Analysis of Beneficial Effects of Flavonoids in Patients with Atherosclerosis Risk on Blood Pressure or Cholesterol during Random Controlled Trials: A Systematic Review and Meta-Analysis
Abstract
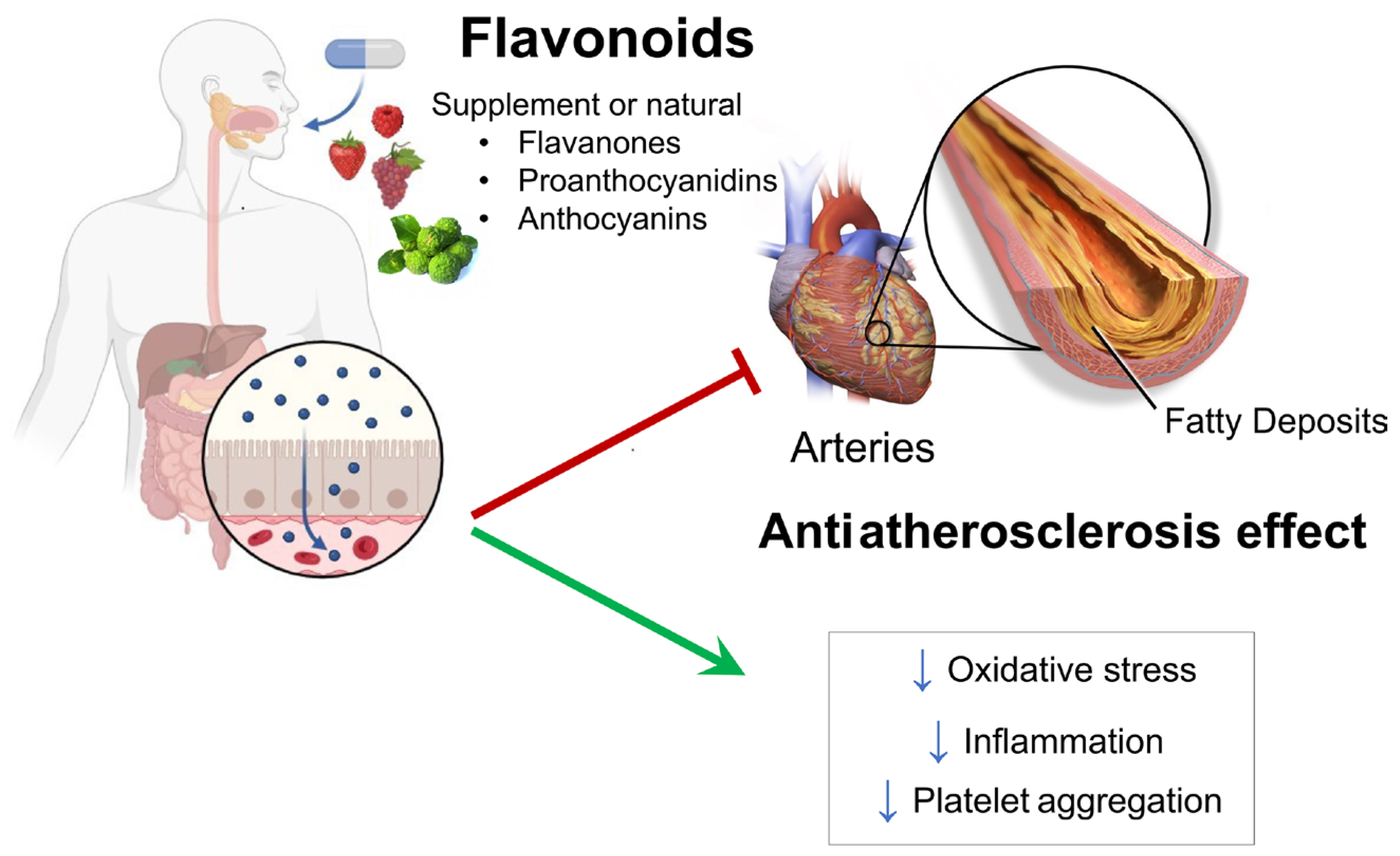
:1. Introduction
2. Materials and Methods
2.1. Data Extraction and Quality Assessment Tool
2.2. Outcome Measures
2.3. Risk of Bias in Individual Studies
