The Potential Role of Lithium as an Antiviral Agent against SARS-CoV-2 via Membrane Depolarization: Review and Hypothesis
Abstract
1. Introduction
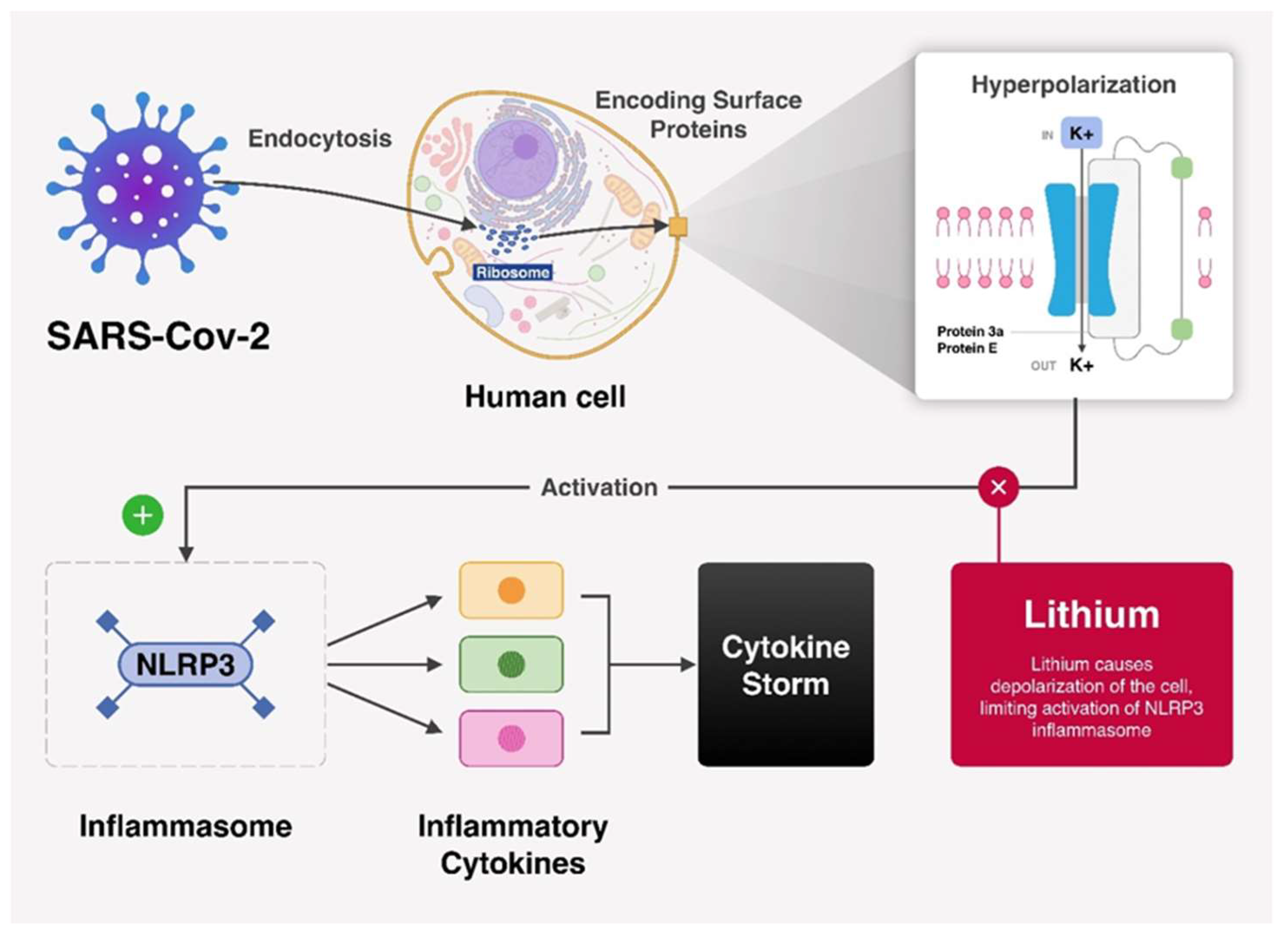
2. SARS-CoV-2 Can Hyperpolarize the Membrane Potential to Enhance Its Own Pathogenesis and Release
3. The Potential Role of Lithium in Fighting SARS-CoV-2 via Membrane Depolarization
4. COVID-19 Patients on Lithium: Expectations, Probabilities, and the Anti-Viral Action of Lithium
5. The Relevant Safety of Lithium and Its Administration
6. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Adhikari, S.P.; Meng, S.; Wu, Y.J.; Mao, Y.P.; Ye, R.X.; Wang, Q.Z.; Sun, C.; Sylvia, S.; Rozelle, S.; Raat, H.; et al. Epidemiology, causes, clinical manifestation and diagnosis, prevention and control of coronavirus disease (COVID-19) during the early outbreak period: A scoping review. Infect. Dis. Poverty 2020, 9, 1–2. [Google Scholar] [CrossRef] [PubMed]
- WHO Says Coronavirus Situation ‘Worsening’ Worldwide. Available online: https://medicalxpress.com/news/2020-06-coronavirus-situation-worsening-worldwide.html (accessed on 11 June 2020).
