Urinary Metabolomics Validates Metabolic Differentiation Between Renal Cell Carcinoma Stages and Reveals a Unique Metabolic Profile for Oncocytomas
Abstract
1. Introduction
2. Results
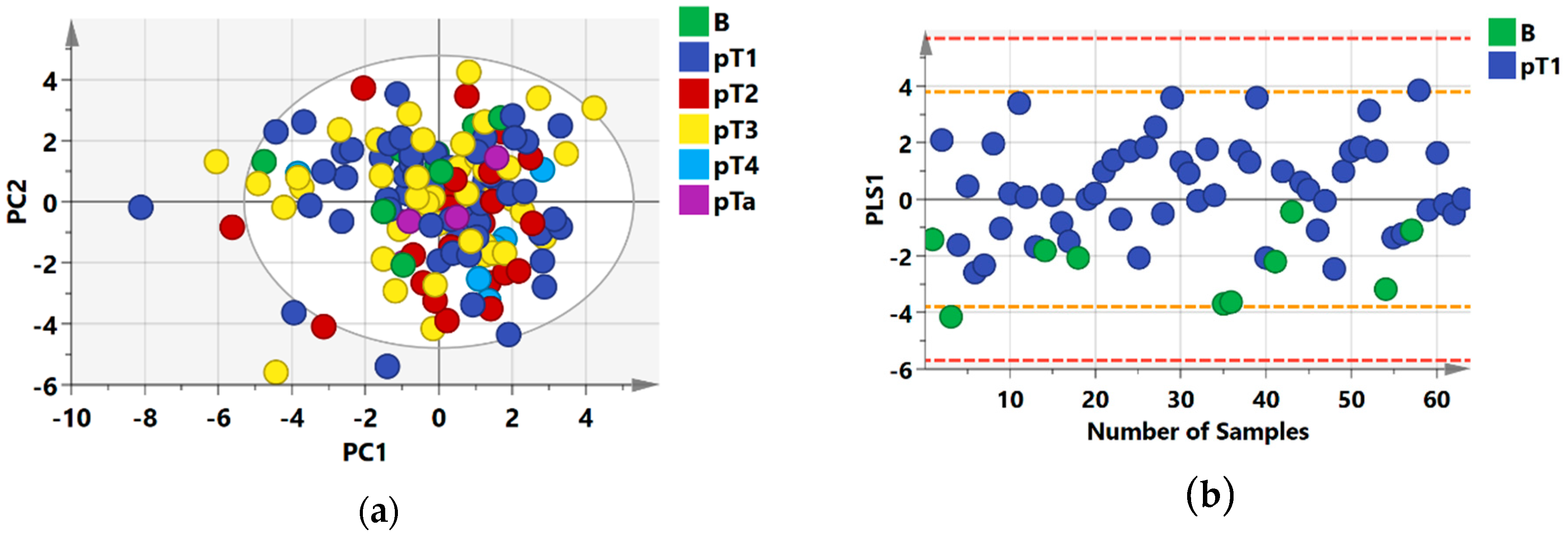
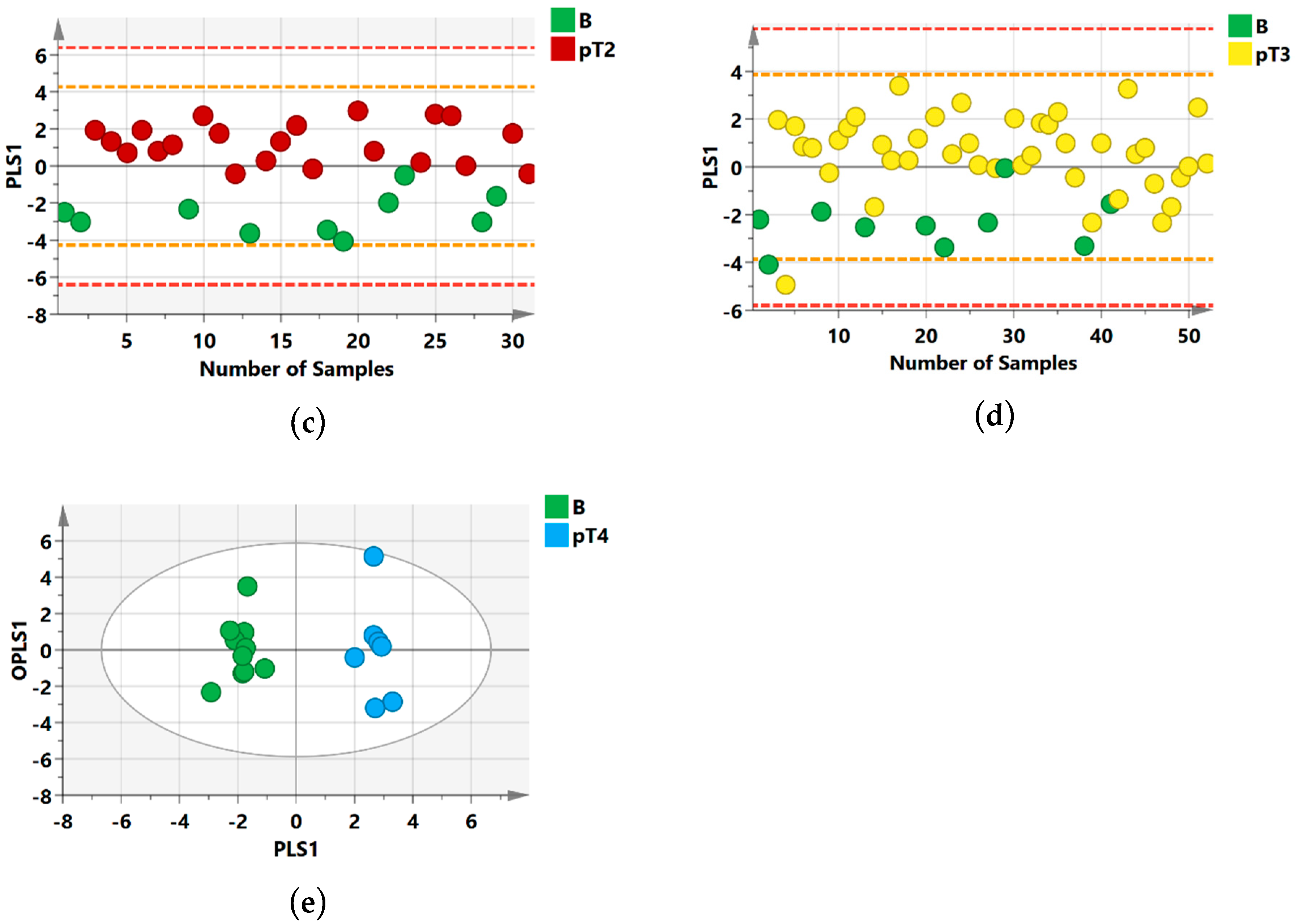
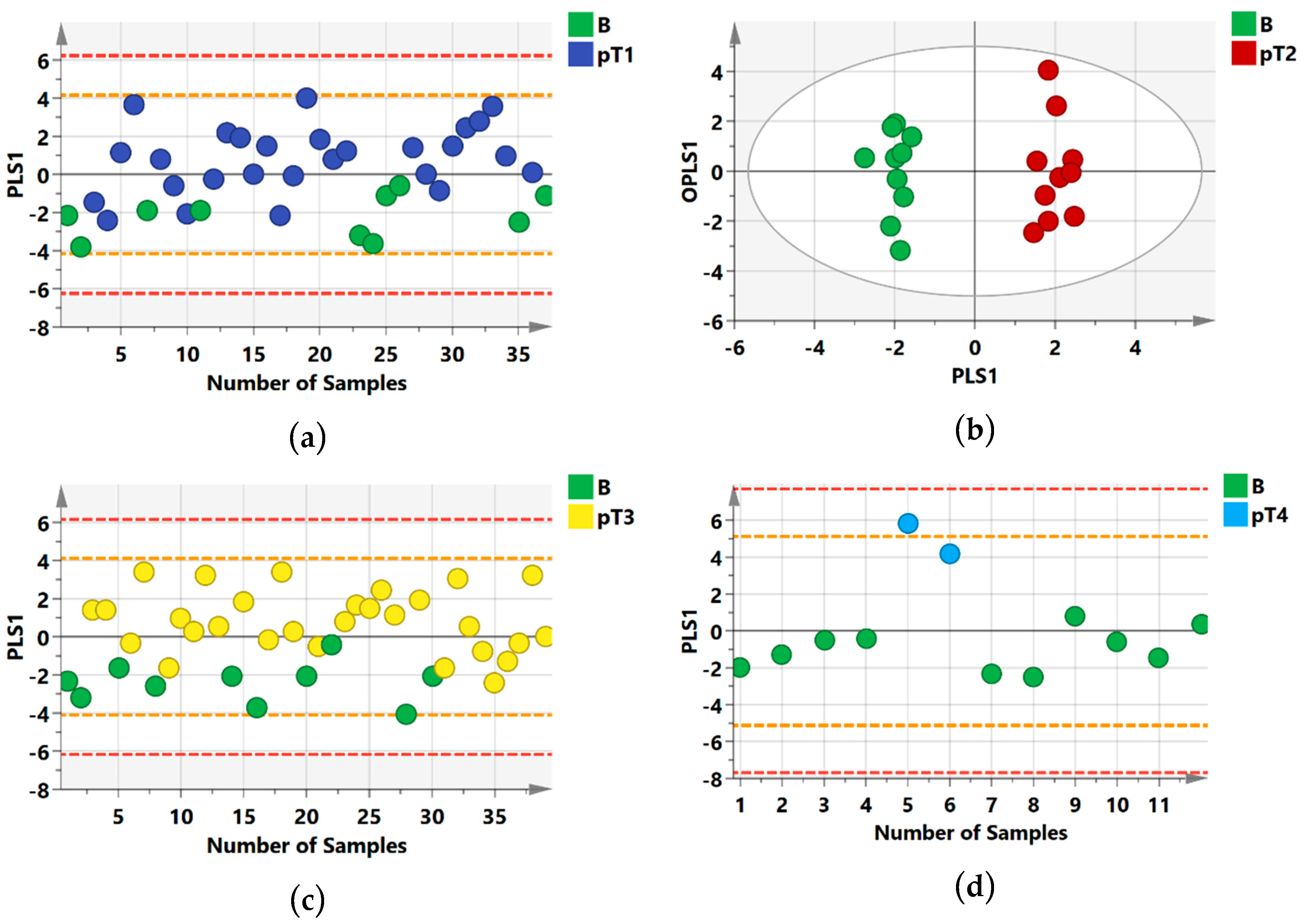
2.1. 1H NMR Models Separate Benign Lesions from All RCC Stages
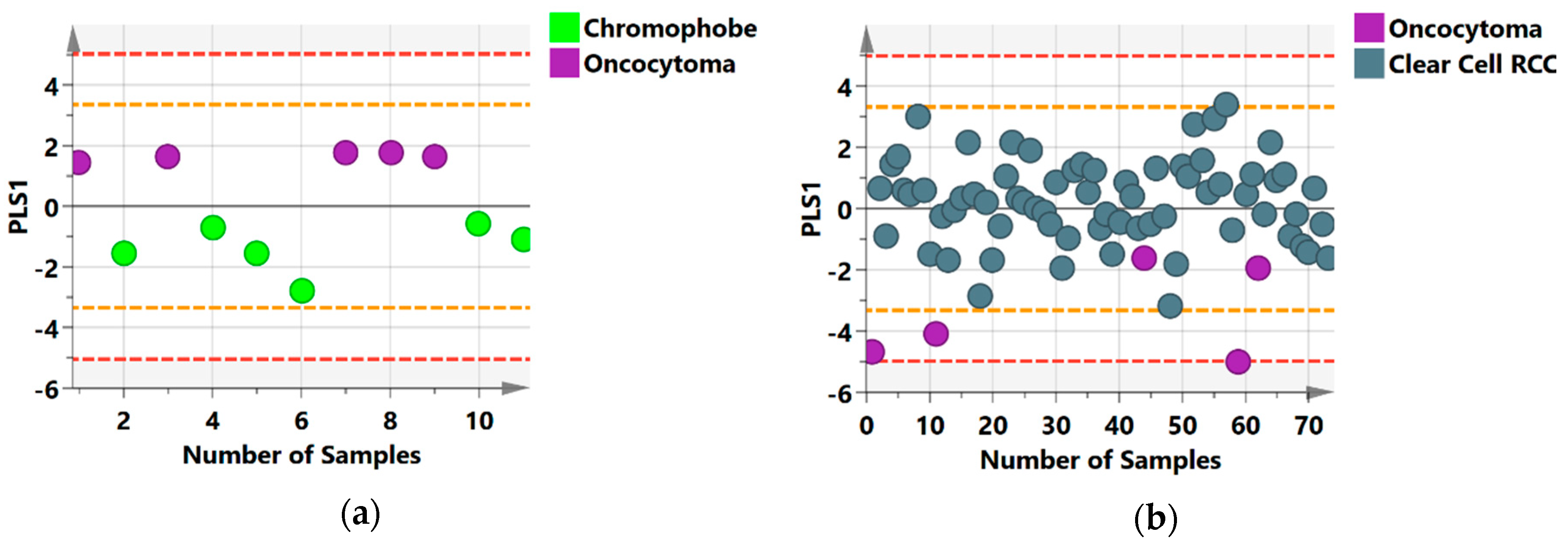
2.2. Group Separation between RCC Histology Subtypes
2.3. Confirmed Differential Metabolites; Potential RCC Biomarkers
2.4. Metabolic Distinction between Histological Subtypes
3. Discussion
4. Materials and Methods
4.1. Patient Enrollment and Sample Collection
4.2. 1H NMR Spectral Collection
4.3. Multivariate Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Model type | R2 | Q2 | CV ANOVA (p-Value) | Q2 Intercept |
|---|---|---|---|---|
| All Histology Subtypes | ||||
| pT1 vs pT2 | 0.30 | 0.19 | 6.7 × 10−4 | −0.20 |
| pT2 vs pT3 | 0.41 | 0.26 | 1.1 × 10−4 | −0.23 |
| pT1 vs pT4 | 0.33 | 0.12 | 2.5 × 10−2 | −0.23 |
| pT2 vs pT4 | 0.74 | 0.42 | 1.0 × 10−2 | −0.57 |
| pT3 vs pT4 | 0.37 | 0.16 | 1.8 × 10−2 | −0.24 |
| ccRCC Histology Only | ||||
| pT1 vs pT2 | 0.52 | 0.29 | 2.9 × 10−3 | −0.31 |
| pT2 vs pT3 | 0.41 | 0.16 | 4.8 × 10−2 | −0.30 |

References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.L.; Chang, A.; Perazella, M.A.; Okusa, M.D.; Jaimes, E.A.; Weiss, R.H. The Nephrologist’s Tumor: Basic Biology and Management of Renal Cell Carcinoma. J. Am. Soc. Nephrol. 2016, 27, 2227. [Google Scholar] [CrossRef] [PubMed]
- Wobker, S.E.; Williamson, S.R. Modern Pathologic Diagnosis of Renal Oncocytoma. J. Kidney Cancer VHL 2017, 4, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Jinzaki, M.; Silverman, S.G.; Akita, H.; Mikami, S.; Oya, M. Diagnosis of Renal Angiomyolipomas: Classic, Fat-Poor, and