Metabolomics in the Identification of Biomarkers of Asthma
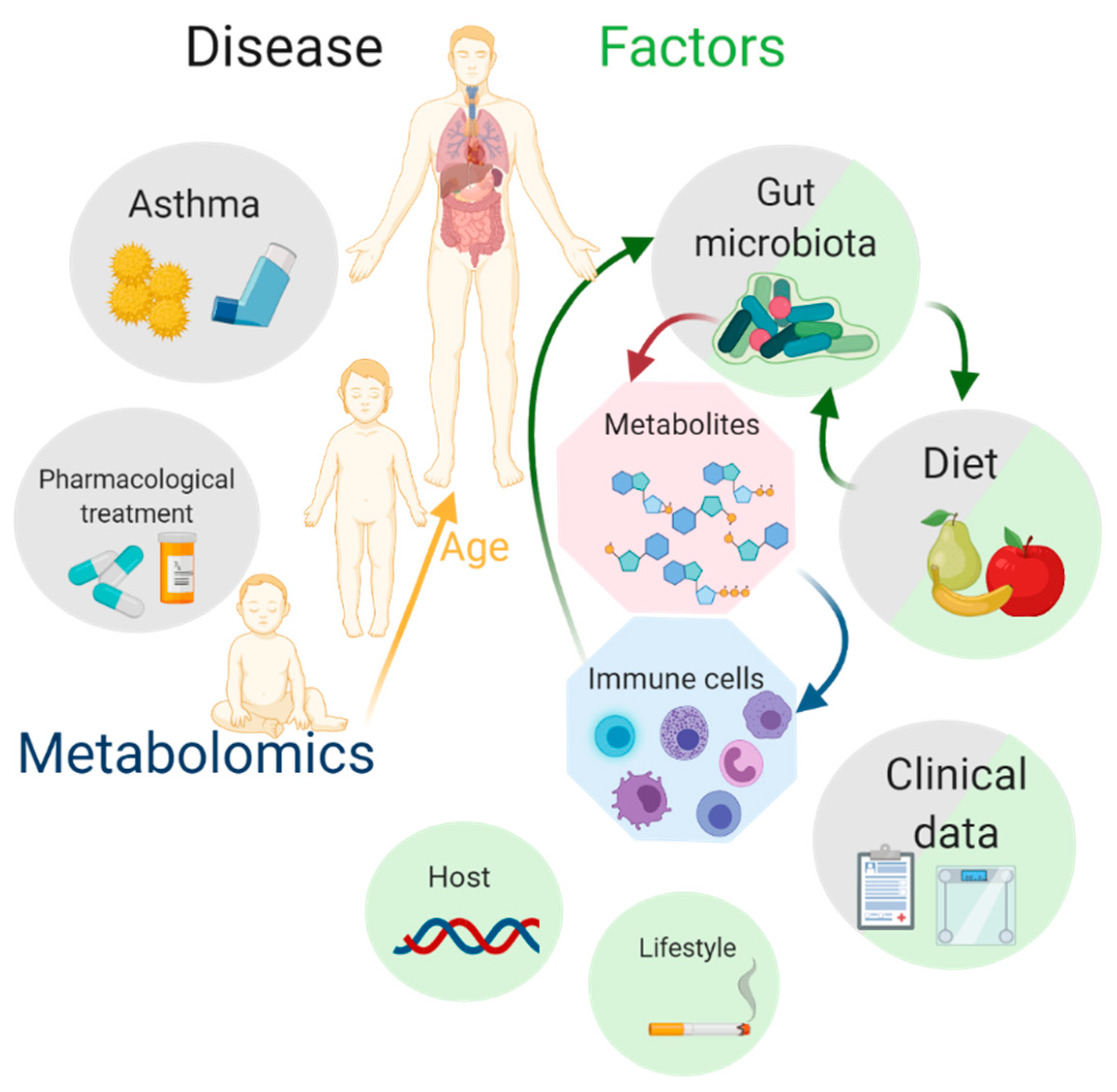
1. Asthma
2. Metabolomics
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Villaseñor, A.; Rosace, D.; Obeso, D.; Pérez-Gordo, M.; Chivato, T.; Barbas, C.; Barber, D.; Escribese, M.M. Allergic Asthma: An Overview of Metabolomic Strategies Leading to the Identification of Biomarkers in the Field. Clin. Exp. Allergy 2017, 47, 442–456. [Google Scholar] [CrossRef] [PubMed]
- Traister, R.S.; Wenzel, S.E. Inflammatory Phenotypes in Asthma Pathogenesis. Drug Discov. Today Dis. Mech. 2012, 9, e75–e81. [Google Scholar] [CrossRef]
- Kuruvilla, M.E.; Lee, F.E.-H.; Lee, G.B. Understanding Asthma Phenotypes, Endotypes, and Mechanisms of Disease. Clin. Rev. Allergy Immunol. 2019, 56, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Diamant, Z.; Vijverberg, S.; Alving, K.; Bakirtas, A.; Bjermer, L.; Custovic, A.; Dahlen, S.; Gaga, M.; Van Wijk, R.G.; Del Giacco, S.; et al. Toward Clinically Applicable Biomarkers for Asthma: An EAACI Position Paper. Allergy 2019, 74, 1835–1851. [Google Scholar] [CrossRef] [PubMed]
- Eguiluz-Gracia, I.; Mathioudakis, A.G.; Bartel, S.; Vijverberg, S.J.H.; Fuertes, E.; Comberiati, P.; Cai, Y.S.; Tomazic, P.V.; Diamant, Z.; Vestbo, J.; et al. The Need for Clean Air: The Way Air Pollution and Climate Change Affect Allergic Rhinitis and Asthma. Allergy 2020, 75, 2170–2184. [Google Scholar] [CrossRef] [PubMed]
- Wheelock, C.E.; Rappaport, S.M. The Role of Gene-Environment Interactions in Lung Disease: The Urgent Need for the Exposome. Eur. Respir. J. 2020, 55, 1902064. [Google Scholar] [CrossRef] [PubMed]
- Papamichael, M.; Katsardis, C.; Sarandi, E.; Georgaki, S.; Frima, E.-S.; Varvarigou, A.; Tsoukalas, D. Application of Metabolomics in Pediatric Asthma: Prediction, Diagnosis and Personalized Treatment. Metabolites 2021, 11, 251. [Google Scholar] [CrossRef] [PubMed]
