Involvement of Lactate and Pyruvate in the Anti-Inflammatory Effects Exerted by Voluntary Activation of the Sympathetic Nervous System
Abstract
1. Introduction
2. Results
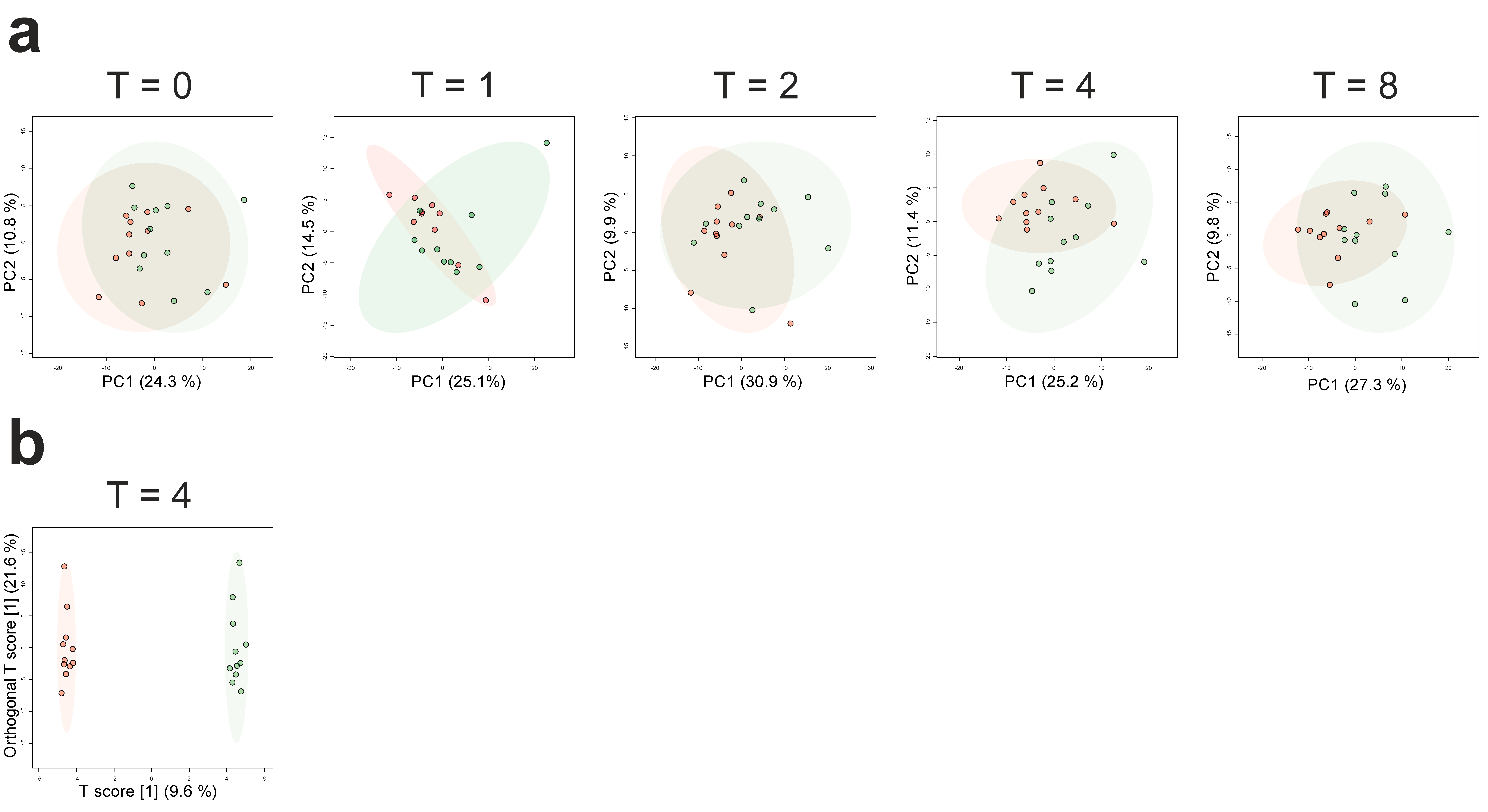
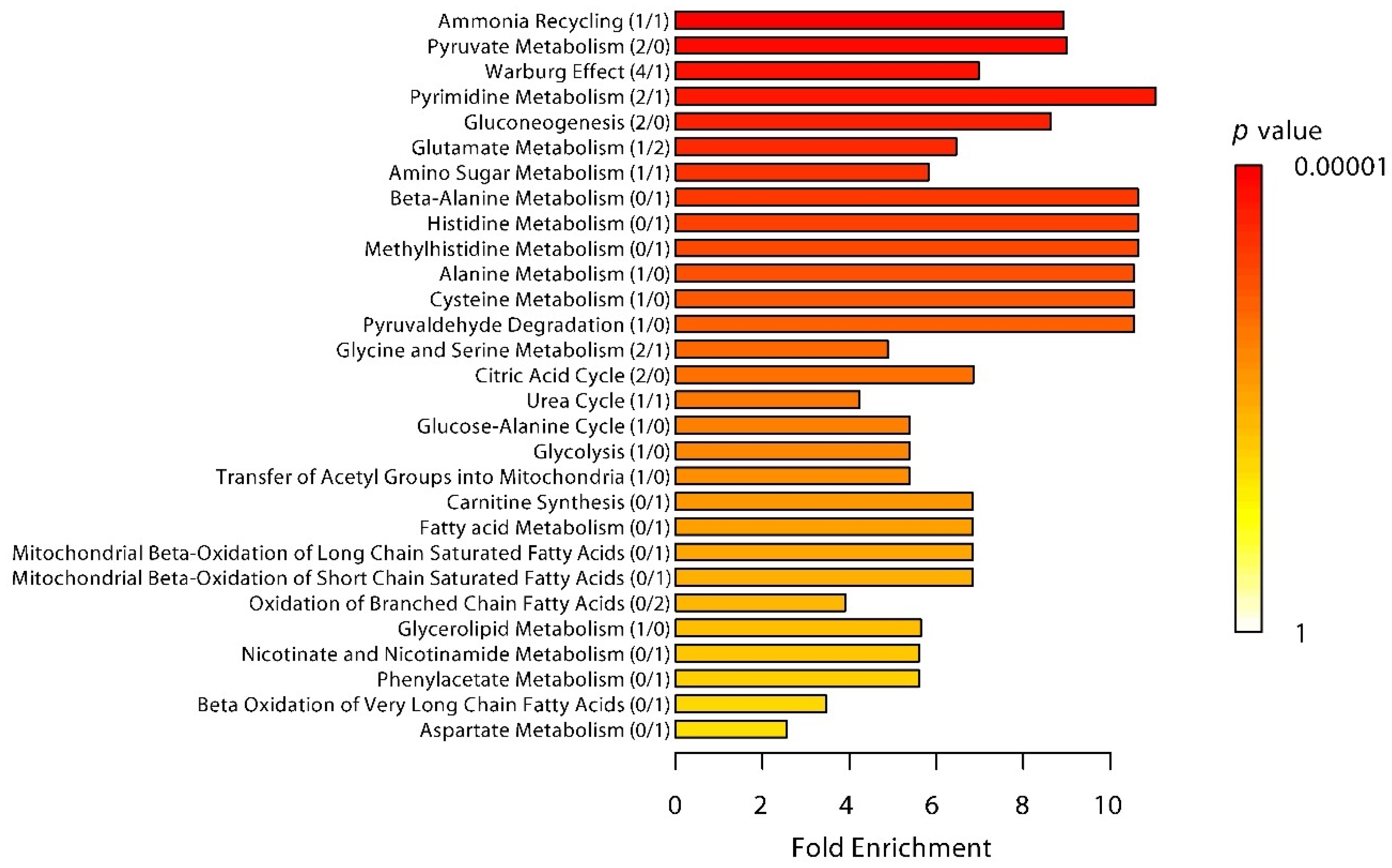
2.1. Effects of LPS Administration on Plasma Metabolites
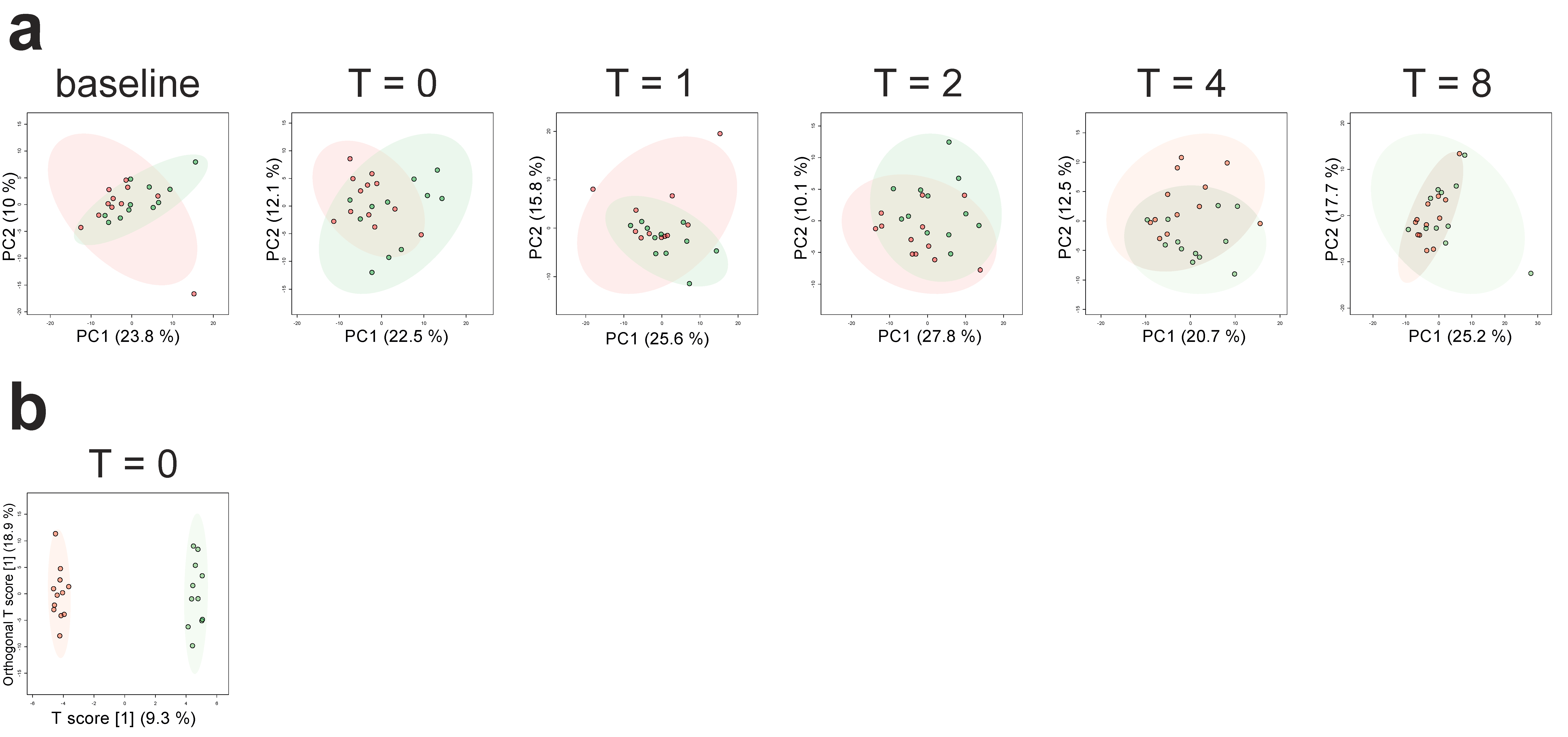
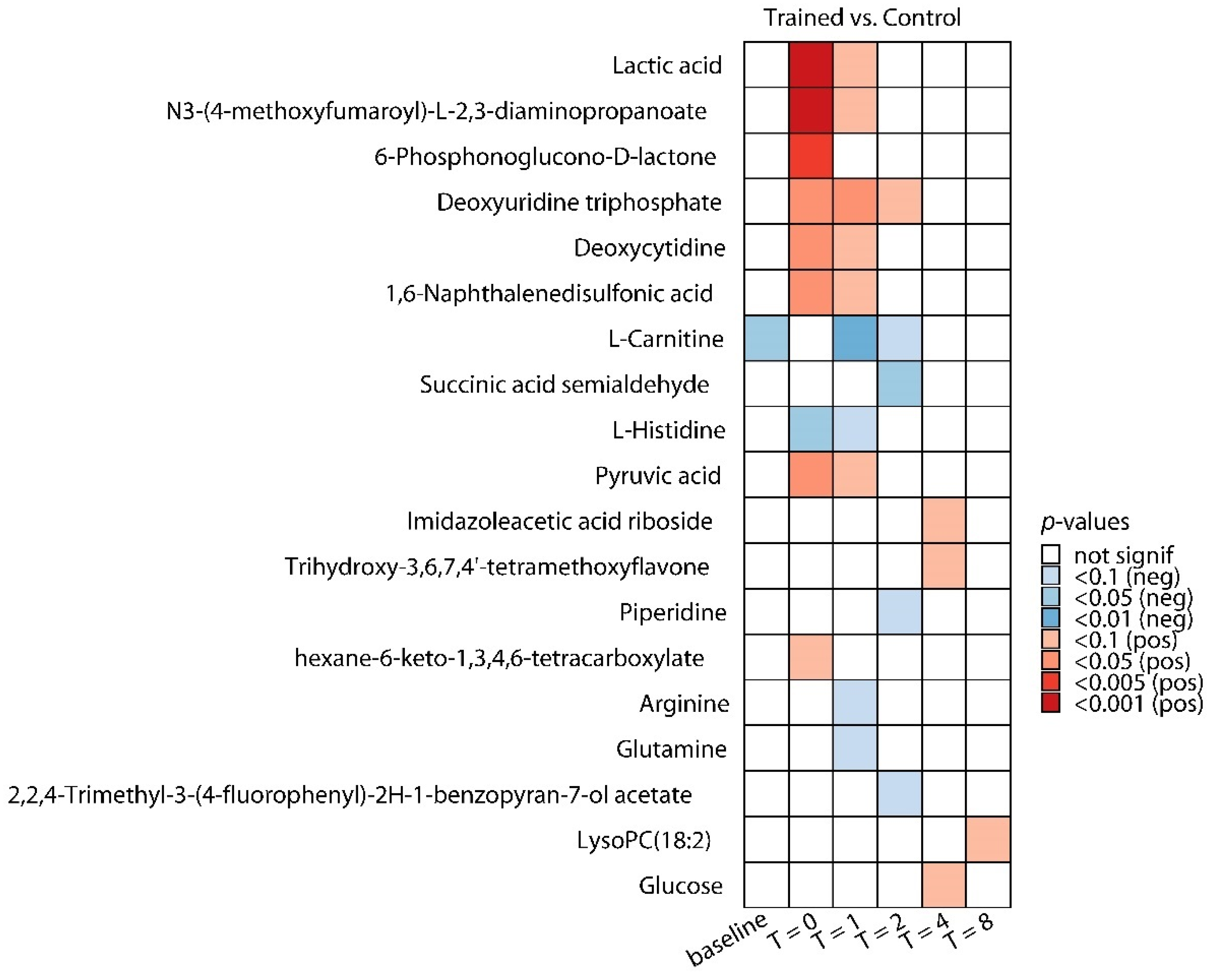
2.2. Differences in Plasma Metabolites in Trained and Untrained Individuals
2.3. Relationship between Differentially Regulated Metabolites and Inflammatory Response Mediators
3. Discussion
4. Materials and Methods
4.1. Subjects and Experimental Design
4.2. Sample Preparation for LC-MS Analysis
4.3. LC-MS Analysis
4.4. Cytokine and Lactate Determinations
4.5. PBMC Stimulation Experiments
4.6. Raw Data Processing and Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kox, M.; van Eijk, L.T.; Zwaag, J.; van den Wildenberg, J.; Sweep, F.C.; van der Hoeven, J.G.; Pickkers, P. Voluntary activation of the sympathetic nervous system and attenuation of the innate immune response in humans. Proc. Natl. Acad. Sci. USA 2014, 111, 7379–7384. [Google Scholar] [CrossRef] [PubMed]
- Bravo, E.L. Metabolic factors and the sympathetic nervous system. Am. J. Hypertens. 1989, 2, 339S–344S. [Google Scholar] [CrossRef] [PubMed]
- Stienstra, R.; Netea-Maier, R.T.; Riksen, N.P.; Joosten, L.A.B.; Netea, M.G. Specific and Complex Reprogramming of Cellular Metabolism in Myeloid Cells during Innate Immune Responses. Cell Metab. 2017, 26, 142–156. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, L.A.; Kishton, R.J.; Rathmell, J. A guide to immunometabolism for immunologists. Nat. Rev. Immunol. 2016, 16, 553–565. [Google Scholar] [CrossRef]
- Schwertner, H.A.; Kong, S.B. Determination of modafinil in plasma and urine by reversed phase high-performance liquid-chromatography. J. Pharm. Biomed. Anal. 2005, 37, 475–479. [Google Scholar] [CrossRef][Green Version]
