A Simple GC-MS/MS Method for Determination of Smoke Taint-Related Volatile Phenols in Grapes
Abstract
1. Introduction
2. Results
2.1. Optimization of GC-MS/MS Method
2.2. Selecting the Optimal Protocol for Extracting Free and Conjugated Phenols
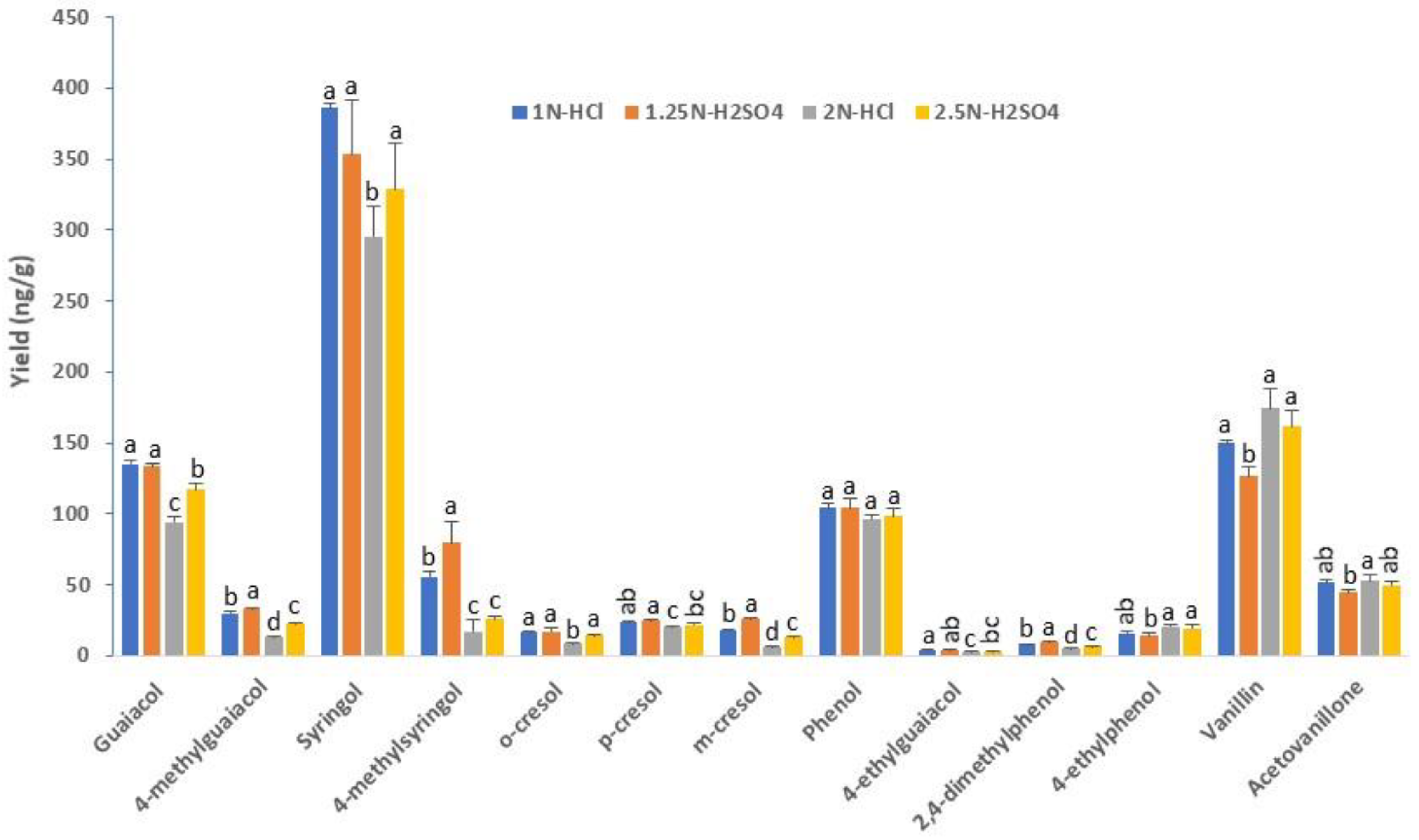
2.3. Selecting the Optimal Conditions for Phenol Glycoside Hydrolysis
2.4. Method Accuracy
2.5. Method Reproducibility
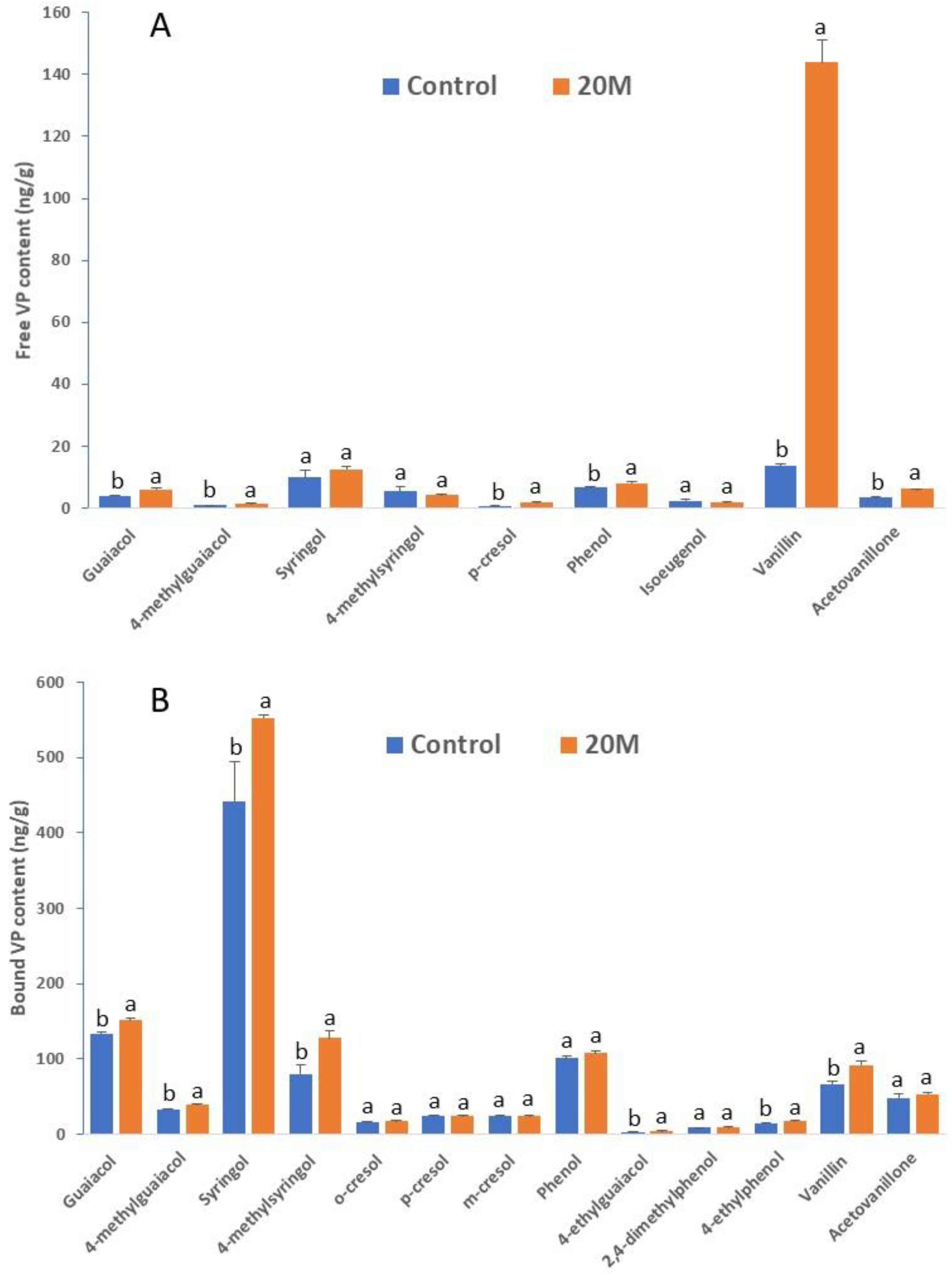
2.6. Method Application
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Chemicals and Reagents
4.3. Extraction of Free and Glycosylated Phenols from Grapes
4.4. Sample Preparation for GC-MS
4.5. GC-MS/MS Settings
4.6. LC-MS Analysis of Phenol Glycosides
4.7. Method Validation
4.8. Statistical Analysis of Data
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kristic, M.P.; Johnson, D.L.; Herderich, M.J. Review of smoke taint in wine: Smoke-derived volatile phenols and their glycosidic metabolites in grapes and vines as biomarkers for smoke exposure and their role in the sensory perception of smoke taint. Aust. J. Grape Wine Res. 2015, 21, 537–553. [Google Scholar] [CrossRef]
- Kennison, K.R.; Wilkinson, K.L.; Williams, H.G.; Smith, J.H.; Gibberd, M.R. Smoke-derived taint in wine: Effect of postharvest smoke exposure of grapes on the chemical composition and sensory characteristics of wine. J. Agric. Food Chem. 2007, 55, 10897–10901. [Google Scholar] [CrossRef] [PubMed]
- Parker, M.; Osidacz, P.; Baldock, G.A.; Hayasaka, Y.; Black, C.A.; Pardon, K.H.; Jeffery, D.W.; Geue, J.P.; Herderich, M.J.; Francis, I.L. Contribution of several volatile phenols and their glycoconjugates to smoke-related sensory properties of red wine. J. Agric. Food Chem. 2012, 60, 2629–2637. [Google Scholar] [CrossRef]
