Crosstalk Between Skeletal Muscle and Proximal Connective Tissues in Lipid Dysregulation in Obesity and Type 2 Diabetes
Abstract
1. Introduction
2. Physiological Regulation of Lipid Metabolism at the Skeletal Muscle–Connective Tissue Interface
2.1. Healthy Skeletal Muscle Lipid Uptake, Oxidation, and Storage
2.2. Lipid Droplet Dynamics in Skeletal Muscle
2.3. Regulation of Lipid Metabolism in Skeletal Muscle
2.4. Adipose Tissue Depots and Adipocyte Phenotypes in Physiological Lipid Metabolism
2.5. Physiological Roles of Bone in Lipid Metabolism
2.6. Emerging Role of Fascia in Local Adipogenesis and Lipid Metabolic Regulation
3. Lipid Dysregulation in Skeletal Muscle and Local Connective Tissues in Obesity and T2DM
3.1. Pathological Alterations in Skeletal Muscle Lipid Handling in Obesity and T2DM
3.2. Adipose Tissue Remodeling and Depot-Specific Dysfunction in Obesity and T2DM
3.3. Bone Marrow Adiposity and Lipid Metabolic Dysfunction in Obesity and T2DM
3.4. Fascia and Fibro-Adipogenic Remodeling in Obesity and T2DM
4. Molecular Mediators and Mechanisms of Local Crosstalk Driving Lipid Dysregulation and Insulin Resistance
4.1. Interleukins in Local Muscle–Adipose Crosstalk During Lipid Dysregulation
4.2. Other Myokines and Muscle-Derived Factors in Local Lipid Dysregulation
4.3. Leptin and Adiponectin in Local Lipid Dysregulation and Adipose–Muscle Crosstalk
4.4. Other Adipokines Involved in Local Lipid Metabolism and Insulin Sensitivity
4.5. Lipid Intermediates and Signaling Lipids Shaping Local Metabolic Communication
4.6. miRNAs and Exosomes in Local Adipose–Muscle Crosstalk
4.7. Compartment-Specific Adipose Secretomes Influence Local Muscle and Stromal Metabolism
4.8. Roles of Myokines, Osteokines, and Adipokines in Muscle–Bone–Adipose Metabolic Interactions
5. Conclusions and Future Directions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 3-HIB | 3-hydroxyisobutyrate |
| AMPK | AMP-activated protein kinase |
| ATGL | adipose triglyceride lipase |
| BAIBA | β-aminoisobutyric acid |
| BAT | brown adipose tissue |
| BCAA | branched-chain amino acid |
| DAGs | diacylglycerols |
| ER | endoplasmic reticulum |
| FA | fatty acid |
| FABP4 | Fatty acid-binding protein 4 |
| FAPs | fibro-adipogenic progenitors |
| FATP4 | Fatty Acid Transport Protein 4 |
| FFA | free fatty acid |
| FGF2 | fibroblast growth factor 2 |
| FGF21 | fibroblast growth factor 21 |
| GWAS | Genome-wide association studies |
| HFD | high-fat diet |
| HSL | hormone-sensitive lipase |
| IGF-1 | insulin-like growth factor 1 |
| IL-1 | interleukin-1 |
| IL-6 | interleukin-6 |
| IMAT | Intermuscular adipose tissue |
| IMCL | intramyocellular lipids |
| IRS-1 | insulin receptor substrate 1 |
| LCFA | long-chain fatty acid |
| LDs | lipid droplets |
| NEFA | non-esterified fatty acid |
| nPKC | novel protein kinase C |
| OCN | Osteocalcin |
| PAHSAs | palmitic acid esters of hydroxystearic acid |
| PGC1α | PPARγ-coactivator 1α |
| PLIN5 | perilipin 5 |
| PPAR | peroxisome proliferator-activated receptor |
| RANKL | receptor activator of nuclear factor κB ligand |
| RBP4 | Retinol-binding protein 4 |
| ROS | reactive oxygen species |
| SAT | subcutaneous adipose tissue |
| Sost | sclerostin |
| T2DM | type 2 diabetes mellitus |
| TAG | triacylglycerol |
| TNF-α | tumor necrosis factor-alpha |
| TRAF6 | TNF receptor-associated factor 6 |
| UCP1 | uncoupling protein 1 |
| ucOCN | undercarboxylated osteocalcin |
| VAT | visceral adipose tissue |
| WAT | white adipose tissue |
References
- Kerr, J.A.; Cini, K.I.; Francis, K.L.; Sawyer, S.M.; Azzopardi, P.S.; Patton, G.C.; Dhungel, B.; Jebasingh, F.K.; Abate, Y.H.; Abbas, N.; et al. Global, Regional, and National Prevalence of Adult Overweight and Obesity, 1990–2021, with Forecasts to 2050: A Forecasting Study for the Global Burden of Disease Study 2021. Lancet 2025, 405, 813–838. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Saeedi, P.; Karuranga, S.; Pinkepank, M.; Ogurtsova, K.; Duncan, B.B.; Stein, C.; Basit, A.; Chan, J.C.N.; Mbanya, J.C.; et al. IDF Diabetes Atlas: Global, Regional and Country-Level Diabetes Prevalence Estimates for 2021 and Projections for 2045. Diabetes Res. Clin. Pract. 2022, 183, 109119. [Google Scholar] [CrossRef] [PubMed]
- Blüher, M. Obesity: Global Epidemiology and Pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef]
- Van Herpen, N.A.; Schrauwen-Hinderling, V.B. Lipid Accumulation in Non-Adipose Tissue and Lipotoxicity. Physiol. Behav. 2008, 94, 231–241. [Google Scholar] [CrossRef]
- Umek, N.; Horvat, S.; Cvetko, E. Skeletal Muscle and Fiber Type-Specific Intramyocellular Lipid Accumulation in Obese Mice. Bosn. J. Basic. Med. Sci. 2021, 21, 729–737. [Google Scholar] [CrossRef]
- Yazıcı, D.; Demir, S.Ç.; Sezer, H. Insulin Resistance, Obesity, and Lipotoxicity. Adv. Exp. Med. Biol. 2024, 1460, 391–430. [Google Scholar] [CrossRef]
- Brøns, C.; Grunnet, L.G. Skeletal Muscle Lipotoxicity in Insulin Resistance and Type 2 Diabetes: A Causal Mechanism or an Innocent Bystander? Eur. J. Endocrinol. 2017, 176, R67–R78. [Google Scholar] [CrossRef] [PubMed]
- Kamrani, P.; Marston, G.; Arbor, T.C.; Jan, A. Anatomy, Connective Tissue; StatPearls: Petersburg, FL, USA, 2023. [Google Scholar]
- Weber, K.T.; Sun, Y.; Katwa, L.C.; Cleutjens, J.P.M. Connective Tissue: A Metabolic Entity? J. Mol. Cell Cardiol. 1995, 27, 107–120. [Google Scholar] [CrossRef]
- O’Rourke, R.W.; Metcalf, M.D.; White, A.E.; Madala, A.; Winters, B.R.; Maizlin, I.I.; Jobe, B.A.; Roberts, C.T.; Slifka, M.K.; Marks, D.L. Depot-Specific Differences in Inflammatory Mediators and a Role for NK Cells and IFN-γ in Inflammation in Human Adipose Tissue. Int. J. Obes. 2009, 33, 978. [Google Scholar] [CrossRef]
- Bigornia, S.J.; Farb, M.G.; Mott, M.M.; Hess, D.T.; Carmine, B.; Fiscale, A.; Joseph, L.; Apovian, C.M.; Gokce, N. Relation of Depot-Specific Adipose Inflammation to Insulin Resistance in Human Obesity. Nutr. Diabetes 2012, 2, e30. [Google Scholar] [CrossRef]
- Xiao, H.; Li, W.; Qin, Y.; Lin, Z.; Qian, C.; Wu, M.; Xia, Y.; Bai, J.; Geng, D. Crosstalk between Lipid Metabolism and Bone Homeostasis: Exploring Intricate Signaling Relationships. Research 2024, 7, 0447. [Google Scholar] [CrossRef]
- Jia, G.; Sowers, J.R. Increased Fibro-Adipogenic Progenitors and Intramyocellular Lipid Accumulation in Obesity-Related Skeletal Muscle Dysfunction. Diabetes 2019, 68, 18–20. [Google Scholar] [CrossRef]
- Uezumi, A.; Ito, T.; Morikawa, D.; Shimizu, N.; Yoneda, T.; Segawa, M.; Yamaguchi, M.; Ogawa, R.; Matev, M.M.; Miyagoe-Suzuki, Y.; et al. Fibrosis and Adipogenesis Originate from a Common Mesenchymal Progenitor in Skeletal Muscle. J. Cell Sci. 2011, 124, 3654–3664. [Google Scholar] [CrossRef] [PubMed]
- Jani, S.; Da Eira, D.; Hadday, I.; Bikopoulos, G.; Mohasses, A.; de Pinho, R.A.; Ceddia, R.B. Distinct Mechanisms Involving Diacylglycerol, Ceramides, and Inflammation Underlie Insulin Resistance in Oxidative and Glycolytic Muscles from High Fat-Fed Rats. Sci. Rep. 2021, 11, 19160. [Google Scholar] [CrossRef]
- Szendroedi, J.; Yoshimura, T.; Phielix, E.; Koliaki, C.; Marcucci, M.; Zhang, D.; Jelenik, T.; Müller, J.; Herder, C.; Nowotny, P.; et al. Role of Diacylglycerol Activation of PKCθ in Lipid-Induced Muscle Insulin Resistance in Humans. Proc. Natl. Acad. Sci. USA 2014, 111, 9597–9602. [Google Scholar] [CrossRef]
- Li, X.; Fang, L.; Zhou, R.; Yao, L.; Clayton, S.W.; Muscat, S.; Kamm, D.R.; Wang, C.; Liu, C.J.; Qin, L.; et al. Current Cutting-Edge Omics Techniques on Musculoskeletal Tissues and Diseases. Bone Res. 2025, 13, 59. [Google Scholar] [CrossRef]
- Liu, P.; Li, D.; Zhang, J.; He, M.; Li, Y.; Liu, R.; Li, M. Transcriptomic and Lipidomic Profiling of Subcutaneous and Visceral Adipose Tissues in 15 Vertebrates. Sci. Data 2023, 10, 453. [Google Scholar] [CrossRef]
- Umek, N.; Meznarič, M.; Šink, Ž.; Blagotinšek Cokan, K.; Prosenc Zmrzljak, U.; Horvat, S. In Situ Spatial Transcriptomic Analysis of Human Skeletal Muscle Using the Xenium Platform. Cell Tissue Res. 2025, 399, 291–302. [Google Scholar] [CrossRef]
- Kolnes, K.J.; Nilsen, E.T.F.; Brufladt, S.; Meadows, A.M.; Jeppesen, P.B.; Skattebo, Ø.; Johansen, E.I.; Birk, J.B.; Højlund, K.; Hingst, J.; et al. Effects of Seven Days’ Fasting on Physical Performance and Metabolic Adaptation during Exercise in Humans. Nat. Commun. 2025, 16, 122. [Google Scholar] [CrossRef] [PubMed]
- Naëgel, A.; Viallon, M.; Ratiney, H.; Nguyen, T.; Leporq, B.; Kennouche, D.; Grenier, T.; Grundler, F.; Mesnage, R.; Guy, J.M.; et al. Impact of Long-Term Fasting on Skeletal Muscle: Structure, Energy Metabolism and Function Using 31P/1H MRS and MRI. J. Cachexia Sarcopenia Muscle 2025, 16, e13773. [Google Scholar] [CrossRef] [PubMed]
- Katare, P.B.; Dalmao-Fernandez, A.; Mengeste, A.M.; Hamarsland, H.; Ellefsen, S.; Bakke, H.G.; Kase, E.T.; Thoresen, G.H.; Rustan, A.C. Energy Metabolism in Skeletal Muscle Cells from Donors with Different Body Mass Index. Front. Physiol. 2022, 13, 982842. [Google Scholar] [CrossRef] [PubMed]
- Schiaffino, S.; Chemello, F.; Reggiani, C. The Diversity of Skeletal Muscle Fiber Types. Cold Spring Harb. Perspect. Biol. 2025, 17, a041477. [Google Scholar] [CrossRef]
- Murgia, M.; Nogara, L.; Baraldo, M.; Reggiani, C.; Mann, M.; Schiaffino, S. Protein Profile of Fiber Types in Human Skeletal Muscle: A Single-Fiber Proteomics Study. Skelet. Muscle 2021, 11, 24. [Google Scholar] [CrossRef]
- Jang, C.; Oh, S.F.; Wada, S.; Rowe, G.C.; Liu, L.; Chan, M.C.; Rhee, J.; Hoshino, A.; Kim, B.; Ibrahim, A.; et al. A Branched-Chain Amino Acid Metabolite Drives Vascular Fatty Acid Transport and Causes Insulin Resistance. Nat. Med. 2016, 22, 421–426. [Google Scholar] [CrossRef]
- Hagberg, C.E.; Falkevall, A.; Wang, X.; Larsson, E.; Huusko, J.; Nilsson, I.; Van Meeteren, L.A.; Samen, E.; Lu, L.; Vanwildemeersch, M.; et al. Vascular Endothelial Growth Factor B Controls Endothelial Fatty Acid Uptake. Nature 2010, 464, 917–921. [Google Scholar] [CrossRef] [PubMed]
