Influence of Dietary Ingredients on Lean Body Percent, Uremic Toxin Concentrations, and Kidney Function in Senior-Adult Cats
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Study Design
2.2. Foods
2.3. Chemical Analyses for Biomarkers and Metabolites
2.4. Statistical Methods
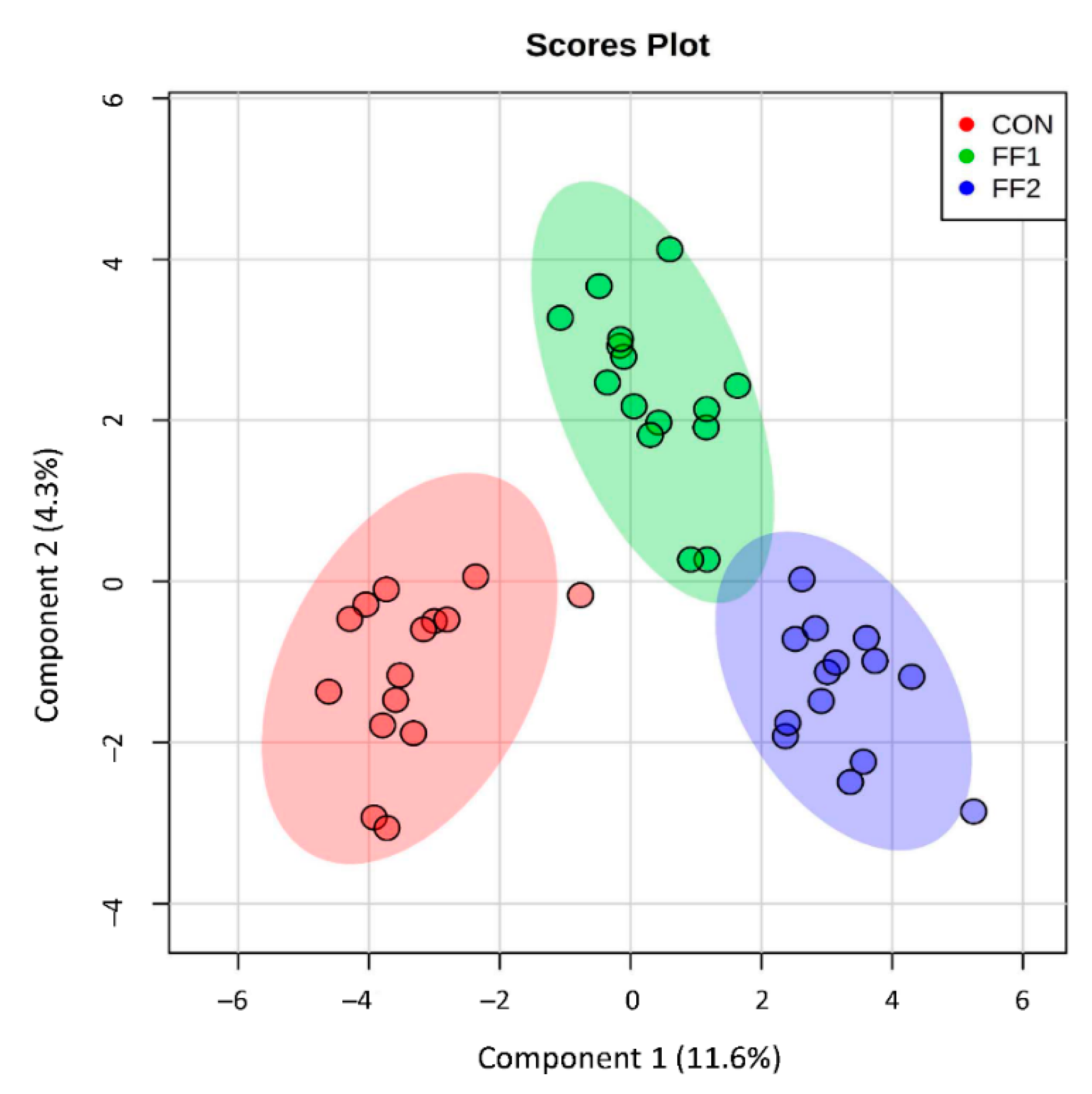
3. Results
3.1. Effect of Age on Lean Body Percent, GFR, and Serum Biochemistries
3.2. Effect of Feeding Renal Protective Foods on Body Weight, Lean Body Percent, GFR, Serum Biomarkers, and PGE2 across Time
3.3. Effect of Feeding Renal Protective Foods on Major Serum Fatty Acid Concentrations across Time
3.4. Effect of Functional Foods on Plasma Metabolite Concentrations of Antioxidants and Methylation Substrates after a Three-Month Feeding Period
3.5. Effect of Functional Foods on Plasma Metabolite Concentrations of Compounds Produced by Gut Microbial Metabolism after a Three-Month Feeding Period
4. Discussion
4.1. Effects of Age on Lean Body Percent and Renal Function
4.2. Effects of Functional Foods on Lean Body Percent, Renal Function, and Serum Fatty Acids and PGE2 Concentrations
4.3. Effects of Functional Foods on Selected Plasma Metabolite Concentrations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Hall, J.A.; Yerramilli, M.; Obare, E.; Yerramilli, M.; Yu, S.; Jewell, D.E. Comparison of serum concentrations of symmetric dimethylarginine and creatinine as kidney function biomarkers in healthy geriatric cats fed reduced protein foods enriched with fish oil, l-carnitine, and medium-chain triglycerides. Vet. J. 2014, 202, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.A.; Yerramilli, M.; Obare, E.; Yerramilli, M.; Jewell, D.E. Comparison of serum concentrations of symmetric dimethylarginine and creatinine as kidney function biomarkers in cats with chronic kidney disease. J. Vet. Intern. Med. 2014, 28, 1676–1683. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.H.; Mitch, W.E. Mechanisms of muscle wasting in chronic kidney disease. Nat. Rev. Nephrol. 2014, 10, 504–516. [Google Scholar] [CrossRef] [PubMed]
- Vlassara, H.; Torreggiani, M.; Post, J.B.; Zheng, F.; Uribarri, J.; Striker, G.E. Role of oxidants/inflammation in declining renal function in chronic kidney disease and normal aging. Kidney Int. Suppl. 2009, 114, S3–S11. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.A.; Yerramilli, M.; Obare, E.; Panickar, K.S.; Bobe, G.; Jewell, D.E. Nutritional interventions that slow the age-associated decline in renal function in a canine geriatric model for elderly humans. J. Nutr. Health Aging 2016, 20, 1010–1023. [Google Scholar] [CrossRef]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.; Stehle, P.; Teta, D.; et al. Evidence-based recommendations for optimal dietary protein intake in older people: A position paper from the prot-age study group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef]
- Polzin, D.J. Evidence-based step-wise approach to managing chronic kidney disease in dogs and cats. J. Vet. Emerg. Crit. Care (San Antonio) 2013, 23, 205–215. [Google Scholar] [CrossRef]
- Forrester, S.; Adams, L.; Allen, T. Chronic kidney disease. In Small Animal Clinical Nutrition, 5th ed.; Hand, M., Thatcher, C., Remillard, R., Roudebush, P., Novotny, B., Eds.; Mark Morris Institute: Topeka, KS, USA, 2010; pp. 765–810. [Google Scholar]
- Bartges, J.W. Chronic kidney disease in dogs and cats. Vet. Clin. N. Am. Small Anim. Pract. 2012, 42, 669–692. [Google Scholar] [CrossRef]
