Gut Microbiota in Acute Myeloid Leukemia: From Biomarkers to Interventions
Abstract
1. Acute Myeloid Leukemia (AML) and Gut Microbiota
1.1. AML
1.2. Gut Microbiota in AML
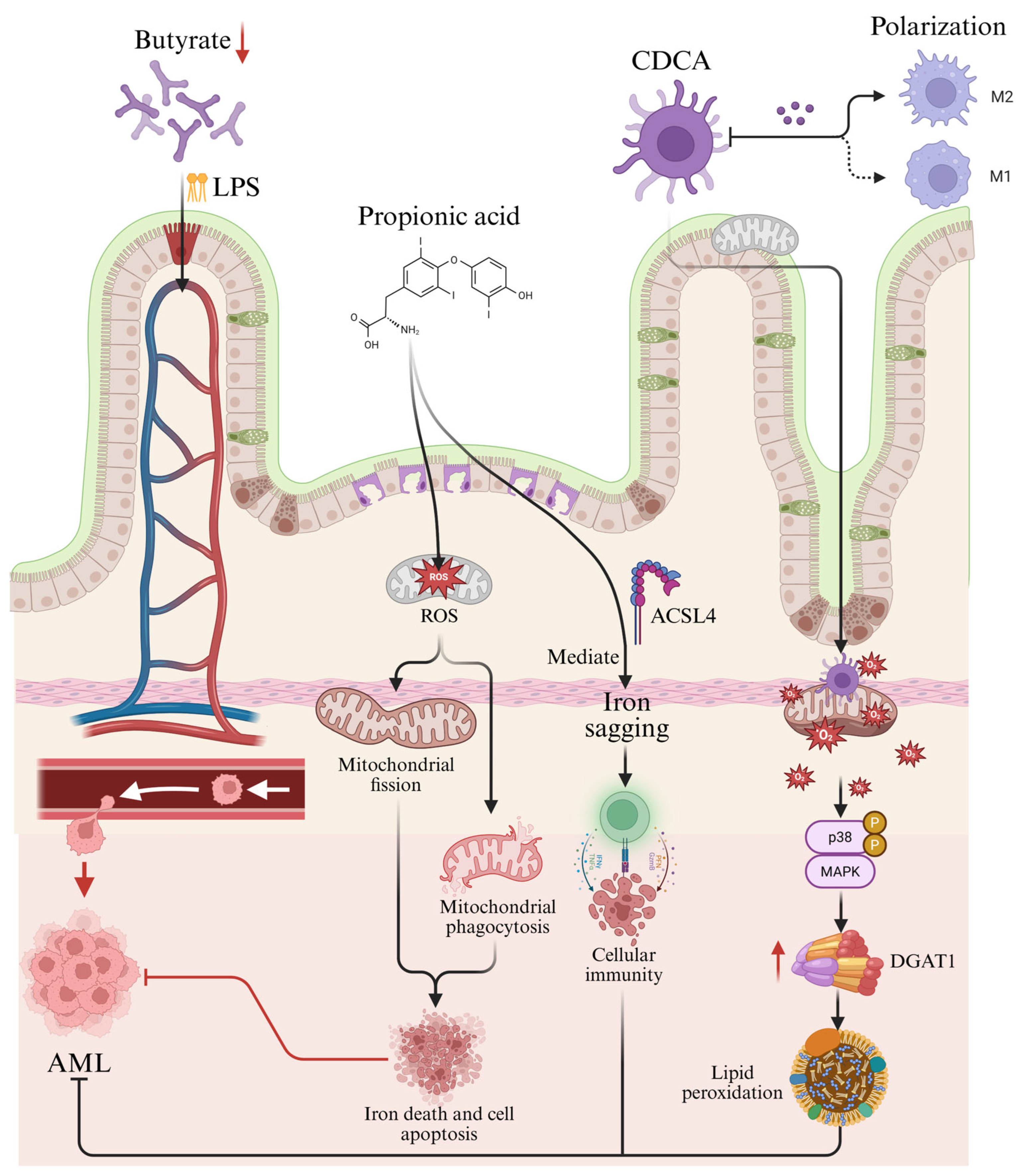
2. Potential Mechanisms of Gut Microbiota in AML Pathogenesis
3. Gut Microbiota in Biomarker Hunting of AML
3.1. Gut Microbiota Composition
3.2. Gut Microbial Metabolic Products
4. Prognosis and Intervention
4.1. Gut Microbiota in Prognosis of AML
| Class | Study Subjects | Therapeutic Interventions | Intervention-Associated Alterations | Reference |
|---|---|---|---|---|
| Population Studies | AML patients | Intensive chemotherapy |
| [16] |
| AML patients | Chemotherapy |
| [20] | |
| AML patients | Induction chemotherapy |
| [21] | |
| AML patients | Stem cell transplant |
| [27] | |
| AML patients | Post-chemotherapy tigecycline therapy |
| [28] | |
| AML patients, WT mice | Intensive chemotherapy |
| [29] | |
| AML patients | CPX-352 |
| [31] | |
| AML patients | Induction Therapy |
| [32] | |
| AML patients | Induction therapy, subcutaneous granulocyte colony-stimulating factor therapy, intensive chemotherapy |
| [33] | |
| Animal Experiment | AML mice | Curcumin |
| [30] |
4.2. Gut Microbiota in the Intervention of AML
5. Perspectives and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lagunas-Rangel, F.A.; Chávez-Valencia, V.; Gómez-Guijosa, M.Á.; Cortes-Penagos, C. Acute Myeloid Leukemia-Genetic Alterations and Their Clinical Prognosis. Int. J. Hematol. Oncol. Stem Cell Res. 2017, 11, 328–339. [Google Scholar]
- Sasaki, K.; Ravandi, F.; Kadia, T.M.; DiNardo, C.D.; Short, N.J.; Borthakur, G.; Jabbour, E.; Kantarjian, H.M. De novo acute myeloid leukemia: A population-based study of outcome in the United States based on the Surveillance, Epidemiology, and End Results (SEER) database, 1980 to 2017. Cancer 2021, 127, 2049–2061. [Google Scholar] [CrossRef]
- Lachowiez, C.A.; Loghavi, S.; Kadia, T.M.; Daver, N.; Borthakur, G.; Pemmaraju, N.; Naqvi, K.; Alvarado, Y.; Yilmaz, M.; Short, N.; et al. Outcomes of older patients with NPM1-mutated AML: Current treatments and the promise of venetoclax-based regimens. Blood Adv. 2020, 4, 1311–1320. [Google Scholar] [CrossRef]
- Mims, A.S.; Kohlschmidt, J.; Borate, U.; Blachly, J.S.; Orwick, S.; Eisfeld, A.-K.; Papaioannou, D.; Nicolet, D.; Mrόzek, K.; Stein, E.; et al. A precision medicine classification for treatment of acute myeloid leukemia in older patients. J. Hematol. Oncol. 2021, 14, 96. [Google Scholar] [CrossRef] [PubMed]
- Bennett, J.M.; Catovsky, D.; Daniel, M.T.; Flandrin, G.; Galton, D.A.; Gralnick, H.R.; Sultan, C. Proposals for the classification of the acute leukaemias. French-American-British (FAB) co-operative group. Br. J. Haematol. 1976, 33, 451–458. [Google Scholar] [CrossRef]