- Gemmink, A.; Daemen, S.; Brouwers, B.; Huntjens, P.R.; Schaart, G.; Moonen-Kornips, E.; Jörgensen, J.; Hoeks, J.; Schrauwen, P.; Hesselink, M.K.C. Dissociation of Intramyocellular Lipid Storage and Insulin Resistance in Trained Athletes and Type 2 Diabetes Patients; Involvement of Perilipin 5? J. Physiol. 2018, 596, 857–868. [Google Scholar] [CrossRef] [PubMed]
- Maunder, E.; Rothschild, J.A.; Fritzen, A.M.; Jordy, A.B.; Kiens, B.; Brick, M.J.; Leigh, W.B.; Chang, W.L.; Kilding, A.E. Skeletal Muscle Proteins Involved in Fatty Acid Transport Influence Fatty Acid Oxidation Rates Observed during Exercise. Pflug. Arch. 2023, 475, 1061. [Google Scholar] [CrossRef]
- Conte, M.; Armani, A.; Conte, G.; Serra, A.; Franceschi, C.; Mele, M.; Sandri, M.; Salvioli, S. Muscle-specific Perilipin2 Down-regulation Affects Lipid Metabolism and Induces Myofiber Hypertrophy. J. Cachexia Sarcopenia Muscle 2018, 10, 95. [Google Scholar] [CrossRef]
- Nakamura, N.K.; Tokunaga, D.S.; Ha, H.Y.; Polgar, N. The Exocyst Is Required for CD36 Fatty Acid Translocase Trafficking and Free Fatty Acid Uptake in Skeletal Muscle Cells. Cells 2022, 11, 2440. [Google Scholar] [CrossRef]
- Benninghoff, T.; Espelage, L.; Eickelschulte, S.; Zeinert, I.; Sinowenka, I.; Müller, F.; Schöndeling, C.; Batchelor, H.; Cames, S.; Zhou, Z.; et al. The RabGAPs TBC1D1 and TBC1D4 Control Uptake of Long-Chain Fatty Acids Into Skeletal Muscle via Fatty Acid Transporter SLC27A4/FATP4. Diabetes 2020, 69, 2281–2293. [Google Scholar] [CrossRef]
- Irshad, Z.; Lund, J.; Sillars, A.; Løvsletten, N.G.; Gharanei, S.; Salt, I.P.; Freeman, D.J.; Gill, J.M.R.; Thoresen, G.H.; Rustan, A.C.; et al. The Roles of DGAT1 and DGAT2 in Human Myotubes Are Dependent on Donor Patho-Physiological Background. FASEB J. 2023, 37, e23209. [Google Scholar] [CrossRef]
- Shaw, C.S.; Swinton, C.; Morales-Scholz, M.G.; McRae, N.; Erftemeyer, T.; Aldous, A.; Murphy, R.M.; Howlett, K.F. Impact of Exercise Training Status on the Fiber Type-Specific Abundance of Proteins Regulating Intramuscular Lipid Metabolism. J. Appl. Physiol. 2020, 128, 379–389. [Google Scholar] [CrossRef]
- Gemmink, A.; Daemen, S.; Kuijpers, H.J.H.; Schaart, G.; Duimel, H.; López-Iglesias, C.; van Zandvoort, M.A.M.J.; Knoops, K.; Hesselink, M.K.C. Super-Resolution Microscopy Localizes Perilipin 5 at Lipid Droplet-Mitochondria Interaction Sites and at Lipid Droplets Juxtaposing to Perilipin 2. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2018, 1863, 1423–1432. [Google Scholar] [CrossRef]
- Whytock, K.L.; Shepherd, S.O.; Wagenmakers, A.J.M.; Strauss, J.A. Hormone-Sensitive Lipase Preferentially Redistributes to Lipid Droplets Associated with Perilipin-5 in Human Skeletal Muscle during Moderate-Intensity Exercise. J. Physiol. 2018, 596, 2077–2090. [Google Scholar] [CrossRef]
- Daemen, S.; Gemmink, A.; Brouwers, B.; Meex, R.C.R.; Huntjens, P.R.; Schaart, G.; Moonen-Kornips, E.; Jörgensen, J.; Hoeks, J.; Schrauwen, P.; et al. Distinct Lipid Droplet Characteristics and Distribution Unmask the Apparent Contradiction of the Athlete’s Paradox. Mol. Metab. 2018, 17, 71–81. [Google Scholar] [CrossRef]
- Boeszoermenyi, A.; Nagy, H.M.; Arthanari, H.; Pillip, C.J.; Lindermuth, H.; Luna, R.E.; Wagner, G.; Zechner, R.; Zangger, K.; Oberer, M. Structure of a CGI-58 Motif Provides the Molecular Basis of Lipid Droplet Anchoring. J. Biol. Chem. 2015, 290, 26361–26372. [Google Scholar] [CrossRef] [PubMed]
- Vieira-Lara, M.A.; Bakker, B.M. The Paradox of Fatty-Acid β-Oxidation in Muscle Insulin Resistance: Metabolic Control and Muscle Heterogeneity. Biochim. Biophys. Acta BBA Mol. Basis Dis. 2024, 1870, 167172. [Google Scholar] [CrossRef] [PubMed]
- Sachs, S.; Zarini, S.; Kahn, D.E.; Harrison, K.A.; Perreault, L.; Phang, T.; Newsom, S.A.; Strauss, A.; Kerege, A.; Schoen, J.A.; et al. Intermuscular Adipose Tissue Directly Modulates Skeletal Muscle Insulin Sensitivity in Humans. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E866–E879. [Google Scholar] [CrossRef] [PubMed]
- Sparks, L.M.; Goodpaster, B.H.; Bergman, B.C. The Metabolic Significance of Intermuscular Adipose Tissue: Is IMAT a Friend or a Foe to Metabolic Health? Diabetes 2021, 70, 2457–2467. [Google Scholar] [CrossRef]
- Chrzanowski-Smith, O.J.; Edinburgh, R.M.; Smith, E.; Thomas, M.P.; Walhin, J.-P.; Koumanov, F.; Williams, S.; Betts, J.A.; Gonzalez, J.T. Resting Skeletal Muscle PNPLA2 (ATGL) and CPT1B Are Associated with Peak Fat Oxidation Rates in Men and Women but Do Not Explain Observed Sex Differences. Exp. Physiol. 2021, 106, 1208–1223. [Google Scholar] [CrossRef]
- Alsted, T.J.; Nybo, L.; Schweiger, M.; Fledelius, C.; Jacobsen, P.; Zimmermann, R.; Zechner, R.; Kiens, B. Adipose Triglyceride Lipase in Human Skeletal Muscle Is Upregulated by Exercise Training. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E445–E453. [Google Scholar] [CrossRef]
- Knudsen, J.G.; Bertholdt, L.; Joensen, E.; Lassen, S.B.; Hidalgo, J.; Pilegaard, H. Skeletal Muscle Interleukin-6 Regulates Metabolic Factors in IWAT during HFD and Exercise Training. Obes. Silver Spring 2015, 23, 1616–1624. [Google Scholar] [CrossRef]
- Pierce, J.R.; Maples, J.M.; Hickner, R.C. IL-15 Concentrations in Skeletal Muscle and Subcutaneous Adipose Tissue in Lean and Obese Humans: Local Effects of IL-15 on Adipose Tissue Lipolysis. Am. J. Physiol. Endocrinol. Metab. 2015, 308, E1131–E1139. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Huang, Q.; Tominaga, T.; Liu, C.; Suzuki, K. An 8-Week Ketogenic Diet Alternated Interleukin-6, Ketolytic and Lipolytic Gene Expression, and Enhanced Exercise Capacity in Mice. Nutrients 2018, 10, 1696. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Yu, H.; Liang, J.; Zhang, Q.; Sun, J.; Yang, H.; Yan, H.; Zhang, S.; Li, Y.; Jin, Y.; et al. Increased FGF-21 Improves Ectopic Lipid Deposition in the Liver and Skeletal Muscle. Nutrients 2024, 16, 1254. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xu, S.; Zhang, X.; Yi, Z.; Cichello, S. Skeletal Intramyocellular Lipid Metabolism and Insulin Resistance. Biophys. Rep. 2015, 1, 90–98. [Google Scholar] [CrossRef]
- Stanford, K.I.; Lynes, M.D.; Takahashi, H.; Baer, L.A.; Arts, P.J.; May, F.J.; Lehnig, A.C.; Middelbeek, R.J.W.; Richard, J.J.; So, K.; et al. 12,13-DiHOME: An Exercise-Induced Lipokine That Increases Skeletal Muscle Fatty Acid Uptake. Cell Metab. 2018, 27, 1111. [Google Scholar] [CrossRef]
- Lee, E.J.; Ahmad, S.S.; Lim, J.H.; Ahmad, K.; Shaikh, S.; Lee, Y.S.; Park, S.J.; Jin, J.O.; Lee, Y.H.; Choi, I. Interaction of Fibromodulin and Myostatin to Regulate Skeletal Muscle Aging: An Opposite Regulation in Muscle Aging, Diabetes, and Intracellular Lipid Accumulation. Cells 2021, 10, 2083. [Google Scholar] [CrossRef]
- Liu, X.; Yang, Y.; Shao, H.; Liu, S.; Niu, Y.; Fu, L. Globular Adiponectin Ameliorates Insulin Resistance in Skeletal Muscle by Enhancing the LKB1-Mediated AMPK Activation via SESN2. Sports Med. Health Sci. 2022, 5, 34–41. [Google Scholar] [CrossRef]
- Jung, T.W.; Park, H.S.; Choi, G.H.; Kim, D.; Lee, T. β-Aminoisobutyric Acid Attenuates LPS-Induced Inflammation and Insulin Resistance in Adipocytes through AMPK-Mediated Pathway. J. Biomed. Sci. 2018, 25, 27. [Google Scholar] [CrossRef]
- Kilpiö, T.; Skarp, S.; Perjés, Á.; Swan, J.; Kaikkonen, L.; Saarimäki, S.; Szokodi, I.; Penninger, J.M.; Szabó, Z.; Magga, J.; et al. Apelin Regulates Skeletal Muscle Adaptation to Exercise in a High-Intensity Interval Training Model. Am. J. Physiol. Cell Physiol. 2024, 326, C1437–C1450. [Google Scholar] [CrossRef]
- Fritzen, A.M.; Lundsgaard, A.M.; Kiens, B. Tuning Fatty Acid Oxidation in Skeletal Muscle with Dietary Fat and Exercise. Nat. Rev. Endocrinol. 2020, 16, 683–696. [Google Scholar] [CrossRef]
- O’Neill, H.M.; Lally, J.S.; Galic, S.; Thomas, M.; Azizi, P.D.; Fullerton, M.D.; Smith, B.K.; Pulinilkunnil, T.; Chen, Z.; Samaan, M.C.; et al. AMPK Phosphorylation of ACC2 Is Required for Skeletal Muscle Fatty Acid Oxidation and Insulin Sensitivity in Mice. Diabetologia 2014, 57, 1693–1702. [Google Scholar] [CrossRef]
- Kalenta, H.; Kilroe, S.P.; Romsdahl, T.B.; Marchant, E.D.; Maroto, R.; Linares, J.J.; Russell, W.K.; Rasmussen, B.B. Constitutively Active MTORC1 Signaling Modifies the Skeletal Muscle Metabolome and Lipidome Response to Exercise. J. Appl. Physiol. 2025, 138, 1173–1186. [Google Scholar] [CrossRef] [PubMed]
- Zizola, C.; Kennel, P.J.; Akashi, H.; Ji, R.; Castillero, E.; George, I.; Homma, S.; Schulze, P.C. Activation of PPARδ Signaling Improves Skeletal Muscle Oxidative Metabolism and Endurance Function in an Animal Model of Ischemic Left Ventricular Dysfunction. Am. J. Physiol. Heart Circ. Physiol. 2015, 308, H1078. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.S.; Ng, C.F.; Pang, B.P.S.; Hang, M.; Tse, M.C.L.; Iu, E.C.Y.; Ooi, X.C.; Yang, X.; Kim, J.K.; Lee, C.W.; et al. Exercise-Induced BDNF Promotes PPARδ-Dependent Reprogramming of Lipid Metabolism in Skeletal Muscle during Exercise Recovery. Sci. Signal 2024, 17, eadh2783. [Google Scholar] [CrossRef] [PubMed]
- Frigolet, M.E.; Gutiérrez-Aguilar, R. The Colors of Adipose Tissue. Gac. Med. Mex. 2020, 156, 142–149. [Google Scholar] [CrossRef]
- Masoodi, M.; Kuda, O.; Rossmeisl, M.; Flachs, P.; Kopecky, J. Lipid Signaling in Adipose Tissue: Connecting Inflammation & Metabolism. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2015, 1851, 503–518. [Google Scholar] [CrossRef]
- Ibrahim, M.M. Subcutaneous and Visceral Adipose Tissue: Structural and Functional Differences. Obes. Rev. 2010, 11, 11–18. [Google Scholar] [CrossRef]
- Kershaw, E.E.; Flier, J.S. Adipose Tissue as an Endocrine Organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556. [Google Scholar] [CrossRef]
- Singh, A.K.; Aryal, B.; Chaube, B.; Rotllan, N.; Varela, L.; Horvath, T.L.; Suárez, Y.; Fernández-Hernando, C. Brown Adipose Tissue Derived ANGPTL4 Controls Glucose and Lipid Metabolism and Regulates Thermogenesis. Mol. Metab. 2018, 11, 59–69. [Google Scholar] [CrossRef]
