Drought-Induced Xylem Sulfate Activates the ABA-Mediated Regulation of Sulfate Assimilation and Glutathione Redox in Brassica napus Leaves
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Growth and Treatments
2.2. Measurement of Leaf Water Potential
2.3. Collection of Xylem and Phloem
2.4. Measurement of H2O2 Concentration
2.5. Determination of ABA Concentration
2.6. Sulfate and Cys Concentration
2.7. Measurement of GSH/GSSG
2.8. Measurement of NADPH/NADP+
2.9. Measurements of Antioxidant Enzyme Activities
2.10. RNA Extraction and Quantitative Real-Time PCR Analysis
2.11. Statistical Analysis
3. Results
3.1. Leaf Water Potential, H2O2 Concentration and Antioxidant Enzyme Activities
3.2. ABA and Sulfate Concentrations in Leaves, Roots, and Xylem
3.3. Cys and GSH Concentration, and the Expression of Sulfate Assimilation-Related Genes
3.4. Redox Status under Drought Stress
3.5. Correlation of Sulfate or ABA with Physiological Parameters Influenced by Drought Stress
4. Discussion
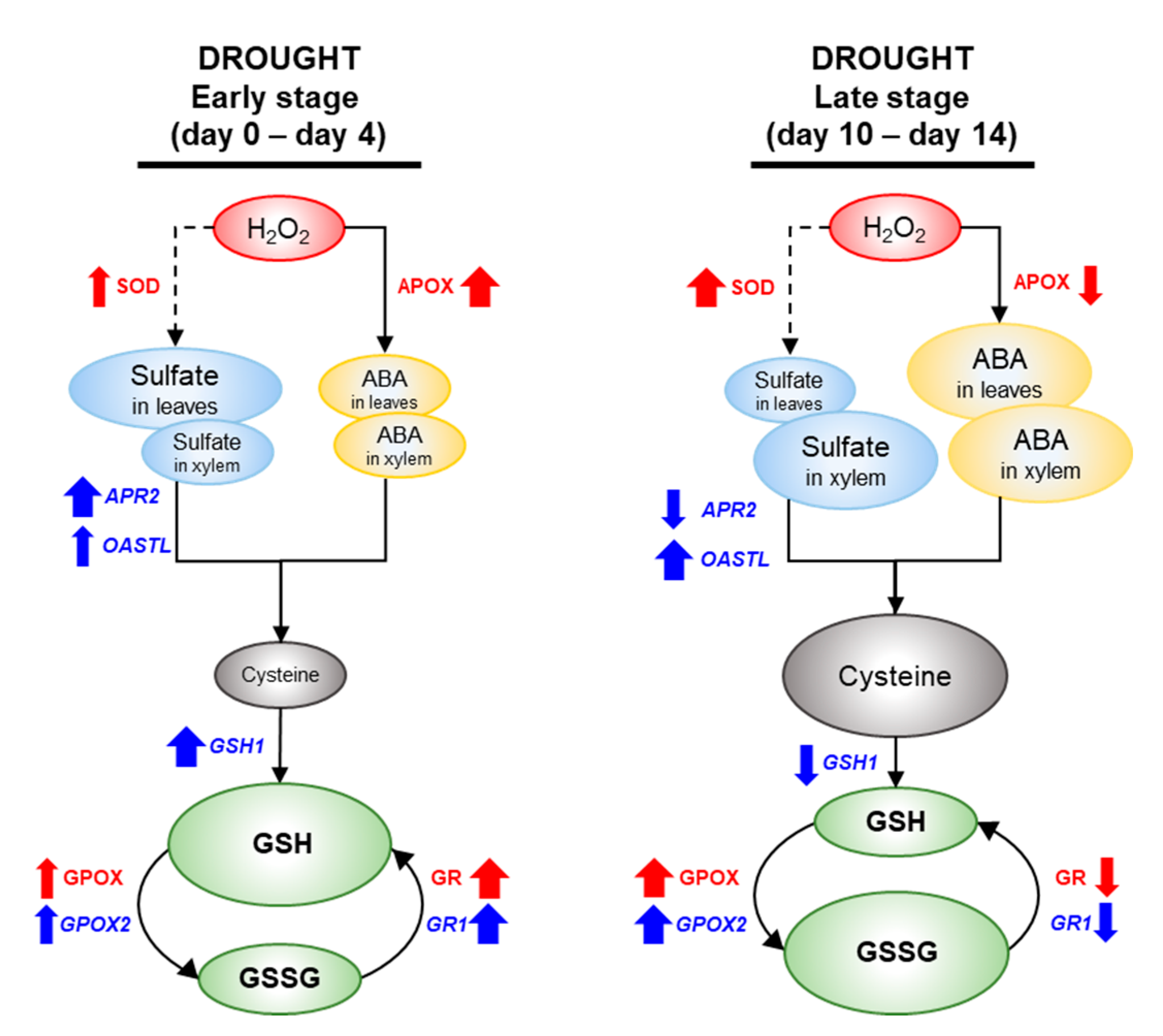
4.1. Drought Intensity-Responsive H2O2 Accumulation and Antioxidative Enzymatic Activity
4.2. Drought Intensity-Responsive Changes in Sulfate, ABA Allocation, and S-Assimilates in Leaves
4.3. Sulfate and/or ABA Roles in Regulating Sulfate Assimilation
4.4. Interactions between Sulfate and ABA in GSH-Based Redox Control
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gilbert, S.M.; Clarkson, D.T.; Cambridge, M.; Lambers, H.; Hawkesford, M.J. SO42– deprivation has an early effect on the content of ribulose-1,5-biphosphate carboxylase/oxygenase and photosynthesis in young leaves of wheat. Plant Physiol. 1997, 115, 1231–1239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, B.R.; Muneer, S.; Jung, W.J.; Avice, J.C.; Ourry, A.; Kim, T.H. Partitioning of newly absorbed and previously stored nitrogen and sulphur under sulphate deficient nutrition. J. Plant Nutr. 2014, 37, 1702–1716. [Google Scholar] [CrossRef]
- Muneer, S.; Lee, B.R.; Kim, K.Y.; Park, S.H.; Zhang, Q.; Kim, T.H. Involvement of sulphur nutrition in modulating iron deficiency responses in photosynthetic organelles of oilseed rape (Brassica napus L.). Photosynth. Res. 2014, 119, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.R.; Zaman, R.; Avice, J.C.; Ourry, A.; Kim, T.H. Sulfur use efficiency is a significant determinant of drought stress tolerance in relation to photosynthetic activity in Brassica napus cultivars. Front. Plant Sci. 2016, 7, 459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foyer, C.H.; Noctor, G. Redox signaling in plants. Antioxid. Redox Signal. 2013, 18, 2087–2090. [Google Scholar] [CrossRef]
