Past Experiences for Future Applications of Metabolomics in Critically Ill Patients with Sepsis and Septic Shocks
Abstract
1. Introduction
2. The Role of Microbiota and Its Metabolites in the Development of Sepsis and Septic Shock
3. Metabolomics for Sepsis Diagnosis
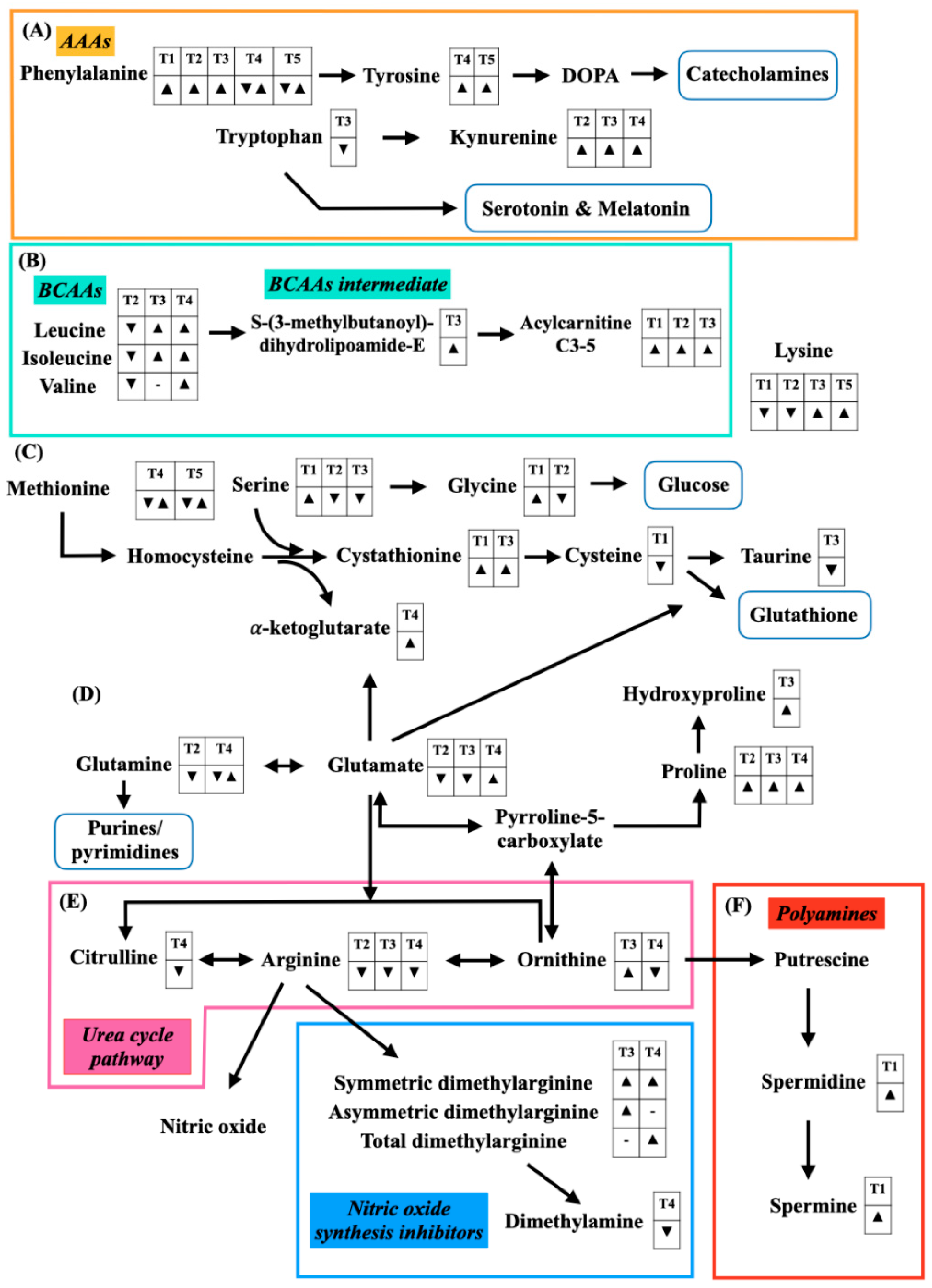
3.1. Alterations of Amino Acids and Amines in Sepsis
3.1.1. A Decrease in Cysteine and Lysine
3.1.2. An Increase in Glycine, Serine, Polyamines, and Amino Acid-Derived Acylcarnitines
3.2. Alterations of Fatty Acids and Their Related Metabolites in Sepsis
3.2.1. A Decrease of Eicosanoids
3.2.2. An Increase in Free Fatty Acids
3.2.3. An Alteration of Fatty Acid-Derived Acylcarnitines and Ceramides
3.3. Alterations of Phospholipids in Sepsis
3.3.1. A Decrease in Sphingomyelines and Lysophosphatidylcholines
3.3.2. An Increase in Cardiolipins
3.3.3. An Alteration of Phosphatidylcholines
4. Metabolomics for Septic Shock Diagnosis
4.1. Alterations of Amino Acids and Amines in Septic Shock
4.1.1. A Decrease in Branched-Chain Amino Acids, Glutamine, Glutamate, Arginine, and Proline
4.1.2. An Increase in Aromatic Amino Acids
4.2. Alterations of Glycolysis-Related Metabolites in Septic Shock
5. Metabolomics for Prognostication Patients with Sepsis
5.1. Alterations of Amino Acids and Amines in Sepsis Non-Survivors
5.1.1. A Decrease in Taurine, Tryptophan, Glutamate, Arginine, and Serine
5.1.2. An Increase in S-(3-Methyl-butanoyl)-dihydrolipoamide-E, Amino Acid-Derived Acylcarnitines, and Symmetric Dimethylarginine and Asymmetric Dimethylarginine
5.2. Alterations of Fatty Acids and Their Related Metabolites in Sepsis Non-Survivors
5.3. Alteration of Phospholipids in Sepsis Non-Survivors
5.4. Alterations of Glycolysis-Related Metabolites in Sepsis Non-Survivors
5.5. Alterations of Aromatic Microbial Metabolites in Sepsis Non-Survivors
6. Metabolomics for Prognostication Patients with Septic Shock
6.1. Alterations of Amino Acids and Amines in Septic Shock Non-Survivors
6.1.1. A Decrease in Dimethylamine and Citrulline
6.1.2. An Increase in Symmetric Dimethylarginine, Total Dimethylarginine, and Tyrosine
6.1.3. An Alternation of Phenylalanine and Methionine
6.2. Alterations of Fatty Acid-Related Metabolites in Septic Shock Non-Survivors
6.3. Alterations of Phospholipids in Septic Shock Non-Survivors
6.4. Alterations of Glycolysis-Related Metabolites and TCA Cycle Metabolites in Septic Shock Non-Survivors
7. Metabolomics for Monitoring Treatment Response in Sepsis and Septic Shock
8. Conclusions
9. Limitation and Future Direction of the Metabolomic Research in Sepsis
Author Contributions
Funding
Conflicts of Interest
References
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Vincent, J.-L.; Marshall, J.C.; A Ñamendys-Silva, S.A.; François, B.; Martin-Loeches, I.; Lipman, J.; Reinhart, K.; Antonelli, M.; Pickkers, P.; Njimi, H.; et al. Assessment of the worldwide burden of critical illness: The Intensive Care Over Nations (ICON) audit. Lancet Respir. Med. 2014, 2, 380–386. [Google Scholar] [CrossRef]
- Levy, M.M.; Evans, L.E.; Rhodes, A. The Surviving Sepsis Campaign Bundle: 2018 update. Intensiv. Care Med. 2018, 44, 925–928. [Google Scholar] [CrossRef] [PubMed]
- Zurfluh, S.; Baumgartner, T.; Meier, M.A.; Ottiger, M.; Voegeli, A.; Bernasconi, L.; Neyer, P.; Mueller, B.; Schuetz, P. The role of metabolomic markers for patients with infectious diseases: Implications for risk stratification and therapeutic modulation. Expert Rev. Anti-Infect. Ther. 2018, 16, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Serkova, N.J.; Standiford, T.J.; Stringer, K.A. The Emerging Field of Quantitative Blood Metabolomics for Biomarker Discovery in Critical Illnesses. Am. J. Respir. Crit. Care Med. 2011, 184, 647–655. [Google Scholar] [CrossRef]
- Carré, J.E.; Singer, M. Cellular energetic metabolism in sepsis: The need for a systems approach. Biochim. Biophys. Acta (BBA)—Bioenerg. 2008, 1777, 763–771. [Google Scholar] [CrossRef]
- Beloborodova, N.V.; Olenin, A.Y.; Pautova, A.K. Metabolomic findings in sepsis as a damage of host-microbial metabolism integration. J. Crit. Care 2018, 43, 246–255. [Google Scholar] [CrossRef] [PubMed]
- Drobnik, W.; Liebisch, G.; Audebert, F.-X.; Fröhlich, D.; Glück, T.; Vogel, P.; Rothe, G.; Schmitz, G. Plasma ceramide and lysophosphatidylcholine inversely correlate with mortality in sepsis patients. J. Lipid Res. 2003, 44, 754–761. [Google Scholar] [CrossRef]
- Schmerler, D.; Neugebauer, S.; Ludewig, K.; Bremer-Streck, S.; Brunkhorst, F.M.; Kiehntopf, M. Targeted metabolomics for discrimination of systemic inflammatory disorders in critically ill patients. J. Lipid Res. 2012, 53, 1369–1375. [Google Scholar] [CrossRef]
- Su, L.; Huang, Y.; Zhu, Y.; Xia, L.; Wang, R.; Xiao, K.; Wang, H.; Yan, P.; Wen, B.; Cao, L.; et al. Discrimination of sepsis stage metabolic profiles with an LC/MS-MS-based metabolomics approach. BMJ Open Respir. Res. 2014, 1, e000056. [Google Scholar] [CrossRef]
- Claussnitzer, M.; Dankel, S.N.; Kim, K.-H.; Quon, G.; Meuleman, W.; Haugen, C.; Glunk, V.; Sousa, I.S.; Beaudry, J.L.; Puviindran, V.; et al. FTO Obesity Variant Circuitry and Adipocyte Browning in Humans. N. Engl. J. Med. 2015, 373, 895–907. [Google Scholar] [CrossRef] [PubMed]