- Singh, D.P.; Chong, H.H.; Pitt, K.M.; Cleary, M.; Dokoozlian, N.K.; Downey, M.O. Guaiacol and 4-methylguaiacol accumulate in wines made from smoke-affected fruit because of hydrolysis of their conjugates. Aust. J. Grape Wine Res. 2011, 17, S13–S21. [Google Scholar] [CrossRef]
- Caffrey, A.; Lerno, L.; Rumbaugh, A.; Girardello, R.; Zweigenbaum, J.; Oberholster, A.; Ebeler, S.E. Changes in smoke-taint volatile-phenol glycosides in wildfire smoke-exposed Cabernet Sauvignon grapes throughout winemaking. Am. J. Enol. Vitic. 2019, 70, 373–381. [Google Scholar] [CrossRef]
- Caboni, P.; Sarais, G.; Cabras, M.; Angioni, A. Determination of 4-ethylphenol and 4-ethylquaiacol in wines by LC-MS-MS and HPLC-DAD-fluorescence. J. Agric. Food Chem. 2007, 55, 7288–7293. [Google Scholar] [CrossRef]
- Wilkinson, K.L.; Ristic, R.; Pinchbeck, K.A.; Fudge, A.L.; Singh, D.P.; Pitt, K.M.; Downey, M.O.; Baldock, G.A.; Hayasaka, Y.; Parker, M.; et al. Comparison of methods for the analysis of smoke related phenols and their conjugates in grapes and wine. Aust. J. Grape Wine Res. 2011, 17, S22–S28. [Google Scholar] [CrossRef]
- Pizarro, C.; Pérez-del-Notario, N.; González-Sáiz, J.M. Optimisation of a simple and reliable method based on headspace solid-phase microextraction for the determination of volatile phenols in beer. J. Chromatogr. A 2010, 1217, 6013–6021. [Google Scholar] [CrossRef]
- Zhou, Q.; Qian, Y.; Qian, M.C. Analysis of volatile phenols in alcoholic beverage by ethylene glycol-polydimethylsiloxane based stir bar sorptive extraction and gas chromatography–mass spectrometry. J. Chromatogr. A 2015, 1390, 22–27. [Google Scholar] [CrossRef]
- Allen, D.; Bui, A.D.; Cain, N.; Rose, G.; Downey, M. Analysis of free and bound phenolics in wine and grapes by GC-MS after automated SPE. Anal. Bioanal. Chem. 2013, 405, 9869–9877. [Google Scholar] [CrossRef]
