Acute Exercise and the Systemic and Airway Inflammatory Response to a High-Fat Meal in Young and Older Adults
Abstract
:1. Introduction
2. Materials and Methods
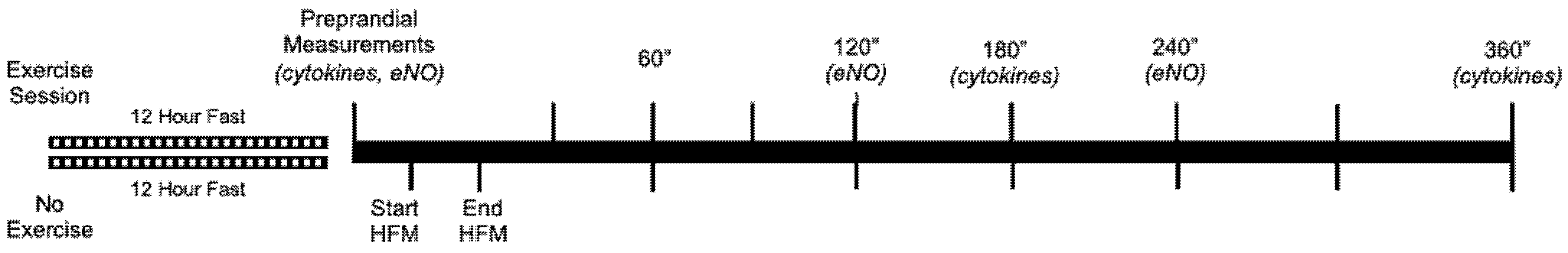
2.1. Experimental Design
2.2. Initial Visit
2.3. Pre-Prandial Exercise Bout
2.4. HFM Protocol
2.5. Cytokine Analysis
2.6. Airway Inflammation
2.7. Statistical Analysis
3. Results
3.1. Participant Characteristics and Exercise Sessions
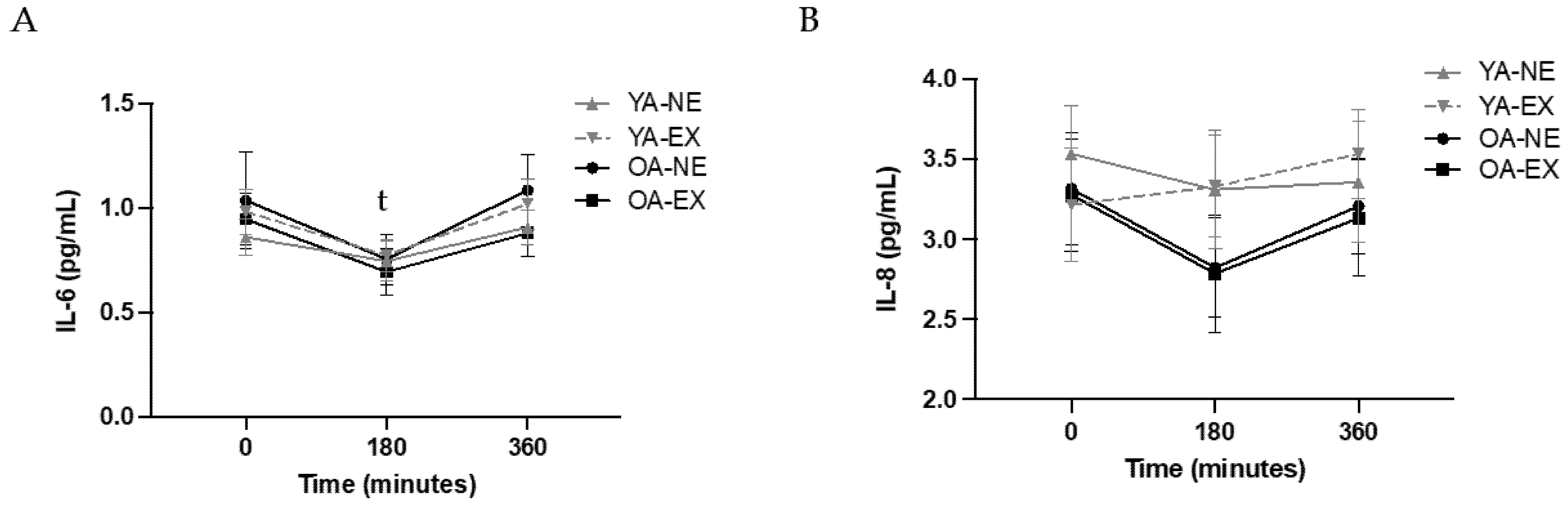
3.2. Systemic Markers of Inflammation: Primary Outcomes
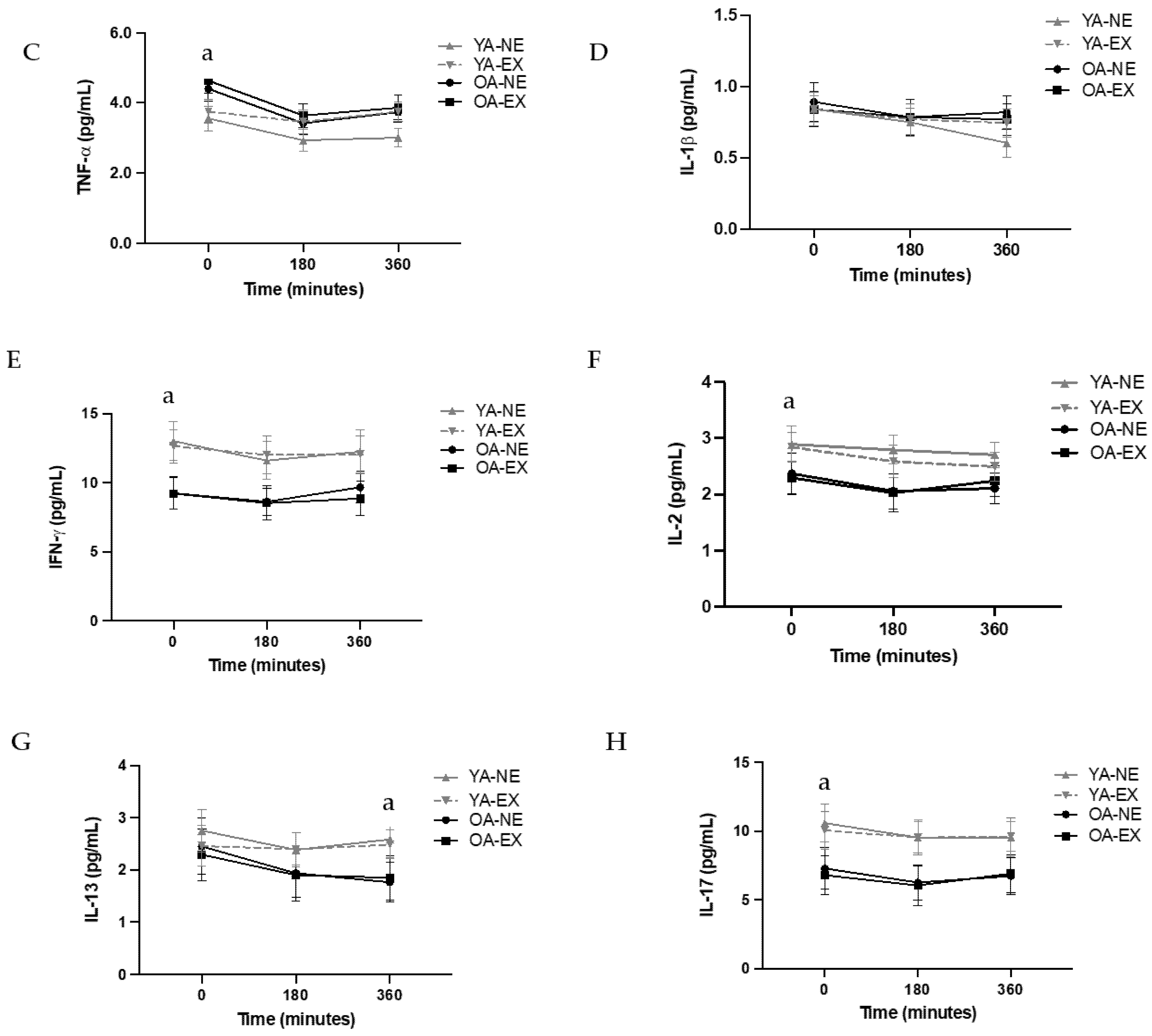
3.3. Secondary Cytokine Outcomes
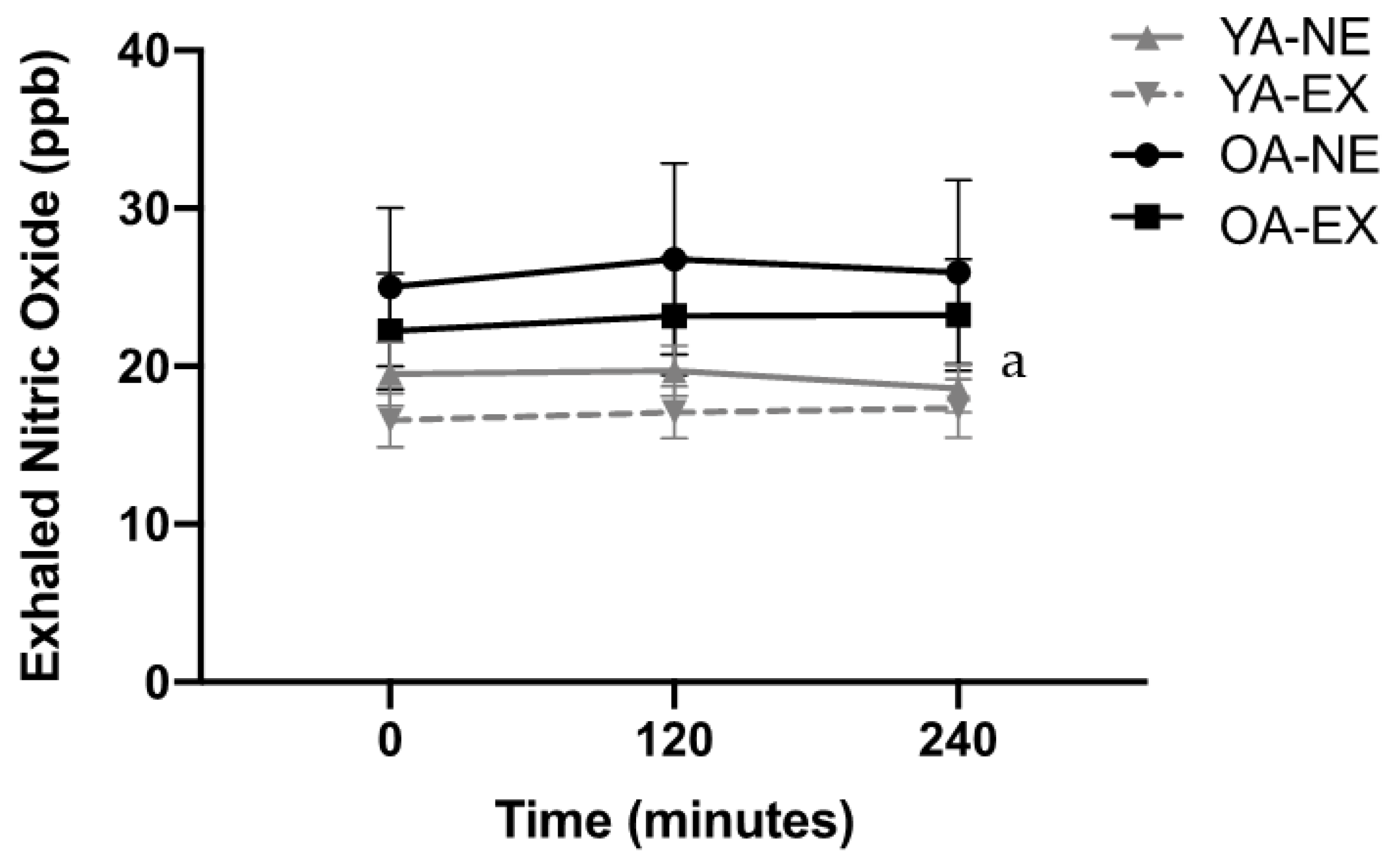
3.4. eNO
4. Discussion
4.1. The Impact of Age and Exercise on Systemic Inflammation
4.2. Post-Prandial Airway Inflammation, Aging, and Exercise
4.3. Experimental Considerations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Keirns, B.H.; Sciarrillo, C.M.; Poindexter, K.L.; Emerson, S.R. Daily Triglyceride Kinetics When Consuming a Realistic Western Diet in At-Risk Individuals across the Metabolic Spectrum: A Case Study. Curr. Dev. Nutr. 2021, 5, 505. [Google Scholar] [CrossRef]
- Hu, F.B.; Rimm, E.B.; Stampfer, M.J.; Ascherio, A.; Spiegelman, D.; Willett, C.W. Prospective Study of Major Dietary Patterns and Risk of Coronary Heart Disease in Men. Am. J. Clin. Nutr. 2000, 72, 912–921. [Google Scholar] [CrossRef] [PubMed]
- Wood, L.G.; Gibson, P.G. Dietary Factors Lead to Innate Immune Activation in Asthma. Pharmacol. Ther. 2009, 123, 37–53. [Google Scholar] [CrossRef] [PubMed]
- Hotamisligil, G.S. Inflammation and Metabolic Disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef]
- Schell, J.; Betts, N.M.; Lyons, T.J.; Basu, A. Raspberries Improve Postprandial Glucose and Acute and Chronic Inflammation in Adults with Type 2 Diabetes. Ann. Nutr. Metab. 2019, 74, 165–174. [Google Scholar] [CrossRef]