- Neugebauer, S.; Giamarellos-Bourboulis, E.J.; Pelekanou, A.; Marioli, A.; Baziaka, F.; Tsangaris, I.; Bauer, M.; Kiehntopf, M. Metabolite Profiles in Sepsis. Crit. Care Med. 2016, 44, 1649–1662. [Google Scholar] [CrossRef]
- Mecatti, G.C.; Messias, M.C.F.; Paiola, R.M.S.; Angolini, C.F.F.; Cunha, I.B.D.S.; Eberlin, M.N.; de Carvalho, P. Lipidomic Profiling of Plasma and Erythrocytes From Septic Patients Reveals Potential Biomarker Candidates. Biomark. Insights 2018, 13, 1177271918765137. [Google Scholar] [CrossRef]
- Brun, J.; A Gray, D. Targeting the ubiquitin proteasome pathway for the treatment of septic shock in patients. Crit. Care 2009, 13, 311. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M.; Chioléro, R.L. Antioxidant supplementation in sepsis and systemic inflammatory response syndrome. Crit. Care Med. 2007, 35, S584–S590. [Google Scholar] [CrossRef]
- Li, P.; Yin, Y.-L.; Li, D.; Kim, S.W.; Wu, G. Amino acids and immune function. Br. J. Nutr. 2007, 98, 237–252. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, W.; Han, D.; Meng, J.; Wang, H.; Cao, G. L–lysine ameliorates sepsis-induced acute lung injury in a lipopolysaccharide-induced mouse model. Biomed. Pharmacother. 2019, 118, 109307. [Google Scholar] [CrossRef]
- Mauriz, J.L.; Matilla, B.; Culebras, J.; González, P.; González-Gallego, J. Dietary glycine inhibits activation of nuclear factor kappa B and prevents liver injury in hemorrhagic shock in the rat. Free. Radic. Biol. Med. 2001, 31, 1236–1244. [Google Scholar] [CrossRef]
- Zhong, Z.; Wheeler, M.D.; Li, X.; Froh, M.; Schemmer, P.; Yin, M.; Bunzendaul, H.; Bradford, B.; Lemasters, J.J. l-Glycine: A novel antiinflammatory, immunomodulatory, and cytoprotective agent. Curr. Opin. Clin. Nutr. Metab. Care 2003, 6, 229–240. [Google Scholar] [CrossRef]
- Razak, M.A.; Begum, P.S.; Viswanath, B.; Rajagopal, S. Multifarious Beneficial Effect of Nonessential Amino Acid, Glycine: A Review. Oxidative Med. Cell. Longev. 2017, 2017, 1–8. [Google Scholar] [CrossRef]
- Rodriguez, A.E.; Ducker, G.S.; Billingham, L.K.; Martinez, C.A.; Mainolfi, N.; Suri, V.; Friedman, A.; Manfredi, M.G.; Weinberg, S.; Rabinowitz, J.D.; et al. Serine Metabolism Supports Macrophage IL-1β Production. Cell Metab. 2019, 29, 1003–1011.e4. [Google Scholar] [CrossRef]
- Ma, E.H.; Bantug, G.; Griss, T.; Condotta, S.; Johnson, R.M.; Samborska, B.; Mainolfi, N.; Suri, V.; Guak, H.; Balmer, M.L.; et al. Serine Is an Essential Metabolite for Effector T Cell Expansion. Cell Metab. 2017, 25, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Mickiewicz, B.; Duggan, G.E.; Winston, B.W.; Doig, C.; Kubes, P.; Vogel, H.J. Metabolic Profiling of Serum Samples by 1H Nuclear Magnetic Resonance Spectroscopy as a Potential Diagnostic Approach for Septic Shock. Crit. Care Med. 2014, 42, 1140–1149. [Google Scholar] [CrossRef]
- Firpo, M.R.; Mounce, B.C. Diverse Functions of Polyamines in Virus Infection. Biomol. 2020, 10, 628. [Google Scholar] [CrossRef] [PubMed]
- Moinard, C.; Cynober, L.; De Bandt, J.-P. Polyamines: Metabolism and implications in human diseases. Clin. Nutr. 2005, 24, 184–197. [Google Scholar] [CrossRef]
- Sagar, N.; Tarafdar, S.; Agarwal, S.; Tarafdar, A.; Sharma, S. Polyamines: Functions, Metabolism, and Role in Human Disease Management. Med. Sci. 2021, 9, 44. [Google Scholar] [CrossRef]
- Yoshida, M.; Kashiwagi, K.; Shigemasa, A.; Taniguchi, S.; Yamamoto, K.; Makinoshima, H.; Ishihama, A.; Igarashi, K. A Unifying Model for the Role of Polyamines in Bacterial Cell Growth, the Polyamine Modulon. J. Biol. Chem. 2004, 279, 46008–46013. [Google Scholar] [CrossRef]
- McCann, M.; De la Rosa, M.G.; Rosania, G.; Stringer, K. l-Carnitine and Acylcarnitines: Mitochondrial Biomarkers for Precision Medicine. Metabolites 2021, 11, 51. [Google Scholar] [CrossRef]
- Newgard, C.B. Interplay between Lipids and Branched-Chain Amino Acids in Development of Insulin Resistance. Cell Metab. 2012, 15, 606–614. [Google Scholar] [CrossRef]
- Marik, P.E.; Bellomo, R. Stress hyperglycemia: An essential survival response! Crit. Care 2013, 17, 305–307. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.L.; Goh, Y.Y. The role of group IIA secretory phospholipase A2 (sPLA2-IIA) as a biomarker for the diagnosis of sepsis and bacterial infection in adults—A systematic review. PLoS ONE 2017, 12, e0180554. [Google Scholar] [CrossRef]
- Van Wyngene, L.; Vandewalle, J.; Libert, C. Reprogramming of basic metabolic pathways in microbial sepsis: Therapeutic targets at last? EMBO Mol. Med. 2018, 10, e8712. [Google Scholar] [CrossRef] [PubMed]
- Delogu, G.; Famularo, G.; Amati, F.; Signore, L.; Antonucci, A.; Trinchieri, V.; Di Marzio, L.; Cifone, M.G. Ceramide concentrations in septic patients: A possible marker of multiple organ dysfunction syndrome. Crit. Care Med. 1999, 27, 2413–2417. [Google Scholar] [CrossRef] [PubMed]
