Abstract
Metabolic syndrome (MetS) is a group of metabolic abnormalities, which together lead to increased risk of coronary heart disease (CHD) and type 2 diabetes mellitus (T2DM), as well as reduced quality of life. Dietary nitrate, betalains and anthocyanins may improve risk factors for MetS and reduce the risk of development of CHD and T2DM. Beetroot is a rich source of dietary nitrate, and anthocyanins are present in high concentrations in blackcurrants. This narrative review considers the efficacy of beetroot and blackcurrant compounds as potential agents to improve MetS risk factors, which could lead to decreased risk of CHD and T2DM. Further research is needed to establish the mechanisms through which these outcomes may occur, and chronic supplementation studies in humans may corroborate promising findings from animal models and acute human trials.
Keywords:
anthocyanins; dietary nitrate; betalains; hyperglycemia; hypertension; dyslipidemia; diabetes; glucose control 1. Introduction
The term metabolic syndrome (MetS) is used to describe a group of metabolic abnormalities associated with an increased risk of coronary heart disease (CHD), cardiovascular disease (CVD), stroke, and type 2 diabetes mellitus (T2DM) [1,2]. These metabolic abnormalities include glucose intolerance, insulin resistance, central obesity, dyslipidemia and hypertension [3]. Three different definitions for MetS were in use from 2001–2009, those of the World Health Organization (WHO) [4], the Adult Treatment Panel (ATP III) [5], and the European Group for the Study of Insulin Resistance (EGIR) [6]. Each organization’s definition and measures varied slightly. Still, all agreed on the basic components of glucose intolerance, obesity, hypertension and dyslipidemia. In 2009, an international collaborative effort, including numerous health federations, developed a unified definition for MetS (Table 1) to enable and simplify data comparisons between nations and organizations [7].

Table 1.
Harmonized criteria for the clinical diagnosis of metabolic syndrome (Adapted from [7]).
As a result of the historical differences in the definitions for MetS, before 2009, it was difficult to establish and compare prevalence data, as different studies employed different measures. Despite this, in 2007, the International Diabetes Federation estimated that around one-quarter of the worldwide adult population (1.9 billion people) had MetS, and further increases were expected due to increasing obesity in developed and developing nations [8]. Furthermore, a 2007 study of MetS prevalence (using the 2001 ATP III definition) in Auckland (NZ), including 1006 Māori, 996 Pacific people, and 2020 of other ethnicities (mostly Europeans), showed that 16% of people aged 35–74 years had MetS, with higher rates in Māori (32%) and Pacific peoples (39%) [9], showing a clear difference in MetS risk between ethnic groups. Disparities between ethnicities are also shown in the US population, with non-Hispanic black women 1.2 times more likely to have MetS than non-Hispanic white women [10]. This study by Moore and colleagues [10] also showed an association between education level and MetS risk. These differences may also be explained through education and deprivation levels, with Māori and Pacific populations in NZ experiencing greater deprivation than NZ Europeans [11] and non-Hispanic black populations having a lower mean education level than non-Hispanic white populations in the US [12].
The global prevalence of T2DM rose from 108 million in 1980 to 422 million in 2014, and current projections estimate that worldwide, 591.9 million people will have T2DM by 2035 [13,14]. Early-onset of T2DM is associated with increased mortality. Data indicate that a 10 year earlier onset of T2DM results in a 1.6 times increased death rate from CVD [15]. MetS itself is associated with a higher relative risk (RR) for mortality from CVD (RR 1.74) [16]. Predictably, several studies have indicated that the presence of MetS is a significant indicator for future diabetes [2,17], with a 2013 study [18] finding a Cox proportional hazard ratio of 2.71 (95% CI 2.30–3.18). Clearly, reducing the incidence of MetS is needed to curb mortality from both T2DM and CVD.
The dietary intervention has been shown to reverse insulin resistance and T2DM [19], improve lipid profiles [20,21] and reduce cardiovascular risk [22,23]. Recent evidence suggests a role for “functional” foods and beverages containing flavonoids in reducing MetS risk [24]. Particular interest has been shown in consuming the anthocyanin subclass of flavonoids, which is associated with reduced T2DM and CVD risk [25,26,27]. Furthermore, foods high in dietary nitrate may be effective in reducing T2DM risk by improving blood glucose control [28]. In addition, dietary nitrate has been shown to lower blood pressure [29,30,31] and improve lipid profiles [32], positively impacting MetS and CVD risk factors.
While various anthocyanins are present in many common berries, blackcurrants have been shown to be a particularly good source of anthocyanins [33], a type of flavonoid responsible for the red/purple color of many berries. Anthocyanins have been shown to improve blood glucose control and other risk factors associated with MetS [34].
Common sources of dietary nitrate include leafy green vegetables and certain root vegetables. Of these, beetroot has gained renown for its unusually high nitrate content. However, it also contains high quantities of betalain pigments and other antioxidants, such as carotenoids, ascorbic acid, flavonoids, and phenolic acids [35], which may also play a role in improving risk factors for chronic disease.
Both Beta vulgaris (beetroot) and Ribes nigrum (blackcurrant) have been studied for their bioactive potential in improving postprandial blood glucose [28,36,37,38,39,40,41,42,43] and other risk factors associated with MetS [44,45,46]. This review, therefore, focuses upon and explores the current evidence for bioactive compounds in beetroot and blackcurrant and their impact on MetS risk factors. Particular focus is given to the compounds nitrate and betalains in beetroot, and anthocyanins in blackcurrant, due to their particular abundance in these foods compared to other sources, as well as the evidence for their efficacy as highlighted in the following review.
1.1. Bioactive Compounds in Beetroot
Beetroot contains many micronutrients in varying abundance. Beetroot contains notable quantities of thiamine (Vit. B1; 0.27 mg, 26% RDI), riboflavin (Vit. B2; 0.27 mg, 20% RDI), folate (Vit. B9; 80 µg, 20% RDI) and iron (0.79 mg, 9.8% RDI) per 100 g beetroot [47,48]. While these compounds are involved in many important metabolic processes, the focus of this review is on the bioactive effects of nitrates and betalains, as these compounds may have a significant effect on MetS risk factors.
1.1.1. Dietary Nitrate
Beetroot stores nitrate in their roots, and along with rocket, radish and celery, are some of the foods most abundant in nitrate. The nitrate content of beetroot has been consistently recorded at over 2500 mg/kg [49,50], with values varying depending on the beetroot cultivar and growing conditions. For instance, beetroot juices prepared from 7 different cultivars showed nitrate content of between 564 ± 129 mg/L and 4626 ± 658 mg/L [50]. Furthermore, beetroot harvested in the summer contains lower nitrate concentrations than those harvested in autumn, as light causes a reduction in nitrate accumulation [51]. The nitrate content of the surrounding soil is also a key variable affecting the nitrate content of the beetroot [52], with low-soil nitrate resulting in reduced uptake into the vegetable.
Dietary nitrate may be a key bioactive within beetroot, as nitrate can be broken down into nitric oxide (NO), which plays a pivotal role in regulating vascular tone, blood pressure [30,53,54], glucose metabolism [55], and lipid peroxidation [32]. Previous studies have shown that patients with T2DM generate less NO from l-arginine compared to healthy controls [56] due to the inhibition of NO synthase by advanced glycation end products (AGEs) [57], resulting in poorer vascular tone regulation, increased blood pressure and impaired glucose metabolism.
The enterosalivary circuit acts as the pathway through which dietary nitrate is reduced into NO. Once ingested, nitrate is absorbed readily across the gut wall and transported to the blood plasma. Around 60–75% of this nitrate is then lost to excretion within 48 h of consumption [58]. Nevertheless, around 25% of nitrate in the circulation is actively concentrated by the sialin transporter to the salivary glands, where it reenters the oral cavity [59]. Here, nitrate is reduced to nitrite through nitrite-producing bacteria, such as Staphylococcus sciuri [60] and then swallowed before being absorbed across the gut wall to increase circulatory nitrite levels [61]. Once in circulation, nitrite is further reduced to NO by nitrite reductase [62] to function as a potent vasodilator, controlling vascular tone and blood pressure [30]. Nitric oxide also mediates glucose uptake from the intestines and skeletal muscle and could play an important role in the regulation of blood glucose levels [55].
Previous research in animal models shows that dietary nitrate supplementation increases circulating NO in animals with impaired NO production brought about by MetS [28,36]. Therefore, the high nitrate content of beetroot may offer a viable pathway to increase circulating NO in humans, particularly in groups who may have impaired NO production, such as people living with diabetes and/or MetS [57].
1.1.2. Betalains
Betalains are divided into two subclasses, betacyanins, which are red pigments and betaxanthines, which are yellow pigments. Beetroot and prickly pear cacti are the only edible food products currently known to contain betalains [35]. Beetroot contains around 120 mg/100 g dry weight of betalains [63], and beetroot juice contains between 700 and 1300 mg/L of betalains depending on the cultivar and growing methods [50].
Structurally, betalains are similar to anthocyanins (Figure 1); pigments, which give foods, such as berries their bright purple, blue and/or red colors, and within plants, betalains perform analogous functions to anthocyanins, meaning the two never naturally coexist [64]. Anthocyanins have been shown to have strong antioxidant effects, and given the similarity in structure, it seems likely that betalains will have equivalent effects. In support of this, both betacyanins and betaxanthines have been shown to have a radical-scavenging capacity three to four-fold greater than ascorbic acid, catechin and rutin [65], with other work demonstrating that the antioxidant capacity of beetroot is positively associated with betalain concentration [66].
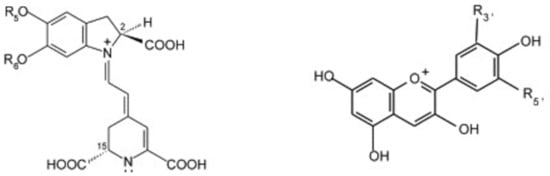
Figure 1.
Typical structure of betalains (left—betacyanin) and anthocyanins (right—anthocyanidin).