- Hall, J.A.; Jewell, D.E. Feeding healthy beagles medium-chain triglycerides, fish oil, and carnitine offsets age-related changes in serum fatty acids and carnitine metabolites. PLoS ONE 2012, 7, e49510. [Google Scholar] [CrossRef]
- Vogt, A.H.; Rodan, I.; Brown, M.; Brown, S.; Buffington, C.A.; Forman, M.J.; Neilson, J.; Sparkes, A.; European Society of Feline Medicine; Feline Advisory Bureaus Well Cat for Life. AAFP-AAHA: Feline life stage guidelines. J. Am. Anim. Hosp. Assoc. 2010, 46, 70–85. [Google Scholar] [CrossRef]
- NRC. Guide for the Care and Use of Laboratory Animals; National Academy Press: Washington, DC, USA, 2011. [Google Scholar]
- Hall, J.A.; Melendez, L.D.; Jewell, D.E. Using gross energy improves metabolizable energy predictive equations for pet foods whereas undigested protein and fiber content predict stool quality. PLoS ONE 2013, 8, e54405. [Google Scholar] [CrossRef] [PubMed]
- Roush, J.K.; Dodd, C.E.; Fritsch, D.A.; Allen, T.A.; Jewell, D.E.; Schoenherr, W.D.; Richardson, D.C.; Leventhal, P.S.; Hahn, K.A. Multicenter veterinary practice assessment of the effects of omega-3 fatty acids on osteoarthritis in dogs. J. Am. Vet. Med. Assoc. 2010, 236, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- De Baere, S.; Smets, P.; Finch, N.; Heiene, R.; De Backer, P.; Daminet, S.; Croubels, S. Quantitative determination of exo- and endo-iohexol in canine and feline samples using high performance liquid chromatography with ultraviolet detection. J. Pharm. Biomed. Anal. 2012, 61, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Chong, J.; Soufan, O.; Li, C.; Caraus, I.; Li, S.; Bourque, G.; Wishart, D.S.; Xia, J. Metaboanalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 2018, 46, W486–W494. [Google Scholar] [CrossRef] [PubMed]
- Santanasto, A.J.; Goodpaster, B.H.; Kritchevsky, S.B.; Miljkovic, I.; Satterfield, S.; Schwartz, A.V.; Cummings, S.R.; Boudreau, R.M.; Harris, T.B.; Newman, A.B. Body composition remodeling and mortality: The health aging and body composition study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2017, 72, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, E.; Chiles Shaffer, N.; Gonzalez-Freire, M.; Shardell, M.D.; Zoli, M.; Studenski, S.A.; Ferrucci, L. Early body composition, but not body mass, is associated with future accelerated decline in muscle quality. J. Cachexia Sarcopenia Muscle 2017, 8, 490–499. [Google Scholar] [CrossRef]
- Rantanen, T.; Harris, T.; Leveille, S.G.; Visser, M.; Foley, D.; Masaki, K.; Guralnik, J.M. Muscle strength and body mass index as long-term predictors of mortality in initially healthy men. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2000, 55, M168–M173. [Google Scholar] [CrossRef]
- Wang, X.L.; Vrtiska, T.J.; Avula, R.T.; Walters, L.R.; Chakkera, H.A.; Kremers, W.K.; Lerman, L.O.; Rule, A.D. Age, kidney function, and risk factors associate differently with cortical and medullary volumes of the kidney. Kidney Int. 2014, 85, 677–685. [Google Scholar] [CrossRef]
- Abdulkader, R.; Burdmann, E.A.; Lebrao, M.L.; Duarte, Y.A.O.; Zanetta, D.M.T. Aging and decreased glomerular filtration rate: An elderly population-based study. PLoS ONE 2017, 12, e0189935. [Google Scholar] [CrossRef]
- Hommos, M.S.; Glassock, R.J.; Rule, A.D. Structural and functional changes in human kidneys with healthy aging. J. Am. Soc. Nephrol. 2017, 28, 2838–2844. [Google Scholar] [CrossRef] [PubMed]
