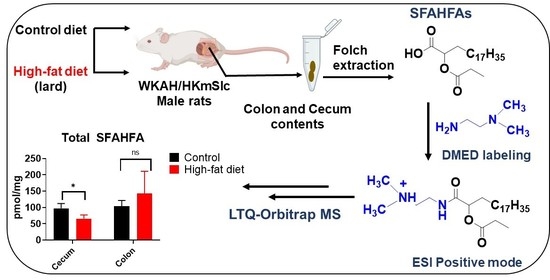
Chemical Labeling Assisted Detection and Identification of Short Chain Fatty Acid Esters of Hydroxy Fatty Acid in Rat Colon and Cecum Contents
Abstract
1. Introduction
2. Results and Discussion
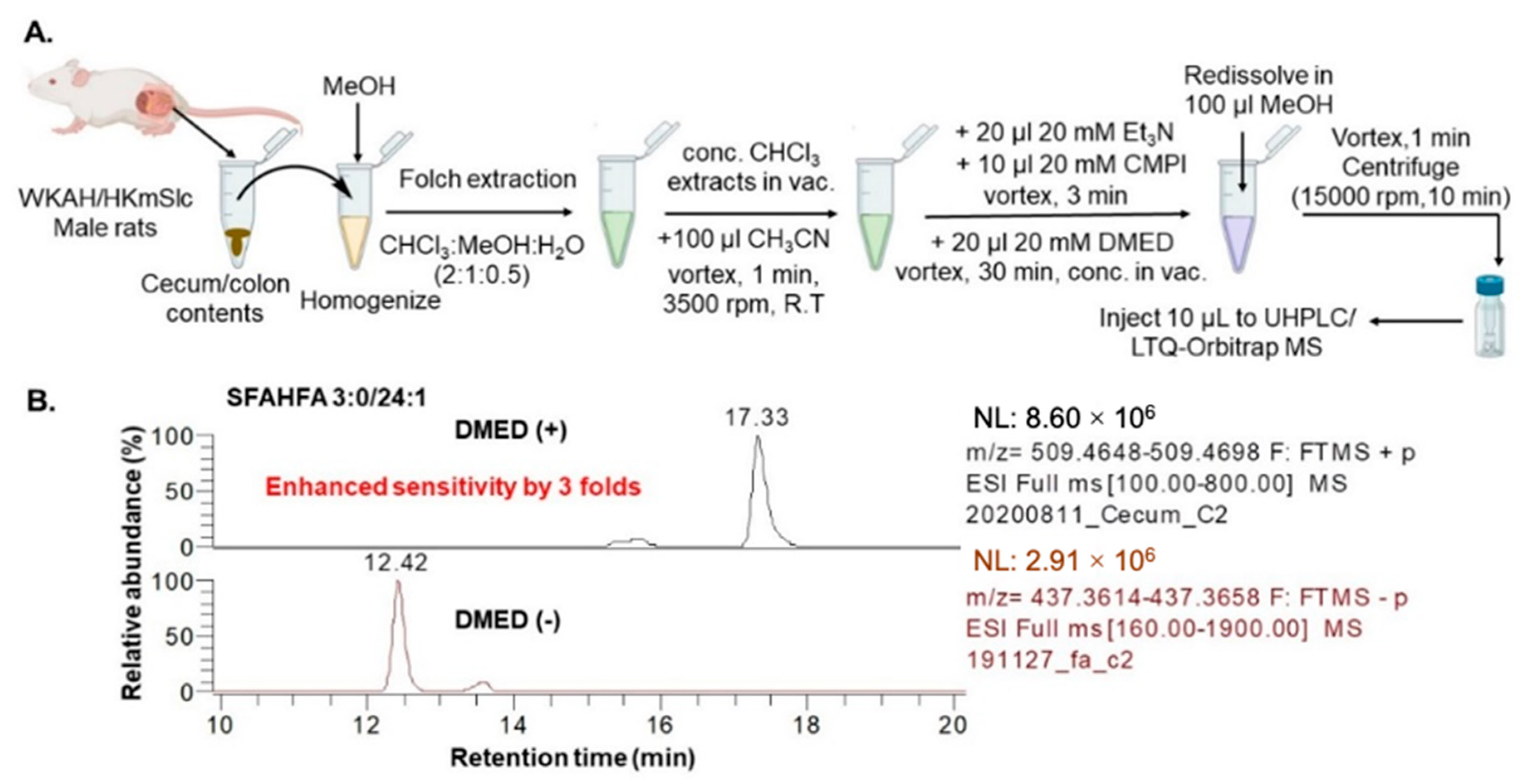
2.1. Chemical Labeling Assisted Detection and Identification of SFAHFAs
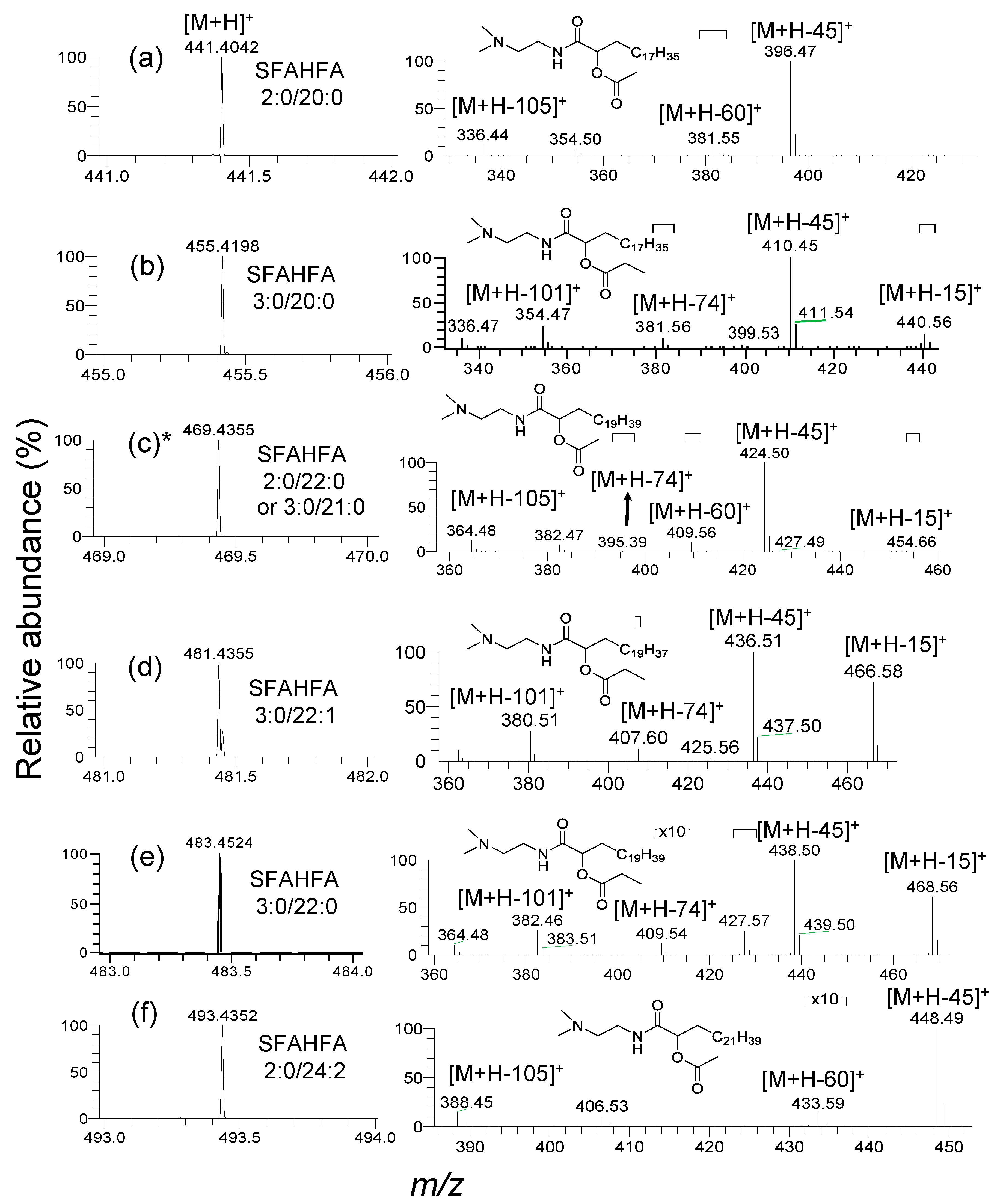
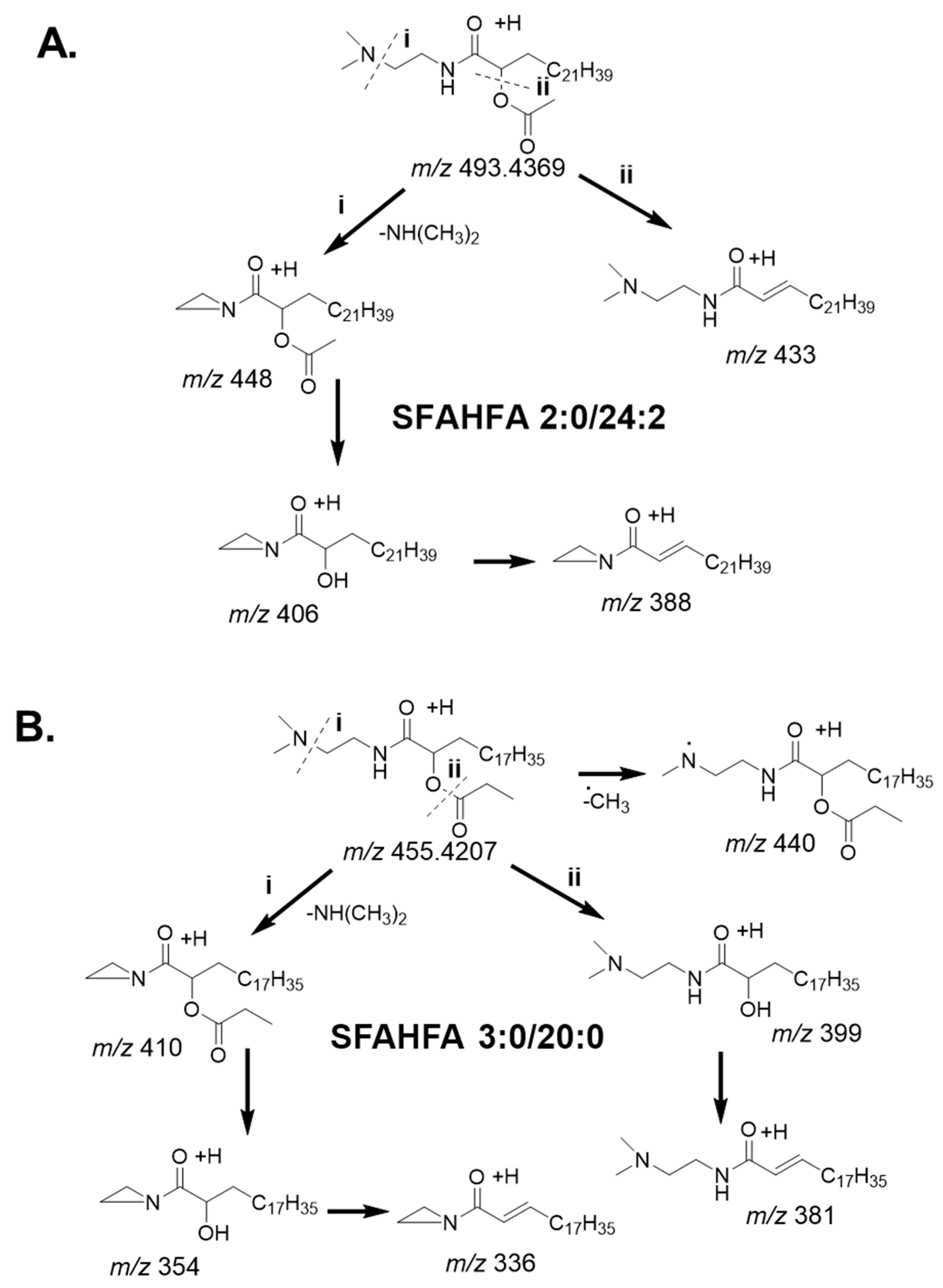
2.2. Annotation of DMED-labeled-SFAHFAs by MS/MS Analysis
2.3. Fragmentation Pattern of DMED-labeled SFAHFAs
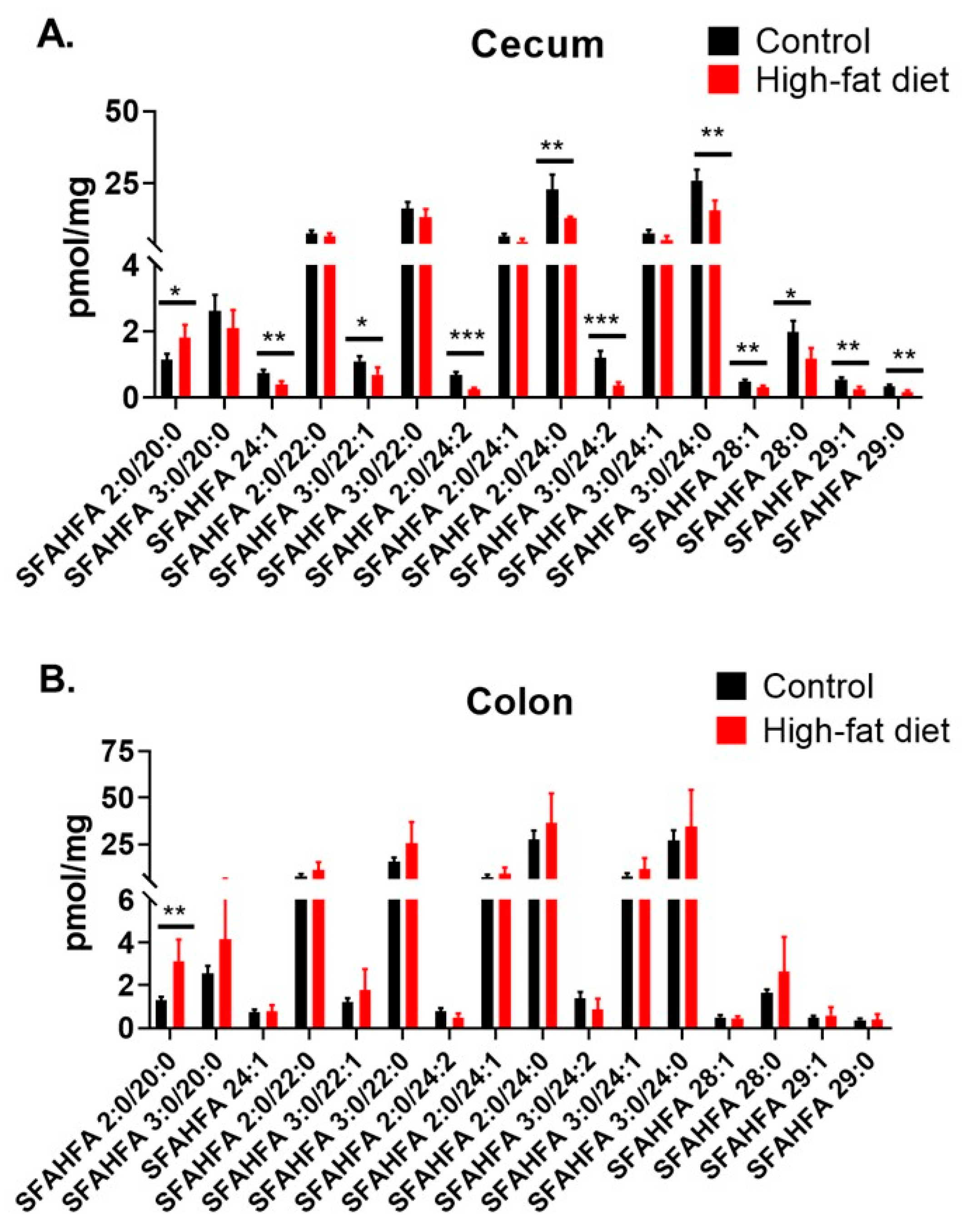
2.4. Altered Levels of SFAHFAs as Possible Biomarkers of Obesity
3. Materials and Methods
3.1. Chemicals
3.2. Animal Samples
3.3. Sample Preparation for LC-MS
3.4. LC-MS Analysis
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Boden, G. Obesity and Free Fatty Acids. Endocrinol. Metab. Clin. N. Am. 2008, 37, 635–646. [Google Scholar] [CrossRef] [PubMed]
- Halade, G.V.; Kain, V. Obesity and cardiometabolic defects in heart failure pathology. Compr. Physiol. 2017, 7, 1463–1477. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo, M.G.; Lytle, K.A.; Spooner, M.H.; Jump, D.B. A lipidomic analysis of docosahexaenoic acid (22:6, ω3) mediated attenuation of western diet induced nonalcoholic steatohepatitis in male ldlr -/- mice. Metabolites 2019, 9, 252. [Google Scholar] [CrossRef] [PubMed]
