 | [87] | |
| Candida albicans | 30.2–1000 | [51,91,96,98,158] | |
| Cryptococcus neoformans | 313 | [158] | |
| Epidermophyton floccosum | 30 | [96] | |
| Microsporum canis | 2.5–30.2 | [68,96] | |
| Microsporum gypseum | 2.5 | [68] | |
| Penicillium expansum | 5000 | [87] | |
| Penicillium ochrochloron | 470 | [91] | |
| Penicillium funiculosum | 570 | [91] | |
| Trichophyton erinacei | 1.5 | [68] | |
| Trichophyton mentagrophytes | 5–15.3 | [68,96] | |
| Trichophyton rubrum | 15–256 | [51,96] | |
| Trichophyton terrestre | 5 | [68] | |
| Trichophyton tonsurans | 15.2 | [96] | |
| Salvia fruticosa Miller | Candida albicans | 512 | [51] |
| Fusarium oxysporum f. sp. dianthi | >2000 | [99] | |
| Fusarium proliferatum | >2000 | [99] | |
| Fusarium solani f. sp. cucurbitae | >2000 | [99] | |
| Malassezia furfur | >4 | [99] | |
| Rhizoctonia solani | >2000 | [99] | |
| Sclerotinia sclerotiorum | >2000 | [99] | |
| Trichophyton rubrum | 2–256 | [49,99] | |
| Trichosporon beigelii | 4 | [49] | |
| Salvia mirzayanii Rech. f. and Esfand | Candida albicans | 0.5–2 | [100] |
| Candida krusei | 1 | [100] | |
| Candida dubliniensis | 0.06–0.5 | [100] | |
| Candida glabrata | 0.06–1 | [100] | |
| Candida parapsilosis | 0.25–1 | [100] | |
| Candida tropicalis | 0.25–2 | [100] | |
| Trichosporon sp. | 1 | [100] | |
| Salvia officinalis L. | Aspergillus flavus | 5–10 | [101] |
| Aspergillus fumigatus | 2.5–5 | [101] | |
| Aspergillus niger | 5–1250 | [101,158] | |
| Candida albicans | 2.5–2780 | [96,101,158,177] | |
| Candida guillermondii | 1.25–2.5 | [101] | |
| Candida krusei | 2.5–5 | [101] | |
| Candida parapsilosis | 5 | [101] | |
| Candida tropicalis | 5 | [101] | |
| Cryptococcus neoformans | 0.64–625 | [101,158] | |
| Epidermophyton floccosum | 0.64–100 | [96,101] | |
| Microsporum canis | 1.25–100.2 | [96,101] | |
| Microsporum gypseum | 1.25–2.5 | [101] | |
| Trichophyton mentagrophytes | 1.25–60 | [96,101] | |
| Trichophyton mentagrophytes var. interdigitale | 1.25 | [101] | |
| Trichophyton rubrum | 0.64–60 | [96,101] | |
| Trichophyton tonsurans | 60 | [96] | |
| Trichophyton verrucosum | 1.25–2.5 | [101] | |
| Salvia sclarea L. | Aspergillus niger | 1250 | [158] |
| Candida albicans | 1250 | [158] | |
| Cryptococcus neoformans | 313 | [158] | |
| Fusarium delphinoides | 1800–3600 | [85] | |
| Fusarium incarnatum-equiseti | 1800–3600 | [85] | |
| Fusarium napiforme | 1800–3600 | [85] | |
| Fusarium oxysporum | 1800–3600 | [85] | |
| Fusarium solani | 3600–7200 | [85] | |
| Fusarium verticillioides | 1800 | [85] | |
| Satureja hortensis L. | Alternaria alternata | 62.5 | [103] |
| Aspergillus flavus | 31.25–500 | [103,104,117] | |
| Aspergillus niger | 471 | [117] | |
| Aspergillus ochraceus | 423 | [117] | |
| Aspergillus parasiticus | 373 | [117] | |
| Aspergillus terreus | 389 | [117] | |
| Aspergillus variecolor | 125 | [103] | |
| Candida albicans | 200–400 | [103,178] | |
| Fusarium culmorum | 125 | [103] | |
