Detailing the Ten Main Professional Roles of a Pharmacist to Provide the Scope of Professional Functions
Abstract
1. Introduction
2. Materials and Methods
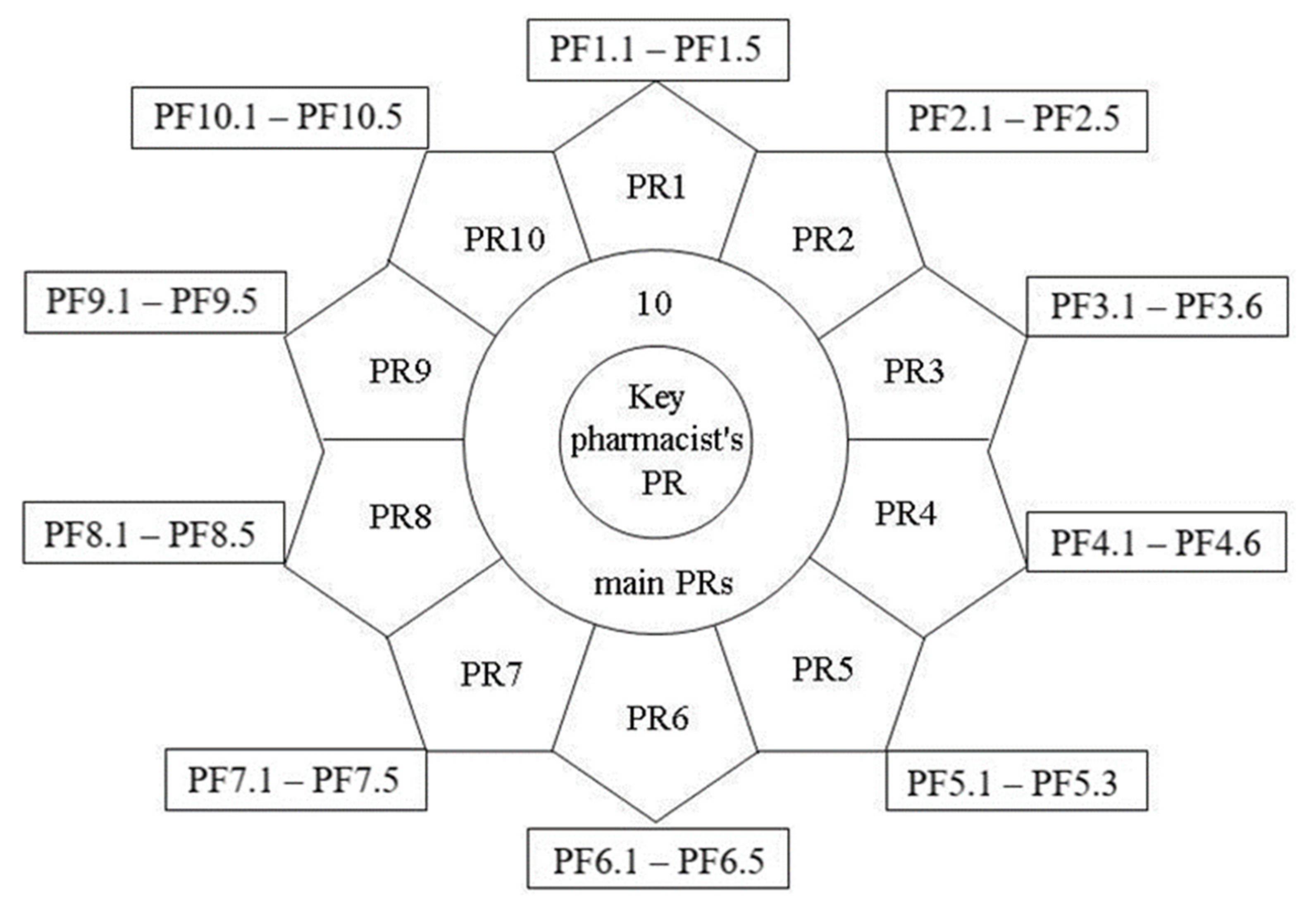
3. Results
3.1. Caregiver
- –
- assess the state of health, the level of his medical literacy, and physiological needs for the quality of pharmaceutical care (PF1.1);
- –
- to ensure the implementation of possible diagnostic measures (measurement of blood pressure, heart rate, blood sugar, etc.) in a pharmaceutical institution (PF1.2);
- –
- provide quality pharmaceutical care through a set of its characteristics (appropriate therapeutic concordance, patient-oriented results, economic and physical accessibility, rationality, safety, effectiveness, timeliness, absence (minimization) of drug-related mistakes, continuity) (PF1.3);
- –
- have the skills of telepharmacy, i.e., remote (distant) provision of quality pharmaceutical care with the assistance of a range of organizational and financial measures, information and telecommunications technologies, and infrastructure (PF1.4);
- –
- take professional responsibility for the quality of pharmaceutical care, including in emergencies, pandemics, and peacetime and wartime emergencies, as well as for reducing environmental risk throughout the drug supply circulation chain (PF1.5).
3.2. Decision Maker
- –
- rational, cost-effective procurement and efficient, safe, and economical use of all resources in the pharmaceutical organization (medicines, medical devices, active pharmaceutical ingredients, excipients, etc.) (PF2.1);
- –
- decision-making on the proper organization of the pharmaceutical organization and participation in the production (manufactured) of medicines in the conditions of pharmaceutical enterprises and pharmacies (PF2.2);
- –
- determination of factors influencing the processes of absorption, distribution, deposition, metabolism, and excretion of drugs due to the condition, features of the human body, and physicochemical properties of drugs (PF2.3);
- –
- the adoption of safe and logical decisions based on proper evaluation of evidence-based medicine and pharmacy data and professional judgments, including the choice of a rational type of pharmaceutical care, which minimizes the risk of pharmacotherapy for humans and promotes the provision of quality pharmaceutical care (PF2.4);
- –
- influence on the implementation of the national medical policy, through active participation in the activities of pharmaceutical public and professional organizations, as a consequence of promoting the improvement of the regulatory framework of the pharmaceutical sector of the health sector (PF2.5).
