An Integrated Analytical Approach Reveals Trichome Acylsugar Metabolite Diversity in the Wild Tomato Solanum pennellii
Abstract
:1. Introduction
2. Results
2.1. Experimental Design
2.2. Untargeted Metabolomics Reveals Acylsugars and Flavonoids in Trichomes
2.3. Acylsugar Core Composition Varies across the S. pennellii Geographic Range
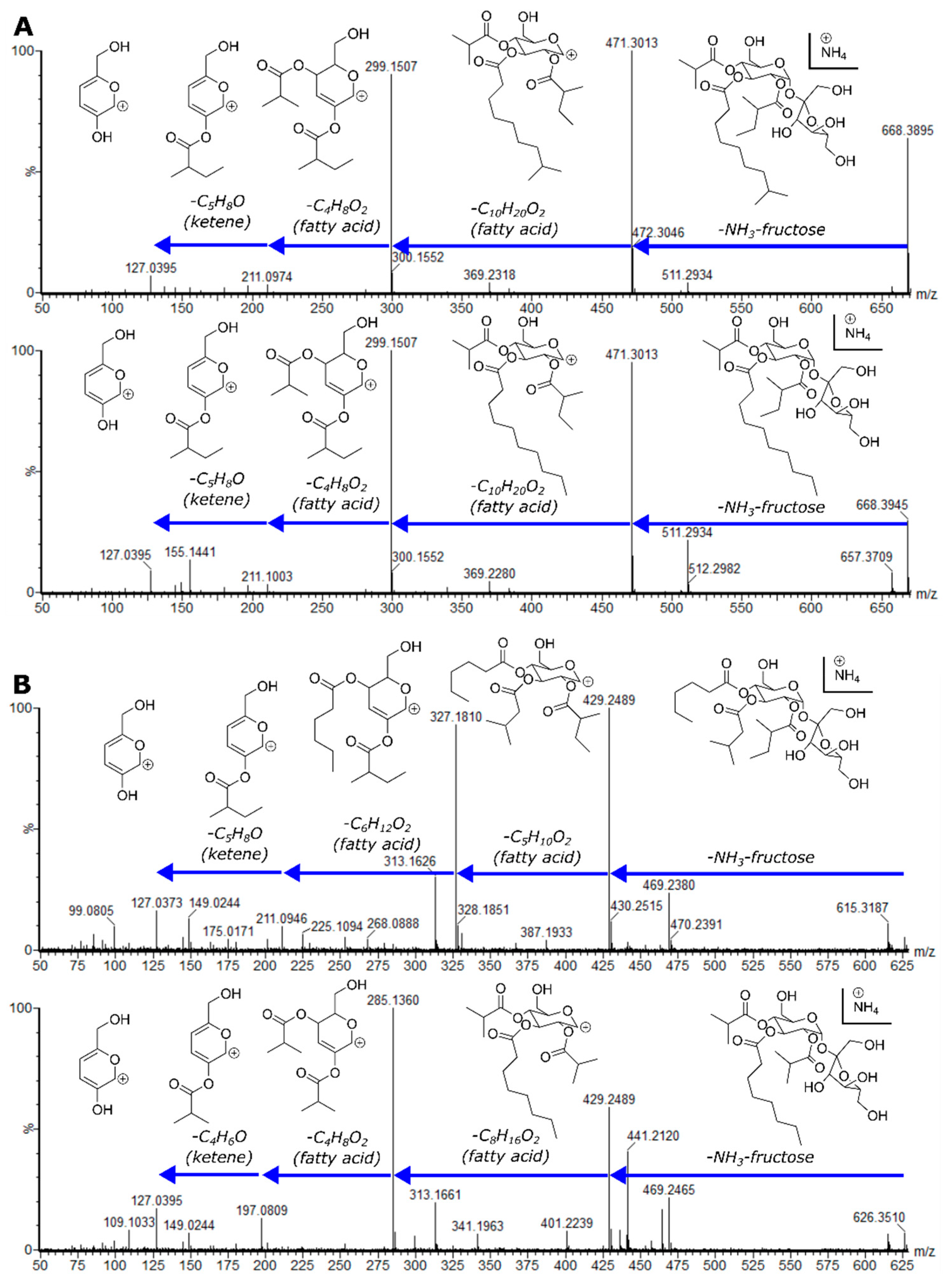
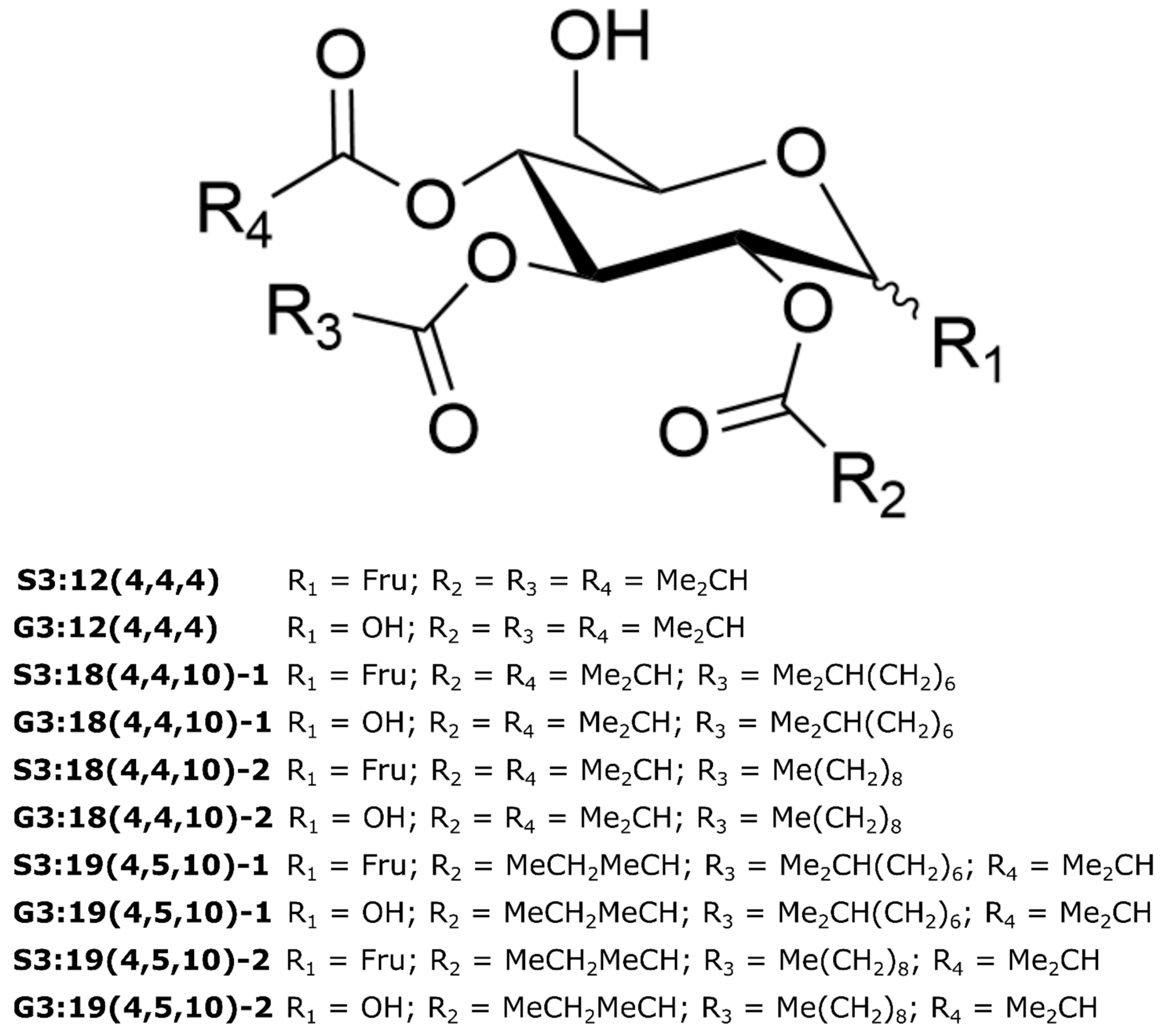
2.4. Variable Acyl Chain and Sugar Composition Yield Acylsugar Diversity
2.5. NMR Spectroscopy Resolves Structural Relationships between Acylsugars
2.6. Flavonoids Vary by Core and Degree of Methylation
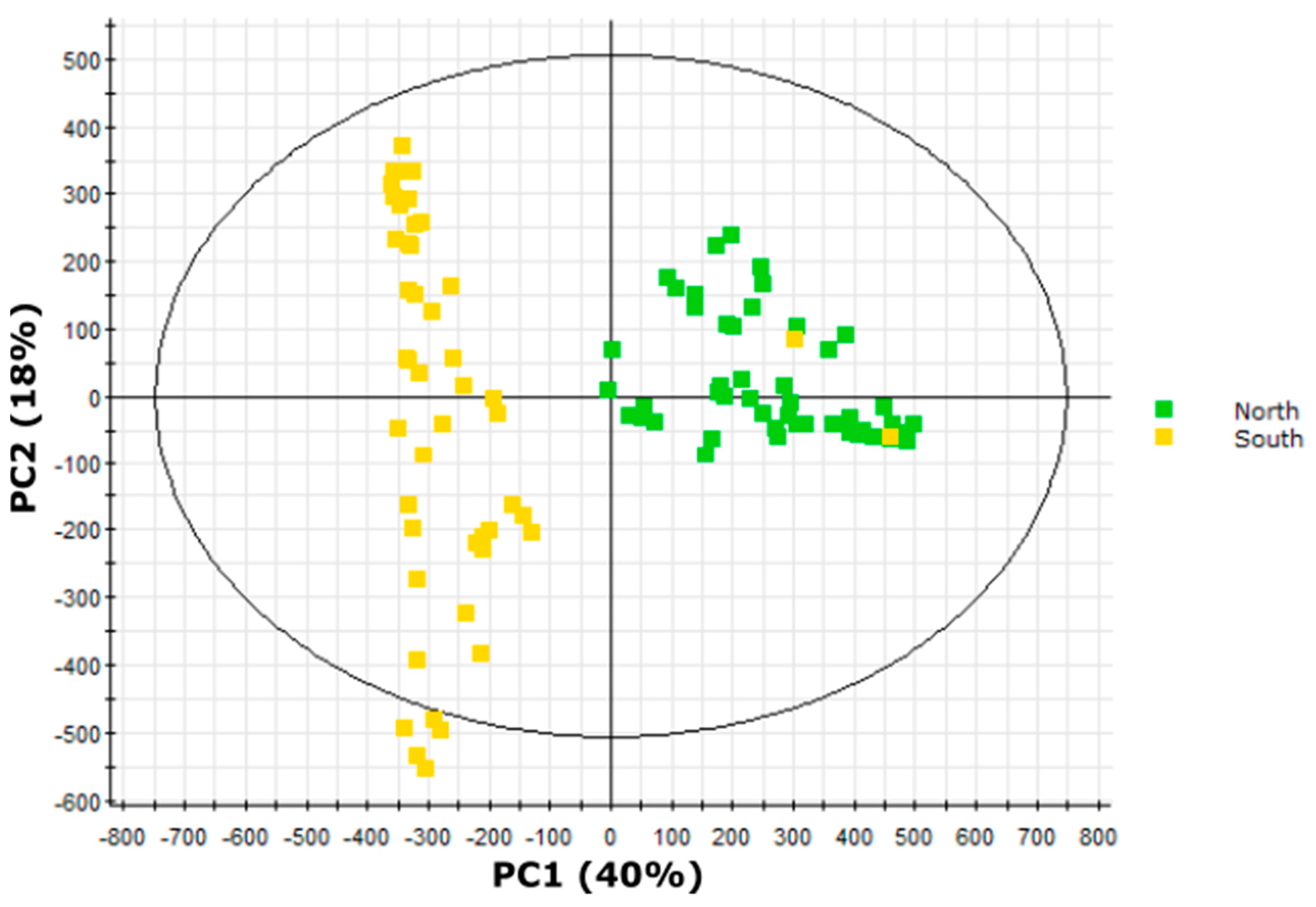
2.7. Multivariate Analysis Implicates Short-Branched Acyl Chains in North-South Acylsugar Variation
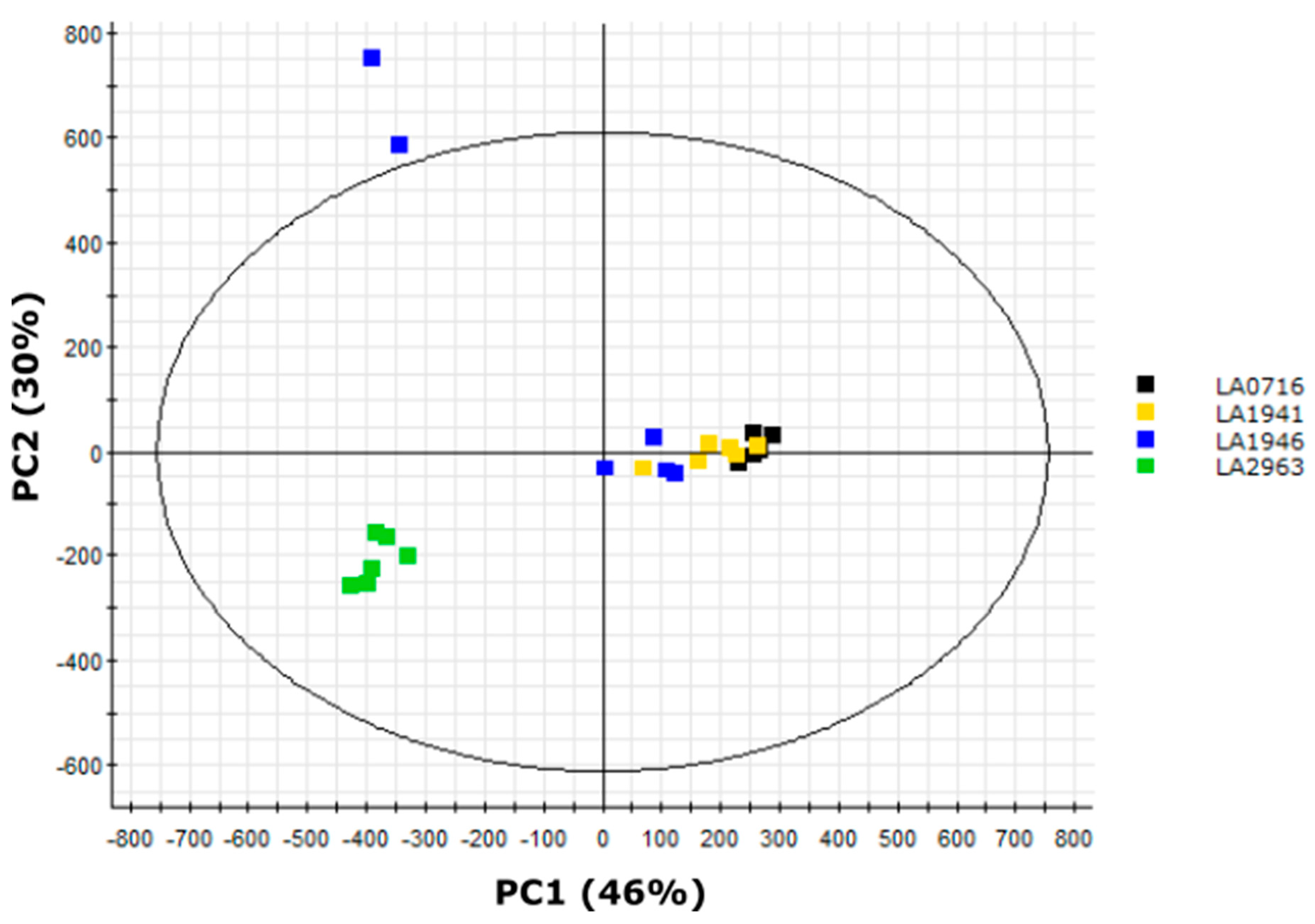
2.8. Variation in Medium-Length Acyl Chains Drives Variation within the South Range
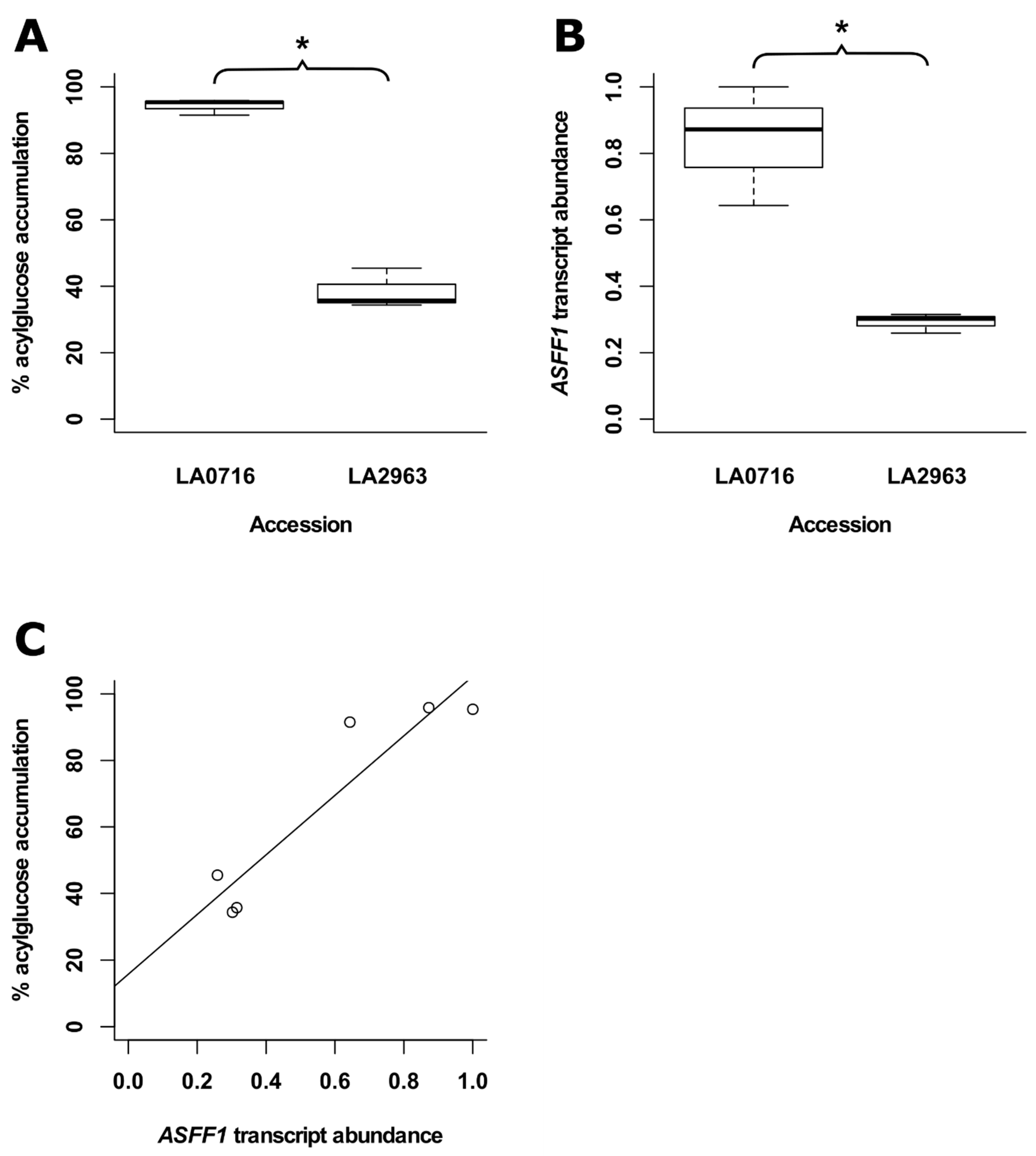
2.9. LA2963 Segregates from Other Atico Region Accessions Due to High Acylsucrose Content
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Acylsugar Extraction
4.3. Metabolomic Analysis by UHPLC–MS
4.4. Untargeted Metabolomics Data Processing
4.5. Acylsugar Quantification
4.6. RNA Extraction, cDNA Synthesis, and qPCR
4.7. Acylsugar Purification
4.8. NMR Spectroscopy
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pichersky, E.; Lewinsohn, E. Convergent evolution in plant specialized metabolism. Annu. Rev. Plant Biol. 2011, 62, 549–566. [Google Scholar] [CrossRef] [Green Version]
- Pichersky, E.; Raguso, R.A. Why do plants produce so many terpenoid compounds? New Phytol. 2018, 220, 692–702. [Google Scholar] [CrossRef] [PubMed]
- Tahara, S. A journey of twenty-five years through the ecological biochemistry of flavonoids. Biosci. Biotechnol. Biochem. 2007, 71, 1387–1404. [Google Scholar] [CrossRef]
- Milo, R.; Last, R.L. Achieving diversity in the face of constraints: Lessons from metabolism. Science 2012, 336, 1663–1667. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Jing, S.-X.; Luo, S.-H.; Li, S.-H. Non-volatile natural products in plant glandular trichomes: Chemistry, biological activities and biosynthesis. Nat. Prod. Rep. 2019, 36, 626–665. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.-H.; Liu, G.; Shi, F.; Jones, A.D.; Beaudry, R.M.; Howe, G.A. The tomato odorless-2 mutant is defective in trichome-based production of diverse specialized metabolites and broad-spectrum resistance to insect herbivores. Plant Physiol. 2010, 154, 262–272. [Google Scholar] [CrossRef] [Green Version]
