Gut Microbiota Metabolites in Major Depressive Disorder—Deep Insights into Their Pathophysiological Role and Potential Translational Applications
Abstract
1. Introduction
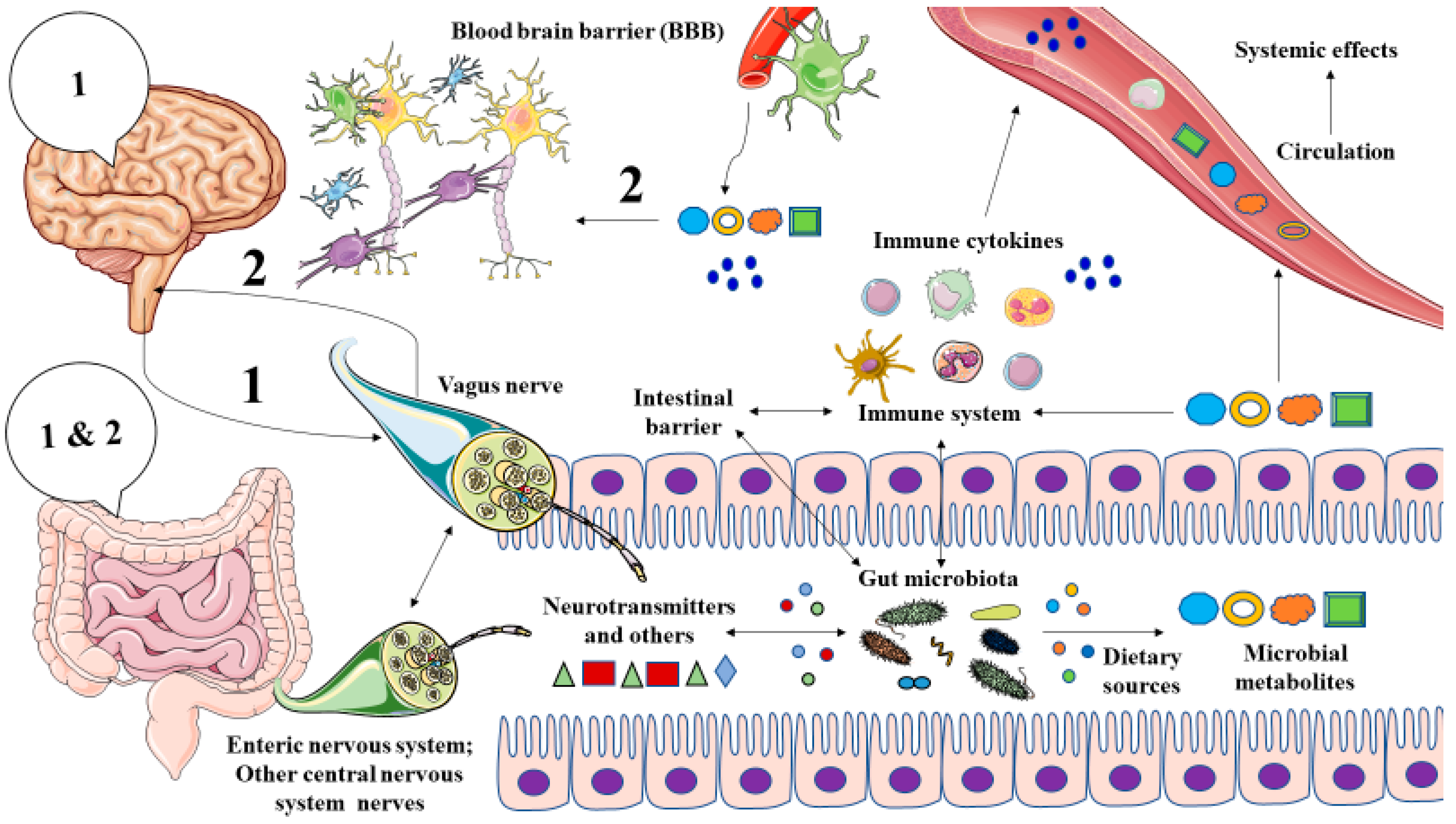
2. The Role of Gut Microbiota in Major Depression: The Microbiota-Gut-Brain Axis
3. Microbial Metabolites in MDD
3.1. Biological Basis of MDD
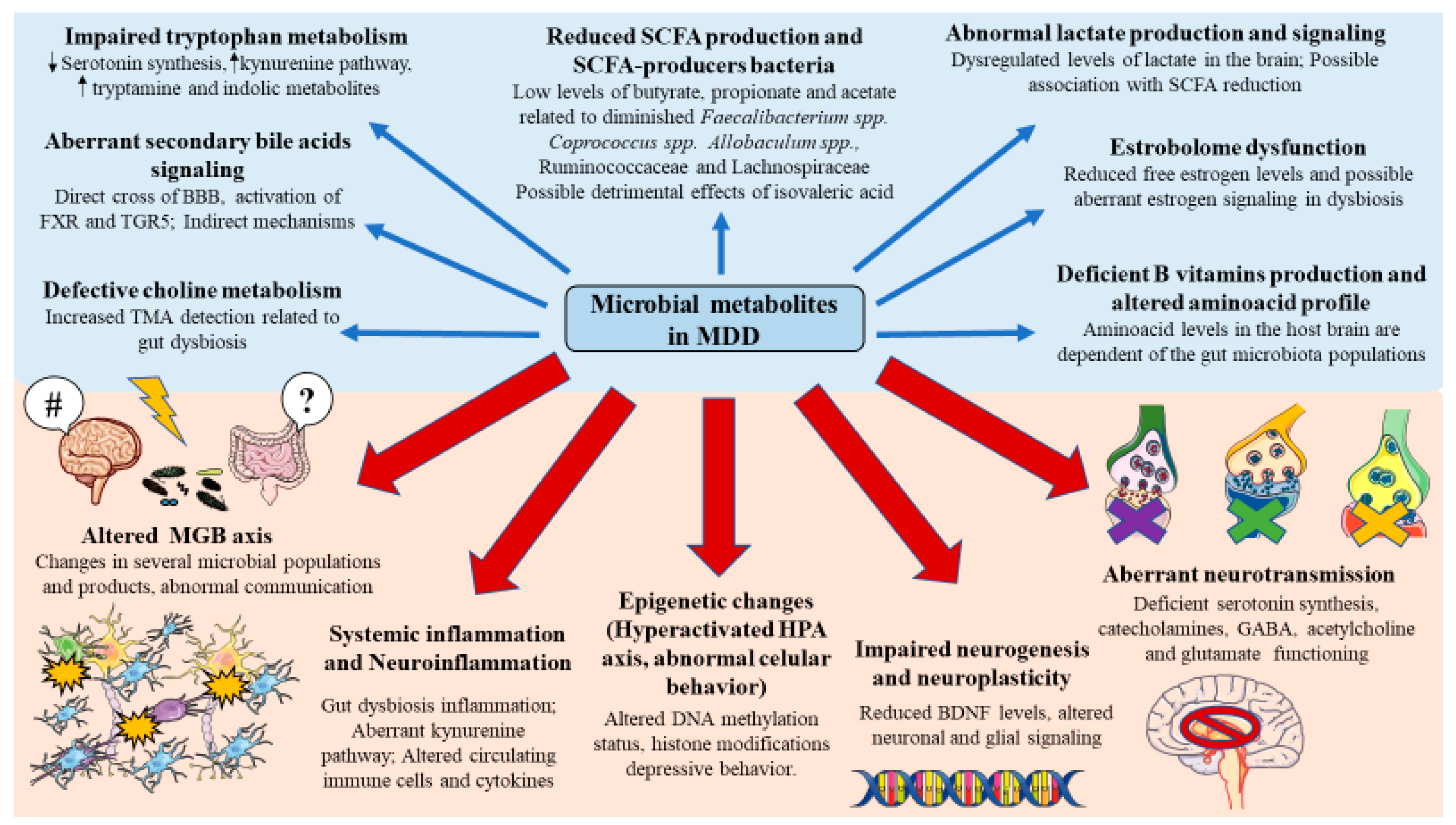
3.2. Microbial Metabolites Involved in the Pathophysiology of MDD
3.2.1. Short-Chain Fatty Acids
3.2.2. Lactate
3.2.3. Tryptophan Metabolites
3.2.4. Microbial Influence in Other Amino Acids
3.2.5. Secondary Bile Acids
3.2.6. Vitamins
3.2.7. Choline-Derived Metabolites
3.2.8. Estrobolome
4. Translational Applications for the Depressed Patient
4.1. The Importance of Boosting Individual’s Microbial Metabolome
4.2. Emulation of Microbial Metabolism
4.2.1. Adjuvant Therapy
Supplementation with Probiotics
Supplementation with Prebiotics
Supplementation with SCFAs and Derivatives
Supplementation with Nutraceuticals
4.2.2. Lifestyle Intervention
Diet
Exercise
Sleep
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Malley, M.A. From Endosymbiosis to Holobionts: Evaluating a Conceptual Legacy. J. Theor. Biol. 2017, 434, 34–41. [Google Scholar] [CrossRef]
- Baedke, J.; Fábregas-Tejeda, A.; Nieves Delgado, A. The Holobiont Concept before Margulis. J. Exp. Zool. Part B Mol. Dev. Evol. 2020, 334, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Hamady, M.; Fraser-Liggett, C.M.; Knight, R.; Gordon, J.I. The Human Microbiome Project. Nature 2007, 449, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef]
- Baquero, F.; Nombela, C. The Microbiome as a Human Organ. Clin. Microbiol. Infect. 2012, 18 (Suppl. 4), 2–4. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.M.; et al. Enterotypes of the Human Gut Microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef]
- Xu, C.; Zhu, H.; Qiu, P. Aging Progression of Human Gut Microbiota. BMC Microbiol. 2019, 19, 236. [Google Scholar] [CrossRef]
- Iebba, V.; Totino, V.; Gagliardi, A.; Santangelo, F.; Cacciotti, F.; Trancassini, M.; Mancini, C.; Cicerone, C.; Corazziari, E.; Pantanella, F.; et al. Eubiosis and Dysbiosis: The Two Sides of the Microbiota-PubMed. New Microbiol. 2016, 39, 1–12. [Google Scholar]
- Vamanu, E.; Rai, S.N.; Plaza-Díaz, J.; Battino, M. The Link between Obesity, Microbiota Dysbiosis, and Neurodegenerative Pathogenesis. Diseases 2021, 9, 45. [Google Scholar] [CrossRef] [PubMed]
- Lazar, V.; Ditu, L.M.; Pircalabioru, G.G.; Gheorghe, I.; Curutiu, C.; Holban, A.M.; Picu, A.; Petcu, L.; Chifiriuc, M.C. Aspects of Gut Microbiota and Immune System Interactions in Infectious Diseases, Immunopathology, and Cancer. Front. Immunol. 2018, 9, 1. [Google Scholar] [CrossRef]
- Giulio, P. The Intestinal Microbiota: Towards a Multifactorial Integrative Model. Eubiosis and Dysbiosis in Morbid Physical and Psychological Conditions. Arch. Clin. Gastroenterol. 2021, 7, 24–35. [Google Scholar] [CrossRef]
- GBD Results Tool|GHDx. Available online: http://ghdx.healthdata.org/gbd-results-tool?params=gbd-api-2019-permalink/d780dffbe8a381b25e1416884959e88b (accessed on 7 December 2021).
- Depression. Available online: https://www.who.int/news-room/fact-sheets/detail/depression (accessed on 7 December 2021).