- Naqvi, A.A.; Fatima, K.; Mohammad, T.; Fatima, U.; Singh, I.K.; Singh, A.; Atif, S.M.; Hariprasad, G.; Hasan, G.M.; Hassan, M.I. Insights into SARS-CoV-2 genome, structure, evolution, pathogenesis and therapies: Structural genomics approach. Biochimic. Biophys. Acta BBA Mol. Basis Dis. 2020, 1866, 165878. [Google Scholar] [CrossRef]
- Yang, Y.; Peng, F.; Wang, R.; Guan, K.; Jiang, T.; Xu, G.; Sun, J.; Chang, C. The deadly coronaviruses: The 2003 SARS pandemic and the 2020 novel coronavirus epidemic in China. J. Autoimmun. 2020, 109, 102434. [Google Scholar] [CrossRef] [PubMed]
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; Di Napoli, R. Features, evaluation and treatment coronavirus (COVID-19). In StatPearls; StatPearls Publishing: Tampa, FL, USA, 2020. [Google Scholar]
- Castaño-Rodriguez, C.; Honrubia, J.M.; Gutiérrez-Álvarez, J.; DeDiego, M.L.; Nieto-Torres, J.L.; Jimenez-Guardeño, J.M.; Regla-Nava, J.A.; Fernandez-Delgado, R.; Verdia-Báguena, C.; Queralt-Martín, M.; et al. Role of severe acute respiratory syndrome coronavirus viroporins E, 3a, and 8a in replication and pathogenesis. MBio 2018, 9, e02325–e02417. [Google Scholar] [CrossRef]
- Lu, W.; Zheng, B.J.; Xu, K.; Schwarz, W.; Du, L.; Wong, C.K.; Chen, J.; Duan, S.; Deubel, V.; Sun, B. Severe acute respiratory syndrome-associated coronavirus 3a protein forms an ion channel and modulates virus release. Proc. Natl. Acad. Sci. USA 2006, 103, 12540–12545. [Google Scholar] [CrossRef] [PubMed]
- Abdul Kadir, L.; Stacey, M.; Barrett-Jolley, R. Emerging roles of the membrane potential: Action beyond the action potential. Front. Physiol. 2018, 9, 1661. [Google Scholar] [CrossRef]
- Blackiston, D.J.; McLaughlin, K.A.; Levin, M. Bioelectric controls of cell proliferation: Ion channels, membrane voltage and the cell cycle. Cell Cycle 2009, 8, 3527–3536. [Google Scholar] [CrossRef]
- Urrego, D.; Tomczak, A.P.; Zahed, F.; Stühmer, W.; Pardo, L.A. Potassium channels in cell cycle and cell proliferation. Philos Trans. R Soc. B 2014, 369, 20130094. [Google Scholar] [CrossRef]
- Kito, H.; Yamamura, H.; Suzuki, Y.; Ohya, S.; Asai, K.; Imaizumi, Y. Membrane hyperpolarization induced by endoplasmic reticulum stress facilitates Ca2+ influx to regulate cell cycle progression in brain capillary endothelial cells. J. Pharmacol. Sci. 2014, 14002SC. [Google Scholar] [CrossRef]
- Muñoz-Planillo, R.; Kuffa, P.; Martínez-Colón, G.; Smith, B.L.; Rajendiran, T.M.; Núñez, G. K+ efflux is the common trigger of NLRP3 inflammasome activation by bacterial toxins and particulate matter. Immunity 2013, 38, 1142–1153. [Google Scholar] [CrossRef]
- Fung, S.Y.; Yuen, K.S.; Ye, Z.W.; Chan, C.P.; Jin, D.Y. A tug-of-war between severe acute respiratory syndrome coronavirus 2 and host antiviral defence: Lessons from other pathogenic viruses. Emerg. Microbes Infect. 2020, 9, 558–570. [Google Scholar] [CrossRef]