| Fusarium oxysporum | 250 | [103] | |
| Microsporum canis | 62.5 | [103] | |
| Moniliania fructicola | 31.25 | [103] | |
| Penicillium spp. | 125 | [103] | |
| Rhizoctonia solani | 125 | [103] | |
| Rhizopus spp. | 250 | [103] | |
| Sclerotinia minor | 250 | [103] | |
| Sclerotinia sclerotiorum | 125 | [103] | |
| Trichophyton mentagrophytes | 62.5 | [103] | |
| Trichophyton rubrum | 31.25 | [103] | |
| Satureja montana L. | Microsporum canis | 0.5 | [68] |
| Microsporum gypseum | 2 | [68] | |
| Trichophyton erinacei | 2 | [68] | |
| Trichophyton mentagrophytes | 2 | [68] | |
| Trichophyton terrestre | 3 | [68] | |
| Satureja thymbra L. | Aspergillus flavus | 25 | [105] |
| Aspergillus fumigatus | 1.25–25 | [105,179] | |
| Aspergillus niger | 2.5–25 | [105,179] | |
| Aspergillus ochraceus | 2.5–25 | [105,179] | |
| Aspergillus versicolor | 1.25 | [179] | |
| Candida albicans | 25–128 | [51,105] | |
| Penicillium funiculosum | 2.5–25 | [105,179] | |
| Penicillium ochrochloron | 1–1.25 | [105,179] | |
| Trichoderma viride | 1.25–25 | [105,179] | |
| Trichophyton rubrum | 128 | [51] | |
| Stachys cretica L. | Candida albicans | 625 | [106] |
| Stachys officinalis (L.) Trevis | Aspergillus niger | 2500 | [107] |
| Candida albicans | 5000 | [107] | |
| Stachys pubescens Ten. | Alternaria alternata | 1 | [108] |
| Aspergillus flavus | 0–5 | [108] | |
| Fusarium oxysporum | 1 | [108] | |
| Teucrium sauvagei Le Houerou | Aspergillus fumigatus | >1000 | [109] |
| Candida albicans | >1000 | [109] | |
| Cryptococcus neoformans | >1000 | [109] | |
| Epidermophyton floccosum | 850 | [109] | |
| Microsporum canis | 800 | [109] | |
| Microsporum gypseum | 900 | [109] | |
| Scopulariopsis brevicaulis | >1000 | [109] | |
| Scytalidium dimidiatum | >1000 | [109] | |
| Trichophyton mentagrophytes var. interdigitale | 950 | [109] | |
| Trichophyton mentagrophytes var. mentagrophytes | 900 | [109] | |
| Trichophyton rubrum | 800 | [109] | |
| Trichophyton soudanense | 800 | [109] | |
| Teucrium yemense Deflers. | Aspergillus niger | 313 | [77] |
| Botrytis cinerea | 313 | [77] | |
| Candida albicans | 1250 | [77] | |
| Thymbra capitata (L.) Cav. = Thymus capitatus (L.) Hoffmanns. and Link = Coridothymus capitatus (L.) Rchb.f. Solms | Aspergillus flavus | 0.32 | [111] |
| Aspergillus fumigatus | 0.16–0.32 | [111] | |
| Aspergillus niger | 0.1–0.16 | [111,180] | |
| Aspergillus oryzae | 0.2 | [180] | |
| Candida albicans | 0.16–128 | [51,110,111,112] | |
| Candida glabrata | 0.32 | [111,112] | |
| Candida guilliermondii | 0.16–0.32 | [111,112] | |
| Candida krusei | 0.32 | [111] | |
| Candida parapsilosis | 0.32 | [111,112] | |
| Candida tropicalis | 0.32 | [111,112] | |
| Epidermophyton floccosum | 0.08 | [111] | |
| Fusarium solani | 0.2 | [180] | |
| Microsporum canis | 0.08 | [111] | |
| Microsporum gypseum | 0.08 | [111] | |
| Penicillium digitatum | 0.5 | [180] | |
| Trichophyton mentagrophytes | 0.08 | [111] | |
