Pharmacokinetic Simulation Study: Exploring the Impact of Clinical Parameters on Lamotrigine for Different Patient Populations with Implications for Liver Function Assessment and Therapeutic Drug Monitoring
Abstract
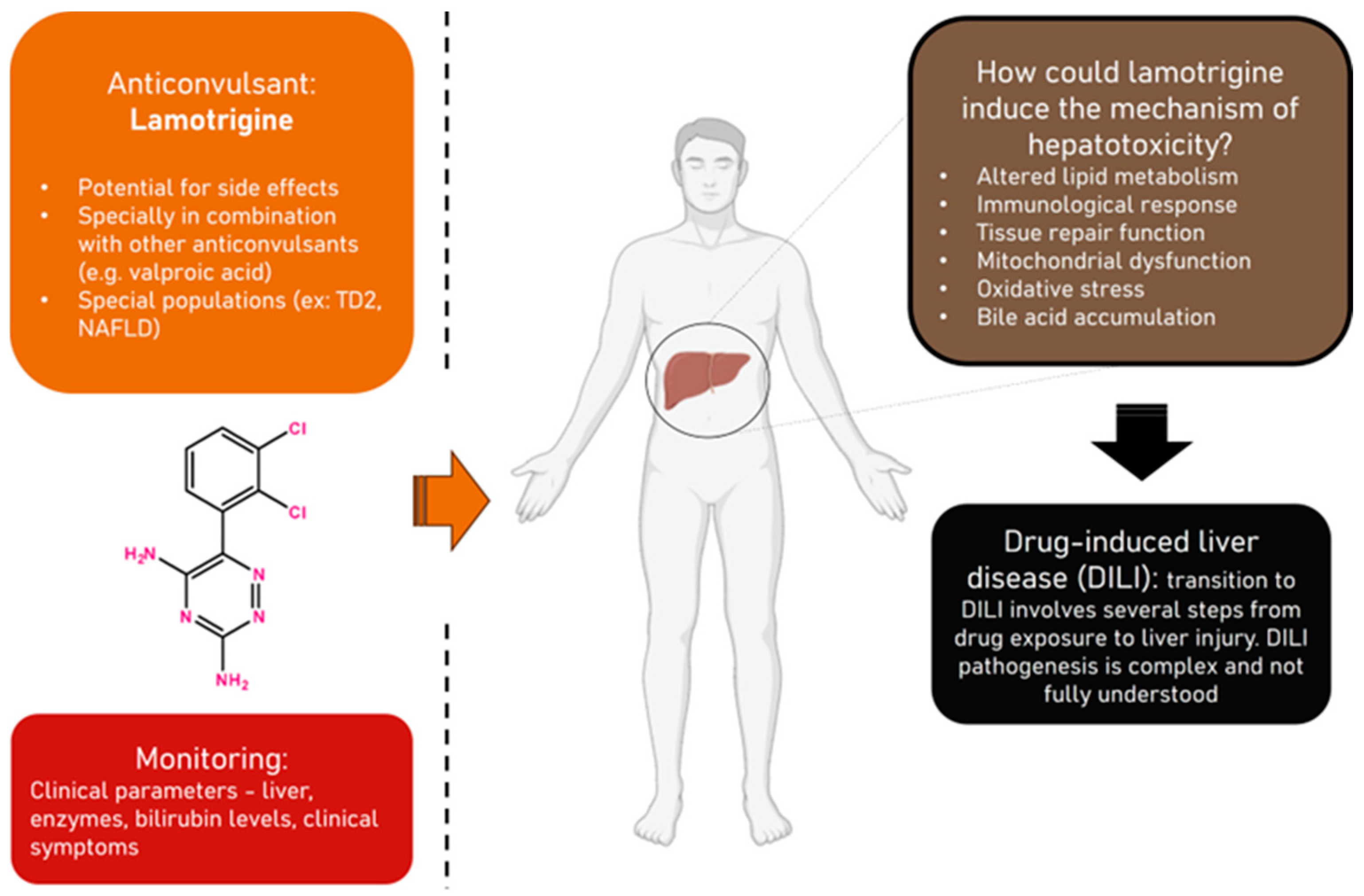
1. Introduction
2. Methods
2.1. Clinical Data Collection and Statistical Analysis
2.2. In Silico Prediction of Pharmacokinetic and Physicochemical Properties of Lamotrigine and Physiologically Based Pharmacokinetic (PBPK) Modeling Development
2.3. DILIsym Simulation Mode within GastroPlus
3. Results
3.1. Hospital Santo António Data Analysis
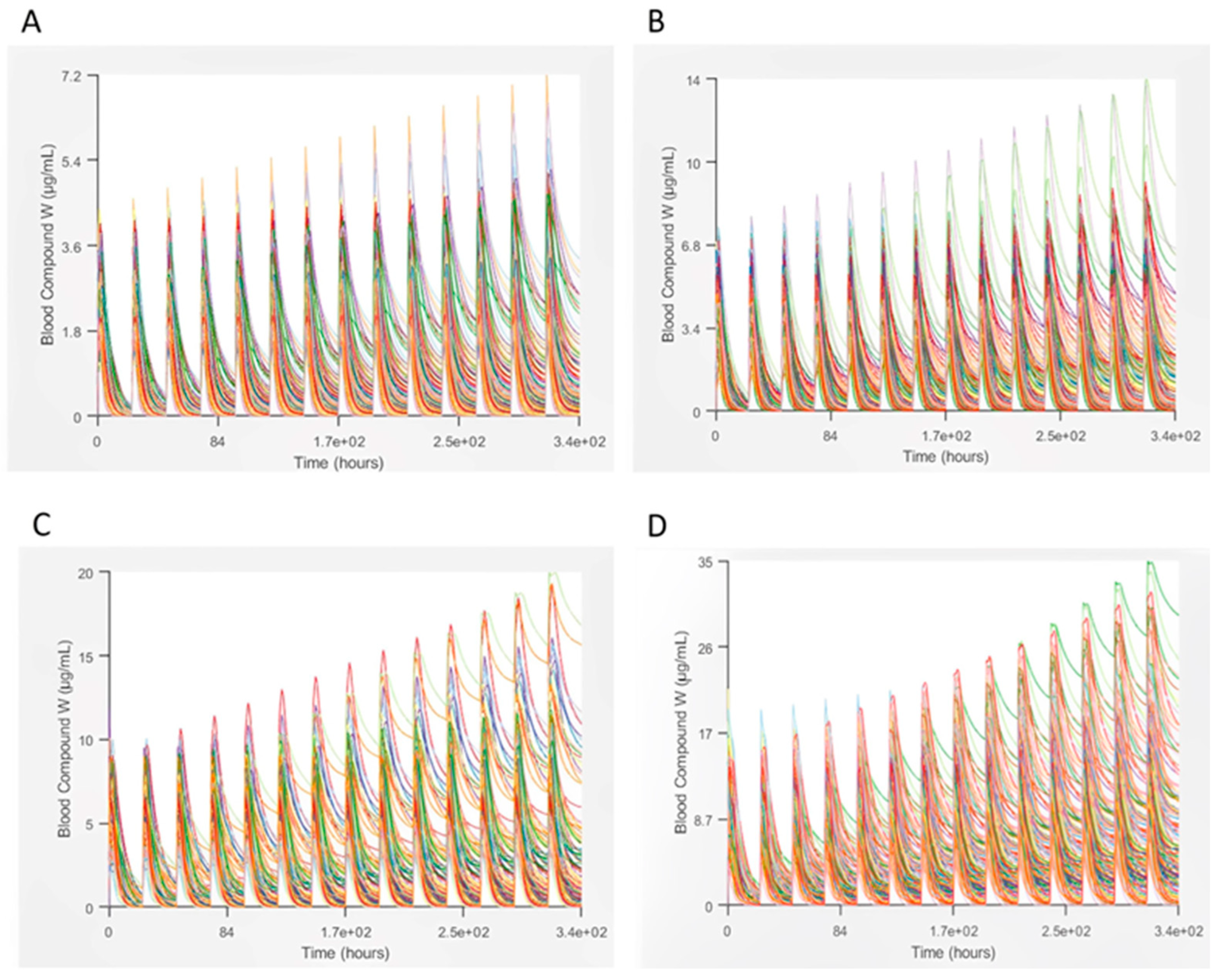
3.2. Lamotrigine PBPK Model and Population Simulation Results
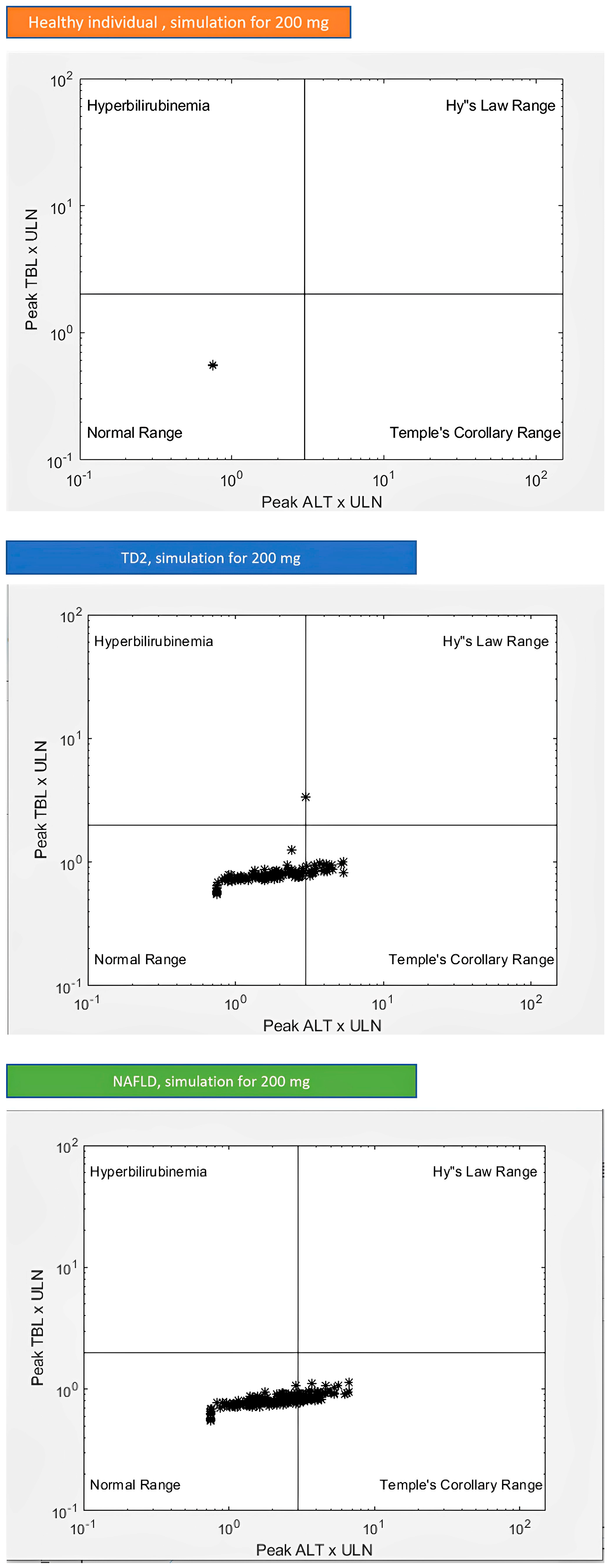
3.3. DILI Potential of Lamotrigine for Different Individuals
4. Discussion
4.1. Interplay of Lamotrigine, Vitamin B12, Creatinine, and Lipid Metabolism: Implications for Renal and Hepatic Systems
4.2. Pharmacokinetic Evaluation of Lamotrigine and Implications for Liver Function
4.3. Overall Liver Function Assessment
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Medicines Agency. Lamictal—Referral. Available online: https://www.ema.europa.eu/en/medicines/human/referrals/lamictal (accessed on 9 January 2024).
- FDA. Lamictal Label—Highlights of Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/020241s045s051lbl.pdf (accessed on 9 January 2024).
- FDA. FDA Drug Safety Communication: FDA Warns of Serious Immune System Reaction with Seizure and Mental Health Medicine Lamotrigine (Lamictal). Available online: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-warns-serious-immune-system-reaction-seizure-and-mental-health (accessed on 9 January 2024).