- Mirsaeidi, M.; Banoei, M.M.; Nienow, C.K.; Abassi, T.; Hakim, A.; Schraufnagel, D.; Winston, B.W.; Sweiss, N.; Baughman, R.; Garcia, J.G.; et al. Plasma metabolomic profile in fibrosing pulmonary sarcoidosis. Sarcoidosis Vasc. Diffus. Lung Dis. Off. J. WASOG World Assoc. Sarcoidosis Granul. Disord. 2016, 33, 29–38. [Google Scholar]
- Van de Beek, M.C.; Dijkstra, I.M.; van Lenthe, H.; Ofman, R.; Goldhaber-Pasillas, D.; Schauer, N.; Schackmann, M.; Engelen-Lee, J.Y.; Vaz, F.M.; Kulik, W.; et al. C26:0-Carnitine Is a New Biomarker for X-Linked Adrenoleukodystrophy in Mice and Man. PLoS ONE 2016, 11, e0154597. [Google Scholar] [CrossRef]
- Van Eijk, L.T.; Pickkers, P.; Smits, P.; Bouw, M.P.; van der Hoeven, J.G. Severe vagal response after endotoxin administration in humans. Intensive Care Med. 2004, 30, 2279–2281. [Google Scholar] [CrossRef]
- Xia, J.; Wishart, D.S. Web-based inference of biological patterns, functions and pathways from metabolomic data using MetaboAnalyst. Nat. Protoc. 2011, 6, 743–760. [Google Scholar] [CrossRef]
- Nolt, B.; Tu, F.; Wang, X.; Ha, T.; Winter, R.; Williams, D.L.; Li, C. Lactate and Immunosuppression in Sepsis. Shock 2018, 49, 120–125. [Google Scholar] [CrossRef]
- Fink, M.P. Ethyl pyruvate: A novel anti-inflammatory agent. J. Intern. Med. 2007, 261, 349–362. [Google Scholar] [CrossRef] [PubMed]
- DeForge, L.E.; Remick, D.G. Kinetics of TNF, IL-6, and IL-8 gene expression in LPS-stimulated human whole blood. Biochem. Biophys. Res. Commun. 1991, 174, 18–24. [Google Scholar] [CrossRef]
- Cohen, J.D.; Strock, D.J.; Teik, J.E.; Katz, T.B.; Marcel, P.D. Deoxycytidine in human plasma: Potential for protecting leukemic cells during chemotherapy. Cancer Lett. 1997, 116, 167–175. [Google Scholar] [CrossRef]
