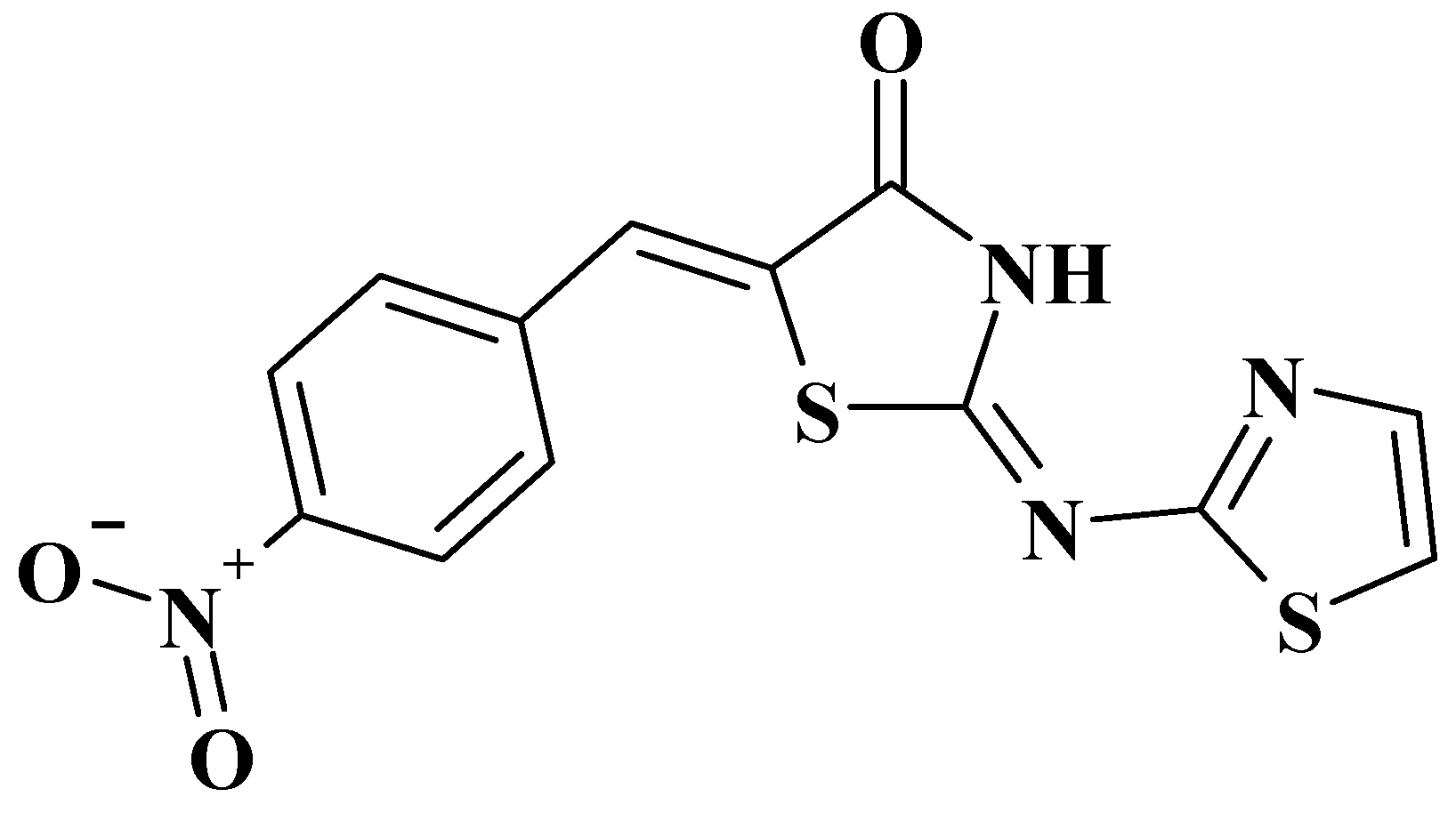
Evaluation of 5-[(Z)-(4-nitrobenzylidene)]-2-(thiazol-2-ylimino)-4-thiazolidinone (Les-6222) as Potential Anticonvulsant Agent
Abstract
1. Introduction
2. Materials and Methods
2.1. Pharmacology Assay
2.1.1. Animals
2.1.2. PTZ-Induced Kindling
2.1.3. Immunochemical Studies
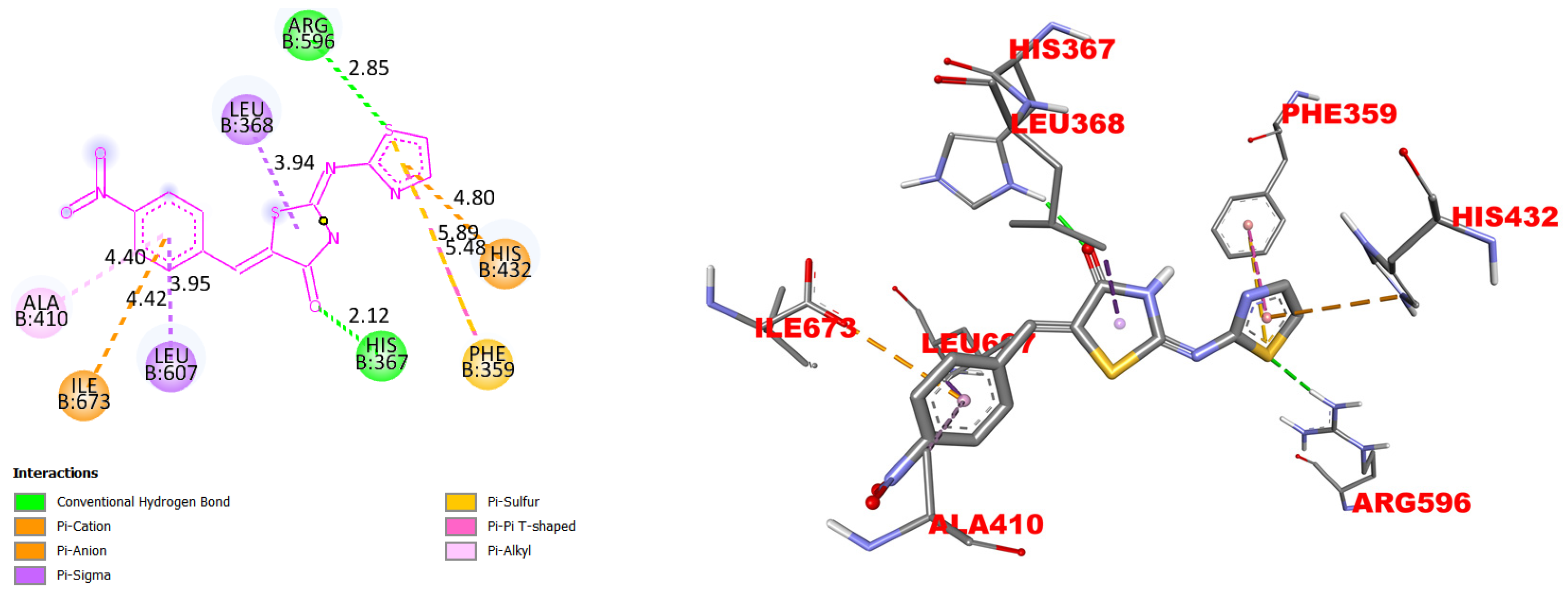
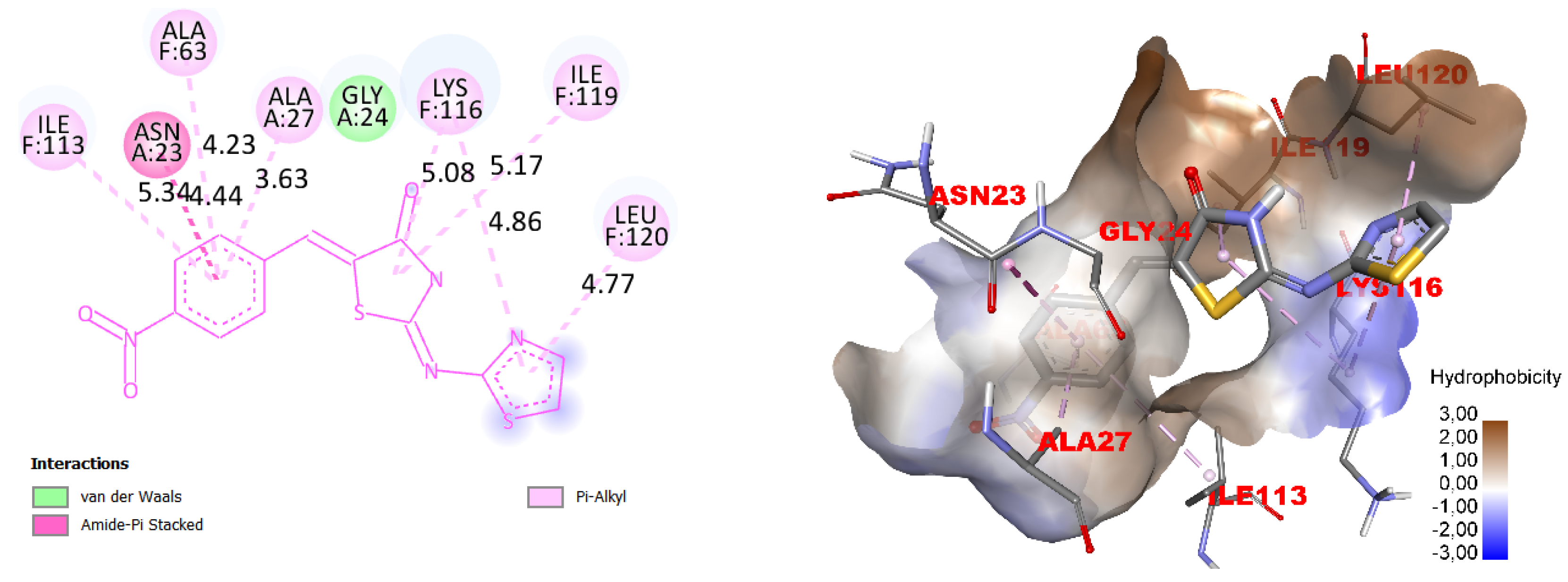
2.2. Molecular Docking
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thijs, R.D.; Surges, R.; O’Brien, T.J.; Sander, J.W. Epilepsy in adults. Lancet 2019, 393, 689–701. [Google Scholar] [CrossRef]
- Rosillo-de la Torre, A.; Luna-Bárcenas, G.; Orozco-Suárez, S.; Salgado-Ceballos, H.; García, P.; Lazarowski, A.; Rocha, L. Pharmacoresistant epilepsy and nanotechnology. Front. Biosci. (Elite Ed.) 2014, 6, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Weaver, D.F.; Pohlmann-Eden, B. Pharmacoresistant epilepsy: Unmet needs in solving the puzzle(s). Epilepsia 2013, 54, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Nidhi, S.; Manveen, B.; Sumitra, S. A review on chemical induced kindling models of epilepsy. J. Vet. Med. Res. 2016, 3, 1–6. [Google Scholar]
- de Vries, E.E.; van den Munckhof, B.; Braun, K.P.; van Royen-Kerkhof, A.; de Jager, W.; Jansen, F.E. Inflammatory mediators in human epilepsy: A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2016, 63, 177–190. [Google Scholar] [CrossRef]
- Arisi, G.M.; Foresti, M.L.; Katki, K.; Shapiro, L.A. Increased CCL2, CCL3, CCL5, and IL-1β cytokine concentration in piriform cortex, hippocampus, and neocortex after pilocarpine-induced seizures. J. Neuroinflammation 2015, 12, 129. [Google Scholar] [CrossRef]
- Vezzani, A.; Balosso, S.; Ravizza, T. Neuroinflammatory pathways as treatment targets and biomarkers in epilepsy. Nat. Rev. Neurol. 2019, 15, 459–472. [Google Scholar] [CrossRef]
- Oliveira, M.S.; Furian, A.F.; Royes, L.F.; Fighera, M.R.; Fiorenza, N.G.; Castelli, M.; Machado, P.; Bohrer, D.; Veiga, M.; Ferreira, J.; et al. Cyclooxygenase-2/PGE2 pathway facilitates pentylenetetrazol-induced seizures. Epilepsy Res. 2008, 79, 14–21. [Google Scholar] [CrossRef]
