Antiaging Properties of the Ethanol Fractions of Clove (Syzygium aromaticum L.) Bud and Leaf at the Cellular Levels: Study in Yeast Schizosaccharomyces pombe
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeast Cell Culture
2.2. Fractionations of Clove Extract
2.3. Total Flavonoid Content
2.4. Antioxidant Activity Based on 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Assay
2.5. Antioxidant Activity Based on 2,2’-Azino-Bis(3-Ethylbenzothiazoline-6-Sulphonic Acid) (ABTS) Assay
2.6. Antioxidant Activity Based on Thiobarbituric Acid (TBA) Assay
2.7. Antiglycation Assay
2.8. Yeast Viability Assay (Spot Test Assay)
2.9. Yeast Oxidative Stress Response Assay
2.10. Chronological Lifespan (CLS) Assay
2.11. Mitochondria Activity Assay
2.12. Cell Cycle Assay
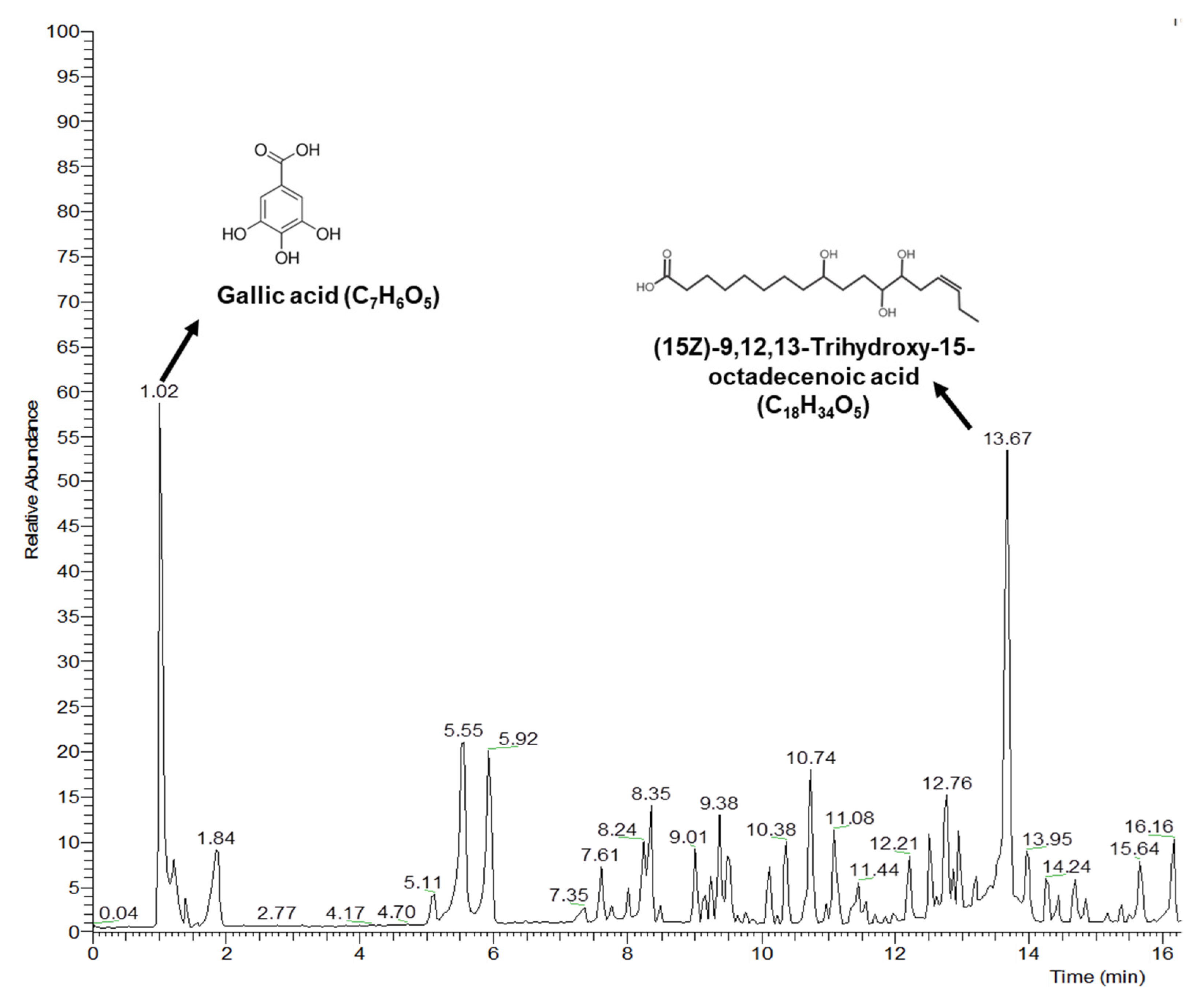
2.13. Liquid Chromatography-Mass Spectrometry Analysis
3. Results
3.1. Total Flavonoid, Antioxidant, and Antiglycation Activities
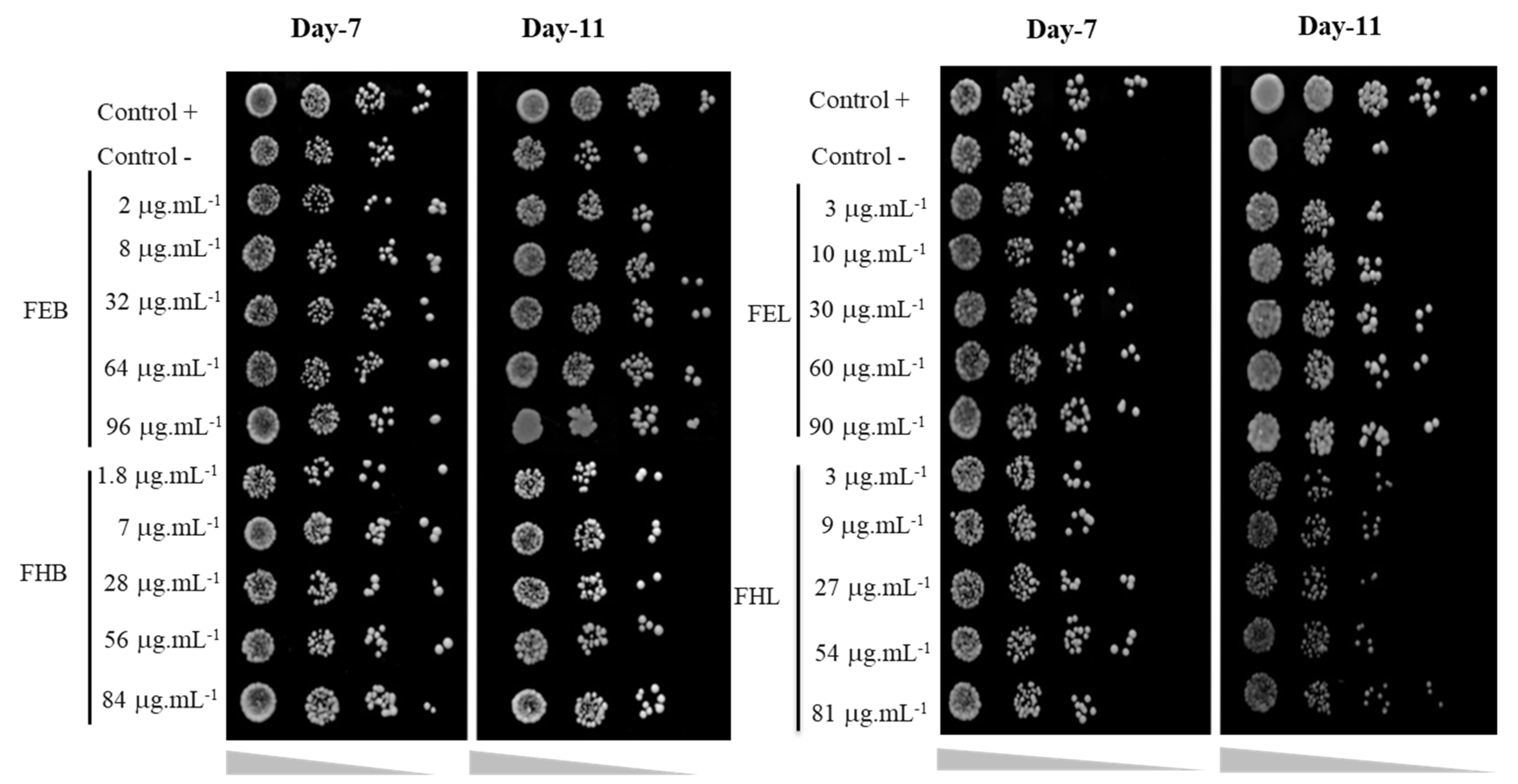
3.2. Yeast Viability Assay
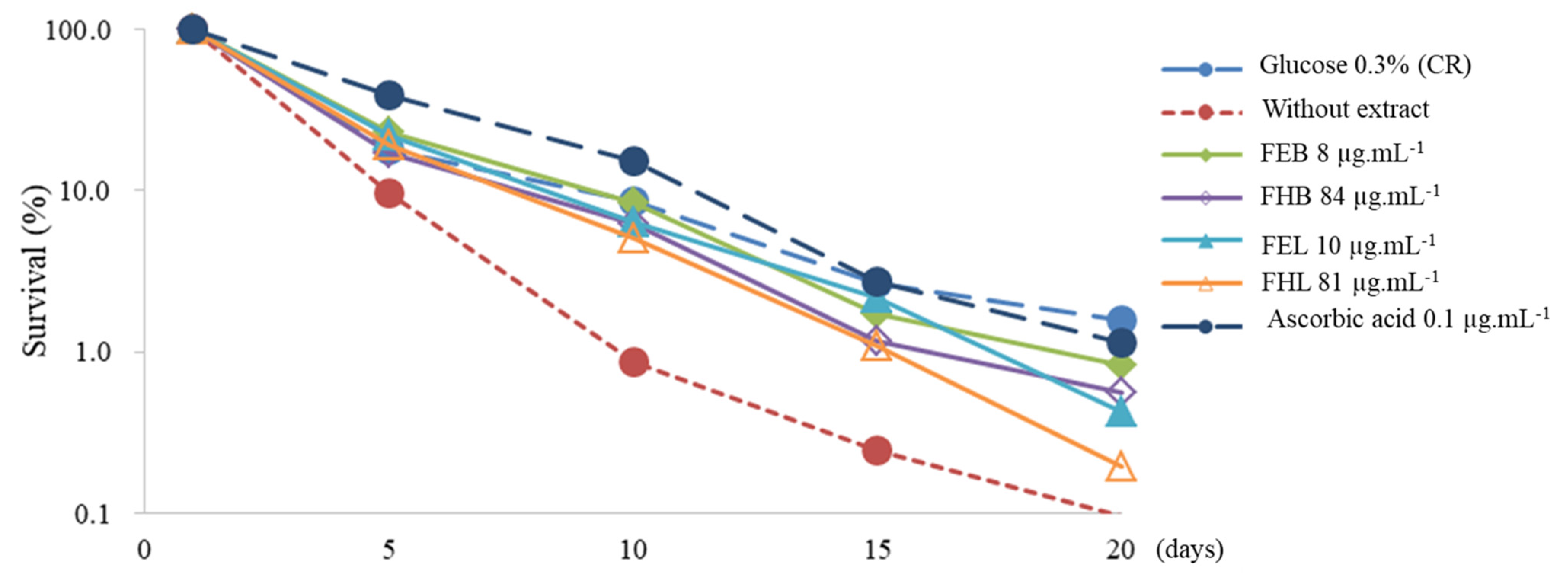
3.3. Antiaging Analysis Based on Chronological Lifespan (CLS)
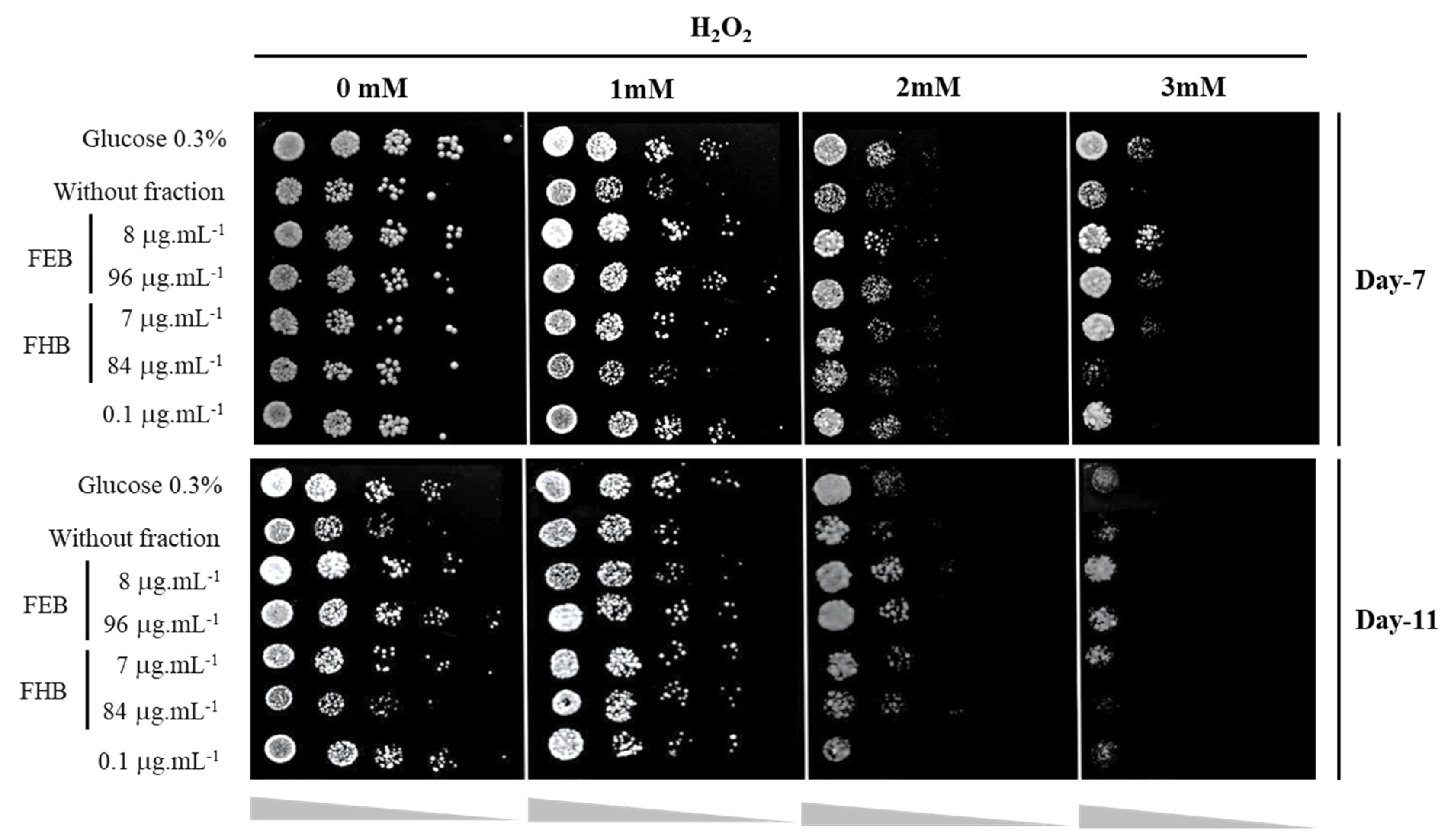
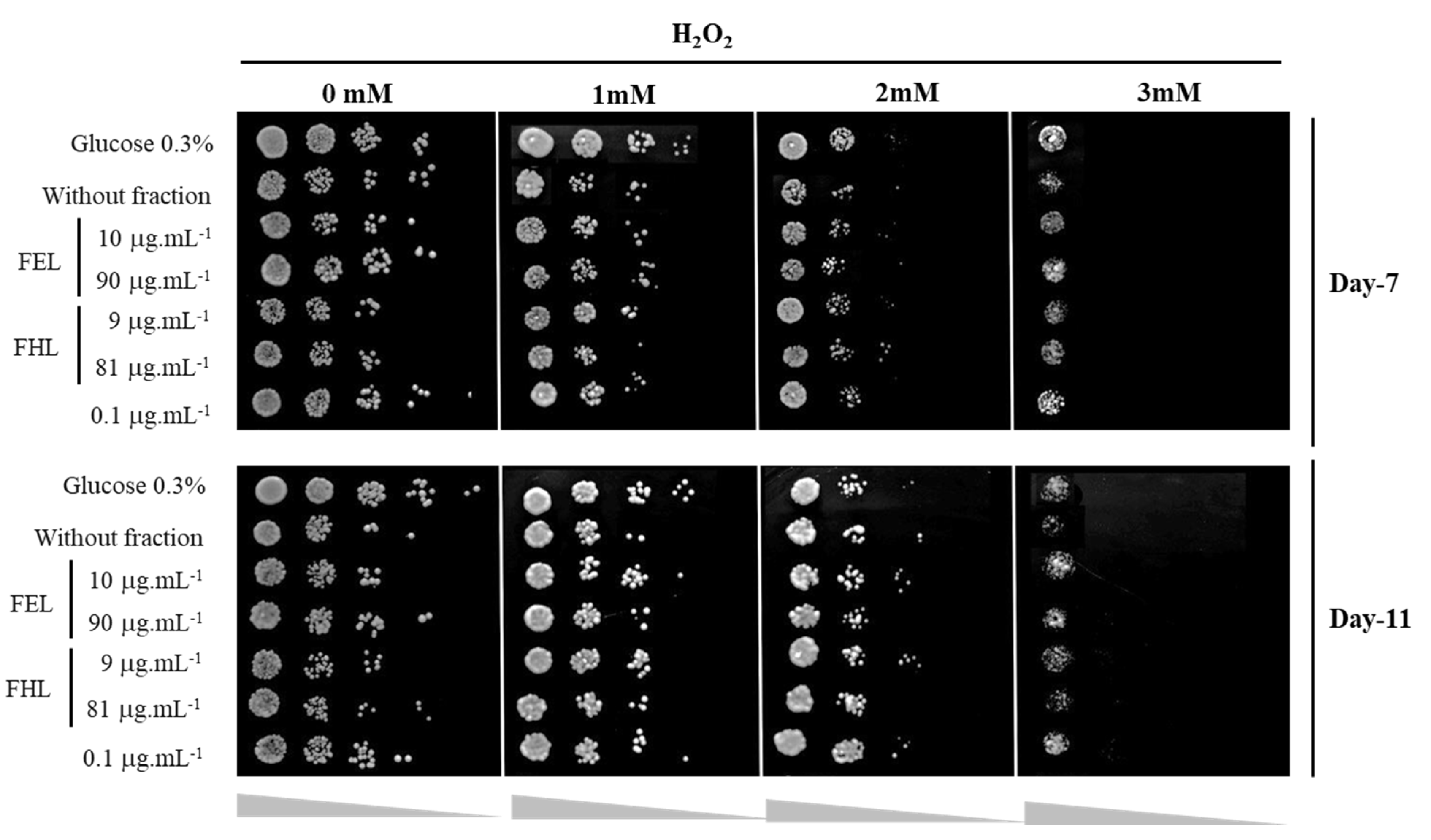
3.4. Oxidative Stress Response Assay
3.5. Mitochondrial Activity Assay
3.6. Cell Cycle Analysis
3.7. LC-MS Data Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef]
- Kwa, M.; Welty, L.J.; Xu, S. Adverse Events Reported to the US Food and Drug Administration for Cosmetics and Personal Care Products. JAMA Intern. Med. 2017, 177, 1202–1204. [Google Scholar] [CrossRef]
- Ganceviciene, R.; Liakou, A.I.; Theodoridis, A.; Makrantonaki, E.; Zouboulis, C.C. Skin anti-aging strategies. Dermato-Endocrinology 2012, 4, 308–319. [Google Scholar] [CrossRef]