- Albert, P.R. Why Is Depression More Prevalent in Women? J. Psychiatry Neurosci. 2015, 40, 219. [Google Scholar] [CrossRef] [PubMed]
- Ogrodniczuk, J.S.; Oliffe, J.L. Men and Depression. Can. Fam. Physician 2011, 57, 153. [Google Scholar]
- The Lancet Global Health. Mental Health Matters. Lancet Glob. Health 2020, 8, e1352. [Google Scholar] [CrossRef]
- Thakur, V.; Jain, A. COVID 2019-Suicides: A Global Psychological Pandemic. Brain Behav. Immun. 2020, 88, 952. [Google Scholar] [CrossRef]
- Santomauro, D.F.; Mantilla Herrera, A.M.; Shadid, J.; Zheng, P.; Ashbaugh, C.; Pigott, D.M.; Abbafati, C.; Adolph, C.; Amlag, J.O.; Aravkin, A.Y.; et al. Global Prevalence and Burden of Depressive and Anxiety Disorders in 204 Countries and Territories in 2020 Due to the COVID-19 Pandemic. Lancet 2021, 398, 1700–1712. [Google Scholar] [CrossRef]
- Sibley, S.; Sauers, L.; Daltry, R. Humanity and Resilience Project: The Development of a New Outreach Program for Counseling Centers at Colleges and Universities. J. Coll. Stud. Psychother. 2018, 33, 67–74. [Google Scholar] [CrossRef]
- Rao, G. Mens Sana in Corpore Sano. PDA J. Pharm. Sci. Technol. 2013, 67, 568. [Google Scholar] [CrossRef] [PubMed]
- Gianaros, P.J.; Wager, T.D. Brain-Body Pathways Linking Psychological Stress and Physical Health. Curr. Dir. Psychol. Sci. 2015, 24, 313–321. [Google Scholar] [CrossRef]
- Renoir, T.; Hasebe, K.; Gray, L. Mind and Body: How the Health of the Body Impacts on Neuropsychiatry. Front. Pharmacol. 2013, 4, 158. [Google Scholar] [CrossRef]
- Nemeroff, C.B. Psychoneuroimmunoendocrinology: The Biological Basis of Mind-Body Physiology and Pathophysiology. Depress. Anxiety 2013, 30, 285–287. [Google Scholar] [CrossRef]
- Daemen, M.J.A.P. The Heart and the Brain: An Intimate and Underestimated Relation. Neth. Heart J. 2013, 21, 53. [Google Scholar] [CrossRef]
- Pedersen, B.K. Physical Activity and Muscle–Brain Crosstalk. Nat. Rev. Endocrinol. 2019, 15, 383–392. [Google Scholar] [CrossRef]
- Ridaura, V.; Belkaid, Y. Gut Microbiota: The Link to Your Second Brain. Cell 2015, 161, 193–194. [Google Scholar] [CrossRef]
- Cryan, J.F.; O’riordan, K.J.; Cowan, C.S.M.; Sandhu, K.V.; Bastiaanssen, T.F.S.; Boehme, M.; Codagnone, M.G.; Cussotto, S.; Fulling, C.; Golubeva, A.V.; et al. The Microbiota-Gut-Brain Axis. Physiol. Rev. 2019, 99, 1877–2013. [Google Scholar] [CrossRef] [PubMed]
- Furness, J.B.; Callaghan, B.P.; Rivera, L.R.; Cho, H.J. The Enteric Nervous System and Gastrointestinal Innervation: Integrated Local and Central Control. Adv. Exp. Med. Biol. 2014, 817, 39–71. [Google Scholar] [CrossRef] [PubMed]
- Louwies, T.; Johnson, A.C.; Orock, A.; Yuan, T.; Greenwood-Van Meerveld, B. The Microbiota-Gut-Brain Axis: An Emerging Role for the Epigenome. Exp. Biol. Med. 2020, 245, 138–145. [Google Scholar] [CrossRef] [PubMed]
- García-Montero, C.; Fraile-Martínez, O.; Gómez-Lahoz, A.M.; Pekarek, L.; Castellanos, A.J.; Noguerales-Fraguas, F.; Coca, S.; Guijarro, L.G.; García-Honduvilla, N.; Asúnsolo, A.; et al. Nutritional Components in Western Diet Versus Mediterranean Diet at the Gut Microbiota-Immune System Interplay. Implications for Health and Disease. Nutrients 2021, 13, 699. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Gomez-Lahoz, A.M.; Orozco, A.; Lahera, G.; Sosa-Reina, M.D.; Diaz, D.; Albillos, A.; Quintero, J.; Molero, P.; Monserrat, J.; et al. Blunted Expansion of Regulatory T Lymphocytes Is Associated With Increased Bacterial Translocation in Patients With Major Depressive Disorder. Front. Psychiatry 2021, 11, 1530. [Google Scholar] [CrossRef]
- Álvarez-Mon, M.A.; Gómez-Lahoz, A.M.; Orozco, A.; Lahera, G.; Diaz, D.; Ortega, M.A.; Albillos, A.; Quintero, J.; Aubá, E.; Monserrat, J.; et al. Expansion of CD4 T Lymphocytes Expressing Interleukin 17 and Tumor Necrosis Factor in Patients with Major Depressive Disorder. J. Pers. Med. 2021, 11, 220. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Gómez, A.M.; Orozco, A.; Lahera, G.; Sosa, M.D.; Diaz, D.; Auba, E.; Albillos, A.; Monserrat, J.; Alvarez-Mon, M. Abnormal Distribution and Function of Circulating Monocytes and Enhanced Bacterial Translocation in Major Depressive Disorder. Front. Psychiatry 2019, 10, 812. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Eguilaz, M.; Ramon-Traperom, J.L.; Perez-Martinez, L.; Blanco, J.R. El Eje Microbiota-Intestino-Cerebro y Sus Grandes Proyecciones [The Microbiota-Gut-Brain Axis and Its Great Projections]. Rev. Neurol. 2019, 68, 111–117. [Google Scholar]
- Caspani, G.; Swann, J. Small Talk: Microbial Metabolites Involved in the Signaling from Microbiota to Brain. Curr. Opin. Pharmacol. 2019, 48, 99–106. [Google Scholar] [CrossRef]
- Parker, A.; Fonseca, S.; Carding, S.R. Gut Microbes and Metabolites as Modulators of Blood-Brain Barrier Integrity and Brain Health. Gut Microbes 2020, 11, 135–157. [Google Scholar] [CrossRef] [PubMed]
- Margolis, K.G.; Cryan, J.F.; Mayer, E.A. The Microbiota-Gut-Brain Axis: From Motility to Mood. Gastroenterology 2021, 160, 1486–1501. [Google Scholar] [CrossRef] [PubMed]
- Generoso, J.S.; Giridharan, V.V.; Lee, J.; Macedo, D.; Barichello, T. The Role of the Microbiota-Gut-Brain Axis in Neuropsychiatric Disorders. Rev. Bras. Psiquiatr. 2021, 43, 293–305. [Google Scholar] [CrossRef]
- Petra, A.I.; Panagiotidou, S.; Hatziagelaki, E.; Stewart, J.M.; Conti, P.; Theoharides, T.C. Gut-Microbiota-Brain Axis and Its Effect on Neuropsychiatric Disorders With Suspected Immune Dysregulation. Clin. Ther. 2015, 37, 984–995. [Google Scholar] [CrossRef]
- Levey, D.F.; Stein, M.B.; Wendt, F.R.; Pathak, G.A.; Zhou, H.; Aslan, M.; Quaden, R.; Harrington, K.M.; Nuñez, Y.Z.; Overstreet, C.; et al. Bi-Ancestral Depression GWAS in the Million Veteran Program and Meta-Analysis in >1.2 Million Individuals Highlight New Therapeutic Directions. Nat. Neurosci. 2021, 24, 954–963. [Google Scholar] [CrossRef]
- Flint, J.; Kendler, K.S. The Genetics of Major Depression. Neuron 2014, 81, 484–503. [Google Scholar] [CrossRef]
- Syed, S.A.; Nemeroff, C.B. Early Life Stress, Mood, and Anxiety Disorders. Chronic Stress 2017, 1, 2470547017694461. [Google Scholar] [CrossRef] [PubMed]
- LeMoult, J.; Humphreys, K.L.; Tracy, A.; Hoffmeister, J.A.; Ip, E.; Gotlib, I.H. Meta-Analysis: Exposure to Early Life Stress and Risk for Depression in Childhood and Adolescence. J. Am. Acad. Child Adolesc. Psychiatry 2020, 59, 842–855. [Google Scholar] [CrossRef]
- Li, M.; Fu, X.; Xie, W.; Guo, W.; Li, B.; Cui, R.; Yang, W. Effect of Early Life Stress on the Epigenetic Profiles in Depression. Front. Cell Dev. Biol. 2020, 8, 867. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhao, Y.; Wang, Y.; Liu, L.; Zhang, X.; Li, B.; Cui, R. The Effects of Psychological Stress on Depression. Curr. Neuropharmacol. 2015, 13, 494. [Google Scholar] [CrossRef]
- Noble, R.E. Depression in Women. Metab. Clin. Exp. 2005, 54, 49–52. [Google Scholar] [CrossRef]
- Alegría, M.; NeMoyer, A.; Falgàs Bagué, I.; Wang, Y.; Alvarez, K. Social Determinants of Mental Health: Where We Are and Where We Need to Go. Curr. Psychiatry Rep. 2018, 20, 95. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.F.; Peng, W.; Sweeney, J.A.; Jia, Z.Y.; Gong, Q.Y. Brain Structure Alterations in Depression: Psychoradiological Evidence. CNS Neurosci. Ther. 2018, 24, 994. [Google Scholar] [CrossRef]
- Lee, B.H.; Kim, Y.K. The Roles of BDNF in the Pathophysiology of Major Depression and in Antidepressant Treatment. Psychiatry Investig. 2010, 7, 231. [Google Scholar] [CrossRef]
- Rajkowska, G.; Miguel-Hidalgo, J.J. Gliogenesis and Glial Pathology in Depression. CNS Neurol. Disord. Drug Targets 2007, 6, 219. [Google Scholar] [CrossRef] [PubMed]
- Rial, D.; Lemos, C.; Pinheiro, H.; Duarte, J.M.; Gonçalves, F.Q.; Real, J.I.; Prediger, R.D.; Gonçalves, N.; Gomes, C.A.; Canas, P.M.; et al. Depression as a Glial-Based Synaptic Dysfunction. Front. Cell. Neurosci. 2015, 9, 521. [Google Scholar] [CrossRef]
- Owens, M.J. Selectivity of Antidepressants: From the Monoamine Hypothesis of Depression to the SSRI Revolution and beyond-PubMed. J. Clin. Psychiatry 2004, 65, 5–10. [Google Scholar]
- Hirschfeld, R.M.A. History and Evolution of the Monoamine Hypothesis of Depression. J. Clin. Psychiatry 2000, 61, 4–6. [Google Scholar] [PubMed]
- Heninger, G.R.; Delgado, P.L.; Charney, D.S. The Revised Monoamine Theory of Depression: A Modulatory Role for Monoamines, Based on New Findings from Monoamine Depletion Experiments in Humans. Pharmacopsychiatry 1996, 29, 2–11. [Google Scholar] [CrossRef]
- Neumann, I.D.; Landgraf, R. Balance of Brain Oxytocin and Vasopressin: Implications for Anxiety, Depression, and Social Behaviors. Trends Neurosci. 2012, 35, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Keller, J.; Gomez, R.; Williams, G.; Lembke, A.; Lazzeroni, L.; Murphy, G.M.; Schatzberg, A.F. HPA Axis in Major Depression: Cortisol, Clinical Symptomatology, and Genetic Variation Predict Cognition. Mol. Psychiatry 2017, 22, 527. [Google Scholar] [CrossRef] [PubMed]
- Van Den Eede, F.; Claes, S.J. Mechanisms of Depression: Role of the HPA Axis. Drug Discov. Today Dis. Mech. 2004, 1, 413–418. [Google Scholar] [CrossRef]
- Quera Salva, M.A.; Hartley, S.; Barbot, F.; Alvarez, J.C.; Lofaso, F.; Guilleminault, C. Circadian Rhythms, Melatonin and Depression. Curr. Pharm. Des. 2011, 17, 1459–1470. [Google Scholar] [CrossRef]
- Maria Michel, T.; Pulschen, D.; Thome, J. The Role of Oxidative Stress in Depressive Disorders. Curr. Pharm. Des. 2012, 18, 5890–5899. [Google Scholar] [CrossRef]
- Bajpai, A.; Verma, A.K.; Srivastava, M.; Srivastava, R. Oxidative Stress and Major Depression. J. Clin. Diagn. Res. 2014, 8, CC04. [Google Scholar] [CrossRef]
- Bakunina, N.; Pariante, C.M.; Zunszain, P.A. Immune Mechanisms Linked to Depression via Oxidative Stress and Neuroprogression. Immunology 2015, 144, 365. [Google Scholar] [CrossRef]
- Brites, D.; Fernandes, A. Neuroinflammation and Depression: Microglia Activation, Extracellular Microvesicles and MicroRNA Dysregulation. Front. Cell. Neurosci. 2015, 9, 476. [Google Scholar] [CrossRef]
- Troubat, R.; Barone, P.; Leman, S.; Desmidt, T.; Cressant, A.; Atanasova, B.; Brizard, B.; El Hage, W.; Surget, A.; Belzung, C.; et al. Neuroinflammation and Depression: A Review. Eur. J. Neurosci. 2021, 53, 151–171. [Google Scholar] [CrossRef]
- Du, Y.; Gao, X.R.; Peng, L.; Ge, J.F. Crosstalk between the Microbiota-Gut-Brain Axis and Depression. Heliyon 2020, 6, e04097. [Google Scholar] [CrossRef]
- Sittipo, P.; Shim, J.W.; Lee, Y.K. Microbial Metabolites Determine Host Health and the Status of Some Diseases. Int. J. Mol. Sci. 2019, 20, 5296. [Google Scholar] [CrossRef] [PubMed]
- Martinez, K.B.; Leone, V.; Chang, E.B. Microbial Metabolites in Health and Disease: Navigating the Unknown in Search of Function. J. Biol. Chem. 2017, 292, 8553. [Google Scholar] [CrossRef] [PubMed]
- Morrison, D.J.; Preston, T. Formation of Short Chain Fatty Acids by the Gut Microbiota and Their Impact on Human Metabolism. Gut Microbes 2016, 7, 189. [Google Scholar] [CrossRef]
- Louis, P.; Young, P.; Holtrop, G.; Flint, H.J. Diversity of Human Colonic Butyrate-Producing Bacteria Revealed by Analysis of the Butyryl-CoA:Acetate CoA-Transferase Gene. Environ. Microbiol. 2010, 12, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, E.; Grootaert, C.; Verstraete, W.; Van de Wiele, T. Propionate as a Health-Promoting Microbial Metabolite in the Human Gut. Nutr. Rev. 2011, 69, 245–258. [Google Scholar] [CrossRef]
- Deleu, S.; Machiels, K.; Raes, J.; Verbeke, K.; Vermeire, S. Short Chain Fatty Acids and Its Producing Organisms: An Overlooked Therapy for IBD? EBioMedicine 2021, 66, 103293. [Google Scholar] [CrossRef] [PubMed]
- Layden, B.T.; Angueira, A.R.; Brodsky, M.; Durai, V.; Lowe, W.L. Short Chain Fatty Acids and Their Receptors: New Metabolic Targets. Transl. Res. J. Lab. Clin. Med. 2013, 161, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Wang, Q.; Wu, Q.; Mao-Draayer, Y.; Kim, C.H. Bidirectional Regulatory Potentials of Short-Chain Fatty Acids and Their G-Protein-Coupled Receptors in Autoimmune Neuroinflammation. Sci. Rep. 2019, 9, 8837. [Google Scholar] [CrossRef]