- Park, S.H.; Lee, B.R.; La, V.H.; Mamun, M.A.; Bae, D.W.; Kim, T.H. Characterization of salicylic acid- and abscisic acid-mediated photosynthesis, Ca2+ and H2O2 accumulation in two distinct phases of drought stress intensity in Brassica napus. Environ. Exp. Bot. 2021, 186, 104434. [Google Scholar] [CrossRef]
- Baxter, A.; Mittler, R.; Suzuki, N. ROS as key players in plant stress signaling. J. Exp. Bot. 2014, 65, 1229–1240. [Google Scholar] [CrossRef]
- Choudhury, F.K.; Rivero, R.M.; Blumwald, E.; Mittler, R. Reactive oxygen species, abiotic stress and stress combination. Plant J. 2017, 90, 856–867. [Google Scholar] [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405410. [Google Scholar] [CrossRef]
- Lee, B.R.; Jin, Y.L.; Avice, J.C.; Cliquet, J.B.; Ourry, A.; Kim, T.H. Increased proline loading to phloem and its effects on nitrogen uptake and assimilation in water-stressed white clover (Trifolium repens). New Phytol. 2009, 182, 654–663. [Google Scholar] [CrossRef]
- Khan, N.A.; Anjum, N.A.; Nazar, R.; Iqbal, N. Increased activity of ATP sulfurylase and increased contents of cysteine and glutathione reduce high cadmium-induced oxidative stress in mustard cultivar with high photosynthetic potential. Russ. J. Plant Physiol. 2009, 56, 670–677. [Google Scholar] [CrossRef]
- Szalai, G.; Kellős, T.; Galiba, G.; Kocsy, G. Glutathione as an antioxidant and regulatory molecule in plants under abiotic stress conditions. J. Plant Growth Regul. 2009, 28, 66–80. [Google Scholar] [CrossRef]
- Meyer, A.J. The integration of glutathione homeostasis and redox signaling. J. Plant Physiol. 2008, 165, 1390–1403. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Ascorbate and glutathione: The heart of the redox hub. Plant Physiol. 2011, 155, 2–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, N.; Malagoli, M.; Wirtz, M.; Hell, R. Drought stress in maize causes differential acclimation responses of glutathione and sulfur metabolism in leaves and roots. BMC Plant Biol. 2016, 16, 247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Astolfi, S.; Zuchi, S. Adequate S supply protects barley plants from adverse effects of salinity stress by increasing thiol contents. Acta Physiol. Plant. 2013, 35, 175–181. [Google Scholar] [CrossRef]