2.4. Statistical Analysis
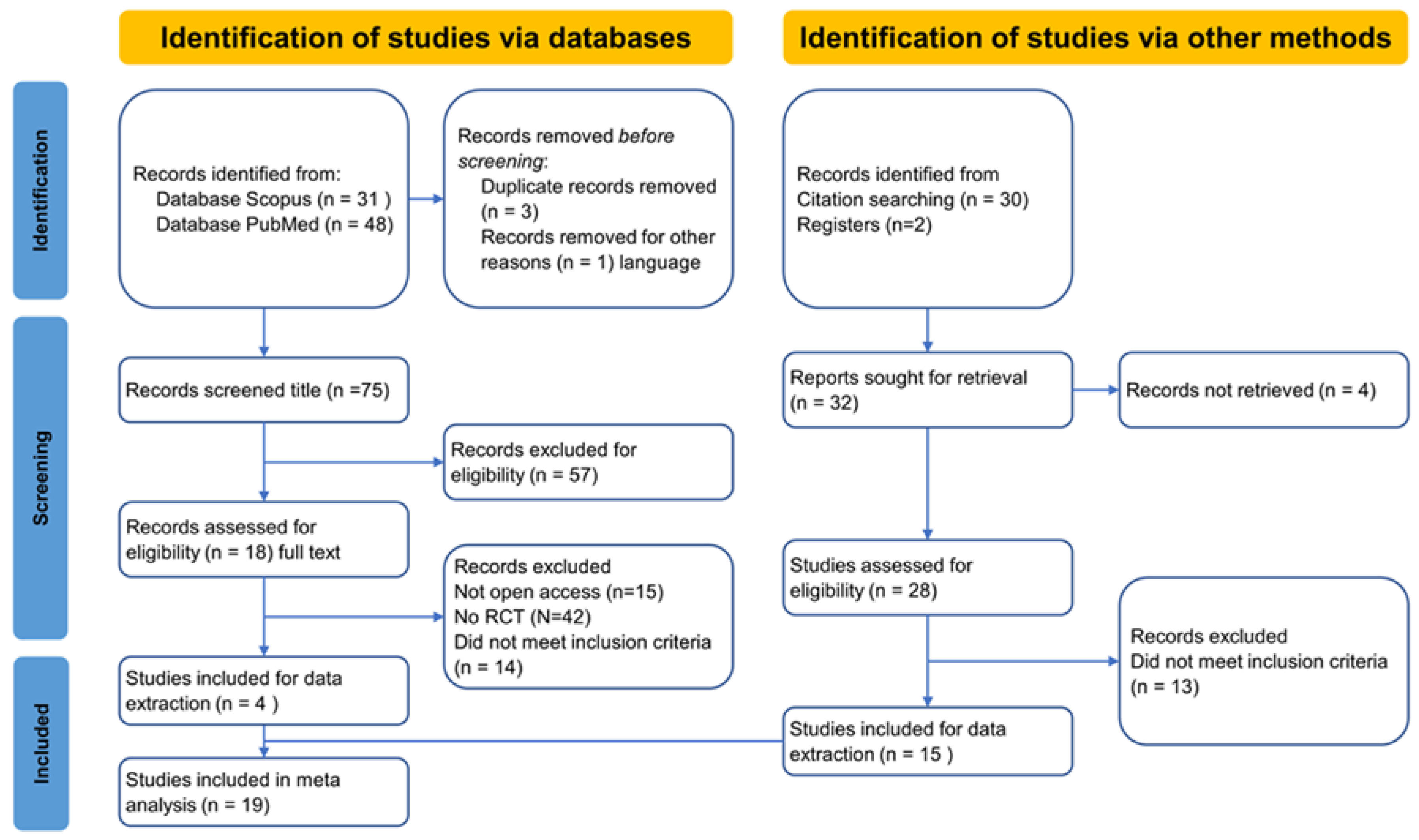
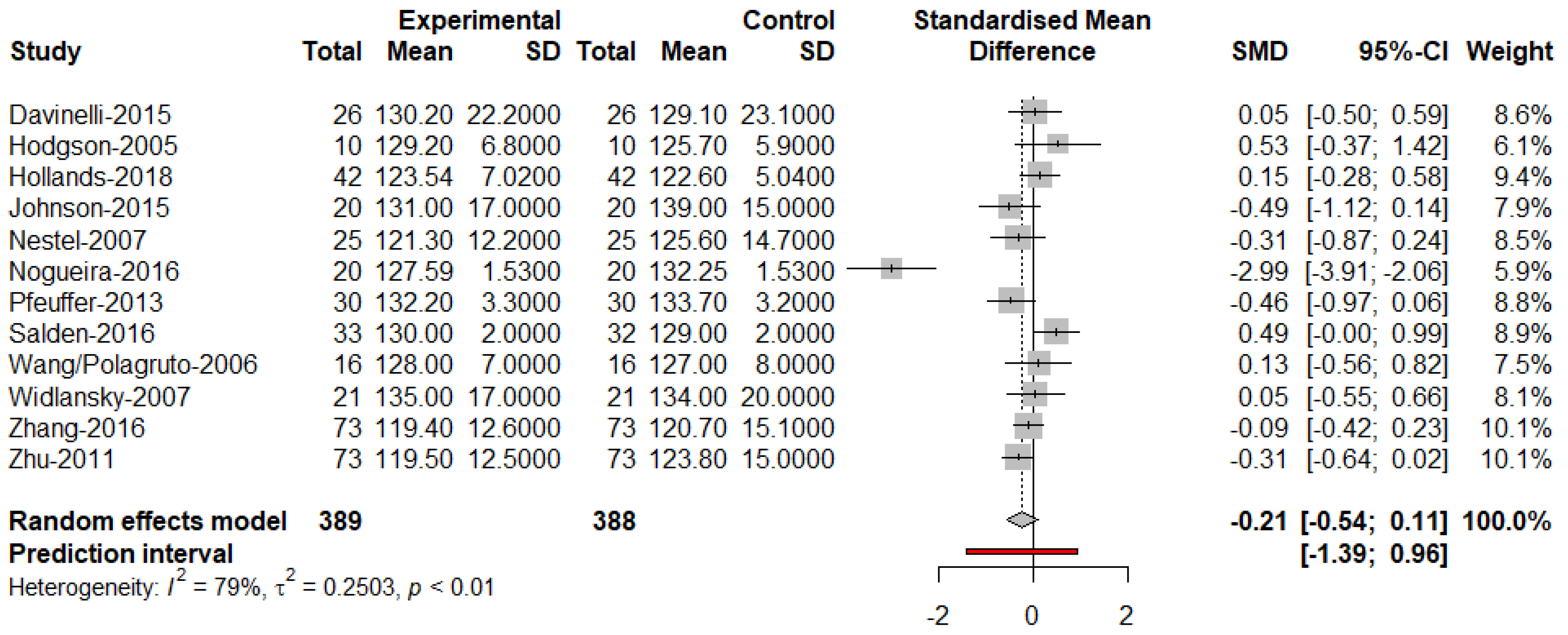
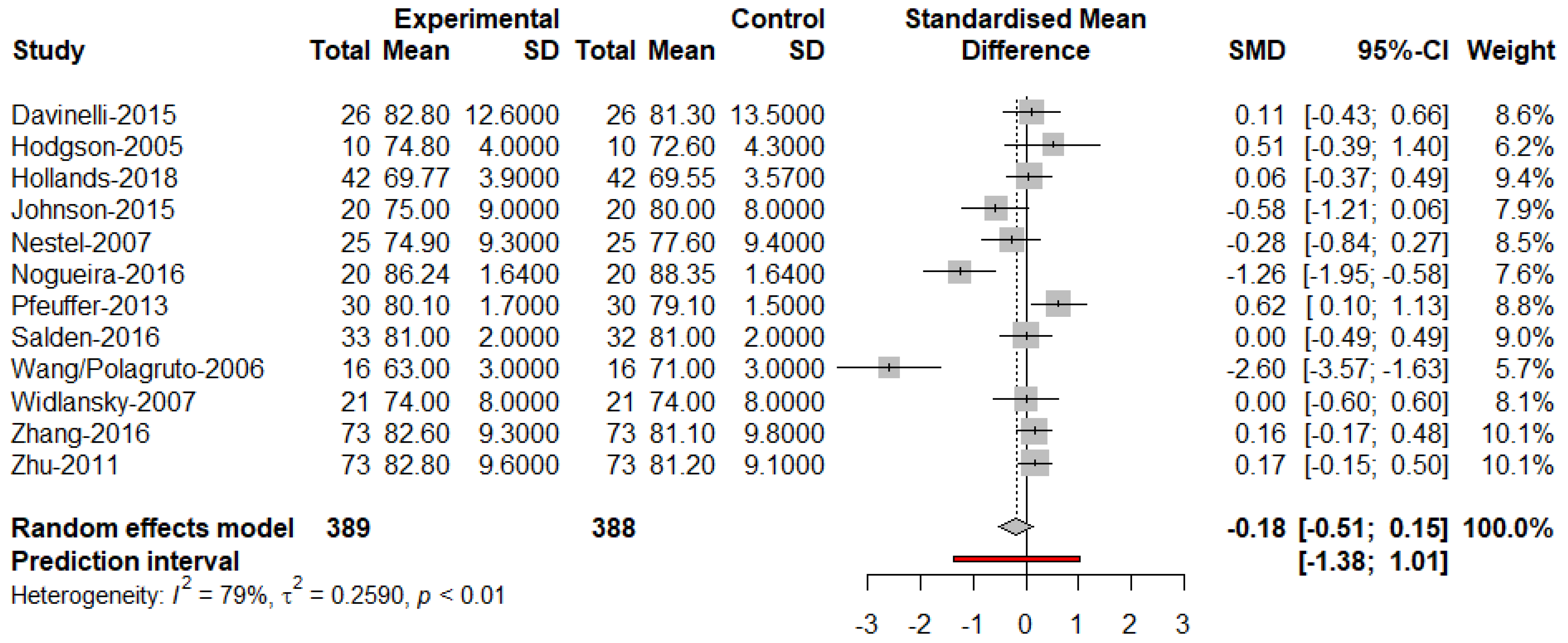
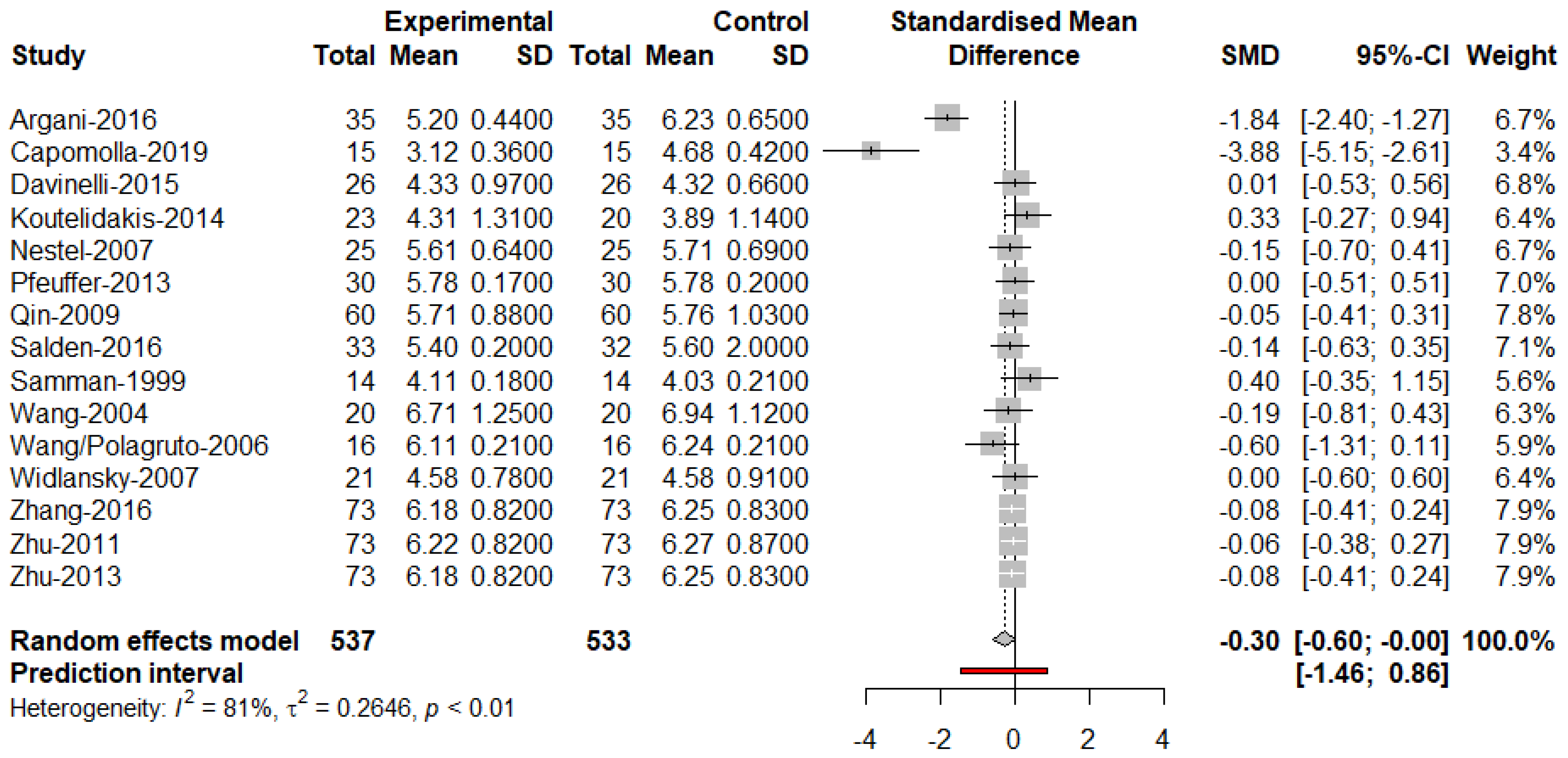