- Calvani, R.; Miccheli, A.; Landi, F.; Bossola, M.; Cesari, M.; Leeuwenburgh, C.; Sieber, C.C.; Bernabei, R.; Marzetti, E. Current nutritional recommendations and novel dietary strategies to manage sarcopenia. J. Frailty Aging 2013, 2, 38–53. [Google Scholar] [PubMed]
- Shlisky, J.; Bloom, D.E.; Beaudreault, A.R.; Tucker, K.L.; Keller, H.H.; Freund-Levi, Y.; Fielding, R.A.; Cheng, F.W.; Jensen, G.L.; Wu, D.; et al. Nutritional considerations for healthy aging and reduction in age-related chronic disease. Adv. Nutr. 2017, 8, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Reilly, R.F. Effects of dietary interventions on incidence and progression of CKD. Nat. Rev. Nephrol 2014, 10, 712–724. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Sjogren, P.; Arnlov, J.; Banerjee, T.; Cederholm, T.; Riserus, U.; Lindholm, B.; Lind, L.; Carrero, J.J. A proinflammatory diet is associated with systemic inflammation and reduced kidney function in elderly adults. J. Nutr. 2015, 145, 729–735. [Google Scholar] [CrossRef]
- Ringseis, R.; Keller, J.; Eder, K. Mechanisms underlying the anti-wasting effect of l-carnitine supplementation under pathologic conditions: Evidence from experimental and clinical studies. Eur. J. Nutr. 2013, 52, 1421–1442. [Google Scholar] [CrossRef]
- Juliet, P.A.; Balasubramaniam, D.; Balasubramaniam, N.; Panneerselvam, C. Carnitine: A neuromodulator in aged rats. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2003, 58, 970–974. [Google Scholar] [CrossRef]
- Thangasamy, T.; Subathra, M.; Sittadjody, S.; Jeyakumar, P.; Joyee, A.G.; Mendoza, E.; Chinnakkanu, P. Role of L-carnitine in the modulation of immune response in aged rats. Clin. Chim. Acta Int. J. Clin. Chem. 2008, 389, 19–24. [Google Scholar] [CrossRef]
- Owen, L.; Sunram-Lea, S.I. Metabolic agents that enhance atp can improve cognitive functioning: A review of the evidence for glucose, oxygen, pyruvate, creatine, and l-carnitine. Nutrients 2011, 3, 735–755. [Google Scholar] [CrossRef]
- Takahashi, N.; Morimoto, S.; Okigaki, M.; Seo, M.; Someya, K.; Morita, T.; Matsubara, H.; Sugiura, T.; Iwasaka, T. Decreased plasma level of vitamin C in chronic kidney disease: Comparison between diabetic and non-diabetic patients. Nephrol. Dial. Transpl. 2011, 26, 1252–1257. [Google Scholar] [CrossRef]
- Fletcher, A.E.; Breeze, E.; Shetty, P.S. Antioxidant vitamins and mortality in older persons: Findings from the nutrition add-on study to the medical research council trial of assessment and management of older people in the community. Am. J. Clin. Nutr. 2003, 78, 999–1010. [Google Scholar] [CrossRef] [PubMed]
- Karamouzis, I.; Sarafidis, P.A.; Karamouzis, M.; Iliadis, S.; Haidich, A.B.; Sioulis, A.; Triantos, A.; Vavatsi-Christaki, N.; Grekas, D.M. Increase in oxidative stress but not in antioxidant capacity with advancing stages of chronic kidney disease. Am. J. Nephrol. 2008, 28, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Boudouris, G.; Verginadis, I.I.; Simos, Y.V.; Zouridakis, A.; Ragos, V.; Karkabounas, S.; Evangelou, A.M. Oxidative stress in patients treated with continuous ambulatory peritoneal dialysis (CAPD) and the significant role of vitamin C and E supplementation. Int. Urol. Nephrol. 2013, 45, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Moffitt, T.A.; Garrett, P.J.; Hannon-Fletcher, M.P. Micronutrient supplementation in maintenance haemodialysis patients enhances activity of antioxidant enzymes. J. Nephrol. 2013, 26, 403–411. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Small, D.M.; Coombes, J.S.; Bennett, N.; Johnson, D.W.; Gobe, G.C. Oxidative stress, anti-oxidant therapies and chronic kidney disease. Nephrology (Carlton) 2012, 17, 311–321. [Google Scholar] [CrossRef]