| Trichophyton rubrum | 0.16–64 | [51,111] | |
| Thymbra spicata L. | Aspergillus fumigatus | 0.3 | [179] |
| Aspergillus niger | 0.6 | [179] | |
| Aspergillus versicolor | 0.3 | [179] | |
| Aspergillus ochraceus | 0.6 | [179] | |
| Candida albicans | 1.12–3750 | [51,113,114] | |
| Candida krusei | 1.12 | [114] | |
| Candida parapsilosis | 0.6–1.12 | [114] | |
| Penicillium funiculosum | 0.3 | [179] | |
| Penicillium ochrochloron | 0.3 | [179] | |
| Trichoderma viride | 0.3 | [179] | |
| Trichophyton rubrum | 64 | [51] | |
| Thymus bovei Benth. | Candida albicans | 250 | [115] |
| Thymus daenensis Celak. | Alternaria alternata | >8 | [108] |
| Aspergillus flavus | 1 | [108] | |
| Fusarium oxysporum | 4 | [108] | |
| Thymus kotschyanus Boiss. and Hohen. | Alternaria alternata | 1 | [108] |
| Aspergillus flavus | 0.5 | [108] | |
| Fusarium oxysporum | 0–5 | [108] | |
| Thymus mastichina (L.) L. | Candida albicans | 1.25–2.5 | [116] |
| Candida glabrata | 1.25–1.5 | [116] | |
| Candida guilliermondii | 1.25 | [116] | |
| Candida krusei | 1.25–2.5 | [116] | |
| Candida parapsilosis | 2.5–5 | [116] | |
| Candida tropicalis | 2.5–10 | [116] | |
| Thymus migricus Klokov et Des.-Shost. | Aspergillus flavus | 452 | [117] |
| Aspergillus niger | 460 | [117] | |
| Aspergillus ochraceus | 430 | [117] | |
| Aspergillus parasiticus | 581 | [117] | |
| Aspergillus terreus | 447 | [117] | |
| Thymus pulegioides L. | Aspergillus flavus | 0.32 | [119] |
| Aspergillus fumigatus | 0.16 | [119] | |
| Aspergillus niger | 0.32 | [119] | |
| Candida albicans | 0.32–0.64 | [119] | |
| Candida glabrata | 0.32–0.64 | [119] | |
| Candida guilliermondii | 0.32 | [119] | |
| Candida krusei | 0.32–0.64 | [119] | |
| Candida parapsilosis | 0.64 | [119] | |
| Candida tropicalis | 0.32–0.64 | [119] | |
| Epidermophyton floccosum | 0.16 | [119] | |
| Microsporum canis | 0.16 | [119] | |
| Microsporum gypseum | 0.16 | [119] | |
| Trichophyton mentagrophytes | 0.16 | [119] | |
| Trichophyton rubrum | 0.32 | [119] | |
| Thymus schimperi Ronninger | Aspergillus minutus | 0.512–2 | [120] |
| Aspergillus niger | 0.16 | [181] | |
| Aspergillus tubingensis | 1–4 | [120] | |
| Beauveria bassiana | 0.128–1 | [120] | |
| Candida albicans | 0.16 | [181] | |
| Microsporum spp. | 0.08 | [181] | |
| Microsporum gypseum | 0.128–1 | [120] | |
| Penicillium chrysogenum | 0.512–2 | [120] | |
| Rhodotorula spp. | 0.08 | [181] | |
| Tricophyton spp. | 0.08–0.31 | [181] | |
| Verticillium sp. | 0.512–2 | [120] | |
| Thymus serpyllum L. | Aspergillus carbonarius | 1.25 | [182] |
| Aspergillus ochraceus | 0.625 | [182] | |
| Aspergillus niger | 2.5 | [182] | |
| Microsporum canis | 0.025 | [68] | |
| Microsporum gypseum | 0.25 | [68] | |
| Trichophyton erinacei | 0.1 | [68] | |
| Trichophyton mentagrophytes | 0.2 | [68] | |
| Trichophyton terrestre | 0.1 | [68] | |
| Thymus striatus Vahl. | Alternaria alternata | 1 | [121] |
| Aspergillus flavus | 1.5 | [121] | |
