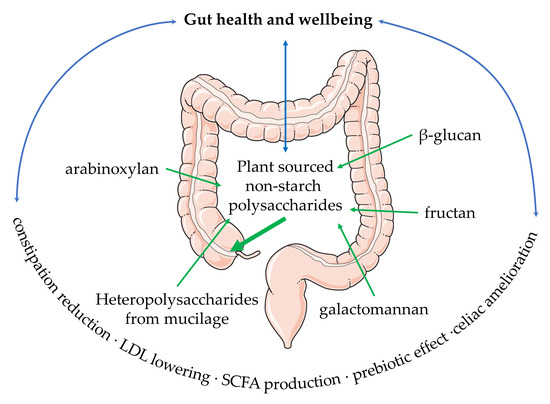
Trends in the Use of Plant Non-Starch Polysaccharides within Food, Dietary Supplements, and Pharmaceuticals: Beneficial Effects on Regulation and Wellbeing of the Intestinal Tract
Abstract
1. Introduction
2. Non-Starch Polysaccharides in Cereal Grains and their Application in Healthy Products
3. Galactomannan in Legumes and their Beneficial Effects in the Well-Being of the Intestinal Tract
4. Plant-Based Fructans, Technological Use, and Prebiotic Effects
5. Heteropolysaccarides from Plant Mucilages and their Potential for Use in Healthy Products
6. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Praznik, W.; Loeppert, R.; Viernstein, H.; Haslberger, A.G.; Unger, F.M. Dietary fiber and prebiotics. In Polysaccharides: Bioactivity and Biotechnology; Ramawat, K.G., Mérillon, J.-M., Eds.; Springer International Publishing: Cham, Germany, 2015; pp. 891–925. [Google Scholar]
- Kasper, H. Ernährungsmedizin und Diätetik, 9th ed.; Elsevier-Urban & Fischer: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Hofmann, A.F. Overview of bile secretion. In Comprehensive Physiology; American Physiological Society: Rockville, MD, USA, 2011; pp. 549–566. [Google Scholar]
- Gibson, G.R.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [PubMed]
- Roberfroid, M. Prebiotics and synbiotics: Concepts and nutritional properties. Br. J. Nutr. 1998, 80, S197–S202. [Google Scholar] [PubMed]
- Roberfroid, M.; Slavin, J. Nondigestible oligosaccharides. Crit. Rev. Food Sci. Nutr. 2000, 40, 461–480. [Google Scholar] [CrossRef] [PubMed]
- Hague, A.; Singh, B.; Paraskeva, C. Butyrate acts as a survival factor for colonic epithelial cells: Further fuel for the in vivo versus in vitro debate. Gastroenterology 1997, 112, 1036–1040. [Google Scholar] [CrossRef] [PubMed]
- Cieslik, E.; Topolska, K.; Pisulewski, P.M. Effect of inulin-type fructans on body weight gain and selected biochemical parameters at calcium hypoalimentation in rats. Pol. J. Food Nutr. Sci. 2009, 59, 163–168. [Google Scholar]
- Sotnikova, N.; Antsiferova, I.; Malyshkina, A. Cytokine network of eutopic and ectopic endometrium in women with adenomyosis. Am. J. Reprod. Immunol. 2002, 47, 251–255. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Diet, Nutrition and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation; World Health Organization: Geneva, Switzerland, 2003; pp. 54–71. [Google Scholar]
- Christodoulides, S.; Dimidi, E.; Fragkos, K.C.; Farmer, A.D.; Whelan, K.; Scott, S.M. Systematic review with meta-analysis: Effect of fibre supplementation on chronic idiopathic constipation in adults. Aliment. Pharmacol. Ther. 2016, 44, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Elleuch, M.; Bedigian, D.; Roiseux, O.; Besbes, S.; Blecker, C.; Attia, H. Dietary fibre and fibre-rich by-products of food processing: Characterisation, technological functionality and commercial applications: A review. Food Chem. 2011, 124, 411–421. [Google Scholar] [CrossRef]
- Dodevska, M.S.; Djordjevic, B.I.; Sobajic, S.S.; Miletic, I.D.; Djordjevic, P.B.; Dimitrijevic-Sreckovic, V.S. Characterisation of dietary fibre components in cereals and legumes used in serbian diet. Food Chem. 2013, 141, 1624–1629. [Google Scholar] [CrossRef] [PubMed]
- Decker, E.A.; Rose, D.J.; Stewart, D. Processing of oats and the impact of processing operations on nutrition and health benefits. Br. J. Nutr. 2014, 112, S58–S64. [Google Scholar] [CrossRef] [PubMed]
- Åman, P.; Graham, H. Analysis of total and insoluble mixed-linked (1→3),(1→4)-β-d-glucans in barley and oats. J. Agric. Food Chem. 1987, 35, 704–709. [Google Scholar] [CrossRef]