- French, J.A.; Perucca, E.; Sander, J.W.; Bergfeldt, L.; Baulac, M.; Auerbach, D.S.; Keezer, M.; Thijs, R.D.; Devinsky, O.; Vossler, D.G.; et al. FDA Safety Warning on the Cardiac Effects of Lamotrigine: An Advisory from the Ad Hoc ILAE/AES Task Force. Epilepsia Open 2021, 6, 45–48. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Kotake, K.; Watanabe, N.; Fujiwara, T.; Sakamoto, S. Lamotrigine in the Maintenance Treatment of Bipolar Disorder. Cochrane Database Syst. Rev. 2021. [Google Scholar] [CrossRef]
- Chouchana, M.; Smati, J.; Bloch, V.; Fontan, J.-E.; Etain, B.; Delage, C. Lamotrigine in Mood Disorders: Flash Survey on Prescribing Habits and Blood Tests Practices. Encephale 2023, 49, 640–644. [Google Scholar] [CrossRef]
- Avancini, J.; Maragno, L.; Santi, C.G.; Criado, P.R. Drug Reaction with Eosinophilia and Systemic Symptoms/Drug-Induced Hypersensitivity Syndrome: Clinical Features of 27 Patients. Clin. Exp. Dermatol. 2015, 40, 851–859. [Google Scholar] [CrossRef]
- Tempark, T.; John, S.; Rerknimitr, P.; Satapornpong, P.; Sukasem, C. Drug-Induced Severe Cutaneous Adverse Reactions: Insights into Clinical Presentation, Immunopathogenesis, Diagnostic Methods, Treatment, and Pharmacogenomics. Front. Pharmacol. 2022, 13, 832048. [Google Scholar] [CrossRef]
- Lens, S.; Crespo, G.; Carrión, J.A.; Miquel, R.; Navasa, M. Severe Acute Hepatitis in the Dress Syndrome: Report of Two Cases. Ann. Hepatol. 2010, 9, 198–201. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Cabriales, S.A.; Shear, N.H.; Gonzalez-Moreno, E.I. Liver Involvement in the Drug Reaction, Eosinophilia, and Systemic Symptoms Syndrome. World J. Clin. Cases 2019, 7, 705–716. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Checa, J.C.; Bagnaninchi, P.; Ye, H.; Sancho-Bru, P.; Falcon-Perez, J.M.; Royo, F.; Garcia-Ruiz, C.; Konu, O.; Miranda, J.; Lunov, O.; et al. Advanced Preclinical Models for Evaluation of Drug-Induced Liver Injury—Consensus Statement by the European Drug-Induced Liver Injury Network [PRO-EURO-DILI-NET]. J. Hepatol. 2021, 75, 935–959. [Google Scholar] [CrossRef] [PubMed]
- Weber, S.; Gerbes, A.L. Challenges and Future of Drug-Induced Liver Injury Research—Laboratory Tests. Int. J. Mol. Sci. 2022, 23, 6049. [Google Scholar] [CrossRef]
- Im, S.G.; Yoo, S.H.; Park, Y.M.; Lee, S.J.; Jang, S.K.; Jeon, D.O.; Cho, H.J.; Oh, M.J. Liver Dysfunction Induced by Systemic Hypersensitivity Reaction to Lamotrigine: Case Report. Clin. Mol. Hepatol. 2015, 21, 180. [Google Scholar] [CrossRef] [PubMed]
- Iriki, H.; Ouchi, T.; Ito, H.; Sawada, M.; Mukai, M.; Nomura, H.; Baba, Y.; Adachi, T.; Funakoshi, T.; Amagai, M.; et al. Case of Lamotrigine-induced Drug Adverse Reaction under Tocilizumab Treatment with Clinical and Virological Features of Drug-induced Hypersensitivity Syndrome. J. Dermatol. 2018, 45, 738–741. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, Y.; Azukizawa, H.; Asada, H.; Niihara, H.; Morita, E.; Yamauchi, T.; Mizukawa, Y.; Kusakabe, Y.; Numazawa, S.; Izumi, M.; et al. Drug-induced Hypersensitivity Syndrome/Drug Reaction with Eosinophilia and Systemic Symptoms Due to Lamotrigine Differs from That Due to Other Drugs. J. Dermatol. 2019, 46, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Marie, S.; Frost, K.L.; Hau, R.K.; Martinez-Guerrero, L.; Izu, J.M.; Myers, C.M.; Wright, S.H.; Cherrington, N.J. Predicting Disruptions to Drug Pharmacokinetics and the Risk of Adverse Drug Reactions in Non-Alcoholic Steatohepatitis Patients. Acta Pharm. Sin. B 2023, 13, 1–28. [Google Scholar] [CrossRef]
- Massart, J.; Begriche, K.; Moreau, C.; Fromenty, B. Role of Nonalcoholic Fatty Liver Disease as Risk Factor for Drug-Induced Hepatotoxicity. J. Clin. Transl. Res. 2017, 3, 212–232. [Google Scholar] [CrossRef]
- Lee, C.; Lui, D.T.; Lam, K.S. Non-alcoholic Fatty Liver Disease and Type 2 Diabetes: An Update. J. Diabetes Investig. 2022, 13, 930–940. [Google Scholar] [CrossRef]
- Kosmalski, M.; Ziółkowska, S.; Czarny, P.; Szemraj, J.; Pietras, T. The Coexistence of Nonalcoholic Fatty Liver Disease and Type 2 Diabetes Mellitus. J. Clin. Med. 2022, 11, 1375. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, C.G.; Rau, M.; Jahn, D.; Geier, A. Changes in Drug Transport and Metabolism and Their Clinical Implications in Non-Alcoholic Fatty Liver Disease. Expert. Opin. Drug Metab. Toxicol. 2017, 13, 625–640. [Google Scholar] [CrossRef]
- Perez-Lloret, S.; Olmos, L.; De Mena, F.; Pieczanski, P.; Moncalvo, J.R. Bioequivalence of lamotrigine 50-mg tablets in healthy male volunteers: A randomized, single-dose, 2-period, 2-sequence crossover study. Arzneimittelforschung 2012, 62, 470–476. [Google Scholar] [CrossRef]
