The Obesogenic and Glycemic Effect of Bariatric Surgery in a Family with a Melanocortin 4 Receptor Loss-of-Function Mutation
Abstract
:1. Introduction
2. Materials and Methods
3. Results
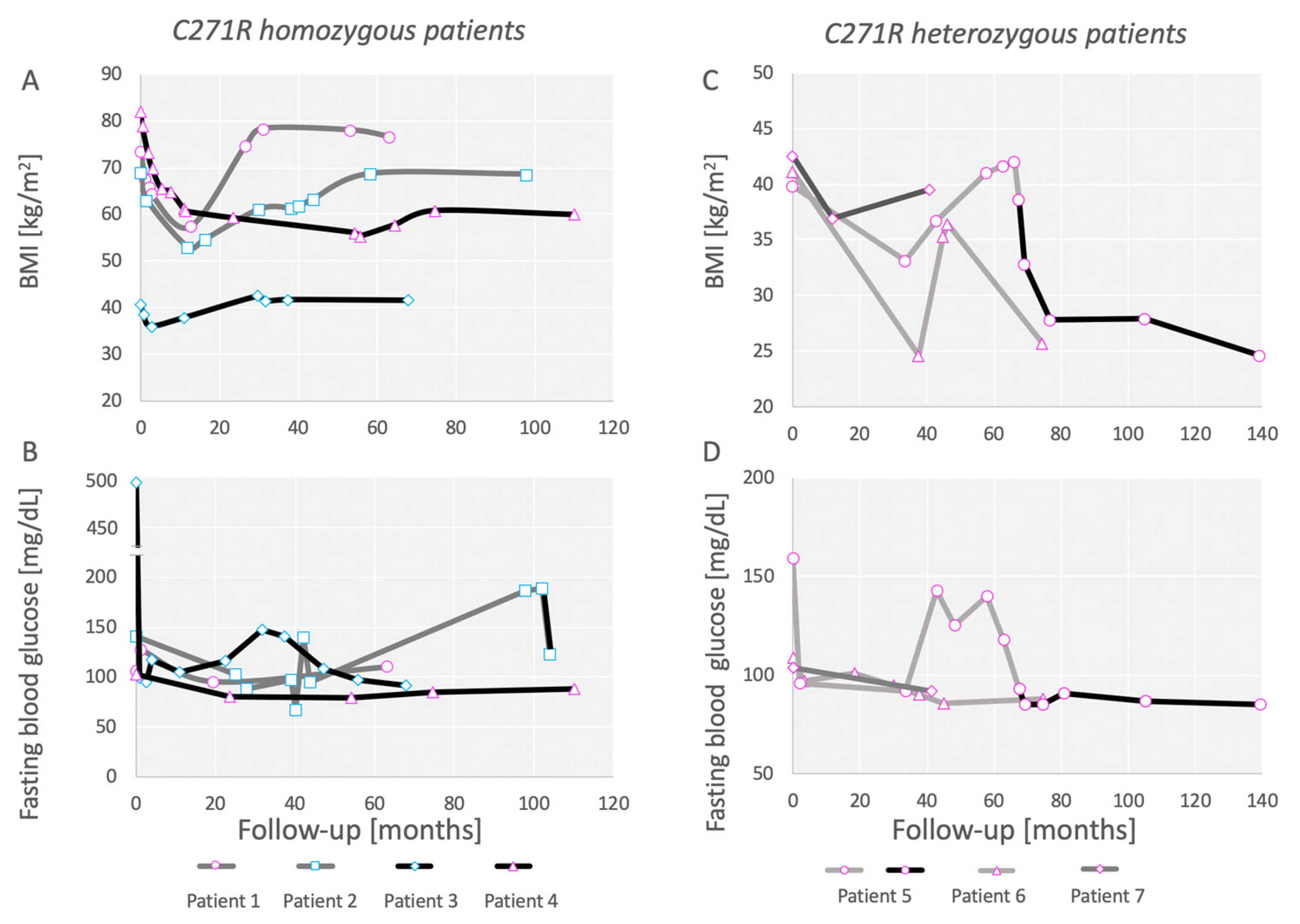
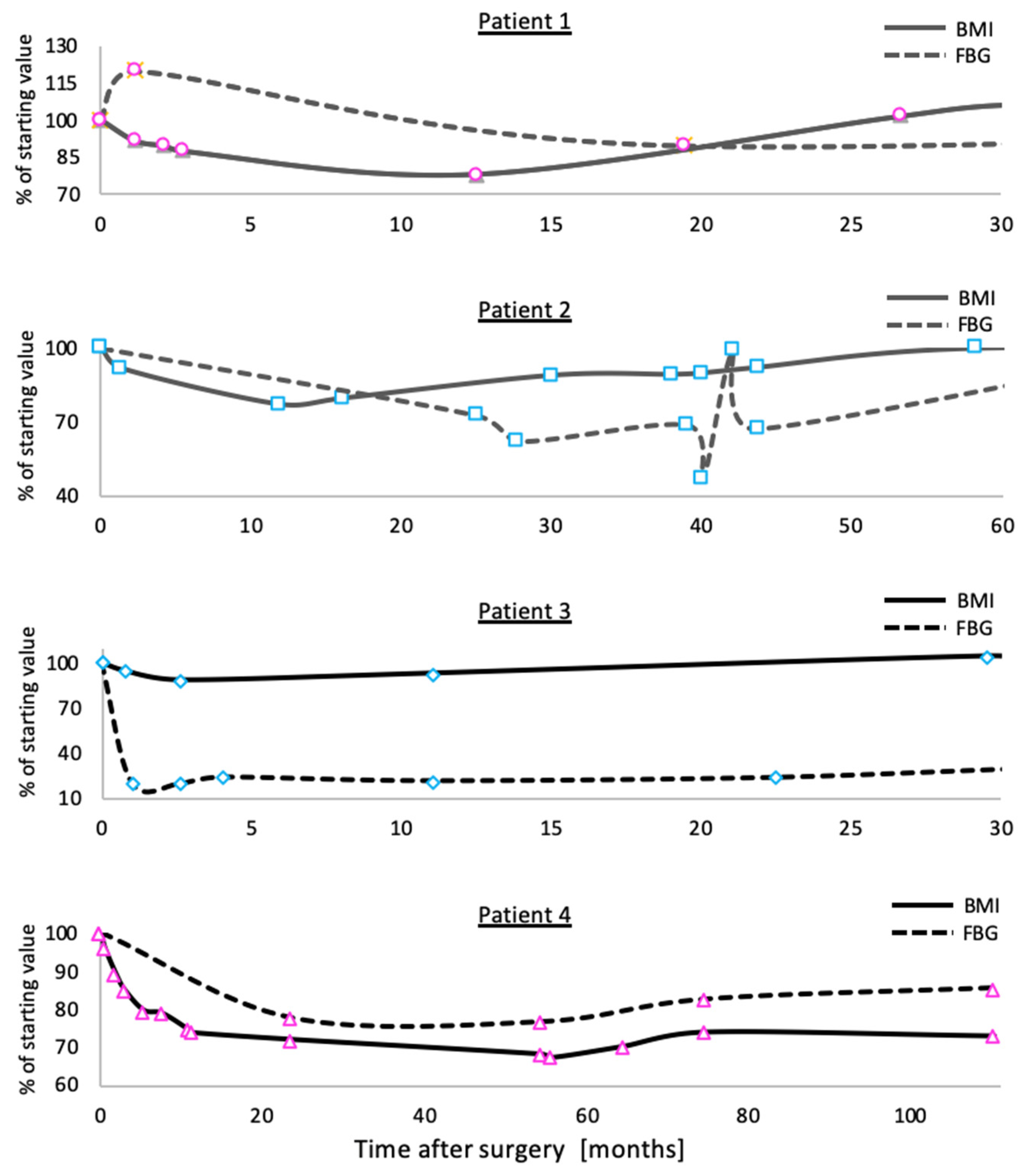
3.1. Outcomes of Bariatric Surgery in a Family Carrying the C271R Mutation in MC4R
3.2. Patients Homozygous for C271R Mutation in MC4R
3.3. Patients Heterozygous for C271R Mutation in MC4R
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tao, Y.-X. The Melanocortin-4 Receptor: Physiology, Pharmacology, and Pathophysiology. Endocr. Rev. 2010, 31, 506–543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oswal, A.; Yeo, G.S.H. The leptin melanocortin pathway and the control of body weight: Lessons from human and murine genetics. Obes. Rev. 2007, 8, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Garfield, A.S.; Li, C.; Madara, J.C.; Shah, B.P.; Webber, E.; Steger, J.S.; Campbell, J.N.; Gavrilova, O.; Lee, C.E.; Olson, D.P.; et al. A neural basis for melanocortin-4 receptor–regulated appetite. Nat. Neurosci. 2015, 18, 863–871. [Google Scholar] [CrossRef] [Green Version]
- Rossi, J.; Balthasar, N.; Olson, D.; Scott, M.; Berglund, E.; Lee, C.E.; Choi, M.J.; Lauzon, D.; Lowell, B.B.; Elmquist, J.K. Melanocortin-4 Receptors Expressed by Cholinergic Neurons Regulate Energy Balance and Glucose Homeostasis. Cell Metab. 2011, 13, 195–204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shah, B.P.; Vong, L.; Olson, D.P.; Koda, S.; Krashes, M.J.; Ye, C.; Yang, Z.; Fuller, P.M.; Elmquist, J.K.; Lowell, B.B. MC4R-expressing glutamatergic neurons in the paraventricular hypothalamus regulate feeding and are synaptically connected to the parabrachial nucleus. Proc. Natl. Acad. Sci. USA 2014, 111, 13193–13198. [Google Scholar] [CrossRef] [Green Version]
- Balthasar, N.; Dalgaard, L.; Lee, C.E.; Yu, J.; Funahashi, H.; Williams, T.; Ferreira, M.; Tang, V.; McGovern, R.A.; Kenny, C.D.; et al. Divergence of Melanocortin Pathways in the Control of Food Intake and Energy Expenditure. Cell 2005, 123, 493–505. [Google Scholar] [CrossRef]
- Farooqi, I.S.; Keogh, J.M.; Yeo, G.S.H.; Lank, E.J.; Cheetham, T.; O’Rahilly, S. Clinical Spectrum of Obesity and Mutations in the Melanocortin 4 Receptor Gene. N. Engl. J. Med. 2003, 348, 1085–1095. [Google Scholar] [CrossRef] [Green Version]