- Zhang, Z.; Smith, C.; Li, W. Extraction and modification technology of arabinoxylans from cereal by-products: A critical review. Food Res. Int. 2014, 65, 423–436. [Google Scholar] [CrossRef]
- Buksa, K.; Praznik, W.; Loeppert, R.; Nowotna, A. Characterization of water and alkali extractable arabinoxylan from wheat and rye under standardized conditions. J. Food Sci. Technol. 2016, 53, 1389–1398. [Google Scholar] [CrossRef] [PubMed]
- Buksa, K.; Nowotna, A.; Ziobro, R.; Praznik, W. Molecular properties of arabinoxylan fractions isolated from rye grain of different quality. J. Cereal Sci. 2014, 60, 368–373. [Google Scholar] [CrossRef]
- Roubroeks, J.; Andersson, R.; Åman, P. Structural features of (1→3),(1→4)-β-d-glucan and arabinoxylan fractions isolated from rye bran. Carbohydr. Polym. 2000, 42, 3–11. [Google Scholar] [CrossRef]
- Buksa, K.; Nowotna, A.; Praznik, W.; Gambuś, H.; Ziobro, R.; Krawontka, J. The role of pentosans and starch in baking of wholemeal rye bread. Food Res. Int. 2010, 43, 2045–2051. [Google Scholar] [CrossRef]
- Ahmad, Z.; Butt, M.S.; Ahmed, A.; Riaz, M.; Sabir, S.M.; Farooq, U.; Rehman, F.U. Effect of aspergillus niger xylanase on dough characteristics and bread quality attributes. J. Food Sci. Technol. 2014, 51, 2445–2453. [Google Scholar] [CrossRef] [PubMed]
- Capriles, V.D.; Arêas, J.A.G. Novel approaches in gluten-free breadmaking: Interface between food science, nutrition, and health. Compr. Rev. Food Sci. Food Saf. 2014, 13, 871–890. [Google Scholar] [CrossRef]
- Mansberger, A.; D’Amico, S.; Novalin, S.; Schmidt, J.; Tömösközi, S.; Berghofer, E.; Schoenlechner, R. Pentosan extraction from rye bran on pilot scale for application in gluten-free products. Food Hydrocoll. 2014, 35, 606–612. [Google Scholar] [CrossRef]
- Schuppan, D. Zöliakie. World Food Regul. Rev. 2016, 59, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Van Buul, V.J.; Brouns, F.J.P.H. Health effects of wheat lectins: A review. J. Cereal Sci. 2014, 59, 112–117. [Google Scholar] [CrossRef]
- De Punder, K.; Pruimboom, L. The dietary intake of wheat and other cereal grains and their role in inflammation. Nutrients 2013, 5, 771–787. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Jubete, L.; Arendt, E.; Gallagher, E. Nutritive value of pseudocereals and their increasing use as functional gluten-free ingredients. Trends Food Sci. Technol. 2010, 21, 106–113. [Google Scholar] [CrossRef]
- Torbica, A.; Hadnađev, M.; Dapčević, T. Rheological, textural and sensory properties of gluten-free bread formulations based on rice and buckwheat flour. Food Hydrocoll. 2010, 24, 626–632. [Google Scholar] [CrossRef]
- Bender, D.; Regner, M.; D’Amico, S.; Jäger, H.; Tömösközi, S.; Schoenlechner, R. Effect of differently extracted arabinoxylan on gluten-free sourdough-bread properties. J. Food Qual. 2018, 1–10. [Google Scholar] [CrossRef]
- Gambuś, H.; Sikora, M.; Ziobro, R. The effect of composition of hydrocolloids on properties of gluten-free bread. Acta Sci. Pol. Technol. Aliment. 2007, 6, 61–74. [Google Scholar]
- Falck, P.; Aronsson, A.; Grey, C.; Stålbrand, H.; Karlsson, E.N.; Adlercreutz, P. Production of arabinoxylan-oligosaccharide mixtures of varying composition from rye bran by a combination of process conditions and type of xylanase. Bioresour. Technol. 2014, 174, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Hartemink, R.; Schoustra, S.E.; Rombouts, F.M. Degradation of guar gum by intestinal bacteria. Biosci. Microflora 1999, 18, 17–25. [Google Scholar] [CrossRef]