- Tan, Y.J.; Lim, S.G.; Hong, W. Regulation of cell death during infection by the severe acute respiratory syndrome coronavirus and other coronaviruses. Cell Microbiol. 2007, 9, 2552–2561. [Google Scholar] [CrossRef]
- Ren, Y.; Shu, T.; Wu, D.; Mu, J.; Wang, C.; Huang, M.; Han, Y.; Zhang, X.Y.; Zhou, W.; Qiu, Y.; et al. The ORF3a protein of SARS-CoV-2 induces apoptosis in cells. Cell. Mol. Immunol. 2020, 17, 881–883. [Google Scholar] [CrossRef]
- Chen, I.Y.; Moriyama, M.; Chang, M.F.; Ichinohe, T. Severe acute respiratory syndrome coronavirus viroporin 3a activates the NLRP3 inflammasome. Front. Microbiol. 2019, 10, 50. [Google Scholar] [CrossRef] [PubMed]
- Schoeman, D.; Fielding, B.C. Coronavirus envelope protein: Current knowledge. Virol. J. 2019, 16, 69. [Google Scholar] [CrossRef] [PubMed]
- Verdiá-Báguena, C.; Nieto-Torres, J.L.; Alcaraz, A.; DeDiego, M.L.; Torres, J.; Aguilella, V.M.; Enjuanes, L. Coronavirus E protein forms ion channels with functionally and structurally-involved membrane lipids. Virology 2012, 432, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Torres, J.L.; Verdiá-Báguena, C.; Jimenez-Guardeño, J.M.; Regla-Nava, J.A.; Castaño-Rodriguez, C.; Fernandez-Delgado, R.; Torres, J.; Aguilella, V.M.; Enjuanes, L. Severe acute respiratory syndrome coronavirus E protein transports calcium ions and activates the NLRP3 inflammasome. Virology 2015, 485, 330–339. [Google Scholar] [CrossRef]
- Li, S.; Yuan, L.; Dai, G.; Chen, R.A.; Liu, D.X.; Fung, T.S. Regulation of the ER Stress Response by the Ion Channel Activity of the Infectious Bronchitis Coronavirus Envelope Protein Modulates Virion Release, Apoptosis, Viral Fitness, and Pathogenesis. Front. Microbiol. 2020, 10, 3022. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.S.; Liu, D.X. Coronavirus infection, ER stress, apoptosis and innate immunity. Front. Microbiol. 2014, 5, 296. [Google Scholar] [CrossRef]
- Minakshi, R.; Padhan, K.; Rani, M.; Khan, N.; Ahmad, F.; Jameel, S. The SARS Coronavirus 3a protein causes endoplasmic reticulum stress and induces ligand-independent downregulation of the type 1 interferon receptor. PLoS ONE 2009, 4, e8342. [Google Scholar] [CrossRef]
- Versteeg, G.A.; van de Nes, P.S.; Bredenbeek, P.J.; Spaan, W.J. The coronavirus spike protein induces endoplasmic reticulum stress and upregulation of intracellular chemokine mRNA concentrations. J. Virol. 2007, 81, 10981–10990. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, M.; Benvenuto, D.; Giovanetti, M.; Angeletti, S.; Ciccozzi, M.; Pascarella, S. Sars-CoV-2 Envelope and Membrane Proteins: Structural Differences Linked to Virus Characteristics? BioMed Res. Int. 2020. [Google Scholar] [CrossRef] [PubMed]
