Metabolic Perturbations in a Bacillus subtilis clpP Mutant during Glucose Starvation
Abstract
1. Introduction
2. Results
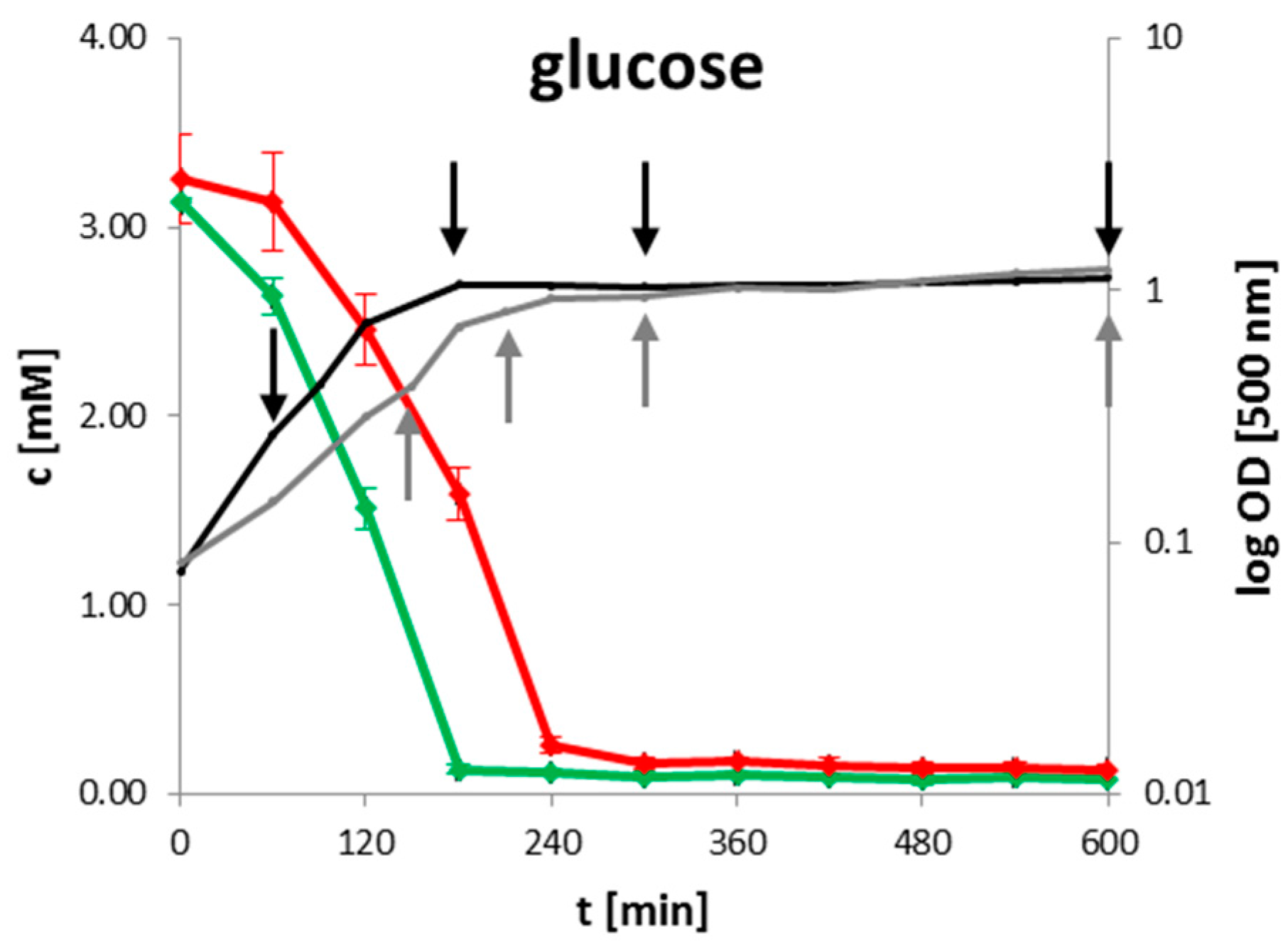
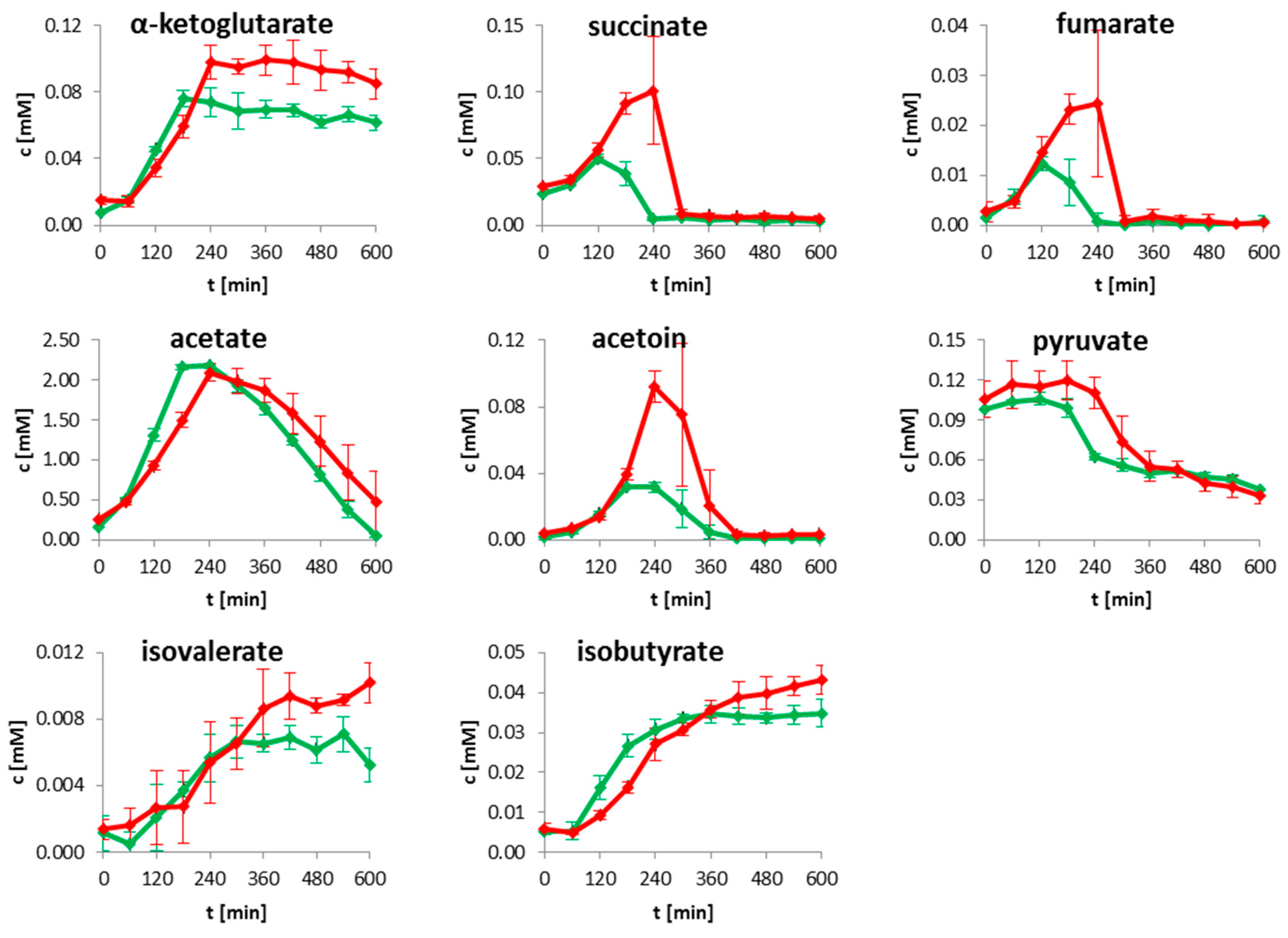
2.1. Extracellular Metabolites
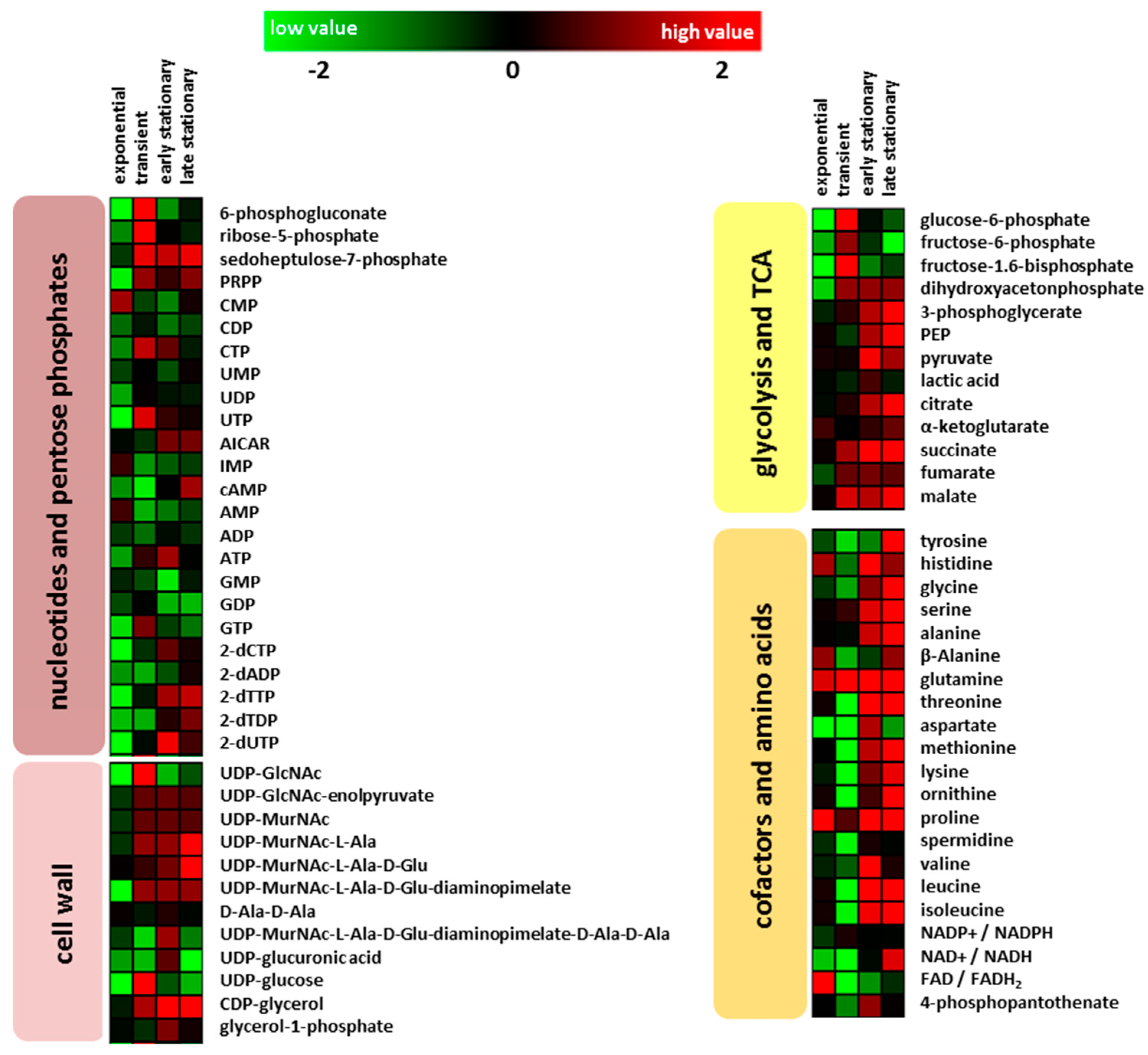
2.2. Intracellular Metabolites
2.2.1. Glycolysis and Tricarboxylic Acid Cycle
2.2.2. Amino Acids and Nucleotides
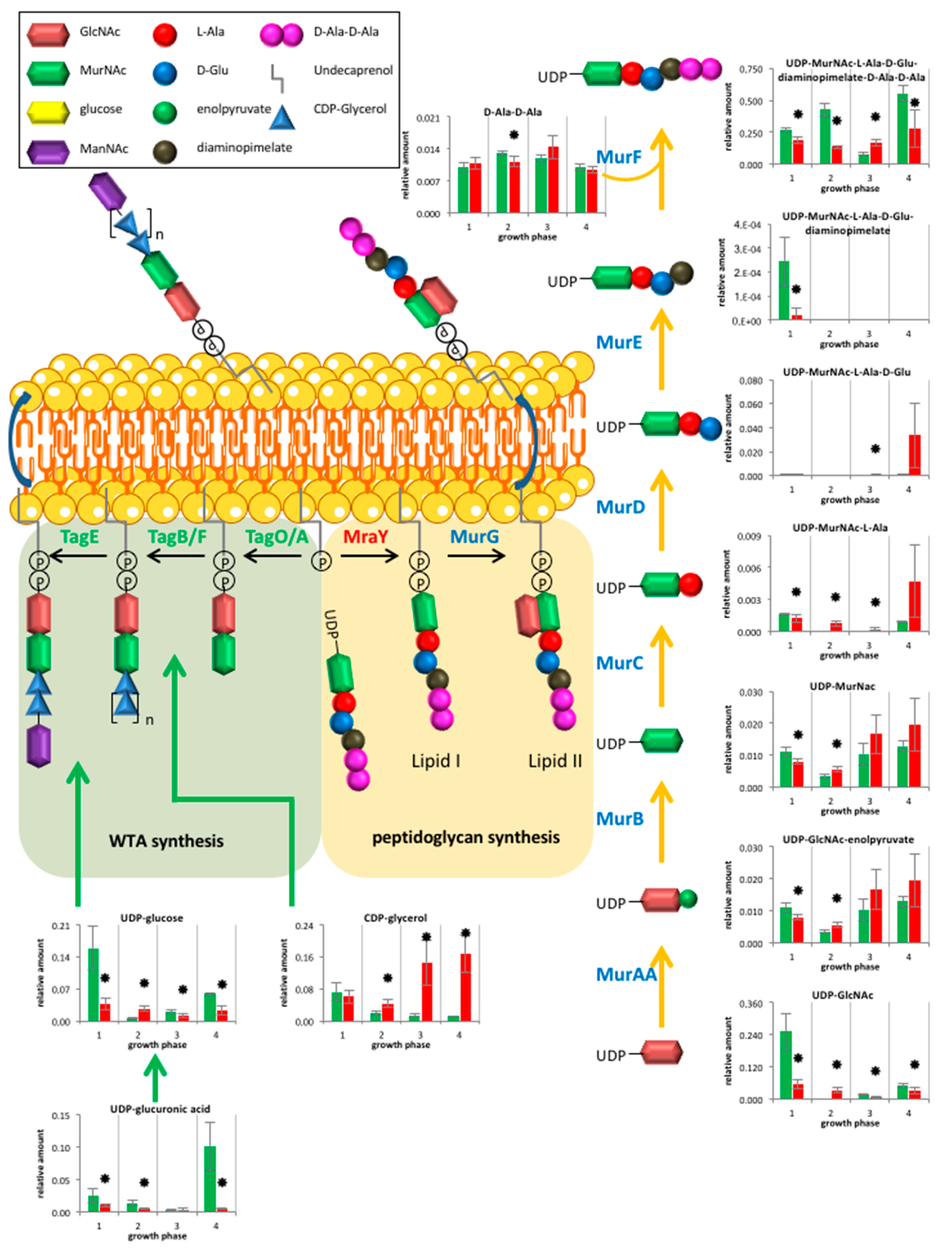
2.2.3. Cell Wall Metabolism
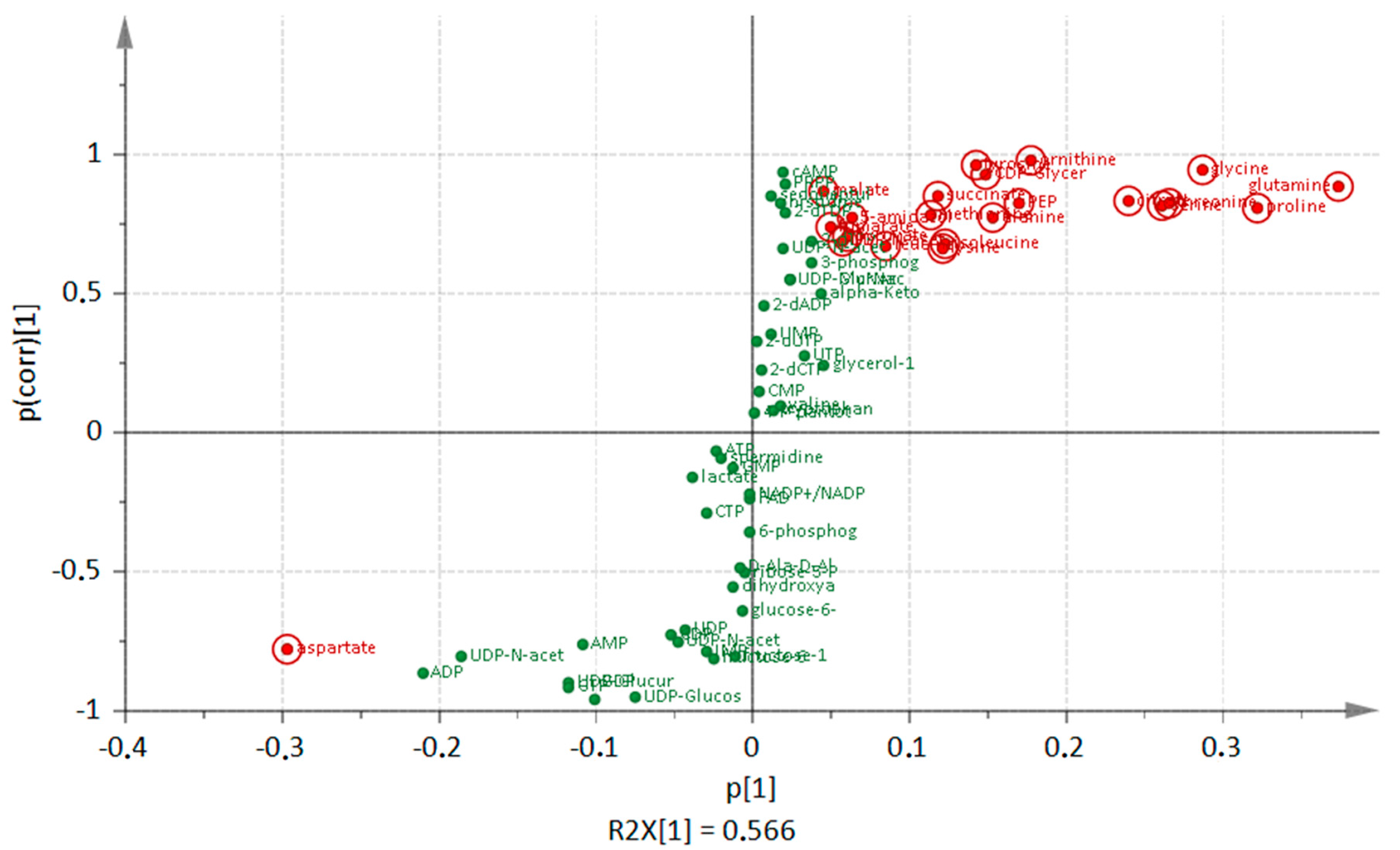
3. Discussion
3.1. Comparison of Metabolic Alterations in Wild Type and clpP Mutant Cells
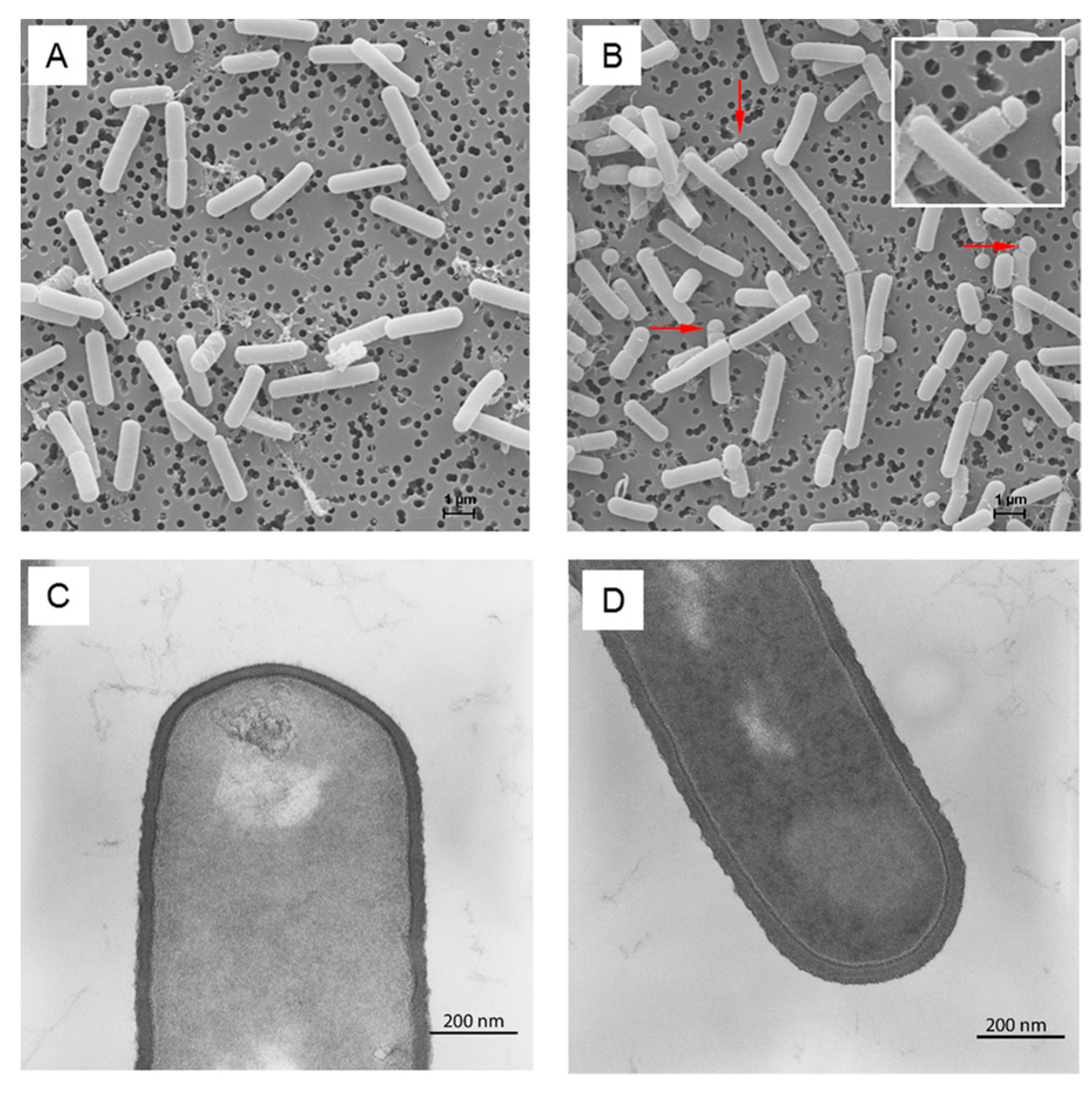
3.2. Cell Wall Metabolism and Morphology
4. Materials and Methods
4.1. Cultivation
4.2. Sampling of Extracellular Metabolites
4.3. 1H-NMR Spectroscopic Analysis of Extracellular Metabolites
4.4. Sampling of Intracellular Metabolites
4.5. GC-MS Analysis of Intracellular Metabolites
4.6. HPLC-MC Analysis of Intracellular Metabolites
4.7. Statistical Analysis and Visualization
4.8. Transmission Electron Microscopy
4.9. Scanning Electron Microscopy
4.10. Disk Diffusion Experiments
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lupas, A.; Flanagan, J.M.; Tamura, T.; Baumeister, W. Self-compartmentalizing proteases. Trends Biochem. Sci. 1997, 22, 399–404. [Google Scholar] [CrossRef]
- Young, J.C.; Agashe, V.R.; Siegers, K.; Hartl, F.U. Pathways of chaperone-mediated protein folding in the cytosol. Nat. Rev. Mol. Cell Biol. 2004, 5, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Kock, H.; Gerth, U.; Hecker, M. The ClpP peptidase is the major determinant of bulk protein turnover in Bacillus subtilis. J. Bacteriol. 2004, 186, 5856–5864. [Google Scholar] [CrossRef] [PubMed]
