Plasma Ceramides Pathophysiology, Measurements, Challenges, and Opportunities
Abstract
:1. Introduction
2. Ceramide Chemical Structure and Biological Activity
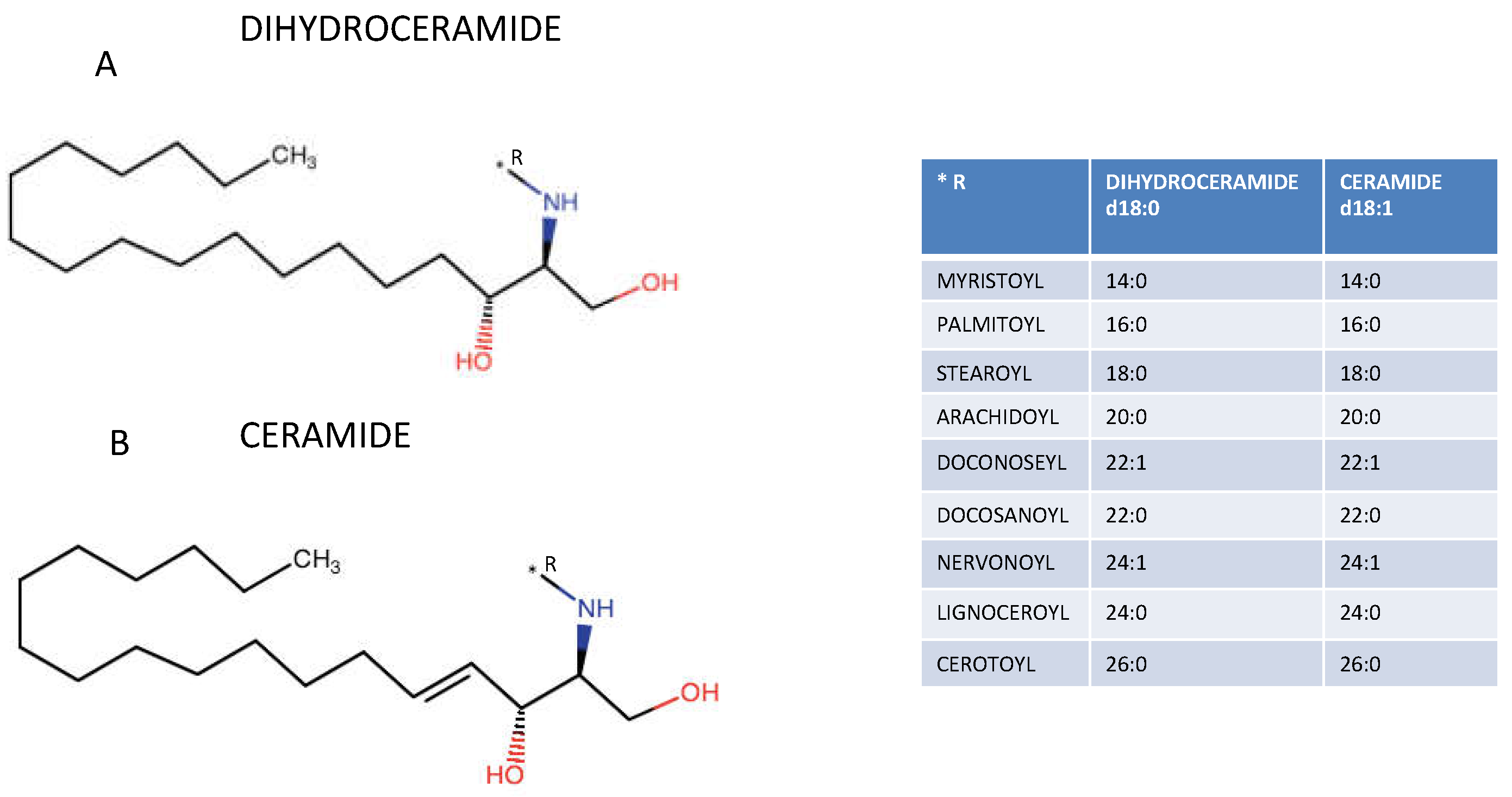
2.1. Chemical Structure and Production
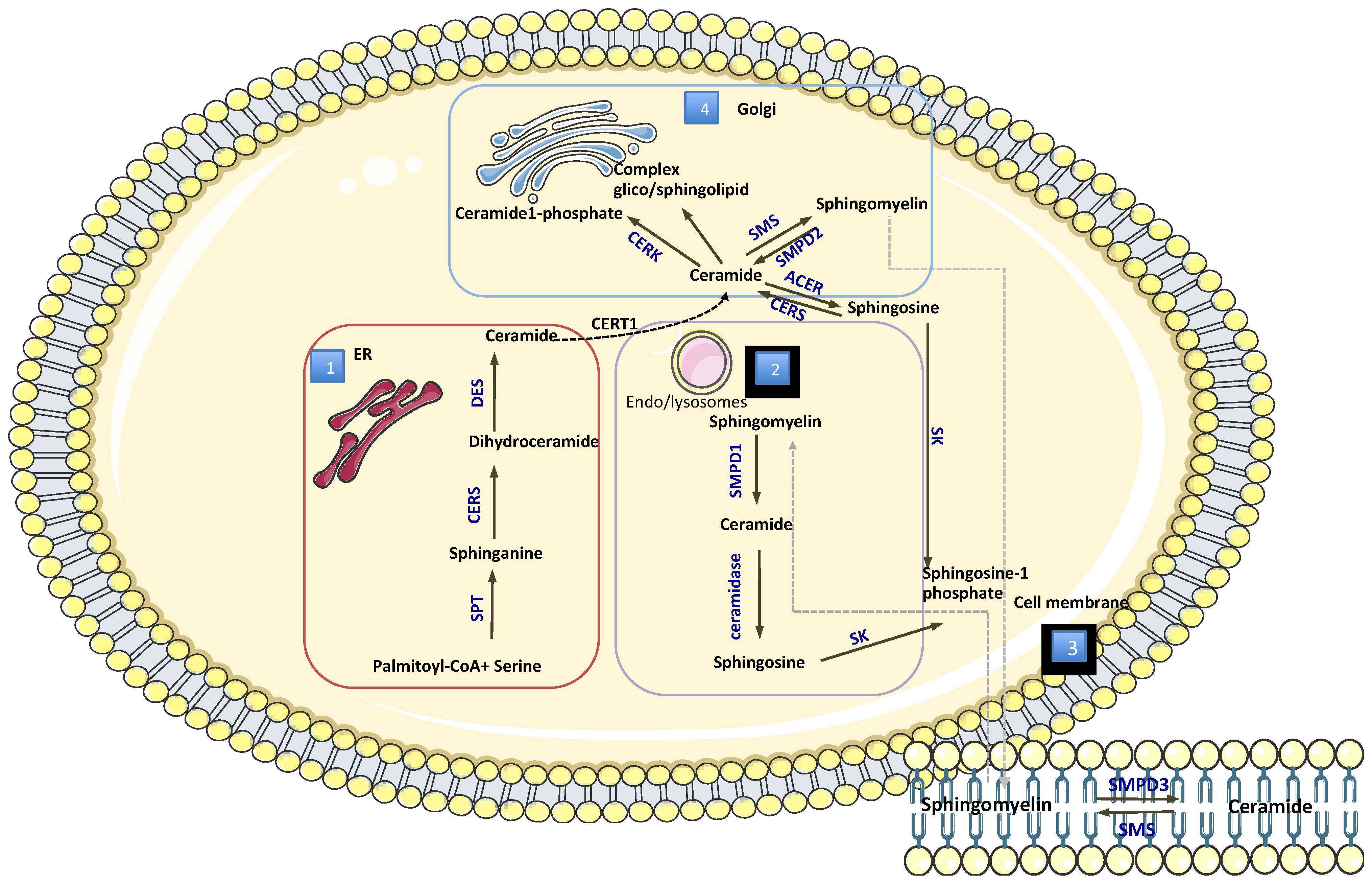
2.2. Ceramides as Structural Elements of Biological Membranes as Well as Bioactive Molecules
3. Modulation of Ceramides by Diet and Endurance Exercise
4. Ceramides Scores as Opportunity for Practical Application
5. What Is in Store for the Future?
Author Contributions
Funding
Conflicts of Interest
References
- Hannun, Y.A.; Obeid, L.M. Sphingolipids and their metabolism in physiology and disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Choi, R.H.; Tatum, S.M.; Symons, J.D.; Summers, S.A.; Holland, W.L. Ceramides and other sphingolipids as drivers of cardiovascular disease. Nat. Rev. Cardiol. 2021, 18, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Yun, H.; Sun, L.; Wu, Q.; Zong, G.; Qi, Q.; Li, H.; Zheng, H.; Zeng, R.; Liang, L.; Lin, X. Associations among circulating sphingolipids, β-cell function, and risk of developing type 2 diabetes: A population-based cohort study in China. PLoS Med. 2020, 17, e1003451. [Google Scholar] [CrossRef] [PubMed]
- Ayub, M.; Jin, H.K.; Bae, J.S. Novelty of sphingolipids in the central nervous system physiology and disease: Focusing on the sphingolipid hypothesis of neuroinflammation and neurodegeneration. Int. J. Mol. Sci. 2021, 22, 7353. [Google Scholar] [CrossRef]
- Piazzesi, A.; Afsar, S.Y.; van Echten-Deckert, G. Sphingolipid metabolism in the development and progression of cancer: One cancer’s help is another’s hindrance. Mol. Oncol. 2021. [Google Scholar] [CrossRef]
- Custodia, A.; Aramburu-Núñez, M.; Correa-Paz, C.; Posado-Fernández, A.; Gómez-Larrauri, A.; Castillo, J.; Gómez-Muñoz, A.; Sobrino, T.; Ouro, A. Ceramide metabolism and parkinson’s disease—Therapeutic targets. Biomolecules 2021, 11, 945. [Google Scholar] [CrossRef]
- Eakin, K.A.; Saleem, M.; Herrmann, N.; Cogo-Moreira, H.; Mielke, M.M.; Oh, P.I.; Haughey, N.J.; Venkata, S.L.V.; Lanctôt, K.L.; Swardfager, W. Plasma Sphingolipids Mediate a Relationship between Type 2 Diabetes and Memory Outcomes in Patients with Coronary Artery Disease Undertaking Exercise. J. Alzheimer’s Dis. 2019, 69, 717–727. [Google Scholar] [CrossRef]
- Zabielski, P.; Hady, H.R.; Chacinska, M.; Roszczyc, K.; Gorski, J.; Blachnio-Zabielska, A.U. The effect of high fat diet and metformin treatment on liver lipids accumulation and their impact on insulin action. Sci. Rep. 2018, 8, 7249. [Google Scholar] [CrossRef] [Green Version]