- Loos, R.J.F.; Lindgren, C.M.; Li, S.; Wheeler, E.; Zhao, J.H.; Prokopenko, I.; Inouye, M.; Freathy, R.M.; Attwood, A.P.; Beckmann, J.S.; et al. Common variants near MC4R are associated with fat mass, weight and risk of obesity. Nat. Genet. 2008, 40, 768–775. [Google Scholar] [CrossRef] [Green Version]
- Mägi, R.; Manning, S.; Yousseif, A.; Pucci, A.; Santini, F.; Karra, E.; Querci, G.; Pelosini, C.; McCarthy, M.I.; Lindgren, C.M.; et al. Contribution of 32 GWAS-Identified Common Variants to Severe Obesity in European Adults Referred for Bariatric Surgery. PLoS ONE 2013, 8, e70735. [Google Scholar] [CrossRef] [Green Version]
- Lotta, L.A.; Mokrosinski, J.; De Oliveira, E.M.; Li, C.; Sharp, S.J.; Luan, J.; Brouwers, B.; Ayinampudi, V.; Bowker, N.; Kerrison, N.; et al. Human Gain-of-Function MC4R Variants Show Signaling Bias and Protect against Obesity. Cell 2019, 177, 597–607e9. [Google Scholar] [CrossRef] [Green Version]
- Sjöström, L.; Lindroos, A.-K.; Peltonen, M.; Torgerson, J.; Bouchard, C.; Carlsson, B.; Dahlgren, S.; Larsson, B.; Narbro, K.; Sjöström, C.D.; et al. Lifestyle, Diabetes, and Cardiovascular Risk Factors 10 Years after Bariatric Surgery. N. Engl. J. Med. 2004, 351, 2683–2693. [Google Scholar] [CrossRef]
- Inge, T.H.; Courcoulas, A.P.; Jenkins, T.M.; Michalsky, M.; Helmrath, M.A.; Brandt, M.L.; Harmon, C.M.; Zeller, M.H.; Chen, M.K.; Xanthakos, S.A.; et al. Weight Loss and Health Status 3 Years after Bariatric Surgery in Adolescents. N. Engl. J. Med. 2016, 374, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Mingrone, G.; Panunzi, S.; De Gaetano, A.; Guidone, C.; Iaconelli, A.; Leccesi, L.; Nanni, G.; Pomp, A.; Castagneto, M.; Ghirlanda, G.; et al. Bariatric Surgery versus Conventional Medical Therapy for Type 2 Diabetes. N. Engl. J. Med. 2012, 366, 1577–1585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sjöström, L. Review of the key results from the Swedish Obese Subjects (SOS) trial—A prospective controlled intervention study of bariatric surgery. J. Intern. Med. 2013, 273, 219–234. [Google Scholar] [CrossRef] [PubMed]