- Hayasaka, Y.; Parker, M.; Baldock, G.A.; Pardon, K.H.; Black, C.A.; Jeffery, D.W. Assessing the impact of smoke exposure in grapes: Development and validation of a HPLC-MS/MS Method for the quantitative analysis of smoke-derived phenolic glycosides in grapes and wine. J. Agric. Food Chem. 2013, 61, 25–33. [Google Scholar] [CrossRef]
- Van der Hulst, L.; Munguia, P.; Culbert, J.A.; Ford, C.M.; Burton, R.A.; Wilkinson, K.L. Accumulation of volatile phenol glycoconjugates in grapes following grapevine exposure to smoke and potential mitigation of smoke taint by foliar application of kaolin. Planta 2019, 249, 941–952. [Google Scholar] [CrossRef] [PubMed]
- Kennison, K.R.; Gibberd, M.R.; Pollnitz, A.P.; Wilkinson, K.L. Smoke-derived taint in wine: The release of smoke-derived volatile phenols during fermentation of Merlot juice following grapevine exposure to smoke. J. Agric. Food Chem. 2008, 56, 7379–7383. [Google Scholar] [CrossRef] [PubMed]
- Hayasaka, Y.; Baldock, G.A.; Parker, M.; Pardon, K.H.; Black, C.A.; Herderich, M.J.; Jeffery, D.W. Glycosylation of smoke-derived volatile phenols in grapes as a consequence of grapevine exposure to bushfire smoke. J. Agric. Food Chem. 2010, 58, 10989–10998. [Google Scholar] [CrossRef]
- Hayasaka, Y.; Dungey, K.A.; Baldock, G.A.; Kennison, K.R.; Wilkinson, K.L. Identification of a β-D-glucopyranoside precursor to guaiacol in grape juice following grapevine exposure to smoke. Anal. Chim. Acta 2010, 660, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.P.; Zerihun, A.; Kelly, D.; Cain, N.M.; Nankervis, P.; Downey, M.O. A GC-MS based analytical method for detection of smoke taint associated phenols in smoke affected wines. Curr. Bioact. Compd. 2012, 8, 190–199. [Google Scholar] [CrossRef]