3.3. Communicator
- –
- possess and apply in practice verbal and nonverbal skills of interpersonal communication and fundamental principles of pharmaceutical ethics and deontology (PF3.1);
- –
- establish regular bilateral and multilateral communications in the process of pharmaceutical care aimed at doctors, medical staff, patients, or their caregivers (PF3.2);
- –
- show tolerance, loyalty, respect, compassion, and understanding to the patient regardless of nationality, political and religious beliefs, property status, gender, age, and social status (PF3.3);
- –
- keep confidential information about the condition, health, and diagnosis of the patient, which is a professional secret, except as provided by law (PF3.4);
- –
- ensure that information for patients, doctors, other health professionals, and the public is based on evidence-based medicine and is objective, effective, unbiased, understandable, non-promotional, accurate, and adequate (PF3.5);
- –
- to carry out information work among the population to prevent the use of potentially harmful substances for recreational purposes, to prevent substance abuse, prevent common diseases, prevent dangerous infectious, viral and parasitic diseases, as well as to promote timely detection and maintenance of treatment for these diseases, their medical and biological characteristics and microbiological features (PF3.6).
3.4. Manager
- –
- to carry out planning, i.e., determine the prospects and future state (landmark of future activities) of the pharmaceutical organization (PF4.1);
- –
- to be able to organize, i.e., create an adequate structure of the pharmaceutical organization by dividing it into units according to goals and strategies, and to establish the relationship of powers of higher and lower levels of government and opportunities for division and coordination of tasks (PF4.2);
- –
- to motivate, i.e., to ensure the process of motivating yourself and other members of the pharmaceutical organization to activities aimed at achieving goals, both personal and for the whole organization (PF4.3);
- –
- to control, i.e., determine how to make correct management decisions, and determine, as well, the need to make certain adjustments (PF4.4);
- –
- to regulate, i.e., eliminate deviations, failures, shortcomings, etc., in the pharmaceutical organization by developing and implementing appropriate measures (PF4.5);
- –
- to adequately respond to managerial influence in the vertical and horizontal division of managerial activity (PF4.6).