- Robert, G.; Wagner, J.R. ROS-Induced DNA Damage as an Underlying Cause of Aging. Adv. Geriatr. Med. Res. 2020, 2, e200024. [Google Scholar]
- Astuti, R.I.; Listyowati, S.; Wahyuni, W.T. Life Span Extension of Model Yeast Saccharomyces Cerevisiae upon Ethanol Derived-Clover Bud Extract Treatment. IOP Conf. Ser. Earth Environ. Sci. 2019, 299, 012059. [Google Scholar] [CrossRef]
- Fauzya, A.F.; Astuti, R.I.; Mubarik, N.R. Effect of Ethanol-Derived Clove Leaf Extract on the Oxidative Stress Response in Yeast Schizosaccharomyces pombe. Int. J. Microbiol. 2019, 2019, 2145378. [Google Scholar] [CrossRef]
- Hasim; Andrianto, D.; Islamiati, W.; Wahdah Ham, A.F.; Nur Farida, D. Antioxidant Activity of Ethanol Extract of Red Yeast Rice and Its Fractionation Products. Res. J. Phytochem. 2018, 12, 52–59. [Google Scholar]
- Andrianto, D.; Katayama, T.; Suzuki, T. Screening of Antioxidant and Antihyperlipidemic Potencies of Indonesian Fruits. J. For. Biomass Util. Soc. 2011, 1, 19–25. [Google Scholar]
- Prastya, M.E.; Astuti, R.I.; Batubara, I.; Wahyudi, A.A.T. Antioxidant, Antiglycation and in vivo Antiaging Effects of Metabolite Extracts from Marine Sponge-associated Bacteria. Indian J. Pharm. Sci. 2019, 81, 344–353. [Google Scholar] [CrossRef]
- Kikuzaki, H.; Nakatani, N. Antioxidant Effects of Some Ginger Constituents. J. Food Sci. 1993, 58, 1407–1410. [Google Scholar] [CrossRef]
- Povichit, N.; Phrutivorapongkul, A.; Suttajit, M.; Leelapornpisid, P. Antiglycation and Antioxidant Activities of Oxyresveratrol Extracted from the Heartwood of Artocarpus lakoocha Roxb. Maejo Int. J. Sci. Technol. 2010, 4, 454–461. [Google Scholar]
