Protective Role of Vitamin D in Renal Tubulopathies
Abstract
1. Introduction
1.1. Vitamin D: A Polymorphous Agent with Heterogeneous Functions
1.2. Vitamin D Protective Role in Tubular Damage in CKD
2. Megalin, Cubilin and Vitamin D: A Synergy into Proximal Tubule Cells for Body Homeostasis
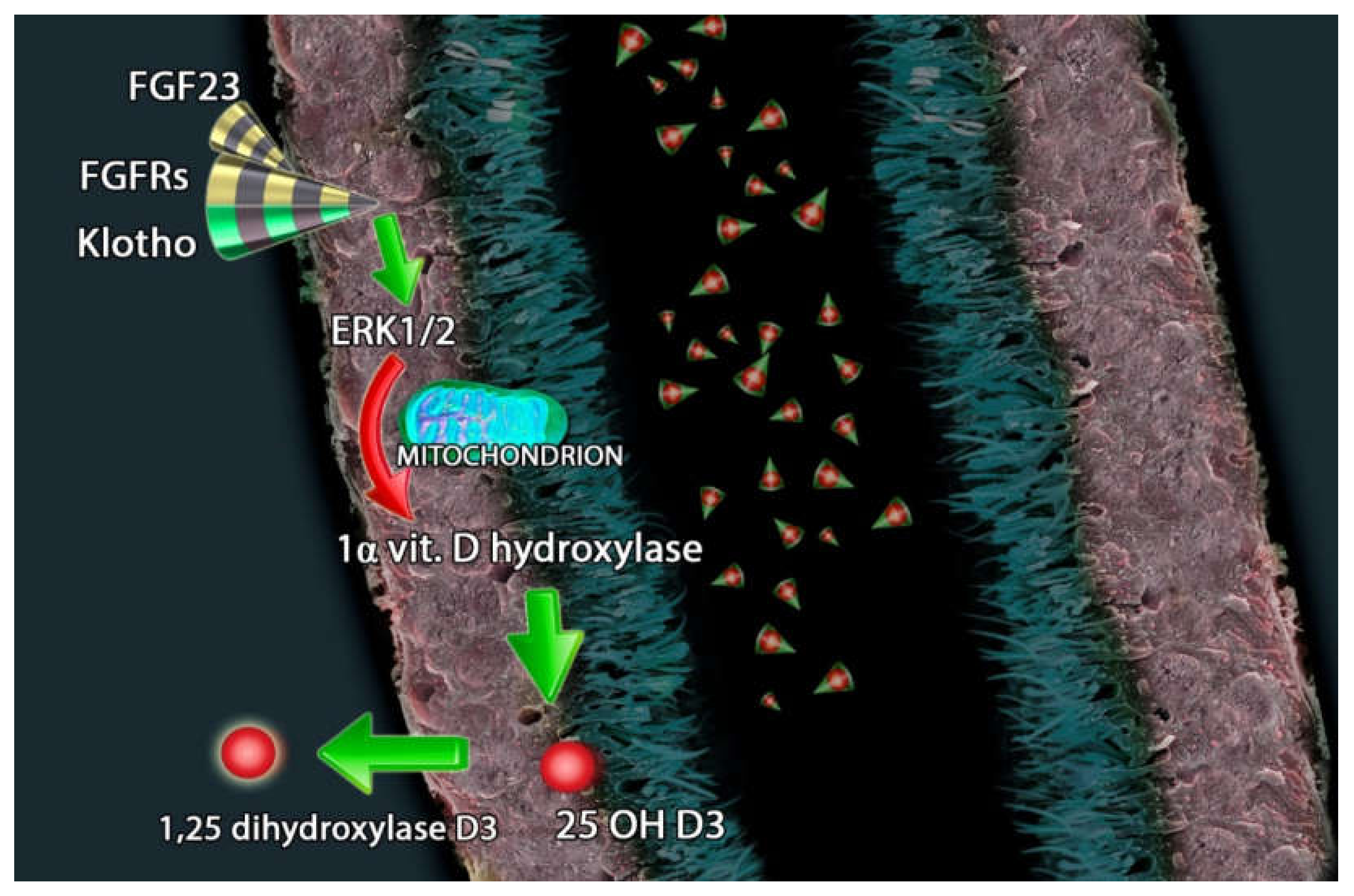
3. FGF23-Klotho and Vitamin D Axis in Endocrine-Paracrine Renal Loop
4. Vitamin D in Primitive Renal Tubular Disorders
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| [1,25 (OH)2D3] | 1,25-dihydroxyvitamin D3 |
| [25(OH)D] | 25-hydroxyvitamin D |
| ADAM17 | a disintegrin and metalloproteinase |
| AKT/UCP2 | protein kinase B/mitochondrial uncoupling protein 2 |
| Calcidiol | 25-hydroxyvitamin D |
| Calcitriol | 1,25-dihydroxy vitamin D |
| CKD | Chronic Kidney Disease |
| CI | Confidence Interval |
| DB/FOAR | Donnai-Barrow/Facio-Oculo-Acustico-Renal |
| DBP | Vitamin D Binding Protein |
| eGFR | estimated glomerular filtration rate |
| EMT/EndMT | epithelial/endothelial-mesenchymal |
| ERK 1/2 | extracellular signal-regulated kinase ½ |
| FGF23 | Fibroblast Growth Factor 23 |
| FGFRs | FGF receptors |
| HK2 | renal tubular epithelial cell line |
| HRPTEpiC | human renal PTECs |
| IL-6 | cytokine interleukin-6 |
| LPS | Lipopolysaccharide |
| Lrp2 | low-density lipoprotein-related protein 2 |
| MCP-1 | monocyte chemoattractant protein |
| MIP-1β | monocyte inflammatory protein |
| NaPi-II | sodium-phosphate cotransporter |
| NF-κB | nuclear factor-κB |
| PTH | parathyroid hormone |
| PPARs | peroxisome proliferator-activated receptors |
| PDGF | platelet-derived growth factor |
| PICs | pro-inflammatory cytokines |
| PTECs | Proximal Tubular Epithelial Cells |
| RAAS | renin–angiotensin–aldosterone system |
| RANTES | regulated on activation, normal T cell expressed and secreted |
| ROCK | Rho protein kinase pathway |
| ROS | reactive oxygen species |
| SAP | serum amyloid P |
| TGFβ | transforming growth factor β |
| VDR | Vitamin D receptor |
| VEGF | vascular endothelial growth factor |
| XLH | X-linked hypophosphatemia |
References
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium; Ross, A.C.; Taylor, C.L.; Yaktine, A.L. (Eds.) Dietary Reference Intakes for Calcium and Vitamin D; National Academies Press (US): Washington, DC, USA, 2011; p. 3, Overview of Vitamin D. Available online: https://www.ncbi.nlm.nih.gov/books/NBK56061/ (accessed on 20 December 2019).
- National Osteoporosis Society. Vitamin D and Bone Health: A Practical Clinical Guideline for Patient Management; National Osteoporosis Society: Camerton, UK, 2013. [Google Scholar]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int. Suppl. 2009, 113, S1–S130. [Google Scholar]
- Coudroy, G.; Gburek, J.; Kozyraki, R.; Madsen, M.; Trugnan, G.; Moestrup, S.K.; Verroust, P.J.; Maurice, M. Contribution of cubilin and amnionless to processing and membrane targeting of cubilin-amnionless complex. J. Am. Soc. Nephrol. 2005, 16, 2330–2337. [Google Scholar] [CrossRef]
- Erben, R.G. α-Klotho’s effects on mineral homeostasis are fibroblast growth factor-23 dependent. Curr. Opin. Nephrol. Hypertens. 2018, 27, 229–235. [Google Scholar] [CrossRef]
- Hoenderop, J.G.; Nilius, B.; Bindels, R.J. Calcium absorption across epithelia. Physiol. Rev. 2005, 85, 373–422. [Google Scholar] [CrossRef]
- Sabbagh, Y.; Giral, H.; Caldas, Y.; Levi, M.; Schiavi, S.C. Intestinal phosphate transport. Adv. Chronic. Kidney Dis. 2011, 18, 85–90. [Google Scholar] [CrossRef]
- Lambers, T.T.; Bindels, R.J.; Hoenderop, J.G. Coordinated control of renal Ca2+ handling. Kidney Int. 2006, 69, 650–654. [Google Scholar] [CrossRef]