Epithelioid Types. Semin Ultrasound CT MR 2017, 38, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Pollard, P.J.; Briere, J.J.; Alam, N.A.; Barwell, J.; Barclay, E.; Wortham, N.C.; Hunt, T.; Mitchell, M.; Olpin, S.; Moat, S.J.; et al. Accumulation of Krebs Cycle Intermediates and Over-Expression of HIF1alpha in Tumours Which Result from Germline FH and SDH Mutations. Hum. Mol. Genet. 2005, 14, 2231–2239. [Google Scholar] [CrossRef] [PubMed]
- Ooi, A.; Wong, J.C.; Petillo, D.; Roossien, D.; Perrier-Trudova, V.; Whitten, D.; Min, B.W.; Tan, M.H.; Zhang, Z.; Yang, X.J.; et al. An Antioxidant Response Phenotype Shared between Hereditary and Sporadic Type 2 Papillary Renal Cell Carcinoma. Cancer Cell 2011, 20, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Laba, P.; Wang, J.; Zhang, J. Low Level of Isocitrate Dehydrogenase 1 Predicts Unfavorable Postoperative Outcomes in Patients with Clear Cell Renal Cell Carcinoma. BMC Cancer 2018, 18, 852. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Aronov, P.; Zakharkin, S.O.; Anderson, D.; Perroud, B.; Thompson, I.M.; Weiss, R.H. Urine Metabolomics Analysis for Kidney Cancer Detection and Biomarker Discovery. Mol. Cell Proteom. 2009, 8, 558–570. [Google Scholar] [CrossRef]
- Ganti, S.; Weiss, R.H. Urine Metabolomics for Kidney Cancer Detection and Biomarker Discovery. Urol Oncol 2011, 29, 551–557. [Google Scholar] [CrossRef]
- Zheng, H.; Ji, J.; Zhao, L.; Chen, M.; Shi, A.; Pan, L.; Huang, Y.; Zhang, H.; Dong, B.; Gao, H. Prediction and Diagnosis of Renal Cell Carcinoma Using Nuclear Magnetic Resonance-Based Serum Metabolomics and Self-Organizing Maps. Oncotarget 2016, 7, 59189–59198. [Google Scholar] [CrossRef]
- Van Oostenbrugge, T.J.; Fütterer, J.J.; Mulders, P.F.A. Diagnostic Imaging for Solid Renal Tumors: A Pictorial Review. Kidney Cancer 2018, 2, 79–93. [Google Scholar] [CrossRef] [PubMed]