- Rubino, F.; Nathan, D.M.; Eckel, R.H.; Schauer, P.R.; Alberti, K.G.M.M.; Zimmet, P.Z.; Del Prato, S.; Ji, L.; Sadikot, S.M.; Herman, W.H.; et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: A joint statement by international diabetes organizations. Diabetes Care 2016, 39, 861–877. [Google Scholar] [CrossRef] [Green Version]
- Adams, T.D.; Davidson, L.E.; Litwin, S.E.; Kim, J.; Kolotkin, R.; Nanjee, M.N.; Gutierrez, J.M.; Frogley, S.J.; Ibele, A.R.; Brinton, E.A.; et al. Weight and Metabolic Outcomes 12 Years after Gastric Bypass. N. Engl. J. Med. 2017, 377, 1143–1155. [Google Scholar] [CrossRef]
- Ben-Zvi, D.; Meoli, L.; Abidi, W.M.; Nestoridi, E.; Panciotti, C.; Castillo, E.; Pizarro, P.; Shirley, E.; Gourash, W.F.; Thompson, C.C.; et al. Time-Dependent Molecular Responses Differ between Gastric Bypass and Dieting but Are Conserved Across Species. Cell Metab. 2018, 28, 310–323.e6. [Google Scholar] [CrossRef] [Green Version]
- Liou, A.P.; Paziuk, M.; Luevano, J.-M., Jr.; Machineni, S.; Turnbaugh, P.J.; Kaplan, L.M. Conserved Shifts in the Gut Microbiota Due to Gastric Bypass Reduce Host Weight and Adiposity. Sci. Transl. Med. 2013, 5, 178ra41. [Google Scholar] [CrossRef] [Green Version]
- Saeidi, N.; Meoli, L.; Nestoridi, E.; Gupta, N.K.; Kvas, S.; Kucharczyk, J.; Bonab, A.A.; Fischman, A.J.; Yarmush, M.L.; Stylopoulos, N. Reprogramming of Intestinal Glucose Metabolism and Glycemic Control in Rats After Gastric Bypass. Science 2013, 341, 406–410. [Google Scholar] [CrossRef] [Green Version]
- Arble, D.M.; Sandoval, D.A.; Seeley, R.J. Mechanisms underlying weight loss and metabolic improvements in rodent models of bariatric surgery. Diabetologia 2015, 58, 211–220. [Google Scholar] [CrossRef] [Green Version]
- Rubino, F.; Forgione, A.; Cummings, D.E.; Vix, M.; Gnuli, D.; Mingrone, G.; Castagneto, M.; Marescaux, J. The Mechanism of Diabetes Control After Gastrointestinal Bypass Surgery Reveals a Role of the Proximal Small Intestine in the Pathophysiology of Type 2 Diabetes. Ann. Surg. 2006, 244, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Miras, A.; Le Roux, C. Mechanisms underlying weight loss after bariatric surgery. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Abu-Gazala, S.; Horwitz, E.; Schyr, R.B.-H.; Bardugo, A.; Israeli, H.; Hija, A.; Schug, J.; Shin, S.; Dor, Y.; Kaestner, K.H.; et al. Sleeve Gastrectomy Improves Glycemia Independent of Weight Loss by Restoring Hepatic Insulin Sensitivity. Diabetes 2018, 67, 1079–1085. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mirshahi, U.L.; Still, C.D.; Masker, K.K.; Gerhard, G.S.; Carey, D.J.; Mirshahi, T. TheMC4R(I251L) Allele Is Associated with Better Metabolic Status and More Weight Loss after Gastric Bypass Surgery. J. Clin. Endocrinol. Metab. 2011, 96, E2088–E2096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Toro-Martín, J.; Guénard, F.; Tchernof, A.; Pérusse, L.; Marceau, S.; Vohl, M.-C. Polygenic risk score for predicting weight loss after bariatric surgery. JCI Insight 2018, 3, e122011. [Google Scholar] [CrossRef] [Green Version]
- Censani, M.; Conroy, R.; Deng, L.; Oberfield, S.E.; McMahon, D.J.; Zitsman, J.L.; Leibel, R.L.; Chung, W.K.; Fennoy, I. Weight loss after bariatric surgery in morbidly obese adolescents with MC4R mutations. Obesity 2014, 22, 225–231. [Google Scholar] [CrossRef] [Green Version]
- Hatoum, I.J.; Stylopoulos, N.; Vanhoose, A.M.; Boyd, K.L.; Yin, D.P.; Ellacott, K.; Ma, L.L.; Blaszczyk, K.; Keogh, J.M.; Cone, R.D.; et al. Melanocortin-4 Receptor Signaling is Required for Weight Loss after Gastric Bypass Surgery. J. Clin. Endocrinol. Metab. 2012, 97, E1023–E1031. [Google Scholar] [CrossRef]
- Valette, M.; Poitou, C.; Le Beyec, J.; Bouillot, J.-L.; Clement, K.; Czernichow, S. Melanocortin-4 Receptor Mutations and Polymorphisms Do Not Affect Weight Loss after Bariatric Surgery. PLoS ONE 2012, 7, e48221. [Google Scholar] [CrossRef] [Green Version]
- Hao, Z.; Münzberg, H.; Rezai-Zadeh, K.; Keenan, M.; Coulon, D.; Lu, H.; Berthoud, H.-R.; Ye, J. Leptin deficient ob/ob mice and diet-induced obese mice responded differently to Roux-en-Y bypass surgery. Int. J. Obes. 2015, 39, 798–805. [Google Scholar] [CrossRef] [Green Version]
- Aslan, I.R.; Ranadive, S.A.; Ersoy, B.A.; Rogers, S.J.; Lustig, R.H.; Vaisse, C. Bariatric surgery in a patient with complete MC4R deficiency. Int. J. Obes. 2011, 35, 457–461. [Google Scholar] [CrossRef] [Green Version]