- Aoyama, K.; Nakaki, T. Glutathione in Cellular Redox Homeostasis: Association with the Excitatory Amino Acid Carrier 1 (EAAC1). Molecules 2015, 20, 8742–8758. [Google Scholar] [CrossRef] [Green Version]
- Ernst, L.; Goodger, J.Q.D.; Alvarez, S.; Marsh, E.L.; Berla, B.; Lockhart, E.; Jung, J.; Li, P.; Bohnert, H.J.; Schachtman, D.P. Sulphate as a xylem-born chemical signal precedes the expression of ABA biosynthetic genes in maize roots. J. Exp. Bot. 2010, 61, 3395–3405. [Google Scholar] [CrossRef] [Green Version]
- Cao, M.J.; Wang, Z.; Zhao, Q.; Mao, J.L.; Speiser, A.; Wirtz, M.; Hell, R.; Zhu, J.K.; Xinag, C.B. Sulfate availability affect ABA levels and germination response to ABA and salt stress in Arabidopsis thaliana. Plant J. 2014, 77, 604–615. [Google Scholar] [CrossRef]
- Batool, S.; Uslu, V.V.; Rajab, H.; Ahmad, N.; Waadt, R.; Geiger, D.; Malagoli, M.; Xiang, C.B.; Hedrich, R.; Rennenberg, H.; et al. Sulfate is incorporated into cysteine to trigger ABA production and stomatal closure. Plant Cell 2018, 30, 2973–2987. [Google Scholar] [CrossRef]
- Sierla, M.; Waszczak, C.; Vahisalu, T.; Kangasjarvi, J. Reactive oxygen species in the regulation of stomatal movements. Plant Physiol. 2016, 170, 1569–1580. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malcheska, F.; Ahmad, A.; Batool, S.; Muller, H.M.; Ludwig-Muller, J.; Kreuzwieser, J.; Randewig, D.; Hänsch, R.; Mendel, R.R.; Hell, R.; et al. Drought enhanced xylem sap sulfate closes stomata by affecting ALMT12 and guard cell ABA synthesis. Plant Physiol. 2017, 174, 798–814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okuma, E.; Jahan, M.S.; Munemasa, S.; Hossain, M.A.; Muroyama, D.; Islam, M.M.; Ogawa, K.; Watanabe-Sugimoto, M.; Nakamura, Y.; Shimoishi, Y.; et al. Negative regulation of abscisic acid-induced stomatal closure by glutathione in Arabidopsis. J. Plant Physiol. 2011, 168, 2048–2055. [Google Scholar] [CrossRef] [PubMed]