- Falegan, O.S.; Ball, M.W.; Shaykhutdinov, R.A.; Pieroraio, P.M.; Farshidfar, F.; Vogel, H.J.; Allaf, M.E.; Hyndman, M.E. Urine and Serum Metabolomics Analyses May Distinguish between Stages of Renal Cell Carcinoma. Metabolites 2017, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Sreekumar, A.; Poisson, L.M.; Rajendiran, T.M.; Khan, A.P.; Cao, Q.; Yu, J.; Laxman, B.; Mehra, R.; Lonigro, R.J.; Li, Y.; et al. Metabolomic Profiles Delineate Potential Role for Sarcosine in Prostate Cancer Progression. Nature 2009, 457, 910–914. [Google Scholar] [CrossRef] [PubMed]
- Amelio, I.; Cutruzzola, F.; Antonov, A.; Agostini, M.; Melino, G. Serine and Glycine Metabolism in Cancer. Trends Biochem. Sci. 2014, 39, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.; Nilsson, R.; Sharma, S.; Madhusudhan, N.; Kitami, T.; Souza, A.L.; Kafri, R.; Kirschner, M.W.; Clish, C.B.; Mootha, V.K. Metabolite Profiling Identifies a Key Role for Glycine in Rapid Cancer Cell Proliferation. Science 2012, 336, 1040–1044. [Google Scholar] [CrossRef] [PubMed]
- Hakimi, A.A.; Reznik, E.; Lee, C.-H.; Creighton, C.J.; Brannon, A.R.; Luna, A.; Aksoy, B.A.; Liu, E.M.; Shen, R.; Lee, W.; et al. An Integrated Metabolic Atlas of Clear Cell Renal Cell Carcinoma. Cancer Cell 2016, 29, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas Research Network. Comprehensive Molecular Characterization of Clear Cell Renal Cell Carcinoma. Nature 2013, 499, 43–49. [Google Scholar] [CrossRef]
- Mather, A.; Pollock, C. Glucose Handling by the Kidney. Kidney Int. 2011, 79, S1–S6. [Google Scholar] [CrossRef]
- Ragone, R.; Sallustio, F.; Piccinonna, S.; Rutigliano, M.; Vanessa, G.; Palazzo, S.; Lucarelli, G.; Ditonno, P.; Battaglia, M.; Fanizzi, F.; et al. Renal Cell Carcinoma: A Study through NMR-Based Metabolomics Combined with Transcriptomics. Diseases 2016, 4, 7. [Google Scholar] [CrossRef]