- Hartemink, R.; Rombouts, F.M. Comparison of media for the detection of bifidobacteria, lactobacilli and total anaerobes from faecal samples. J. Microbiol. Methods 1999, 36, 181–192. [Google Scholar] [CrossRef]
- Ramsden, L. Plant and algal gums and mucilages. In Chemical and Functional Properties of Food Saccharides; Tomasik, P., Ed.; CRC Press: Boca Raton, FL, USA, 2003; p. 231. [Google Scholar]
- Prajapati, V.D.; Jani, G.K.; Moradiya, N.G.; Randeria, N.P.; Nagar, B.J.; Naikwadi, N.N.; Variya, B.C. Galactomannan: A versatile biodegradable seed polysaccharide. Int. J. Biol. Macromol. 2013, 60, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.J.; Smith, B.G. Plant cell walls and cell-wall polysaccharides: Structures, properties and uses in food products. Int. J. Food Sci. Tech. 2006, 41, 129–143. [Google Scholar] [CrossRef]
- Wielinga, W.C. Galactomannans. In Handbook of Hydrocolloids; Phillips, G.O., Williams, P.A., Eds.; Woodhead Publishing: Shaston, UK, 2000; pp. 413–423. [Google Scholar]
- Yoko, K. Guar Gum; Joint FAO/WHO Expert Committee on Food Additives: Rome, Italy, 2008; p. 4. [Google Scholar]
- Moreira, L. An overview of mannan structure and mannan-degrading enzyme systems. Appl. Microbiol. Biotechnol. 2008, 79, 165. [Google Scholar] [CrossRef] [PubMed]
- Guillon, F.; Champ, M.-J. Carbohydrate fractions of legumes: Uses in human nutrition and potential for health. Br. J. Nutr. 2002, 88, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.-J.; Chu, D.-C.; Juneja, L.R. Chemical and physical properties, safety and application of partially hydrolized guar gum as dietary fiber. J. Clin. Biochem. Nutr. 2008, 42, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zartl, B.; Silberbauer, K.; Loeppert, R.; Viernstein, H.; Praznik, W.; Mueller, M. Fermentation of non-digestible raffinose family oligosaccharides and galactomannans by probiotics. Food Funct. 2018, 9, 1638–1646. [Google Scholar] [CrossRef] [PubMed]
- Praznik, W.; Cieslik, E.; Huber, A. Fructans: Occurrence and application in food. In Chemical and Functional Properties of Food Saccharides; Tomasik, P., Ed.; CRC Press: Boca Raton, FL, USA, 2003; pp. 197–215. [Google Scholar]
- Praznik, W.; Löppert, R.; Huber, A. Analysis and molecular composition of fructans from different plant sources. In Recent Advances in Fructooligosaccharides Research; Shiomi, N., Benkeblia, N., Onodera, S., Eds.; Research Signpost: Kerala, India, 2007; pp. 93–117. [Google Scholar]
- French, A.; Waterhouse, A. Chemical structure and characteristics. In Science and Technology of Fructans; Suzuki, M., Chatterton, N.J., Eds.; CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar]
- Praznik, W.; Löppert, R.; Cruz Rubio, J.M.; Zangger, K.; Huber, A. Structure of fructo-oligosaccharides from leaves and stem of agave tequilana weber, var. Azul. Carbohydr. Res. 2013, 381, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M.B. Introducing inulin-type fructans. Br. J. Nutr. 2005, 93, S13–S25. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M. Dietary fiber, inulin, and oligofructose: A review comparing their physiological effects. Crit. Rev. Food Sci. Nutr. 1993, 33, 103–148. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Beatty, E.R.; Wang, X.; Cummings, J.H. Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. Gastroenterology 1995, 108, 975–982. [Google Scholar] [CrossRef]
- Moser, M.; Sentko, A.; Alexiou, H. Inulin and health benefits. In Polysaccharides: Bioactivity and Biotechnology; Ramawat, K.G., Mérillon, J.-M., Eds.; Springer International Publishing: Cham, Germany, 2015; pp. 675–715. [Google Scholar]