- Luu, V.T.; Weinhold, A.; Ullah, C.; Dressel, S.; Schoettner, M.; Gase, K.; Gaquerel, E.; Xu, S.; Baldwin, I.T. O-acyl sugars protect a wild tobacco from both native fungal pathogens and a specialist herbivore. Plant Physiol. 2017, 174, 370–386. [Google Scholar] [CrossRef] [Green Version]
- Weinhold, A.; Baldwin, I.T. Trichome-derived O-acyl sugars are a first meal for caterpillars that tags them for predation. Proc. Natl. Acad. Sci. USA 2011, 108, 7855–7859. [Google Scholar] [CrossRef] [Green Version]
- Yang, T.; Stoopen, G.; Wiegers, G.; Mao, J.; Wang, C.; Dicke, M.; Jongsma, M.A. Pyrethrins protect pyrethrum leaves against attack by western flower thrips, Frankliniella occidentalis. J. Chem. Ecol. 2012, 38, 370–377. [Google Scholar] [CrossRef] [Green Version]
- McDowell, E.T.; Kapteyn, J.; Schmidt, A.; Li, C.; Kang, J.-H.; Descour, A.; Shi, F.; Larson, M.; Schilmiller, A.; An, L.; et al. Comparative functional genomic analysis of Solanum glandular trichome types. Plant Physiol. 2011, 155, 524–539. [Google Scholar] [CrossRef] [Green Version]
- Nakashima, T.; Wada, H.; Morita, S.; Erra-Balsells, R.; Hiraoka, K.; Nonami, H. Single-cell metabolite profiling of stalk and glandular cells of intact trichomes with internal electrode capillary pressure probe electrospray ionization mass spectrometry. Anal. Chem. 2016, 88, 3049–3057. [Google Scholar] [CrossRef] [PubMed]
- Fobes, J.F.; Mudd, J.B.; Marsden, M.P.F. Epicuticular lipid accumulation on the leaves of Lycopersicon pennellii (Corr.) D’Arcy and Lycopersicon esculentum Mill. Plant Physiol. 1985, 77, 567–570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leckie, B.M.; D’Ambrosio, D.A.; Chappell, T.M.; Halitschke, R.; De Jong, D.M.; Kessler, A.; Kennedy, G.G.; Mutschler, M.A. Differential and synergistic functionality of acylsugars in suppressing oviposition by insect herbivores. PLoS ONE 2016, 11, e0153345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mirnezhad, M.; Romero-González, R.R.; Leiss, K.A.; Choi, Y.H.; Verpoorte, R.; Klinkhamer, P.G.L. Metabolomic analysis of host plant resistance to thrips in wild and cultivated tomatoes. Phytochem. Anal. 2010, 21, 110–117. [Google Scholar] [CrossRef]
- Smeda, J.R.; Schilmiller, A.L.; Anderson, T.; Ben-Mahmoud, S.; Ullman, D.E.; Chappell, T.M.; Kessler, A.; Mutschler, M.A. Combination of acylglucose QTL reveals additive and epistatic genetic interactions and impacts insect oviposition and virus infection. Mol. Breed. 2018, 38, 3. [Google Scholar] [CrossRef]
- Leckie, B.M.; De Jong, D.M.; Mutschler, M.A. Quantitative trait loci increasing acylsugars in tomato breeding lines and their impacts on silverleaf whiteflies. Mol. Breed. 2012, 30, 1621–1634. [Google Scholar] [CrossRef]