- Seo, W.; Oh, H. Comparisons of acute physiological parameters influencing outcome in patients with traumatic brain injury and hemorrhagic stroke. Worldviews Evid.-Based Nurs. 2009, 6, 36–43. [Google Scholar] [CrossRef]
- Niizuma, K.; Endo, H.; Chan, P.H. Oxidative stress and mitochondrial dysfunction as determinants of ischemic neuronal death and survival. J. Neurochem. 2009, 109, 133–138. [Google Scholar] [CrossRef]
- Wu, L.; Xiong, X.; Wu, X.; Ye, Y.; Jian, Z.; Zhi, Z.; Gu, L. Targeting oxidative stress and inflammation to prevent ischemia-reperfusion injury. Front. Mol. Neurosci. 2020, 13, 28. [Google Scholar] [CrossRef] [PubMed]
- Anand, N.; Stead, L.G. Neuron-specific enolase as a marker for acute ischemic stroke: A systematic review. Cerebrovasc. Dis. 2005, 20, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Mu, R.Z.; Liu, S.; Liang, K.G.; Jiang, D.; Huang, Y.J. A meta-analysis of neuron-specific enolase levels in cerebrospinal fluid and serum in children with epilepsy. Front. Mol. Neurosci. 2020, 13, 24. [Google Scholar] [CrossRef] [PubMed]
- Saini, S. Synthesis and anticonvulsant studies of thiazolidinone and azetidinone derivatives from indole moiety. Drug Res. 2019, 69, 445–450. [Google Scholar] [CrossRef]
- Senthilraja, M.; Alagarsamy, V. Synthesis and Pharmacological Investigation of 2-(4-Dimethylaminophenyl)-3, 5-disubstituted thiazolidin-4-ones as Anticonvulsants. Arch. Pharm. 2012, 345, 827–833. [Google Scholar] [CrossRef]
- Mech, D.; Kurowska, A.; Trotsko, N. The Bioactivity of Thiazolidin-4-Ones: A Short Review of the Most Recent Studies. Int. J. Mol. Sci. 2021, 22, 11533. [Google Scholar] [CrossRef]
- Mishchenko, M.; Shtrygol, S.; Kaminskyy, D.; Lesyk, R. Thiazole-bearing 4-thiazolidinones as new anticonvulsant agents. Sci. Pharm. 2020, 88, 16. [Google Scholar] [CrossRef]
- Mishchenko, M.V.; Shtrygol, S.Y.; Lesyk, R.B.; Lozynskyi, A.V.; Holota, S.M. Screening study of new thiazolidinone derivatives for anticonvulsant activity. Zaporozhye Med. J. 2020, 22, 840–846. [Google Scholar] [CrossRef]