- Timmermans, K.; Kox, M.; Scheffer, G.J.; Pickkers, P. Plasma Nuclear and Mitochondrial DNA Levels, and Markers of Inflammation, Shock, and Organ Damage in Patients with Septic Shock. Shock 2016, 45, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Kamisoglu, K.; Sleight, K.E.; Calvano, S.E.; Coyle, S.M.; Corbett, S.A.; Androulakis, I.P. Temporal metabolic profiling of plasma during endotoxemia in humans. Shock 2013, 40, 519–526. [Google Scholar] [CrossRef]
- Quehenberger, O.; Dennis, E.A. The human plasma lipidome. N. Engl. J. Med. 2011, 365, 1812–1823. [Google Scholar] [CrossRef]
- Dinkla, S.; van Eijk, L.T.; Fuchs, B.; Schiller, J.; Joosten, I.; Brock, R.; Pickkers, P.; Bosman, G.J. Inflammation-associated changes in lipid composition and the organization of the erythrocyte membrane. BBA Clin. 2016, 5, 186–192. [Google Scholar] [CrossRef]
- Zheng, J.I.E. Energy metabolism of cancer: Glycolysis versus oxidative phosphorylation (Review). Oncol. Lett. 2012, 4, 1151–1157. [Google Scholar] [CrossRef]
- Cheng, S.C.; Joosten, L.A.; Netea, M.G. The interplay between central metabolism and innate immune responses. Cytokine Growth Factor Rev. 2014, 25, 707–713. [Google Scholar] [CrossRef]
- Tannahill, G.M.; Curtis, A.M.; Adamik, J.; Palsson-McDermott, E.M.; McGettrick, A.F.; Goel, G.; Frezza, C.; Bernard, N.J.; Kelly, B.; Foley, N.H.; et al. Succinate is an inflammatory signal that induces IL-1beta through HIF-1alpha. Nature 2013, 496, 238–242. [Google Scholar] [CrossRef]
- Longo, N.; Frigeni, M.; Pasquali, M. Carnitine transport and fatty acid oxidation. Biochim. Biophys. Acta 2016, 1863, 2422–2435. [Google Scholar] [CrossRef] [PubMed]
- Sacca, L.; Vigorito, C.; Cicala, M.; Corso, G.; Sherwin, R.S. Role of gluconeogenesis in epinephrine-stimulated hepatic glucose production in humans. Am. J. Physiol. 1983, 245, E294–E302. [Google Scholar] [CrossRef] [PubMed]