Following beetroot juice consumption, betalain concentrations in urine are only 0.3–0.9% of that ingested [67,68], suggesting a high absorption and an alternative method of elimination, such as metabolism. Furthermore, at least 12 betalain derivatives (5 native betalains and 7 betalain metabolites) have been identified in blood plasma and urine after beetroot juice consumption [66]. Interestingly, while the betalain concentrations in blood plasma increase with beetroot consumption, after 2 weeks of chronic supplementation, the process of adaptation favoring betalain metabolism occurs, so that plasma betalain levels drop to less than 10% of that observed during the first week of supplementation [66]. This suggests that as supplementation continues, the breakdown and utilization of betalains and their metabolites increases.
1.2. Bioactive Compounds of Blackcurrant Juice
Blackcurrants contain many bioactive ingredients, and they are particularly potent sources of vitamin C, with just 25 g containing 100% of the Australian and New Zealand recommended daily intake (45 mg) [69]. In addition, blackcurrants contain notable quantities of potassium (322 mg; 8.5% RDI), iron (1.54 mg; 19.3% RDI), manganese (0.256 mg; 4.7% RDI) and phosphorous (59 mg; 5.9% RDI)/100 g. This review, however, focuses on the anthocyanins present in blackcurrants and their effects on markers of MetS, as these pigments are found in greater quantities in blackcurrants than most other berries and edible plants (Table 2).

Table 2.
Content of edible berries/plants for the anthocyanins delphinidin 3-O-glucoside, cyanidin 3-O-glucoside, cyanidin 3-O-rutinoside and delphinidin 3-O-rutinoside.
While the anthocyanins listed in Table 2 are not unique to blackcurrants, they are present at higher levels than in other berries and plants, with blackcurrants containing 476 mg/100 g of anthocyanins, compared with 386 mg/100 g, 140 mg/100 g and 122 mg/100 g in blueberries, cranberries and cherries, respectively [46]. Furthermore, certain New Zealand cultivars are particularly high in anthocyanins, with the total anthocyanin content in New Zealand blackcurrant juices ranging between 346 and 850 mg/100 mL [70,71], compared to 179–310 mg/100 mL for non-New Zealand cultivars [72].
The absorption and bioavailability of anthocyanins are complex since they are highly susceptible to degradation by heat and pH variation [84]. Anthocyanins may undergo several transformations before excretion through liver microsomes and epithelial gut bacteria and via enzymatic degradation [85]. The effects of anthocyanins on health may be due to both the bioactivity of their metabolites [86] as well as their absorbed intact structures. The most abundant anthocyanins present in blackcurrants are delphinidin-3-O-rutinoside and cyanidin-3-O-rutinoside (Table 2). Röhrig et al. [87] investigated the bioavailability of these anthocyanins and their degradation products gallic acid and protocatechuic acid and found that plasma and urine concentrations peaked 2 h following ingestion, with recoveries of 0.040% and 0.048% for delphinidin-3-O-rutinoside and cyanidin-3-O-rutinoside, respectively [87]. In this study, significant quantities of bioactive degradation products were present in the plasma and urine following ingestion of blackcurrant, indicating an abundance of breakdown pathways for anthocyanins and their degradation products [87].
2. Effects of Dietary Nitrate and Beetroot Juice on MetS
2.1. Glucose Homeostasis
While investigations into the potential of beetroot juice supplementation to improve glucose control are limited (Table 3), previous studies using animal models have shown that nitrate supplementation is effective in improving blood glucose metabolism. In a placebo-controlled study, Khalifi et al. [28] divided 32 rats into 4 groups (n = 8): control (C), control + nitrate (CN), diabetes (D), and diabetes + nitrate (DN), and supplemented them with either sodium nitrate (CN and DN) or tap water (C and D) for 8 weeks. Before the intervention, all diabetic rats had lower serum nitrite and nitrate levels, as well as raised systolic blood pressure, compared to controls. Following 8 weeks of supplementation, in the group of diabetic rats supplemented with nitrate (DN), serum nitrite and nitrate and systolic blood pressure returned to values similar to both control groups (C and D) following 8 weeks of supplementation. Furthermore, increases in serum glucose during intravenous glucose tolerance tests for the diabetic, nitrate-supplemented rats were significantly lower than those in the diabetes control group (24.1% vs. 90.2%) [28].

Table 3.
Details of beetroot and dietary nitrate studies investigating glucose homeostasis.
Gheibi et al. [36] used a similar methodology to supplement diabetic and non-diabetic rats and demonstrated improved glucose tolerance, lipid profiles and insulin resistance during glucose tolerance tests, fasting lipids tests and insulin tolerance tests, respectively, in T2DM rats following ad libitum nitrate supplementation with sodium nitrate in drinking water. These effects were associated with decreased gluconeogenesis, inflammation and oxidative stress, and most importantly, increased expression of GLUT4 transporter proteins in insulin-sensitive tissues due to increased activation of AMP-activated protein kinase (AMPK). Before supplementation, mRNA expression and protein levels of GLUT4 were significantly lower in the soleus muscle (54% and 34%, respectively) and adipose tissue (67% and 41%, respectively) of the diabetic rats vs. controls. Supplementation increased mRNA expression and protein levels of GLUT4 translocators in the soleus (215% and 17%, respectively) and adipose tissue (344% and 22%, respectively). This is interesting, as the AMPK and GLUT4 translocation pathways are similar to those currently targeted by common antidiabetic drugs, including metformin [88].
Insulin release also causes increases in NO production, which in turn leads to the dilation of terminal arterioles to increase capillary recruitment (the number of perfused capillaries), as well as relaxation of larger blood vessels to increase peripheral blood flow [89,90]. It is thought that between 25 and 40% of glucose uptake caused by insulin release can be attributed to NO-dependent increases in blood flow to skeletal muscles [91]. As previously stated, oxidative stress from insulin resistance and glucose spikes can downregulate this response [92]. However, supplementation with dietary nitrate through beetroot juice has been shown to counteract the impairment of endothelial function typically associated with ingestion of a mixed macronutrient meal [93].
While these studies show that dietary nitrate can improve glucose metabolism in diabetic animal models, it is yet to be established if these effects can be translated to humans. Furthermore, an effective method of delivery of dietary nitrate in humans other than drinking water would be of greater significance given the lifestyle nature of MetS and T2DM. Beetroot may provide a viable and effective method of delivering dietary nitrate through lifestyle intervention.
While both beetroot juice and whole beetroot contain dietary nitrate and betalains, beetroot juice has been the preferred method of intervention in trials to date. Beetroot juice contains less dietary fiber than the whole beetroot, although dietary fiber may have beneficial effects on glucose metabolism itself [94]. However, beetroot juice does not need to be cooked, which may degrade the betalain due to heat treatment [95,96].
Five studies have investigated the effects of beetroot juice supplementation on glucose control during an acute supplementation trial [37,38,39,40,41] (Table 3). Wootton-Beard et al. [37] recruited 16 healthy volunteers to complete an oral glucose tolerance test (OGTT) with either placebo or beetroot juice (990 mg nitrate) intake in a repeated-measures design. In this study, early postprandial insulin (0–60 min) and glucose responses (0–30 min) were lowered, with a lower peak in glucose response in the beetroot supplemented (22.0 mmol/L/min) vs. the matched control (28.3 mmol/L/min). Fuchs et al. [38] recruited 16 obese, insulin-dependent patients to consume 100 mL beetroot juice (300 mg nitrate) or water but found no effect on postprandial glucose and insulin responses [38]. It is worth noting that Fuchs et al. [38] used far lower levels of nitrate supplementation (100 mL of beetroot juice containing 300 mg nitrate) than used by Wootton-Beard and colleagues [37] (225 mL beetroot juice containing 990 mg nitrate). Furthermore, Fuchs and colleagues [38] only took measures of glucose every 30 min. Hence they may not have had the sensitivity to detect differences in early phase glucose response, such as those found by Wootton-Beard et al. [37], who took measures at baseline, 5, 15, 30, 45, 60, 90, 120 and 150 min. Shepherd et al. [39] recruited 31 healthy participants to complete a 3 h OGTT with either beetroot juice (738 mg nitrate) or nitrate-depleted beetroot juice and found no differences in plasma glucose C-peptide or incretin levels. However, these measures were taken hourly, which may also have led to the same sensitivity issues as Fuchs and colleagues [38]. Furthermore, of note is that the nitrate-depleted juice acted as a control, and while depleted of the bioactive compound nitrate, this juice likely contained high betalain concentrations and therefore, both the intervention and control drinks may have had a hypoglycemic effect on glucose metabolism due to the presence of betalains.
Chang and colleagues [41] recruited 10 healthy volunteers in a crossover trial and administered either 270 mL beetroot juice (nitrate values not available) or a sugar-matched control with white bread to make 50 g of total carbohydrate. The beetroot juice intervention reduced blood glucose levels at 15, 30, 90 and 180 min compared to control, showing a reduction in the early phase and peak glucose, as well as a delay in glycemic response [41]. Furthermore, Holy and colleagues [40] administered 300 g of carbohydrate in a meal with either 250 mL of beetroot juice (nitrate not specified) or 250 mL of water and found that blood glucose levels at 2 h post-meal were significantly lowered in the test condition than in the control group.
Data from a recent longer-term supplementation study involving 30 healthy participants showed that daily consumption of a 10% beetroot juice beverage (9808 mg GAE/100 mL; volume and nitrate concentration not supplied) resulted in a 34.5% decrease in plasma glucose following 4 weeks of supplementation [97]. While these results are promising, the lack of detail in the methods for this study means that comparison is not possible, and it is difficult to draw concrete conclusions based on this research. Further research should be conducted to corroborate these findings, and interventions lasting more than 90 days would be particularly useful to determine how long-term supplementation may affect HbA1c, the gold standard measure of long-term diabetes status.