- Mishchenko, M.V.; Shtrygol, S.Y. Spectrum of anticonvulsant activity and acute toxicity of 5-[(Z)-(4-nitro-benzylidene)]-2-(thiazol-2-ilimino)-4-thiazolidinone. Pharmacol. Med. Toxicol. 2020, 14, 389–396. (In Ukrainian) [Google Scholar] [CrossRef]
- Bespalov, A.; Michel, M.C.; Steckler, T. Good Research Practice in Non-Clinical Pharmacology and Biomedicine; Springer: Cham, Switzerland, 2020; 423p. [Google Scholar]
- Mishchenko, M.; Shtrygol, S.; Lozynskyi, A.; Khomyak, S.; Novikov, V.; Karpenko, O.; Holota, S.; Lesyk, R. Evaluation of anticonvulsant activity of dual COX-2/5-LOX inhibitor darbufelon and its novel analogues. Sci. Pharm. 2021, 89, 22. [Google Scholar] [CrossRef]
- Geyer, M.A.; Markou, A. The role of preclinical models in the development of psychotropic drugs. In Neuropsychopharmacology: The Fifth Generation of Progress; Davis, K.L., Charney, D., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2002; Volume 30, pp. 445–455. [Google Scholar]
- Chen, Y.Z.; Zhi, D.G. Ligand-protein inverse docking and its potential use in the computer search of protein targets of a small molecule. Proteins 2001, 43, 217–226. [Google Scholar] [CrossRef]
- Bland, M. An Introduction to Medical Statistics, 4th ed.; Oxford University Press: Oxford, UK, 2015; 427p. [Google Scholar]
- Bertram, E. The relevance of kindling for human epilepsy. Epilepsia 2007, 48, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Mokuno, K.; Kato, K.; Kawai, K.; Matsuoka, Y.; Yanag, T.; Sobue, I. Neuron-specific enolase and S-100 protein levels in cerebrospinal fluid of patients with various neurological diseases. J. Neurol. Sci. 1983, 60, 443–451. [Google Scholar] [CrossRef]
- Liu, Z.; Li, J.; Yang, F.; Hu, Y.; Liu, J.; Hu, H.; Su, W. Sodium valproate combined with levetiracetam in pediatric epilepsy and its influence on NSE, IL-6, hs-CRP and electroencephalogram improvement. Exp. Ther. Med. 2020, 20, 2043–2048. [Google Scholar] [CrossRef]