- Leckie, B.M.; De Jong, D.M.; Mutschler, M.A. Quantitative trait loci regulating sugar moiety of acylsugars in tomato. Mol. Breed. 2013, 31, 957–970. [Google Scholar] [CrossRef]
- Leckie, B.M.; Halitschke, R.; De Jong, D.M.; Smeda, J.R.; Kessler, A.; Mutschler, M.A. Quantitative trait loci regulating the fatty acid profile of acylsugars in tomato. Mol. Breed. 2014, 34, 1201–1213. [Google Scholar] [CrossRef]
- Fan, P.; Miller, A.M.; Liu, X.; Jones, A.D.; Last, R.L. Evolution of a flipped pathway creates metabolic innovation in tomato trichomes through BAHD enzyme promiscuity. Nat. Commun. 2017, 8, 2080. [Google Scholar] [CrossRef]
- Fan, P.; Wang, P.; Lou, Y.-R.; Leong, B.J.; Moore, B.M.; Schenck, C.A.; Combs, R.; Cao, P.; Brandizzi, F.; Shiu, S.-H.; et al. Evolution of a plant gene cluster in Solanaceae and emergence of metabolic diversity. eLife 2020, 9, e56717. [Google Scholar] [CrossRef]
- Ning, J.; Moghe, G.; Leong, B.; Kim, J.; Ofner, I.; Wang, Z.; Adams, C.; Jones, A.D.; Zamir, D.; Last, R.L. A feedback insensitive isopropylmalate synthase affects acylsugar composition in cultivated and wild tomato. Plant Physiol. 2015, 169, 1821–1835. [Google Scholar] [CrossRef] [Green Version]
- Leong, B.J.; Lybrand, D.B.; Lou, Y.-R.; Fan, P.; Schilmiller, A.L.; Last, R.L. Evolution of metabolic novelty: A trichome-expressed invertase creates specialized metabolic diversity in wild tomato. Sci. Adv. 2019, 5, eaaw3754. [Google Scholar] [CrossRef] [Green Version]
- Schilmiller, A.L.; Gilgallon, K.; Ghosh, B.; Jones, A.D.; Last, R.L. Acylsugar acylhydrolases: Carboxylesterase-catalyzed hydrolysis of acylsugars in tomato trichomes. Plant Physiol. 2016, 170, 1331–1344. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Jones, A.D. Profiling of stable isotope enrichment in specialized metabolites using liquid chromatography and multiplexed nonselective collision-induced dissociation. Anal. Chem. 2014, 86, 10600–10607. [Google Scholar] [CrossRef]
- Wang, Z. Dynamics, Distribution and Development of Specialized Metabolism in Glandular Trichome of Tomato and Its Wild Relatives. Ph.D. Thesis, Michigan State University, East Lansing, MI, USA, 2015. [Google Scholar]
- Shapiro, J.A.; Steffens, J.C.; Mutschler, M.A. Acylsugars of the wild tomato Lycopersicon pennellii in relation to geographic distribution of the species. Biochem. Syst. Ecol. 1994, 22, 545–561. [Google Scholar] [CrossRef]
- Slocombe, S.P.; Schauvinhold, I.; McQuinn, R.P.; Besser, K.; Welsby, N.A.; Harper, A.; Aziz, N.; Li, Y.; Larson, T.R.; Giovannoni, J.; et al. Transcriptomic and reverse genetic analyses of branched-chain fatty acid and acyl sugar production in Solanum pennellii and Nicotiana benthamiana. Plant Physiol. 2008, 148, 1830–1846. [Google Scholar] [CrossRef] [Green Version]