- Kruger, E.; Witt, E.; Ohlmeier, S.; Hanschke, R.; Hecker, M. The Clp proteases of Bacillus subtilis are directly involved in degradation of misfolded proteins. J. Bacteriol. 2000, 182, 3259–3265. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.G.; Kim, M.; Song, H. Structural insights into the conformational diversity of ClpP from Bacillus subtilis. Mol. Cell. 2011, 32, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Gerth, U.; Kock, H.; Kusters, I.; Michalik, S.; Switzer, R.L.; Hecker, M. Clp-dependent proteolysis down-regulates central metabolic pathways in glucose-starved Bacillus subtilis. J. Bacteriol. 2008, 190, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Derre, I.; Rapoport, G.; Devine, K.; Rose, M.; Msadek, T. ClpE, a novel type of HSPhsp100 ATPase, is part of the CtsR heat shock regulon of Bacillus subtilis. Mol. Microbiol. 1999, 32, 581–593. [Google Scholar] [CrossRef] [PubMed]
- Gottesman, S. Proteolysis in bacterial regulatory circuits. Annu. Rev. Cell Dev. Biol. 2003, 19, 565–587. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.M.; Hartling, J.A.; Flanagan, J.M. The structure of ClpP at 2.3 angstrom resolution suggests a model for ATP-dependent proteolysis. Cell 1997, 91, 447–456. [Google Scholar] [CrossRef]
- Kirstein, J.; Strahl, H.; Moliere, N.; Hamoen, L.W.; Turgay, K. Localization of general and regulatory proteolysis in Bacillus subtilis cells. Mol. Microbiol. 2008, 70, 682–694. [Google Scholar] [CrossRef] [PubMed]
- Kramer, G.; Boehringer, D.; Ban, N.; Bukau, B. The ribosome as a platform for co-translational processing, folding and targeting of newly synthesized proteins. Nat. Struct. Mol. Biol. 2009, 16, 589–597. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Garsin, D.A.; Losick, R. Self-reinforcing activation of a cell-specific transcription factor by proteolysis of an anti-sigma factor in B. Subtilis. Mol. Cell 2001, 8, 873–883. [Google Scholar] [CrossRef]