- Bonnefond, A.; Keller, R.; Meyre, D.; Stutzmann, F.; Thuillier, D.; Stefanov, D.G.; Froguel, P.; Horber, F.F.; Kral, J.G. Eating Behavior, Low-Frequency Functional Mutations in the Melanocortin-4 Receptor (MC4R) Gene, and Outcomes of Bariatric Operations: A 6-Year Prospective Study. Diabetes Care 2016, 39, 1384–1392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cooiman, M.; Kleinendorst, L.; Aarts, E.; Janssen, I.; van Amstel, H.P.; Blakemore, A.; Hazebroek, E.; Meijers-Heijboer, H.; van der Zwaag, B.; Berends, F.; et al. Genetic Obesity and Bariatric Surgery Outcome in 1014 Patients with Morbid Obesity. Obes. Surg. 2020, 30, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Jelin, E.B.; Daggag, H.; Speer, A.L.; Hameed, N.; Lessan, N.; Barakat, M.; Nadler, E.P. Melanocortin-4 receptor signaling is not required for short-term weight loss after sleeve gastrectomy in pediatric patients. Int. J. Obes. 2016, 40, 550–553. [Google Scholar] [CrossRef] [PubMed]
- Elkhenini, H.F.; New, J.P.; Syed, A.A. Five-year outcome of bariatric surgery in a patient with melanocortin-4 receptor mutation. Clin. Obes. 2014, 4, 121–124. [Google Scholar] [CrossRef]
- Vos, N.; Oussaada, S.M.; Cooiman, M.I.; Kleinendorst, L.; Ter Horst, K.W.; Hazebroek, E.J.; Romijn, J.A.; Serlie, M.J.; Mannens, M.M.A.M.; Van Haelst, M.M. Bariatric Surgery for Monogenic Non-syndromic and Syndromic Obesity Disorders. Curr. Diabetes Rep. 2020, 20, 44. [Google Scholar] [CrossRef]
- Israeli, H.; Degtjarik, O.; Fierro, F.; Chunilal, V.; Gill, A.K.; Roth, N.J.; Botta, J.; Prabahar, V.; Peleg, Y.; Chan, L.F.; et al. Structure reveals the activation mechanism of the MC4 receptor to initiate satiation signaling. Science 2021, 372, 808–814. [Google Scholar] [CrossRef]
- Agranat-Meged, A.; Ghanadri, Y.; Eisenberg, I.; Ben Neriah, Z.; Kieselstein-Gross, E.; Mitrani-Rosenbaum, S. Attention deficit hyperactivity disorder in obese melanocortin-4-receptor (MC4R) deficient subjects: A newly described expression of MC4R deficiency. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2008, 147B, 1547–1553. [Google Scholar] [CrossRef]
- Mulla, C.M.; Middelbeek, R.J.; Patti, M.-E. Mechanisms of weight loss and improved metabolism following bariatric surgery. Ann. N. Y. Acad. Sci. 2018, 1411, 53–64. [Google Scholar] [CrossRef]
- Hutch, C.R.; Sandoval, D. The Role of GLP-1 in the Metabolic Success of Bariatric Surgery. Endocrinology 2017, 158, 4139–4151. [Google Scholar] [CrossRef] [Green Version]
