Multiblock Analysis to Relate Polyphenol Targeted Mass Spectrometry and Sensory Properties of Chocolates and Cocoa Beans
Abstract
1. Introduction
2. Results and Discussion
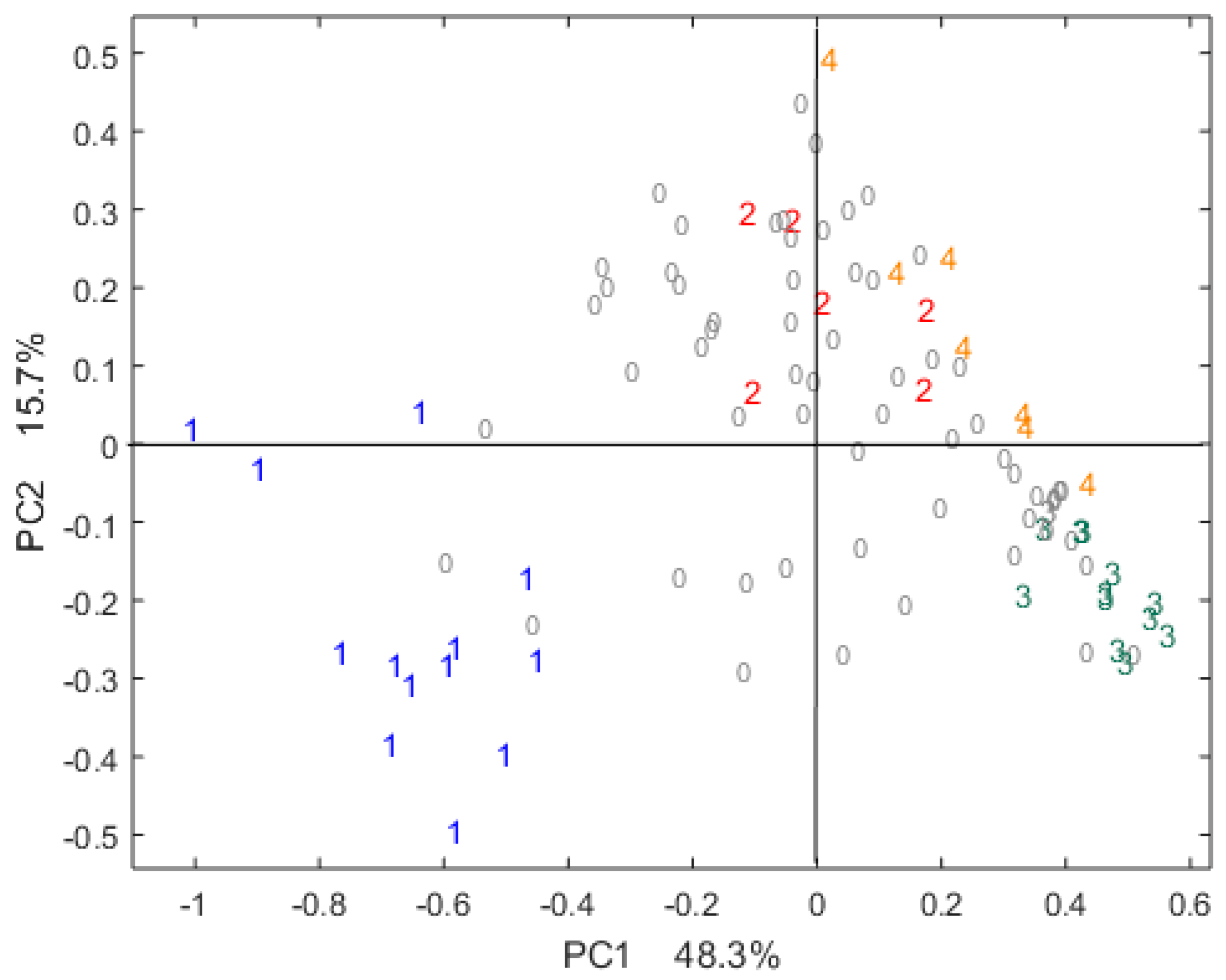
2.1. Differentiation of Chocolate Samples in the Sensory Space
2.2. Identification of the Polyphenolic Compounds in the Chocolate Samples Linked to the Sensory Analysis
2.3. Exploring the Link between Cocoa and Chocolate Polyphenols Contents
3. Materials and Methods
3.1. Chocolate and Cocoa Samples
3.2. Polyphenols Analysis by UPLC-ESI-TQ-MS
3.3. Sensory Analysis of Chocolate Samples
3.4. Data Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Deuscher, Z.; Andriot, I.; Sémon, E.; Repoux, M.; Preys, S.; Roger, J.-M.; Boulanger, R.; Labouré, H.; Le Quéré, J.L. Volatile compounds profiling by using proton transfer reaction-time of flight-mass spectrometry (PTR-ToF-MS). The case study of dark chocolates organoleptic differences. J. Mass Spectrom. 2019, 54, 92–119. [Google Scholar] [CrossRef] [PubMed]
- Deuscher, Z.; Gourrat, K.; Repoux, M.; Boulanger, R.; Labouré, H.; Le Quéré, J.L. Key Aroma Compounds of Dark Chocolates Differing in Organoleptic Properties: A GC-O Comparative Study. Molecules 2020, 25, 1809. [Google Scholar] [CrossRef]
- Seyfried, C.; Granvogl, M. Characterization of the Key Aroma Compounds in Two Commercial Dark Chocolates with High Cocoa Contents by Means of the Sensomics Approach. J. Agric. Food Chem. 2019, 67, 5827–5837. [Google Scholar] [CrossRef]
- Chetschik, I.; Pedan, V.; Chatelain, K.; Kneubühl, M.; Hühn, T. Characterization of the Flavor Properties of Dark Chocolates Produced by a Novel Technological Approach and Comparison with Traditionally Produced Dark Chocolates. J. Agric. Food Chem. 2019, 67, 3991–4001. [Google Scholar] [CrossRef] [PubMed]
- Arnoldi, A.; Arnoldi, C.; Baldi, O.; Griffini, A. Flavor components in the Maillard reaction of different amino acids with fructose in cocoa butter-water Qualitative and quantitative analysis of pyrazines. J. Agric. Food Chem. 1988, 36, 988–992. [Google Scholar] [CrossRef]
- Hashim, P.; Selamat, J.; Muhammad, S.K.S.; Ali, A. Changes in free amino acid, peptide-N, sugar and pyrazine concentration during cocoa fermentation. J. Sci. Food Agric. 1998, 78, 535–542. [Google Scholar] [CrossRef]