- Turgay, K.; Hahn, J.; Burghoorn, J.; Dubnau, D. Competence in Bacillus subtilis is controlled by regulated proteolysis of a transcription factor. EMBO J. 1998, 17, 6730–6738. [Google Scholar] [PubMed]
- Msadek, T.; Dartois, V.; Kunst, F.; Herbaud, M.L.; Denizot, F.; Rapoport, G. ClpP of Bacillus subtilis is required for competence development, motility, degradative enzyme synthesis, growth at high temperature and sporulation. Mol. Microbiol. 1998, 27, 899–914. [Google Scholar] [CrossRef] [PubMed]
- Boyle-Vavra, S.; Jones, M.; Gourley, B.L.; Holmes, M.; Ruf, R.; Balsam, A.R.; Boulware, D.R.; Kline, S.; Jawahir, S.; Devries, A.; et al. Comparative genome sequencing of an isogenic pair of USA800 clinical methicillin-resistant Staphylococcus aureus isolates obtained before and after daptomycin treatment failure. Antimicrob. Agents Chemother. 2011, 55, 2018–2025. [Google Scholar] [CrossRef] [PubMed]
- Butala, M.; Zgur-Bertok, D.; Busby, S.J.W. The bacterial LexA transcriptional repressor. Cell. Mol. Life Sci. 2009, 66, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Frees, D.; Qazi, S.N.A.; Hill, P.J.; Ingmer, H. Alternative roles of ClpX and ClpP in Staphylococcus aureus stress tolerance and virulence. Mol. Microbiol. 2003, 48, 1565–1578. [Google Scholar] [CrossRef] [PubMed]
- Kock, H.; Gerth, U.; Hecker, M. MurAA, catalysing the first committed step in peptidoglycan biosynthesis, is a target of Clp-dependent proteolysis in Bacillus subtilis. Mol. Microbiol. 2004, 51, 1087–1102. [Google Scholar] [CrossRef] [PubMed]
- Michalik, S.; Bernhardt, J.; Otto, A.; Moche, M.; Becher, D.; Meyer, H.; Lalk, M.; Schurmann, C.; Schluter, R.; Kock, H.; et al. Life and death of proteins: A case study of glucose-starved Staphylococcus aureus. Mol. Cell Proteom. 2012, 11, 558–570. [Google Scholar] [CrossRef] [PubMed]