- Ryan, K.K.; Tremaroli, V.; Clemmensen, C.; Kovatcheva-Datchary, P.; Myronovych, A.; Karns, R.; Wilson-Pérez, H.E.; Sandoval, D.A.; Kohli, R.; Bäckhed, F.; et al. FXR is a molecular target for the effects of vertical sleeve gastrectomy. Nature 2014, 509, 183–188. [Google Scholar] [CrossRef] [Green Version]
- Stefater, M.A.; Wilson-Pérez, H.E.; Chambers, A.P.; Sandoval, D.A.; Seeley, R.J. All Bariatric Surgeries Are Not Created Equal: Insights from Mechanistic Comparisons. Endocr. Rev. 2012, 33, 595–622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cavin, J.-B.; Couvelard, A.; Lebtahi, R.; Ducroc, R.; Arapis, K.; Voitellier, E.; Cluzeaud, F.; Gillard, L.; Hourseau, M.; Mikail, N.; et al. Differences in Alimentary Glucose Absorption and Intestinal Disposal of Blood Glucose After Roux-en-Y Gastric Bypass vs Sleeve Gastrectomy. Gastroenterology 2016, 150, 454–464.e9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, Y.; Huang, Z.-P.; Liu, C.-Q.; Qi, L.; Sheng, Y.; Zou, D.-J. Modulation of the gut microbiome: A systematic review of the effect of bariatric surgery. Eur. J. Endocrinol. 2018, 178, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Pratt, J.S.A.; Browne, A.; Browne, N.T.; Bruzoni, M.; Cohen, M.; Desai, A.; Inge, T.; Linden, B.C.; Mattar, S.G.; Michalsky, M.; et al. ASMBS pediatric metabolic and bariatric surgery guidelines, 2018. Surg. Obes. Relat. Dis. 2018, 14, 882–901. [Google Scholar] [CrossRef]

| # | Year | Age | Sex | Weight (kg) | Height (cm) | Surgery | C271 Mutation |
|---|---|---|---|---|---|---|---|
| 1 | 2013 | 12 | F | 159 | 147 | LSG | Hom |
| 2 | 2011 | 12 | M | 167 | 156 | LSG | Hom |
| 3 | 2010 | 17 | M | 111 | 165 | LRYGB | Hom |
| 4 | 2005 | 16 | F | 200 | 157 | LRYGB | Hom |
| 5 | 2007 | 35 | F | 102 | 167 | LAGB | Het |
| 6 | 2010 | 41 | F | 99 | 155 | LAGB | Het |
| 7 | 2016 | 18 | F | 109 | 160 | LSG | Het |
| General | Main Clinical Parameters at the Surgery | Main Clinical Parameters at the Latest Time Point | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| # | C271R Mutation | Sex | Bariatric Surgery | Year, Age at Surgery | BMI (kg/m2) | Glucose (mg/dL) | A1c (%) | Blood Pressure (mmHg) | Major Morbidities | Followup (Months) | BMI (kg/m2) | Glucose (mg/dL) | A1c (%) | Blood Pressure (mmHg) | Major Morbidities |
| 1 | Hom | F | LSG | 2013, 12 | 73.6 | 106 | 5.8 | 149/118 | Anemia, asthma, OSA, hypertension, tibia vara | 63 | 76.7 | 110 | 5.1 | 123/72 | OSA, asthma, tibia vara |
