Are Steroid Hormones Dysregulated in Autistic Girls?
Abstract
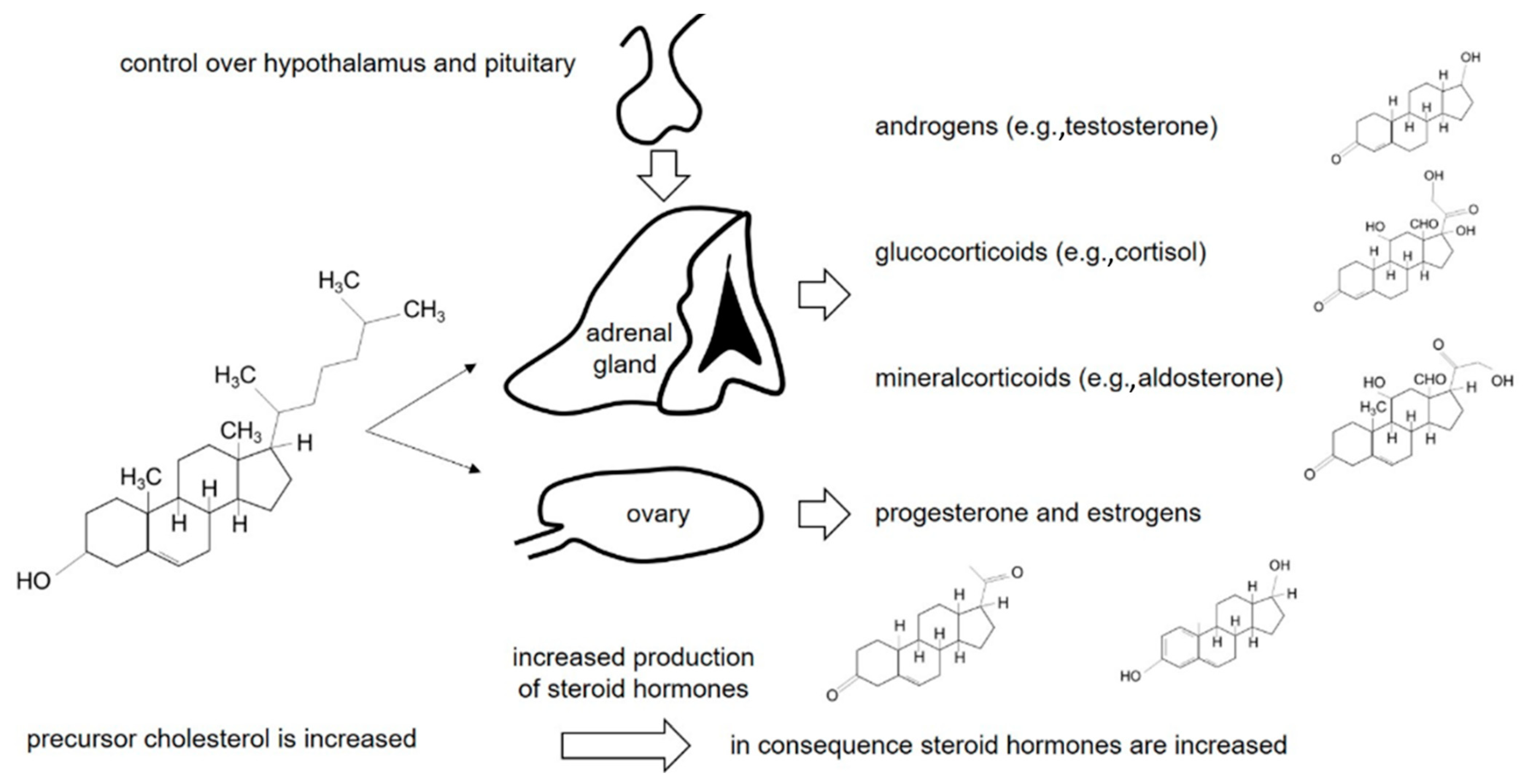
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Methods
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Spratt, E.; Nicholas, J.S.; Brady, K.T.; Carpenter, L.; Hatcher, C.R.; Meekins, K.A.; Furlanetto, R.W.; Charles, J.M. Enhanced Cortisol Response to Stress in Children in Autism. J. Autism Dev. Disord. 2012, 42, 75–81. [Google Scholar] [CrossRef]
- Hollocks, M.; Howlin, P.; Papadopoulos, A.S.; Khondoker, M.; Simonoff, E. Differences in HPA-axis and heart rate responsiveness to psychosocial stress in children with autism spectrum disorders with and without co-morbid anxiety. Psychoneuroendocrinology 2014, 46, 32–45. [Google Scholar] [CrossRef]
- Jacobson, L. Hypothalamic-Pituitary-Adrenocortical Axis: Neuropsychiatric Aspects. Compr. Physiol. 2014, 4, 715–738. [Google Scholar] [PubMed]
- Taylor, J.L.; Corbett, B.A. A review of rhythm and responsiveness of cortisol in individuals with autism spectrum disorders. Psychoneuroendocrinology 2014, 49, 207–228. [Google Scholar] [CrossRef] [PubMed]
- Tani, P.; Lindberg, N.; Matto, V.; Appelberg, B.; Wendt, T.N.-V.; Von Wendt, L.; Porkka-Heiskanen, T. Higher plasma ACTH levels in adults with Asperger syndrome. J. Psychosom. Res. 2005, 58, 533–536. [Google Scholar] [CrossRef] [PubMed]
- Coleman, M.; Gillberg, C. The Autisms, 4th ed.; Oxford University Press: New York, NY, USA, 2012. [Google Scholar]
- Gillberg, C.; Fernell, E.; Kočovská, E.; Minnis, H.; Bourgeron, T.; Thompson, L.; Allely, C.S. The role of cholesterol metabolism and various steroid abnormalities in autism spectrum disorders: A hypothesis paper. Autism Res. 2017, 10, 1022–1044. [Google Scholar] [CrossRef]
- Gillberg, C. Debate and Argument: Is Autism a Pervasive Developmental Disorder? J. Child Psychol. Psychiatry 1991, 32, 1169–1170. [Google Scholar] [CrossRef]
- Bejerot, S.; Eriksson, J. Sexuality and Gender Role in Autism Spectrum Disorder: A Case Control Study. PLoS ONE 2014, 9, e87961. [Google Scholar] [CrossRef]
- Posserud, M.B.; Lundervold, A.J.; Gillberg, C. Autistic features in a total population of 7–9-year-old children assessed by the ASSQ (Autism Spectrum Screening Questionnaire). J. Child Psychology Psychiatry 2006, 47, 167–175. [Google Scholar] [CrossRef]
- Crider, A.; Thakkar, R.; Ahmed, A.O.; Pillai, A. Dysregulation of estrogen receptor beta (ERβ), aromatase (CYP19A1), and ER co-activators in the middle frontal gyrus of autism spectrum disorder subjects. Mol. Autism 2014, 5, 46. [Google Scholar] [CrossRef]