- Jee, A.; Sernoskie, S.C.; Uetrecht, J. Idiosyncratic Drug-Induced Liver Injury: Mechanistic and Clinical Challenges. Int. J. Mol. Sci. 2021, 22, 2954. [Google Scholar] [CrossRef]
- Karaoulanis, S.E.; Syngelakis, M.; Fokas, K. Rhabdomyolysis after Lamotrigine Overdose: A Case Report and Review of the Literature. Ann. Gen. Psychiatry 2016, 15, 6. [Google Scholar] [CrossRef]
- Fontana, R.J. Pathogenesis of Idiosyncratic Drug-Induced Liver Injury and Clinical Perspectives. Gastroenterology 2014, 146, 914–928.e1. [Google Scholar] [CrossRef] [PubMed]
- McGill, M.R.; Jaeschke, H. Biomarkers of Drug-Induced Liver Injury: Progress and Utility in Research, Medicine, and Regulation. Expert. Rev. Mol. Diagn. 2018, 18, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Wu, D.; Jiang, W.; Li, J.; Long, J.; Jia, C.; Zhou, T. Molecular Biomarkers in Drug-Induced Liver Injury: Challenges and Future Perspectives. Front. Pharmacol. 2020, 10, 1667. [Google Scholar] [CrossRef]
- Nicoletti, P.; Barrett, S.; McEvoy, L.; Daly, A.K.; Aithal, G.; Lucena, M.I.; Andrade, R.J.; Wadelius, M.; Hallberg, P.; Stephens, C.; et al. Shared Genetic Risk Factors Across Carbamazepine-Induced Hypersensitivity Reactions. Clin. Pharmacol. Ther. 2019, 106, 1028–1036. [Google Scholar] [CrossRef]
- Costa, B.; Vale, N. Understanding Lamotrigine’s Role in the CNS and Possible Future Evolution. Int. J. Mol. Sci. 2023, 24, 6050. [Google Scholar] [CrossRef]
- Matsuo, F.; Gay, P.; Madsen, J.; Tolman, K.G.; Rollins, D.E.; Risner, M.E.; Lai, A.A. Lamotrigine High-Dose Tolerability and Safety in Patients with Epilepsy: A Double-Blind, Placebo-Controlled, Eleven-Week Study. Epilepsia 1996, 37, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Patsalos, P.N.; Berry, D.J.; Bourgeois, B.F.D.; Cloyd, J.C.; Glauser, T.A.; Johannessen, S.I.; Leppik, I.E.; Tomson, T.; Perucca, E. Antiepileptic Drugs—Best Practice Guidelines for Therapeutic Drug Monitoring: A Position Paper by the Subcommission on Therapeutic Drug Monitoring, ILAE Commission on Therapeutic Strategies. Epilepsia 2008, 49, 1239–1276. [Google Scholar] [CrossRef] [PubMed]
- Garnett, W.R. Lamotrigine: Pharmacokinetics. J. Child. Neurol. 1997, 12, S10–S15. [Google Scholar] [CrossRef]
- Milosheska, D.; Lorber, B.; Vovk, T.; Kastelic, M.; Dolžan, V.; Grabnar, I. Pharmacokinetics of Lamotrigine and Its Metabolite N-2-Glucuronide: Influence of Polymorphism of UDP-Glucuronosyltransferases and Drug Transporters. Br. J. Clin. Pharmacol. 2016, 82, 399–411. [Google Scholar] [CrossRef]
- Douglas-Hall, P.; Dzahini, O.; Gaughran, F.; Bile, A.; Taylor, D. Variation in Dose and Plasma Level of Lamotrigine in Patients Discharged from a Mental Health Trust. Ther. Adv. Psychopharmacol. 2017, 7, 17–24. [Google Scholar] [CrossRef]
- Mitra-Ghosh, T.; Callisto, S.P.; Lamba, J.K.; Remmel, R.P.; Birnbaum, A.K.; Barbarino, J.M.; Klein, T.E.; Altman, R.B. PharmGKB Summary: Lamotrigine Pathway, Pharmacokinetics and Pharmacodynamics. Pharmacogenet Genom. 2020, 30, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Pennazio, F.; Brasso, C.; Villari, V.; Rocca, P. Current Status of Therapeutic Drug Monitoring in Mental Health Treatment: A Review. Pharmaceutics 2022, 14, 2674. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.; Grover, S.; Rao, G.P. Clinical Practice Guidelines for Management of Bipolar Disorder. Indian. J. Psychiatry 2017, 59, S51–S66. [Google Scholar] [CrossRef]
- St Louis, E.K.; Rosenfeld, W.E.; Bramley, T. Antiepileptic Drug Monotherapy: The Initial Approach in Epilepsy Management. Curr. Neuropharmacol. 2009, 7, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Li, C. Little’s Test of Missing Completely at Random. Stata J. Promot. Commun. Stat. Stata 2013, 13, 795–809. [Google Scholar] [CrossRef]
- van Dijkman, S.C.; de Jager, N.C.B.; Rauwé, W.M.; Danhof, M.; Della Pasqua, O. Effect of Age-Related Factors on the Pharmacokinetics of Lamotrigine and Potential Implications for Maintenance Dose Optimisation in Future Clinical Trials. Clin. Pharmacokinet. 2018, 57, 1039–1053. [Google Scholar] [CrossRef]
- Conner, T.M.; Reed, R.C.; Zhang, T. A Physiologically Based Pharmacokinetic Model for Optimally Profiling Lamotrigine Disposition and Drug–Drug Interactions. Eur. J. Drug Metab. Pharmacokinet. 2018, 44, 389–408. [Google Scholar] [CrossRef]
- Porat, D.; Azran, C.; Kais, H.; Dahan, A. Managing the Unpredictable: Mechanistic Analysis and Clinical Recommendations for Lamotrigine Treatment after Bariatric Surgery. J. Clin. Med. 2021, 10, 5627. [Google Scholar] [CrossRef]
- Rambeck, B.; Wolf, P. Lamotrigine Clinical Pharmacokinetics. Clin. Pharmacokinet. 1993, 25, 433–443. [Google Scholar] [CrossRef]