- Kern, D.M.; Sorum, B.; Hoel, C.M.; Sridharan, S.; Remis, J.P.; Toso, D.B.; Brohawn, S.G. Cryo-EM structure of the SARS-CoV-2 3a ion channel in lipid nanodiscs. BioRxiv 2020. [Google Scholar] [CrossRef]
- Tomar, P.P.; Arkin, I.T. SARS-CoV-2 E protein is a potential ion channel that can be inhibited by Gliclazide and Memantine. Biochem. Biophys. Res. Commun. 2020, 530, 10–14. [Google Scholar] [CrossRef]
- Channappanavar, R.; Perlman, S. Pathogenic human coronavirus infections: Causes and consequences of cytokine storm and immunopathology. In Seminars in Immunopathology; Springer: Berlin/Heidelberg, Germany, 2017; Volume 39, pp. 529–539. [Google Scholar]
- Carmeliet, E.E. Influence of lithium ions on the transmembrane potential and cation content of cardiac cells. J. Gen. Physiol. 1964, 47, 501–530. [Google Scholar] [CrossRef]
- Qaswal, A.B. Quantum Electrochemical Equilibrium: Quantum Version of the Goldman–Hodgkin–Katz Equation. Quantum Rep. 2020, 2, 266–277. [Google Scholar]
- Qaswal, A.B. Lithium stabilizes the mood of bipolar patients by depolarizing the neuronal membrane via quantum tunneling through the sodium channels. Clin. Psychopharmacol. Neurosci. 2020, 18, 214. [Google Scholar] [CrossRef]
- Plotnikov, E.Y.; Silachev, D.N.; Zorova, L.D.; Pevzner, I.B.; Jankauskas, S.S.; Zorov, S.D.; Babenko, V.A.; Skulachev, M.V.; Zorov, D.B. Lithium salts—Simple but magic. Biochemistry 2014, 79, 740–749. [Google Scholar] [CrossRef] [PubMed]
- Malhi, G.S.; Tanious, M.; Das, P.; Coulston, C.M.; Berk, M. Potential mechanisms of action of lithium in bipolar disorder. CNS Drugs 2013, 27, 135–153. [Google Scholar]
- Cone, C.D.; Cone, C.M. Induction of mitosis in mature neurons in central nervous system by sustained depolarization. Science 1976, 192, 155–158. [Google Scholar] [CrossRef]
- Stillwell, E.F.; Cone, C.M.; JUNC. Stimulation of DNA synthesis in CNS neurones by sustained depolarisation. Nat. New Biol. 1973, 246, 110. [Google Scholar] [CrossRef]
- Vidali, S.; Aminzadeh-Gohari, S.; Vatrinet, R.; Iommarini, L.; Porcelli, A.M.; Kofler, B.; Feichtinger, R.G. Lithium and Not Acetoacetate Influences the Growth of Cells Treated with Lithium Acetoacetate. Int. J. Mol. Sci. 2019, 20, 3104. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Huang, K.; Liu, X.; Liu, J.; Lu, X.; Tao, K.; Wang, G.; Wang, J. Lithium chloride suppresses colorectal cancer cell survival and proliferation through ROS/GSK-3β/NF-κB signaling pathway. Oxid. Med. Cell Longev. 2014. [Google Scholar] [CrossRef] [PubMed]
- Maddu, N.; Raghavendra, P.B. Review of lithium effects on immune cells. Immunopharmacol. Immunotoxicol. 2015, 37, 111–125. [Google Scholar] [CrossRef] [PubMed]
- Bartnikowski, M.; Moon, H.J.; Ivanovski, S. Release of lithium from 3D printed polycaprolactone scaffolds regulates macrophage and osteoclast response. Biomed. Mater. 2018, 13, 065003. [Google Scholar] [CrossRef]
