An Overview of Metabolic Activity, Beneficial and Pathogenic Aspects of Burkholderia Spp.
Abstract
1. Genus Burkholderia
1.1. Discovering, Classification and Distribution
1.2. Species of Burkholderia
2. Plant Growth Promoting Effect of Burkholderia Spp.
3. Use of Burkholderia Spp. as Biocontrol Agent
4. Induction of Plant Systemic Resistance (ISR)
5. Burkholderia’ Diseases on Human and Animals
6. Microbial Secondary Metabolites
6.1. Secondary Metabolites Produced by Genus Burkholderia
6.1.1. Burkholderia cepacia
6.1.2. Burkholderia andropogonis
6.1.3. Burkholderia plantarii
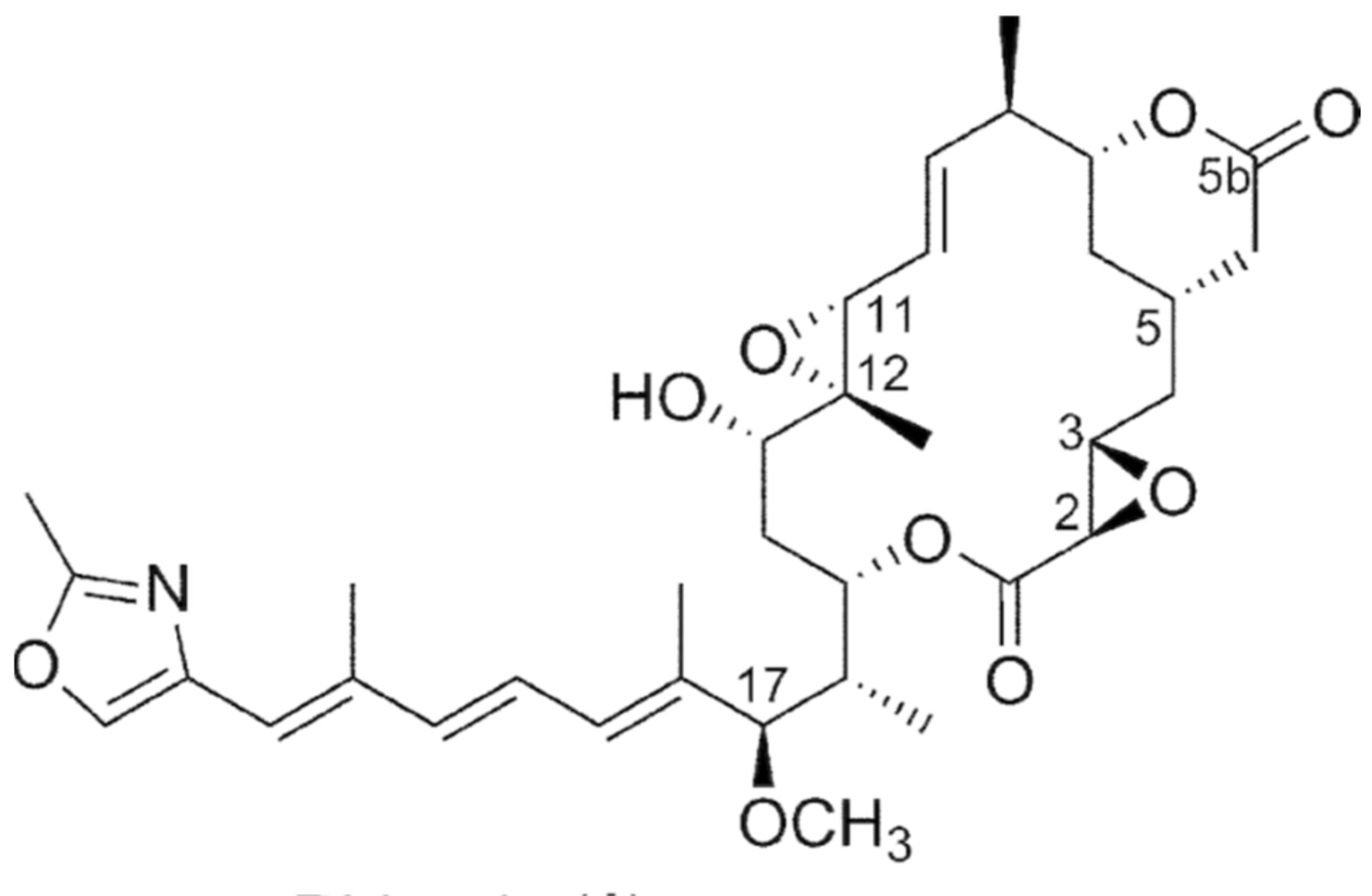
6.1.4. Burkholderia rhizoxinica
6.1.5. Burkholderia glumae
6.1.6. Burkholderia caryophylli
6.1.7. Burkholderia gladioli
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Burkholder, W.H. Three bacterial plant pathogens. Phytomonas caryophylli sp.n., Phytomonas alliicola. sp.n., and Phytomonas manihotis (Arthaud-Berthet et Sondar) Viégas. Phytopathology 1942, 32, 141–149. [Google Scholar]
- Burkholder, W.H. Sour skin, a bacterial rot of onion bulbs. Phytopathology 1950, 40, 115–117. [Google Scholar]
- Ferreira, A.S.; Silva, I.N.; Moreira, L.M. Mechanisms controlling the expression of the exopolysaccharide of Burkholderia and role in niche adaptation. Biotechnol. Biopolym. 2011, 8, 147–164. [Google Scholar]
- Wiedmann, M.; Weilmeier, D.; Dineen, S.S.; Ralyea, R.; Boor, K.J. Molecular and Phenotypic Characterization of Pseudomonas spp. Isolated from Milk. Appl. Environ. Microbiol. 2000, 66, 2085–2095. [Google Scholar] [CrossRef] [PubMed]
- Compant, S.; Kaplan, H.; Sessitsch, A.; Nowak, J.; Ait Barka, E.; Clement, C. Endophytic colonization of Vitis vinifera L. by Burkholderia phytofirmans strain PsJN: From the rhizosphere to inflorescence tissues. FEMS Microbiol. Ecol. 2008, 63, 84–93. [Google Scholar] [CrossRef]
- Yabuuchi, E.; Kosako, Y.; Oyaizu, H.; Yano, I.; Hotta, H.; Hashimoto, Y.; Ezaki, T.; Arakawa, M. Proposal of Burkholderia gen. nov. and transfer of seven species of the genus Pseudomonas homology group II to the new genus, with the type species Burkholderia cepacia (Palleroni and Holmes 1981) comb. nov. Microbiol. Immunol. 1992, 36, 1251–1275. [Google Scholar] [CrossRef] [PubMed]
- Stoyanova, M.; Pavlina, I.; Moncheva, P.; Bogatzevska, N. Biodiversity and Incidence of Burkholderia Species. Biotechnol. Biotechnol. 2007, 47, 306–310. [Google Scholar] [CrossRef]
- Compant, S.M.; Nowak, J.; Coenye, T.; Clement, C.; Ait Barka, E. Diversity and occurrence of Burkholderia spp. In the natural environment. FEMS Microbiol. Rev. 2008, 32, 607–626. [Google Scholar] [CrossRef] [PubMed]
- Graves, M.; Robin, T.; Chipman, A.M.; Wong, J.; Khashe, S.; Janda, J.M. Four additional cases of Burkholderia gladioli infection with microbiological correlates and review. Clin. Infect. 1997, 25, 838–842. [Google Scholar] [CrossRef]
- Furuya, N.; Ura, H.; Iiyama, K.; Matsumoto, M.; Takeshita, M.; Takanami, Y. Specific oligonucleotide primers based on sequences of the 16S-23S rDNA spacer region for the detection of Burkholderia gladioli by PCR. J. Gen. Plant Pathol. 2002, 68, 220–224. [Google Scholar] [CrossRef]
- Parte, A.C.; Sardà Carbasse, J.; Meier-Kolthoff, J.P.; Reimer, L.C.; Göker, M. List of prokaryotic names with standing in nomenclature (LPSN) moves to the DSMZ. Int. J. Syst. Evol. Microbiol. 2020, 70, 5607–5612. [Google Scholar] [CrossRef]
- Coenye, T.; Vandamme, P. Diversity and significance of Burkholderia species occupying diverse ecological niches. Environ. Microbiol. 2003, 5, 719–729. [Google Scholar] [CrossRef]
- Bevivino, A.; Tabacchioni, S.; Chiarini, L.; Carusi, M.V.; Del Gallo, M.; Visca, P. Phenotypic comparison between rhizosphere and clinical isolated of Burkholderia cepacia. Microbiology 1994, 140, 1069–1077. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Baldani, V.L.; Baldani, J.I.; Dobereiner, J. Inoculation of rice plants with the endophytic diazotrophs Herbaspirillum seropedicae and Burkholderia spp. Biol. Fert. Soils 2000, 30, 485–491. [Google Scholar] [CrossRef]
- Compant, S.; Duffy, B.; Nowak, J.; Clement, C.; Barka, E. Use of plant growth-promoting bacteria for biocontrol of plant diseases: Principles, mechanisms of action, and future prospects. Appl. Environ. Microbiol. 2005, 71, 4951–4959. [Google Scholar] [CrossRef]
- Meyer, J.M.; Tran van, V.; Strintzi, A.; Berge, O.; Winkelmann, G. Ornibactin production and transport proprieties in strains of Burkholderia vietnamensis and Burkholderia cepacia. Biometals 1995, 8, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.; Kang, S.C.; Maheshwari, D.K. Isolation of endophytic plant growth promoting Burkholderia sp. MSSP from root nodules of Mimosa pudica. Curr. Sci. 2005, 98, 177–180. [Google Scholar]
- Honma, M.; Shimomura, T. Metabolism of 1-aminocyclopropane-1- carboxylic acid. Agr. Biol. Chem. 1978, 42, 1825–1831. [Google Scholar]
- Estrade-de los, S.; Bustillos-Cristales, R.; Caballero-Mellado, J. Burkholderia, a genus rich in plant- associated nitrogen fixers with wide environmental and geographic distribution. Appl. Environ. Microbiol. 2001, 67, 2790–2798. [Google Scholar] [CrossRef] [PubMed]
