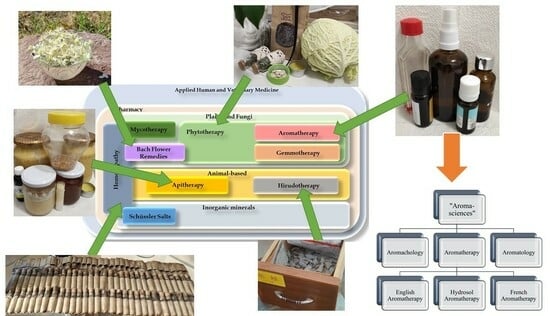
Complementary Practices in Pharmacy and Their Relation to Glaucoma—Classification, Definitions, and Limitations
Abstract
1. Introduction
2. Challenges in Glaucoma
3. Materials and Methods
4. Phytotherapy
4.1. Definitions and Description
4.2. Limitations
4.3. Application in Glaucoma
5. Aromatherapy
5.1. Definitions and Description
5.2. Limitations
5.3. Application in Glaucoma
6. Gemmotherapy
6.1. Definitions and Description
6.2. Limitations
6.3. Application in Glaucoma
7. Bach Flower Therapy
7.1. Definitions and Description
7.2. Limitations
7.3. Application in Glaucoma
8. Mycotherapy
8.1. Definitions and Description
8.2. Limitations
8.3. Application in Glaucoma
9. Apitherapy
9.1. Definitions and Descriptions
9.2. Limitations
9.3. Application in Glaucoma
10. Hirudotherapy
10.1. Definitions and Description
10.2. Limitations
10.3. Application in Glaucoma
11. Systematic Overview and Future Prospects
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AMD | Age-Related Macular Degeneration |
| BFRs | Bach Flower Remedies |
| EBM | Evidence-Based Medicine |
| EMA | European Medicines Agency |
| ESCOP | European Scientific Cooperative on Phytotherapy |
| ET-1 | Endothelin-1 |
| IOP | Intraocular Pressure |
| MMP-9 | Matrix Metalloproteinase-9 |
| NF-κB | Nuclear Factor Kappa B |
| NTG | Normotension Glaucoma |
| PACG | Primary Angle Closure Glaucoma |
| POAG | Primary Open-Angle Glaucoma |
| RPE | Retinal Pigment Epithelium |
| TCM | Traditional Chinese Medicine |
| WHO | World Health Organization |
References
- WHO. WHO Global Report on Traditional and Complementary Medicine; World Health Organization: Geneva, Switzerland, 2019; ISBN 978-92-4-151543-6. [Google Scholar]
- de Carvalho Resende, M.M.; de Carvalho Costa, F.E.; Gardona, R.G.B.; Araújo, R.G.; Mundim, F.G.L.; de Carvalho Costa, M.J. Preventive Use of Bach Flower Rescue Remedy in the Control of Risk Factors for Cardiovascular Disease in Rats. Complement. Ther. Med. 2014, 22, 719–723. [Google Scholar] [CrossRef] [PubMed]
- Sen, T.; Samanta, S.K. Medicinal Plants, Human Health and Biodiversity: A Broad Review. Adv. Biochem. Eng. Biotechnol. 2014, 147, 59–110. [Google Scholar] [CrossRef]
- Kovács-Valasek, A.; Rák, T.; Pöstyéni, E.; Csutak, A.; Gábriel, R. Three Major Causes of Metabolic Retinal Degenerations and Three Ways to Avoid Them. Int. J. Mol. Sci. 2023, 24, 8728. [Google Scholar] [CrossRef] [PubMed]
- Colalto, C. What Phytotherapy Needs: Evidence-Based Guidelines for Better Clinical Practice. Phyther. Res. 2018, 32, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Donno, D.; Mellano, M.G.; Cerutti, A.K.; Beccaro, G.L. Biomolecules and Natural Medicine Preparations: Analysis of New Sources of Bioactive Compounds from Ribes and Rubus spp. Buds. Pharmaceuticals 2016, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Wan, M.J.; Daniel, S.; Kassam, F.; Mutti, G.; Butty, Z.; Kasner, O.; Trope, G.E.; Buys, Y.M. Survey of Complementary and Alternative Medicine Use in Glaucoma Patients. J. Glaucoma 2010, 21, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Trumbeckaite, S.; Dauksiene, J.; Bernatoniene, J.; Janulis, V. Knowledge, Attitudes, and Usage of Apitherapy for Disease Prevention and Treatment among Undergraduate Pharmacy Students in Lithuania. Evid. Based. Complement. Alternat. Med. 2015, 2015, 172502. [Google Scholar] [CrossRef]