- Wood, L.G.; Garg, M.L.; Gibson, P.G. A High-Fat Challenge Increases Airway Inflammation and Impairs Bronchodilator Recovery in Asthma. J. Allergy Clin. Immunol. 2011, 127, 1133–1140. [Google Scholar] [CrossRef]
- Emerson, S.R.; Kurti, S.P.; Harms, C.A.; Haub, M.D.; Melgarejo, T.; Logan, C.; Rosenkranz, S.K. Magnitude and Timing of the Postprandial Inflammatory Response to a High-Fat Meal in Healthy Adults: A Systematic Review. Adv. Nutr. 2017, 8, 213–225. [Google Scholar] [CrossRef]
- Cowan, S.; Gibson, S.; Sinclair, A.J.; Truby, H.; Dordevic, A.L. Meals That Differ in Nutrient Composition and Inflammatory Potential Do Not Differentially Impact Postprandial Circulating Cytokines in Older Adults above a Healthy Weight. Nutrients 2022, 14, 1470. [Google Scholar] [CrossRef]
- Kurti, S.P.; Emerson, S.R.; Rosenkranz, S.K.; Teeman, C.S.; Emerson, E.M.; Cull, B.J.; Smith, J.R.; Harms, C.A. Post-Prandial Systemic 8-Isoprostane Increases after Consumption of Moderate and High-Fat Meals in Insufficiently Active Males. Nutr. Res. 2017, 39, 61–68. [Google Scholar] [CrossRef]
- Ade, C.J.; Rosenkranz, S.K.; Harms, C.A. The Effects of Short-Term Fish Oil Supplementation on Pulmonary Function and Airway Inflammation Following a High-Fat Meal. Eur. J. Appl. Physiol. 2014, 114, 675–682. [Google Scholar] [CrossRef] [Green Version]
- Johnson, A.M.; Kurti, S.P.; Smith, J.R.; Rosenkranz, S.K.; Harms, C.A. Effects of an Acute Bout of Moderate-Intensity Exercise on Postprandial Lipemia and Airway Inflammation. Appl. Physiol. Nutr. Metab. 2015, 41, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, S.K.; Townsend, D.K.; Steffens, S.E.; Harms, C.A. Effects of a High-Fat Meal on Pulmonary Function in Healthy Subjects. Eur. J. Appl. Physiol. 