- Karakurt, H.; Aslantas, R. Effects of some plant growth promoting Rhizobacteria (PGPR) strains on plant Growth and leaf nutrient content of apple. J. Fruit Ornamen. Plant Res. 2010, 18, 101–110. [Google Scholar]
- Barka, E.; Gognies, S.; Nowak, J.; Audran, J.C.; Belarbi, A. Inhibitory effect of endophyte bacteria on Botrytis cinerea and its influence to promote the grapevine growth. Biol. Control 2002, 24, 135–142. [Google Scholar] [CrossRef]
- Cain, C.C.; Henry, A.T.; Waldo, R.H.; Casida, L.J.; Falkinham, J.O. Identification and characteristics of a novel Burkholderia strain with broad-spectrum antimicrobial activity. Appl. Environ. Microbiol. 2000, 66, 4139–4141. [Google Scholar] [CrossRef][Green Version]
- Perin, L.; Martinez-aguilar, L.; Paredes-Valdez, G.; Baldani, J.I.; Estrada-de los, S.; Reis, V.M.; Caballero-Mellado, J. Burkholderia silvatlantica sp. nov. a diazotrophic bacterium associated with sugar cane and maize. Int. J. Syst. Evol. Micriobiol. 2006, 56, 1931–1937. [Google Scholar] [CrossRef]
- Yu, J.; Ahmedna, M.; Goktepe, I. Effects of processing methods and extraction solvents on concentration and antioxidant activity of peanut skin phenolics. Food Chem. 2005, 90, 199–206. [Google Scholar] [CrossRef]
- El-Banna, N.; Winkelmann, G. Pyrrolnitrin from Burkholderia cepacia: Antibiotic activity against fungi and novel activities against streptomycetes. J. Appl. Microbiol. 1998, 85, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Holmes, A.; Govan, J.; Goldstein, R. Agricultural use of Burkholderia (Pseudomonas) cepacia: A threat to human health. Emerg. Infect. Dis. 1998, 4, 221–227. [Google Scholar] [CrossRef]
- Kang, J.W.; Doty, S.L. Co-metabolic degradation of trichloroethylene by Burkholderia cepacia G4 with poplar leaf homogenate. Can. J. Microbiol. 2014, 60, 487–490. [Google Scholar] [CrossRef] [PubMed]
- Vandamme, P.; Opelt, K.; Knochel, N.; Berg, C.; Schonmann, S.; De Brandt, E.; Eberl, L.; Falsen, E.; Berg, G. Burkholderia bryophyla sp. nov. and Burkholderia megapolitana sp. nov., moss-associated species with antifungal and plant growthpromoting properties. Int. J. Syst. Evol. Microbiol. 2007, 57, 2228–2235. [Google Scholar] [CrossRef] [PubMed]
- Elshafie, H.S.; Camele, I.; Racioppi, R.; Scrano, L.; Iacobellis, N.S.; Bufo, S.A. In vitro antifungal activity of Burkholderia gladioli pv. agaricicola against some phytopathogenic fungi. Int. J. Mol. Sci. 2012, 13, 6291. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Racioppi, R.; Bufo, S.A.; Camele, I. In vitro study of biological activity of four strains of Burkholderia gladioli pv. agaricicola and identification of their bioactive metabolites using GC–MS. Saudia J. Biol. Sci. 2017, 24, 295–301. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Viggiani, L.; Mostafa, M.S.; El-Hashash, M.A.; Bufo, S.A.; Camele, I. Biological activity and chemical identification of ornithine lipid produced by Burkholderia gladioli pv. agaricicola ICMP 11096 using LC-MS and NMR analyses. J. Biol. Res. 2017, 9, 96–103. [Google Scholar] [CrossRef]
- Walker, R.; Innes, C.M.; Allan, E.J. The potential biocontrol agent Pseudomonas antimicrobica inhibits germination of conidia and outgrowth of Botrytis cinerea. Lett. Appl. Microbiol. 2001, 32, 346–348. [Google Scholar] [CrossRef]
- Altindag, M.; Sahin, M.; Esitken, A.; Ercisli, S.; Guleryuz, M.; Donmez, M.F. Biological control of brown rot (Moniliana laxa Ehr.) on apricot (Prunus armeniaca L. cv. Hacihaliloglu) by Bacillus, Burkholderia and Pseudomonas application under in vitro and in vivo conditions. Biol. Control 2006, 38, 369–372. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Sakr, S.; Bufo, S.A.; Camele, I. An attempt of biocontrol the tomato-wilt disease caused by Verticillium dahliae using Burkholderia gladioli pv. agaricicola and its bioactive secondary metabolites. Int. J. Plant Biol. 2017, 8, 57–60. [Google Scholar] [CrossRef][Green Version]
- Van Loon, L.C. Plant responses to plant growth-promoting rhizobacteria. Eur. J. Plant Pathol. 2007, 119, 243–254. [Google Scholar] [CrossRef]
- Van Peer, R.; Niemann, G.J.; Schippers, B. Induced resistance and phytoalexin accumulation in biological control of fusarium wilt of carnation by Pseudomonas sp. strain WCS417r. Phytopathology 1991, 81, 728–734. [Google Scholar] [CrossRef]
- Zhang, S.; Moyne, A.L.; Reddy, M.S.; Kloepper, J.W. The role of salicylic acid in induced systemic resistance elicited by plant growth promoting rhizobacteria against blue mould of tobacco. Biol. Control 2002, 25, 288–296. [Google Scholar] [CrossRef]
- Wang, Y.; Zeng, Q.; Zhang, Z.; Yan, R.; Zhu, D. Antagonistic bioactivity of an endophytic bacterium H- 6. Afr. J. Biotechnol. 2010, 9, 6140–6145. [Google Scholar]
- Compant, S.; Reiter, B.; Sessitsch, A.; Nowak, J.; Clement, C.; Barka, E. Endophytic colonization of Vitis vinifera L. by plant growth-promoting bacterium Burkholderia sp. strain PsJN. Appl. Environ. Microbiol. 2005, 71, 1685–1693. [Google Scholar] [CrossRef]
- Sharma, V.K.; Nowak, J. Enhancement of verticillium wilt resistance in tomato transplants by in vitro co-culture of seedlings with a plant growth-promoting rhizobacterium (Pseudomonas sp. strain PsJN). Can. J. Microbiol. 1998, 44, 528–536. [Google Scholar] [CrossRef]