- Monteiro, M.; Moreira, N.; Pinto, J.; Pires-Luís, A.S.; Henrique, R.; Jerónimo, C.; Bastos, M.L.; Gil, A.M.; Carvalho, M.; Guedes de Pinho, P. GC-MS Metabolomics-based Approach for the Identification of a Potential VOC-biomarker Panel in the Urine of Renal Cell Carcinoma Patients. J. Cell. Mol. Med. 2017, 21, 2092–2105. [Google Scholar] [CrossRef]
- Ng, K.L.; Rajandram, R.; Morais, C.; Yap, N.Y.; Samaratunga, H.; Gobe, G.C.; Wood, S.T. Differentiation of Oncocytoma from Chromophobe Renal Cell Carcinoma (RCC): Can Novel Molecular Biomarkers Help Solve an Old Problem? J. Clin. Pathol. 2014, 67, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Ng, K.L.; Morais, C.; Bernard, A.; Saunders, N.; Samaratunga, H.; Gobe, G.; Wood, S. A Systematic Review and Meta-Analysis of Immunohistochemical Biomarkers That Differentiate Chromophobe Renal Cell Carcinoma from Renal Oncocytoma. J. Clin. Pathol. 2016, 69, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Demirovic, A.; Cesarec, S.; Spajic, B.; Tomas, D.; Bulimbasic, S.; Milosevic, M.; Marusic, Z.; Kruslin, B. Can Renal Oncocytoma Be Distinguished from Chromophobe Renal Cell Carcinoma by the Presence of Fibrous Capsule? Virchows Arch. 2010, 456, 85–89. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nizioł, J.; Bonifay, V.; Ossoliński, K.; Ossoliński, T.; Ossolińska, A.; Sunner, J.; Beech, I.; Arendowski, A.; Ruman, T. Metabolomic Study of Human Tissue and Urine in Clear Cell Renal Carcinoma by LC-HRMS and PLS-DA. Anal. Bioanal. Chem. 2018, 410, 3859–3869. [Google Scholar] [CrossRef] [PubMed]