- Owusu, M.; Petersen, M.A.; Heimdal, H. Relationship of sensory and instrumental aroma measurements of dark chocolate as influenced by fermentation method, roasting and conching conditions. J. Food Sci. Technol. 2013, 50, 909–917. [Google Scholar] [CrossRef] [PubMed]
- Hinneh, M.; Semanhyia, E.; Van de Walle, D.; De Winne, A.; Tzompa-Sosa, D.A.; Scalone, G.L.; De Meulenaer, B.; Messens, K.; Van Durme, J.; Afoakwa, E.O.; et al. Assessing the influence of pod storage on sugar and free amino acid profiles and the implications on some Maillard reaction related flavor volatiles in Forastero cocoa beans. Food Res. Int. 2018, 111, 607–620. [Google Scholar] [CrossRef] [PubMed]
- Guinard, J.X.; Mazzucchelli, R. Effects of sugar and fat on the sensory properties of milk chocolate: Descriptive analysis and instrumental measurements. J. Sci. Food Agric. 1999, 79, 1331–1339. [Google Scholar] [CrossRef]
- Adeyeye, E.I.; Akinyeye, R.O.; Ogunlade, I.; Olaofe, O.; Boluwade, J.O. Effect of farm and industrial processing on the amino acid profile of cocoa beans. Food Chem. 2010, 118, 357–363. [Google Scholar] [CrossRef]
- Afoakwa, E.O.; Paterson, A.; Fowler, M.; Ryan, A. Flavor formation and character in cocoa and chocolate: A critical review. Crit. Rev. Food Sci. Nutr. 2008, 48, 840–857. [Google Scholar] [CrossRef] [PubMed]
- Afoakwa, E.O.; Quao, J.; Budu, A.S.; Takrama, J.; Saalia, F.K. Effect of pulp preconditioning on acidification, proteolysis, sugars and free fatty acids concentration during fermentation of cocoa (Theobroma cacao) beans. Int. J. Food Sci. Nutr. 2011, 62, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Aprotosoaie, A.; Luca, S.V.; Miron, A. Flavor Chemistry of Cocoa and Cocoa Products—An Overview. Compr. Rev. Food Sci. Food Saf. 2016, 15, 73–91. [Google Scholar] [CrossRef]
- Kongor, J.E.; Hinneh, M.; de Walle, D.V.; Afoakwa, E.O.; Boeckx, P.; Dewettinck, K. Factors influencing quality variation in cocoa (Theobroma cacao) bean flavour profile—A review. Food Res. Int. 2016, 82, 44–52. [Google Scholar] [CrossRef]
- Perez-Jimenez, J.; Neveu, V.; Vos, F.; Scalbert, A. Systematic Analysis of the Content of 502 Polyphenols in 452 Foods and Beverages: An Application of the Phenol-Explorer Database. J. Agric. Food Chem. 2010, 58, 4959–4969. [Google Scholar] [CrossRef]
- Wollgast, J.; Anklam, E. Review on polyphenols in Theobroma cacao: Changes in composition during the manufacture of chocolate and methodology for identification and quantification. Food Res. Int. 2000, 33, 423–447. [Google Scholar] [CrossRef]