- Smedley, S.R.; Schroeder, F.C.; Weibel, D.B.; Meinwald, J.; Lafleur, K.A.; Renwick, J.A.; Rutowski, R.; Eisner, T. Mayolenes: Labile defensive lipids from the glandular hairs of a caterpillar (Pieris rapae). Proc. Natl. Acad. Sci. USA 2002, 99, 6822–6827. [Google Scholar] [CrossRef]
- Yore, M.M.; Syed, I.; Moraes-Vieira, P.M.; Zhang, T.; Herman, M.A.; Homan, E.A.; Patel, R.T.; Lee, J.; Chen, S.; Peroni, O.D.; et al. Discovery of a class of endogenous mammalian lipids with anti-diabetic and anti-inflammatory effects. Cell 2014, 159, 318–332. [Google Scholar] [CrossRef]
- Kuda, O.; Brezinova, M.; Rombaldova, M.; Slavikova, B.; Posta, M.; Beier, P.; Janovska, P.; Veleba, J.; Kopecky, J.; Kudova, E.; et al. Docosahexaenoic acid-derived fatty acid esters of hydroxy fatty acids (FAHFAS) with anti-inflammatory properties. Diabetes 2016, 65, 2580–2590. [Google Scholar] [CrossRef]
- Moraes-Vieira, P.M.; Saghatelian, A.; Kahn, B.B. GLUT4 expression in adipocytes regulates de novo lipogenesis and levels of a novel class of lipids with antidiabetic and anti-inflammatory effects. Diabetes 2016, 65, 1808–1815. [Google Scholar] [CrossRef]