- Wettersten, H.I.; Hakimi, A.A.; Morin, D.; Bianchi, C.; Johnstone, M.E.; Donohoe, D.R.; Trott, J.F.; Aboud, O.A.; Stirdivant, S.; Neri, B.; et al. Grade-Dependent Metabolic Reprogramming in Kidney Cancer Revealed by Combined Proteomics and Metabolomics Analysis. Cancer Res. 2015, 75, 2541. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, C.; Meregalli, C.; Bombelli, S.; Di Stefano, V.; Salerno, F.; Torsello, B.; De Marco, S.; Bovo, G.; Cifola, I.; Mangano, E.; et al. The Glucose and Lipid Metabolism Reprogramming Is Grade-Dependent in Clear Cell Renal Cell Carcinoma Primary Cultures and Is Targetable to Modulate Cell Viability and Proliferation. Oncotarget 2017, 8, 113502–113515. [Google Scholar] [CrossRef] [PubMed]
- Mickiewicz, B.; Tam, P.; Jenne, C.N.; Leger, C.; Wong, J.; Winston, B.W.; Doig, C.; Kubes, P.; Vogel, H.J. Integration of Metabolic and Inflammatory Mediator Profiles as a Potential Prognostic Approach for Septic Shock in the Intensive Care Unit. Crit. Care 2015, 19, 11. [Google Scholar] [CrossRef]
- Mickiewicz, B.; Thompson, G.C.; Blackwood, J.; Jenne, C.N.; Winston, B.W.; Vogel, H.J.; Joffe, A.R. Development of Metabolic and Inflammatory Mediator Biomarker Phenotyping for Early Diagnosis and Triage of Pediatric Sepsis. Crit. Care 2015, 19, 320. [Google Scholar] [CrossRef]
- Nicholson, J.K.; Foxall, P.J.; Spraul, M.; Farrant, R.D.; Lindon, J.C. 750 MHz 1H and 1H-13C NMR Spectroscopy of Human Blood Plasma. Anal. Chem. 1995, 67, 793–811. [Google Scholar] [CrossRef]
- Weljie, A.M.; Newton, J.; Mercier, P.; Carlson, E.; Slupsky, C.M. Targeted Profiling: Quantitative Analysis of 1H NMR Metabolomics Data. Anal. Chem 2006, 78, 4430–4442. [Google Scholar] [CrossRef]
- Dieterle, F.; Ross, A.; Schlotterbeck, G.; Senn, H. Probabilistic Quotient Normalization as Robust Method to Account for Dilution of Complex Biological Mixtures. Application in 1H NMR Metabonomics. Anal. Chem. 2006, 78, 4281–4290. [Google Scholar] [CrossRef]