- Speiser, A.; Silbermann, M.; Dong, Y.; Haberland, S.; Uslu, V.V.; Wang, S.; Banagsh, S.A.K.; Reichelt, M.; Meyer, A.J.; Wirtz, M.; et al. Sulfur partitioning between glutathione and protein synthesis determines plant growth. Plant Physiol. 2018, 177, 927–937. [Google Scholar] [CrossRef] [Green Version]
- Lee, B.R.; Jin, Y.L.; Park, S.H.; Zaman, R.; Zhang, Q.; Avice, J.C.; Ourry, A.; Kim, T.H. Genotypic variation in N uptake and assimilation estimated by 15N tracing in water deficit-stressed Brassica napus. Environ. Exp. Bot. 2015, 109, 73–79. [Google Scholar] [CrossRef]
- Lee, B.R.; Kim, K.Y.; Jung, W.J.; Avice, J.C.; Ourry, A.; Kim, T.H. Peroxidases and lignification in relation to the intensity of water-deficit stress in white clover (Trifolium repens L.). J. Exp. Bot. 2007, 58, 1271–1279. [Google Scholar] [CrossRef] [Green Version]
- La, V.H.; Lee, B.R.; Islam, M.T.; Park, S.H.; Jung, H.I.; Bae, D.W.; Kim, T.H. Characterization of salicylic acid-mediated modulation of the drought stress responses: Reactive oxygen species, proline, and redox state in Brassica napus. Environ. Exp. Bot. 2019, 157, 1–10. [Google Scholar] [CrossRef]
- Gaitonde, M. A spectrophotometric method for the direct determination of cysteine in the presence of other naturally occurring amino acids. Biochem. J. 1967, 104, 627–633. [Google Scholar] [CrossRef] [Green Version]
- Hasanuzzaman, M.; Hossain, M.A.; Fujita, M. Nitric oxide modulates antioxidant defense and the methylglyoxal detoxification system and reduces salinity-induced damage of wheat seedlings. Plant Biotecnol. Rep. 2011, 5, 353–365. [Google Scholar] [CrossRef]
- Lee, B.R.; Jin, Y.L.; Jung, W.J.; Avice, J.C.; Bertrand, A.M.; Ourry, A.; Park, C.W.; Kim, T.H. Water-deficit accumulates sugars by starch degradation—Not by de novo synthesis—In white clover leaves (Trifolium repens L.). Physiol. Plant. 2008, 134, 403–411. [Google Scholar] [CrossRef]
- Pinheiro, C.; Chaves, M.M. Photosynthesis and drought: Can we make metabolic connections from available data? J. Exp. Bot. 2011, 62, 869–882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bista, D.R.; Heckathorn, S.A.; Jayawardena, D.A.; Mishra, S.; Boldt, J.K. Effects of drought on nutrient uptake and the levels of nutrient-uptake proteins in roots of drought-sensitive and -tolerant grasses. Plants 2018, 7, 28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cruz de Carvalho, M.H.; Contour-Anse, D. (h)GR, beans and drought stress. Plant Signal. Behav. 