- Khoury, J.D.; Solary, E.; Abla, O.; Akkari, Y.; Alaggio, R.; Apperley, J.F.; Bejar, R.; Berti, E.; Busque, L.; Chan, J.K.C.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia 2022, 36, 1703–1719. [Google Scholar] [CrossRef]
- Döhner, H.; Wei, A.H.; Appelbaum, F.R.; Craddock, C.; DiNardo, C.D.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Godley, L.A.; Hasserjian, R.P.; et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood 2022, 140, 1345–1377. [Google Scholar] [CrossRef]
- Eisfeld, A.-K.; Mardis, E.R. Acute Myeloid Leukemia Genomics: Impact on Care and Remaining Challenges. Clin. Chem. 2024, 70, 4–12. [Google Scholar] [CrossRef] [PubMed]
- Awada, H.; Mustafa Ali, M.K.; Thapa, B.; Awada, H.; Seymour, L.; Liu, L.; Gurnari, C.; Kishtagari, A.; Wang, E.; Baer, M.R. A Focus on Intermediate-Risk Acute Myeloid Leukemia: Sub-Classification Updates and Therapeutic Challenges. Cancers 2022, 14, 4166. [Google Scholar] [CrossRef] [PubMed]
- Sallman, D.A.; McLemore, A.F.; Aldrich, A.L.; Komrokji, R.S.; McGraw, K.L.; Dhawan, A.; Geyer, S.; Hou, H.-A.; Eksioglu, E.A.; Sullivan, A.; et al. TP53 mutations in myelodysplastic syndromes and secondary AML confer an immunosuppressive phenotype. Blood 2020, 136, 2812–2823. [Google Scholar] [CrossRef]
- Zhang, J.; Gu, Y.; Chen, B. Mechanisms of drug resistance in acute myeloid leukemia. Onco Targets Ther. 2019, 12, 1937–1945. [Google Scholar] [CrossRef]
- Takiishi, T.; Fenero, C.I.M.; Câmara, N.O.S. Intestinal barrier and gut microbiota: Shaping our immune responses throughout life. Tissue Barriers 2017, 5, e1373208. [Google Scholar] [CrossRef]
- Fan, Y.; Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 2021, 19, 55–71. [Google Scholar] [CrossRef]
- Qiao, S.; Liu, C.; Sun, L.; Wang, T.; Dai, H.; Wang, K.; Bao, L.; Li, H.; Wang, W.; Liu, S.-J.; et al. Gut Parabacteroides merdae protects against cardiovascular damage by enhancing branched-chain amino acid catabolism. Nat. Metab. 2022, 4, 1271–1286. [Google Scholar] [CrossRef]
- Wang, R.; Yang, X.; Liu, J.; Zhong, F.; Zhang, C.; Chen, Y.; Sun, T.; Ji, C.; Ma, D. Gut microbiota regulates acute myeloid leukaemia via alteration of intestinal barrier function mediated by butyrate. Nat. Commun. 2022, 13, 2522. [Google Scholar] [CrossRef] [PubMed]
- Pötgens, S.A.; Lecop, S.; Havelange, V.; Li, F.; Neyrinck, A.M.; Neveux, N.; Maertens, J.; Walter, J.; Schoemans, H.; Delzenne, N.M.; et al. Gut microbiota alterations induced by intensive chemotherapy in acute myeloid leukaemia patients are associated with gut barrier dysfunction and body weight loss. Clin. Nutr. 2023, 42, 2214–2228. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wei, Y.; Jia, W.; Can, C.; Wang, R.; Yang, X.; Gu, C.; Liu, F.; Ji, C.; Ma, D. Chenodeoxycholic acid suppresses AML progression through promoting lipid peroxidation via ROS/p38 MAPK/DGAT1 pathway and inhibiting M2 macrophage polarization. Redox Biol. 2022, 56, 102452. [Google Scholar] [CrossRef]