- Hoffer, L.J. Cori cycle contribution to plasma glucose appearance in man. JPEN J. Parenter. Enter. Nutr. 1990, 14, 646–648. [Google Scholar] [CrossRef] [PubMed]
- Ratter, J.M.; Rooijackers, H.M.M.; Hooiveld, G.J.; Hijmans, A.G.M.; de Galan, B.E.; Tack, C.J.; Stienstra, R. In vitro and in vivo Effects of Lactate on Metabolism and Cytokine Production of Human Primary PBMCs and Monocytes. Front. Immunol. 2018, 9, 2564. [Google Scholar] [CrossRef]
- Yu, D.H.; Noh, D.H.; Song, R.H.; Park, J. Ethyl pyruvate downregulates tumor necrosis factor alpha and interleukin (IL)-6 and upregulates IL-10 in lipopolysaccharide-stimulated canine peripheral blood mononuclear cells. J. Vet. Med. Sci. 2010, 72, 1379–1381. [Google Scholar] [CrossRef]
- Venkataraman, R.; Kellum, J.A.; Song, M.; Fink, M.P. Resuscitation with Ringer’s ethyl pyruvate solution prolongs survival and modulates plasma cytokine and nitrite/nitrate concentrations in a rat model of lipopolysaccharide-induced shock. Shock 2002, 18, 507–512. [Google Scholar] [CrossRef]
- Van Eijk, L.T.; Dorresteijn, M.J.; Smits, P.; van der Hoeven, J.G.; Netea, M.G.; Pickkers, P. Gender differences in the innate immune response and vascular reactivity following the administration of endotoxin to human volunteers. Crit. Care Med. 2007, 35, 1464–1469. [Google Scholar] [CrossRef]
- O’Brien, S.M.; Fitzgerald, P.; Scully, P.; Landers, A.; Scott, L.V.; Dinan, T.G. Impact of gender and menstrual cycle phase on plasma cytokine concentrations. Neuroimmunomodulation 2007, 14, 84–90. [Google Scholar] [CrossRef]
- Gornicsar, K.; Mozes, T.; Grosz, A.; Biro, E.; Ladi, S.; Clayton, P. TNFalpha Variation During the Menstrual Cycle and Thereafter: A New Explanation for Gender-Based Disparities in ICU Admission Rates, Trauma Outcomes, and General Mortality. Shock 2017, 47, 416–421. [Google Scholar] [CrossRef]