- Burke, B.A.; Goldsby, G.; Mudd, B.J. Polar epicuticular lipids of Lycopersicon pennellii. Phytochemistry 1987, 26, 2567–2571. [Google Scholar] [CrossRef]
- Correll, D.S. The Potato and Its Wild Relatives; Contributions from Texas Research Foundation; Texas Research Foundation: Renner, TX, USA, 1962. [Google Scholar]
- Ghangas, G.S.; Steffens, J.C. UDPglucose: Fatty acid transglucosylation and transacylation in triacylglucose biosynthesis. Proc. Natl. Acad. Sci. USA 1993, 90, 9911–9915. [Google Scholar] [CrossRef] [Green Version]
- Kuai, J.P.; Ghangas, G.S.; Steffens, J.C. Regulation of triacylglucose fatty acid composition (uridine diphosphate glucose: Fatty acid glucosyltransferases with overlapping chain-length specificity). Plant Physiol. 1997, 115, 1581–1587. [Google Scholar] [CrossRef] [Green Version]
- Li, A.X.; Steffens, J.C. An acyltransferase catalyzing the formation of diacylglucose is a serine carboxypeptidase-like protein. Proc. Natl. Acad. Sci. USA 2000, 97, 6902–6907. [Google Scholar] [CrossRef] [Green Version]
- Li, A.X.; Eannetta, N.; Ghangas, G.S.; Steffens, J.C. Glucose polyester biosynthesis. Purification and characterization of a glucose acyltransferase. Plant Physiol. 1999, 121, 453–460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, P.; Miller, A.M.; Schilmiller, A.L.; Liu, X.; Ofner, I.; Jones, A.D.; Zamir, D.; Last, R.L. In vitro reconstruction and analysis of evolutionary variation of the tomato acylsucrose metabolic network. Proc. Natl. Acad. Sci. USA 2016, 113, E239–E248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, B.; Westbrook, T.C.; Jones, A.D. Comparative structural profiling of trichome specialized metabolites in tomato (Solanum lycopersicum) and S. habrochaites: Acylsugar profiles revealed by UHPLC/MS and NMR. Metabolomics 2014, 10, 496–507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.; Enright, M.; Barry, C.S.; Jones, A.D. Profiling, isolation and structure elucidation of specialized acylsucrose metabolites accumulating in trichomes of Petunia species. Metabolomics 2017, 13, 85. [Google Scholar] [CrossRef]
- Nadakuduti, S.S.; Uebler, J.B.; Liu, X.; Jones, A.D.; Barry, C.S. Characterization of trichome-expressed bahd acyltransferases in Petunia axillaris reveals distinct acylsugar assembly mechanisms within the Solanaceae. Plant Physiol. 2017, 175, 36–50. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.; Kang, K.; Gonzales-Vigil, E.; Shi, F.; Jones, A.D.; Barry, C.S.; Last, R.L. Striking natural diversity in glandular trichome acylsugar composition is shaped by variation at the acyltransferase2 locus in the wild tomato Solanum habrochaites. Plant Physiol. 2012, 160, 1854–1870. [Google Scholar] [CrossRef] [Green Version]