- Wei, Y.; Liu, W.; Wang, R.; Chen, Y.; Liu, J.; Guo, X.; Can, C.; Yang, X.; Wang, D.; Hu, X.; et al. Propionate promotes ferroptosis and apoptosis through mitophagy and ACSL4-mediated ferroptosis elicits anti-leukemia immunity. Free Radic. Biol. Med. 2024, 213, 36–51. [Google Scholar] [CrossRef]
- de Martel, C.; Ferlay, J.; Franceschi, S.; Vignat, J.; Bray, F.; Forman, D.; Plummer, M. Global burden of cancers attributable to infections in 2008: A review and synthetic analysis. Lancet Oncol. 2012, 13, 607–615. [Google Scholar] [CrossRef]
- Xu, J.; Kang, Y.; Zhong, Y.; Ye, W.; Sheng, T.; Wang, Q.; Zheng, J.; Yang, Q.; Yi, P.; Li, Z. Alteration of gut microbiome and correlated amino acid metabolism are associated with acute myelocytic leukemia carcinogenesis. Cancer Med. 2023, 12, 16431–16443. [Google Scholar] [CrossRef] [PubMed]
- Rattanathammethee, T.; Tuitemwong, P.; Thiennimitr, P.; Sarichai, P.; Na Pombejra, S.; Piriyakhuntorn, P.; Hantrakool, S.; Chai-Adisaksopha, C.; Rattarittamrong, E.; Tantiworawit, A.; et al. Gut microbiota profiles of treatment-naïve adult acute myeloid leukemia patients with neutropenic fever during intensive chemotherapy. PLoS ONE 2020, 15, e0236460. [Google Scholar] [CrossRef]
- Wu, B.; Xu, Y.; Tang, M.; Jiang, Y.; Zhang, T.; Huang, L.; Wang, S.; Hu, Y.; Zhou, K.; Zhang, X.; et al. A Metabolome and Microbiome Analysis of Acute Myeloid Leukemia: Insights into the Carnosine-Histidine Metabolic Pathway. Toxics 2023, 12, 14. [Google Scholar] [CrossRef]
- Pötgens, S.A.; Havelange, V.; Lecop, S.; Li, F.; Neyrinck, A.M.; Bindels, F.; Neveux, N.; Demoulin, J.-B.; Moors, I.; Kerre, T.; et al. Gut microbiome alterations at acute myeloid leukemia diagnosis are associated with muscle weakness and anorexia. Haematologica 2024, 109, 3194–3208. [Google Scholar] [CrossRef]
- Yu, D.; Yu, X.; Ye, A.; Xu, C.; Li, X.; Geng, W.; Zhu, L. Profiling of gut microbial dysbiosis in adults with myeloid leukemia. FEBS Open Bio 2021, 11, 2050–2059. [Google Scholar] [CrossRef]
- Recharla, N.; Geesala, R.; Shi, X.-Z. Gut Microbial Metabolite Butyrate and Its Therapeutic Role in Inflammatory Bowel Disease: A Literature Review. Nutrients 2023, 15, 2275. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.K.; Millman, S.E.; Zhang, L. Metabolism in acute myeloid leukemia: Mechanistic insights and therapeutic targets. Blood 2023, 141, 1119–1135. [Google Scholar] [CrossRef] [PubMed]
- Rashidi, A.; Ebadi, M.; Rehman, T.U.; Elhusseini, H.; Halaweish, H.F.; Kaiser, T.; Holtan, S.G.; Khoruts, A.; Weisdorf, D.J.; Staley, C. Lasting shift in the gut microbiota in patients with acute myeloid leukemia. Blood Adv. 2022, 6, 3451–3457. [Google Scholar] [CrossRef] [PubMed]