- Zabielski, P.; Chacinska, M.; Charkiewicz, K.; Baranowski, M.; Gorski, J.; Blachnio-Zabielska, A.U. Effect of metformin on bioactive lipid metabolism in insulin-resistant muscle. J. Endocrinol. 2017, 233, 329–340. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.D.; Toledo, E.; Hruby, A.; Rosner, B.A.; Willett, W.C.; Sun, Q.; Razquin, C.; Zheng, Y.; Ruiz-Canela, M.; Guasch-Ferré, M.; et al. Plasma ceramides, mediterranean diet, and incident cardiovascular disease in the PREDIMED trial (prevención con dieta mediterránea). Circulation 2017, 135, 2028–2040. [Google Scholar] [CrossRef] [Green Version]
- Walker, M.E.; Xanthakis, V.; Peterson, L.R.; Duncan, M.S.; Lee, J.; Ma, J.; Bigornia, S.; Moore, L.L.; Quatromoni, P.A.; Vasan, R.S.; et al. Dietary patterns, ceramide ratios, and risk of all-cause and cause-specific mortality: The framingham offspring study. J. Nutr. 2020, 150, 2994–3004. [Google Scholar] [CrossRef]
- Maltais, A.; Lemieux, I.; Alméras, N.; Tremblay, A.; Bergeron, J.; Poirier, P.; Després, J.P. One-Year Lifestyle Intervention, Muscle Lipids, and Cardiometabolic Risk. Med. Sci. Sports Exerc. 2019, 51, 2156–2165. [Google Scholar] [CrossRef] [PubMed]
- Coll-Risco, I.; Borges-Cosic, M.; Acosta-Manzano, P.; Camiletti-Moirón, D.; Aranda, P.; Aparicio, V.A. Effects of concurrent exercise on cardiometabolic status during perimenopause: The FLAMENCO Project. Climacteric 2018, 21, 559–565. [Google Scholar] [CrossRef]
- Wun, C.H.; Zhang, M.J.; Ho, B.H.; McGeough, K.; Tan, F.; Aziz, A.R. Efficacy of a six-week dispersed wingate-cycle training protocol on peak aerobic power, leg strength, insulin sensitivity, blood lipids and quality of life in healthy adults. Int. J. Environ. Res. Public Health 2020, 17, 4860. [Google Scholar] [CrossRef] [PubMed]
- Gaggini, M.; Sabatino, L.; Vassalle, C. Conventional and innovative methods to assess oxidativestressbiomarkersintheclinicalcardiovascular setting. Biotechniques 2020, 68, 223–231. [Google Scholar] [CrossRef] [Green Version]
- Reidy, P.T.; Mahmassani, Z.S.; McKenzie, A.I.; Petrocelli, J.J.; Summers, S.A.; Drummond, M.J. Influence of exercise training on skeletal muscle insulin resistance in aging: Spotlight on muscle ceramides. Int. J. Mol. Sci. 2020, 21, 1514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaggini, M.; Vassalle, C.; Carli, F.; Maltinti, M.; Sabatino, L.; Buzzigoli, E.; Mastorci, F.; Sbrana, F.; Gastaldelli, A.; Pingitore, A. Changes in plasma bioactive lipids and inflammatory markers during a half-marathon in trained athletes. Appl. Sci. 2021, 11, 4622. [Google Scholar] [CrossRef]
- Kasumov, T.; Solomon, T.P.J.; Hwang, C.; Huang, H.; Haus, J.M.; Zhang, R.; Kirwan, J.P. Improved insulin sensitivity after exercise training is linked to reduced plasma C14:0 ceramide in obesity and type 2 diabetes. Obesity 2015, 23, 1414–1421. [Google Scholar] [CrossRef]