- Agabeyoglu, I.; Incecayir, T. Pharmacokinetic Modelling of Lamotrigine from Plasma Concentrations in Healthy Volunteers. J. Bioanal. Biomed. 2009, 1, 41–45. [Google Scholar] [CrossRef]
- Argikar, U.A.; Remmel, R.P. Variation in Glucuronidation of Lamotrigine in Human Liver Microsomes. Xenobiotica 2009, 39, 355–363. [Google Scholar] [CrossRef]
- Polepally, A.R.; Brundage, R.C.; Remmel, R.P.; Leppik, I.E.; Pennell, P.B.; White, J.R.; Ramsay, R.E.; Kistner, B.M.; Birnbaum, A.K. Lamotrigine Pharmacokinetics following Oral and Stable-labeled Intravenous Administration in Young and Elderly Adult Epilepsy Patients: Effect of Age. Epilepsia 2018, 59, 1718–1726. [Google Scholar] [CrossRef]
- Cohen, A.F.; Land, G.S.; Breimer, D.D.; Yuen, W.C.; Winton, C.; Peck, A.W. Lamotrigine, a New Anticonvulsant: Pharmacokinetics in Normal Humans. Clin. Pharmacol. Ther. 1987, 42, 535–541. [Google Scholar] [CrossRef]
- Battino, D.; Estienne, M.; Avanzini, G. Clinical Pharmacokinetics of Antiepileptic Drugs in Paediatric Patients. Clin. Pharmacokinet. 1995, 29, 341–369. [Google Scholar] [CrossRef]
- Mahmoud, S.H.; Zhou, X.Y.; Ahmed, S.N. Managing the Patient with Epilepsy and Renal Impairment. Seizure 2020, 76, 143–152. [Google Scholar] [CrossRef]
- McMahon, G.M.; Hwang, S.-J.; Tanner, R.M.; Jacques, P.F.; Selhub, J.; Muntner, P.; Fox, C.S. The Association between Vitamin B12, Albuminuria and Reduced Kidney Function: An Observational Cohort Study. BMC Nephrol. 2015, 16, 7. [Google Scholar] [CrossRef]
- Linnebank, M.; Moskau, S.; Semmler, A.; Widman, G.; Stoffel-Wagner, B.; Weller, M.; Elger, C.E. Antiepileptic Drugs Interact with Folate and Vitamin B12 Serum Levels. Ann. Neurol. 2011, 69, 352–359. [Google Scholar] [CrossRef]
- Sugihara, T.; Koda, M.; Matono, T.; Okamoto, K.; Murawaki, Y.; Isomoto, H.; Tokunaga, S. Risk Assessment of Hepatocellular Carcinoma in General Population by Liver Stiffness in Combination with Controlled Attenuation Parameter Using Transient Elastography: A Cross Sectional Study. Yonago Acta Med. 2017, 60, 106–112. [Google Scholar] [CrossRef]
- Wu, H.H.L.; Wang, A.Y.-M. Vitamin B12 and Chronic Kidney Disease. In Vitamins and Hormones; Academic Press: Cambridge, MA, USA, 2022; pp. 325–353. [Google Scholar]
- Dinç, D.; Schulte, P.F.J. The Use of Anticonvulsants and the Levels of Folate, Vitamin B12 and Homocysteine. Tijdschr. Psychiatr. 2018, 60, 20–28. [Google Scholar]
- Callaghan, F.M.; Leishear, K.; Abhyankar, S.; Demner-Fushman, D.; McDonald, C.J. High Vitamin B12 Levels Are Not Associated with Increased Mortality Risk for ICU Patients after Adjusting for Liver Function: A Cohort Study. ESPEN J. 2014, 9, e76–e83. [Google Scholar] [CrossRef]
- Romain, M.; Sviri, S.; Linton, D.M.; Stav, I.; van Heerden, P.V. The Role of Vitamin B12 in the Critically Ill—A Review. Anaesth. Intensive Care 2016, 44, 447–452. [Google Scholar] [CrossRef]
- Tal, S.; Shavit, Y.; Stern, F.; Malnick, S. Association Between Vitamin B12 Levels and Mortality in Hospitalized Older Adults. J. Am. Geriatr. Soc. 2010, 58, 523–526. [Google Scholar] [CrossRef]
- Hauser, E.; Seidl, R.; Freilinger, M.; Male, C.; Herkner, K. Hematologic Manifestations and Impaired Liver Synthetic Function during Valproate Monotherapy. Brain Dev. 1996, 18, 105–109. [Google Scholar] [CrossRef]
- Jacobsen, N.O.; Mosekilde, L.; Myhre-Jensen, O.; Pedersen, E.; Wildenhoff, K.E. Liver Biopsies in Epileptics during Anticonvulsant Therapy. Acta Med. Scand. 1976, 199, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Elkhatib, I.; Cao, W.; Rao, S.; Fryer, J.; Buchman, A.L. Serum B12 Concentration Is Elevated in Patients Receiving Chronic Parenteral Nutrition, But Is Not a Marker of Intestinal Failure-Associated Liver Disease. J. Clin. Gastroenterol. 2010, 44, 571–574. [Google Scholar] [CrossRef] [PubMed]
- Al-Musharaf, S.; Aljuraiban, G.S.; Danish Hussain, S.; Alnaami, A.M.; Saravanan, P.; Al-Daghri, N. Low Serum Vitamin B12 Levels Are Associated with Adverse Lipid Profiles in Apparently Healthy Young Saudi Women. Nutrients 2020, 12, 2395. [Google Scholar] [CrossRef] [PubMed]
- Adaikalakoteswari, A.; Jayashri, R.; Sukumar, N.; Venkataraman, H.; Pradeepa, R.; Gokulakrishnan, K.; Anjana, R.M.; McTernan, P.G.; Tripathi, G.; Patel, V.; et al. Vitamin B12 Deficiency Is Associated with Adverse Lipid Profile in Europeans and Indians with Type 2 Diabetes. Cardiovasc. Diabetol. 2014, 13, 129. [Google Scholar] [CrossRef] [PubMed]
- Dickerman, H.W.; Redfield, B.G.; Bieri, J.G.; Weissbach, H. Studies on the role of vitamin b12 for the synthesis of methionine in liver. Ann. N. Y Acad. Sci. 1964, 112, 791–798. [Google Scholar] [CrossRef]
- Ge, Y.; Zadeh, M.; Mohamadzadeh, M. Vitamin B12 Regulates the Transcriptional, Metabolic, and Epigenetic Programing in Human Ileal Epithelial Cells. Nutrients 2022, 14, 2825. [Google Scholar] [CrossRef] [PubMed]