3.5. Life-Long Learner
- –
- identify their learning needs and be individually responsible for the systematic acquisition, maintenance, development, and expansion of program competencies throughout their professional activities (PF5.1);
- –
- be active participants in the two stages of continuing pharmaceutical education (specialist training and continuing professional development) (PF5.2);
- –
- use three forms (formal, non-technical, and informal) of implementation and two forms (institutionalized and non-institutionalized) of education (PF5.3).
3.6. Teacher
- –
- transfer to the new generation of pharmaceutical specialists their knowledge, skills, and abilities, as well as encourage them to demonstrate independent cognitive activity, including through mentoring (PF6.1);
- –
- be able to properly organize the educational process in the pharmaceutical organization, which would be convenient for the new generation of pharmaceuticals, and would not interfere with the work of employees of this organization and their patients (PF6.2);
- –
- participate in educating patients on issues related to the protection and promotion of health, medical literacy, and effectively promote a healthy lifestyle using scientifically sound methods (PF6.3);
- –
- provide training to empower patients and their communities in self-care in health care, ultimately to help optimize the use of resources and costs for health care and to improve outcomes for patients and health care systems (PF6.4);
- –
- teach patients the rules of counteracting the distribution of counterfeit medicines and the disposal of pharmaceutical waste as an element of pharmaceutical practice (PF6.5).
3.7. Leader
- –
- have organizational skills, be able to gain trust and persuade members of the interdisciplinary medical team and the staff of the pharmaceutical organization, combine their efforts to achieve the goals of the multidisciplinary team and the pharmaceutical institution (PF7.1);
- –
- establish, maintain and coordinate favorable personal, professional, and socio-psychological relations within the team of the pharmaceutical organization and between team members, doctors, nurses and patients, and their caregivers in compliance with professional ethics, resolve conflicts in professional activities, and also support their colleagues at all stages of the work (PF7.2);
- –
- consciously introduce new, constructive ideas for the functioning and development of the staff of the pharmaceutical organization, including the formulation of new goals and objectives, justifying priorities in the development of the team, and tactical ways and methods of achieving them (PF7.3);
- –
- promote the achievement of the goals of the pharmaceutical organization, high performance, positive changes in colleagues, and be responsible for the results of collective activities (PF7.4);
- –
- promote the profession’s prestige, particularly its life position and professional competence, thus serving as a model of behavior for colleagues, members of a multidisciplinary medical team, and patients (PF7.5).
3.8. Researcher
- –
- understand the concept of construction and facilitate various types of research, make hypotheses, build theories, and obtain facts to implement the results of pharmaceutical research in the evidence base and practice (PF8.1);
- –
- search for and develop new or improve existing drugs, identify the advantages and disadvantages (unknown in practice, undesirable, threatening side effects) of drugs of different pharmacological groups, and recognize the need to apply the principles of pharmacovigilance (PF8.2);
- –
- develop new methods of quality control of medicines, including active pharmaceutical ingredients, medicinal plant raw materials, and excipients, predict and determine the impact of environmental factors on the quality of medicines and consumer characteristics of related products following their physicochemical properties, and recognize that these will be affected by the individual size, weight, gender, and age of consumers (PF8.3);
- –
- based on audit and evaluation of provided pharmaceutical services, develop and implement new approaches to the implementation of professional pharmaceutical activities, in particular through the quality management system in the pharmaceutical organization, and ensure their effective updating (PF8.4);
- –
- effectively use the modern system of evidence on medicines to provide recommendations for the rational use of medicines in a multidisciplinary medical team, and contribute to the evidence base by accumulating an array of evidence-based scientific and pharmaceutical information, disseminating it in multidisciplinary cooperation to improve the treatment of patients (PF8.5).
