Ethanolic Extract of Artemisia vulgaris Leaf Promotes Apoptotic Cell Death in Non-Small-Cell Lung Carcinoma A549 Cells through Inhibition of the Wnt Signaling Pathway
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Plant Sample and Extract Preparation
2.2.2. Cell Culture
2.2.3. GC-MS Analysis
2.2.4. Cell Viability Assay
2.2.5. Morphological Evaluation
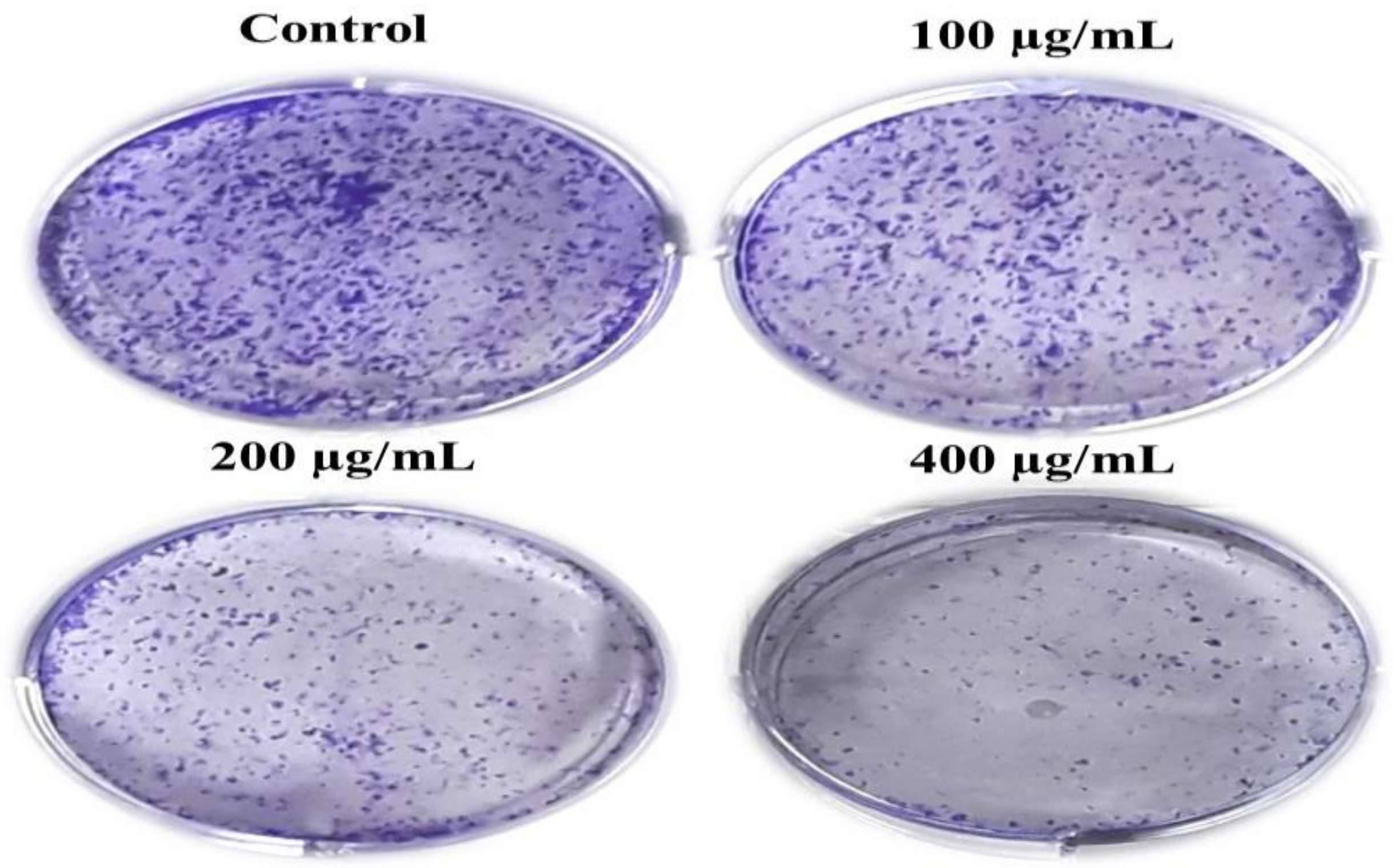
2.2.6. Colony Formation Assay
2.2.7. Hoechst/PI Staining
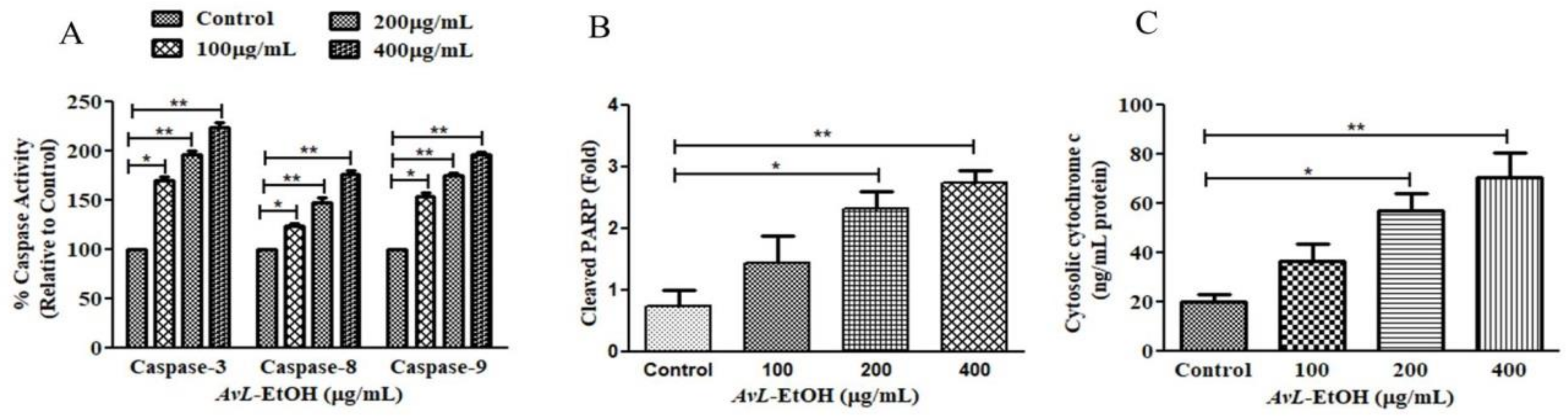
2.2.8. Measurement of Caspase-8, -9, and -3 Activity
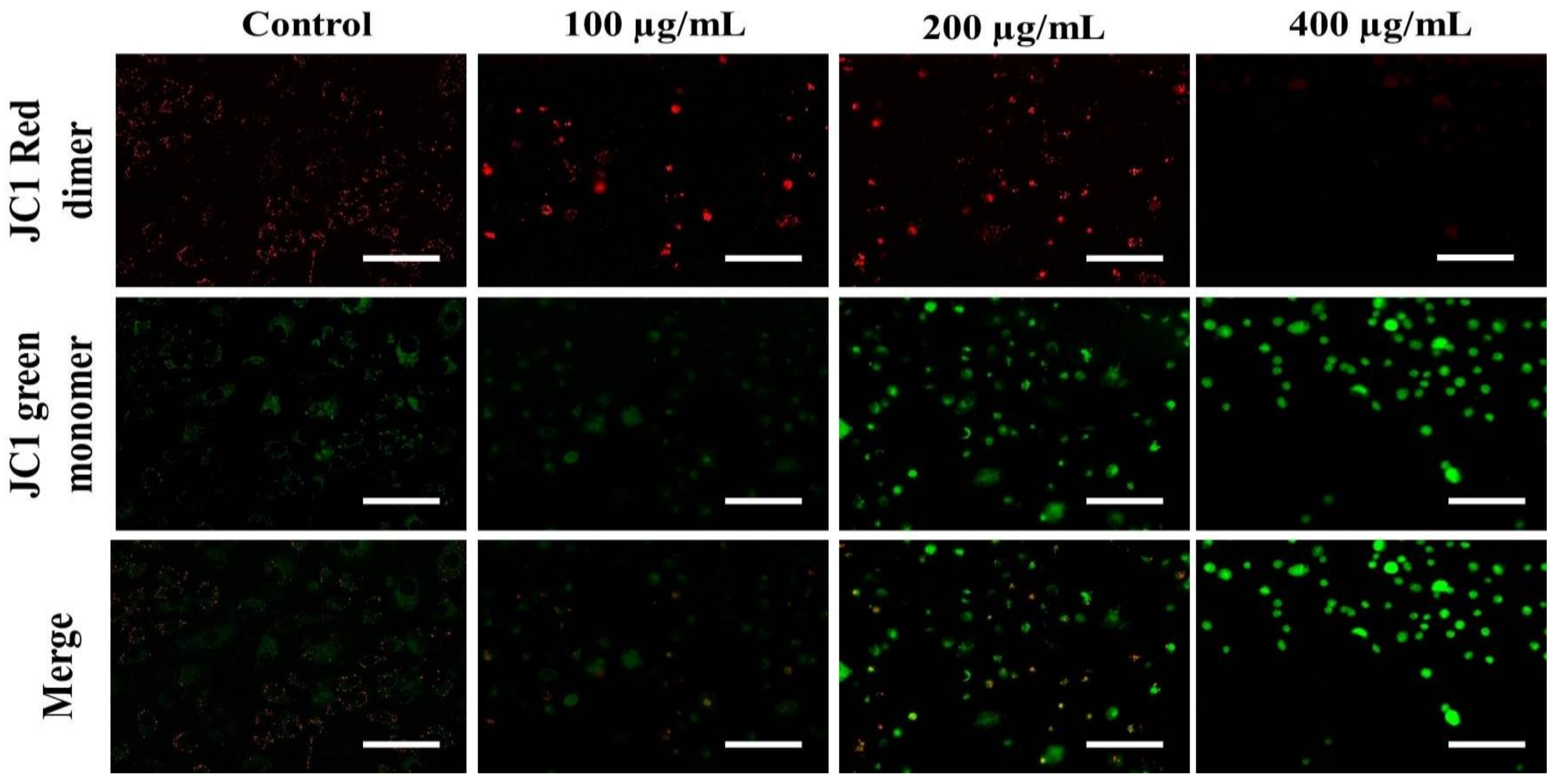
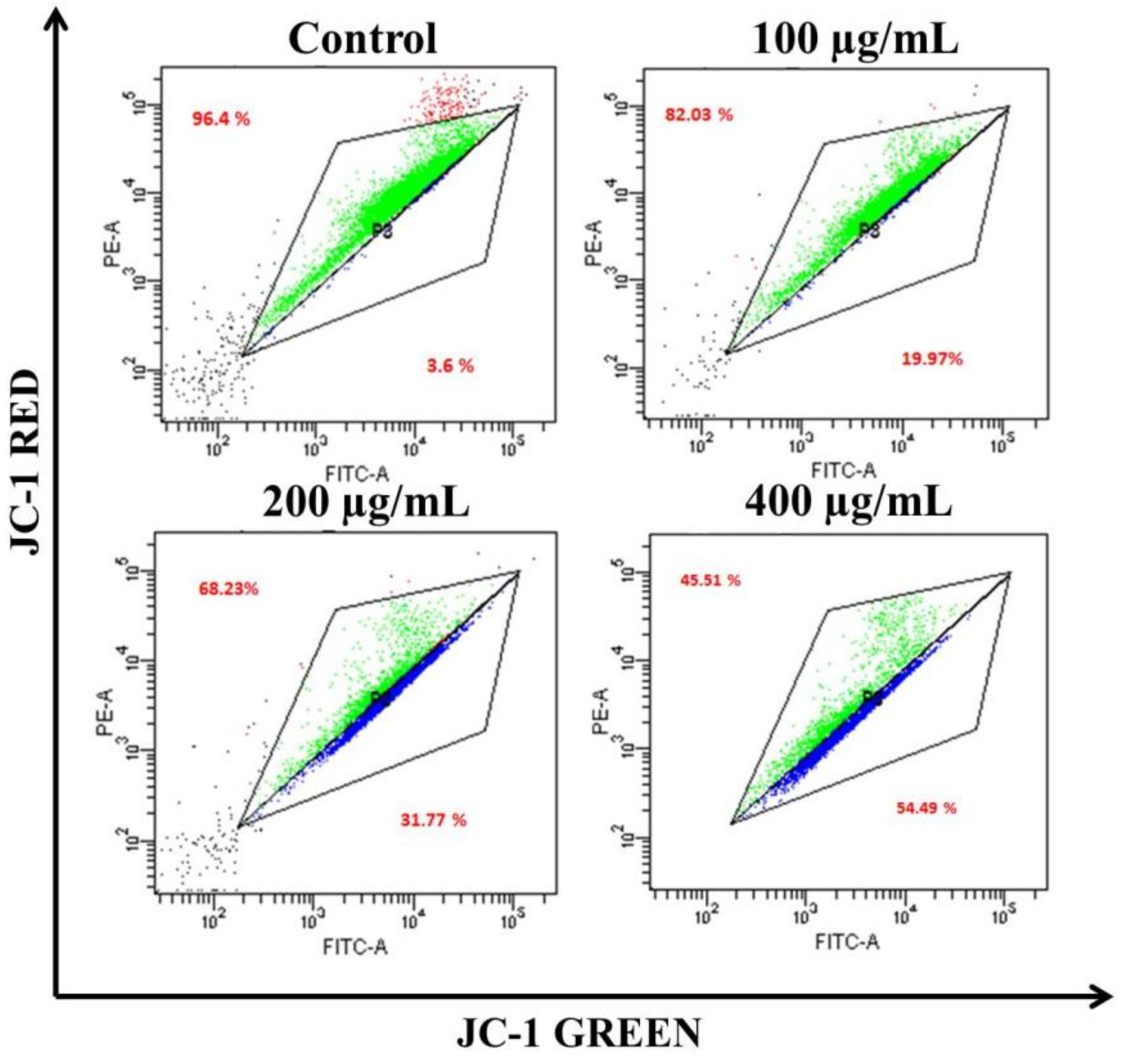
2.2.9. Mitochondrial Membrane Potential Quantification