- Lee, J.; Moraes-Vieira, P.M.; Castoldi, A.; Aryal, P.; Yee, E.U.; Vickers, C.; Parnas, O.; Donaldson, C.J.; Saghatelian, A.; Kahn, B.B. Branched fatty acid esters of hydroxy fatty acids (FAHFAs) protect against colitis by regulating gut innate and adaptive immune responses. J. Biol. Chem. 2016, 291, 22207–22217. [Google Scholar] [CrossRef]
- Gowda, S.G.B.; Fuda, H.; Tsukui, T.; Chiba, H.; Hui, S.P. Discovery of eicosapentaenoic acid esters of hydroxy fatty acids as potent nrf2 activators. Antioxidants 2020, 9, 397. [Google Scholar] [CrossRef]
- Zhu, Q.F.; Yan, J.W.; Gao, Y.; Zhang, J.W.; Yuan, B.F.; Feng, Y.Q. Highly sensitive determination of fatty acid esters of hydroxyl fatty acids by liquid chromatography-mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2017, 1061, 34–40. [Google Scholar] [CrossRef]
- Zhu, Q.F.; Yan, J.W.; Zhang, T.Y.; Xiao, H.M.; Feng, Y.Q. Comprehensive Screening and Identification of Fatty Acid Esters of Hydroxy Fatty Acids in Plant Tissues by Chemical Isotope Labeling-Assisted Liquid Chromatography-Mass Spectrometry. Anal. Chem. 2018, 90, 10056–10063. [Google Scholar] [CrossRef] [PubMed]
- Gowda, S.G.B.; Liang, C.; Gowda, D.; Hou, F.; Kawakami, K.; Fukiya, S.; Yokota, A.; Chiba, H.; Hui, S.P. Identification of short-chain fatty acid esters of hydroxy fatty acids (SFAHFAs) in a murine model by nontargeted analysis using ultra-high-performance liquid chromatography/linear ion trap quadrupole-Orbitrap mass spectrometry. Rapid Commun. Mass Spectrom. 2020, 34, e8831. [Google Scholar] [CrossRef] [PubMed]