- Blaak, E.E.; Canfora, E.E.; Theis, S.; Frost, G.; Groen, A.K.; Mithieux, G.; Nauta, A.; Scott, K.; Stahl, B.; van Harsselaar, J.; et al. Short Chain Fatty Acids in Human Gut and Metabolic Health. Benef. Microbes 2020, 11, 411–455. [Google Scholar] [CrossRef]
- Deng, Z.; Luo, X.M.; Liu, J.; Wang, H. Quorum Sensing, Biofilm, and Intestinal Mucosal Barrier: Involvement the Role of Probiotic. Front. Cell. Infect. Microbiol. 2020, 10, 504. [Google Scholar] [CrossRef]
- van der Hee, B.; Wells, J.M. Microbial Regulation of Host Physiology by Short-Chain Fatty Acids. Trends Microbiol. 2021, 29, 700–712. [Google Scholar] [CrossRef]
- Silva, Y.P.; Bernardi, A.; Frozza, R.L. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front. Endocrinol. 2020, 11, 25. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Ling, Z.; Zhang, Y.; Mao, H.; Ma, Z.; Yin, Y.; Wang, W.; Tang, W.; Tan, Z.; Shi, J.; et al. Altered Fecal Microbiota Composition in Patients with Major Depressive Disorder. Brain Behav. Immun. 2015, 48, 186–194. [Google Scholar] [CrossRef]
- Valles-Colomer, M.; Falony, G.; Darzi, Y.; Tigchelaar, E.F.; Wang, J.; Tito, R.Y.; Schiweck, C.; Kurilshikov, A.; Joossens, M.; Wijmenga, C.; et al. The Neuroactive Potential of the Human Gut Microbiota in Quality of Life and Depression. Nat. Microbiol. 2019, 4, 623–632. [Google Scholar] [CrossRef]
- Wu, M.; Tian, T.; Mao, Q.; Zou, T.; Zhou, C.J.; Xie, J.; Chen, J.J. Associations between Disordered Gut Microbiota and Changes of Neurotransmitters and Short-Chain Fatty Acids in Depressed Mice. Transl. Psychiatry 2020, 10, 350. [Google Scholar] [CrossRef] [PubMed]
- Tsuruta, T.; Saito, S.; Osaki, Y.; Hamada, A.; Aoki-Yoshida, A.; Sonoyama, K. Organoids as an Ex Vivo Model for Studying the Serotonin System in the Murine Small Intestine and Colon Epithelium. Biochem. Biophys. Res. Commun. 2016, 474, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Engevik, M.A.; Luck, B.; Visuthranukul, C.; Ihekweazu, F.D.; Engevik, A.C.; Shi, Z.; Danhof, H.A.; Chang-Graham, A.L.; Hall, A.; Endres, B.T.; et al. Human-Derived Bifidobacterium Dentium Modulates the Mammalian Serotonergic System and Gut-Brain Axis. Cell. Mol. Gastroenterol. Hepatol. 2021, 11, 221–248. [Google Scholar] [CrossRef] [PubMed]
- Yano, J.M.; Yu, K.; Donaldson, G.P.; Shastri, G.G.; Ann, P.; Ma, L.; Nagler, C.R.; Ismagilov, R.F.; Mazmanian, S.K.; Hsiao, E.Y. Indigenous Bacteria from the Gut Microbiota Regulate Host Serotonin Biosynthesis. Cell 2015, 161, 264–276. [Google Scholar] [CrossRef] [PubMed]
- Reigstad, C.S.; Salmonson, C.E.; Rainey, J.F.; Szurszewski, J.H.; Linden, D.R.; Sonnenburg, J.L.; Farrugia, G.; Kashyap, P.C. Gut Microbes Promote Colonic Serotonin Production through an Effect of Short-Chain Fatty Acids on Enterochromaffin Cells. FASEB J. 2015, 29, 1395–1403. [Google Scholar] [CrossRef]
- Skonieczna-żydecka, K.; Grochans, E.; Maciejewska, D.; Szkup, M.; Schneider-Matyka, D.; Jurczak, A.; Łoniewski, I.; Kaczmarczyk, M.; Marlicz, W.; Czerwińska-Rogowska, M.; et al. Faecal Short Chain Fatty Acids Profile Is Changed in Polish Depressive Women. Nutrients 2018, 10, 1939. [Google Scholar] [CrossRef]
- Müller, B.; Rasmusson, A.J.; Just, D.; Jayarathna, S.; Moazzami, A.; Novicic, Z.K.; Cunningham, J.L. Fecal Short-Chain Fatty Acid Ratios as Related to Gastrointestinal and Depressive Symptoms in Young Adults. Psychosom. Med. 2021, 83, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Byrne, C.S.; Chambers, E.S.; Alhabeeb, H.; Chhina, N.; Morrison, D.J.; Preston, T.; Tedford, C.; Fitzpatrick, J.; Irani, C.; Busza, A.; et al. Increased Colonic Propionate Reduces Anticipatory Reward Responses in the Human Striatum to High-Energy Foods. Am. J. Clin. Nutr. 2016, 104, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Gorwood, P. Neurobiological Mechanisms of Anhedonia. Dialogues Clin. Neurosci. 2008, 10, 291. [Google Scholar] [CrossRef] [PubMed]
- Van de Wouw, M.; Boehme, M.; Lyte, J.M.; Wiley, N.; Strain, C.; O’Sullivan, O.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Short-chain Fatty Acids: Microbial Metabolites That Alleviate Stress-induced Brain-Gut Axis Alterations. J. Physiol. 2018, 596, 4923. [Google Scholar] [CrossRef]
- Li, H.; Xiang, Y.; Zhu, Z.; Wang, W.; Jiang, Z.; Zhao, M.; Cheng, S.; Pan, F.; Liu, D.; Ho, R.C.M.; et al. Rifaximin-Mediated Gut Microbiota Regulation Modulates the Function of Microglia and Protects against CUMS-Induced Depression-like Behaviors in Adolescent Rat. J. Neuroinflamm. 2021, 18, 254. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.; Tsai, S.J. Epigenetics and Depression: An Update. Psychiatry Investig. 2019, 16, 654. [Google Scholar] [CrossRef] [PubMed]
- Ortega, M.A.; Alvarez-Mon, M.A.; García-Montero, C.; Fraile-Martinez, O.; Lahera, G.; Monserrat, J.; Muñoz-Merida, L.; Mora, F.; Rodríguez-Jiménez, R.; Fernandez-Rojo, S.; et al. MicroRNAs as Critical Biomarkers of Major Depressive Disorder: A Comprehensive Perspective. Biomedicines 2021, 9, 1659. [Google Scholar] [CrossRef]
- Sun, H.; Kennedy, P.J.; Nestler, E.J. Epigenetics of the Depressed Brain: Role of Histone Acetylation and Methylation. Neuropsychopharmacology 2012, 38, 124–137. [Google Scholar] [CrossRef]
- Kaur, H.; Singh, Y.; Singh, S.; Singh, R.B. Gut Microbiome-Mediated Epigenetic Regulation of Brain Disorder and Application of Machine Learning for Multi-Omics Data Analysis. Genome 2021, 64, 355–371. [Google Scholar] [CrossRef] [PubMed]
- Zalar, B.; Haslberger, A.; Peterlin, B. The Role of Microbiota in Depression-a Brief Review. Psychiatr. Danub. 2018, 30, 136–141. [Google Scholar] [CrossRef]
- Szczesniak, O.; Hestad, K.A.; Hanssen, J.F.; Rudi, K. Isovaleric Acid in Stool Correlates with Human Depression. Nutr. Neurosci. 2016, 19, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Adeva-Andany, M.; López-Ojén, M.; Funcasta-Calderón, R.; Ameneiros-Rodríguez, E.; Donapetry-García, C.; Vila-Altesor, M.; Rodríguez-Seijas, J. Comprehensive Review on Lactate Metabolism in Human Health. Mitochondrion 2014, 17, 76–100. [Google Scholar] [CrossRef]
- Ríos-Covián, D.; Ruas-Madiedo, P.; Margolles, A.; Gueimonde, M.; De los Reyes-Gavilán, C.G.; Salazar, N. Intestinal Short Chain Fatty Acids and Their Link with Diet and Human Health. Front. Microbiol. 2016, 7, 185. [Google Scholar] [CrossRef]
- Magistretti, P.J.; Allaman, I. Lactate in the Brain: From Metabolic End-Product to Signalling Molecule. Nat. Rev. Neurosci. 2018, 19, 235–249. [Google Scholar] [CrossRef]
- Riske, L.; Thomas, R.K.; Baker, G.B.; Dursun, S.M. Lactate in the Brain: An Update on Its Relevance to Brain Energy, Neurons, Glia and Panic Disorder. Ther. Adv. Psychopharmacol. 2017, 7, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Bradley, K.A.L.; Mao, X.; Case, J.A.C.; Kang, G.; Shungu, D.C.; Gabbay, V. Increased Ventricular Cerebrospinal Fluid Lactate in Depressed Adolescents. Eur. Psychiatry J. Assoc. Eur. Psychiatr. 2016, 32, 1–8. [Google Scholar] [CrossRef]
- Machado-Vieira, R.; Zanetti, M.V.; Otaduy, M.C.; De Sousa, R.T.; Soeiro-De-Souza, M.G.; Costa, A.C.; Carvalho, A.F.; Leite, C.C.; Busatto, G.F.; Zarate, C.A.; et al. Increased Brain Lactate during Depressive Episodes and Reversal Effects by Lithium Monotherapy in Drug-Naive Bipolar Disorder: A 3T 1H-MRS Study. J. Clin. Psychopharmacol. 2017, 37, 40. [Google Scholar] [CrossRef]
- Chen, J.-J.; Zhou, C.-J.; Zheng, P.; Cheng, K.; Wang, H.-Y.; Li, J.; Zeng, L.; Xie, P. Differential Urinary Metabolites Related with the Severity of Major Depressive Disorder. Behav. Brain Res. 2017, 332, 280–287. [Google Scholar] [CrossRef]
- Carrard, A.; Cassé, F.; Carron, C.; Burlet-Godinot, S.; Toni, N.; Magistretti, P.J.; Martin, J.L. Role of Adult Hippocampal Neurogenesis in the Antidepressant Actions of Lactate. Mol. Psychiatry 2021, 76. [Google Scholar] [CrossRef]
- Karnib, N.; El-Ghandour, R.; El Hayek, L.; Nasrallah, P.; Khalifeh, M.; Barmo, N.; Jabre, V.; Ibrahim, P.; Bilen, M.; Stephan, J.S.; et al. Lactate Is an Antidepressant That Mediates Resilience to Stress by Modulating the Hippocampal Levels and Activity of Histone Deacetylases. Neuropsychopharmacology 2019, 44, 1152–1162. [Google Scholar] [CrossRef]
- Carrard, A.; Elsayed, M.; Margineanu, M.; Boury-Jamot, B.; Fragnière, L.; Meylan, E.M.; Petit, J.M.; Fiumelli, H.; Magistretti, P.J.; Martin, J.L. Peripheral Administration of Lactate Produces Antidepressant-like Effects. Mol. Psychiatry 2018, 23, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Scaglia, F. The Role of Mitochondrial Dysfunction in Psychiatric Disease. Dev. Disabil. Res. Rev. 2010, 16, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Dalton, A.; Mermier, C.; Zuhl, M. Exercise Influence on the Microbiome–Gut–Brain Axis. Gut Microbes 2019, 10, 555. [Google Scholar] [CrossRef]
- Modoux, M.; Rolhion, N.; Mani, S.; Sokol, H. Tryptophan Metabolism as a Pharmacological Target. Trends Pharmacol. Sci. 2021, 42, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Agus, A.; Planchais, J.; Sokol, H. Gut Microbiota Regulation of Tryptophan Metabolism in Health and Disease. Cell Host Microbe 2018, 23, 716–724. [Google Scholar] [CrossRef]
- Alkhalaf, L.M.; Ryan, K.S. Biosynthetic Manipulation of Tryptophan in Bacteria: Pathways and Mechanisms. Chem. Biol. 2015, 22, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Mu, C.L.; Farzi, A.; Zhu, W.Y. Tryptophan Metabolism: A Link Between the Gut Microbiota and Brain. Adv. Nutr. 2020, 11, 709–723. [Google Scholar] [CrossRef]
- Kaur, H.; Bose, C.; Mande, S.S. Tryptophan Metabolism by Gut Microbiome and Gut-Brain-Axis: An in Silico Analysis. Front. Neurosci. 2019, 13, 1365. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, T.A.; Nguyen, J.C.D.; Polglaze, K.E.; Bertrand, P.P. Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis. Nutrients 2016, 8, 56. [Google Scholar] [CrossRef]
- Tian, P.; Wang, G.; Zhao, J.; Zhang, H.; Chen, W. Bifidobacterium with the Role of 5-Hydroxytryptophan Synthesis Regulation Alleviates the Symptom of Depression and Related Microbiota Dysbiosis. J. Nutr. Biochem. 2019, 66, 43–51. [Google Scholar] [CrossRef]
- Soh, N.L.; Walter, G. Tryptophan and Depression: Can Diet Alone Be the Answer? Acta Neuropsychiatr. 2011, 23, 3–11. [Google Scholar] [CrossRef]
- Lukić, I.; Getselter, D.; Koren, O.; Elliott, E. Role of Tryptophan in Microbiota-Induced Depressive-like Behavior: Evidence from Tryptophan Depletion Study. Front. Behav. Neurosci. 2019, 13, 123. [Google Scholar] [CrossRef] [PubMed]
- Miura, H.; Ozaki, N.; Sawada, M.; Isobe, K.; Ohta, T.; Nagatsu, T. A Link between Stress and Depression: Shifts in the Balance between the Kynurenine and Serotonin Pathways of Tryptophan Metabolism and the Etiology and Pathophysiology of Depression. Stress 2008, 11, 198–209. [Google Scholar] [CrossRef]
- O’Connor, J.C.; Lawson, M.A.; André, C.; Moreau, M.; Lestage, J.; Castanon, N.; Kelley, K.W.; Dantzer, R. Lipopolysaccharide-Induced Depressive-like Behavior Is Mediated by Indoleamine 2,3-Dioxygenase Activation in Mice. Mol. Psychiatry 2009, 14, 511–522. [Google Scholar] [CrossRef]
- Marx, W.; McGuinness, A.J.; Rocks, T.; Ruusunen, A.; Cleminson, J.; Walker, A.J.; Gomes-da-Costa, S.; Lane, M.; Sanches, M.; Diaz, A.P.; et al. The Kynurenine Pathway in Major Depressive Disorder, Bipolar Disorder, and Schizophrenia: A Meta-Analysis of 101 Studies. Mol. Psychiatry 2020, 26, 4158–4178. [Google Scholar] [CrossRef] [PubMed]
- Ogyu, K.; Kubo, K.; Noda, Y.; Iwata, Y.; Tsugawa, S.; Omura, Y.; Wada, M.; Tarumi, R.; Plitman, E.; Moriguchi, S.; et al. Kynurenine Pathway in Depression: A Systematic Review and Meta-Analysis. Neurosci. Biobehav. Rev. 2018, 90, 16–25. [Google Scholar] [CrossRef]
- Dantzer, R. Role of the Kynurenine Metabolism Pathway in Inflammation-Induced Depression–Preclinical Approaches. Curr. Top. Behav. Neurosci. 2017, 31, 117. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.J.; Huang, X.F.; Newell, K.A. The Kynurenine Pathway in Major Depression: What We Know and Where to Next. Neurosci. Biobehav. Rev. 2021, 127, 917–927. [Google Scholar] [CrossRef]