- Khundmiri, S.J.; Murray, R.D.; Lederer, E.P.T.H. Vitamin, D. Compr. Physiol. 2016, 6, 561–601. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C. Renoprotective effects of vitamin D analogs. Kidney Int. 2010, 78, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Gembillo, G.; Cernaro, V.; Salvo, A.; Siligato, R.; Laudani, A.; Buemi, M.; Santoro, D. Role of Vitamin D Status in Diabetic Patients with Renal Disease. Medicina (Kaunas) 2019, 6, E273. [Google Scholar] [CrossRef] [PubMed]
- Santoro, D.; Sebekova, K.; Teta, D.; De Nicola, L. Extraskeletal Functions of Vitamin, D. Biomed. Res. Int. 2015, 2015, 294719. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Li, Y.; Liu, Y. Paricalcitol attenuates renal interstitial fibrosis in obstructive nephropathy. J. Am. Soc. Nephrol. 2006, 17, 3382–3393. [Google Scholar] [CrossRef] [PubMed]
- Tsuprykov, O.; Chen, X.; Hocher, C.F.; Skoblo, R.; Lianghong Yin Hocher, B. Why should we measure free 25(OH) vitamin D? J. Steroid Biochem. Mol. Biol. 2018, 180, 87–104. [Google Scholar] [CrossRef] [PubMed]
- Jean, G.; Souberbielle, J.C.; Chazot, C. Vitamin D in Chronic Kidney Disease and Dialysis Patients. Nutrients 2017, 9, E328. [Google Scholar] [CrossRef]
- Kochupillai, N. The physiology of vitamin D: Current concepts. Indian J. Med. Res. 2008, 127, 256–262. [Google Scholar]
- Lucisano, S.; Buemi, M.; Passantino, A.; Aloisi, C.; Cernaro, V.; Santoro, D. New insights on the role of vitamin D in the progression of renal damage. Kidney Blood Press Res. 2013, 37, 667–678. [Google Scholar] [CrossRef]
- Yang, S.; Li, A.; Wang, J.; Liu, J.; Han, Y.; Zhang, W.; Li, Y.C.; Zhang, H. Vitamin D Receptor: A Novel Therapeutic Target for Kidney Diseases. Curr. Med. Chem. 2018, 25, 3256–3271. [Google Scholar] [CrossRef]
- Hu, H.; Xu, S.; Hu, S.; Gao, Y.; Shui, H. Effect of 1,25(OH)(2)D(3) on transdifferentiation of rat renal tubular epithelial cells induced by high glucose. Biomed. Rep. 2016, 5, 699–704. [Google Scholar] [CrossRef]
- Ahmed, A.; Sims, R.V. Proximal renal tubular acidosis associated with osteomalacia. South. Med. J. 2001, 94, 536–539. [Google Scholar] [CrossRef]
- Garcia Nieto, V.; Sánchez Almeida, E.; García García, M. Renal tubular dysfunction of Vitamin D deficiency rickets. Nephron 1996, 72, 364. [Google Scholar] [CrossRef]
- Muldowney, F.P.; Freaney, R.; McGeeney, D. Renal tubular acidosis and amino-aciduria in osteomalacia of dietary or intestinal origin. Q J. Med. 1968, 37, 517–539. [Google Scholar] [PubMed]
- Skelton, L.A.; Boron, W.F.; Zhou, Y. Acid-base transport by the renal proximal tubule. J. Nephrol. 2010, 23 (Suppl. 16), S4–S18. [Google Scholar] [PubMed]
- Santoro, D.; Caccamo, D.; Gagliostro, G.; Ientile, R.; Benvenga, S.; Bellinghieri, G.; Savica, V. Vitamin D metabolism and activity as well as genetic variants of the vitamin D receptor (VDR) in chronic kidney disease patients. J. Nephrol. 2013, 26, 636–644. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, H.; Kurokawa, K. Localization of receptors for 1,25(OH)2D3 along the rat nephron. J. Biol. Chem. 1982, 257, 13428–13432. [Google Scholar]
- Akiba, T.; Endou, H.; Koseki, C.; Sakai, F.; Horiuchi, N.; Suda, T. Localization of 1-a-hydroxylase activity in the mammalian kidney. Biochem. Biophys. Res. Commun. 1980, 94, 313–318. [Google Scholar] [CrossRef]
- Takezawa, K.; Moorthy, B.; Mandel, M.L.; Garancis, J.C.; Ghazarian, J.G. Antigenic and catalytic disparity in the distribution of cytochrome P-450 dependent 25-hydroxyvitamin D3-1-a-hydroxylase and 24-hydroxylase. Histochemistry 1990, 95, 37–42. [Google Scholar] [CrossRef]