- Ige, M.; Liu, J. Focus: Plant-Based Medicine and Pharmacology: Herbal Medicines in Glaucoma Treatment. Yale J. Biol. Med. 2020, 93, 347. [Google Scholar]
- Rák, T.; Kovács-Valasek, A.; Pöstyéni, E.; Csutak, A.; Gábriel, R. Complementary Approaches to Retinal Health Focusing on Diabetic Retinopathy. Cells 2023, 12, 2699. [Google Scholar] [CrossRef]
- Hassen, G.; Belete, G.; Carrera, K.G.; Iriowen, R.O.; Araya, H.; Alemu, T.; Solomon, N.; Bam, D.S.; Nicola, S.M.; Araya, M.E.; et al. Clinical Implications of Herbal Supplements in Conventional Medical Practice: A US Perspective. Cureus 2022, 14, e26893. [Google Scholar] [CrossRef]
- West, A.L.; Oren, G.A.; Moroi, S.E. Evidence for the Use of Nutritional Supplements and Herbal Medicines in Common Eye Diseases. Am. J. Ophthalmol. 2006, 141, 157–166. [Google Scholar] [CrossRef]
- Sim, R.H.; Sirasanagandla, S.R.; Das, S.; Teoh, S.L. Treatment of Glaucoma with Natural Products and Their Mechanism of Action: An Update. Nutrients 2022, 14, 534. [Google Scholar] [CrossRef] [PubMed]
- Petkova, V.; Hadzhieva, B.; Nedialkov, P. Phytotherapeutic Approaches to Treatment and Prophylaxis in Pediatric Practice. Pharmacia 2019, 66, 115–119. [Google Scholar] [CrossRef]
- Falzon, C.C.; Balabanova, A. Phytotherapy: An Introduction to Herbal Medicine. Prim. Care-Clin. Off. Pract. 2017, 44, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Reis, D.; Jones, T. Aromatherapy: Using Essential Oils as a Supportive Therapy. Clin. J. Oncol. Nurs. 2017, 21, 16–19. [Google Scholar] [CrossRef] [PubMed]
- Fazlollahpour-Rokni, F.; Shorofi, S.A.; Mousavinasab, N.; Ghafari, R.; Esmaeili, R. The Effect of Inhalation Aromatherapy with Rose Essential Oil on the Anxiety of Patients Undergoing Coronary Artery Bypass Graft Surgery. Complement. Ther. Clin. Pract. 2019, 34, 201–207. [Google Scholar] [CrossRef]
- Boehm, K.; Büssing, A.; Ostermann, T. Aromatherapy as an Adjuvant Treatment in Cancer Care—A Descriptive Systematic Review. Afr. J. Tradit. Complement. Altern. Med. 2012, 9, 503–518. [Google Scholar] [CrossRef] [PubMed]
- Farrar, A.J.; Farrar, F.C. Clinical Aromatherapy. Nurs. Clin. N. Am. 2020, 55, 489–504. [Google Scholar] [CrossRef] [PubMed]
- Posadzki, P.; Alotaibi, A.; Ernst, E. Adverse Effects of Aromatherapy: A Systematic Review of Case Reports and Case Series. Int. J. Risk Saf. Med. 2012, 24, 147–161. [Google Scholar] [CrossRef]
- Wang, C.X.; Chen, S.L. Aromachology and Its Application in the Textile Field. Fibres Text. East. Eur. 2005, 13, 41–44. [Google Scholar]
- Georgiev, V.; Ananga, A.; Dincheva, I.; Badjakov, I.; Gochev, V.; Tsolova, V. Chemical Composition, in Vitro Antioxidant Potential, and Antimicrobial Activities of Essential Oils and Hydrosols from Native American Muscadine Grapes. Molecules 2019, 24, 3355. [Google Scholar] [CrossRef]
- Edris, A.E. Identification and Absolute Quantification of the Major Water-Soluble Aroma Components Isolated from the Hydrosols of Some Aromatic Plants. J. Essent. Oil-Bearing Plants 2009, 12, 155–161. [Google Scholar] [CrossRef]