- Campderrós, L.; Moure, R.; Cairó, M.; Gavaldà-Navarro, A.; Quesada-López, T.; Cereijo, R.; Giralt, M.; Villarroya, J.; Villarroya, F. Brown Adipocytes Secrete GDF15 in Response to Thermogenic Activation. Obesity 2019, 27, 1606–1616. [Google Scholar] [CrossRef]
- Quesada-López, T.; Cereijo, R.; Turatsinze, J.V.; Planavila, A.; Cairó, M.; Gavaldà-Navarro, A.; Peyrou, M.; Moure, R.; Iglesias, R.; Giralt, M.; et al. The Lipid Sensor GPR120 Promotes Brown Fat Activation and FGF21 Release from Adipocytes. Nat. Commun. 2016, 7, 13479. [Google Scholar] [CrossRef]
- Stanford, K.I.; Middelbeek, R.J.W.; Townsend, K.L.; An, D.; Nygaard, E.B.; Hitchcox, K.M.; Markan, K.R.; Nakano, K.; Hirshman, M.F.; Tseng, Y.H.; et al. Brown Adipose Tissue Regulates Glucose Homeostasis and Insulin Sensitivity. J. Clin. Investig. 2013, 123, 215–223. [Google Scholar] [CrossRef]
- Rosenwald, M.; Perdikari, A.; Rülicke, T.; Wolfrum, C. Bi-Directional Interconversion of Brite and White Adipocytes. Nat. Cell Biol. 2013, 15, 659–667. [Google Scholar] [CrossRef]
- Wu, J.; Boström, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G.; et al. Beige Adipocytes Are a Distinct Type of Thermogenic Fat Cell in Mouse and Human. Cell 2012, 150, 366–376. [Google Scholar] [CrossRef]
- Altshuler-Keylin, S.; Shinoda, K.; Hasegawa, Y.; Ikeda, K.; Hong, H.; Kang, Q.; Yang, Y.; Perera, R.M.; Debnath, J.; Kajimura, S. Beige Adipocyte Maintenance Is Regulated by Autophagy-Induced Mitochondrial Clearance. Cell Metab. 2016, 24, 402–419. [Google Scholar] [CrossRef]
- O’Mara, A.E.; Johnson, J.W.; Linderman, J.D.; Brychta, R.J.; McGehee, S.; Fletcher, L.A.; Fink, Y.A.; Kapuria, D.; Cassimatis, T.M.; Kelsey, N.; et al. Chronic Mirabegron Treatment Increases Human Brown Fat, HDL Cholesterol, and Insulin Sensitivity. J. Clin. Investig. 2020, 130, 2209–2219. [Google Scholar] [CrossRef] [PubMed]
- Blondin, D.P.; Nielsen, S.; Kuipers, E.N.; Severinsen, M.C.; Jensen, V.H.; Miard, S.; Jespersen, N.Z.; Kooijman, S.; Boon, M.R.; Fortin, M.; et al. Human Brown Adipocyte Thermogenesis Is Driven by Β2-AR Stimulation. Cell Metab. 2020, 32, 287–300.e7. [Google Scholar] [CrossRef] [PubMed]
- Cero, C.; Lea, H.J.; Zhu, K.Y.; Shamsi, F.; Tseng, Y.H.; Cypess, A.M. Β3-Adrenergic Receptors Regulate Human Brown/Beige Adipocyte Lipolysis and Thermogenesis. JCI Insight 2021, 6, e139160. [Google Scholar] [CrossRef] [PubMed]
- Wibmer, A.G.; Becher, T.; Eljalby, M.; Crane, A.; Andrieu, P.C.; Jiang, C.S.; Vaughan, R.; Schöder, H.; Cohen, P. Brown Adipose Tissue Is Associated with Healthier Body Fat Distribution and Metabolic Benefits Independent of Regional Adiposity. Cell Rep. Med. 2021, 2, 100332. [Google Scholar] [CrossRef]
- Becher, T.; Palanisamy, S.; Kramer, D.J.; Eljalby, M.; Marx, S.J.; Wibmer, A.G.; Butler, S.D.; Jiang, C.S.; Vaughan, R.; Schöder, H.; et al. Brown Adipose Tissue Is Associated with Cardiometabolic Health. Nat. Med. 2021, 27, 58–65. [Google Scholar] [CrossRef]
- Karsenty, G.; Mera, P. Molecular Bases of the Crosstalk between Bone and Muscle. Bone 2018, 115, 43–49. [Google Scholar] [CrossRef]
- Mera, P.; Laue, K.; Ferron, M.; Confavreux, C.; Wei, J.; Galán-Díez, M.; Lacampagne, A.; Mitchell, S.J.; Mattison, J.A.; Chen, Y.; et al. Osteocalcin Signaling in Myofibers Is Necessary and Sufficient for Optimum Adaptation to Exercise. Cell Metab. 2016, 23, 1078. [Google Scholar] [CrossRef]
- Delgado-Calle, J.; Bellido, T. New Insights Into the Local and Systemic Functions of Sclerostin: Regulation of Quiescent Bone Lining Cells and Beige Adipogenesis in Peripheral Fat Depots. J. Bone Miner. Res. 2017, 32, 889–891. [Google Scholar] [CrossRef] [PubMed]
- Fulzele, K.; Lai, F.; Dedic, C.; Saini, V.; Uda, Y.; Shi, C.; Tuck, P.; Aronson, J.L.; Liu, X.; Spatz, J.M.; et al. Osteocyte-Secreted Wnt Signaling Inhibitor Sclerostin Contributes to Beige Adipogenesis in Peripheral Fat Depots. J. Bone Min. Res. 2017, 32, 373. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Yu, R.; Jiang, F.; Hou, X.; Wei, L.; Bao, Y.; Jia, W. Osteocalcin and Risks of Incident Diabetes and Diabetic Kidney Disease: A 4.6-Year Prospective Cohort Study. Diabetes Care 2022, 45, 830–836. [Google Scholar] [CrossRef]
- Lacombe, J.; Rifai, O.A.; Loter, L.; Moran, T.; Turcotte, A.F.; Grenier-Larouche, T.; Tchernof, A.; Biertho, L.; Carpentier, A.C.; Prud’homme, D.; et al. Measurement of Bioactive Osteocalcin in Humans Using a Novel Immunoassay Reveals Association with Glucose Metabolism and β-Cell Function. Am. J. Physiol. Endocrinol. Metab. 2020, 318, E381–E391. [Google Scholar] [CrossRef]
- Liu, H.; Liu, L.; Rosen, C.J. Bone Marrow Adipocytes as Novel Regulators of Metabolic Homeostasis: Clinical Consequences of Bone Marrow Adiposity. Curr. Obes. Rep. 2025, 14, 9. [Google Scholar] [CrossRef]
- Suchacki, K.J.; Tavares, A.A.S.; Mattiucci, D.; Scheller, E.L.; Papanastasiou, G.; Gray, C.; Sinton, M.C.; Ramage, L.E.; McDougald, W.A.; Lovdel, A.; et al. Bone Marrow Adipose Tissue Is a Unique Adipose Subtype with Distinct Roles in Glucose Homeostasis. Nat. Commun. 2020, 11, 3097. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Tian, L.; Majumdar, A.; Scheller, E.L. Function and Regulation of Bone Marrow Adipose Tissue in Health and Disease: State of the Field and Clinical Considerations. Compr. Physiol. 2024, 14, 5521–5579. [Google Scholar] [CrossRef]
- Zhao, Y.; Peng, X.; Wang, Q.; Zhang, Z.; Wang, L.; Xu, Y.; Yang, H.; Bai, J.; Geng, D. Crosstalk Between the Neuroendocrine System and Bone Homeostasis. Endocr. Rev. 2024, 45, 95–124. [Google Scholar] [CrossRef]
- Zhang, Y.; Su, X.; Dong, Y.; Chen, T.; Zhang, Y.; Wu, B.; Li, H.; Sun, X.; Xia, L.; Zhang, D.; et al. Cytological and Functional Characteristics of Fascia Adipocytes in Rats: A Unique Population of Adipocytes. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2020, 1865, 158585. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Lyu, Y.; Wang, W.; Zhang, Y.; Li, D.; Wei, S.; Du, C.; Geng, B.; Sztalryd, C.; Xu, G. Fascia Origin of Adipose Cells. Stem Cells 2016, 34, 1407–1419. [Google Scholar] [CrossRef]
- Chen, T.; Zhang, Y.; Dong, Y.; Zhang, D.; Xia, L.; Sun, X.; Li, H.; Han, C.; Wang, H.; Xu, G. Mast Cell and Heparin Promote Adipogenesis in Superficial Fascia of Rats. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2021, 1866, 159024. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Dong, Y.; Chen, T.; Xu, G. Generation of Functional Fat Organoid from Rat Superficial Fascia. Adipocyte 2022, 11, 287–300. [Google Scholar] [CrossRef]
- Fede, C.; Petrelli, L.; Pirri, C.; Neuhuber, W.; Tiengo, C.; Biz, C.; De Caro, R.; Schleip, R.; Stecco, C. Innervation of Human Superficial Fascia. Front. Neuroanat. 2022, 16, 981426. [Google Scholar] [CrossRef]
- Albertin, G.; Astolfi, L.; Fede, C.; Simoni, E.; Contran, M.; Petrelli, L.; Tiengo, C.; Guidolin, D.; De Caro, R.; Stecco, C. Detection of Lymphatic Vessels in the Superficial Fascia of the Abdomen. Life 2023, 13, 836. [Google Scholar] [CrossRef] [PubMed]
- Fede, C.; Petrelli, L.; Pirri, C.; Tiengo, C.; De Caro, R.; Stecco, C. Detection of Mast Cells in Human Superficial Fascia. Int. J. Mol. Sci. 2023, 24, 11599. [Google Scholar] [CrossRef]
- Uapinyoying, P.; Hogarth, M.; Battacharya, S.; Mázala, D.A.G.; Panchapakesan, K.; Bönnemann, C.G.; Jaiswal, J.K. Single-Cell Transcriptomic Analysis of the Identity and Function of Fibro/Adipogenic Progenitors in Healthy and Dystrophic Muscle. iScience 2023, 26, 107479. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, G.; Turiel, G.; Gorski, T.; Soro-Arnaiz, I.; Zhang, J.; Casartelli, N.C.; Masschelein, E.; Maffiuletti, N.A.; Sutter, R.; Leunig, M.; et al. MME+ Fibro-Adipogenic Progenitors Are the Dominant Adipogenic Population during Fatty Infiltration in Human Skeletal Muscle. Commun. Biol. 2023, 6, 111. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Li, Y.; Zhang, J.; Qi, B.; Chen, J.; Sun, Y. Role of Aberrant Activated Fibro/Adipogenic Progenitors and Suppressed Ferroptosis in Disused Skeletal Muscle Atrophy and Fatty Infiltration. J. Mol. Med. 2025, 103, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Joe, A.W.B.; Yi, L.; Natarajan, A.; Le Grand, F.; So, L.; Wang, J.; Rudnicki, M.A.; Rossi, F.M.V. Muscle Injury Activates Resident Fibro/Adipogenic Progenitors That Facilitate Myogenesis. Nat. Cell Biol. 2010, 12, 153–163. [Google Scholar] [CrossRef]
- Fu, C.; Chin-Young, B.; Park, G.Y.; Guzmán-Seda, M.; Laudier, D.; Han, W.M. WNT7A Suppresses Adipogenesis of Skeletal Muscle Mesenchymal Stem Cells and Fatty Infiltration through the Alternative Wnt-Rho-YAP/TAZ Signaling Axis. Stem Cell Rep. 2023, 18, 999–1014. [Google Scholar] [CrossRef]
- Giuliani, G.; Rosina, M.; Reggio, A. Signaling Pathways Regulating the Fate of Fibro/Adipogenic Progenitors (FAPs) in Skeletal Muscle Regeneration and Disease. FEBS J. 2022, 289, 6484–6517. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Hayashi, M.; Uchida, Y.; Cheng, X.W.; Nakayama, T.; Matsushita, T.; Murohara, T.; Takeshita, K. Notch1 Haploinsufficiency in Mice Accelerates Adipogenesis. Sci. Rep. 2021, 11, 16761. [Google Scholar] [CrossRef]
- Dennis, K.M.J.H.; Gopal, K.; Montes Aparicio, C.N.; Aaron Zhang, J.; Castro-Guarda, M.; Nicol, T.; Devereux, R.M.; Carter, R.D.; Azizi, S.A.; Lan, T.; et al. FoxO1-ZDHHC4-CD36 S-Acylation Axis Drives Metabolic Dysfunction in Diabetes. Circ. Res. 2025, 136, 1545. [Google Scholar] [CrossRef]
- Zhu, B.; Li, M.Y.; Lin, Q.; Liang, Z.; Xin, Q.; Wang, M.; He, Z.; Wang, X.; Wu, X.; Chen, G.G.; et al. Lipid Oversupply Induces CD36 Sarcolemmal Translocation via Dual Modulation of PKCζ and TBC1D1: An Early Event Prior to Insulin Resistance. Theranostics 2020, 10, 1332. [Google Scholar] [CrossRef]
- Hao, J.W.; Wang, J.; Guo, H.; Zhao, Y.Y.; Sun, H.H.; Li, Y.F.; Lai, X.Y.; Zhao, N.; Wang, X.; Xie, C.; et al. CD36 Facilitates Fatty Acid Uptake by Dynamic Palmitoylation-Regulated Endocytosis. Nat. Commun. 2020, 11, 4765. [Google Scholar] [CrossRef]