3.9. Entrepreneur
- –
- take the initiative to combine financial, production, material, raw materials, human, informational, intellectual, and other resources in the process of circulation of medicines (PF9.1);
- –
- generate innovations (novelty), master new medicines, technologies, and forms of pharmaceutical business organization, search for new markets, means of meeting consumer needs, the transition from traditional to new forms of management (PF9.2);
- –
- risk one’s property, ownership, invested funds, and one’s labor, time, and business reputation in the course of entrepreneurial activity (PF9.3);
- –
- make decisions at the stages of circulation of medicines, which are aimed at achieving the success of the pharmaceutical organization, but do not guarantee it due to the uncertainty and variability of the economic situation (PF9.4);
- –
- implement the trinity of the pharmaceutical business to obtain its economic benefits and ensure the economic well-being of its employees and the maximum satisfaction of consumer needs (PF9.5).
3.10. Agent of Positive Change
- –
- in-depth analysis of the state of development of pharmaceutical practice, identification of strategic and common priorities in ensuring the quality of pharmaceutical services, participation in the creation or improvement of national professional responsibilities, guidelines, and legislation that guide the pharmaceutical community to these priorities (PF10.1);
- –
- education of the pharmaceutical community on essential issues in the development of pharmaceutical practice, advising on difficult issues to which the answers are not obvious, cultivating a spirit of expertise, and involving other experts in creating common knowledge bases to improve patient care (PF10.2);
- –
- involvement of members of the pharmaceutical community in projects aimed at improving patient care, and quality of pharmaceutical services, using modern methods of online and offline channels (PF10.3);
- –
- active adaptation to new hybrid challenges of modern times, acting socially responsibly, in particular through participation in various public and social events guiding the transformation of human behavior to actively overcome complex threats (PF10.4);
- –
- motivation to work in public and professional pharmaceutical organizations on a volunteer basis (PF10.5).
4. Discussion
- three partial PF are characteristic of a life-long-learner;
- five partial PF—for a caregiver, a decision-maker, a teacher, a leader, a researcher, an entrepreneur, and an agent of positive change;
- six partial PF—for a communicator and a manager.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dubiel-Zielińska, P. Moral dilemmas in professions of public trust and the assumptions of ethics of social consequences. Ethics Bioeth. 2016, 6, 19–32. [Google Scholar] [CrossRef]
- FIP Statement of Policy on Good Pharmacy Education Practice. Approved by FIP Council in Vienna. 2000, pp. 7–8. Available online: https://www.fip.org/file/1518 (accessed on 14 April 2022).
- The Role of the Pharmacist in the Health Care System: Preparing the Future Pharmacist: Curricular Development: Report of a Third WHO Consultative Group on the Role of the Pharmacist, Vancouver, Canada, 27–29 August 1997. p. 49. Available online: https://apps.who.int/iris/handle/10665/63817 (accessed on 14 April 2022).