- Sung, C.C.; Hsu, Y.C.; Chen, C.C.; Lin, Y.F.; Wu, C.C. Oxidative stress and nucleic acid oxidation in patients with chronic kidney disease. Oxid. Med. Cell. Longev. 2013, 2013, 301982. [Google Scholar] [CrossRef]
- Calder, P.C.; Yaqoob, P. Understanding omega-3 polyunsaturated fatty acids. Postgrad. Med. 2009, 121, 148–157. [Google Scholar] [CrossRef]
- Hall, J.A.; Brockman, J.A.; Jewell, D.E. Dietary fish oil alters the lysophospholipid metabolomic profile and decreases urinary 11-dehydro thromboxane B(2) concentration in healthy beagles. Vet. Immunol. Immunopathol. 2011, 144, 355–365. [Google Scholar] [CrossRef]
- Brown, S.A. Oxidative stress and chronic kidney disease. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 157–166. [Google Scholar] [CrossRef]
- Farsijani, S.; Morais, J.A.; Payette, H.; Gaudreau, P.; Shatenstein, B.; Gray-Donald, K.; Chevalier, S. Relation between mealtime distribution of protein intake and lean mass loss in free-living older adults of the nuage study. Am. J. Clin. Nutr. 2016, 104, 694–703. [Google Scholar] [CrossRef]
- Rebrin, I.; Sohal, R.S. Pro-oxidant shift in glutathione redox state during aging. Adv. Drug Deliv. Rev. 2008, 60, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Dello, S.A.; Neis, E.P.; de Jong, M.C.; van Eijk, H.M.; Kicken, C.H.; Olde Damink, S.W.; Dejong, C.H. Systematic review of ophthalmate as a novel biomarker of hepatic glutathione depletion. Clin. Nutr. 2013, 32, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Reddy, M.A.; Natarajan, R. Recent developments in epigenetics of acute and chronic kidney diseases. Kidney Int. 2015, 88, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Park, L.K.; Friso, S.; Choi, S.W. Nutritional influences on epigenetics and age-related disease. Proc. Nutr. Soc. 2012, 71, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, S.; Jao, T.M.; Inagi, R. Dietary metabolites and chronic kidney disease. Nutrients 2017, 9, 358. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.N.; Chou, C.C.; Tsai, P.S.J.; Lee, Y.J. Plasma indoxyl sulfate concentration predicts progression of chronic kidney disease in dogs and cats. Vet. J. 2018, 232, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Nataatmadja, M.; Cho, Y.; Campbell, K.; Johnson, D.W. The roles of indoxyl sulphate and p-cresyl sulphate in patients with chronic kidney disease: A review of therapeutic options. In Chronic Kidney Disease; Rath, T., Ed.; IntechOpen: London, UK, 2018. [Google Scholar]
- Pimpao, R.C.; Ventura, M.R.; Ferreira, R.B.; Williamson, G.; Santos, C.N. Phenolic sulfates as new and highly abundant metabolites in human plasma after ingestion of a mixed berry fruit puree. Br. J. Nutr. 2015, 113, 454–463. [Google Scholar] [CrossRef]
- Wikoff, W.R.; Anfora, A.T.; Liu, J.; Schultz, P.G.; Lesley, S.A.; Peters, E.C.; Siuzdak, G. Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc. Natl. Acad. Sci. USA 2009, 106, 3698–3703. [Google Scholar] [CrossRef]
| Demographics | Control Food (n = 15) | Functional Food 1 (n = 15) | Functional Food 2 (n = 14) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Range | Mean | SD | Range | Mean | SD | Range | |
| Age, years | 12.0 | 0.9 | 10.9–13.7 | 12.3 | 0.5 | 10.9–14.0 | 12.2 | 1.0 | 10.7–14.0 |