- Segre, E.; Fullerton, J.N. Stimulated Whole Blood Cytokine Release as a Biomarker of Immunosuppression in the Critically Ill: The Need for a Standardized Methodology. Shock 2016, 45, 490–494. [Google Scholar] [CrossRef]
- Dorresteijn, M.J.; van Eijk, L.T.; Netea, M.G.; Smits, P.; van der Hoeven, J.G.; Pickkers, P. Iso-osmolar prehydration shifts the cytokine response towards a more anti-inflammatory balance in human endotoxemia. J. Endotoxin Res. 2005, 11, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Van Lier, D.; Geven, C.; Leijte, G.P.; Pickkers, P. Experimental human endotoxemia as a model of systemic inflammation. Biochimie 2018, 159, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.A.; Want, E.J.; O’Maille, G.; Abagyan, R.; Siuzdak, G. XCMS: Processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Anal. Chem. 2006, 78, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Libiseller, G.; Dvorzak, M.; Kleb, U.; Gander, E.; Eisenberg, T.; Madeo, F.; Neumann, S.; Trausinger, G.; Sinner, F.; Pieber, T.; et al. IPO: A tool for automated optimization of XCMS parameters. BMC Bioinform. 2015, 16, 118. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org (accessed on 16 June 2016).
- Creek, D.J.; Jankevics, A.; Burgess, K.E.; Breitling, R.; Barrett, M.P. IDEOM: An Excel interface for analysis of LC-MS-based metabolomics data. Bioinformatics 2012, 28, 1048–1049. [Google Scholar] [CrossRef]
- Brand, B.; Hadlich, F.; Brandt, B.; Schauer, N.; Graunke, K.L.; Langbein, J.; Repsilber, D.; Ponsuksili, S.; Schwerin, M. Temperament type specific metabolite profiles of the prefrontal cortex and serum in cattle. PLoS ONE 2015, 10, e0125044. [Google Scholar] [CrossRef]
- Stoessel, D.; Schulte, C.; Teixeira Dos Santos, M.C.; Scheller, D.; Rebollo-Mesa, I.; Deuschle, C.; Walther, D.; Schauer, N.; Berg, D.; Nogueira da Costa, A.; et al. Promising Metabolite Profiles in the Plasma and CSF of Early Clinical Parkinson’s Disease. Front. Aging Neurosci. 2018, 10, 51. [Google Scholar] [CrossRef]