- Wing, L. Sex ratios in early childhood autism and related conditions. Psychiatry Res. 1981, 5, 129–137. [Google Scholar] [CrossRef]
- Bitsika, V.; Sharpley, C.; Sweeney, J.A.; McFarlane, J.R. HPA and SAM axis responses as correlates of self- vs parental ratings of anxiety in boys with an Autistic Disorder. Physiol. Behav. 2014, 127, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kočovská, E.; Fernell, E.; Billstedt, E.; Minnis, H.; Gillberg, C. Vitamin D and autism: Clinical review. Res. Dev. Disabil. 2012, 33, 1541–1550. [Google Scholar] [CrossRef] [PubMed]
- Kocovska, E.; Andosdottir, G.; Weihe, P.; Halling, J.; Fernell, E.; Stóra, T.; Biskupstø, R.; Gillberg, I.; Shea, R.L.; Billstedt, E.; et al. Vitamin d in the general population of young adults with autism in the faroe islands. J. Autism Dev. Disord. 2014, 44, 2996–3005. [Google Scholar] [CrossRef] [PubMed]
- Fernell, E.; Bejerot, S.; Westerlund, J.; Miniscalco, C.; Simila, H.; Eyles, D.W.; Gillberg, C.; Humble, M.B. Autism spectrum disorder and low vitamin D at birth: A sibling control study. Mol. Autism 2015, 6, 3. [Google Scholar] [CrossRef]
- Hamza, R.T.; Hewedi, D.H.; Ismail, M.A. Basal and Adrenocorticotropic Hormone Stimulated Plasma Cortisol Levels Among Egyptian Autistic Children: Relation to Disease Severity. Ital. J. Pediatr. 2010, 36, 71. [Google Scholar] [CrossRef]
- Brosnan, M.; Turner-Cobb, J.; Munro-Naan, Z.; Jessop, D. Absence of a normal Cortisol Awakening Response (CAR) in adolescent males with Asperger Syndrome (AS). Psychoneuroendocrinology 2009, 34, 1095–1100. [Google Scholar] [CrossRef]
- Marinović-Ćurin, J.; Marinović-Terzić, I.; Bujas-Petković, Z.; Zekan, L.; Škrabić, V.; Đogaš, Z.; Terzić, J. Slower cortisol response during ACTH stimulation test in autistic children. Eur. Child Adolesc. Psychiatry 2007, 17, 39–43. [Google Scholar] [CrossRef]
- Hoshino, Y.; Ohno, Y.; Murata, S.; Yokoyama, F.; Kaneko, M.; Kumashiro, H. Dexamethasone Suppression Test in Autistic Children. Psychiatry Clin. Neurosci. 1984, 38, 445–449. [Google Scholar] [CrossRef]
- Hoshino, Y.; Yokoyama, F.; Watanabe, M.; Murata, S.; Kaneko, M.; Kumashiro, H. The Diurnal Variation and Response to Dexamethasone Suppression Test of Saliva Cortisol Level in Autistic Children. Psychiatry Clin. Neurosci. 1987, 41, 227–235. [Google Scholar] [CrossRef]