- Neavin, D.R.; Liu, D.; Ray, B.; Weinshilboum, R.M. The Role of the Aryl Hydrocarbon Receptor (AHR) in Immune and Inflammatory Diseases. Int. J. Mol. Sci. 2018, 19, 3851. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Ray, B.; Neavin, D.R.; Zhang, J.; Athreya, A.P.; Biernacka, J.M.; Bobo, W.V.; Hall-Flavin, D.K.; Skime, M.K.; Zhu, H.; et al. Beta-Defensin 1, Aryl Hydrocarbon Receptor and Plasma Kynurenine in Major Depressive Disorder: Metabolomics-Informed Genomics. Transl. Psychiatry 2018, 8, 10. [Google Scholar] [CrossRef] [PubMed]
- Mousseau, D.D. Tryptamine: A Metabolite of Tryptophan Implicated in Various Neuropsychiatric Disorders. Metab. Brain Dis. 1993, 8, 1–44. [Google Scholar] [CrossRef]
- Williams, B.B.; Van Benschoten, A.H.; Cimermancic, P.; Donia, M.S.; Zimmermann, M.; Taketani, M.; Ishihara, A.; Kashyap, P.C.; Fraser, J.S.; Fischbach, M.A. Discovery and Characterization of Gut Microbiota Decarboxylases That Can Produce the Neurotransmitter Tryptamine. Cell Host Microbe 2014, 16, 495. [Google Scholar] [CrossRef]
- Gao, J.; Xu, K.; Liu, H.; Liu, G.; Bai, M.; Peng, C.; Li, T.; Yin, Y. Impact of the Gut Microbiota on Intestinal Immunity Mediated by Tryptophan Metabolism. Front. Cell. Infect. Microbiol. 2018, 8, 13. [Google Scholar] [CrossRef]
- Jaglin, M.; Rhimi, M.; Philippe, C.; Pons, N.; Bruneau, A.; Goustard, B.; Daugé, V.; Maguin, E.; Naudon, L.; Rabot, S. Indole, a Signaling Molecule Produced by the Gut Microbiota, Negatively Impacts Emotional Behaviors in Rats. Front. Neurosci. 2018, 12, 216. [Google Scholar] [CrossRef] [PubMed]
- Pappolla, M.A.; Perry, G.; Fang, X.; Zagorski, M.; Sambamurti, K.; Poeggeler, B. Indoles as Essential Mediators in the Gut-Brain Axis. Their Role in Alzheimer’s Disease. Neurobiol. Dis. 2021, 156, 105403. [Google Scholar] [CrossRef]
- Beloborodova, N.V.; Chernevskaya, E.A.; Getsina, M.L. Indolic Structure Metabolites as Potential Biomarkers of Non-Infectious Diseases. Curr. Pharm. Des. 2021, 27, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Roiser, J.P.; McLean, A.; Ogilvie, A.D.; Blackwell, A.D.; Bamber, D.J.; Goodyer, I.; Jones, P.B.; Sahakian, B.J. The Subjective and Cognitive Effects of Acute Phenylalanine and Tyrosine Depletion in Patients Recovered from Depression. Neuropsychopharmacology 2005, 30, 775. [Google Scholar] [CrossRef]
- Zheng, S.; Yu, M.; Lu, X.; Huo, T.; Ge, L.; Yang, J.; Wu, C.; Li, F. Urinary Metabonomic Study on Biochemical Changes in Chronic Unpredictable Mild Stress Model of Depression. Clin. Chim. Acta Int. J. Clin. Chem. 2010, 411, 204–209. [Google Scholar] [CrossRef]
- Inoshita, M.; Umehara, H.; Watanabe, S.Y.; Nakataki, M.; Kinoshita, M.; Tomioka, Y.; Tajima, A.; Numata, S.; Ohmori, T. Elevated Peripheral Blood Glutamate Levels in Major Depressive Disorder. Neuropsychiatr. Dis. Treat. 2018, 14, 945. [Google Scholar] [CrossRef]
- Averina, O.V.; Zorkina, Y.A.; Yunes, R.A.; Kovtun, A.S.; Ushakova, V.M.; Morozova, A.Y.; Kostyuk, G.P.; Danilenko, V.N.; Chekhonin, V.P. Bacterial Metabolites of Human Gut Microbiota Correlating with Depression. Int. J. Mol. Sci. 2020, 21, 9234. [Google Scholar] [CrossRef] [PubMed]
- Mazzoli, R.; Pessione, E. The Neuro-Endocrinological Role of Microbial Glutamate and GABA Signaling. Front. Microbiol. 2016, 7, 1934. [Google Scholar] [CrossRef]
- Altaib, H.; Nakamura, K.; Abe, M.; Badr, Y.; Yanase, E.; Nomura, I.; Suzuki, T. Differences in the Concentration of the Fecal Neurotransmitters GABA and Glutamate Are Associated with Microbial Composition among Healthy Human Subjects. Microorganisms 2021, 9, 378. [Google Scholar] [CrossRef]
- Luck, B.; Horvath, T.D.; Engevik, K.A.; Ruan, W.; Haidacher, S.J.; Hoch, K.M.; Oezguen, N.; Spinler, J.K.; Haag, A.M.; Versalovic, J.; et al. Neurotransmitter Profiles Are Altered in the Gut and Brain of Mice Mono-Associated with Bifidobacterium Dentium. Biomolecules 2021, 11, 1091. [Google Scholar] [CrossRef] [PubMed]
- Duranti, S.; Ruiz, L.; Lugli, G.A.; Tames, H.; Milani, C.; Mancabelli, L.; Mancino, W.; Longhi, G.; Carnevali, L.; Sgoifo, A.; et al. Bifidobacterium Adolescentis as a Key Member of the Human Gut Microbiota in the Production of GABA. Sci. Rep. 2020, 10, 14112. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus Strain Regulates Emotional Behavior and Central GABA Receptor Expression in a Mouse via the Vagus Nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef] [PubMed]
- Strandwitz, P.; Kim, K.H.; Terekhova, D.; Liu, J.K.; Sharma, A.; Levering, J.; McDonald, D.; Dietrich, D.; Ramadhar, T.R.; Lekbua, A.; et al. GABA-Modulating Bacteria of the Human Gut Microbiota. Nat. Microbiol. 2019, 4, 396–403. [Google Scholar] [CrossRef]
- Otaru, N.; Ye, K.; Mujezinovic, D.; Berchtold, L.; Constancias, F.; Cornejo, F.A.; Krzystek, A.; de Wouters, T.; Braegger, C.; Lacroix, C.; et al. GABA Production by Human Intestinal Bacteroides spp.: Prevalence, Regulation, and Role in Acid Stress Tolerance. Front. Microbiol. 2021, 12, 860. [Google Scholar] [CrossRef]
- Martins-de-Souza, D. Proteomics, Metabolomics, and Protein Interactomics in the Characterization of the Molecular Features of Major Depressive Disorder. Dialogues Clin. Neurosci. 2014, 16, 63–73. [Google Scholar] [CrossRef]
- Li, J.; Jia, X.; Wang, C.; Wu, C.; Qin, X. Altered Gut Metabolome Contributes to Depression-like Behaviors in Rats Exposed to Chronic Unpredictable Mild Stress. Transl. Psychiatry 2019, 9, 40. [Google Scholar] [CrossRef]
- Kawase, T.; Nagasawa, M.; Ikeda, H.; Yasuo, S.; Koga, Y.; Furuse, M. Gut Microbiota of Mice Putatively Modifies Amino Acid Metabolism in the Host Brain. Br. J. Nutr. 2017, 117, 775–783. [Google Scholar] [CrossRef]
- Steffens, D.C.; Jiang, W.; Krishnan, K.R.R.; Karoly, E.D.; Mitchell, M.W.; O’Connor, C.M.; Kaddurah-Daouk, R. Metabolomic Differences in Heart Failure Patients with and without Major Depression. J. Geriatr. Psychiatry Neurol. 2010, 23, 138–146. [Google Scholar] [CrossRef]
- Ridlon, J.M.; Kang, D.J.; Hylemon, P.B.; Bajaj, J.S. Bile Acids and the Gut Microbiome. Curr. Opin. Gastroenterol. 2014, 30, 332–338. [Google Scholar] [CrossRef]
- Chiang, J.Y.L. Bile Acid Metabolism and Signaling. Compr. Physiol. 2013, 3, 1191. [Google Scholar] [CrossRef] [PubMed]
- Ridlon, J.M.; Harris, S.C.; Bhowmik, S.; Kang, D.J.; Hylemon, P.B. Consequences of Bile Salt Biotransformations by Intestinal Bacteria. Gut Microbes 2016, 7, 22. [Google Scholar] [CrossRef]
- Zeng, H.; Umar, S.; Rust, B.; Lazarova, D.; Bordonaro, M. Secondary Bile Acids and Short Chain Fatty Acids in the Colon: A Focus on Colonic Microbiome, Cell Proliferation, Inflammation, and Cancer. Int. J. Mol. Sci. 2019, 20, 1214. [Google Scholar] [CrossRef] [PubMed]
- Staley, C.; Weingarden, A.R.; Khoruts, A.; Sadowsky, M.J. Interaction of Gut Microbiota with Bile Acid Metabolism and Its Influence on Disease States. Appl. Microbiol. Biotechnol. 2017, 101, 47. [Google Scholar] [CrossRef] [PubMed]
- Chen, I.; Cassaro, S. Physiology, Bile Acids; StatPearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Martinot, E.; Sèdes, L.; Baptissart, M.; Lobaccaro, J.M.; Caira, F.; Beaudoin, C.; Volle, D.H. Bile Acids and Their Receptors. Mol. Asp. Med. 2017, 56, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Monteiro-Cardoso, V.F.; Corlianò, M.; Singaraja, R.R. Bile Acids: A Communication Channel in the Gut-Brain Axis. Neuromolecular Med. 2021, 23, 99–117. [Google Scholar] [CrossRef]
- Mulak, A. Bile Acids as Key Modulators of the Brain-Gut-Microbiota Axis in Alzheimer’s Disease. J. Alzheimer’s Dis. 2021, 84, 461–477. [Google Scholar] [CrossRef]
- Jia, W.; Rajani, C.; Kaddurah-Daouk, R.; Li, H. Expert Insights: The Potential Role of the Gut Microbiome-Bile Acid-Brain Axis in the Development and Progression of Alzheimer’s Disease and Hepatic Encephalopathy. Med. Res. Rev. 2020, 40, 1496–1507. [Google Scholar] [CrossRef]
- Li, P.; Killinger, B.A.; Ensink, E.; Beddows, I.; Yilmaz, A.; Lubben, N.; Lamp, J.; Schilthuis, M.; Vega, I.E.; Woltjer, R.; et al. Gut Microbiota Dysbiosis Is Associated with Elevated Bile Acids in Parkinson’s Disease. Metabolites 2021, 11, 29. [Google Scholar] [CrossRef] [PubMed]
- Mertens, K.L.; Kalsbeek, A.; Soeters, M.R.; Eggink, H.M. Bile Acid Signaling Pathways from the Enterohepatic Circulation to the Central Nervous System. Front. Neurosci. 2017, 11, 617. [Google Scholar] [CrossRef] [PubMed]
- Trzeciak, P.; Herbet, M. Role of the Intestinal Microbiome, Intestinal Barrier and Psychobiotics in Depression. Nutrients 2021, 13, 927. [Google Scholar] [CrossRef]
- Caspani, G.; Kennedy, S.; Foster, J.A.; Swann, J. Gut Microbial Metabolites in Depression: Understanding the Biochemical Mechanisms. Microb. Cell 2019, 6, 454. [Google Scholar] [CrossRef] [PubMed]
- Dethloff, F.; Vargas, F.; Elijah, E.; Quinn, R.; Park, D.I.; Herzog, D.P.; Müller, M.B.; Gentry, E.C.; Knight, R.; Gonzalez, A.; et al. Paroxetine Administration Affects Microbiota and Bile Acid Levels in Mice. Front. Psychiatry 2020, 11, 518. [Google Scholar] [CrossRef]
- Anglin, R.E.S.; Samaan, Z.; Walter, S.D.; Sarah, D.M. Vitamin D Deficiency and Depression in Adults: Systematic Review and Meta-Analysis. Br. J. Psychiatry J. Ment. Sci. 2013, 202, 100–107. [Google Scholar] [CrossRef]
- Rao, T.S.S.; Asha, M.R.; Ramesh, B.N.; Rao, K.S.J. Understanding Nutrition, Depression and Mental Illnesses. Indian J. Psychiatry 2008, 50, 77. [Google Scholar] [CrossRef]
- Ramakrishna, B.S. Role of the Gut Microbiota in Human Nutrition and Metabolism. J. Gastroenterol. Hepatol. 2013, 28 (Suppl. 4), 9–17. [Google Scholar] [CrossRef]
- Singh, P.; Rawat, A.; Alwakeel, M.; Sharif, E.; Al Khodor, S. The Potential Role of Vitamin D Supplementation as a Gut Microbiota Modifier in Healthy Individuals. Sci. Rep. 2020, 10, 21641. [Google Scholar] [CrossRef]
- Kennedy, D.O. B Vitamins and the Brain: Mechanisms, Dose and Efficacy—A Review. Nutrients 2016, 8, 68. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Prakash, M.; Apostolopoulos, V. The Effects of Vitamin B on the Immune/Cytokine Network and Their Involvement in Depression. Maturitas 2017, 96, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Young, L.M.; Pipingas, A.; White, D.J.; Gauci, S.; Scholey, A. A Systematic Review and Meta-Analysis of B Vitamin Supplementation on Depressive Symptoms, Anxiety, and Stress: Effects on Healthy and ‘At-Risk’ Individuals. Nutrients 2019, 11, 2232. [Google Scholar] [CrossRef] [PubMed]
- Uebanso, T.; Shimohata, T.; Mawatari, K.; Takahashi, A. Functional Roles of B-Vitamins in the Gut and Gut Microbiome. Mol. Nutr. Food Res. 2020, 64, 2000426. [Google Scholar] [CrossRef]
- Reininghaus, E.Z.; Platzer, M.; Kohlhammer-Dohr, A.; Hamm, C.; Mörkl, S.; Bengesser, S.A.; Fellendorf, F.T.; Lahousen-Luxenberger, T.; Leitner-Afschar, B.; Schöggl, H.; et al. PROVIT: Supplementary Probiotic Treatment and Vitamin B7 in Depression-A Randomized Controlled Trial. Nutrients 2020, 12, 3422. [Google Scholar] [CrossRef]
- Vennemann, F.B.C.; Ioannidou, S.; Valsta, L.M.; Dumas, C.; Ocké, M.C.; Mensink, G.B.M.; Lindtner, O.; Virtanen, S.M.; Tlustos, C.; D’Addezio, L.; et al. Dietary Intake and Food Sources of Choline in European Populations. Br. J. Nutr. 2015, 114, 2046–2055. [Google Scholar] [CrossRef]
- Barrea, L.; Annunziata, G.; Muscogiuri, G.; Di Somma, C.; Laudisio, D.; Maisto, M.; de Alteriis, G.; Tenore, G.C.; Colao, A.; Savastano, S. Trimethylamine-N-Oxide (TMAO) as Novel Potential Biomarker of Early Predictors of Metabolic Syndrome. Nutrients 2018, 10, 1971. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Zhang, P.; Luo, J.; Shen, L.; Zhang, S.; Gu, H.; He, J.; Wang, L.; Zhao, X.; Gan, M.; et al. Dietary Betaine Prevents Obesity through Gut Microbiota-Drived MicroRNA-378a Family. Gut Microbes 2021, 13, 1862612. [Google Scholar] [CrossRef]
- Chen, Q.; Wang, Y.; Jiao, F.; Shi, C.; Pei, M.; Wang, L.; Gong, Z. Betaine Inhibits Toll-like Receptor 4 Responses and Restores Intestinal Microbiota in Acute Liver Failure Mice. Sci. Rep. 2020, 10, 21850. [Google Scholar] [CrossRef]