- Kokotou, M.G. Analytical methods for the determination of fatty acid esters of hydroxy fatty acids (FAHFAs) in biological samples, plants and foods. Biomolecules 2020, 10, 1092. [Google Scholar] [CrossRef] [PubMed]
- Kolar, M.J.; Nelson, A.T.; Chang, T.; Ertunc, M.E.; Christy, M.P.; Ohlsson, L.; Härröd, M.; Kahn, B.B.; Siegel, D.; Saghatelian, A. Faster Protocol for Endogenous Fatty Acid Esters of Hydroxy Fatty Acid (FAHFA) Measurements. Anal. Chem. 2018, 90, 5358–5365. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Wang, M.; Duan, Q.; Han, X. Sensitive analysis of fatty acid esters of hydroxy fatty acids in biological lipid extracts by shotgun lipidomics after one-step derivatization. Anal. Chim. Acta 2020, 1105, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Kind, T.; Zhu, Q.F.; Wang, Y.; Yan, J.W.; Fiehn, O.; Feng, Y.Q. In-Silico-Generated Library for Sensitive Detection of 2-Dimethylaminoethylamine Derivatized FAHFA Lipids Using High-Resolution Tandem Mass Spectrometry. Anal. Chem. 2020, 92, 5960–5968. [Google Scholar] [CrossRef] [PubMed]
- Pfeuffer, M.; Jaudszus, A. Pentadecanoic and Heptadecanoic Acids: Multifaceted Odd-Chain Fatty Acids. Adv. Nutr. Int. Rev. J. 2016, 15, 730–734. [Google Scholar] [CrossRef]
