Evaluation of the Potential Allergenicity of Strawberries in Response to Different Farming Practices
Abstract
1. Introduction
2. Materials and Methods
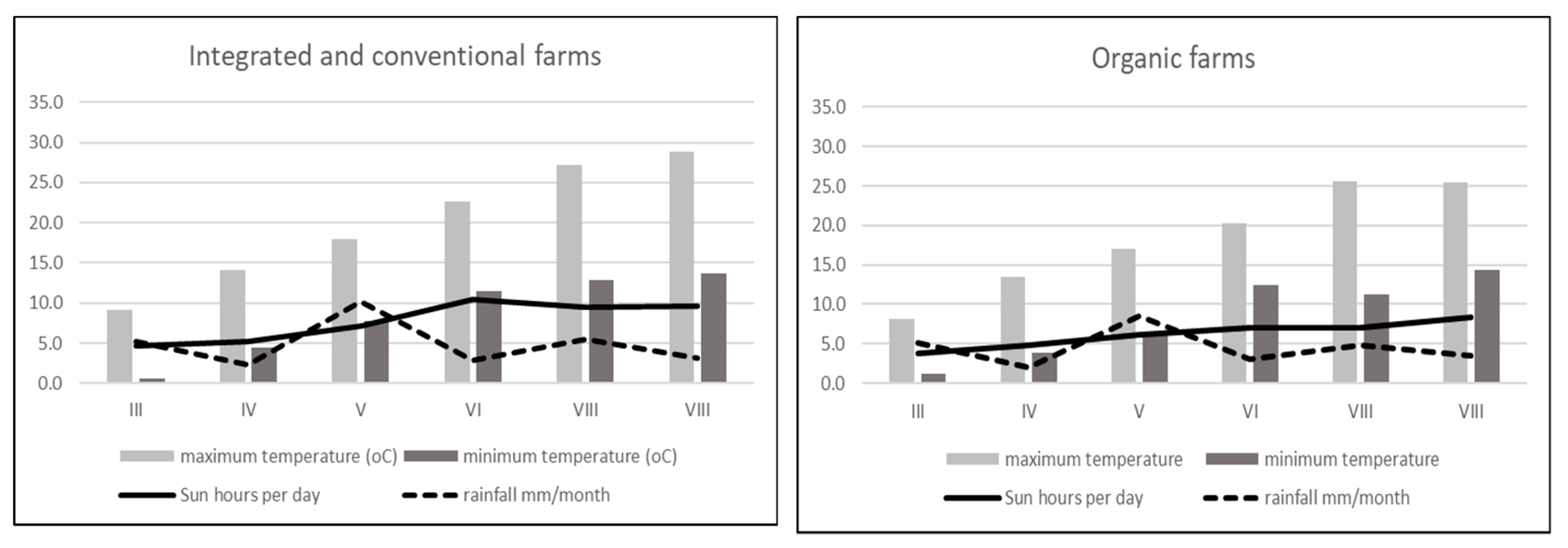
2.1. Fruits Origins
2.2. Plant Material Preparation
2.3. Dry Matter Content
2.4. Flavonoids Content
2.5. Anthocyanins Content
2.6. Potential Allergenicity Analysis
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Auerbach, B.J.; Littman, A.J.; Tinker, L.; Larson, J.; Krieger, J.; Young, B.; Neuhouser, M. Associations of 100% fruit juice versus whole fruit with hypertension and diabetes risk in postmenopausal women: Results from the Women’s Health Initiative. Prev. Med. 2017, 105, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Qi-Jun, W.; Lang, W.; Li-Qiang, Z.; Xin, X.; Chao, J.; Ting-Ting, G. Consumption of fruit and vegetables reduces risk of pancreatic cancer: Evidence from epidemiological studies. Eur. J. Cancer Prev. 2016, 25, 196–205. [Google Scholar]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef] [PubMed]
- Wagner, M.G.; Rhee, Y.; Honrath, K.; Salafia, E.H.B. Nutrition education effective in increasing fruit and vegetable, consumption among overweight and obese adults. Appetite 2016, 100, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Chavesa, V.C.; Calvete, E.; Reginattoa, F.H. Quality properties and antioxidant activity of seven strawberry (Fragaria x ananassa duch) cultivars. Sci. Hort. 2017, 225, 293–298. [Google Scholar] [CrossRef]
- Fernandes, F.C.; Domingues, V.F.; de Freitas, F.; Delerue-Matos, C.; Mateus, N. Strawberries from integrated pest management and organic farming: Phenolic composition and antioxidant properties. Food Chem. 2012, 134, 1926–1931. [Google Scholar] [CrossRef]
- Forbes-Hernandez, T.Y.; Gasparrini, M.; Afrin, S.; Bompadre, S.; Mezzetti, B.; Quiles, J.L.; Giampieri, F.; Battino, M. The healthy effects of strawberry polyphenols: Which strategy behind antioxidant capacity? Crit. Rev. Food Sci. Nutr. 2016, 56, 46–59. [Google Scholar] [CrossRef]
- Council Regulation (EC) No 834/2007 of 28 June 2007 on Organic Production and Labelling of Organic Products and Repealing Regulation (EEC) No 2092/91. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32007R0834 (accessed on 15 December 2019).