- Hilvo, M.; Vasile, V.C.; Donato, L.J.; Hurme, R.; Laaksonen, R. Ceramides and Ceramide Scores: Clinical Applications for Cardiometabolic Risk Stratification. Front. Endocrinol. 2020, 11, 570628. [Google Scholar] [CrossRef]
- Skácel, J.; Slusher, B.S.; Tsukamoto, T. Small Molecule Inhibitors Targeting Biosynthesis of Ceramide, the Central Hub of the Sphingolipid Network. J. Med. Chem. 2021, 64, 279–297. [Google Scholar] [CrossRef]
- Morad, S.A.F.; Cabot, M.C. Ceramide-orchestrated signalling in cancer cells. Nat. Rev. Cancer 2013, 13, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Raichur, S. Ceramide Synthases Are Attractive Drug Targets for Treating Metabolic Diseases. Front. Endocrinol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Simons, K.; Ehehalt, R. Cholesterol, lipid rafts, and disease. J. Clin. Investig. 2002, 110, 597–603. [Google Scholar] [CrossRef] [PubMed]
- Karanth, S.; Azinfar, A.; Helm, C.A.; Delcea, M. Identification of a critical lipid ratio in raft-like phases exposed to nitric oxide: An AFM study. Biophys. J. 2021, 120, 3103–3111. [Google Scholar] [CrossRef]
- Ladjohounlou, R.; Louati, S.; Lauret, A.; Gauthier, A.; Ardail, D.; Magne, N.; Alphonse, G.; Rodriguez-Lafrasse, C. Ceramide-enriched membrane domains contribute to targeted and nontargeted effects of radiation through modulation of pi3k/akt signaling in hnscc cells. Int. J. Mol. Sci. 2020, 21, 7200. [Google Scholar] [CrossRef]
- Miyaji, M.; Jin, Z.X.; Yamaoka, S.; Amakawa, R.; Fukuhara, S.; Sato, S.B.; Kobayashi, T.; Domae, N.; Mimori, T.; Bloom, E.T.; et al. Role of membrane sphingomyelin and ceramide in platform formation for Fas-mediated apoptosis. J. Exp. Med. 2005, 202, 249–259. [Google Scholar] [CrossRef] [Green Version]
- Yun, S.H.; Park, E.S.; Shin, S.W.; Ju, M.H.; Han, J.Y.; Jeong, J.S.; Kim, S.H.; Stonik, V.A.; Kwak, J.Y.; Park, J.I. By activating Fas/ceramide synthase 6/p38 kinase in lipid rafts, Stichoposide D inhibits growth of leukemia xenografts. Oncotarget 2015, 6, 27596–27612. [Google Scholar] [CrossRef] [Green Version]
- McGraw, K.; List, A. Erythropoietin Receptor Signaling and Lipid Rafts. Vitam. Horm. 2017, 105, 79–100. [Google Scholar]
- Bekhite, M.; González-Delgado, A.; Hübner, S.; Haxhikadrija, P.; Kretzschmar, T.; Müller, T.; Wu, J.M.F.; Bekfani, T.; Franz, M.; Wartenberg, M.; et al. The role of ceramide accumulation in human induced pluripotent stem cell-derived cardiomyocytes on mitochondrial oxidative stress and mitophagy. Free Radic. Biol. Med. 2021, 167, 66–80. [Google Scholar] [CrossRef]
- Adhikari, B.; De Silva, B.; Molina, J.A.; Allen, A.; Peck, S.H.; Lee, S.Y. Neuronal ceroid lipofuscinosis related ER membrane protein CLN8 regulates PP2A activity and ceramide levels. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 322–328. [Google Scholar] [CrossRef]