- Bordiec, S.; Paquis, S.; Lacroix, H.; Dhond, S.; Barka, E.A.; Kauffmann, S.; Jeandet, P.; Gourbeyre, F.M.; Clement, C.; Baillieul, F.; et al. Comparative analysis of defence responses induced by the endophytic plant growth-promoting rhizobacterium Burkholderia phytofirmans strain PsJN and the non-host bacterium Pseudomonas syringae pv. pisi in grapevine cell suspensions. J. Exp. Bot. 2010, 62, 595–603. [Google Scholar] [CrossRef]
- Chain, P.S.G.; Denef, V.J.; Konstantinidis, K.T.; Vergez, L.M.; Agulló, L.; Reyes, V.L.; Hauser, L.; Córdova, M.; Gómez, L.; González, M.; et al. Burkholderia xenovorans LB400 harbors a multi-replicon, 9.73-Mbp genome shaped for versatility. Proc. Natl. Acad. Sci. USA 2006, 103, 15280–15287. [Google Scholar] [CrossRef]
- European and Mediterranean Plant Protection Organization. Burkholderia caryophylli. OEPP/EPPO Bull. 2006, 36, 95–98.
- OEPP/EPPO, EPPO Standard PP 2/1(1) Guideline on good plant protection practice: Principles of good plant protection practice. OEPP/EPPO Bull. 1994, 24, 233–240. [CrossRef]
- Dance, D.A.B. Melioidosis And Glanders as Possible Biological Weapons. In Bioterrorism and Infectious Agents: A New Dilemma for the 21st Century; Fong, I.W., Alibek, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 99–145. [Google Scholar]
- Whitlock, G.C.; Estes, D.M.; Torres, A.G. Glanders: Off to the races with Burkholderia mallei. FEMS Microbiol. Lett. 2007, 277, 115–122. [Google Scholar] [CrossRef]
- Ngamdee, W.; Tandhavanant, S.; Wikraiphat, C.; Reamtong, O.; Wuthiekanun, V.; Salje, J.; Low, D.A.; Peacock, S.J.; Chantratita, N. Competition between Burkholderia pseudomallei and B. thailandensis. BMC Microbiol. 2015, 15, 56. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mahenthiralingam, E.; Urban, T.A.; Goldberg, J.B. The multifarious, multireplicon Burkholderia cepacia complex. Nat. Rev. Microbiol. 2005, 3, 144–156. [Google Scholar] [CrossRef] [PubMed]
- Lipuma, J.J. Update on the Burkholderia cepacia complex. Curr. Opin. Pulm. Med. 2005, 11, 528–533. [Google Scholar] [CrossRef]
- Peeters, C.; Zlosnik, J.E.; Spilker, T.; Hird, T.J.; LiPuma, J.J.; Vandamme, P. Burkholderia pseudomultivorans sp. nov., a novel Burkholderia cepacian complex species from human respiratory samples and the rhizosphere. Syst. Appl. Microbiol. 2013, 36, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Bourgaud, F.; Gravot, A.; Milesi, S.; Gontier, E. Production of plant secondary metabolites: A historical perspective. Plant Sci. 2001, 161, 839–851. [Google Scholar] [CrossRef]
- Wright, J.L. Attack and Defend: The Function and Evolution of Bioactive or Toxic Metabolites. In Proceedings of the Xth International Conference on Harmful Algae, St. Petersburg, FL, USA, 21–25 October 2002. [Google Scholar]
- Croteau, R.; Kutchan, T.M.; Lewis, N.G. “Chapter 24: Natural Products (Secondary Metabolites)”. In Natural Products in Chemical Biology; Civjan, N., Ed.; Wiley: Hoboken, NJ, USA, 2012; pp. 1250–1319. ISBN 978-1-118-10117-9. [Google Scholar]
- Sanchez, S.; Demain, A.L. Comprehensive Biotechnology, 3rd ed.; Moo-Young, M., Ed.; Pergamon: Oxford, UK, 2011; pp. 131–143. [Google Scholar]
- Camele, I.; Elshafie, H.S.; Caputo, L.; Sakr, S.H.; De Feo, V. Bacillus mojavensis: Biofilm formation and biochemical investigation of its bioactive metabolites. J. Biol. Res. 2019, 92, 39–45. [Google Scholar] [CrossRef]
- Samson, R.A.; Pitt, J.I. Integration of Modern Taxonomic Methods for Penicillium and Aspergillus Classification; CRC Press: Boca Raton, FL, USA, 2000; p. 66. [Google Scholar]
- Subhan, M.; Faryal, R.; Macreadie, I. Exploitation of Aspergillus terreus for the Production of Natural Statins. J. Fungi 2016, 2, 13. [Google Scholar] [CrossRef]
- Panda, B.; Javed, S.; Ali, M. Optimization of fermentation parameters for higher lovastatin production in red mold rice through co-culture of Monascus purpureus and Monascus ruber. Food Bioprocess Technol. 2010, 3, 373–378. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef]
- Mast, Y.; Stegmann, E. Actinomycetes: The Antibiotics Producers. Antibiotics 2019, 8, 105. [Google Scholar] [CrossRef]
- Hu, F.; Young, J.M. Biocidal activity in plant pathogenic Acidovorax, Burkholderia, Herbaspirillum, Ralstonia, and Xanthomonas sp. J. Appl. Microbiol. 1998, 84, 263–271. [Google Scholar] [CrossRef]
- Depoorter, E.; Bull, M.J.; Peeters, C.; Coenye, T.; Vandamme, P.; Mahenthiralingam, E. Burkholderia: An update on taxonomy and biotechnological potential as antibiotic producers. Appl. Microbiol. Biotechnol. 2016, 100, 5215–5229. [Google Scholar] [CrossRef] [PubMed]
- Kunakom, S.; Eustaquio, A.S. Burkholderia as a source of natural products. J. Nat. Prod. 2019, 82, 2018–2037. [Google Scholar] [CrossRef] [PubMed]
- Al-ahmadi, J.K.; Yazdi, T.M.; Najafi, F.M.; Shahverdi, A.R.; Faramarzi, M.A.; Zarrini, G.; Behravan, J. Optimization of medium and cultivation conditions for chitinase production by the newly isolated: Aeromonas sp. Biotechnology 2008, 7, 266–272. [Google Scholar]
- Wang, S.; Hwang, J. Microbial reclamation of shellfish wastes for the production of chitinases. Enzyme. Microb. Technol. 2001, 28, 376–382. [Google Scholar] [CrossRef]