- Payandemehr, B.; Khoshneviszadeh, M.; Varastehmoradi, B.; Gholizadeh, R.; Bahremand, T.; Attar, H.; Bahremand, A.; Dehpour, A.R. A COX/5-LOX inhibitor licofelone revealed anticonvulsant properties through iNOS diminution in mice. Neurochem. Res. 2015, 40, 1819–1828. [Google Scholar] [CrossRef]
- Rojas, A.; Chen, D.; Ganesh, T.; Varvel, N.H.; Dingledine, R. The COX-2/prostanoid signaling cascades in seizure disorders. Expert Opin. Ther. Targets 2019, 23, 1–13. [Google Scholar] [CrossRef]
- Ricciotti, E.; FitzGerald, G.A. Prostaglandins and inflammation. ATVB 2011, 31, 986–1000. [Google Scholar] [CrossRef]
- Rojas, A.; Jiang, J.; Ganesh, T.; Yang, M.S.; Lelutiu, N.; Gueorguieva, P.; Dingledine, R. Cyclooxygenase-2 in epilepsy. Epilepsia 2014, 55, 17–25. [Google Scholar] [CrossRef]
- Geronikaki, A.A.; Lagunin, A.A.; Hadjipavlou-Litina, D.I.; Eleftheriou, P.T.; Filimonov, D.A.; Poroikov, V.V.; Alam, I.; Saxena, A.K. Computer-aided discovery of anti-inflammatory thiazolidinones with dual cyclooxygenase/lipoxygenase inhibition. J. Med. Chem. 2008, 51, 1601–1609. [Google Scholar] [CrossRef]
- Zhu, X.; Yao, Y.; Yang, J.; Zhengxie, J.; Li, X.; Hu, S.; Zhang, A.; Dong, J.; Zhang, C.; Gan, G. COX-2-PGE2 signaling pathway contributes to hippocampal neuronal injury and cognitive impairment in PTZ-kindled epilepsy mice. Int. Immunopharmacol. 2020, 87, 106801. [Google Scholar] [CrossRef]
- Elgarhi, R.; Shehata, M.M.; Abdelsameea, A.A.; Salem, A.E. Effects of Diclofenac Versus Meloxicam in Pentylenetetrazol-Kindled Mice. Neurochem. Res. 2020, 45, 1913–1919. [Google Scholar] [CrossRef]
- Tanaka, S.; Nakamura, T.; Sumitani, K.; Takahashi, F.; Konishi, R.; Itano, T.; Miyamoto, O. Stage- and region-specific cyclooxygenase expression and effects of a selective COX-1 inhibitor in the mouse amygdala kindling model. Neurosci. Res. 2009, 65, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Barbalho, P.G.; Carvalho, B.S.; Lopes-Cendes, I.; Maurer-Morelli, C.V. Cyclooxygenase-1 as a potential therapeutic target for seizure suppression: Evidences from zebrafish pentylenetetrazole-seizure model. Front. Neurol. 2016, 7, 200. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Yang, M.S.; Quan, Y.; Gueorguieva, P.; Ganesh, T.; Dingledine, R. Therapeutic window for cyclooxygenase-2 related anti-inflammatory therapy after status epilepticus. Neurobiol. Dis. 2015, 76, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Sang, N.; Zhang, J.; Marcheselli, V.; Bazan, N.G.; Chen, C. Postsynaptically synthesized prostaglandin E2 (PGE2) modulates hippocampal synaptic transmission via a presynaptic PGE2 EP2 receptor. J. Neurosci. 2005, 25, 9858–9870. [Google Scholar] [CrossRef]
- Chen, C.; Bazan, N.G. Endogenous PGE2 regulates membrane excitability and synaptic transmission in hippocampal CA1 pyramidal neurons. J. Neurophysiol. 2005, 93, 929–941. [Google Scholar] [CrossRef]
- Bezzi, P.; Carmignoto, G.; Pasti, L.; Vesce, S.; Rossi, D.; Rizzini, B.L.; Pozzan, T.; Volterra, A. Prostaglandins stimulate calcium-dependent glutamate release in astrocytes. Nature 1998, 391, 281–285. [Google Scholar] [CrossRef]
- Akaike, A.; Kaneko, S.; Tamura, Y.; Nakata, N.; Shiomi, H.; Ushikubi, F.; Narumiya, S. Prostaglandin E2 protects cultured cortical neurons against N-methyl-D-aspartate receptor-mediated glutamate cytotoxicity. Brain Res. 1994, 663, 237–243. [Google Scholar] [CrossRef]
- Yagami, T.; Koma, H.; Yamamoto, Y. Pathophysiological Roles of Cyclooxygenases and Prostaglandins in the Central Nervous System. Mol. Neurobiol. 2015, 53, 4754–4771. [Google Scholar] [CrossRef]
- Gryglewski, R.J. Prostacyclin as a circulatory hormone. Biochem. Pharmacol. 1979, 28, 3161–3166. [Google Scholar] [CrossRef]
- Steinhauer, H.B.; Anhut, H.; Hertting, G. The synthesis of prostaglandins and thromboxane in the mouse brain in vivo. Influence of drug induced convulsions, hypoxia and the anticonvulsants trimethadione and diazepam. Naunyn-Schmiedeb. Arch. Pharmacol. 1979, 310, 53–58. [Google Scholar] [CrossRef]
- Manev, H.; Uz, T.; Sugaya, K.; Qu, T. Putative role of neuronal 5-lipoxygenase in an aging brain. FASEB J. 2000, 14, 1464–1469. [Google Scholar] [CrossRef]