- Liu, F.; Li, X.; Lu, C.; Bai, A.; Bielawski, J.; Bielawska, A.; Marshall, B.; Schoenlein, P.V.; Lebedyeva, I.O.; Liu, K. Ceramide activates lysosomal cathepsin B and cathepsin D to attenuate autophagy and induces ER stress to suppress myeloidderived suppressor cells. Oncotarget 2016, 7, 83907–83925. [Google Scholar] [CrossRef] [Green Version]
- Colombini, M. Membrane channels formed by ceramide. Handb. Exp. Pharmacol. 2013, 215, 109–126. [Google Scholar] [CrossRef]
- Green, C.D.; Maceyka, M.; Cowart, L.A.; Spiegel, S. Sphingolipids in metabolic disease: The good, the bad, and the unknown. Cell Metab. 2021, 33, 1293–1306. [Google Scholar] [CrossRef]
- Šrámek, J.; Němcová-fürstová, V.; Kovář, J. Molecular mechanisms of apoptosis induction and its regulation by fatty acids in pancreatic β-cells. Int. J. Mol. Sci. 2021, 22, 4285. [Google Scholar] [CrossRef]
- Stiban, J.; Perera, M. Very long chain ceramides interfere with C16-ceramide-induced channel formation: A plausible mechanism for regulating the initiation of intrinsic apoptosis. Biochim. Biophys. Acta Biomembr. 2015, 1848, 561–567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartmann, D.; Wegner, M.S.; Wanger, R.A.; Ferreirós, N.; Schreiber, Y.; Lucks, J.; Schiffmann, S.; Geisslinger, G.; Grösch, S. The equilibrium between long and very long chain ceramides is important for the fate of the cell and can be influenced by co-expression of CerS. Int. J. Biochem. Cell Biol. 2013, 45, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Lang, F.; Ullrich, S.; Gulbins, E. Ceramide formation as a target in beta-cell survival and function. Expert Opin. Ther. Targets 2011, 15, 1061–1071. [Google Scholar] [CrossRef] [PubMed]
- Gomez, L.; Paillard, M.; Price, M.; Chen, Q.; Teixeira, G.; Spiegel, S.; Lesnefsky, E.J. A novel role for mitochondrial sphingosine-1-phosphate produced by sphingosine kinase-2 in PTP-mediated cell survival during cardioprotection. Basic Res. Cardiol. 2011, 106, 1341–1353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Honbo, N.; Goetzl, E.J.; Chatterjee, K.; Karliner, J.S.; Gray, M.O. Signals from type 1 sphingosine 1-phosphate receptors enhance adult mouse cardiac myocyte survival during hypoxia. Am. J. Physiol. Hear. Circ. Physiol. 2007, 293, H3150–H3158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurz, J.; Parnham, M.J.; Geisslinger, G.; Schiffmann, S. Ceramides as Novel Disease Biomarkers. Trends Mol. Med. 2019, 25, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Siddique, M.M.; Li, Y.; Chaurasia, B.; Kaddai, V.A.; Summers, S.A. Dihydroceramides: From bit players to lead actors. J. Biol. Chem. 2015, 290, 15371–15379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muñoz-Guardiola, P.; Casas, J.; Megías-Roda, E.; Solé, S.; Perez-Montoyo, H.; Yeste-Velasco, M.; Erazo, T.; Diéguez-Martínez, N.; Espinosa-Gil, S.; Muñoz-Pinedo, C.; et al. The anti-cancer drug ABTL0812 induces ER stress-mediated cytotoxic autophagy by increasing dihydroceramide levels in cancer cells. Autophagy 2021, 17, 1349–1366. [Google Scholar] [CrossRef] [PubMed]