- Frees, D.; Gerth, U.; Ingmer, H. Clp chaperones and proteases are central in stress survival, virulence and antibiotic resistance of Staphylococcus aureus. Int. J. Med. Microbiol. 2014, 304, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Renna, M.C.; Najimudin, N.; Winik, L.R.; Zahler, S.A. Regulation of the Bacillus subtilis AlsS, AlsD, and AlsR genes involved in post-exponential-phase production of acetoin. J. Bacteriol. 1993, 175, 3863–3875. [Google Scholar] [CrossRef] [PubMed]
- Schilling, O.; Frick, O.; Herzberg, C.; Ehrenreich, A.; Heinzle, E.; Wittmann, C.; Stulke, J. Transcriptional and metabolic responses of Bacillus subtilis to the availability of organic acids: Transcription regulation is important but not sufficient to account for metabolic adaptation. Appl. Environ. Microbiol. 2007, 73, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.O.; Bignon, J.; Rapoport, G.; Debarbouille, M. Regulation of the acetoin catabolic pathway is controlled by sigma l in Bacillus subtilis. J. Bacteriol. 2001, 183, 2497–2504. [Google Scholar] [PubMed]
- Zhang, Y.; Agrebi, R.; Bellows, L.E.; Collet, J.F.; Kaever, V.; Grundling, A. Evolutionary adaptation of the essential tRNA methyltransferase TrmD to the signaling molecule 3′,5′-cAMP in bacteria. J. Biol. Chem. 2017, 292, 313–327. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.S.; Sweitzer, T.D.; Dixon, J.E.; Kent, C. Expression, purification, and characterization of CTP: Glycerol-3-phosphate cytidylyltransferase from Bacillus subtilis. J. Biol. Chem. 1993, 268, 16648–16654. [Google Scholar] [PubMed]