Currently, there is evidence from human trial data to suggest that beetroot juice may improve acute blood glucose responses through inhibition of salivary enzymes, delayed uptake of glucose across the intestinal wall and increased GLUT-4 translocation. While this may not lead to an improvement in incremental area under the curve during OGTT tests, there does appear to be a decreased peak in blood glucose, which may limit production of AGEs and oxidative damage associated with hyperglycemic episodes. Future studies should analyze incretins, such as gastric inhibitory polypeptide (GIP) and glucagon-like peptide-1 (GLP-1), to establish mechanisms of action or utilize interstitial glucose measurement to give a clearer picture of how these compounds are affecting glucose responses. In a chronic setting, evidence is limited to rat models [28,36] and one 6 week intervention study [97]. However, results show promise in both settings (Table 3). Chronic studies should investigate changes in HbA1C levels for examining the impact on glycemic control, and currently, dosing requirements are unclear since studies are limited and lacking detail. Dietary nitrate appears to have long-term benefits in an animal model. However, the effect of betalains is difficult to observe independently of nitrate. Studies investigating nitrate-depleted beetroot juice vs. placebo may give insight into the action of betalains on blood glucose responses and insulin sensitivity, particularly given the findings of Shepherd and colleagues [39], who found no difference between nitrate-depleted beetroot juice and normal beetroot juice on OGTT outcomes, although, as previously discussed, the sensitivity of these findings may be lower than those of Wooton-Beard and colleagues [37].
2.2. Hypertension
To date, several studies have investigated the effects of beetroot juice supplementation on blood pressure (Table 4), with most studies finding a significant reduction in blood pressure following both acute [31,98,99] and longer-term (1–8 weeks) supplementation [46,100,101,102,103]; however, other studies have shown no positive effects [104,105,106]. Several factors may have influenced the outcomes from these studies, including participants’ age, gender, BMI and/or beetroot dosage.

Table 4.
Details of beetroot trials investigating hypertension.
Most studies have shown that supplementation with beetroot juice has a greater effect on blood pressure in males [105,107] since premenopausal females tend to have a lower initial blood pressure than males of the same age and BMI [108]. One study showed decreased systolic blood pressure in males of 4–5 mmHg compared to 2–3 mmHg in females, 6 h after supplementation with 500 g of beetroot and apple juice (465 mg nitrate) [107]. Furthermore, a recent meta-analysis [109] showed that subjects with a BMI > 25 had greater decreases in blood pressure following beetroot juice consumption compared to subjects with a normal BMI.
The mechanisms through which beetroot supplementation may reduce blood pressure are well described, with dietary nitrate potentially increasing NO content in the vasculature through nitrate and nitrite-reducing enzyme activity. NO then stimulates cyclic 3′, 5′-guanosine monophosphate in the vascular smooth muscles, which initiates Ca2+ release, resulting in increased vasodilation and decreased blood pressure [52]. Interestingly, Bahadoran et al. [109], in a meta-analysis of 27 studies using nitrate-depleted beetroot juice as a control, showed that beetroot juice seems to have blood pressure-lowering effects (SBP, −2.91 mmHg: DBP −0.91 mmHg) independent of nitrate, which may suggest a role for betalains in the responses observed in previous trials.
To date, findings suggest that beetroot juice supplementation improves blood pressure regulation through increased NO content in the vasculature. Furthermore, evidence suggests there are hypotensive effects of beetroot juice independent of nitrate, meaning betalains may have an important role to play. Further investigation is needed to understand the hypotensive effect of betalains since studies to date have tended to focus on the nitrate content of their interventions and do not report betalain content. In addition, promising data from studies using anthocyanins [44,110], the structurally similar counterparts of betalains found in berries and other plants, suggests betalains may work through similar mechanisms as anthocyanins. However, these mechanisms are not presently fully understood.
2.3. Dyslipidemia
Different aspects of lipid profiles have been investigated following supplementation with beetroot (Table 5) in healthy individuals [40], physically fit soldiers [111] and hypercholesterolemic rats [112]. Early in vivo studies by Al-Dosari and colleagues [113] involving rats with hypercholesterolemia showed that administration of 250 mg/kg body weight of freeze-dried beetroot extract (anthocyanin data not given) significantly decreased total cholesterol and triglycerides in the intervention group vs. control. Al-Dosari and colleagues [113] also found a significant increase in high-density lipoprotein (HDL) in the intervention group vs. control.

Table 5.
Details of beetroot and dietary nitrate trials investigating dyslipidemia.
In human trials, Holy et al. [40] showed that in healthy subjects, an acute dose of beetroot juice (250 mL, nitrate and betalains not specified) with a carbohydrate meal (300 g) lowers blood triglycerides, total cholesterol and low-density lipoprotein (LDL) [40]. Research by Singh et al. [111] involved daily supplementation of 30 soldiers with 400 mL of beetroot (nitrate and betalains not specified) juice for 15 days in a before-after study. HDL was significantly increased (1.109 ± 0.214 mmol/L to 1.298 ± 0.253 mmol/L) and LDL was significantly reduced (3.354 ± 2.128 mmol/L to 3.090 ± 2.048 mmol/L) from baseline to 15 days [111]. However, Singh et al. [111] did not include a control group, meaning conclusions may be difficult to draw as other variables, such as training status in their military rotation cycle and changes in diet due to the intervention, may have affected these results. Taken together, the results from these studies [40,111,113] show that beetroot juice may be beneficial in improving dyslipidemia both acutely and following longer-term consumption, however more robust methodology, such as randomized control trials, is needed in longer-term trials to corroborate the work of Singh and colleagues [111].
The mechanism(s) by which beetroot supplementation improves lipid profiles is not yet clear. It may be that both the increased NO production from nitrate and the potential antioxidant effects of the betalains from beetroot consumption are leading to improvements in the lipid profile. Previous work has shown that isolated betanin inhibits lipid peroxidation, leading to fewer reactive oxygen species and less oxidative damage [68]. In contrast, others [28] have shown improved lipid profiles following dietary nitrate supplementation in the absence of betalains. Combined, this may show independent effects of both betalains and nitrate in beetroot in improving lipid profiles.
It is possible that a “functional food” high in both dietary nitrate and betalains may have a marked effect on dyslipidemia in people living with MetS. Future research should focus on establishing mechanisms of action and establishing the efficacy of the intervention in a chronic trial, using a placebo-controlled crossover design. A Latin square design, including nitrate-depleted beetroot juice, may also help clarify the effects of betalains in this complex equation. Furthermore, dosing studies using different concentrations of both nitrate and betalains may aid in our understanding of the mechanisms of action through which beetroot may improve dyslipidemia.
3. Effects of Blackcurrant Anthocyanins on MetS
3.1. Glucose Homeostasis
The effects of blackcurrant anthocyanins (mostly delivered in studies as juice) on blood glucose responses have been extensively researched in recent years (Table 6), with randomized crossover and controlled trials conducted in both healthy [34,42] and overweight adults [43]. Following consumption of blackcurrant juice and blackcurrant juice fortified with crowberry powder, Törrönen et al. [114] conducted OGTT trials with 14 participants. They found that, whereas glucose and insulin responses in the early phase (0–30 min) were improved in the blackcurrant with crowberry extract group, there was no significant difference in overall AUC (area under the curve) for glucose (0–120 min). This is similar to findings in beetroot juice trials [37,41] and may indicate that carbohydrate absorption is delayed, rather than reduced, following acute supplementation.

Table 6.
Details of trials investigating blackcurrant juice and extracts on glucose homeostasis.
Castro-Acosta and colleagues [34] showed that drinks containing blackcurrant extract providing 600 mg of anthocyanins effectively reduced plasma glucose responses to a high-carbohydrate meal at 0–30 min post-meal. Furthermore, plasma insulin, plasma GIP and GLP-1 were all significantly reduced following blackcurrant extract consumption compared to control. In a subsequent study [42], 25 healthy participants were supplemented with placebo, apple extract, or apple extract and blackcurrant extract in association with a starch and sucrose meal. The iAUC (incremental area under the curve; 0–30 min) improved in the apple vs. placebo trials, while apple and blackcurrant improved iAUC vs. both placebo and apple extract [42]. As apple extract has been shown to inhibit sodium-glucose transport protein-1 (SGLT-1) action [115] and proanthocyanidins in blackcurrant, inhibit alpha-amylase [116] and alpha-glucosidase [117], combining different polyphenol-rich foods may result in a synergistic effect, acting on multiple pathways, to impact on postprandial glucose responses; however further research is needed to corroborate these findings.
Watson et al. [118] took glucose measurements at 60 and 150 min following consumption of blackcurrant fruit juice (525 mg polyphenols/60 kg BW; 0.91 g carbohydrate/kg BW) following an overnight fast and observed elevated blood glucose at both time points in the intervention vs. control. While these results appear to show increased blood glucose following blackcurrant supplementation, this supports the findings of Castro-Acosta [34,42] and Törrönen [119], where the early phase blood glucose response was inhibited, with elevated glucose readings occurring at 60 and 150 min. It is possible that if glucose measurements had been taken in the early phase (0–30 min), a blunted blood glucose would have been seen at this time point. However, this can only be speculated upon. Furthermore, in this trial [118], the intervention drinks were not consumed as part of a meal but on their own, which may have affected the speed of absorption of the carbohydrates.
Several chronic supplementation studies using blackcurrant anthocyanins to investigate glucose regulation have also been conducted. Nolan et al. [43] conducted a two-experiment intervention, including an acute supplementation and a short-term (8 days) supplementation trial. They demonstrated improvements in free-living individuals’ glucose control using continuous glucose monitoring over 8 days of supplementation with a New Zealand blackcurrant extract (300 mg ingested before breakfast, 300 mg ingested before dinner; 600 mg anthocyanins per day) in overweight and obese adults [43]. Compared to placebo, insulin sensitivity was improved by 22%, and postprandial glucose responses to breakfast and lunch were reduced by 9% and 8%, respectively, in the intervention group [43].
In the acute supplementation trial, they found significant reductions in postprandial blood glucose at 45 min, 60 min and 90 min, as well as decreased AUC for glucose and insulin following acute ingestion of blackcurrant extract containing 105 mg anthocyanins with a mixed macronutrient meal [43]. These data suggest that even short-term supplementation with New Zealand blackcurrant extract can improve insulin sensitivity. This has been shown previously with blueberries [121]. However, this is the first study of its kind conducted using an anthocyanin-rich blackcurrant extract.