- Cooper, K.; Donovan, J.; Waterhouse, A.; Williamson, G. Cocoa and health: A decade of research. Br. J. Nutr. 2008, 99, 1–11. [Google Scholar] [CrossRef]
- Misnawi, A.; Jinap, S.M.; Jamilah, B.; Nazamid, S. Effects of incubation and polyphenol oxidase enrichment on colour, fermentation index, procyanidins and astringency of unfermented and partly fermented cocoa beans. Int. J. Food Sci. Technol. 2003, 38, 285–295. [Google Scholar] [CrossRef]
- Noor-Soffalina, S.S.; Jinap, S.; Nazamid, S.; Nazimah, S.A.H. Effect of polyphenol and pH on cocoa Maillard-related flavour precursors in a lipidic model system. Int. J. Food Sci. Technol. 2009, 44, 168–180. [Google Scholar] [CrossRef]
- Perez-Jimenez, J.; Fezeu, L.; Touvier, M.; Arnault, N.; Manach, C.; Hercberg, S.; Galan, P.; Scalbert, A. Dietary intake of 337 polyphenols in French adults. Am. J. Clin. Nutr. 2011, 93, 1220–1228. [Google Scholar] [CrossRef]
- Godos, J.; Marventano, S.; Mistretta, A.; Galvano, F.; Grosso, G. Dietary sources of polyphenols in the Mediterranean healthy Eating, Aging and Lifestyle (MEAL) study cohort. Int. J. Food Sci. Nutr. 2017, 68, 750–756. [Google Scholar] [CrossRef] [PubMed]
- Ellam, S.; Williamson, G. Cocoa and Human Health. Annu. Rev. Nutr. 2013, 33, 105–128. [Google Scholar] [CrossRef]
- Williamson, G. The role of polyphenols in modern nutrition. Nutr. Bull. 2017, 42, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Peleg, H.; Gacon, K.; Schlich, P.; Noble, A.C. Bitterness and astringency of flavan-3-ol monomers, dimers and trimers. J. Sci. Food Agric. 1999, 79, 1123–1128. [Google Scholar] [CrossRef]
- Serra Bonvehi, J.; Coll, F.V. Evaluation of bitterness and astringency of polyphenolic compounds in cocoa powder. Food Chem. 1997, 60, 365–370. [Google Scholar] [CrossRef]
- Soares, S.; Brandão, E.; Guerreiro, C.; Soares, S.; Mateus, N.; de Freitas, V. Tannins in Food: Insights into the Molecular Perception of Astringency and Bitter Taste. Molecules 2020, 25, 2590. [Google Scholar] [CrossRef] [PubMed]
- Stark, T.; Bareuther, S.; Hofmann, T. Sensory-guided decomposition of roasted cocoa nibs (Theobroma cacao) and structure determination of taste-active polyphenols. J. Agric. Food Chem. 2005, 53, 5407–5418. [Google Scholar] [CrossRef]