- Van Den Berg, R.A.; Hoefsloot, H.C.; Westerhuis, J.A.; Smilde, A.K.; Van Der Werf, M.J. Centering, Scaling, and Transformations: Improving the Biological Information Content of Metabolomics Data. BMC Genom. 2006, 7, 142. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, L.; Johansson, E.; Kettaneh-Wold, N.; Trygg, J.; Wikstrom, C.; Wold, S. Multi-and Megavariate Data Analysis Part I: Basic Principles Applications; Umetrics Inc.: Umea, Sweden, 2006. [Google Scholar]
- Madsen, R.; Lundstedt, T.; Trygg, J. Chemometrics in Metabolomics—A Review in Human Disease Diagnosis. Anal. Chim. Acta 2010, 659, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Li, D.; Meng, J.; Wang, H. Introduction to SIMCA-P and Its Application In Handbook of Partial Least Squares: Concepts, Methods and Applications; Esposito Vinzi, V., Chin, W.W., Henseler, J., Wang, H., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 757–774. [Google Scholar]
- Galindo-Prieto, B.; Eriksson, L.; Trygg, J. Variable Influence on Projection (VIP) for Orthogonal Projections to Latent Structures (OPLS). J. Chemom. 2014, 28, 623–632. [Google Scholar] [CrossRef]
- Trygg, J.; Holmes, E.; Lundstedt, T. Chemometrics in Metabonomics. J. Proteome Res. 2007, 6, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, M.; Johnson, K. Over-Fitting and Model Tuning. In Applied Predictive Modeling; Springer: New York, NY, USA, 2013; pp. 61–92. [Google Scholar]
- Edgington, E.S. Randomized Single-Subject Experiments and Statistical Tests. J. Couns. Psychol. 1987, 34, 437–442. [Google Scholar] [CrossRef]
| Sample Group | Number of Samples | Age at Surgery (Range in Years) | Mean Age (Years) | Number of Men | Number of Women |
|---|---|---|---|---|---|
| Benign Control | 10 | 47–89 | 63 | 6 | 4 |
| Angiomyolipoma | 5 | 47–89 | 66 | 2 | 3 |
| Oncocytoma | 5 | 55–73 | 61 | 4 | 1 |
| RCC | 134 | 35–99 | 61 | 96 | 38 |
| ccRCC | 74 | 35–99 | 59 | 51 | 23 |