- Bindels, R.J.M.; Hartog, A.; Timmermans, J.; van Os, C.H. Active Ca21 transport in primary cultures of rabbit kidney CCD: Stimulation by 1,25(OH)2D3 and PTH. Am. J. Physiol. Renal Fluid Electrolyte Physiol. 1991, 261, F799–F807. [Google Scholar] [CrossRef]
- Bouhtiauy, D.L.; Lajeunesse, D.; Brunette, M.G. Effect of vitamin D depletion on calcium transport by the luminal and basolateral membranes of the proximal and distal nephrons. Endocrinology 1993, 132, 115–120. [Google Scholar] [CrossRef]
- Siaw, E.K.; Walters, M.R. 1,25-Dihydroxyvitamin D-stimulated calmodulin binding proteins: A sustained effect on distal tubules. Am. J. Physiol. Renal Physiol. 2002, 282, F77–F84. [Google Scholar] [CrossRef]
- Cernaro, V.; Lacquaniti, A.; Donato, V.; Fazio, M.R.; Buemi, A.; Buemi, M. Fibrosis, regeneration and cancer: What is the link? Nephrol. Dial. Transplant. 2012, 27, 21–27. [Google Scholar] [CrossRef][Green Version]
- Carthy, J.M. TGFβ signaling and the control of myofibroblast differentiation: Implications for chronic inflammatory disorders. J. Cell Physiol. 2018, 233, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Li, Y.; Liu, Y. Therapeutic role and potential mechanisms of active Vitamin D in renal interstitial fibrosis. J. Steroid Biochem. Mol. Biol. 2007, 103, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A. Cellular and molecular mechanisms of fibrosis. J. Pathol. 2008, 214, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Duffield, J.S. Cellular and molecular mechanisms in kidney fibrosis. J. Clin. Invest. 2014, 124, 2299–2306. [Google Scholar] [CrossRef]
- Arfian, N.; Muflikhah, K.; Soeyono, S.K.; Sari, D.C.; Tranggono, U.; Anggorowati, N.; Romi, M.M. Vitamin D Attenuates Kidney Fibrosis via Reducing Fibroblast Expansion, Inflammation, and Epithelial Cell Apoptosis. Kobe J. Med. Sci. 2016, 62, E38–E44. [Google Scholar]
- Goncalves, J.G.; de Braganca, A.C.; Canale, D.; Shimizu MH, M.; Sanches, T.R.; Moysés RM, A.; Volpini, R.A. Vitamin D deficiency aggravates chronic kidney disease progression after ischemic acute kidney injury. PLoS ONE 2014, 9, e107228. [Google Scholar] [CrossRef]
- Inoue, K.; Matsui, I.; Hamano, T.; Fujii, N.; Shimomura, A.; Nakano, C.; Kusunoki, Y.; Takabatake, Y.; Hirata, M.; Nishiyama, A.; et al. Maxacalcitol ameliorates tubulointerstitial fibrosis in obstructed kidneys by recruiting PPM1A/VDR complex to pSmad3. Lab. Invest. 2012, 92, 1686–1697. [Google Scholar] [CrossRef]
- Lee, J.W.; Kim, S.C.; Ko, Y.S.; Lee, H.Y.; Cho, E.; Kim, M.G.; Jo, S.K.; Cho, W.Y.; Kim, H.K. Renoprotective effect of paricalcitol via a modulation of the TLR4-NF-κB pathway in ischemia/reperfusion-induced acute kidney injury. Biochem. Biophys. Res. Commun. 2014, 444, 121–127. [Google Scholar] [CrossRef]
- Nakhoul, N.; Batuman, V. Role of proximal tubules in the pathogenesis of kidney disease. Contrib. Nephrol. 2011, 169, 37–50. [Google Scholar] [CrossRef]
- Chesney, R.W. Interactions of vitamin D and the proximal tubule. Pediatr. Nephrol. 2016, 31, 7–14. [Google Scholar] [CrossRef]
- Zhu, X.; Wu, S.; Guo, H. Active Vitamin D and Vitamin D Receptor Help Prevent High Glucose Induced Oxidative Stress of Renal Tubular Cells via AKT/UCP2 Signaling Pathway. Biomed. Res. Int. 2019, 2019, 9013904. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Zheng, Y.; Huang, J.; Peng, W.; Chen, X.; Kang, X.; Zeng, Q. UCP2 ameliorates mitochondrial dysfunction, inflammation, and oxidative stress in lipopolysaccharide-induced acute kidney injury. Int. Immunopharmacol. 2019, 71, 336–349. [Google Scholar] [CrossRef] [PubMed]