- Vyas, M.V.; Davidson, B.A.; Escalaya, L.; Costella, J.; Saposnik, G.; Burneo, J.G. Antiepileptic Drug Use for Treatment of Epilepsy and Dyslipidemia: Systematic Review. Epilepsy Res. 2015, 113, 44–67. [Google Scholar] [CrossRef] [PubMed]
- Mintzer, S.; Skidmore, C.T.; Abidin, C.J.; Morales, M.C.; Chervoneva, I.; Capuzzi, D.M.; Sperling, M.R. Effects of Antiepileptic Drugs on Lipids, Homocysteine, and C-reactive Protein. Ann. Neurol. 2009, 65, 448–456. [Google Scholar] [CrossRef]
- Mintzer, S.; Trinka, E.; Kraemer, G.; Chervoneva, I.; Werhahn, K.J. Impact of Carbamazepine, Lamotrigine, and Levetiracetam on Vascular Risk Markers and Lipid-lowering Agents in the Elderly. Epilepsia 2018, 59, 1899–1907. [Google Scholar] [CrossRef] [PubMed]
- de Almeida, K.M.; Moreira, C.L.R.L.; Lafer, B. Metabolic Syndrome and Bipolar Disorder: What Should Psychiatrists Know? CNS Neurosci. Ther. 2012, 18, 160–166. [Google Scholar] [CrossRef]
- Suzuki, T.; Mihara, K.; Nagai, G.; Kagawa, S.; Nakamura, A.; Nemoto, K.; Kondo, T. Relationship Between UGT1A4 and UGT2B7 Polymorphisms and the Steady-State Plasma Concentrations of Lamotrigine in Patients With Treatment-Resistant Depressive Disorder Receiving Lamotrigine as Augmentation Therapy. Ther. Drug Monit. 2019, 41, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, G.D. Variability in the Human Drug Response. Thromb. Res. 1983, 29, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Alabi, A.; Todd, A.; Husband, A.; Reilly, J. Safety Profile of Lamotrigine in Overdose. Ther. Adv. Psychopharmacol. 2016, 6, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Alyahya, B.; Friesen, M.; Nauche, B.; Laliberté, M. Acute Lamotrigine Overdose: A Systematic Review of Published Adult and Pediatric Cases. Clin. Toxicol. 2017, 56, 81–89. [Google Scholar] [CrossRef]
- Bondareva, I. Individualizing Antiepileptic Therapy for Patients. In Individualized Drug Therapy for Patients; Elsevier: Amsterdam, The Netherlands, 2017; pp. 327–371. [Google Scholar]
- Chong, E.; Dupuis, L.L. Therapeutic Drug Monitoring of Lamotrigine. Ann. Pharmacother. 2002, 36, 917–920. [Google Scholar] [CrossRef]
- Tien, N.; Wu, T.-Y.; Lin, C.-L.; Chu, F.-Y.; Wang, C.C.N.; Hsu, C.Y.; Tsai, F.-J.; Fang, Y.-J.; Lim, Y.-P. Association of Epilepsy, Anti-Epileptic Drugs (AEDs), and Type 2 Diabetes Mellitus (T2DM): A Population-Based Cohort Retrospective Study, Impact of AEDs on T2DM-Related Molecular Pathway, and via Peroxisome Proliferator-Activated Receptor γ Transactivation. Front. Endocrinol. 2023, 14, 1156952. [Google Scholar] [CrossRef]
- Luef, G.; Rauchenzauner, M.; Waldmann, M.; Sturm, W.; Sandhofer, A.; Seppi, K.; Trinka, E.; Unterberger, I.; Ebenbichler, C.F.; Joannidis, M.; et al. Non-Alcoholic Fatty Liver Disease (NAFLD), Insulin Resistance and Lipid Profile in Antiepileptic Drug Treatment. Epilepsy Res. 2009, 86, 42–47. [Google Scholar] [CrossRef]
- Shlobin, N.A.; Sander, J.W. Drivers for the Comorbidity of Type 2 Diabetes Mellitus and Epilepsy: A Scoping Review. Epilepsy Behav. 2020, 106, 107043. [Google Scholar] [CrossRef]
- Howell, B.A.; Yang, Y.; Kumar, R.; Woodhead, J.L.; Harrill, A.H.; Clewell, H.J.; Andersen, M.E.; Siler, S.Q.; Watkins, P.B. In Vitro to in Vivo Extrapolation and Species Response Comparisons for Drug-Induced Liver Injury (DILI) Using DILIsymTM: A Mechanistic, Mathematical Model of DILI. J. Pharmacokinet. Pharmacodyn. 2012, 39, 527–541. [Google Scholar] [CrossRef] [PubMed]
- Eichenbaum, G.; Yang, K.; Gebremichael, Y.; Howell, B.A.; Murray, F.J.; Jacobson-Kram, D.; Jaeschke, H.; Kuffner, E.; Gelotte, C.K.; Lai, J.C.K.; et al. Application of the DILIsym® Quantitative Systems Toxicology Drug-Induced Liver Injury Model to Evaluate the Carcinogenic Hazard Potential of Acetaminophen. Regul. Toxicol. Pharmacol. 2020, 118, 104788. [Google Scholar] [CrossRef]
- Brennan, P.N.; Cartlidge, P.; Manship, T.; Dillon, J.F. Guideline Review: EASL Clinical Practice Guidelines: Drug-Induced Liver Injury (DILI). Frontline Gastroenterol. 2021, 13, 332–336. [Google Scholar] [CrossRef]
- Chopra, S.; Saxena, R. Drug-Induced Liver Injury—Perspectives from Pathology. Curr. Pharmacol. Rep. 2018, 4, 182–192. [Google Scholar] [CrossRef]
- Teschke, R.; Danan, G. Advances in Idiosyncratic Drug-Induced Liver Injury Issues: New Clinical and Mechanistic Analysis Due to Roussel Uclaf Causality Assessment Method Use. Int. J. Mol. Sci. 2023, 24, 10855. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.H. Drug-Induced Liver Injury Throughout the Drug Development Life Cycle: Where We Have Been, Where We Are Now, and Where We Are Headed. Perspectives of a Clinical Hepatologist. Pharmaceut Med. 2013, 27, 165–191. [Google Scholar] [CrossRef]