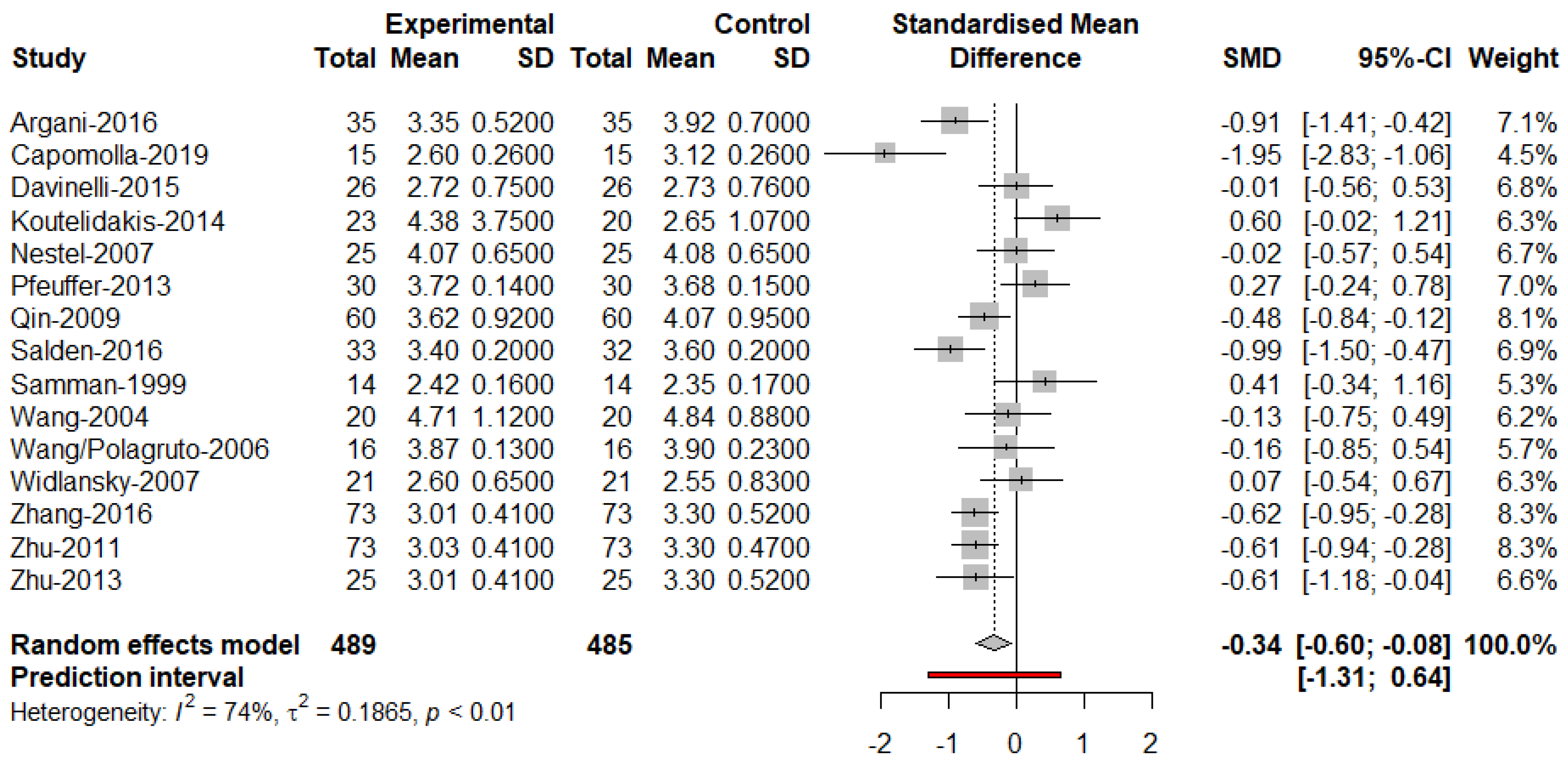
3. Results
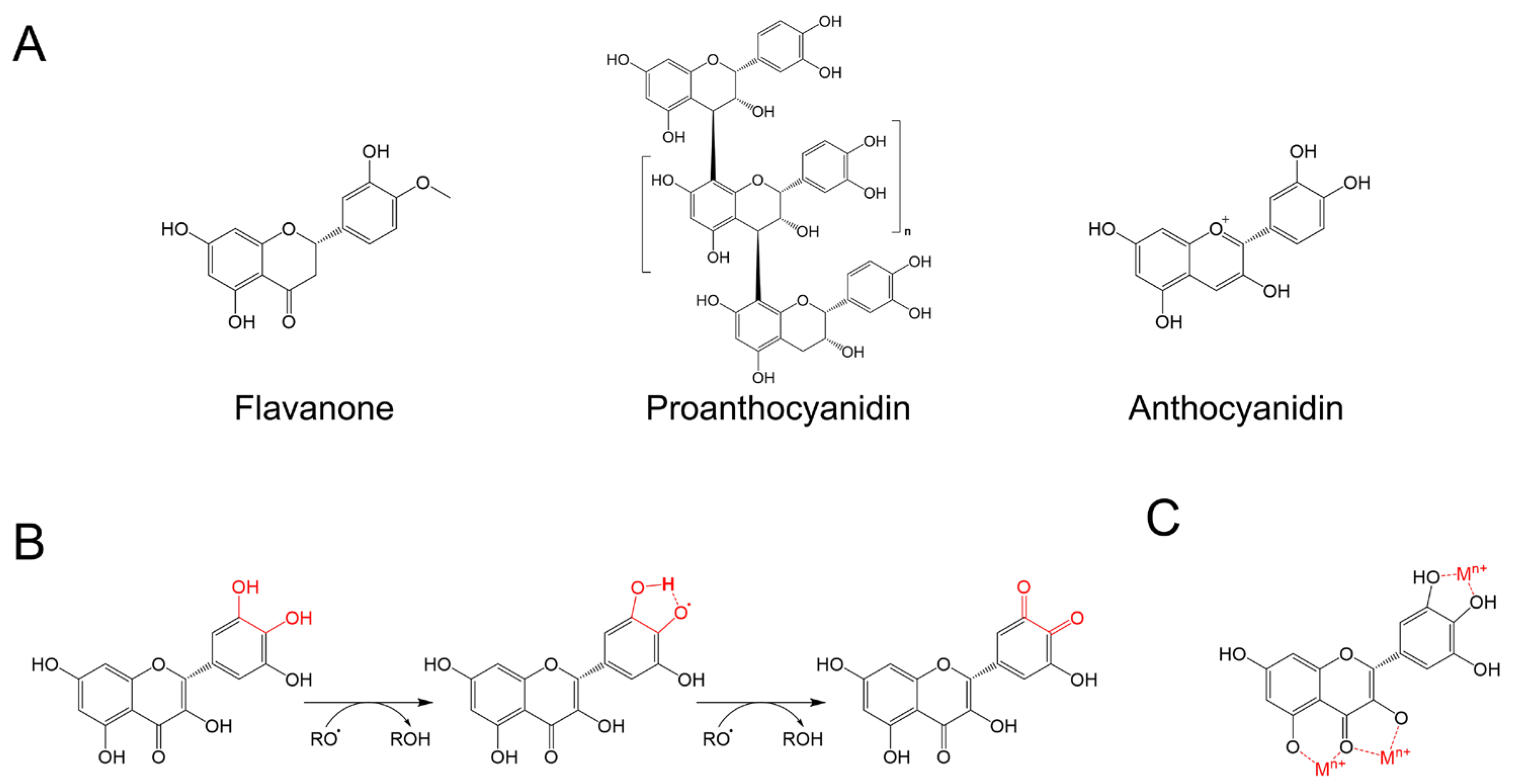
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Available online: https://www.who.int (accessed on 11 March 2023).