- Monroe, J.G.; Cambier, J.C. B cell activation. I. Anti-immunoglobulin-induced receptor cross-linking results in a decrease in the plasma membrane potential of murine B lymphocytes. J. Exp. Med. 1983, 157, 2073–2086. [Google Scholar] [CrossRef]
- Pandit, V.; Nesbitt, S.R.; Kim, D.Y.; Mixon, A.; Kotha, S.P. Combinatorial therapy using negative pressure and varying lithium dosage for accelerated wound healing. J. Mech. Behav. Biomed. 2015, 44, 173–178. [Google Scholar] [CrossRef]
- Bose, S.; Fielding, G.; Tarafder, S.; Bandyopadhyay, A. Understanding of dopant-induced osteogenesis and angiogenesis in calcium phosphate ceramics. Trends Biotechnol. 2013, 31, 594–605. [Google Scholar] [CrossRef]
- Chatterjee, S.; Browning, E.A.; Hong, N.; DeBolt, K.; Sorokina, E.M.; Liu, W.; Birnbaum, M.J.; Fisher, A.B. Membrane depolarization is the trigger for PI3K/Akt activation and leads to the generation of ROS. Am. J. Physiol. Heart Circ. Physiol. 2012, 302, H105–H114. [Google Scholar]
- Lee, Y.I.; Seo, M.; Kim, Y.; Kim, S.Y.; Kang, U.G.; Kim, Y.S.; Juhnn, Y.S. Membrane depolarization induces the undulating phosphorylation/dephosphorylation of glycogen synthase kinase 3β, and this dephosphorylation involves protein phosphatases 2A and 2B in SH-SY5Y human neuroblastoma cells. J. Biol. Chem. 2005, 280, 22044–22052. [Google Scholar] [CrossRef]
- Chuang, D.M.; Wang, Z.; Chiu, C.T. GSK-3 as a target for lithium-induced neuroprotection against excitotoxicity in neuronal cultures and animal models of ischemic stroke. Front. Mol. Neurosci. 2011, 4, 15. [Google Scholar] [CrossRef]
- Feske, S.; Wulff, H.; Skolnik, E.Y. Ion channels in innate and adaptive immunity. Annu. Rev. Immunol. 2015, 33, 291–353. [Google Scholar] [CrossRef]
- Erndt-Marino, J.; Hahn, M.S. Membrane potential controls macrophage activation. Front. Bioeng. Biotechnol. 2016, 4, 10–3389. [Google Scholar]
- Haslberger, A.; Romanin, C.; Koerber, R. Membrane potential modulates release of tumor necrosis factor in lipopolysaccharide-stimulated mouse macrophages. Mol. Biol. Cell 1992, 3, 451–460. [Google Scholar] [PubMed]
- Hsueh, P.R.; Huang, L.M.; Chen, P.J.; Kao, C.L.; Yang, P.C. Chronological evolution of IgM, IgA, IgG and neutralisation antibodies after infection with SARS-associated coronavirus. Clin. Microbiol. Infect. 2004, 10, 1062–1066. [Google Scholar] [CrossRef]
- Kucharz, E.J.; Sierakowski, S.; Staite, N.D.; Goodwin, J.S. Mechanism of lithium-induced augmentation of T-cell proliferation. Int. J. Immunopharmacol. 1988, 10, 253–259. [Google Scholar] [CrossRef]
- Borkowsky, W.; Shenkman, L.; Rausen, A. T-lymphocyte cycling in human cyclic neutropenia: Effects of lithium in vitro and in vivo. Clin. Immunol. Immunopathol. 1982, 23, 586–592. [Google Scholar] [CrossRef]