- Wu, J.L.; Leung, E.L.H.; Zhou, H.; Liu, L.; Li, N. Metabolite analysis of toosendanin by an ultra-high performance liquid chromatography-quadrupole-time of flight mass spectrometry technique. Molecules 2013, 18, 12144–12153. [Google Scholar] [CrossRef]
- Li, J.; Xu, J.; Zhang, R.; Hao, Y.; He, J.; Chen, Y.; Jiao, G.; Abliz, Z. Strategy for Global Profiling and Identification of 2- and 3-Hydroxy Fatty Acids in Plasma by UPLC–MS/MS. Anal. Chem. 2020, 92, 5143–5151. [Google Scholar] [CrossRef]
- Hiraki, T.; Kubota, M.; Hirano, T.; Niwa, H.; Ohashi, M. FAB-CID Mass Spectrometry of Imidazolone Derivatives Related to the Chromophore of Aequorea Green Fluorescent Protein. J. Mass Spectrom. Soc. Jpn. 1998, 46, 37–43. [Google Scholar] [CrossRef][Green Version]
- Hirata, S.I.; Kunisawa, J. Gut microbiome, metabolome, and allergic diseases. Allergol. Int. 2017, 66, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.Y.; Leelasinjaroen, P.; Gillilland, M.G.; Zhou, H.; Owyang, C. Su2125 High fat diet induced activation of autophagy in paneth cells is responsible for gut dysbiosis and intestinal inflammation in rats. Gastroenterology 2013, 144, S564. [Google Scholar] [CrossRef]
- Araújo, J.R.; Tomas, J.; Brenner, C.; Sansonetti, P.J. Impact of high-fat diet on the intestinal microbiota and small intestinal physiology before and after the onset of obesity. Biochimie 2017, 141, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Li, J.; Tang, R.; Zhang, G.; Zeng, H.; Wood, R.J.; Liu, Z. High Fat Diet Alters Gut Microbiota and the Expression of Paneth Cell-Antimicrobial Peptides Preceding Changes of Circulating Inflammatory Cytokines. Mediat. Inflamm. 2017, 2017, 9474896. [Google Scholar] [CrossRef] [PubMed]
- Jakobsdottir, G.; Xu, J.; Molin, G.; Ahrné, S.; Nyman, M. High-fat diet reduces the formation of butyrate, but increases succinate, inflammation, liver fat and cholesterol in rats, while dietary fibre counteracts these effects. PLoS ONE 2013, 8, e80476. [Google Scholar] [CrossRef]
- Brezinova, M.; Kuda, O.; Hansikova, J.; Rombaldova, M.; Balas, L.; Bardova, K.; Durand, T.; Rossmeisl, M.; Cerna, M.; Stranak, Z.; et al. Levels of palmitic acid ester of hydroxystearic acid (PAHSA) are reduced in the breast milk of obese mothers. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2018, 1863, 126–131. [Google Scholar] [CrossRef]
- Mallett, A.K.; Rowland, I.R.; Wise, A. Dietary fat and cecal microbial activity in the rat. Nutr. Cancer 2009, 6, 86–91. [Google Scholar] [CrossRef]
- Gowda, S.G.B.; Gao, Z.-J.; Chen, Z.; Abe, T.; Hori, S.; Fukiya, S.; Ishizuka, S.; Yokota, A.; Chiba, H.; Hui, S.-P. Untargeted lipidomic analysis of plasma from high fat diet-induced obese rats using UHPLC- linear trap quadrupole -Orbitrap MS. Anal. Sci. 2020, 36, 821–828. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