- Dayan, F.E.; Cantrell, C.L.; Duke, S.O. Natural products in crop protection. Bioorg. Med. Chem. 2009, 17, 4022–4034. [Google Scholar] [CrossRef]
- Young, J.E.; Zhao, X.; Carey, E.E.; Welti, R.; Yang, S.-S.; Wang, W. Phytochemical phenolics in organically grown vegetables. Mol. Nutr. Food Res. 2005, 49, 1136–1142. [Google Scholar] [CrossRef]
- Valiante, D.; Sirtori, I.; Cossa, S.; Corengia, L.; Pedretti, M.; Cavallaroa, L.; Vignoli, L.; Galvagni, A.; Gomarasca, S.; Pesce, G.R.; et al. Environmental impact of strawberry production in Italy and Switzerland with different cultivation practices. Sci. Total Environ. 2019, 664, 249–261. [Google Scholar] [CrossRef]
- Hallmann, E.; Piotrowska, A.; Świąder, K. The effect of organic practices on the bioactive compounds content in strawberry fruits. J. Res. Appl. Agric. Engng. 2016, 61, 176–179. [Google Scholar]
- Roussos, P.A.; Triantafilidis, A.; Kepolas, E. Strawberry fruits production and quality under conventional, integrated and organic management. Acta Hort. 2010, 916, 125–126. [Google Scholar] [CrossRef]
- Barański, M.; Średnicka-Tober, D.; Volakakis, N.; Seal, C.; Sanderson, R.; Stewart, G.B.; Benbrook, C.; Biavati, B.; Markellou, E.; Giotis, C.; et al. Higher antioxidant and lower cadmium concentrations and lower incidence of pesticide residues in organically grown crops: A systematic literature review and meta-analyses. Br. J. Nutrit. 2012, 112, 794–811. [Google Scholar] [CrossRef]
- Laimer, M.; Maghuly, E. Awareness and knowledge of allergens: A need and a challenge to assure a safe and healthy consumption of small fruits. J. Berry Res. 2010, 1, 61–71. [Google Scholar] [CrossRef]
- Sicherer, S.H.; Sampson, H.A. Food allergy. J. Allergy Clin. Immunol. 2010, S116–S125. [Google Scholar] [CrossRef]
- Guarino, C.; Sciarrillo, R. The identification of allergen proteins in two different varieties of strawberry by two different approaches: Proteomic and western blotting method. Ann. Agric. Sci. 2018, 63, 181–189. [Google Scholar] [CrossRef]
- Kovacova-Hanuskova, E.; Buday, T.; Gavliakova, S.; Plevkova, J. Histamine, histamine intoxication and intolerance. Allergol. Immunopathol. 2015, 43, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Vlieg-Boerstra, B.J.; Van der Heide, S.; Oude, J.N.G.; Kluin-Nelemans, J.C.; Dubois, A.E. Mastocytosis and adverse reactions to biogenic amines and histamine-releasing foods. What is the evidence? Neth. J. Med. 2005, 63, 244–249. [Google Scholar] [PubMed]
- Maintz, L.; Novak, N. Histamine and histamine intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef]
- Sánchez-Pérez, S.; Comas-Basté, O.; Rabell-González, J.; Veciana-Nogués, M.T.; Latorre-Moratalla, M.L.; Vidal-Carou, M.C. Biogenic amines in plant-origin foods: Are they frequently underestimated in low-histamine diets? Foods 2018, 7, 205. [Google Scholar] [CrossRef]