- Bijlsma, S.; Bobeldijk, I.; Verheij, E.R.; Ramaker, R.; Kochhar, S.; Macdonald, I.A.; van Ommen, B.; Smilde, A.K. Large-scale human metabolomics studies: A strategy for data (pre-) processing and validation. Anal. Chem. 2006, 78, 567–574. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Kamisoglu, K.; Calvano, S.E.; Coyle, S.M.; Corbett, S.A.; Androulakis, I.P. Integrated transcriptional and metabolic profiling in human endotoxemia. Shock 2014, 42, 499–508. [Google Scholar] [CrossRef]
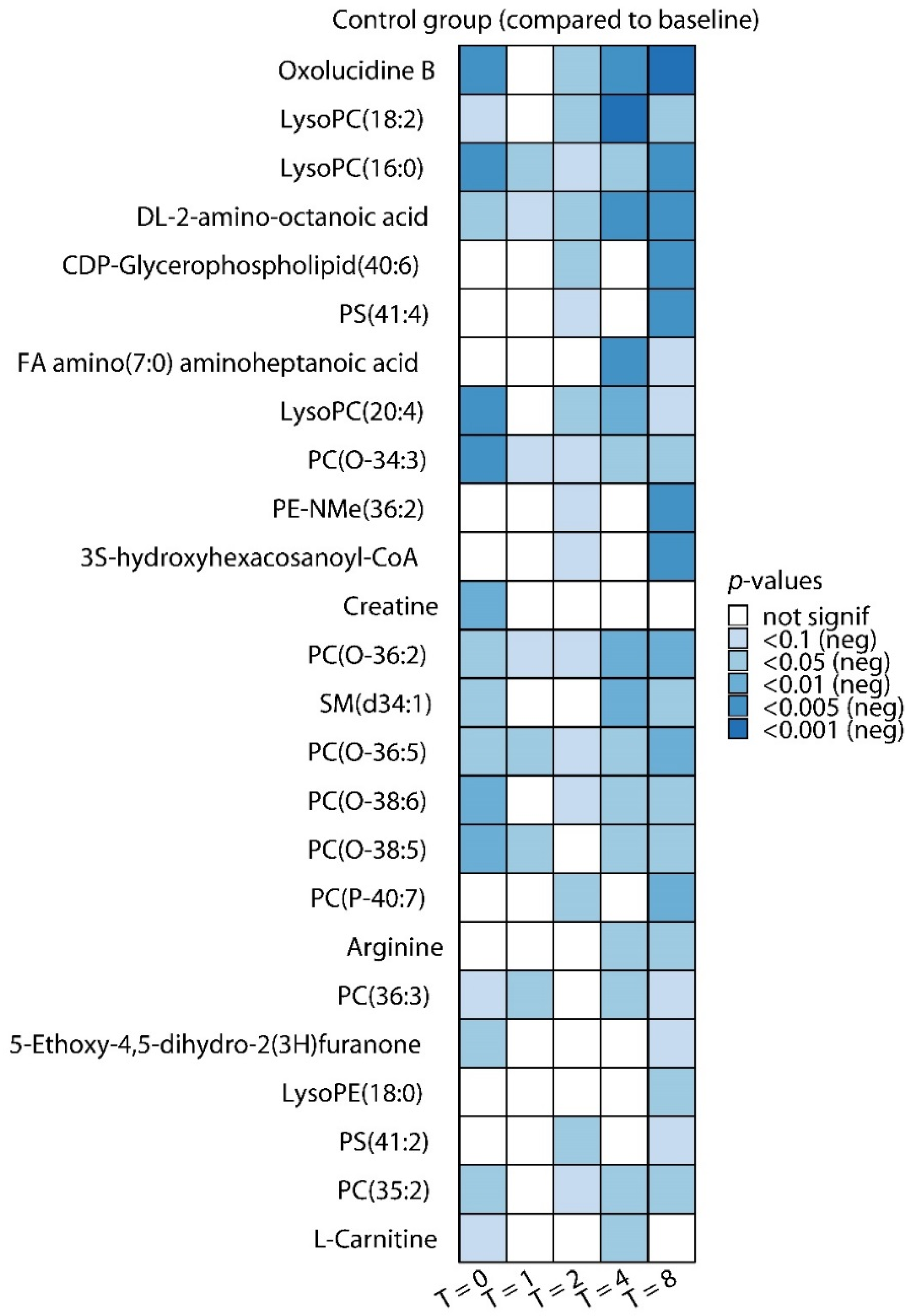

| Metabolite | Class | Fold Change Versus Baseline 1 | FDR Adjusted p-Value 2 |
|---|---|---|---|
| Increased | |||
| 5,8-Dihydro-6-(4-methyl-3-pentenyl)-1,2,3,4-tetrathiocin | Prenol lipids | +1.25 | 0.044 |
| 6-Phosphonoglucono-D-lactone | Monosaccharides | +3.31 | 0.051 |
| Deoxycytidine | Nucleosides, nucleotides, and derivatives | +24.77 | 0.061 |
| Artonin K | Flavonoids | +1.43 | 0.065 |
| Hexane-6-keto-1,3,4,6-tetracarboxylate | Unknown | +3.91 | 0.065 |
| Cis-(homo)2aconitate | Metabolism of cofactors and vitamins | +1.25 | 0.071 |
| Deoxyuridine triphosphate | Nucleosides, nucleotides, and derivatives | +4.46 | 0.076 |
| Imidazoleacetic acid riboside | Nucleosides, nucleotides, and derivatives | +1.31 | 0.076 |
| Lactic acid | Hydroxy acids and derivatives | +2.03 | 0.091 |
| Gnidicin | Unknown | +6.87 | 0.096 |
| Decreased | |||
| LysoPC(18:2) | Glycerophospholipids | −2.22 | 0.0008 |
| dl-2-amino-octanoic acid | Amino acids and derivatives | −1.51 | 0.002 |
| Oxolucidine B | Unknown | −2.27 | 0.005 |
| FA amino(7:0) aminoheptanoic acid | Fatty acyls | −1.59 | 0.005 |
| LysoPC(20:4) | Glycerophospholipids | −2.85 | 0.007 |
| PC(O−36:2) | Glycerophospholipids | −1.26 | 0.007 |