- Curin, J.M.; Terzić, J.; Petković, Z.B.; Zekan, L.; Terzić, I.M.; Susnjara, I.M. Lower cortisol and higher ACTH levels in individuals with autism. J. Autism Dev. Disord. 2003, 33, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Iwata, K.; Matsuzaki, H.; Miyachi, T.; Shimmura, C.; Suda, S.; Tsuchiya, K.J.; Matsumoto, K.; Suzuki, K.; Iwata, Y.; Nakamura, K.; et al. Investigation of the serum levels of anterior pituitary hormones in male children with autism. Mol. Autism 2011, 2, 16. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-J.; Shou, X.-J.; Li, J.; Jia, M.-X.; Zhang, J.-S.; Guo, Y.; Wei, Q.-Y.; Zhang, X.-T.; Han, S.-P.; Zhang, R.; et al. Mothers of Autistic Children: Lower Plasma Levels of Oxytocin and Arg-Vasopressin and a Higher Level of Testosterone. PLoS ONE 2013, 8, e74849. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Auyeung, B.; Nørgaard-Pedersen, B.; Hougaard, D.M.; Abdallah, M.W.; Melgaard, L.; Cohen, A.S.; Chakrabarti, B.; Ruta, L.; Lombardo, M.V. Elevated fetal steroidogenic activity in autism. Mol. Psychiatry 2014, 20, 369–376. [Google Scholar] [CrossRef] [PubMed]
- El-Baz, F.; Hamza, R.T.; Ayad, M.S.; Mahmoud, N.H. Hyperandrogenemia in male autistic children and adolescents: Relation to disease severity. Int. J. Adolesc. Med. Heal. 2014, 26, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Ruta, L.; Ingudomnukul, E.; Taylor, K.; Chakrabarti, B.; Baron-Cohen, S. Increased serum androstenedione in adults with autism spectrum conditions. Psychoneuroendocrinology 2011, 36, 1154–1163. [Google Scholar] [CrossRef]
- Ingudomnukul, E.; Baron-Cohen, S.; Wheelwright, S.J.; Knickmeyer, R. Elevated rates of testosterone-related disorders in women with autism spectrum conditions. Horm. Behav. 2007, 51, 597–604. [Google Scholar] [CrossRef]
- Knickmeyer, R.; Baron-Cohen, S.; Fane, B.A.; Wheelwright, S.J.; Mathews, G.A.; Conway, G.S.; Brook, C.G.; Hines, M. Androgens and autistic traits: A study of individuals with congenital adrenal hyperplasia. Horm. Behav. 2006, 50, 148–153. [Google Scholar] [CrossRef]
- Majewska, M.D.; Hill, M.; Urbanowicz, E.; Rok-Bujko, P.; Bieńkowski, P.; Namysłowska, I.; Mierzejewski, P. Marked elevation of adrenal steroids, especially androgens, in saliva of prepubertal autistic children. Eur. Child Adolesc. Psychiatry 2013, 23, 485–498. [Google Scholar] [CrossRef]
- Takagishi, H.; Takahashi, T.; Yamagishi, T.; Shinada, M.; Inukai, K.; Tanida, S.; Mifune, N.; Horita, Y.; Hashimoto, H.; Yang, Y.; et al. Salivary testosterone levels and autism-spectrum quotient in adults. Neuro Endocrinol. Lett. 2010, 31, 837–841. [Google Scholar]