- Zhong, C.; Lu, Z.; Che, B.; Qian, S.; Zheng, X.; Wang, A.; Bu, X.; Zhang, J.; Ju, Z.; Xu, T.; et al. Choline Pathway Nutrients and Metabolites and Cognitive Impairment after Acute Ischemic Stroke. Stroke 2021, 52, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Nurk, E.; Refsum, H.; Bjelland, I.; Drevon, C.A.; Tell, G.S.; Ueland, P.M.; Vollset, S.E.; Engedal, K.; Nygaard, H.A.; Smith, D.A. Plasma Free Choline, Betaine and Cognitive Performance: The Hordaland Health Study. Br. J. Nutr. 2013, 109, 511–519. [Google Scholar] [CrossRef]
- Riley, C.A.; Renshaw, P.F. Brain Choline in Major Depression: A Review of the Literature. Psychiatry Res. Neuroimaging 2018, 271, 142–153. [Google Scholar] [CrossRef]
- Skonieczna-żydecka, K.; Jakubczyk, K.; Maciejewska-Markiewicz, D.; Janda, K.; Kaźmierczak-Siedlecka, K.; Kaczmarczyk, M.; Łoniewski, I.; Marlicz, W. Gut Biofactory-Neurocompetent Metabolites within the Gastrointestinal Tract. A Scoping Review. Nutrients 2020, 12, 3369. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-T.; Hsieh, C.-P.; Lee, M.-Y.; Chen, L.-C.; Huang, C.-M.; Chen, H.-H.; Chan, M.-H. Betaine Prevents and Reverses the Behavioral Deficits and Synaptic Dysfunction Induced by Repeated Ketamine Exposure in Mice. Biomed. Pharmacother. 2021, 144, 112369. [Google Scholar] [CrossRef]
- Wang, Z.; Tang, W.H.W.; Buffa, J.A.; Fu, X.; Britt, E.B.; Koeth, R.A.; Levison, B.S.; Fan, Y.; Wu, Y.; Hazen, S.L. Prognostic Value of Choline and Betaine Depends on Intestinal Microbiota-Generated Metabolite Trimethylamine-N-Oxide. Eur. Heart J. 2014, 35, 904–910. [Google Scholar] [CrossRef]
- Rath, S.; Heidrich, B.; Pieper, D.H.; Vital, M. Uncovering the Trimethylamine-Producing Bacteria of the Human Gut Microbiota. Microbiome 2017, 5, 54. [Google Scholar] [CrossRef] [PubMed]
- Arias, N.; Arboleya, S.; Allison, J.; Kaliszewska, A.; Higarza, S.G.; Gueimonde, M.; Arias, J.L. The Relationship between Choline Bioavailability from Diet, Intestinal Microbiota Composition, and Its Modulation of Human Diseases. Nutrients 2020, 12, 2340. [Google Scholar] [CrossRef]
- Meinitzer, S.; Baranyi, A.; Holasek, S.; Schnedl, W.J.; Zelzer, S.; Mangge, H.; Herrmann, M.; Meinitzer, A.; Enko, D. Sex-Specific Associations of Trimethylamine-N-Oxide and Zonulin with Signs of Depression in Carbohydrate Malabsorbers and Nonmalabsorbers. Dis. Markers 2020, 2020, 7897240. [Google Scholar] [CrossRef] [PubMed]
- Romano, K.A.; Martinez-del Campo, A.; Kasahara, K.; Chittim, C.L.; Vivas, E.I.; Amador-Noguez, D.; Balskus, E.P.; Rey, F.E. Metabolic, Epigenetic, and Transgenerational Effects of Gut Bacterial Choline Consumption. Cell Host Microbe 2017, 22, 279–290.e7. [Google Scholar] [CrossRef]
- Hammond, G.L. Plasma Steroid-Binding Proteins: Primary Gatekeepers of Steroid Hormone Action. J. Endocrinol. 2016, 230, R13. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-R.; Kim, T.-H.; Choi, K.-C. Functions and Physiological Roles of Two Types of Estrogen Receptors, ERα and ERβ, Identified by Estrogen Receptor Knockout Mouse. Lab. Anim. Res. 2012, 28, 71. [Google Scholar] [CrossRef] [PubMed]
- Sui, Y.; Wu, J.; Chen, J. The Role of Gut Microbial β-Glucuronidase in Estrogen Reactivation and Breast Cancer. Front. Cell Dev. Biol. 2021, 9, 2067. [Google Scholar] [CrossRef] [PubMed]
- Parida, S.; Sharma, D. The Microbiome–Estrogen Connection and Breast Cancer Risk. Cells 2019, 8, 1642. [Google Scholar] [CrossRef]
- Flores, R.; Shi, J.; Fuhrman, B.; Xu, X.; Veenstra, T.D.; Gail, M.H.; Gajer, P.; Ravel, J.; Goedert, J.J. Fecal Microbial Determinants of Fecal and Systemic Estrogens and Estrogen Metabolites: A Cross-Sectional Study. J. Transl. Med. 2012, 10, 253. [Google Scholar] [CrossRef]
- Hussain, T.; Murtaza, G.; Kalhoro, D.H.; Kalhoro, M.S.; Metwally, E.; Chughtai, M.I.; Mazhar, M.U.; Khan, S.A. Relationship between Gut Microbiota and Host-Metabolism: Emphasis on Hormones Related to Reproductive Function. Anim. Nutr. 2021, 7, 1. [Google Scholar] [CrossRef]
- Baker, J.M.; Al-Nakkash, L.; Herbst-Kralovetz, M.M. Estrogen–Gut Microbiome Axis: Physiological and Clinical Implications. Maturitas 2017, 103, 45–53. [Google Scholar] [CrossRef]
- Freeman, E.W. Treatment of Depression Associated with the Menstrual Cycle: Premenstrual Dysphoria, Postpartum Depression, and the Perimenopause. Dialogues Clin. Neurosci. 2002, 4, 177–191. [Google Scholar] [CrossRef]
- Hantsoo, L.; Epperson, C.N. Premenstrual Dysphoric Disorder: Epidemiology and Treatment. Curr. Psychiatry Rep. 2015, 17, 87. [Google Scholar] [CrossRef]
- Cutter, W.J.; Norbury, R.; Murphy, D.G.M. Oestrogen, Brain Function, and Neuropsychiatric Disorders. J. Neurol. Neurosurg. Psychiatry 2003, 74, 837–840. [Google Scholar] [CrossRef][Green Version]
- Flores-Ramos, M.; Burrola-Suárez, M.A.; Guiza-Zayas, R.; Enciso-Araujo, J.M.; Islas-Preciado, D.; Camarena, E.E. Evaluation of Hormonal and Metabolic Factors Related to Depression in Reproductive Age Women. Salud Ment. 2020, 43, 35–41. [Google Scholar] [CrossRef]
- Markle, J.G.M.; Frank, D.N.; Mortin-Toth, S.; Robertson, C.E.; Feazel, L.M.; Rolle-Kampczyk, U.; Von Bergen, M.; McCoy, K.D.; Macpherson, A.J.; Danska, J.S. Sex Differences in the Gut Microbiome Drive Hormone-Dependent Regulation of Autoimmunity. Science 2013, 339, 1084–1088. [Google Scholar] [CrossRef] [PubMed]
- Franzosa, E.A.; Huang, K.; Meadow, J.F.; Gevers, D.; Lemon, K.P.; Bohannan, B.J.M.; Huttenhower, C. Identifying Personal Microbiomes Using Metagenomic Codes. Proc. Natl. Acad. Sci. USA 2015, 112, E2930–E2938. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, Stability and Resilience of the Human Gut Microbiota. Nature 2012, 489, 220. [Google Scholar] [CrossRef]
- Cowan, C.S.M.; Dinan, T.G.; Cryan, J.F. Annual Research Review: Critical Windows-the Microbiota-Gut-Brain Axis in Neurocognitive Development. J. Child Psychol. Psychiatry Allied Discip. 2020, 61, 353–371. [Google Scholar] [CrossRef] [PubMed]
- Elrakaiby, M.; Dutilh, B.E.; Rizkallah, M.R.; Boleij, A.; Cole, J.N.; Aziz, R.K. Pharmacomicrobiomics: The Impact of Human Microbiome Variations on Systems Pharmacology and Personalized Therapeutics. J. Integr. Biol. 2014, 18, 402. [Google Scholar] [CrossRef] [PubMed]
- Rizkallah, M.; Saad, R.; Aziz, R. The Human Microbiome Project, Personalized Medicine and the Birth of Pharmacomicrobiomics. Curr. Pharm. Pers. Med. 2012, 8, 182–193. [Google Scholar] [CrossRef]
- Gupta, V.K.; Paul, S.; Dutta, C. Geography, Ethnicity or Subsistence-Specific Variations in Human Microbiome Composition and Diversity. Front. Microbiol. 2017, 8, 1162. [Google Scholar] [CrossRef] [PubMed]
- Ursell, L.K.; Haiser, H.J.; Van Treuren, W.; Garg, N.; Reddivari, L.; Vanamala, J.; Dorrestein, P.C.; Turnbaugh, P.J.; Knight, R. The Intestinal Metabolome: An Intersection between Microbiota and Host. Gastroenterology 2014, 146, 1470–1476. [Google Scholar] [CrossRef]
- Kosmides, A.K.; Kamisoglu, K.; Calvano, S.E.; Corbett, S.A.; Androulakis, I.P. Metabolomic Fingerprinting: Challenges and Opportunities. Crit. Rev. Biomed. Eng. 2013, 41, 205. [Google Scholar] [CrossRef]
- Riscuta, G.; Xi, D.; Pierre-Victor, D.; Starke-Reed, P.; Khalsa, J.; Duffy, L. Diet, Microbiome, and Epigenetics in the Era of Precision Medicine. Methods Mol. Biol. 2018, 1856, 141–156. [Google Scholar] [CrossRef]
- Hullar, M.A.J.; Fu, B.C. Diet, the Gut Microbiome, and Epigenetics. Cancer J. 2014, 20, 170. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, J.A.; Blaser, M.J.; Caporaso, J.G.; Jansson, J.K.; Lynch, S.V.; Knight, R. Current Understanding of the Human Microbiome. Nat. Med. 2018, 24, 392. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.; Jin, G.; Wang, G.; Liu, T.; Liu, X.; Wang, B.; Cao, H. Current Sampling Methods for Gut Microbiota: A Call for More Precise Devices. Front. Cell. Infect. Microbiol. 2020, 10, 151. [Google Scholar] [CrossRef]
- María Remes Troche, J.; Coss Adame, E.; Ángel Valdovinos Díaz, M.; Gómez Escudero, O.; Eugenia Icaza Chávez, M.; Antonio Chávez-Barrera, J.; Zárate Mondragón, F.; Antonio Ruíz Velarde Velasco, J.; Rafael Aceves Tavares, G.; Antonio Lira Pedrín, M.; et al. Lactobacillus Acidophilus LB: A Useful Pharmabiotic for the Treatment of Digestive Disorders. Ther. Adv. Gastroenterol. 2020, 13, 1756284820971201. [Google Scholar] [CrossRef]
- Flux, M.C.; Lowry, C.A. Finding Intestinal Fortitude: Integrating the Microbiome into a Holistic View of Depression Mechanisms, Treatment, and Resilience. Neurobiol. Dis. 2020, 135, 104578. [Google Scholar] [CrossRef]
- Gawlik-Kotelnicka, O.; Strzelecki, D. Probiotics as a Treatment for “Metabolic Depression”? A Rationale for Future Studies. Pharmaceuticals 2021, 14, 384. [Google Scholar] [CrossRef] [PubMed]
- Skonieczna-żydecka, K.; Marlicz, W.; Misera, A.; Koulaouzidis, A.; Łoniewski, I. Microbiome—The Missing Link in the Gut-Brain Axis: Focus on Its Role in Gastrointestinal and Mental Health. J. Clin. Med. 2018, 7, 521. [Google Scholar] [CrossRef] [PubMed]
- Tian, P.; O’Riordan, K.J.; Lee, Y.-K.; Wang, G.; Zhao, J.; Zhang, H.; Cryan, J.F.; Chen, W. Towards a Psychobiotic Therapy for Depression: Bifidobacterium Breve CCFM1025 Reverses Chronic Stress-Induced Depressive Symptoms and Gut Microbial Abnormalities in Mice. Neurobiol. Stress 2020, 12, 100216. [Google Scholar] [CrossRef]
- Barros-Santos, T.; Silva, K.S.O.; Libarino-Santos, M.; Cata-Preta, E.G.; Reis, H.S.; Tamura, E.K.; De Oliveira-Lima, A.J.; Berro, L.F.; Uetanabaro, A.P.T.; Marinho, E.A.V. Effects of Chronic Treatment with New Strains of Lactobacillus Plantarum on Cognitive, Anxiety- and Depressive-like Behaviors in Male Mice. PLoS ONE 2020, 15, e0234037. [Google Scholar] [CrossRef] [PubMed]
- Steenbergen, L.; Sellaro, R.; van Hemert, S.; Bosch, J.A.; Colzato, L.S. A Randomized Controlled Trial to Test the Effect of Multispecies Probiotics on Cognitive Reactivity to Sad Mood. Brain Behav. Immun. 2015, 48, 258–264. [Google Scholar] [CrossRef]
- Lach, G.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. Anxiety, Depression, and the Microbiome: A Role for Gut Peptides. Neurotherapeutics 2018, 15, 36–59. [Google Scholar] [CrossRef]
- Buffington, S.A.; Di Prisco, G.V.; Auchtung, T.A.; Ajami, N.J.; Petrosino, J.F.; Costa-Mattioli, M. Microbial Reconstitution Reverses Maternal Diet-Induced Social and Synaptic Deficits in Offspring. Cell 2016, 165, 1762–1775. [Google Scholar] [CrossRef]
- Varian, B.J.; Poutahidis, T.; DiBenedictis, B.T.; Levkovich, T.; Ibrahim, Y.; Didyk, E.; Shikhman, L.; Cheung, H.K.; Hardas, A.; Ricciardi, C.E.; et al. Microbial Lysate Upregulates Host Oxytocin. Brain Behav. Immun. 2017, 61, 36. [Google Scholar] [CrossRef] [PubMed]
- McGaughey, K.D.; Yilmaz-Swenson, T.; Elsayed, N.M.; Cruz, D.A.; Rodriguiz, R.M.; Kritzer, M.D.; Peterchev, A.V.; Roach, J.; Wetsel, W.C.; Williamson, D.E. Relative Abundance of Akkermansia spp. and Other Bacterial Phylotypes Correlates with Anxiety- and Depressive-like Behavior Following Social Defeat in Mice. Sci. Rep. 2019, 9, 3281. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.T.; Lai, J.B.; Du, Y.L.; Xu, Y.; Ruan, L.M.; Hu, S.H. Current Understanding of Gut Microbiota in Mood Disorders: An Update of Human Studies. Front. Genet. 2019, 10, 98. [Google Scholar] [CrossRef]
- Yaghoubfar, R.; Behrouzi, A.; Zare Banadkoki, E.; Ashrafian, F.; Lari, A.; Vaziri, F.; Nojoumi, S.A.; Fateh, A.; Khatami, S.; Siadat, S.D. Effect of Akkermansia Muciniphila, Faecalibacterium Prausnitzii, and Their Extracellular Vesicles on the Serotonin System in Intestinal Epithelial Cells. Probiotics Antimicrob. Proteins 2021, 13, 1546–1556. [Google Scholar] [CrossRef]
- Yaghoubfar, R.; Behrouzi, A.; Fateh, A.; Nojoumi, S.A.; Vaziri, F.; Khatami, S.; Siadat, S.D. Effects of Akkermansia Muciniphila and Faecalibacterium Prausnitzii on Serotonin Transporter Expression in Intestinal Epithelial Cells. J. Diabetes Metab. Disord. 2021, 20, 1–5. [Google Scholar] [CrossRef]
- Lukić, I.; Getselter, D.; Ziv, O.; Oron, O.; Reuveni, E.; Koren, O.; Elliott, E. Antidepressants Affect Gut Microbiota and Ruminococcus Flavefaciens Is Able to Abolish Their Effects on Depressive-like Behavior. Transl. Psychiatry 2019, 9, 133. [Google Scholar] [CrossRef]
- Zou, R.; Tian, P.; Xu, M.; Zhu, H.; Zhao, J.; Zhang, H.; Chen, W.; Wang, G. Psychobiotics as a Novel Strategy for Alleviating Anxiety and Depression. J. Funct. Foods 2021, 86, 1756–4646. [Google Scholar] [CrossRef]