From the current evidence, it is clear that blackcurrant supplementation has an acute effect on blood glucose levels, delaying blood glucose responses and decreasing peak glucose during OGTTs by inhibiting carbohydrate absorption during the early phase (0–30 min). Further work needs to be undertaken to understand the appropriate doses for achieving long-term improvements in blood glucose responses, as previous work has utilized anthocyanin levels ranging from 210 mg [44] to 1200 mg [42], and have been consumed at different times of the day and at different times concerning OGTT/meal consumption.
3.2. Hypertension
The potential effects of blackcurrant anthocyanins on hypertension are currently not well established (Table 7). However, a recent study [44] showed that ingestion of 600 mg/day of anthocyanins from a NZ blackcurrant extract over a 7 day intervention period resulted in decreases in systolic (6 mmHg) and diastolic (6 mmHg) blood pressure in a cohort of 14 healthy older adults. Such changes are sufficient to improve health outcomes. Reducing systolic blood pressure of 6 mmHg contributes about 35–40% less stroke and 20–25% less coronary heart disease mortality [122] after just one week of supplementation.

Table 7.
Details of trials investigating blackcurrant juice and extracts on hypertension and dyslipidemia.
Anthocyanins are proposed to lower blood pressure through three main mechanisms. First, anthocyanins increase endothelial-derived NO through modulation of endothelial NO synthase (eNOS) expression [123]. As previously stated, NO contributes to endothelium-dependent vasorelaxation through increased stimulation of cyclic 3′, 5′-guanosine monophosphate, in turn leading to increased blood vessel vasodilation. Second, due to their high antioxidant activity, anthocyanins can protect against reactive oxygen species, which normally interact rapidly with NO to form other radical intermediates, resulting in lower NO availability, leading to vasoconstriction and hypertension. The protective effect of anthocyanins reduces this NO conversion, thus preventing oxidative damage [124]. Third, anthocyanins are proposed to inhibit production of vasoconstriction-promoting molecules, such as angiotensin II and thromboxanes, through inhibition of the enzymes (angiotensin-converting enzyme and cyclooxygenase, respectively), which catalyze production of these vasoconstrictors [125]. Although yet to be confirmed in human trials, Park [45] also showed that blackcurrant downregulated endothelin-1 expression, another vasoconstrictor following blackcurrant supplementation in rats, and this could be another contributing mechanism to the effects observed in humans in previous trials.
While current crossover studies are limited (Table 7), the mechanisms through which blackcurrant supplementation may regulate vascular tone are well understood. More studies need to be conducted in human populations, both in an acute and chronic setting, to corroborate the findings of Cook et al. [44] and establish ideal dosage for chronic studies.
3.3. Dyslipidemia
As well as improving both glucose tolerance and blood pressure, consumption of blackcurrant extract also has been shown to have beneficial effects on lipid profiles in both rats [45] and human trials [119] (Table 7). Park et al. [45] induced MetS in Sprague-Dawley rats and supplemented them with placebo, 100 mg/kg/day or 300 mg/kg/day of blackcurrant extract for 4 weeks to investigate lipid profile responses. Following supplementation, rats ingesting 100 mg/kg/day and 300 mg/kg/day of blackcurrant showed significant decreases in triglyceride and LDL levels and a significant increase in HDL levels vs. control.
More recently, Nanashima et al. fed ovariectomized rats ad libitum either a regular diet or one containing 3% blackcurrant extract and showed decreased bodyweight, visceral fat weight, serum triglycerides, total cholesterol and LDL cholesterol in the extract-treated rats [126]. The findings of this study [126] show promise for the efficacy of blackcurrant anthocyanins to decrease dyslipidemia in a menopausal animal model. Postmenopausal women with low estrogen activity show an increased risk of dyslipidemia [127]. However, blackcurrant anthocyanins may attenuate this response through phytoestrogen signaling in estrogen receptors α [128] and β [129].
In human trials, Törrönen et al. [119] investigated blood glucose and free fatty acid (FFA) responses in 20 healthy females (aged 25–69 years) in a randomized, crossover, acute meal study, consuming blackcurrants or lingonberries as a puree with 35 g sucrose added or 35 g sucrose with water. Ingestion of the berry meals decreased the concentration of FFAs compared to the control meal, despite there being an overall higher carbohydrate load in the berry meal [119].
While there is evidence that blackcurrant extract supplementation can improve blood lipid profiles in an acute setting and mechanisms, including phytoestrogen signaling through receptors, chronic studies have yet to be conducted in humans, and dosages are yet to be established.
4. Future Perspectives
This review has focused on the efficacy of beetroot and blackcurrants to improve risk factors for MetS. While there is currently strong evidence to suggest the compounds present within these foods may have positive effects on MetS and its risk factors, some key gaps in the literature need to be addressed before it can be reliably concluded that blackcurrant and beetroot compounds improve MetS risk factors.
First, current literature investigating beetroot and MetS risk factors focuses on dietary nitrate as the active ingredient, despite evidence suggesting that betalains may have an additive or synergistic effect through other, currently unclear, pathways. It is important that future studies report both the betalain content as well as the nitrate content of interventions to ensure results can be compared between studies and to enable the sources of any observed effects to be established. Following this, studies investigating nitrate-depleted beetroot may be useful in determining the potential of betalains to affect MetS outcomes. Nitrate-depleted beetroot juice has already been suggested to improve hypertension [109]. However, further work needs to be conducted to investigate its effects on hyperglycemia and dyslipidemia.
Interestingly, research by Sawicki and colleagues [130], who supplemented volunteers with red beetroot juice for 6 weeks, found that betalain concentrations were highest in the plasma after 1 week and urine after 2 weeks. However, the lowest levels for plasma and urine were found in weeks 3 and 4, respectively. The authors suggest that this results from a process of adaptation towards betalain metabolism, as has been shown for other phytochemicals [131]. This may mean that to elicit changes in MetS risk factors, chronic supplementation with beetroot juice is more effective than acute supplementation. The shift in metabolism can only take place after 2 weeks of exposure to betalains. Trials investigating chronic supplementation should be conducted in the future, measuring plasma and urine betalain concentrations as well as MetS risk factors to establish whether this is the case.
Furthermore, in both blackcurrant and beetroot trials, there is little information regarding the appropriate doses of bioactive ingredients to elicit changes, with anthocyanin levels in blackcurrant studies ranging from 210 mg [44] to 1200 mg [42] and nitrate content administration in beetroot juice studies ranging between 68.2 mg [98] and 990 mg [37]. Future studies, particularly in areas such as hypertension, where considerable evidence currently exists, should establish appropriate minimum dosages for eliciting the improvements in MetS risk factors. This would allow more appropriate administration of any lifestyle intervention, which may increase compliance within an at-risk population.
Recent work has shown that the methods for extraction and processing of beetroot [132] and blackcurrant [133] compounds can significantly impact their betalain and anthocyanin content, respectively. Betalains [132] and anthocyanins [84] are particularly sensitive toward heat, pH, light and oxygen, leading to poor stability of these compounds during processing. Beetroot, for example, is often blanched to prolong its freshness for commercial sale, degrading the betalains [134]. This means beetroot products used in intervention trials may not be reflective of those commercially available and affordable. Recently, however, investigations into alternative processing methods have shown that high-pressure processing retains betalains and vitamin C more successfully than blanching [135]. Furthermore, studies should look to use commercially available juices and extracts when conducting trials, as these are reflective of what is applicable for the consumer. Most of the studies evaluating the effects of blackcurrant anthocyanins on biomarkers of MetS have used blackcurrants and blackcurrant extracts from New Zealand, which have reported higher anthocyanin levels [71] than non-New Zealand fruit/juice [33]. It is speculative to suggest that New Zealand blackcurrant could be more beneficial in managing MetS and its risk factors, but more research on this possibility in well-designed RCTs is required.
5. Conclusions
This review aimed to collect and highlight scientific evidence regarding the role of beetroot and blackcurrant consumption in the possible prevention of MetS and its associated risk factors. The current evidence demonstrates that beetroot and blackcurrant positively affects management of several MetS risk factors, including dyslipidemia, hyperglycemia and hypertension. Beetroot and blackcurrant may improve glucose uptake by activating AMPK, GLUT-4 and SGLT-1, while also inhibiting the breakdown of glucose in the intestines and saliva, leading to overall improvements in glucose control. Beetroot exhibits hypotensive effects through increasing NO availability through nitrate. However, betalains may also have a role, as has been shown for structurally similar blackcurrant anthocyanins, through modulation of endothelial NO synthase (eNOS) expression and inhibition of vasoconstriction-promoting molecule production. However, more research is required to demonstrate this conclusively. Finally, beetroot and blackcurrants may improve lipid profiles. However, the mechanisms through which this takes place are not clear, and more mechanistic as well as RCTs are needed to strengthen the currently limited evidence.
Author Contributions
C.H., K.R.-M., A.A., R.P. and R.H. contributed to the conceptualization of the manuscript; C.H., performed the literature search and paper selection for the review; C.H., completed the original draft preparation; C.H., K.R.-M., A.A., R.P. and R.H. reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
Hurst has dual associations with commercial organizations that may benefit financially from the sale of products made from New Zealand blackcurrants: as Principal Scientist for the New Zealand Institute for Plant and Food Research Limited: and as Chief Science Advisor for 2before Performance Nutrition Limited. These associations did not influence the representation or interpretation of reported data and research findings.