- Santarsiero, A.; Onzo, A.; Pascale, R.; Acquavia, M.A.; Coviello, M.; Convertini, P.; Todisco, S.; Marsico, M.; Pifano, C.; Iannece, P.; et al. Pistacia Lentiscus Hydrosol: Untargeted Metabolomic Analysis and Anti-Inflammatory Activity Mediated by NF- κ B and the Citrate Pathway. Oxid. Med. Cell. Longev. 2020, 2020, 4264815. [Google Scholar] [CrossRef] [PubMed]
- Ali, B.; Al-Wabel, N.A.; Shams, S.; Ahamad, A.; Khan, S.A.; Anwar, F. Essential Oils Used in Aromatherapy: A Systemic Review. Asian Pac. J. Trop. Biomed. 2015, 5, 601–611. [Google Scholar] [CrossRef]
- Liu, J.; Cao, L.; Yang, G.; Zhou, R. Effects of Non-Pharmacological Interventions on Anxiety, Depression, and Sleep Quality in Patients with Postoperative Glaucoma: A Protocol for Systematic Review and Network Meta-Analysis. Medicine 2021, 100, E27090. [Google Scholar] [CrossRef] [PubMed]
- Sabel, B.A.; Wang, J.; Cárdenas-Morales, L.; Faiq, M.; Heim, C. Mental Stress as Consequence and Cause of Vision Loss: The Dawn of Psychosomatic Ophthalmology for Preventive and Personalized Medicine. EPMA J. 2018, 9, 133. [Google Scholar] [CrossRef]
- Shin, D.Y.; Jung, K.I.; Park, H.Y.L.; Park, C.K. The Effect of Anxiety and Depression on Progression of Glaucoma. Sci. Rep. 2021, 11, 1769. [Google Scholar] [CrossRef]
- Southwell, I. Backhousia Citriodora F. Muell. (Lemon Myrtle), an Unrivalled Source of Citral. Foods 2021, 10, 1596. [Google Scholar] [CrossRef]
- Lis-Balchin, M. The Safety Issue in Aromatherapy. Aromather. Sci. Guid. Healthc. Prof. 2005, 17, 86–87. [Google Scholar]
- Rodger, F.C.; Grover, A.D.; Saiduzzafar, H. The Effect of Citral on Intraocular Dynamics in Monkeys. AMA. Arch. Ophthalmol. 1960, 63, 77–83. [Google Scholar] [CrossRef]
- Saiduzzafar, H. The Effect of Citral on Variations in the Aqueous Outflow Facility of Rabbits. Indian J. Ophthalmol. 1959, 7, 39. [Google Scholar]
- Sarkova, A.; Sarek, M. EAV and Gemmotherapy—Medicine for the next Millennium? (Technique as a Means to Link Eastern and Western Medicine). In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology, Shanghai, China, 17–18 January 2006; Volume 7, pp. 4943–4946. [Google Scholar]
- Cao, L.; Park, Y.; Lee, S.; Kim, D.O. Extraction, Identification, and Health Benefits of Anthocyanins in Blackcurrants (Ribes nigrum L.). Appl. Sci. 2021, 11, 1863. [Google Scholar] [CrossRef]
- Ernst, E. Bach Flower Remedies: A Systematic Review of Randomised Clinical Trials. Swiss Med. Wkly. 2010, 140, 29–31. [Google Scholar] [CrossRef]
- Thaler, K.; Kaminski, A.; Chapman, A.; Langley, T.; Gartlehner, G. Bach Flower Remedies for Psychological Problems and Pain: A Systematic Review. BMC Complement. Altern. Med. 2009, 9, 16. [Google Scholar] [CrossRef]
- Rivas-Suárez, S.R.; Águila-Vázquez, J.; Suárez-Rodríguez, B.; Vázquez-León, L.; Casanova-Giral, M.; Morales-Morales, R.; Rodríguez-Martín, B.C. Exploring the Effectiveness of External Use of Bach Flower Remedies on Carpal Tunnel Syndrome: A Pilot Study. J. Evid.-Based Complement. Altern. Med. 2017, 22, 18–24. [Google Scholar] [CrossRef]
- Howard, J. Do Bach Flower Remedies Have a Role to Play in Pain Control? A Critical Analysis Investigating Therapeutic Value beyond the Placebo Effect, and the Potential of Bach Flower Remedies as a Psychological Method of Pain Relief. Complement. Ther. Clin. Pract. 2007, 13, 174–183. [Google Scholar] [CrossRef]