| Aspergillus niger | 1 | [121] | |
| Aspergillus ochraceus | 1 | [121] | |
| Aspergillus terreus | 1 | [121] | |
| Aspergillus versicolor | 1 | [121] | |
| Cladosporium cladosporioides | 0.5 | [121] | |
| Epidermophyton floccosum | 1 | [121] | |
| Microsporum canis | 1.5 | [121] | |
| Penicillium funiculosum | 2 | [121] | |
| Penicillium ochrochloron | 2 | [121] | |
| Phomopsis helianthi | 0.5 | [121] | |
| Trichoderma viride | 2 | [121] | |
| Trichophyton mentagrophytes | 1 | [121] | |
| Thymus vulgaris L. | Absidia spp. | 7 ± 4 | [122] |
| Alternaria spp. | 9.4 ± 4.5 | [122] | |
| Alternaria alternata | 4.7–500 | [122,183] | |
| Aspergillus spp. | 3.2 | [122] | |
| Aspergillus flavus | 9.35–1500 | [64,104,122,125,184] | |
| Aspergillus fumigatus | 144–1000 | [124,184] | |
| Aspergillus niger | 9.35–1250 | [64,122,158,184] | |
| Aspergillus ochraceus | 2.5–750 | [164,184] | |
| Aspergillus parasiticus | 1250 | [64] | |
| Aspergillus sulphureus | 10.88 ± 3.1 | [122] | |
| Aspergillus versicolor | 9.6 ± 9.25 | [122] | |
| Botrytis cinerea | 312 | [87] | |
| Candida albicans | 0.16–313 | [73,74,116,158] | |
| Candida glabrata | 0.16–0.32 | [116] | |
| Candida krusei | 0.08–0.16 | [116] | |
| Candida guillermondii | 0.16 | [116] | |
| Candida parapsilosis | 0.16–0.32 | [116] | |
| Candida tropicalis | 0.16–0.32 | [116] | |
| Chaetomium globosum | 1.6 | [122] | |
| Cladosporium spp. | 12.8 | [122] | |
| Cladosporium sphaerospermum | 19.6 | [122] | |
| Cryptococcus neoformans | 78 | [158] | |
| Epidermophyton floccosum | 4 | [74] | |
| Fusarium spp. | 62.5 | [185] | |
| Fusarium delphinoides | 900–1800 | [85] | |
| Fusarium incarnatum-equiseti | 450–3600 | [85] | |
| Fusarium napiforme | 900 | [85] | |
| Fusarium oxysporum | 5–900 | [85,126] | |
| Fusarium solani | 1800–3600 | [85] | |
| Fusarium verticillioides | 900 | [85] | |
| Malassezia furfur | 920 | [174] | |
| Microsporum canis | 2.2 | [74] | |
| Mortierella spp. | 250 | [185] | |
| Mucor spp. | 50.2 ± 8.4 | [122] | |
| Penicilium spp. | 18.95–500 | [122,185] | |
| Penicilium brevicompactum | 19.6 | [122] | |
| Penicillium chrysogenum | 312.5–1750 | [64,184] | |
| Penicilium chrysogenum | 19.6 | [122] | |
| Penicillium citrinum | 1250 | [184] | |
| Penicillium expansum | 625 | [87] | |
| Penicillium griseofulvum | 19.6 | [122] | |
| Rhizopus spp. | 12.6 | [122] | |
| Rhodotorula glutinis | 72 | [73] | |
| Rhizopus oryzae | 256–512 | [123] | |
| Saccharomyces cerevisiae | 72 | [73] | |
| Schizosaccharomyces pombe | 36 | [73] | |
| Stachybotrys chartarum | 6.2 | [122] | |
| Trichoderma spp. | 16.8 | [122] | |
| Trichophyton mentagrophytes | 2.2 | [74] | |
| Trichophyton rubrum | 2–72 | [74,124] | |
| Trichophyton tonsurans | 2.2 | [74] | |
| Ulocladium spp. | 5.45 ± 1.5 | [122] | |
| Yarrowia lypolytica | 36 | [73] | |
| Thymus zygis L. | Candida albicans | 0.16–0.32 | [116] |
| Candida glabrata | 0.32 | [116] | |
| Candida krusei | 0.16–0.32 | [116] | |