2008, 3, 834–835. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foyer, C.H.; Noctor, G. Redox sensing and signalling associated with reactive oxygen in chloroplasts, peroxisomes and mitochondria. Physiol. Plant. 2003, 119, 355–364. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Wang, X.; Hu, Y.; Hu, W.; Bi, Y. Glucose-6-phosphate dehydrogenase plays a pivotal role in tolerance to drought stress in soybean roots. Plant Cell Rep. 2013, 32, 415–429. [Google Scholar] [CrossRef]
- Fortunato, A.A.; Debona, D.; Bernardeli, A.M.A.; Rodrigues, F.A. Changes in the antioxidant system in soybean leaves infected by Corynespora cassiicola. Phytopathology 2015, 105, 1050–1058. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Wu, M.; Teng, Y.; Jia, S.; Yu, D.; Wei, T.; Chen, C.; Song, W. Overexpression of the glutathione peroxidase 5 (RcGPOX5) gene from Rhodiola crenulata increases drought tolerance in Salvia miltiorrhiza. Front. Plant Sci. 2019, 9, 1950. [Google Scholar] [CrossRef]
- Mittova, V.; Theodoulou, F.L.; Kiddle, G.; Gómez, L.; Volokita, M.; Tal, M.; Foyer, C.H.; Guy, M. Coordinate induction of glutathione biosynthesis and glutathione-metabolizing enzymes is correlated with salt tolerance in tomato. FEBS Lett. 2003, 554, 417–421. [Google Scholar] [CrossRef] [Green Version]
- Rouphael, Y.; Cardarelli, M.; Schwarz, D.; Franken, P.; Colla, G. Effects of drought on nutrient uptake and assimilation in vegetable crops. In Plant Responses to Drought Stress, 1st ed.; Aroca, R., Ed.; Springer: Berlin, Germany, 2012; pp. 171–195. [Google Scholar]
- Herrera, K.; Cahoon, R.E.; Kumaran, S.; Jez, J. Reaction mechanism of glutathione synthetase from Arabidopsis thaliana: Site-directed mutagenesis of active site residues. J. Biol. Chem. 2007, 282, 17157–17165. [Google Scholar] [CrossRef] [Green Version]
- Barroso, C.; Romero, L.C.; Cejudo, F.J.; Vega, J.M.; Gotor, C. Salt-specific regulation of the cytosolic O-acetylserine(thiol)lyase gene from Arabidopsis thaliana is dependent on abscisic acid. Plant Mol. Biol. 1999, 40, 729–736. [Google Scholar] [CrossRef]