- Mueller, M.; Reiner, J.; Fleischhacker, L.; Viernstein, H.; Loeppert, R.; Praznik, W. Growth of selected probiotic strains with fructans from different sources relating to degree of polymerization and structure. J. Funct. Foods 2016, 24, 264–275. [Google Scholar] [CrossRef]
- Mueller, M.; Schwarz, S.; Viernstein, H.; Loeppert, R.; Praznik, W. Growth of selected probiotic strains with fructans from agaves and chicory. Agro. Food Industry Hi. Tech. 2016, 27, 40–43. [Google Scholar]
- Peshev, D.; Van den Ende, W. Fructans: Prebiotics and immunomodulators. J. Funct. Foods 2014, 8, 348–357. [Google Scholar] [CrossRef]
- Orafti. Description, production, natural occurrence and history of inulin and oligofructose. Orafti Product Book 08/99, Tienen, Belgium, 1999. [Google Scholar]
- Praznik, W.; Cieślik, E.; Filipiak-Florkiewicz, A. Soluble dietary fibres in jerusalem artichoke powders: Composition and application in bread. Food Nahrung 2002, 46, 151–157. [Google Scholar] [CrossRef]
- Loeppert, R.; Huber, A.; Villalvazo Naranjo, J.; Praznik, W. Molecular and Physicochemical Characteristics of Fructan during Technological Processing of Agave Tequilana Weber Var. Azul, Proceedings of the 6th International Fructan Symposium, Hokkaido, Japan,27–31 July 2008; Benkeblia, N., Shiomi, N., Eds.; Dynamic Biochemistry, Process Biotechnology and Molecular Biology: Hokkaido, Japan, 2009; pp. 22–33. [Google Scholar]
- Allsopp, P.; Possemiers, S.; Campbell, D.; Oyarzábal, I.S.; Gill, C.; Rowland, I. An exploratory study into the putative prebiotic activity of fructans isolated from agave angustifolia and the associated anticancer activity. Anaerobe 2013, 22, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Koenen, M.E.; Cruz Rubio, J.M.; Mueller, M.; Venema, K. The effect of agave fructan products on the activity and composition of the microbiota determined in a dynamic in vitro model of the human proximal large intestine. J. Funct. Foods 2016, 22, 201–210. [Google Scholar] [CrossRef]
- Lopez-Velazquez, G.; Parra-Ortiz, M.; Mora Ide, L.; Garcia-Torres, I.; Enriquez-Flores, S.; Alcantara-Ortigoza, M.A.; Angel, A.G.; Velazquez-Aragon, J.; Ortiz-Hernandez, R.; Cruz-Rubio, J.M.; et al. Effects of fructans from mexican agave in newborns fed with infant formula: A randomized controlled trial. Nutrients 2015, 7, 8939–8951. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.H.; Yu, N.; Gray, G.R.; Ralph, J.; Anderson, L.; Marlett, J.A. The gel-forming polysaccharide of psyllium husk (plantago ovata forsk). Carbohydr. Res. 2004, 339, 2009–2017. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Pandey, J.; Kumar, P.; Raj, V. Psyllium mucilage and its use in pharmaceutical field. Curr. Syn. Sys. Bio. 2017, 5, 134. [Google Scholar] [CrossRef]
- Yu, L.; Yakubov, G.E.; Zeng, W.; Xing, X.; Stenson, J.; Bulone, V.; Stokes, J.R. Multi-layer mucilage of plantago ovata seeds: Rheological differences arise from variations in arabinoxylan side chains. Carbohydr. Polym. 2017, 165, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.W.; Zettwoch, N.; Feldman, T.; Tietyen-Clark, J.; Oeltgen, P.; Bishop, C.W. Cholesterol-lowering effects of psyllium hydrophilic mucilloid for hypercholesterolemic men. Arch. Intern. Med. 1988, 148, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.H.; Wang, H.; Chen, X.Y.; Wang, B.S.; Rong, Z.X.; Wang, B.S.; Su, B.H.; Chen, H.Z. Time- and dose-dependent effect of psyllium on serum lipids in mild-to-moderate hypercholesterolemia: A meta-analysis of controlled clinical trials. Eur. J. Clin. Nutr. 2008, 63, 821. [Google Scholar] [CrossRef] [PubMed]