- Bradstreet, J.J.; Smith, S.; Granpeesheh, R.; El-Dahr, J.M.; Rossignol, D. Spironolactone might be a desirable immunologic and hormonal intervention in autism spectrum disorders. Med. Hypotheses 2007, 68, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Kolkhof, P.; Bärfacker, L. 30 YEARS OF THE MINERALOCORTICOID RECEPTOR: Mineralocorticoid receptor antagonists: 60 years of research and development. J. Endocrinol. 2017, 234, T125–T140. [Google Scholar] [CrossRef] [PubMed]
- Beggiato, A.; Peyre, H.; Maruani, A.; Scheid, I.; Rastam, M.; Amsellem, F.; Gillberg, C.I.; Leboyer, M.; Bourgeron, T.; Gillberg, C.; et al. Gender differences in autism spectrum disorders: Divergence among specific core symptoms. Autism Res. 2016, 10, 680–689. [Google Scholar] [CrossRef] [PubMed]
- Hindström, S. The knowledge about autism in girls must increase. Lakartidningen 2017, 114. [Google Scholar]
- Popper, K.R. Logik der Forschung. Tübingen: Mohr Siebeck; Original Work Published: Tuebingen, Germany, 1934. [Google Scholar]
- Aufdenblatten, M.; Baumann, M.; Raio, L.; Dick, B.; Frey, B.M.; Schneider, H.; Surbek, D.; Hocher, B.; Mohaupt, M. Prematurity Is Related to High Placental Cortisol in Preeclampsia. Pediatr. Res. 2009, 65, 198–202. [Google Scholar] [CrossRef]
- Henschkowski, J.; Stuck, A.E.; Frey, B.M.; Gillmann, G.; Dick, B.; Frey, F.J.; Mohaupt, M.G. Age-dependent decrease in 11β-hydroxysteroid dehydrogenase type 2 (11β-HSD2) activity in hypertensive patients. Am. J. Hypertens. 2008, 21, 644–649. [Google Scholar] [CrossRef]
- Krone, N.; Hughes, B.A.; Lavery, G.G.; Stewart, P.M.; Arlt, W.; Shackleton, C.H. Gas chromatography/mass spectrometry (GC/MS) remains a pre-eminent discovery toOl in clinical steroid investigations even in the era of fast liquid chromatography tandem mass spectrometry (LC/MS/MS). J Steroid Biochem. Mol. Biol. 2010, 121, 496–504. [Google Scholar] [CrossRef]
- Brämswig, J.; Dübbers, A. Disorders of Pubertal Development. Dtsch. Aerzteblatt Online 2009, 106, 295–304. [Google Scholar] [CrossRef]
- Brix, N.; Ernst, A.; Lauridsen, L.L.B.; Parner, E.; Støvring, H.; Olsen, J.; Henriksen, T.B.; Ramlau-Hansen, C.H. Timing of puberty in boys and girls: A population-based study. Paediatr. Périnat. Epidemiol. 2018, 33, 70–78. [Google Scholar] [CrossRef]
- Tordjman, S.; Anderson, G.M.; McBride, P.A.; Hertzig, M.E.; Snow, M.E.; Hall, L.M.; Ferrari, P.; Cohen, D.J. Plasma androgens in autism. J. Autism Dev. Disord. 1995, 25, 295–304. [Google Scholar] [CrossRef]
- Lachenbruch, P.A.; Cohen, J. Statistical Power Analysis for the Behavioral Sciences (2nd ed.). J. Am. Stat. Assoc. 1989, 84, 1096. [Google Scholar] [CrossRef]
- Uanhoro, J.O. Effect size calculators. 2017. Available online: https://effect-size-calculator.herokuapp.com/ (accessed on 2 March 2020).