2.2.10. Determination of Intracellular Reactive Oxygen Species Levels
2.2.11. Quantification of Apoptosis
2.2.12. DNA Damage Assay
2.2.13. Quantification of Cytosolic Cytochrome-c Levels
2.2.14. Quantification of PARP Cleavage
2.2.15. Reverse Transcriptase qPCR Evaluations
2.2.16. Statistical Analysis
3. Results
3.1. GC-MS Analysis of Ethanolic Extract of A. vulgaris
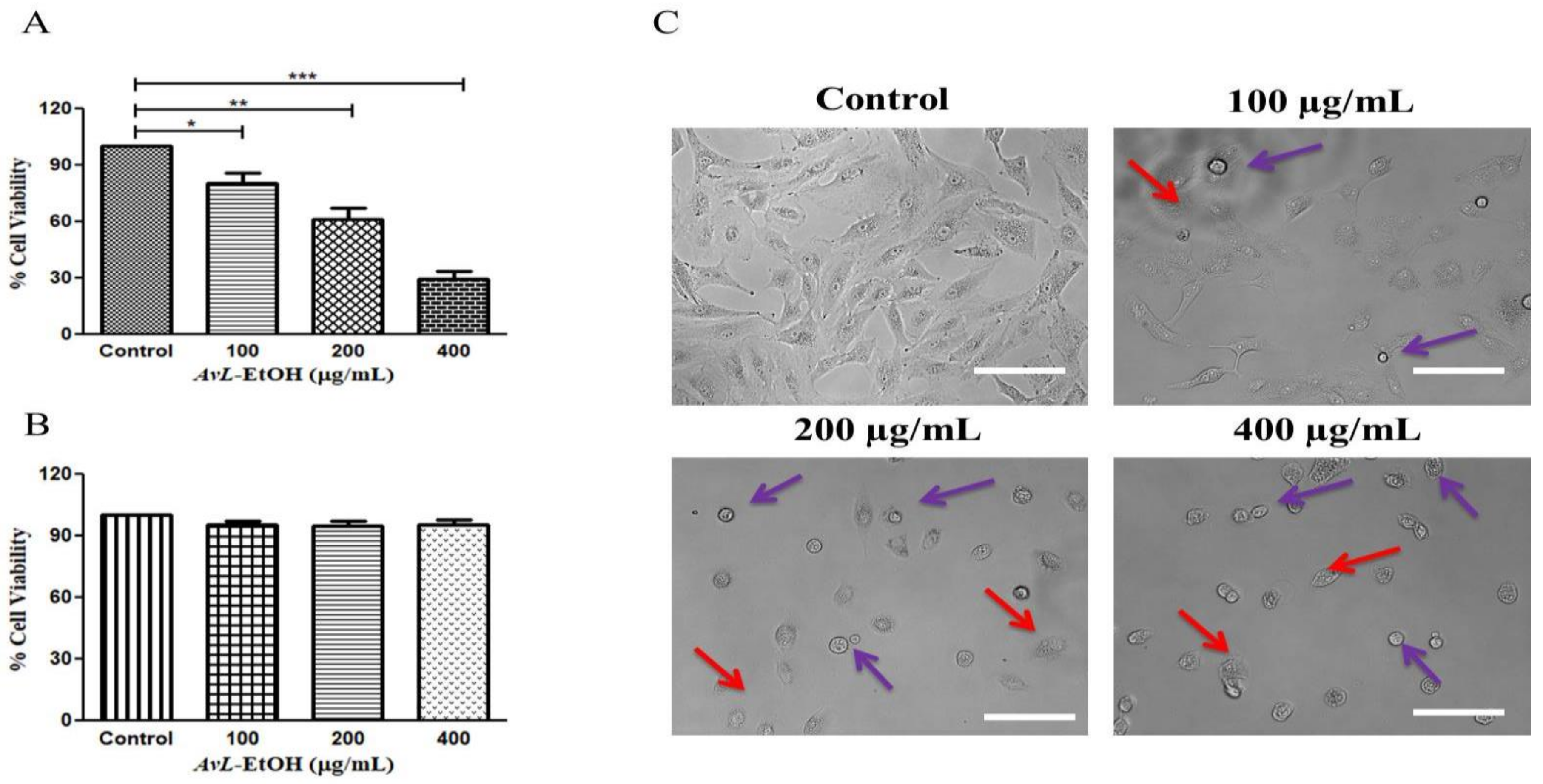