- Huberts, D.H.E.W.; González, J.; Lee, S.S.; Litsios, A.; Hubmann, G.; Wit, E.C.; Heinemann, M. Calorie restriction does not elicit a robust extension of replicative lifespan in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 2014, 111, 11727–11731. [Google Scholar] [CrossRef]
- Wahyudi, A.T.; Prastya, M.; Astuti, R.I.; Batubara, I. Bacillus sp. SAB E-41-derived extract shows antiaging properties via ctt1-mediated oxidative stress tolerance response in yeast Schizosaccharomyces pombe. Asian Pac. J. Trop. Biomed. 2018, 8, 533. [Google Scholar] [CrossRef]
- Astuti, R.I.; Watanabe, D.; Takagi, H. Nitric oxide signaling and its role in oxidative stress response in Schizosaccharomyces pombe. Nitric Oxide 2016, 52, 29–40. [Google Scholar] [CrossRef]
- Chen, B.-R.; Runge, K.W. A New Schizosaccharomyces pombe Chronological Lifespan Assay Reveals That Caloric Restriction Promotes Efficient Cell Cycle Exit and Extends Longevity. Exp. Gerontol. 2009, 44, 493–502. [Google Scholar] [CrossRef]
- Khalil, A.A.; Rahman, U.U.; Khan, M.R.; Sahar, A.; Mehmood, T.; Khan, M. Essential oil eugenol: Sources, extraction techniques and nutraceutical perspectives. RSC Adv. 2017, 7, 32669–32681. [Google Scholar] [CrossRef]
- Bezerra, D.P.; Militão, G.C.G.; De Morais, M.C.; De Sousa, D.P. The Dual Antioxidant/Prooxidant Effect of Eugenol and Its Action in Cancer Development and Treatment. Nutrients 2017, 9, 1367. [Google Scholar] [CrossRef] [PubMed]
- Adefegha, S.A.; Oboh, G.; Adefegha, O.M.; Boligon, A.A.; Athayde, M.L. Antihyperglycemic, Hypolipidemic, Hepatoprotective and Antioxidative Effects of Dietary Clove (Szyzgium aromaticum) Bud Powder in a High-Fat Diet/Streptozotocin-Induced Diabetes Rat Model. J. Sci. Food Agric. 2014, 94, 2726–2737. [Google Scholar] [CrossRef] [PubMed]