- Chrysant, S.G. A New Paradigm in the Treatment of the Cardiovascular Disease Continuum: Focus on Prevention. Hippokratia 2011, 15, 7–11. [Google Scholar] [PubMed]
- Libby, P. Inflammation and Cardiovascular Disease Mechanisms. Am. J. Clin. Nutr. 2006, 83, 456S–460S. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Bai, N.; Ma, Y.; Zhong, P.-Y.; Shang, Y.-S.; Wang, Z.-L. Safety and Efficacy of Anti-Inflammatory Therapy in Patients with Coronary Artery Disease: A Systematic Review and Meta-Analysis. BMC Cardiovasc. Disord. 2022, 22, 84. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Rahman, T.; Bukhari, S.M.A.; Herrera, E.C.; Awuah, W.A.; Lawrence, J.; de Andrade, H.; Patel, N.; Shah, R.; Shaikh, R.; Capriles, C.A.A.; et al. Lipid Lowering Therapy: An Era Beyond Statins. Curr. Probl. Cardiol. 2022, 47, 101342. [Google Scholar] [CrossRef] [PubMed]
- Szczepańska, E.; Białek-Dratwa, A.; Janota, B.; Kowalski, O. Dietary Therapy in Prevention of Cardiovascular Disease (CVD)-Tradition or Modernity? A Review of the Latest Approaches to Nutrition in CVD. Nutrients 2022, 14, 2649. [Google Scholar] [CrossRef]
- Sharif, H.; Akash, M.S.H.; Rehman, K.; Irshad, K.; Imran, I. Pathophysiology of Atherosclerosis: Association of Risk Factors and Treatment Strategies Using Plant-Based Bioactive Compounds. J. Food Biochem. 2020, 44, e13449. [Google Scholar] [CrossRef]
- Hoensch, H.P.; Oertel, R. The Value of Flavonoids for the Human Nutrition: Short Review and Perspectives. Clin. Nutr. Exp. 2015, 3, 8–14. [Google Scholar] [CrossRef]
- Pangestika, I.; Oksal, E.; Tengku Muhammad, T.S.; Amir, H.; Syamsumir, D.F.; Wahid, M.E.A.; Andriani, Y. Inhibitory effects of tangeretin and trans-ethyl caffeate on the HMG-CoA reductase activity: Potential agents for reducing cholesterol levels. Saudi Journal of Biological Sciences. Saudi J. Biol. Sci. 2020, 27, 1947–1960. [Google Scholar] [CrossRef]
- Ma, C.; Zhang, J.; Yang, S.; Hua, Y.; Su, J.; Shang, Y.; Fan, G. Astragalus Flavone Ameliorates Atherosclerosis and Hepatic Steatosis via Inhibiting Lipid-Disorder and Inflammation in apoE-/- Mice. Front. Pharmacol. 2020, 11, 610550. [Google Scholar] [CrossRef]
- Georgiev, V.; Ananga, A.; Tsolova, V. Recent Advances and Uses of Grape Flavonoids as Nutraceuticals. Nutrients 2014, 6, 391–415. [Google Scholar] [CrossRef]
- Côrtes, S.F.; Rezende, B.A.; Corriu, C.; Medeiros, I.A.; Teixeira, M.M.; Lopes, M.J.; Lemos, V.S. Pharmacological Evidence for the Activation of Potassium Channels as the Mechanism Involved in the Hypotensive and Vasorelaxant Effect of Dioclein in Rat Small Resistance Arteries. Br. J. Pharmacol. 2001, 133, 849–858. [Google Scholar] [CrossRef]
- Neveu, V.; Perez-Jiménez, J.; Vos, F.; Crespy, V.; du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An Online Comprehensive Database on Polyphenol Contents in Foods. Database 2010, 2010, bap024. [Google Scholar] [CrossRef] [PubMed]
- Esmaillzadeh, A.; Azadbakht, L. Major Dietary Patterns in Relation to General Obesity and Central Adiposity among Iranian Women. J. Nutr. 2008, 138, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Habauzit, V.; Verny, M.-A.; Milenkovic, D.; Barber-Chamoux, N.; Mazur, A.; Dubray, C.; Morand, C. Flavanones Protect from Arterial Stiffness in Postmenopausal Women Consuming Grapefruit Juice for 6 Mo: A Randomized, Controlled, Crossover Trial. Am. J. Clin. Nutr. 2015, 102, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.; Kroon, P.A.; Rimm, E.B.; Cohn, J.S.; Harvey, I.; Le Cornu, K.A.; Ryder, J.J.; Hall, W.L.; Cassidy, A. Flavonoids, flavonoid-rich foods, and cardiovascular risk: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2008, 88, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Wei, H.; He, B. Dietary flavonoids intake and the risk of coronary heart disease: A dose-response meta-analysis of 15 prospective studies. Thromb. Res. 2015, 135, 459–463. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.M.; Liu, Y.J.; Huang, Y.; Yu, H.J.; Yuan, S.; Tang, B.W.; Wang, P.G.; He, Q.Q. Dietary total flavonoids intake and risk of mortality from all causes and cardiovascular disease in the general population: A systematic review and meta-analysis of cohort studies. Mol. Nutr. Food Res. 2017, 61, 6. [Google Scholar] [CrossRef] [PubMed]
- Micek, A.; Godos, J.; Del Rio, D.; Galvano, F.; Grosso, G. Dietary Flavonoids and Cardiovascular Disease: A Comprehensive Dose-Response Meta-Analysis. Mol. Nutr. Food Res. 2021, 65, e2001019. [Google Scholar] [CrossRef]
- Thiriet, C.; Mahjoub, K.; Courte, G.; Labroca, P.; Cravoisy, A.; Lemarie, J.; Conrad, M.; Nace, L.; Bollaert, P.-E.; Gibot, S. Automated Measurement of Neutrophil CD64 Expression for Diagnosing Sepsis in Critically Ill Patients. Minerva Anestesiol. 2019, 85, 943–950. [Google Scholar] [CrossRef]
- Zhu, Y.; Xia, M.; Yang, Y.; Liu, F.; Li, Z.; Hao, Y.; Mi, M.; Jin, T.; Ling, W. Purified Anthocyanin Supplementation Improves Endothelial Function via NO-CGMP Activation in Hypercholesterolemic Individuals. Clin. Chem. 2011, 57, 1524–1533. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- R: A Language and Environment for Statistical Computing. Available online: https://www.gbif.org/tool/81287/r-a-language-and-environment-for-statistical-computing (accessed on 30 August 2023).