- Swoboda, J.G.; Campbell, J.; Meredith, T.C.; Walker, S. Wall teichoic acid function, biosynthesis, and inhibition. Chembiochem Eur. J. Chem. Biol. 2010, 11, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Gerth, U.; Kruger, E.; Derre, I.; Msadek, T.; Hecker, M. Stress induction of the Bacillus subtilis ClpP gene encoding a homologue of the proteolytic component of the Clp protease and the involvement of ClpP and ClpX in stress tolerance. Mol. Microbiol. 1998, 28, 787–802. [Google Scholar] [CrossRef] [PubMed]
- Brill, J.; Hoffmann, T.; Bleisteiner, M.; Bremer, E. Osmotically controlled synthesis of the compatible solute proline is critical for cellular defense of Bacillus subtilis against high osmolarity. J. Bacteriol. 2011, 193, 5335–5346. [Google Scholar] [CrossRef] [PubMed]
- Ignatova, Z.; Gierasch, L.M. Inhibition of protein aggregation in vitro and in vivo by a natural osmoprotectant. Proc. Natl. Acad. Sci. USA 2006, 103, 13357–13361. [Google Scholar] [CrossRef] [PubMed]
- D'Elia, M.A.; Millar, K.E.; Beveridge, T.J.; Brown, E.D. Wall teichoic acid polymers are dispensable for cell viability in Bacillus subtilis. J. Bacteriol. 2006, 188, 8313–8316. [Google Scholar] [CrossRef] [PubMed]
- Lunderberg, J.M.; Liszewski Zilla, M.; Missiakas, D.; Schneewind, O. Bacillus anthracis tagO is required for vegetative growth and secondary cell wall polysaccharide synthesis. J. Bacteriol. 2015, 197, 3511–3520. [Google Scholar] [CrossRef] [PubMed]
- Siebring, J.; Elema, M.J.H.; Vega, F.D.; Kovacs, A.T.; Haccou, P.; Kuipers, O.P. Repeated triggering of sporulation in Bacillus subtilis selects against a protein that affects the timing of cell division. ISME J. 2014, 8, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Chien, A.C.; Hill, N.S.; Levin, P.A. Cell size control in bacteria. Curr. Biol. 2012, 22, R340–R349. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.J.; Blackman, S.A.; Foster, S.J. Autolysins of Bacillus subtilis: Multiple enzymes with multiple functions. Microbiology 2000, 146, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Rubio, A.; Jayaswal, R.K.; Silverman, J.A.; Wilkinson, B.J. Additional routes to Staphylococcus aureus daptomycin resistance as revealed by comparative genome sequencing, transcriptional profiling, and phenotypic studies. PLoS ONE 2013, 8, e58469. [Google Scholar] [CrossRef] [PubMed]
- Vanalstyne, D.; Simon, M.I. Division mutants of Bacillus-subtilis—Isolation and PBS1 transduction of division-specific markers. J. Bacteriol. 1971, 108, 1366–1379. [Google Scholar]
- van Baarle, S.; Bramkamp, M. The MinCDJ system in Bacillus subtilis prevents minicell formation by promoting divisome disassembly. PLoS ONE 2010, 5. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.Y.; Michalik, S.; Varming, A.N.; Andersen, J.H.; Albrecht, D.; Jelsbak, L.; Krieger, S.; Ohlsen, K.; Hecker, M.; Gerth, U.; et al. Trapping and proteomic identification of cellular substrates of the ClpP protease in Staphylococcus aureus. J. Proteome Res. 2013, 12, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Sass, P.; Josten, M.; Famulla, K.; Schiffer, G.; Sahl, H.G.; Hamoen, L.; Brotz-Oesterhelt, H. Antibiotic acyldepsipeptides activate ClpP peptidase to degrade the cell division protein Ftsz. Proc. Natl. Acad. Sci. USA 2011, 108, 17474–17479. [Google Scholar] [CrossRef] [PubMed]
- Harwood, C.R.; Cutting, S.M. Molecular Biological Methods for Bacillus; Wiley: Chichester, NY, USA, 1990. [Google Scholar]
- Meyer, H.; Weidmann, H.; Lalk, M. Methodological approaches to help unravel the intracellular metabolome of Bacillus subtilis. Microb. Cell Fact. 2013, 12, 69. [Google Scholar] [CrossRef] [PubMed]
- Dorries, K.; Lalk, M. Metabolic footprint analysis uncovers strain specific overflow metabolism and D-isoleucine production of Staphylococcus aureus COL and HG001. PLoS ONE 2013, 8, e81500. [Google Scholar] [CrossRef] [PubMed]