- Jocken, J.W.E.; Goossens, G.H.; Boon, H.; Mason, R.R.; Essers, Y.; Havekes, B.; Watt, M.J.; Van Loon, L.J.; Blaak, E.E. Insulin-Mediated Suppression of Lipolysis in Adipose Tissue and Skeletal Muscle of Obese Type 2 Diabetic Men and Men with Normal Glucose Tolerance. Diabetologia 2013, 56, 2255–2265. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, B.A.; Young, M.; Nakamura, N.; Ha, H.; Carter, L.; Pitts, M.W.; Torres, D.; Noh, H.L.; Suk, S.; Kim, J.K.; et al. Disrupted Glucose Homeostasis and Skeletal-Muscle-Specific Glucose Uptake in an Exocyst Knockout Mouse Model. J. Biol. Chem. 2021, 296, 100482. [Google Scholar] [CrossRef]
- Bonen, A.; Campbell, S.E.; Benton, C.R.; Chabowski, A.; Coort, S.L.M.; Han, X.-X.; Koonen, D.P.Y.; Glatz, J.F.C.; Luiken, J.J.F.P. Regulation of Fatty Acid Transport by Fatty Acid Translocase/CD36. Proc. Nutr. Soc. 2004, 63, 245–249. [Google Scholar] [CrossRef]
- Ahuja, P.; Ng, C.F.; Pang, B.P.S.; Chan, W.S.; Tse, M.C.L.; Bi, X.; Kwan, H.L.R.; Brobst, D.; Herlea-Pana, O.; Yang, X.; et al. Muscle-Generated BDNF (Brain Derived Neurotrophic Factor) Maintains Mitochondrial Quality Control in Female Mice. Autophagy 2022, 18, 1367–1384. [Google Scholar] [CrossRef]
- Li, Q.; Jiang, X.; Zhou, Y.; Gu, Y.; Ding, Y.; Luo, J.; Pang, N.; Sun, Y.; Pei, L.; Pan, J.; et al. Improving Mitochondrial Function in Skeletal Muscle Contributes to the Amelioration of Insulin Resistance by Nicotinamide Riboside. Int. J. Mol. Sci. 2023, 24, 10015. [Google Scholar] [CrossRef] [PubMed]
- Field, J.T.; Chapman, D.; Hai, Y.; Ghavami, S.; West, A.R.; Ozerklig, B.; Saleem, A.; Kline, J.; Mendelson, A.A.; Kindrachuk, J.; et al. The Mitophagy Receptor BNIP3L/Nix Coordinates Nuclear Calcium Signaling to Modulate the Muscle Phenotype. Autophagy 2025, 21, 1544–1555. [Google Scholar] [CrossRef] [PubMed]
- Hoehn, K.L.; Salmon, A.B.; Hohnen-Behrens, C.; Turner, N.; Hoy, A.J.; Maghzal, G.J.; Stocker, R.; Van Remmen, H.; Kraegen, E.W.; Cooney, G.J.; et al. Insulin Resistance Is a Cellular Antioxidant Defense Mechanism. Proc. Natl. Acad. Sci. USA 2009, 106, 17787. [Google Scholar] [CrossRef] [PubMed]
- Turpin-Nolan, S.M.; Hammerschmidt, P.; Chen, W.; Jais, A.; Timper, K.; Awazawa, M.; Brodesser, S.; Brüning, J.C. CerS1-Derived C18:0 Ceramide in Skeletal Muscle Promotes Obesity-Induced Insulin Resistance. Cell Rep. 2019, 26, 1–10.e7. [Google Scholar] [CrossRef]
- Hammerschmidt, P.; Steculorum, S.M.; Bandet, C.L.; Del Río-Martín, A.; Steuernagel, L.; Kohlhaas, V.; Feldmann, M.; Varela, L.; Majcher, A.; Quatorze Correia, M.; et al. CerS6-Dependent Ceramide Synthesis in Hypothalamic Neurons Promotes ER/Mitochondrial Stress and Impairs Glucose Homeostasis in Obese Mice. Nat. Commun. 2023, 14, 7824. [Google Scholar] [CrossRef]
- Houstis, N.; Rosen, E.D.; Lander, E.S. Reactive Oxygen Species Have a Causal Role in Multiple Forms of Insulin Resistance. Nature 2006, 440, 944–948. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Endoplasmic Reticulum Stress and the Inflammatory Basis of Metabolic Disease. Cell 2010, 140, 900–917. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.; Dun, Y.; Cheng, J.; Ripley-Gonzalez, J.W.; Jiang, W.; You, B.; Liu, S. Exercise Activates Autophagy and Regulates Endoplasmic Reticulum Stress in Muscle of High-Fat Diet Mice to Alleviate Insulin Resistance. Biochem. Biophys. Res. Commun. 2022, 601, 45–51. [Google Scholar] [CrossRef]
- Kwon, H.J.; Yang, H.M.; Won, S.Y. Intramuscular Innervation Patterns of the Splenius Capitis and Splenius Cervicis and Their Clinical Implications for Botulinum Toxin Injections. Clin. Anat. 2020, 33, 1138–1143. [Google Scholar] [CrossRef]
- Li, L.; Lin, J.; Huang, C.; Liu, J.; Yuan, Y.; Liu, Z.; Li, Y.; Li, W.; Diao, A. The TFEB Activator Clomiphene Citrate Ameliorates Lipid Metabolic Syndrome Pathology by Activating Lipophagy and Lipolysis. Biochem. Pharmacol. 2025, 232, 116694. [Google Scholar] [CrossRef]
- Wang, Y.; Nguyen, H.P.; Xue, P.; Xie, Y.; Yi, D.; Lin, F.; Dinh, J.; Viscarra, J.A.; Ibe, N.U.; Duncan, R.E.; et al. ApoL6 Associates with Lipid Droplets and Disrupts Perilipin1-HSL Interaction to Inhibit Lipolysis. Nat. Commun. 2024, 15, 186. [Google Scholar] [CrossRef] [PubMed]
- Goodpaster, B.H.; He, J.; Watkins, S.; Kelley, D.E. Skeletal Muscle Lipid Content and Insulin Resistance: Evidence for a Paradox in Endurance-Trained Athletes. J. Clin. Endocrinol. Metab. 2001, 86, 5755–5761. [Google Scholar] [CrossRef] [PubMed]
- Labbé, K.; LeBon, L.; King, B.; Vu, N.; Stoops, E.H.; Ly, N.; Lefebvre, A.E.Y.T.; Seitzer, P.; Krishnan, S.; Heo, J.M.; et al. Specific Activation of the Integrated Stress Response Uncovers Regulation of Central Carbon Metabolism and Lipid Droplet Biogenesis. Nat. Commun. 2024, 15, 8301. [Google Scholar] [CrossRef] [PubMed]
- Serrano, N.; Colenso-Semple, L.M.; Lazauskus, K.K.; Siu, J.W.; Bagley, J.R.; Lockie, R.G.; Costa, P.B.; Galpin, A.J. Extraordinary Fast-Twitch Fiber Abundance in Elite Weightlifters. PLoS ONE 2019, 14, e0207975. [Google Scholar] [CrossRef]
- Plotkin, D.L.; Roberts, M.D.; Haun, C.T.; Schoenfeld, B.J. Muscle Fiber Type Transitions with Exercise Training: Shifting Perspectives. Sports 2021, 9, 127. [Google Scholar] [CrossRef]
- Sergi, D.; Naumovski, N.; Heilbronn, L.K.; Abeywardena, M.; O’Callaghan, N.; Lionetti, L.; Luscombe-Marsh, N. Mitochondrial (Dys)Function and Insulin Resistance: From Pathophysiological Molecular Mechanisms to the Impact of Diet. Front. Physiol. 2019, 10, 532. [Google Scholar] [CrossRef]
- Chen, G.; Long, J.; Zhang, Y.; Zhou, X.; Gao, B.; Qin, Z.; Zhu, Y.; Song, B.; Cui, Z.; liu, Z.; et al. Fascia-Derived Stem Cells Enhance Fat Graft Retention by Promoting Vascularization through the HMOX1-HIF-1α Pathway. Stem Cell Res. Ther. 2025, 16, 92. [Google Scholar] [CrossRef]
- Contreras, O.; Soliman, H.; Theret, M.; Rossi, F.M.V.; Brandan, E. TGF-β-Driven Downregulation of the Transcription Factor TCF7L2 Affects Wnt/β-Catenin Signaling in PDGFRα+ Fibroblasts. J. Cell Sci. 2020, 133, jcs242297. [Google Scholar] [CrossRef] [PubMed]
- Shibao, C.A.; Peche, V.S.; Pietka, T.A.; Samovski, D.; Williams, I.M.; Abumrad, N.N.; Gamazon, E.R.; Goldberg, I.J.; Wasserman, D.H.; Abumrad, N.A. Microvascular Insulin Resistance with Enhanced Muscle Glucose Disposal in CD36 Deficiency. Diabetologia 2025, 68, 662–675. [Google Scholar] [CrossRef] [PubMed]
- Nie, Y.; Sato, Y.; Garner, R.T.; Kargl, C.; Wang, C.; Kuang, S.; Gilpin, C.J.; Gavin, T.P. Skeletal Muscle-Derived Exosomes Regulate Endothelial Cell Functions via Reactive Oxygen Species-Activated Nuclear Factor-ΚB Signalling. Exp. Physiol. 2019, 104, 1262–1273. [Google Scholar] [CrossRef]
- Görgens, S.W.; Eckardt, K.; Jensen, J.; Drevon, C.A.; Eckel, J. Exercise and Regulation of Adipokine and Myokine Production. Prog. Mol. Biol. Transl. Sci. 2015, 135, 313–336. [Google Scholar] [CrossRef]
- Boström, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Boström, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1-α-Dependent Myokine That Drives Brown-Fat-like Development of White Fat and Thermogenesis. Nature 2012, 481, 463–468. [Google Scholar] [CrossRef]
- He, J.; Watkins, S.; Kelley, D.E. Skeletal Muscle Lipid Content and Oxidative Enzyme Activity in Relation to Muscle Fiber Type in Type 2 Diabetes and Obesity. Diabetes 2001, 50, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Uezumi, A.; Fukada, S.; Yamamoto, N.; Ikemoto-Uezumi, M.; Nakatani, M.; Morita, M.; Yamaguchi, A.; Yamada, H.; Nishino, I.; Hamada, Y.; et al. Identification and Characterization of PDGFRα+ Mesenchymal Progenitors in Human Skeletal Muscle. Cell Death Dis. 2014, 5, e1186. [Google Scholar] [CrossRef]
- Karastergiou, K.; Fried, S.K. Multiple Adipose Depots Increase Cardiovascular Risk via Local and Systemic Effects Topical Collection on Clinical Trials and Their Interpretations. Curr. Atheroscler. Rep. 2013, 15, 361. [Google Scholar] [CrossRef]
- Manolopoulos, K.N.; Karpe, F.; Frayn, K.N. Gluteofemoral Body Fat as a Determinant of Metabolic Health. Int. J. Obes. 2010, 34, 949–959. [Google Scholar] [CrossRef]
- Snijder, M.B.; Dekker, J.M.; Visser, M.; Yudkin, J.S.; Stehouwer, C.D.A.; Bouter, L.M.; Heine, R.J.; Nijpels, G.; Seidell, J.C. Larger Thigh and Hip Circumferences Are Associated with Better Glucose Tolerance: The Hoorn Study. Obes. Res. 2003, 11, 104–111. [Google Scholar] [CrossRef]
- Tchkonia, T.; Thomou, T.; Zhu, Y.; Karagiannides, I.; Pothoulakis, C.; Jensen, M.D.; Kirkland, J.L. Mechanisms and Metabolic Implications of Regional Differences among Fat Depots. Cell Metab. 2013, 17, 644–656. [Google Scholar] [CrossRef]
- Alser, M.; Naja, K.; Elrayess, M.A. Mechanisms of Body Fat Distribution and Gluteal-Femoral Fat Protection against Metabolic Disorders. Front. Nutr. 2024, 11, 1368966. [Google Scholar] [CrossRef]
- Brand, T.; Van Den Munckhof, I.C.L.; Van Der Graaf, M.; Schraa, K.; Dekker, H.M.; Joosten, L.A.B.; Netea, M.G.; Riksen, N.P.; De Graaf, J.; Rutten, J.H.W. Superficial vs Deep Subcutaneous Adipose Tissue: Sex-Specific Associations With Hepatic Steatosis and Metabolic Traits. J. Clin. Endocrinol. Metab. 2021, 106, e3881. [Google Scholar] [CrossRef]
- Dhokte, S.; Czaja, K. Visceral Adipose Tissue: The Hidden Culprit for Type 2 Diabetes. Nutrients 2024, 16, 1015. [Google Scholar] [CrossRef] [PubMed]
- Kahn, D.; Macias, E.; Zarini, S.; Garfield, A.; Zemski Berry, K.; Gerszten, R.; Schoen, J.; Cree-Green, M.; Bergman, B.C. Quantifying the Inflammatory Secretome of Human Intermuscular Adipose Tissue. Physiol. Rep. 2022, 10, e15424. [Google Scholar] [CrossRef]
- Tuttle, L.J.; Sinacore, D.R.; Cade, W.T.; Mueller, M.J. Lower Physical Activity Is Associated with Higher Intermuscular Adipose Tissue in People with Type 2 Diabetes and Peripheral Neuropathy. Phys. Ther. 2011, 91, 923–930. [Google Scholar] [CrossRef]
- Guo, F.; Xu, S.; Zhu, Y.; Zheng, X.; Lu, Y.; Tu, J.; He, Y.; Jin, L.; Li, Y. PPARγ Transcription Deficiency Exacerbates High-Fat Diet-Induced Adipocyte Hypertrophy and Insulin Resistance in Mice. Front. Pharmacol. 2020, 11, 1285. [Google Scholar] [CrossRef] [PubMed]
- Garin-Shkolnik, T.; Rudich, A.; Hotamisligil, G.S.; Rubinstein, M. FABP4 Attenuates PPARγ and Adipogenesis and Is Inversely Correlated with PPARγ in Adipose Tissues. Diabetes 2014, 63, 900–911. [Google Scholar] [CrossRef]