- Nadeem, M.F.; Samanta, S.; Mustafa, F. Is the paradigm of community pharmacy practice expected to shift due to COVID-19? Res. Soc. Adm. Pharm. 2021, 17, 2046. [Google Scholar] [CrossRef] [PubMed]
- Chambliss, W.G.; Carroll, W.A.; Kennedy, D.; Levine, D.; Moné, M.A.; Ried, L.D.; Shepherd, M.; Yelvigi, M. Role of the pharmacist in preventing distribution of counterfeit medications. J. Am. Pharm. Assoc. 2012, 52, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Pottie, K.; Haydt, S.; Farrell, B.; Kennie, N.; Sellors, C.; Martin, C.; Dolovich, L. Pharmacist’s identity development within multidisciplinary primary health care teams in Ontario; qualitative results from the IMPACT project. Res. Soc. Adm. Pharm. 2009, 5, 319. [Google Scholar] [CrossRef]
- Gregory, P.A.M.; Teixeira, B.; Austin, Z. What does it take to change practice? Perspectives of pharmacists in Ontario. Can. Pharm. J. 2017, 151, 43–50. [Google Scholar] [CrossRef]
- Sam, A.T.; Parasuraman, S. The nine-star pharmacist: An overview. J. Young Pharm. 2015, 7, 281. [Google Scholar] [CrossRef]
- Kremin, Y.; Zaprutko, T.; Kus, K.; Hromovyk, B. Comparative analysis of the understanding the “ten-star pharmacist” concept by pharmaceutical specialists and pharmacy students of medical universities of Ukraine and Poland. JHPOR 2022, 2. [Google Scholar] [CrossRef]
- Mattingly, T.J., 2nd; Mullins, C.D.; Melendez, D.R.; Boyden, K.; Eddington, N.D. A Systematic Review of Entrepreneurship in Pharmacy Practice and Education. Am. J. Pharm. Educ. 2019, 83, 7233. [Google Scholar] [CrossRef]
- Hallit, S.; Hajj, A.; Sacre, H.; Zeenny, R.M.; Akel, M.; Sili, G.; Salameh, P. Emphasizing the Role of Pharmacist as a Researcher: The Lebanese Order of Pharmacists' Perspective. J. Res. Pharm. Pract. 2019, 8, 229–230. [Google Scholar] [CrossRef]
- Ghibu, S.; Juncan, A.M.; Rus, L.L.; Frum, A.; Dobrea, C.M.; Chiş, A.A.; Gligor, F.G.; Morgovan, C. The Particularities of Pharmaceutical Care in Improving Public Health Service during the COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2021, 18, 9776. [Google Scholar] [CrossRef]
- Grosman-Dziewiszek, P.; Wiatrak, B.; Jęśkowiak, I.; Szeląg, A. Patients’ Habits and the Role of Pharmacists and Telemedicine as Elements of a Modern Health Care System during the COVID-19 Pandemic. J. Clin. Med. 2021, 10, 4211. [Google Scholar] [CrossRef] [PubMed]
- DeRemer, C.E.; Reiter, J.; Olson, J.L. Transitioning ambulatory care pharmacy services to telemedicine while maintaining multidisciplinary collaborations. Am. J. Health Syst. Pharm. 2021, 78, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Abousheishaa, A.A.; Sulaiman, A.H.; Huri, H.Z.; Zaini, S.; Othman, N.A.; Aladdin, Z.; Guan, N.C. Global Scope of Hospital Pharmacy Practice: A Scoping Review. Healthcare 2020, 8, 143. [Google Scholar] [CrossRef] [PubMed]
- Khan, M. Role of Clinical Pharmacist in Context of World Health Organization against COVID-19. J. Pharm. Pract. Community Med. 2020, 6, 44–45. [Google Scholar] [CrossRef]
- Abubakar, U.; Sulaiman, S.A.; Usman, M.N.; Umar, M.D. Nigerian pharmacists' self-perceived competence and confidence to plan and conduct pharmacy practice research. Pharm. Pract. 2018, 16, 1152. [Google Scholar] [CrossRef]
- Nissen, L.; Singleton, J. Chapter 18-Leadership in Pharmacy Education. In Pharmacy Education in the Twenty First Century and Beyond; Academic Press: Cambridge, MA, USA, 2018; pp. 297–309. [Google Scholar] [CrossRef]
- Vears, D.F.; Gillam, L. Inductive content analysis: A guide for beginning qualitative researchers. Health Prof. Educ. 2022, 23, 111–127. [Google Scholar] [CrossRef]