- Ibranji, A.; Nikolla, E.; Loloci, G.; Mingomataj, E. A case report on transitory histamine intolerance from strawberry intake in a 15 month old child with acute gastroenteritis. Clin. Transl. Allergy 2015, 5, 61. [Google Scholar] [CrossRef]
- Barre, A.; Simplicien, M.; Benoist, H.; Rougé, P. Fruit allergies: Beware of the seed allergens! Allergies aux fruits: Attention aux allerg?nes des grains. Rev. Fr. Allergol. 2018, 58, 308–317. [Google Scholar] [CrossRef]
- Muñoz, C.; Hoffmann, T.; Escobar, N.M.; Ludemann, F.; Botella, M.A.; Valpuesta, V.; Schwab, W. The strawberry fruit Fra a allergen functions in flavonoid biosynthesis. Mol. Plant. 2010, 3, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Hyun, T.K.; Kim, J.-S. Genomic identification of putative allergen genes in woodland strawberry (Fragaria vesca) and mandarin orange (Citrus clementina). Plant Omics 2011, 4, 428–434. [Google Scholar]
- PN-EN 12145:2001 Polish Standard Committee. Available online: https://infostore.saiglobal.com/en-us/Standards/PN-EN-12145-2001-926782_SAIG_PKN_PKN_2186561/ (accessed on 15 December 2019).
- Hallmann, E.; Rozpara, E.; Słowianek, M.; Leszczyńska, J. The effect of organic and conventional farm management on the allergenic potency and bioactive compounds status of apricots (Prunus armeniaca L.). Food Chem. 2019, 279, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Dóka, O.; Ficzek, G.; Bicanic, D.; Spruijt, R.; Luterotti, S.; Tóth, M.; Buijnsters, J.G.; Vegvari, G. Direct phytochemical techniques for rapid quantification of total anthocyanins content in sour cherry cultivars. Talanta 2011, 84, 341–346. [Google Scholar] [CrossRef]
- Crecente-Campo, J.; Nunes-Damaceno, M.; Romero-Rodríguez, M.A.; Vázquez-Odériz, M.L. Color, anthocyanin pigment, ascorbic acid and total phenolic compound determination in organic versus conventional strawberries (Fragaria x ananassa Duch, cv Selva). J. Food Compos. Anal. 2012, 28, 23–30. [Google Scholar] [CrossRef]
- Giampieri, F.; Tulipani, S.; Alvarez-Suarez, J.M.; Quiles, J.L.; Mezzetti, B.; Battino, M. The strawberry: Composition, nutritional quality, and impact on human health. Nutrition 2012, 28, 9–19. [Google Scholar] [CrossRef]
- Tulipani, S.; Marzban, G.; Herndl, A.; Laimer, M.; Mezzetti, B.; Battino, M. Influence of environmental and genetic factors on health-related compounds in strawberry. Food Chem. 2011, 124, 906–913. [Google Scholar] [CrossRef]
- Gendron, M.; Gravel, V.; Carisse, O. Assessment tool to compare the environmental, economic and social sustainability of strawberry production systems in Quebec. Acta Hort. 2017, 1156, 256–262. [Google Scholar] [CrossRef]
- Abountiolas, M.; Kelly, K.; Yagiz, Y.; Li, Z.; Mahnken, G.; Borejsza-Wysocki, W.; Marshall, M.; Sims, C.A.; Peres, N.; do Nascimento Nunes, M.C. Sensory quality, physicochemical attributes, polyphenol profiles, and residual fungicides in strawberries from different disease-control treatments. J. Agric. Food Chem. 2018, 66, 6986–6996. [Google Scholar] [CrossRef]