- Opal, S.M. Endotoxins and Other Sepsis Triggers. Contrib. Nephrol. 2010, 167, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. The 2008 ESPEN Sir David Cuthbertson lecture: Fatty acids and inflammation—From the membrane to the nucleus and from the laboratory bench to the clinic. Clin. Nutr. 2010, 29, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Goñi, F.M.; Alonso, A. Sphingomyelinases: Enzymology and membrane activity. FEBS Lett. 2002, 531, 38–46. [Google Scholar] [CrossRef]
- Ahn, W.-G.; Jung, J.-S.; Kwon, H.Y.; Song, D.-K. Alteration of Lysophosphatidylcholine-Related Metabolic Parameters in the Plasma of Mice with Experimental Sepsis. Inflammation 2016, 40, 537–545. [Google Scholar] [CrossRef]
- Umezu-Goto, M.; Kishi, Y.; Taira, A.; Hama, K.; Dohmae, N.; Takio, K.; Yamori, T.; Mills, G.B.; Inoue, K.; Aoki, J.; et al. Autotaxin has lysophospholipase D activity leading to tumor cell growth and motility by lysophosphatidic acid production. J. Cell Biol. 2002, 158, 227–233. [Google Scholar] [CrossRef]
- Zou, C.; Synan, M.J.; Li, J.; Xiong, S.; Manni, M.L.; Liu, Y.; Chen, B.B.; Zhao, Y.; Shiva, S.; Tyurina, Y.Y.; et al. LPS impairs oxygen utilization in epithelia by triggering degradation of the mitochondrial enzyme Alcat1. J. Cell Sci. 2016, 129, 51–64. [Google Scholar] [CrossRef]
- Pizzuto, M.; Pelegrin, P. Cardiolipin in Immune Signaling and Cell Death. Trends Cell Biol. 2020, 30, 892–903. [Google Scholar] [CrossRef]
- Van Der Veen, J.N.; Kennelly, J.P.; Wan, S.; Vance, J.E.; Vance, D.E.; Jacobs, R.L. The critical role of phosphatidylcholine and phosphatidylethanolamine metabolism in health and disease. Biochim. et Biophys. Acta (BBA)—Biomembr. 2017, 1859, 1558–1572. [Google Scholar] [CrossRef] [PubMed]
- Mickiewicz, B.; Tam, P.; Jenne, C.N.; Leger, C.; Wong, J.; Winston, B.W.; Doig, C.; Kubes, P.; Vogel, H.J.; Alberta Sepsis Network. Integration of metabolic and inflammatory mediator profiles as a potential prognostic approach for septic shock in the intensive care unit. Crit. Care 2015, 19, 1–12. [Google Scholar] [CrossRef]
- HoleČek, M. Branched-chain amino acids in health and disease: Metabolism, alterations in blood plasma, and as supplements. Nutr. Metab. 2018, 15, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Mattick, J.; Kamisoglu, K.; Ierapetritou, M.G.; Androulakis, I.P.; Berthiaume, F. Branched-chain amino acid supplementation: Impact on signaling and relevance to critical illness. Wiley Interdiscip. Rev. Syst. Biol. Med. 2013, 5, 449–460. [Google Scholar] [CrossRef]
- Tapiero, H.; Mathé, G.; Couvreur, P.; Tew, K., II. Glutamine and glutamate. Biomed. Pharmacother. 2002, 56, 446–457. [Google Scholar] [CrossRef]
- Poeze, M.; Luiking, Y.C.; Breedveld, P.; Manders, S.; Deutz, N.E. Decreased plasma glutamate in early phases of septic shock with acute liver dysfunction is an independent predictor of survival. Clin. Nutr. 2008, 27, 523–530. [Google Scholar] [CrossRef]
- Machado, M.C.C.; Da Silva, F.P. Hyperammonemia due to urea cycle disorders: A potentially fatal condition in the intensive care setting. J. Intensiv. Care 2014, 2, 22. [Google Scholar] [CrossRef]
- Straaten, H.M.O.-V.; van Zanten, A.R. Glutamine supplementation in the critically ill: Friend or foe? Crit. Care 2014, 18, 143. [Google Scholar] [CrossRef] [PubMed]
- Luiking, Y.C.; Poeze, M.; Ramsay, G.; Deutz, N.E. Reduced citrulline production in sepsis is related to diminished de novo arginine and nitric oxide production. Am. J. Clin. Nutr. 2008, 89, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, M.D.; Carrott, P.W.; Patel, J.; Kiraly, L.; Martindale, R.G. Parenteral or Enteral Arginine Supplementation Safety and Efficacy. J. Nutr. 2016, 146, 2594S–2600S. [Google Scholar] [CrossRef]
- Marik, P.E. Arginine: Too much of a good thing may be bad! Crit. Care Med. 2006, 34, 2844–2847. [Google Scholar] [CrossRef]
- Heyland, D.K.; Novak, F.; Drover, J.W.; Jain, M.; Su, X.; Suchner, U. Should Immunonutrition Become Routine in Critically Ill Patients? JAMA 2001, 286, 944–953. [Google Scholar] [CrossRef]
- Otto, G.P.; Neugebauer, S.; A Claus, R.; Sossdorf, M. Arginine metabolism is markedly impaired in polymicrobial infected mice. Crit. Care 2012, 16, 412. [Google Scholar] [CrossRef]
- Druml, W.; Heinzel, G.; Kleinberger, G. Amino acid kinetics in patients with sepsis. Am. J. Clin. Nutr. 2001, 73, 908–913. [Google Scholar] [CrossRef] [PubMed]
- Ploder, M.; Neurauter, G.; Spittler, A.; Schroecksnadel, K.; Roth, E.; Fuchs, D. Serum phenylalanine in patients post trauma and with sepsis correlate to neopterin concentrations. Amino Acids 2007, 35, 303–307. [Google Scholar] [CrossRef]
- Pastor, C.M.; Williams, D.; Yoneyama, T.; Hatakeyama, K.; Singleton, S.; Naylor, E.; Billiar, T.R. Competition for Tetrahydrobiopterin between Phenylalanine Hydroxylase and Nitric Oxide Synthase in Rat Liver. J. Biol. Chem. 1996, 271, 24534–24538. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Meininger, C.J.; Haynes, T.E.; Hatakeyama, K.; Wu, G. Regulation of Tetrahydrobiopterin Synthesis and Bioavailability in Endothelial Cells. Cell Biophys. 2004, 41, 415–434. [Google Scholar] [CrossRef]