| 2 | Hom | M | LSG | 2012, 12 | 68.9 | 141 | 6.9 | 141/113 | T2D, OSA, hypertension, tibia vara | 98 | 68.6 | 189 | 6.8 | 128/65 | T2D, OSA, tibia vara |
| LOAGB | 2020, 20 | 68.6 | 189 | 6.8 | 128/65 | T2D, OSA, tibia vara | 1 § | 67.5 | 123 | - | - | OSA, tibia vara | |||
| 3 | Hom | M | LRYGB | 2010, 17 | 40.8 | 495 | 12.9 | 139/78 | T2D, hypertension, hyperlipidemia, tibia vara | 68 | 41.7 | 92 | 6.2 | 133/72 | Tibia vara |
| 4 | Hom | F | LRYGB | 2005, 16 | 82.2 | 76 | - | 140/105 | Hypertension | 110 | 60 | 88 | - | 142/80 | Anemia, hypertension |
| 5 | Het | F | LAGB | 2007, 35 | 39.8 | 159 | 6.5 | 125/73 | T2D, asthma | 67 | 38.7 | 93 | 6.2 | 138/87 | Hypertension |
| 5 | LRYGB | 2013, 41 | 38.7 | 93 | 6.2 | 138/87 | Hypertension | 72 § | 24.6 | 85 | 5 | 141/87 | Hypertension, anemia | ||
| 6 | Het | F | LAGB | 2010, 41 | 41.2 | 109 | 5.1 | 125/75 | IFG, asthma | 75 | 25.8 | 88 | 4.5 | - | |
| 7 | Het | F | LSG | 2016, 18 | 42.6 | 104 | 5.6 | 133/76 | IFG | 41 | 39.5 | 92 | 4.5 | 126/70 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grinbaum, R.; Beglaibter, N.; Mitrani-Rosenbaum, S.; Kaplan, L.M.; Ben-Zvi, D. The Obesogenic and Glycemic Effect of Bariatric Surgery in a Family with a Melanocortin 4 Receptor Loss-of-Function Mutation. Metabolites 2022, 12, 430. https://doi.org/10.3390/metabo12050430
Grinbaum R, Beglaibter N, Mitrani-Rosenbaum S, Kaplan LM, Ben-Zvi D. The Obesogenic and Glycemic Effect of Bariatric Surgery in a Family with a Melanocortin 4 Receptor Loss-of-Function Mutation. Metabolites. 2022; 12(5):430. https://doi.org/10.3390/metabo12050430
Chicago/Turabian StyleGrinbaum, Ronit, Nahum Beglaibter, Stella Mitrani-Rosenbaum, Lee M. Kaplan, and Danny Ben-Zvi. 2022. "The Obesogenic and Glycemic Effect of Bariatric Surgery in a Family with a Melanocortin 4 Receptor Loss-of-Function Mutation" Metabolites 12, no. 5: 430. https://doi.org/10.3390/metabo12050430
APA StyleGrinbaum, R., Beglaibter, N., Mitrani-Rosenbaum, S., Kaplan, L. M., & Ben-Zvi, D. (2022). The Obesogenic and Glycemic Effect of Bariatric Surgery in a Family with a Melanocortin 4 Receptor Loss-of-Function Mutation. Metabolites, 12(5), 430. https://doi.org/10.3390/metabo12050430