- Rojas, D.F.C.; Souza, C.R.F.; Oliveira, W.P. Clove (Syzygium aromaticum): A precious spice. Asian Pac. J. Trop. Biomed. 2014, 4, 90–96. [Google Scholar] [CrossRef]
- Aboelmaati, M.F.; Fawzy, M.; Hassanien, R. Antioxidant Properties of Different Extracts from Five Medicinal Plants. Zagazig J. Agric. Res. 2012, 39, 1–13. [Google Scholar]
- Shojaii, A.; Kefayati, Z.; Motamed, S.M.; Noori, M.; Ghods, R. Antioxidant activity and phenolic and flavonoid contents of the extract and subfractions of Euphorbia splendida Mobayen. Pharmacogn. Res. 2017, 9, 362–365. [Google Scholar] [CrossRef] [PubMed]
- Ismail, N.Z.; Arsad, H.; Samian, M.R.; Hamdan, M.R. Determination of Phenolic and Flavonoid Contents, Antioxidant Activities and GC-MS Analysis of Clinacanthus nutans (Acanthaceae) in Different Locations. AGRIVITA J. Agric. Sci. 2017, 39. [Google Scholar] [CrossRef][Green Version]
- Zhang, Q.-W.; Lin, L.-G.; Ye, W.-C. Techniques for extraction and isolation of natural products: A comprehensive review. Chin. Med. 2018, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- El Ghallab, Y.; Al Jahid, A.; Eddine, J.J.; Said, A.A.H.; Zarayby, L.; Derfoufi, S. Syzygium aromaticum L.: Phytochemical investigation and comparison of the scavenging activity of essential oil, extracts and eugenol. Adv. Tradit. Med. 2019, 20, 153–158. [Google Scholar] [CrossRef]
- Gülçin, I. Antioxidant Activity of Eugenol: A Structure–Activity Relationship Study. J. Med. Food 2011, 14, 975–985. [Google Scholar] [CrossRef]