- Noestheden, M.; Thiessen, K.; Dennis, E.G.; Tiet, B.; Zandberg, W.F. Quantitating organoleptic volatile phenols in smoke-exposed Vitis vinifera berries. J. Agric. Food. Chem. 2017, 65, 8418–8425. [Google Scholar] [CrossRef]
- Pollnitz, A.P.; Pardon, K.H.; Sefton, M.A. Quantitative analysis of 4-ethylphenol and 4-ethylguaiacol in red wine. J. Chromatogr. A 2000, 874, 101–109. [Google Scholar] [CrossRef]
- Pollnitz, A.P.; Pardon, K.H.; Sykes, M.; Sefton, M.A. The effects of sample preparation and gas chromatograph injection techniques on the accuracy of measuring guaiacol, 4-methylguaiacol and other volatile oak compounds in oak extracts by stable isotope dilution analyses. J. Agric. Food Chem. 2004, 52, 3244–3252. [Google Scholar] [CrossRef]
- Hayasaka, Y.; Baldock, G.A.; Pardon, K.H.; Jeffery, D.W.; Herderich, M.J. Investigation into the formation of Guaiacol conjugates in berries and leaves of grapevine Vitis vinifera L. Cv. Cabernet sauvignon using stable isotope tracers combined with HPLC-MS and MS/MS analysis. J. Agric. Food Chem. 2010, 58, 2076–2081. [Google Scholar]
- Noestheden, M.; Dennis, E.G.; Zandberg, W.F. Quantitating volatile phenols in Cabernet Franc berries and wine after on-vine exposure to smoke from a simulated forest fire. J. Agric. Food Chem. 2018, 66, 695–703. [Google Scholar] [CrossRef]
- Liu, Z.; Rochfort, S. Recent progress in polar metabolite quantification in plants using liquid chromatography–mass spectrometry. J. Integr. Plant. Biol. 2014, 56, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Ristic, R.; Fudge, A.L.; Pinchbeck, K.A.; de Bei, R.; Fuentes, S.; Hayasaka, Y.; Tyerman, S.D.; Wilkinson, K.L. Impact of grapevine exposure to smoke on vine physiology and the composition and sensory properties of wine. Exp. Plant. Physiol. 2016, 28, 67–83. [Google Scholar] [CrossRef]