- Qin, N.; Cai, T.; Ke, Q.; Yuan, Q.; Luo, J.; Mao, X.; Jiang, L.; Cao, H.; Wen, P.; Zen, K.; et al. UCP2-dependent improvement of mitochondrial dynamics protects against acute kidney injury. J. Pathol. 2019, 247, 392–405. [Google Scholar] [CrossRef] [PubMed]
- Chung, B.H.; Kim, B.M.; Doh, K.C.; Cho, M.L.; Kim, K.W.; Yang, C.W. Protective effect of 1α,25-dihydroxyvitamin D3 on effector CD4+ T cell induced injury in human renal PTECs. PLoS ONE 2017, 12, e0172536. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Xia, Y.; Gan, H. Calcitriol Ameliorates AngiotensinII-Induced Renal Injury Partly via Upregulating A20. Inflammation 2017, 40, 1884–1893. [Google Scholar] [CrossRef] [PubMed]
- Jaattela, M.; Mouritzen, H.; Elling, F.; Bastholm, L. A20 zinc finger protein inhibits TNF and IL-1 signaling. J. Immunol. 1996, 156, 1166–1173. [Google Scholar]
- Zhang, W.; Yi, B.; Zhang, K.; Li, A.; Yang, S.; Huang, J.; Liu, J.; Zhang, H. 1,25-(OH)(2)D(3) and its analogue BXL-628 inhibit high glucose-induced activation of RhoA/ROCK pathway in HK-2 cells. Exp. Ther. Med. 2017, 13, 1969–1976. [Google Scholar] [CrossRef][Green Version]
- Zhang, K.; Zhang, H.; Xiang, H.; Liu, J.; Liu, Y.; Zhang, X.; Wang, J.; Tang, Y. TGF-β1 induces the dissolution of tight junctions in human renal proximal tubular cells: Role of the RhoA/ROCK signaling pathway. Int J. Mol. Med. 2013, 32, 464–468. [Google Scholar] [CrossRef]
- Lu, Q.; Chen, Y.B.; Yang, H.; Wang, W.W.; Li, C.C.; Wang, L.; Wang, J.; Du, L.; Yin, X.X. Inactivation of TSC1 promotes epithelial-mesenchymal transition of renal tubular epithelial cells in mouse diabetic nephropathy. Acta Pharm. Sin. 2019, 40, 1555–1567. [Google Scholar] [CrossRef]
- Krüger, S.; Kreft, B. 1,25-dihydroxyvitamin D3 differentially regulates IL-1alpha-stimulated IL-8 and MCP-1 mRNA expression and chemokine secretion by human primary PTECs. Exp. Nephrol. 2001, 9, 223–228. [Google Scholar]
- Jacobson, S.H.; Hylander, B.; Wretlind, B.; Brauner, A. Interleukin-6 and interleukin-8 in serum and urine in patients with acute pyelonephritis in relation to bacterial-virulence-associated traits and renal function. Nephron 1994, 67, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Yung, S.; Ng, C.Y.; Au, K.Y.; Cheung, K.F.; Zhang, Q.; Zhang, C.; Yap, D.Y.; Chau, M.K.; Chan, T.M. Binding of anti-dsDNA antibodies to PTECs contributes to renal tubulointerstitial inflammation. Clin. Sci. (Lond.) 2017, 131, 49–67. [Google Scholar] [CrossRef] [PubMed]
- Wada, T.; Yokoyama, H.; Su, S.B.; Mukaida, N.; Iwano, M.; Dohi, K.; Takahashi, Y.; Sasaki, T.; Furuichi, K.; Segawa, C.; et al. Monitoring urinary levels of monocyte chemotactic and activating factor reflects disease activity of lupus nephritis. Kidney Int. 1996, 49, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.S.; Joo, S.Y.; Lee, K.E.; Choi, J.S.; Bae, E.H.; Ma, S.K.; Kim, S.H.; Lee, J.; Kim, S.W. Paricalcitol attenuates 4-hydroxy-2-hexenal-induced inflammation and epithelial-mesenchymal transition in human renal PTECs. PLoS ONE 2013, 8, e63186. [Google Scholar] [CrossRef]
- Du, J.; Jiang, S.; Hu, Z.; Tang, S.; Sun, Y.; He, J.; Li, Z.; Yi, B.; Wang, J.; Zhang, H.; et al. Vitamin D receptor activation protects against lipopolysaccharide-induced acute kidney injury through suppression of tubular cell apoptosis. Am. J. Physiol. Renal Physiol. 2019, 316, F1068–F1077. [Google Scholar] [CrossRef]
- Tan, X.; Wen, X.; Liu, Y. Paricalcitol inhibits renal inflammation by promoting vitamin D receptor-mediated sequestration of NF-kappaB signaling. J. Am. Soc. Nephrol. 2008, 19, 1741–1752. [Google Scholar] [CrossRef]