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. Introduction to Meta-Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2021; ISBN 978-1-119-55838-5. [Google Scholar]
- Samman, S.; Lyons Wall, P.M.; Chan, G.S.; Smith, S.J.; Petocz, P. The effect of supplementation with isoflavones on plasma lipids and oxidisability of low density lipoprotein in premenopausal women. Atherosclerosis 1999, 147, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jones, P.J.; Ausman, L.M.; Lichtenstein, A.H. Soy protein reduces triglyceride levels and triglyceride fatty acid fractional synthesis rate in hypercholesterolemic subjects. Atherosclerosis 2004, 173, 269–275. [Google Scholar] [CrossRef]
- Hodgson, J.M.; Burke, V.; Puddey, I.B. Acute Effects of Tea on Fasting and Postprandial Vascular Function and Blood Pressure in Humans. J. Hypertens. 2005, 23, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Wang-Polagruto, J.F.; Villablanca, A.C.; Polagruto, J.A.; Lee, L.; Holt, R.R.; Schrader, H.R.; Ensunsa, J.L.; Steinberg, F.M.; Schmitz, H.H.; Keen, C.L. Chronic Consumption of Flavanol-Rich Cocoa Improves Endothelial Function and Decreases Vascular Cell Adhesion Molecule in Hypercholesterolemic Postmenopausal Women. J. Cardiovasc. Pharmacol. 2006, 47 (Suppl. S2), S177–S186. [Google Scholar] [CrossRef] [PubMed]
- Nestel, P.; Fujii, A.; Zhang, L. An Isoflavone Metabolite Reduces Arterial Stiffness and Blood Pressure in Overweight Men and Postmenopausal Women. Atherosclerosis 2007, 192, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Widlansky, M.E.; Hamburg, N.M.; Anter, E.; Holbrook, M.; Kahn, D.F.; Elliott, J.G.; Keaney, J.F.; Vita, J.A. Acute EGCG Supplementation Reverses Endothelial Dysfunction in Patients with Coronary Artery Disease. J. Am. Coll. Nutr. 2007, 26, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Xia, M.; Ma, J.; Hao, Y.; Liu, J.; Mou, H.; Cao, L.; Ling, W. Anthocyanin Supplementation Improves Serum LDL- and HDL-Cholesterol Concentrations Associated with the Inhibition of Cholesteryl Ester Transfer Protein in Dyslipidemic Subjects. Am. J. Clin. Nutr. 2009, 90, 485–492. [Google Scholar] [CrossRef]
- Pfeuffer, M.; Auinger, A.; Bley, U.; Kraus-Stojanowic, I.; Laue, C.; Winkler, P.; Rüfer, C.E.; Frank, J.; Bösch-Saadatmandi, C.; Rimbach, G.; et al. Effect of Quercetin on Traits of the Metabolic Syndrome, Endothelial Function and Inflammation in Men with Different APOE Isoforms. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 403–409. [Google Scholar] [CrossRef]
- Zhu, Y.; Ling, W.; Guo, H.; Song, F.; Ye, Q.; Zou, T.; Li, D.; Zhang, Y.; Li, G.; Xiao, Y.; et al. Anti-Inflammatory Effect of Purified Dietary Anthocyanin in Adults with Hypercholesterolemia: A Randomized Controlled Trial. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 843–849. [Google Scholar] [CrossRef]
- Koutelidakis, A.E.; Rallidis, L.; Koniari, K.; Panagiotakos, D.; Komaitis, M.; Zampelas, A.; Anastasiou-Nana, M.; Kapsokefalou, M. Effect of green tea on postprandial antioxidant capacity, serum lipids, C-reactive protein and glucose levels in patients with coronary artery disease. Eur. J. Nutr. 2014, 53, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Davinelli, S.; Bertoglio, J.C.; Zarrelli, A.; Pina, R.; Scapagnini, G. A Randomized Clinical Trial Evaluating the Efficacy of an Anthocyanin-Maqui Berry Extract (Delphinol®) on Oxidative Stress Biomarkers. J. Am. Coll. Nutr. 2015, 34 (Suppl. S1), 28–33. [Google Scholar] [CrossRef]
- Johnson, S.A.; Figueroa, A.; Navaei, N.; Wong, A.; Kalfon, R.; Ormsbee, L.T.; Feresin, R.G.; Elam, M.L.; Hooshmand, S.; Payton, M.E.; et al. Daily blueberry consumption improves blood pressure and arterial stiffness in postmenopausal women with pre- and stage 1-hypertension: A randomized, double-blind, placebo-controlled clinical trial. J. Acad. Nutr. Diet. 2015, 115, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhu, Y.; Song, F.; Yao, Y.; Ya, F.; Li, D.; Ling, W.; Yang, Y. Effects of Purified Anthocyanin Supplementation on Platelet Chemokines in Hypocholesterolemic Individuals: A Randomized Controlled Trial. Nutr. Metab. 2016, 13, 86. [Google Scholar] [CrossRef]
- Argani, H.; Ghorbanihaghjo, A.; Vatankhahan, H.; Rashtchizadeh, N.; Raeisi, S.; Ilghami, H. The Effect of Red Grape Seed Extract on Serum Paraoxonase Activity in Patients with Mild to Moderate Hyperlipidemia. Sao Paulo Med. J. 2016, 134, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, L.P.; Nogueira, J.F.; Klein, M.R.S.T.; Sanjuliani, A.F. Short-term Effects of Green Tea on Blood Pressure, Endothelial Function, and Metabolic Profile in Obese Prehypertensive Women: A Crossover Randomized Clinical Trial. J. Am. Coll. Nutr. 2016, 36, 108–115. [Google Scholar] [CrossRef]