- Gibb, R.D.; McRorie, J.J.W.; Russell, D.A.; Hasselblad, V.; D’Alessio, D.A. Psyllium fiber improves glycemic control proportional to loss of glycemic control: A meta-analysis of data in euglycemic subjects, patients at risk of type 2 diabetes mellitus, and patients being treated for type 2 diabetes mellitus. Am. J. Clin. Nutr. 2015, 102, 1604–1614. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Kaur, R.; Punia, S. Characterization of mucilages extracted from different flaxseed (linum usitatissiumum L.) cultivars: A heteropolysaccharide with desirable functional and rheological properties. Int. J. Biol. Macromol. 2018, 117, 919–927. [Google Scholar] [CrossRef] [PubMed]
- Qian, K.Y.; Cui, S.W.; Wu, Y.; Goff, H.D. Flaxseed gum from flaxseed hulls: Extraction, fractionation, and characterization. Food Hydrocoll. 2012, 28, 275–283. [Google Scholar] [CrossRef]
- Kaewmanee, T.; Bagnasco, L.; Benjakul, S.; Lanteri, S.; Morelli, C.F.; Speranza, G.; Cosulich, M.E. Characterisation of mucilages extracted from seven italian cultivars of flax. Food Chem. 2014, 148, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Naran, R.; Chen, G.; Carpita, N.C. Novel rhamnogalacturonan i and arabinoxylan polysaccharides of flax seed mucilage. Plant Physiol. 2008, 148, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Fedeniuk, R.W.; Biliaderis, C.G. Composition and physicochemical properties of linseed (Linum usitatissimum L.) mucilage. J. Agric. Food Chem. 1994, 42, 240–247. [Google Scholar] [CrossRef]
- Ray, S.; Paynel, F.; Morvan, C.; Lerouge, P.; Driouich, A.; Ray, B. Characterization of mucilage polysaccharides, arabinogalactanproteins and cell-wall hemicellulosic polysaccharides isolated from flax seed meal: A wealth of structural moieties. Carbohydr. Polym. 2013, 93, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Praznik, W.; Cavarkapa, A.; Unger, F.M.; Loeppert, R.; Holzer, W.; Viernstein, H.; Mueller, M. Molecular dimensions and structural features of neutral polysaccharides from the seed mucilage of hyptis suaveolens L. Food Chem. 2017, 221, 1997–2004. [Google Scholar] [CrossRef] [PubMed]
- Aspinall, G.O.; Capek, P.; Carpenter, R.C.; Gowda, D.C.; Szafranek, J. A novel l-fuco-4-o-methyl-d-glucurono-d-xylan from hyptis suaveolens. Carbohydr. Res. 1991, 214, 107–113. [Google Scholar] [CrossRef]
- Gowda, D.C. Polysaccharide components of the seed-coat mucilage from hyptis suaveolens. Phytochemistry 1984, 23, 337–338. [Google Scholar] [CrossRef]
- Ngozi, L.U.; Ugochukwu, N.; Ifeoma, P.U.; Charity, E.A.; Chinyelu, I.E. The efficacy of hyptis suaveolens: A review of its nutritional and medicinal applications. European J. Med. Plants 2014, 4, 661–674. [Google Scholar] [CrossRef]
- Prince, P.S.; Ram, K.R.; Chaudhary, A.; Dinesh, G.; Sharma, V.K. Hyptis suaveolens (L.) poit: A phyto-pharmacological review. Int. J. Chem. Pharm. Sci. 2013, 4, 1–11. [Google Scholar]
- Mueller, M.; Čavarkapa, A.; Unger, F.M.; Viernstein, H.; Praznik, W. Prebiotic potential of neutral oligo- and polysaccharides from seed mucilage of hyptis suaveolens. Food Chem. 2017, 221, 508–514. [Google Scholar] [CrossRef] [PubMed]
- McCleary, B.V.; Matheson, N.K. Action patterns and substrate-binding requirements of β-d-mannanase with mannosaccharides and mannan-type polysaccharides. Carbohydr. Res. 1983, 119, 191–219. [Google Scholar] [CrossRef]
- Lin, K.-Y.; Daniel, J.R.; Whistler, R.L. Structure of chia seed polysaccharide exudate. Carbohydr. Polym. 1994, 23, 13–18. [Google Scholar] [CrossRef]
- Muñoz, L.A.; Cobos, A.; Diaz, O.; Aguilera, J.M. Chia seeds: Microstructure, mucilage extraction and hydration. J. Food Eng. 2012, 108, 216–224. [Google Scholar] [CrossRef]