- Mandal, S.; Ji, W.; McKnight, T.D. Candidate gene networks for acylsugar metabolism and plant defense in wild tomato Solanum pennellii. Plant Cell 2020, 32, 81–99. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, A.; Li, C.; Shi, F.; Jones, A.D.; Pichersky, E. Polymethylated myricetin in trichomes of the wild tomato species Solanum habrochaites and characterization of trichome-specific 3′/5′- and 7/4′-myricetin O-methyltransferases. Plant Physiol. 2011, 155, 1999–2009. [Google Scholar] [CrossRef] [Green Version]
- Sumner, L.W.; Amberg, A.; Barrett, D.; Beale, M.H.; Beger, R.; Daykin, C.A.; Fan, T.W.-M.; Fiehn, O.; Goodacre, R.; Griffin, J.L.; et al. Proposed minimum reporting standards for chemical analysis. Metabolomics 2007, 3, 211–221. [Google Scholar] [CrossRef] [Green Version]
- Schilmiller, A.L.; Moghe, G.D.; Fan, P.; Ghosh, B.; Ning, J.; Jones, A.D.; Last, R.L. functionally divergent alleles and duplicated loci encoding an acyltransferase contribute to acylsugar metabolite diversity in Solanum Trichomes. Plant Cell 2015, 27, 1002–1017. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, A.; Li, C.; Jones, A.D.; Pichersky, E. Characterization of a flavonol 3-O-methyltransferase in the trichomes of the wild tomato species Solanum habrochaites. Planta 2012, 236, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Matsuba, Y.; Ning, J.; Schilmiller, A.L.; Hammar, D.; Jones, A.D.; Pichersky, E.; Last, R.L. Analysis of natural and induced variation in tomato glandular trichome flavonoids identifies a gene not present in the reference genome. Plant Cell 2014, 26, 3272–3285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, C.; Schmidt, A.; Pichersky, E.; Shi, F.; Jones, A.D. Identification of methylated flavonoid regioisomeric metabolites using enzymatic semisynthesis and liquid chromatography-tandem mass spectrometry. Metabolomics 2013, 9, 92–101. [Google Scholar] [CrossRef]
- Maluf, W.R.; Maciel, G.M.; Gomes, L.A.A.; Cardoso, M.D.G.; Gonçalves, L.D.; da Silva, E.C.; Knapp, M. Broad-spectrum arthropod resistance in hybrids between high- and low-acylsugar tomato lines. Crop Sci. 2010, 50, 439–450. [Google Scholar] [CrossRef]
- Alba, J.M.; Montserrat, M.; Fernández-Muñoz, R. Resistance to the two-spotted spider mite (Tetranychus urticae) by acylsucroses of wild tomato (Solanum pimpinellifolium) trichomes studied in a recombinant inbred line population. Exp. Appl. Acarol. 2009, 47, 35–47. [Google Scholar] [CrossRef]