References
- Isomaa, B.; Almgren, P.; Tuomi, T.; Forsen, B.; Lahti, K.; Nissen, M.; Taskinen, M.R.; Groop, L. Cardiovascular Morbidity and Mortality Associated With the Metabolic Syndrome. Diabetes Care 2001, 24, 683–689. [Google Scholar]
- Shin, J.-A.; Lee, J.-H.; Lim, S.-Y.; Ha, H.-S.; Kwon, H.-S.; Park, Y.-M.; Lee, W.-C.; Kang, M.-I.; Yim, H.-W.; Yoon, K.-H.; et al. Metabolic syndrome as a predictor of type 2 diabetes, and its clinical interpretations and usefulness. J. Diabet. Investig. 2013, 4, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Eckel, R.H.; Alberti, K.G.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2010, 375, 181–183. [Google Scholar] [CrossRef]
- Alberti, K.G.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus. Provisional report of a WHO consultation. Diabet. Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). J. Am. Med. Assoc. 2001, 285, 2486. [Google Scholar] [CrossRef] [PubMed]
- Balkau, B. Comment on the provisional report from the WHO consultation. European Group for the Study of Insulin Resistance (EGIR). Diabet. Med. 1999, 16, 442–443. [Google Scholar] [PubMed]
- Alberti, K.G.M.M.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.-C.; James, P.T.; Loria, C.M.; Smith, S.C. Harmonizing the metabolic syndrome. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef]
- Desroches, S.; Lamarche, B. The evolving definitions and increasing prevalence of the metabolic syndrome. Appl. Physiol. Nutr. Metab. 2007, 32, 23–32. [Google Scholar] [CrossRef]
- Gentles, D.; Metcalf, P.; Dyall, L.; Sundborn, G.; Schaaf, D.; Black, P.; Scragg, R.; Jackson, R. Metabolic syndrome prevalence in a multicultural population in Auckland, New Zealand. N. Z. Med. J. 2007, 120, U2399. [Google Scholar]
- Moore, J.X.; Chaudhary, N.; Akinyemiju, T. Metabolic Syndrome Prevalence by Race/Ethnicity and Sex in the United States, National Health and Nutrition Examination Survey, 1988–2012. Prev. Chronic Dis. 2017, 14, E24. [Google Scholar] [CrossRef]
- Utter, J.; Denny, S.; Crengle, S.; Ameratunga, S.; Robinson, E.; Clark, T.; Percival, T.; Maddison, R. Overweight among New Zealand adolescents: Associations with ethnicity and deprivation. Int. J. Pediatric Obes. 2010, 5, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Assari, S.; Caldwell, C.H.; Bazargan, M. Parental Educational Attainment and Black-White Adolescents’ Achievement Gap: Blacks’ Diminished Returns. Open J. Soc. Sci. 2020, 08, 282–297. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Lu, Y.; Hajifathalian, K.; Bentham, J.; Di Cesare, M.; Danaei, G.; Bixby, H.; Cowan, M.; Ali, M.; Taddei, C.; et al. Worldwide trends in diabetes since 1980: A pooled analysis of 751 population-based studies with 4·4 million participants. Lancet 2016, 387, 1513–1530. [Google Scholar] [CrossRef]
- Guariguata, L.; Whiting, D.R.; Hambleton, I.; Beagley, J.; Linnenkamp, U.; Shaw, J.E. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res. Clin. Pract. 2014, 103, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Best Practice Journal New Zealand (BPAC NZ). A Rising Tide of Type 2 Diabetes in Younger People: What Can Primary Care Do? Available online: https://bpac.org.nz/2018/docs/diabetes.pdf (accessed on 10 October 2020).
- Galassi, A.; Reynolds, K.; He, J. Metabolic syndrome and risk of cardiovascular disease: A meta-analysis. Am. J. Med. 2006, 119, 812–819. [Google Scholar] [CrossRef]
- Laaksonen, D.E.; Lakka, H.-M.; Niskanen, L.K.; Kaplan, G.A.; Salonen, J.T.; Lakka, T.A. Metabolic syndrome and development of diabetes mellitus: Application and validation of recently suggested definitions of the metabolic syndrome in a prospective cohort study. Am. J. Epidemiol. 2002, 156, 1070–1077. [Google Scholar] [CrossRef] [PubMed]
- Povel, C.M.; Beulens, J.W.; Van Der Schouw, Y.T.; Dolle, M.E.T.; Spijkerman, A.M.W.; Verschuren, W.M.M.; Feskens, E.J.M.; Boer, J.M.A. Metabolic Syndrome Model Definitions Predicting Type 2 Diabetes and Cardiovascular Disease. Diabetes Care 2013, 36, 362–368. [Google Scholar] [CrossRef]
- Gow, M.L.; Baur, L.A.; Johnson, N.A.; Cowell, C.T.; Garnett, S.P. Reversal of type 2 diabetes in youth who adhere to a very-low-energy diet: A pilot study. Diabetologia 2017, 60, 406–415. [Google Scholar] [CrossRef]
- Tierney, A.C.; McMonagle, J.; Shaw, D.I.; Gulseth, H.L.; Helal, O.; Saris, W.H.M.; Paniagua, J.A.; Gołąbek-Leszczyñska, I.; Defoort, C.; Williams, C.M.; et al. Effects of dietary fat modification on insulin sensitivity and on other risk factors of the metabolic syndrome—LIPGENE: A European randomized dietary intervention study. Int. J. Obes. 2011, 35, 800–809. [Google Scholar] [CrossRef]
- Lankinen, M.; Schwab, U.; Kolehmainen, M.; Paananen, J.; Nygren, H.; Seppänen-Laakso, T.; Poutanen, K.; Hyötyläinen, T.; Risérus, U.; Savolainen, M.J. A healthy Nordic diet alters the plasma lipidomic profile in adults with features of metabolic syndrome in a multicenter randomized dietary intervention. J. Nutr. 2015, 146, 662–672. [Google Scholar] [CrossRef]
- Abbate, M.; Gallardo-Alfaro, L.; Bibiloni, M.D.M.; Tur, J.A. Efficacy of dietary intervention or in combination with exercise on primary prevention of cardiovascular disease: A systematic review. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 1080–1093. [Google Scholar] [CrossRef] [PubMed]
- Apostolidou, C.; Adamopoulos, K.; Lymperaki, E.; Iliadis, S.; Papapreponis, P.; Kourtidou-Papadeli, C. Cardiovascular risk and benefits from antioxidant dietary intervention with red wine in asymptomatic hypercholesterolemics. Clin. Nutr. ESPEN 2015, 10, e224–e233. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, S. Functional foods against metabolic syndrome (obesity, diabetes, hypertension and dyslipidemia) and cardiovasular disease. Trends Food Sci. Technol. 2014, 35, 114–128. [Google Scholar] [CrossRef]
- Cassidy, A.; Bertoia, M.; Chiuve, S.; Flint, A.; Forman, J.; Rimm, E.B. Habitual intake of anthocyanins and flavanones and risk of cardiovascular disease in men. Am. J. Clin. Nutr. 2016, 104, 587–594. [Google Scholar] [CrossRef]
- Wedick, N.M.; Pan, A.; Cassidy, A.; Rimm, E.B.; Sampson, L.; Rosner, B.; Willett, W.; Hu, F.B.; Sun, Q.; Van Dam, R.M. Dietary flavonoid intakes and risk of type 2 diabetes in US men and women. Am. J. Clin. Nutr. 2012, 95, 925–933. [Google Scholar] [CrossRef]
- Muraki, I.; Imamura, F.; Manson, J.E.; Hu, F.B.; Willett, W.C.; Van Dam, R.M.; Sun, Q. Fruit consumption and risk of type 2 diabetes: Results from three prospective longitudinal cohort studies. BMJ 2013, 347, f5001. [Google Scholar] [CrossRef]
- Khalifi, S.; Rahimipour, A.; Jeddi, S.; Ghanbari, M.; Kazerouni, F.; Ghasemi, A. Dietary nitrate improves glucose tolerance and lipid profile in an animal model of hyperglycemia. Nitric Oxide 2015, 44, 24–30. [Google Scholar] [CrossRef]
- Gilchrist, M.; Winyard, P.G.; Aizawa, K.; Anning, C.; Shore, A.; Benjamin, N. Effect of dietary nitrate on blood pressure, endothelial function, and insulin sensitivity in type 2 diabetes. Free Radic. Biol. Med. 2013, 60, 89–97. [Google Scholar] [CrossRef]
- Larsen, F.J.; Ekblom, B.; Sahlin, K.; Lundberg, J.O.; Weitzberg, E. Effects of Dietary Nitrate on Blood Pressure in Healthy Volunteers. N. Engl. J. Med. 2006, 355, 2792–2793. [Google Scholar] [CrossRef]
- Stanaway, L.; Rutherfurd-Markwick, K.; Page, R.; Wong, M.; Jirangrat, W.; Teh, K.H.; Ali, A. Acute Supplementation with Nitrate-Rich Beetroot Juice Causes a Greater Increase in Plasma Nitrite and Reduction in Blood Pressure of Older Compared to Younger Adults. Nutrients 2019, 11, 1683. [Google Scholar] [CrossRef]
- Carlström, M.; Larsen, F.J.; Nyström, T.; Hezel, M.; Borniquel, S.; Weitzberg, E.; Lundberg, J.O. Dietary inorganic nitrate reverses features of metabolic syndrome in endothelial nitric oxide synthase-deficient mice. Proc. Natl. Acad. Sci. USA 2010, 107, 17716–17720. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Gebhardt, S.E.; Prior, R.L. Concentrations of anthocyanins in common foods in the United States and estimation of normal consumption. J. Agric. Food Chem. 2006, 54, 4069–4075. [Google Scholar] [CrossRef]
- Castro-Acosta, M.L.; Smith, L.; Miller, R.J.; McCarthy, D.I.; Farrimond, J.A.; Hall, W.L. Drinks containing anthocyanin-rich blackcurrant extract decrease postprandial blood glucose, insulin and incretin concentrations. J. Nutr. Biochem. 2016, 38, 154–161. [Google Scholar] [CrossRef]