| SM(d34:1) | Sphingolipids | −1.21 | 0.009 |
| Arginine | Amino acids and derivatives | −1.40 | 0.013 |
| PC(36:3) | Glycerophospholipids | −1.29 | 0.015 |
| PC(O-36:5) | Glycerophospholipids | −1.24 | 0.019 |
| PC(O-34:3) | Glycerophospholipids | −1.28 | 0.021 |
| l-Carnitine | Alkylamines | −1.32 | 0.022 |
| Succinic acid semialdehyde | Fatty acids and conjugates | −1.47 | 0.022 |
| PC(O-38:5) | Glycerophospholipids | −1.20 | 0.023 |
| PS(21:0) | Glycerophospholipids | −3.43 | 0.024 |
| l-Acetylcarnitine | Fatty acid esters | −1.67 | 0.026 |
| PC(O-38:6) | Glycerophospholipids | −1.21 | 0.027 |
| Glutamine | Amino acids and derivatives | −1.45 | 0.028 |
| PC(36:5) | Glycerophospholipids | −1.65 | 0.035 |
| PC(36:4) | Glycerophospholipids | −1.20 | 0.035 |
| LysoPC(16:0) | Glycerophospholipids | −2.20 | 0.038 |
| PC(P-40:6) | Glycerophospholipids | −1.19 | 0.045 |
| PC(35:2) | Organic phosphoric acids and derivatives | −1.25 | 0.045 |
| Narciclasine | Unknown | −1.86 | 0.045 |
| PC(40:6) | Glycerophospholipids | −1.69 | 0.045 |
| Lenticin | Unknown | −1.35 | 0.065 |
| PC(36:2) | Glycerophospholipids | −1.35 | 0.066 |
| PC(34:3) | Glycerophospholipids | −1.23 | 0.071 |
| PC(38:4) | Glycerophospholipids | −1.20 | 0.071 |
| PE(39:1) | Glycerophospholipids | −1.33 | 0.071 |
| PC(O-34:2) | Glycerophospholipids | −1.30 | 0.071 |
| SM(d34:2) | Sphingolipids | −1.20 | 0.078 |
| PC(38:5) | Glycerophospholipids | −1.17 | 0.087 |
| TG(41:0) | Glycerolipids | −1.20 | 0.091 |
| Metabolite | Class | Fold-Change (Trained/Control) | FDR-Adjusted p-Value 1 |
|---|---|---|---|
| Increased | |||
| N3-(4-methoxyfumaroyl)-l-2,3-diaminopropanoate | Unknown | +3.15 | 0.0007 |
| Lactic acid | Carbohydrate metabolism | +3.00 | 0.0007 |
| 6-Phosphonoglucono-d-lactone | Lactones | +3.19 | 0.005 |
| Deoxyuridine triphosphate | Nucleosides, nucleotides, and derivatives | +4.91 | 0.006 |