- Jaffer, I.; Riederer, M.; Shah, P.; Peters, P.; Quehenberger, F.; Wood, A.; Scharnagl, H.; März, W.; Kostner, K.M.; Kostner, G.M. Expression of Fat Mobilizing Genes in Human Epicardial Adipose Tissue. Atherosclerosis 2012, 220, 122–127. [Google Scholar] [CrossRef]
- Zhang, T.; Li, J.; Li, X.; Liu, Y. Intermuscular Adipose Tissue in Obesity and Related Disorders: Cellular Origins, Biological Characteristics and Regulatory Mechanisms. Front. Endocrinol. 2023, 14, 1280853. [Google Scholar] [CrossRef] [PubMed]
- Trayhurn, P.; Beattie, J.H. Physiological Role of Adipose Tissue: White Adipose Tissue as an Endocrine and Secretory Organ. Proc. Nutr. Soc. 2001, 60, 329–339. [Google Scholar] [CrossRef]
- McKie, G.L.; Wright, D.C. Biochemical Adaptations in White Adipose Tissue Following Aerobic Exercise: From Mitochondrial Biogenesis to Browning. Biochem. J. 2020, 477, 1061–1081. [Google Scholar] [CrossRef]
- Roden, M.; Shulman, G.I. The Integrative Biology of Type 2 Diabetes. Nature 2019, 576, 51–60. [Google Scholar] [CrossRef]
- Gornicka, A.; Fettig, J.; Eguchi, A.; Berk, M.P.; Thapaliya, S.; Dixon, L.J.; Feldstein, A.E. Adipocyte Hypertrophy Is Associated with Lysosomal Permeability Both in Vivo and in Vitro: Role in Adipose Tissue Inflammation. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E597–E606. [Google Scholar] [CrossRef]
- Alkhouri, N.; Gornicka, A.; Berk, M.P.; Thapaliya, S.; Dixon, L.J.; Kashyap, S.; Schauer, P.R.; Feldstein, A.E. Adipocyte Apoptosis, a Link between Obesity, Insulin Resistance, and Hepatic Steatosis. J. Biol. Chem. 2010, 285, 3428–3438. [Google Scholar] [CrossRef]
- Yaghootkar, H.; Lotta, L.A.; Tyrrell, J.; Smit, R.A.J.; Jones, S.E.; Donnelly, L.; Beaumont, R.; Campbell, A.; Tuke, M.A.; Hayward, C.; et al. Genetic Evidence for a Link between Favorable Adiposity and Lower Risk of Type 2 Diabetes, Hypertension, and Heart Disease. Diabetes 2016, 65, 2448–2460. [Google Scholar] [CrossRef] [PubMed]
- Abraham, A.; Cule, M.; Thanaj, M.; Basty, N.; Hashemloo, M.A.; Sorokin, E.P.; Whitcher, B.; Burgess, S.; Bell, J.D.; Sattar, N.; et al. Genetic Evidence for Distinct Biological Mechanisms That Link Adiposity to Type 2 Diabetes: Toward Precision Medicine. Diabetes 2024, 73, 1012–1025. [Google Scholar] [CrossRef] [PubMed]
- Shungin, D.; Winkler, T.; Croteau-Chonka, D.C.; Ferreira, T.; Locke, A.E.; Mägi, R.; Strawbridge, R.J.; Pers, T.H.; Fischer, K.; Justice, A.E.; et al. New Genetic Loci Link Adipose and Insulin Biology to Body Fat Distribution. Nature 2015, 518, 187–196. [Google Scholar] [CrossRef]
- Jilo, D.D.; Abebe, B.K.; Wang, J.; Guo, J.; Li, A.; Zan, L. Long Non-Coding RNA (LncRNA) and Epigenetic Factors: Their Role in Regulating the Adipocytes in Bovine. Front. Genet. 2024, 15, 1405588. [Google Scholar] [CrossRef] [PubMed]
- Fassio, A.; Idolazzi, L.; Rossini, M.; Gatti, D.; Adami, G.; Giollo, A.; Viapiana, O. The Obesity Paradox and Osteoporosis. Eat. Weight. Disord. 2018, 23, 293–302. [Google Scholar] [CrossRef]
- Dai, X.; Liu, B.; Hou, Q.; Dai, Q.; Wang, D.; Xie, B.; Sun, Y.; Wang, B. Global and Local Fat Effects on Bone Mass and Quality in Obesity: Insights from Lipidomics and 3D Imaging Assessment. Bone Jt. Res. 2023, 12, 580. [Google Scholar] [CrossRef]
- He, H.; Zhang, Y.; Sun, Y.; Zhang, Y.; Xu, J.; Yang, Y.; Chen, J. Folic Acid Attenuates High-Fat Diet-Induced Osteoporosis Through the AMPK Signaling Pathway. Front. Cell Dev. Biol. 2022, 9, 791880. [Google Scholar] [CrossRef]
- Kang, Y.S.; Kim, J.C.; Kim, J.S.; Kim, S.H. Effects of Swimming Exercise on Serum Irisin and Bone FNDC5 in Rat Models of High-Fat Diet-Induced Osteoporosis. J. Sports Sci. Med. 2019, 18, 596. [Google Scholar]
- Walsh, M.C.; Kim, G.K.; Maurizio, P.L.; Molnar, E.E.; Choi, Y. TRAF6 Autoubiquitination-Independent Activation of the NFκB and MAPK Pathways in Response to IL-1 and RANKL. PLoS ONE 2008, 3, e4064. [Google Scholar] [CrossRef]
- Wu, W.J.; Xia, C.L.; Ou, S.J.; Yang, Y.; Ma, Y.F.; Hou, Y.L.; Yang, Q.P.; Zhang, J.; Li, J.W.; Qi, Y.; et al. Novel Elongator Protein 2 Inhibitors Mitigating Tumor Necrosis Factor- α Induced Osteogenic Differentiation Inhibition. Biomed. Res. Int. 2021, 2021, 3664564. [Google Scholar] [CrossRef]
- Yao, Z.; Getting, S.J.; Locke, I.C. Regulation of Tnf-Induced Osteoclast Differentiation. Cells 2022, 11, 132. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Q.; Rao, E.; Sun, Y.; Grossmann, M.E.; Morris, R.J.; Cleary, M.P.; Li, B. Epidermal Fatty Acid Binding Protein Promotes Skin Inflammation Induced by High-Fat Diet. Immunity 2015, 42, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Leanza, G.; Conte, C.; Cannata, F.; Isgrò, C.; Piccoli, A.; Strollo, R.; Quattrocchi, C.C.; Papalia, R.; Denaro, V.; Maccarrone, M.; et al. Oxidative Stress in Postmenopausal Women with or without Obesity. Cells 2023, 12, 1137. [Google Scholar] [CrossRef]
- Cojocaru, K.-A.; Luchian, I.; Goriuc, A.; Antoci, L.-M.; Ciobanu, C.-G.; Popescu, R.; Vlad, C.-E.; Blaj, M.; Foia, L.G. Mitochondrial Dysfunction, Oxidative Stress, and Therapeutic Strategies in Diabetes, Obesity, and Cardiovascular Disease. Antioxidants 2023, 12, 658. [Google Scholar] [CrossRef] [PubMed]
- Lubkowska, A.; Dudzińska, W.; Pluta, W. Antioxidant Enzyme Activity and Serum HSP70 Concentrations in Relation to Insulin Resistance and Lipid Profile in Lean and Overweight Young Men. Antioxidants 2023, 12, 655. [Google Scholar] [CrossRef] [PubMed]
- Xia, B.; Zhu, R.; Zhang, H.; Chen, B.; Liu, Y.; Dai, X.; Ye, Z.; Zhao, D.; Mo, F.; Gao, S.; et al. Lycopene Improves Bone Quality and Regulates AGE/RAGE/NF-κB Signaling Pathway in High-Fat Diet-Induced Obese Mice. Oxid. Med. Cell Longev. 2022, 2022, 3697067. [Google Scholar] [CrossRef]
- Wang, Y.N.; Jia, T.T.; Feng, Y.; Liu, S.Y.; Zhang, W.J.; Zhang, D.J.; Xu, X. Hyperlipidemia Impairs Osseointegration via the ROS/Wnt/β-Catenin Pathway. J. Dent. Res. 2021, 100, 658–665. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.; Ambrogini, E.; Han, L.; Manolagas, S.C.; Jilka, R.L. Increased Lipid Oxidation Causes Oxidative Stress, Increased Peroxisome Proliferator-Activated Receptor-γ Expression, and Diminished pro-Osteogenic Wnt Signaling in the Skeleton. J. Biol. Chem. 2009, 284, 27438–27448. [Google Scholar] [CrossRef]
- Xiao, Y.; Cui, J.; Li, Y.X.; Shi, Y.H.; Wang, B.; Le, G.W.; Wang, Z.P. Dyslipidemic High-Fat Diet Affects Adversely Bone Metabolism in Mice Associated with Impaired Antioxidant Capacity. Nutrition 2011, 27, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Park, J.N.; Park, H.J.; Suh, J.H.; Choi, H.S. High Cholesterol-Induced Bone Loss Is Attenuated by Arctiin via an Action in Osteoclasts. Nutrients 2022, 14, 4483. [Google Scholar] [CrossRef]
- Patsch, J.M.; Li, X.; Baum, T.; Yap, S.P.; Karampinos, D.C.; Schwartz, A.; Link, T.M. Bone Marrow Fat Composition as a Novel Imaging Biomarker in Postmenopausal Women with Prevalent Fragility Fractures. J. Bone Min. Res. 2013, 28, 1721–1728. [Google Scholar] [CrossRef]
- Sheu, Y.; Amati, F.; Schwartz, A.V.; Danielson, M.E.; Li, X.; Boudreau, R.; Cauley, J.A. Vertebral Bone Marrow Fat, Bone Mineral Density and Diabetes: The Osteoporotic Fractures in Men (MrOS) Study. Bone 2017, 97, 299. [Google Scholar] [CrossRef]
- Kim, T.Y.; Schwartz, A.V.; Li, X.; Xu, K.; Kazakia, G.J.; Grunfeld, C.; Nissenson, R.A.; Shoback, D.M.; Schafer, A.L. Bone Marrow Adipose Tissue Composition and Glycemic Improvements after Gastric Bypass Surgery. Bone Rep. 2022, 17, 101596. [Google Scholar] [CrossRef]
- Gassert, F.T.; Kufner, A.; Gassert, F.G.; Leonhardt, Y.; Kronthaler, S.; Schwaiger, B.J.; Boehm, C.; Makowski, M.R.; Kirschke, J.S.; Baum, T.; et al. MR-Based Proton Density Fat Fraction (PDFF) of the Vertebral Bone Marrow Differentiates between Patients with and without Osteoporotic Vertebral Fractures. Osteoporos. Int. 2022, 33, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.-H.; Joseph, G.; Saeed, I.; Pirmoazen, A.M.; Kenny, K.; Kim, T.Y.; Schafer, A.L.; Schwartz, A.V.; Li, X.; Link, T.M.; et al. Bone Marrow Adiposity Alterations in Type 2 Diabetes Are Sex-Specific and Associated with Serum Lipid Levels. J. Bone Miner. Res. 2023, 38, 1877–1884. [Google Scholar] [CrossRef]
- Saroha, A.; Saran, S.; Saxena, S.; Kant, R.; Bhadoria, A.S. Ultrasonographic Evaluation of Thickness and Stiffness of Achilles Tendon and Plantar Fascia in Type 2 Diabetics Patients: A Cross-Sectional Observation Study. J. Med. Ultrasound 2023, 31, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Pirri, C.; Biz, C.; Pirri, N.; Macchi, V.; Porzionato, A.; De Caro, R.; Ruggieri, P.; Stecco, C. Crural and Plantar Fasciae Changes in Chronic Charcot Diabetic Foot: A Cross-Sectional Ultrasound Imaging Study-An Evidence of Fascial Continuity. J. Clin. Med. 2023, 12, 4664. [Google Scholar] [CrossRef] [PubMed]
- Rungratanawanich, W.; Qu, Y.; Wang, X.; Essa, M.M.; Song, B.J. Advanced Glycation End Products (AGEs) and Other Adducts in Aging-Related Diseases and Alcohol-Mediated Tissue Injury. Exp. Mol. Med. 2021, 53, 168–188. [Google Scholar] [CrossRef]
- Ugwoke, C.K.; Cvetko, E.; Umek, N. Pathophysiological and Therapeutic Roles of Fascial Hyaluronan in Obesity-Related Myofascial Disease. Int. J. Mol. Sci. 2022, 23, 11843. [Google Scholar] [CrossRef]
- Wang, K.; Yang, J.; An, Y.; Wang, J.; Tan, S.; Xu, H.; Dong, Y. MST1/2 Regulates Fibro/Adipogenic Progenitor Fate Decisions in Skeletal Muscle Regeneration. Stem Cell Rep. 2024, 19, 501–514. [Google Scholar] [CrossRef]
- Lemos, D.R.; Babaeijandaghi, F.; Low, M.; Chang, C.K.; Lee, S.T.; Fiore, D.; Zhang, R.H.; Natarajan, A.; Nedospasov, S.A.; Rossi, F.M.V. Nilotinib Reduces Muscle Fibrosis in Chronic Muscle Injury by Promoting TNF-Mediated Apoptosis of Fibro/Adipogenic Progenitors. Nat. Med. 2015, 21, 786–794. [Google Scholar] [CrossRef] [PubMed]
- Giordani, L.; He, G.J.; Negroni, E.; Sakai, H.; Law, J.Y.C.; Siu, M.M.; Wan, R.; Corneau, A.; Tajbakhsh, S.; Cheung, T.H.; et al. High-Dimensional Single-Cell Cartography Reveals Novel Skeletal Muscle-Resident Cell Populations. Mol. Cell 2019, 74, 609–621.e6. [Google Scholar] [CrossRef]