- Yong, S.J.; Tong, T.; Chew, J.; Lim, W.L. Antidepressive Mechanisms of Probiotics and Their Therapeutic Potential. Front. Neurosci. 2019, 13, 1361. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.; Bhati, A.; Priya, K.; Sharma, K.K.; Singhal, B. Precision Postbiotics and Mental Health: The Management of Post-COVID-19 Complications. Probiotics Antimicrob. Proteins 2021, 1, 1. [Google Scholar] [CrossRef]
- Vindigni, S.M.; Surawicz, C.M. Fecal Microbiota Transplantation. Gastroenterol. Clin. N. Am. 2017, 46, 171–185. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.W.; Kuo, C.H.; Kuo, F.C.; Wang, Y.K.; Hsu, W.H.; Yu, F.J.; Hu, H.M.; Hsu, P.I.; Wang, J.Y.; Wu, D.C. Fecal Microbiota Transplantation: Review and Update. J. Formos. Med. Assoc. 2019, 118 (Suppl. 1), S23–S31. [Google Scholar] [CrossRef]
- Chinna Meyyappan, A.; Forth, E.; Wallace, C.J.K.; Milev, R. Effect of Fecal Microbiota Transplant on Symptoms of Psychiatric Disorders: A Systematic Review. BMC Psychiatry 2020, 20, 299. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert Consensus Document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods 2019, 8, 92. [Google Scholar] [CrossRef] [PubMed]
- Corzo, N.; Alonso, J.L.; Azpiroz, F.; Calvo, M.A.; Cirici, M.; Leis, R.; Lombó, F.; Mateos-Aparicio, I.; Plou, F.J.; Ruas-Madiedo, P.; et al. Prebiotics: Concept, Properties and Beneficial Effects. Nutr. Hosp. 2015, 31 (Suppl. 1), 99–118. [Google Scholar] [CrossRef]
- Liu, R.T.; Walsh, R.F.L.; Sheehan, A.E. Prebiotics and Probiotics for Depression and Anxiety: A Systematic Review and Meta-Analysis of Controlled Clinical Trials. Neurosci. Biobehav. Rev. 2019, 102, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Noonan, S.; Zaveri, M.; Macaninch, E.; Martyn, K.; Sanjay, M. Food & Mood: A Review of Supplementary Prebiotic and Probiotic Interventions in the Treatment of Anxiety and Depression in Adults. BMJ Nutr. Prev. Health 2020, 3, 351. [Google Scholar] [CrossRef] [PubMed]
- Paiva, I.H.R.; Duarte-Silva, E.; Peixoto, C.A. The Role of Prebiotics in Cognition, Anxiety, and Depression. Eur. Neuropsychopharmacology 2020, 34, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Varzakas, T.; Kandylis, P.; Dimitrellou, D.; Salamoura, C.; Zakynthinos, G.; Proestos, C. Innovative and Fortified Food: Probiotics, Prebiotics, GMOs, and Superfood. In Preparation and Processing of Religious and Cultural Foods; Woodhead Publishing: Sawston, UK, 2018; pp. 67–129. [Google Scholar] [CrossRef]
- Cunningham, M.; Azcarate-Peril, M.A.; Barnard, A.; Benoit, V.; Grimaldi, R.; Guyonnet, D.; Holscher, H.D.; Hunter, K.; Manurung, S.; Obis, D.; et al. Shaping the Future of Probiotics and Prebiotics. Trends Microbiol. 2021, 29, 667–685. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, J.M. The Food Matrix: Implications in Processing, Nutrition and Health. Crit. Rev. Food Sci. Nutr. 2019, 59, 3612–3629. [Google Scholar] [CrossRef] [PubMed]
- Şanlier, N.; Gökcen, B.B.; Sezgin, A.C. Health Benefits of Fermented Foods. Crit. Rev. Food Sci. Nutr. 2019, 59, 506–527. [Google Scholar] [CrossRef]
- Stiemsma, L.T.; Nakamura, R.E.; Nguyen, J.G.; Michels, K.B. Does Consumption of Fermented Foods Modify the Human Gut Microbiota? J. Nutr. 2020, 150, 1680–1692. [Google Scholar] [CrossRef] [PubMed]
- Aslam, H.; Green, J.; Jacka, F.N.; Collier, F.; Berk, M.; Pasco, J.; Dawson, S.L. Fermented Foods, the Gut and Mental Health: A Mechanistic Overview with Implications for Depression and Anxiety. Nutr. Neurosci. 2020, 23, 659–671. [Google Scholar] [CrossRef]
- Wei, Y.B.; Melas, P.A.; Wegener, G.; Mathe, A.A.; Lavebratt, C. Antidepressant-like Effect of Sodium Butyrate Is Associated with an Increase in TET1 and in 5-Hydroxymethylation Levels in the Bdnf Gene. Int. J. Neuropsychopharmacol. 2014, 18, pyv026. [Google Scholar] [CrossRef]
- Han, A.; Sung, Y.B.; Chung, S.Y.; Kwon, M.S. Possible Additional Antidepressant-like Mechanism of Sodium Butyrate: Targeting the Hippocampus. Neuropharmacology 2014, 81, 292–302. [Google Scholar] [CrossRef]
- Peng, J. Effects of Sodium Butyrate on Spatial Learning and Memory Ability in Chronic Stress Model Rats. China Pharm. 2016, 27, 2233–2235. [Google Scholar] [CrossRef]
- Valvassori, S.; Varela, R.; Arent, C.; Dal-Pont, G.; Bobsin, T.; Budni, J.; Reus, G.; Quevedo, J. Sodium Butyrate Functions as an Antidepressant and Improves Cognition with Enhanced Neurotrophic Expression in Models of Maternal Deprivation and Chronic Mild Stress. Curr. Neurovascular Res. 2014, 11, 359–366. [Google Scholar] [CrossRef]
- Valvassori, S.; Resende, W.; Budni, J.; Dal-Pont, G.; Bavaresco, D.; Reus, G.; Carvalho, A.; Goncalves, C.; Furlanetto, C.; Streck, E.; et al. Sodium Butyrate, a Histone Deacetylase Inhibitor, Reverses Behavioral and Mitochondrial Alterations in Animal Models of Depression Induced by Early- or Late-Life Stress. Curr. Neurovascular Res. 2015, 12, 312–320. [Google Scholar] [CrossRef]
- Huang, W.; Hu, W.; Cai, L.; Zeng, G.; Fang, W.; Dai, X.; Ye, Q.; Chen, X.; Zhang, J. Acetate Supplementation Produces Antidepressant-like Effect via Enhanced Histone Acetylation. J. Affect. Disord. 2021, 281, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S.; Jasbi, P.; Jin, Y.; Bauer, S.; Williams, S.; Fessler, S.N.; Gu, H. Daily Vinegar Ingestion Improves Depression Scores and Alters the Metabolome in Healthy Adults: A Randomized Controlled Trial. Nutrients 2021, 13, 4020. [Google Scholar] [CrossRef]
- Hao, C.; Gao, Z.; Liu, X.J.; Rong, Z.; Jia, J.; Kang, K.; Guo, W.; Li, J. Intravenous Administration of Sodium Propionate Induces Antidepressant or Prodepressant Effect in a Dose Dependent Manner. Sci. Rep. 2020, 10, 19917. [Google Scholar] [CrossRef]
- Das, L.; Bhaumik, E.; Raychaudhuri, U.; Chakraborty, R. Role of Nutraceuticals in Human Health. J. Food Sci. Technol. 2012, 49, 173–183. [Google Scholar] [CrossRef]
- Sarris, J.; Murphy, J.; Mischoulon, D.; Papakostas, G.I.; Fava, M.; Berk, M.; Ng, C.H. Adjunctive Nutraceuticals for Depression: A Systematic Review and Meta-Analyses. Am. J. Psychiatry 2016, 173, 575–587. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Ortega, M.A.; García-Montero, C.; Fraile-Martinez, O.; Monserrat, J.; Lahera, G.; Mora, F.; Rodriguez-Quiroga, A.; Fernandez-Rojo, S.; Quintero, J.; et al. Exploring the Role of Nutraceuticals in Major Depressive Disorder (MDD): Rationale, State of the Art and Future Prospects. Pharmaceuticals 2021, 14, 821. [Google Scholar] [CrossRef]
- Malaguarnera, L. Vitamin D and Microbiota: Two Sides of the Same Coin in the Immunomodulatory Aspects. Int. Immunopharmacol. 2020, 79, 106112. [Google Scholar] [CrossRef] [PubMed]
- Lauinger, L.; Kaiser, P. Sensing and Signaling of Methionine Metabolism. Metabolites 2021, 11, 83. [Google Scholar] [CrossRef]
- Mentch, S.J.; Locasale, J.W. One-Carbon Metabolism and Epigenetics: Understanding the Specificity. Ann. N. Y. Acad. Sci. 2016, 1363, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Jabs, S.; Biton, A.; Bécavin, C.; Nahori, M.A.; Ghozlane, A.; Pagliuso, A.; Spanò, G.; Guérineau, V.; Touboul, D.; Giai Gianetto, Q.; et al. Impact of the Gut Microbiota on the M6A Epitranscriptome of Mouse Cecum and Liver. Nat. Commun. 2020, 11, 1344. [Google Scholar] [CrossRef]
- Miousse, I.R.; Pathak, R.; Garg, S.; Skinner, C.M.; Melnyk, S.; Pavliv, O.; Hendrickson, H.; Landes, R.D.; Lumen, A.; Tackett, A.J.; et al. Short-Term Dietary Methionine Supplementation Affects One-Carbon Metabolism and DNA Methylation in the Mouse Gut and Leads to Altered Microbiome Profiles, Barrier Function, Gene Expression and Histomorphology. Genes Nutr. 2017, 12, 22. [Google Scholar] [CrossRef]
- Zheng, Y.; Cantley, L.C. Toward a Better Understanding of Folate Metabolism in Health and Disease. J. Exp. Med. 2019, 216, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Engevik, M.A.; Morra, C.N.; Röth, D.; Engevik, K.; Spinler, J.K.; Devaraj, S.; Crawford, S.E.; Estes, M.K.; Kalkum, M.; Versalovic, J. Microbial Metabolic Capacity for Intestinal Folate Production and Modulation of Host Folate Receptors. Front. Microbiol. 2019, 10, 2305. [Google Scholar] [CrossRef]
- Costantini, L.; Molinari, R.; Farinon, B.; Merendino, N. Impact of Omega-3 Fatty Acids on the Gut Microbiota. Int. J. Mol. Sci. 2017, 18, 2645. [Google Scholar] [CrossRef] [PubMed]
- Westfall, S.; Pasinetti, G.M. The Gut Microbiota Links Dietary Polyphenols With Management of Psychiatric Mood Disorders. Front. Neurosci. 2019, 13, 1196. [Google Scholar] [CrossRef]
- Marques, C.; Fernandes, I.; Meireles, M.; Faria, A.; Spencer, J.P.E.; Mateus, N.; Calhau, C. Gut Microbiota Modulation Accounts for the Neuroprotective Properties of Anthocyanins. Sci. Rep. 2018, 8, 11341. [Google Scholar] [CrossRef]
- Park, M.; Choi, J.; Lee, H.J. Flavonoid-Rich Orange Juice Intake and Altered Gut Microbiome in Young Adults with Depressive Symptom: A Randomized Controlled Study. Nutrients 2020, 12, 1815. [Google Scholar] [CrossRef]
- Bajinka, O.; Tan, Y.; Abdelhalim, K.A.; Özdemir, G.; Qiu, X. Extrinsic Factors Influencing Gut Microbes, the Immediate Consequences and Restoring Eubiosis. AMB Express 2020, 10, 130. [Google Scholar] [CrossRef] [PubMed]
- Koh, A.; De Vadder, F.; Kovatcheva-Datchary, P.; Bäckhed, F. From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites. Cell 2016, 165, 1332–1345. [Google Scholar] [CrossRef]
- Gantenbein, K.V.; Kanaka-Gantenbein, C. Mediterranean Diet as an Antioxidant: The Impact on Metabolic Health and Overall Wellbeing. Nutrients 2021, 13, 1951. [Google Scholar] [CrossRef] [PubMed]
- Myhrstad, M.C.W.; Tunsjø, H.; Charnock, C.; Telle-Hansen, V.H. Dietary Fiber, Gut Microbiota, and Metabolic Regulation—Current Status in Human Randomized Trials. Nutrients 2020, 12, 859. [Google Scholar] [CrossRef] [PubMed]
- Opara, E.I.; Chohan, M. Culinary Herbs and Spices: Their Bioactive Properties, the Contribution of Polyphenols and the Challenges in Deducing Their True Health Benefits. Int. J. Mol. Sci. 2014, 15, 19183. [Google Scholar] [CrossRef] [PubMed]
- Galli, C. Bioactive Components in Mediterranean Diets. Nutrafoods 2012, 11, 11–17. [Google Scholar] [CrossRef]
- Oliver, A.; Chase, A.B.; Weihe, C.; Orchanian, S.B.; Riedel, S.F.; Hendrickson, C.L.; Lay, M.; Sewall, J.M.; Martiny, J.B.H.; Whiteson, K. High-Fiber, Whole-Food Dietary Intervention Alters the Human Gut Microbiome but Not Fecal Short-Chain Fatty Acids. Msystems 2021, 6, e00115-21. [Google Scholar] [CrossRef]
- Baxter, N.T.; Schmidt, A.W.; Venkataraman, A.; Kim, K.S.; Waldron, C.; Schmidt, T.M. Dynamics of Human Gut Microbiota and Short-Chain Fatty Acids in Response to Dietary Interventions with Three Fermentable Fibers. mBio 2019, 10, e02566-18. [Google Scholar] [CrossRef]
- Akagawa, S.; Akagawa, Y.; Nakai, Y.; Yamagishi, M.; Yamanouchi, S.; Kimata, T.; Chino, K.; Tamiya, T.; Hashiyada, M.; Akane, A.; et al. Fiber-Rich Barley Increases Butyric Acid-Producing Bacteria in the Human Gut Microbiota. Metabolites 2021, 11, 559. [Google Scholar] [CrossRef]
- Precup, G.; Vodnar, D.C. Gut Prevotella as a Possible Biomarker of Diet and Its Eubiotic versus Dysbiotic Roles: A Comprehensive Literature Review. Br. J. Nutr. 2019, 122, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.M. The Immune Response to Prevotella Bacteria in Chronic Inflammatory Disease. Immunology 2017, 151, 363. [Google Scholar] [CrossRef]
- Nehlig, A. The Neuroprotective Effects of Cocoa Flavanol and Its Influence on Cognitive Performance. Br. J. Clin. Pharmacol. 2013, 75, 716. [Google Scholar] [CrossRef]
- Martín-Peláez, S.; Camps-Bossacoma, M.; Massot-Cladera, M.; Rigo-Adrover, M.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Effect of Cocoa’s Theobromine on Intestinal Microbiota of Rats. Mol. Nutr. Food Res. 2017, 61, 1700238. [Google Scholar] [CrossRef]
- O’neill Rothenberg, D.; Zhang, L. Mechanisms Underlying the Anti-Depressive Effects of Regular Tea Consumption. Nutrients 2019, 11, 1361. [Google Scholar] [CrossRef]