- Roitman, J.N.; Mahoney, N.E.; Janisiewicz, W.J. Production and composition of phenylpyrrole metabolites produced by Pseudomonas cepacia. Appl. Microbiol. Biotechnol. 1990, 34, 381–386. [Google Scholar] [CrossRef]
- Lavermicocca, P.; Iacobellis, N.S.; Simmaco, M.; Graniti, A. Biological properties and spectrum of activity of Pseudomonas syringae pv. syringae toxins. Physiol. Mol. Plant Pathol. 1997, 50, 129–140. [Google Scholar] [CrossRef]
- Cartwright, D.K.; Chilton, W.S.; Benson, D.M. Pyrrolnitrin and phenazine production by Pseudomonas cepacia, strain 5.5B, a biocontrol agent of Rhizoctonia solani. Appl. Microbiol. Biotechnol. 1995, 43, 211–216. [Google Scholar] [CrossRef]
- Liu, X.; Bimerew, M.; Ma, Y.; Muller, H.; Ovadis, M.; Eberl, L.; Berg, G.; Chernin, L. Quorum-sensing signaling is required for production of the antibiotic pyrrolnitrin in a rhizospheric biocontrol strain of Serratia plymuthica. FEMS Microbiol. Lett. 2007, 270, 299–305. [Google Scholar] [CrossRef]
- Rahman, M.A.; Mahmud, T.M.; Kadir, J.; Abdul Rahman, R.; Begum, M.M. Screening of antagonistic bacteria for biocontrol activities on Colletotrichum gloeosporioides in papaya. Asian J. Plant Sci. 2007, 6, 12–20. [Google Scholar] [CrossRef]
- Arima, K.; Imanaki, H.; Kousaka, M.; Fukuda, A.; Tamura, G. Studies on pyrrolnitrin, a new antibiotic. I. Isolation and properties of pyrrolnitrin. J. Antibiot. 1965, 8, 201–204. [Google Scholar]
- Arima, K.; Imanaki, H.; Kousaka, M.; Fukuta, A.; Tamura, G. Pyrrolnitrin, a new antibiotic substance, produced by Psneudomonas. Agr. Biol. Chem. 1964, 28, 575–576. [Google Scholar] [CrossRef]
- Lee, C.; Kim, S.; Hyun, B.; Suh, J.; Yon, C.; Kim, C.; Lim, Y.; Kim, C. Cepacidine A, a novel antifungal antibiotic produced by Pseudomonas cepacia. J. Antibiot. 1994, 47, 1402–1405. [Google Scholar] [CrossRef] [PubMed]
- Parker, W.L.; Rathnum, M.L.; Seiner, V.; Trejo, W.H.; Principe, P.A.; Sykes, R.B. Cepacin A and Cepacin B, two new antibiotics produced by Pseudomonas cepacia. J. Antibiot. 1984, 37, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Santos-Villalobos, S.; Barrera-Galicia, G.C.; Miranda-Salcedo, M.A.; Peña-Cabriales, J.J. Burkholderia cepacia XXVI siderophore with biocontrol capacity against Colletotrichum gloeosporioides. World. J. Microbiol. Biotechnol. 2012, 28, 2615–2623. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.F. Bacteria in relation to plant diseases. Carnegie Inst Washington Publ. 1911, 2, 1–368. [Google Scholar]
- Takahashi, Y.; Takahashi, K.; Watanabe, K.; Kawano, T. Bacterial black spot caused by Burkholderia andropogonis on Ondontoglossum and intergeneric hybrid orchids. J. Gen. Plant Pathol. 2004, 70, 284–287. [Google Scholar] [CrossRef]
- Gillis, M.; Van, T.; Bardin, R.; Goor, M.; Hebbar, P.; Willems, A.; Segers, P.; Kersters, K.; Heulin, T.; Fernandez, M.P. Polyphasic taxonomy in the genus Burkholderia leading to an emended description of the genus and proposition of Burkholderia vietnamiensis sp. nov. for N2 Fixing isolates from rice in Vietnam. Int. J. Syst. Bacteriol. 1995, 45, 274–289. [Google Scholar] [CrossRef]
- Vidaver, A.K.; Carlson, R.R. Leaf spot of field corn caused by Pseudomonas andropogonis. Plant Dis. Rep. 1978, 62, 213–216. [Google Scholar]
- Coenye, T.; Laevens, S.; Gillis, M.; Vandamme, P. Genotypic and chemotaxonomic evidence for the reclassification of Pseudomonas woodsii (Smith 1911) Stevens 1925 as Burkholderia andropogonis (Smith 1911) Gillis et al. 1995. Int. J. Syst. Evol. Microbiol. 2001, 51, 183–185. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Owens, L.D.; Thompson, J.F.; Pitcher, R.G.; Williams, T. Structure of rhizobitoxine, an antimetabolic enol-ether amino acid from Rhizobium japonicum. J. Chem. Soc. Chem. Commun. 1972, 12, 714. [Google Scholar] [CrossRef]
- Owens, L.D.; Lieberman, M.; Kunishi, A. Inhibition of ethylene production by rhizobitoxine. Plant Physiol. 1971, 48, 1–4. [Google Scholar] [CrossRef]
- Sugawara, M.; Okazaki, S.; Nukui, N.; Ezura, H.; Mitsui, H.; Minamisawa, K. Rhizobitoxine modulates plant-microbe interactions by ethylene inhibition. Biotech. Adv. 2006, 24, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Duodu, S.; Bhuvaneswari, T.V.; Stokkermans, T.J.W.; Peters, N.K. A Positive role for rhizobitoxine in Rhizobium-legume symbiosis. Mol. Plant-Microbe Interact. 1999, 12, 1082–1089. [Google Scholar] [CrossRef]
- Yasuta, T.; Satoh, S.; Minamisawa, K. New assay for rhizobitoxine based on inhibition of 1-Aminocyclopropane-1-Carboxylate Synthase. Appl. Environ. Microbiol. 1999, 65, 849–852. [Google Scholar] [CrossRef] [PubMed]
- Azegami, K.; Nishiyama, K.; Kato, H. Effect of iron limitation on Pseudomonas plantarii growth and tropolone and protein production. Appl. Environ. Microbiol. 1988, 54, 844–847. [Google Scholar] [CrossRef] [PubMed]
- Trust, T.J. Antibacterial Activity of Tropolone. Antimicrob. Agen. Chemoth. 1975, 7, 500–506. [Google Scholar] [CrossRef]
- Mitchell, R.E.; Katrina, L.T. Antibacterial iminopyrrolidines from Burkholderia plantarii, a bacterial pathogen of rice. Org. Biomol. Chem. 2005, 3, 3540–3543. [Google Scholar] [CrossRef]