- McGurk, K.A.; Keavney, B.D.; Nicolaou, A. Circulating ceramides as biomarkers of cardiovascular disease: Evidence from phenotypic and genomic studies. Atherosclerosis 2021, 327, 18–30. [Google Scholar] [CrossRef]
- Roszczyc-Owsiejczuk, K.; Zabielski, P. Sphingolipids as a Culprit of Mitochondrial Dysfunction in Insulin Resistance and Type 2 Diabetes. Front. Endocrinol. 2021, 12, 143. [Google Scholar] [CrossRef]
- Ismaeel, A.; Franco, M.E.; Lavado, R.; Papoutsi, E.; Casale, G.P.; Fuglestad, M.; Mietus, C.J.; Haynatzki, G.R.; Smith, R.S.; Bohannon, W.T.; et al. Altered metabolomic profile in patients with peripheral artery disease. J. Clin. Med. 2019, 8, 1463. [Google Scholar] [CrossRef] [Green Version]
- Bergman, B.C.; Brozinick, J.T.; Strauss, A.; Bacon, S.; Kerege, A.; Bui, H.H.; Sanders, P.; Siddall, P.; Kuo, M.S.; Perreault, L. Serum sphingolipids: Relationships to insulin sensitivity and changes with exercise in humans. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E398–E408. [Google Scholar] [CrossRef] [Green Version]
- Lankinen, M.; Schwab, U.; Kolehmainen, M.; Paananen, J.; Nygren, H.; Seppänen-Laakso, T.; Poutanen, K.; Hyötyläinen, T.; Risérus, U.; Savolainen, M.J.; et al. A healthy Nordic diet alters the plasma lipidomic profile in adults with features of metabolic syndrome in a multicenter randomized dietary intervention. J. Nutr. 2016, 146, 662–672. [Google Scholar] [CrossRef] [Green Version]
- Görski, J.; Dobrzyn, A.; Zendzian-Piotrowska, M. The sphingomyelin-signaling pathway in skeletal muscles and its role in regulation of glucose uptake. Ann. N. Y. Acad. Sci. 2002, 967, 236–248. [Google Scholar] [CrossRef] [PubMed]
- Dubé, J.J.; Amati, F.; Toledo, F.G.S.; Stefanovic-Racic, M.; Rossi, A.; Coen, P.; Goodpaster, B.H. Effects of weight loss and exercise on insulin resistance, and intramyocellular triacylglycerol, diacylglycerol and ceramide. Diabetologia 2011, 54, 1147–1156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shepherd, S.O.; Cocks, M.; Meikle, P.J.; Mellett, N.A.; Ranasinghe, A.M.; Barker, T.A.; Wagenmakers, A.J.M.; Shaw, C.S. Lipid droplet remodelling and reduced muscle ceramides following sprint interval and moderate-intensity continuous exercise training in obese males. Int. J. Obes. 2017, 41, 1745–1754. [Google Scholar] [CrossRef] [PubMed]