- Stark, T.; Bareuther, S.; Hofmann, T. Molecular Definition of the Taste of Roasted Cocoa Nibs (Theobroma cacao) by Means of Quantitative Studies and Sensory Experiments. J. Agric. Food Chem. 2006, 54, 5530–5539. [Google Scholar] [CrossRef]
- Stark, T.; Hofmann, T. Application of a molecular sensory science approach to alkalized cocoa (Theobroma cacao): Structure determination and sensory activity of nonenzymatically C-glycosylated flavan-3-ols. J. Agric. Food Chem. 2006, 54, 9510–9521. [Google Scholar] [CrossRef]
- Misnawi, A.; Jinap, S.; Jamilah, B.; Nazamid, S. Sensory properties of cocoa liquor as affected by polyphenol concentration and duration of roasting. Food Qual. Prefer. 2004, 15, 403–409. [Google Scholar] [CrossRef]
- Zyzelewicz, D.; Krysiak, W.; Oracz, J.; Sosnowska, D.; Budryn, G.; Nebesny, E. The influence of the roasting process conditions on the polyphenol content in cocoa beans, nibs and chocolates. Food Res. Int. 2016, 89, 918–929. [Google Scholar] [CrossRef]
- Elwers, S.; Zambrano, A.; Rohsius, C.; Lieberei, R. Histological features of phenolic compounds in fine and bulk cocoa seed (Theobroma cacao L.). J. Appl. Bot. Food Qual. 2010, 83, 182–188. [Google Scholar]
- Tuenter, E.; Delbaere, C.; de Winne, A.; Bijttebier, S.; Custers, D.; Foubert, K.; van Durme, J.; Messens, K.; Dewettinck, K.; Pieters, L. Non-volatile and volatile composition of West African bulk and Ecuadorian fine-flavor cocoa liquor and chocolate. Food Res. Int. 2020, 130, 108943. [Google Scholar] [CrossRef] [PubMed]
- Wolfender, J.; Nuzillard, J.; van der Hooft, J.; Renault, J.; Bertrand, S. Accelerating Metabolite Identification in Natural Product Research: Toward an Ideal Combination of Liquid Chromatography–High-Resolution Tandem Mass Spectrometry and NMR Profiling, in Silico Databases, and Chemometrics. Anal. Chem. 2019, 91, 704–742. [Google Scholar] [CrossRef] [PubMed]
- De Villiers, A.; Venter, P.; Pasch, H. Recent advances and trends in the liquid-chromatography−mass spectrometry analysis of flavonoids. J. Chrom. A 2016, 1430, 16–78. [Google Scholar] [CrossRef] [PubMed]
- Fayeulle, N.; Meudec, E.; Boulet, J.C.; Vallverdu-Queralt, A.; Hue, C.; Boulanger, R.; Cheynier, V.; Sommerer, N. Fast Discrimination of Chocolate Quality Based on Average-Mass-Spectra Fingerprints of Cocoa Polyphenols. J. Agric. Food Chem. 2019, 67, 2723–2731. [Google Scholar] [CrossRef]