- Hörmann, B.; Müller, M.M.; Syldatk, C.; Hausmann, R. Rhamnolipid production by Burkholderia plantarii DSM 9509T. Eur. J. Lipid Sci. Technol. 2010, 112, 674–680. [Google Scholar] [CrossRef]
- Laila, P.; Hertweck, C. Pathogenic fungus harbours endosymbiotic bacteria for toxin production. Nature 2005, 437, 884–888. [Google Scholar]
- Partida-Martinez, L.P.; Groth, I.; Schmitt, I.; Richter, W.; Roth, M.; Hertweck, C. Burkholderia rhizoxinica sp. nov. and Burkholderia endofungorum sp. nov., bacterial endosymbionts of the plant-pathogenic fungus Rhizopus microsporus. Int. J. Sys. Evolut. Microbiol. 2007, 57, 2583–2590. [Google Scholar] [CrossRef]
- Sawana, A.; Adeolu, M.; Gupta, R.S. Molecular signatures and phylogenomic analysis of the genus Burkholderia: Proposal for division of this genus into the emended genus Burkholderia containing pathogenic organisms and a new genus Paraburkholderia gen. nov. harboring environmental species. Front. Genet. 2014, 5, 429. [Google Scholar] [CrossRef]
- Jennessen, J.; Nielsen, K.F.; Houbraken, J.; Lyhne, E.K.; Schnrer, J.; Frisvad, J.C.; Samson, R.A. Secondary metabolite and mycotoxin production by the Rhizopus microsporus group. J. Agric. Food Chem. 2005, 53, 1833–1840. [Google Scholar] [CrossRef]
- Gee, J.E.; Glass, M.B.; Lackner, G.; Helsel, L.O.; Daneshvar, M.; Hollis, D.G.; Jordan, J.; Morey, R.; Steigerwalt, A.; Hertweck, C. Characterization of Burkholderia rhizoxinica and Burkholderia endofungorum isolated from clinical specimens. PLoS ONE 2011, 6, e15731. [Google Scholar] [CrossRef] [PubMed]
- Lackner, G.; Moebius, N.; Partida-Martinez, L.; Hertweck, C. Complete genome sequence of Burkholderia rhizoxinica, an endosymbiont of Rhizopus microspores. J. Bacteriol. 2011, 193, 783–784. [Google Scholar] [CrossRef] [PubMed]
- Ikubo, S.; Takigawa, N.; Ueoka, H.; Kiura, K.; Tabata, M.; Shibayama, T.; Chikamori, M.; Aoe, K.; Matsushita, A.; Harada, M. In vitro evaluation of antimicrotubule agents in human small-cell lung cancer cell lines. Anticancer Res. 1999, 19, 3985–3988. [Google Scholar]
- Loper, J.E.; Henkels, M.D.; Shaffer, B.; Valeriote, F.A.; Gross, H. Isolation and identification of rhizoxin analogs from Pseudomonas fluorescens Pf-5 by using a genomic mining strategy. App. Environ. Microbiol. 2008, 74, 3085–3093. [Google Scholar] [CrossRef]
- Partida-Martinez, L.P.; Hertweck, C. A gene cluster encoding rhizoxin biosynthesis in “Burkholderia rhizoxina”,the bacterial endosymbiont of the fungus Rhizopus Microsporus. Chem. Biochem. 2007, 8, 41–45. [Google Scholar]
- Agbagala, M.L. Occurrence and Survival of Burkholderia glumae (Kurita and Tabei, Comb. nov.) Yabuuchi et.al. in Rice (Oryza sativa L.) Seeds. Master’s Thesis, University of Philippines, Los Baños, Laguna, Philippines, 1997. [Google Scholar]
- Pedraza, L.A.; Bautista, J.; Uribe-Vélez, D. Seed-born Burkholderia glumae infects rice seedling and maintains bacterial population during vegetative and reproductive growth stage. Plant Pathol. J. 2018, 34, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Yeonhwa, J.; Jinwoo, K.; Suhyun, K.; Yongsung, K.; Tomohisa, N.; Tsushima, O. Toxoflavin Produced by Burkholderia glumae causing rice grain rot is responsible for inducing bacterial wilt in many field crops. Plant Dis. 2007, 87, 890–895. [Google Scholar]
- Lee, J.; Park, J.; Kim, S.; Park, I.; Seo, Y. Differential regulation of toxoflavin production and its role in the enhanced virulence of Burkholderia gladioli. Mol. Plant Pathol. 2016, 17, 65–76. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J.G.; Kang, Y.; Nagamatsu, T.; Hwang, I. Quorum sensing and the Lys-R type transcriptional activator ToxR regulate toxoflavin biosynthesis and transport in Burkholderia glumae. Mol. Microbiol. 2004, 54, 21–34. [Google Scholar] [CrossRef]
- Kusumi, T.; Ohtani, I.; Nishiyamaa, K.; KLakisawa, H. Caryoynencins, potent antibiotics from a plant pathogen Pseudomonas caryophylli. Tetrahedron Lett. 1987, 34, 3981–3984. [Google Scholar] [CrossRef]
- Gregory, C.L. Carnation bacterial wilt in Indiana. USDA Plant Dis. Rep. 1942, 26, 413. [Google Scholar]
- Scientific Opinion on the risk to plant health posed by Burkholderia caryophylli for the EU territory with the identification and evaluation of risk reduction options. EFSA J. 2013, 11, 3071. [CrossRef]
- Yamaguchi, M.; Park, H.; Ishizuka, S.; Omata, K.; Hirama, M. Chemistry and antimicrobial activity of caryoynencins analogs. Afric. J. Biotechnol. 1995, 7, 2227–2231. [Google Scholar] [CrossRef] [PubMed]
- Coenye, T.; Vandamme, P. Burkholderia: Molecular Microbiology and Genomics; Horizon Bioscience: Norfolk, UK, 2007; pp. 203–228. [Google Scholar]
- Chowdhury, P.R.; Heinemann, J.A. The General secretory pathway of Burkholderia gladioli pv. agaricicola BG164R is necessary for cavity disease in white button mushrooms . Appl. Environ. Microbiol. 2006, 72, 3558–3565. [Google Scholar]
- Wilsher, M.L.; Kolbe, J.; Morris, A.J.; Welch, D.F. Nosocomial acquisition of Burkholderia gladioli in patients with cystic fibrosis. Am. J. Resp. Crit. Care Med. 1997, 156, 1436–1440. [Google Scholar] [CrossRef]
- UK Cystic Fibrosis Trust Infection Control Group. The Burkholderia Cepacia Complex. In Suggestions for Prevention and Infection Control, 2nd ed.; 2004; p. 32.