| Papillary | 22 | 51–84 | 66 | 18 | 4 |
| Chromophobe | 6 | 53–68 | 61 | 3 | 3 |
| RCC mixed type | 4 | 53–70 | 61 | 2 | 2 |
| Collecting duct carcinoma | 1 | 62 | - | - | 1 |
| Transitional cell carcinoma | 27 | 44–82 | 65 | 22 | 5 |
| Unclassified | 1 | 35 | - | - | 1 |
| Non-invasive papillary urothelial carcinoma (pTa) | 4 | 50–66 | 58 | 3 | 1 |
| Stage I (pT1) | 59 | 35–99 | 63 | 41 | 18 |
| Stage II (pT2) | 24 | 35–84 | 58 | 17 | 7 |
| Stage III (pT3) | 45 | 37–82 | 62 | 30 | 15 |
| Stage IV (pT4) | 7 | 37–71 | 62 | 7 | 0 |
| BMI 19–25 | 40 | 35–89 | 63 | 2 | 19 |
| BMI above 25 | 102 | 35–81 | 61 | 79 | 23 |
| BMI unknown | 3 | 41–61 | 52 | 2 | 1 |
| Model Type | R2Y | Q2 | CV ANOVA p-Value | Q2 Intercept | Sensitivity | Specificity |
|---|---|---|---|---|---|---|
| Benign vs RCC (All histology types included) | ||||||
| B vs. pT1 | 0.30 | 0.15 | 7.5e−3 | −0.21 | 1.00 | 0.30 |
| B vs. pT2 | 0.75 | 0.64 | 5.4e−7 | −0.37 | 1.00 | 0.90 |
| B vs. pT3 | 0.37 | 0.12 | 4.4e−2 | −0.24 | 0.98 | 0.40 |
| B vs. pT4 | 0.97 | 0.67 | 4.5e−2 | −0.41 | 1.00 | 1.00 |
| Benign vs RCC (ccRCC only) | ||||||
| B vs. pT1 | 0.42 | 0.27 | 4.3e−3 | −0.25 | 0.89 | 0.70 |
| B vs. pT2 | 0.96 | 0.82 | 3.3e−4 | −0.65 | 1.00 | 1.00 |
| B vs. pT3 | 0.48 | 0.32 | 1.0e−3 | −0.32 | 0.93 | 0.80 |
| B vs. pT4 | 0.82 | 0.65 | 8.6e−3 | −0.42 | 1.00 | 1.00 |
| Histology Comparisons | ||||||
| ONC vs. Chromophobe | 0.88 | 0.77 | 3.0e−2 | −0.51 | 1.00 | 1.00 |
| ONC vs. ccRCC | 0.32 | 0.20 | 4.8e−4 | −0.24 | 1.00 | 0.20 |
| Comparison | Increased in Cancer Relative to Benign | Decreased in Cancer Relative to Benign Samples |
|---|---|---|
| B vs. pT1 | O-acetylcarnitine | Glycine |
| Carnitine | Citrate | |
| B vs. pT2 | Gluconate | Citrate |
| Carnitine | Creatinine | |
| - | Glycine | |
| - | Propylene Glycol | |
| B vs. pT3 | Carnitine | Pyridoxine |