- Liu, Z.; Rochfort, S. A simple method for simultaneous quantification of total arabinoxylans and fructans in wheat flour. J. Agric. Food Chem. 2014, 62, 6319–6324. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Rochfort, S. A fast liquid chromatography-mass spectrometry (LC-MS) method for quantification of major polar metabolites in plants. J. Chromatogr. B 2013, 912, 8–15. [Google Scholar] [CrossRef]
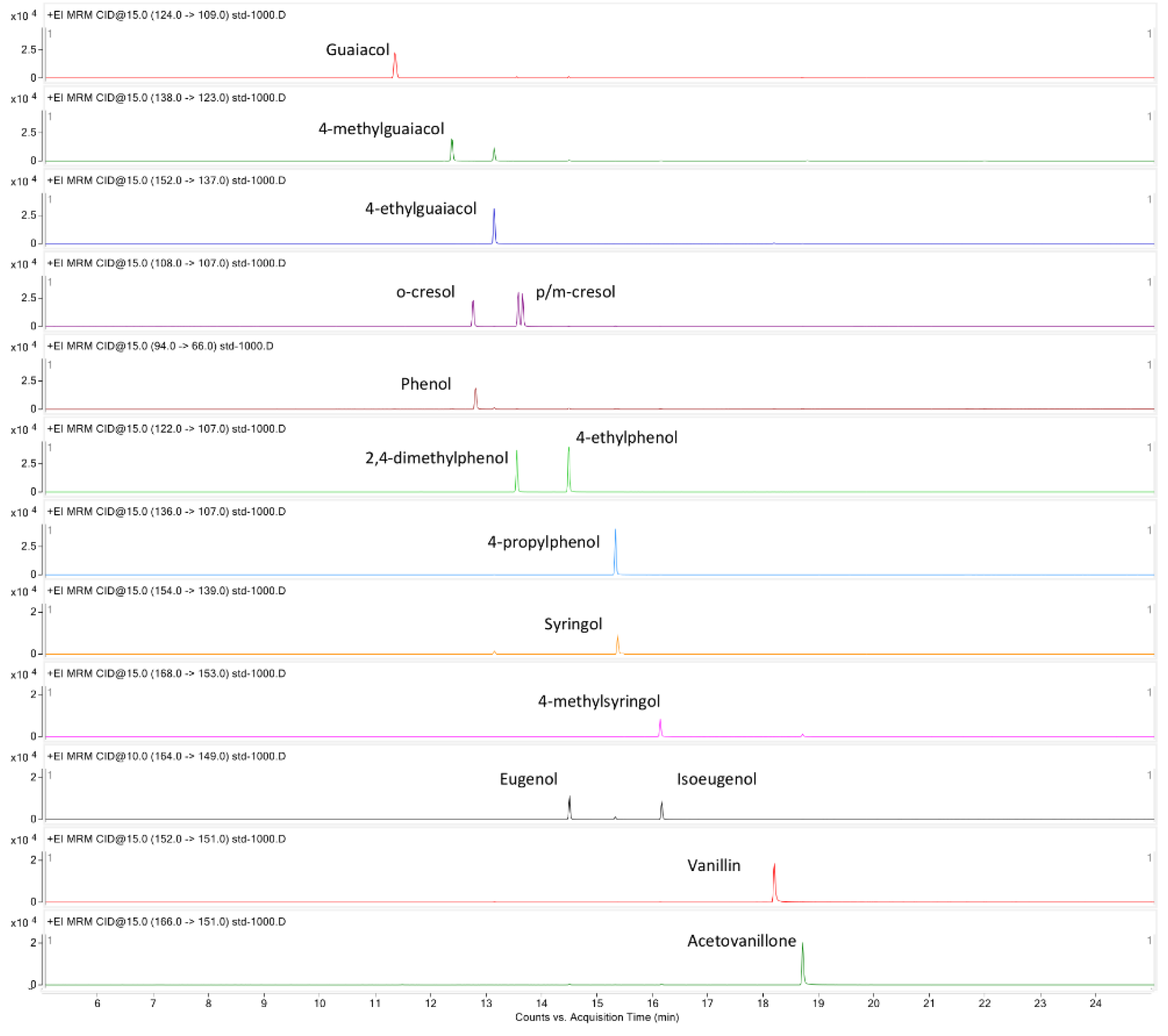
| Name | Precursor (m/z) | Production (m/z) | C. E. (eV) | R.T. (min) | LOD (ng/mL) | LOQ (ng/mL) | Linear Range (ng/mL) | R2 |
|---|---|---|---|---|---|---|---|---|
| Guaiacol | 124 | 109 | 15 | 11.307 | 0.33 | 1 | 1–1000 | 0.9998 |
| d3-guaiacol (IS) | 127 | 109 | 15 | 11.292 | 0.33 | 1 | 1–1000 | 0.9998 |
| 4-methylguaiacol | 138 | 123 | 15 | 12.326 | 0.33 | 1 | 1–1000 | 0.9998 |
| Syringol | 154 | 139 | 15 | 15.320 | 0.33 | 1 | 1–1000 | 0.9990 |
| 4-methylsyringol | 168 | 153 | 15 | 16.081 | 1 | 3.3 | 3.3–1000 | 0.9981 |
| p-cresol | 108 | 107 | 15 | 12.712 | 0.33 | 1 | 1–1000 | 0.9995 |
| o-cresol | 108 | 107 | 15 | 13.529 | 0.33 | 1 | 1–1000 | 0.9996 |
| m-cresol | 108 | 107 | 15 | 13.611 | 0.33 | 1 | 1–1000 | 0.9992 |
| Phenol | 94 | 66 | 15 | 12.754 | 0.33 | 1 | 1–1000 | 0.9993 |
| 4-ethylguaiacol | 152 | 137 | 15 | 13.101 | 0.33 | 1 | 1–1000 | 0.9999 |
| Eugenol | 164 | 149 | 10 | 14.447 | 1 | 3.3 | 3.3–1000 | 1.0000 |
| Isoeugenol | 164 | 149 | 10 | 16.110 | 1 | 3.3 | 3.3–1000 | 0.9998 |
| 2,4-dimethylphenol | 122 | 107 | 15 | 13.501 | 0.33 | 1 | 1–1000 | 0.9996 |
| 4-ethylphenol | 122 | 107 | 15 | 14.442 | 0.33 | 1 | 1–1000 | 0.9996 |
| 4-n-propylphenol | 136 | 107 | 15 | 15.284 | 0.33 | 1 | 1–1000 | 0.9997 |