- Suantawee, T.; Wesarachanon, K.; Anantsuphasak, K.; Daenphetploy, T.; Thien-Ngern, S.; Thilavech, T.; Pasukamonset, P.; Ngamukote, S.; Adisakwattana, S. Protein glycation inhibitory activity and antioxidant capacity of clove extract. J. Food Sci. Technol. 2014, 52, 3843–3850. [Google Scholar] [CrossRef]
- Neveu, V.; Pérez-Jiménez, J.; Vos, F.; Crespy, V.; du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010, 2010, bap024. [Google Scholar] [CrossRef]
- Huyut, Z.; Beydemir, Ş.; Gülçin, I. Antioxidant and Antiradical Properties of Selected Flavonoids and Phenolic Compounds. Biochem. Res. Int. 2017, 2017, 7616791. [Google Scholar] [CrossRef]
- Liu, H.; Schmitz, J.C.; Wei, J.; Cao, S.; Beumer, J.; Strychor, S.; Cheng, L.; Liu, M.; Wang, C.; Wu, N.; et al. Clove Extract Inhibits Tumor Growth and Promotes Cell Cycle Arrest and Apoptosis. Oncol. Res. Featur. Preclin. Clin. Cancer Ther. 2014, 21, 247–259. [Google Scholar] [CrossRef]
- Batubara, I.; Astuti, R.I.; Prastya, M.E.; Ilmiawati, A.; Maeda, M.; Suzuki, M.; Hamamoto, A.; Takemori, H. The Antiaging Effect of Active Fractions and Ent-11α-Hydroxy-15-Oxo-Kaur-16-En-19-Oic Acid Isolated from Adenostemma lavenia (L.) o. Kuntze at the Cellular Level. Antioxidants 2020, 9, 719. [Google Scholar] [CrossRef]
- Vivancos, A.P.; Jara, M.; Zuin, A.; Sansó, M.; Hidalgo, E. Oxidative Stress in Schizosaccharomyces pombe: Different H2O2 Levels, Different Response Pathways. Mol. Genet. Genom. 2006, 276, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Astuti, R.I.; Nasuno, R.; Takagi, H. Chapter Two—Nitric Oxide Signalling in Yeast. Adv. Microb. Physiol. 2018, 72, 29–63. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Schroeder, E.A.; Ocampo, A.; Barrientos, A.; Shadel, G.S. Regulation of Yeast Chronological Life Span by TORC1 via Adaptive Mitochondrial ROS Signaling. Cell Metab. 2011, 13, 668–678. [Google Scholar] [CrossRef] [PubMed]