- Scherer, P.E.; Hill, J.A. Obesity, diabetes, and cardiovascular diseases. Circ. Res. 2016, 118, 1703–1705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boon, J.; Hoy, A.J.; Stark, R.; Brown, R.D.; Meex, R.C.; Henstridge, D.C.; Schenk, S.; Meikle, P.J.; Horowitz, J.F.; Kingwell, B.A.; et al. Ceramides Contained in LDL Are Elevated in Type 2 Diabetes and Promote Inflammation and Skeletal Muscle Insulin Resistance. Diabetes 2013, 62, 401–410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chavez, J.A.; Knotts, T.A.; Wang, L.P.; Li, G.; Dobrowsky, R.T.; Florant, G.L.; Summers, S.A. A role for ceramide, but not diacylglycerol, in the antagonism of insulin signal transduction by saturated fatty acids. J. Biol. Chem. 2003, 278, 10297–10303. [Google Scholar] [CrossRef] [Green Version]
- Baranowski, M.; Zabielski, P.; Blachnio, A.; Gorski, J. Effect of exercise duration on ceramide metabolism in the rat heart. Acta Physiol. 2008, 192, 519–529. [Google Scholar] [CrossRef]
- Błachnio-Zabielska, A.; Baranowski, M.; Zabielski, P.; Górski, J. Effect of exercise duration on the key pathways of ceramide metabolism in rat skeletal muscles. J. Cell. Biochem. 2008, 105, 776–784. [Google Scholar] [CrossRef]
- Baranowski, M.; Charmas, M.; Długołecka, B.; Górski, J. Exercise increases plasma levels of sphingoid base-1 phosphates in humans. Acta Physiol. 2011, 203, 373–380. [Google Scholar] [CrossRef]
- Baranowski, M.; Górski, J.; Klapcinska, B.; Waskiewicz, Z.; Sadowska-Krepa, E. Ultramarathon run markedly reduces plasma sphingosine-1-phosphate concentration. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 148–156. [Google Scholar] [CrossRef]
- Baranowski, M.; Błachnio-Zabielska, A.U.; Charmas, M.; Helge, J.W.; Dela, F.; Książek, M.; Długołęcka, B.; Klusiewicz, A.; Chabowski, A.; Górski, J. Exercise increases sphingoid base-1-phosphate levels in human blood and skeletal muscle in a time- and intensity-dependent manner. Eur. J. Appl. Physiol. 2015, 115, 993–1003. [Google Scholar] [CrossRef] [Green Version]
- Søgaard, D.; Baranowski, M.; Larsen, S.; Lund, M.T.; Scheuer, C.M.; Abildskov, C.V.; Dideriksen, S.G.; Dela, F.; Helge, J.W. Muscle-saturated bioactive lipids are increased with aging and influenced by high-intensity interval training. Int. J. Mol. Sci. 2019, 20, 1240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mardare, C.; Krüger, K.; Liebisch, G.; Seimetz, M.; Couturier, A.; Ringseis, R.; Wilhelm, J.; Weissmann, N.; Eder, K.; Mooren, F.C. Endurance and resistance training affect high fat diet-induced increase of ceramides, inflammasome expression, and systemic inflammation in mice. J. Diabetes Res. 2016, 2016, 4536470. [Google Scholar] [CrossRef] [Green Version]
- Mietus-Snyder, M.; Narayanan, N.; Krauss, R.M.; Laine-Graves, K.; McCann, J.C.; Shigenaga, M.K.; McHugh, T.H.; Ames, B.N.; Suh, J.H. Randomized nutrient bar supplementation improves exercise-associated changes in plasma metabolome in adolescents and adult family members at cardiometabolic risk. PLoS ONE 2020, 15, e0240437. [Google Scholar] [CrossRef]