- Kocsy, G.; Szalai, G.; Galiba, G. Effect of osmotic stress on glutathione and hydroxymethyl-glutathione accumulation in wheat. J. Plant Physiol. 2004, 161, 785–794. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.J.; Zhou, Y.H.; Shi, K.; Zhou, J.; Foyer, C.H.; Yu, J.Q. Interplay between reactive oxygen species and hormones in the control of plant development and stress tolerance. J. Exp. Bot. 2015, 66, 2839–2856. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hieno, A.; Naznin, H.A.; Inaba-Hasegawa, K.; Yokogawa, T.; Hayami, N.; Nomoto, M.; Tada, Y.; Yokogawa, T.; Higuchi-Takeuchi, M.; Hanada, K.; et al. Transcriptome analysis and identification of a transcriptional regulatory network in the response to H2O2. Plant Physiol. 2019, 180, 1629–1646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- La, V.H.; Lee, B.R.; Islam, M.T.; Park, S.H.; Lee, H.; Bae, D.W.; Kim, T.H. Antagonistic shifting from abscisic acid- to salicylic acid-mediated sucrose accumulation contributes to drought tolerance in Brassica napus. Enviorn. Exp. Bot. 2019, 162, 38–47. [Google Scholar] [CrossRef]
- Lee, B.R.; La, V.H.; Park, S.H.; Mamun, M.A.; Bae, D.W.; Kim, T.H. H2O2-responsive hormonal status involves oxidative burst signaling and proline metabolism in Rapeseed leaves. Antioxidants 2022, 11, 566. [Google Scholar] [CrossRef]
- Rentel, M.C.; Lecourieux, D.; Ouaked, F.; Usher, S.L.; Petersen, L.; Okamoto, H.; Knight, H.; Peck, S.C.; Grierson, C.S.; Hirt, H.; et al. OXI1 kinase is necessary for oxidative burst-mediated signaling in Arabidopsis. Nature 2004, 427, 858–861. [Google Scholar] [CrossRef]
- Koprivova, A.; North, K.A.; Kopriva, S. Complex signaling network in regulation of adenosine 5’-phosphosulfate reductase by salt stress in Arabidopsis roots. Plant Physiol. 2008, 146, 1408–1420. [Google Scholar] [CrossRef] [Green Version]
- Rajab, H.; Khan, M.S.; Malagoli, M.; Hell, R.; Wirtz, M. Sulfate-induced stomata closure requires the canonical ABA signal transduction machinery. Plants 2019, 8, 21. [Google Scholar] [CrossRef] [Green Version]
- Tausz, M.; Sircelj, H.; Grill, D. The glutathione system as a stress marker in plant ecophysiology: Is a stress–response concept valid? J. Expt. Bot. 2004, 55, 1955–1962. [Google Scholar] [CrossRef]
- Marty, L.; Siala, W.; Schwarzlander, M.; Fricker, M.D.; Wirtz, M.; Sweetlove, L.J.; Meyer, Y.; Meyer, A.J.; Reichheld, J.P.; Hell, R. The NADPH-dependent thioredoxin system constitutes a functional backup for cytosolic glutathione reductase in Arabidopsis. Proc. Natl. Acad. Sci. USA 2009, 106, 9109–9114. [Google Scholar] [CrossRef]