- Karamac, M.; Kosinska, A.; Pegg, R. Content of Gallic Acid in Selected Plant Extracts. Pol. J. Food Nutr. Sci. 2006, 15, 55–58. [Google Scholar]
- Prastya, M.E.; Astuti, R.I.; Batubara, I.; Takagi, H.; Wahyudi, A.T. Chemical Screening Identifies an Extract from Marine Pseudomonas Sp.-PTR-08 as an Anti-Aging Agent That Promotes Fission Yeast Longevity by Modulating the Pap1—Ctt1+ Pathway and the Cell Cycle. Mol. Biol. Rep. 2020, 47, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Prastya, M.; Astuti, R.I.; Batubara, I.; Takagi, H.; Wahyudi, A.T. Natural extract and its fractions isolated from the marine bacterium Pseudoalteromonas flavipulchra STILL-33 have antioxidant and antiaging activities in Schizosaccharomyces pombe. FEMS Yeast Res. 2020, 20, foaa014. [Google Scholar] [CrossRef]
- Sarima, A.R.I.; Meryandini, A. Modulation of Aging in Yeast Saccharomyces Cerevisiae by Roselle Petal Extract (Hibiscus Sabdariffa L.). Am. J. Biochem. Biotechnol. 2019, 15, 23–32. [Google Scholar] [CrossRef]
- Chang, W.-T.; Huang, S.-C.; Cheng, H.-L.; Chen, S.-C.; Hsu, C.-L. Rutin and Gallic Acid Regulates Mitochondrial Functions via the SIRT1 Pathway in C2C12 Myotubes. Antioxidants 2021, 10, 286. [Google Scholar] [CrossRef]
- Rahimifard, M.; Baeeri, M.; Bahadar, H.; Moini-Nodeh, S.; Khalid, M.; Haghi-Aminjan, H.; Mohammadian, H.; Abdollahi, M. Therapeutic Effects of Gallic Acid in Regulating Senescence and Diabetes; An In Vitro Study. Molecules 2020, 25, 5875. [Google Scholar] [CrossRef]