| O-acetylcarnitine | Adipate | |
| O-cresol | Citrate | |
| Methylhistidine | Glycine | |
| B vs. pT4 | Histamine | Citrate |
| O-acetylcarnitine | Succinate | |
| Taurine | Glycine | |
| Carnitine | Glycerol | |
| 5-aminolevulinate | - | |
| Carnitine | - | |
| Methionine | - | |
| ONC vs. Chromophobe | Trans-aconitate | Citrate |
| Succinate | - | |
| Methylhistidine | - | |
| Carnitine | - | |
| ONC vs. ccRCC | Tartrate | 1-methylnicotinamide |
| Trans-aconitate | Glycine | |
| Histamine | 2-hydroxyisobutyrate | |
| Carnitine | Citrate |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Falegan, O.S.; Arnold Egloff, S.A.; Zijlstra, A.; Hyndman, M.E.; Vogel, H.J. Urinary Metabolomics Validates Metabolic Differentiation Between Renal Cell Carcinoma Stages and Reveals a Unique Metabolic Profile for Oncocytomas. Metabolites 2019, 9, 155. https://doi.org/10.3390/metabo9080155
Falegan OS, Arnold Egloff SA, Zijlstra A, Hyndman ME, Vogel HJ. Urinary Metabolomics Validates Metabolic Differentiation Between Renal Cell Carcinoma Stages and Reveals a Unique Metabolic Profile for Oncocytomas. Metabolites. 2019; 9(8):155. https://doi.org/10.3390/metabo9080155
Chicago/Turabian StyleFalegan, Oluyemi S., Shanna A. Arnold Egloff, Andries Zijlstra, M. Eric Hyndman, and Hans J. Vogel. 2019. "Urinary Metabolomics Validates Metabolic Differentiation Between Renal Cell Carcinoma Stages and Reveals a Unique Metabolic Profile for Oncocytomas" Metabolites 9, no. 8: 155. https://doi.org/10.3390/metabo9080155
APA StyleFalegan, O. S., Arnold Egloff, S. A., Zijlstra, A., Hyndman, M. E., & Vogel, H. J. (2019). Urinary Metabolomics Validates Metabolic Differentiation Between Renal Cell Carcinoma Stages and Reveals a Unique Metabolic Profile for Oncocytomas. Metabolites, 9(8), 155. https://doi.org/10.3390/metabo9080155