| Vanillin | 152 | 151 | 15 | 18.183 | 1 | 3.3 | 3.3–1000 | 0.9999 |
| Acetovanillone | 166 | 151 | 15 | 18.668 | 1 | 3.3 | 3.3–1000 | 0.9999 |
| VPs | Control | 20M |
|---|---|---|
| Guaiacol | 69.0±1.6 b | 81.0±0.3 a |
| 4-methylguaiacol | 65.4±2.4 b | 77.2±1.2 a |
| Syringol | 71.3±5.0 a | 79.6±4.3 a |
| 4-methylsyringol | 73.8±3.6 a | 76.1±4.2 a |
| p-cresol | 48.3±0.5 b | 62.9±2.0 a |
| o-cresol | 63.9±2.2 b | 78.6±2.4 a |
| m-cresol | 62.6±2.8 b | 75.3±3.2 a |
| Phenol | 55.3±2.5 b | 74.2±2.6 a |
| 4-ethylguaiacol | 52.8±1.2 b | 71.4±3.9 a |
| Eugenol | 59.4±1.3 b | 74.0±3.2 a |
| Isoeugenol | 43.9±1.1 b | 62.2±5.8 a |
| 2,4-dimethylphenol | 46.4±1.1 b | 69.7±2.6 a |
| 4-ethylphenol | 19.5±0.5 b | 51.4±2.0 a |
| 4-n-propylphenol | 10.9±0.6 b | 38.6±2.3 a |
| Vanillin | 54.6±9.8 b | 84.9±7.8 a |
| Acetovanillone | 48.5±6.7 b | 83.0±4.6 a |
| VPs | VP Form | Lab-A (µg/L) * | Lab-B (µg/g) ** | Lab-C (µg/g) | Our Lab (µg/g) |
|---|---|---|---|---|---|
| Guaiacol | Free | 6.7 | 9 | 7 | 6 |
| Bound | 8.3 | 3.7 | 51 | 16 | |
| 4-methylguaiacol | Free | <1 | 1.7 | <5 | 1.1 |
| Bound | 2 | - | <5 | 3.5 | |
| Syringol | Free | 1 | <2 | <3 | 5.1 |
| Bound | 52 | 37 | 48 | 41 | |
| 4-methylsyringol | Free | <1 | <2 | <5 | < 1 |
| Bound | 9.7 | 3 | 8 | 9.0 | |
| o-cresol | Free | 4 | 7.7 | <3 | 3.5 |
| Bound | <1 | - | 4 | 2.7 | |
| p-cresol | Free | <1 | 2 | 7 | 1 |
| Bound | 1.3 | - | 4 | 5.5 | |
| m-cresol | Free | 3 | 6 | <3 | 2.6 |
| Bound | 2 | - | 8 | 3.6 |
| VPs | Free VPs | Bound VPs | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Shiraz | Chardonnay | Pinot | Shiraz | Chardonnay | Pinot | |||||||
| Mean | RSD | Mean | RSD | Mean | RSD | Mean | RSD | Mean | RSD | Mean | RSD | |
| (ng/g) | (%) | (ng/g) | (%) | (ng/g) | (%) | (ng/g) | (%) | (ng/g) | (%) | (ng/g) | (%) | |