- Pallant, J.F. SPSS Survival Manual: A Step by Step Guide to Data Analysis Using SPSS for Windows; Allen & Unwin.: Crows Nest, Australia, 2005. [Google Scholar]
- Ishimoto, H.; Ginzinger, D.G.; Jaffe, R.B. Adrenocorticotropin Preferentially Up-Regulates Angiopoietin 2 in the Human Fetal Adrenal Gland: Implications for Coordinated Adrenal Organ Growth and Angiogenesis. J. Clin. Endocrinol. Metab. 2006, 91, 1909–1915. [Google Scholar] [CrossRef] [PubMed]
- Ishimoto, H.; Minegishi, K.; Higuchi, T.; Furuya, M.; Asai, S.; Kim, S.H.; Tanaka, M.; Yoshimura, Y.; Jaffe, R. The periphery of the human fetal adrenal gland is a site of angiogenesis: Zonal differential expression and regulation of angiogenic factors. J. Clin. Endocrinol. Metab. 2008, 93, 2402–2408. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hitoshi, I.; Robert, B. Development and Function of the Human Fetal Adrenal Cortex: A Key Component in the Feto-Placental Unit. Endocr. Rev. 2011, 32, 317–355. [Google Scholar]
- Hu, V.; Sarachana, T.; Sherrard, R.M.; Kocher, K. Investigation of sex differences in the expression of RORA and its transcriptional targets in the brain as a potential contributor to the sex bias in autism. Mol. Autism 2015, 6, 7. [Google Scholar] [CrossRef]
- Sarachana, T.; Xu, M.; Wu, R.-C.; Hu, V. Sex Hormones in Autism: Androgens and Estrogens Differentially and Reciprocally Regulate RORA, a Novel Candidate Gene for Autism. PLoS ONE 2011, 6, e17116. [Google Scholar] [CrossRef]
- Sarachana, T.; Hu, V. Differential recruitment of coregulators to the RORA promoter adds another layer of complexity to gene (dys) regulation by sex hormones in autism. Mol. Autism 2013, 4, 39. [Google Scholar] [CrossRef]
- Chakrabarti, B.; Dudbridge, F.; Kent, L.; Wheelwright, S.J.; Hill-Cawthorne, G.A.; Allison, C.; Banerjee-Basu, S.; Baron-Cohen, S. Genes related to sex steroids, neural growth, and social-emotional behavior are associated with autistic traits, empathy, and Asperger syndrome. Autism Res. 2009, 2, 157–177. [Google Scholar] [CrossRef]
- The Autism Genome Project. Consortium. Mapping autism risk loci using genetic linkage and chromosomal rearrangements. Nat. Genet. 2007, 39, 319–328. [Google Scholar] [CrossRef]
- Jacquemont, M.-L.; Sanlaville, D.; Redon, R.; Raoul, O.; Cormier-Daire, V.; Lyonnet, S.; Amiel, J.; Le Merrer, M.; Heron, D.; De Blois, M.-C.; et al. Array-based comparative genomic hybridisation identifies high frequency of cryptic chromosomal rearrangements in patients with syndromic autism spectrum disorders. J. Med. Genet. 2006, 43, 843–849. [Google Scholar] [CrossRef]
- Cook, E.H., Jr.; Scherer, S.W. Copy-number variations associated with neuropsychiatric conditions. Nature 2008, 455, 919–923. [Google Scholar] [CrossRef] [PubMed]
- Levy, D.; Ronemus, M.; Yamrom, B.; Lee, Y.-H.; Leotta, A.; Kendall, J.; Marks, S.; Lakshmi, B.; Pai, D.; Ye, K.; et al. Rare De Novo and Transmitted Copy-Number Variation in Autistic Spectrum Disorders. Neuron 2011, 70, 886–897. [Google Scholar] [CrossRef] [PubMed]