- Fayeulle, N.; Vallverdu-Queralt, A.; Meudec, E.; Hue, C.; Boulanger, R.; Cheynier, V.; Sommerer, N. Characterization of new flavan-3-ol derivatives in fermented cocoa beans. Food Chem. 2018, 259, 207–212. [Google Scholar] [CrossRef]
- D’Souza, R.; Grimbs, S.; Behrends, B.; Bernaert, H.; Ullrich, M.; Kuhnert, N. Origin-based polyphenolic fingerprinting of Theobroma cacao in unfermented and fermented beans. Food Res. Int. 2017, 99, 550–559. [Google Scholar] [CrossRef]
- Sánchez-Rabaneda, F.; Jáuregui, O.; Casals, I.; Andrés-Lacueva, C.; Izquierdo-Pulido, M.; Lamuela-Raventós, R.M. Liquid chromatographic/electrospray ionization tandem mass spectrometric study of the phenolic composition of cocoa (Theobroma cacao). J. Mass Spectrom. 2003, 38, 35–42. [Google Scholar] [CrossRef]
- Sukha, D.A.; Butler, D.R.; Umaharan, P.; Boult, E. The use of an optimised organoleptic assessment protocol to describe and quantify different flavour attributes of cocoa liquors made from Ghana and Trinitario beans. Eur. Food Res. Technol. 2008, 226, 405–413. [Google Scholar] [CrossRef]
- Qannari, E.; Wakeling, I.; Courcoux, P.; MacFie, H. Defining the underlying sensory dimensions. Food Qual. Pref. 2000, 11, 151–154. [Google Scholar] [CrossRef]
- Boselli, E.; Boulton, R.; Thorngate, J.; Frega, N. Chemical and Sensory Characterization of DOC Red Wines from Marche (Italy) Related to Vintage and Grape Cultivars. J. Agric. Food Chem. 2004, 52, 3843–3854. [Google Scholar] [CrossRef] [PubMed]
- Preys, S.; Mazerolles, G.; Courcoux, P.; Samson, A.; Fischer, U.; Hanafi, M.; Bertrand, D.; Cheynier, V. Relationship between polyphenolic composition and some sensory properties in red wines using multiway analyses. Anal. Chim. Acta 2006, 563, 126–136. [Google Scholar] [CrossRef]
- Bernaert, H.; Blondeel, I.; Allegaert, L.; Lohmueller, T. Industrial Treatment of Cocoa in Chocolate Production: Health Implications. In Chocolate and Health; Paoletti, R., Poli, A., Conti, A., Visioli, F., Eds.; Springer: Milan, Italy, 2012; pp. 17–32. [Google Scholar]
- Counet, C.; Callemien, D.; Ouwerx, C.; Collin, S. Use of gas chromatography-olfactometry to identify key odorant compounds in dark chocolate. Comparison of samples before and after conching. J. Agric. Food Chem. 2002, 50, 2385–2391. [Google Scholar] [CrossRef] [PubMed]
- Rigaud, J.; Escribano-Bailon, M.; Prieur, C.; Souquet, J.; Cheynier, V. Normal-phase high-performance liquid chromatographic separation of procyanidins from cacao beans and grape seeds. J. Chrom. A 1993, 654, 255–260. [Google Scholar] [CrossRef]