- Schilmiller, A.L.; Charbonneau, A.L.; Last, R.L. Identification of a BAHD acetyltransferase that produces protective acyl sugars in tomato trichomes. Proc. Natl. Acad. Sci. USA 2012, 109, 16377–16382. [Google Scholar] [CrossRef] [Green Version]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]



| Name | MSI | RT | Formula | m/zacc | m/zex | Δm (ppm) | Fragment m/z |
|---|---|---|---|---|---|---|---|
| Triacylsucroses | |||||||
| S3:12(4,4,4) | 1 | 2.21 | C24H40O14 | 570.2778 | 570.2756 | 3.9 | 373.1872, 285.1326, 197.0809, 127.0395 |
| S3:13(4,4,5) | 3 | 2.34 | C25H42O14 | 584.2926 | 584.2913 | 2.2 | 387.2010, 299.1507, 197.0809, 127.0395 |
| S3:14(4,5,5) | 3 | 2.59 | C26H44O14 | 598.3075 | 598.3069 | 1.0 | 401.2178, 313.1668, 211.0951, 127.0396 |
| S3:15(5,5,5) | 3 | 3.00 | C27H46O14 | 612.3229 | 612.3226 | 0.5 | 415.2348, 313.1661, 211.0974, 127.0395 |
| S3:16(5,5,6) | 3 | 3.44 | C28H48O14 | 626.3392 | 626.3382 | 1.6 | 429.2489, 327.1810, 211.0946, 127.0373 |
| S3:16(4,4,8) | 3 | 3.67 | C28H48O14 | 626.3387 | 626.3382 | 0.8 | 429.2489, 285.1360, 197.0809, 127.0395 |
| S3:17(4,5,8) | 3 | 4.23 | C29H50O14 | 640.3543 | 640.3539 | 0.6 | 443.2710, 299.1541, 211.0974, 127.0395 |
| S3:17(4,4,9) | 3 | 4.43 | C29H50O14 | 640.3536 | 640.3539 | −0.5 | 443.2646, 285.1341, 197.0773, 127.0396 |
| S3:18(4,4,10)-1 | 1 | 5.45 | C30H52O14 | 654.3699 | 654.3695 | 0.6 | 457.2864, 285.1360, 197.0837, 127.0395 |
| S3:18(4,4,10)-2 | 1 | 5.71 | C30H52O14 | 654.3699 | 654.3695 | 0.6 | 457.2864, 285.1360, 197.0837, 127.0395 |
| S3:19(4,5,10)-1 | 1 | 6.24 | C31H54O14 | 668.3856 | 668.3852 | 0.6 | 471.3013, 299.1507, 211.0974, 127.0395 |
| S3:19(4,5,10)-2 | 1 | 6.52 | C31H54O14 | 668.3855 | 668.3852 | 0.5 | 471.3013, 299.1507, 211.1003, 127.0395 |
| S3:20(5,5,10) | 3 | 7.57 | C32H56O14 | 682.4011 | 682.4008 | 0.4 | 485.3103, 313.1661, 211.0974, 127.0395 |
| S3:20(4,4,12) | 3 | 8.20 | C32H56O14 | 682.4009 | 682.4008 | 0.2 | 485.3146, 285.1360, 197.0837, 127.0395 |
| S3:21(5,5,11) | 3 | 8.56 | C33H58O14 | 696.4166 | 696.4165 | 0.1 | 499.3259, 313.1661, 211.0974, 127.0395 |
| S3:21(4,5,12) | 3 | 9.14 | C33H58O14 | 696.4161 | 696.4165 | −0.6 | 499.3259, 299.1472, 211.0974, 127.0395 |
| S3:22(5,5,12) | 3 | 10.26 | C34H60O14 | 710.4319 | 710.4321 | −0.3 | 513.3442, 313.1661, 211.0974, 127.0395 |
| Triacylsucroses (cont’d) | |||||||
| S3:23(5,6,12) | 3 | 11.24 | C35H62O14 | 724.4471 | 724.4478 | −1.0 | 527.3616, 327.1810, 211.0946, 127.0373 |
| Triacylglucoses | |||||||
| G3:12(4,4,4) | 1 | 2.76; 2.84 | C18H30O9 | 408.2235 | 408.2228 | 1.7 | 373.1872, 285.1326, 197.0809, 127.0395 |