- Anney, R.; Klei, L.; Pinto, D.; Regan, R.; Conroy, J.; Magalhães, T.R.; Correia, C.; Abrahams, B.S.; Sykes, N.; Pagnamenta, A.T.; et al. A genome-wide scan for common alleles affecting risk for autism. Hum. Mol. Genet. 2010, 19, 4072–4082. [Google Scholar] [CrossRef] [PubMed]
- Myers, R.A.; Casals, F.; Gauthier, J.; Hamdan, F.F.; Keebler, J.; Boyko, A.R.; Bustamante, C.D.; Piton, A.; Spiegelman, D.; Henrion, E.; et al. A Population Genetic Approach to Mapping Neurological Disorder Genes Using Deep Resequencing. PLoS Genet. 2011, 7, e1001318. [Google Scholar] [CrossRef] [PubMed]
- Iossifov, I.; Ronemus, M.; Levy, D.; Wang, Z.; Hakker, I.; Rosenbaum, J.; Yamrom, B.; Lee, Y.-H.; Narzisi, G.; Leotta, A.; et al. De Novo Gene Disruptions in Children on the Autistic Spectrum. Neuron 2012, 74, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Neale, B.M.; Kou, Y.; Liu, L.; Ma’Ayan, A.; Samocha, K.; Sabo, A.; Lin, C.-F.; Stevens, C.; Wang, L.-S.; Makarov, V.; et al. Patterns and rates of exonic de novo mutations in autism spectrum disorders. Nature 2012, 485, 242–245. [Google Scholar] [CrossRef]
- O’Roak, B.; Vives, L.; Girirajan, S.; Karakoc, E.; Krumm, N.; Coe, B.P.; Levy, R.; Ko, A.; Lee, C.; Smith, J.D.; et al. Sporadic autism exomes reveal a highly interconnected protein network of de novo mutations. Nature 2012, 485, 246–250. [Google Scholar] [CrossRef]
- Sanders, S.J.; Murtha, M.T.; Gupta, A.R.; Murdoch, J.D.; Raubeson, M.J.; Willsey, A.J.; Ercan-Sencicek, A.G.; DiLullo, N.M.; Parikshak, N.N.; Stein, J.; et al. De novo mutations revealed by whole-exome sequencing are strongly associated with autism. Nature 2012, 485, 237–241. [Google Scholar] [CrossRef]
- Menashe, I.; Grange, P.; Larsen, E.C.; Banerjee-Basu, S.; Mitra, P.P. Co-expression Profiling of Autism Genes in the Mouse Brain. PLoS Comput. Boil. 2013, 9, 1003128. [Google Scholar] [CrossRef]
- Basu, S.N.; Kollu, R.; Banerjee-Basu, S. AutDB: A gene reference resource for autism research. Nucleic Acids Res. 2008, 37, D832–D836. [Google Scholar] [CrossRef]
- Vorstman, J.A.S.; Parr, J.R.; Moreno-De-Luca, D.; Anney, R.; Nurnberger, J.I., Jr.; Hallmayer, J.F. Autism genetics: Opportunities and challenges for clinical translation. Nat. Rev. Genet. 2017, 18, 362–376. [Google Scholar] [CrossRef] [PubMed]
- Garber, K. Neuroscience - Autism’s cause may reside in abnormalities at the synapse. Science 2007, 317, 190–191. [Google Scholar] [CrossRef] [PubMed]
- Bourgeron, T. A synaptic trek to autism. Curr. Opin. Neurobiol. 2009, 19, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Durand, C.M.; Betancur, C.; Boeckers, T.M.; Bockmann, J.; Chaste, P.; Fauchereau, F.; Nygren, G.; Rastam, M.; Gillberg, I.C.; Anckarsäter, H.; et al. Mutations in the gene encoding the synaptic scaffolding protein SHANK3 are associated with autism spectrum disorders. Nat. Genet. 2006, 39, 25–27. [Google Scholar] [CrossRef]