- Alañón, M.; Castle, S.; Siswanto, P.; Cifuentes-Gómez, T.; Spencer, J. Assessment of Flavanol Stereoisomers and Caffeine and Theobromine Content in Commercial Chocolates. Food Chem. 2016, 208, 177–184. [Google Scholar] [CrossRef]
- De Taeye, C.; Kankolongo Cibaka, M.L.; Jerkovic, V.; Collin, S. Degradation of (−)-Epicatechin and Procyanidin B2 in Aqueous and Lipidic Model Systems. First Evidence of “Chemical” Flavan-3-ol Oligomers in Processed Cocoa. J. Agric. Food Chem. 2014, 62, 9002–9016. [Google Scholar] [CrossRef]
- Cooper, K.A.; ECampos-Giménez Alvarez, D.J.; Nagy, K.; Donovan, J.L.; Williamson, G. Rapid Reversed Phase Ultra-Performance Liquid Chromatography Analysis of the Major Cocoa Polyphenols and Inter-relationships of Their Concentrations in Chocolate. J. Agric. Food Chem. 2007, 55, 2841–2847. [Google Scholar] [CrossRef]
- Hammerstone, J.; Lazarus, S.; Mitchell, A.; Rucker, R.; Schmitz, H. Identification of Procyanidins in Cocoa (Theobroma Cacao) and Chocolate Using High-Performance Liquid Chromatography/Mass Spectrometry. J. Agric. Food Chem. 1999, 47, 490–496. [Google Scholar] [CrossRef]
- Counet, C.; Collin, S. Effect of the number of flavanol units on the antioxidant activity of procyanidin fractions isolated from chocolate. J. Agric. Food Chem. 2003, 51, 6816–6822. [Google Scholar] [CrossRef]
- Vidal, S.; Francis, L.; Noble, A.; Kwiatkowski, M.; Cheynier, V.; Waters, E. Taste and mouth-feel properties of different types of tannin-like polyphenolic compounds and anthocyanins in wine. Anal. Chim. Acta 2004, 513, 57–65. [Google Scholar] [CrossRef]
- De Taeye, C.; Caullet, G.; Evina, V.E.; Collin, S. Procyanidin A2 and Its Degradation Products in Raw, Fermented, and Roasted Cocoa. J. Agric. Food Chem. 2017, 65, 1715–1723. [Google Scholar] [CrossRef] [PubMed]
- Lavit, C. Analyse Conjointe de Tableaux Quatitatifs; Masson: Paris, France, 1988. [Google Scholar]
- Robert, P.; Escoufier, Y. A Unifying Tool for Linear Multivariate Statistical Methods: The RV- Coefficient. J. R. Stat. Soc. Ser. C (Appl. Stat.) 1976, 25, 257–265. [Google Scholar] [CrossRef]
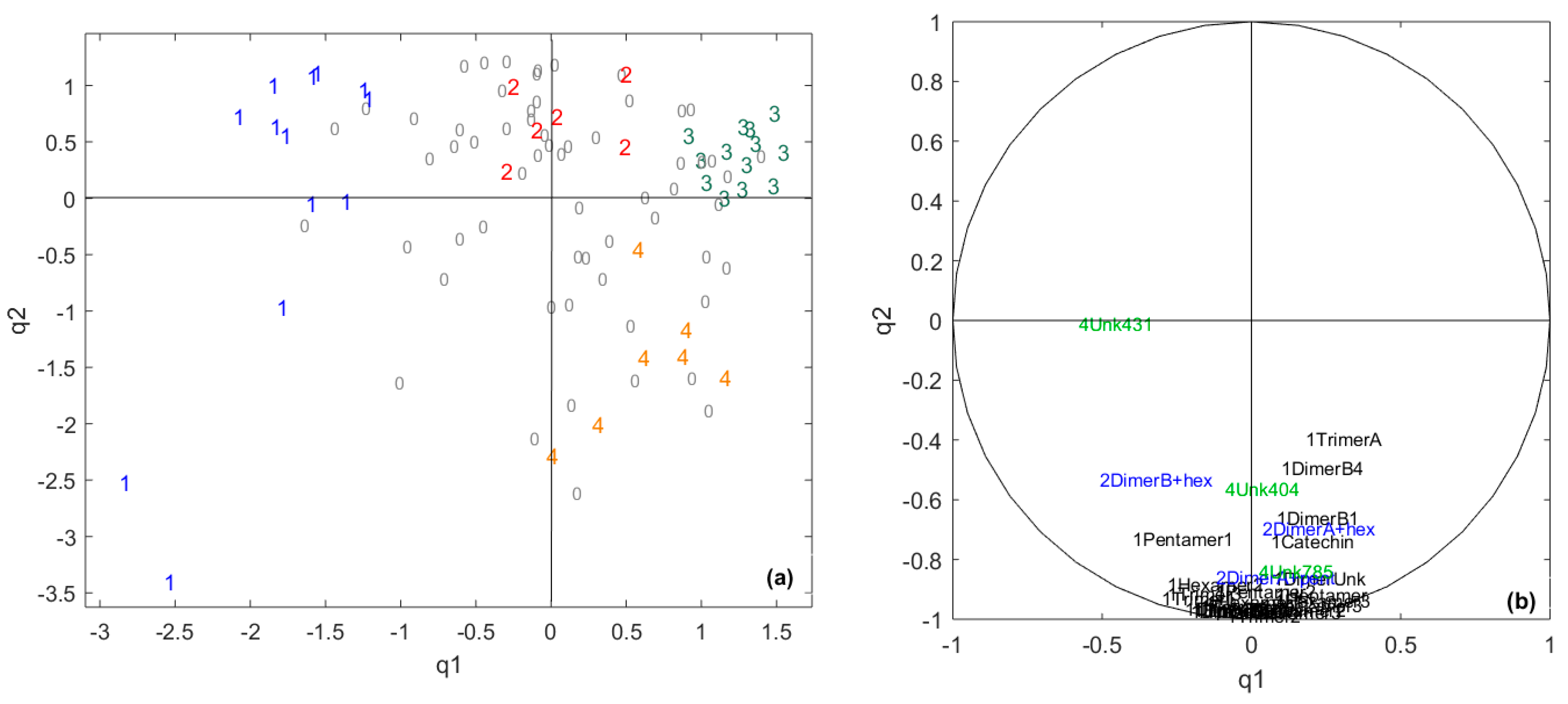
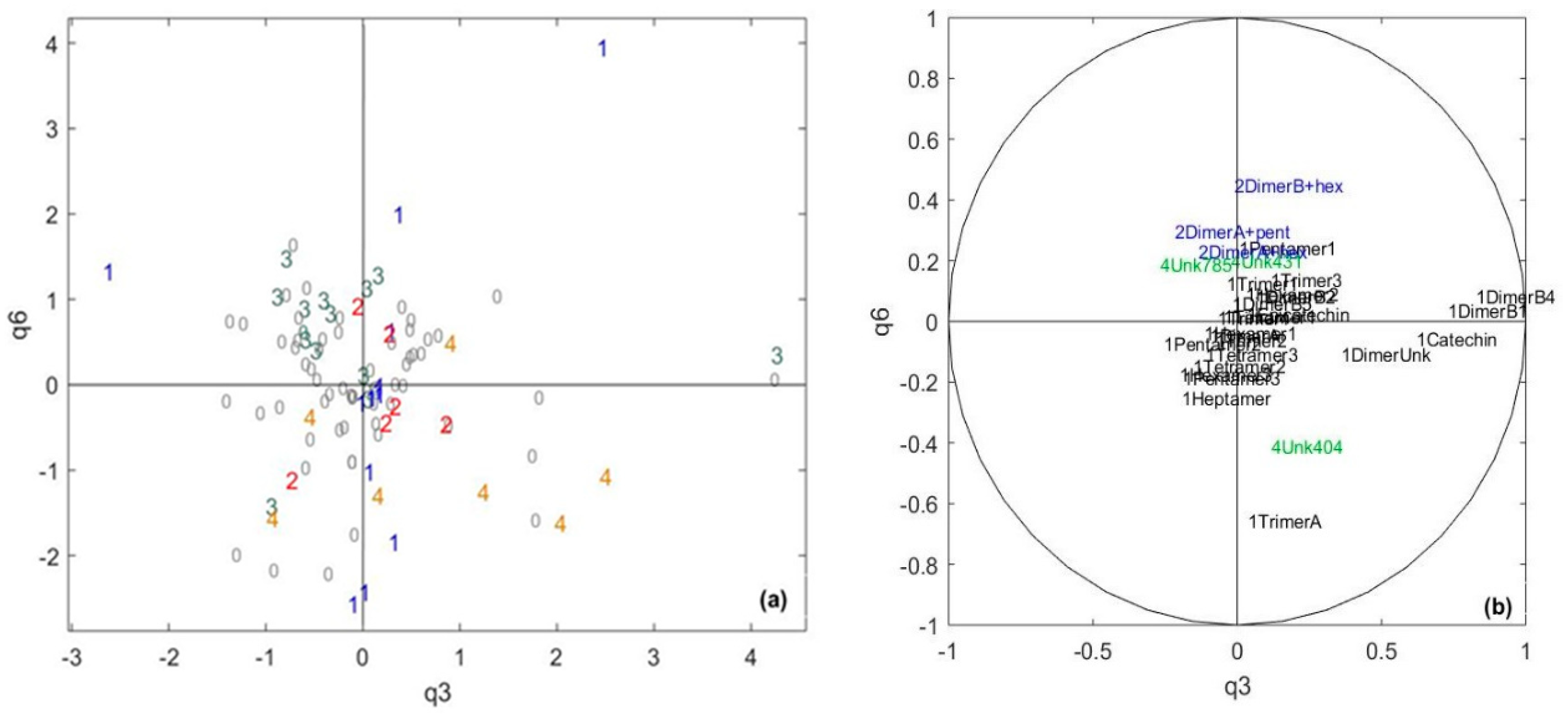
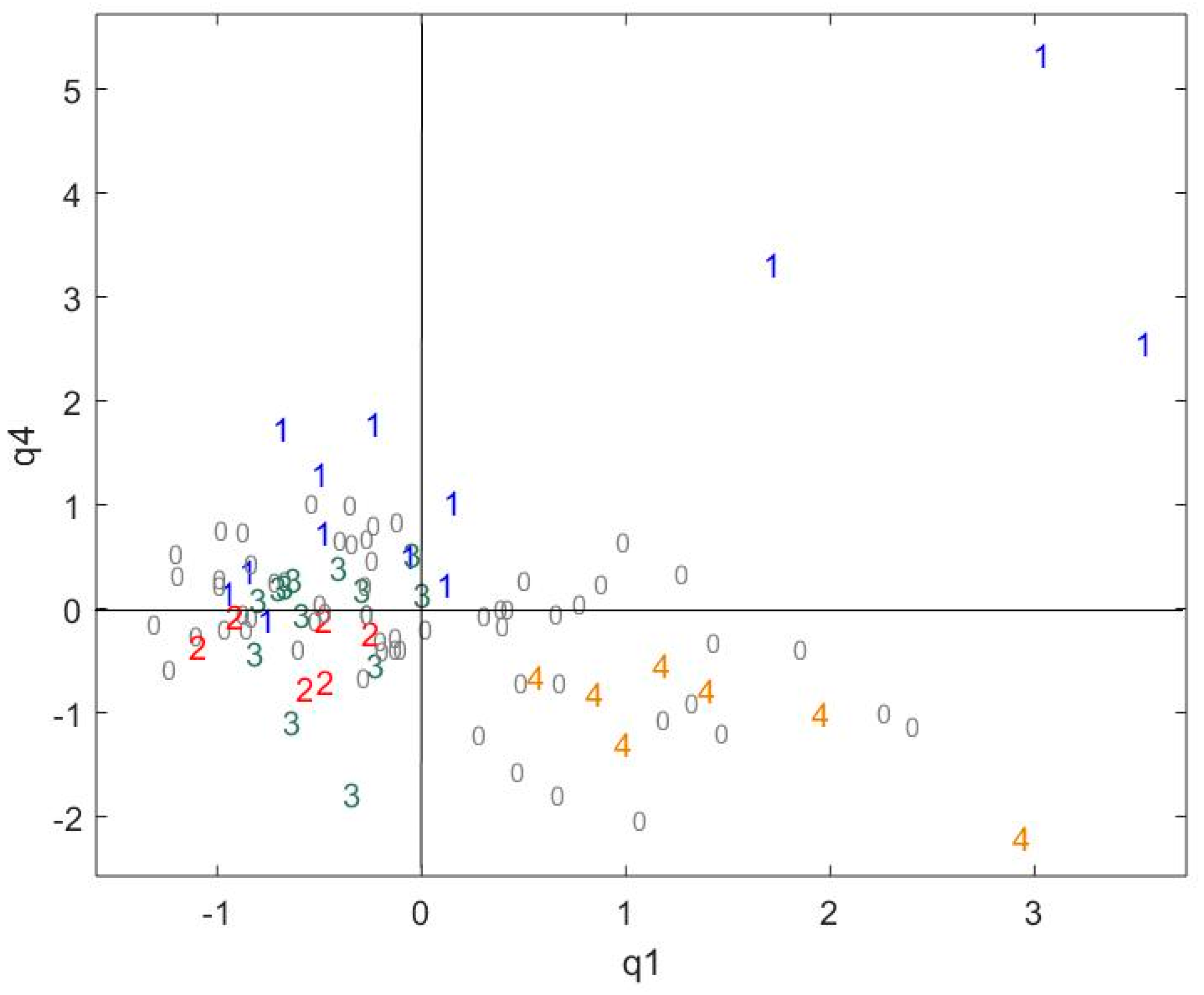
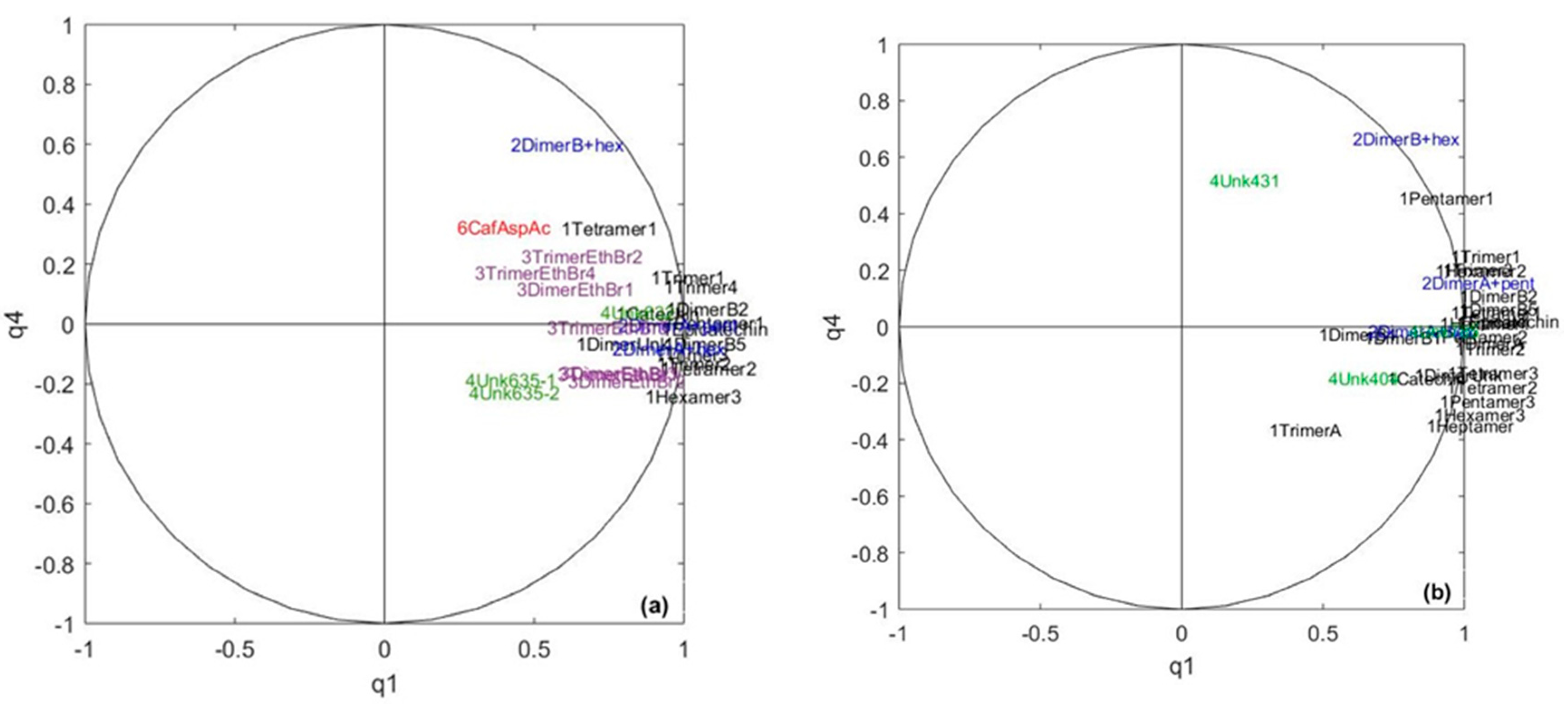
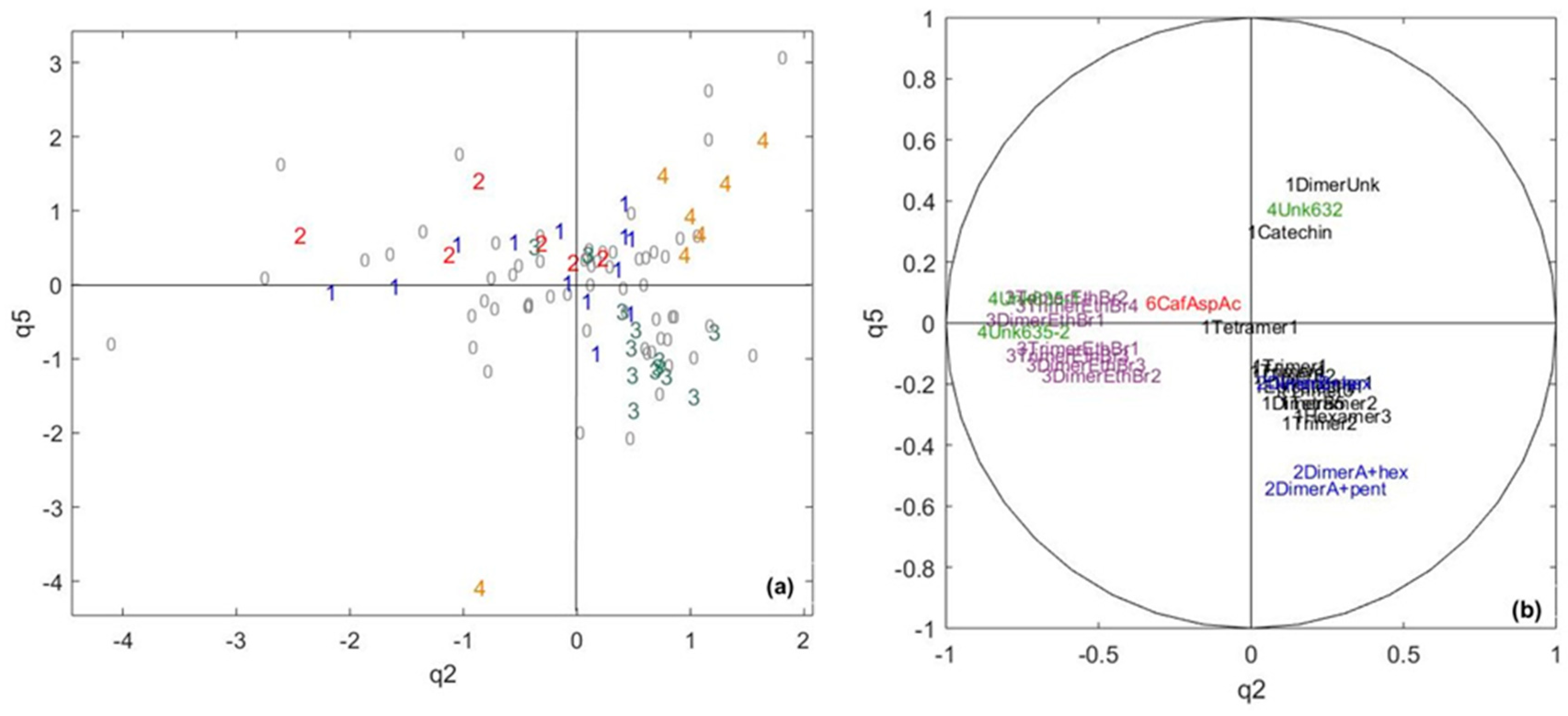