- Salden, B.N.; Troost, F.J.; de Groot, E.; Stevens, Y.R.; Garcés-Rimón, M.; Possemiers, S.; Winkens, B.; Masclee, A.A. Randomized Clinical Trial on the Efficacy of Hesperidin 2S on Validated Cardiovascular Biomarkers in Healthy Overweight Individuals. Am. J. Clin. Nutr. 2016, 104, 1523–1533. [Google Scholar] [CrossRef] [PubMed]
- Hollands, W.J.; Tapp, H.; Defernez, M.; Perez Moral, N.; Winterbone, M.S.; Philo, M.; Lucey, A.J.; Kiely, M.E.; Kroon, P.A. Lack of Acute or Chronic Effects of Epicatechin-Rich and Procyanidin-Rich Apple Extracts on Blood Pressure and Cardiometabolic Biomarkers in Adults with Moderately Elevated Blood Pressure: A Randomized, Placebo-Controlled Crossover Trial. Am. J. Clin. Nutr. 2018, 108, 1006–1014. [Google Scholar] [CrossRef]
- Capomolla, A.S.; Janda, E.; Paone, S.; Parafati, M.; Sawicki, T.; Mollace, R.; Ragusa, S.; Mollace, V. Atherogenic Index Reduction and Weight Loss in Metabolic Syndrome Patients Treated with A Novel Pectin-Enriched Formulation of Bergamot Polyphenols. Nutrients 2019, 11, 1271. [Google Scholar] [CrossRef]
- Grijalva-Guiza, R.E.; Jiménez-Garduño, A.M.; Hernández, L.R. Potential Benefits of Flavonoids on the Progression of Atherosclerosis by Their Effect on Vascular Smooth Muscle Excitability. Molecules 2021, 26, 3557. [Google Scholar] [CrossRef]
- Weggemans, R.M.; Trautwein, E.A. Relation between soy-associated isoflavones and LDL and HDL cholesterol concentrations in humans: A meta-analysis. Eur. J. Clin. Nutr. 2003, 57, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, X.G.; Melby, M.K.; Watanabe, S. Soy isoflavone intake lowers serum LDL cholesterol: A meta-analysis of 8 randomized controlled trials in humans. J. Nutr. 2004, 134, 2395–2400. [Google Scholar] [CrossRef] [PubMed]
- Taku, K.; Umegaki, K.; Sato, Y.; Taki, Y.; Endoh, K.; Watanabe, S. Soy isoflavones lower serum total and LDL cholesterol in humans: A meta-analysis of 11 randomized controlled trials. Am. J. Clin. Nutr. 2007, 85, 1148–1156, Correction in Am. J. Clin. Nutr. 2007, 86, 809.. [Google Scholar] [CrossRef] [PubMed]
- Raman, G.; Avendano, E.E.; Chen, S.; Wang, J.; Matson, J.; Gayer, B.; Novotny, J.A.; Cassidy, A. Dietary Intakes of Flavan-3-Ols and Cardiometabolic Health: Systematic Review and Meta-Analysis of Randomized Trials and Prospective Cohort Studies. Am. J. Clin. Nutr. 2019, 110, 1067–1078. [Google Scholar] [CrossRef]
- Serban, M.-C.; Sahebkar, A.; Zanchetti, A.; Mikhailidis, D.P.; Howard, G.; Antal, D.; Andrica, F.; Ahmed, A.; Aronow, W.S.; Muntner, P.; et al. Effects of Quercetin on Blood Pressure: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Am. Heart Assoc. 2016, 5, e002713. [Google Scholar] [CrossRef]
- Daneshzad, E.; Shab-Bidar, S.; Mohammadpour, Z.; Djafarian, K. Effect of Anthocyanin Supplementation on Cardio-Metabolic Biomarkers: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Clin. Nutr. 2019, 38, 1153–1165. [Google Scholar] [CrossRef]
- Sone, T.; Kuriyama, S.; Nakaya, N.; Hozawa, A.; Shimazu, T.; Nomura, K.; Rikimaru, S.; Tsuji, I. Randomized Controlled Trial for an Effect of Catechin-Enriched Green Tea Consumption on Adiponectin and Cardiovascular Disease Risk Factors. Food Nutr. Res. 2011, 55, 8326. [Google Scholar] [CrossRef]
| Author | Flavonoid or Class of Flavonoid | Participant | Treatment | RCT Analysis Style | Participants Characteristics | ||
|---|---|---|---|---|---|---|---|
| Sex | Age | Length | Dose mg/d | ||||
| Samman et al., 1999 [23] | Isoflavones | F | 18–45 | 4 months | 86 | Single blind, crossover | Premenopausal women |
| Wang et al., 2004 [24] | Isoflavones | M, F | Over fifty | 6 weeks | 92 | Double blind, crossover | Hypercholesterolemic subjects |
| Hodgson, 2005 [25] | Catechins | M, F | 45–70 | 4 weeks | 300 | Single blind, parallel | History of coronary artery disease |
| Wang-Polagruto, 2006 [26] | Flavanols | F | 53–59 | 6 weeks | 446 | Double blind, parallel | Hypercholescterolemic postmenopausal women |
| Nestel et al., 2007 [27] | Isoflavones | M, F | 50–64 | 5 weeks | 1000 | Double blind, crossover | Overweight men and postmenopausal women |
| Widlansky et al., 2007 [28] | Catechin | M, F | 48–68 | 2 weeks | 300 | Double blind | Men and women with coronary artery disease |
| Qin et al., 2009 [29] | Anthocyanins | M, F | 40–65 | 12 weeks | 320 | Double blind, parallel | Dyslipidemic patients |