- Bray, J.; Turner, A.R.; Dusel, F. Lithium and the mitogenic response of human lymphocytes. Clin. Immunol. Immunopathol. 1981, 19, 284–288. [Google Scholar]
- Kiefer, H.; Blume, A.J.; Kaback, H.R. Membrane potential changes during mitogenic stimulation of mouse spleen lymphocytes. Proc. Natl. Acad. Sci. USA 1980, 77, 2200–2204. [Google Scholar] [CrossRef]
- Daniele, R.P.; Holian, S.K. A potassium ionophore (valinomycin) inhibits lymphocyte proliferation by its effects on the cell membrane. Proc. Natl Acad Sci. USA 1976, 73, 3599–3602. [Google Scholar] [CrossRef]
- Akdis, M.; Aab, A.; Altunbulakli, C.; Azkur, K.; Costa, R.A.; Crameri, R.; Duan, S.; Eiwegger, T.; Eljaszewicz, A.; Ferstl, R.; et al. Interleukins (from IL-1 to IL-38), interferons, transforming growth factor β, and TNF-α: Receptors, functions, and roles in diseases. J. Allergy Clin. Immunol. 2016, 138, 984–1010. [Google Scholar]
- Nassar, A.; Azab, A.N. Effects of lithium on inflammation. ACS Chem. Neurosci. 2014, 5, 451–458. [Google Scholar] [CrossRef] [PubMed]
- J. De-Paula, V.; S. Kerr, D.; Scola, G.; F. Gattaz, W.; V. Forlenza, O. Lithium distinctly modulates the secretion of pro-and anti-inflammatory interleukins in co-cultures of neurons and glial cells at therapeutic and sub-therapeutic concentrations. Curr. Alzheimer Res. 2016, 13, 848–852. [Google Scholar] [CrossRef]
- Himmerich, H.; Bartsch, S.; Hamer, H.; Mergl, R.; Schönherr, J.; Petersein, C.; Munzer, A.; Kirkby, K.C.; Bauer, K.; Sack, U. Impact of mood stabilizers and antiepileptic drugs on cytokine production in-vitro. J. Psychiatr. Res. 2013, 47, 1751–1759. [Google Scholar] [CrossRef]
- Dribben, W.H.; Eisenman, L.N.; Mennerick, S. Magnesium induces neuronal apoptosis by suppressing excitability. Cell Death Dis. 2010, 1, e63. [Google Scholar] [CrossRef]
- Stanojevic, M.; Lopicic, S.; Spasic, S.; Vukovic, I.; Nedeljkov, V.; Prostran, M. Effects of high extracellular magnesium on electrophysiological properties of membranes of Retzius neurons in leech Haemopis sanguisuga. J. Elementol. 2016, 21, 221–230. [Google Scholar] [CrossRef]
- Barjas Qaswal, A. Magnesium Ions Depolarize the Neuronal Membrane via Quantum Tunneling through the Closed Channels. Quantum Rep. 2020, 2, 57–63. [Google Scholar]
- Sun, Y.M.; Favre, I.; Schild, L.; Moczydlowski, E. On the structural basis for size-selective permeation of organic cations through the voltage-gated sodium channel: Effect of alanine mutations at the DEKA locus on selectivity, inhibition by Ca2+ and H+, and molecular sieving. J. Gen. Physiol. 1997, 110, 693–715. [Google Scholar] [CrossRef] [PubMed]
- Rybakowski, J.K.; Nassar, A.; Azab, A.N. The effect of lithium on the immune system. Hum. Psychopharmacol. Clin. Exp. 1999, 14, 345–353. [Google Scholar] [CrossRef]
- COVID-19 CORONAVIRUS PANDEMIC. Available online: https://www.worldometers.info/coronavirus/ (accessed on 19 January 2021).