| Data Table/Common Component | q1 | q2 | q3 | q4 | q5 | q6 | q7 | q8 | q9 |
|---|---|---|---|---|---|---|---|---|---|
| Sensory | 0.48 | 0.08 | 0.00 | 0.15 | 0.06 | 0.00 | 0.00 | 0.04 | 0.00 |
| Polyphenols in chocolates | 0.04 | 0.70 | 0.07 | 0.00 | 0.00 | 0.04 | 0.03 | 0.00 | 0.01 |
| Specific Weights | q1 | q2 | q3 | q4 | q5 | q6 | q7 | q8 | q9 |
|---|---|---|---|---|---|---|---|---|---|
| Polyphenols in cocoa beans | 0.50 | 0.22 | 0.01 | 0.03 | 0.06 | 0.03 | 0.00 | 0.02 | 0.02 |
| Polyphenols in chocolates | 0.68 | 0.02 | 0.07 | 0.05 | 0.01 | 0.01 | 0.03 | 0.00 | 0.00 |
| Codes | Molecules Studied | Presence | Molecule Family |
|---|---|---|---|
| Catechin | Catechin | Cocoa + chocolate | Flavan-3-ol |
| Epicatechin | Epicatechin | Cocoa + chocolate | Flavan-3-ol |
| DimerB1 | Procyanidin dimerB1 | Chocolate | Flavan-3-ol |
| DimerB2 | Procyanidin dimer B2 | Cocoa + chocolate | Flavan-3-ol |
| DimerB4 | Procyanidin dimer B4 | Chocolate | Flavan-3-ol |
| DimerB5 | Procyanidin dimer B5 | Cocoa + chocolate | Flavan-3-ol |
| DimerUnk | Unknown procyanidin dimer | Cocoa + chocolate | Flavan-3-ol |
| A-type Dimer | Procyanidin A type dimer | Chocolate | Flavan-3-ol |
| Trimer1 | Procyanidin trimer | Cocoa + chocolate | Flavan-3-ol |
| Trimer2 | Procyanidin trimer C1 | Cocoa + chocolate | Flavan-3-ol |
| Trimer3 | Procyanidin trimer | Cocoa + chocolate | Flavan-3-ol |
| Trimer4 | Procyanidin trimer | Cocoa + chocolate | Flavan-3-ol |
| TrimerA | A type procyanidin trimer | Chocolate | Flavan-3-ol |
| Tetramer1 | Procyanidin tetramer | Cocoa + chocolate | Flavan-3-ol |
| Tetramer2 | Procyanidin tetramer | Cocoa + chocolate | Flavan-3-ol |
| Tetramer3 | Procyanidin tetramer | Chocolate | Flavan-3-ol |
| Pentamer1 | Procyanidin pentamer | Cocoa + chocolate | Flavan-3-ol |
| Pentamer2 | Procyanidin pentamer | Chocolate | Flavan-3-ol |
| Pentamer3 | Procyanidin pentamer | Chocolate | Flavan-3-ol |
| Hexamer1 | Procyanidin hexamer | Chocolate | Flavan-3-ol |
| Hexamer2 | Procyanidin hexamer | Chocolate | Flavan-3-ol |
| Hexamer3 | Procyanidin hexamer | Cocoa + chocolate | Flavan-3-ol |
| Heptamer | Procyanidin heptamer | Chocolate | Flavan-3-ol |
| DimerB+hex | B type procyanidin dimer C-hexoside | Cocoa + chocolate | Glycosylated flavan-3-ol |
| DimerA+hex | A type procyanidin dimer O-hexoside | Cocoa + chocolate | Glycosylated flavan-3-ol |