- Rane, M.J.; Song, Y.; Jin, S.; Barati, M.T.; Wu, R.; Kausar, H.; Li, X. Interplay between Akt and p38 MAPK pathways in the regulation of renal tubular cell apoptosis associated with diabetic nephropathy. Am. J. Physiol.-Renal Physiol. 2010, 298, F49–F61. [Google Scholar] [CrossRef]
- Hong, Y.A.; Yang, K.J.; Jung, S.Y.; Chang, Y.K.; Park, C.W.; Yang, C.W.; Kim, S.Y.; Hwang, H.S. Paricalcitol attenuates lipopolysaccharide-induced inflammation and apoptosis in proximal tubular cells through the prostaglandin E(2) receptor EP4. Kidney Res. Clin. Pract. 2017, 36, 145–158. [Google Scholar] [CrossRef]
- Morgado-Pascual, J.L.; Rayego-Mateos, S.; Valdivielso, J.M.; Ortiz, A.; Egido, J.; Ruiz-Ortega, M. Paricalcitol Inhibits Aldosterone-Induced Proinflammatory Factors by Modulating Epidermal Growth Factor Receptor Pathway in Cultured Tubular Epithelial Cells. Biomed. Res. Int. 2015, 2015, 783538. [Google Scholar] [CrossRef]
- Rayego-Mateos, S.; Morgado-Pascual, J.L.; Sanz, A.B.; Ramos, A.M.; Eguchi, S.; Batlle, D.; Ruiz-Ortega, M. TWEAK transactivation of the epidermal growth factor receptor mediates renal inflammation. J. Pathol. 2013, 231, 480–494. [Google Scholar] [CrossRef]
- Gao, Y.; Zhou, S.; Luu, S.; Glowacki, J. Megalin mediates 25-hydroxyvitamin D(3) actions in human mesenchymal stem cells. Faseb. J. 2019, 33, 7684–7693. [Google Scholar] [CrossRef] [PubMed]
- Christensen, E.I.; Willnow, T.E. Essential role of megalin in renal proximal tubule for vitamin homeostasis. J. Am. Soc. Nephrol. 1999, 10, 2224–2236. [Google Scholar] [PubMed]
- Matarese, V.; Lodish, H.F. Specific uptake of retinol-binding protein by variant F9 cell lines. J. Biol. Chem. 1993, 268, 18859–18865. [Google Scholar] [PubMed]
- Rowling, M.J.; Kemmis, C.M.; Taffany, D.A.; Welsh, J. Megalin-mediated endocytosis of vitamin D binding protein correlates with 25-hydroxycholecalciferol actions in human mammary cells. J. Nutr. 2006, 136, 2754–2759. [Google Scholar] [CrossRef] [PubMed]
- Saito, A.; Iino, N.; Takeda, T.; Gejyo, F. Role of megalin, a proximal tubular endocytic receptor, in calcium and phosphate homeostasis. Ther. Apher. Dial. 2007, 11 (Suppl. 1), S23–S26. [Google Scholar] [CrossRef]
- Chapron, B.D.; Chapron, A.; Phillips, B.; Okoli, M.C.; Shen, D.D.; Kelly, E.J.; Himmelfarb, J.; Thummel, K.E. Reevaluating the role of megalin in renal vitamin D homeostasis using a human cell-derived microphysiological system. ALTEX 2018, 35, 504–515. [Google Scholar] [CrossRef]
- Storm, T.; Tranebjærg, L.; Frykholm, C.; Birn, H.; Verroust, P.J.; Nevéus, T.; Sundelin, B.; Hertz, J.M.; Holmström, G.; Ericson, K.; et al. Renal phenotypic investigations of megalin-deficient patients: Novel insights into tubular proteinuria and albumin filtration. Nephrol. Dial. Transplant. 2013, 28, 585–591. [Google Scholar] [CrossRef]
- Willnow, T.E.; Christ, A. Endocytic receptor LRP2/megalin-of holoprosencephaly and renal Fanconi syndrome. Pflug. Arch. 2017, 469, 907–916. [Google Scholar] [CrossRef]
- Beydoun, M.A.; Ding, E.L.; Beydoun, H.A.; Tanaka, T.; Ferrucci, L.; Zonderman, A.B. Vitamin D receptor and megalin gene polymorphisms and their associations with longitudinal cognitive change in US adults. Am. J. Clin. Nutr. 2012, 95, 163–178. [Google Scholar] [CrossRef]
- De, S.; Kuwahara, S.; Hosojima, M.; Ishikawa, T.; Kaseda, R.; Sarkar, P.; Toba, K. Exocytosis-Mediated Urinary Full-Length Megalin Excretion Is Linked With the Pathogenesis of Diabetic Nephropathy. Diabetes 2017, 66, 1391–1404. [Google Scholar] [CrossRef]