| Candida guillermondii | 0.16 | [116] | |
| Candida parapsilosis | 0.32 | [116] | |
| Candida tropicalis | 0.16–0.32 | [116] | |
| Penicillium corylophilum | 0.3125–0.625 | [165] | |
| Vitex agnus-castus L. | Candida albicans | 0.53–512 | [51,129] |
| Candida dubliniensis | 0.27 | [129] | |
| Candida famata | 2.13 | [129] | |
| Candida glabrata | 0.27 | [129] | |
| Candida krusei | 0.27 | [129] | |
| Candida lusitaniae | 2.13 | [129] | |
| Candida parapsilosis | 1.06 | [129] | |
| Candida tropicalis | 0.13 | [129] | |
| Epidermophyton floccosum | 0.64–2.5 | [128] | |
| Microsporum canis | 0.64–5 | [128] | |
| Microsporum gypseum | 1.25–10 | [128] | |
| Trichophyton mentagrophytes | 1.25–10 | [128] | |
| Trichophyton rubrum | 0.64–512 | [51,128] | |
| Zataria multiflora Boiss. | Aspergillus flavus | 358 | [117] |
| Aspergillus niger | 358 | [117] | |
| Aspergillus ochraceus | 341 | [117] | |
| Aspergillus parasiticus | 367 | [117] | |
| Aspergillus terreus | 447 | [117] | |
| Microsporum canis | 0.125–0.25 | [130] | |
| Microsporum gypseum | 0.03–0.06 | [130] | |
| Trichophyton mentagrophytes | 0.03 | [130] | |
| Trichophyton rubrum | 0.03–0.06 | [130] | |
| Trichophyton schoenleinii | 0.125–0.6 | [130] | |
| Ziziphora clinopodioides Lam. | Aspergillus flavus | 48.82 | [184,186] |
| Aspergillus fumigatus | 1750 | [184] | |
| Aspergillus niger | 3000 | [184] | |
| Aspergillus ochraceus | 1500 | [184] | |
| Aspergillus parasiticus | 48.82 | [186] | |
| Penicillium chrysogenum | 3000 | [184] | |
| Penicillium citrinum | 1750 | [184] | |
| Ziziphora tenuior L. | Aspergillus flavus | 1.25 | [133] |
| Aspergillus fumigatus | 0.64 | [133] | |
| Aspergillus niger | 0.64 | [133] | |
| Candida albicans | 1.25 | [133] | |
| Candida guillermondii | 1.25 | [133] | |
| Candida krusei | 1.25 | [133] | |
| Candida parapsilosis | 1.25 | [133] | |
| Candida tropicalis | 1.25 | [133] | |
| Cryptococcus neoformans | 0.16 | [133] | |
| Epidermophyton floccosum | 0.64 | [133] | |
| Microsporum canis | 0.64–1.25 | [133] | |
| Microsporum gypseum | 1.25 | [133] | |
| Trichophyton mentagrophytes | 1.25 | [133] | |
| Trichophyton mentagrophytes var. interdigitale | 1.254 | [133] | |
| Trichophyton rubrum | 0.64 | [133] | |
| Trichophyton verrucosum | 0.64 | [133] |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karpiński, T.M. Essential Oils of Lamiaceae Family Plants as Antifungals. Biomolecules 2020, 10, 103. https://doi.org/10.3390/biom10010103
Karpiński TM. Essential Oils of Lamiaceae Family Plants as Antifungals. Biomolecules. 2020; 10(1):103. https://doi.org/10.3390/biom10010103
Chicago/Turabian StyleKarpiński, Tomasz M. 2020. "Essential Oils of Lamiaceae Family Plants as Antifungals" Biomolecules 10, no. 1: 103. https://doi.org/10.3390/biom10010103
APA StyleKarpiński, T. M. (2020). Essential Oils of Lamiaceae Family Plants as Antifungals. Biomolecules, 10(1), 103. https://doi.org/10.3390/biom10010103