- Zhong, F.-L.; He, J.-J.; Bai, K.-H.; Shao, R.-N.; Wu, G.-Y.; Tian, X.-P.; Wang, D.-W.; Dai, Y.-J.; Chen, S.-L. Tigecycline-induced coagulation gene prognostic prediction model and intestinal flora signature in AML. Front. Immunol. 2024, 15, 1486592. [Google Scholar] [CrossRef]
- Hueso, T.; Ekpe, K.; Mayeur, C.; Gatse, A.; Joncquel-Chevallier Curt, M.; Gricourt, G.; Rodriguez, C.; Burdet, C.; Ulmann, G.; Neut, C.; et al. Impact and consequences of intensive chemotherapy on intestinal barrier and microbiota in acute myeloid leukemia: The role of mucosal strengthening. Gut Microbes 2020, 12, 1800897. [Google Scholar] [CrossRef]
- Liu, J.; Luo, W.; Chen, Q.; Chen, X.; Zhou, G.; Sun, H. Curcumin sensitizes response to cytarabine in acute myeloid leukemia by regulating intestinal microbiota. Cancer Chemother. Pharmacol. 2022, 89, 243–253. [Google Scholar] [CrossRef]
- Renga, G.; Nunzi, E.; Stincardini, C.; Pariano, M.; Puccetti, M.; Pieraccini, G.; Di Serio, C.; Fraziano, M.; Poerio, N.; Oikonomou, V.; et al. CPX-351 exploits the gut microbiota to promote mucosal barrier function, colonization resistance, and immune homeostasis. Blood 2024, 143, 1628–1645. [Google Scholar] [CrossRef]
- Galloway-Peña, J.R.; Shi, Y.; Peterson, C.B.; Sahasrabhojane, P.; Gopalakrishnan, V.; Brumlow, C.E.; Daver, N.G.; Alfayez, M.; Boddu, P.C.; Khan, M.A.W.; et al. Gut Microbiome Signatures Are Predictive of Infectious Risk Following Induction Therapy for Acute Myeloid Leukemia. Clin. Infect. Dis. 2020, 71, 63–71. [Google Scholar] [CrossRef]
- Salvestrini, V.; Conti, G.; D’Amico, F.; Cristiano, G.; Candela, M.; Cavo, M.; Turroni, S.; Curti, A. Gut Microbiome as a Potential Marker of Hematologic Recovery Following Induction Therapy in Acute Myeloid Leukemia Patients. Cancer Med. 2025, 14, e70501. [Google Scholar] [CrossRef] [PubMed]
- Yadegar, A.; Bar-Yoseph, H.; Monaghan, T.M.; Pakpour, S.; Severino, A.; Kuijper, E.J.; Smits, W.K.; Terveer, E.M.; Neupane, S.; Nabavi-Rad, A.; et al. Fecal microbiota transplantation: Current challenges and future landscapes. Clin. Microbiol. Rev. 2024, 37, e0006022. [Google Scholar] [CrossRef] [PubMed]
- Rashidi, A.; Ebadi, M.; Rehman, T.U.; Elhusseini, H.; Kazadi, D.; Halaweish, H.; Khan, M.H.; Hoeschen, A.; Cao, Q.; Luo, X.; et al. Randomized Double-Blind Phase II Trial of Fecal Microbiota Transplantation Versus Placebo in Allogeneic Hematopoietic Cell Transplantation and AML. J. Clin. Oncol. 2023, 41, 5306–5319. [Google Scholar] [CrossRef] [PubMed]
- Malard, F.; Vekhoff, A.; Lapusan, S.; Isnard, F.; D’Incan-Corda, E.; Rey, J.; Saillard, C.; Thomas, X.; Ducastelle-Lepretre, S.; Paubelle, E.; et al. Gut microbiota diversity after autologous fecal microbiota transfer in acute myeloid leukemia patients. Nat. Commun. 2021, 12, 3084. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Ishikawa, D.; Nomura, K.; Fukuda, N.; Haraikawa, M.; Haga, K.; Shibuya, T.; Mita, T.; Nagahara, A. Donor Screening Revisions of Fecal Microbiota Transplantation in Patients with Ulcerative Colitis. J. Clin. Med. 2022, 11, 1055. [Google Scholar] [CrossRef]