| G3:13(4,4,5) | 3 | 3.12; 3.24 | C19H32O9 | 422.2392 | 422.2385 | 1.7 | 387.2014, 299.1501, 197.0801, 127.0396 |
| G3:14(4,5,5) | 3 | 3.70; 3.83 | C20H34O9 | 436.2547 | 436.2541 | 1.4 | 401.2178, 299.1466, 211.0951, 127.0374 |
| G3:15(5,5,5) | 3 | 4.42; 4.58 | C21H36O9 | 450.2705 | 450.2698 | 1.6 | 415.2308, 313.1626, 211.0974, 127.0395 |
| G3:16(5,5,6) | 3 | 5.23; 5.41 | C22H38O9 | 464.2859 | 464.2854 | 1.1 | 429.2489, 327.1810, 211.0946, 127.0395 |
| G3:16(4,4,8)-1 | 3 | 5.56; 5.80 | C22H38O9 | 464.2861 | 464.2854 | 1.5 | 429.2529, 285.1360, 197.0809, 127.0395 |
| G3:16(4,4,8)-2 | 3 | 5.80; 6.04 | C22H38O9 | 464.2857 | 464.2854 | 0.7 | 429.2529, 285.1360, 197.0809, 127.0395 |
| G3:17(4,5,8)-1 | 3 | 6.46; 6.72 | C23H40O9 | 478.3011 | 478.3011 | 0.0 | 443.2628, 299.1472, 211.0946, 127.0395 |
| G3:17(4,5,8)-2 | 3 | 6.71; 6.99 | C23H40O9 | 478.3008 | 478.3011 | −0.6 | 443.2628, 299.1472, 197.0781, 127.0373 |
| G3:18(4,4,10)-1 | 1 | 8.03; 8.33 | C24H42O9 | 492.3168 | 492.3167 | 0.2 | 457.2779, 285.1326, 197.0809, 127.0395 |
| G3:18(4,4,10)-2 | 1 | 8.33; 8.64 | C24H42O9 | 492.3170 | 492.3167 | 0.6 | 457.2779, 285.1326, 197.0809, 127.0395 |
| G3:19(4,5,10)-1 | 1 | 9.05; 9.34 | C25H44O9 | 506.3328 | 506.3324 | 0.8 | 471.2970, 299.1472, 211.0974, 127.0395 |
| G3:19(4,5,10)-2 | 1 | 9.34; 9.66 | C25H44O9 | 506.3328 | 506.3324 | 0.8 | 471.2970, 299.1507, 211.0946, 127.0395 |
| G3:20(5,5,10) | 3 | 10.47; 10.72 | C26H46O9 | 520.3486 | 520.3480 | 1.2 | 485.3146, 313.1661, 211.0974, 127.0395 |
| G3:20(4,4,12) | 3 | 11.10; 11.42 | C26H46O9 | 520.3483 | 520.3480 | 0.6 | 485.3103, 285.1326, 197.0809, 127.0395 |
| G3:21(5,5,11) | 3 | 11.47; 11.75 | C27H48O9 | 534.3637 | 534.3637 | 0.0 | 499.3215, 313.1626, 211.0974, 127.0373 |
| G3:21(4,5,12) | 3 | 12.10; 12.40 | C27H48O9 | 534.3636 | 534.3637 | −0.2 | 499.3290, 299.1507, 211.0974, 127.0395 |
| Triacylglucoses (cont’d) | |||||||
| G3:22(5,5,12) | 3 | 13.10; 13.36 | C28H50O9 | 548.3794 | 548.3793 | 0.2 | 513.3442, 313.1661, 211.0974, 127.0395 |
| G3:23(5,6,12) | 3 | 14.02; 14.29 | C29H52O9 | 562.3938 | 562.3950 | −2.1 | 527.3471, 327.1848, 211.0960, 127.0372 |
| Tetraacylglucoses | |||||||
| G4:14(2,4,4,4) | 3 | 3.54; 3.79 | C20H32O10 | 450.2343 | 450.2334 | 2.0 | 415.1946, 327.1417, 239.0891, 197.0809, 127.0373 |
| G4:15(2,4,4,5) | 3 | 4.10; 4.45 | C21H34O10 | 464.2502 | 464.2491 | 2.4 | 429.2162, 341.1562, 239.0922, 197.0837, 127.0395 |
| Name | MSI | RT | Formula | m/zacc | m/zex | Δm (ppm) | Core | # Me |
|---|---|---|---|---|---|---|---|---|
| Flavonoid A | 3 | 3.04 | C17H14O6 | 315.0869 | 315.0863 | 1.9 | kaempferol | 2 |