- Dorries, K.; Schlueter, R.; Lalk, M. Impact of antibiotics with various target sites on the metabolome of Staphylococcus aureus. Antimicrob. Agents Chemother. 2014, 58, 7151–7163. [Google Scholar] [CrossRef] [PubMed]
- Gierok, P.; Harms, M.; Richter, E.; Hildebrandt, J.P.; Lalk, M.; Mostertz, J.; Hochgrafe, F. Staphylococcus aureus alpha-toxin mediates general and cell type-specific changes in metabolite concentrations of immortalized human airway epithelial cells. PLoS ONE 2014, 9, e94818. [Google Scholar] [CrossRef] [PubMed]
- Wishart, D.S.; Jewison, T.; Guo, A.C.; Wilson, M.; Knox, C.; Liu, Y.; Djoumbou, Y.; Mandal, R.; Aziat, F.; Dong, E.; et al. HMDB 3.0—The human metabolome database in 2013. Nucleic Acids Res. 2013, 41, D801–D807. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, D.E. The energy charge of the adenylate pool as a regulatory parameter. Interaction with feedback modifiers. Biochemistry 1968, 7, 4030–4034. [Google Scholar] [CrossRef] [PubMed]
- Klukas, C.; Schreiber, F. Integration of -omics data and networks for biomedical research with vanted. J. Int. Bioinform. 2010, 7, 112. [Google Scholar]
- Saeed, A.I.; Sharov, V.; White, J.; Li, J.; Liang, W.; Bhagabati, N.; Braisted, J.; Klapa, M.; Currier, T.; Thiagarajan, M.; et al. TM4: A free, open-source system for microarray data management and analysis. BioTechniques 2003, 34, 374–378. [Google Scholar] [PubMed]
- Cao, M.; Bernat, B.A.; Wang, Z.; Armstrong, R.N.; Helmann, J.D. FosB, a cysteine-dependent fosfomycin resistance protein under the control of sigma(W), an extracytoplasmic-function sigma factor in Bacillus subtilis. J. Bacteriol. 2001, 183, 2380–2383. [Google Scholar] [CrossRef] [PubMed]