- Schroeder, M.A.; Atherton, H.J.; Dodd, M.; Lee, P.; Cochlin, L.E.; Radda, G.K.; Clarke, K.; Tyler, D. The Cycling of Acetyl-Coenzyme A Through Acetylcarnitine Buffers Cardiac Substrate Supply. Circ. Cardiovasc. Imaging 2012, 5, 201–209. [Google Scholar] [CrossRef]
- Jennaro, T.S.; Puskarich, M.; McCann, M.R.; Gillies, C.E.; Pai, M.P.; Karnovsky, A.; Evans, C.R.; Jones, A.E.; Stringer, K.A. Using l-Carnitine as a Pharmacologic Probe of the Interpatient and Metabolic Variability of Sepsis. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2020, 40, 913–923. [Google Scholar] [CrossRef]
- Dalli, J.; Colas, R.A.; Quintana, C.; Barragan-Bradford, D.; Hurwitz, S.; Levy, B.D.; Choi, A.M.; Serhan, C.N.; Baron, R.M. Human Sepsis Eicosanoid and Proresolving Lipid Mediator Temporal Profiles. Crit. Care Med. 2017, 45, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Langley, R.J.; Tsalik, E.L.; van Velkinburgh, J.C.; Glickman, S.W.; Rice, B.J.; Wang, C.; Chen, B.; Carin, L.; Suarez, A.; Mohney, R.P.; et al. An Integrated Clinico-Metabolomic Model Improves Prediction of Death in Sepsis. Sci. Transl. Med. 2013, 5, 195ra95. [Google Scholar] [CrossRef]
- Rogers, A.; McGeachie, M.; Baron, R.M.; Gazourian, L.; Haspel, J.A.; Nakahira, K.; Fredenburgh, L.E.; Hunninghake, G.M.; Raby, B.A.; Matthay, M.A.; et al. Metabolomic Derangements Are Associated with Mortality in Critically Ill Adult Patients. PLoS ONE 2014, 9, e87538. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.-P.; Chen, G.-Y.; Chuang, T.-Y.; Huang, Y.-T.; Chang, H.-T.; Chen, Y.-F.; Liu, W.-L.; Chen, Y.-J.; Hsu, C.-L.; Huang, M.-T.; et al. Increased Plasma Acetylcarnitine in Sepsis Is Associated With Multiple Organ Dysfunction and Mortality. Crit. Care Med. 2019, 47, 210–218. [Google Scholar] [CrossRef]
- Wang, J.; Sun, Y.; Teng, S.; Li, K. Prediction of sepsis mortality using metabolite biomarkers in the blood: A meta-analysis of death-related pathways and prospective validation. BMC Med. 2020, 18, 1–15. [Google Scholar] [CrossRef]
- Huang, S.-S.; Lin, J.-Y.; Chen, W.-S.; Liu, M.-H.; Cheng, C.-W.; Cheng, M.-L.; Wang, C.-H. Phenylalanine- and leucine-defined metabolic types identify high mortality risk in patients with severe infection. Int. J. Infect. Dis. 2019, 85, 143–149. [Google Scholar] [CrossRef]
- Winkler, M.S.; Nierhaus, A.; Rösler, G.; Lezius, S.; Harlandt, O.; Schwedhelm, E.; Böger, R.H.; Kluge, S. Symmetrical (SDMA) and asymmetrical dimethylarginine (ADMA) in sepsis: High plasma levels as combined risk markers for sepsis survival. Crit. Care 2018, 22, 216. [Google Scholar] [CrossRef]
- Ekremoğlu, M.; Türközkan, N.; Erdamar, H.; Kurt, Y.; Yaman, H. Protective effect of taurine on respiratory burst activity of polymorphonuclear leukocytes in endotoxemia. Amino Acids 2006, 32, 413–417. [Google Scholar] [CrossRef]
- Chiarla, C.; Giovannini, I.; Siegel, J.H.; Boldrini, G.; Castagneto, M. The Relationship between Plasma Taurine and Other Amino Acid Levels in Human Sepsis. J. Nutr. 2000, 130, 2222–2227. [Google Scholar] [CrossRef] [PubMed]
- Elmokadem, E.M.; Sabri, N.A.; Roshdy, T.A.; Hasanin, A.M. Clinical outcomes study of critically-ill septic patients given taurine supplemented enteral nutrition. Int. J. Pharm. Sci. Res. 2015, 6, 3544–3553. [Google Scholar] [CrossRef]
- Ploder, M.; Spittler, A.; Schroecksnadel, K.; Neurauter, G.; Pelinka, L.E.; Roth, E.; Fuchs, D. Accelerated Tryptophan Degradation in Trauma and Sepsis Patients is Related to Pro-inflammatory Response and to the Diminished in vitro Response of Monocytes. Pteridines 2009, 20, 54–61. [Google Scholar] [CrossRef]
- Changsirivathanathamrong, D.; Wang, Y.; Rajbhandari, D.; Maghzal, G.J.; Mak, W.M.; Woolfe, C.; Duflou, J.; Gebski, V.; dos Remedios, C.G.; Celermajer, D.S.; et al. Tryptophan metabolism to kynurenine is a potential novel contributor to hypotension in human sepsis. Crit. Care Med. 2011, 39, 2678–2683. [Google Scholar] [CrossRef] [PubMed]
- Zulpaite, R.; Miknevicius, P.; Leber, B.; Strupas, K.; Stiegler, P.; Schemmer, P. Tryptophan Metabolism via Kynurenine Pathway: Role in Solid Organ Transplantation. Int. J. Mol. Sci. 2021, 22, 1921. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, T.A.; Nguyen, J.C.D.; Polglaze, K.E.; Bertrand, P.P. Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis. Nutrients 2016, 8, 56. [Google Scholar] [CrossRef]
- Telias, I.; Wilcox, M.E. Sleep and Circadian Rhythm in Critical Illness. Crit. Care 2019, 23, 1–8. [Google Scholar] [CrossRef]
- Panyushkina, A.; Matyushkina, D.; Pobeguts, O. Understanding Stress Response to High-Arsenic Gold-Bearing Sulfide Concentrate in Extremely Metal-Resistant Acidophile Sulfobacillus thermotolerans. Microorganisms 2020, 8, 1076. [Google Scholar] [CrossRef]
- Lambden, S. Bench to bedside review: Therapeutic modulation of nitric oxide in sepsis—An update. Intensiv. Care Med. Exp. 2019, 7, 1–14. [Google Scholar] [CrossRef]
- Böger, R.H. The emerging role of asymmetric dimethylarginine as a novel cardiovascular risk factor. Cardiovasc. Res. 2003, 59, 824–833. [Google Scholar] [CrossRef]
- Yoshikai, Y. Roles of prostaglandins and leukotrienes in acute inflammation caused by bacterial infection. Curr. Opin. Infect. Dis. 2001, 14, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Aronoff, D.M. Cyclooxygenase Inhibition in Sepsis: Is There Life after Death? Mediat. Inflamm. 2012, 2012, 1–7. [Google Scholar] [CrossRef]