- Li, W.; Huai, Z.; Li, P.; Raza, A.; Mubarik, M.S.; Habib, M.; Fiaz, S.; Zhang, B.; Pan, J.; Khan, R.S.A. Genome-wide characterization of glutathione peroxidase (GPX) gene family in rapeseed (Brassica napus L.) revealed their role in multiple abiotic stress response and hormone signaling. Antioxidants 2021, 10, 1481. [Google Scholar] [CrossRef] [PubMed]

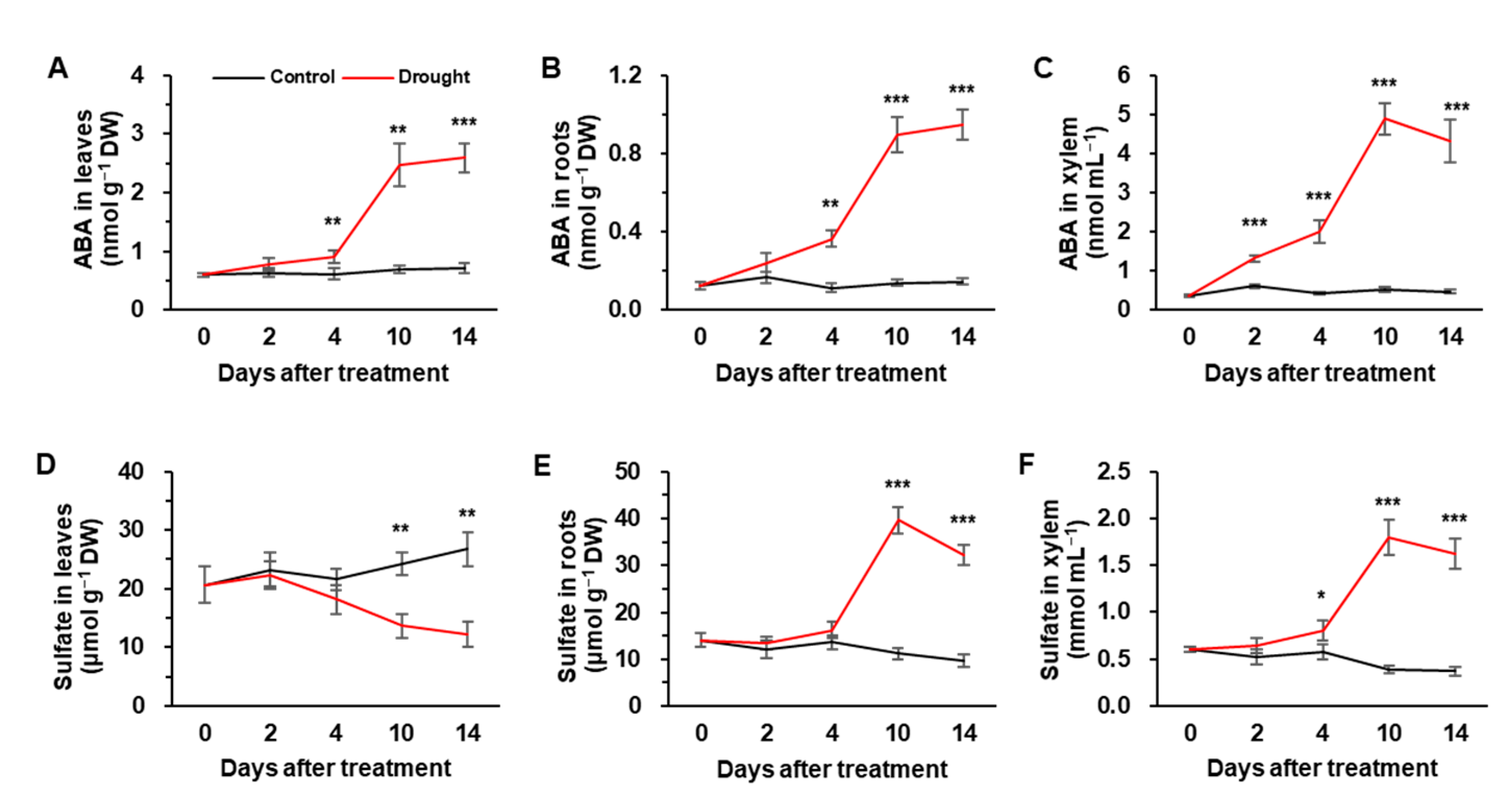
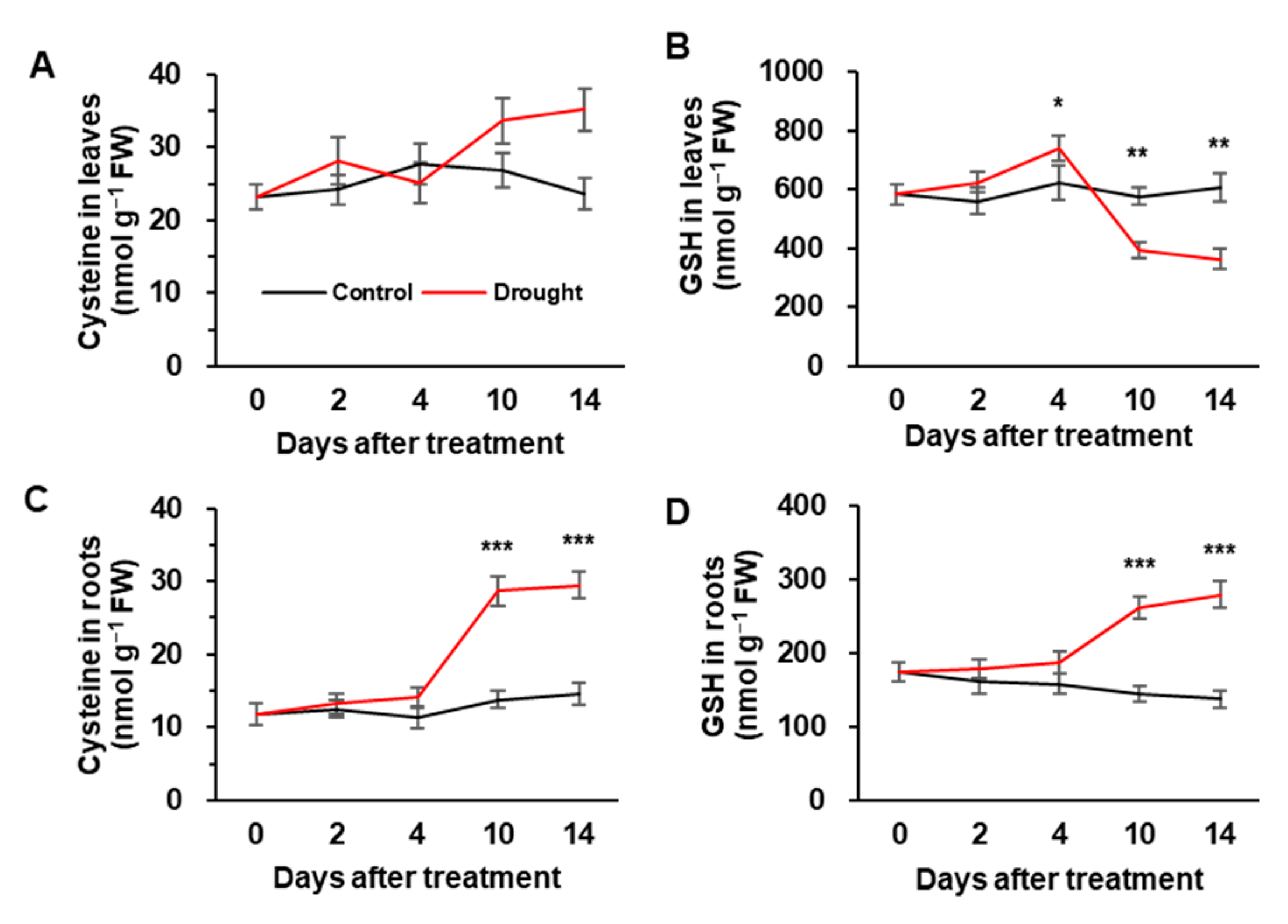
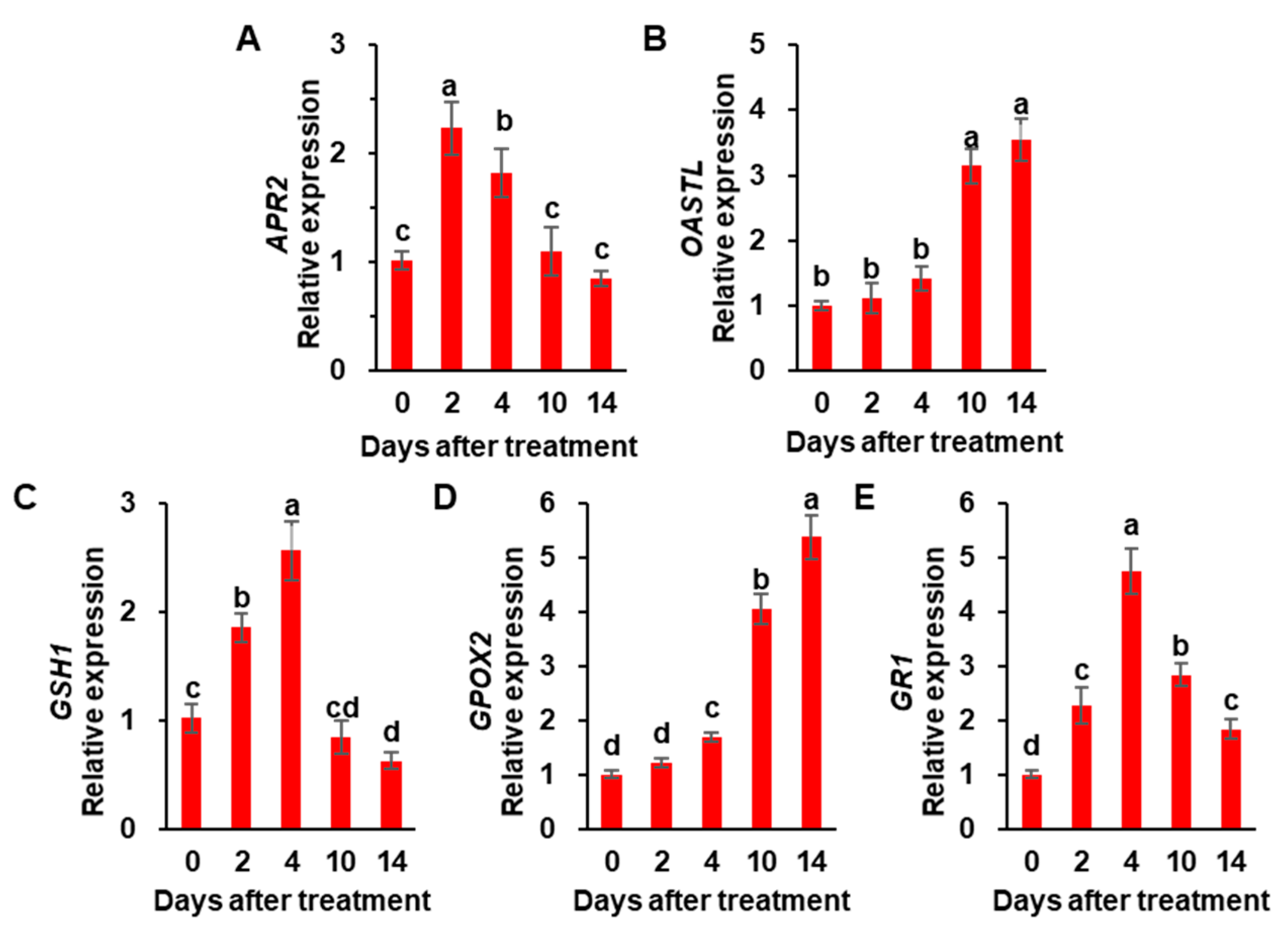
| Physiological Parameters | Days after Treatment | ||||
|---|---|---|---|---|---|
| /Treatment | 0 | 2 | 4 | 10 | 14 |
| Leaf water potential (Ψw, MPa) | |||||
| Control | −0.39 | −0.42 | −0.45 | −0.54 | −0.53 |
| Drought | −0.39 | −0.49 | −0.57 * | −1.12 ** | −1.34 *** |
| H2O2 concentration (nmol g−1 FW) | |||||
| Control | 2.97 | 3.06 | 3.16 | 3.08 | 3.47 |
| Drought | 2.97 | 3.54 | 3.85 | 7.86 *** | 8.67 *** |
| Leaf biomass (Dry matter, g plant−1) | |||||
| Control | 8.23 | 8.56 | 10.89 | 14.07 | 16.48 |
| Drought | 8.23 | 8.47 | 8.92 ** | 9.49 *** | 9.46 *** |
| Root biomass (Dry matter, g plant−1) | |||||
| Control | 5.86 | 5.09 | 6.28 | 7.14 | 8.16 |
| Drought | 5.86 | 5.35 | 6.05 | 6.64 | 7.23 |
| GSH | GSSG | GSH/GSSG | NADPH | NADP+ | NADPH/NADP+ | ||
|---|---|---|---|---|---|---|---|
| Day | Treatment | (nmol g−1 FW) | (nmol g−1 FW) | Ratio | (nmol g−1 FW) | (nmol g−1 FW) | Ratio |
| 0 | Control | 582 ± 35 | 44.3 ± 3.7 | 13.2 ± 0.7 | 3.2 ± 0.3 | 6.0 ± 0.4 | 0.53 ± 0.04 |
| Drought | 582 ± 35 | 44.3 ± 3.7 | 13.2 ± 0.7 | 3.2 ± 0.3 | 6.0 ± 0.4 | 0.53 ± 0.04 | |
| 2 | Control | 560 ± 46 | 45.5 ± 5.7 | 12.4 ± 0.6 | 3.7 ± 0.3 | 7.1 ± 0.6 | 0.53 ± 0.04 |
| Drought | 697 ± 37 * | 50.4 ± 4.3 | 13.9 ± 0.5 * | 5.0 ± 0.4 * | 6.9 ± 0.5 | 0.73 ± 0.09 * | |
| 4 | Control | 620 ± 58 | 48.1 ± 4.6 | 12.9 ± 0.8 | 3.3 ± 0.4 | 6.5 ± 0.4 | 0.51 ± 0.07 |
| Drought | 741 ± 42 * | 63.3 ± 5.2 * | 11.7 ± 0.4 | 4.9 ± 0.3 ** | 7.3 ± 0.4 | 0.67 ± 0.07 | |
| 10 | Control | 575 ± 30 | 51.2 ± 4.8 | 11.3 ± 0.5 | 3.6 ± 0.4 | 5.6 ± 0.5 | 0.65 ± 0.11 |
| Drought | 392 ± 28 ** | 78.2 ± 5.9 ** | 5.1 ± 0.7 *** | 3.0 ± 0.4 | 9.2 ± 0.8 ** | 0.33 ± 0.02 ** | |
| 14 | Control | 606 ± 48 | 51.8 ± 4.9 | 11.7 ± 0.7 | 3.9 ± 0.3 | 5.4 ± 0.3 | 0.73 ± 0.08 |