- Van Den Ameele, S.; Van Diermen, L.; Staels, W.; Coppens, V.; Dumont, G.; Sabbe, B. The effect of mood-stabilizing drugs on cytokine levels in bipolar disorder: A systematic review. J. Affect. Disord. 2016, 203, 364–373. [Google Scholar] [CrossRef]
- Neurath, A.R.; Stasny, J.T.; Rubin, B.A. Disruption of adenovirus type 7 by lithium iodide resulting in the release of viral deoxyribonucleic acid. J. Virol. 1970, 5, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Skinner, G.R.; Hartley, C.; Buchan, A.; Harper, L.; Gallimore, P. The effect of lithium chloride on the replication of herpes simplex virus. Med. Microbiol. Immunol. 1980, 168, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Ziaie, Z.; Kefalides, N.A. Lithium chloride restores host protein synthesis in herpes simplex virus-infected endothelial cells. Biochem. Biophys. Res. Commun. 1989, 160, 1073–1078. [Google Scholar] [CrossRef]
- Cernescu, C.; Popescu, L.; Constantinescu, S.T.; Cernescu, S. Antiviral effect of lithium chloride. Virology 1988, 39, 93–101. [Google Scholar]
- Amsterdam, J.D.; Maislin, G.; Hooper, M.B. Suppression of herpes simplex virus infections with oral lithium carbonate—A possible antiviral activity. Pharm. J. Hum. Pharm. Drug 1996, 16, 1070–1075. [Google Scholar]
- Hartley, C.E.; Buchan, A.; Randall, S.; Skinner, G.R.B.; Osborne, M.; Tomkins, L.M. The effects of lithium and potassium on macromolecular synthesis in herpes simplex virus-infected cells. J. Gen. Virol. 1993, 74, 1519–1525. [Google Scholar] [CrossRef]
- Chen, Y.; Yan, H.; Zheng, H.; Shi, Y.; Sun, L.; Wang, C.; Sun, J. Antiviral effect of lithium chloride on infection of cells by porcine parvovirus. Arch. Virol. 2015, 160, 1015–1020. [Google Scholar] [CrossRef]
- Zhou, P.; Fu, X.; Yan, Z.; Fang, B.; Huang, S.; Fu, C.; Hong, M.; Li, S. Antiviral effect of lithium chloride on infection of cells by canine parvovirus. Arch. Virol. 2015, 160, 2799–2805. [Google Scholar]
- Harrison, S.M.; Tarpey, I.; Rothwell, L.; Kaiser, P.; Hiscox, J.A. Lithium chloride inhibits the coronavirus infectious bronchitis virus in cell culture. Avian Pathol. 2007, 36, 109–114. [Google Scholar] [CrossRef][Green Version]
- Ren, X.; Meng, F.; Yin, J.; Li, G.; Li, X.; Wang, C.; Herrler, G. Action mechanisms of lithium chloride on cell infection by transmissible gastroenteritis coronavirus. PLoS ONE. 2011, 6, e18669. [Google Scholar] [CrossRef]
- Benítez-Rangel, E.; García, L.; Namorado, M.C.; Reyes, J.L.; Guerrero-Hernández, A. Ion channel inhibitors block caspase activation by mechanisms other than restoring intracellular potassium concentration. Cell Death Dis. 2011, 2, e113. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cui, J.; Xie, J.; Gao, M.; Zhou, H.; Chen, Y.; Cui, T.; Bai, X.; Wang, H.; Zhang, G. Inhibitory effects of lithium chloride on replication of type II porcine reproductive and respiratory syndrome virus in vitro. Antiviral Ther. 2015, 20, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Qian, K.; Cheng, X.; Zhang, D.; Shao, H.; Yao, Y.; Nair, V.; Qin, A. Antiviral effect of lithium chloride on replication of avian leukosis virus subgroup J in cell culture. Arch. Virol. 2018, 163, 987–995. [Google Scholar] [CrossRef]
- Puertas, M.C.; Salgado, M.; Morón-López, S.; Ouchi, D.; Muñoz-Moreno, J.A.; Moltó, J. Effect of lithium on HIV-1 expression and proviral reservoir size in the CD4 + T cells of antiretroviral therapy suppressed patients. AIDS 2014, 28, 2157–2159. [Google Scholar] [CrossRef] [PubMed]