3.2. AvL-EtOH Inhibits the Cell Viability of A549 Cells
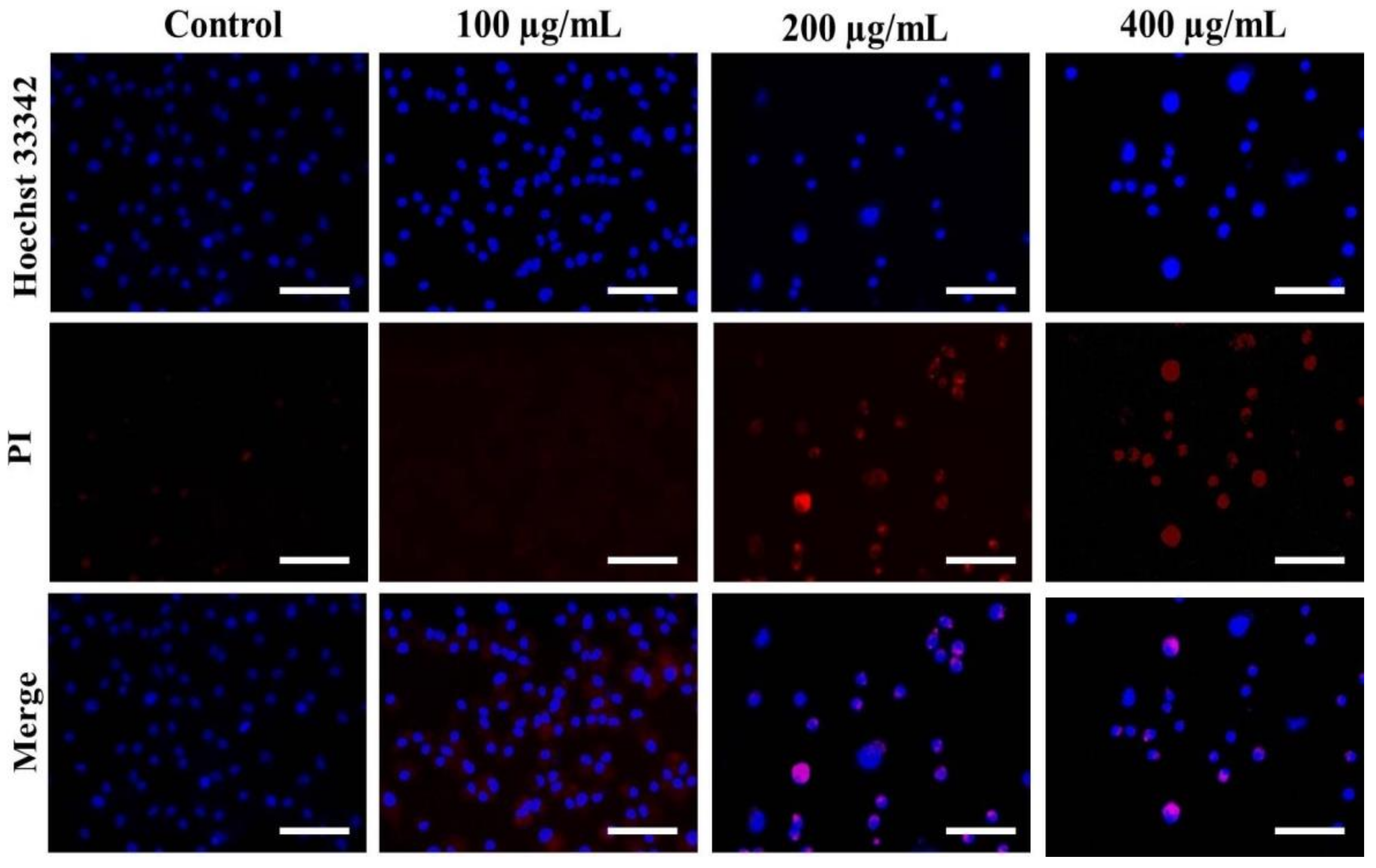
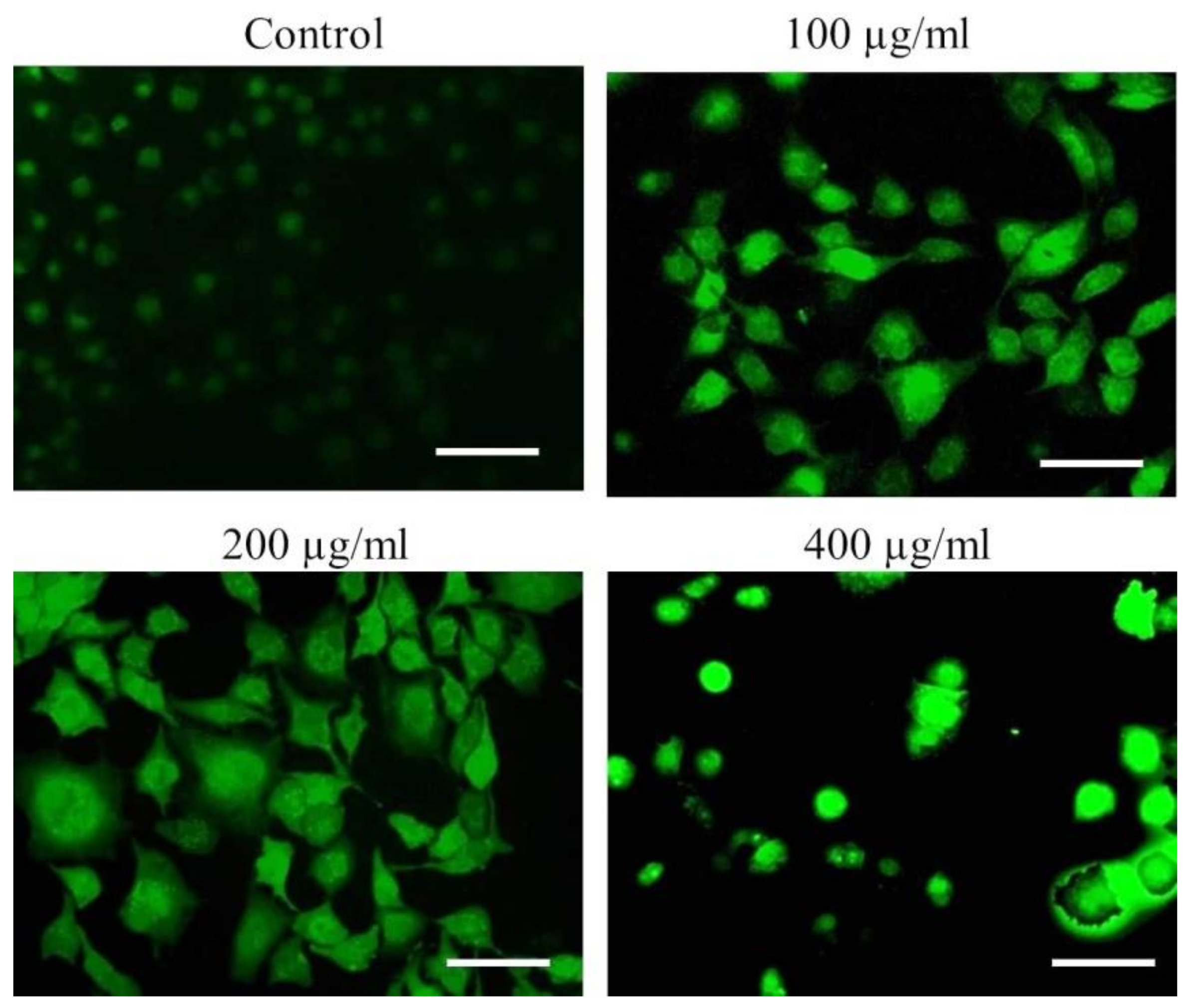
3.3. Nuclear Condensation in A549 Cells after AvL-EtOH Exposure
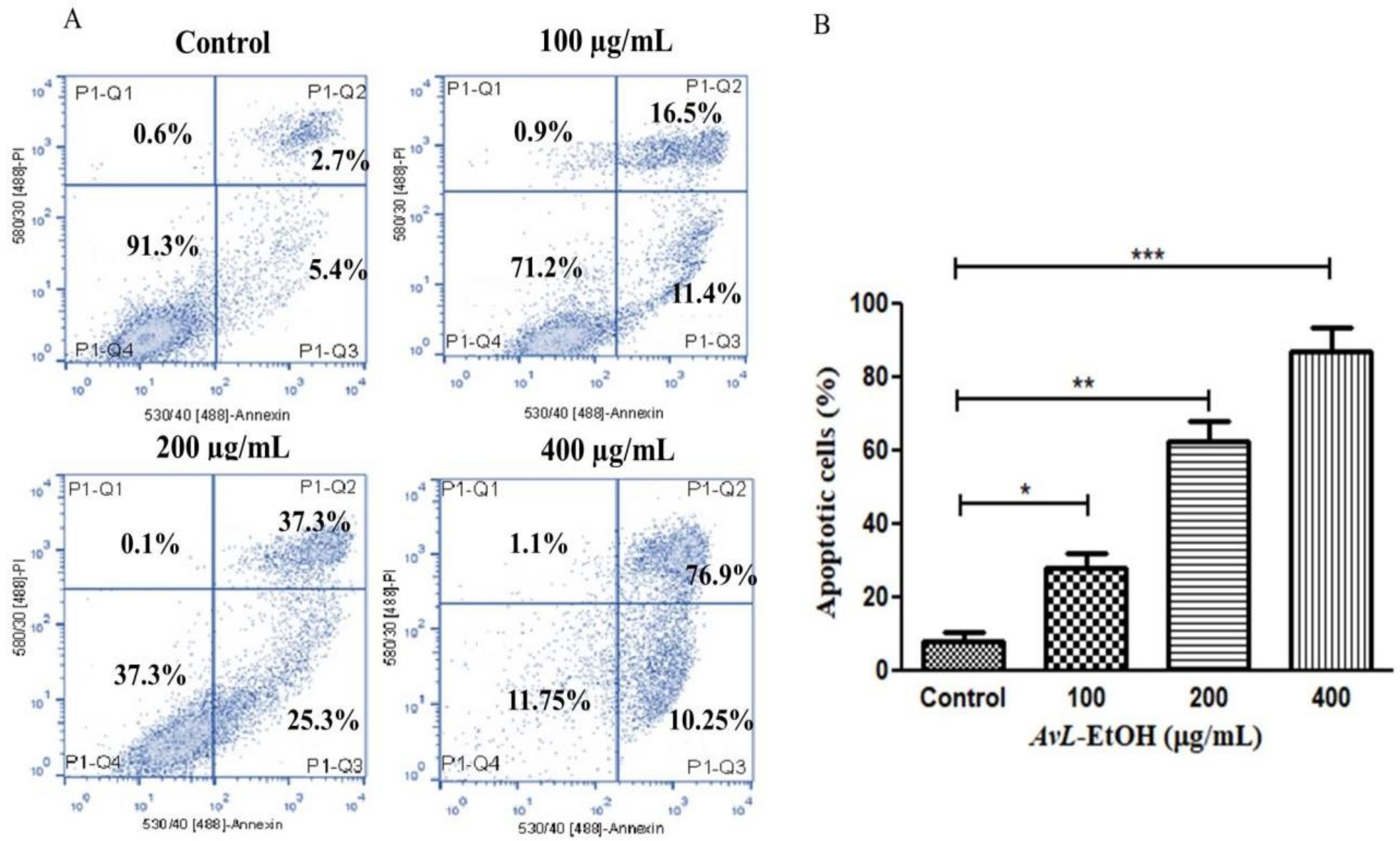
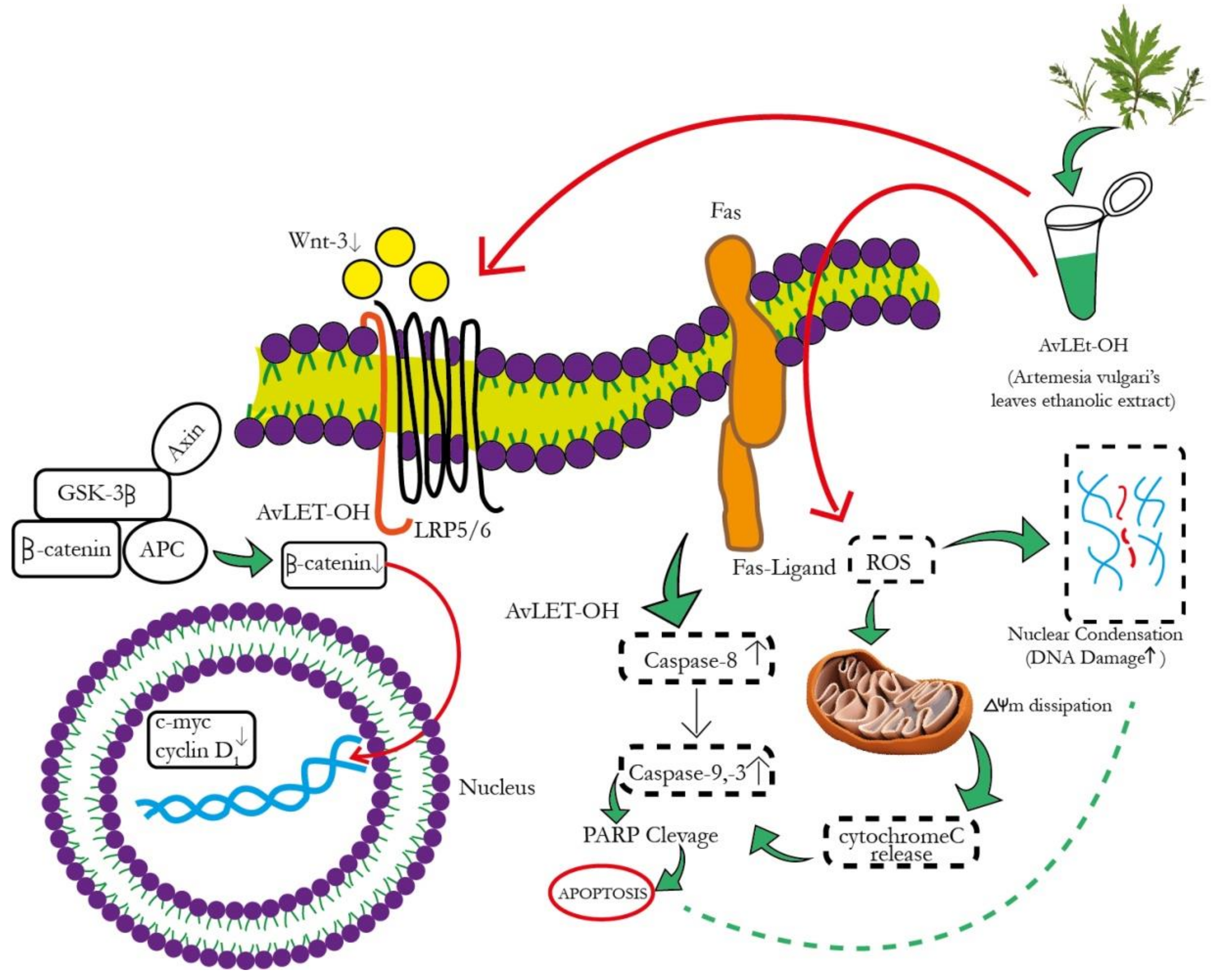
3.4. AvL-EtOH Induced Apoptosis through Activation of Intrinsic as well as Extrinsic Pathway