| Drought | 363 ± 33 ** | 81.4 ± 6.2 ** | 4.5 ± 0.6 *** | 3.8 ± 0.3 | 10.3 ± 0.7 *** | 0.37 ± 0.04 ** |
| Xylem | Leaves | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ψw | Sulfate | ABA | Sulfate | ABA | H2O2 | GR | GPOX | OASTL | GSH1 | Cys | GSH | GSH/ GSSG | NADPH | NADPH/ NADP+ | |
| Ψw | - | 0.922 *** | −0.915 *** | 0.866 *** | −0.972 *** | −0.965 *** | 0.372 | −0.966 *** | −0.974 *** | 0.616 ** | −0.830 *** | 0.845 *** | 0.958 *** | 0.358 | 0.789 *** |
| Sulfate in xylem | - | - | 0.963 *** | −0.847 *** | 0.978 *** | 0.959 *** | −0.405 | 0.975 *** | 0.949 *** | −0.607 ** | 0.86 3 *** | −0.880 *** | −0.974 *** | −0.467 * | −0.826 *** |
| ABA in xylem | - | - | - | −0.821 *** | 0.945 *** | 0.929 *** | −0.234 | 0.955 *** | 0.934 *** | −0.461 * | 0.855 *** | −0.771 *** | −0.952 *** | −0.349 | −0.765 *** |
| Sulfate in leaves | - | - | - | - | −0.837 *** | −0.874 *** | 0.509 * | −0.857 *** | −0.898 *** | 0.538 * | −0.694 *** | 0.769 *** | 0.864 *** | 0.358 | 0.708 *** |
| ABA in leaves | - | - | - | - | - | 0.971 *** | −0.389 | 0.983 *** | 0.960 *** | −0.635 ** | 0.868 *** | −0.887 *** | −0.976 *** | −0.426 | −0.816 *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, B.-R.; Park, S.-H.; La, V.H.; Bae, D.-W.; Kim, T.-H. Drought-Induced Xylem Sulfate Activates the ABA-Mediated Regulation of Sulfate Assimilation and Glutathione Redox in Brassica napus Leaves. Metabolites 2022, 12, 1190. https://doi.org/10.3390/metabo12121190
Lee B-R, Park S-H, La VH, Bae D-W, Kim T-H. Drought-Induced Xylem Sulfate Activates the ABA-Mediated Regulation of Sulfate Assimilation and Glutathione Redox in Brassica napus Leaves. Metabolites. 2022; 12(12):1190. https://doi.org/10.3390/metabo12121190
Chicago/Turabian StyleLee, Bok-Rye, Sang-Hyun Park, Van Hien La, Dong-Won Bae, and Tae-Hwan Kim. 2022. "Drought-Induced Xylem Sulfate Activates the ABA-Mediated Regulation of Sulfate Assimilation and Glutathione Redox in Brassica napus Leaves" Metabolites 12, no. 12: 1190. https://doi.org/10.3390/metabo12121190
APA StyleLee, B.-R., Park, S.-H., La, V. H., Bae, D.-W., & Kim, T.-H. (2022). Drought-Induced Xylem Sulfate Activates the ABA-Mediated Regulation of Sulfate Assimilation and Glutathione Redox in Brassica napus Leaves. Metabolites, 12(12), 1190. https://doi.org/10.3390/metabo12121190