2010, 109, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Kurti, S.P.; Smith, J.R.; Rosenkranz, S.K.; Emerson, S.R.; Edwards, E.S.; Jurrens, K.; Laughlin, A.; Harms, C.A. Deep Inspirations Attenuate Postprandial Airway Inflammation in College-Aged Adults with Elevated Baseline Exhaled Nitric Oxide: A Pilot Study. Exp. Lung Res. 2020, 46, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Kurti, S.P.; Rosenkranz, S.K.; Levitt, M.; Cull, B.J.; Teeman, C.S.; Emerson, S.R.; Harms, C.A.; De Andrade, E.L. Does Moderate Intensity Exercise Attenuate the Postprandial Lipemic and Airway Inflammatory Response to a High-Fat Meal? BioMed Res. Int. 2015, 2015, 647952. [Google Scholar] [CrossRef] [PubMed]
- Kurti, S.P.; Rosenkranz, S.K.; Chapes, S.K.; Teeman, C.S.; Cull, B.J.; Emerson, S.R.; Levitt, M.H.; Smith, J.R.; Harms, C.A. Does Chronic Physical Activity Level Modify the Airway Inflammatory Response to an Acute Bout of Exercise in the Postprandial Period? Appl. Physiol. Nutr. Metab. 2017, 42, 173–180. [Google Scholar] [CrossRef]
- Woods, J.A.; Wilund, K.R.; Martin, S.A.; Kistler, B.M. Exercise, Inflammation and Aging. Aging Dis. 2012, 3, 130–140. [Google Scholar]
- Kurti, S.P.; Frick, H.; Wisseman, W.S.; Malin, S.K.; Edwards, D.A.; Emerson, S.R.; Edwards, E.S. Acute Exercise Improves Glucose and TAG Metabolism in Young and Older Adults Following High-Fat, High-Carbohydrate Meal Intake. Br. J. Nutr. 2022, 127, 687–695. [Google Scholar] [CrossRef]
- Emerson, S.R.; Kurti, S.P.; Snyder, B.S.; Sitaraman, K.; Haub, M.D.; Rosenkranz, S.K. Effects of Thirty and Sixty Minutes of Moderate-Intensity Aerobic Exercise on Postprandial Lipemia and Inflammation in Overweight Men: A Randomized Cross-over Study. J. Int. Soc. Sports Nutr. 2016, 13, 26. [Google Scholar] [CrossRef]