- Bindu, S.; Mazumder, S.; Bandyopadhyay, U. Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: A current perspective. Biochem. Pharmacol. 2020, 180, 114147. [Google Scholar] [CrossRef]
- Srisawat, N.; Kulvichit, W.; Mahamitra, N.; Hurst, C.; Praditpornsilpa, K.; Lumlertgul, N.; Chuasuwan, A.; Trongtrakul, K.; Tasnarong, A.; Champunot, R.; et al. The epidemiology and characteristics of acute kidney injury in the Southeast Asia intensive care unit: A prospective multicentre study. Nephrol. Dial. Transplant. 2020, 35, 1729–1738. [Google Scholar] [CrossRef]
- Trongtrakul, K.; Sawawiboon, C.; Wang, Y.; Chitsomkasem, A.; Limphunudom, P.; Kurathong, S.; Prommool, S.; Trakarnvanich, T.; Srisawat, N. Acute kidney injury in critically ill surgical patients: Epidemiology, risk factors and outcomes. Nephrology 2017, 24, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.; Hoppel, C.L.; Lok, K.H.; Zhao, L.; Wong, S.W.; Minkler, P.E.; Hwang, D.H.; Newman, J.; Garvey, W.T. Plasma Acylcarnitine Profiles Suggest Incomplete Long-Chain Fatty Acid β-Oxidation and Altered Tricarboxylic Acid Cycle Activity in Type 2 Diabetic African-American Women. J. Nutr. 2009, 139, 1073–1081. [Google Scholar] [CrossRef]
- Garcia-Alvarez, M.; Marik, P.; Bellomo, R. Sepsis-associated hyperlactatemia. Crit. Care 2014, 18, 1–11. [Google Scholar] [CrossRef]
- Beloborodova, N.; Sarshor, Y.N.; Bedova, A.Y.; Chernevskaya, E.; Pautova, A.K. Involvement of Aromatic Metabolites in the Pathogenesis of Septic Shock. Shock 2018, 50, 273–279. [Google Scholar] [CrossRef]
- Liu, Z.; Triba, M.N.; Amathieu, R.; Lin, X.; Bouchemal, N.; Hantz, E.; Le Moyec, L.; Savarin, P. Nuclear magnetic resonance-based serum metabolomic analysis reveals different disease evolution profiles between septic shock survivors and non-survivors. Crit. Care 2019, 23, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yin, P.; Amathieu, R.; Savarin, P.; Xu, G. Application of LC-MS-based metabolomics method in differentiating septic survivors from non-survivors. Anal. Bioanal. Chem. 2016, 408, 7641–7649. [Google Scholar] [CrossRef]
- Ferrario, M.; Cambiaghi, A.; Brunelli, L.; Giordano, S.; Caironi, P.; Guatteri, L.; Raimondi, F.; Gattinoni, L.; Latini, R.; Masson, S.; et al. Mortality prediction in patients with severe septic shock: A pilot study using a target metabolomics approach. Sci. Rep. 2016, 6, 20391. [Google Scholar] [CrossRef] [PubMed]
- Cambiaghi, A.; Díaz, R.; Martinez, J.B.; Odena, A.; Brunelli, L.; Caironi, P.; Masson, S.; Baselli, G.; Ristagno, G.; Gattinoni, L.; et al. An Innovative Approach for The Integration of Proteomics and Metabolomics Data In Severe Septic Shock Patients Stratified for Mortality. Sci. Rep. 2018, 8, 6681. [Google Scholar] [CrossRef]
- Garcia-Simon, M.; Morales, J.M.; Modesto-Alapont, V.; Gonzalez-Marrachelli, V.; Vento-Rehues, R.; Jorda-Miñana, A.; Blanquer-Olivas, J.; Monleon, D. Prognosis Biomarkers of Severe Sepsis and Septic Shock by 1H NMR Urine Metabolomics in the Intensive Care Unit. PLoS ONE 2015, 10, e0140993. [Google Scholar] [CrossRef]
- Evans, C.R.; Karnovsky, A.; Puskarich, M.; Michailidis, G.; Jones, A.E.; Stringer, K.A. Untargeted Metabolomics Differentiates l-Carnitine Treated Septic Shock 1-Year Survivors and Nonsurvivors. J. Proteome Res. 2019, 18, 2004–2011. [Google Scholar] [CrossRef] [PubMed]
- Tsikas, D. Urinary Dimethylamine (DMA) and Its Precursor Asymmetric Dimethylarginine (ADMA) in Clinical Medicine, in the Context of Nitric Oxide (NO) and Beyond. J. Clin. Med. 2020, 9, 1843. [Google Scholar] [CrossRef]
- Wexler, O.; Gough, M.S.; Morgan, M.A.M.; Mack, C.M.; Apostolakos, M.J.; Doolin, K.P.; Mooney, R.A.; Arning, E.; Bottiglieri, T.; Pietropaoli, A.P. Methionine Metabolites in Patients With Sepsis. J. Intensiv. Care Med. 2016, 33, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Tvrzicka, E.; Kremmyda, L.-S.; Stankova, B.; Zak, A. Fatty acids as biocompounds: Their role in human metabolism, health and disease—a review. Part 1: Classification, dietary sources and biological functions. Biomed. Pap. 2011, 155, 117–130. [Google Scholar] [CrossRef]
- Nuzzo, E.; Berg, K.M.; Andersen, L.W.; Balkema, J.; Montissol, S.; Cocchi, M.N.; Liu, X.; Donnino, M.W. Pyruvate Dehydrogenase Activity Is Decreased in the Peripheral Blood Mononuclear Cells of Patients with Sepsis. A Prospective Observational Trial. Ann. Am. Thorac. Soc. 2015, 12, 1662–1666. [Google Scholar] [CrossRef]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial Reactive Oxygen Species (ROS) and ROS-Induced ROS Release. Physiol. Rev. 2014, 94, 909–950. [Google Scholar] [CrossRef]
- Martínez-Reyes, I.; Chandel, N.S. Mitochondrial TCA cycle metabolites control physiology and disease. Nat. Commun. 2020, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Galley, H.F. Oxidative stress and mitochondrial dysfunction in sepsis. Br. J. Anaesth. 2011, 107, 57–64. [Google Scholar] [CrossRef]