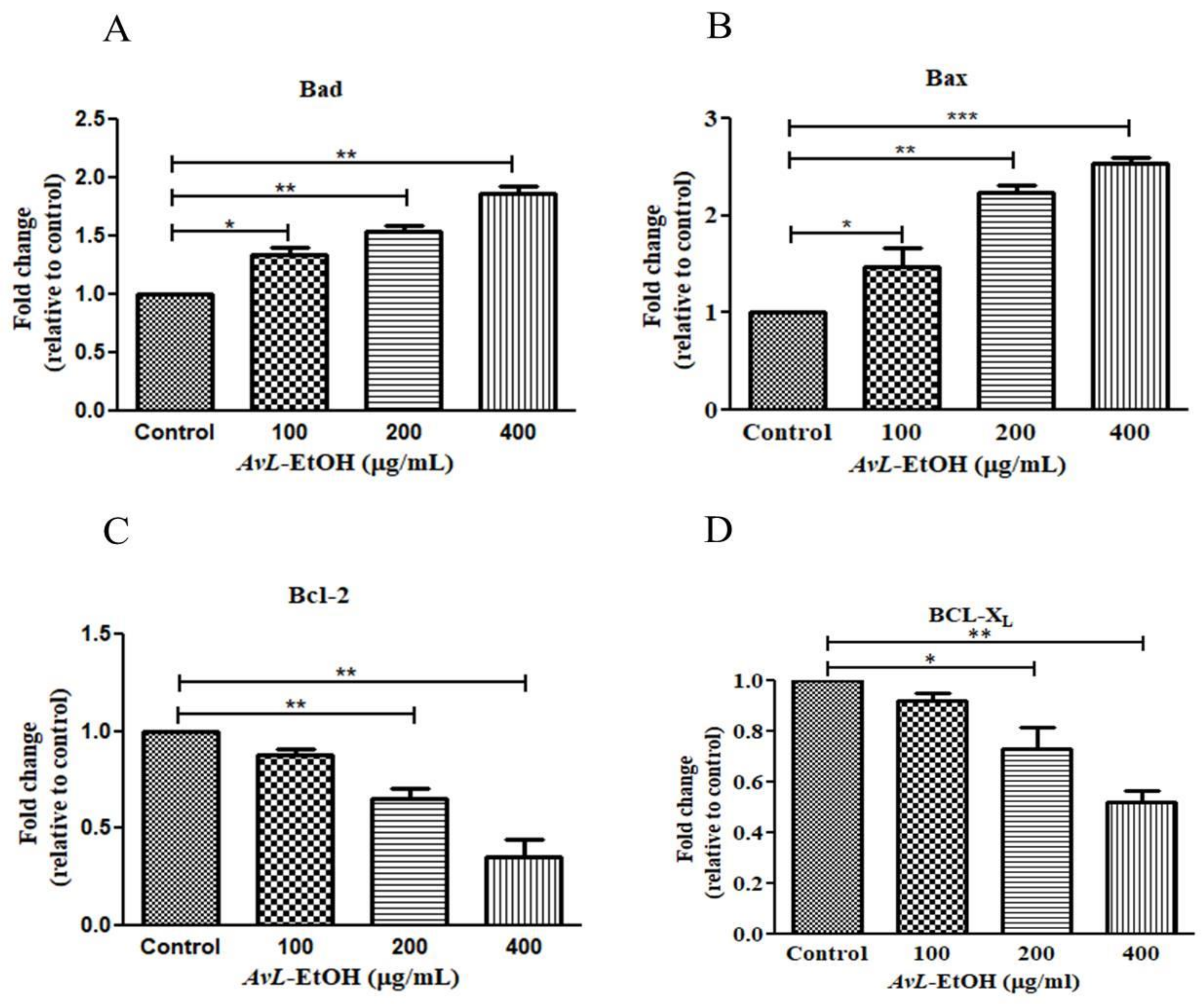
3.5. Modulation of Wnt/β-Catenin Signaling Pathway by AvL-EtOH
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Global Cancer Observatory, Cancer Factsheet. Available online: https://gco.iarc.fr/today/data/factsheets/cancers/15-Lung-fact-sheet.pdf (accessed on 25 August 2022).