- Borland, C.; Cox, Y.; Higenbottam, T. Measurement of Exhaled Nitric Oxide in Man. Thorax 1993, 48, 1160–1162. [Google Scholar] [CrossRef]
- Kharitonov, S.; Alving, K.; Barnes, P.J. Exhaled and Nasal Nitric Oxide Measurements: Recommendations. The European Respiratory Society Task Force. Eur. Respir. J. 1997, 10, 1683–1693. [Google Scholar] [CrossRef]
- Emerson, S.R.; Sciarrillo, C.M.; Kurti, S.P.; Emerson, E.M.; Rosenkranz, S.K. High-Fat Meal–Induced Changes in Markers of Inflammation and Angiogenesis in Healthy Adults Who Differ by Age and Physical Activity Level. Curr. Dev. Nutr. 2019, 3, nzy098. [Google Scholar] [CrossRef]
- Teeman, C.S.; Kurti, S.P.; Cull, B.J.; Emerson, S.R.; Haub, M.D.; Rosenkranz, S.K. The Effect of Moderate Intensity Exercise in the Postprandial Period on the Inflammatory Response to a High-Fat Meal: An Experimental Study. Nutr. J. 2016, 15, 24. [Google Scholar] [CrossRef] [PubMed]
- Dekker, M.J.; Wright, A.J.; Mazurak, V.C.; Marangoni, A.G.; Rush, J.W.E.; Graham, T.E.; Robinson, L.E. Fasting Triacylglycerol Status, but Not Polyunsaturated/Saturated Fatty Acid Ratio, Influences the Postprandial Response to a Series of Oral Fat Tolerance Tests. J. Nutr. Biochem. 2009, 20, 694–704. [Google Scholar] [CrossRef] [PubMed]
- Izawa, S.; Miki, K.; Liu, X.; Ogawa, N. The Diurnal Patterns of Salivary Interleukin-6 and C-Reactive Protein in Healthy Young Adults. Brain. Behav. Immun. 2013, 27, 38–41. [Google Scholar] [CrossRef] [PubMed]
- Redwine, L.; Hauger, R.L.; Gillin, J.C.; Irwin, M. Effects of Sleep and Sleep Deprivation on Interleukin-6, Growth Hormone, Cortisol, and Melatonin Levels in Humans. J. Clin. Endocrinol. Metab. 2000, 85, 3597–3603. [Google Scholar] [CrossRef]