- Kujala, T.; Vienola, M.; Klika, K.; Loponen, J.; Pihlaja, K. Betalain and phenolic compositions of four beetroot (Beta vulgaris) cultivars. Eur. Food Res. Technol. 2002, 214, 505–510. [Google Scholar] [CrossRef]
- Gheibi, S.; Jeddi, S.; Carlström, M.; Gholami, H.; Ghasemi, A. Effects of long-term nitrate supplementation on carbohydrate metabolism, lipid profiles, oxidative stress, and inflammation in male obese type 2 diabetic rats. Nitric Oxide 2018, 75, 27–41. [Google Scholar] [CrossRef]
- Wootton-Beard, P.C.; Brandt, K.; Fell, D.; Warner, S.; Ryan, L. Effects of a beetroot juice with high neobetanin content on the early-phase insulin response in healthy volunteers. J. Nutr. Sci. 2014, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, D.; Nyakayiru, J.; Draijer, R.; Mulder, T.P.; Hopman, M.T.; Eijsvogels, T.M.; Thijssen, D.H. Impact of flavonoid-rich black tea and beetroot juice on postprandial peripheral vascular resistance and glucose homeostasis in obese, insulin-resistant men: A randomized controlled trial. Nutr. Metab. 2016, 13, 34. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, A.I.; Wilkerson, D.P.; Fulford, J.; Winyard, P.G.; Benjamin, N.; Shore, A.C.; Gilchrist, M. Effect of nitrate supplementation on hepatic blood flow and glucose homeostasis: A double-blind, placebo-controlled, randomized control trial. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 311, G356–G364. [Google Scholar] [CrossRef]
- Holy, B.I.N.; Briggs, N. Post-prandial effect of beetroot (beta vulgaris) juice on glucose and lipids levels of apparently healthy subjects. Eur. J. Pharm. Med. Res. 2017, 4, 60–62. [Google Scholar]
- Chang, P.; Hafiz, M.; Boesch, C. Beetroot juice attenuates glycaemic response in healthy volunteers. Proc. Nutr. Soc. 2018, 77, E165. [Google Scholar] [CrossRef]
- Castro-Acosta, M.L.; Stone, S.G.; Mok, J.E.; Mhajan, R.K.; Fu, C.-I.; Lenihan-Geels, G.N.; Corpe, C.P.; Hall, W.L. Apple and blackcurrant polyphenol-rich drinks decrease postprandial glucose, insulin and incretin response to a high-carbohydrate meal in healthy men and women. J. Nutr. Biochem. 2017, 49, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Nolan, A.; Brett, R.; Strauss, J.A.; Stewart, C.E.; Shepherd, S.O. Short-term, but not acute, intake of New Zealand blackcurrant extract improves insulin sensitivity and free-living postprandial glucose excursions in individuals with overweight or obesity. Eur. J. Nutr. 2020, 60, 1253–1262. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.D.; Sandu, B.; Kaur, A.; Joyce, P. Jennifer Patricia, Effect of New Zealand Blackcurrant on Blood Pressure, Cognitive Function and Functional Performance in Older Adults. J. Nutr. Gerontol. Geriatr. 2020, 39, 99–113. [Google Scholar] [CrossRef]
- Park, J.H.; Kho, M.C.; Kim, H.Y.; Ahn, Y.M.; Lee, Y.J.; Kang, D.G.; Lee, H.S. Blackcurrant Suppresses Metabolic Syndrome Induced by High-Fructose Diet in Rats. Evid. Based Complement. Altern. Med. 2015, 2015, 1–11. [Google Scholar] [CrossRef]
- Capper, T.; Houghton, D.; Stewart, C.J.; Blain, A.P.; McMahon, N.; Siervo, M.; West, D.J.; Stevenson, E.J. Whole beetroot consumption reduces systolic blood pressure and modulates diversity and composition of the gut microbiota in older participants. NFS J. 2020, 21, 28–37. [Google Scholar] [CrossRef]
- Yashwant, K. Beetroot: A super food. IJESTA 2015, 1, 20–26. [Google Scholar]
- National Health and Medical Research Council. Nutrient Reference Values for Australia and New Zealand: Executive Summary; NHMRC: Canberra, Australia, 2006.
- Santamaria, P. Nitrate in vegetables: Toxicity, content, intake and EC regulation. J. Sci. Food Agric. 2006, 86, 10–17. [Google Scholar] [CrossRef]
- Wruss, J.; Waldenberger, G.; Huemer, S.; Uygun, P.; Lanzerstorfer, P.; Müller, U.; Höglinger, O.; Weghuber, J. Compositional characteristics of commercial beetroot products and beetroot juice prepared from seven beetroot varieties grown in Upper Austria. J. Food Compos. Anal. 2015, 42, 46–55. [Google Scholar] [CrossRef]
- Umar, S.; Iqbal, M.; Abrol, Y. Are nitrate concentrations in leafy vegetables within safe limits? Curr. Sci. 2007, 92, 355–360. [Google Scholar]
- Gee, L.C.; Ahluwalia, A. Dietary Nitrate Lowers Blood Pressure: Epidemiological, Pre-clinical Experimental and Clinical Trial Evidence. Curr. Hypertens. Rep. 2016, 18, 17. [Google Scholar] [CrossRef]
- Dharmashankar, K.; Widlansky, M.E. Vascular Endothelial Function and Hypertension: Insights and Directions. Curr. Hypertens. Rep. 2010, 12, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Shesely, E.G.; Maeda, N.; Kim, H.S.; Desai, K.M.; Krege, J.H.; Laubach, V.E.; Sherman, P.A.; Sessa, W.C.; Smithies, O. Elevated blood pressures in mice lacking endothelial nitric oxide synthase. Proc. Natl. Acad. Sci. USA 1996, 93, 13176–13181. [Google Scholar] [CrossRef] [PubMed]
- Roy, D.; Perreault, M.; Marette, A. Insulin stimulation of glucose uptake in skeletal muscles and adipose tissues in vivo is NO dependent. Am. J. Physiol. Endocrinol. Metab. 1998, 274, E692–E699. [Google Scholar] [CrossRef] [PubMed]
- Avogaro, A.; Toffolo, G.; Kiwanuka, E.; De Kreutzenberg, S.V.; Tessari, P.; Cobelli, C. L-Arginine-Nitric Oxide Kinetics in Normal and Type 2 Diabetic Subjects: A Stable-Labelled 15N Arginine Approach. Diabetes 2003, 52, 795–802. [Google Scholar] [CrossRef] [PubMed]
- Amore, A.; Cirina, P.; Mitola, S.; Peruzzi, L.; Gianoglio, B.; Rabbone, I.; Sacchetti, C.; Cerutti, F.; Grillo, C.; Coppo, R. Nonenzymatically glycated albumin (Amadori adducts) enhances nitric oxide synthase activity and gene expression in endothelial cells. Kidney Int. 1997, 51, 27–35. [Google Scholar] [CrossRef]
- Wagner, D.A.; Schultz, D.S.; Deen, W.M.; Young, V.R.; Tannenbaum, S.R. Metabolic fate of an oral dose of 15N-labeled nitrate in humans: Effect of diet supplementation with ascorbic acid. Cancer Res. 1983, 43, 1921–1925. [Google Scholar]
- Qin, L.; Liu, X.; Sun, Q.; Fan, Z.; Xia, D.; Ding, G.; Ong, H.L.; Adams, D.; Gahl, W.A.; Zheng, C.; et al. Sialin (SLC17A5) functions as a nitrate transporter in the plasma membrane. Proc. Natl. Acad. Sci. USA 2012, 109, 13434–13439. [Google Scholar] [CrossRef]
- Li, H.; Duncan, C.; Townend, J.; Killham, K.; Smith, L.M.; Johnston, P.; Dykhuizen, R.; Kelly, D.; Golden, M.; Benjamin, N. Nitrate-reducing bacteria on rat tongues. Appl. Environ. Microbiol. 1997, 63, 924–930. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Govoni, M. Inorganic nitrate is a possible source for systemic generation of nitric oxide. Free Radic. Biol. Med. 2004, 37, 395–400. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Weitzberg, E.; Gladwin, M.T. The nitrate–nitrite–nitric oxide pathway in physiology and therapeutics. Nat. Rev. Drug Fisc. 2008, 7, 156–167. [Google Scholar] [CrossRef]
- Lee, E.J.; An, D.; Nguyen, C.T.; Patil, B.S.; Kim, J.; Yoo, K.S. Betalain and betaine composition of greenhouse-or field-produced beetroot (Beta vulgaris L.) and inhibition of HepG2 cell proliferation. J. Agric. Food Chem. 2014, 62, 1324–1331. [Google Scholar] [CrossRef]
- Stafford, H.A. Anthocyanins and betalains: Evolution of the mutually exclusive pathways. Plant. Sci. 1994, 101, 91–98. [Google Scholar] [CrossRef]
- Cai, Y.; Sun, M.; Corke, H. Antioxidant activity of betalains from plants of the Amaranthaceae. J. Agric. Food Chem. 2003, 51, 2288–2294. [Google Scholar] [CrossRef] [PubMed]
- Sawicki, T.; Bączek, N.; Wiczkowski, W. Betalain profile, content and antioxidant capacity of red beetroot dependent on the genotype and root part. J. Funct. Foods 2016, 27, 249–261. [Google Scholar] [CrossRef]
- Frank, T.; Stintzing, F.C.; Carle, R.; Bitsch, I.; Quaas, D.; Straß, G.; Bitsch, R.; Netzel, M. Urinary pharmacokinetics of betalains following consumption of red beet juice in healthy humans. Pharmacol. Res. 2005, 52, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Kanner, J.; Harel, S.; Granit, R. Betalains a new class of dietary cationized antioxidants. J. Agric. Food Chem. 2001, 49, 5178–5185. [Google Scholar] [CrossRef] [PubMed]
- Haytowitz, D.; Ahuja, J.; Wu, X.; Khan, M.; Somanchi, M.; Nickle, M.; Nguyen, Q.; Roseland, J.; Williams, J.; Patterson, K. USDA National Nutrient Database for Standard Reference, Legacy. In USDA National Nutrient Database for Standard Reference; The United States Department of Agriculture: Washington, DC, USA, 2018. [Google Scholar]
- Parkar, S.G.; Redgate, E.L.; McGhie, T.K.; Hurst, R.D. In vitro studies of modulation of pathogenic and probiotic bacterial proliferation and adhesion to intestinal cells by blackcurrant juices. J. Funct. Foods 2014, 8, 35–44. [Google Scholar] [CrossRef]