- Henry, D.A.; Mahenthiralingam, E.; Vandamme, P.; Coenye, T.; Speert, D.P. Phenotypic methods for determining genomovar status of the Burkholderia cepacia Complex. J. Clin. Microbiol. 2001, 39, 1073–1078. [Google Scholar] [CrossRef][Green Version]
- Bradbury, J. Guide to Plant Pathogenic Bacteria; CAB International: Wallingford, UK, 1986. [Google Scholar]
- Jiao, Z.; Kawamura, Y.; Mishima, N.; Yang, R.; Li, N.; Liu, X.; Ezaki, T. Need to differentiate lethal toxin-producing strains of Burkholderia gladioli, which cause severe food poisoning: Description of Burkholderia gladioli pv. cocovenenans and an emended description of Burkholderia gladioli. Microbiol. Immunol. 2003, 47, 915–925. [Google Scholar] [CrossRef]
- Uchida, J. Bacterial Diseases of Dendrobium; HITAHR, College of Tropical Agriculture and Human Resources, University of Hawaii: Honolulu, HI, USA, 1995; p. 158. [Google Scholar]
- Keith, L.M.; Kewake, S.; Zee, F.T. Isolation and characterization of Burkholderia gladioli from orchids in Hawaii. Plant Dis. 2005, 89, 1273–1278. [Google Scholar] [CrossRef] [PubMed]
- Ura, H.; Furuya, N.; Iiyama, K.; Hidaka, M.; Tsuchiya, K.; Matsuyama, N. Burkholderia gladioli associated with symptoms of bacterial grain rot and leaf- sheath browning of rice plants. J. Gen. Pl. Pathol. 2006, 72, 98–103. [Google Scholar] [CrossRef]
- Scortichini, M. Pseudomonas gladioli pv. gladioli causal agent of bacterial scab of Gladiolus in Lazio. Informatore Fitopatologico 1993, 43, 49–51. [Google Scholar]
- Chase, A.R.; Miller, J.W.; Jones, J.B. Leaf spot and blight of Asplenium nidus caused by Pseudomonas gladioli. Plant Dis. 1984, 68, 344–347. [Google Scholar] [CrossRef]
- Lamovšek, J.; Gerič Stare, B.; Žerjav, M.; Urek, G. Soft rot of onion bulbs caused by Burkholderia gladioli pv. alliicola in slovenia. J. Plant Pathol. 2016, 98, 369–377. [Google Scholar]
- Wong, J.W.; Lai, K.M.; Wan, C.K.; Ma, K.K.; Fang, M. Isolation and optimisation of PAH-degradative bacteria from contaminated soil for PAH bioremediation. Water Air Soil Pollut. 2002, 139, 1–13. [Google Scholar] [CrossRef]
- Levenberg, B.; Linton, S.N. On biosynthesis of toxoflavin, an azapteridine antibiotic produced by Pseudomonas cocovenenans. J. Biolog. Chem. 1966, 241, 846–852. [Google Scholar] [CrossRef]
- Gill, W.M. Bacterial diseases of Agaricus Mushrooms. Rep. Tottori. Mycol. Inst. 1995, 33, 34–55. [Google Scholar]
- Elshafie, H.S.; Devescovi, G.; Venturi, V.; Camele, I.; Bufo, S.A. Study of the regulatory role of N-Acyl homoserine lactones mediated quorum sensing in the biological activity of Burkholderia gladioli pv. agaricicola causing soft rot of Agaricus spp. Front. Microbiol. 2019, 10, 2695. [Google Scholar] [CrossRef] [PubMed]
- Gill, W.M.; Tsuneda, A. The interaction of the soft rot bacterium Pseudomonas gladioli pv. agaricicola with Japanese cultivated mushrooms. Can. J. Microbiol. 1997, 43, 639–648. [Google Scholar] [CrossRef]
- Chowdhury, P.R. Biology and Genetic Characterization of Burkholderia gladioli pv. agaricicola, the Causal Organism of ‘Cavity Disease’ of White Button Mushrooms. Ph.D. Thesis, School of Biological Sciences at the University of Canterbury, Christchurch, New Zealand, 2004; p. 208. [Google Scholar]