| Urinary Steroid Hormone Metabolites | Autistic Girls | Control Girls | 95% CI | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| (μg/mmol Creatinine) | Median | Mean | SEM | p-Value | Median | Mean | SEM | Effect Size | Lower Limit | Upper Limit |
| Androgen-Metabolites | ||||||||||
| Androsterone | 28.5 | 71.7 | 21.4 | 0.15 | 35.2 | 42.1 | 9.1 | 0.46 | −0.25 | 1.16 |
| Etiocholanolone | 24.6 | 57.8 | 16.6 | 0.17 | 26.4 | 38.8 | 14.3 | 0.32 | −0.39 | 1.00 |
| Androstenediol | 1.7 | 2.2 | 0.4 | 0.02 | 1.2 | 1.3 | 0.2 | 0.71 | 0.01 | 1.44 |
| 11-Oxo-Etiocholanolon | 30.4 | 35.1 | 6.7 | 0.11 | 19.2 | 20.9 | 3.0 | 0.67 | −0.04 | 1.39 |
| 11b-Hydroxy-Androsteron | 29.5 | 36.6 | 5.5 | 0.08 | 24.4 | 22.1 | 3.2 | 0.77 | 0.09 | 1.53 |
| 11b-Hydroxy-Etiocholanolon | 28.9 | 26.0 | 5.2 | 0.06 | 12.5 | 13.1 | 2.0 | 0.78 | 0.06 | 1.50 |
| Dehydroepiandrosteron (Wilcoxon) | 2.8 | 15.2 | 8.4 | 0.17 | 3.7 | 4.3 | 1.0 | 0.47 | ||
| 5-Androstene-3b,17b-diol | 3.4 | 6.8 | 2.6 | 0.10 | 1.8 | 2.2 | 0.4 | 0.62 | −0.09 | 1.33 |
| 16a-Hydroxy-DHEA | 8.2 | 57.6 | 36.9 | 0.20 | 6.0 | 8.0 | 1.5 | 0.48 | −0.33 | 1.07 |
| 5-Androstene-3b,16a,17b-triol | 8.1 | 36.8 | 16.8 | 0.12 | 7.2 | 8.6 | 1.9 | 0.59 | −0.11 | 1.31 |
| 5-Pregnene-3b, 16a,17b-triol | 4.4 | 11.6 | 4.4 | 0.70 | 6.8 | 9.7 | 1.7 | 0.14 | −0.50 | 0.89 |
| Testosterone | 0.8 | 0.8 | 0.1 | 0.02 | 0.5 | 0.5 | 0.1 | 0.83 | 0.26 | 1.73 |
| 5a-Dihydrotestosteron | 1.2 | 1.2 | 0.2 | 0.94 | 1.2 | 1.2 | 0.1 | 0.03 | −0.51 | 0.87 |
| Oestrogen-Metabolites | ||||||||||
| Estriol | 0.1 | 0.6 | 0.4 | 0.11 | 0.1 | 0.4 | 0.1 | 0.19 | −0.60 | 0.78 |
| 17b-Estradiol (Wilcoxon) | 0.046 | 0.1 | 0.0 | 0.10 | 0.1 | 0.2 | 0.1 | −0.49 | ||
| Progesterone-Metabolites | ||||||||||
| 17-Hydroxypregnanolon (Wilcoxon) | 2.7 | 5.5 | 1.4 | 0.33 | 3.3 | 3.5 | 0.6 | 0.47 | ||
| Pregnanediol (Wilcoxon) | 11.6 | 18.9 | 4.6 | 0.50 | 10.8 | 12.7 | 2.4 | 0.44 | ||
| Pregnanetriol | 18.4 | 33.0 | 9.2 | 0.17 | 19.3 | 20.5 | 3.5 | 0.46 | −0.23 | 1.18 |
| 11-Oxo-Pregnanetriol (Wilcoxon) | 1.0 | 1.2 | 0.2 | 0.99 | 1.1 | 1.4 | 0.4 | −0.20 | ||
| Aldosterone-Metabolites | ||||||||||
| Tetrahydroaldosterone | 1.9 | 2.2 | 0.5 | 0.05 | 4.1 | 1.1 | 0.1 | 0.72 | 0.04 | 1.48 |
| Corticosterone-Metabolites | ||||||||||
| TetrahydroDOC | 0.5 | 0.6 | 0.1 | 0.71 | 1.0 | 0.5 | 0.1 | 0.14 | −0.50 | 0.89 |
| Tetrahydrodehydrocorticosterone | 9.5 | 10.2 | 1.6 | 0.71 | 0.4 | 9.4 | 1.2 | 0.14 | −0.59 | 0.80 |
| Tetrahydrocorticosterone (Wilcoxon) | 9.3 | 9.2 | 1.0 | 0.4965 | 9.7 | 9.6 | 2.1 | −0.07 | ||
| 5a-Tetrahydrocorticosteron | 18.6 | 18.9 | 2.8 | 0.43 | 8.2 | 24.8 | 6.7 | −0.30 | −1.00 | 0.40 |
| 18-Hydroxy-Tetrahydrocompound A (Wilcoxon) | 1.1 | 1.9 | 0.6 | 0.04 | 24.8 | 7.1 | 2.1 | −0.81 | ||
| Cortisone | 9.0 | 10.2 | 0.9 | 0.70 | 3.9 | 10.9 | 1.5 | −0.15 | −0.89 | 0.50 |
| Cortisone-Metabolites | ||||||||||
| Tetrahydrocortisone | 176.6 | 203.4 | 22.6 | 0.75 | 10.3 | 194.1 | 25.0 | 0.10 | −0.60 | 0.79 |
| a-Cortolon | 65.2 | 70.2 | 7.7 | 0.23 | 194.1 | 56.4 | 7.0 | 0.48 | −0.22 | 1.18 |
| b-Cortolon | 169.0 | 220.4 | 69.1 | 0.02 | 62.2 | 43.3 | 5.5 | 0.85 | 0.17 | 1.63 |