- Toi, N.; Inaba, M.; Ishimura, E.; Tsugawa, N.; Imanishi, Y.; Emoto, M.; Hirayama, Y.; Nakatani, S.; Saito, A.; Yamada, S. Significance of urinary C-megalin excretion in vitamin D metabolism in pre-dialysis CKD patients. Sci. Rep. 2019, 9, 2207. [Google Scholar] [CrossRef]
- De, S.; Kuwahara, S.; Saito, A. The endocytic receptor megalin and its associated proteins in proximal tubule epithelial cells. Membranes (Basel) 2014, 4, 333–355. [Google Scholar] [CrossRef]
- Christensen, E.I.; Nielsen, R.; Birn, H. From bowel to kidneys: The role of cubilin in physiology and disease. Nephrol. Dial. Transpl. 2013, 28, 274–281. [Google Scholar] [CrossRef]
- Christensen, E.I.; Birn, H.; Storm, T.; Weyer, K.; Nielsen, R. Endocytic receptors in the renal proximal tubule. Physiology (Bethesda) 2012, 27, 223–236. [Google Scholar] [CrossRef]
- Nykjaer, A.; Fyfe, J.C.; Kozyraki, R.; Leheste, J.R.; Jacobsen, C.; Nielsen, M.S.; Ray, R. Cubilin dysfunction causes abnormal metabolism of the steroid hormone 25(OH) vitamin D(3). Proc. Natl. Acad. Sci. USA 2001, 98, 13895–13900. [Google Scholar] [CrossRef]
- Kaseda, R.; Hosojima, M.; Sato, H.; Saito, A. Role of Megalin and Cubilin in the Metabolism of Vitamin D3. Ther. Apher. Dial. 2011, 15, 14–17. [Google Scholar] [CrossRef]
- Bonnet, L.; Karkeni, E.; Couturier, C.; Astier, J.; Dalifard, J.; Defoort, C.; Svilar, L.; Martin, J.C.; Tourniaire, F.; Landrier, J.F. Gene Expression Pattern in Response to Cholecalciferol Supplementation Highlights Cubilin as a Major Protein of 25(OH)D Uptake in Adipocytes and Male Mice White Adipose Tissue. Endocrinology 2018, 159, 957–966. [Google Scholar] [CrossRef]
- Landrier, J.F.; Marcotorchino, J.; Tourniaire, F. Lipophilic micronutrients and adipose tissue biology. Nutrients 2012, 4, 1622–1649. [Google Scholar] [CrossRef]
- Amsellem, S.; Gburek, J.; Hamard, G.; Nielsen, R.; Willnow, T.E.; Devuyst, O.; Nexo, E.; Verroust, P.J.; Christensen, E.I.; Kozyraki, R. Cubilin is essential for albumin reabsorption in the renal proximal tubule. J. Am. Soc. Nephrol. 2010, 21, 1859–1867. [Google Scholar] [CrossRef]
- Bikle, D.D.; Schwartz, J. Vitamin D Binding Protein, Total and Free Vitamin D Levels in Different Physiological and Pathophysiological Conditions. Front. Endocrinol. (Lausanne) 2019, 10, 317. [Google Scholar] [CrossRef]
- Kuro, O.M. A phosphate-centric paradigm for pathophysiology and therapy of chronic kidney disease. Kidney Int. Suppl. 2013, 3, 420–426. [Google Scholar] [CrossRef]
- Hu, M.C.; Shiizaki, K.; Kuro-o, M.; Moe, O.W. Fibroblast Growth Factor 23 and Klotho: Physiology and Pathophysiology of an Endocrine Network of Mineral Metabolism. Annu. Rev. Physiol. 2013, 75, 503–533. [Google Scholar] [CrossRef]
- Mace, M.L.; Gravesen, E.; Hofman-Bang, J.; Olgaard, K.; Lewin, E. Key role of the kidney in the regulation of fibroblast growth factor 23. Kidney Int. 2015, 88, 1304–1313. [Google Scholar] [CrossRef]
- Noonan, M.L.; White, K.E. FGF23 Synthesis and Activity. Curr. Mol. Biol. Rep. 2019, 5, 18–25. [Google Scholar] [CrossRef]
- Tan, S.J.; Smith, E.R.; Hewitson, T.D.; Holt, S.G.; Toussaint, N.D. The importance of klotho in phosphate metabolism and kidney disease. Nephrology(Carlton) 2014, 19, 439–449. [Google Scholar] [CrossRef]
- Razzaque, M.S. FGF23, klotho and vitamin D interactions: What have we learned from in vivo mouse genetics studies? Adv. Exp. Med. Biol. 2012, 728, 84–91. [Google Scholar] [CrossRef]
- Czaya, B.; Faul, C. The Role of Fibroblast Growth Factor 23 in Inflammation and Anemia. Int J. Mol. Sci. 2019, 20, E4195. [Google Scholar] [CrossRef]
- Fukumoto, S. FGF23 and Bone and Mineral Metabolism. Handb. Exp. Pharmacol. 2019. [Google Scholar] [CrossRef]