| DimerA+pent | A type procyanidin dimer O-pentoside | Cocoa + chocolate | Glycosylated flavan-3-ol |
| TrimerEthBr1 | Ethyl Bridged procyanidin trimer | Cocoa | Ethyl bridged flavan-3-ol |
| TrimerEthBr2 | Ethyl Bridged procyanidin trimer | Cocoa | Ethyl bridged flavan-3-ol |
| TrimerEthBr3 | Ethyl Bridged procyanidin trimer | Cocoa | Ethyl bridged flavan-3-ol |
| TrimerEthBr4 | Ethyl Bridged procyanidin trimer | Cocoa | Ethyl bridged flavan-3-ol |
| DimerEthBr1 | Ethyl Bridged procyanidin dimer | Cocoa | Ethyl bridged flavan-3-ol |
| DimerEthBr2 | Ethyl Bridged procyanidin dimer | Cocoa | Ethyl bridged flavan-3-ol |
| DimerEthBr3 | Ethyl Bridged procyanidin dimer | Cocoa | Ethyl bridged flavan-3-ol |
| CafAspAc | Caffeoyl aspartic acid | Cocoa | Hydroxycinnamic acid |
| Unk632 | Unknown compound at m/z 632 | Cocoa | Unknown |
| Unk635-1 | Unknown compound at m/z 635 | Cocoa | Unknown |
| Unk635-2 | Unknown compound at m/z 635 | Cocoa | Unknown |
| Unk404 | Unknown compound at m/z 404 | Chocolate | Unknown |
| Unk431 | Unknown compound at m/z 431 | Chocolate | Unknown |
| Unk785 | Unknown compound at m/z 785 | Chocolate | Unknown |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fayeulle, N.; Preys, S.; Roger, J.-M.; Boulanger, R.; Hue, C.; Cheynier, V.; Sommerer, N. Multiblock Analysis to Relate Polyphenol Targeted Mass Spectrometry and Sensory Properties of Chocolates and Cocoa Beans. Metabolites 2020, 10, 311. https://doi.org/10.3390/metabo10080311
Fayeulle N, Preys S, Roger J-M, Boulanger R, Hue C, Cheynier V, Sommerer N. Multiblock Analysis to Relate Polyphenol Targeted Mass Spectrometry and Sensory Properties of Chocolates and Cocoa Beans. Metabolites. 2020; 10(8):311. https://doi.org/10.3390/metabo10080311
Chicago/Turabian StyleFayeulle, Noémie, Sébastien Preys, Jean-Michel Roger, Renaud Boulanger, Clotilde Hue, Véronique Cheynier, and Nicolas Sommerer. 2020. "Multiblock Analysis to Relate Polyphenol Targeted Mass Spectrometry and Sensory Properties of Chocolates and Cocoa Beans" Metabolites 10, no. 8: 311. https://doi.org/10.3390/metabo10080311
APA StyleFayeulle, N., Preys, S., Roger, J.-M., Boulanger, R., Hue, C., Cheynier, V., & Sommerer, N. (2020). Multiblock Analysis to Relate Polyphenol Targeted Mass Spectrometry and Sensory Properties of Chocolates and Cocoa Beans. Metabolites, 10(8), 311. https://doi.org/10.3390/metabo10080311