- de Oliveira, F.N.; Okuyama, C.E.; de Souza Marquez, A.; Fernandes, K.B.; Dalaqua, P.V.; do Carmo, A.C.; Branco-Barreiro, F.C. Use of Bach Flower Remedies in Children and Adolescents with Attention Deficit Hyperactivity Disorder: Systematic Review and Meta-Analysis. Adv. Integr. Med. 2023, 10, 93–100. [Google Scholar] [CrossRef]
- Yadav, S.K.; Ir, R.; Jeewon, R.; Doble, M.; Hyde, K.D.; Kaliappan, I.; Jeyaraman, R.; Reddi, R.N.; Krishnan, J.; Li, M.; et al. A Mechanistic Review on Medicinal Mushrooms-Derived Bioactive Compounds: Potential Mycotherapy Candidates for Alleviating Neurological Disorders. Planta Med. 2020, 86, 1161–1175. [Google Scholar] [CrossRef]
- Zmitrovich, I.V.; Belova, N.V.; Balandaykin, M.E.; Bondartseva, M.A.; Wasser, S.P. Cancer without Pharmacological Illusions and a Niche for Mycotherapy (Review). Int. J. Med. Mushrooms 2019, 21, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Rossi, P.; Difrancia, R.; Quagliariello, V.; Savino, E.; Tralongo, P.; Randazzo, C.L.; Berretta, M. B-Glucans from Grifola Frondosa and Ganoderma Lucidum in Breast Cancer: An Example of Complementary and Integrative Medicine. Oncotarget 2018, 9, 24837–24856. [Google Scholar] [CrossRef] [PubMed]
- Venturella, G.; Ferraro, V.; Cirlincione, F.; Gargano, M.L. Medicinal Mushrooms: Bioactive Compounds, Use, and Clinical Trials. Int. J. Mol. Sci. 2021, 22, 634. [Google Scholar] [CrossRef]
- Amen, Y.; Zhu, Q.; Tran, H.B.; Afifi, M.S.; Halim, A.F.; Ashour, A.; Shimizu, K. Partial Contribution of Rho-Kinase Inhibition to the Bioactivity of Ganoderma Lingzhi and Its Isolated Compounds: Insights on Discovery of Natural Rho-Kinase Inhibitors. J. Nat. Med. 2017, 71, 380–388. [Google Scholar] [CrossRef]
- Lin, F.L.; Cheng, Y.W.; Yu, M.; Ho, J.D.; Kuo, Y.C.; Chiou, G.C.Y.; Chang, H.M.; Lee, T.H.; Hsiao, G. The Fungus-Derived Retinoprotectant Theissenolactone C Improves Glaucoma-like Injury Mediated by MMP-9 Inhibition. Phytomedicine 2019, 56, 207–214. [Google Scholar] [CrossRef]
- Hellner, M.; Winter, D.; Von Georgi, R.; Münstedt, K. Apitherapy: Usage and Experience in German Beekeepers. Evid.-Based Complement. Altern. Med. 2008, 5, 475–479. [Google Scholar] [CrossRef]
- Münstedt, K.; Funk, D.; Riepen, T.; Berkes, E.; Hübner, J. Acceptance of Apitherapeutic Methods in Patients Consulting General Physicians or Gynaecologists. Complement. Ther. Clin. Pract. 2019, 35, 154–157. [Google Scholar] [CrossRef]
- Kavurmaci, M.; Tan, M. Determination of Knowledge and Attitudes of Nurses about Apitherapy. Complement. Ther. Clin. Pract. 2019, 36, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Fratellone, P.M.; Tsimis, F.; Fratellone, G. Apitherapy Products for Medicinal Use. J. Altern. Complement. Med. 2016, 22, 1020–1022. [Google Scholar] [CrossRef] [PubMed]
- Inanc, B.B. The Quality Specialities in Turkiye’s Honies for Apitherapy. J. Pharmacopunct. 2021, 23, 194–200. [Google Scholar]
- Cornara, L.; Biagi, M.; Xiao, J.; Burlando, B. Therapeutic Properties of Bioactive Compounds from Different Honeybee Products. Front. Pharmacol. 2017, 8, 412. [Google Scholar] [CrossRef] [PubMed]
- Machado, A.M.; Miguel, M.G.; Vilas-Boas, M.; Figueiredo, A.C. Honey Volatiles as a Fingerprint for Botanical Origin—A Review on Their Occurrence on Monofloral Honeys. Molecules 2020, 25, 374. [Google Scholar] [CrossRef] [PubMed]