- Schrage, B.; Stevenson, D.; Wells, R.W.; Lyall, K.; Holmes, S.; Deng, D.; Hurst, R.D. Evaluating the health benefits of fruits for physical fitness: A research platform. J. Berry Res. 2010, 1, 35–44. [Google Scholar] [CrossRef]
- Moyer, R.A.; Hummer, K.E.; Finn, C.E.; Frei, B.; Wrolstad, R.E. Anthocyanins, phenolics, and antioxidant capacity in diverse small fruits: Vaccinium, Rubus, and Ribes. J. Agric. Food Chem. 2002, 50, 519–525. [Google Scholar] [CrossRef]
- Lätti, A.K.; Riihinen, K.R.; Kainulainen, P.S. Analysis of Anthocyanin Variation in Wild Populations of Bilberry (Vaccinium myrtillus L.) in Finland. J. Agric. Food Chem. 2008, 56, 190–196. [Google Scholar] [CrossRef]
- Wu, X.; Gu, L.; Prior, R.L.; McKay, S. Characterization of anthocyanins and proanthocyanidins in some cultivars of Ribes, Aronia, and Sambucus and their antioxidant capacity. J. Agric. Food Chem. 2004, 52, 7846–7856. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.J.; Howard, L.R.; Prior, R.L.; Clark, J.R. Flavonoid glycosides and antioxidant capacity of various blackberry, blueberry and red grape genotypes determined by high-performance liquid chromatography/mass spectrometry. J. Sci. Food Agric. 2004, 84, 1771–1782. [Google Scholar] [CrossRef]
- Gao, L.; Mazza, G. Quantitation and Distribution of Simple and Acylated Anthocyanins and Other Phenolics in Blueberries. J. Food Sci. 1994, 59, 1057–1059. [Google Scholar] [CrossRef]
- Romani, A.; Vignolini, P.; Galardi, C.; Mulinacci, N.; Benedettelli, S.; Heimler, D. Germplasm Characterization of Zolfino Landraces (Phaseolus vulgarisL.) by Flavonoid Content. J. Agric. Food Chem. 2004, 52, 3838–3842. [Google Scholar] [CrossRef] [PubMed]
- Kaack, K.; Austed, T. Interaction of vitamin C and flavonoids in elderberry (Sambucus nigraL.) during juice processing. Plant. Foods Hum. Nutr. 1998, 52, 187–198. [Google Scholar] [CrossRef]
- Gao, L.; Mazza, G. Characterization, quantitation, and distribution of anthocyanins and colorless phenolics in sweet cherries. J. Agric. Food Chem. 1995, 43, 343–346. [Google Scholar] [CrossRef]
- Romani, A.; Mulinacci, N.; Pinelli, P.; Vincieri, F.F.; Cimato, A. Polyphenolic content in five tuscany cultivars of Olea europaea L. J. Agric. Food Chem. 1999, 47, 964–967. [Google Scholar] [CrossRef]
- Kim, D.-O.; Chun, O.K.; Kim, Y.J.; Moon, H.-Y.; Lee, C.Y. Quantification of Polyphenolics and Their Antioxidant Capacity in Fresh Plums. J. Agric. Food Chem. 2003, 51, 6509–6515. [Google Scholar] [CrossRef]
- De Rosso, V.V.; Mercadante, A.Z. HPLC–PDA–MS/MS of Anthocyanins and Carotenoids from Dovyalis and Tamarillo Fruits. J. Agric. Food Chem. 2007, 55, 9135–9141. [Google Scholar] [CrossRef]
- Sadilova, E.; Stintzing, F.C.; Carle, R. Anthocyanins, Colour and Antioxidant Properties of Eggplant (Solanum melongena L.) and Violet Pepper (Capsicum annuum L.) Peel Extracts. Z. Nat. C 2006, 61, 527–535. [Google Scholar] [CrossRef]
- Welch, C.R.; Wu, Q.; Simon, J.E. Recent advances in anthocyanin analysis and characterization. Curr. Anal. Chem. 2008, 4, 75–101. [Google Scholar] [CrossRef]
- Fleschhut, J.; Kratzer, F.; Rechkemmer, G.; Kulling, S.E. Stability and biotransformation of various dietary anthocyanins in vitro. Eur. J. Nutr. 2006, 45, 7–18. [Google Scholar] [CrossRef]
- de Ferrars, R.M.; Czank, C.; Saha, S.; Needs, P.W.; Zhang, Q.; Raheem, K.S.; Botting, N.P.; Kroon, P.A.; Kay, C.D. Methods for isolating, identifying, and quantifying anthocyanin metabolites in clinical samples. Anal. Chem. 2014, 86, 10052–10058. [Google Scholar] [CrossRef]
- Roöhrig, T.; Kirsch, V.; Schipp, D.; Galan, J.; Richling, E. Absorption of anthocyanin rutinosides after consumption of a blackcurrant (Ribes nigrum L.) Extract. J. Agric. Food Chem. 2019, 67, 6792–6797. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Carlström, M.; Weitzberg, E. Metabolic Effects of Dietary Nitrate in Health and Disease. Cell Metab. 2018, 28, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Vincent, M.A.; Dawson, D.; Clark AD, H.; Lindner, J.R.; Rattigan, S.; Clark, M.G.; Barrett, E.J. Skeletal Muscle Microvascular Recruitment by Physiological Hyperinsulinemia Precedes Increases in Total Blood Flow. Diabetes 2002, 51, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Baron, A.D.; Brechtel-Hook, G.; Johnson, A.; Cronin, J.; Leaming, R.; Steinberg, H.O. Effect of perfusion rate on the time course of insulin-mediated skeletal muscle glucose uptake. Am. J. Physiol. Endocrinol. Metab. 1996, 271, E1067–E1072. [Google Scholar] [CrossRef]
- Mather, K.; Laakso, M.; Edelman, S.; Hook, G.; Baron, A. Evidence for physiological coupling of insulin-mediated glucose metabolism and limb blood flow. Am. J. Physiol. Endocrinol. Metab. 2000, 279, E1264–E1270. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-A.; Koh, K.K.; Quon, M.J. The Union of Vascular and Metabolic Actions of Insulin in Sickness and in Health. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 889–891. [Google Scholar] [CrossRef][Green Version]
- Joris, P.J.; Mensink, R.P. Beetroot juice improves in overweight and slightly obese men postprandial endothelial function after consumption of a mixed meal. Atherosclerosis 2013, 231, 78–83. [Google Scholar] [CrossRef]
- Cassidy, Y.M.; McSorley, E.M.; Allsopp, P.J. Effect of soluble dietary fibre on postprandial blood glucose response and its potential as a functional food ingredient. J. Funct. Foods 2018, 46, 423–439. [Google Scholar] [CrossRef]
- Prieto-Santiago, V.; Cavia, M.M.; Alonso-Torre, S.R.; Carrillo, C. Relationship between color and betalain content in different thermally treated beetroot products. J. Food Sci. Technol. 2020, 57, 3305–3313. [Google Scholar] [CrossRef] [PubMed]
- Herbach, K.; Stintzing, F.; Carle, R. Impact of thermal treatment on color and pigment pattern of red beet (Beta vulgaris L.) preparations. J. Food Sci. 2004, 69, C491–C498. [Google Scholar] [CrossRef]
- Olumese, F.; Oboh, H. Effects of Daily Intake of Beetroot Juice on Blood Glucose and Hormones in Young Healthy Subjects. Niger. Q. J. Hosp. Med. 2016, 26, 455–462. [Google Scholar]
- Hobbs, D.A.; Goulding, M.G.; Nguyen, A.; Malaver, T.; Walker, C.F.; George, T.W.; Methven, L.; Lovegrove, J.A. Acute ingestion of beetroot bread increases endothelium-independent vasodilation and lowers diastolic blood pressure in healthy men: A randomized controlled trial. J. Nutr. 2013, 143, 1399–1405. [Google Scholar] [CrossRef] [PubMed]
- Raubenheimer, K.; Hickey, D.; Leveritt, M.; Fassett, R.; Ortiz de Zevallos Munoz, J.; Allen, J.D.; Briskey, D.; Parker, T.J.; Kerr, G.; Peake, J.M. Acute effects of nitrate-rich beetroot juice on blood pressure, hemostasis and vascular inflammation markers in healthy older adults: A randomized, placebo-controlled crossover study. Nutrients 2017, 9, 1270. [Google Scholar] [CrossRef]
- McDonagh, S.T.; Wylie, L.J.; Webster, J.M.; Vanhatalo, A.; Jones, A.M. Influence of dietary nitrate food forms on nitrate metabolism and blood pressure in healthy normotensive adults. Nitric Oxide 2018, 72, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Asgary, S.; Afshani, M.R.; Sahebkar, A.; Keshvari, M.; Taheri, M.; Jahanian, E.; Rafieian-Kopaei, M.; Malekian, F.; Sarrafzadegan, N. Improvement of hypertension, endothelial function and systemic inflammation following short-term supplementation with red beet (Beta vulgaris L.) juice: A randomized crossover pilot study. J. Hum. Hypertens. 2016, 30, 627–632. [Google Scholar] [CrossRef]
- Ashor, A.; Jajja, A.; Sutyarjoko, A.; Brandt, K.; Qadir, O.; Lara, J.; Siervo, M. Effects of beetroot juice supplementation on microvascular blood flow in older overweight and obese subjects: A pilot randomised controlled study. J. Hum. Hypertens. 2015, 29, 511–513. [Google Scholar] [CrossRef]
- Erisna, M.; Arma, M.R.; Sumarni, S.; Triana, H. In Does Beetroot Juice Lower Blood Pressure? A Systematic Review. In Proceedings of the International Conference on Applied Science and Health, Nakhon Pathom, Thailand, 23–24 July 2019; pp. 271–281. [Google Scholar]
- Ormesher, L.; Myers, J.E.; Chmiel, C.; Wareing, M.; Greenwood, S.L.; Tropea, T.; Lundberg, J.O.; Weitzberg, E.; Nihlen, C.; Sibley, C.P. Effects of dietary nitrate supplementation, from beetroot juice, on blood pressure in hypertensive pregnant women: A randomised, double-blind, placebo-controlled feasibility trial. Nitric Oxide 2018, 80, 37–44. [Google Scholar] [CrossRef]