| 20a-Dihydrocortison | 1.1 | 1.2 | 0.1 | 0.87 | 47.2 | 1.2 | 0.2 | 0.07 | −0.69 | 0.69 |
| 20b-Dihydrocortison | 3.4 | 3.7 | 0.5 | 0.49 | 1.2 | 3.2 | 0.5 | 0.29 | −0.44 | 0.95 |
| Cortisol | 5.9 | 6.3 | 0.6 | 0.02 | 3.0 | 4.1 | 0.5 | 0.41 | 0.26 | 1.73 |
| Cortisol-Metabolites | ||||||||||
| Tetrahydrocortisol | 61.9 | 68.4 | 8.5 | 0.62 | 4.7 | 61.5 | 9.0 | 0.21 | −0.49 | 0.90 |
| 5a-Tetrahydrocortisol | 56.7 | 77.0 | 16.1 | 0.54 | 52.4 | 96.7 | 23.7 | -0.25 | −0.94 | 0.45 |
| a-Cortol | 10.8 | 13.5 | 1.8 | 0.65 | 89.4 | 12.1 | 1.9 | 0.20 | −0.55 | 0.84 |
| b-Cortol | 12.8 | 15.2 | 2.3 | 0.08 | 12.0 | 26.0 | 4.4 | -0.75 | −1.52 | -0.08 |
| 20a-Dihydrocortisol (Wilcoxon) | 2.3 | 3.2 | 0.6 | 0.40 | 3.2 | 3.6 | 0.7 | -0.15 | ||
| 6b-Hydroxycortisol | 9.3 | 11.1 | 1.5 | 0.55 | 9.8 | 9.8 | 1.4 | 0.23 | −0.47 | 0.92 |
| 18-Hydroxycortisol | 27.0 | 35.4 | 4.8 | 0.02 | 19.9 | 21.3 | 3.1 | 0.83 | −0.47 | 0.92 |
| Total | ||||||||||
| Total Androgens | 150.0 | 359.4 | 105.1 | 0.08 | 122.0 | 172.8 | 29.8 | 0.60 | −0.11 | 1.31 |
| Total of Cortisol and Cortisone | 610.9 | 740.9 | 101.3 | 0.13 | 605.2 | 552.1 | 75.9 | 0.53 | −0.17 | 1.24 |
| Total - All Measured Metabolites | 982.2 | 1206.3 | 170.2 | 0.08 | 835.2 | 814.4 | 96.3 | 0.70 | 0.03 | 1.47 |
| Autistic Girls | Control Girls | ||||
|---|---|---|---|---|---|
| Mean | SEM | p-Value | Mean | SEM | |
| 18OHTHA/THALDO | 1.175 | 0.399 | 0.014 | 1.595 | 0.419 |
| 21-Hydroxylase Deficit | |||||
| 17HP/(THE + THF + 5aTHF) | 0.017 | 0.003 | 0.026 | 0.010 | 0.002 |
| PT/(THE + THF + 5aTHF) | 0.098 | 0.016 | 0.064 | 0.073 | 0.013 |
| 100 × PT’ONE/(THE + THF + 5aTHF) | 0.358 | 0.043 | 0.644 | 0.404 | 0.099 |
| 17-Hydroxylase Deficit | |||||
| (THA + THB + 5aTHB)/(THE + THF + 5aTHF) | 0.256 | 0.012 | 0.407 | 0.287 | 0.021 |
| 100 × THDOC/(THE + THF + 5aTHF) | 0.257 | 0.069 | 0.157 | 0.145 | 0.018 |
| 11-Hydroxylase Deficit | |||||
| 100 × THS/(THE + THF + 5aTHF) | 1.682 | 0.152 | 0.330 | 1.864 | 0.151 |
| 100 × THDOC/(THE + THF + 5aTHF) | 0.257 | 0.069 | 0.157 | 0.145 | 0.018 |
| 11-Beta Hydroxylase | |||||
| F/E | 0.608 | 0.035 | 0.002 | 0.445 | 0.026 |
| (THF + 5aTHF)/THE | 0.642 | 0.058 | 0.290 | 0.044 | 0.004 |
| (F + E)/THE + THF + 5aTHF | 0.057 | 0.005 | 0.788 | 0.044 | 0.004 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gasser, B.A.; Kurz, J.; Dick, B.; Mohaupt, M.G. Are Steroid Hormones Dysregulated in Autistic Girls? Diseases 2020, 8, 6. https://doi.org/10.3390/diseases8010006
Gasser BA, Kurz J, Dick B, Mohaupt MG. Are Steroid Hormones Dysregulated in Autistic Girls? Diseases. 2020; 8(1):6. https://doi.org/10.3390/diseases8010006
Chicago/Turabian StyleGasser, Benedikt Andreas, Johann Kurz, Bernhard Dick, and Markus Georg Mohaupt. 2020. "Are Steroid Hormones Dysregulated in Autistic Girls?" Diseases 8, no. 1: 6. https://doi.org/10.3390/diseases8010006
APA StyleGasser, B. A., Kurz, J., Dick, B., & Mohaupt, M. G. (2020). Are Steroid Hormones Dysregulated in Autistic Girls? Diseases, 8(1), 6. https://doi.org/10.3390/diseases8010006