- Ristow, M.; Schmeisser, S. Extending life span by increasing oxidative stress. Free. Radic. Biol. Med. 2011, 51, 327–336. [Google Scholar] [CrossRef]
- Kim, B.-R.; Kim, H.M.; Jin, C.H.; Kang, S.-Y.; Kim, J.-B.; Jeon, Y.G.; Park, K.Y.; Lee, I.-S.; Han, A.-R. Composition and Antioxidant Activities of Volatile Organic Compounds in Radiation-Bred Coreopsis Cultivars. Plants 2020, 9, 717. [Google Scholar] [CrossRef] [PubMed]
- Leonov, A.; Feldman, R.; Piano, A.; Arlia-Ciommo, A.; Lutchman, V.; Ahmadi, M.; Elsaser, S.; Fakim, H.; Heshmati-Moghaddam, M.; Hussain, A.; et al. Caloric restriction extends yeast chronological lifespan via a mechanism linking cellular aging to cell cycle regulation, maintenance of a quiescent state, entry into a non-quiescent state and survival in the non-quiescent state. Oncotarget 2017, 8, 69328–69350. [Google Scholar] [CrossRef] [PubMed]
- Ou, T.-T.; Wang, C.-J.; Lee, Y.-S.; Wu, C.-H.; Lee, H.-J. Gallic acid induces G2/M phase cell cycle arrest via regulating 14-3-3β release from Cdc25C and Chk2 activation in human bladder transitional carcinoma cells. Mol. Nutr. Food Res. 2010, 54, 1781–1790. [Google Scholar] [CrossRef] [PubMed]


| No | Sample | Total Flavonoid (mgQE·g−1 Fractions) | IC50 (μg·mL−1) | |||
|---|---|---|---|---|---|---|
| DPPH | ABTS | TBA | Antiglycation | |||
| 1 | Ascorbic acid | NA | 6.25 ± 0.13 a | 11.07 ± 0.18 a | NA | NA |
| 2 | α-Tocoferol | NA | NA | NA | 36.75 ± 6.20 c | NA |
| 3 | Aminoguanidine | NA | NA | NA | NA | 2.35 ± 0.47 a |
| 4 | FHB | 7.58 ± 0.08 b | 6.88 ± 0.20 ab | 41.81 ± 0.88 e | 13.38 ± 1.98 b | 41.69 ± 1.26 b |
| 5 | FEB | 2.90 ± 0.31 a | 8.37 ± 0.99 bc | 30.64 ± 2.26 d | 2.61 ± 0.80 a | 35.64 ± 2.83 b |