- Pooley, H.M.; Karamata, D. Incorporation of [2-3H]glycerol into cell surface components of Bacillus subtilis 168 and thermosensitive mutants affected in wall teichoic acid synthesis: Effect of tunicamycin. Microbiology 2000, 146, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Farha, M.A.; Leung, A.; Sewell, E.W.; D'Elia, M.A.; Allison, S.E.; Ejim, L.; Pereira, P.M.; Pinho, M.G.; Wright, G.D.; Brown, E.D. Inhibition of WTA synthesis blocks the cooperative action of PBPs and sensitizes MRSA to β-lactams. ACS Chem. Biol. 2013, 8, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Adimpong, D.B.; Sorensen, K.I.; Thorsen, L.; Stuer-Lauridsen, B.; Abdelgadir, W.S.; Nielsen, D.S.; Derkx, P.M.; Jespersen, L. Antimicrobial susceptibility of Bacillus strains isolated from primary starters for African traditional bread production and characterization of the bacitracin operon and bacitracin biosynthesis. Appl. Environ. Microbiol. 2012, 78, 7903–7914. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schultz, D.; Schlüter, R.; Gerth, U.; Lalk, M. Metabolic Perturbations in a Bacillus subtilis clpP Mutant during Glucose Starvation. Metabolites 2017, 7, 63. https://doi.org/10.3390/metabo7040063
Schultz D, Schlüter R, Gerth U, Lalk M. Metabolic Perturbations in a Bacillus subtilis clpP Mutant during Glucose Starvation. Metabolites. 2017; 7(4):63. https://doi.org/10.3390/metabo7040063
Chicago/Turabian StyleSchultz, Daniel, Rabea Schlüter, Ulf Gerth, and Michael Lalk. 2017. "Metabolic Perturbations in a Bacillus subtilis clpP Mutant during Glucose Starvation" Metabolites 7, no. 4: 63. https://doi.org/10.3390/metabo7040063
APA StyleSchultz, D., Schlüter, R., Gerth, U., & Lalk, M. (2017). Metabolic Perturbations in a Bacillus subtilis clpP Mutant during Glucose Starvation. Metabolites, 7(4), 63. https://doi.org/10.3390/metabo7040063