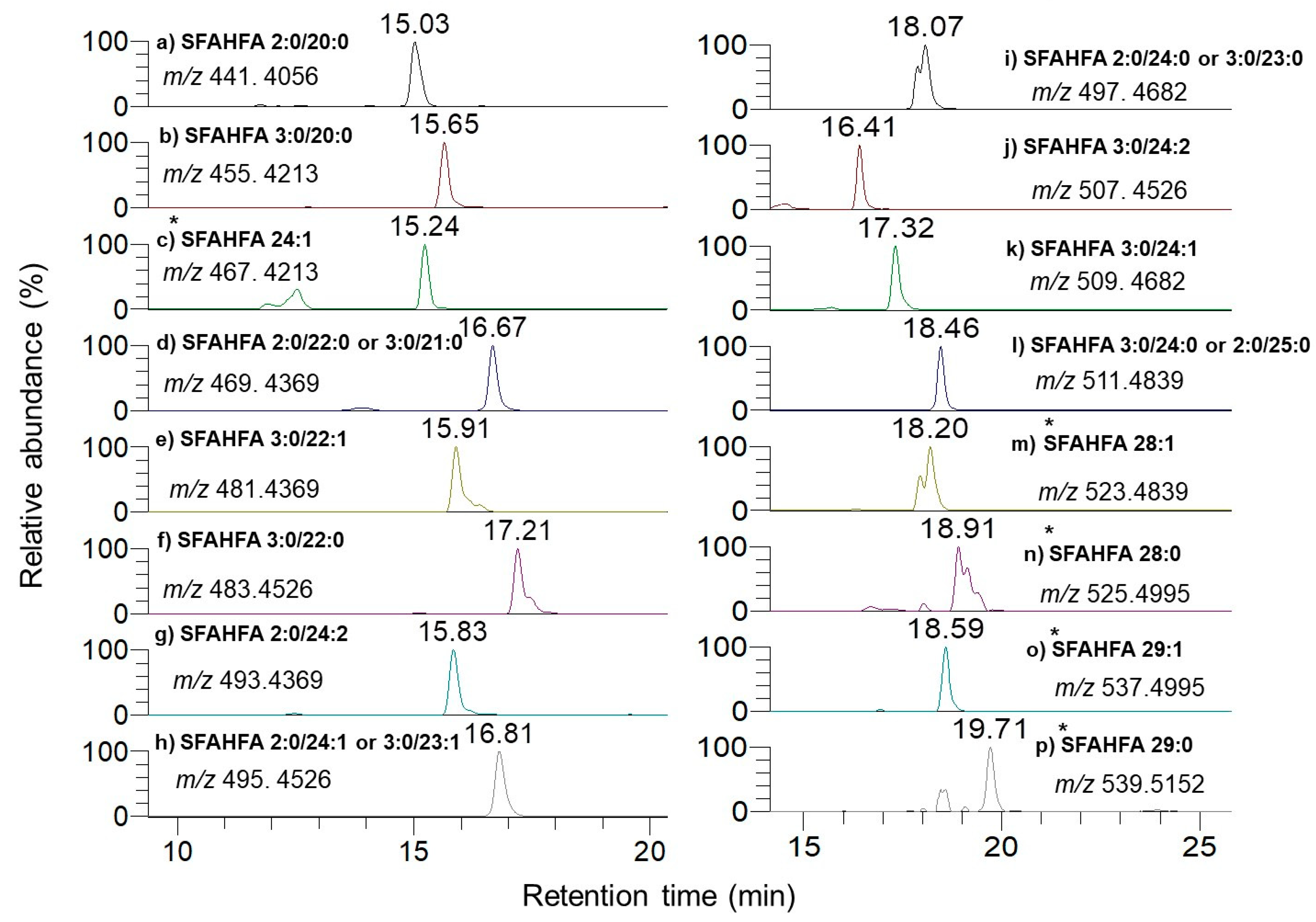

| SI No | Type of SFAHFA | Theoretical m/z | Experimental m/z | Mass Error ppm | Identified |
|---|---|---|---|---|---|
| 1 | SFAHFA 2:0/20:0 | 441.4056 | 441.4042 | −3.1 | [12] |
| 2 | SFAHFA 3:0/20:0 | 455.4213 | 455.4198 | −3.2 | This study |
| 3 | SFAHFA 24:1 | 467.4213 | 467.4197 | −3.4 | [12] |
| 4 | SFAHFA 2:0/22:0 or 3:0/21:0 | 469.4369 | 469.4355 | −2.9 | [12] |
| 5 | SFAHFA 3:0/22:1 | 481.4369 | 481.4355 | −2.9 | This study |
| 6 | SFAHFA 3:0/22:0 | 483.4526 | 483.4524 | −0.4 | [12] |
| 7 | SFAHFA 2:0/24:2 | 493.4369 | 493.4352 | −3.4 | This study |
| 8 | SFAHFA 2:0/24:1 or 3:0/23:1 | 495.4526 | 495.4507 | −3.8 | [12] |
| 9 | SFAHFA 2:0/24:0 or 3:0/23:0 | 497.4682 | 497.4677 | −1.0 | [12] |
| 10 | SFAHFA 3:0/24:2 | 507.4526 | 507.4514 | −2.3 | [12] |
| 11 | SFAHFA 3:0/24:1 | 509.4682 | 509.4673 | −1.7 | [12] |
| 12 | SFAHFA 3:0/24:0 or 2:0/25:0 | 511.4839 | 511.4833 | −1.1 | [12] |
| 13 | SFAHFA 28:1 | 523.4839 | 523.4830 | −1.7 | This study |
| 14 | SFAHFA 28:0 | 525.4995 | 525.4989 | −1.1 | [12] |
| 15 | SFAHFA 29:1 | 537.4995 | 537.4990 | −0.9 | This study |
| 16 | SFAHFA 29:0 | 539.5152 | 539.5150 | −0.4 | This study |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