- Batbold, U.; Liu, J.J. Novel Insights of Herbal Remedy into NSCLC Suppression through Inducing Diverse Cell Death Pathways via Affecting Multiple Mediators. Appl. Sci. 2022, 12, 4868. [Google Scholar] [CrossRef]
- Hardy, D.; Cormier, J.N.; Xing, Y.; Liu, C.C.; Xia, R.; Du, X.L. Chemotherapy-associated toxicity in a large cohort of elderly patients with non-small cell lung cancer. J. Thorac. Oncol. 2010, 5, 90–98. [Google Scholar] [CrossRef]
- Holohan, C.; Van Schaeybroeck, S.; Longley, D.B.; Johnston, P.G. Cancer drug resistance: An evolving paradigm. Nat. Rev. Cancer 2013, 13, 714–726. [Google Scholar] [CrossRef]
- Longley, D.B.; Johnston, P.G. Molecular mechanisms of drug resistance. J. Pathol. 2005, 205, 275–292. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.; Bahadur, S.; Harwansh, R.; Biswas, S.; Banerjee, S. Paradigm shift in natural product research: Traditional medicine inspired approaches. Phytochem. Rev. 2016, 16, 803–826. [Google Scholar] [CrossRef]
- Venkatesha, S.H.; Berman, B.M.; Moudgil, K.D. Herbal medicinal products target defined biochemical and molecular mediators of inflammatory autoimmune arthritis. Bioorg. Med. Chem. 2011, 19, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Koni, M.; Pinnarò, V.; Brizzi, M.F. The Wnt Signalling Pathway: A Tailored Target in Cancer. Int. J. Mol. Sci. 2020, 21, 7697. [Google Scholar] [CrossRef]
- Hafeez, B.B.; Siddiqui, I.A.; Asim, M.; Malik, A.; Afaq, F.; Adhami, V.M.; Saleem, M.; Din, M.; Mukhtar, H. A dietary anthocyanidin delphinidin induces apoptosis of human prostate cancer PC3 cells in vitro and in vivo: Involvement of nuclear factor-kappaB signaling. Cancer Res. 2008, 68, 8564–8572. [Google Scholar] [CrossRef]
- Du, J.H.; Zhang, H.D.; Ma, Z.J.; Ji, K.M. Artesunate induces oncosis-like cell death in vitro and has antitumor activity against pancreatic cancer xenografts in vivo. Cancer Chemother. Pharmacol. 2010, 65, 895–902. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Shimada, H.; Matsuda, H.; Yamahara, J.; Murakami, N. Bioactive constituents of Chinese natural medicines. I. New sesquiterpene ketones with vasorelaxant effect from Chinese moxa, the processed leaves of Artemisia argyi Levl. et Vant.: Moxartenone and moxartenolide. Chem. Pharm. Bull. 1996, 44, 1656–1662. [Google Scholar] [CrossRef]
- Afsar, S.K.; Kumar, K.R.; Gopal, J.V.; Raveesha, P. Assessment of antiinfammatory activity of Artemisia vulgaris leaves by cotton pellet granuloma method in Wistar albino rats. J. Pharm. Res. 2013, 7, 463–467. [Google Scholar]
- Jakovljević, M.R.; Grujičić, D.; Vukajlović, J.T.; Marković, A.; Milutinović, M.; Stanković, M.; Milošević-Djordjević, O. In vitro study of genotoxic and cytotoxic activities of methanol extracts of Artemisia vulgaris L. and Artemisia alba Turra. S. Afr. J. Bot. 2020, 132, 117–126. [Google Scholar] [CrossRef]
- Ahmad, A.; Tiwari, R.K.; Almeleebia, T.M.; Al Fayi, M.S.; Alshahrani, M.Y.; Ahmad, I.; Abohassan, M.S.; Saeed, M.; Ansari, I.A. Swertia chirayita suppresses the growth of non-small cell lung cancer A549 cells and concomitantly induces apoptosis via downregulation of JAK1/STAT3 pathway. Saudi J. Biol. Sci. 2021, 28, 6279–6288. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, R.K.; Singh, S.; Gupta, C.L.; Bajpai, P. Microglial TLR9: Plausible Novel Target for Therapeutic Regime Against Glioblastoma Multiforme. Cell Mol. Neurobiol. 2021, 41, 1391–1393. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.K.; Bhardwaj, R.; Dwarakanath, B.S.; Varshney, R. Metabolic oxidative stress induced by a combination of 2-DG and 6-AN enhances radiation damage selectively in malignant cells via non-coordinated expression of antioxidant enzymes. Cancer Lett. 2010, 295, 154–166. [Google Scholar] [CrossRef]
- Ahmad, A.; Tiwari, R.K.; Saeed, M.; Ahmad, I.; Ansari, I.A. Glycyrrhizin Mediates Downregulation of Notch Pathway Resulting in Initiation of Apoptosis and Disruption in the Cell Cycle Progression in Cervical Cancer Cells. Nutr Cancer. 2022, 74, 622–639. [Google Scholar] [CrossRef]
- Ahmad, A.; Ansari, I.A. Carvacrol Exhibits Chemopreventive Potential against Cervical Cancer Cells via Caspase-Dependent Apoptosis and Abrogation of Cell Cycle Progression. Anticancer Agents Med. Chem. 2021, 21, 2224–2235. [Google Scholar] [CrossRef]
- Pandey, B.P.; Thapa, R.; Upreti, A. Chemical composition, antioxidant and antibacterial activities of essential oil and methanol extract of Artemisia vulgaris and Gaultheria fragrantissima collected from Nepal. Asian Pac. J. Trop. Med. 2017, 10, 952–959. [Google Scholar] [CrossRef]
- Van Nguyen Thien, T.; Tran, L.T.K.; Nhu, N.T.T.; Duc, T.P.; Do, L.T.M.; Tu, D.D.; That, Q.T. A new eudesmane-type sesquiterpene from the leaves of Artemisia vulgaris. Chem. Nat. Compd. 2018, 54, 66–68. [Google Scholar] [CrossRef]
- Ekiert, H.; Pajor, J.; Klin, P.; Rzepiela, A.; Ślesak, H.; Szopa, A. Significance of Artemisia vulgaris L. (Common Mugwort) in the History of Medicine and Its Possible Contemporary Applications Substantiated by Phytochemical and Pharmacological Studies. Molecules 2020, 25, 4415. [Google Scholar] [CrossRef]
- Saleh, A.M.; Aljada, A.; Rizvi, S.A.; Nasr, A.; Alaskar, A.S.; Williams, J.D. In vitro cytotoxicity of Artemisia vulgaris L. essential oil is mediated by a mitochondria-dependent apoptosis in HL-60 leukemic cell line. BMC Complement. Altern. Med. 2014, 14, 226. [Google Scholar] [CrossRef]
- Vahdati-Mashhadian, N.; Emami, S.; Oghazian, M.; Vosough, R. The cytotoxicity evaluation of seven species of Artemisia on human tumor cell lines. Pharmacologyonline 2009, 1, 229–242. [Google Scholar]
- Rasheed, T.; Bilal, M.; Iqbal, H.M.N.; Li, C. Green biosynthesis of silver nanoparticles using leaves extract of Artemisia vulgaris and their potential biomedical applications. Colloids Surf. B Biointerfaces 2017, 158, 408–415. [Google Scholar] [CrossRef]
- Yip, H.Y.K.; Papa, A. Signaling Pathways in Cancer: Therapeutic Targets, Combinatorial Treatments, and New Developments. Cells 2021, 10, 659. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, B.A.; El-Deiry, W.S. Targeting apoptosis in cancer therapy. Nat. Rev. Clin. Oncol. 2020, 17, 395–417. [Google Scholar] [CrossRef] [PubMed]