- Trefflich, I.; Marschall, H.U.; di Giuseppe, R.; Ståhlman, M.; Michalsen, A.; Lampen, A.; Abraham, K.; Weikert, C. Associations between Dietary Patterns and Bile Acids—Results from a Cross-Sectional Study in Vegans and Omnivores. Nutrients 2020, 12, 47. [Google Scholar] [CrossRef]
- Socała, K.; Szopa, A.; Serefko, A.; Poleszak, E.; Wlaź, P. Neuroprotective Effects of Coffee Bioactive Compounds: A Review. Int. J. Mol. Sci. 2020, 22, 107. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Liu, X.; Zhang, D. Fish Consumption and Risk of Depression: A Meta-Analysis. J. Epidemiol. Community Health 2016, 70, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Klimova, B.; Novotny, M.; Valis, M. The Impact of Nutrition and Intestinal Microbiome on Elderly Depression—A Systematic Review. Nutrients 2020, 12, 710. [Google Scholar] [CrossRef]
- Ezra-Nevo, G.; Henriques, S.F.; Ribeiro, C. The Diet-Microbiome Tango: How Nutrients Lead the Gut Brain Axis. Curr. Opin. Neurobiol. 2020, 62, 122–132. [Google Scholar] [CrossRef]
- Koopman, M.; Daniels, J.K.; Spitzer, C.; Lampe, A.; El Aidy, S. Depressed Gut? The Microbiota-Diet-Inflammation Trialogue in Depression. Curr. Opin. Psychiatry 2017, 30, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Monda, V.; Villano, I.; Messina, A.; Valenzano, A.; Esposito, T.; Moscatelli, F.; Viggiano, A.; Cibelli, G.; Chieffi, S.; Monda, M.; et al. Exercise Modifies the Gut Microbiota with Positive Health Effects. Oxidative Med. Cell. Longev. 2017, 2017, 3831972. [Google Scholar] [CrossRef]
- Gubert, C.; Kong, G.; Renoir, T.; Hannan, A.J. Exercise, Diet and Stress as Modulators of Gut Microbiota: Implications for Neurodegenerative Diseases. Neurobiol. Dis. 2020, 134, 104621. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.-F.; Ferreira Rocha, N.; Paes, F.; Arias-Carrión, O.; Machado, S.; de Sá Filho, A. Neural Mechanisms of Exercise: Effects on Gut Miccrobiota and Depression. CNS Neurol. Disord. Drug Targets 2015, 14, 1312–1314. [Google Scholar] [CrossRef] [PubMed]
- Sheng, L.; Wang, Y.; Jiang, A.; Zhou, Y.; Zhou, H. Effect of Regular Physical Exercise on Gut Microbiota and Depressive Behaviors in Rats. J. Food Qual. 2021, 2021, 1210089. [Google Scholar] [CrossRef]
- Tabone, M.; Bressa, C.; García-Merino, J.A.; Moreno-Pérez, D.; Van, E.C.; Castelli, F.A.; Fenaille, F.; Larrosa, M. The Effect of Acute Moderate-Intensity Exercise on the Serum and Fecal Metabolomes and the Gut Microbiota of Cross-Country Endurance Athletes. Sci. Rep. 2021, 11, 3558. [Google Scholar] [CrossRef] [PubMed]
- Proia, P.; di Liegro, C.M.; Schiera, G.; Fricano, A.; Di Liegro, I. Lactate as a Metabolite and a Regulator in the Central Nervous System. Int. J. Mol. Sci. 2016, 17, 1450. [Google Scholar] [CrossRef]
- Nalbandian, M.; Takeda, M. Lactate as a Signaling Molecule That Regulates Exercise-Induced Adaptations. Biology 2016, 5, 38. [Google Scholar] [CrossRef]
- Scheiman, J.; Luber, J.M.; Chavkin, T.A.; MacDonald, T.; Tung, A.; Pham, L.D.; Wibowo, M.C.; Wurth, R.C.; Punthambaker, S.; Tierney, B.T.; et al. Meta-Omics Analysis of Elite Athletes Identifies a Performance-Enhancing Microbe That Functions via Lactate Metabolism. Nat. Med. 2019, 25, 1104–1109. [Google Scholar] [CrossRef]
- Clark, A.; Mach, N. Exercise-Induced Stress Behavior, Gut-Microbiota-Brain Axis and Diet: A Systematic Review for Athletes. J. Int. Soc. Sports Nutr. 2016, 13, 43. [Google Scholar] [CrossRef]
- Intlekofer, K.A.; Berchtold, N.C.; Malvaez, M.; Carlos, A.J.; McQuown, S.C.; Cunningham, M.J.; Wood, M.A.; Cotman, C.W. Exercise and Sodium Butyrate Transform a Subthreshold Learning Event into Long-Term Memory via a Brain-Derived Neurotrophic Factor-Dependent Mechanism. Neuropsychopharmacology 2013, 38, 2027–2034. [Google Scholar] [CrossRef]
- O’Sullivan, O.; Cronin, O.; Clarke, S.F.; Murphy, E.F.; Molloy, M.G.; Shanahan, F.; Cotter, P.D. Exercise and the Microbiota. Gut Microbes 2015, 6, 131. [Google Scholar] [CrossRef]
- Matenchuk, B.A.; Mandhane, P.J.; Kozyrskyj, A.L. Sleep, Circadian Rhythm, and Gut Microbiota. Sleep Med. Rev. 2020, 53, 101340. [Google Scholar] [CrossRef]
- Li, Y.; Hao, Y.; Fan, F.; Zhang, B. The Role of Microbiome in Insomnia, Circadian Disturbance and Depression. Front. Psychiatry 2018, 9, 669. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.P.; Easson, C.; Lyle, S.M.; Kapoor, R.; Donnelly, C.P.; Davidson, E.J.; Parikh, E.; Lopez, J.V.; Tartar, J.L. Gut Microbiome Diversity Is Associated with Sleep Physiology in Humans. PLoS ONE 2019, 14, e0222394. [Google Scholar] [CrossRef]
- Zhang, Q.; Yun, Y.; An, H.; Zhao, W.; Ma, T.; Wang, Z.; Yang, F. Gut Microbiome Composition Associated With Major Depressive Disorder and Sleep Quality. Front. Psychiatry 2021, 12, 551. [Google Scholar] [CrossRef]
- Mortaş, H.; Bilici, S.; Karakan, T. The Circadian Disruption of Night Work Alters Gut Microbiota Consistent with Elevated Risk for Future Metabolic and Gastrointestinal Pathology. Chronobiol. Int. 2020, 37, 1067–1081. [Google Scholar] [CrossRef]
- Nutt, D.J.; Wilson, S.; Paterson, L. Sleep Disorders as Core Symptoms of Depression. Dialogues Clin. Neurosci. 2008, 10, 329. [Google Scholar] [CrossRef] [PubMed]
- Haynes, P.L.; Mcquaid, J.R.; Ancoli-Israel, S.; Martin, J.L. Disrupting Life Events and the Sleep-Wake Cycle in Depression. Psychol. Med. 2006, 36, 1363–1373. [Google Scholar] [CrossRef] [PubMed]
- Yoshii, K.; Hosomi, K.; Sawane, K.; Kunisawa, J. Metabolism of Dietary and Microbial Vitamin B Family in the Regulation of Host Immunity. Front. Nutr. 2019, 6, 48. [Google Scholar] [CrossRef]
| Microbial Metabolite | Microorganisms Implicated | Microbiota-Gut-Brain Axis | Status In MDD | Translational Approaches | References |
|---|---|---|---|---|---|
| Short-chain fatty acids (SCFAs) | Eubacterium rectale, Roseburia faecis, Eubacterium hallii, and Faecalibacterium prausnitziiappears to be the major producers of butyrate in the gut. Veilonella spp., Lactobacillus spp., Bacteroides spp., and Propionibacterium spp. are some of the most representative bacteria involved in propionate production. Conversely, acetate production is commonly spread among numerous bacterial classes | SCFAs are shown to be a central mechanism of gut-brain communication. They can cross the BBB activating several mechanisms in the brain, modulating the levels of neurotrophic factors, neurotransmitters, neurogenesis, and reducing neuroinflammation and glial dysfunction. SCFAs interact with enteroendocrine cells and promote indirect signaling to the brain by inducing the production of several hormones and neurotransmitters like GABA and serotonin in the gut. There is a positive association between Allobaculum, acetate, and serotonin levels. | Acetate, propionate, butyrate: Downregulated Isovalerate: Upregulated Reduced levels of butyrate producers like Faecalibacterium and Coprococcus spp. are correlated with MDD severity. Fecal measures of SCFAs also found a significant association between acetate, propionate, and butyrate levels with both depressive and gastrointestinal symptoms in young adults, therefore supporting that SCFAs are importantly related to the development of MDD. A significant reduction of fecal SCFAs between depressed and non-depressed women was also proposed to be a potential contributor of suffering from MDD with a possible inverse association with depression severity. Depressed patients show a prominent epigenetic footprint in comparison to non-depressed subjects, including critical alterations in DNA methylation, histone modifications, and non-coding RNAs (i.e., microRNAs). SCFAs act as histone deacetylases (HDAC) inhibitors altering gene expression. Propionate production is associated with a decreased anticipatory response of the reward system, related to the monoamine dopamine. Altered dopaminergic neurotransmission is a major feature underpinning MDD, which is related to anhedonia. | SCFA-mediated inhibition of HDAC may lead to the hyperacetylation of histones H3/H4 and hence increase BDNF expression, showing antidepressant effects in mice. This epigenetic modulation is mostly responsible for the immunomodulatory action of SCFAs. In the CNS, SCFAs are major mediators of microglial maturation and function whereas, in the gut, SCFAs induce the differentiation of Treg cells. Overall, the epigenetic and immunomodulatory effects of acetate, and more prominently propionate and butyrate exert promising antidepressant effects, as they are mostly correlated with favorable microbial populations. Prior studies have proven the anti-depressants effect of butyrate by reversing behavioral alterations in mouse models like anhedonia, low energy, cognitive and social abilities. Similarly, the use of rifaximin was related to a decreased depressive-like behavior due to its favorable effects on Ruminococcaceae and Lachnospiraceae, which positively correlated with butyrate production. Conversely, another type of SCFA, isovaleric acid has been shown to be correlated with certain bacterial populations related to MDD and augmented cortisol levels. A proposed mechanism of this pro-depressive effect of isovaleric acid is through interfering with synaptic neurotransmitter release. | [68,69,70,76,77,78,79] |
| Lactate | Lactic acid bacteria, bifidobacteria, or proteobacteria. | Lactate can cross the BBB to match the energetic needs of the brain, influencing many neuronal functions such as excitability, plasticity, and memory consolidation. | The role of lactate in the etiopathogenesis of MDD is poorly understood. However, a plausible explanation is an accumulation of lactate in the brain due to an impaired mitochondrial function -a major feature of different psychiatric diseases-. In MDD, there is increased ventricular lactate. Patients with severe depression show a significant detection of lactate in the urine in comparison to moderate and non-depressed subjects. | Peripheral administration of lactate reverts the effects of stress and exerts antidepressant effects, supporting hippocampal neurogenesis related to changes in serotonin receptor trafficking. Physical exercise may be a powerful link between lactate and antidepressant effects influencing not only the muscle production of lactate but also the MGB axis. | [97,98,99,100,101,102,103,104,105,106,107] |
| Tryptophan (Trp) metabolites | Some bacterial communities may synthesize Trp like Escherichia coli. 5 phyla are involved in Trp metabolism: Firmicutes, Bacteroidetes, Actinobacteria, Proteobacteria, and Fusobacteria, with a central role of Clostridium, Burkholderia, Streptomyces, Pseudomonas, and Bacillus genera | Trp is importantly implicated in the synthesis of serotonin and other critical metabolites, including those belonging to the kynurenine pathway; tryptamine; indole, and derivates. All these components exert signaling actions in the brain, involved in its proper functioning. | Serotonin synthesis from Trp is impaired An altered kynurenine pathway is reported in patients with MDD, with an upregulation of the enzyme indoleamine 2 3-dioxygenase (IDO) and neurotoxic components like 3-hydroxykynurenine and quinolinic acid along with a reduction of neuroprotective members like kynurenine and kynurenic acid Increased tryptamine levels and indole metabolism | Probiotic bacteria (Bifidobacterium longum subsp. infantis E41 and Bifidobacterium breve M2CF22M7) were associated with an increased 5-hydroxytryptophan, tph1, serotonin, and BDNF in the brain. Current clinical trials are evaluating the efficacy of different probiotic strains with prebiotics and antidepressants in the Trp metabolism, kynurenine pathway, and other pathophysiological mechanisms involved in MDD. Anthocyanins, a group of polyphenols used as nutraceutical appears to modulate host Trp metabolism increasing neuroprotective kynurenic acid Ensuring an adequate nutritional and dietary context is crucial to prevent gut dysbiosis and Trp metabolism, although the evidence has failed to find benefits from increasing dietary Trp or through supplementation | [30,112,113,114,115,116,117,119,120,121,125,126,127,128,129,261] |