| Zhu et al., 2011 [21] | Anthocyanin | M, F | 40–65 | 12 weeks | 320 | Double blind, parallel | Hypercholesterolemic individuals |
| Pfeuffer et al., 2013 [30] | Flavonols | M | 48–68 | 8 weeks | 150 | Double blind, crossover | Men with APOE4 polymorphism |
| Zhu et al., 2013 [31] | Anthocyanin | M, F | 40–65 | 24 weeks | 320 | Double blind, parallel | Subjects with hypercholesterolemia |
| Koutelidakis et al., 2014 [32] | Catechins | M, F | 45–70 | 3 hours | 400 | Single blind, parallel | Patients with coronary artery disease |
| Davinelli et al., 2015 [33] | Anthocyanin | M, F | 45–65 | 4 weeks | 162 | Double blind, parallel | Overweight and smokers |
| Johnson et al., 2015 [34] | Anthocyanin | F | 45–65 | 8 weeks | 103.2 | Double blind, parallel | Promenopausal women with pre and stage 1 hypertension |
| Zhang et al., 2016 [35] | Anthocyanin | M, F | 40–65 | 24 weeks | 320 | Double blind, parallel | Hypercholesterolemic individuals |
| Argani et al., 2016 [36] | Proanthocyanidins | M, F | 21–64 | 8 weeks | 190 | Double blind, parallel | Moderate hyperlipidemia |
| Nogueira et al., 2016 [37] | Catechins | F | 18–59 | 4 weeks | 780 | Double blind, crossover | Prehypertensive obese individuals |
| Salden et al., 2016 [38] | Flavanone | M, F | 40–68 | 6 weeks | 450 | Double blind, parallel | Overweight individuals |
| Hollands et al., 2018 [39] | Procyanidin | M, F | 56–70 | 4 weeks | 130 | Double blind, crossover | Men and Women with moderately high blood pressure |
| Capomolla et al., 2019 [40] | Flavanones | M, F | 40–80 | 90 days | 650, 1300 | Double blind | Metabolic syndrome, obese, and mild hyperglycemia |
| Risk of Bias | From the Randomization Process | Due to Deviations from Intended Interventions | Due to Missing Outcome Data | In Measurements of the Outcome | In Selection of the Reported Result | |
|---|---|---|---|---|---|---|
| Study | ||||||
| Argani et al., 2016 [38] | + | + | + | + | + | |
| Davinelli et al., 2015 [35] | + | + | + | + | + | |
| Hodgson 2005 [27] | + | + | + | + | + | |
| Hollands et al., 2018 [41] | ? | + | + | + | + | |
| Johnson et al., 2015 [36] | + | + | + | + | + | |
| Koutelidakis et al., 2014 [34] | + | + | + | + | + | |
| Nestel et al., 2007 [29] | + | + | + | + | + | |
| Nogueira et al., 2016 [39] | + | + | + | + | + | |
| Pfeuffer et al., 2013 [32] | ? | ? | + | + | + | |
| Qin et al., 2009 [31] | + | + | + | + | + | |
| Salden et al., 2016 [40] | + | + | + | + | + | |
| Samman et al., 1999 [25] | + | + | + | + | + | |
| Capomolla et al., 2019 [42] | ? | + | + | + | + | |
| Wang et al., 2004 [26] | + | + | + | + | + | |
| Wang-Polagruto 2006 [28] | + | + | + | + | + | |
| Widlansky et al., 2007 [30] | + | + | + | + | + | |
| Zhang et al., 2016 [37] | + | + | + | + | + | |
| Zhu et al., 2011 [21] | ? | + | + | + | + | |
| Zhu et al., 2013 [33] | + | + | + | + | + | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grijalva-Guiza, R.E.; Grijalva-Montano, T.L.; Cuautle, M.; Quiroga-González, E.; Hernández, L.R.; Ortega Aguilar, A.; Jiménez-Garduño, A.M. Analysis of Beneficial Effects of Flavonoids in Patients with Atherosclerosis Risk on Blood Pressure or Cholesterol during Random Controlled Trials: A Systematic Review and Meta-Analysis. Sci. Pharm. 2023, 91, 55. https://doi.org/10.3390/scipharm91040055
Grijalva-Guiza RE, Grijalva-Montano TL, Cuautle M, Quiroga-González E, Hernández LR, Ortega Aguilar A, Jiménez-Garduño AM. Analysis of Beneficial Effects of Flavonoids in Patients with Atherosclerosis Risk on Blood Pressure or Cholesterol during Random Controlled Trials: A Systematic Review and Meta-Analysis. Scientia Pharmaceutica. 2023; 91(4):55. https://doi.org/10.3390/scipharm91040055
Chicago/Turabian StyleGrijalva-Guiza, Rosa Edith, Thais Lucía Grijalva-Montano, Mariana Cuautle, Enrique Quiroga-González, Luis Ricardo Hernández, Alicia Ortega Aguilar, and Aura Matilde Jiménez-Garduño. 2023. "Analysis of Beneficial Effects of Flavonoids in Patients with Atherosclerosis Risk on Blood Pressure or Cholesterol during Random Controlled Trials: A Systematic Review and Meta-Analysis" Scientia Pharmaceutica 91, no. 4: 55. https://doi.org/10.3390/scipharm91040055
APA StyleGrijalva-Guiza, R. E., Grijalva-Montano, T. L., Cuautle, M., Quiroga-González, E., Hernández, L. R., Ortega Aguilar, A., & Jiménez-Garduño, A. M. (2023). Analysis of Beneficial Effects of Flavonoids in Patients with Atherosclerosis Risk on Blood Pressure or Cholesterol during Random Controlled Trials: A Systematic Review and Meta-Analysis. Scientia Pharmaceutica, 91(4), 55. https://doi.org/10.3390/scipharm91040055