- Razavi, S.M.; Khayatan, D.; Arab, Z.N.; Momtaz, S.; Zare, K.; Jafari, R.M.; Dehpour, A.R.; Abdolghaffari, A.H. Licofelone, a potent COX/5-LOX inhibitor and a novel option for treatment of neurological disorders. Prostaglandins Other Lipid Mediat. 2021, 157, 106587. [Google Scholar] [CrossRef] [PubMed]
- Qu, T.; Uz, T.; Manev, H. Inflammatory 5-LOX mRNA and protein are increased in brain of aging rats. Neurobiol. Aging 2000, 21, 647–652. [Google Scholar] [CrossRef]
- Aguiar, C.C.T.; Almeida, A.B.; Araújo, P.V.P.; Abreu, R.N.D.C.D.; Chaves, E.M.C.; Vale, O.C.D.; Macêdo, S.M.; Woods, D.J.; de França Fonteles Fonteles, M.M.; Vasconcelos, S.M.M. Oxidative stress and epilepsy: Literature review. Oxid. Med. Cell. Longev. 2012, 2012, 795259. [Google Scholar] [CrossRef]
- Garga, N.; Lowenstein, D.H. Posttraumatic epilepsy: A major problem in desperate need of major advances. Epilepsy Curr. 2006, 6, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Mishchenko, M.V.; Shtrigol, S.Y.; Lozinskyi, A.V.; Lesyk, R.B. Psychotropic properties of the potential anticonvulsant 5-[(Z)-(4-nitrobenzylidene)]-2-(thiazol-2-ylimino)-4-thiazolidinone. News Pharm. 2021, 2, 96–103. [Google Scholar] [CrossRef]
| Experimental Data | Group of Animals | |||
|---|---|---|---|---|
| Control Pathology (Pentylenetetrazole), n = 9 | Sodium Valproate, n = 8 | Celecoxib, n = 7 | Les-6222, n = 9 | |
| Dose, mg/kg | 30 | 300 | 4 | 100 |
| Latent period of seizures, days | 4 | 6 | 4 | 4 |
| % of mice with convulsions, the average value for the entire period | 29.83 ± 7.22 | 9.08 ± 3.44 | 29.84 ± 6.71 | 7.29 ± 2.79 |
| Number of days with seizures | 13/16 | 8/16 | 13/16 | 7/16 |
| Number of days without seizures | 3/16 | 8/16 | 3/16 | 9/16 |
| Group | Dose, mg/kg | COX-1, pkg/g of Tissue | COX-2, ng/g Tissue | PGE2, pkg/g of Tissue | PGF2a, pkg/g of Tissue | PGI2, ng/g of Tissue | TXB2, pkg/g of Tissue | 8-isopro- stane, nM/g | NSE, ng/h |
|---|---|---|---|---|---|---|---|---|---|
| Intact control | – | 794.86 ± 4.59 | 132.16 ± 3.44 | 704.70 ± 4.57 | 988.79 ± 15.15 | 6.15 ± 0.05 | 131.66 ± 1.32 | 19.65 ± 0.23 | 4.04 ± 0.05 |