- Chien, H.Y.; Shih, A.T.; Yang, B.S.; Hsiao, V.K.S. Fast Honey Classification Using Infrared Spectrum and Machine Learning. Math. Biosci. Eng. 2019, 16, 6874–6891. [Google Scholar] [CrossRef]
- Nikolić-Pavljašević, S.; Redžepagić-Dervišević, E. Diseases of the eye and the therapeutic effect of honey. Hrana Zdr. Boles. Znan. časopis za Nutr. i dijetetiku 2016, 5, 27–31. [Google Scholar]
- Sforcin, J.M. Biological Properties and Therapeutic Applications of Propolis. Phyther. Res. 2016, 30, 894–905. [Google Scholar] [CrossRef]
- Denisow, B.; Denisow-Pietrzyk, M. Biological and Therapeutic Properties of Bee Pollen: A Review. J. Sci. Food Agric. 2016, 96, 4303–4309. [Google Scholar] [CrossRef] [PubMed]
- Pasupuleti, V.R.; Sammugam, L.; Ramesh, N.; Gan, S.H. Honey, Propolis, and Royal Jelly: A Comprehensive Review of Their Biological Actions and Health Benefits. Oxid. Med. Cell. Longev. 2017, 2017, 1259510. [Google Scholar] [CrossRef] [PubMed]
- Yucel, B.; Acikgoz, Z.; Bayraktar, H.; Seremet, C. The Effects of Apilarnil (Drone Bee Larvae) Administration on Growth Performance and Secondary Sex Characteristics of Male Broilers. J. Anim. Vet. Adv. 2011, 10, 2263–2266. [Google Scholar]
- Doğanyiğit, Z.; Okan, A.; Kaymak, E.; Pandır, D.; Silici, S. Investigation of Protective Effects of Apilarnil against Lipopolysaccharide Induced Liver Injury in Rats via TLR 4/HMGB-1/NF-ΚB Pathway. Biomed. Pharmacother. 2020, 125, 109967. [Google Scholar] [CrossRef] [PubMed]
- Nyman, G.S.A.; Tang, M.; Inerot, A.; Osmancevic, A.; Malmberg, P.; Hagvall, L. Contact Allergy to Beeswax and Propolis among Patients with Cheilitis or Facial Dermatitis. Contact Dermat. 2019, 81, 110–116. [Google Scholar] [CrossRef]
- Wehbe, R.; Frangieh, J.; Rima, M.; Obeid, D.; Sabatier, J.M.; Fajloun, Z. Bee Venom: Overview of Main Compounds and Bioactivities for Therapeutic Interests. Molecules 2019, 24, 2997. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Liu, Y.; Ye, Y.; Wang, X.R.; Lin, L.T.; Xiao, L.Y.; Zhou, P.; Shi, G.X.; Liu, C.Z. Bee Venom Therapy: Potential Mechanisms and Therapeutic Applications. Toxicon 2018, 148, 64–73. [Google Scholar] [CrossRef]
- Vazquez-Revuelta, P.; Madrigal-Burgaleta, R. Death Due to Live Bee Acupuncture Apitherapy. J. Investig. Allergol. Clin. Immunol. 2018, 28, 45–46. [Google Scholar] [CrossRef]
- Farkas, Á.; Molnár, R.; Morschhauser, T.; Hahn, I. Variation in Nectar Volume and Sugar Concentration of Allium ursinum L. ssp. Ucrainicum in Three Habitats. Sci. World J. 2012, 2012, 138579. [Google Scholar] [CrossRef]
- Rong, P.; Zhang, J.; Zhang, B. Applied TCM Ophthalmology [使用中医眼科学]; Zhong Guo Zhong Yi Yao Chu Ban She: Beijing, China, 2020; ISBN 9787513261678. [Google Scholar]
- Pourrahimi, M.; Abdi, M.; Ghods, R. Complications of Leech Therapy. Avicenna J. Phytomed. 2020, 10, 222–234. [Google Scholar]
- Kulbida, R.; Mathes, A.; Loeser, J. Beneficial Effects of Hirudotherapy in a Chronic Case of Complex Regional Pain Syndrome. J. Integr. Med. 2019, 17, 383–386. [Google Scholar] [CrossRef]
- Şenel, E.; Taylan Özkan, A.; Mumcuoglu, K.Y. Scientometric Analysis of Medicinal Leech Therapy. J. Ayurveda Integr. Med. 2019, 11, 534–538. [Google Scholar] [CrossRef]