- Keirns, B.H.; Hart, S.M.; Sciarrillo, C.M.; Poindexter, K.L.; Clarke, S.L.; Emerson, S.R. Postprandial Triglycerides, Flow-Mediated Dilation, and the Inflammatory Cytokine Milieu in Metabolically Healthy Obesity: A Cross-Sectional Pilot Study. Obesities 2021, 1, 58–71. [Google Scholar] [CrossRef]
- Jankord, R.; Jemiolo, B. Influence of Physical Activity on Serum IL-6 and IL-10 Levels in Healthy Older Men. Med. Sci. Sports Exerc. 2004, 36, 960–964. [Google Scholar] [CrossRef]
- Nappo, F.; Esposito, K.; Cioffi, M.; Giugliano, G.; Molinari, A.M.; Paolisso, G.; Marfella, R.; Giugliano, D. Postprandial Endothelial Activation in Healthy Subjects and in Type 2 Diabetic Patients: Role of Fat and Carbohydrate Meals. J. Am. Coll. Cardiol. 2002, 39, 1145–1150. [Google Scholar] [CrossRef]
- Milan, A.M.; Pundir, S.; Pileggi, C.A.; Markworth, J.F.; Lewandowski, P.A.; Cameron-Smith, D. Comparisons of the Postprandial Inflammatory and Endotoxaemic Responses to Mixed Meals in Young and Older Individuals: A Randomised Trial. Nutrients 2017, 9, 354. [Google Scholar] [CrossRef]
- Wu, Y.; Zhou, B.P. TNF-α/NFκ-B/Snail Pathway in Cancer Cell Migration and Invasion. Br. J. Cancer 2010, 102, 639–644. [Google Scholar] [CrossRef]
- Wei, J.; Xu, H.; Davies, J.L.; Hemmings, G.P. Increase of Plasma IL-6 Concentration with Age in Healthy Subjects. Life Sci. 1992, 51, 1953–1956. [Google Scholar] [CrossRef]
- Michaud, M.; Balardy, L.; Moulis, G.; Gaudin, C.; Peyrot, C.; Vellas, B.; Cesari, M.; Nourhashemi, F. Proinflammatory Cytokines, Aging, and Age-Related Diseases. J. Am. Med. Dir. Assoc. 2013, 14, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Rane, M. Interleukin-6 Signaling and Anti-Interleukin-6 Therapeutics in Cardiovascular Disease. Circ. Res. 2021, 128, 1728–1746. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an Endocrine Organ: Focus on Muscle-Derived Interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Steensberg, A.; Fischer, C.; Keller, C.; Keller, P.; Plomgaard, P.; Wolsk-Petersen, E.; Febbraio, M. The Metabolic Role of IL-6 Produced during Exercise: Is IL-6 an Exercise Factor? Proc. Nutr. Soc. 2004, 63, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Olin, A.C.; Rosengren, A.; Thelle, D.S.; Lissner, L.; Bake, B.; Torén, K. Height, Age, and Atopy Are Associated with Fraction of Exhaled Nitric Oxide in a Large Adult General Population Sample. Chest 2006, 130, 1319–1325. [Google Scholar] [CrossRef] [PubMed]
- Teng, K.T.; Chang, C.Y.; Chang, L.F.; Nesaretnam, K. Modulation of Obesity-Induced Inflammation by Dietary Fats: Mechanisms and Clinical Evidence. Nutr. J. 2014, 13, 12. [Google Scholar] [CrossRef]
- Ricciardolo, F.L.M.; Sterk, P.J.; Gaston, B.; Folkerts, G. Nitric Oxide in Health and Disease of the Respiratory System. Physiol. Rev. 2004, 84, 731–765. [Google Scholar] [CrossRef]
- Laprise, C.; Laviolette, M.; Boutet, M.; Boulet, L.P. Asymptomatic Airway Hyperresponsiveness: Relationships with Airway Inflammation and Remodelling. Eur. Respir. J. 1999, 14, 63–73. [Google Scholar] [CrossRef]
- Scott, H.A.; Latham, J.R.; Callister, R.; Pretto, J.J.; Baines, K.; Saltos, N.; Upham, J.W.; Wood, L.G. Acute Exercise Is Associated with Reduced Exhaled Nitric Oxide in Physically Inactive Adults with Asthma. Ann. Allergy Asthma Immunol. 2015, 114, 470–479. [Google Scholar] [CrossRef]
- Alving, K.; Anolik, R.; Crater, G.; LaForce, C.F.; Rickard, K. Validation of a New Portable Exhaled Nitric Oxide Analyzer, NIOX VERO®: Randomized Studies in Asthma. Pulm. Ther. 2017, 3, 207–218. [Google Scholar] [CrossRef] [Green Version]