- Aykul, S.; Huang, L.; Wang, L.; Das, N.M.; Reisman, S.; Ray, Y.; Zhang, Q.; Rothman, N.; Nannuru, K.C.; Kamat, V.; et al. Anti-ACVR1 Antibodies Exacerbate Heterotopic Ossification in Fibrodysplasia Ossificans Progressiva (FOP) by Activating FOP-Mutant ACVR1. J. Clin. Investig. 2022, 132, e153792. [Google Scholar] [CrossRef]
- Buras, E.D.; Converso-Baran, K.; Davis, C.S.; Akama, T.; Hikage, F.; Michele, D.E.; Brooks, S.V.; Chun, T.H. Fibro-Adipogenic Remodeling of the Diaphragm in Obesity-Associated Respiratory Dysfunction. Diabetes 2019, 68, 45–56. [Google Scholar] [CrossRef]
- Arrighi, N.; Moratal, C.; Clément, N.; Giorgetti-Peraldi, S.; Peraldi, P.; Loubat, A.; Kurzenne, J.Y.; Dani, C.; Chopard, A.; Dechesne, C.A. Characterization of Adipocytes Derived from Fibro/Adipogenic Progenitors Resident in Human Skeletal Muscle. Cell Death Dis. 2015, 6, e1733. [Google Scholar] [CrossRef]
- Chen, W.; Wang, L.; You, W.; Shan, T. Myokines Mediate the Cross Talk between Skeletal Muscle and Other Organs. J. Cell Physiol. 2021, 236, 2393–2412. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, R.; Thurmond, D.C. Mechanisms by Which Skeletal Muscle Myokines Ameliorate Insulin Resistance. Int. J. Mol. Sci. 2022, 23, 4636. [Google Scholar] [CrossRef] [PubMed]
- Pelczyńska, M.; Miller-Kasprzak, E.; Piątkowski, M.; Mazurek, R.; Klause, M.; Suchecka, A.; Bucoń, M.; Bogdański, P. The Role of Adipokines and Myokines in the Pathogenesis of Different Obesity Phenotypes—New Perspectives. Antioxidants 2023, 12, 2046. [Google Scholar] [CrossRef]
- Townsend, L.K.; Wright, D.C. Looking on the “Brite” Side Exercise-Induced Browning of White Adipose Tissue. Pflug. Arch. 2019, 471, 455–465. [Google Scholar] [CrossRef] [PubMed]
- Mitrou, P.; Lambadiari, V.; Maratou, E.; Boutati, E.; Komesidou, V.; Papakonstantinou, A.; Raptis, S.A.; Dimitriadis, G. Skeletal Muscle Insulin Resistance in Morbid Obesity: The Role of Interleukin-6 and Leptin. Exp. Clin. Endocrinol. Diabetes 2011, 119, 484–489. [Google Scholar] [CrossRef]
- Holmes, A.G.; Mesa, J.L.; Neill, B.A.; Chung, J.; Carey, A.L.; Steinberg, G.R.; Kemp, B.E.; Southgate, R.J.; Lancaster, G.I.; Bruce, C.R.; et al. Prolonged Interleukin-6 Administration Enhances Glucose Tolerance and Increases Skeletal Muscle PPARα and UCP2 Expression in Rats. J. Endocrinol. 2008, 198, 367–374. [Google Scholar] [CrossRef]
- Carey, A.L.; Steinberg, G.R.; Macaulay, S.L.; Thomas, W.G.; Holmes, A.G.; Ramm, G.; Prelovsek, O.; Hohnen-Behrens, C.; Watt, M.J.; James, D.E.; et al. Interleukin-6 Increases Insulin-Stimulated Glucose Disposal in Humans and Glucose Uptake and Fatty Acid Oxidation in Vitro via AMP-Activated Protein Kinase. Diabetes 2006, 55, 2688–2697. [Google Scholar] [CrossRef]
- Ellingsgaard, H.; Hauselmann, I.; Schuler, B.; Habib, A.M.; Baggio, L.L.; Meier, D.T.; Eppler, E.; Bouzakri, K.; Wueest, S.; Muller, Y.D.; et al. Interleukin-6 Enhances Insulin Secretion by Increasing Glucagon-like Peptide-1 Secretion from L Cells and Alpha Cells. Nat. Med. 2011, 17, 1481–1489. [Google Scholar] [CrossRef]
- Nielsen, A.R.; Hojman, P.; Erikstrup, C.; Fischer, C.P.; Plomgaard, P.; Mounier, R.; Mortensen, O.H.; Broholm, C.; Taudorf, S.; Krogh-Madsen, R.; et al. Association between Interleukin-15 and Obesity: Interleukin-15 as a Potential Regulator of Fat Mass. J. Clin. Endocrinol. Metab. 2008, 93, 4486–4493. [Google Scholar] [CrossRef]
- Quinn, L.S.; Anderson, B.G.; Strait-Bodey, L.; Stroud, A.M.; Argués, J.M. Oversecretion of Interleukin-15 from Skeletal Muscle Reduces Adiposity. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E191–E202. [Google Scholar] [CrossRef]
- Garneau, L.; Mulvihill, E.E.; Smith, S.R.; Sparks, L.M.; Aguer, C. Myokine Secretion Following an Aerobic Exercise Intervention in Individuals with Type 2 Diabetes with or without Exercise Resistance. Int. J. Mol. Sci. 2024, 25, 4889. [Google Scholar] [CrossRef]
- Banitalebi, E.; Kazemi, A.R.; Faramarzi, M.; Nasiri, S.; Haghighi, M.M. Effects of Sprint Interval or Combined Aerobic and Resistance Training on Myokines in Overweight Women with Type 2 Diabetes: A Randomized Controlled Trial. Life Sci. 2019, 217, 101–109. [Google Scholar] [CrossRef]
- Kim, W.K.; Choi, H.R.; Park, S.G.; Ko, Y.; Bae, K.H.; Lee, S.C. Myostatin Inhibits Brown Adipocyte Differentiation via Regulation of Smad3-Mediated β-Catenin Stabilization. Int. J. Biochem. Cell Biol. 2012, 44, 327–334. [Google Scholar] [CrossRef]
- Han, X.; Møller, L.L.V.; De Groote, E.; Bojsen-Møller, K.N.; Davey, J.; Henríquez-Olguin, C.; Li, Z.; Knudsen, J.R.; Jensen, T.E.; Madsbad, S.; et al. Mechanisms Involved in Follistatin-Induced Hypertrophy and Increased Insulin Action in Skeletal Muscle. J. Cachexia Sarcopenia Muscle 2019, 10, 1241–1257. [Google Scholar] [CrossRef]
- Brown, M.L.; Bonomi, L.; Ungerleider, N.; Zina, J.; Kimura, F.; Mukherjee, A.; Sidis, Y.; Schneyer, A. Follistatin and Follistatin Like-3 Differentially Regulate Adiposity and Glucose Homeostasis. Obesity 2011, 19, 1940–1949. [Google Scholar] [CrossRef]
- Brandt, C.; Hansen, R.H.; Hansen, J.B.; Olsen, C.H.; Galle, P.; Mandrup-Poulsen, T.; Gehl, J.; Pedersen, B.K.; Hojman, P. Over-Expression of Follistatin-like 3 Attenuates Fat Accumulation and Improves Insulin Sensitivity in Mice. Metabolism 2015, 64, 283–295. [Google Scholar] [CrossRef]
- Kurdiova, T.; Balaz, M.; Vician, M.; Maderova, D.; Vlcek, M.; Valkovic, L.; Srbecky, M.; Imrich, R.; Kyselovicova, O.; Belan, V.; et al. Effects of Obesity, Diabetes and Exercise on Fndc5 Gene Expression and Irisin Release in Human Skeletal Muscle and Adipose Tissue: In Vivo and in Vitro Studies. J. Physiol. 2014, 592, 1091–1107. [Google Scholar] [CrossRef]
- Moreno-Navarrete, J.M.; Ortega, F.; Serrano, M.; Guerra, E.; Pardo, G.; Tinahones, F.; Ricart, W.; Fernández-Real, J.M. Irisin Is Expressed and Produced by Human Muscle and Adipose Tissue in Association with Obesity and Insulin Resistance. J. Clin. Endocrinol. Metab. 2013, 98, E769–E778. [Google Scholar] [CrossRef] [PubMed]
- Norheim, F.; Langleite, T.M.; Hjorth, M.; Holen, T.; Kielland, A.; Stadheim, H.K.; Gulseth, H.L.; Birkeland, K.I.; Jensen, J.; Drevon, C.A. The Effects of Acute and Chronic Exercise on PGC-1α, Irisin and Browning of Subcutaneous Adipose Tissue in Humans. FEBS J. 2014, 281, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.D.; Boström, P.; O’Sullivan, J.F.; Schinzel, R.T.; Lewis, G.D.; Dejam, A.; Lee, Y.K.; Palma, M.J.; Calhoun, S.; Georgiadi, A.; et al. β-Aminoisobutyric Acid Induces Browning of White Fat and Hepatic β-Oxidation and Is Inversely Correlated with Cardiometabolic Risk Factors. Cell Metab. 2014, 19, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Barlow, J.P.; Solomon, T.P. Do Skeletal Muscle-Secreted Factors Influence the Function of Pancreatic β-Cells? Am. J. Physiol. Endocrinol. Metab. 2018, 314, E297–E307. [Google Scholar] [CrossRef]
- Lee, C.; Zeng, J.; Drew, B.G.; Sallam, T.; Martin-Montalvo, A.; Wan, J.; Kim, S.J.; Mehta, H.; Hevener, A.L.; De Cabo, R.; et al. The Mitochondrial-Derived Peptide MOTS-c Promotes Metabolic Homeostasis and Reduces Obesity and Insulin Resistance. Cell Metab. 2015, 21, 443–454. [Google Scholar] [CrossRef]
- Lee, C.; Kim, K.H.; Cohen, P. MOTS-c: A Novel Mitochondrial-Derived Peptide Regulating Muscle and Fat Metabolism. Free Radic. Biol. Med. 2016, 100, 182–187. [Google Scholar] [CrossRef]
- Münzberg, H.; Morrison, C.D. Structure, Production and Signaling of Leptin. Metabolism 2015, 64, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Pereira, S.; Cline, D.L.; Glavas, M.M.; Covey, S.D.; Kieffer, T.J. Tissue-Specific Effects of Leptin on Glucose and Lipid Metabolism. Endocr. Rev. 2021, 42, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Perakakis, N.; Farr, O.M.; Mantzoros, C.S. Leptin in Leanness and Obesity: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2021, 77, 745–760. [Google Scholar] [CrossRef]
- Suzuki, A.; Okamoto, S.; Lee, S.; Saito, K.; Shiuchi, T.; Minokoshi, Y. Leptin Stimulates Fatty Acid Oxidation and Peroxisome Proliferator-Activated Receptor α Gene Expression in Mouse C2C12 Myoblasts by Changing the Subcellular Localization of the α 2 Form of AMP-Activated Protein Kinase. Mol. Cell Biol. 2007, 27, 4317–4327. [Google Scholar] [CrossRef]
- Perry, R.J.; Wang, Y.; Cline, G.W.; Rabin-Court, A.; Song, J.D.; Dufour, S.; Zhang, X.M.; Petersen, K.F.; Shulman, G.I. Leptin Mediates a Glucose-Fatty Acid Cycle to Maintain Glucose Homeostasis in Starvation. Cell 2018, 172, 234–248.e17. [Google Scholar] [CrossRef]
- Myers, M.G.; Cowley, M.A.; Münzberg, H. Mechanisms of Leptin Action and Leptin Resistance. Annu. Rev. Physiol. 2008, 70, 537–556. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J.M.; Halaas, J.L. Leptin and the Regulation of Body Weight in Mammals. Nature 1998, 395, 763–770. [Google Scholar] [CrossRef]
- Spranger, J.; Kroke, A.; Möhlig, M.; Bergmann, M.M.; Ristow, M.; Boeing, H.; Pfeiffer, A.F.H. Adiponectin and Protection against Type 2 Diabetes Mellitus. Lancet 2003, 361, 226–228. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Minokoshi, Y.; Ito, Y.; Waki, H.; Uchida, S.; Yamashita, S.; Noda, M.; Kita, S.; Ueki, K.; et al. Adiponectin Stimulates Glucose Utilization and Fatty-Acid Oxidation by Activating AMP-Activated Protein Kinase. Nat. Med. 2002, 8, 1288–1295. [Google Scholar] [CrossRef]
- Kadowaki, T.; Yamauchi, T.; Kubota, N.; Hara, K.; Ueki, K.; Tobe, K. Adiponectin and Adiponectin Receptors in Insulin Resistance, Diabetes, and the Metabolic Syndrome. J. Clin. Investig. 2006, 116, 1784–1792. [Google Scholar] [CrossRef]
- Holland, W.L.; Adams, A.C.; Brozinick, J.T.; Bui, H.H.; Miyauchi, Y.; Kusminski, C.M.; Bauer, S.M.; Wade, M.; Singhal, E.; Cheng, C.C.; et al. An FGF21-Adiponectin-Ceramide Axis Controls Energy Expenditure and Insulin Action in Mice. Cell Metab. 2013, 17, 790–797. [Google Scholar] [CrossRef]