| Sex | 9 females; 6 males | 9 females; 6 males | 7 females; 7 males | ||||||
| Body weight, kg | 4.45 | 0.84 | 3.31–5.82 | 4.73 | 1.11 | 3.22–6.62 | 4.23 | 0.78 | 3.11–5.82 |
| Body lean, kg | 3.29 | 0.46 | 2.67–3.96 | 3.53 | 0.74 | 2.52–4.81 | 3.38 | 0.56 | 2.66–4.58 |
| Body fat, kg | 1.05 | 0.58 | 0.30–2.15 | 1.08 | 0.51 | 0.31–1.96 | 0.74 | 0.37 | 0.27–1.45 |
| Food/Nutrient | Pre-Trial Food | Control Food | Functional Food 1 | Functional Food 2 |
|---|---|---|---|---|
| Added fish oil, % | 0 | 0.1 | 0.5 | 0.5 |
| Fruit and vegetables, % | 0 | 0 | 1.9 | 4.0 |
| Pea protein, % | 0 | 0 | 19.7 | 43.6 |
| Wet meat chicken, % | 0 | 0 | 16.0 | 15.5 |
| Chicken meal, % | 28.0 | 19.4 | 0 | 0 |
| Corn gluten meal, % | 15.7 | 19.2 | 16.0 | 0 |
| Moisture | 6.50 | 6.77 | 6.78 | 6.34 |
| Protein | 33.43 | 32.58 | 30.24 | 31.77 |
| Fat | 21.12 | 20.84 | 16.17 | 15.25 |
| Atwater Energy, 5 kcal/kg | 4092 | 4019 | 3810 | 3746 |
| Ash | 4.94 | 5.16 | 5.57 | 6.21 |
| Crude fiber | 1.8 | 3.0 | 1.9 | 2.2 |
| Calcium | 0.78 | 0.85 | 0.80 | 0.78 |
| Phosphorus | 0.84 | 0.74 | 0.76 | 0.83 |
| Sodium | 0.46 | 0.30 | 0.36 | 0.36 |
| Total tocopherols, IU/kg | 49 | 1059 | 1137 | 1268 |
| Vitamin C, mg/kg | 97 | 192 | 231 | 231 |
| Palmitic acid [16:0] | 4.27 | 4.11 | 2.97 | 3.06 |
| Stearic acid [18:0] | 2.01 | 1.90 | 0.69 | 0.72 |
| linoleic acid (LA) [18:2 (n − 6)] | 3.70 | 3.72 | 3.55 | 3.49 |
| alpha linolenic acid (αLA) [18:3 (n − 3)] | 0.18 | 0.20 | 0.28 | 0.33 |
| arachidonic acid (ARA) [20:4 (n − 6)] | 0.12 | 0.12 | 0.04 | 0.04 |
| eicosapentaenoic acid (EPA) [20:5 (n − 3)] | 0.01 | 0.03 | 0.09 | 0.10 |
| docosapentaenoic acid (DPA) [22:5 (n − 3)] | 0.01 | 0.02 | 0.02 | 0.02 |
| docosahexaenoic acid (DHA) [22:6 (n − 3)] | 0.01 | 0.02 | 0.06 | 0.06 |
| saturated fatty acids (SFA) 6 | 6.69 | 6.42 | 3.90 | 4.02 |
| monounsaturated fatty acids (MUFA) 7 | 8.01 | 7.71 | 5.69 | 5.60 |
| polyunsaturated fatty acids (PUFA) 8 | 4.22 | 4.07 | 4.12 | 4.03 |
| (n − 6) fatty acids (FA 9) | 4.03 | 3.80 | 3.67 | 3.61 |
| (n − 3) fatty acids (FA) 10 | 0.19 | 0.27 | 0.45 | 0.42 |
| (n − 6):(n − 3) ratio | 21.2 | 14.1 | 8.2 | 8.6 |
| Variables | Young-Adult Cats | Senior-Adult Cats at Baseline (T0) and after Feeding for Six months (T6) | SEM | p–Values 5 Senior-Adult Cats vs. Young-Adult Cats | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Senior-Adult Cats (T0) | Control (T6) | FF1 (T6) | FF2 (T6) | Senior-Adult Cats (T0) | Control (T6) | FF1 (T6) | FF2 (T6) | |||
| Body Mass and Composition: | ||||||||||
| Body Weight, kg 6 | 4.53 | 4.47 | 4.44 | 4.63 | 4.20 | 0.18 | 0.66 | 0.76 | 0.70 | 0.25 |
| Lean Body Mass, kg 6 | 3.70 | 3.40 | 3.33 | 3.57 | 3.60 * | 0.02 | 0.06 | 0.09 | 0.53 | 0.52 |
| Fat Body Mass, kg 6 | 0.70 | 0.96 | 0.99 | 0.95 | 0.53 * | 0.11 | 0.03 | 0.04 | 0.08 | 0.21 |
| Lean Body, % | 81.6 | 76.9 | 75.9 | 78.2 | 84.9 | 1.9 | 0.02 | 0.02 | 0.14 | 0.16 |
| Fat Body,% | 14.4 | 20.6 | 21.6 | 19.2 * | 12.4 * | 1.79 | <0.01 | <0.01 | 0.04 | 0.39 |
| Renal Function: | ||||||||||
| Glomerular Filtration Rate, mL/min/kg 7 | 2.08 7 | 1.92 | 2.09 * | 2.32 * | 2.13 * | 0.13 | 0.09 | 0.91 | 0.21 | 0.87 |
| Serum Metabolites: | ||||||||||
| Creatinine, mg/dL | 1.31 | 1.22 | 1.25 | 0.99 * | 1.17 | 0.049 | 0.11 | <0.01 | <0.01 | <0.01 |
| Symmetric dimethylarginine (SDMA), μg/dL | 11.5 | 11.1 | 10.3 * | 9.1 * | 10.8 | 0.4 | 0.79 | 0.01 | <0.01 | 0.15 |