- Church, R.J.; Watkins, P.B. In Silico Modeling to Optimize Interpretation of Liver Safety Biomarkers in Clinical Trials. Exp. Biol. Med. 2018, 243, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Overstreet, K.; Costanza, C.; Behling, C.; Hassanin, T.; Masliah, E. Fatal Progressive Hepatic Necrosis Associated with Lamotrigine Treatment: A Case Report and Literature Review. Dig. Dis. Sci. 2002, 47, 1921–1925. [Google Scholar] [CrossRef]
- Bresnahan, R.; Panebianco, M.; Marson, A.G. Lamotrigine Add-on Therapy for Drug-Resistant Generalised Tonic-Clonic Seizures. Cochrane Database Syst. Rev. 2020, 2020, CD007783. [Google Scholar] [CrossRef]
- Davidson, M.D.; Ballinger, K.R.; Khetani, S.R. Long-Term Exposure to Abnormal Glucose Levels Alters Drug Metabolism Pathways and Insulin Sensitivity in Primary Human Hepatocytes. Sci. Rep. 2016, 6, 28178. [Google Scholar] [CrossRef] [PubMed]
| Value | Unit | Reference | |
|---|---|---|---|
| Blood to plasma | 1.137 | Dimensionless | ADMET Predictor |
| Fraction unbound plasma | 0.45 | Dimensionless | ADMET Predictor |
| Molecular weight | 256.1 | g/mol | ADMET Predictor |
| LogP | 1.98 | o: w | ADMET Predictor |
| Acid-base switch | Monoprotic base | - | [40,42] |
| pKa | 5.5 | Dimensionless | [40,42] |
| Renal clearance | 2000 | mL/hour/kg0.75 | [40] |
| K(ab)—absorption rate | 0.85 | 1/hour | [43] |
| Peff in human | 7.761 | 10−4 cm/s | ADMET Predictor |
| Fraction unbound in enterocytes | 0.9 | Dimensionless | ADMET Predictor |
| Km metabolite (UGT1A3) | 700 | (μM) | [40,44] |
| Vmax (UGT1A3) | 17 | (pmol/min/mg of microsomal protein) | [40,44] |
| Km metabolite (UGT1A4) | 550 | (μM) | [40,44] |
| Vmax (UGT1A4) | 153 | (pmol/min/mg of microsomal protein) | [40,44] |
| Total (n = 37, Mean) | Median | Q1 | Q2 | IQR | |
|---|---|---|---|---|---|
| Sex | |||||
| Male | 12 (32.43%) | Female | |||
| Female | 25 (67.57%) | NA | NA | NA | |
| Age | |||||
| Children (≤15 years) | 9 (24.32%) | 9 | 8 | 13 | 5 |
| Youth (>15>25 years) | 11 (29.72%) | 20 | 18.5 | 24 | 5.5 |
| Adult (≥25>65 years | 17 (45.95%) | 40 | 38 | 59 | 21 |
| Senior (≥65 years) | 0 (0%) | NA | NA | NA | NA |
| Lamotrigine dose group | |||||
| Very Low (<100 mg) | 7 (21.21%) | 100 | 25 | 100 | 75 |
| Low (100–200 mg) | 9 (27.27%) | 175 | 150 | 200 | 50 |
| Average (200–400 mg) | 16 (48.5%) | 375 | 300 | 400 | 100 |
| High (>400 mg) | 1 (3.03%) | 600 | 600 | 600 | 0 |
| Reported drug combination therapy | |||||
| Yes | 23 (62.16%) | Yes | NA | NA | NA |
| No | 14 (37.84%) |
| Parameters Measured | Number of Individuals with Out-of-Range Parameter Values | Percent of Total (n = 37) |
|---|---|---|
| Uric acid | 0 | 0.00% |
| Bicarbonates | 4 | 10.81% |
| Glucose | 4 | 10.81% |
| Creatinine | 16 | 43.24% |
| Urea | 1 | 2.70% |
| Bilirubin total | 8 | 21.62% |
| Aminotransferase aspartate | 0 | 0.00% |
| Aminotransferase alanine | 6 | 16.21% |
| Phosphatase alkaline | 3 | 8.10% |
| Gamma-glutamyl transferase | 9 | 24.32% |
| Lactate dehydrogenase | 5 | 13.51% |
| Total creatine kinase (CK) | 5 | 13.51% |
| Ammonia | 5 | 13.51% |
| Total cholesterol | 3 | 8.10% |
| Triglycerides | 1 | 2.70% |
| High density lipoprotein (HDL) cholesterol | 2 | 5.40% |
| Non-HDL cholesterol | 0 | 0.00% |
| Low-density lipopro-tein (LDL) cholesterol | 1 | 2.70% |
| Very-low-density lipoprotein (VLDL) cholesterol | 1 | 2.70% |
| C-reactive protein | 1 | 2.70% |
| Iron | 2 | 5.41% |
| Albumin | 14 | 37.83% |
| Total proteins | 2 | 5.41% |
| Sodium | 0 | 0.00% |
| Potassium | 2 | 5.41% |
| Chlorides | 5 | 13.51% |
| Vitamin B12 | 7 | 18.92% |
| Variable 1 | Variable 2 | rho | p-Value | Strength of Correlation |
|---|---|---|---|---|
| Dose | ASAT | −0.5360653 | 0.0007523 | Moderate correlation |
| Gamma-glutamyl transferase | 0.4472029 | 0.009073 | Low to moderate | |
| VLDL cholesterol | 0.5018162 | 0.02417 | Moderate correlation | |
| Chlorates | −0.4058334 | 0.0357 | Low to moderate correlation | |
| Vitamin B12 | 0.6738606 | 0.04657 | Strong correlation | |
| Triglycerides | 0.5039345 | 0.02348 | Moderate correlation | |
| Trough concentration | 0.5548736 | 0.0004445 | Strong correlation | |
| Trough concentration | Phosphatase alkaline | −0.4746656 | 0.005253 | Low to moderate correlation |
| Sodium | −0.4580201 | 0.01425 | Low to moderate correlation | |
| Chlorates | −0.5701696 | 0.002357 | Strong correlation | |
| Vitamin B12 | 0.9333333 | 0.0007496 | Very strong correlation | |
| Total cholesterol | 0.5016958 | 0.02421 | Moderate correlation | |
| Non-HDL cholesterol | 0.5101582 | 0.02155 | Moderate correlation | |