- Fang, Z.; Pyne, S.; Pyne, N.J. Ceramide and sphingosine 1-phosphate in adipose dysfunction. Prog. Lipid Res. 2019, 74, 145–159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, L.C.; Martens, C.R.; Santos-Parker, J.R.; Bassett, C.J.; Strahler, T.R.; Cruickshank-Quinn, C.; Reisdorph, N.; McQueen, M.B.; Seals, D.R. Amino acid and lipid associated plasma metabolomic patterns are related to healthspan indicators with ageing. Clin. Sci. 2018, 132, 1765–1777. [Google Scholar] [CrossRef] [PubMed]
- Noerman, S.; Klåvus, A.; Järvelä-Reijonen, E.; Karhunen, L.; Auriola, S.; Korpela, R.; Lappalainen, R.; Kujala, U.M.; Puttonen, S.; Kolehmainen, M.; et al. Plasma lipid profile associates with the improvement of psychological well-being in individuals with perceived stress symptoms. Sci. Rep. 2020, 10, 2143. [Google Scholar] [CrossRef]
- Berkowitz, L.; Henríquez, M.P.; Salazar, C.; Rojas, E.; Echeverría, G.; Love, G.D.; Rigotti, A.; Coe, C.L.; Ryff, C.D. Association between serum sphingolipids and eudaimonic well-being in white U.S. adults. Sci. Rep. 2021, 11, 13139. [Google Scholar] [CrossRef] [PubMed]
- Turpin, S.M.; Nicholls, H.T.; Willmes, D.M.; Mourier, A.; Brodesser, S.; Wunderlich, C.M.; Mauer, J.; Xu, E.; Hammerschmidt, P.; Brönneke, H.S.; et al. Obesity-induced CerS6-dependent C16:0 ceramide production promotes weight gain and glucose intolerance. Cell Metab. 2014, 20, 678–686. [Google Scholar] [CrossRef] [Green Version]
- Raichur, S.; Wang, S.T.; Chan, P.W.; Li, Y.; Ching, J.; Chaurasia, B.; Dogra, S.; Öhman, M.K.; Takeda, K.; Sugii, S.; et al. CerS2 haploinsufficiency inhibits β-oxidation and confers susceptibility to diet-induced steatohepatitis and insulin resistance. Cell Metab. 2014, 20, 687–695. [Google Scholar] [CrossRef] [Green Version]
- Grebe, S.K.G.; Singh, R.J. LC-MS/MS in the clinical laboratory—Where to from here? Clin. Biochem. Rev. 2011, 32, 5–31. [Google Scholar]
- Laaksonen, R.; Ekroos, K.; Sysi-Aho, M.; Hilvo, M.; Vihervaara, T.; Kauhanen, D.; Suoniemi, M.; Hurme, R.; März, W.; Scharnagl, H.; et al. Plasma ceramides predict cardiovascular death in patients with stable coronary artery disease and acute coronary syndromes beyond LDL-cholesterol. Eur. Heart J. 2016, 37, 1967–1976. [Google Scholar] [CrossRef]
- Havulinna, A.S.; Sysi-Aho, M.; Hilvo, M.; Kauhanen, D.; Hurme, R.; Ekroos, K.; Salomaa, V.; Laaksonen, R. Circulating Ceramides Predict Cardiovascular Outcomes in the Population-Based FINRISK 2002 Cohort. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 2424–2430. [Google Scholar] [CrossRef] [Green Version]