| Blood urea nitrogen (BUN), mg/dL | 21.82 | 20.33 | 21.30 * | 22.35 * | 23.21 * | 0.45 | 0.02 | 0.56 | 0.55 | 0.12 |
| Total Protein, mg/dL | 6.71 | 6.68 | 6.97 | 7.37 * | 7.42 * | 0.14 | 0.67 | 0.17 | <0.01 | <0.01 |
| Albumin, mg/dL | 3.31 | 2.82 | 2.67 * | 2.78 | 2.68 | 0.07 | <0.01 | <0.01 | <0.01 | <0.01 |
| Prostaglandin, pg/dL | NA 8 | 162 | 116 * | 94 * | 149 | 8.5 | NA | NA | NA | NA |
| Variables | Renal-Protective Foods | SEM | p-Values for Food Effect 4 | p-Values for Food Comparisons 5 | p-Values for Time Effect 4 | |||
|---|---|---|---|---|---|---|---|---|
| Control | FF1 | FF2 | FF1 vs. Control | FF2 vs. Control | ||||
| Body Mass and Composition: | ||||||||
| Body Weight, kg 6 | ||||||||
| Initial, T0 | 4.45 | 4.73 | 4.23 | 0.25 | ||||
| Change, T6 − T0 | −0.07 | −0.10 | −0.03 | 0.07 | 0.58 | 0.37 | 0.83 | 0.98 |
| Lean Body, % 7 | ||||||||
| Initial, T0 | 75 | 75.4 | 80.4 | 2.1 | ||||
| Change, T6 − T0 | 0.9 | 2.7 | 4.5 | 1.0 | <0.01 | <0.01 | <0.01 | <0.01 |
| Renal Function: | ||||||||
| Glomerular Filtration Rate, mL/min/kg | ||||||||
| Initial, T0 | 1.85 | 2.01 | 1.85 | 0.04 | ||||
| Change, T6 − T0 | 0.23 | 0.30 | 0.27 | 0.11 | 0.86 | 0.65 | 0.82 | <0.01 |
| Serum Biochemistries: | ||||||||
| Creatinine, mg/dL | ||||||||
| Initial, T0 | 1.15 | 1.13 | 1.20 | 0.062 | ||||
| Change, T6 − T0 | 0.04 | −0.19 | −0.05 | 0.047 | 0.07 | <0.01 | 0.2 | <0.01 |
| SDMA, μg/dL | ||||||||
| Initial, T0 | 10.9 | 10.9 | 11.1 | 0.29 | ||||
| Change, T6 − T0 | −0.67 | −1.76 | −0.31 | 0.33 | <0.01 | <0.01 | 0.85 | <0.01 |
| BUN, mg/dL | ||||||||
| Initial, T0 | 19.8 | 20.1 | 20.3 | 0.6 | ||||
| Change, T6 − T0 | 1.52 | 1.32 | 3.32 | 0.6 | 0.09 | 0.85 | 0.08 | <0.01 |
| Total Protein, mg/dL | ||||||||
| Initial, T0 | 7.22 | 6.44 | 6.39 | 0.05 | ||||
| Change, T6 − T0 | −0.10 | 0.82 | 1.02 | 0.11 | <0.01 | <0.01 | <0.01 | <0.01 |
| Albumin, mg/dL | ||||||||
| Initial, T0 | 2.83 | 2.8 | 2.77 | 0.05 | ||||
| Change, T6 − T0 | −0.33 | 0.04 | 0.01 | 0.05 | <0.01 | <0.01 | <0.01 | 0.04 |
| Fatty Acids (mg/dL) | Renal Protective Foods | SEM | p-Values for Food Effect 4 | p-Values for Food Comparisons 5 | p-Values for Time Effect 4 | |||
|---|---|---|---|---|---|---|---|---|
| Control | FF1 | FF2 | FF1 vs. Control | FF2 vs. Control | ||||
| Individual: | ||||||||
| C16:0, T0 | 24.5 | 24.3 | 22.5 | 1.2 | ||||
| Change, T6 − T0 | +1.1 | +0.61 | +0.53 | 1.0 | 0.19 | 0.52 | 0.25 | 0.51 |
| C16:1, T0 | 0.76 | 0.63 | 0.66 | 0.04 | ||||
| Change, T6 − T0 | +0.57 | +0.31 | +0.50 | 0.06 | 0.07 | <0.01 | 0.34 | <0.01 |
| C18:0, T0 | 43.9 | 39.7 | 37.1 | 1.1 | ||||
| Change, T6 − T0 | +0.5 | +2.2 | −0.4 | 0.7 | <0.01 | 0.10 | 0.36 | 0.07 |
| C18:1, T0 | 22.5 | 24.6 | 21.4 | 1.1 | ||||
| Change, T6 − T0 | +3.5 | +1.5 | +2.3 | 0.7 | 0.33 | 0.01 | 0.14 | <0.01 |
| C18:2 (n − 6), T0 | 48.4 | 51.6 | 42.7 | 1.2 | ||||
| Change, T6 − T0 | +4.6 | +0.7 | +2.3 | 1.1 | 0.26 | 0.01 | 0.13 | <0.01 |
| C18:3 (n − 3), T0 | 0.00 | 0.00 | 0.00 | 0.00 | ||||
| Change, T6 − T0 | +0.07 | +0.08 | +0.06 | 0.01 | 0.88 | 0.45 | 0.19 | <0.01 |
| C20:4 (n − 6), T0 | 18.1 | 16.7 | 15.6 | 0.5 | ||||
| Change, T6 − T0 | −1.4 | −4.1 | −4.6 | 0.5 | <0.01 | <0.01 | <0.01 | <0.01 |
| C20:5 (n − 3), T0 | 0.09 | 0.09 | 0.09 | 0.01 | ||||
| Change, T6 − T0 | +0.77 | +1.79 | +1.81 | 0.17 | <0.01 | <0.01 | <0.01 | <0.01 |
| C22:5 (n − 3), T0 | 0.62 | 0.56 | 0.63 | 0.03 | ||||
| Change, T6 − T0 | +0.56 | +1.18 | +0.98 | 0.11 | <0.01 | <0.01 | 0.01 | <0.01 |
| C22:6 (n − 3), T0 | 2.05 | 2.53 | 2.41 | 0.09 | ||||