- Hung, J.; McQuillan, B.M.; Thompson, P.L.; Beilby, J.P. Circulating Adiponectin Levels Associate with Inflammatory Markers, Insulin Resistance and Metabolic Syndrome Independent of Obesity. Int. J. Obes. 2008, 32, 772–779. [Google Scholar] [CrossRef]
- Bokarewa, M.; Nagaev, I.; Dahlberg, L.; Smith, U.; Tarkowski, A. Resistin, an Adipokine with Potent Proinflammatory Properties. J. Immunol. 2005, 174, 5789–5795. [Google Scholar] [CrossRef] [PubMed]
- Kusminski, C.M.; Da Silva, N.F.; Creely, S.J.; Fisher, F.M.; Harte, A.L.; Baker, A.R.; Kumar, S.; McTernan, P.G. The in Vitro Effects of Resistin on the Innate Immune Signaling Pathway in Isolated Human Subcutaneous Adipocytes. J. Clin. Endocrinol. Metab. 2007, 92, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Park, S.E.; Rhee, E.J.; Lee, W.Y.; Kim, W.J.; Yoo, S.H.; Bae, J.C.; Choi, E.S.; Park, C.Y.; Oh, K.W.; Park, S.W.; et al. The Role of Serum Adipocyte Fatty Acid-Binding Protein on the Development of Metabolic Syndrome Is Independent of pro-Inflammatory Cytokines. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 525–532. [Google Scholar] [CrossRef]
- Nowak, C.; Sundström, J.; Gustafsson, S.; Giedraitis, V.; Lind, L.; Ingelsson, E.; Fall, T. Protein Biomarkers for Insulin Resistance and Type 2 Diabetes Risk in Two Large Community Cohorts. Diabetes 2016, 65, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Tso, A.W.K.; Xu, A.; Sham, P.C.; Wat, N.M.S.; Wang, Y.; Fong, C.H.Y.; Cheung, B.M.Y.; Janus, E.D.; Lam, K.S.L. Serum Adipocyte Fatty Acid-Binding Protein as a New Biomarker Predicting the Development of Type 2 Diabetes: A 10-Year Prospective Study in a Chinese Cohort. Diabetes Care 2007, 30, 2667–2672. [Google Scholar] [CrossRef]
- Fan, J.; Hu, J. Retinol Binding Protein 4 and Type 2 Diabetes: From Insulin Resistance to Pancreatic β-Cell Function. Endocrine 2024, 85, 1020–1034. [Google Scholar] [CrossRef]
- Ülgen, F.; Herder, C.; Kühn, M.C.; Willenberg, H.S.; Schott, M.; Scherbaum, W.A.; Schinner, S. Association of Serum Levels of Retinol-Binding Protein 4 with Male Sex but Not with Insulin Resistance in Obese Patients. Arch. Physiol. Biochem. 2010, 116, 57–62. [Google Scholar] [CrossRef]
- Oh, J.; Park, C.; Kim, S.; Kim, M.; Kim, C.S.; Jo, W.; Park, S.; Yi, G.S.; Park, J. High Levels of Intracellular Endotrophin in Adipocytes Mediate COPII Vesicle Supplies to Autophagosome to Impair Autophagic Flux and Contribute to Systemic Insulin Resistance in Obesity. Metabolism 2023, 145, 155629. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Park, J.; Gupta, O.T.; Holland, W.L.; Auerbach, P.; Zhang, N.; Goncalves Marangoni, R.; Nicoloro, S.M.; Czech, M.P.; Varga, J.; et al. Endotrophin Triggers Adipose Tissue Fibrosis and Metabolic Dysfunction. Nat. Commun. 2014, 5, 3485. [Google Scholar] [CrossRef] [PubMed]
- Karsdal, M.A.; Henriksen, K.; Genovese, F.; Leeming, D.J.; Nielsen, M.J.; Riis, B.J.; Christiansen, C.; Byrjalsen, I.; Schuppan, D. Serum Endotrophin Identifies Optimal Responders to PPARγ Agonists in Type 2 Diabetes. Diabetologia 2017, 60, 50–59. [Google Scholar] [CrossRef]
- Sparding, N.; Rasmussen, D.G.K.; Genovese, F.; Karsdal, M.A.; Hornum, M.; Feldt-Rasmussen, B.; Packington, R.; Selby, N.M. Circulating Levels of Endotrophin Are Prognostic for Long-Term Mortality after AKI. Kidney360 2022, 3, 809–817. [Google Scholar] [CrossRef]
- Nedeva, I.S.; Assyov, Y.; Karamfilova, V.; Vodenicharov, V.; Gerganova, A.; Hristova, J.; Kamenov, Z. Circulating Asprosin Concentrations in Patients with Obesity and Carbohydrate Disturbances. Horm. Metab. Res. 2022, 55, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Chen, C.; Zhou, N.; Fu, Y.; Cheng, X. Circulating Asprosin Concentrations Are Increased in Type 2 Diabetes Mellitus and Independently Associated with Fasting Glucose and Triglyceride. Clin. Chim. Acta 2019, 489, 183–188. [Google Scholar] [CrossRef]
- Lo, J.C.; Ljubicic, S.; Leibiger, B.; Kern, M.; Leibiger, I.B.; Moede, T.; Kelly, M.E.; Chatterjee Bhowmick, D.; Murano, I.; Cohen, P.; et al. Adipsin Is an Adipokine That Improves β Cell Function in Diabetes. Cell 2014, 158, 41–53. [Google Scholar] [CrossRef]
- Itani, S.I.; Ruderman, N.B.; Schmieder, F.; Boden, G. Lipid-Induced Insulin Resistance in Human Muscle Is Associated with Changes in Diacylglycerol, Protein Kinase C, and IκB-α. Diabetes 2002, 51, 2005–2011. [Google Scholar] [CrossRef]
- Perseghin, G.; Scifo, P.; De Cobelli, F.; Pagliato, E.; Battezzati, A.; Arcelloni, C.; Vanzulli, A.; Testolin, G.; Pozza, G.; Del Maschio, A.; et al. Intramyocellular Triglyceride Content Is a Determinant of in Vivo Insulin Resistance in Humans: A 1H-13C Nuclear Magnetic Resonance Spectroscopy Assessment in Offspring of Type 2 Diabetic Parents. Diabetes 1999, 48, 1600–1606. [Google Scholar] [CrossRef]
- Turpin, S.M.; Nicholls, H.T.; Willmes, D.M.; Mourier, A.; Brodesser, S.; Wunderlich, C.M.; Mauer, J.; Xu, E.; Hammerschmidt, P.; Brönneke, H.S.; et al. Obesity-Induced CerS6-Dependent C16:0 Ceramide Production Promotes Weight Gain and Glucose Intolerance. Cell Metab. 2014, 20, 678–686. [Google Scholar] [CrossRef]
- Raichur, S.; Brunner, B.; Bielohuby, M.; Hansen, G.; Pfenninger, A.; Wang, B.; Bruning, J.C.; Larsen, P.J.; Tennagels, N. The Role of C16:0 Ceramide in the Development of Obesity and Type 2 Diabetes: CerS6 Inhibition as a Novel Therapeutic Approach. Mol. Metab. 2019, 21, 36–50. [Google Scholar] [CrossRef]
- Chavez, J.A.; Knotts, T.A.; Wang, L.P.; Li, G.; Dobrowsky, R.T.; Florant, G.L.; Summers, S.A. A Role for Ceramide, but Not Diacylglycerol, in the Antagonism of Insulin Signal Transduction by Saturated Fatty Acids. J. Biol. Chem. 2003, 278, 10297–10303. [Google Scholar] [CrossRef]
- Stratford, S.; Hoehn, K.L.; Liu, F.; Summers, S.A. Regulation of Insulin Action by Ceramide: Dual Mechanisms Linking Ceramide Accumulation to the Inhibition of Akt/Protein Kinase B. J. Biol. Chem. 2004, 279, 36608–36615. [Google Scholar] [CrossRef] [PubMed]
- Haus, J.M.; Kashyap, S.R.; Kasumov, T.; Zhang, R.; Kelly, K.R.; Defronzo, R.A.; Kirwan, J.P. Plasma Ceramides Are Elevated in Obese Subjects with Type 2 Diabetes and Correlate with the Severity of Insulin Resistance. Diabetes 2009, 58, 337–343. [Google Scholar] [CrossRef]
- Kasumov, T.; Solomon, T.P.J.; Hwang, C.; Huang, H.; Haus, J.M.; Zhang, R.; Kirwan, J.P. Improved Insulin Sensitivity after Exercise Training Is Linked to Reduced Plasma C14:0 Ceramide in Obesity and Type 2 Diabetes. Obesity 2015, 23, 1414–1421. [Google Scholar] [CrossRef]
- Lee, J.Y.; Cho, H.K.; Kwon, Y.H. Palmitate Induces Insulin Resistance without Significant Intracellular Triglyceride Accumulation in HepG2 Cells. Metabolism 2010, 59, 927–934. [Google Scholar] [CrossRef] [PubMed]
- Fayyaz, S.; Henkel, J.; Japtok, L.; Krämer, S.; Damm, G.; Seehofer, D.; Püschel, G.P.; Kleuser, B. Involvement of Sphingosine 1-Phosphate in Palmitate-Induced Insulin Resistance of Hepatocytes via the S1P2 Receptor Subtype. Diabetologia 2014, 57, 373–382. [Google Scholar] [CrossRef]
- Wei, Y.; Wang, D.; Topczewski, F.; Pagliassotti, M.J. Saturated Fatty Acids Induce Endoplasmic Reticulum Stress and Apoptosis Independently of Ceramide in Liver Cells. Am. J. Physiol. Endocrinol. Metab. 2006, 291, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Wong, S.; Xie, W.; Lei, T.; Luo, Z. Palmitate Modulates Intracellular Signaling, Induces Endoplasmic Reticulum Stress, and Causes Apoptosis in Mouse 3T3-L1 and Rat Primary Preadipocytes. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E576–E586. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Lee, S.Y.; Bae, Y.S. Functional Roles of Sphingolipids in Immunity and Their Implication in Disease. Exp. Mol. Med. 2023, 55, 1110–1130. [Google Scholar] [CrossRef]
- Puig, N.; Estruch, M.; Jin, L.; Sanchez-quesada, J.L.; Benitez, S. The Role of Distinctive Sphingolipids in the Inflammatory and Apoptotic Effects of Electronegative LDL on Monocytes. Biomolecules 2019, 9, 300. [Google Scholar] [CrossRef]
- D’Souza, K.; Paramel, G.V.; Kienesberger, P.C. Lysophosphatidic Acid Signaling in Obesity and Insulin Resistance. Nutrients 2018, 10, 399. [Google Scholar] [CrossRef]
- Yore, M.M.; Syed, I.; Moraes-Vieira, P.M.; Zhang, T.; Herman, M.A.; Homan, E.A.; Patel, R.T.; Lee, J.; Chen, S.; Peroni, O.D.; et al. Discovery of a Class of Endogenous Mammalian Lipids with Anti-Diabetic and Anti-Inflammatory Effects. Cell 2014, 159, 318–332. [Google Scholar] [CrossRef]
- Albracht-Schulte, K.; Kalupahana, N.S.; Ramalingam, L.; Wang, S.; Rahman, S.M.; Robert-McComb, J.; Moustaid-Moussa, N. Omega-3 Fatty Acids in Obesity and Metabolic Syndrome: A Mechanistic Update. J. Nutr. Biochem. 2018, 58, 1–16. [Google Scholar] [CrossRef]
- Koves, T.R.; Ussher, J.R.; Noland, R.C.; Slentz, D.; Mosedale, M.; Ilkayeva, O.; Bain, J.; Stevens, R.; Dyck, J.R.B.; Newgard, C.B.; et al. Mitochondrial Overload and Incomplete Fatty Acid Oxidation Contribute to Skeletal Muscle Insulin Resistance. Cell Metab. 2008, 7, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.H.; Hoppel, C.L.; Lok, K.H.; Zhao, L.; Wong, S.W.; Minkler, P.E.; Hwang, D.H.; Newman, J.W.; Garvey, W.T. Plasma Acylcarnitine Profiles Suggest Incomplete Long-Chain Fatty Acid β-Oxidation and Altered Tricarboxylic Acid Cycle Activity in Type 2 Diabetic African-American Women. J. Nutr. 2009, 139, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Chimal-Vega, B.; Maldonado-Arvizu, J.E.; Avalos, A.D.H.; Díaz-Villanueva, J.F.; Avila-Barrientos, L.P.; González, V.G.G. Inter-Tissue Communication Mechanisms via Exosomes and Their Implications in Metabolic Diseases: Opportunities for Pharmacological Regulation. Future Pharmacol. 2025, 5, 11. [Google Scholar] [CrossRef]
- Guay, C.; Regazzi, R. Exosomes as New Players in Metabolic Organ Cross-Talk. Diabetes Obes. Metab. 2017, 19, 137–146. [Google Scholar] [CrossRef]
- Vechetti, I.J.; Peck, B.D.; Wen, Y.; Walton, R.G.; Valentino, T.R.; Alimov, A.P.; Dungan, C.M.; Van Pelt, D.W.; von Walden, F.; Alkner, B.; et al. Mechanical Overload-Induced Muscle-Derived Extracellular Vesicles Promote Adipose Tissue Lipolysis. FASEB J. 2021, 35, e21644. [Google Scholar] [CrossRef] [PubMed]