- Lunch, K.H.; Dennis, J.J. Burkholderia. In Molecular Detection of Foodborn Pathogens; Taylor and Francis Group, Liu, D., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 331–340. [Google Scholar]
- Steindler, L.; Venturi, V. Detection of quorum-sensing N-acyl homoserine lactone signal molecules by bacterial biosensors. FEMS Microbiol. Lett. 2007, 266, 1–9. [Google Scholar] [CrossRef]
- Camele, I.; Elshafie, H.S.; De Feo, V.; Caputo, L. Anti-quorum sensing and antimicrobial effect of mediterranean plant essential oils against phytopathogenic bacteria. Front. Microbiol. 2019, 10, 2019. [Google Scholar]
- Withers, H.; Swift, S.; Williams, P. Quorum sensing as an integral component of gene regulatory networks in Gram-negative bacteria. Curr. Opin. Microbiol. 2001, 4, 186–193. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Bufo, S.A.; Racioppi, R.; Camele, I. Biochemical characterization of volatile secondary metabolites produced by Burkholderia gladioli pv. agaricicola. Int. J. Drug Discov. 2013, 5, 181–184. [Google Scholar]
- Ayoola, G.A.; Johnson, O.O.; Adelowotan, T.; Aibinu, I.E.; Adenipekun, E.; Adepoju-Bello, A.A.; Coker, H.A.; Odugbemi, T.O. Evaluation of the chemical constituents and the antimicrobial activity of the volatile oil of Citrus reticulata fruit (Tangerine fruit peel) from South West Nigeria. Afr. J. Biotechnol. 2008, 7, 2227–2231. [Google Scholar]
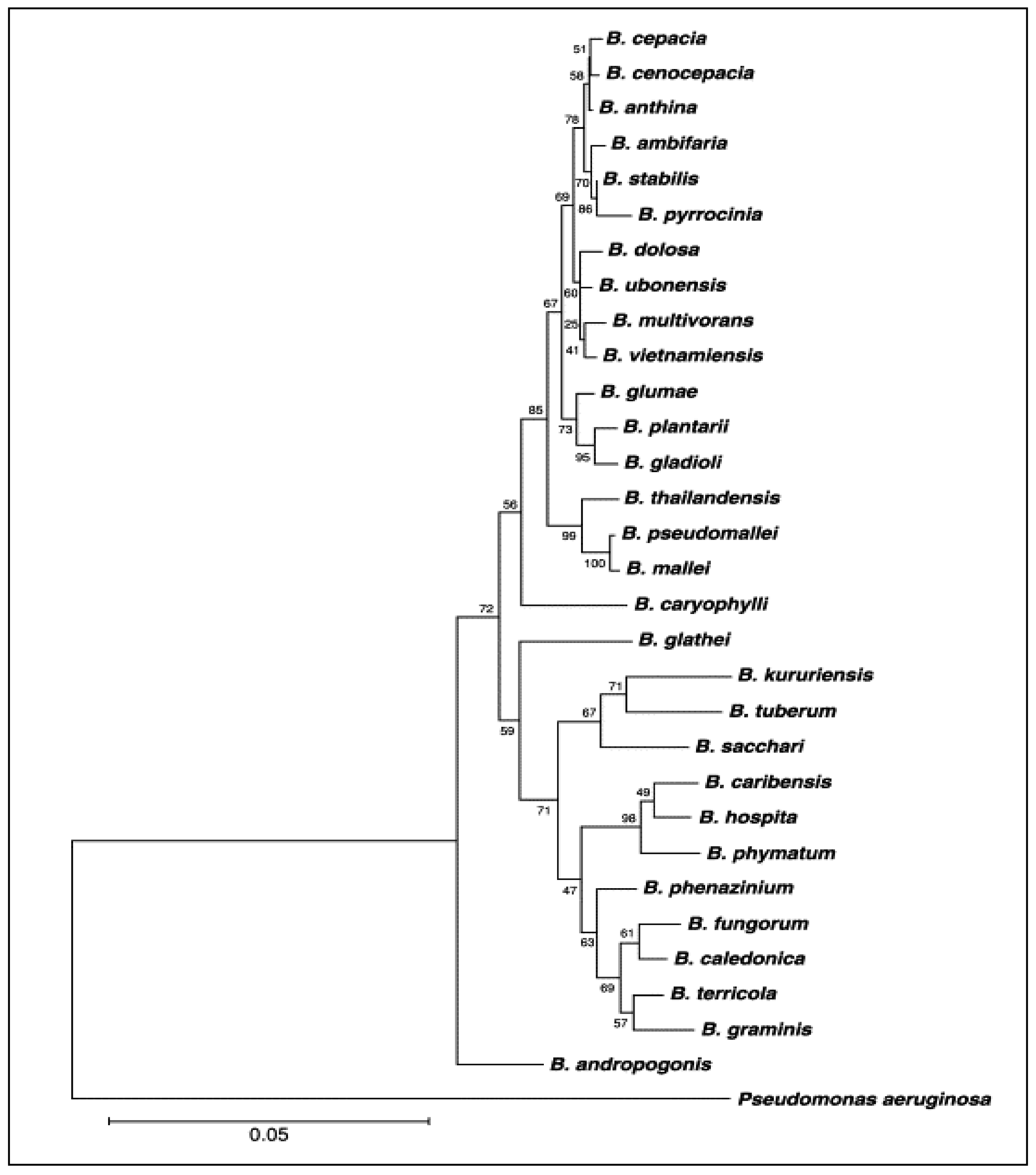


| Species of Burkholderia | Author | Year of Description | Disease | Host | |
|---|---|---|---|---|---|
| 1 | B. alpina | Weber and King | 2017 | - | - |
| 2 | B. ambifaria | Coenye et al. | 2001 | belongs to B. cepacia complex | |
| 3 | B. andropogonis | Smith (Gillis et al.) | 1911, 1995 | bacterial leaf stripe | sorghum and corn |
| 4 | B. anthina | Vandamme et al. | 2002 | belongs to B. cepacia complex | |
| 5 | B. arboris | Vanlaere et al. | 2008 | belongs to B. cepacia complex | |
| 6 | B. caryophylli | Burkholder (Yabuuchi et al.) | 1942, 1993 | wilt, stem cracking and rot of stems and roots | carnation |
| 7 | B. cenocepacia | Vandamme et al. | 2003 | cystic fibrosis | humans |
| 8 | B. cepacia | Palleroni and Holmes (Yabuuchi et al.) | 1981, 1993 |
cystic fibrosis soft-rotting |
humans onion |
| 9 | B. contaminans | Vanlaere et al. | 2009 | belongs to B. cepacia complex | |
| 10 | B. diffusa | Vanlaere et al. | 2008 | belongs to B. cepacia complex | |
| 11 | B. dolosa | Vermis et al. | 2004 | belongs to B. cepacia complex | |
| 12 | B. gladioli | Severini (Yabuuchi et al.) | 1931, 1993 | a. Scabdisease b. severe pulmonary infections c. soft rot | - gladiolus corms - humans - mushroom |
| 13 | B. glumae | Kurita and Tabei (Urakami et al.) | 1967, 1994 | panicle blight | rice |
| 14 | B. humptydooensis | Vanlaere et al. | 2009 | melioidosis disease | humans and animals |
| 15 | B. lata | Vanlaere et al. | 2009 | belongs to B. cepacia complex | |
| 16 | B. latens | Vanlaere et al. | 2008 | belongs to B. cepacia complex | |
| 17 | B. mallei | Zopf (Yabuuchi et al.) | 1885, 1993 | glanders disease | animals |