B. Gowda, S.G.; Gowda, D.; Liang, C.; Li, Y.; Kawakami, K.; Fukiya, S.; Yokota, A.; Chiba, H.; Hui, S.-P. Chemical Labeling Assisted Detection and Identification of Short Chain Fatty Acid Esters of Hydroxy Fatty Acid in Rat Colon and Cecum Contents. Metabolites 2020, 10, 398. https://doi.org/10.3390/metabo10100398
B. Gowda SG, Gowda D, Liang C, Li Y, Kawakami K, Fukiya S, Yokota A, Chiba H, Hui S-P. Chemical Labeling Assisted Detection and Identification of Short Chain Fatty Acid Esters of Hydroxy Fatty Acid in Rat Colon and Cecum Contents. Metabolites. 2020; 10(10):398. https://doi.org/10.3390/metabo10100398
Chicago/Turabian StyleB. Gowda, Siddabasave Gowda, Divyavani Gowda, Chongsheng Liang, Yonghan Li, Kentaro Kawakami, Satoru Fukiya, Atsushi Yokota, Hitoshi Chiba, and Shu-Ping Hui. 2020. "Chemical Labeling Assisted Detection and Identification of Short Chain Fatty Acid Esters of Hydroxy Fatty Acid in Rat Colon and Cecum Contents" Metabolites 10, no. 10: 398. https://doi.org/10.3390/metabo10100398
APA StyleB. Gowda, S. G., Gowda, D., Liang, C., Li, Y., Kawakami, K., Fukiya, S., Yokota, A., Chiba, H., & Hui, S.-P. (2020). Chemical Labeling Assisted Detection and Identification of Short Chain Fatty Acid Esters of Hydroxy Fatty Acid in Rat Colon and Cecum Contents. Metabolites, 10(10), 398. https://doi.org/10.3390/metabo10100398