| Flavonoid C | 3 | 3.17 | C18H16O7 | 345.0980 | 345.0969 | 3.2 | quercetin | 3 |
| Flavonoid D | 3 | 4.00 | C19H18O7 | 359.1137 | 359.1125 | 3.3 | quercetin | 4 |
| Flavonoid B | 3 | 4.90 | C18H16O6 | 329.1025 | 329.1020 | 1.5 | kaempferol | 3 |
| Accession | Total Acylsugars (µmol/g DW) | Tukey’s MST | % Acylglucose | Tukey’s MST | Range | Region |
|---|---|---|---|---|---|---|
| LA1809 | 136 ± 27 | B | 69 ± 4 | CDE | North | |
| LA2657 | 133 ± 28 | B | 56 ± 6 | EF | North | |
| LA2560 | 340 ± 64 | A | 65 ± 6 | CDE | North | |
| LA1773 | 237 ± 98 | AB | 66 ± 2 | CDE | North | |
| LA1376 | 261 ± 105 | AB | 70 ± 7 | CDE | North | |
| LA1523 | 158 ± 68 | B | 65 ± 8 | CDE | North | |
| LA1272 | 163 ± 109 | B | 58 ± 4 | DEF | North | |
| LA2719 | 218 ± 44 | AB | 70 ± 3 | BCDE | North | |
| LA1340 | 166 ± 39 | B | 80 ± 10 | ABC | South | Pisco |
| LA1693 | 193 ± 88 | AB | 77 ± 7 | ABCD | South | Pisco |
| LA1674 | 248 ± 83 | AB | 90 ± 4 | A | South | Pisco |
| LA1656 | 269 ± 54 | AB | 90 ± 4 | AB | South | Pisco |
| LA1946 | 257 ± 94 | AB | 82 ± 20 | ABC | South | Atico |
| LA1941 | 244 ± 66 | AB | 95 ± 2 | A | South | Atico |
| LA2963 | 183 ± 38 | B | 42 ± 6 | F | South | Atico |
| LA0716 | 238 ± 75 | AB | 95 ± 2 | A | South | Atico |
| n | % Correct | % Incorrect | % Unknown | |
|---|---|---|---|---|
| Full range (N = 96) | ||||
| North | 48 | 100 | 0 | 0 |
| South | 48 | 94 | 4 | 2 |
| South range (N = 48) | ||||
| Pisco | 24 | 67 | 4 | 29 |
| Atico | 24 | 77 | 4 | 19 |
| Atico region (N = 24) | ||||
| LA0716/LA1941/LA1946 | 18 | 97 | 0 | 3 |
| LA2963 | 6 | 100 | 0 | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lybrand, D.B.; Anthony, T.M.; Jones, A.D.; Last, R.L. An Integrated Analytical Approach Reveals Trichome Acylsugar Metabolite Diversity in the Wild Tomato Solanum pennellii. Metabolites 2020, 10, 401. https://doi.org/10.3390/metabo10100401
Lybrand DB, Anthony TM, Jones AD, Last RL. An Integrated Analytical Approach Reveals Trichome Acylsugar Metabolite Diversity in the Wild Tomato Solanum pennellii. Metabolites. 2020; 10(10):401. https://doi.org/10.3390/metabo10100401
Chicago/Turabian StyleLybrand, Daniel B., Thilani M. Anthony, A. Daniel Jones, and Robert L. Last. 2020. "An Integrated Analytical Approach Reveals Trichome Acylsugar Metabolite Diversity in the Wild Tomato Solanum pennellii" Metabolites 10, no. 10: 401. https://doi.org/10.3390/metabo10100401
APA StyleLybrand, D. B., Anthony, T. M., Jones, A. D., & Last, R. L. (2020). An Integrated Analytical Approach Reveals Trichome Acylsugar Metabolite Diversity in the Wild Tomato Solanum pennellii. Metabolites, 10(10), 401. https://doi.org/10.3390/metabo10100401