| 18 | B. metallica | Vanlaere et al. | 2008 | belongs to B. cepacia complex | |
| 19 | B. multivorans | Vandamme et al. | 1997 | belongs to B. cepacia complex | |
| 20 | B. oklahomensis | Glass et al. | 2006 | melioidosis | humans |
| 21 | B. plantarii | Azegami et al. (Urakami et al.) | 1987, 1994 | seedling blight | rice |
| 22 | B. pseudomallei | Whitmore (Yabuuchi et al.) | 1913, 1993 | melioidosis disease | humans and animals |
| 23 | B. pseudomultivorans | Peeters et al. | 2014 | belongs to B. cepacia complex | |
| 24 | B. puraquae | Martina et al. | 2018 | belongs to B. cepacia complex | |
| 25 | B. pyrrocinia | Imanaka et al. (Vandamme et al.), (Storms et al.) | 1965, 1997, 2004 | cystic fibrosis | humans |
| 26 | B. rhizoxinica | Partida-Martinez et al. | 2007 | rice seedling blight, associated with Rhizopus microsporus | rice |
| 27 | B. seminalis | Vanlaere et al. | 2008 | belongs to B. cepacia complex | |
| 28 | B. singaporensis | Wang et al. | 2003 | - | - |
| 29 | B. singularis | Vandamme et al. | 2017 | respiratory system disease | humans |
| 30 | B. stabilis | Vandamme et al. | 2000 | belongs to B. cepacia complex | |
| 31 | B. stagnalis | De Smet et al. | 2015 | B. stagnalis | |
| 32 | B. territorii | De Smet et al. | 2015 | belongs to B. cepacia complex | |
| 33 | B. thailandensis | Brett et al. | 1998 | melioidosis disease | humans and animals |
| 34 | B. ubonensis | Yabuuchi et al. | 2000 | - | - |
| 35 | B. vietnamiensis | Gillis et al. | 1995 | cystic fibrosis | humans |
| No. | Species | Synthesized Metabolites | References |
|---|---|---|---|
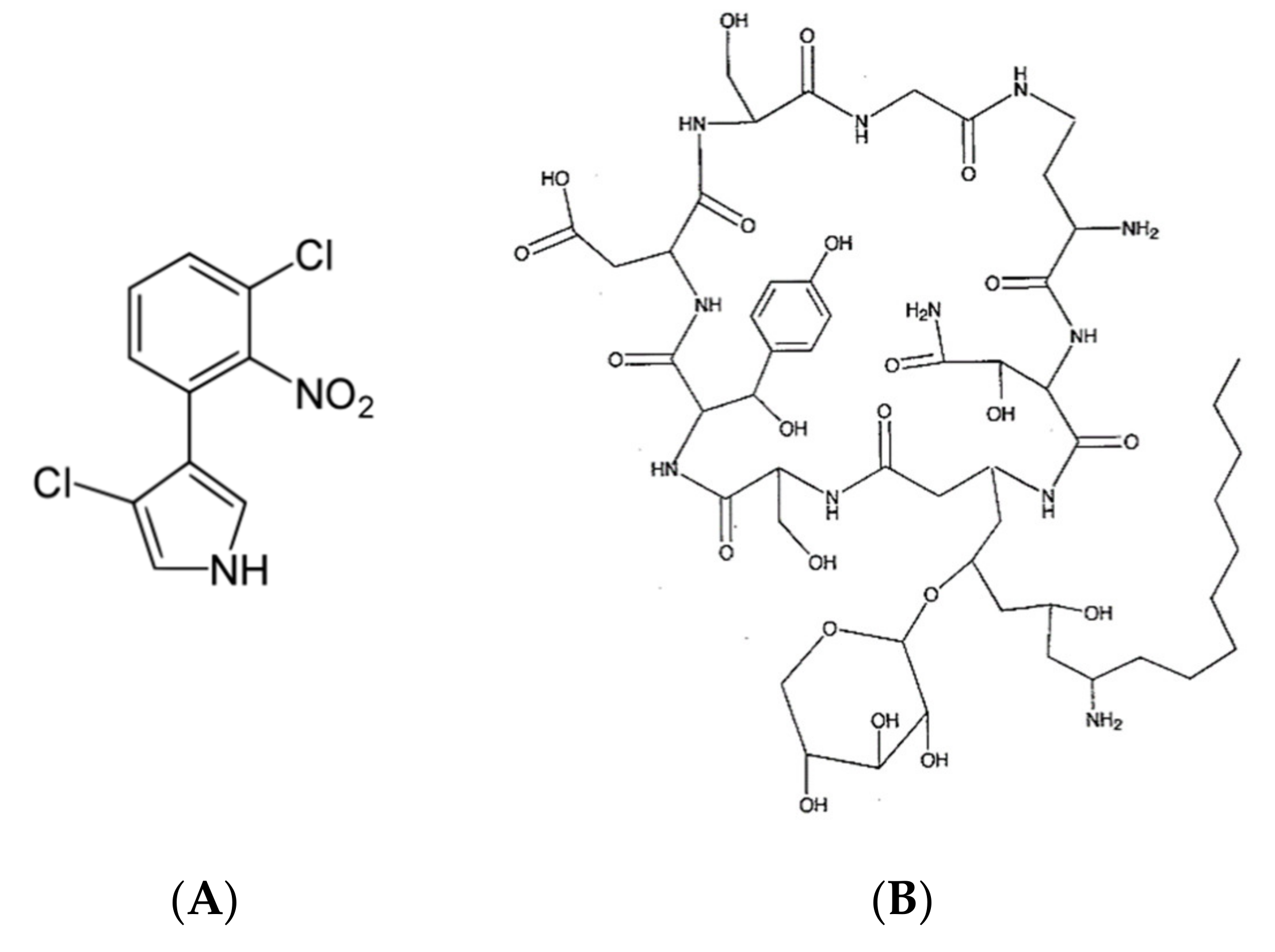
| 1 | B. cepacia | Pyrrolnitrin | [26,72] |
| Cepacidine A | [75,76] | ||
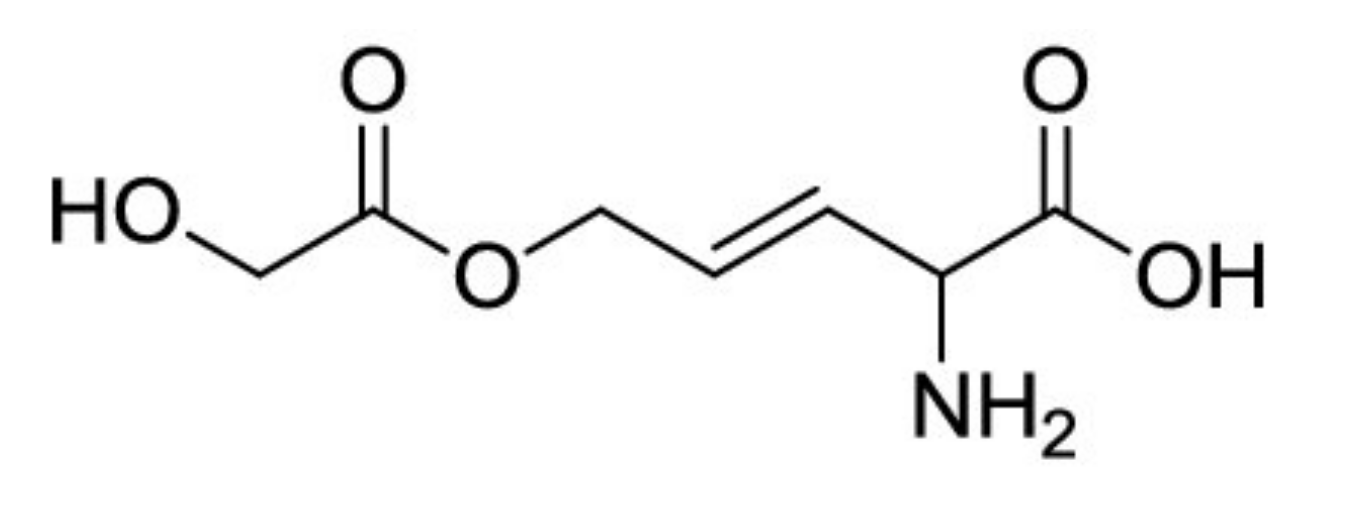
| 2 | B. andropogonis | Rhizobitoxine | [81,82] |
| 3 | B. plantarii | Tropolone | [87] |
| 4 | B. plantarii strain DSM 9509 | Rhamnolipids | [88] |
| 5 | B. rhizoxinica | Rhizoxin | [89,90] |
| 6 | B. glumae | Toxoflavin | [97,98] |
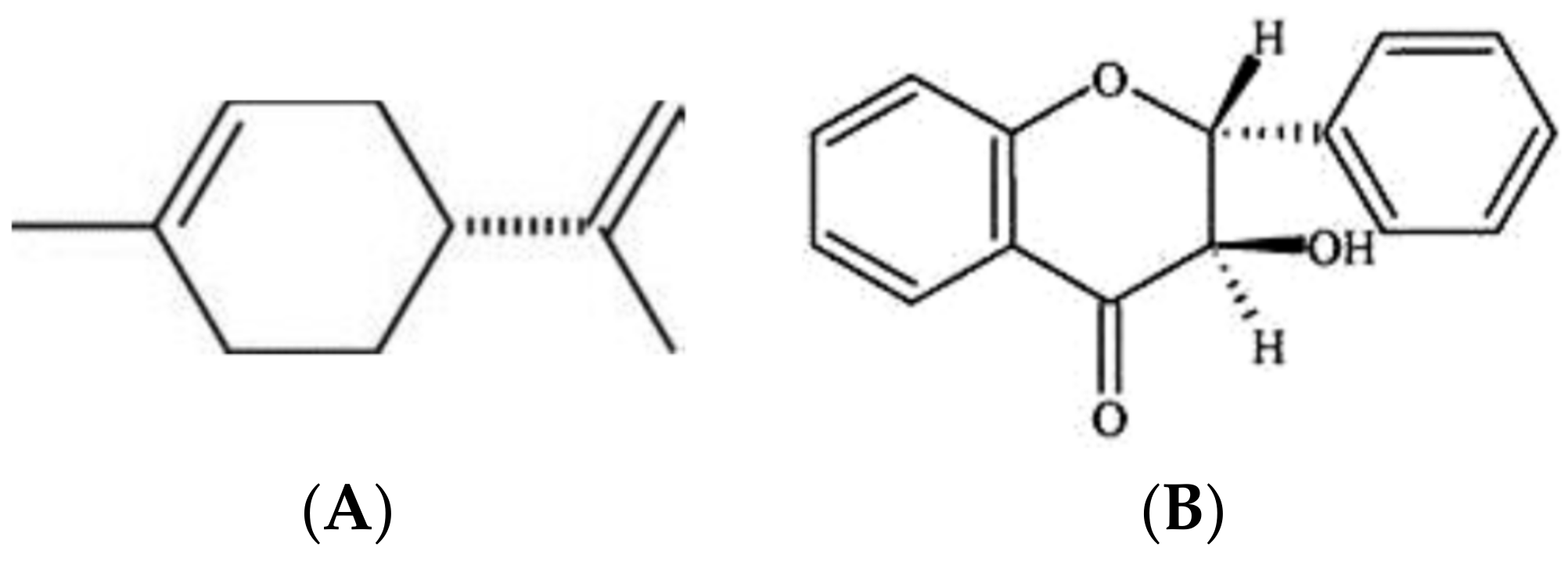
| 7 | B. caryophylli | Caryoynencine | [99,101] |
| 8 | B. gladioli pv. cocovenenas | Bongkrekic acid and toxoflavin | [108,116] |
| 9 | B. gladioli pv. agaricicola | d-Limonene | [127] |
| 4-Flavanone | [128] | ||
| Ornithine lipid | [32] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elshafie, H.S.; Camele, I. An Overview of Metabolic Activity, Beneficial and Pathogenic Aspects of Burkholderia Spp. Metabolites 2021, 11, 321. https://doi.org/10.3390/metabo11050321
Elshafie HS, Camele I. An Overview of Metabolic Activity, Beneficial and Pathogenic Aspects of Burkholderia Spp. Metabolites. 2021; 11(5):321. https://doi.org/10.3390/metabo11050321
Chicago/Turabian StyleElshafie, Hazem S., and Ippolito Camele. 2021. "An Overview of Metabolic Activity, Beneficial and Pathogenic Aspects of Burkholderia Spp." Metabolites 11, no. 5: 321. https://doi.org/10.3390/metabo11050321
APA StyleElshafie, H. S., & Camele, I. (2021). An Overview of Metabolic Activity, Beneficial and Pathogenic Aspects of Burkholderia Spp. Metabolites, 11(5), 321. https://doi.org/10.3390/metabo11050321