- Lim, B.; Greer, Y.; Lipkowitz, S.; Takebe, N. Novel Apoptosis-Inducing Agents for the Treatment of Cancer, a New Arsenal in the Toolbox. Cancers 2019, 11, 1087. [Google Scholar] [CrossRef] [PubMed]
- Greenwell, M.; Rahman, P.K. Medicinal Plants: Their Use in Anticancer Treatment. Int. J. Pharm. Sci. Res. 2015, 6, 4103–4112. [Google Scholar]
- Cragg, G.M.; Newman, D.J. Plants as a source of anti-cancer agents. J. Ethnopharmacol. 2005, 100, 72–79. [Google Scholar] [CrossRef]
- Fetrow, C.W.; Avila, J.R. Professional’s Handbook of Complementary & Alternative Medicines; Springhouse Publishing Company: Alphen aan den Rijn, Netherlands, 2001. [Google Scholar]
- Gruenwald, J.; Brendler, T.; Jaenicke, C. Physicians’ Desk Reference (PDR) for Herbal Medicines, 13th ed.; Thomson Medical Economics Company: Montvale, NJ, USA, 2004. [Google Scholar]
- Tang, J.; Zhao, J.J.; Li, Z.H. Ethanol extract of Artemisia sieversiana exhibits anticancer effects and induces apoptosis through a mitochondrial pathway involving DNA damage in COLO-205 colon carcinoma cells. Bangladesh J. Pharmacol. 2015, 10, 518–523. [Google Scholar] [CrossRef]
- Emami, S.A.; Vahdati-Mashhadian, N.; Vosough, R.; Oghazian, M.B. The anticancer activity of five species of Artemisia on Hep2 and HepG2 cell lines. Pharmacologyonline 2009, 3, 327–339. [Google Scholar]
- Sharmila, K.; Padma, P.R. Anticancer activity of Artemisia vulgaris on hepatocellular carcinoma (HepG2) cells. Int. J. Pharm. Pharm. Sci. 2013, 5, 479–483. [Google Scholar]
- Abdelhamed, S.; Yokoyama, S.; Hafiyani, L.; Kalauni, S.K.; Hayakawa, Y.; Awale, S.; Saiki, I. Identification of plant extracts sensitizing breast cancer cells to TRAIL. Oncol. Rep. 2013, 29, 1991–1998. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.C.; Deng, T.; Lehal, R.S.; Kim, J.; Zacksenhaus, E. Identification of tumorsphere- and tumor-initiating cells in HER2/Neu-induced mammary tumors. Cancer Res. 2007, 67, 8671–8681. [Google Scholar] [CrossRef]
- Abad, M.J.; Bedoya, L.M.; Apaza, L.; Bermejo, P. The artemisia L. Genus: A review of bioactive essential oils. Molecules 2012, 17, 2542–2566. [Google Scholar] [CrossRef]
- Tang, H.Q.; Hu, J.; Yang, L.; Tan, R.X. Terpenoids and flavonoids from Artemisia species. Planta Medica 2000, 66, 391–393. [Google Scholar] [CrossRef] [PubMed]
- Turi, C.E.; Shipley, P.R.; Murch, S.J. North American Artemisia species from the subgenus Tridentatae (Sagebrush): A phytochemical, botanical and pharmacological review. Phytochemistry 2014, 98, 9–26. [Google Scholar] [CrossRef]
- Taleghani, A.; Emami, S.A.; Tayarani-Najaran, Z. Artemisia: A promising plant for the treatment of cancer. Bioorganic Med. Chem. 2020, 28, 115180. [Google Scholar] [CrossRef]
- Roos, W.P.; Kaina, B. DNA damage-induced cell death by apoptosis. Trends Mol. Med. 2006, 12, 440–450. [Google Scholar] [CrossRef]
- Yang, J.; Zhao, X.; Tang, M.; Li, L.; Lei, Y.; Cheng, P.; Guo, W.; Zheng, Y.; Wang, W.; Luo, N.; et al. The role of ROS and subsequent DNA-damage response in PUMA-induced apoptosis of ovarian cancer cells. Oncotarget 2017, 8, 23492–23506. [Google Scholar] [CrossRef]
- Srinivas, U.S.; Tan, B.W.Q.; Vellayappan, B.A.; Jeyasekharan, A.D. ROS and the DNA damage response in cancer. Redox Biol. 2019, 25, 101084. [Google Scholar] [CrossRef]
- Bhardwaj, M.; Kim, N.H.; Paul, S.; Jakhar, R.; Han, J.; Kang, S.C. 5-Hydroxy-7-Methoxyflavone Triggers Mitochondrial-Associated Cell Death via Reactive Oxygen Species Signaling in Human Colon Carcinoma Cells. PLoS ONE 2016, 11, e0154525. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Lai, H.; Chen, J.; Li, L.; Wong, Y.S.; Chen, T.; Li, X. Natural borneol, a monoterpenoid compound, potentiates selenocystine-induced apoptosis in human hepatocellular carcinoma cells by enhancement of cellular uptake and activation of ROS-mediated DNA damage. PLoS ONE 2013, 8, e63502. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.L.; Sedlic, F.; Bosnjak, Z.; Nilakantan, V. SOD1 and MitoTEMPO partially prevent mitochondrial permeability transition pore opening, necrosis, and mitochondrial apoptosis after ATP depletion recovery. Free Radic. Biol. Med. 2010, 49, 1550–1560. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Wei, J.; Chen, Y.; He, P.; Lin, J.; Tan, S.; Nie, J.; Lu, S.; He, M.; Lu, Z.; et al. Isoorientin from Gypsophila elegans induces apoptosis in liver cancer cells via mitochondrial-mediated pathway. J. Ethnopharmacol. 2016, 187, 187–194. [Google Scholar] [CrossRef]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol. Rev. 2014, 94, 909–950. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.; Huang, J.; Dong, Q. Wnt signaling regulation of stem-like properties in human lung adenocarcinoma cell lines. Med. Oncol. 2015, 32, 157. [Google Scholar] [CrossRef]
- Stewart, D.J. Wnt signaling pathway in non-small cell lung cancer. J. Natl. Cancer Inst. 2014, 106, djt356. [Google Scholar] [CrossRef]
- Xu, X.; Sun, P.L.; Li, J.Z.; Jheon, S.; Lee, C.T.; Chung, J.H. Aberrant Wnt1/β-catenin expression is an independent poor prognostic marker of non-small cell lung cancer after surgery. J. Thorac. Oncol. 2014, 6, 716–724. [Google Scholar] [CrossRef]
- David, M.D.; Cantí, C.; Herreros, J. Wnt-3a and Wnt-3 differently stimulate proliferation and neurogenesis of spinal neural precursors and promote neurite outgrowth by canonical signaling. J. Neurosci. Res. 2010, 88, 3011–3023. [Google Scholar] [CrossRef]
- Okoye, U.C.; Malbon, C.C.; Wang, H.Y. Wnt and Frizzled RNA expression in human mesenchymal and embryonic (H7) stem cells. J. Mol. Signal. 2008, 3, 16. [Google Scholar] [CrossRef]
- Zhang, M.; Shi, J.; Huang, Y.; Lai, L. Expression of canonical WNT/β-CATENIN signaling components in the developing human lung. BMC Dev. Biol. 2012, 12, 21. [Google Scholar] [CrossRef] [PubMed]



| Genes | Forward Sequence | Reverse Sequence |
|---|---|---|
| GAPDH | GAAATCCCATCACCATCTTCCAGG | GAGCCCCAGCCTTCTCCATG |
| Bax | GCCCTTTTGCTTCAGGGTTT | TCCAATGTCCAGCCCATGAT |