- Villagrán-Silva, F.; Loren, P.; Sandoval, C.; Lanas, F.; Salazar, L.A. Circulating MicroRNAs as Potential Biomarkers of Overweight and Obesity in Adults: A Narrative Review. Genes 2025, 16, 349. [Google Scholar] [CrossRef]
- Heyn, G.S.; Corrêa, L.H.; Magalhães, K.G. The Impact of Adipose Tissue-Derived MiRNAs in Metabolic Syndrome, Obesity, and Cancer. Front. Endocrinol. 2020, 11, 563816. [Google Scholar] [CrossRef]
- Al-Rawaf, H.A. Circulating MicroRNAs and Adipokines as Markers of Metabolic Syndrome in Adolescents with Obesity. Clin. Nutr. 2019, 38, 2231–2238. [Google Scholar] [CrossRef]
- Chen, Y.; Pan, R.; Pfeifer, A. Regulation of Brown and Beige Fat by MicroRNAs. Pharmacol. Ther. 2017, 170, 1–7. [Google Scholar] [CrossRef]
- Thomou, T.; Mori, M.A.; Dreyfuss, J.M.; Konishi, M.; Sakaguchi, M.; Wolfrum, C.; Rao, T.N.; Winnay, J.N.; Garcia-Martin, R.; Grinspoon, S.K.; et al. Adipose-Derived Circulating MiRNAs Regulate Gene Expression in Other Tissues. Nature 2017, 542, 450–455. [Google Scholar] [CrossRef]
- Crewe, C.; Scherer, P.E. Intercellular and Interorgan Crosstalk through Adipocyte Extracellular Vesicles. Rev. Endocr. Metab. Disord. 2022, 23, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Crewe, C.; Joffin, N.; Rutkowski, J.M.; Kim, M.; Zhang, F.; Towler, D.A.; Gordillo, R.; Scherer, P.E. An Endothelial-to-Adipocyte Extracellular Vesicle Axis Governed by Metabolic State. Cell 2018, 175, 695–708.e13. [Google Scholar] [CrossRef] [PubMed]
- Kahn, D.; Macias, E.; Zarini, S.; Garfield, A.; Zemski Berry, K.; Maclean, P.; Gerszten, R.E.; Libby, A.; Solt, C.; Schoen, J.; et al. Exploring Visceral and Subcutaneous Adipose Tissue Secretomes in Human Obesity: Implications for Metabolic Disease. Endocrinology 2022, 163, bqac140. [Google Scholar] [CrossRef]
- Fain, J.N.; Madan, A.K.; Hiler, M.L.; Cheema, P.; Bahouth, S.W. Comparison of the Release of Adipokines by Adipose Tissue, Adipose Tissue Matrix, and Adipocytes from Visceral and Subcutaneous Abdominal Adipose Tissues of Obese Humans. Endocrinology 2004, 145, 2273–2282. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscles, Exercise and Obesity: Skeletal Muscle as a Secretory Organ. Nat. Rev. Endocrinol. 2012, 8, 457–465. [Google Scholar] [CrossRef]
- Zhu, C.; Ding, X.; Chen, M.; Feng, J.; Zou, J.; Zhang, L. Exercise-Mediated Skeletal Muscle-Derived IL-6 Regulates Bone Metabolism: A New Perspective on Muscle–Bone Crosstalk. Biomolecules 2025, 15, 893. [Google Scholar] [CrossRef]
- Dong, Y.; Yuan, H.; Ma, G.; Cao, H. Bone-Muscle Crosstalk under Physiological and Pathological Conditions. Cell Mol. Life Sci. 2024, 81, 310. [Google Scholar] [CrossRef]
- Weitzmann, M.N.; Cenci, S.; Rifas, L.; Brown, C.; Pacifici, R. Interleukin-7 Stimulates Osteoclast Formation by up-Regulating the T-Cell Production of Soluble Osteoclastogenic Cytokines. Blood 2000, 96, 1873–1878. [Google Scholar] [CrossRef]
- Dankbar, B.; Fennen, M.; Brunert, D.; Hayer, S.; Frank, S.; Wehmeyer, C.; Beckmann, D.; Paruzel, P.; Bertrand, J.; Redlich, K.; et al. Myostatin Is a Direct Regulator of Osteoclast Differentiation and Its Inhibition Reduces Inflammatory Joint Destruction in Mice. Nat. Med. 2015, 21, 1085–1090. [Google Scholar] [CrossRef]
- Qin, Y.; Peng, Y.; Zhao, W.; Pan, J.; Ksiezak-Reding, H.; Cardozo, C.; Wu, Y.; Pajevic, P.D.; Bonewald, L.F.; Bauman, W.A.; et al. Myostatin Inhibits Osteoblastic Differentiation by Suppressing Osteocyte-Derived Exosomal MicroRNA-218: A Novel Mechanism in Muscle-Bone Communication. J. Biol. Chem. 2017, 292, 11021–11033. [Google Scholar] [CrossRef] [PubMed]
- Xiao, G.; Gopalakrishnan, R.; Jiang, D.; Reith, E.; Benson, M.D.; Franceschi, R.T. Bone Morphogenetic Proteins, Extracellular Matrix, and Mitogen-Activated Protein Kinase Signaling Pathways Are Required for Osteoblast-Specific Gene Expression and Differentiation in MC3T3-E1 Cells. J. Bone Miner. Res. 2002, 17, 101–110. [Google Scholar] [CrossRef]
- Yakar, S.; Rosen, C.J.; Beamer, W.G.; Ackert-Bicknell, C.L.; Wu, Y.; Liu, J.-L.; Ooi, G.T.; Setser, J.; Frystyk, J.; Boisclair, Y.R.; et al. Circulating Levels of IGF-1 Directly Regulate Bone Growth and Density. J. Clin. Investig. 2002, 110, 771–781. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Dutchak, P.A.; Wang, X.; Ding, X.; Wang, X.; Bookout, A.L.; Goetz, R.; Mohammadi, M.; Gerard, R.D.; Dechow, P.C.; et al. Fibroblast Growth Factor 21 Promotes Bone Loss by Potentiating the Effects of Peroxisome Proliferator-Activated Receptor γ. Proc. Natl. Acad. Sci. USA 2012, 109, 3143–3148. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Stanislaus, S.; Asuncion, F.; Niu, Q.T.; Chinookoswong, N.; Villasenor, K.; Wang, J.; Wong, P.; Boyce, R.; Dwyer, D.; et al. FGF21 Is Not a Major Mediator for Bone Homeostasis or Metabolic Actions of PPARα and PPARγ Agonists. J. Bone Miner. Res. 2017, 32, 834–845. [Google Scholar] [CrossRef]
- Colaianni, G.; Cuscito, C.; Mongelli, T.; Pignataro, P.; Buccoliero, C.; Liu, P.; Lu, P.; Sartini, L.; Comite, M.D.; Mori, G.; et al. The Myokine Irisin Increases Cortical Bone Mass. Proc. Natl. Acad. Sci. USA 2015, 112, 12157–12162. [Google Scholar] [CrossRef] [PubMed]
- Levinger, I.; Brennan-Speranza, T.C.; Zulli, A.; Parker, L.; Lin, X.; Lewis, J.R.; Yeap, B.B. Multifaceted Interaction of Bone, Muscle, Lifestyle Interventions and Metabolic and Cardiovascular Disease: Role of Osteocalcin. Osteoporos. Int. 2017, 28, 2265–2273. [Google Scholar] [CrossRef]
- Yi, H.S. Sclerostin as a Putative Myokine in Sarcopenia. Endocrinol. Metab. 2022, 37, 430–431. [Google Scholar] [CrossRef]
- Kim, J.A.; Roh, E.; Hong, S.H.; Lee, Y.B.; Kim, N.H.; Yoo, H.J.; Seo, J.A.; Kim, N.H.; Kim, S.G.; Baik, S.H.; et al. Association of Serum Sclerostin Levels with Low Skeletal Muscle Mass: The Korean Sarcopenic Obesity Study (KSOS). Bone 2019, 128, 115053. [Google Scholar] [CrossRef]
- Kirk, B.; Feehan, J.; Lombardi, G.; Duque, G. Muscle, Bone, and Fat Crosstalk: The Biological Role of Myokines, Osteokines, and Adipokines. Curr. Osteoporos. Rep. 2020, 18, 388–400. [Google Scholar] [CrossRef] [PubMed]
- Scheller, E.L.; Doucette, C.R.; Learman, B.S.; Cawthorn, W.P.; Khandaker, S.; Schell, B.; Wu, B.; Ding, S.Y.; Bredella, M.A.; Fazeli, P.K.; et al. Region-Specific Variation in the Properties of Skeletal Adipocytes Reveals Regulated and Constitutive Marrow Adipose Tissues. Nat. Commun. 2015, 6, 7808. [Google Scholar] [CrossRef]
- Li, F.; Li, Y.; Duan, Y.; Hu, C.A.A.; Tang, Y.; Yin, Y. Myokines and Adipokines: Involvement in the Crosstalk between Skeletal Muscle and Adipose Tissue. Cytokine Growth Factor. Rev. 2017, 33, 73–82. [Google Scholar] [CrossRef]
- Sirico, F.; Bianco, A.; D’Alicandro, G.; Castaldo, C.; Montagnani, S.; Spera, R.; Di Meglio, F.; Nurzynska, D. Effects of Physical Exercise on Adiponectin, Leptin, and Inflammatory Markers in Childhood Obesity: Systematic Review and Meta-Analysis. Child. Obes. 2018, 14, 207. [Google Scholar] [CrossRef]
- Becic, T.; Studenik, C.; Hoffmann, G. Exercise Increases Adiponectin and Reduces Leptin Levels in Prediabetic and Diabetic Individuals: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Med. Sci. 2018, 6, 97. [Google Scholar] [CrossRef] [PubMed]
- De Micheli, A.J.; Spector, J.A.; Elemento, O.; Cosgrove, B.D. A Reference Single-Cell Transcriptomic Atlas of Human Skeletal Muscle Tissue Reveals Bifurcated Muscle Stem Cell Populations. Skelet. Muscle 2020, 10, 19. [Google Scholar] [CrossRef]
- Martínez Mir, C.; Pisterzi, P.; De Poorter, I.; Rilou, M.; van Kranenburg, M.; Heijs, B.; Alemany, A.; Sage, F.; Geijsen, N. Spatial Multi-Omics in Whole Skeletal Muscle Reveals Complex Tissue Architecture. Commun. Biol. 2024, 7, 1272. [Google Scholar] [CrossRef]
- Zhang, H.; Lu, K.H.; Ebbini, M.; Huang, P.; Lu, H.; Li, L. Mass Spectrometry Imaging for Spatially Resolved Multi-Omics Molecular Mapping. npj Imaging 2024, 2, 20. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.; Ramírez-Pardo, I.; Isern, J.; An, J.; Perdiguero, E.; Serrano, A.L.; Li, J.; García-Domínguez, E.; Segalés, J.; Guo, P.; et al. Multimodal Cell Atlas of the Ageing Human Skeletal Muscle. Nature 2024, 629, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Massier, L.; Jalkanen, J.; Elmastas, M.; Zhong, J.; Wang, T.; Nono Nankam, P.A.; Frendo-Cumbo, S.; Bäckdahl, J.; Subramanian, N.; Sekine, T.; et al. An Integrated Single Cell and Spatial Transcriptomic Map of Human White Adipose Tissue. Nat. Commun. 2023, 14, 1438. [Google Scholar] [CrossRef] [PubMed]
- Kedlian, V.R.; Wang, Y.; Liu, T.; Chen, X.; Bolt, L.; Tudor, C.; Shen, Z.; Fasouli, E.S.; Prigmore, E.; Kleshchevnikov, V.; et al. Human Skeletal Muscle Aging Atlas. Nat. Aging 2024, 4, 727–744. [Google Scholar] [CrossRef]
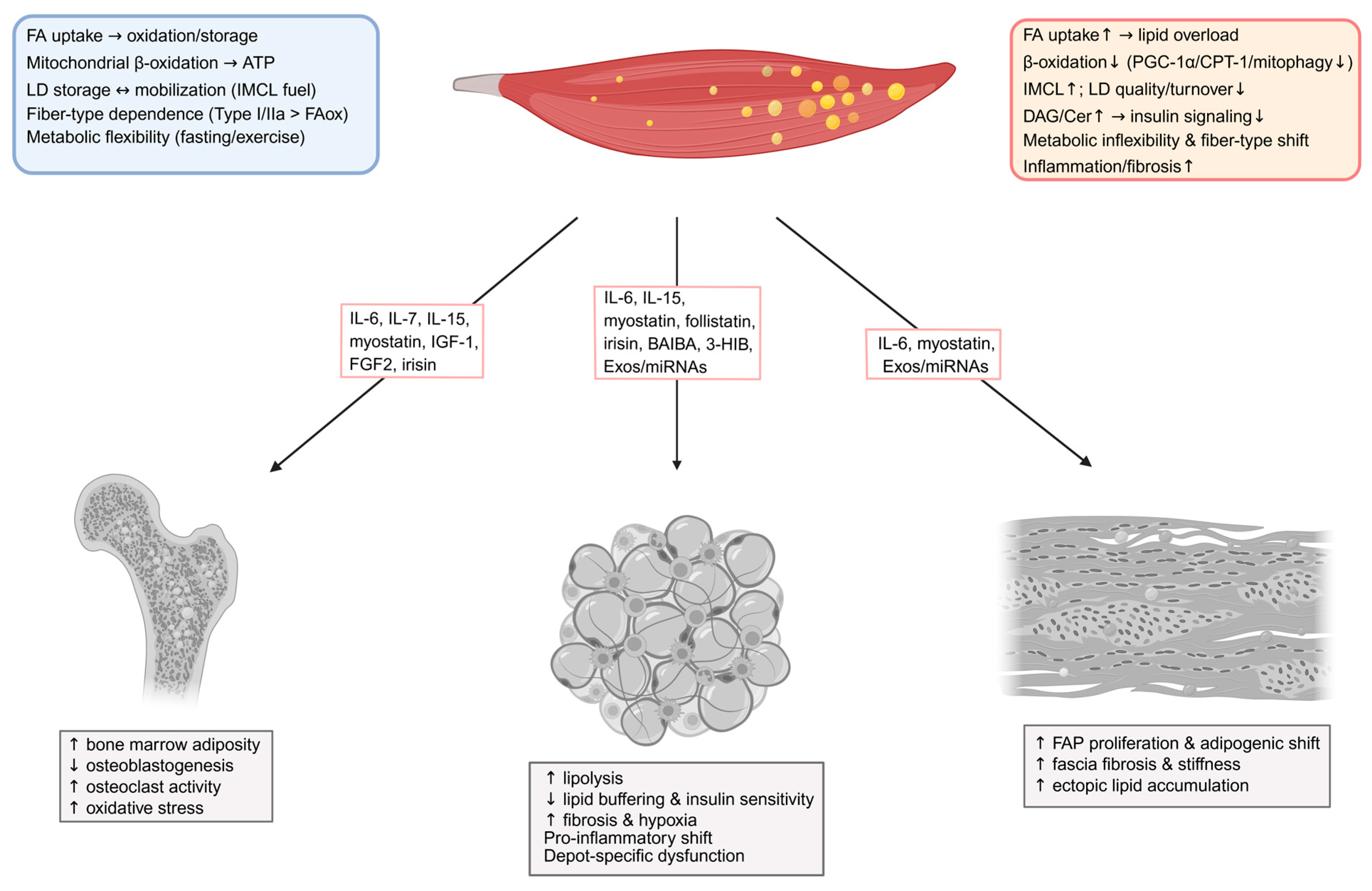
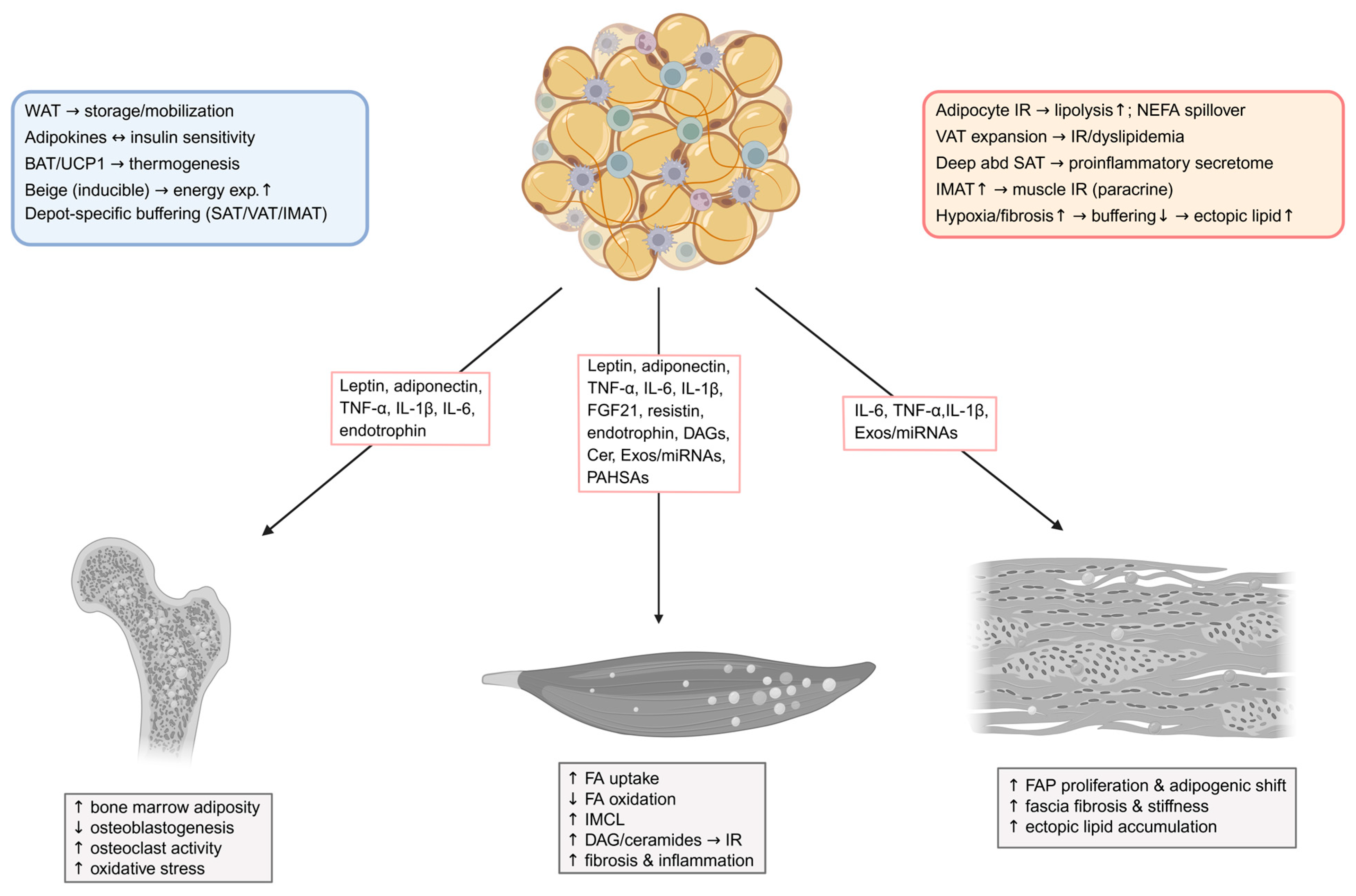
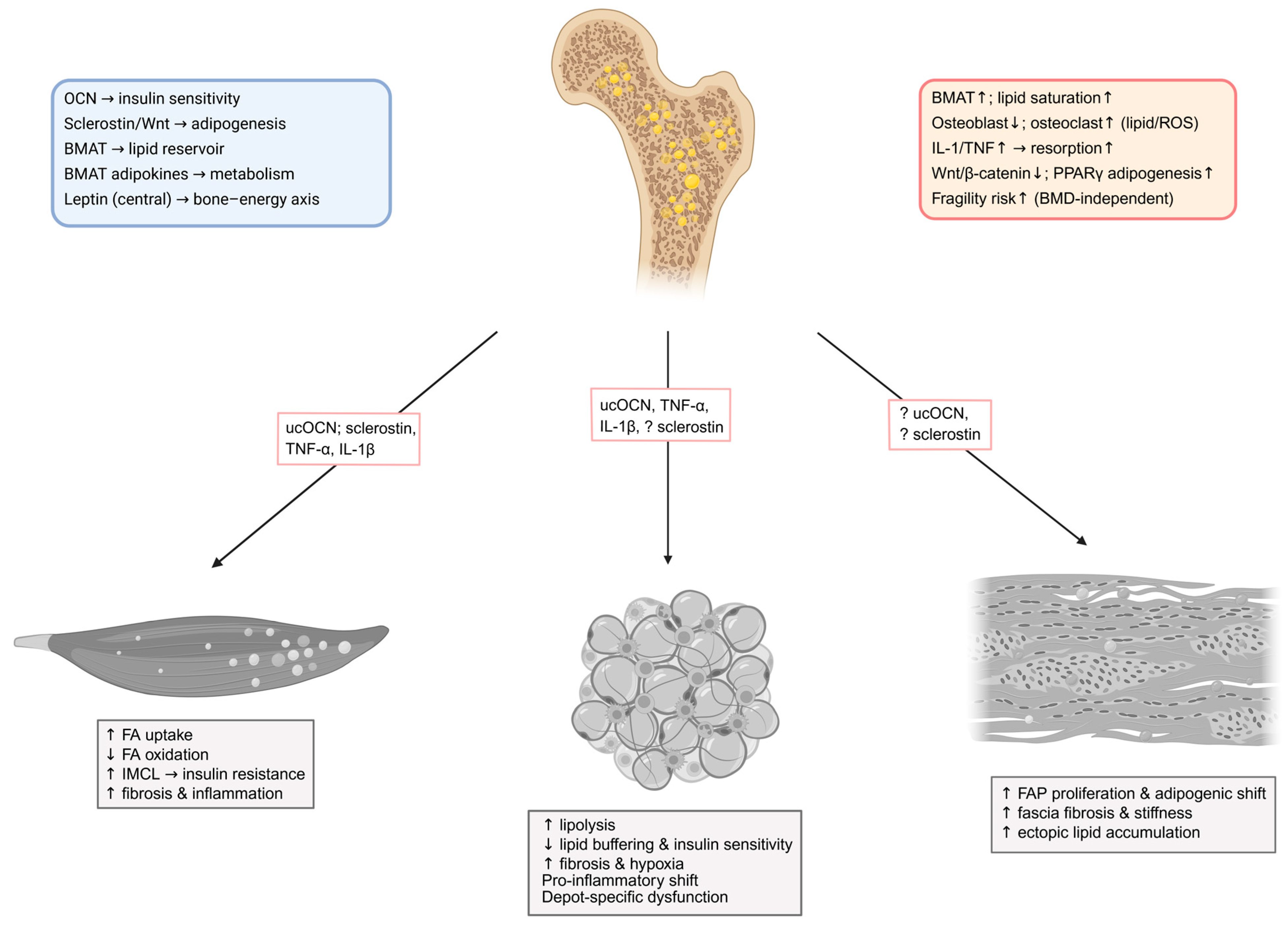
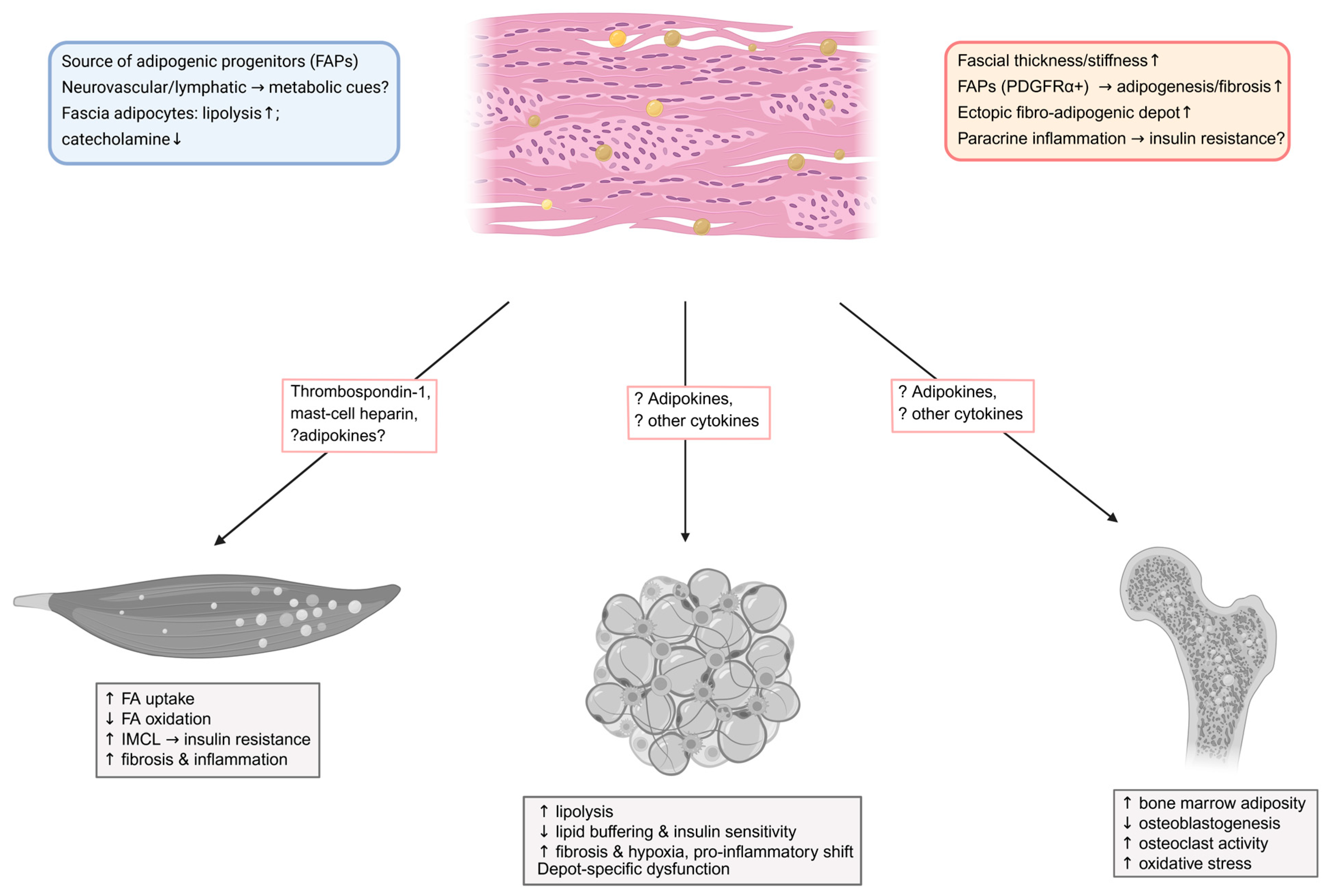
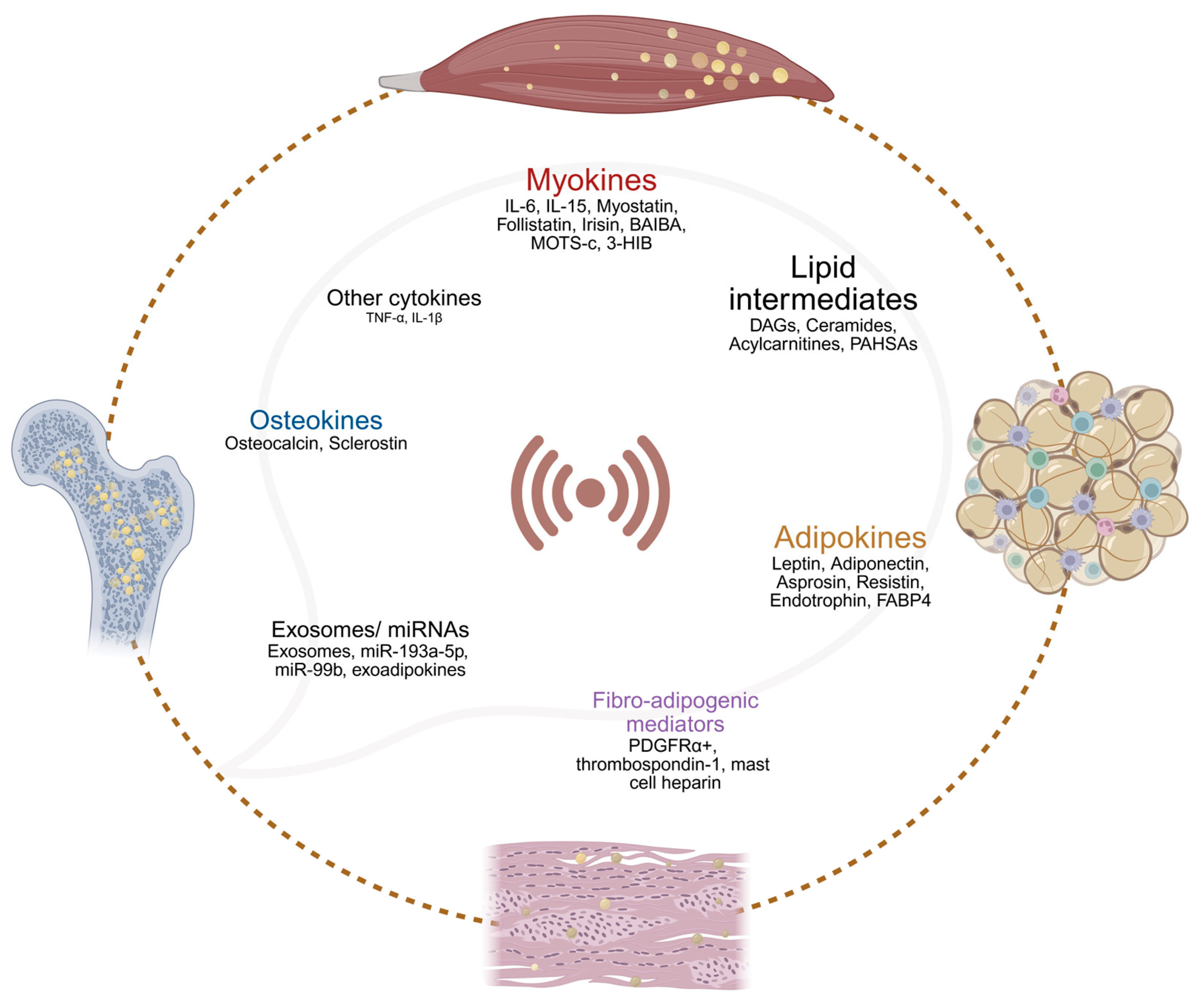
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pollak, N.; Janežič, E.G.; Šink, Ž.; Ugwoke, C.K. Crosstalk Between Skeletal Muscle and Proximal Connective Tissues in Lipid Dysregulation in Obesity and Type 2 Diabetes. Metabolites 2025, 15, 581. https://doi.org/10.3390/metabo15090581
Pollak N, Janežič EG, Šink Ž, Ugwoke CK. Crosstalk Between Skeletal Muscle and Proximal Connective Tissues in Lipid Dysregulation in Obesity and Type 2 Diabetes. Metabolites. 2025; 15(9):581. https://doi.org/10.3390/metabo15090581
Chicago/Turabian StylePollak, Nataša, Efua Gyakye Janežič, Žiga Šink, and Chiedozie Kenneth Ugwoke. 2025. "Crosstalk Between Skeletal Muscle and Proximal Connective Tissues in Lipid Dysregulation in Obesity and Type 2 Diabetes" Metabolites 15, no. 9: 581. https://doi.org/10.3390/metabo15090581
APA StylePollak, N., Janežič, E. G., Šink, Ž., & Ugwoke, C. K. (2025). Crosstalk Between Skeletal Muscle and Proximal Connective Tissues in Lipid Dysregulation in Obesity and Type 2 Diabetes. Metabolites, 15(9), 581. https://doi.org/10.3390/metabo15090581