- Puskarich, M.A.; Finkel, M.A.; Karnovsky, A.; Jones, A.E.; Trexel, J.; Harris, B.N.; Stringer, K.A. Pharmacometabolomics ofl-Carnitine Treatment Response Phenotypes in Patients with Septic Shock. Ann. Am. Thorac. Soc. 2015, 12, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Cambiaghi, A.; Pinto, B.B.; Brunelli, L.; Falcetta, F.; Aletti, F.; Bendjelid, K.; Pastorelli, R.; Ferrario, M. Characterization of a metabolomic profile associated with responsiveness to therapy in the acute phase of septic shock. Sci. Rep. 2017, 7, 1–16. [Google Scholar] [CrossRef]
- Ruggieri, A.J.; Levy, R.J.; Deutschman, C.S. Mitochondrial Dysfunction and Resuscitation in Sepsis. Crit. Care Clin. 2010, 26, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Maslove, D.M.; Wong, H.R. Gene expression profiling in sepsis: Timing, tissue, and translational considerations. Trends Mol. Med. 2014, 20, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Robinson, R.A.S. The role of proteomics in understanding biological mechanisms of sepsis. Proteom.—Clin. Appl. 2013, 8, 35–52. [Google Scholar] [CrossRef] [PubMed]



| Age (Sample Size) | APACHE-II Score | Samples Since Admission | Methods | Major Findings in Sepsis Group | Interpretation | Citation | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Serum | Plasma | Others | Metabolic Pathways | Decreased | Increased | |||||
| N/A (102) vs. N/A (56) | N/A vs. N/A | ✓ D1 | Targeted (LC-MS/MS) | At D1 | Patients with sepsis had increased ceramides, but decreased phospholipids when compared to patients without sepsis | [8] | ||||
| Fatty acids |
|
| ||||||||
| Phospholipids |
| |||||||||
| T: 64 ± 11 (30) vs. 67 ± 10 (33) V: 64 ± 15 (39) vs. 67 ± 10 (41) | 23 ± 8 vs. 18 ± 7 26 ± 9 vs. 19 ± 7 | ✓ within 24 h/ at onset of SIRS | Targeted (LC-MS/MS) | Fatty acids | - |
| Fatty acids and phospholipids are potential markers for discriminating sepsis from SIRS | [9] | ||
| Phospholipids | - |
| ||||||||
| 64 ± 17 (35) vs. 59 ± 19 (15) | 22 ± 7 vs. 11 ± 9 | ✓ within 24 h | Targeted (LC-MS/MS) | Amino acids and amine |
|
| Amino acids and lactitol dihydrate could differentiate sepsis from SIRS | [10] | ||
| Others |
|
| ||||||||
| 57 ± 22 (35) vs. 47 ± 13 (14) | 18 ± 8 vs. 9 ± 3 | ✓ within 24 h | Targeted (LC-MS/MS) | Amino acids and amine |
|
| Critically ill patients with sepsis had a wide range of amino acid spectral changes that differ from SIRS | [11] | ||
| T: 70 ± 17 (123) vs. 63 ± 16 (42) V: 64 ± 24 (59) vs. 60 ± 18 (2) | 11 ± 6 vs. 11 ± 8 13 ± 11 vs. 9 ± 6 | ✓ within 24 h | Targeted (LC-MS/MS) | Amino acids and amine | - |
| Amino acids, fatty acids, and phospholipids can potentially be used as sepsis biomarkers | [12] | ||
| Fatty acids |
|
| ||||||||
| Phospholipids |
|
| ||||||||
| 56 ± 18 (20) vs. 58 ± 11 (20) | 15 ± 6 vs. N/A | ✓ within 36 h | Targeted (LC-MS and GC-MS) | Phospholipids |
C16:0/20:1, C16:0/20:3
|
C16:0/18:2, C16:0/20:5 | Fatty acids and phospholipids detected in plasma and erythrocytes could signal sepsis vs. non-sepsis | [13] | ||
| Erythrocytes ✓ within 36 h | GC-MS | Fatty acids |
DHA (C22:6 n-3) |
| ||||||
| Phospholipids |
|
C16:0/20:4, C16:0/20:5
| ||||||||
| Age (Age Range) (Sample Size) | APACH-II Score | Samples Since Admission | Methods | Major Findings in Septic Shock Groups | Interpretation | Citation | |||
|---|---|---|---|---|---|---|---|---|---|
| Serum | Plasma | Metabolic Pathways | Decreased | Increased | |||||
| 62 (55–73) (39) vs. 66 (56–71) (20) | 23 (16–31) vs. 14 (13–17) | ✓ within 24 h | Targeted (1H-NMRS) | Amino acids and amines |
|
| Septic shock patients had different patterns in amino acids, fatty acids, and TCA cycle metabolites | [23] | |
| Fatty acids | - |
| |||||||
| Glycolysis |
|
| |||||||
| TCA cycle | - |
| |||||||
| 62 (56–73) (37) vs. 66 (56–71) (20) | 23 (16–31) vs. 14 (13–17) | ✓ within 24 h | ✓ within 24 h | Targeted (1H- NMRS) | Amino acids and amines |
|
| Septic shock patients had different patterns of metabolites, particularly amino acids | [42] |
| Glycolysis |
|
| |||||||
| Settings | Age (Age Range) (Sample Size) | APACHE-II Score | Samples Since Admission | Methods | Major Findings in Non-Survivors | Interpretation | Citation | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Serum | Plasma | Blood | Metabolic Pathways | Decreased | Increased | ||||||
| 48-H mortality | 67 ± 15 (9) vs. 63 ± 18 (26) | 26 ± 6 vs. 20 ± 8 | ✓ within 48-H before death | Targeted (LC-MS/MS) | Amino acids and amines | - |
| Amino acids and phospholipids could indicate the possibility of death within 48-H in patients with sepsis | [10] | ||
| Phospholipids | - |
| |||||||||
| 7-D mortality | 60 (36–80) (9) vs. 60 (27–84) (13) | 31 (16–46) vs. 22 (14–38) | ✓ within 48 h = D1, D3, and D7 | Targeted (LC-MS/MS) | Persisted D1 to D7 | Fatty acids and proresolving lipids signal 7-D mortality in critically-ill patients with sepsis | [60] | ||||
| Fatty acids | - |
| |||||||||
| At D1 | |||||||||||
| Fatty acids | - |
| |||||||||
| - |
| ||||||||||
| At D3 | |||||||||||
| Fatty acids | - |
| |||||||||
| - |
| ||||||||||
| At D7 | |||||||||||
| Fatty acids | - |
| |||||||||
| - |
| ||||||||||