- Rodelo-Haad, C.; Santamaria, R.; Muñoz-Castañeda, J.R.; Pendón-Ruiz de Mier, M.V.; Martin-Malo, A.; Rodriguez, M. FGF23, Biomarker or Target? Toxins (Basel) 2019, 11, E175. [Google Scholar] [CrossRef]
- Nguyen-Yamamoto, L.; Karaplis, A.C.; St-Arnaud, R.; Goltzman, D. Fibroblast Growth Factor 23 Regulation by Systemic and Local Osteoblast-Synthesized 1,25-Dihydroxyvitamin D.J. Am. Soc. Nephrol. 2017, 28, 586–597. [Google Scholar] [CrossRef]
- Liu, E.S.; Martins, J.S.; Raimann, A.; Chae, B.T.; Brooks, D.J.; Jorgetti, V.; Bouxsein, M.L.; Demay, M.B. 1,25-Dihydroxyvitamin D Alone Improves Skeletal Growth, Microarchitecture, and Strength in a Murine Model of XLH, Despite Enhanced FGF23 Expression. J. Bone Miner. Res. 2016, 31, 929–939. [Google Scholar] [CrossRef]
- Liu, S.; Quarles, L.D. How fibroblast growth factor 23 works. J. Am. Soc. Nephrol. 2007, 18, 1637–1647. [Google Scholar] [CrossRef]
- Erben, R.G.; Andrukhova, O. FGF23-Klotho signaling axis in the kidney. Bone 2017, 100, 62–68. [Google Scholar] [CrossRef]
- Vervloet, M. Renal and extrarenal effects of fibroblast growth factor 23. Nat. Rev. Nephrol. 2019, 15, 109–120. [Google Scholar] [CrossRef]
- Prié, D.; Friedlander, G. Reciprocal control of 1,25- Dihydroxyvitamin D and FGF23 formation involving the FGF23/ Klotho system. Clin. J. Am. Soc. Nephrol. 2010, 5, 1717–1722. [Google Scholar] [CrossRef]
- Erben, R.G. Physiological actions of fibroblast growth factor23. Front. Endocrinol. 2018, 9, 267. [Google Scholar] [CrossRef]
- D’Arrigo, G.; Pizzini, P.; Cutrupi, S.; Tripepi, R.; Tripepi, G.; Mallamaci, F.; Zoccali, C. FGF23 and the PTH response to paricalcitol in chronic kidney disease. Eur. J. Clin. Invest. 2019, 50, e13196. [Google Scholar] [CrossRef]
- Charoenngam, N.; Rujirachun, P.; Holick, M.F.; Ungprasert, P. Oral vitamin D(3) supplementation increases serum fibroblast growth factor 23 concentration in vitamin D-deficient patients: A systematic review and meta-analysis. Osteoporos Int. 2019, 30, 2183–2193. [Google Scholar] [CrossRef]
- Ozeki, M.; Fujita, S.; Kizawa, S.; Morita, H.; Sohmiya, K.; Hoshiga, M.; Ishizaka, N. Association of serum levels of FGF23 and α-Klotho with glomerular filtration rate and proteinuria among cardiac patients. BMC Nephrol. 2014, 15, 147. [Google Scholar] [CrossRef]
- Erben, R.G. Update on FGF23 and Klotho signaling. Mol. Cell Endocrinol. 2016, 432, 56–65. [Google Scholar] [CrossRef]
- Hu, M.C.; Shi, M.; Zhang, J.; Pastor, J.; Nakatani, T.; Lanske, B.; Razzaque, M.S.; Rosenblatt, K.P.; Baum, M.G.; Kuro-o, M.; et al. Klotho: A novel phosphaturic substance acting as an autocrine enzyme in the renal proximal tubule. FASEB J. 2010, 24, 3438–3450. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Guo, X.; Che, L.; Guan, X.; Wu, B.; Lu, R.; Zhu, M.; Pang, H.; Yan, Y.; Ni, Z.; et al. Klotho Reduces Necroptosis by Targeting Oxidative Stress Involved in Renal Ischemic-Reperfusion Injury. Cell Physiol. Biochem. 2018, 45, 2268–2282. [Google Scholar] [CrossRef] [PubMed]
- Song, J.H.; Lee, M.Y.; Kim, Y.J.; Park, S.R.; Kim, J.; Ryu, S.Y.; Jung, J.Y. Developmental immunolocalization of the Klotho protein in mouse kidney epithelial cells. Eur. J. Histochem. 2014, 58, 2256. [Google Scholar] [CrossRef]
- Van’T Hoff, W.G. Molecular developments in renal tubulopathies. Arch. Dis. Child. 2000, 83, 189–191. [Google Scholar]
- Foreman, J.W. Fanconi Syndrome. Pediatr. Clin. North. Am. 2019, 66, 159–167. [Google Scholar] [CrossRef]
- Kashoor, I.; Batlle, D. Proximal renal tubular acidosis with and without Fanconi syndrome. Kidney Res. Clin. Pr. 2019, 38, 267–281. [Google Scholar] [CrossRef]