| Combination | Uric acid | 0.785328 | 0.000874 | Strong correlation |
| Total cholesterol | 0.5919496 | 0.005967 | Moderate correlation | |
| Non-HDL cholesterol | 0.4639278 | 0.03935 | Low to moderate correlation | |
| Time | Total cholesterol | −0.8571429 | 0.02381 | Strong correlation |
| Non-HDL cholesterol | −0.8108437 | 0.02692 | Strong correlation | |
| Creatinine | Urea | 0.616872 | 4.781 × 10−5 | Strong correlation |
| Total bilirubin | 0.6655658 | 3.228 × 10−5 | Strong correlation | |
| Total creatoquinase | 0.5361997 | 0.008353 | Moderate correlation | |
| Sodium | 0.4112316 | 0.02668 | Low to moderate correlation | |
| Chlorates | 0.4914554 | 0.009231 | Moderate correlation | |
| Vitamin B12 | −0.9333333 | 0.0007496 | Very strong correlation |
| Variable 1 | Variable 2 | rho | p-Value | Strength of Correlations |
|---|---|---|---|---|
| Vitamin B12 | Dose | 0.6738606 | 0.04657 | Moderate correlation |
| Trough concentration | 0.9333333 | 0.0007496 | Very strong correlation | |
| Creatinine | −0.9333333 | 0.0007496 | Very strong correlation | |
| LDL-cholesterol | 0.7619048 | 0.03676 | Strong correlation | |
| VDL-cholestrol | 0.9047619 | 0.004563 | Very strong correlation | |
| Albumin | −0.8809524 | 0.007242 | Strong correlation | |
| Total cholesterol | 0.7425283 | 0.03486 | Strong correlation | |
| Triglycerides | 0.9047619 | 0.004563 | Very strong correlation | |
| Non-HDL-cholesterol | 0.7425283 | 0.03486 | Strong correlation |
| Dose | Results for Pharmacokinetic Parameter | |||||
|---|---|---|---|---|---|---|
| F (%) | Cmax (µg/mL) | Tmax (h) | AUC0–inf (µg h/mL) | AUC0–t (µg h/mL) | ||
| 200 | Observed (mean or geo. mean ± SD) | 98 | 2.46 ± 0.41–2.91 ± 0.26 | 1.11 | - | 94.6 ± 21.6–123 ± 14 |
| Single simulation | 99.268 | 1.7306 | 1.68 | 61.218 | 27.137 | |
| DILIsym simulation output (90% CI) | 98.267 (97.92–98.62) | 1.62 (1.576–1.664) | 1.7188 (1.537–1.900) | 60.218 (58.24–62.20) | 25.161 (24.64–25.68) | |
| 400 | Single simulation | 98.636 | 3.097 | 2.4 | 121.68 | 53.315 |
| DILIsym simulation output (90% CI) | 95.459 (94.68–96.38) | 3.0257 (2.918–3.133) | 1.7108 (1.539–1.882) | 124.65 (120.6–128.7) | 48.889 (47.73–50.05) | |
| 600 | Single simulation | 97.192 | 4.3525 | 0.48 | 180.05 | 77.604 |
| DILIsym simulation output (90% CI) | 90.359 | 3.9353 | 1.8835 | 186.48 | 67.06 | |
| 1200 | Single simulation | 84.868 | 8.0976 | 0.48 | 362.2 | 130.8 |
| DILIsym simulation output (90% CI) | 77.406 (75.39–79.57) | 7.089 (6.778–7.400) | 1.2443 (0.959–1.530) | 353.29 (337.1–369.5) | 113.49 (109.5–117.5) | |
| Group | Subgroup | Outcome Variable | Metric | Healthy Individuals | TD2 Individuals | NAFLD Individuals | Units |
|---|---|---|---|---|---|---|---|
| Outcomes | Outcomes | Number of deaths | Count | 0 | 0 | 0 | dimensionless |
| Outcomes | Outcomes | ALT at or over 3 × ULN | Count | 0 | 24 | 64 | dimensionless |
| Outcomes | Outcomes | Bilirubin over 2 × ULN | Count | 0 | 1 | 0 | dimensionless |
| Outcomes | Outcomes | Hy’s law cases | Count | 0 | 0 | 0 | dimensionless |
| Outcomes | Outcomes | ALT at least 5 × ULN | Count | 0 | 3 | 0 | dimensionless |
| Outcomes | Outcomes | Bilirubin greater than 2 × ULN | Count | 0 | 1 | 0 | dimensionless |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costa, B.; Silva, I.; Oliveira, J.C.; Reguengo, H.; Vale, N. Pharmacokinetic Simulation Study: Exploring the Impact of Clinical Parameters on Lamotrigine for Different Patient Populations with Implications for Liver Function Assessment and Therapeutic Drug Monitoring. Sci. Pharm. 2024, 92, 15. https://doi.org/10.3390/scipharm92010015
Costa B, Silva I, Oliveira JC, Reguengo H, Vale N. Pharmacokinetic Simulation Study: Exploring the Impact of Clinical Parameters on Lamotrigine for Different Patient Populations with Implications for Liver Function Assessment and Therapeutic Drug Monitoring. Scientia Pharmaceutica. 2024; 92(1):15. https://doi.org/10.3390/scipharm92010015
Chicago/Turabian StyleCosta, Bárbara, Isabel Silva, José Carlos Oliveira, Henrique Reguengo, and Nuno Vale. 2024. "Pharmacokinetic Simulation Study: Exploring the Impact of Clinical Parameters on Lamotrigine for Different Patient Populations with Implications for Liver Function Assessment and Therapeutic Drug Monitoring" Scientia Pharmaceutica 92, no. 1: 15. https://doi.org/10.3390/scipharm92010015
APA StyleCosta, B., Silva, I., Oliveira, J. C., Reguengo, H., & Vale, N. (2024). Pharmacokinetic Simulation Study: Exploring the Impact of Clinical Parameters on Lamotrigine for Different Patient Populations with Implications for Liver Function Assessment and Therapeutic Drug Monitoring. Scientia Pharmaceutica, 92(1), 15. https://doi.org/10.3390/scipharm92010015