- Asami, D.K.; Hong, Y.-J.; Barrett, D.M.; Mitchell, A.E. Comparison of the total phenolic and ascorbic acid content of freeze-dried and air-dried marionberry, strawberry, and corn grown using conventional, organic, and sustainable agricultural practices. J. Agric. Food Chem. 2003, 51, 1237–1241. [Google Scholar] [CrossRef]
- Conti, S.; Villari, G.; Faugnoa, S.; Melchionna, G.; Somma, S.; Caruso, G. Effects of organic vs. conventional farming system on yield and quality of strawberry grown as an annual or biennial crop in southern Italy. Sci. Hort. 2014, 180, 63–71. [Google Scholar] [CrossRef]
- Zhou, S.; Bhatti, S.; Wei, S.; Chen, F.-C. Organic strawberry production in Tennessee, USA, and areas of comparable climate in China. In Organic Farming for Sustainable Agriculture; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Abrol, D.P.; Gorka, A.K.; Ansari, M.J.; Al-Ghamdi, A.; Al-Kahtani, S. Impact of insect pollinators on yield and fruit quality of strawberry. Saudi J. Biol. Sci. 2019, 26, 524–530. [Google Scholar] [CrossRef]
- Bloksma, J.; Northolt, M.; Huber, M.; van der Burgt, G.J.; van de Vijver, L. A new food quality concept based on life processes. In Handbook of Organic Food Safety and Quality; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Vagiri, M.; Johansson, E.; Rumpunen, K. Phenolic compounds in black currant leaves—An interaction between the plant and foliar diseases? J. Plant Interact. 2017, 12, 193–199. [Google Scholar] [CrossRef]
- Bautista, I.; Boscaiu Neagu, M.T.; Lidón, A.; Llinares Palacios, J.V.; Lull, C.; Donat-Torres, M.P.; Mayoral García-Berlanga, O.; Vicented, O. Environmentally induced changes in antioxidant phenolic compounds levels in wild plants. Acta Physiol. Plant. 2016, 38, 1–15. [Google Scholar] [CrossRef]
- Fernández-Lara, R.; Gordillo, B.; Rodríguez-Pulido, F.J.; González-Miret, M.L.; del Villar-Martínez, A.A.; Dávila-Ortiz, G.; Heredia, F.J. Assessment of the differences in the phenolic composition and color characteristics of new strawberry (Fragaria x ananassa Duch.) cultivars by HPLC-MS and Imaging Tristimulus Colorimetry. Food Res. Int. 2015, 76, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Ariza, M.T.; Reboredo-Rodríguez, P.; Mazzoni, L.; Forbes-Hernández, T.Y.; Giampieri, F.; Afrin, S.; Gasparrini, M.; Soria, C.; Martínez-Ferri, E.; Battino, M.; et al. Strawberry achenes are an important source of bioactive compounds for human health. Int. J. Mol. Sci. 2016, 17, 1103. [Google Scholar] [CrossRef] [PubMed]
- Meinhart, A.D.; Damina, F.M.; Caldeirão, L.; Filhoa, M.J.; da Silva, M.C.; Constant, L.S.; Filho, J.T.; Wagner, R.; Godoya, H.T. Chlorogenic and caffeic acids in 64 fruits consumed in Brazil. Food Chem. 2019, 286, 51–63. [Google Scholar] [CrossRef]