| Change, T6 − T0 | +1.18 | +2.99 | +2.30 | 0.28 | <0.01 | <0.01 | <0.01 | <0.01 |
| Sums: | ||||||||
| SFA 6, T0 | 68.5 | 64.1 | 59.7 | 1.7 | ||||
| Change, T6 − T0 | +1.8 | +3.1 | +0.3 | 1.1 | 0.19 | 0.44 | 0.36 | <0.01 |
| MUFA 7, T0 | 23.2 | 25.2 | 22.1 | 0.6 | ||||
| Change, T6 − T0 | +3.8 | +2.0 | +2.8 | 0.6 | 0.51 | 0.04 | 0.23 | <0.01 |
| PUFA 8, T0 | 72.5 | 75.3 | 64.5 | 1.8 | ||||
| Change, T6 − T0 | +6.2 | +2.1 | +2.5 | 1.4 | 0.02 | 0.04 | 0.07 | <0.01 |
| (n − 6) PUFA 9, T0 | 70.4 | 72.7 | 62.0 | 1.7 | ||||
| Change, T6 − T0 | +4.2 | −2.7 | −1.7 | 1.4 | <0.01 | <0.01 | <0.01 | 0.35 |
| (n − 3) PUFA 10, T0 | 2.1 | 2.6 | 2.5 | 0.2 | ||||
| Change, T6 − T0 | +2.0 | +4.9 | +4.2 | 0.5 | <0.01 | <0.01 | <0.01 | <0.01 |
| Ratios: | ||||||||
| (n − 6):(n − 3), T0 | 35.4 | 28.5 | 26.3 | 2.0 | ||||
| Change, T6 − T0 | −13.7 | −14.3 | −13.0 | 1.8 | 0.96 | 0.79 | 0.79 | <0.01 |
| PUFA:SFA, T0 | 1.06 | 1.19 | 1.08 | 0.01 | ||||
| Change, T6 − T0 | +0.06 | −0.03 | +0.043 | 0.01 | <0.01 | <0.01 | 0.14 | 0.12 |
| Metabolite Class 4 | Control | Functional Food 1 | Functional Food 2 | One Way-ANOVA on T3 − T0 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T3 − T0 DELTA | T3 − T0 DELTA | T3 − T0 DELTA | p-Value | q-Value | Tukey’s HSD 6 Post Hoc | |||||||||
| Mean | SEM 5 | Paired t-Test p-Value | Mean | SEM 5 | Paired t-Test p-Value | Mean | SEM 5 | Paired t-Test p-Value | Control vs. FF1 | Control vs. FF2 | FF1 vs. FF2 | |||
| Glutathione: | ||||||||||||||
| ophthalmate | 0.15 | 0.19 | 0.39 | −0.43 | 0.26 | 0.07 | −1.59 | 0.44 | 6.3 × 10−5 | 9.0 × 10−5 | 2.5 × 10−4 | 0.12 | 5.5 × 10−5 | 0.02 |
| pyroglutamine | 0.23 | 0.18 | 0.12 | −1.80 | 1.08 | 4.6 × 10−3 | −1.61 | 0.91 | 0.01 | 2.7 × 10−3 | 3.9 × 10−3 | 0.01 | 0.01 | 0.98 |
| glutathione, oxidized (GSSG) | −0.10 | 0.15 | 0.69 | −0.41 | 0.11 | 4.5 × 10−4 | −0.55 | 0.10 | 9.6 × 10−6 | 7.3 × 10−4 | 1.5 × 10−3 | 0.03 | 0.00 | 0.28 |
| cysteine-glutathione disulfide | −0.03 | 0.07 | 0.65 | −0.12 | 0.08 | 0.03 | −0.22 | 0.04 | 2.3 × 10−5 | 0.01 | 0.01 | 0.42 | 4.7 × 10−3 | 0.10 |
| homocysteine | −0.14 | 0.17 | 0.36 | 0.40 | 0.15 | 0.01 | 0.31 | 0.20 | 0.31 | 0.05 | 0.04 | 0.04 | 0.31 | 0.59 |
| Methylation: | ||||||||||||||
| glycine | −0.16 | 0.07 | 0.01 | −0.12 | 0.10 | 0.32 | −0.21 | 0.08 | 0.10 | 0.21 | 0.10 | 0.95 | 0.35 | 0.22 |
| sarcosine (N-methylglycine) | −0.03 | 0.14 | 0.85 | 0.21 | 0.10 | 0.09 | 0.19 | 0.18 | 0.80 | 0.47 | 0.15 | 0.49 | 0.98 | 0.60 |
| betaine | 1.09 | 0.25 | 1.1 × 10−6 | −0.32 | 0.08 | 7.0 × 10−5 | −0.02 | 0.09 | 0.95 | 1.9 × 10−10 | 4.5 × 10−9 | 3.5 × 10−10 | 4.6 × 10−6 | 0.01 |
| 5-methylcytidine | −0.03 | 0.06 | 0.57 | 0.95 | 0.27 | 1.3 × 10−3 | 4.99 | 0.49 | 1.4 × 10−12 | 2.1 × 10−14 | 7.6 × 10−13 | 2.3 × 10−4 | 2.3 × 10−10 | 4.2 × 10−9 |
| 5-methylcytosine | −0.05 | 0.09 | 0.51 | 0.37 | 0.14 | 0.03 | 2.05 | 0.33 | 4.7 × 10−5 | 1.4 × 10−5 | 4.4 × 10−5 | 0.11 | 8.4 × 10−6 | 4.5 × 10−3 |
| 5-hydroxymethylcytosine | −0.08 | 0.06 | 0.19 | 0.32 | 0.08 | 3.4 × 10−3 | 0.66 | 0.12 | 1.1 × 10−3 | 1.8 × 10−4 | 4.4 × 10−4 | 0.02 | 1.3 × 10−4 | 0.20 |
| 5-methyl-2′-deoxycytidine | −0.09 | 0.07 | 0.03 | 0.19 | 0.08 | 0.03 | 0.35 | 0.12 | 0.01 | 2.1 × 10−3 | 3.4 × 10−3 | 0.06 | 1.6 × 10−3 | 0.35 |
| Putrefactive Postbiotics: | ||||||||||||||
| 3-indoxyl sulfate | −0.36 | 0.15 | 0.03 | −0.84 | 0.24 | 0.01 | −1.16 | 0.18 | 9.5 × 10−5 | 0.01 | 0.01 | 0.42 | 0.01 | 0.16 |