- Obeso, D.; Zubeldia-Varela, E.; Villaseñor, A. Uncovering the Influence of Diet and Gut Microbiota in Human Serum Metabolome. Allergy 2021. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z., Jr.; Camargo, A.C.; Hasegawa, K. Metabolomics in the Prevention and Management of Asthma. Expert Rev. Respir. Med. 2019, 13, 1135–1138. [Google Scholar] [CrossRef] [PubMed]
- Reinke, S.N.; Gallart-Ayala, H.; Gómez, C.; Checa, A.; Fauland, A.; Naz, S.; Kamleh, M.A.; Djukanović, R.; Hinks, T.S.; Wheelock, C.E. Metabolomics Analysis Identifies Different Metabotypes of Asthma Severity. Eur. Respir. J. 2017, 49, 1601740. [Google Scholar] [CrossRef] [PubMed]
- Lee-Sarwar, K.A.; Lasky-Su, J.; Kelly, R.S.; Litonjua, A.A.; Weiss, S.T. Gut Microbial-Derived Metabolomics of Asthma. Metabolites 2020, 10, 97. [Google Scholar] [CrossRef] [PubMed]
- Rago, D.; Pedersen, C.-E.T.; Huang, M.; Kelly, R.S.; Gürdeniz, G.; Brustad, N.; Knihtilä, H.; Lee-Sarwar, K.A.; Morin, A.; Rasmussen, M.A.; et al. Characteristics and Mechanisms of a Sphingolipid-Associated Childhood Asthma Endotype. Am. J. Respir. Crit. Care Med. 2021, 203, 853–863. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villaseñor, A.; Eguiluz-Gracia, I.; Moreira, A.; Wheelock, C.E.; Escribese, M.M. Metabolomics in the Identification of Biomarkers of Asthma. Metabolites 2021, 11, 346. https://doi.org/10.3390/metabo11060346
Villaseñor A, Eguiluz-Gracia I, Moreira A, Wheelock CE, Escribese MM. Metabolomics in the Identification of Biomarkers of Asthma. Metabolites. 2021; 11(6):346. https://doi.org/10.3390/metabo11060346
Chicago/Turabian StyleVillaseñor, Alma, Ibon Eguiluz-Gracia, André Moreira, Craig E. Wheelock, and María M Escribese. 2021. "Metabolomics in the Identification of Biomarkers of Asthma" Metabolites 11, no. 6: 346. https://doi.org/10.3390/metabo11060346
APA StyleVillaseñor, A., Eguiluz-Gracia, I., Moreira, A., Wheelock, C. E., & Escribese, M. M. (2021). Metabolomics in the Identification of Biomarkers of Asthma. Metabolites, 11(6), 346. https://doi.org/10.3390/metabo11060346