- D’Evoli, L.; Lucarini, M.; del Pulgar, J.S.; Aguzzi, A.; Gabrielli, A.; Gambelli, L.; Lombardi-Boccia, G. Phenolic acids content and nutritional quality of conventional, organic and biodynamic cultivations of the tomato CXD271BIO breeding line (Solanum lycopersicum L.). Food Nutr. Sci. 2016, 7, 1112–1121. [Google Scholar] [CrossRef]
- Xie, Z.; Charles, M.T.; Fan, J.; Charlebois, D.; Khanizadeh, S.; Rolland, D.; Roussel, D.; Deschênesa, M.; Dubéa, C. Effects of preharvest ultraviolet-C irradiation on fruit phytochemical profiles and antioxidant capacity in three strawberry (Fragaria × ananassa Duch.) cultivars. J. Sci. Food Agric. 2015, 95, 2996–3002. [Google Scholar] [CrossRef]
- De Oliveira, I.R.; Rodrigues, G.; Crizel, G.R.; Severo, J.; Renard, C.M.G.C.; Chaves, F.C.; Rombaldi, C.V. Preharvest UV-C radiation influences physiological, biochemical, and transcriptional changes in strawberry cv. Camarosa. Plant Phys. Biochem. 2016, 108, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Hjernoe, K.; Alm, R.; Canbäck, B.; Matthiesen, R.; Trajkovski, K.; Björk, L.; Roepstorff, P.; Emanuelsson, C. Down-regulation of the strawberry Bet v1 homologous in the concert with the flavonoids biosynthesis pathway in colorless strawberry mutant. Proteomics 2006, 6, 1574–1587. [Google Scholar] [CrossRef] [PubMed]
- Hoppin, J.A.; Umbach, D.M.; Long, S.; London, S.J.; Henneberger, P.K.; Blair, A.; Alavanja, M.; Freeman, L.E.B.; Sandler, D.P. Pesticides are associated with allergic and non-allergic wheeze among male farmers. Environ. Health Perspect. 2017, 125, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Nyambok, E.; Robinson, C. The role of food additives and chemicals in food allergy. Ann. J. Food Process. Pres. 2016, 1, 1006–1009. [Google Scholar]
- Tsujia, M.; Kawamoto, T.; Koriyama, C.; Yamamoto, M.; Tsuchiy, T.; Matsumura, F. Association of PCBs and allergies in children. Pestic. Biochem. Phys. 2015, 120, 21–26. [Google Scholar] [CrossRef]
- Blair, A.; Ritz, B.; Wesseling, C.; Freeman, L.B. Pesticides and human health. Occup. Environ. Med. 2014, 1, 1–3. [Google Scholar] [CrossRef]
- Reganold, J.P.; Andrews, P.K.; Reeve, J.R.; Carpenter-Boggs, L.; Schadt, C.W.; Alldredge, J.R.; Ross, C.F.; Davies, N.M.; Zhou, J. Fruit and soil quality of organic and conventional strawberry agroecosystems. PLoS ONE. 2010, 5, 123–146. [Google Scholar] [CrossRef]
- Słowianek, M.; Skorupa, M.; Hallmann, E.; Rembiałkowska, E.; Leszczyńska, J. Allergenicity of tomatoes cultivated in organic and conventional systems. Plant Food Hum. Nutr. 2016, 71, 35–41. [Google Scholar] [CrossRef]
- Franz, K.; Eberlein, B.; Hücherig, S.; Edelmann, K.; Besbes, F.; Ring, J.; Darsow, U.; Schwab, W. Breeding of hypoallergenic strawberry fruit. J. Berry Res. 2013, 3, 197–201. [Google Scholar] [CrossRef]




| Crop Production System | Localization | Soil Type | Forecrop | Irrigation | Organic and Mineral Fertilizers | Plant Protection |
|---|---|---|---|---|---|---|
| Organic Farms | Leokadiów (51°24’ N21°48’ E) | Luvisol, pH(KCl) 5.6 | Cereals | No | Cow manure 5 t ha−1 | No |