- Amsterdam, J.D.; García-España, F.; Rybakowski, J. Rates of flu-like infection in patients with affective illness. J. Affect. Disord. 1998, 47, 177–182. [Google Scholar] [CrossRef]
- Landen, M.; Lichtenstein, P.; Larsson, H.; Song, J. Respiratory infection during lithium and valproate medication: A within-individual prospective study of 50,000 patients with bipolar disorder. medRxiv 2020. [Google Scholar] [CrossRef]
- Spuch, C.; López-García, M.; Rivera-Baltanás, T.; Rodrígues-Amorím, D.; Olivares, J.M. Does Lithium Deserve a Place in the Treatment Against COVID-19? A Preliminary Observational Study in Six Patients, Case Report. Front. Pharmacol. 2020, 11, 1347. [Google Scholar] [CrossRef] [PubMed]
- Baird-Gunning, J.; Lea-Henry, T.; Hoegberg, L.C.; Gosselin, S.; Roberts, D.M. Lithium poisoning. J. Intensive Care Med. 2017, 32, 249–263. [Google Scholar] [CrossRef]
- Hillert, M.; Zimmermann, M.; Klein, J. Uptake of lithium into rat brain after acute and chronic administration. Neurosci. Lett. 2012, 521, 62–66. [Google Scholar] [CrossRef]
- Lithium: Drug information. Available online: https://www.uptodate.com/contents/lithium-drug-information?search=lithium&source=panel_search_result&selectedTitle=1~148&usage_type=panel&kp_tab=drug_general&display_rank=1 (accessed on 19 January 2021).
- Murru, A.; Manchia, M.; Hajek, T.; Nielsen, R.E.; Rybakowski, J.K.; Sani, G.; Schulze, T.G.; Tondo, L.; Bauer, M.; International Group for The Study of Lithium Treated Patients (IGSLi). Lithium’s antiviral effects: A potential drug for CoViD-19 disease? Int. J. Bipolar Dis. 2020, 8, 1–9. [Google Scholar] [CrossRef]
- Rajkumar, R.P. Lithium as a candidate treatment for COVID-19: Promises and pitfalls. Drug Dev. Res. 2020, 81, 782–785. [Google Scholar] [CrossRef] [PubMed]
- Nowak, J.K.; Walkowiak, J. Lithium and coronaviral infections. A scoping review. F1000Research 2020, 9, 93. [Google Scholar] [CrossRef] [PubMed]
| NLRP3 Suppression | Cell Death Inhibition | Immunomodulation |
|---|---|---|
| Lithium prevents the activation of NLRP3 inflammasome, which is implicated in the release of pro-inflammatory cytokines in the cytokine storm. | Lithium inhibits cell death cascade, resulting in a decrease in viral release, attenuation of immune system over activation, and a decrease in lung parenchymal damage. | Lithium can balance immune system function in such a way that prevents the harmful effects of over-activation, but guarantees a level of activation that can fight and clear the virus. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qaswal, A.B.; Suleiman, A.; Guzu, H.; Harb, T.; Atiyat, B. The Potential Role of Lithium as an Antiviral Agent against SARS-CoV-2 via Membrane Depolarization: Review and Hypothesis. Sci. Pharm. 2021, 89, 11. https://doi.org/10.3390/scipharm89010011
Qaswal AB, Suleiman A, Guzu H, Harb T, Atiyat B. The Potential Role of Lithium as an Antiviral Agent against SARS-CoV-2 via Membrane Depolarization: Review and Hypothesis. Scientia Pharmaceutica. 2021; 89(1):11. https://doi.org/10.3390/scipharm89010011
Chicago/Turabian StyleQaswal, Abdallah Barjas, Aiman Suleiman, Hasan Guzu, Taima’a Harb, and Bashir Atiyat. 2021. "The Potential Role of Lithium as an Antiviral Agent against SARS-CoV-2 via Membrane Depolarization: Review and Hypothesis" Scientia Pharmaceutica 89, no. 1: 11. https://doi.org/10.3390/scipharm89010011
APA StyleQaswal, A. B., Suleiman, A., Guzu, H., Harb, T., & Atiyat, B. (2021). The Potential Role of Lithium as an Antiviral Agent against SARS-CoV-2 via Membrane Depolarization: Review and Hypothesis. Scientia Pharmaceutica, 89(1), 11. https://doi.org/10.3390/scipharm89010011