| 28-D mortality | 69 ± 17 (31) vs. 56 ± 19 (90) | 23 ± 8 vs. 15 ± 7 | ✓ H0 and H24 | ✓ H0 and H24 | - Untargeted (UPLC-MS/MS and GC-MS) - Targeted (UPLC-MS/MS) | Persisted At H0 to H24 | 28-D mortality could be predicted by several amino acids, amines, fatty acids, and glycolysis metabolites | [61] | |||
| Amino acids and amines | - |
| |||||||||
| Fatty acids | - |
| |||||||||
| Glycolysis | - |
| |||||||||
| TCA cycle | - |
| |||||||||
| At H0 | |||||||||||
| Fatty acids | - |
| |||||||||
| At H24 | |||||||||||
| Amino acids and amines | - |
| |||||||||
| Fatty acids | - |
| |||||||||
| Phospholipids |
| - | |||||||||
| 28-D mortality | T: 58 ± 15 (30) vs. 53 ± 14 (60) V: 69 ± 16 (34) vs. 58 ± 17 (115) | 30 ± 11 vs. 23 ± 9 23 ± 8 vs. 15 ± 7 | ✓ H0 | Targeted (GC-MS and LC-MS) | Amino acids and amines | - |
| Non-surviving 28-D sepsis patients had specific changes in amino acids, fatty acids, glycolysis, and bile acids’ metabolic pathways, as well as an increase in aromatic microbial metabolites | [62] | ||
| Fatty acids | - |
| |||||||||
| Phospholipids |
| - | |||||||||
| Glycolysis Aromatic microbial metabolites | - - |
| |||||||||
| 28-D mortality | 61 ± 21 (15) vs. 54 ± 23 (20) | 22 ± 8 vs. 10 ± 5 | ✓ within 24H = D1, D3, D5, D7, D10, and D14 | Targeted (LC-MS/MS) | At certain time points | Amino acids could indicate the possibility of death in septic patients | [11] | ||||
| Amino acids and amines |
|
| |||||||||
| 28-D mortality | 68 (51–75) (31) vs. 63 (53–74) (89) | 12 (8–9) vs. 9 (6–13) | ✓ within 24H = D1, D3, D7 | Targeted (LC-MS/MS) | Amino acids and amines |
| High level of plasma SDMA and ADMA can predict sepsis non-survival | [66] | |||
| 28-D mortality | 70 ± 13 (21) vs. 72 ± 15 (69) | 26 ± 9 vs. 23 ± 8 | ✓ H0 | Targeted (UHPLC-MS) | Glycolysis | - |
| Acetylcarnitine can forecast 28-D mortality in patients with sepsis | [63] | ||
| 28-D mortality | 67 ± 14 (54) vs. 62 ± 19 (134) | 22 (18–30) vs. 18 (13–24) | ✓ H0 | Targeted (LC-MS) | Amino acids and amines | - |
| Particular metabolites can forecast 28-D mortality in sepsis patients | [64] | ||
| Phospholipids |
| - | |||||||||
| Glycolysis | - |
| |||||||||
| 30-D mortality | 55 (17–80) (39) vs. 54 (20–91) (63) | N/A vs. N/A | ✓ D1, D4, and D11 | Targeted (LC-MS/MS) Lipids | Persisted along D1 to D11 | [8] | |||||
| Fatty acids and phospholipids | - |
| |||||||||
| 90-D mortality | 75 ± 13 (30) vs. 71 ± 13 (63) | 9 ± 4 $ vs. 5 ± 4 $ | ✓ D1 | Targeted (UPLC-MS) | Amino acids and amine | - |
| In sepsis patients, 90-D mortality can be expected by phenylalanine and leucine | [65] | ||
| Settings | Age (Age Range) (Sample Size) | APACHE-II Score | Samples Since Admission | Methods | Major Findings in Non-Survivors (NS) | Interpretation | Citation | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Serum | Plasma | Urine | Metabolic Pathways | Decreased | Increased | ||||||
| ICU mortality | 63 (60–77) * (8) * | 26 (18–31) * | ✓ within 24 h | ✓ within 24 h | Targeted (1H-NMRS) | Amino acids and amines |
| - | Non-survivors in septic shock had high levels of 2-Hydrocyiso-valerate and fructose | [42] | |
| Fatty acids | - |
| |||||||||
| Glycolysis | - |
| |||||||||
| 24-H mortality | 72 ± 0.4 (30) vs. 69 ± 0.3 (40) | 12 ± 0.6 $ vs. 11 ± 0.7 $ | ✓ H0 and H24 of vaso-pressor initiation | Targeted (1H-NMRS) | At H0; | Non-surviving patients with 24-H septic shock can be forecasted by amino acids, TCA cycle metabolites, and fatty acids pathways | [86] | ||||
| Amino acids and amines | - |
| |||||||||
| Glycolysis | - |
| |||||||||
| TCA cycle | - |
| |||||||||
| At H24; | |||||||||||
| Amino acids and amines | - |
| |||||||||
| Fatty acids | - |
| |||||||||
| Glycolysis | - |
| |||||||||
| TCA cycle | - |
| |||||||||
| ∆H24-H0 in nonsurvivors; | |||||||||||
| Amino acids and amines | - |
| |||||||||
| Glycolysis | - |
| |||||||||
| TCA cycle | - |
| |||||||||
| Others |
|
| |||||||||
| ∆H24-H0 in survivors; | |||||||||||
| Amino acids and amines |
| - | |||||||||
| Glycolysis |
| - | |||||||||
| TCA cycle |
| - | |||||||||
| 7-D mortality | 66 ± 1 (21) vs. 64 ± 1 (29) | 68 ± 2 # vs. 54 ± 2 # | ✓ H0 | Untargeted (UPLC-MS) | Amino acids and amines |
|
| Non-surviving 7-D septic shock patients demonstrated several precise metabolomics signals from amino acids, TCA cycle, fatty acids, and phospholipids pathways | [87] | ||
| Fatty acids |
|
| |||||||||
| Phospholipids |
|
| |||||||||
| Glycolysis |
|
| |||||||||
| TCA cycle | - |
| |||||||||
| 28-D mortality | 70 ± 12 (11) vs. 61 ± 15 (9) | 12 ± 2 vs. 11 ± 2 | ✓ D1 and D7 | Targeted (LC-MS/MS) | At D1 | Long chain PC and LysoPC metabolites had predictive capability for 28-D mortality patients in septic shock | [88] | ||||
| Phospholipids |
|
| |||||||||
| Glycolysis |
| - | |||||||||
| At D7 | |||||||||||
| Amino acids and amines | - |
| |||||||||
| Phospholipids |
| - | |||||||||
| ∆D7-D1 comparing between NS vs. S | |||||||||||
| ↔ vs. ↓ | |||||||||||
| Amino acids and amines |
| - | |||||||||
| ↔ vs. ↑ | ↑ vs. ↑↑ | ||||||||||
| Phospholipids |
|
| |||||||||
| ↓vs. ↔ | |||||||||||
| Phospholipids |
| - | |||||||||