- Reyes-Caudillo, E.; Tecante, A.; Valdivia-López, M.A. Dietary fibre content and antioxidant activity of phenolic compounds present in mexican chia (salvia hispanica L.) seeds. Food Chem. 2008, 107, 656–663. [Google Scholar] [CrossRef]
- Razavi, S.M.A.; Mortazavi, S.A.; Matia-Merino, L.; Hosseini-Parvar, S.H.; Motamedzadegan, A.; Khanipour, E. Optimisation study of gum extraction from basil seeds (ocimum basilicum L.). Int. J. Food Sci. Tech. 2009, 44, 1755–1762. [Google Scholar] [CrossRef]
- Kadam, P.V.; Yadav, K.N.; Jagdale, S.K.; Shivatare, R.S.; Bhilwade, S.K.; Patil, M.J. Evaluation of ocimum sanctum and ocimum basillicum mucilage- as a pharmaceutical excipient. J. Chem. Pharm. Res. 2012, 4, 1950–1955. [Google Scholar]
- Praznik, W.; Loeppert, R.; Viernstein, H.; Mueller, M. Characterization of Heteropolysaccharides from Seed Mucilage of Chia (salvia hispanica L.), Basil (ocimum basilicum L.) and Flax (linum usitatissimum L.) and Their Healthy Functionality, Proceedings of the 12th International Conference on Polysaccharides-Glycoscience, Prague, Czech, 19–21 October 2016; Chemical Society: Prague, Czech, 2016; pp. 9–11. [Google Scholar]
- Medina-Torres, L.; Brito-De La Fuente, E.; Torrestiana-Sanchez, B.; Katthain, R. Rheological properties of the mucilage gum (opuntia ficus-indica). Food Hydrocoll. 2000, 14, 417–424. [Google Scholar] [CrossRef]
- McGarvie, D.; Parolis, H. Methylation analysis of the mucilage of opuntia ficus-indica. Carbohydr. Res. 1981, 88, 305–314. [Google Scholar] [CrossRef]
- Nuñez-López, M.A.; Paredes-López, O.; Reynoso-Camacho, R. Functional and hypoglycemic properties of nopal cladodes (o. Ficus-indica) at different maturity stages using in vitro and in vivo tests. J. Agric. Food Chem. 2013, 61, 10981–10986. [Google Scholar] [CrossRef] [PubMed]
- Guevara-Arauza, J.C.; de Jesús Ornelas-Paz, J.; Pimentel-González, D.J.; Rosales Mendoza, S.; Soria Guerra, R.E.; Paz Maldonado, L.M.T. Prebiotic effect of mucilage and pectic-derived oligosaccharides from nopal (opuntia ficus-indica). Food Sci. Biotechnol. 2012, 21, 997–1003. [Google Scholar] [CrossRef]
- Douglas, L.C.; Sanders, M.E. Probiotics and prebiotics in dietetics practice. J. Am. Diet. Assoc. 2008, 108, 510–521. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cruz-Rubio, J.M.; Loeppert, R.; Viernstein, H.; Praznik, W. Trends in the Use of Plant Non-Starch Polysaccharides within Food, Dietary Supplements, and Pharmaceuticals: Beneficial Effects on Regulation and Wellbeing of the Intestinal Tract. Sci. Pharm. 2018, 86, 49. https://doi.org/10.3390/scipharm86040049
Cruz-Rubio JM, Loeppert R, Viernstein H, Praznik W. Trends in the Use of Plant Non-Starch Polysaccharides within Food, Dietary Supplements, and Pharmaceuticals: Beneficial Effects on Regulation and Wellbeing of the Intestinal Tract. Scientia Pharmaceutica. 2018; 86(4):49. https://doi.org/10.3390/scipharm86040049
Chicago/Turabian StyleCruz-Rubio, José Manuel, Renate Loeppert, Helmut Viernstein, and Werner Praznik. 2018. "Trends in the Use of Plant Non-Starch Polysaccharides within Food, Dietary Supplements, and Pharmaceuticals: Beneficial Effects on Regulation and Wellbeing of the Intestinal Tract" Scientia Pharmaceutica 86, no. 4: 49. https://doi.org/10.3390/scipharm86040049
APA StyleCruz-Rubio, J. M., Loeppert, R., Viernstein, H., & Praznik, W. (2018). Trends in the Use of Plant Non-Starch Polysaccharides within Food, Dietary Supplements, and Pharmaceuticals: Beneficial Effects on Regulation and Wellbeing of the Intestinal Tract. Scientia Pharmaceutica, 86(4), 49. https://doi.org/10.3390/scipharm86040049