- Van Eck, D.; McAdams, D.A.; Vermaas, P.E. Functional Decomposition in Engineering: A Survey. In Proceedings of the 19th International Conference on Design Theory and Methodology, Las Vegas, NV, USA, 4–7 September 2007; pp. 227–236. [Google Scholar] [CrossRef]
- Payne, G.; Williams, M. Generalization in Qualitative Research. Sociology 2005, 39, 295–314. [Google Scholar] [CrossRef]
- Luk, R.W.P. Understanding Scientific Study via Process Modeling. Found. Sci. 2010, 15, 49–78. [Google Scholar] [CrossRef]
- Yoghourdjian, V.; Archambault, D.; Diehl, S.; Dwyer, T.; Klein, K.; Purchase, H.C.; Wu, H.-Y. Exploring the limits of complexity: A survey of empirical studies on graph visualisation. Vis. Inform. 2018, 2, 264–282. [Google Scholar] [CrossRef]
- Chertes, A.; Crişan, O. Standards for good pharmacy practice—A comparative analysis. Farmacia 2019, 67, 545–550. [Google Scholar] [CrossRef]
- World Health Organization. Available online: https://www.who.int/docs/default-source/medicines/norms-and-standards/guidelines/distribution/trs961-annex8-fipwhoguidelinesgoodpharmacypractice.pdf (accessed on 3 April 2022).
- Guhl, D.; Blankart, K.E.; Stargardt, T. Service quality and perceived customer value in community pharmacies. Health Serv. Manag. Res. 2018, 32, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Newlands, R.S.; Power, A.; Young, L.; Watson, M. Quality improvement of community pharmacy services: A prioritisation exercise. Int. J. Pharm. Pract. 2017, 26, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Kremin, Y.I.; Hromovyk, B.P. Research of the relationship between educational and professional programs of higher pharmaceutical education in Ukraine with the «Ten-star pharmacist» concept. Farmatsevtychnyi Zhurnal 2021, 76, 27–36. [Google Scholar] [CrossRef]
- Model Standards of Practice for Pharmacists and Pharmacy Technicians in Canada. National Association of Pharmacy Regulatory Authorities. 2022, p. 34. Available online: https://www.napra.ca/wp-content/uploads/2022/09/NAPRA-MSOP-Feb-2022-EN-final.pdf (accessed on 12 September 2022).
- Standards for the Initial Education and Training of Pharmacists. General Pharmaceutical Council. 2021, p. 42. Available online: https://www.pharmacyregulation.org/sites/default/files/document/standards-for-the-initial-education-and-training-of-pharmacists-january-2021.pdf (accessed on 9 September 2022).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kremin, Y.; Lesyk, L.; Lesyk, R.; Levytska, O.; Hromovyk, B. Detailing the Ten Main Professional Roles of a Pharmacist to Provide the Scope of Professional Functions. Sci. Pharm. 2023, 91, 5. https://doi.org/10.3390/scipharm91010005
Kremin Y, Lesyk L, Lesyk R, Levytska O, Hromovyk B. Detailing the Ten Main Professional Roles of a Pharmacist to Provide the Scope of Professional Functions. Scientia Pharmaceutica. 2023; 91(1):5. https://doi.org/10.3390/scipharm91010005
Chicago/Turabian StyleKremin, Yuliia, Lilia Lesyk, Roman Lesyk, Oksana Levytska, and Bohdan Hromovyk. 2023. "Detailing the Ten Main Professional Roles of a Pharmacist to Provide the Scope of Professional Functions" Scientia Pharmaceutica 91, no. 1: 5. https://doi.org/10.3390/scipharm91010005
APA StyleKremin, Y., Lesyk, L., Lesyk, R., Levytska, O., & Hromovyk, B. (2023). Detailing the Ten Main Professional Roles of a Pharmacist to Provide the Scope of Professional Functions. Scientia Pharmaceutica, 91(1), 5. https://doi.org/10.3390/scipharm91010005