| 2-oxindole-3-acetate | −0.02 | 0.15 | 0.87 | −0.24 | 0.16 | 0.25 | −1.04 | 0.18 | 3.5 × 10−6 | 4.8 × 10−5 | 1.4 × 10−4 | 0.58 | 6.0 × 10−5 | 1.2 × 10−3 |
| indolepropionate | −0.25 | 0.19 | 0.15 | 0.78 | 0.57 | 0.21 | 0.12 | 0.43 | 0.27 | 0.11 | 0.06 | 0.29 | 0.83 | 0.11 |
| indoleacrylate | 0.02 | 0.20 | 0.86 | 0.45 | 0.27 | 0.08 | 0.24 | 0.31 | 0.66 | 0.58 | 0.17 | 0.56 | 0.95 | 0.76 |
| indolelactate | −0.14 | 0.11 | 0.09 | 0.07 | 0.17 | 0.95 | 0.07 | 0.11 | 0.97 | 0.52 | 0.16 | 0.56 | 0.62 | 1.00 |
| indoleacetate | 0.38 | 0.22 | 0.11 | 0.37 | 0.25 | 0.32 | −0.58 | 0.57 | 0.28 | 0.14 | 0.07 | 1.00 | 0.20 | 0.19 |
| 3-(4-hydroxyphenyl)lactate (HPLA) | 0.01 | 0.07 | 0.64 | −0.19 | 0.06 | 2.6 × 10−3 | −0.41 | 0.07 | 1.3 × 10−8 | 4.5 × 10−8 | 6.4 × 10−7 | 0.01 | 2.2 × 10−8 | 8.0 × 10−4 |
| phenylpropionylglycine | −0.71 | 0.18 | 1.2 × 10−4 | −0.66 | 0.24 | 2.8 × 10−4 | −1.11 | 0.28 | 9.7 × 10−7 | 0.01 | 0.01 | 1.00 | 0.01 | 0.02 |
| 3-phenylpropionate (hydrocinnamate) | −0.88 | 0.36 | 0.02 | −1.18 | 0.47 | 1.3 × 10−3 | −2.57 | 0.79 | 1.0 × 10−7 | 1.5 × 10−4 | 3.8 × 10−4 | 0.67 | 2.0 × 10−4 | 2.4 × 10−3 |
| catechol sulfate | −0.26 | 0.29 | 0.45 | 0.05 | 0.42 | 0.87 | 0.80 | 0.31 | 0.06 | 0.31 | 0.12 | 0.82 | 0.28 | 0.60 |
| 2-hydroxyphenylacetate | −0.12 | 0.14 | 0.38 | 0.05 | 0.12 | 0.79 | 0.16 | 0.13 | 0.36 | 0.40 | 0.14 | 0.67 | 0.37 | 0.86 |
| 3-ethylphenylsulfate | −0.09 | 0.28 | 0.12 | −0.26 | 0.20 | 0.07 | −0.43 | 0.14 | 0.01 | 0.27 | 0.12 | 1.00 | 0.32 | 0.36 |
| p-cresol sulfate | 0.28 | 0.18 | 0.02 | 0.31 | 0.22 | 0.72 | −0.36 | 0.37 | 0.25 | 0.22 | 0.10 | 0.91 | 0.21 | 0.40 |
| phenol sulfate | −0.21 | 0.22 | 0.32 | −0.03 | 0.21 | 0.98 | −0.32 | 0.17 | 0.08 | 0.58 | 0.17 | 0.72 | 0.97 | 0.59 |
| 3-(4-hydroxyphenyl)propionate | 0.63 | 0.44 | 0.69 | −1.29 | 0.81 | 0.44 | −1.30 | 2.05 | 0.34 | 0.36 | 0.13 | 0.71 | 0.79 | 0.33 |
| phenylacetylglutamine | 0.75 | 0.24 | 2.9 × 10−3 | 0.23 | 0.19 | 0.09 | −0.36 | 0.66 | 0.61 | 0.32 | 0.13 | 0.57 | 0.31 | 0.88 |
| phenylacetate | 0.73 | 0.33 | 0.03 | 0.07 | 0.23 | 0.56 | −1.48 | 1.04 | 0.19 | 0.05 | 0.03 | 0.70 | 0.04 | 0.21 |
| phenylacetylglycine | 0.63 | 0.26 | 0.02 | 0.13 | 0.24 | 0.34 | −0.60 | 0.49 | 0.43 | 0.15 | 0.08 | 0.87 | 0.14 | 0.33 |
| phenyllactate (PLA) | −0.46 | 0.27 | 0.01 | −0.20 | 0.09 | 0.03 | 0.09 | 0.18 | 0.64 | 0.08 | 0.05 | 0.90 | 0.09 | 0.19 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hall, J.A.; Jackson, M.I.; Farace, G.; Yerramilli, M.; Jewell, D.E. Influence of Dietary Ingredients on Lean Body Percent, Uremic Toxin Concentrations, and Kidney Function in Senior-Adult Cats. Metabolites 2019, 9, 238. https://doi.org/10.3390/metabo9100238
Hall JA, Jackson MI, Farace G, Yerramilli M, Jewell DE. Influence of Dietary Ingredients on Lean Body Percent, Uremic Toxin Concentrations, and Kidney Function in Senior-Adult Cats. Metabolites. 2019; 9(10):238. https://doi.org/10.3390/metabo9100238
Chicago/Turabian StyleHall, Jean A., Matthew I. Jackson, Giosi Farace, Maha Yerramilli, and Dennis E. Jewell. 2019. "Influence of Dietary Ingredients on Lean Body Percent, Uremic Toxin Concentrations, and Kidney Function in Senior-Adult Cats" Metabolites 9, no. 10: 238. https://doi.org/10.3390/metabo9100238
APA StyleHall, J. A., Jackson, M. I., Farace, G., Yerramilli, M., & Jewell, D. E. (2019). Influence of Dietary Ingredients on Lean Body Percent, Uremic Toxin Concentrations, and Kidney Function in Senior-Adult Cats. Metabolites, 9(10), 238. https://doi.org/10.3390/metabo9100238