- Hilvo, M.; Meikle, P.J.; Pedersen, E.R.; Tell, G.S.; Dhar, I.; Brenner, H.; Schöttker, B.; Lääperi, M.; Kauhanen, D.; Koistinen, K.M.; et al. Development and validation of a ceramide-and phospholipid-based cardiovascular risk estimation score for coronary artery disease patients. Eur. Heart J. 2020, 41, 371–380. [Google Scholar] [CrossRef] [Green Version]
- Hilvo, M.; Wallentin, L.; Lakic, T.G.; Held, C.; Kauhanen, D.; Jylhä, A.; Lindbäck, J.; Siegbahn, A.; Granger, C.B.; Koenig, W.; et al. Prediction of residual risk by ceramide-phospholipid score in patients with stable coronary heart disease on optimal medical therapy. J. Am. Heart Assoc. 2020, 9, e015258. [Google Scholar] [CrossRef] [PubMed]
- Hilvo, M.; Salonurmi, T.; Havulinna, A.S.; Kauhanen, D.; Pedersen, E.R.; Tell, G.S.; Meyer, K.; Teeriniemi, A.M.; Laatikainen, T.; Jousilahti, P.; et al. Ceramide stearic to palmitic acid ratio predicts incident diabetes. Diabetologia 2018, 61, 1424–1434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poss, A.M.; Maschek, J.A.; Cox, J.E.; Hauner, B.J.; Hopkins, P.N.; Hunt, S.C.; Holland, W.L.; Summers, S.A.; Playdon, M.C. Machine learning reveals serum sphingolipids as cholesterol-independent biomarkers of coronary artery disease. J. Clin. Investig. 2020, 130, 1363–1376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quer, G.; Arnaout, R.; Henne, M.; Arnaout, R. Machine Learning and the Future of Cardiovascular Care: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2021, 77, 300–313. [Google Scholar] [CrossRef] [PubMed]
- Petrocelli, J.J.; McKenzie, A.I.; Mahmassani, Z.S.; Reidy, P.T.; Stoddard, G.J.; Poss, A.M.; Holland, W.L.; Summers, S.A.; Drummond, M.J.; Couteur, D. Le Ceramide Biomarkers Predictive of Cardiovascular Disease Risk Increase in Healthy Older Adults after Bed Rest. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2020, 75, 1663–1670. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, M. Plasma ceramides and cardiac risk. Eur. Heart J. 2017, 38, 1359–1360. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaggini, M.; Pingitore, A.; Vassalle, C. Plasma Ceramides Pathophysiology, Measurements, Challenges, and Opportunities. Metabolites 2021, 11, 719. https://doi.org/10.3390/metabo11110719
Gaggini M, Pingitore A, Vassalle C. Plasma Ceramides Pathophysiology, Measurements, Challenges, and Opportunities. Metabolites. 2021; 11(11):719. https://doi.org/10.3390/metabo11110719
Chicago/Turabian StyleGaggini, Melania, Alessandro Pingitore, and Cristina Vassalle. 2021. "Plasma Ceramides Pathophysiology, Measurements, Challenges, and Opportunities" Metabolites 11, no. 11: 719. https://doi.org/10.3390/metabo11110719
APA StyleGaggini, M., Pingitore, A., & Vassalle, C. (2021). Plasma Ceramides Pathophysiology, Measurements, Challenges, and Opportunities. Metabolites, 11(11), 719. https://doi.org/10.3390/metabo11110719