| Bad | CCTCAGGCCTATGCAAAAAG | AAACCCAAAACTTCCGATGG |
| Bcl2 | GATTGTGGCCTTCTTTGAG | CAAACTGAGCAGAGTCTTC |
| Mcl-1 | GGACATCAAAAACGAAGACG | GCAGCTTTCTTGGTTTATGG |
| Cyclin D1 | CCGTCCATGCGGAAGATC | GAAGACCTCCTCCTCGCACT |
| c-myc | AGCGACTCTGAGGAGGAACAAG | GTGGCACCTCTTGAGGACCA |
| Wnt3 | CGCTCAGCTATGAACAAGCA | AAAGTTGGGGGAGTTCTCGT |
| β-catenin | TCTGAGGACAAGCCACAAGATTACA | TGGGCACCAATATCAAGTCCAA |
| Bcl-XL | CAGAGCTTTGAACAGGTAG | GCTCTCGGGTGCTGTATTG |
| Peak No. | Retention Time | Area | Area (%) | Compound Identified | Molecular Formula |
|---|---|---|---|---|---|
| 1. | 4.464 | 71,441 | 0.83 | 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl | C6H8O4 |
| 2. | 6.718 | 379,461 | 4.43 | 2,3,6,10,10-Pentamethyl-1-oxa-spiro(4,5)deca-3 | C14H22O |
| 3. | 7.925 | 59,506 | 0.69 | 3-(Pyrrolidin-1-yl)cyclopent-2-en-1-one | C9H13NO |
| 4. | 8.382 | 561,175 | 6.55 | 2H-1-benzopyran-2-one | C9H6O2 |
| 5. | 8.591 | 546,765 | 6.38 | Guanosine | C10H13N5O5 |
| 6. | 9.000 | 41,899 | 0.49 | 4H-1,2,3,6,7,8,9,9A-octahydroquinolizine-1,4,8-D | C9H12D3N |
| 7. | 9.086 | 27,165 | 0.32 | 2,4-Ditert-butylphenyl 5-hydroxypentanoate | C19H30O3 |
| 8. | 9.241 | 104,843 | 1.22 | Decanoic anhydride | C20H38O3 |
| 9. | 10.063 | 128,609 | 1.50 | 1-(4-Methylphenyl)-1H-pyrrole | C11H11N |
| 10. | 10.942 | 1,664,345 | 19.42 | 1,3,4,5-Tetrahydroxycyclohexanecarboxylic acid | C7H12O6 |
| 11. | 11.269 | 124,502 | 1.45 | 3alpha,7beta-Dihydroxy-5beta,6beta-epoxycholestane | C27H46O3 |
| 12. | 11.356 | 40,194 | 0.47 | 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine | C12H15N |
| 13. | 11.992 | 188,369 | 2.20 | Nonylamine, N,N-di(allyl) | C15H29N |
| 14. | 12.207 | 44,211 | 0.52 | (1R,3E,7E,11R)-1,5,5,8-Tetramethyl-12-oxabicyclo [9.1.0]dodeca-3,7-diene | C7H12O6 |
| 15. | 12.299 | 40,009 | 0.47 | 6-Hydroxy-4,4,7a-trimethyl-5,6,7,7a-tetrahydrobenzofuran-2(4H)- | C11H16O3 |
| 16. | 12.625 | 246,632 | 2.88 | Arctiol | C15H26O2 |
| 17. | 12.721 | 96,591 | 1.13 | Neophytadiene | C20H38 |
| 18. | 12.981 | 24,151 | 0.28 | 10,12-Tetradecadienal; Vernaldehyde | C14H24O |
| 19. | 13.074 | 45,010 | 0.53 | 2-Furanmethanol, tetrahydro-;Dihydropanaxacol | C17H28O3 |
| 20. | 13.168 | 23,344 | 0.27 | 3,7,11,15-Tetramethylhexadec-2-en-1-yl acetate; Cetoleic acid | C2 O22H42 |
| 21. | 13.414 | 123,198 | 1.44 | 2-(Dimethylamino)-N-(2,6-dimethylphenyl) acetamide; Lidocaine | C14H22N2O |
| 22. | 13.563 | 51,472 | 0.60 | Limonene dioxide; Diosphenol | C10H16O2 |
| 23. | 13.780 | 116,889 | 1.36 | 1H-Cyclopropa[a]naphthalene, 1a,2,3,5,6,7a,7b-octahydro-1,1,7,7a-tetramethyl-, 1aR-1aα,7α,7aα,7bβ | C15H24 |
| 24. | 13.995 | 239,211 | 2.79 | 5,10-Diethoxy-2,3,7,8-tetrahydro-1H,6H-dipyrrolo[1,2-a:1’,2’-d]pyrazine; Rivastigmine | C14H22N2O2 |
| 25. | 14.355 | 233,920 | 2.73 | Scopoletin | C10H8O4 |
| 26. | 14.840 | 124,027 | 1.45 | Martynoside | C31H40O15 |
| 27. | 14.980 | 29,151 | 0.34 | Geranic acid | C10H16O2 |
| 28. | 15.042 | 224,828 | 2.62 | Deoxyartemisinin | C15H22O4 |
| 29. | 15.222 | 111,627 | 1.30 | Retinal | C20H28O |
| 30. | 15.341 | 88,859 | 1.04 | 5-oxatricyclo[4.4.0.01,4]decan-7-one; 4-Heptyloxyphenol | C13H20O2 |
| 31. | 15.441 | 239,833 | 2.80 | Phytol isomer | C20H40O |
| 32. | 15.749 | 182,147 | 2.13 | Farnesol | C15H26O |
| 33. | 15.849 | 55,741 | 0.65 | Cycloionone | C13H20O |
| 34. | 16.057 | 58,278 | 0.68 | 1H-2-Indenone,2,4,5,6,7,7a-hexahydro-3-(1-methylethyl) | |
| 35. | 16.339 | 25,000 | 0.29 | 1,8-Diazabicyclo[5.4.0]undec-7-en-11-one; Phenoxypropazine | C9H14N2O |
| 36. | 16.591 | 277,133 | 3.23 | Cyperadione; Farnesoic acid | C15H24O2 |
| 37. | 16.956 | 22,939 | 0.27 | 2-dimethylaminoethyl ester; Hexanoic acid | C10H21NO2 |
| 38. | 18.447 | 64,492 | 0.75 | N-cis-11-eicosaenoylethanolamine; 2-(Dimethylamino)ethyl vaccenoate | C22H43NO2 |
| 39. | 19.034 | 48,587 | 0.57 | Phthalic acid | C24H38O4 |
| 40. | 20.269 | 119,663 | 1.40 | 1-Hexacosanol | C26H54O |
| 41. | 21.024 | 1,122,793 | 13.10 | Erucamide; 13-Docosenamide | C22H43NO |
| 42. | 22.127 | 79,068 | 0.92 | Chloroacetic acid | C14H27ClO2 |
| 43. | 22.726 | 42,958 | 0.50 | Octadecamethylcyclononasiloxane | C18H54O9Si9 |
| 44. | 25.217 | 37,974 | 0.44 | Vitamin E | C29H50O2 |
| 46. | 25.760 | 92,327 | 1.08 | 4-(acetyloxy)-1-(1,5-dimethylhexyl)-3A,6,6,12A-tetramethyl-2,3,3A,3B,5A,6,7,8,9,11,12,12A-dodecahydro-1H-cyclopenta[A]cyclopropa[E]phenanthren-7-yl acetate | C34H53DO4 |
| 47. | 27.639 | 102,832 | 1.20 | Stigmasterone | C29H46O |
| 48. | 28.962 | 77,279 | 0.90 | Stearoyldelicone | C33H54O3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tiwari, R.K.; Ahmad, A.; Khan, A.F.; Al-Keridis, L.A.; Saeed, M.; Alshammari, N.; Alabdallah, N.M.; Ansari, I.A.; Mujeeb, F. Ethanolic Extract of Artemisia vulgaris Leaf Promotes Apoptotic Cell Death in Non-Small-Cell Lung Carcinoma A549 Cells through Inhibition of the Wnt Signaling Pathway. Metabolites 2023, 13, 480. https://doi.org/10.3390/metabo13040480
Tiwari RK, Ahmad A, Khan AF, Al-Keridis LA, Saeed M, Alshammari N, Alabdallah NM, Ansari IA, Mujeeb F. Ethanolic Extract of Artemisia vulgaris Leaf Promotes Apoptotic Cell Death in Non-Small-Cell Lung Carcinoma A549 Cells through Inhibition of the Wnt Signaling Pathway. Metabolites. 2023; 13(4):480. https://doi.org/10.3390/metabo13040480
Chicago/Turabian StyleTiwari, Rohit Kumar, Afza Ahmad, Ahamad Faiz Khan, Lamya Ahmed Al-Keridis, Mohd Saeed, Nawaf Alshammari, Nadiyah M. Alabdallah, Irfan Ahmad Ansari, and Farina Mujeeb. 2023. "Ethanolic Extract of Artemisia vulgaris Leaf Promotes Apoptotic Cell Death in Non-Small-Cell Lung Carcinoma A549 Cells through Inhibition of the Wnt Signaling Pathway" Metabolites 13, no. 4: 480. https://doi.org/10.3390/metabo13040480
APA StyleTiwari, R. K., Ahmad, A., Khan, A. F., Al-Keridis, L. A., Saeed, M., Alshammari, N., Alabdallah, N. M., Ansari, I. A., & Mujeeb, F. (2023). Ethanolic Extract of Artemisia vulgaris Leaf Promotes Apoptotic Cell Death in Non-Small-Cell Lung Carcinoma A549 Cells through Inhibition of the Wnt Signaling Pathway. Metabolites, 13(4), 480. https://doi.org/10.3390/metabo13040480