- Tanabe, Y.; Harada, N.; Ito, J.; Matsuno, K.; Takeshige, T.; Harada, S.; Takemasa, M.; Kotajima, M.; Ishimori, A.; Katsura, Y.; et al. Difference between Two Exhaled Nitric Oxide Analyzers, NIOX VERO® Electrochemical Hand-Held Analyzer and NOA280i ® Chemiluminescence Stationary Analyzer. J. Asthma 2019, 56, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Endo, Y.; Shirai, T.; Akamatsu, T.; Asada, K. Comparison of Fractional Exhaled Nitric Oxide Levels Measured Using the NIOX VERO and NOA 280i. Ann. Allergy Asthma Immunol. 2017, 119, 383–385. [Google Scholar] [CrossRef] [PubMed]
- Svendsen, I.S.; Killer, S.C.; Gleeson, M. Influence of Hydration Status on Changes in Plasma Cortisol, Leukocytes, and Antigen-Stimulated Cytokine Production by Whole Blood Culture Following Prolonged Exercise. ISRN Nutr. 2014, 2014, 561401. [Google Scholar] [CrossRef] [PubMed]
- Klarlund Pedersen, B.; Dyhr Toft, A.; Pedersen, B.K. Reviews EVects of Exercise on Lymphocytes and Cytokines. Br. J. Sports Med. 2000, 34, 246–251. [Google Scholar] [CrossRef]
- Teeman, C.S.; Kurti, S.P.; Cull, B.J.; Emerson, S.R.; Haub, M.D.; Rosenkranz, S.K. Postprandial Lipemic and Inflammatory Responses to High-Fat Meals: A Review of the Roles of Acute and Chronic Exercise. Nutr. Metab. 2016, 13, 80. [Google Scholar] [CrossRef]
- Yamada, T.; Takaoka, A. Tumor Necrosis Factor. Handb. Horm. Comp. Endocrinol. Basic Clin. Res. 2021, 328, 441–445. [Google Scholar] [CrossRef]
| YA (n = 12/5 M, 7 F) | OA (n = 12/8 M, 4 F) | |||||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||
| Age (years) | 23.4 | ± | 3.8 | 67.4 | ± | 5.0 * |
| Height (cm) | 167.5 | ± | 8.1 | 176.0 | ± | 8.9 * |
| Weight (kg) | 71.4 | ± | 17.3 | 80.7 | ± | 15.1 |
| Body Mass Index (BMI) (kg/m2) | 25.3 | ± | 5.0 | 25.8 | ± | 3.1 |
| Total Body Fat (%) | 28.2 | ± | 8.7 | 33.1 | ± | 6.5 |
| Android Body Fat (%) | 30.1 | ± | 13.1 | 40.3 | ± | 9.7 |