| 6 | FHL | 25.65 ± 0.09 b | 8.87 ± 0.75 c | 24.24 ± 0.79 b | 8.99 ± 2.40 bc | 54.12 ± 3.13 c |
| 7 | FEL | 7.31 ± 0.23 c | 9.80 ± 0.62 c | 35.43 ± 0.69 c | 11.07± 3.40 bc | 37.15 ± 2.42 b |
| Flavonoid | DPPH | ABTS | TBA | Antiglycation | |
|---|---|---|---|---|---|
| Flavonoid | 1.00 | - | - | - | - |
| DPPH | 0.18 | 1.00 | - | - | - |
| ABTS | −0.65 | −0.40 | 1.00 | - | - |
| TBA | 0.19 | −0.16 | 0.53 | 1.00 | - |
| Antiglycation | 0.94 | −0.09 | −0.55 | 0.22 | 1.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lesmana, D.; Andrianto, D.; Astuti, R.I. Antiaging Properties of the Ethanol Fractions of Clove (Syzygium aromaticum L.) Bud and Leaf at the Cellular Levels: Study in Yeast Schizosaccharomyces pombe. Sci. Pharm. 2021, 89, 45. https://doi.org/10.3390/scipharm89040045
Lesmana D, Andrianto D, Astuti RI. Antiaging Properties of the Ethanol Fractions of Clove (Syzygium aromaticum L.) Bud and Leaf at the Cellular Levels: Study in Yeast Schizosaccharomyces pombe. Scientia Pharmaceutica. 2021; 89(4):45. https://doi.org/10.3390/scipharm89040045
Chicago/Turabian StyleLesmana, Dedy, Dimas Andrianto, and Rika Indri Astuti. 2021. "Antiaging Properties of the Ethanol Fractions of Clove (Syzygium aromaticum L.) Bud and Leaf at the Cellular Levels: Study in Yeast Schizosaccharomyces pombe" Scientia Pharmaceutica 89, no. 4: 45. https://doi.org/10.3390/scipharm89040045
APA StyleLesmana, D., Andrianto, D., & Astuti, R. I. (2021). Antiaging Properties of the Ethanol Fractions of Clove (Syzygium aromaticum L.) Bud and Leaf at the Cellular Levels: Study in Yeast Schizosaccharomyces pombe. Scientia Pharmaceutica, 89(4), 45. https://doi.org/10.3390/scipharm89040045