| Guaiacol | 6.4 | 7.8 | 4.4 | 13.6 | 7.8 | 4.1 | 143.4 | 5.1 | 50.7 | 2.9 | 41.8 | 4.1 |
| 4-methylguaiacol | 1.9 | 6.8 | 1.3 | 17.3 | 2.4 | 10.2 | 37.2 | 7.9 | 23.3 | 7.2 | 13.2 | 3.6 |
| Syringol | 59.3 | 8.6 | 47.7 | 12.9 | 40.9 | 7.2 | 988.7 | 11.3 | 540.7 | 5.7 | 394.9 | 2.2 |
| 4-methylsyringol | 544.9 | 11.3 | 200.7 | 5.4 | 147.5 | 9.0 | ||||||
| o-cresol | 1.0 | 11.6 | 2.6 | 3.9 | 13.1 | 5.9 | 11.5 | 5.2 | 7.7 | 9.2 | ||
| p-cresol | 1.8 | 7.4 | 1.2 | 10.7 | 2.8 | 3.9 | 16.3 | 6.4 | 16.9 | 3.2 | 14.1 | 4.6 |
| m-cresol | 1.2 | 13.7 | 1.3 | 8.1 | 2.9 | 7.7 | 17.2 | 4.0 | 14.5 | 7.7 | 10.8 | 4.5 |
| Phenol | 12.4 | 5.0 | 11.9 | 6.1 | 17.4 | 3.7 | 79.4 | 4.5 | 72.4 | 3.0 | 61.2 | 2.0 |
| 4-ethylguaiacol | 4.3 | 8.7 | 4.6 | 10.3 | 3.1 | 8.7 | ||||||
| Eugenol | 4.0 | 6.7 | 5.7 | 8.9 | 4.1 | 21.3 | ||||||
| 2,4-dimethylphenol | 5.9 | 5.6 | 6.2 | 7.8 | 3.8 | 10.8 | ||||||
| 4-ethylphenol | 1.4 | 7.7 | 1.4 | 9.8 | 1.7 | 9.3 | 16.7 | 11.0 | 10.5 | 11.2 | 10.6 | 10.2 |
| Vanillin | 242.4 | 10.5 | 206.1 | 7.8 | 200.9 | 3.2 | 188.0 | 4.2 | 120.3 | 6.7 | 133.7 | 2.69 |
| Acetovanillone | 9.6 | 10.5 | 7.4 | 8.2 | 12.9 | 12.8 | 74.3 | 4.7 | 55.7 | 3.6 | 83.6 | 3.4 |
| VPs | Free VPs | Bound VPs | ||||
|---|---|---|---|---|---|---|
| L | M | H | L | M | H | |
| Guaiacol | 2.7 (10.6) | 4.3 (4.7) | 4.6 (5.2) | 24.8 (2.9) | 29.3 (6.9) | 117.6 (2.8) |
| 4-methylguaiacol | <1 | <1 | 1.5 (6.6) | 7.2 (4.8) | 12.6 (25.4) | 19.3 (3.9) |
| Syringol | 6.4 (15.7) | 37.4 (16.4) | 17.7 (24.0) | 47.1 (20.1) | 78.3 (20.9) | 486.3 (1.7) |
| 4-methylsyringol | 4.5 (9.9) | 2.9 (13.6) | 6.6 (14.5) | 32.2 (18.8) | <2.5 | 17.0 (19.6) |
| o-cresol | <1 | <1 | <1 | <2.5 | 2.5 (16.1) | 11.9 (6.1) |
| p-cresol | <1 | <1 | <1 | 6.3 (6.2) | 12.5 (7.1) | 22.4 (3.9) |