- Jha, K.; Garg, A.; Narang, R.; Das, S. Hirudotherapy in Medicine and Dentistry. J. Clin. Diagn. Res. 2015, 9, ZE05. [Google Scholar] [CrossRef]
- Sobczak, N.; Kantyka, M. Hirudotherapy in Veterinary Medicine. Ann. Parasitol. 2014, 60, 89–92. [Google Scholar] [PubMed]
- Dong, H.; Ren, J.X.; Wang, J.J.; Ding, L.S.; Zhao, J.J.; Liu, S.Y.; Gao, H.M. Chinese Medicinal Leech: Ethnopharmacology, Phytochemistry, and Pharmacological Activities. Evid.-Based Complement. Altern. Med. 2016, 2016, 7895935. [Google Scholar] [CrossRef] [PubMed]
- Gileva, O.S.; Mumcuoglu, K.Y. Hirudotherapy. Biother.-Hist. Princ. Pract. 2013, 31–76. [Google Scholar] [CrossRef]
- Khan, J.A.; Parray, S.A. Irsal-e-Alaq (Leech Therapy) in Classical Literature of Unani System of Medicine: A Review. J. Blood Res. 2018, 1. [Google Scholar]
- Münstedt, K.; Riepen, T. Patients’ Decisions Regarding the Treatment of Primary Dysmenorrhoea. Complement. Ther. Med. 2019, 45, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pratiwi, R.; Dipadharma, R.H.F.; Prayugo, I.J.; Layandro, O.A. Recent Analytical Method for Detection of Chemical Adulterants in Herbal Medicine. Molecules 2021, 26, 6606. [Google Scholar] [CrossRef] [PubMed]



| 1. Fear | |||
| 2 | Aspen | Populus tremula L. | Vague fears of unknown objects and events |
| 6 | Cherry Plum | Prunus cerasifera Ehrh. | Fear of losing mental control |
| 20 | Mimulus | Erythranthe guttata (Fisch. DC.) G.L.Nesom | Phobia of known things with withdrawn and nervous personality |
| 25 | Red Chestnut | Aesculus × carnea Zeyh. | Excessive worry and over-anxiety for the welfare of relatives |
| 26 | Rock rose | Helianthemum nummularium (L.) Mill. | Extreme phobia, terror, panic (generalized anxiety disorder), and nightmares (nocturnal panic attack) |
| 2. Uncertainty | |||
| 5 | Cerato | Ceratostigma willmottianum Stapf | Lack of confidence in own decisions; repeatedly seeking advice of others (approval-seeking behavior) |
| 12 | Gentian | Gentiana amarella (L.) Börner | Doubt and discouragement after an unfortunate event; pessimism |
| 13 | Gorse | Ulex europaeus L. | Great hopelessness and despair |
| 17 | Hornbeam | Carpinus betulus L. | Procrastination; lack of the strength to fulfil daily tasks |
| 28 | Scleranthus | Scleranthus annuus L. | Indecisiveness between alternatives |
| 36 | Wild Oat | Bromus ramosus Huds. | Dissatisfaction and uncertainty over one’s direction in life (existential depression) |
| 3. Lack of Interest | |||
| 7 | Chestnut Bud | Aesculus hippocastanum L. | Failure to learn from mistakes and negative experiences |
| 9 | Clematis | Clematis vitalba L. | Daydreaming of the future with insufficient interest in the present; absent-mindedness |
| 16 | Honeysuckle | Lonicera caprifolium L. | Living in the past with overwhelming nostalgia |
| 21 | Mustard | Sinapis arvensis (L.) Andrz. ex Besser | Major depression for no reason |
| 23 | Olive | Olea europaea L. | Fatigue induced by physical and mental exertion |
| 35 | White Chestnut | Aesculus hippocastanum L. | Unwanted worrying and intrusive thoughts; concentration inability |
| 37 | Wild Rose | Rosa canina L. | Resignation, learned helplessness, and apathy |
| 4. Loneliness | |||
| 14 | Heather | Calluna vulgaris (L.) Hull | Fear against loneliness contributes to talkative, self-centered, and self-concerned individuals |