- Taylor, H.C.; Elbadawy, E.H. Renal tubular acidosis type 2 with Fanconi’s syndrome, osteomalacia, osteoporosis, and secondary hyperaldosteronism in an adult consequent to vitamin D and calcium deficiency: Effect of vitamin D and calcium citrate therapy. Endocr. Pr. 2006, 12, 559–567. [Google Scholar] [CrossRef]
- Ali, S.A.; Tariq, M. Successful treatment of proximal renal tubular acidosis and Fanconi syndrome with vitamin D replacement. Saudi J. Kidney Dis. Transpl. 2016, 27, 812–815. [Google Scholar] [CrossRef]
- Baran, D.T.; Marcy, T.W. Evidence for a defect in vitamin D metabolism in a patient with incomplete Fanconi syndrome. J. Clin. Endocrinol. Metab. 1984, 59, 998–1001. [Google Scholar] [CrossRef]
- Cunha, T.D.S.; Heilberg, I.P. Bartter syndrome: Causes, diagnosis, and treatment. Int. J. Nephrol. Renovasc. Dis. 2018, 11, 291–301. [Google Scholar] [CrossRef]
- Li, X.Y.; Jiang, Y.; Xu, L.J.; Duan, L.; Peng, X.Y.; Chen, L.M.; Xia, W.B.; Xing, X.P. A clinical and hereditary analysis of novel complex heterozygous KCNJ1 mutation in a Bartter syndrome type Ⅱ patient. Zhonghua Nei Ke Za Zhi 2017, 56, 760–762. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, S.; Jagadeesh, A. Bartter Syndrome with Nephrogenic Diabetes Insipidus and Vitamin D Resistant Rickets. Indian Pediatr. 2017, 54, 165–166. [Google Scholar] [PubMed]
- Storm, T.; Zeitz, C.; Cases, O.; Amsellem, S.; Verroust, P.J.; Madsen, M.; Benoist, J.F.; Passemard, S.; Lebon, S.; Jønsson, I.M.; et al. Detailed investigations of proximal tubular function in Imerslund-Gräsbeck syndrome. BMC Med. Genet. 2013, 14, 111. [Google Scholar] [CrossRef] [PubMed]
- Ciancio, J.I.R.; Furman, M.; Banka, S.; Grunewald, S. Profound vitamin D deficiency in four siblings with Imerslund-Grasbeck syndrome with homozygous CUBN mutation. Jimd Rep. 2019, 49, 43–47. [Google Scholar] [CrossRef][Green Version]
- Emma, F.; Cappa, M.; Antoniazzi, F.; Bianchi, M.L.; Chiodini, I.; Eller Vainicher, C.; Di Iorgi, N.; Maghnie, M.; Cassio, A.; Balsamo, A.; et al. X-linked hypophosphatemic rickets: An Italian experts’ opinion survey. Ital. J. Pediatr. 2019, 45, 67. [Google Scholar] [CrossRef]
- Haffner, D.; Waldegger, S. Disorders of phosphorus metabolism. In Pediatric Kidney Disease; Geary, D.F., Schaefer, F., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 953–972. [Google Scholar]
- Gohil, A.; Imel, E.A. FGF23 and Associated Disorders of Phosphate Wasting. Pediatr. Endocrinol. Rev. 2019, 17, 17–34. [Google Scholar] [CrossRef]
- Conti, G.; Chirico, V.; Lacquaniti, A.; Silipigni, L.; Fede, C.; Vitale, A.; Fede, C. Vitamin D intoxication in two brothers: Be careful with dietary supplements. J. Pediatr. Endocrinol. Metab. 2014, 27, 763–767. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gembillo, G.; Cernaro, V.; Siligato, R.; Curreri, F.; Catalano, A.; Santoro, D. Protective Role of Vitamin D in Renal Tubulopathies. Metabolites 2020, 10, 115. https://doi.org/10.3390/metabo10030115
Gembillo G, Cernaro V, Siligato R, Curreri F, Catalano A, Santoro D. Protective Role of Vitamin D in Renal Tubulopathies. Metabolites. 2020; 10(3):115. https://doi.org/10.3390/metabo10030115
Chicago/Turabian StyleGembillo, Guido, Valeria Cernaro, Rossella Siligato, Francesco Curreri, Antonino Catalano, and Domenico Santoro. 2020. "Protective Role of Vitamin D in Renal Tubulopathies" Metabolites 10, no. 3: 115. https://doi.org/10.3390/metabo10030115
APA StyleGembillo, G., Cernaro, V., Siligato, R., Curreri, F., Catalano, A., & Santoro, D. (2020). Protective Role of Vitamin D in Renal Tubulopathies. Metabolites, 10(3), 115. https://doi.org/10.3390/metabo10030115