| 28-D mortality | 64 ± 17 (8) vs. 66 ± 14 (9) | D1: 12 ± 3 $ vs. 11 ± 2 $ D7: 9 ± 5 $ vs. 5 ± 2$ | ✓ at Shock Dx | Targeted (LC-MS/MS) | Crude ratio of D7/D1 | The ratios of particular amino acids and phospholipids can determine 28-D mortality in septic shock patients | [89] | ||||
| Amino acids and amines | - |
| |||||||||
| Phospholipids |
|
| |||||||||
| Ratio of D7/D1 discriminated by multivariate analysis | |||||||||||
| Amino acids and amines |
|
| |||||||||
| Phospholipids |
|
| |||||||||
| 30-D mortality | 65 (37–79) (12) vs. 60 (24–80) (48) | 21 ± 5 vs. 19 ± 6 | ✓ H0 and H24 | Untargeted (1H-NMRS) | Amino acids and amines |
| - | Particular amino acids, glycolytic metabolites, and alcohol can predict 30-D mortality in septic shock patients | [90] | ||
| Glycolysis | - |
| |||||||||
| 90-D mortality | 70 ± 12 (11) vs. 61 ± 15 (9) | 12 ± 2 vs. 11 ± 2 | ✓ D1 and D7 | Targeted (LC-MS/MS) | At D1 | Long chain PC and LysoPC metabolites had predictive capability for 90-D mortality in septic shock patients | [88] | ||||
| Phospholipids |
| - | |||||||||
| At D7 | |||||||||||
| Phospholipids |
|
| |||||||||
| ∆D7-D1 comparing between NS vs. S | |||||||||||
| ↔ vs. ↓ | |||||||||||
| Amino acids and amines |
| - | |||||||||
| ↔ vs. ↑ | ↑ vs. ↑↑ | ||||||||||
| Phospholipids |
|
| |||||||||
| ↔ (↓) vs. ↔ (↑) | |||||||||||
| Phospholipids |
| - | |||||||||
| 1-Y mortality | 69 (61–77) (4) vs. 58 (50–65) (7) | 15 (14–17) $ vs. 14 (9–14) $ | ✓ H0, H24, and H48 after l-carnitine infusion | Untargeted (LC-MS) | Amino acids and amines |
|
| 1-Y mortality in septic shock patients can be determined by certain amino acids, fatty acids, and peptide/short chain proteins | [91] | ||
| Fatty acids | - |
| |||||||||
| Settings | Studies Groups | Age (Age Range) (Sample Size) | APACHE- II Score | Samples | Methods | Major Findings in Responder Groups | Interpretation | Citation | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Serum | Plasma | Metabolic Pathway | Decreased | Increased | |||||||
| l-carnitine responders vs. placebo in septic shock patients treated with vasopressor | Low ketones vs. High ketones categorized by 3-hydroxy-butyrate (cut-off= 153 μM) | 60 (52–68) (15) vs. 69 (60–74) (15) | 10 (9–14) $ vs. 13 (8–14) $ | ✓ H0, H24, and H48 after l-carnitine infusion | Untargeted (1H-NMRS) | At H24 | Pharmacometa-bolomics can be used to guide responses to l-carnitine treatment | [99] | |||
| Amino acids and amines | - |
| |||||||||
| Fatty acids |
| - | |||||||||
| Glycolysis |
| - | |||||||||
| At H48 | |||||||||||
| Fatty acids |
| - | |||||||||
| Glycolysis |
| - | |||||||||
| Characterized response to therapy in patients with septic shock | Response (R) vs. Non-response (NR) to therapy | 67 (61–75) (14) vs. 75 (66–82) (7) | 35 (31–38) vs. 38 (37–39) | ✓ H0 and H48 after resus-citation | Untargeted (LC-MS/MS) | At H0, R vs. NR | Metabolomics from particular pathways including amino acids, fatty acids, phospholipids, and TCA cycle had potential roles for treatment monitoring in patients with septic shock | [100] | |||
| Amino acids and amines |
| - | |||||||||
| Fatty acids | - |
| |||||||||
| Glycolysis |
| - | |||||||||
| In R, H48 vs. H0; | |||||||||||
| Amino acids and amines |
|
| |||||||||
| Fatty acids |
| - | |||||||||
| TCA cycle |
| - | |||||||||
| In NR, H48 vs. H0; | |||||||||||
| Amino acids and amines | - |
| |||||||||
| Fatty acids |
| - | |||||||||
| Comparing R vs. NR overtime | |||||||||||
| ↓ vs. ↓↓ | ↓ vs. ↑ | ||||||||||
| Fatty acids |
| ||||||||||
| Targeted (LC-MS) | In R, H48 vs. H0; | ||||||||||
| Amino acids and amines |
|
| |||||||||
| Phospholipids | - |
| |||||||||
| In NR, H48 vs. H0; | |||||||||||
| Amino acids and amines |
|
| |||||||||
| Phospholipids |
|
| |||||||||
| Comparing R vs. NR overtime; | |||||||||||
| ↑ vs. ↑↑ | |||||||||||
| Amino acid and amines |
| - | |||||||||
| ↑ vs. ↓ | ↑↑ vs. ↑ | ||||||||||
| Phospholipids |
|
| |||||||||
| At H48, R vs. NR; | |||||||||||
| Amino acids and amines |
|
| |||||||||
| Phospholipids | - |
| |||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trongtrakul, K.; Thonusin, C.; Pothirat, C.; Chattipakorn, S.C.; Chattipakorn, N. Past Experiences for Future Applications of Metabolomics in Critically Ill Patients with Sepsis and Septic Shocks. Metabolites 2022, 12, 1. https://doi.org/10.3390/metabo12010001
Trongtrakul K, Thonusin C, Pothirat C, Chattipakorn SC, Chattipakorn N. Past Experiences for Future Applications of Metabolomics in Critically Ill Patients with Sepsis and Septic Shocks. Metabolites. 2022; 12(1):1. https://doi.org/10.3390/metabo12010001
Chicago/Turabian StyleTrongtrakul, Konlawij, Chanisa Thonusin, Chaicharn Pothirat, Siriporn C. Chattipakorn, and Nipon Chattipakorn. 2022. "Past Experiences for Future Applications of Metabolomics in Critically Ill Patients with Sepsis and Septic Shocks" Metabolites 12, no. 1: 1. https://doi.org/10.3390/metabo12010001
APA StyleTrongtrakul, K., Thonusin, C., Pothirat, C., Chattipakorn, S. C., & Chattipakorn, N. (2022). Past Experiences for Future Applications of Metabolomics in Critically Ill Patients with Sepsis and Septic Shocks. Metabolites, 12(1), 1. https://doi.org/10.3390/metabo12010001