| Deoxycytidine | Nucleosides, nucleotides, and derivatives | +9.15 | 0.007 |
| 1,6-Naphthalenedisulfonic acid | Unknown | +2.19 | 0.007 |
| Pyruvic acid | Alcohols and polyols | +2.07 | 0.035 |
| Hexane-6-keto-1,3,4,6-tetracarboxylate | Unknown | +3.52 | 0.083 |
| Decreased | |||
| l-Histidine | Amino acids and derivatives | −2.72 | 0.026 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zwaag, J.; ter Horst, R.; Blaženović, I.; Stoessel, D.; Ratter, J.; Worseck, J.M.; Schauer, N.; Stienstra, R.; Netea, M.G.; Jahn, D.; et al. Involvement of Lactate and Pyruvate in the Anti-Inflammatory Effects Exerted by Voluntary Activation of the Sympathetic Nervous System. Metabolites 2020, 10, 148. https://doi.org/10.3390/metabo10040148
Zwaag J, ter Horst R, Blaženović I, Stoessel D, Ratter J, Worseck JM, Schauer N, Stienstra R, Netea MG, Jahn D, et al. Involvement of Lactate and Pyruvate in the Anti-Inflammatory Effects Exerted by Voluntary Activation of the Sympathetic Nervous System. Metabolites. 2020; 10(4):148. https://doi.org/10.3390/metabo10040148
Chicago/Turabian StyleZwaag, Jelle, Rob ter Horst, Ivana Blaženović, Daniel Stoessel, Jacqueline Ratter, Josephine M. Worseck, Nicolas Schauer, Rinke Stienstra, Mihai G. Netea, Dieter Jahn, and et al. 2020. "Involvement of Lactate and Pyruvate in the Anti-Inflammatory Effects Exerted by Voluntary Activation of the Sympathetic Nervous System" Metabolites 10, no. 4: 148. https://doi.org/10.3390/metabo10040148
APA StyleZwaag, J., ter Horst, R., Blaženović, I., Stoessel, D., Ratter, J., Worseck, J. M., Schauer, N., Stienstra, R., Netea, M. G., Jahn, D., Pickkers, P., & Kox, M. (2020). Involvement of Lactate and Pyruvate in the Anti-Inflammatory Effects Exerted by Voluntary Activation of the Sympathetic Nervous System. Metabolites, 10(4), 148. https://doi.org/10.3390/metabo10040148