| Ignaców (51°45’ N 20°38’ E) | Luvisol, pH(KCl) 6.1 | Fabaceae | No | Cow manure 5 t ha−1 Biosol (organic fertiliser) 300 kg ha−1 | No | |
| Cyganówka (51°21’ N 21°37’ E) | Luvisol, pH(KCl) 6.2 | Potato | No | Cow manure 5 t ha−1 | No | |
| Mieczysławów (51°22’ N 21°39’ E) | Luvisol, pH(KCl) 6.3 | Fabaceae | No | Cow manure 10 t−1 nettle manure 1 t ha−1 | No | |
| Conventional Farms | Dobrosławów (51°22’ N 21°39’ E) | Luvisol, pH(KCl) 5.9 | Fabaceae | No | Slurry 25 m3 ha−1 Hydrocomplex 200 kg ha−1 (N 12%, P2O5 11%, K2O 18%) | Fungicide 3.9 kg(L) ha−1: Vaxiplant SL 1.0 kg ha−1 Polyversum WP 0.1 kg ha−1; Dymas 2.0 L ha−1 Luna Sensation 500 S.C. 0.8 L ha−1; Insecticide 2.0 L ha−1: Ortus 0.5 S.C. 2.0 L ha−1 |
| Zarzecze (51°24’ N 21°50’ E) | Luvisol, pH(KCl) 6.2 | Cereals/Fabaceae | Yes | Slurry 20 m3 ha−1 Hydrocomplex 200 kg ha−1 | Fungicide 2.8 kg(L) ha−1: Vaxiplant SL 1.0 kg ha−1 Luna Sensation 500 S.C. 0.8 L ha−1; Scorpion 325 S.C. 1.0 L ha−1; Herbicide 2.0 L ha−1: Targa Super 0.5 EC 2 L ha−1 | |
| Dobrosławów (51°22’ N 21°39’E) | Luvisol, pH(KCl) 6.0 | Fabaceae | No | Slurry 35 m3 ha−1 Hydrocomplex 200 kg ha−1 | Fungicide 5.8 kg(L) ha−1: Vaxiplant SL 1.0 kg ha−1 Luna Sensation 500 S.C. 0.8 L ha−1; Pomarsol Forte 80 WG 4.0 kg ha−1; Insecticide 0.2 kg ha−1: Mospilan 20 SP 0.2 kg ha−1 | |
| Dobrosławów (51°22’ N 21°39’ E) | Luvisol, pH(KCl) 6.0 | Fabaceae | No | Slurry 20 m3 ha−1 Hydrocomplex 200 kg ha−1 | Fungicide 1.8 kg(L) ha−1: Vaxiplant SL 1.0 kg ha−1 Luna Sensation 500 S.C. 0.8 L ha−1; Insecticide 0.2 kg ha−1: Mospilan 20 SP 0.2 kg ha−1; Herbicide 1.0 kg ha−1: Agil 100 EC 1.0 kg ha−1 | |
| Integrated Farms | Janów (51°24’ N 21°51’ E) | Luvisol, pH(KCl) 6.2 | Cereals | Yes | Manure 25 t ha−1 Slurry 20 m3 ha−1 Ammonium sulphate 150 kg ha−1 (32% N) Potassium sulfate 150 kg ha−1 (50% K2O, 45% SO3) Superphosphate 150 kg ha−1 (65% P2O5, 10% CaO) | Fungicide 4.8 kg(L) ha−1: Luna Sensation 500 S.C. 0.8 L ha−1 Pomarsol Forte 80 WG 4.0 kg ha−1 |
| Leokadiów (51° 24’ N 21°48’ E) | Luvisol, pH(KCl) 6.2 | Potatoes | No | Slurry 20 m3 ha−1 Hydrocomplex 200 kg ha−1 | Fungicide 1.5 L ha−1: Teldor 500 S.C. 1.5 L ha−1; Insecticide 0.3 kg ha 1: Mospilan 0.3 kg ha−1 | |
| Polesie (51°24’ N 21°46’E) | Luvisol, pH(KCl) 6.3 | Fabaceae | No | Potassium sulfate 150 kg ha−1 Hydrocomplex 200 kg ha−1 | Instecticide 2.3 kg(L) ha−1: Ortus 0.5 S.C. 2.0 L ha−1 Mospilan 20 SP 0.3 kg ha−1; | |
| Cyganówka (51°21’ N 21°37’ E) | Luvisol, pH(KCl) 6.4 | Potatoes | No | Manure 15 t ha−1; Slurry 20 m3 ha−1 Hydrocomplex 200 kg ha−1 | Fungicide 0.8 L ha−1: Luna Sensation 500 S.C. 0.8 L ha−1 Instecticide 0.3 kg ha−1: Mospilan 20 SP 0.3 kg ha−1 |
| Compounds/Production System | Organically Cultivated ‘Honeyoe’ (n) = 8 | Integrated Cultivated ‘Honeyoe’(n) = 8 | Conventionally Cultivated ‘Honeyoe’(n) = 8 |
|---|---|---|---|
| dry matter (g 100 g-1 FW) | 8.711 ± 0.36 a2 | 8.52 ± 0.14 a | 8.20 ± 0.27 a |