| 18 | Impatiens | Impatiens glandulifera Royle | Impatience, frustration, and irritability |
| 34 | Water Violet | Hottonia palustris L. | Self-reliant individuals; pride and arrogance |
| 5. Over-Sensitivity | |||
| 1 | Agrimony | Agrimonia eupatoria L. | Hidden worries and mental torture masked by a cheerful face (“smiling depression”) |
| 4 | Centaury | Centaurium erythrea Rafn. | Inability to refuse |
| 15 | Holly | Ilex aquifolium L. | Resentment, envy, jealousy, suspicion, and revenge; victim mentality |
| 33 | Walnut | Juglans regia L. | Protection from changing and unwanted influences (at birth, puberty, divorce etc.); strongly influenced by opinions of others |
| 6. Despondency and Despair | |||
| 10 | Crab Apple | Malus pumila Mill. | Fear of contamination (mysophobia), sensation of uncleanliness, also for self-hatred and body image distortion |
| 11 | Elm | Ulmus minor Mill. | Overwhelmed by responsibilities (responsibility fatigue) and pressures of workload (workload paralysis) |
| 19 | Larch | Larix decidua Mill. | Low self-esteem; lack of confidence, fear of failure (maladaptive perfectionism) |
| 22 | Oak | Quercus robur L. | Perseverance to overcome adversity without losing hope |
| 24 | Pine | Pinus sylvestris L. | Self-blame, self-criticism, guilt |
| 29 | Star of Bethlehem | Ornithogalum umbellatum L. | Sensation of shock and loss, traumatic events, grief of tragedy, post-traumatic stress disorder (PTSD) |
| 30 | Sweet Chestnut | Castanea sativa Mill. | Extreme mental anguish and emotional agony; hopelessness despite all attempts |
| 38 | Willow | Salix alba var. vitellina (L.) Stokes | Resentfulness, self-pity, and bitterness |
| 7. Overcare for others | |||
| 3 | Beech | Fagus sylvatica L. | Intolerance and hyperreactivity; critical, inflexible, and perfectionist mindset |
| 8 | Chicory | Cichorium intybus L. | Selfish and egoistic mindset; demand appreciation from others; possessive, controlling, and critical behaviors (narcissistic personality) |
| 27 | Rock water | Aqua petraea | Self-discipline, self-denial, and self-repression; asceticism |
| 31 | Vervain | Verbena officinalis L. | Enthusiastic strain and pressure; perfectionism and overexertion |
| 32 | Vine | Vitis vinifera L. | Dominance-seeking behavior and cognitive inflexibility |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rák, T.; Csutak, A. Complementary Practices in Pharmacy and Their Relation to Glaucoma—Classification, Definitions, and Limitations. Sci. Pharm. 2024, 92, 16. https://doi.org/10.3390/scipharm92010016
Rák T, Csutak A. Complementary Practices in Pharmacy and Their Relation to Glaucoma—Classification, Definitions, and Limitations. Scientia Pharmaceutica. 2024; 92(1):16. https://doi.org/10.3390/scipharm92010016
Chicago/Turabian StyleRák, Tibor, and Adrienne Csutak. 2024. "Complementary Practices in Pharmacy and Their Relation to Glaucoma—Classification, Definitions, and Limitations" Scientia Pharmaceutica 92, no. 1: 16. https://doi.org/10.3390/scipharm92010016
APA StyleRák, T., & Csutak, A. (2024). Complementary Practices in Pharmacy and Their Relation to Glaucoma—Classification, Definitions, and Limitations. Scientia Pharmaceutica, 92(1), 16. https://doi.org/10.3390/scipharm92010016