| m-cresol | <1 | <1 | <1 | 4.4 (4.4) | 5.6 (5.3) | 8.4 (2.4) |
| Phenol | 6.4 (11.2) | 6.2 (1.3) | 6.6 (6.2) | 50.5 (3.7) | 88.9 (2.8) | 94.7 (12.1) |
| 4-ethylguaiacol | <1 | <1 | <1 | <2.5 | <2.5 | 3.9 (16.7) |
| Isoeugenol | 2.7 (2.7) | 3.7 (6.7) | 3.2 (32.4) | |||
| 2,4-dimethylphenol | <2.5 | 2.8 (7.6) | 6.3 (5.7) | |||
| 4-ethylphenol | <1 | <1 | <1 | 29.8 (5.4) | 65.0 (4.4) | 30.5 (3.0) |
| Vanillin | 175.6 (4.8) | 110.5 (7.6) | 219.5 (8.1) | 139.7 (4.6) | 136.1 (4.9) | 160.9 (7.2) |
| Acetovanillone | 5.6 (5.7) | 9.7 (7.4) | 8.0 (2.8) | 43.3 (3.7) | 38.4 (5.7) | 63.1 (9.3) |
| Sum of all VPs | 204.0 | 174.7 | 267.7 | 385.3 | 472.0 | 1042.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Z.; Ezernieks, V.; Reddy, P.; Elkins, A.; Krill, C.; Murphy, K.; Rochfort, S.; Spangenberg, G. A Simple GC-MS/MS Method for Determination of Smoke Taint-Related Volatile Phenols in Grapes. Metabolites 2020, 10, 294. https://doi.org/10.3390/metabo10070294
Liu Z, Ezernieks V, Reddy P, Elkins A, Krill C, Murphy K, Rochfort S, Spangenberg G. A Simple GC-MS/MS Method for Determination of Smoke Taint-Related Volatile Phenols in Grapes. Metabolites. 2020; 10(7):294. https://doi.org/10.3390/metabo10070294
Chicago/Turabian StyleLiu, Zhiqian, Vilnis Ezernieks, Priyanka Reddy, Aaron Elkins, Christian Krill, Kieran Murphy, Simone Rochfort, and German Spangenberg. 2020. "A Simple GC-MS/MS Method for Determination of Smoke Taint-Related Volatile Phenols in Grapes" Metabolites 10, no. 7: 294. https://doi.org/10.3390/metabo10070294
APA StyleLiu, Z., Ezernieks, V., Reddy, P., Elkins, A., Krill, C., Murphy, K., Rochfort, S., & Spangenberg, G. (2020). A Simple GC-MS/MS Method for Determination of Smoke Taint-Related Volatile Phenols in Grapes. Metabolites, 10(7), 294. https://doi.org/10.3390/metabo10070294