| YA (n = 12/5 M, 7 F) | OA (n = 12/8 M, 4 F) | |||||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||
| VO2peak (L/min) | 2.3 | ± | 0.4 | 2.3 | ± | 0.8 |
| VO2peak (mL/kg/min) | 33.4 | ± | 5.3 | 28.3 | ± | 6.7 * |
| Peak Power (Watts) | 199.2 | ± | 42.1 | 198.3 | ± | 67.9 |
| Peak Heart rate (bpm) | 187.9 | ± | 11.7 | 156.6 | ± | 8.5 * |
| Exercise Duration (min) | 88.1 | ± | 15.2 | 105.6 | ± | 25.7 |
| Exercise Bout Expenditure (kcals) | 642.6 | ± | 155.4 | 726.7 | ± | 135.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kurti, S.P.; Wisseman, W.S.; Miller, M.E.; Frick, H.L.; Malin, S.K.; Emerson, S.R.; Edwards, D.A.; Edwards, E.S. Acute Exercise and the Systemic and Airway Inflammatory Response to a High-Fat Meal in Young and Older Adults. Metabolites 2022, 12, 853. https://doi.org/10.3390/metabo12090853
Kurti SP, Wisseman WS, Miller ME, Frick HL, Malin SK, Emerson SR, Edwards DA, Edwards ES. Acute Exercise and the Systemic and Airway Inflammatory Response to a High-Fat Meal in Young and Older Adults. Metabolites. 2022; 12(9):853. https://doi.org/10.3390/metabo12090853
Chicago/Turabian StyleKurti, Stephanie P., William S. Wisseman, Molly E. Miller, Hannah L. Frick, Steven K. Malin, Sam R. Emerson, David A. Edwards, and Elizabeth S. Edwards. 2022. "Acute Exercise and the Systemic and Airway Inflammatory Response to a High-Fat Meal in Young and Older Adults" Metabolites 12, no. 9: 853. https://doi.org/10.3390/metabo12090853
APA StyleKurti, S. P., Wisseman, W. S., Miller, M. E., Frick, H. L., Malin, S. K., Emerson, S. R., Edwards, D. A., & Edwards, E. S. (2022). Acute Exercise and the Systemic and Airway Inflammatory Response to a High-Fat Meal in Young and Older Adults. Metabolites, 12(9), 853. https://doi.org/10.3390/metabo12090853