| Control pathology | 30 | 893.06 ± 7.50 | 216.12 ± 7.98 | 407.82 ± 3.08 | 1258.98 ± 18.42 | 3.05 ± 0.06 | 263.44 ± 1.14 | 54.67 ± 1.94 | 70.84 ± 1.01 |
| Les-6222 | 100 | 843.32 ± 9.35 | 119.94 ± 2.48 | 515.04 ± 3.44 | 1030.58 ± 25.39 | 3.88 ± 0.06 | 148.55 ± 2.78 | 45.75 ± 0.93 | 57.83 ± 0.90 |
| Celecoxib | 4 | 778.25 ± 7.73 | 213.25 ± 3.38 | 609.10 ± 3.64 | 1129.62 ± 16.11 | 2.91 ± 0.05 | 183.93 ± 2.43 | 40.42 ± 0.63 | 68.12 ± 0.48 |
| Sodium valproate | 300 | 760.40 ± 8.55 | 103.31 ± 4.33 | 629.42 ± 26.77 | 1085.49 ± 15.47 | 4.11 ± 0.10 | 142.10 ± 1.65 | 32.04 ± 0.54 | 34.44 ± 0.51 |
| COX-1, kcal/mol | COX-2, kcal/mol | 5-LOX, kcal/mol | FLAP, kcal/mol | |
|---|---|---|---|---|
| Les-6222 | −6.9 | −6.9 | −7.5 | −7.7 |
| Meloxicam | −9.8 | – | – | – |
| Celecoxib | – | −12.4 | – | – |
| Licofelon | −8.0 | – | ||
| MK-886 | – | – | −7.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mishchenko, M.; Shtrygol’, S.; Lozynskyi, A.; Hoidyk, M.; Khyluk, D.; Gorbach, T.; Lesyk, R. Evaluation of 5-[(Z)-(4-nitrobenzylidene)]-2-(thiazol-2-ylimino)-4-thiazolidinone (Les-6222) as Potential Anticonvulsant Agent. Sci. Pharm. 2022, 90, 56. https://doi.org/10.3390/scipharm90030056
Mishchenko M, Shtrygol’ S, Lozynskyi A, Hoidyk M, Khyluk D, Gorbach T, Lesyk R. Evaluation of 5-[(Z)-(4-nitrobenzylidene)]-2-(thiazol-2-ylimino)-4-thiazolidinone (Les-6222) as Potential Anticonvulsant Agent. Scientia Pharmaceutica. 2022; 90(3):56. https://doi.org/10.3390/scipharm90030056
Chicago/Turabian StyleMishchenko, Mariia, Sergiy Shtrygol’, Andrii Lozynskyi, Mykhailo Hoidyk, Dmytro Khyluk, Tatyana Gorbach, and Roman Lesyk. 2022. "Evaluation of 5-[(Z)-(4-nitrobenzylidene)]-2-(thiazol-2-ylimino)-4-thiazolidinone (Les-6222) as Potential Anticonvulsant Agent" Scientia Pharmaceutica 90, no. 3: 56. https://doi.org/10.3390/scipharm90030056
APA StyleMishchenko, M., Shtrygol’, S., Lozynskyi, A., Hoidyk, M., Khyluk, D., Gorbach, T., & Lesyk, R. (2022). Evaluation of 5-[(Z)-(4-nitrobenzylidene)]-2-(thiazol-2-ylimino)-4-thiazolidinone (Les-6222) as Potential Anticonvulsant Agent. Scientia Pharmaceutica, 90(3), 56. https://doi.org/10.3390/scipharm90030056