| total polyphenols (mg 100 g−1 DW) | 478.26 ± 11.37 a | 499.76 ± 4.59 a | 503.49 ± 17.52 a |
| total phenolic acids | 143.76 ± 4.47 b | 184.29 ± 6.49 a | 178.01 ± 9.01 a |
| gallic | 7.40 ± 1.10 b | 8.83 ± 0.65 b | 17.80 ± 0.17 a |
| chlorogenic | 72.03 ± 3.69 b | 100.12 ± 8.10 a | 96.50 ± 6.86 a |
| p-coumaric | 23.79 ± 1.48 a | 20.24 ± 1.16 ab | 19.80 ± 0.74 b |
| ferulic | 19.42 ± 2.41 a | 27.00 ± 3.86 a | 16.88 ± 3.73 a |
| ellagic | 21.12 ± 1.24 b | 28.10 ± 1.83 a | 27.03 ± 0.70 a |
| total flavonoids (mg 100 g−1 DW) | 334.50 ± 12.24 a | 315.47 ± 5.01 a | 325.48 ± 11.13 a |
| quercetin-3-O-rutinoside | 86.07 ± 5.25 a | 59.45 ± 3.67 a | 25.53 ± 2.46 a |
| quercetin-3-O-glucoside | 29.69 ± 11.05 b | 9.09 ± 0.50 b | 90.92 ± 2.90 a |
| kaempferol-3-O-glucoside | 49.83 ± 3.00 a | 35.09 ± 1.67 b | 43.14 ± 2.88 ab |
| myrycetin | 0.55 ± 0.03 b | 0.43 ± 0.02 c | 0.87 ± 0.02 a |
| luteolin | 1.70 ± 0.09 b | 1.03 ± 0.20 c | 2.85 ± 0.16 a |
| quercetin | 6.07 ± 0.33 a | 6.70 ± 1.26 a | 2.81 ± 0.08 b |
| kaempferol | 7.21 ± 1.40 a | 4.40 ± 0.56 ab | 3.84 ± 0.07 b |
| total anthocyanins (mg 100 g−1 DW) | 153.38 ± 3.77 b | 199.27 ± 5.19 a | 155.51 ± 6.92 b |
| cyanidin-3-O-glucoside | 119.52 ± 3.13 b | 163.27 ± 4.77 a | 120.96 ± 6.13 b |
| pelargonidin-3-O-rutinoside | 8.62 ± 0.05 ab | 8.87 ± 0.10 a | 8.47 ± 0.04 b |
| pelargonidin-3-O-glucoside | 25.24 ± 0.66 a | 27.13 ± 1.71 a | 26.07 ± 0.90 a |
| Bet v1 (ng g−1) | 376.37 ± 15.79 b | 678.28 ± 29.03 a | 637.29 ± 42.10 a |
| profilins (ng g−1) | 483.49 ± 40.15 a | 532.61 ± 34.57 a | 417.89 ± 39.15 a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aninowski, M.; Kazimierczak, R.; Hallmann, E.; Rachtan-Janicka, J.; Fijoł-Adach, E.; Feledyn-Szewczyk, B.; Majak, I.; Leszczyńska, J. Evaluation of the Potential Allergenicity of Strawberries in Response to Different Farming Practices. Metabolites 2020, 10, 102. https://doi.org/10.3390/metabo10030102
Aninowski M, Kazimierczak R, Hallmann E, Rachtan-Janicka J, Fijoł-Adach E, Feledyn-Szewczyk B, Majak I, Leszczyńska J. Evaluation of the Potential Allergenicity of Strawberries in Response to Different Farming Practices. Metabolites. 2020; 10(3):102. https://doi.org/10.3390/metabo10030102
Chicago/Turabian StyleAninowski, Mateusz, Renata Kazimierczak, Ewelina Hallmann, Joanna Rachtan-Janicka, Elżbieta Fijoł-Adach, Beata Feledyn-Szewczyk, Iwona Majak, and Joanna Leszczyńska. 2020. "Evaluation of the Potential Allergenicity of Strawberries in Response to Different Farming Practices" Metabolites 10, no. 3: 102. https://doi.org/10.3390/metabo10030102
APA StyleAninowski, M., Kazimierczak, R., Hallmann, E., Rachtan-Janicka, J., Fijoł-Adach, E., Feledyn-Szewczyk, B., Majak, I., & Leszczyńska, J. (2020). Evaluation of the Potential Allergenicity of Strawberries in Response to Different Farming Practices. Metabolites, 10(3), 102. https://doi.org/10.3390/metabo10030102