| Amino acids | Many bacterial groups are involved in the synthesis and metabolism of several amino acids including glutamate, phenylalanine, tyrosine, threonine, isoleucine, alanine, serine, and oxidized proline | Glutamate is an important neurotransmitter as well as GABA, which is synthesized from glutamate by different microbial communities, especially lactic acid bacteria. Phenylalanine is involved in the synthesis of the catecholamines epinephrine, norepinephrine, and dopamine | Glutamate, GABA, and catecholamines functioning in the brain is impaired in patients with MDD Chronic unpredictable mild stress rats, as well as specific pathogen-free (SPF) and germ-free (GF), have unraveled that gut microbiota is critical for serum levels of plenty amino acid, as well as the concentration of different amino acids in the host brain regions Reduced levels of phenylalanine, aspartate, glutamate, and serine are found in patients with MDD and heart failure | SCFAs from vinegar (acetic acid) is associated with an improved amino acid metabolism (Increased levels of glycine, serine, and threonine) in healthy subjects Creatine supplementation (Nutraceutical) may be related to an improved amino acid metabolism Little and controversial evidence is available regarding the benefits from the administration of some amino acids like phenylalanine and tyrosine Targeting gut microbiota and gut dysbiosis through lifestyle interventions seems to be the most adequate strategy to influence amino acid levels and metabolism | [131,132,133,134,136,137,138,139,140,141,143,144,145,247,251] |
| Secondary bile acids (BAs) | Clostridium, Enterococcus, Bifidobacterium, Lactobacillus, Bacteroides genera, as well as E. coli, C. testosteroni, and Ruminococcus spp. Archaea like Methanobrevibacter smithii and Methanosphera stadmanae | BAs exert important signaling actions in the brain through Farnesoid-X-Receptor (FXR) and the G-Protein-Coupled Bile Acid receptor-1 (GPBAR-1). BAs may act directly by crossing the blood–brain barrier or indirectly, activating FXR and GPBAR-1 receptors in the gut, leading to the production of fibroblast growth factor 19 (FGF19) and glucagon-like peptide 1 (GLP-1), signaling to the brain | In neuropsychiatric disorders, the most prominent signaling route of BA is through the FXR. In the hippocampus, an increased FXR expression seems to cause depression-like symptoms and reduced BDNF levels in rats The use of some antidepressants such as paroxetine alter gut microbiota and BA production in mice | The activation of GPBAR-1 by the secondary bile acid tauroursodeoxycholic acid (TUDCA) as well as the activation of other receptors in the brain of BA like vitamin D receptor (VDR) and pregnane X receptor (PXR) activated by lithocholic acid (LCA) appears to present antidepressant effects Higher consumption of animal products and fats is associated with increased BA production, which is in turn inversely correlated with dietary fiber intake. Ensuring an adequate fiber intake, polyphenols, and high-quality fats while limiting overconsumption of red and processed meats, low-quality fats intake, or ultra-processed foods will benefit BA metabolism and actions in the brain | [30,149,150,152,153,157,158,159,160,277] |
| Vitamins | B1 producers are Bacteroides fragilis and Prevotella copri (phylum Bacteroidetes); Clostridium difficile, some Lactobacillus spp., and Ruminococcus lactaris (Firmicutes); Bifidobacterium spp. (Actinobacteria); and Fusobacterium varium. Bacteroides fragilis and Prevotella copri (Bacteroidetes); Clostridium difficile, Lactobacillus plantarum, L. fermentum, and Ruminococcus lactaris (Firmicutes) can synthesize B2 vitamin. Bacteroides fragilis and Prevotella copri (Bacteroidetes); Ruminococcus lactaris, Clostridium difficile (Firmicutes); Bifidobacterium infantis (Actinobacteria); Helicobacter pylori (Proteobacteria); and Fusobacterium varium (Fusobacteria) can produce B3 vitamin. B3 can be synthesized from Trp as well. Bacteroides fragilis and Prevotella copri (Bacteroidetes); some Ruminococcus spp. (R. lactaris and R. torques) (Firmicutes); Salmonella enterica and Helicobacter pylori (Proteobacteria) synthesize B5. B6 producers are Bacteroides fragilis and Prevotella copri (Bacteroidetes), Bifidobacterium longum and, Collinsella aerofaciens (Actinobacteria), and Helicobacter pylori (Proteobacteria). B7 producers are Bacteroides fragilis and Prevotella copri (Bacteroidetes); Fusobacterium varium (Fusobacteria) and Campylobacter coli (Proteobacteria) Folate or B9 is produced by Bacteroides fragilis and Prevotella copri (Bacteroidetes); Clostridium difficile, Lactobacillus plantarum, L. reuteri, L. delbrueckii ssp. bulgaricus, and Streptococcus thermophilus (Firmicutes), some species in Bifidobacterium spp. (Actinobacteria); Fusobacterium varium (Fusobacteria) and Salmonella enterica (Proteobacteria). Finally, B12 is produced by Bacteroides fragilis and Prevotella copri (Bacteroidetes); Clostridium difficile, Faecalibacterium prausnitzii and Ruminococcus lactaris (Firmicutes); Bifidobacterium animalis, B.infantis, and B.longum (Actinobacteria); Fusobacterium varium (Fusobacteria). Lactobacillus plantarum and L. coryniformis from fermented food also produce B12. | Gut dysbiosis impairs the synthesis of these vitamins which are crucial for the proper functioning of the brain. | Patients with MDD show deficiencies of vitamins, especially D and those from B complex. Low levels of vitamin B1 (thiamine), B2 (riboflavin) B3 (niacin), B6 (pyridoxine) B7/8 (biotin), B9 (folate), and B12 (cyanocobalamin) are pivotal B vitamins associated with MDD. | Besides vitamin D supplementation improves gut microbiota in MDD, several studies are confirming the benefits from using certain B vitamins in combination with probiotics, frequently represented by different species and strains of Bifidobacterium and Lactobacillus. In some studies, in comparison to the groups that only received vitamin B7 plus placebo or probiotics plus placebo, patients supplemented with both probiotics and vitamin showed an increased synthesis of vitamin B6 and B7 and improved clinical outcomes. Gut microbiota is crucial for determining the body levels of B vitamins, but an adequate dietary context is needed to obtain the benefits from those bacteria involved in vitamin synthesis, with noteworthy positive outcomes for the brain. | [164,165,166,167,168,169,301] |
| Choline-derived metabolites | Firmicutes phyla (Clostridium cluster XIVa and Eubacterium strains), Actinobacteria, and Proteobacteria | Choline may be transformed into plenty of metabolites like trimethylamine (TMA), betaine, phosphocholine, and acetylcholine (neurotransmitter). The gut microbiota status and the TMA production and its metabolic product derived, trimethylamine N-oxide (TMAO) are crucial biomarkers of choline metabolism and predictors of the beneficial or detrimental effects in the brain | An increased TMA and TMAO levels related to gut dysbiosis are directly correlated depressive symptoms severity in females and males Elevated levels of choline-consuming bacteria are related to an aberrant global DNA methylation pattern in mice and abnormal behavior in mice | Similar to BA, some animal products and fatty foods may contain high levels of choline and then, it may raise TMA/TMAO levels. However, if maintaining a eubiotic environment with an adequate dietary fiber intake, polyphenols, vitamins, high-quality fats while limiting overconsumption of red and processed meats, low-quality fats, excessive salt, additives, or ultra-processed foods TMA levels will not have any detrimental effect. Conversely, the variety of components included in a proper dietary pattern like those included in the Mediterranean diet will benefit the host health and aid to ameliorate depressive symptoms. | [30,170,171,176,177,179,180,181,182,183] |
| Estrobolome | This term refers to a set of bacteria that are able of metabolizing and modulate the estrogen levels in the body. The gut microbiota is a major determinant of the circulating levels of estrogens through different mechanisms. In men and postmenopausal women, gut microbiota diversity and functions influence the levels of non-ovarian estrogens via enterohepatic circulation, where Clostridia taxa and genera from the Ruminococcaceae family could be key microbial populations involved in this regulation. | Gut microbiota may influence estrogen metabolism and signal in the brain. There are some neuroprotective effects of estrogens mediating some neuroplastic actions in the hippocampus and regulating serotonin receptors levels. | Gut dysbiosis may significantly impair the action of gut microbiota in the estrogen metabolism, driving to the reduction of circulating estrogens, which is associated with a plethora of adverse outcomes ranging from obesity, metabolic syndrome, malignancies, endometrial hyperplasia, endometriosis, polycystic ovary syndrome, fertility, cardiovascular disease, and cognitive dysfunction. The evidence supporting the role of altered sexual hormones in the pathogenesis of MDD in women could be found in the existence of three different types of depression related to the menstrual cycle: Premenstrual dysphoric disorder (PMDD), postpartum depression, and perimenopausal depression. The use of estradiol, but not progesterone appears to be effective for PMDD, postpartum, and moderate/severe perimenopausal depression. | Despite neuroprotective effects, the use of estradiol as the first line-treatment is not supported by current scientific evidence Instead, targeting the estrobolome in the prevention and treatment of these specific disorders could be an effective interesting approach, as women may benefit from the actions of these bacteria. Some studies have reported that lower levels of testosterone but estrogen were associated with more severe depressive symptoms in a group of women with MDD. Other reports allege that gut microbiota transplantation of male to female mice led to an augmentation of testosterone levels so future studies may consider the benefits from using testosterone-producer microorganisms to raise testosterone levels. As gut microbial diversity is directly related to the levels of non-ovaric estrogen levels and the intake of certain dietary components like polyphenols may aid in the modulation of beneficial microbial communities and ER activation, nutritional and lifestyle interventions may be a potential strategy for boosting the estrobolome. | [184,188,189,190,191,192,193,194,195] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortega, M.A.; Alvarez-Mon, M.A.; García-Montero, C.; Fraile-Martinez, O.; Guijarro, L.G.; Lahera, G.; Monserrat, J.; Valls, P.; Mora, F.; Rodríguez-Jiménez, R.; et al. Gut Microbiota Metabolites in Major Depressive Disorder—Deep Insights into Their Pathophysiological Role and Potential Translational Applications. Metabolites 2022, 12, 50. https://doi.org/10.3390/metabo12010050
Ortega MA, Alvarez-Mon MA, García-Montero C, Fraile-Martinez O, Guijarro LG, Lahera G, Monserrat J, Valls P, Mora F, Rodríguez-Jiménez R, et al. Gut Microbiota Metabolites in Major Depressive Disorder—Deep Insights into Their Pathophysiological Role and Potential Translational Applications. Metabolites. 2022; 12(1):50. https://doi.org/10.3390/metabo12010050
Chicago/Turabian StyleOrtega, Miguel A., Miguel Angel Alvarez-Mon, Cielo García-Montero, Oscar Fraile-Martinez, Luis G. Guijarro, Guillermo Lahera, Jorge Monserrat, Paula Valls, Fernando Mora, Roberto Rodríguez-Jiménez, and et al. 2022. "Gut Microbiota Metabolites in Major Depressive Disorder—Deep Insights into Their Pathophysiological Role and Potential Translational Applications" Metabolites 12, no. 1: 50. https://doi.org/10.3390/metabo12010050
APA StyleOrtega, M. A., Alvarez-Mon, M. A., García-Montero, C., Fraile-Martinez, O., Guijarro, L. G., Lahera, G., Monserrat, J., Valls, P., Mora, F., Rodríguez-Jiménez, R., Quintero, J., & Álvarez-Mon, M. (2022). Gut Microbiota Metabolites in Major Depressive Disorder—Deep Insights into Their Pathophysiological Role and Potential Translational Applications. Metabolites, 12(1), 50. https://doi.org/10.3390/metabo12010050