- Bondonno, C.P.; Liu, A.H.; Croft, K.D.; Ward, N.C.; Shinde, S.; Moodley, Y.; Lundberg, J.O.; Puddey, I.B.; Woodman, R.J.; Hodgson, J.M. Absence of an effect of high nitrate intake from beetroot juice on blood pressure in treated hypertensive individuals: A randomized controlled trial. Am. J. Clin. Nutr. 2015, 102, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Siervo, M.; Lara, J.; Jajja, A.; Sutyarjoko, A.; Ashor, A.; Brandt, K.; Qadir, O.; Mathers, J.; Benjamin, N.; Winyard, P.G. Ageing modifies the effects of beetroot juice supplementation on 24-h blood pressure variability: An individual participant meta-analysis. Nitric Oxide 2015, 47, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Coles, L.T.; Clifton, P.M. Effect of beetroot juice on lowering blood pressure in free-living, disease-free adults: A randomized, placebo-controlled trial. Nutr. J. 2012, 11, 106. [Google Scholar] [CrossRef] [PubMed]
- Bonilla Ocampo, D.A.; Paipilla, A.F.; Marín, E.; Vargas-Molina, S.; Petro, J.L.; Pérez-Idárraga, A. Dietary nitrate from beetroot juice for hypertension: A systematic review. Biomolecules 2018, 8, 134. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Mirmiran, P.; Kabir, A.; Azizi, F.; Ghasemi, A. The nitrate-independent blood pressure–lowering effect of beetroot juice: A systematic review and meta-analysis. Adv. Nutr. 2017, 8, 830–838. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, T.; Hashimoto, Y.; Kobayashi, R.; Nakazato, K.; Willems ME, T. Effects of blackcurrant extract on arterial functions in older adults: A randomized, double-blind, placebo-controlled, crossover trial. Clin. Exp. Hypertens. 2020, 42, 640–647. [Google Scholar] [CrossRef]
- Singh, A.; Verma, S.; Singh, V.; Nanjappa, C.; Roopa, N.; Raju, P.S.; Singh, S.N. Beetroot juice supplementation increases high density lipoprotein-cholesterol and reduces oxidative stress in physically active individuals. J. Pharm. Nutr. Sci. 2015, 5, 179–185. [Google Scholar] [CrossRef]
- Rabeh, M.N.; Ibrahim, E.M. Antihypercholesterolemic effects of beet (Beta vulgaris L.) root waste extract on hypercholesterolemic rats and its antioxidant potential properties. Pak. J. Nutr. 2014, 13, 500. [Google Scholar] [CrossRef][Green Version]
- Al-Dosari, M.; Alqasoumi, S.; Ahmed, M.; Al-Yahya, M.; Ansari, M.N.; Rafatullah, S. Effect of Beta vulgaris L. on cholesterol rich diet-induced hypercholesterolemia in rats. Farmacia 2011, 59, 669–678. [Google Scholar]
- Törrönen, R.; McDougall, G.J.; Dobson, G.; Stewart, D.; Hellström, J.; Mattila, P.; Pihlava, J.-M.; Koskela, A.; Karjalainen, R. Fortification of blackcurrant juice with crowberry: Impact on polyphenol composition, urinary phenolic metabolites, and postprandial glycemic response in healthy subjects. J. Funct. Foods 2012, 4, 746–756. [Google Scholar] [CrossRef]
- Schulze, C.; Bangert, A.; Kottra, G.; Geillinger, K.E.; Schwanck, B.; Vollert, H.; Blaschek, W.; Daniel, H. Inhibition of the intestinal sodium-coupled glucose transporter 1 (SGLT1) by extracts and polyphenols from apple reduces postprandial blood glucose levels in mice and humans. Mol. Nutr. Food Res. 2014, 58, 1795–1808. [Google Scholar] [CrossRef] [PubMed]
- Grussu, D.; Stewart, D.; McDougall, G.J. Berry Polyphenols Inhibit α-Amylasein Vitro: Identifying Active Components in Rowanberry and Raspberry. J. Agric. Food Chem. 2011, 59, 2324–2331. [Google Scholar] [CrossRef] [PubMed]
- Barik, S.K.; Russell, W.R.; Moar, K.M.; Cruickshank, M.; Scobbie, L.; Duncan, G.; Hoggard, N. The anthocyanins in black currants regulate postprandial hyperglycaemia primarily by inhibiting α-glucosidase while other phenolics modulate salivary α-amylase, glucose uptake and sugar transporters. J. Nutr. Biochem. 2020, 78, 108325. [Google Scholar] [CrossRef] [PubMed]
- Watson, A.W.; Haskell-Ramsay, C.F.; Kennedy, D.O.; Cooney, J.M.; Trower, T.; Scheepens, A. Acute supplementation with blackcurrant extracts modulates cognitive functioning and inhibits monoamine oxidase-B in healthy young adults. J. Funct. Foods 2015, 17, 524–539. [Google Scholar] [CrossRef]
- Törrönen, R.; Kolehmainen, M.; Sarkkinen, E.; Mykkänen, H.; Niskanen, L. Postprandial glucose, insulin, and free fatty acid responses to sucrose consumed with blackcurrants and lingonberries in healthy women. Am. J. Clin. Nutr. 2012, 96, 527–533. [Google Scholar] [CrossRef]
- Benn, T.; Kim, B.; Park, Y.-K.; Yang, Y.; Pham, T.X.; Ku, C.S.; Farruggia, C.; Harness, E.; Smyth, J.A.; Lee, J.-Y. Polyphenol-rich blackcurrant extract exerts hypocholesterolaemic and hypoglycaemic effects in mice fed a diet containing high fat and cholesterol. Br. J. Nutr. 2015, 113, 1697–1703. [Google Scholar] [CrossRef]
- Stull, A.J.; Cash, K.C.; Johnson, W.D.; Champagne, C.M.; Cefalu, W.T. Bioactives in blueberries improve insulin sensitivity in obese, insulin-resistant men and women. J. Nutr. 2010, 140, 1764–1768. [Google Scholar] [CrossRef]
- Collins, R.; Peto, R.; MacMahon, S.; Godwin, J.; Qizilbash, N.; Hebert, P.; Eberlein, K.; Taylor, J.; Hennekens, C.; Fiebach, N. Blood pressure, stroke, and coronary heart disease: Part 2, short-term reductions in blood pressure: Overview of randomised drug trials in their epidemiological context. Lancet 1990, 335, 827–838. [Google Scholar] [CrossRef]
- Bell, D.R.; Gochenaur, K. Direct vasoactive and vasoprotective properties of anthocyanin-rich extracts. J. Appl. Physiol. 2006, 100, 1164–1170. [Google Scholar] [CrossRef]
- Kim, J.N.; Han, S.N.; Ha, T.J.; Kim, H.-K. Black soybean anthocyanins attenuate inflammatory responses by suppressing reactive oxygen species production and mitogen activated protein kinases signaling in lipopolysaccharide-stimulated macrophages. Nutr. Res. Pract. 2017, 11, 357–364. [Google Scholar] [CrossRef]
- Parichatikanond, W.; Pinthong, D.; Mangmool, S. Blockade of the renin-angiotensin system with delphinidin, cyanin, and quercetin. Planta Med. 2012, 78, 1626–1632. [Google Scholar] [CrossRef] [PubMed]
- Nanashima, N.; Horie, K.; Yamanouchi, K.; Tomisawa, T.; Kitajima, M.; Oey, I.; Maeda, H. Blackcurrant (Ribes nigrum) Extract Prevents Dyslipidemia and Hepatic Steatosis in Ovariectomized Rats. Nutrients 2020, 12, 1541. [Google Scholar] [CrossRef]
- Lobo, R.A. Metabolic syndrome after menopause and the role of hormones. Maturitas 2008, 60, 10–18. [Google Scholar] [CrossRef]
- Nanashima, N.; Horie, K.; Tomisawa, T.; Chiba, M.; Nakano, M.; Fujita, T.; Maeda, H.; Kitajima, M.; Takamagi, S.; Uchiyama, D. Phytoestrogenic activity of blackcurrant (Ribes nigrum) anthocyanins is mediated through estrogen receptor alpha. Mol. Nutr. Food Res. 2015, 59, 2419–2431. [Google Scholar] [CrossRef] [PubMed]
- Nanashima, N.; Horie, K.; Maeda, H. Phytoestrogenic activity of blackcurrant anthocyanins is partially mediated through estrogen receptor beta. Molecules 2018, 23, 74. [Google Scholar] [CrossRef] [PubMed]
- Sawicki, T.; Topolska, J.; Romaszko, E.; Wiczkowski, W. Profile and content of betalains in plasma and urine of volunteers after long-term exposure to fermented red beet juice. J. Agric. Food Chem. 2018, 66, 4155–4163. [Google Scholar] [CrossRef]
- Mullen, W.; Edwards, C.A.; Crozier, A. Absorption, excretion and metabolite profiling of methyl-, glucuronyl-, glucosyl-and sulpho-conjugates of quercetin in human plasma and urine after ingestion of onions. Br. J. Nutr. 2006, 96, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Shi, J.; Xie, S.-Y.; Zhang, T.-Y.; Soladoye, O.P.; Aluko, R.M. Red beetroot betalains: Perspectives on extraction, processing, and potential health benefits. J. Agric. Food Chem. 2020, 68, 11595–11611. [Google Scholar] [CrossRef]
- Kruszewski, B.; Zawada, K.; Karpinski, P. Impact of High-Pressure homogenization parameters on physicochemical characteristics, bioactive compounds content, and antioxidant capacity of blackcurrant juice. Molecules 2021, 26, 1802. [Google Scholar] [CrossRef]
- Otalora, C.M.; Bonifazi, E.L.; Fissore, E.N.; Basanta, M.F.; Gerschenson, L.N. Thermal stability of betalains in by-products of the blanching and cutting of Beta vulgaris L. var conditiva. Pol. J. Food Nutr. Sci. 2020, 70, 15–24. [Google Scholar] [CrossRef]
- Paciulli, M.; Medina-Meza, I.G.; Chiavaro, E.; Barbosa-Cánovas, G.V. Impact of thermal and high pressure processing on quality parameters of beetroot (Beta vulgaris L.). LWT Food Sci. Technol. 2016, 68, 98–104. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).