- Lopetuso, L.R.; Deleu, S.; Godny, L.; Petito, V.; Puca, P.; Facciotti, F.; Sokol, H.; Ianiro, G.; Masucci, L.; Abreu, M.; et al. The first international Rome consensus conference on gut microbiota and faecal microbiota transplantation in inflammatory bowel disease. Gut 2023, 72, 1642–1650. [Google Scholar] [CrossRef]
- Nooij, S.; Ducarmon, Q.R.; Laros, J.F.J.; Zwittink, R.D.; Norman, J.M.; Smits, W.K.; Verspaget, H.W.; Keller, J.J.; Terveer, E.M.; Kuijper, E.J. Fecal Microbiota Transplantation Influences Procarcinogenic Escherichia coli in Recipient Recurrent Clostridioides difficile Patients. Gastroenterology 2021, 161, 1218–1228.e5. [Google Scholar] [CrossRef]
| Study Subjects | Compositional Changes in Gut Microbiota | Biomarker Gut Microbiota | Reference |
|---|---|---|---|
| AML patients, WT mice |
| Low diversity of gut microbiota and reduced Faecalibacterium play an important role in the progression of AML. | [15] |
| AML patients |
| Firmicutes, Bacteroidetes, Collinsella, Coriobacteriaceae | [20] |
| AML patients |
| Firmicutes | [21] |
| AML model mice, AML patients |
| Firmicutes, Bacteroidetes | [22] |
| AML patients |
| Eubacterium eligens, Blautia, Parabacteroides, Prevotella, Phenylacetate, and Hippurate | [23] |
| AML patients |
| Actinobacteria, Acidobacteria, Chloroflexi, Tenericutes, Streptococcus, Megamonas, LachnospiraceaeNC2004 group, Prevotella 9, Sphingomonas, Lysobacyer, Helicobacter, Lactobacillus, Enterococcus | [24] |
| Class | Study Subjects | Alterations in Gut Microbiota-Derived Metabolites | Biomarker Metabolites | Reference |
|---|---|---|---|---|
| Population Studies | AML patients, Murine AML cell line, WT mice |
| LPS, butyrate | [15] |
| AML patients, Human AML cell line |
| CDCA | [17] | |
| AML patients |
| hydroxypropionyl-hydroxyproline, prolyl-tyrosine and tyrosyl-proline | [20] | |
| AML patients |
| L-histidine, myostatin | [22] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ji, M.; Ji, M.; Zhong, Y.; Shao, L. Gut Microbiota in Acute Myeloid Leukemia: From Biomarkers to Interventions. Metabolites 2025, 15, 568. https://doi.org/10.3390/metabo15090568
Ji M, Ji M, Zhong Y, Shao L. Gut Microbiota in Acute Myeloid Leukemia: From Biomarkers to Interventions. Metabolites. 2025; 15(9):568. https://doi.org/10.3390/metabo15090568
Chicago/Turabian StyleJi, Meifen, Meixia Ji, Yebo Zhong, and Lewen Shao. 2025. "Gut Microbiota in Acute Myeloid Leukemia: From Biomarkers to Interventions" Metabolites 15, no. 9: 568. https://doi.org/10.3390/metabo15090568
APA StyleJi, M., Ji, M., Zhong, Y., & Shao, L. (2025). Gut Microbiota in Acute Myeloid Leukemia: From Biomarkers to Interventions. Metabolites, 15(9), 568. https://doi.org/10.3390/metabo15090568







